
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Immunol. , 22 December 2023
Sec. Alloimmunity and Transplantation
Volume 14 - 2023 | https://doi.org/10.3389/fimmu.2023.1287456
In the absence of prophylactic therapy, cytomegalovirus (CMV) viremia is a common complication following allogeneic hematopoietic cell transplantation (allo-HCT) and represents a significant cause of morbidity and mortality. Approximately 25% of allo-HCT happen in China, where the development and refinement of the ‘Beijing protocol’ has enabled frequent and increasing use of haploidentical donors. However, refractory CMV infection (an increase by >1 log10 in blood or serum CMV DNA levels after at least 2 weeks of an appropriately dosed anti-CMV medication) is more common among patients with haploidentical donors than with other donor types and has no established standard of care. Here, we review the literature regarding refractory CMV infection following allo-HCT in China.
Allogeneic hematopoietic cell transplantation (allo-HCT) is an extremely important treatment option for a number of hematologic malignancies or other life-threatening conditions. In some cases, allo-HCT is the only potentially curative therapeutic strategy. The Chinese Blood and Marrow Transplantation Registry Group reported that the annual number of allo-HCT has steadily risen (1). In China, there were 3597 allo-HCT estimated in 2015 (2), increasing to 9597 in 2019 (3), meaning that allo-HCT in China may now represent approximately 25% of allo-HCT worldwide. This increase in numbers reflects a steady increase in adoption of allo-HCT with haploidentical donors (HID) following widespread adoption of the conditioning and risk stratification in the ‘Beijing protocol’ pioneered in China (1, 4, 5). While rates of successful allo-HCT in China have increased, the more intense conditioning required for successful HID allo-HCT means that greater recipient immunosuppression is typically necessary to prevent graft-versus-host disease (GVHD) or other complications.
Cytomegalovirus (CMV) viremia is one of the most common complications following allo-HCT, and is a major cause of morbidity and mortality among patients receiving allo-HCT (6–8). CMV viremia often results from reactivation of latent virus in an immunocompromised host. It is estimated that 83% of people globally are seropositive for CMV, with 90% seropositivity estimated in China (9). CMV viremia following allo-HCT may result in poor graft function (10), CMV disease (such as pneumonia, gastroenteritis, or retinitis) (11), and an increased risk of GVHD (6, 12), fungal infection (13, 14), and of death including non-relapse mortality (NRM) (7, 8, 15, 16).
With increasing numbers of allo-HCT and high rates of CMV seropositivity in China, anti-viral therapeutic strategies for Chinese recipients of allo-HCT are needed. CMV therapies typically include monitoring for CMV viremia using sensitive polymerase chain reaction (PCR)-based techniques and pre-emptive antiviral treatment (PET, given after detection of viremia), which may include the anti-viral agents (val)ganciclovir, foscarnet, cidofovir, and others. However, despite advances in CMV PCR and PET, approximately half of patients with CMV infection are estimated to have refractory CMV with an inadequate response to existing PET (6), representing a key clinical challenge.
In this review, we summarize and discuss the evidence to date surrounding treatment of refractory CMV infection in Chinese patients who receive allo-HCT, including how the emerging strategy of primary prophylaxis may play an important role in the prevention of refractory CMV and CMV disease.
Definitions of CMV infection and disease have been developed by the CMV Drug Development forum, first published in 1993, with more recent updates refining definitions (17). Working from these definitions, definitions of refractory CMV and resistant CMV infection have been proposed by the CMV Resistance Working Group of the CMV Drug Development Forum and are commonly used in clinical trials (18).
Refractory CMV viremia is defined as more than 1 log10 increase in CMV DNA levels in blood or serum after at least 2 weeks of antiviral treatment, with probable refractory infection or disease defined as persistent viral load or a lack of improvement in signs and symptoms after 2 weeks of antiviral treatment. Resistant CMV infection is defined as refractory CMV infection in the presence of known CMV gene mutations conferring antiviral drug resistance, such as the viral UL97 (kinase) and UL54 (DNA polymerase) genes. Therefore, clinically refractory CMV infection, in the absence of known resistance mutations, may not always also be defined as drug resistant.
Globally, refractory CMV infection has been reported in up to 39% of patients following allo-HCT; resistant CMV infection is less common, with reports of up to 14.5% of patients also having a clear antiviral drug resistance mutation (19–21). In China, the incidence of refractory CMV infection may be higher, with a reported incidence of approximately 48%, although the incidence of resistant CMV in Chinese patients is less clear (22, 23). A potential cause of a higher incidence of refractory CMV in Chinese patients compared with global reports could be a higher proportion of HID allo-HCT in China (60%) compared with Europe (42%) or the USA, though the use of HID allo-HCT is rising worldwide (4).
Globally, in a systematic review of 26 studies, Giménez et al. found that overall CMV infection detected by PCR was associated with an increased risk of overall mortality (hazard ratio [HR], 1.47; 95% CI, 1.20–1.80), and NRM (HR, 1.68; 95% CI, 1.14–2.49), although the authors highlight the high heterogeneity among the studies (7). Among the 4 studies that used PCR detection, CMV requiring PET had a further increased risk of overall mortality (HR, 2.35; 95% CI, 1.56–3.52) and (HR, 2.16; 95% CI, 1.27–3.69) (7). In a single-center retrospective analysis of 488 patients in China (of whom 397 had an HLA-mismatched related donor), Liu et al. found in multivariate analysis that refractory CMV infection was an independent risk factor for NRM (HR, 8.435; 95% CI, 1.511–47.099) (6). Interestingly, in the same study, acute GVHD was also an independent risk factor for refractory CMV infection (HR, 1.717; 95% CI, 1.102–2.675) and refractory CMV was an independent risk factor for CMV disease (HR, 10.539; 95% CI, 2.467–45.015) (6). These data are supported by earlier work suggesting a bidirectional relationship between acute GVHD and CMV infection (regardless of refractoriness) (12). Patients had an increased risk of acute GVHD during CMV replication (HR, 2.18; 95% CI, 1.30–3.65), and patients with grade II–IV acute GVHD had an increased risk of CMV reactivation (HR, 1.61; 95% CI, 1.11–2.36), potentially due to the immunosuppressive effects of GVHD and its treatment (12).
Prospective data investigating risk factors for refractory CMV in Chinese allo-HCT recipients are lacking, although notable efforts have been made to predict refractory CMV. Shen et al. used a machine-learning–based method in 289 Chinese patients receiving HID allo-HCT for acute leukemia that incorporated age, sex, underlying disease, CD34 graft status, and cumulative steroid dose to produce a model that was predictive of refractory or recurrent CMV (23). Further prospective clinical validation of these findings would aid CMV-related risk stratification for patients undergoing allo-HCT.
In addition, results from multiple single-center, retrospective studies have been reported. One such study including 359 patients found that donor type (HID/matched related donor), receipt of ATG, MMF, corticosteroid receipt within 30 days, and low presence of CD3+ CD8+ immune were associated with increased risk of CMV infection, with donor type and low (<14.825%) CD3+ CD8+ cells retaining significance in multivariate analysis, suggesting independent predictive value (24). A single-center, retrospective study in China found that 84/282 patients (29.8%) developed clinically refractory CMV reactivation, and of the clinical risk factors considered, there was a greater risk of clinically refractory CMV reactivation associated with haploidentical or matched unrelated donors (compared with HLA-matched sibling donors); total-body irradiation (TBI)-containing conditioning; cord-blood or bone-marrow donor sources compared with peripheral-blood containing sources; and high steroid dosages (≥1 mg/kg per day) (25). Some of these factors were also reported in an earlier, multicenter study of patients who underwent cord-blood allo-HCT, with higher steroid dosages notably associated with refractory CMV (26). Further retrospective work in China found that thrombocytopenia (lower-than-median day 90 platelet counts), higher CMV viral loads (above 104 copies/mL), and ATG-based conditioning were significantly associated with refractory CMV reactivation in multivariate analyses (27). Among these studies (24–27), it is notable that clinical factors associated with suppressing immune function tended to have greater incidence of refractory CMV infection and were identified as risk factors.
Interestingly, Liu et al. reported data that suggest that seropositive Chinese patients who have seropositive donors have a lower incidence and duration of CMV viremia and less development of refractory CMV infection or CMV disease compared with seropositive patients who have seronegative donors; however, there was no effect on survival observed (28). Another study in China found that in patients with Philadelphia chromosome positive acute lymphoblastic leukemia (ALL), patients who received haploidentical allo-HCT had a greater risk of CMV infection than those with HLA-matched allo-HCT (38% relative to 14%) (29), and another, long-term single-center study only including patients with HID allo-HCT found a CMV infection incidence of 64% (30). However, relapse rates were found to be lower among patients with HID relative to HLA-matched allo-HCT (19% relative to 45%) (29), similar to the 2-year cumulative incidence of relapse of 18% in the second study (30).
Refractoriness may be conferred by abnormalities in the recipient’s immunity or by mutations in the CMV genome, though notably, the majority of patients with refractory CMV have clinically refractory CMV, as opposed to resistant CMV with known resistance mutations (19, 31).
Data completely characterizing the molecular epidemiology of CMV resistance mutations are lacking in general, and although drug resistance mutations are generally thought to be uncommon, the incidence of such mutations has been reported at up to 14.5% (19, 31–33); data exploring the prevalence of resistance mutations in Chinese patients with refractory CMV following allo-HCT are also scarce (Table 1). A retrospective study of 41 patients with probable refractory CMV infection following allo-HCT in China found that 20 (49%) had a mutation in UL54 or UL97, and patients with mutations had significantly worse outcomes in terms of clearance of CMV DNAemia; however, only one of the 20 mutations had been previously characterized as a drug-resistance mutation (34). Similarly, a small study in pediatric patients in a Chinese hospital reported 10 novel mutations in UL54, and a retrospective multicenter study of 729 patients reported 12 novel UL54 variants and 1 novel UL97 variant (35). Another retrospective study found that among 221 pediatric patients in another Chinese center, 11 novel mutations in UL54 or UL97 were reported (36). These novel variants may suggest that previously unidentified CMV mutations in UL54, UL97, or other CMV genes are yet to be characterized in Chinese patients, particularly with increasing development and use of novel anti-viral agents. Further, prospective study to characterize the prevalence and nature of mutations that have the potential to confer drug resistance in Chinese patients is required to inform treatment decisions and therapeutic advances.
Developments in next-generation sequencing (NGS) technology may allow for more rapid mutation detection and increased sensitivity beyond the ~20 to 30% mutation prevalence required by Sanger-sequencing techniques (37); genomic analyses using NGS have been performed to better understand resistance pathways observed following letermovir prophylaxis (38, 39). However, appropriate precautions involving standardization and accuracy, avoiding sequencing artifacts, are needed in clinical trials (40).
Importantly, many patients in China are now able to receive effective primary prophylactic therapy with the CMV viral terminase complex inhibitor, letermovir. The drug was approved in China (2022) and elsewhere as primary prophylaxis following a phase 3 randomized trial that demonstrated a significant reduction in the risk of clinically significant CMV infection (defined as CMV infection requiring PET or leading to CMV disease) compared with placebo (60.6% vs. 37.5%; P<0.001) (41). The safety and efficacy findings in this trial have been confirmed in multiple real-world and post-marketing surveillance studies globally (42–48), with a recent European Society for Blood and Marrow Transplantation report suggesting that prophylaxis is used in 56% of centers (49), among which 61% used letermovir. Prophylaxis is recommended by the European Conference on Infections in Leukaemia 2017 (50) and American Society for Transplantation and Cellular Therapy guidelines (51).
Data on the impact of letermovir prophylaxis on refractory CMV infection are emerging. In a single-center retrospective study in the USA, primary prophylaxis with letermovir was associated with an 85% reduction in the risk of refractory or resistant CMV infection (HR, 0.15; 95% CI, 0.04–0.52) and a reduction in the risk of 48-week NRM (HR 0.55; 95% CI, 0.32–0.93) (20). This reduction in the risk of refractory disease may be particularly important in China, where HID allo-HCT is most common, as drug resistance has been observed to occur with HID allo-HCT more commonly than with other donor sources (33).
Ganciclovir (or its oral prodrug, valganciclovir) and foscarnet inhibit CMV DNA polymerase and are typically used as PET in China (Table 2), though use of these widely characterized agents is limited by their associated toxicities and potential resistance mutations. Ganciclovir/valganciclovir is often associated with neutropenia and thus potential secondary infections (71, 72), while foscarnet is often associated with electrolyte disturbances and renal toxicity (73). Indeed, it is recommended to modify dosages of both (val)ganciclovir and foscarnet according to renal function (74–76). While the UL97 kinase inhibitor maribavir has shown promise as PET for refractory CMV, with less hematotoxicity observed than valganciclovir (77–79), data on patients receiving maribavir in China are scarce, and the drug is not yet approved in China. Similarly, data on other newer antiviral therapies, brincidofovir and leflunomide, are lacking in Chinese patients. While letermovir has been shown to be effective as primary prophylaxis, data on its use beyond this setting are highly limited, with no approval in China outside of the primary prophylaxis setting.
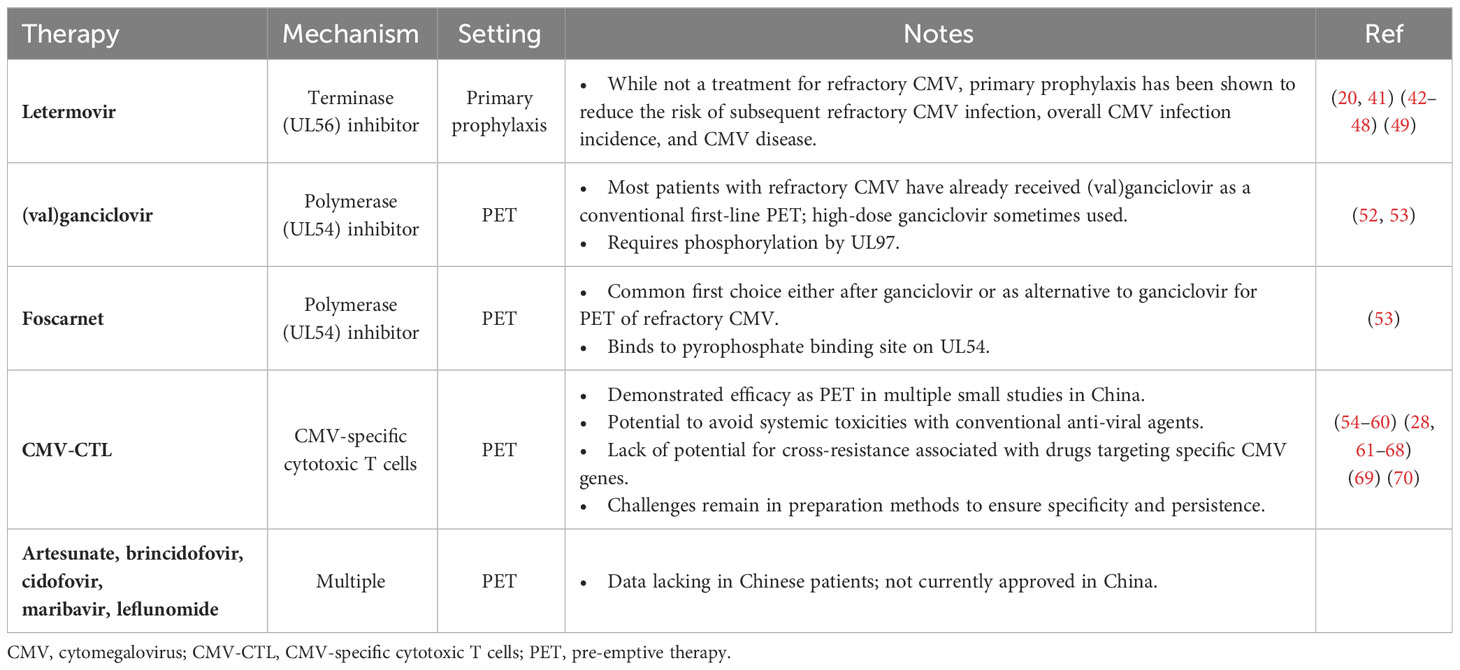
Table 2 Common or emerging therapies for refractory CMV infection in Chinese recipients of allo-HCT.
Combinations of PET agents may be introduced in the case of refractory CMV, such as ganciclovir plus foscarnet, which both inhibit the CMV DNA polymerase, UL54 (32). Limited data are available regarding combinations of PET agents: while a small study (n=32) conducted in Italy found a potential decrease in transplant-related mortality with combined ganciclovir and foscarnet (80), a more recent Chinese study (n=242) found no significant improvements in overall or disease-free survival with the use of combined ganciclovir and foscarnet compared with single-agent PET (22). Further data on combination therapy is required, particularly in patients with refractory CMV.
While uptake of HID allo-HCT continues to increase, the associated intensive conditioning presents a challenge with respect to CMV infection and refractory CMV, as CMV-specific immunity is reduced by delayed or impaired immune reconstitution (81).
Adoptive cell therapy with CMV-specific T cells (CMV-CTL) has been shown to be effective for treatment of CMV infection, including refractory CMV infection, for decades (54–60). Clinical experience of CMV-CTL therapy has been reported in multiple Chinese centers and studies that have suggested encouraging responses (28, 61–68). For example, while a range of efficacy results have been reported, Pei et al. found a CMV clearance rate of 89.5% 6 weeks after infusion (64, 82). For context with conventional antiviral agents, in a phase 3 global randomized study of maribavir compared with investigator’s choice of conventional antiviral agents in patients with refractory CMV, 8-week CMV clearance and symptom resolution was reported in 23.9% of patients who received investigator’s choice and 55.7% of patients who received maribavir (79). While direct comparisons cannot be made between these studies, the efficacy of CMV-CTL is notable and makes CMV-CTL an attractive option for refractory CMV infection; further data, including in Chinese patients, are needed to optimize treatment decisions. It is also notable that CMV-CTL may avoid systemic toxicities associated with treatment using multiple antiviral agents due to its immune-mediated mechanism that may include reversion of T-cell exhaustion (66). Indeed, the 2022 Chinese Society of Hematology consensus recommends CMV-CTL in the event of refractory CMV encephalitis (83). However, the rapid development of anti-viral agents and the intrinsic methodological challenges of generating CMV-CTLs mean that while the uptake of this approach in China is increasing, data on the widespread frequency of its use are not yet known, as CMV-CTL may be restricted to a few specialized centers. Methodologic challenges with CMV-CTLs may include technically challenging ex-vivo co-culture and/or manipulation of antigen-presenting cells (APCs) to ensure CMV specificity, as well as potential challenges to ensure persistence (84). Advances in T-cell generation (54, 62, 85) and development of allogeneic CMV-specific cryopreserved cells in an ‘off-the-shelf’ approach have shown promise to help to mitigate or circumvent T-cell manufacture difficulties and may allow CMV-CTL therapy to become more widely used in future (69). Interestingly, early results from a small Chinese study that generated CMV-specific T cells using T-cell receptors from healthy donors has shown promising anti-CMV efficacy, CMV persistence, and tolerability, and warrants further study in Chinese patients with refractory CMV infection (70).
Detailed identification of T-cell populations to expand and potential lessons from the rapidly growing and potentially complementary field of oncology T-cell manufacture may further optimize CMV-CTL processes and enable an increase in the use of CMV-CTL for refractory CMV infection, particularly if allo-HCT is used to consolidate complete response following T-cell therapy (86–89),. For example, memory T cells have been shown to be critical for CMV-specific T-cell immune reconstitution (90, 91), and detailed analysis suggests that CMV positivity induces T effector memory cell differentiation to a cytotoxic CD4-expressing population (92), further supported by evidence suggesting that durable response to CMV-CTL therapy is greater among patients with higher baseline CD4-positive immune cell counts (93). Further enhancement or enrichment of cell populations during immune reconstitution may enable more optimized CMV-CTL processes and greater CMV-CTL efficacy.
Natural killer (NK) cells have been broadly recognized for their anti-CMV and anti-tumor potential. Studies in China suggest that better NK cell education/licensing as measured by KIR and HLA expression is associated with decreased incidence of CMV reactivation, infection, and CMV disease, and improved relapse-free survival (94, 95), suggesting that KIR ligand matching may be a useful additional tool to optimize outcomes in Chinese patients. However, the practicality of incorporating such additional immunophenotyping into routine clinical practice remains to be seen.
In China, due to the high and rising use of HID allo-HCT, the incidence of refractory CMV infections is high. While therapeutic advances with PET following CMV PCR monitoring have improved outcomes for recipients of allo-HCT, the disease burden, including increased risk of mortality, remains high, particularly for patients with refractory CMV. Data suggest that letermovir prophylaxis may substantially reduce the risk of refractory CMV developing; with the recent approval of letermovir in China, further study of the impact of prophylactic letermovir on the incidence of refractory CMV in Chinese patients is needed to optimize treatment strategies. If treatment of refractory CMV is needed, CMV-specific immunotherapy may provide a useful alternative to established PET agents to avoid cross-resistance and potential treatment-related toxicities. However, as the incidence of resistance mutations is low relative to clinically refractory CMV, further study is needed to understand the mechanisms of refractoriness. Furthermore, due to the high disease burden of refractory CMV, larger, prospective studies involving patients with refractory CMV are needed, as many existing studies are small or retrospective in nature.
DY: Writing – original draft, Writing – review & editing, Conceptualization. YY: Writing – original draft, Writing – review & editing, Conceptualization. YS: Writing – original draft, Writing – review & editing, Conceptualization. EJ: Writing – review & editing, Conceptualization, Supervision.
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This review was funded and sponsored by MSD China. This work was supported by grants from the National Nature Science Foundation of China (No. 82070192 and No. 81670171 to EJ), Haihe Laboratory of Cell Ecosystem Innovation Fund (No. HH22KYZX0034 to EJ), the Non-profit Central Research Institute Fund of Chinese Academy of Medical Sciences (2019XK320076 to DY).
Medical writing support was provided by Adam Gill, MRes, at Rude Health Consulting, and was funded by MSD China.
Authors YY and YS are employees of MSD China.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Zhang XH, Chen J, Han MZ, Huang H, Jiang EL, Jiang M, et al. The consensus from The Chinese Society of Hematology on indications, conditioning regimens and donor selection for allogeneic hematopoietic stem cell transplantation: 2021 update. J Hematol Oncol (2021) 14(1):145. doi: 10.1186/s13045-021-01159-2
2. Xu L-P, Wu DP, Han MZ, Huang H, Liu QF, Liu DH, et al. A review of hematopoietic cell transplantation in China: data and trends during 2008-2016. Bone Marrow Transplant (2017) 52(11):1512–8. doi: 10.1038/bmt.2017.59
3. Xu L-P, Lu PH, Wu DP, Sun ZM, Liu QF, Han MZ, et al. Hematopoietic stem cell transplantation activity in China 2019: a report from the Chinese Blood and Marrow Transplantation Registry Group. Bone Marrow Transplant (2021) 56(12):2940–7. doi: 10.1038/s41409-021-01431-6
4. Apperley J, Niederwieser D, Huang XJ, Nagler A, Fuchs E, Szer J, et al. Haploidentical hematopoietic stem cell transplantation: A global overview comparing asia, the european union, and the United States. Biol Blood Marrow Transplant (2016) 22(1):23–6. doi: 10.1016/j.bbmt.2015.11.001
5. Lv M, Chang YJ, Huang XJ. Update of the “Beijing Protocol” haplo-identical hematopoietic stem cell transplantation. Bone Marrow Transplant (2019) 54(Suppl 2):703–7. doi: 10.1038/s41409-019-0605-2
6. Liu J, Kong J, Chang YJ, Chen H, Chen YH, Han W, et al. Patients with refractory cytomegalovirus (CMV) infection following allogeneic haematopoietic stem cell transplantation are at high risk for CMV disease and non-relapse mortality. Clin Microbiol Infect (2015) 21(12):1121 e9–15. doi: 10.1016/j.cmi.2015.06.009
7. Gimenez E, Torres I, Albert E, Pinana JL, Hernandez-Boluda JC, Solano C, et al. Cytomegalovirus (CMV) infection and risk of mortality in allogeneic hematopoietic stem cell transplantation (Allo-HSCT): A systematic review, meta-analysis, and meta-regression analysis. Am J Transplant (2019) 19(9):2479–94. doi: 10.1111/ajt.15515
8. Teira P, Battiwalla M, Ramanathan M, Barrett AJ, Ahn KW, Chen M, et al. Early cytomegalovirus reactivation remains associated with increased transplant-related mortality in the current era: a CIBMTR analysis. Blood (2016) 127(20):2427–38. doi: 10.1182/blood-2015-11-679639
9. Zuhair M, Smit GSA, Wallis G, Jabbar F, Smith C, Devleesschauwer B, et al. Estimation of the worldwide seroprevalence of cytomegalovirus: A systematic review and meta-analysis. Rev Med Virol (2019) 29(3):e2034. doi: 10.1002/rmv.2034
10. Prabahran A, Koldej R, Chee L, Ritchie D. Clinical features, pathophysiology, and therapy of poor graft function post-allogeneic stem cell transplantation. Blood Adv (2022) 6(6):1947–59. doi: 10.1182/bloodadvances.2021004537
11. Einsele H, Ljungman P, Boeckh M. How I treat CMV reactivation after allogeneic hematopoietic stem cell transplantation. Blood (2020) 135(19):1619–29. doi: 10.1182/blood.2019000956
12. Cantoni N, Hirsch HH, Khanna N, Gerull S, Buser A, Bucher C, et al. Evidence for a bidirectional relationship between cytomegalovirus replication and acute graft-versus-host disease. Biol Blood Marrow Transplant (2010) 16(9):1309–14. doi: 10.1016/j.bbmt.2010.03.020
13. Yong MK, Slavin MA, Kontoyiannis DP. Invasive fungal disease and cytomegalovirus infection: is there an association? Curr Opin Infect Dis (2018) 31(6):481–9. doi: 10.1097/QCO.0000000000000502
14. Yong MK, Ananda-Rajah M, Cameron PU, Morrissey CO, Spencer A, Ritchie D, et al. Cytomegalovirus reactivation is associated with increased risk of late-onset invasive fungal disease after allogeneic hematopoietic stem cell transplantation: A multicenter study in the current era of viral load monitoring. Biol Blood Marrow Transplant (2017) 23(11):1961–7. doi: 10.1016/j.bbmt.2017.07.025
15. Green ML, Leisenring W, Xie H, Mast TC, Cui Y, Sandmaier BM, et al. Cytomegalovirus viral load and mortality after haemopoietic stem cell transplantation in the era of pre-emptive therapy: a retrospective cohort study. Lancet Haematol (2016) 3(3):e119–27. doi: 10.1016/S2352-3026(15)00289-6
16. Zhou X, Jin N, Chen B. Human cytomegalovirus infection: A considerable issue following allogeneic hematopoietic stem cell transplantation. Oncol Lett (2021) 21(4):318. doi: 10.3892/ol.2021.12579
17. Ljungman P, Boeckh M, Hirsch HH, Josephson F, Lundgren J, Nichols G, et al. Definitions of cytomegalovirus infection and disease in transplant patients for use in clinical trials. Clin Infect Dis (2017) 64(1):87–91. doi: 10.1093/cid/ciw668
18. Chemaly RF, Chou S, Einsele H, Griffiths P, Avery R, Razonable RR, et al. Definitions of resistant and refractory cytomegalovirus infection and disease in transplant recipients for use in clinical trials. Clin Infect Dis (2019) 68(8):1420–6. doi: 10.1093/cid/ciy696
19. Khawaja F, Batista MV, El Haddad L, Chemaly RF. Resistant or refractory cytomegalovirus infections after hematopoietic cell transplantation: diagnosis and management. Curr Opin Infect Dis (2019) 32(6):565–74. doi: 10.1097/QCO.0000000000000607
20. Sassine J, Khawaja F, Shigle TL, Handy V, Foolad F, Aitken SL, et al. Refractory and resistant cytomegalovirus after hematopoietic cell transplant in the letermovir primary prophylaxis era. Clin Infect Dis (2021) 73(8):1346–54. doi: 10.1093/cid/ciab298
21. Nichols WG, Corey L, Gooley T, Drew WL, Miner R, Huang M, et al. Rising pp65 antigenemia during preemptive anticytomegalovirus therapy after allogeneic hematopoietic stem cell transplantation: risk factors, correlation with DNA load, and outcomes. Blood (2001) 97(4):867–74. doi: 10.1182/blood.v97.4.867
22. Ma R, He Y, Xu LP, Zhang XH, Wang Y, Liu KY, et al. [Clinical analysis of the efficacies of ganciclovir plus foscarnet and a single antiviral drug for the treatment of cytomegalovirus infection after haploidentical stem cell transplantation]. Zhonghua Nei Ke Za Zhi (2023) 62(1):76–83. doi: 10.3760/cma.j.cn112138-20220118-00058
23. Shen MZ, Hong SD, Wang J, Zhang XH, Xu LP, Wang Y, et al. A predicted model for refractory/recurrent cytomegalovirus infection in acute leukemia patients after haploidentical hematopoietic stem cell transplantation. Front Cell Infect Microbiol (2022) 12:862526. doi: 10.3389/fcimb.2022.862526
24. Chen J, Pang A, Zhao Y, Liu L, Ma R, Wei J, et al. Risk factors for CMV infection within 100 days posttransplantation in patients with acute leukemia. Blood Sci (2022) 04(3):164–9. doi: 10.1097/BS9.0000000000000121
25. Bao X, Zhu Q, Xue S, Xu Y, Ma X, Chen F, et al. Risk factors of clinically refractory CMV reactivation following allogeneic HSCT: a single-center study in China. Bone Marrow Transplant (2016) 51(12):1625–7. doi: 10.1038/bmt.2016.231
26. Tong J, Sun Z, Liu H, Geng L, Zheng C, Tang B, et al. Risk factors of CMV infection in patients after umbilical cord blood transplantation: a multicenter study in China. Chin J Cancer Res (2013) 25(6):695–703. doi: 10.3978/j.issn.1000-9604.2013.11.08
27. Duan Z, Zhang X, Liu Y, Li F, Shen H, Chen R, et al. Risk factors and survival of refractory cytomegalovirus reactivation after allogeneic peripheral blood stem cell transplantation. J Glob Antimicrob Resist 31:279–85. doi: 10.1016/j.jgar.2022.10.009
28. Liu J, Fu Q, Wang Y, Wang FR, Han W, Ma YR, et al. [The effect of donor cytomegalovirus serological status on the outcome of allogeneic stem cell transplantation]. Zhonghua Nei Ke Za Zhi (2021) 60(5):459–65. doi: 10.3760/cma.j.cn112138-20200714-00668
29. Gao L, Zhang C, Gao L, Liu Y, Su Y, Wang S, et al. Favorable outcome of haploidentical hematopoietic stem cell transplantation in Philadelphia chromosome-positive acute lymphoblastic leukemia: a multicenter study in Southwest China. J Hematol Oncol (2015) 8:90. doi: 10.1186/s13045-015-0186-5
30. Wang Y, Liu DH, Liu KY, Xu LP, Zhang XH, Han W, et al. Long-term follow-up of haploidentical hematopoietic stem cell transplantation without in vitro T cell depletion for the treatment of leukemia: nine years of experience at a single center. Cancer (2013) 119(5):978–85. doi: 10.1002/cncr.27761
31. Razonable RR. Drug-resistant cytomegalovirus: clinical implications of specific mutations. Curr Opin Organ Transplant (2018) 23(4):388–94. doi: 10.1097/MOT.0000000000000541
32. El Chaer F, Shah DP, Chemaly RF. How I treat resistant cytomegalovirus infection in hematopoietic cell transplantation recipients. Blood (2016) 128(23):2624–36. doi: 10.1182/blood-2016-06-688432
33. Shmueli E, Or R, Shapira MY, Resnick IB, Caplan O, Bdolah-Abram T, et al. High rate of cytomegalovirus drug resistance among patients receiving preemptive antiviral treatment after haploidentical stem cell transplantation. J Infect Dis (2014) 209(4):557–61. doi: 10.1093/infdis/jit475
34. Yang D, Wen J, Zhang Y, Li G, Zheng Y, Chen S, et al. P1617: Clinical significance of cytomegalovirus drug resistance gene mutations in cytomegalovirus infection after allogeneic hematopoietic stem cell transplantation. HemaSphere (2022) 6:1498–9. doi: 10.1097/01.HS9.0000849324.10688.6e
35. Yang SL, Lin TW, Lin HC, Wang HY, Chang PY, Wang PN, et al. Molecular epidemiology of cytomegalovirus UL97 and UL54 variants in Taiwan. J Microbiol Immunol Infect (2021) 54(5):971–8. doi: 10.1016/j.jmii.2021.01.007
36. Yu U, Wang X, Zhang X, Wang C, Yang C, Zhou X, et al. Cytomegalovirus infection and the implications of drug-resistant mutations in pediatric allogeneic hematopoietic stem cell transplant recipients: A retrospective study from a tertiary hospital in China. Infect Dis Ther (2021) 10(3):1309–22. doi: 10.1007/s40121-021-00452-4
37. Lurain NS, Chou S. Antiviral drug resistance of human cytomegalovirus. Clin Microbiol Rev (2010) 23(4):689–712. doi: 10.1128/CMR.00009-10
38. Komatsu TE, Hodowanec AC, Colberg-Poley AM, Pikis A, Singer ME, O’Rear JJ, et al. In-depth genomic analyses identified novel letermovir resistance-associated substitutions in the cytomegalovirus UL56 and UL89 gene products. Antiviral Res (2019) 169:104549. doi: 10.1016/j.antiviral.2019.104549
39. Chorlton SD, Ritchie G, Lawson T, McLachlan E, Romney MG, Matic N, et al. Next-generation sequencing for cytomegalovirus antiviral resistance genotyping in a clinical virology laboratory. Antiviral Res (2021) 192:105123. doi: 10.1016/j.antiviral.2021.105123
40. Chou S. Advances in the genotypic diagnosis of cytomegalovirus antiviral drug resistance. Antiviral Res (2020) 176:104711. doi: 10.1016/j.antiviral.2020.104711
41. Marty FM, Ljungman P, Chemaly RF, Maertens J, Dadwal SS, Duarte RF, et al. Letermovir prophylaxis for cytomegalovirus in hematopoietic-cell transplantation. N Engl J Med (2017) 377(25):2433–44. doi: 10.1056/NEJMoa1706640
42. Anderson A, Raja M, Vazquez N, Morris M, Komanduri K, Camargo J. Clinical “real-world” experience with letermovir for prevention of cytomegalovirus infection in allogeneic hematopoietic cell transplant recipients. Clin Transplant (2020) 34(7):e13866. doi: 10.1111/ctr.13866
43. Derigs P, Radujkovic A, Schubert ML, Schnitzler P, Schoning T, Muller-Tidow C, et al. Letermovir prophylaxis is effective in preventing cytomegalovirus reactivation after allogeneic hematopoietic cell transplantation: single-center real-world data. Ann Hematol (2021) 100(8):2087–93. doi: 10.1007/s00277-020-04362-2
44. Hiraishi I, Ueno R, Watanabe A, Maekawa S. Safety and effectiveness of letermovir in allogenic hematopoietic stem cell transplantation recipients: interim report of post-marketing surveillance in Japan. Clin Drug Investig (2021) 41(12):1075–86. doi: 10.1007/s40261-021-01096-5
45. Lin A, Maloy M, Su Y, Bhatt V, DeRespiris L, Griffin M, et al. Letermovir for primary and secondary cytomegalovirus prevention in allogeneic hematopoietic cell transplant recipients: Real-world experience. Transpl Infect Dis (2019) 21(6):e13187. doi: 10.1111/tid.13187
46. Martino M, Pitino A, Gori M, Bruno B, Crescimanno A, Federico V, et al. Letermovir prophylaxis for cytomegalovirus infection in allogeneic stem cell transplantation: A real-world experience. Front Oncol (2021) 11:740079. doi: 10.3389/fonc.2021.740079
47. Mori Y, Jinnouchi F, Takenaka K, Aoki T, Kuriyama T, Kadowaki M, et al. Efficacy of prophylactic letermovir for cytomegalovirus reactivation in hematopoietic cell transplantation: a multicenter real-world data. Bone Marrow Transplant (2021) 56(4):853–62. doi: 10.1038/s41409-020-01082-z
48. Serio B, Giudice V, Guariglia R, Fontana R, Pezzullo L, Martorelli MC, et al. Prophylactic letermovir decreases cytomegalovirus reactivation after stem cell transplantation: a single-center real-world evidence study. Infez Med (2021) 29(1):102–13.
49. Cesaro S, Ljungman P, Tridello G, Mikulska M, Wendel L, Styczynski J, et al. New trends in the management of cytomegalovirus infection after allogeneic hematopoietic cell transplantation: a survey of the Infectious Diseases Working Pary of EBMT. Bone Marrow Transplant (2023) 58(2):203–8. doi: 10.1038/s41409-022-01863-8
50. Ljungman P, de la Camara R, Robin C, Crocchiolo R, Einsele H, Hill JA, et al. Guidelines for the management of cytomegalovirus infection in patients with haematological Malignancies and after stem cell transplantation from the 2017 European Conference on Infections in Leukaemia (ECIL 7). Lancet Infect Dis (2019) 19(8):e260–72. doi: 10.1016/S1473-3099(19)30107-0
51. Hakki M, Aitken SL, Danziger-Isakov L, Michaels MG, Carpenter PA, Chemaly RF, et al. American society for transplantation and cellular therapy series: #3-prevention of cytomegalovirus infection and disease after hematopoietic cell transplantation. Transplant Cell Ther (2021) 27(9):707–19. doi: 10.1016/j.jtct.2021.05.001
53. Biron KK. Antiviral drugs for cytomegalovirus diseases. Antiviral Res (2006) 71(2-3):154–63. doi: 10.1016/j.antiviral.2006.05.002
54. Bao L, Dunham K, Stamer M, Mulieri KM, Lucas KG. Expansion of cytomegalovirus pp65 and IE-1 specific cytotoxic T lymphocytes for cytomegalovirus-specific immunotherapy following allogeneic stem cell transplantation. Biol Blood Marrow Transplant (2008) 14(10):1156–62. doi: 10.1016/j.bbmt.2008.07.014
55. Gratama JW, Brooimans RA, van der Holt B, Sintnicolaas K, van Doornum G, Niesters HG, et al. Monitoring cytomegalovirus IE-1 and pp65-specific CD4+ and CD8+ T-cell responses after allogeneic stem cell transplantation may identify patients at risk for recurrent CMV reactivations. Cytomet B Clin Cytom (2008) 74(4):211–20. doi: 10.1002/cyto.b.20420
56. Leen AM, Myers GD, Sili U, Huls MH, Weiss H, Leung KS, et al. Monoculture-derived T lymphocytes specific for multiple viruses expand and produce clinically relevant effects in immunocompromised individuals. Nat Med (2006) 12(10):1160–6. doi: 10.1038/nm1475
57. Walter EA, Greenberg PD, Gilbert MJ, Finch RJ, Watanabe KS, Thomas ED, et al. Reconstitution of cellular immunity against cytomegalovirus in recipients of allogeneic bone marrow by transfer of T-cell clones from the donor. N Engl J Med (1995) 333(16):1038–44. doi: 10.1056/NEJM199510193331603
58. Einsele H, Roosnek E, Rufer N, Sinzger C, Riegler S, Loffler J, et al. Infusion of cytomegalovirus (CMV)-specific T cells for the treatment of CMV infection not responding to antiviral chemotherapy. Blood (2002) 99(11):3916–22. doi: 10.1182/blood.v99.11.3916
59. Feuchtinger T, Opherk K, Bethge WA, Topp MS, Schuster FR, Weissinger EM, et al. Adoptive transfer of pp65-specific T cells for the treatment of chemorefractory cytomegalovirus disease or reactivation after haploidentical and matched unrelated stem cell transplantation. Blood (2010) 116(20):4360–7. doi: 10.1182/blood-2010-01-262089
60. Mehdizadeh M, Karami S, Ghaffari Nazari H, Sankanian G, Hamidpour M, Hajifathali A. Immunotherapy with adoptive cytomegalovirus-specific T cells transfer: Summarizing latest gene engineering techniques. Health Sci Rep (2021) 4(3):e322. doi: 10.1002/hsr2.322
61. Gui RR, Li Z, Zu YL, Wang J, Liu YY, Zhang BL, et al. [CMV-CTL for treatment of refractory CMV infection in 17 patients following alternative donor hematopoietic stem cell transplantation]. Zhonghua Xue Ye Xue Za Zhi (2021) 42(10):865–8. doi: 10.3760/cma.j.issn.0253-2727.2021.10.013
62. Mo W, Liang Y, Wang C, Zhang Y. A simplified CMV-specific T cell transfer targeting drug-resistant CMV retinitis. Ocul Immunol Inflamm (2021) 18:1–3. doi: 10.1080/09273948.2021.1900876
63. Bao L, Cowan MJ, Dunham K, Horn B, McGuirk J, Gilman A, et al. Adoptive immunotherapy with CMV-specific cytotoxic T lymphocytes for stem cell transplant patients with refractory CMV infections. J Immunother (2012) 35(3):293–8. doi: 10.1097/CJI.0b013e31824300a2
64. Pei XY, Zhao XY, Liu XF, Mo XD, Lv M, Xu LP, et al. Adoptive therapy with cytomegalovirus-specific T cells for cytomegalovirus infection after haploidentical stem cell transplantation and factors affecting efficacy. Am J Hematol (2022) 97(6):762–9. doi: 10.1002/ajh.26535
65. Pei XY, Liu XF, Zhao XY, Lv M, Mo XD, Chang YJ, et al. Comparable anti-CMV responses of transplant donor and third-party CMV-specific T cells for treatment of CMV infection after allogeneic stem cell transplantation. Cell Mol Immunol (2022) 19(4):482–91. doi: 10.1038/s41423-021-00829-y
66. Pei XY, Zhao XY, Chang YJ, Liu J, Xu LP, Wang Y, et al. Cytomegalovirus-specific T-cell transfer for refractory cytomegalovirus infection after haploidentical stem cell transplantation: the quantitative and qualitative immune recovery for cytomegalovirus. J Infect Dis (2017) 216(8):945–56. doi: 10.1093/infdis/jix357
67. Xu Z, Huang X, Sun Y, Wang F, Yan C, Zhang X, et al. [Cytomegalovirus specific cytotoxic T lymphocytes for treatment of refractory cytomegalovirus infection in patients following allogeneic hematopoietic stem cell transplantation]. Zhonghua Nei Ke Za Zhi (2015) 54(2):101–5.
68. Wang X, Yu U, Yang C, Wang C, Zhang X, Li Y, et al. Cytomegalovirus (CMV)-specific cytotoxic T lymphocyte therapy resolve CMV diseases and refractory CMV infections in paediatric recipients of allogeneic haematopoietic stem cell transplantation. Bone Marrow Transplant (2022) 57(2):271–5. doi: 10.1038/s41409-021-01499-0
69. Tzannou I, Papadopoulou A, Naik S, Leung K, Martinez CA, Ramos CA, et al. Off-the-shelf virus-specific T cells to treat BK virus, human herpesvirus 6, cytomegalovirus, epstein-barr virus, and adenovirus infections after allogeneic hematopoietic stem-cell transplantation. J Clin Oncol (2017) 35(31):3547–57. doi: 10.1200/JCO.2017.73.0655
70. Liu G, Chen H, Cao X, Jia L, Rui W, Zheng H, et al. Efficacy of pp65-specific TCR-T cell therapy in treating cytomegalovirus infection after hematopoietic stem cell transplantation. Am J Hematol (2022) 97(11):1453–63. doi: 10.1002/ajh.26708
71. Goodrich JM, Mori M, Gleaves CA, Du Mond C, Cays M, Ebeling DF, et al. Early treatment with ganciclovir to prevent cytomegalovirus disease after allogeneic bone marrow transplantation. N Engl J Med (1991) 325(23):1601–7. doi: 10.1056/NEJM199112053252303
72. Liu KY, Wang Y, Han MZ, Huang H, Chen H, Liu QF, et al. Valganciclovir for pre-emptive therapy of cytomegalovirus viraemia after hematopoietic stem cell transplantation: a prospective multi-center trial. Chin Med J (Engl) (2010) 123(16):2199–205.
73. Reusser P, Einsele H, Lee J, Volin L, Rovira M, Engelhard D, et al. Randomized multicenter trial of foscarnet versus ganciclovir for preemptive therapy of cytomegalovirus infection after allogeneic stem cell transplantation. Blood (2002) 99(4):1159–64. doi: 10.1182/blood.v99.4.1159
74. Pfizer. FOSCAVIR US prescribing information (2020). Available at: http://labeling.pfizer.com/ShowLabeling.aspx?id=6521.
75. Cytovene US prescribing information (2017). Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2017/209347s001lbl.pdf.
76. Genentech. Valcyte US prescribing information. 2010 (2022). Available at: https://www.accessdata.fda.gov/drugsatfda_docs/label/2010/021304s008,022257s003lbl.pdf.
77. Maertens J, Cordonnier C, Jaksch P, Poire X, Uknis M, Wu J, et al. Maribavir for preemptive treatment of cytomegalovirus reactivation. N Engl J Med (2019) 381(12):1136–47. doi: 10.1056/NEJMoa1714656
78. Papanicolaou GA, Silveira FP, Langston AA, Pereira MR, Avery RK, Uknis M, et al. Maribavir for refractory or resistant cytomegalovirus infections in hematopoietic-cell or solid-organ transplant recipients: A randomized, dose-ranging, double-blind, phase 2 study. Clin Infect Dis (2019) 68(8):1255–64. doi: 10.1093/cid/ciy706
79. Avery RK, Alain S, Alexander BD, Blumberg EA, Chemaly RF, Cordonnier C, et al. Maribavir for refractory cytomegalovirus infections with or without resistance post-transplant: results from a phase 3 randomized clinical trial. Clin Infect Dis (2022) 75(4):690–701. doi: 10.1093/cid/ciab988
80. Bacigalupo A, Bregante S, Tedone E, Isaza A, Van Lint MT, Trespi G, et al. Combined foscarnet-ganciclovir treatment for cytomegalovirus infections after allogeneic hemopoietic stem cell transplantation. Transplantation (1996) 62(3):376–80. doi: 10.1097/00007890-199608150-00013
81. Luo XH, Zhu Y, Chen YT, Shui LP, Liu L. CMV infection and CMV-specific immune reconstitution following haploidentical stem cell transplantation: an update. Front Immunol (2021) 12:732826. doi: 10.3389/fimmu.2021.732826
82. Stem Cell Application Group CSoHCMA. [The Chinese consensus on the management of cytomegalovirus infection in allogeneic hematopoietic stem cell transplantation patients (2022)]. Zhonghua Xue Ye Xue Za Zhi (2022) 43(8):617–23. doi: 10.3760/cma.j.issn.0253-2727.2022.08.001
83. Xiaojun H. The Chinese consensus on the management of cytomegalovirus infection in allogeneic hematopoietic stem cell transplantation patients (2022). Chin J Hematol (2022) 43(8):617–23. doi: 10.3760/cma.j.issn.0253-2727.2022.08.001
84. Nicholson E, Peggs KS. Cytomegalovirus-specific T-cell therapies: current status and future prospects. Immunotherapy (2015) 7(2):135–46. doi: 10.2217/imt.14.99
85. Meij P, Jedema I, Zandvliet ML, van der Heiden PL, van de Meent M, van Egmond HM, et al. Effective treatment of refractory CMV reactivation after allogeneic stem cell transplantation with in vitro-generated CMV pp65-specific CD8+ T-cell lines. J Immunother (2012) 35(8):621–8. doi: 10.1097/CJI.0b013e31826e35f6
86. Amini L, Silbert SK, Maude SL, Nastoupil LJ, Ramos CA, Brentjens RJ, et al. Preparing for CAR T cell therapy: patient selection, bridging therapies and lymphodepletion. Nat Rev Clin Oncol (2022) 19(5):342–55. doi: 10.1038/s41571-022-00607-3
87. Han L, Zhou J, Li L, Zhou K, Zhao L, Zhu X, et al. Culturing adequate CAR-T cells from less peripheral blood to treat B-cell Malignancies. Cancer Biol Med (2021) 18(4):1066–79. doi: 10.20892/j.issn.2095-3941.2021.0040
88. Yang J, He J, Zhang X, Li J, Wang Z, Zhang Y, et al. Next-day manufacture of a novel anti-CD19 CAR-T therapy for B-cell acute lymphoblastic leukemia: first-in-human clinical study. Blood Cancer J (2022) 12(7):104. doi: 10.1038/s41408-022-00694-6
89. Zhao YL, Liu DY, Sun RJ, Zhang JP, Zhou JR, Wei ZJ, et al. Integrating CAR T-cell therapy and transplantation: comparisons of safety and long-term efficacy of allogeneic hematopoietic stem cell transplantation after CAR T-cell or chemotherapy-based complete remission in B-cell acute lymphoblastic leukemia. Front Immunol (2021) 12:605766. doi: 10.3389/fimmu.2021.605766
90. Luo XH, Huang XJ, Liu KY, Xu LP, Liu DH. Protective immunity transferred by infusion of cytomegalovirus-specific CD8(+) T cells within donor grafts: its associations with cytomegalovirus reactivation following unmanipulated allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant (2010) 16(7):994–1004. doi: 10.1016/j.bbmt.2010.02.007
91. Holtappels R, Podlech J, Freitag K, Lemmermann NA, Reddehase MJ. Memory CD8 T cells protect against cytomegalovirus disease by formation of nodular inflammatory foci preventing intra-tissue virus spread. Viruses (2022) 14(6):1145. doi: 10.3390/v14061145
92. Zhang W, Morris AB, Peek EV, Karadkhele G, Robertson JM, Kissick HT, et al. CMV status drives distinct trajectories of CD4+ T cell differentiation. Front Immunol (2021) 12:620386. doi: 10.3389/fimmu.2021.620386
93. Fabrizio VA, Rodriguez-Sanchez MI, Mauguen A, Dahi PB, Doubrovina E, O’Reilly RJ, et al. Adoptive therapy with CMV-specific cytotoxic T lymphocytes depends on baseline CD4+ immunity to mediate durable responses. Blood Adv (2021) 5(2):496–503. doi: 10.1182/bloodadvances.2020002735
94. Zhao XY, Luo XY, Yu XX, Zhao XS, Han TT, Chang YJ, et al. Recipient-donor KIR ligand matching prevents CMV reactivation post-haploidentical T cell-replete transplantation. Br J Haematol (2017) 177(5):766–81. doi: 10.1111/bjh.14622
Keywords: cytomegalovirus, refractory, China, allogeneic hematopoietic cell transplantation, haploidentical
Citation: Yang D, Yao Y, Sun Y and Jiang E (2023) Refractory cytomegalovirus infections in Chinese patients receiving allogeneic hematopoietic cell transplantation: a review of the literature. Front. Immunol. 14:1287456. doi: 10.3389/fimmu.2023.1287456
Received: 01 September 2023; Accepted: 08 December 2023;
Published: 22 December 2023.
Edited by:
Jamshid Roozbeh, Shiraz University of Medical Sciences, IranReviewed by:
Xiao-Dong Mo, Peking University People’s Hospital, ChinaCopyright © 2023 Yang, Yao, Sun and Jiang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Erlie Jiang, SmlhbmdlcmxpZUBpaGNhbXMuYWMuY24=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.