
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Immunol. , 02 February 2024
Sec. Immunological Tolerance and Regulation
Volume 14 - 2023 | https://doi.org/10.3389/fimmu.2023.1264071
This article is part of the Research Topic Exploring Immunological Tolerance in Allergy Treatment through AIT View all 14 articles
 Yu Tong†
Yu Tong† Lei Wang†
Lei Wang† Lingya Wang
Lingya Wang Jingjing Song
Jingjing Song Junwen Fan
Junwen Fan Chuqiao Lai
Chuqiao Lai Jiali Bao
Jiali Bao Cuiye Weng
Cuiye Weng Yufei Wang
Yufei Wang Jilong Shuai
Jilong Shuai Hui Zhang*
Hui Zhang* Weixi Zhang*
Weixi Zhang*Introduction: Group 2 innate lymphoid cells (ILC2s) play a crucial role in house dust mite (HDM)-induced allergic inflammation, and allergen immunotherapy (AIT) holds promise for treating the disease by reducing the frequency of ILC2s. Despite significant progress in AIT for allergic diseases, there remains a need to improve the control of allergic symptoms.
Methods: We investigated the synergistic effect of the Notch signaling pathway and subcutaneous immunotherapy (SCIT) in treating allergic airway inflammation in mice and their impact on the ratio of ILC2s in lung tissues. This was achieved by establishing the HDM-induced airway allergic disorders (HAAD) model and SCIT model. Additionally, we conducted in vitro investigations into the effect of the Notch signaling pathway on the secretory function of activated ILC2s using fluorescence-activated cell sorting. Furthermore, we explored the coactivation of the Notch signaling pathway with SCIT in vitro by sorting ILC2s from the lung tissues of mice after SCIT modeling.
Results: Previously, our group demonstrated that Notch signaling pathway inhibitors can reduce allergic airway inflammation in mice. Notch signaling induces lineage plasticity of mature ILC2s. In this study, we showed that AIT alleviates allergic airway inflammation and suppresses the frequency of ILC2s induced by HDM. Interestingly, AIT combined with a γ-secretase inhibitor (GSI), an inhibitor of the Notch signaling pathway, significantly inhibited the frequency of ILC2s, reduced airway inflammation, and suppressed Th2-type responses in a mouse model. Furthermore, lung ILC2s from HDM-challenged mice with or without AIT were treated with GSI in vitro, and we found that GSI dramatically reduced the secretion of type 2 inflammatory factors in ILC2s.
Discussion: These findings suggest that Notch signaling pathway inhibitors can be used as adjuvant therapy for AIT and may hold potential treatment value in the cooperative control of allergic airway inflammation during early AIT.
Asthma is an airway disease characterized by airway hyperresponsiveness (AHR), airway inflammation, and increased mucus production. Overall, more than half of asthmatic adults and pediatric patients have allergic asthma (1). House dust mites (HDMs) are the most prevalent allergen among allergic asthma patients (2). Although the rate of allergic asthma decreases as the age of onset increases (3), airway damage from childhood asthma can persist into adulthood (4). Historically, allergic asthma was thought to be an inflammatory disease associated with T-helper 2 (Th2) cells (5). More recently, Group 2 innate lymphoid cells (ILC2s) have also been considered as playing an overwhelming role in the inflammatory response to allergic asthma (6, 7).
Allergen immunotherapy (AIT), which aims to prevent the development of new allergic conditions, is extensively utilized in the clinical treatment of house dust mite (HDM) allergic diseases. This therapy primarily encompasses subcutaneous immunotherapy (SCIT) and sublingual immunotherapy (SLIT) (8). Although much clinical research has demonstrated that AIT has considerable therapeutic effects (9–14), it is not effective for everyone. Therefore, attempts have been made to find new ways to enhance the effect of AIT. AIT is considered to work by reversing allergen-induced modifications in the balance between pro-inflammatory and anti-inflammatory T cells. During allergen immunotherapy (AIT), exposure to high doses of allergens can have dual effects. On one hand, it can induce epithelial cells to secrete anti-inflammatory mediators, such as interleukin (IL)-37 (15) and secretoglobin1A1 (16, 17). These mediators play a crucial role in modulating airway allergic responses by inhibiting pro-inflammatory factors. On the other hand, the presence of transforming growth factor (TGF)-β can induce the proliferation of a specific subset of regulatory T cells (Treg cells). This expansion of Treg cells further contributes to the regulation of immune responses during AIT.These cells decrease Th2 cell responses while inducing a shift to Th1 cell responses (18).
Notably, AIT has been shown to modulate ILC2s. Clinical studies have found a sustained decrease in ILC2s in the peripheral blood of allergic patients after AIT (19–21). Innate lymphoid cells (ILCs), which lack antigen-specific receptors and predominantly reside in mucosal tissues (22, 23), are a subset of ILCs. Among them, ILC2s express the transcription factor GATA-binding Factor 3 (GATA3) and play a role similar to Th2 cells by releasing type 2 cytokines such as IL-13 and IL-5 (24–26). Undeniably, ILC2s play an integral part in allergic asthma (27). When exposed to allergens, epithelial cells release epithelial cell-derived cytokines, such as IL-33, IL-25, and thymic stromal lymphopoietin (TSLP), which activate ILC2s, resulting in a type 2 immune response (28, 29).
The Notch signaling pathway, which is broadly observed in mammals and other animals, plays an integral role in the growth and evolution of lymphocytes (30). In our previous experiments, it was found that γ-secretase inhibitors (GSIs) mitigated airway inflammation by blocking the Notch signaling pathway in OVA-induced asthmatic mice (31, 32). Similarly, the Notch signaling pathway is critical for the development of ILC2s. It has been discovered that Notch signaling is necessary for the accumulation of inflammatory group 2 innate cells in the lung (33). Natural ILC2s were cultivated in vitro in IL-2, IL-7, and IL-25 conditions, and the activation of the Notch signaling pathway dramatically boosted the presence of ILC2s, as well as the expression of IL-13 and IL-5 (34). More crucially, precursor cells, rather than T cells, drive the formation and differentiation of ILC2s under conditions of high Notch signaling intensity (35). However, it remains elusive as to whether the Notch signaling pathway regulates the activation of ILC2s and interferes with the efficacy of AIT treatment for allergic asthma.
In this study, we investigated the effect of GSI on the function of ILC2s in the context of HDM-induced allergic airway inflammation. Our findings revealed that GSI inhibits the secretion of cytokines by ILC2s. We established a mouse model of HDM-induced airway allergic disorders (HAAD) and treated HAAD mice with SCIT, with or without GSI. Our results demonstrated that GSI significantly enhanced the effectiveness of SCIT for HAAD and suppressed the response of ILC2s.
Wild-type BALB/c mice were obtained from Hangzhou Charles River Laboratories, China. All mice were housed in a specific pathogen-free environment, and female mice weighing 18-20 g were used for experiments. All experiments on animals were conducted in accordance with ethical protocols approved by the Laboratory Animal Ethics Committee of Wenzhou Medical University & Laboratory Animal Centre of Wenzhou Medical University.
All mice were randomly assigned to one of four groups: the control group, HDM group, HDM+SCIT group, and HDM+SCIT+GSI group. In this study, we established a model of HAAD and SCIT as described in Figure 1 and Supplementary Table 1. Mice weighing 18-22 g received an intraperitoneal injection of 5 μg HDM (XPB91D3A25, Greer Laboratories, Lenoir, North Carolina, USA) adsorbed on 2.25 mg of aluminum hydroxide in 100 μl PBS on Days 1 and 15. The mice in the HDM+SCIT and HDM+SCIT+GSI groups were desensitized through subcutaneous injection of 250 μg HDM in 100 μl PBS on Days 29, 31, and 33 (36). The mice in the control group were injected with PBS. In the HDM+SCIT+GSI group, GSI L685, 458 (Merck, USA) (0.3 mg/kg) was given to the mice intratracheally 30 minutes (37–39) before each SCIT. Ultimately, the mice were challenged with 25 μg of HDM in 25 μl of PBS via intratracheal direct injection on Days 45, 47, and 49.
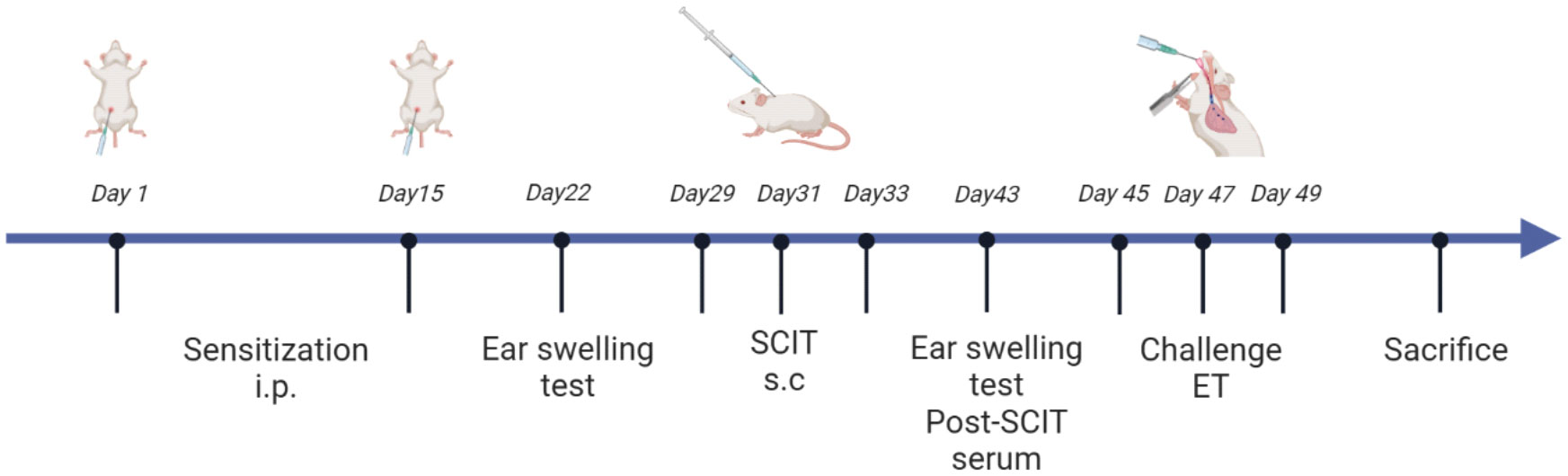
Figure 1 HDM-induced subcutaneous immunotherapy in asthmatic mice as a paradigm, created in BioRender.com.
The ear swelling test (EST) (36) was measured to assess the early phase reaction to HDM before and after SCIT on Days 22 and 32, in order to evaluate the allergic sensitization of the mice. Following anesthesia with isoflurane/oxygen, mice were intradermally injected with 10 μl PBS containing 0.5 μg HDM in the right ear and subcutaneously with 10 μl PBS in the left ear as a control. After 2 hours, the thickness of the mouse ears was measured using a digimatic micrometer (Mitutoyo, Japan), and the thickness of the right ear was subtracted from the thickness of the left ear to calculate the degree of HDM-induced swelling.
After the final HDM challenge, mice were anesthetized via intraperitoneal injection of 1% pentobarbital sodium. Once the mice had completely ceased spontaneous respiration, they were immobilized and the trachea was surgically incised for tracheal intubation. Subsequently, the inserted tracheal tube was connected to SCIREQ Scientific Respiratory Equipment (FlexIVent, Montreal, Canada), and the intermittent ventilation mode (150 breaths/min, PEEP 2 cmH2O) was selected. Baseline airway resistance (Rn) was measured at the stage of saline nebulizing. Then, the mice were challenged with an increasing dose (3.125, 6.25, 12.5, 25, and 50 mg/ml) of methacholine (MCh). And airway Rn was measured at every dose to calculate the percentage of each concentration to baseline.
After evaluating the lung function of the mice, they were cannulated, and the lungs were washed three times with 0.8 ml of cold PBS. The supernatant was collected by centrifuging the bronchoalveolar lavage (BAL) fluid for ELISA, and the cells were resuspended in cold PBS for counting and analyzed by flow cytometry (FCM).
The total protein in lung tissue was isolated and quantified. Protein lysates were electrophoresed using the PAGE Gel Fast Preparation Kit (Epizyme Biotech, China) and then electrotransferred to a polyvinylidene fluoride (PVDF) membrane. The resulting blots were blocked with 5% milk and incubated overnight with the following primary antibodies: anti-Notch1 (Abcam, UK), anti-Hes-1 (Cell Signaling Technology, USA), and anti-tubulin (Affinity Biosciences, USA). Subsequently, goat anti-rabbit IgG-HRP (Affinity, China) as the secondary antibody was incubated at room temperature for 30 minutes.
TRIzol (GlpBio, USA) was employed to extract total RNA from lung tissue. Next, cDNA was synthesized by reverse transcription using qRT Master Mix (TOROIVD, China) according to the manufacturer’s instructions. To examine gene mRNA expression, qPCR (Takara, Japan) was performed with SYBR Green PCR Master Mix. Supplementary Table 3 shows the primer sequences that were employed.
Mouse lung tissues were removed and cleaned in PBS to remove residual blood after right heart lavage with cold PBS under aseptic conditions. Fresh tissues were diced and digested in RPMI1640 medium containing 1 mg/ml Collagenase Type IV (Worthington, USA) and 0.2 mg/ml DNase I (Sigma, UAS) in a 37°C cell incubator for 1 h. Digested tissues were ground and filtered on a 70 μm filter and then further isolated and purified with Ficoll gradient (TBDscience, China). Naturally, the isolated cells were lysed by red blood cell lysis buffer to obtain a single-cell suspension of lung tissues for the following experiments.
Before staining with fluorochrome-conjugated antibodies, which are listed in Supplementary Table 2, single-cell suspensions were blocked with anti-Fc receptor antibodies (anti-mouse CD16/32). For the flow cytometric fluorescence sorting (FACS), a cell viability dye (7-AAD, BD Bioscience, USA) was used to exclude dead cells. Afterward, fluorochrome-conjugated antibodies were used for surface dyeing (lineage antibodies, anti-CD45, anti-ST2, anti-CD127). We defined the population of cells expressing Lin-CD45+CD127+ST2+ as ILC2s. The lineage antibodies we used included anti-CD3e, anti-CD11b, anti-CD45R/B220, anti-TER-119, anti-Ly-6G, and anti-Ly-6C, and then a MoFlo Astrios EQ (Beckman Coulter, USA) was employed for sorting. The purity of isolated ILC2s was up to 96.2%. For intracellular transcription factor staining, sorted ILC2s were handled with the Foxp3/Transcription Factor Staining Buffer Set (eBioscience, USA) according to instructions of the manufacturer after in vitro culture and stained with fluorochrome-conjugated antibodies anti-GATA3 antibodies at 4°C for 1.5 h.
To investigate the function of ILC2s in vitro, lung ILC2s were isolated from the HDM group and HDM+SCIT group, cultured on 96-well plates at a density of 5×103 per well, and processed with GSI (20 μM/ml) or PBS in the presence of mouse IL-2 (10 ng/ml, PeproTech, USA) and IL-7 (10 ng/ml, PeproTech, USA). The intracellular transcription factor GATA3 of ILC2s was analyzed by FCM after stimulation for 16 h, while the cytokines in the culture supernatant were detected by ELISA after stimulation for 48 h.
The BAL fluid and serum from mice, as well as the supernatant from cultured ILC2s, were collected and stored at -80°C. Levels of the cytokines IL-4, IL-5, IL-13, and IgE were determined by ELISA as described by the manufacturer (Thermo Fisher, USA). To detect HDM-sIgE, we coated HDMs (10 μg/plate) on 96-well plates and incubated them overnight at 4°C. After washing with PBST, the coated 96-well plates were blocked with assay diluent buffer for 2 h at room temperature. Then, a 1:80 dilution of serum was added to the plate and incubated at room temperature for 2 h. Subsequently, after washing with PBST, the detection antibody (1:1000 diluted, Biotin Rat Anti-Mouse, BD Pharmingen, USA) was added to the plate and incubated at room temperature for 1 h. Next, 100 μl 1× streptavidin-horseradish peroxidase (HRP) was added to the 96-well plate and incubated for half an hour at room temperature. At last, 100 μl TMB was added to the plate and incubated for 15 min and then the reaction was stopped by adding stop solution. The absorbance of the plate was detected at 450 nm using an enzyme marker. The concentration of total IgE ranged from 4 to 250 ng/ml, with a detection range of 4-500 pg/ml for IL-4, IL-5, and IL-13.
Fresh lung tissue in 4% paraformaldehyde solution fixation for 48 h, followed by paraffin embedding. After tissue sectioning, hematoxylin-eosin (H&E) staining, Masson staining, and Alcian Blue Periodic acid Schiff (AB-PAS) staining were followed. Airway inflammation scores were referenced from our previous report (31, 37).
All data are expressed as the mean ± SEM and statistical significance was determined by one-way ANOVA or Student’s two-tailed t-test. GraphPad Prism 8.0 software was used for statistical analysis, and FlowJo 10.6.2 software was employed for FCM analysis. P<0.05 was recognized as a significant difference.
To investigate the potential impact of GSI on ILC2s function, we primarily evaluated its effect on the production of type 2 cytokines by HDM-activated ILC2s. To promote the expansion of ILC2s, BALB/c mice were pretreated with HDM, and lung ILC2s were isolated as Lin-CD45+ST2+CD127+ cells (Figure 2A) using the sorting gating strategy (S3). We treated sorted lung ILC2s with GSI and observed a notable decrease in the levels of IL-13 and IL-5 in the supernatant of cultured ILC2s, as well as a reduction inintracellular GATA3 expression levels in ILC2s (Figures 2B–E).
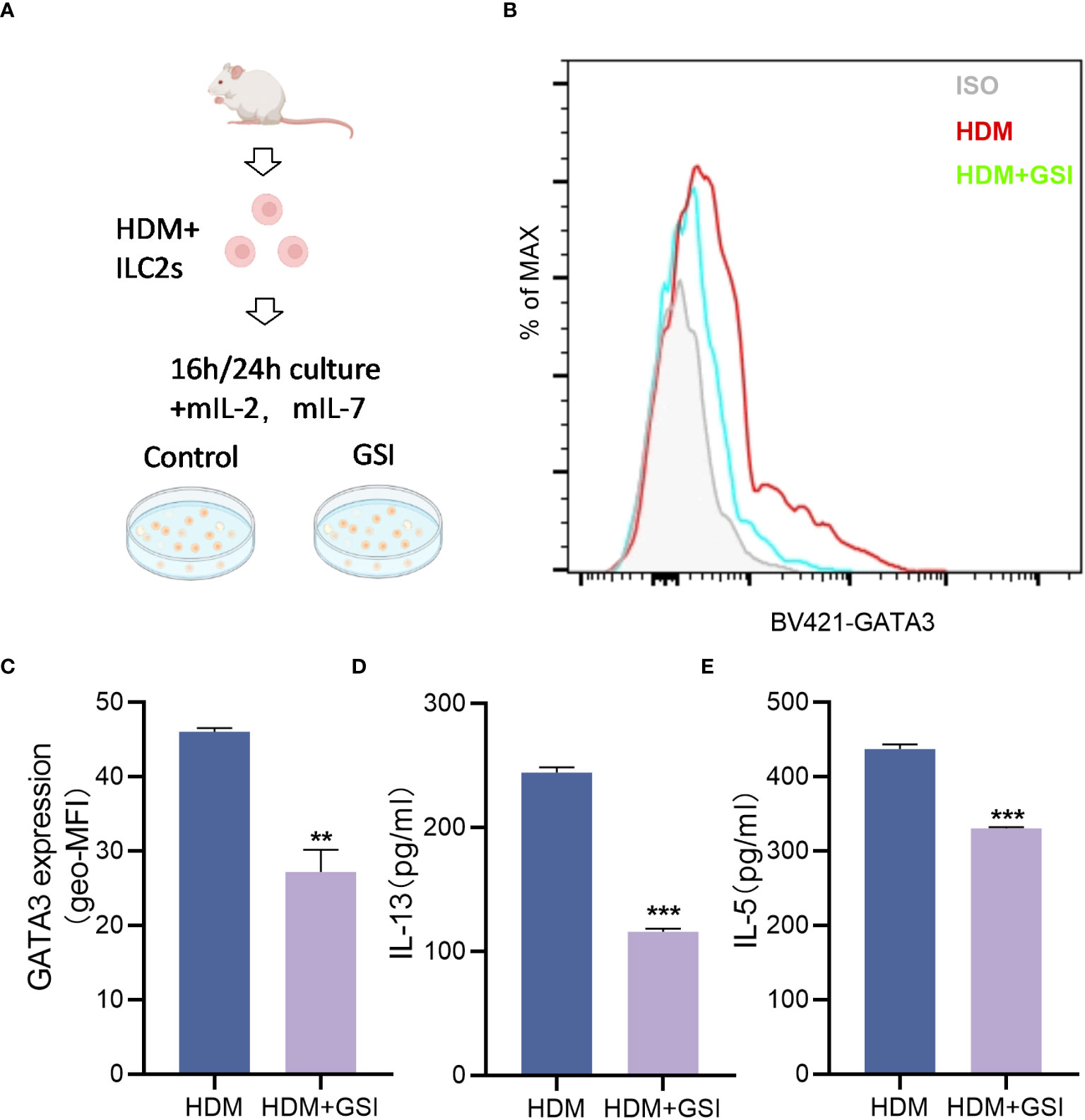
Figure 2 GSI inhibits intracellular GATA3 expression and type 2 cytokine generation in ILC2s in vitro. (A) Diagram showing HDM-induced asthmatic mouse lung tissue extracted and cultivated in vitro using FACS for ILC2s. (B, C) The mean fluorescence intensity (MFI) of GATA3 within ILC2s was measured by FCM after 16 h incubation of ILC2s in the presence of PBS/GSI. (D, E) ILC2s were cultured in the presence of PBS/GSI for 24 h and then the expression levels of IL-5 and IL-13 were measured by ELISA. The data represent three independent experiments, and mean ± SEMs are shown. Student’s two-tailed t-test was used. **P <0.01, ***P <0.001 compared with control group.
To examine the expression of Notch in the HAAD model, lung tissues were harvested and assessed using western blotting and qPCR. As shown in Figure 3, the mRNA expression of Notch1 was significantly higher in the lung tissue of the HDM group compared to the control group. Interestingly, the protein expression of Notch1 and its downstream target Hes-1 was dramatically elevated in the lung tissue of HDM mice compared to the control group. These findings indicate that HDM induces an increase in Notch signaling expression in the lung tissue of asthmatic mice.

Figure 3 Notch1 and Hes-1 protein expression levels, as well as Notch1 mRNA expression levels, were measured. (A) mRNA expression level of Notch1. (B–D) Protein expression level of Notch1 and Hes-1. These data represent at least three independent experiments, and mean ± SEMs were shown. Student’s two-tailed t-test was used. ***P <0.001 compared with the control group.
To begin with, we evaluated the extent of ear swelling in mice before and after SCIT treatment (Figure 4A). We observed that ear swelling was significantly more severe in the asthma group of mice compared to the control group (P<0.001), but it was reduced after SCIT treatment compared to the asthma group (P<0.001). This indicates that SCIT therapy suppresses early allergic reactions in mice. Subsequently, we measured total serum IgE and HDM-specific IgE (HDM-sIgE) levels in mice post-SCIT and challenge (Figures 4B, C). The total serum IgE levels in the SCIT group post-SCIT and challenge were not statistically significant compared to the asthma group. However, serum HDM-sIgE levels in the SCIT group were significantly lower post-challenge than those in the asthma group (P<0.001). These results suggest a successful SCIT model. Airway resistance (Rn) was evaluated after the last challenge. The results (Figures 4D–H) showed that airway resistance was higher in the HDM group of mice compared to the control group (P<0.5), while SCIT significantly reduced Rn (P<0.5). Interestingly, the decrease in airway resistance was more pronounced in the HDM+SCIT+GSI group mice compared to the HDM+SCIT group(P<0.5). These observations suggest that GSI in combination with SCIT can attenuate HDM-induced AHR and early allergic reactions in mice.
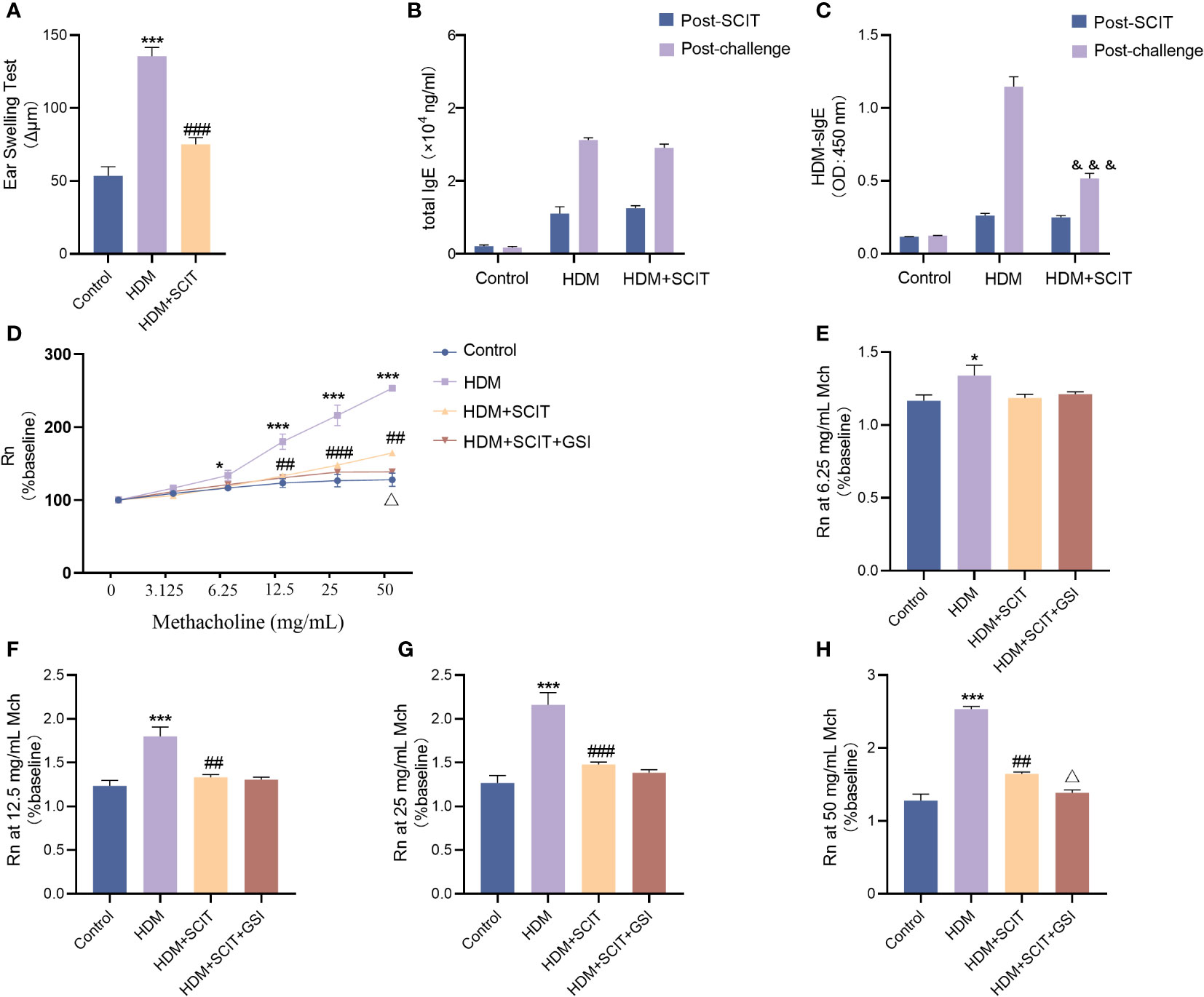
Figure 4 GSI combined with SCIT attenuates AHR in mice of HAAD. Experiment diagram of animal model and changes in airway resistance of each group. (A) EST reaction early allergic reaction. (B) Change in total IgE before and after the challenge. (C) Change in HDM-sIgE before and after the challenge. (D–H) Comparison of airway resistance in each group of mice at different methacholine concentrations. The data represent at least three independent experiments, mean±SEMs were shown. One-way ANOVA test was used for (A, D–H). *P <0.05, ***P <0.001 compared with control group. ##P <0.01, ###P <0.001, compared with HDM group. △P <0.05, compared with HDM+SCIT group. Two-way ANOVA test was used for (B, C). &&&P <0.01, compared to HDM group post challenge.
The lung histopathological sections were stained with H&E(Figures 5A–C). As expected, bronchial wall thickness decreased after SCIT and inflammatory cell infiltration in lung tissue was alleviated (P<0.5). The total cell count and eosinophil count of BAL fluid were significantly reduced (Figures 5F–H). More significantly, inflammatory cell infiltration in the lungs of mice in the HDM+SCIT+GSI group was considerably lower than in the HDM+SCIT group (Figures 5A–C). Similarly, the HDM+SCIT+GSI group showed the most prominent decrease in BALF total cell count and eosinophil count compared to the HDM+SCIT group (Figures 5F–H).
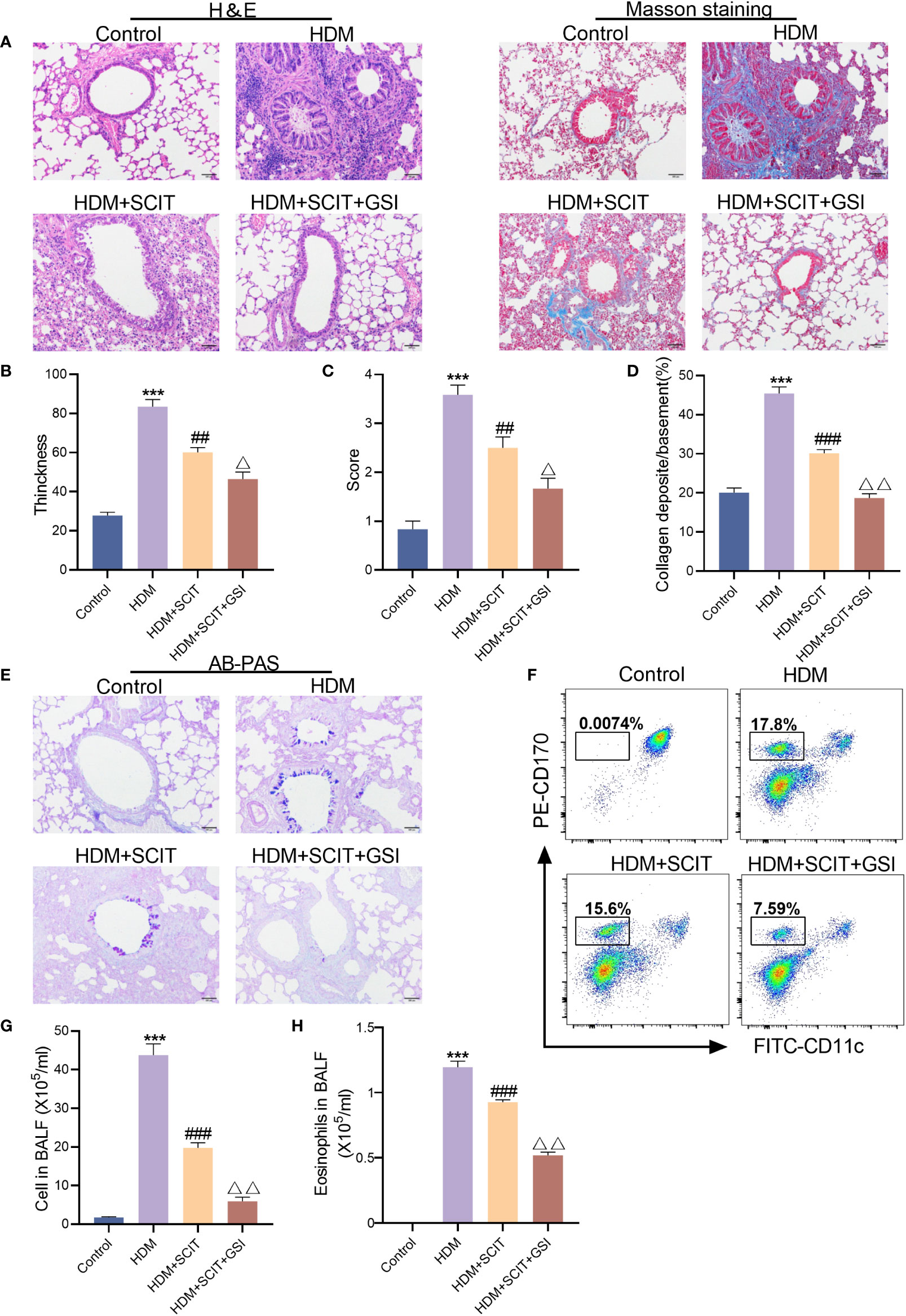
Figure 5 GSI and SCIT together reduce airway inflammation and inhibit airway collagen fibrosis and mucus secretion. (A) H&E staining and Masson staining of lung pathology sections (bars,100 μm). (B) Thickness of tracheal wall. (C) Semi-quantitative analysis to assess lung tissue inflammation. (D) Quantitative analysis of the percentage of peritracheal collagen deposition. (E) AB-PAS staining of lung pathology sections (bars,100 μm). (F) Eosinophil flow gating strategy. (G) Tatal cell in BAL fluid. (H) Eosinophils in BAL fluid. The data represent at least three independent experiments, mean±SEMs were shown. One-way ANOVA test was used. ***P <0.001 compared with control group. ##P <0.01, ###P <0.01, compared with HDM group. △P <0.05, △△P <0.01, compared with HDM+SCIT group.
Afterward, to further investigate the effect of GSI combined with SCIT on asthma, lung tissues of mice were examined using Masson staining (Figures 5A, D) and AB-PAS staining (Figure 5E). We found that SCIT dramatically reduced peritracheal collagen deposition (P<0.5) and decreased airway mucus secretion. Unexpectedly, the percentage of peritracheal collagen deposition in mice in the HDM+SCIT+GSI group (P<0.5) decreased obviously compared with that in mice in the HDM+SCIT group, and airway mucus secretion was also reduced. Overall, these results indicate that GSI combined with SCIT can synergistically alleviate airway inflammation and inhibit airway collagen fibrosis and mucus secretion.
Asthma is closely correlated with the type 2 cytokines interleukin (IL)-4, IL-5, and IL-13 (40). Therefore, we measured IL-4, IL-5, and IL-13 levels in serum and BALF using ELISA. The results (Figure 6) showed that IL-4, IL-5, and IL-13 levels were substantially increased in the HDM group compared to the control group (P<0.5), while the HDM+SCIT group (P<0.5) showed a significant decrease compared to the HDM group. IL-4, IL-5, and IL-13 levels were further reduced by combining SCIT with GSI treatment (P<0.5). These results indicate that combining SCIT and GSI can effectively reduce inflammatory cytokines in BAL fluid and serum.
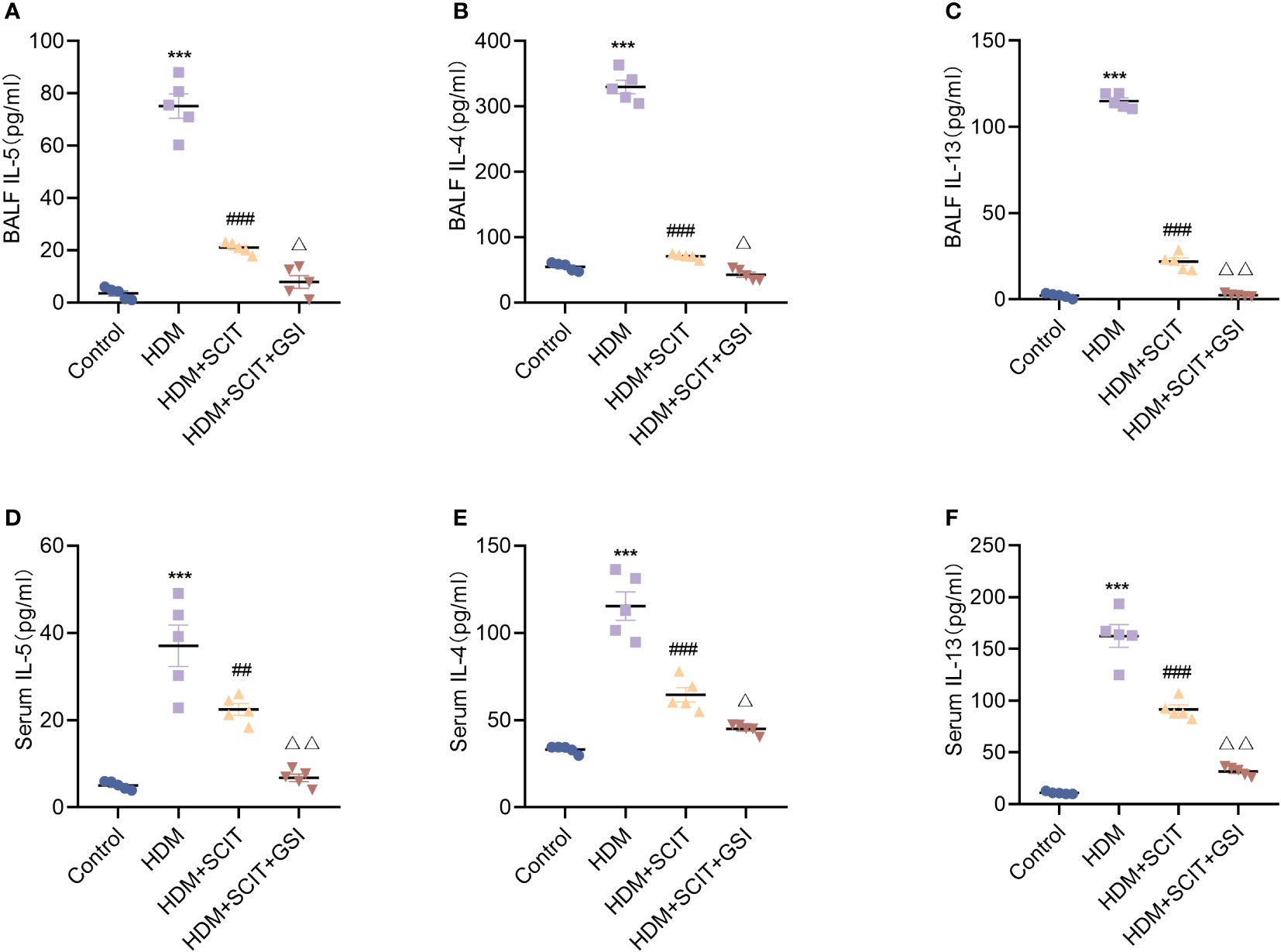
Figure 6 GSI further suppresses Th2-type inflammatory responses based on SCIT. (A–C) Assay of IL-4, IL-5 and IL-13 levels by ELISA in BAL fluid. (D–F) Measurement of IL-4, IL-5 and IL-13 levels by ELISA in serum. The data represent at least three independent experiments, mean±SEMs were shown. One-way ANOVA test was used. ***P <0.001 compared with control group. ##P <0.01, ###P <0.001, compared with HDM group. △P <0.05, △△P <0.01, compared with HDM+SCIT group.
Next, we investigated the effect of SCIT in combination with Notch signaling pathway inhibitors on ILC2s. We first evaluated the ratio of lung ILC2s in the HAAD and SCIT models (Figures 7A, B). Using FCM, we found that the percentage of ILC2s decreased after SCIT compared to the HDM group (P<0.5). When SCIT and GSI were combined (P<0.5), the ILC2s ratio decreased significantly compared to SCIT alone. Furthermore, ILC2s were extracted by FACS from lung tissue of mice in the HDM+SCIT group and cultured in vitro (Figures 7C–G). It was discovered that inhibiting the Notch signaling pathway decreased the MFI of intracellular GATA3 in ILC2s and reduced IL-13 and IL-5 production. Overall, SCIT and GSI treatments have the potential to synergistically suppress the activation of ILC2s in mice with HAAD.
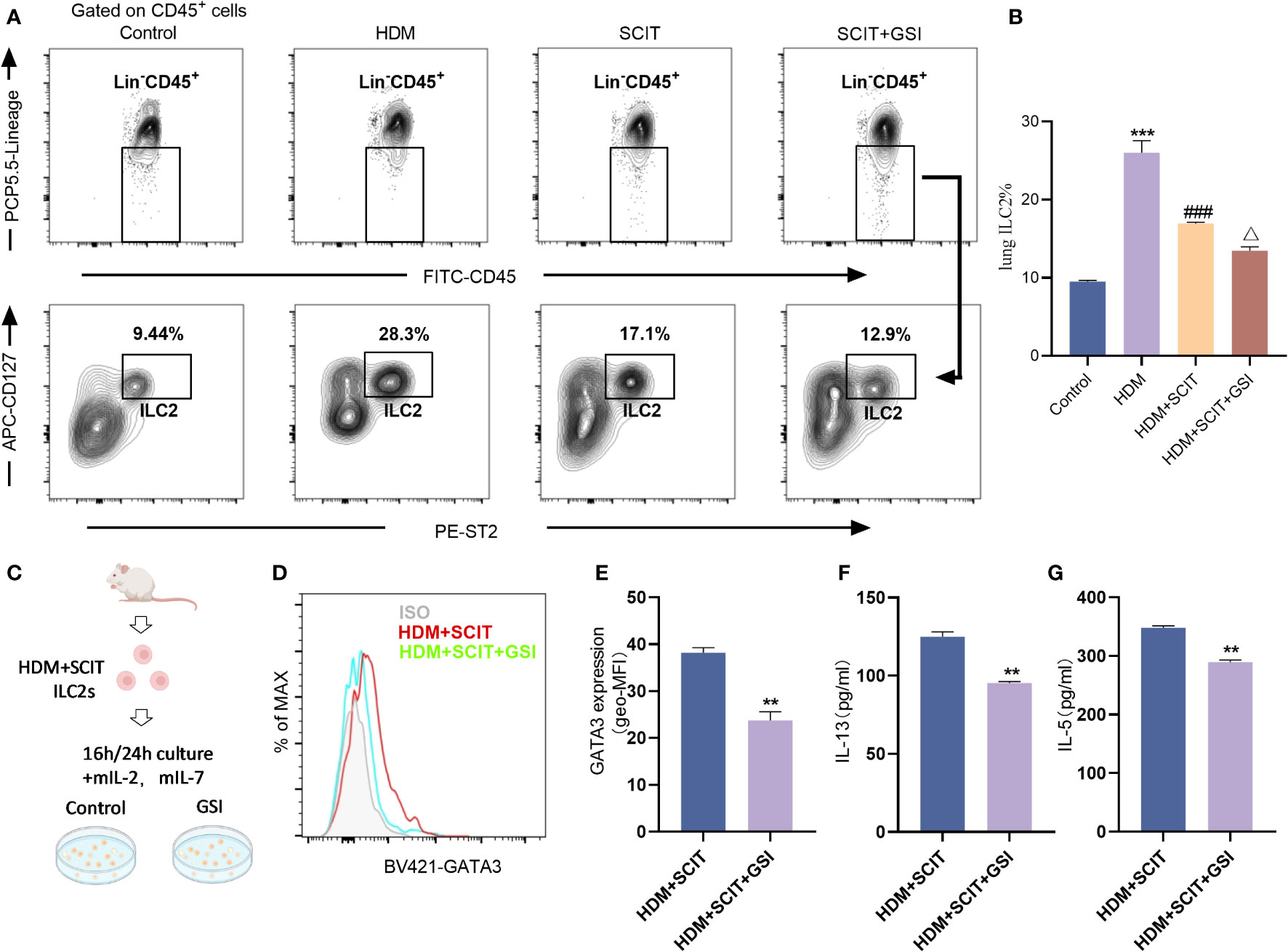
Figure 7 GSI combined with SCIT treatment inhibits the activation of ILC2s in vivo or in vitro. (A) Flow cytometry strategy for gating of ILC2s [Lin-CD45+CD127+ST2+ (41, 42)] in lung tissue. The detailed gating strategy diagram for ILC2s is shown in the Supplementary Material Figure S2. (B) Frequency of ILC2s in mouse lung tissue. The data represent at least three independent experiments, and mean ± SEMs were shown. (C) Schematic diagram of in vitro culture of ILC2s sorted from mice lung tissue after HDM challenge and SCIT treatment. (D, E) The mean fluorescence intensity (MFI) of GATA3 within ILC2s was measured by FCM after 16 h incubation of ILC2s in the presence of PBS/GSI. (F, G) ILC2s were cultured in the presence of PBS/GSI for 24 h and then the expression levels of IL-5 and IL-13 were measured by ELISA. A one-way ANOVA test was used for (B). ***P <0.001 compared with the control group. ###P <0.001, compared with the HDM group. △P <0.05, compared with the HDM+SCIT group. Student’s two-tailed t-test was used for (E–G). **P <0.01, ***P <0.001 compared with control group.
In this study, we found that GSI was able to inhibit the activation of ILC2s in vitro and further reduce the production of IL-5 and IL-13 by ILC2s after SCIT. Additionally, we observed that SCIT therapy reduces airway inflammation in an HDM-induced immunotherapy model for asthma. Interestingly, when combined with SCIT treatment, GSI can further suppress airway inflammation and decrease the generation of inflammatory factors by ILC2s. This study demonstrates that GSI improves the efficacy of SCIT in the treatment of HDM-induced asthma and reduces ILC2s activation.
Allergen combination strategies have introduced novel concepts for the treatment of allergic disorders (43). In this study, we confirmed that HAAD mice with high levels of Notch signaling and activated ILC2s can be effectively treated with a combination of GSI and SCIT. We observed that this combination therapy alleviated HDM-induced airway inflammation and airway hyperresponsiveness in mice. Previous clinical studies have shown that combining anti-IgE monoclonal antibody (omalizumab) with AIT reduced allergy symptoms but did not have a long-term tolerance effect (44, 45). Clinical phase 2a trials have also demonstrated the potential of anti-IL-33 biologics to desensitize peanut-allergic patients (46). However, monoclonal antibodies are expensive and may not be suitable for everyone (47, 48). In contrast, our study provides a new and feasible approach to allergen combination therapy, which could be a valuable addition to adjuvant therapy for AIT.
A study suggests that AIT may targetthe prostaglandin EP3 receptor, leading to a reduction in IL-13+ILC2s (49). Several studies have also reported that the Notch signaling pathway plays a role in regulating ILC2s and type 2 inflammation (33, 34). In this study we investigated the Notch signaling pathway in lung ILC2s. Inhibiting the Notch signaling pathway effectively blocked the activation of ILC2s induced by HDM, as evidenced by decreased GATA3 expression and reduced production of IL-13 and IL-5. GATA3 is a central regulator of ILC2s (50). Furthermore, GSI decreased the expression of GATA3 and the secretion of type 2 inflammatory factors in ILC2s isolated from HDM-challenged mice receiving SCIT. These findings suggest that GSI may enhance the efficacy of SCIT by modulating the function of ILC2s.
Overall, SCIT and GSI treatments have the potential to synergistically suppress the activation of ILC2s in HDM-induced mice. In a previous study using a sensitized mouse model, we demonstrated that the Notch signaling pathway regulates the proliferation and activation of CD4+ T lymphocytes (32). Blocking this pathway with an anti-Dll4 antibody reduced allergic airway inflammation in mice (32). Our clinical trial revealed a correlation between Th17/Treg dysregulation and increased Notch expression in the peripheral blood of children with allergic asthma (51), highlighting the importance of the Notch signaling pathway in asthma pathogenesis. Furthermore, we found that GSI treatment alleviated allergic airway inflammation in mouse model (32) and reduced airway hyperresponsiveness in obese mice (37). These findings suggest that GSI could be a potential therapeutic strategy in allergic asthma. In our current study, we showed that combining GSI with SCIT not only reduced airway inflammation and AHR but also decreased the frequency of ILC2s in lung tissue in vivo experiments. Additionally, inhibiting the Notch signaling pathway in vitro significantly reduced the secretion of type 2 inflammatory factors by ILC2s following SCIT treatment. These results highlight the potential of combining GSI with SCIT as a novel approach to allergen combination therapy, offering new insights for the clinical treatment of asthma.
However, there are still several unresolved issues. The use of HDM to induction of allergic airway inflammation in our study brings us closer to a clinically relevant model compared to previous OVA-induced asthma models. Moreover, the AIT model established in this experiment better simulates the effects observed in humans. However, recent research has proposed the use of low-dose allergens combined with adjuvants as an AIT strategy (52), which provides us with a new idea for model optimization. Additionally, one study found that immunotherapy promoted the production of IL-10-expressing ILC2s in vivo (53), which may be related to the establishment of immune tolerance in the late stages of AIT. Therefore, further investigation is needed to understand the changes in ILC2s at each stage of AIT treatment. Third, the Notch signaling pathway has been found to contribute to the establishment of sustained unresponsiveness (SU) induced by oral immunotherapy in food allergy immunotherapy (54). This may be because inhibition of the Notch signaling pathway in the early stages of AIT reduces the production of activated ILC2s, which secrete type 2 inflammatory factors. However, during immune tolerance, inhibition of the Notch signaling pathway further limits the production of immunosuppressive cells in the bone marrow, leading to different outcomes. In addition, it has been found that immunotherapy inhibits the secretion of Th2 and Th9 cells as well as TGF-β, which decreases the induction of Th2 and Th9 cells and increases Treg cells. Interestingly, neutralizing TGF-β does not affect Treg cells, leading to immune dysregulation (55). Our team’s clinical studies have found that immunotherapy suppresses inflammation by increasing Treg and reversing the Th17/Treg ratio (56). However, it remains to be investigated whether GSI combined with AIT treatment affects these cells, as well as TGF-β.
In conclusion, our study revealed the cooperative effect of GSI in the treatment of asthma by SCIT, providing a new idea for the synergistic effect of GSI on the treatment of asthma by SCIT.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The animal study was approved by Laboratory Animal Ethics Committee of Wenzhou Medical University & Laboratory Animal Centre of Wenzhou Medical University. The study was conducted in accordance with the local legislation and institutional requirements.
Conception: HZ, and LW, LYW. Methodology: YT and LYW. Experiments: YT, LW, LYW, JJS, JF, CL, JB, CW YW, and JLS. Data analysis: YT, LW. Statistics: LYW, JJS and JF. Writing of the manuscript: YT and HZ. Study supervision, project administration, and funding acquisition: HZ and WZ. All authors contributed to the article and approved the submitted version.
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was financially supported by the Medical Science and Technology Project of Zhejiang Province (WKJ-ZJ-2133), the Natural Science Foundation of Zhejiang Province (LQ23H310007), and Wenzhou Municipal Science and Technology Bureau (Y20220266).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2023.1264071/full#supplementary-material.
1. Porsbjerg C, Melen E, Lehtimaki L, Shaw D. Asthma. Lancet (2023). 401:858–73. doi: 10.1016/S0140-6736(22)02125-0
2. Sanchez-Borges M, Fernandez-Caldas E, Thomas WR, Chapman MD, Lee BW, Caraballo L, et al. International consensus (ICON) on: clinical consequences of mite hypersensitivity, a global problem. World Allergy Organ J (2017) 10(1):14. doi: 10.1186/s40413-017-0145-4
3. Honkamaki J, Hisinger-Molkanen H, Ilmarinen P, Piirila P, Tuomisto LE, Andersen H, et al. Age- and gender-specific incidence of new asthma diagnosis from childhood to late adulthood. Respir Med (2019) 154:56–62. doi: 10.1016/j.rmed.2019.06.003
4. Agusti A, Melen E, DeMeo DL, Breyer-Kohansal R, Faner R. Pathogenesis of chronic obstructive pulmonary disease: understanding the contributions of gene-environment interactions across the lifespan. Lancet Respir Med (2022) 10(5):512–24. doi: 10.1016/S2213-2600(21)00555-5
5. Lloyd CM, Hessel EM. Functions of T cells in asthma: more than just T(H)2 cells. Nat Rev Immunol (2010) 10(12):838–48. doi: 10.1038/nri2870
6. Kato A. Group 2 innate lymphoid cells in airway diseases. Chest (2019) 156(1):141–9. doi: 10.1016/j.chest.2019.04.101
7. Maazi H, Akbari O. Type two innate lymphoid cells: the Janus cells in health and disease. Immunol Rev (2017) 278(1):192–206. doi: 10.1111/imr.12554
8. Alvaro-Lozano M, Akdis CA, Akdis M, Alviani C, Angier E, Arasi S, et al. EAACI allergen immunotherapy user’s guide. Pediatr Allergy Immunol (2020) 31 Suppl 25(Suppl 25):1–101. doi: 10.1111/pai.13189
9. Karakoc-Aydiner E, Eifan AO, Baris S, Gunay E, Akturk E, Akkoc T, et al. Long-term effect of sublingual and subcutaneous immunotherapy in dust mite-allergic children with asthma/rhinitis: A 3-year prospective randomized controlled trial. J Investig Allergol Clin Immunol (2015) 25(5):334–42.
10. Zielen S, Kardos P, Madonini E. Steroid-sparing effects with allergen-specific immunotherapy in children with asthma: a randomized controlled trial. J Allergy Clin Immunol (2010) 126(5):942–9. doi: 10.1016/j.jaci.2010.06.002
11. Tanaka A, Tohda Y, Okamiya K, Azuma R, Terada I, Adachi M. Efficacy and safety of HDM SLIT tablet in Japanese adults with allergic asthma. J Allergy Clin Immunol Pract (2020) 8(2):710–720 e714. doi: 10.1016/j.jaip.2019.09.002
12. Bush RK, Swenson C, Fahlberg B, Evans MD, Esch R, Morris M, et al. House dust mite sublingual immunotherapy: results of a US trial. J Allergy Clin Immunol (2011) 127(4):974–981 e971-977. doi: 10.1016/j.jaci.2010.11.045
13. Arroabarren E, Tabar AI, Echechipia S, Cambra K, Garcia BE, Alvarez-Puebla MJ. Optimal duration of allergen immunotherapy in children with dust mite respiratory allergy. Pediatr Allergy Immunol (2015) 26(1):34–41. doi: 10.1111/pai.12296
14. Farraia M, Paciencia I, Castro Mendes F, Cavaleiro Rufo J, Shamji M, Agache I, et al. Allergen immunotherapy for asthma prevention: A systematic review and meta-analysis of randomized and non-randomized controlled studies. Allergy (2022) 77(6):1719–35. doi: 10.1111/all.15295
15. Schröder A, Lunding LP, Zissler UM, Vock C, Webering S, Ehlers JC, et al. IL-37 regulates allergic inflammation by counterbalancing pro-inflammatory IL-1 and IL-33. Allergy (2022) 77(3):856–69. doi: 10.1111/all.15072
16. Zissler UM, Jakwerth CA, Guerth F, Lewitan L, Rothkirch S, Davidovic M, et al. Allergen-specific immunotherapy induces the suppressive secretoglobin 1A1 in cells of the lower airways. Allergy (2021) 76(8):2461–74. doi: 10.1111/all.14756
17. Mootz M, Jakwerth CA, Schmidt-Weber CB, Zissler UM. Secretoglobins in the big picture of immunoregulation in airway diseases. Allergy (2022) 77(3):767–77. doi: 10.1111/all.15033
18. Shamji MH, Layhadi JA, Sharif H, Penagos M, Durham SR. Immunological responses and biomarkers for allergen-specific immunotherapy against inhaled allergens. J Allergy Clin Immunol Pract (2021) 9(5):1769–78. doi: 10.1016/j.jaip.2021.03.029
19. Mitthamsiri W, Pradubpongsa P, Sangasapaviliya A, Boonpiyathad T. Decreased CRTH2 expression and response to allergen re-stimulation on innate lymphoid cells in patients with allergen-specific immunotherapy. Allergy Asthma Immunol Res (2018) 10(6):662–74. doi: 10.4168/aair.2018.10.6.662
20. Lao-Araya M, Steveling E, Scadding GW, Durham SR, Shamji MH. Seasonal increases in peripheral innate lymphoid type 2 cells are inhibited by subcutaneous grass pollen immunotherapy. J Allergy Clin Immunol (2014) 134(5):1193–1195 e1194. doi: 10.1016/j.jaci.2014.07.029
21. Eljaszewicz A, Ruchti F, Radzikowska U, Globinska A, Boonpiyathad T, Gschwend A, et al. Trained immunity and tolerance in innate lymphoid cells, monocytes, and dendritic cells during allergen-specific immunotherapy. J Allergy Clin Immunol (2021) 147(5):1865–77. doi: 10.1016/j.jaci.2020.08.042
22. Bartemes KR, Kita H. Roles of innate lymphoid cells (ILCs) in allergic diseases: The 10-year anniversary for ILC2s. J Allergy Clin Immunol (2021) 147(5):1531–47. doi: 10.1016/j.jaci.2021.03.015
23. Barlow JL, McKenzie ANJ. Innate lymphoid cells of the lung. Annu Rev Physiol (2019) 81:429–52. doi: 10.1146/annurev-physiol-020518-114630
24. Klein Wolterink RG, Serafini N, van Nimwegen M, Vosshenrich CA, de Bruijn MJ, Fonseca Pereira D, et al. Essential, dose-dependent role for the transcription factor Gata3 in the development of IL-5+ and IL-13+ type 2 innate lymphoid cells. Proc Natl Acad Sci USA (2013) 110(25):10240–5. doi: 10.1073/pnas.1217158110
25. Hoyler T, Klose CS, Souabni A, Turqueti-Neves A, Pfeifer D, Rawlins EL, et al. The transcription factor GATA-3 controls cell fate and maintenance of type 2 innate lymphoid cells. Immunity (2012) 37(4):634–48. doi: 10.1016/j.immuni.2012.06.020
26. Mjosberg J, Bernink J, Golebski K, Karrich JJ, Peters CP, Blom B, et al. The transcription factor GATA3 is essential for the function of human type 2 innate lymphoid cells. Immunity (2012) 37(4):649–59. doi: 10.1016/j.immuni.2012.08.015
27. Rodriguez-Rodriguez N, Gogoi M, McKenzie ANJ. Group 2 innate lymphoid cells: team players in regulating asthma. Annu Rev Immunol (2021) 39:167–98. doi: 10.1146/annurev-immunol-110119-091711
28. Gurram RK, Zhu J. Orchestration between ILC2s and Th2 cells in shaping type 2 immune responses. Cell Mol Immunol (2019) 16(3):225–35. doi: 10.1038/s41423-019-0210-8
29. Gon Y, Hashimoto S. Role of airway epithelial barrier dysfunction in pathogenesis of asthma. Allergol Int (2018) 67(1):12–7. doi: 10.1016/j.alit.2017.08.011
30. Radtke F, MacDonald HR, Tacchini-Cottier F. Regulation of innate and adaptive immunity by Notch. Nat Rev Immunol (2013) 13(6):427–37. doi: 10.1038/nri3445
31. Zhang W, Zhang X, Sheng A, Weng C, Zhu T, Zhao W, et al. gamma-secretase inhibitor alleviates acute airway inflammation of allergic asthma in mice by downregulating th17 cell differentiation. Med Inflamm (2015) 2015:258168. doi: 10.1155/2015/258168
32. Zhang W, Nie Y, Chong L, Cai X, Zhang H, Lin B, et al. PI3K and Notch signal pathways coordinately regulate the activation and proliferation of T lymphocytes in asthma. Life Sci (2013) 92(17-19):890–5. doi: 10.1016/j.lfs.2013.03.005
33. Webb LM, Tait Wojno ED. Notch signaling orchestrates helminth-induced type 2 inflammation. Trends Immunol (2019) 40(6):538–52. doi: 10.1016/j.it.2019.04.003
34. Zhang K, Xu X, Pasha MA, Siebel CW, Costello A, Haczku A, et al. Cutting edge: notch signaling promotes the plasticity of group-2 innate lymphoid cells. J Immunol (2017) 198(5):1798–803. doi: 10.4049/jimmunol.1601421
35. Gentek R, Munneke JM, Helbig C, Blom B, Hazenberg MD, Spits H, et al. Modulation of signal strength switches notch from an inducer of T cells to an inducer of ILC2. Front Immunol (2013) 4:334. doi: 10.3389/fimmu.2013.00334
36. Hesse L, van Ieperen N, Habraken C, Petersen AH, Korn S, Smilda T, et al. Subcutaneous immunotherapy with purified Der p1 and 2 suppresses type 2 immunity in a murine asthma model. Allergy (2018) 73(4):862–74. doi: 10.1111/all.13382
37. Zeng Z, Wang L, Ma W, Zheng R, Zhang H, Zeng X, et al. Inhibiting the Notch signaling pathway suppresses Th17-associated airway hyperresponsiveness in obese asthmatic mice. Lab Invest (2019) 99(12):1784–94. doi: 10.1038/s41374-019-0294-x
38. Zeng Z, Lin X, Zheng R, Zhang H, Zhang W. Celastrol alleviates airway hyperresponsiveness and inhibits th17 responses in obese asthmatic mice. Front Pharmacol (2018) 9:49. doi: 10.3389/fphar.2018.00049
39. Kang JH, Kim BS, Uhm TG, Lee SH, Lee GR, Park CS, et al. Gamma-secretase inhibitor reduces allergic pulmonary inflammation by modulating Th1 and Th2 responses. Am J Respir Crit Care Med (2009) 179(10):875–82. doi: 10.1164/rccm.200806-893OC
40. Lambrecht BN, Hammad H, Fahy JV. The cytokines of asthma. Immunity (2019) 50(4):975–91. doi: 10.1016/j.immuni.2019.03.018
41. Helou DG, Shafiei-Jahani P, Lo R, Howard E, Hurrell BP, Galle-Treger L, et al. PD-1 pathway regulates ILC2 metabolism and PD-1 agonist treatment ameliorates airway hyperreactivity. Nat Commun (2020) 11(1):3998. doi: 10.1038/s41467-020-17813-1
42. Thio CL, Lai AC, Chi PY, Webster G, Chang YJ. Toll-like receptor 9-dependent interferon production prevents group 2 innate lymphoid cell-driven airway hyperreactivity. J Allergy Clin Immunol (2019) 144(3):682–697 e689. doi: 10.1016/j.jaci.2019.03.008
43. Durham SR, Shamji MH. Allergen immunotherapy: past, present and future. Nat Rev Immunol (2023) 23(5):317–28. doi: 10.1038/s41577-022-00786-1
44. Casale TB, Busse WW, Kline JN, Ballas ZK, Moss MH, Townley RG, et al. Omalizumab pretreatment decreases acute reactions after rush immunotherapy for ragweed-induced seasonal allergic rhinitis. J Allergy Clin Immunol (2006) 117(1):134–40. doi: 10.1016/j.jaci.2005.09.036
45. Dantzer JA, Wood RA. The use of omalizumab in allergen immunotherapy. Clin Exp Allergy (2018) 48(3):232–40. doi: 10.1111/cea.13084
46. Chinthrajah S, Cao S, Liu C, Lyu SC, Sindher SB, Long A, et al. Phase 2a randomized, placebo-controlled study of anti-IL-33 in peanut allergy. JCI Insight (2019) 4(22). doi: 10.1172/jci.insight.131347
47. Whittington MD, McQueen RB, Ollendorf DA, Tice JA, Chapman RH, Pearson SD, et al. Assessing the value of mepolizumab for severe eosinophilic asthma: a cost-effectiveness analysis. Ann Allergy Asthma Immunol (2017) 118(2):220–5. doi: 10.1016/j.anai.2016.10.028
48. Agache I, Beltran J, Akdis C, Akdis M, Canelo-Aybar C, Canonica GW, et al. Efficacy and safety of treatment with biologicals (benralizumab, dupilumab, mepolizumab, omalizumab and reslizumab) for severe eosinophilic asthma. A systematic review for the EAACI Guidelines - recommendations on the use of biologicals in severe asthma. Allergy (2020) 75(5):1023–42. doi: 10.1111/all.14221
49. Jakwerth CA, Chaker AM, Guerth F, Oelsner M, Pechtold L, Zur Bonsen LS, et al. Sputum microRNA-screening reveals Prostaglandin EP3 receptor as selective target in allergen-specific immunotherapy. Clin Exp Allergy (2021) 51(12):1577–91. doi: 10.1111/cea.14013
50. Stadhouders R, Li BWS, de Bruijn MJW, Gomez A, Rao TN, Fehling HJ, et al. Epigenome analysis links gene regulatory elements in group 2 innate lymphocytes to asthma susceptibility. J Allergy Clin Immunol (2018) 142(6):1793–807. doi: 10.1016/j.jaci.2017.12.1006
51. Li C, Sheng A, Jia X, Zeng Z, Zhang X, Zhao W, et al. Th17/Treg dysregulation in allergic asthmatic children is associated with elevated notch expression. J Asthma (2018) 55(1):1–7. doi: 10.1080/02770903.2016.1266494
52. Heldner A, Alessandrini F, Russkamp D, Heine S, Schnautz B, Chaker A, et al. Immunological effects of adjuvanted low-dose allergoid allergen-specific immunotherapy in experimental murine house dust mite allergy. Allergy (2022) 77(3):907–19. doi: 10.1111/all.15012
53. Golebski K, Layhadi JA, Sahiner U, Steveling-Klein EH, Lenormand MM, Li RCY, et al. Induction of IL-10-producing type 2 innate lymphoid cells by allergen immunotherapy is associated with clinical response. Immunity (2021) 54(2):291–307.e297. doi: 10.1016/j.immuni.2020.12.013
54. Yoneyama T, Nakano N, Hara M, Yamada H, Izawa K, Uchida K, et al. Notch signaling contributes to the establishment of sustained unresponsiveness to food allergens by oral immunotherapy. J Allergy Clin Immunol (2021) 147(3):1063–1076 e1069. doi: 10.1016/j.jaci.2020.07.011
55. Musiol S, Alessandrini F, Jakwerth CA, Chaker AM, Schneider E, Guerth F, et al. TGF-β1 drives inflammatory th cell but not treg cell compartment upon allergen exposure. Front Immunol (2021) 12:763243. doi: 10.3389/fimmu.2021.763243
Keywords: asthma, ILC2s, AIT, notch signaling pathway, HDM
Citation: Tong Y, Wang L, Wang L, Song J, Fan J, Lai C, Bao J, Weng C, Wang Y, Shuai J, Zhang H and Zhang W (2024) Allergen immunotherapy combined with Notch pathway inhibitors improves HDM-induced allergic airway inflammation and inhibits ILC2 activation. Front. Immunol. 14:1264071. doi: 10.3389/fimmu.2023.1264071
Received: 20 July 2023; Accepted: 29 December 2023;
Published: 02 February 2024.
Edited by:
Caterina Vizzardelli, Medical University of Vienna, AustriaReviewed by:
Ulrich Matthias Zissler, Technical University of Munich, GermanyCopyright © 2024 Tong, Wang, Wang, Song, Fan, Lai, Bao, Weng, Wang, Shuai, Zhang and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Weixi Zhang, emhhbmd3ZWl4aTExMkAxNjMuY29t; Hui Zhang, emhoODU1QDEyNi5jb20=
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.