- 1Instituto de Microbiologia Paulo de Góes, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil
- 2Laboratório de Pesquisa em Malária, Instituto Oswaldo Cruz (FIOCRUZ), Rio de Janeiro, Brazil
- 3Laboratório de Imunoparasitologia, Instituto Oswaldo Cruz (FIOCRUZ), Rio de Janeiro, Brazil
- 4Faculdade de Medicina, Universidade Federal do Rio de Janeiro, Rio de Janeiro, Brazil
Eryptosis is a programmed cell death-like process that occurs in red blood cells. Although the red blood cells are anucleated, there are similarities between eryptosis and apoptosis, such as increased calcium efflux, calpain activation, phosphatidylserine exposure, cell blebbing and cell shrinkage. Eryptosis occurs physiologically in red blood cells, as a consequence of the natural senescence process of these cells, but it can also be stimulated in pathological situations such as metabolic syndromes, uremic syndromes, polycythemia vera, anemias such as sickle cell anemia and thalassemia, and infectious processes including Plasmodium infection. Infection-induced eryptosis is believed to contribute to damage caused by Plasmodium, but it’s still a topic of debate in the literature. In this review, we provided an overview of eryptosis mechanisms and its possible pathogenic role in malaria.
Introduction
Apoptosis is a program cell death process that is a common mechanism among many cell types in physiology and disease. Apoptosis typically does not induce inflammation, or tissue scaring (1). The intrinsic pathways and machinery involved in this process is already well understood. A series of genetically regulated steps are essential for the process, with the activation of pro-apoptotic genes, biochemical and morphological changes within cells. These includes DNA fragmentation, cell surface externalization of phosphatidylserine and activation of caspases. In mammalian cells there are two major pathways to caspase-mediated cell death: the extrinsic and the mitochondria-dependent pathway (2).
Despite the absence of nucleus and mitochondria, in 2001, Bratosin and collaborators described a process similar to apoptosis occurring in red blood cells, later called eryptosis. Eryptosis has several similarities with apoptosis, regardless of the trigger, induction of an eryptotic state generally involves the entry of extracellular calcium into the cell, with activation of caspases and calpains, which induce membrane phosphatidylserine exposure, cell blebbing and cell shrinkage (3).
Eryptosis has been associated with several pathologies and can be triggered by several signals, including extended storage after blood donation (4), osmotic shock (5), xenobiotics, oxidative stress and energy depletion (6). Osmotic shock as well as oxidative stress induce the formation of Prostaglandin E2 (PGE2) that activates a calcium-permeable cation channel in the erythrocyte membrane, increasing the cytosolic calcium content, while diminishing chloride content and, consequently, induces cell shrinkage and phosphatidylserine exposure (7). During hyperosmotic shock, p38 MAPK (Mitogen-activated protein kinase) is expressed in erythrocytes and participates in the machinery triggering eryptosis (8).
One of the first events during eryptosis is the influx of calcium into the erythrocyte. In physiological situations, its concentration is much lower than the concentration of calcium in the blood plasma, at least 40,000 times (9). After stimulation, such as hyperosmotic shock and oxidative stress, the calcium channels present in the erythrocyte membrane open, increasing their influx into the cell (9).
These differences of calcium concentrations are maintained by the enzyme Plasma Membrane Ca2+ Pump (PMCP), with energy expenditure. This enzyme is able to sense the increase in cytosolic calcium levels through the calcium/calmodulin complex. Upon activation, calcium influx occurs mainly through nonspecific ion channels, as the transient receptor potential channel TRPC6, which is expressed in erythrocytes (10). Once into the cell, calcium binds to several proteins, associated with different cellular, physiological or pathophysiological processes, including clamodulin, which forms a complex with calcium regulating the function of several other proteins, such as the cytoskeletal proteins, PMCP itself, endothelial NO synthase (eNOS) and protein kinase C (PKC) (11) that will participate in the mechanisms of eryptosis (12).
The externalization of phosphatidylserine is also an event dependent on calcium concentrations. The proteins responsible for the symmetry of phosphatidylserine in the cellular membrane are calcium-dependent proteins. Flipases are examples of transmembrane proteins responsible for translocating phospholipids, maintaining plasma membrane asymmetry. They translocate phosphatidylserine from the outer to the inner side of the membrane. The scramblases are a distinct form of flipases, but also translocate phospholipids between the two monolayers of a lipid bilayer of a cell membrane. Scramblases translocate phosphatidylserine in the opposite direction to the translocation made by flipases, from the inside out. Therefore, exposure of phosphatidylserine in the outer layer of the plasma membrane is a consequence of high levels of calcium that inhibit flipases activity and activate scramblases (9).
Another important events during eryptosis is cell shrinkage and blebbing, which is also controlled by calcium influx. Increased cytosolic calcium activity further activates the cysteine endopeptidase calpain, an enzyme degrading the cytoskeleton and thus leading to cell membrane blebbing (13). The high calcium concentration induces the formation of the calcium/calmodulin complex that is also associated with potassium channels called Gardos channels. This channel induces the hyperpolarization of the membrane which in turn promotes the efflux of chlorine, potassium and water, resulting in cell shrinkage (9).
Ceramide is a sphingolipid that stimulates eryptosis. It is generated after osmotic shock by breaking sphingomyelin and works as a sensitizer to the effect of calcium influx. Ceramide is located in lipid rafts and destabilizes the interaction of cytoskeleton proteins with the plasma membrane, increasing membrane instability, and facilitating the erythrocyte cell membrane scrambling and exposure of phosphatidylserine (13).
Caspase activation is another event that occurs during eryptosis, however unlike what occurs in nucleated cells, in erythrocytes, caspase activation is not a central event. Calcium influx and calcium-induced phosphatidylserine exposure do not appear to be dependent on caspase activation (6). Although there are studies showing caspase 3 activation associated with phosphatidylserine exposure in erythrocytes under oxidative stress (14), they do not play a central role.
Eryptosis and Physiology
Eryptosis is an event that occurs physiologically for the control and elimination of senescent red blood cells. The senescence process occurs through the accumulation of cellular damage caused by oxidative stress and the impossibility of recovering the already existing molecules. This process promotes morphological alterations in cell volume, density, and shape, as well as quantitative and qualitative changes on cell plasma membrane, making the cells targeted by the phagocytic cells in the liver and spleen, and thus being removed from the bloodstream.
Alterations reach critical levels after approximately 120 days after red blood cell entry into the circulation (15). The mechanical filtration of red blood also occurs in the spleen, due to its three-dimensional structure. In the red pulp there is a structure, smaller than capillaries, called interendothelial slit, which do not allow the passage of red blood cells whose volume, shape and density ratio are abnormal. In this way, red blood cells whose geometric patterns are modified, such as during eryptosis, are retained in the organ (16).
Red blood cells also present surface molecules that signal their state of senescence, indicating the right moment when they should be eliminated. Oxidative stress, for example, promotes the formation of band 3 protein aggregates (one of the most abundant transmembrane proteins in red blood cells), which, when stabilized by oxidized hemoglobin molecules (hemicromes), are recognized as antigens by autologous IgG antibodies and complement system. With the deposition of a critical density of antibodies and complement molecules, senescent red blood cells are recognized and eliminated by macrophages. This process also destabilizes the interaction of the cytoskeleton with the band 3 proteins, promoting the formation of vesicles, less fluidity and phospholipid scrambling of the plasma membrane (15).
Another way of eliminating senescent red blood cells is the exposure of phosphatidylserine in the outer portion of its plasma membrane, a sign that indicates that the cell must be phagocytosed, because in healthy cells this phospholipid is actively maintained in the cytoplasmic portion of the plasma membrane. Concomitantly, there is negative regulation of the CD47 molecule, a transmembrane protein whose normal expression indicates a sign of “do not eat me”. Thus, the reduction of CD47 expression and the increase of phosphatidylserine in the outer portion of the plasma membrane stimulates phagocytosis and the elimination of these red cells (17).
When erythrocytes suffer some stress or damage, before their natural senescence period, eryptosis is a possible mechanism for eliminating these damaged cells, instead of hemolysis. The lysis of red blood cells promotes the accumulation of hemoglobin and heme in the blood, which consequently react rapidly with nitric oxide (NO) reducing their availability. This reduction in NO levels can cause vasoconstriction, increased expression of adhesion molecules and endothelial activation. These adhesion molecules act as pro-inflammatory ligands of innate immune receptors, promoting platelet aggregation, and then recruiting cells of the innate response, which will initiate an inflammatory process with the release of pro-inflammatory cytokines and chemokines (18). In addition, the accumulation of hemoglobin to be filtered in the renal glomeruli can cause occlusion of them (19). Therefore, the elimination of red blood cells by eryptosis can prevent even greater damage to the organism.
Eryptosis in Malaria
Eryptosis has also been associated with several pathologies, including metabolic syndromes, uremic syndromes, polycythemia vera (20) and anemias such as sickle cell anemia and thalassemia. Infectious diseases also induce eryptosis, including Plasmodium infection (21). In humans, infection by the protozoan of the genus Plasmodium causes malaria. Its transmission occurs through an arthropod vector, the mosquito of the genus Anopheles. The species that parasitize humans are: Plasmodium falciparum, P. vivax, P. malariae, P. ovale and P. knowlesi. However, the species responsible for most cases are P. falciparum and P. vivax. There are also four species of rodent parasitic plasmodia, which are great tools for the studies of malaria in animal models, such as testing new drugs, studies on drug resistance mechanisms, vaccine development, among others; they are: P. yoelii, P. berghei, P. chabaudi and P. vinckei (22).
The Plasmodium life cycle corresponds to a sexual phase in the invertebrate host, and an asexual phase in the vertebrate human host. The life cycle of Plasmodium in the human host begins when the female mosquito transfers the form of the parasite called sporozoite directly to the skin. After reaching the blood vessels, the sporozoites travel to the liver where they will infect the hepatocytes. In these cells, sporozoites multiply by schizogony, differentiating into schizonts full of merozoites, the infective forms of red blood cells, which will enter the bloodstream, initiating the blood stage of infection, in which signs and symptoms of the disease occur (23).
In the blood phase, the parasites multiply intraerythrocytically by schizogony, passing through three successive and morphologically distinct blood stages: the young (ring-shaped) trophozoite, the mature trophozoite and, finally, the schizont. Some blood merozoites, however, differentiate into gametocytes responsible for the continuity of the cycle in the Anopheles mosquito, after the blood meal in the vertebrate host (24). Clinical symptoms of malaria are associated with the blood cycle of the disease, characterized by infection and lysis of red blood cells. The disease manifests itself in an asymptomatic to mild form, with symptoms such as fever, headache, nausea, drowsiness, among others. As the infection progresses, without timely diagnosis and adequate treatment, malaria can progress to its severe forms, which mainly include severe anemia, cerebral malaria and metabolic acidosis (25).
In malaria, eryptotic process has been better studied in infection by P. falciparum, which induces oxidative stress in the host red blood cell, promoting the opening of calcium channels and inducing eryptosis. During infection, the parasite, indeed, digests hemoglobin to obtain nutrients and, consequently, hydrogen peroxide is generated, and OH radicals induce oxidative stress (26–28). Therefore, eryptosis in P. falciparum-infected red blood cells occurs as a consequence of normal parasite development, with progressive induction of PS exteriorization mediated by increase of cytoplasmic calcium influx in these host cells. It is hypothesized however that the parasite sequesters calcium in its own cytosol, preventing the deflagration of eryptosis during the earlier stages of the intraerythrocytic and allowing the maturation of parasite forms. Parallelly, P. falciparum also increases the activity of sphingomyelinase, catalyzing the synthesis of ceramide from sphingomyelin. In turn, increased production of ceramide, even with low concentrations of calcium, can be responsible for eryptotic events, including externalization of phosphatidylserine (17) (Figure 1).
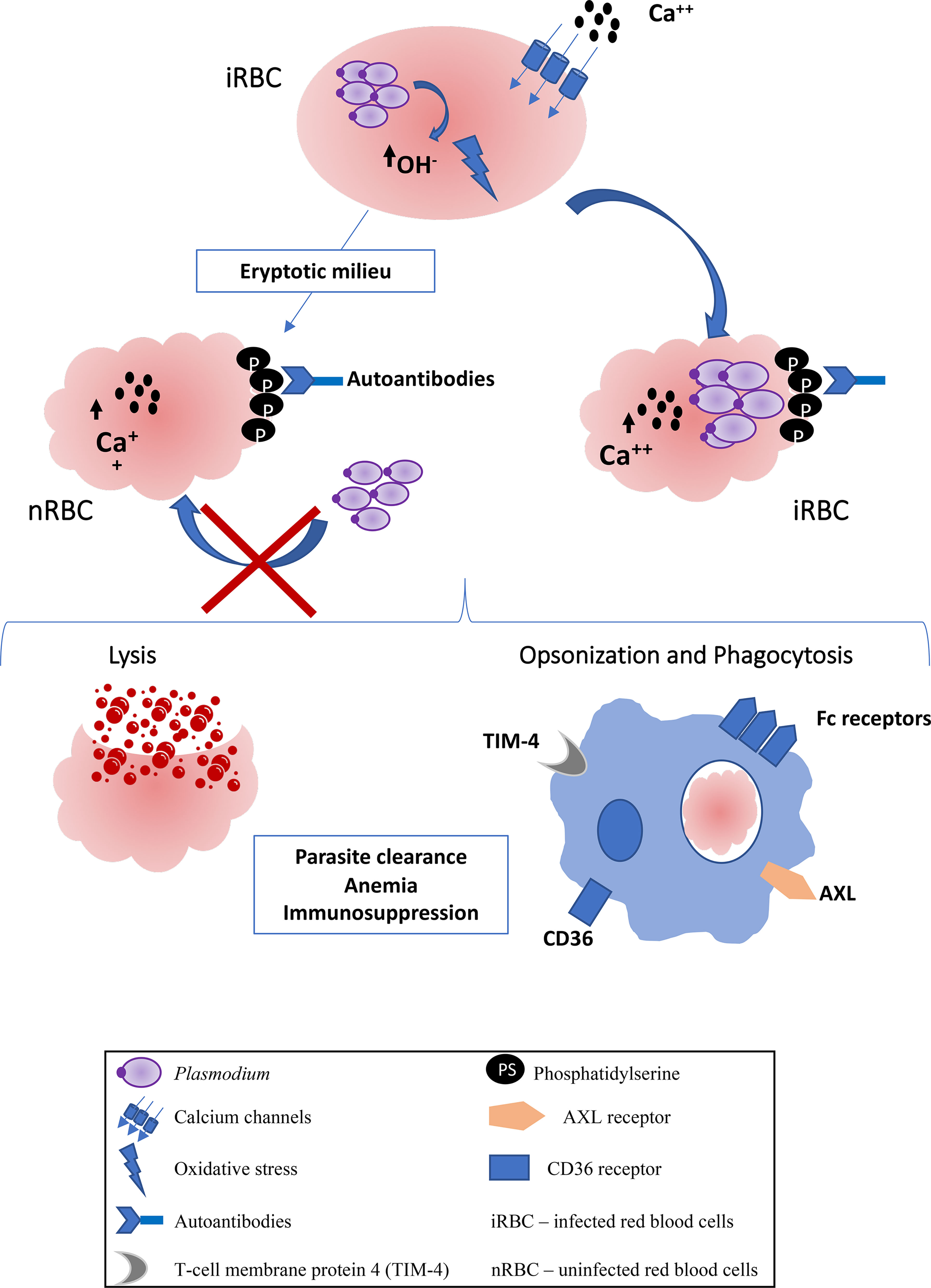
Figure 1 Scheme of eryptosis in Plasmodium infection. The metabolism of the parasite induces the production of OH-radicals that promote oxidative stress in red blood cells. There is the opening of calcium channels, an increase in intracellular calcium and, consequently, the exposure of phosphatidylserine. Uninfected red blood cells also suffer eryptosis, probably from eryptotic factors released by infected red blood cells, becoming refractory to infection. Both uninfected and infected RBCs are eliminated by lysis mediated by activation of the complement system, or by prophagocytosis mediated by Fc receptors on phagocytes, due to the presence of autoantibodies against phosphatidylserine. There are also specific receptors against phosphatidylserine that can promote uptake by phagocytes, such as CD36, TIM-4 and AXL. The consequences of eryptosis can be, parasite clearence, anemia and immunosuppression.
Some host pro-apoptotic factors appear to be associated with eryptosis induction during Plasmodium infection, such as hematin (29), granzyme B (30), soluble FAS ligand (sFAS-L) (31), anti-erythrocyte (32) and anti-phosphatidylserine antibodies (33–37), as well as some anti-apoptotic factors that are reduced during infection, such as nitric oxide (NO), erythropoietin and vitamin E, and thus can contribute to eryptosis (38–40).
Pathological and/or Beneficial Roles of Eryptosis in Malaria
Eryptosis also appears to be induced in uninfected red blood cells (Figure 1), in infections by P. yoelii, P. berghei and by P. falciparum (41–43). Infected and uninfected red blood cells are likely to suffer eryptosis, the latter case being responsible for a worsening of the anemia caused by the infection. On the other hand, eryptotic red blood cells become refractory to infection, which is a possible mechanism of host defense, thus controlling parasitemia (41, 44). It is believed that eryptosis has an important role in the pathogenesis of malaria, especially in severe anemia, however, this role is still controversial. Severe anemia during malaria can be attributed to the lysis of red blood cells during the blood plasma cycle, or the elimination of uninfected blood cells (45, 46).
In addition, during infection there is an inefficient production of red blood cells by the bone marrow (47, 48), demonstrated by the increasing numbers of reticulocytes in the periphery, consequence of the great loss of red blood cells (49). It is believed that the uptake of red blood cells by macrophages, in addition to directly contributing to their elimination, may contribute to the inefficient production of red blood cells, as it would limit the bioavailability of iron for the erythropoiesis process (50). It has been argued that phosphatidylserine exposure in infected red blood cells may play a role in cytoadherence during infection (51), contributing to the adhesion and sequestration of these cells in the vascular endothelium, as well as for cell aggregation and the formation of a structure containing infected and uninfected red blood cells, called rosette (52–54).
Consequently, the increased exposure of phosphatidylserine in infected red blood cells has harmful effects for patients with malaria: the sequestered and adherent erythrocytes prevent the immune elimination of Plasmodium in the spleen; adherence to the endothelium and other cells induces thrombus occlusion, which leads to severe disease; and an a anemia, that is also a main consequence of eryptosis. All events are well characterized in the case of infections by P. falciparum and have until now been attributed mainly to exposure on the cell surface of PfEMP1 (P. falciparum erythrocyte membrane protein 1). Interestingly, in the case of P. vivax, which does not express PfEMP1, there is some evidence that red blood cells infected with P. vivax are able to adhere to the endothelial walls, suggesting the participation of phosphatidylserine (55). However, eryptosis in P. vivax-infected red blood cells has not yet been studied and it has been demonstrated that, differently from plasma of patients infected with P. falciparum, those of P. vivax patients are not able to induce eryptosis in uninfected red blood cells, as determined by PS staining (29).
Alternatively, the preference of P. vivax to infect reticulocytes, whose PS exposition is observed at the later stages of maturation into red blood cells, could be involved in the cytoadherent phenomena occurring with P. vivax-infected red blood cells, contributing to thrombotic events related to malaria, as suggested in sickle cell disease (56). The presence of autoantibodies is frequently reported in patients with malaria, including autoantibodies against phosphatidylserine (33–35, 37). The binding of these antibodies to phosphatidylserine in the outer membrane of eryptotic red blood cells, whether infected or not (34), can promote their lysis due to complement system activation, as well as promote phagocytosis mediated by Fc receptors, present in macrophages (35). The elimination of infected red blood cells contributes to the reduction of parasitemia. However, at the same time that eryptosis eliminates infected cells, it ends up inducing anemia, because through the same mechanisms it promotes the elimination of uninfected red blood cells (35).
Another hypotheses for the contribution of uninfected eryptotic red blood cells is the interaction between phosphatidylserine and the CD36 receptor present in phagocytic cells such as DCs and macrophages, which would recognize eryptotic red blood cells, infected or not, and eliminate them (57). Probably the same can occur with other phosphatidylserine receptors present in phagocytic cells, whose participation in the elimination of apoptotic cells is known, such as TIM-4 (58), MERKT e AXL (59). These data suggest that eryptosis contributes to the severity of anemia during the course of the disease. But eryptosis can be a host mechanism to fight malaria (42). In addition to eliminating infected red blood cells, and consequently interrupting the Plasmodium life cycle, eryptosis makes uninfected red blood cells refractory to infection by P. falciparum, which preferentially invades non-eryptotic red blood cells, suggesting that the increased eryptosis correlates with a lower parasitic load (42) (Figure 1).
Several studies show that the immune response during malaria is deficient (60–63). Some of these works indicate that Plasmodium induces a deficient immune response, possibly as a mechanism for the parasite to sustain its life cycle in the human host, allowing the transmission of sexual forms to the mosquito and continuing its life cycle (64). Apoptosis has been described in several human pathologies, infectious or not, related to immunoregulation. Phagocytosis of apoptotic cells would induce a suppressive profile in macrophages and dendritic cells (DCs) (2, 65).
There are no studies showing the direct relationship between eryptosis and immunomodulation during malaria, but Urban and collaborators, in 1999, described that the infected red blood cells that adhere to CD36 on DCs inhibit their maturation. At this time, it was believed that the PfEMP1 protein was responsible for the interaction between the infected red blood cell and DCs, and that in this way the red blood cells would be imitating the immunosuppressive effects induced by apoptotic cells (66). Today we know that eryptotic red blood cells, infected or not, through phosphatidylserine, can interact with CD36 on DCs and macrophages contributing to their elimination, possibly also contributing to the development of a suppressor phenotype in DCs (57) (Figure 1).
Eryptosis in Other Pathologies
Lang and colleagues observed that the red blood cells of patients suffering from thalassemia, sickle cell anemia and glucose-6-phosphate dehydrogenase deficiency are more sensitive to glucose depletion and oxidative stress, and that the red blood cells of patients with sickle cell anemia and glucose-6-phosphate dehydrogenase deficiency are also more sensitive to osmotic shock. Thus, they observed that the red cells of these patients are more likely to undergo eryptotic process (67). The elimination of these infected eryptotic red blood cells occurs by opsonization via activation of the complement system, and is one of the mechanisms responsible for the protection observed in these patients against Plasmodium infection (68).
In addition to infections by protozoa of the genus Plasmodium, eryptosis has already been described in other infections, such as infection by Mycoplasma suis (69) as a representative for hemotrophic mycoplasmas to study anemia pathogenesis caused by pathogens, and during sepsis (70). The same was observed in vivo during infection with the Trypanosoma brucei (71), and in viral infections such as with LCMV (Lymphocytic Choriomeningitis virus) (72). The production of autoantibodies against phosphatidylserine promotes the elimination of uninfected red blood cells, causing anemia.
In sepsis, the release of molecules derived from pathogens such as peptidoglycans, lipopeptides, α-haemolysin, pyocyanin and listeriolysin are capable of inducing eryptosis, also causing anemia (70). Pyocyanin, a virulence factor of Pseudomonas aeruginosa, induces eryptosis through the accumulation of reactive oxygen species in red blood cells (73). The enhanced eryptosis may contribute to an aggravated microcirculatory derangement associated with sepsis (74). An increase in the number of eryptotic red blood cells is also observed in diabetes. This is believed to be due to a by-product of glycolysis, the dicarbonyl compound methylglyoxal (75). Erythrocytes from diabetic patients show increased superoxide dismutase activity and ROS production, factors that favor phospholipid scrambling of the erythrocyte cell membrane (76).
Concluding Remarks
Eryptosis is an apoptosis-like process that occurs physiologically in senescent red blood cells (15). Infectious processes such as Plasmodium infection are capable of inducing eryptosis (21). Plasmodium promotes oxidative stress that induces the opening of calcium channels, increasing their concentration in the cytoplasm of red blood cells. Consequently, the increase in calcium induces the exposure of phosphatidylserine in the plasma membrane of red blood cells (26–28).
Thus, eryptosis and particularly phosphatidylserine exposure has two main consequences on the severity of malaria. First, we can discuss eryptosis as a protective mechanism of the host against infection. This is because during Plasmodium infection, not only infected red blood cells undergo eryptosis, but also uninfected red blood cells, which become refractory to infection (41–43). We could say that this would be a mechanism of the host to contain the infection. In addition, the presence of antibodies against phosphatidylserine promotes the elimination of infected red blood cells by phagocytes through Fc receptors, which interrupts the parasite cycle, reducing the parasite load (42), similarly to occurring in other infections such as African trypanosomes (71). On the other hand, this same mechanism also promotes cytoadherent phenomena of parasitized red blood cells, as well as the elimination of non-parasitized red blood cells, contributing to the vascular damage and anemia reported in malaria, respectively (35). Moreover, eryptosis may be associated with immunomodulation of the immune response to the parasite, inhibiting the maturation of DCs and promoting a suppressive phenotype in them, that is, inducing an insufficient response to combat the disease (57, 66).
Author Contributions
AMS and AM conceived the manuscript. AMS, PT and AM wrote the manuscript. AMS, PT and AM participated in the preparation of the manuscript.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
This work was funded by grants from Fiocruz/INOVA COVID-19 (VPPCB-005-FIO-20), Conselho Nacional de Desenvolvimento Científico e Tecnológico do Brasil (CNPq), Fundação de Amparo à Pesquisa do Estado do Rio de Janeiro (FAPERJ), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), and it was developed in the frameworks of the Rio de Janeiro Research Network on Neuroinflammation.
References
1. Jacobson MD, Weil M, Raff MC. Programmed Cell Death in Animal Development. Cell (1997) 88(3):347–54. doi: 10.1016/S0092-8674(00)81873-5
2. Fadeel B, Orrenius S. Apoptosis: A Basic Biological Phenomenon With Wide-Ranging Implications in Human Disease. J Intern Med (2005) 258(6):479–517. doi: 10.1111/j.1365-2796.2005.01570.x
3. Bratosin D, Estaquier J, Petit F, Arnoult D, Quatannens B, Tissier J-P, et al. Programmed Cell Death in Mature Erythrocytes: A Model for Investigating Death Effector Pathways Operating in the Absence of Mitochondria. Cell Death Differ (2001) 8(12):1143–56. doi: 10.1038/sj.cdd.4400946
4. Lang E, Pozdeev VI, Xu HC, Shinde PV, Behnke K, Hamdam JM, et al. Storage of Erythrocytes Induces Suicidal Erythrocyte Death. Cell Physiol Biochem (2016) 39(2):668–76. doi: 10.1159/000445657
5. Lang KS, Duranton C, Poehlmann H, Myssina S, Bauer C, Lang F, et al. Cation Channels Trigger Apoptotic Death of Erythrocytes. Cell Death Differ (2003) 10(2):249–56. doi: 10.1038/sj.cdd.4401144
6. Lang E, Lang F. Triggers, Inhibitors, Mechanisms, and Significance of Eryptosis: The Suicidal Erythrocyte Death. BioMed Res Int (2015) 2015:513518. doi: 10.1155/2015/513518
7. Lang PA, Kempe DS, Myssina S, Tanneur V, Birka C, Laufer S, et al. PGE(2) in the Regulation of Programmed Erythrocyte Death. Cell Death Differ (2005) 12(5):415–28. doi: 10.1038/sj.cdd.4401561
8. Gatidis S, Zelenak C, Fajol A, Lang E, Jilani K, Michael D, et al. P38 MAPK Activation and Function Following Osmotic Shock of Erythrocytes. Cell Physiol Biochem (2011) 28(6):1279–86. doi: 10.1159/000335859
9. Föller M, Lang F. Ion Transport in Eryptosis, the Suicidal Death of Erythrocytes. Front Cell Dev Biol (2020) 8:597. doi: 10.3389/fcell.2020.00597
10. Foller M, Kasinathan RS, Koka S, Lang C, Shumilina E, Birnbaumer L, et al. TRPC6 Contributes to the Ca(2+) Leak of Human Erythrocytes. Cell Physiol Biochem (2008) 21(1-3):183–92. doi: 10.1159/000113760
11. Bogdanova A, Makhro A, Wang J, Lipp P, Kaestner L. Calcium in Red Blood Cells-a Perilous Balance. Int J Mol Sci (2013) 14(5):9848–72. doi: 10.3390/ijms14059848
12. De Jong K, Rettig MP, Low PS, Kuypers FA. Protein Kinase C Activation Induces Phosphatidylserine Exposure on Red Blood Cells. Biochemistry (2002) 41(41):12562–7. doi: 10.1021/bi025882o
13. Lang E, Bissinger R, Gulbins E, Lang F. Ceramide in the Regulation of Eryptosis, the Suicidal Erythrocyte Death. Apoptosis (2015) 20(5):758–67. doi: 10.1007/s10495-015-1094-4
14. Mandal D, Moitra PK, Saha S, Basu J. Caspase 3 Regulates Phosphatidylserine Externalization and Phagocytosis of Oxidatively Stressed Erythrocytes. FEBS Lett (2002) 513(2-3):184–8. doi: 10.1016/S0014-5793(02)02294-9
15. Arese P, Turrini F, Schwarzer E. Band 3/Complement-Mediated Recognition and Removal of Normally Senescent and Pathological Human Erythrocytes. Cell Physiol Biochem (2005) 16(4-6):133–46. doi: 10.1159/000089839
16. Pivkin IV, Peng Z, Karniadakis GE, Buffet PA, Dao M, Suresh S. Biomechanics of Red Blood Cells in Human Spleen and Consequences for Physiology and Disease. Proc Natl Acad Sci USA (2016) 2016(28):7804–9. doi: 10.1073/pnas.1606751113
17. Boulet C, Doerig CD, Carvalho TG. Manipulating Eryptosis of Human Red Blood Cells: A Novel Antimalarial Strategy? Front Cell Infect Microbiol (2018) 8:419. doi: 10.3389/fcimb.2018.00419
18. Rapido F. The Potential Adverse Effects of Haemolysis. Blood Transfus (2017) 15(3):218–21. doi: 10.2450/2017.0311-16
19. Lang F, Lang E, Föller M. Physiology and Pathophysiology of Eryptosis. Transfus Med Hemother (2012) 39(5):308–14. doi: 10.1159/000342534
20. Fujita H, Sakuma R, Tomiyama J, Hamaki T, Ohwada A, Kurosawa S, et al. Increased Phosphatidylserine Exposure on the Erythrocyte Membrane in Patients With Polycythaemia Vera. Br J Haematol (2011) 152(2):238–40. doi: 10.1111/j.1365-2141.2010.08379.x
21. Lang F, Qadri SM. Mechanisms and Significance of Eryptosis, the Suicidal Death of Erythrocytes. Blood Purif (2012) 33(1-3):125–30. doi: 10.1159/000334163
22. Rich SM, Ayala FJ. Progress in Malaria Research: The Case for Phylogenetics. Adv Parasitol (2003) 54:255–80. doi: 10.1016/S0065-308X(03)54005-2
23. Cowman AF, Tonkin CJ, Tham W-H, Duraisingh MT. The Molecular Basis of Erythrocyte Invasion by Malaria Parasites. Cell Host Microbe (2017) 22(2):232–45. doi: 10.1016/j.chom.2017.07.003
24. Mueller I, Galinski MR, Baird JK, Carlton JM, Kochar DK, Alonso PL, et al. Key Gaps in the Knowledge of Plasmodium Vivax, a Neglected Human Malaria Parasite. Lancet Infect Dis (2009) 9(9):555–66. doi: 10.1016/S1473-3099(09)70177-X
25. Cowman AF, Healer J, Marapana D, Marsh K. Malaria: Biology and Disease. Cell (2016) 167(3):610–24. doi: 10.1016/j.cell.2016.07.055
26. Zarchin S, Krugliak M, Ginsburg H. Digestion of the Host Erythrocyte by Malaria Parasites is the Primary Target for Quinoline-Containing Antimalarials. Biochem Pharmacol (1986) 35(14):2435–42. doi: 10.1016/0006-2952(86)90473-9
27. Atamna H, Ginsburg H. Origin of Reactive Oxygen Species in Erythrocytes Infected With Plasmodium Falciparum. Mol Biochem Parasitol (1993) 61(2):231–41. doi: 10.1016/0166-6851(93)90069-A
28. Ginsburg H, Atamna H. The Redox Status of Malaria-Infected Erythrocytes: An Overview With an Emphasis on Unresolved Problems. Parasite (1994) 1(1):5–13. doi: 10.1051/parasite/1994011005
29. Dalko E, Das B, Herbert F, Fesel C, Pathak S, Tripathy R, et al. Multifaceted Role of Heme During Severe Plasmodium Falciparum Infections in India. Infect Immun (2015) 83(10):3793–9. doi: 10.1128/IAI.00531-15
30. Hermsen CC, Konijnenberg Y, Mulder L, Loé C, Van Deuren M, Van DerMeer JWM, et al. Circulating Concentrations of Soluble Granzyme A and B Increase During Natural and Experimental Plasmodium Falciparum Infections. Clin Exp Immunol (2003) 132(3):467–72. doi: 10.1046/j.1365-2249.2003.02160.x
31. Issifou S, Mavoungou E, Borrmann S, Bouyou-Akotet MK, Matsiegui P-B, Kremsner PG, et al. Severe Malarial Anemia Associated With Increased Soluble Fas Ligand (Sfasl) Concentrations in Gabonese Children. Eur Cytokine Netw (2003) 14(4):238–41.
32. Mourão LC, de PaulaBaptista R, de Almeida ZB, Grynberg P, Pucci MM, Castro-Gomes T, et al. Anti-Band 3 and Anti-Spectrin Antibodies Are Increased in Plasmodium Vivax Infection and Are Associated With Anemia. Sci Rep (2018) 8(1):8762. doi: 10.1038/s41598-018-27109-6
33. Fernandez-Arias C, Rivera-Correa J, Gallego-Delgado J, Rudlaff R, Fernandez C, Roussel C, et al. Anti-Self Phosphatidylserine Antibodies Recognize Uninfected Erythrocytes Promoting Malarial Anemia. Cell Host Microbe (2016) 19(2):194–203. doi: 10.1016/j.chom.2016.01.009
34. Rivera-Correa J, Conroy AL, Opoka RO, Batte A, Namazzi R, Ouma B, et al. Autoantibody Levels Are Associated With Acute Kidney Injury, Anemia and Post-Discharge Morbidity and Mortality in Ugandan Children With Severe Malaria. Sci Rep (2019) 9(1):14940. doi: 10.1038/s41598-019-51426-z
35. Mourão LC, Cardoso-Oliveira GP, Braga É. Autoantibodies and Malaria: Where We Stand? Insights Into Pathogenesis and Protection. Front Cell Infect Microbiol (2020) 10:262. doi: 10.3389/fcimb.2020.00262
36. Rivera-Correa J, Mackroth MS, Jacobs T, Wiesch JSZ, Rolling T, Rodriguez A. Atypical Memory B-Cells Are Associated With. Elife (2019) 8:1–21. doi: 10.7554/eLife.48309
37. Rivera-Correa J, Yasnot-Acosta MF, Tovar NC, Velasco-Pareja MCEaston ARodriguez A. Atypical Memory B-Cells and Autoantibodies Correlate With Anemia During Plasmodium Vivax Complicated Infections. PloS Negl Trop Dis (2020) 14(7):e0008466. doi: 10.1371/journal.pntd.0008466
38. Burgmann H, Looareesuwan S, Kapiotis C, Viravan S, Vanijanonta U, Hollenstein E, et al. Serum Levels of Erythropoietin in Acute Plasmodium Falciparum Malaria. Am J Trop Med Hyg (1996) 54(3):280–3. doi: 10.4269/ajtmh.1996.54.280
39. Kulkarni AG, Suryakar AN, Sardeshmukh AS, Rathi DB. Studies on Biochemical Changes With Special Reference to Oxidant and Antioxidants in Malaria Patients. Indian J Clin Biochem (2003) 18(2):136–49. doi: 10.1007/BF02867380
40. Sobolewski P, Gramaglia I, Frangos J, Intaglietta M, van derHeyde HC. Nitric Oxide Bioavailability in Malaria. Trends Parasitol (2005) 21(9):415–22. doi: 10.1016/j.pt.2005.07.002
41. Totino PR, et al. Apoptosis of non-Parasitised Red Blood Cells in Plasmodium Yoelii Malaria. Mem Inst Oswaldo Cruz (2013) 108(6):686–90. doi: 10.1590/0074-0276108062013003
42. Apoptosis of non-Parasitized Red Blood Cells in Malaria: A Putative Mechanism Involved in the Pathogenesis of Anaemia. Malar J (2010) 9:350. doi: 10.1186/1475-2875-9-350
43. Totino PRR, dos SantosdeSouza HA, Correa EHC, Daniel-Ribeiro CT, de Fátima Ferreira-da-Cruz M. Eryptosis of non-Parasitized Erythrocytes Is Related to Anemia in Plasmodium Berghei Low Parasitema Malaria of Wistar Rats. Parasitol Res (2019) 118(1):377–82. doi: 10.1007/s00436-018-6167-1
44. Totino PR, Daniel-Ribeiro CT, Ferreira-Da-Cruz MEF. Refractoriness of Eryptotic Red Blood Cells to Plasmodium Falciparum Infection: A Putative Host Defense Mechanism Limiting Parasitaemia. PloS One (2011) 6(10):e26575. doi: 10.1371/journal.pone.0026575
45. Evans KJ, Hansen DS, van Rooijen N, Schofield LABL. Severe Malarial Anemia of Low Parasite Burden in Rodent Models Results From Accelerated Clearance of Uninfected Erythrocytes. Blood (2006) 107(3):1192–9. doi: 10.1182/blood-2005-08-3460
46. Jakeman GN, Saul A, Hogarth WL, Collins WE. Anaemia of Acute Malaria Infections in Non-Immune Patients Primarily Results From Destruction of Uninfected Erythrocytes. Parasitology (1999) 119(Pt 2):127–33. doi: 10.1017/S0031182099004564
47. Casals-Pascual C, Kai O, Cheung JOP, Williams S, Lowe B, Nyanoti M, et al. Suppression of Erythropoiesis in Malarial Anemia Is Associated With Hemozoin In Vitro and In Vivo. Blood (2006) 108(8):2569–77. doi: 10.1182/blood-2006-05-018697
48. Thawani N, Tam M, Bellemare M-J, Bohle DS, Olivier M, de Souza JB, et al. Plasmodium Products Contribute to Severe Malarial Anemia by Inhibiting Erythropoietin-Induced Proliferation of Erythroid Precursors. J Infect Dis (2014) 209(1):140–9. doi: 10.1093/infdis/jit417
49. Chang KH, Tam M, Stevenson MM. Inappropriately Low Reticulocytosis in Severe Malarial Anemia Correlates With Suppression in the Development of Late Erythroid Precursors. Blood (2004) 103(10):3727–35. doi: 10.1182/blood-2003-08-2887
50. Spottiswoode N, Duffy PE, Drakesmith H. Iron, Anemia and Hepcidin in Malaria. Front Pharmacol (2014) 5:125. doi: 10.3389/fphar.2014.00125
51. Eda S, Sherman IW. Cytoadherence of Malaria-Infected Red Blood Cells Involves Exposure of Phosphatidylserine. Cell Physiol Biochem (2002) 12(5-6):373–84. doi: 10.1159/000067908
52. Totino PR, Lopes SC. Insights Into the Cytoadherence Phenomenon of. Front Immunol (2017) 8:1148. doi: 10.3389/fimmu.2017.01148
53. Ho M, Davis TM, K Silamut, Bunnag D, White NJ. Rosette Formation of Plasmodium Falciparum-Infected Erythrocytes From Patients With Acute Malaria. Infect Immun (1991) 59(6):2135–9. doi: 10.1128/iai.59.6.2135-2139.1991
54. David PH, Handunnetti SM, Leech JH, Gamage P, Mendis KN. Rosetting: A New Cytoadherence Property of Malaria-Infected Erythrocytes. Am J Trop Med Hyg (1988) 38(2):289–97. doi: 10.4269/ajtmh.1988.38.289
55. Alves FA, Pelajo-Machado M, Totino PRR, Souza MT, Gonçalves EC, Schneider MPC. Splenic Architecture Disruption and Parasite-Induced Splenocyte Activation and Anergy in Plasmodium Falciparum-Infected Saimiri Sciureus Monkeys. Malar J (2015) 14:128. doi: 10.1186/s12936-015-0641-3
56. Mankelow TJ, Griffiths RE, Trompeter S, Flatt JF, Cogan NM, Massey EJ, et al. Autophagic Vesicles on Mature Human Reticulocytes Explain Phosphatidylserine-Positive Red Cells in Sickle Cell Disease. Blood (2015) 126(15):1831–4. doi: 10.1182/blood-2015-04-637702
57. Totino PR, Daniel-Ribeiro CT, Ferreira-Da-Cruz MF. Evidencing the Role of Erythrocytic Apoptosis in Malarial Anemia. Front Cell Infect Microbiol (2016) 6(176). doi: 10.3389/fcimb.2016.00176
58. Miyanishi M, Tada K, Koike M, Uchiyama Y, Kitamura T, Nagata S. Identification of Tim4 as a Phosphatidylserine Receptor. Nature (2007) 450(7168):435–9. doi: 10.1038/nature06307
59. Lemke G. How Macrophages Deal With Death. Nat Rev Immunol (2019) 19(9):539–49. doi: 10.1038/s41577-019-0167-y
60. Amorim KN, Chagas DCG, Sulczewski FB, Boscardin SB. Dendritic Cells and Their Multiple Roles During Malaria Infection. J Immunol Res (2016) 2016: 2926436. doi: 10.1155/2016/2926436
61. Ocaña-Morgner C, Wong KA, Lega F, Dotor J, Borras-Cuesta F, Rodriguez A. Role of TGF-Beta and PGE2 in T Cell Responses During Plasmodium Yoelii Infection. Eur J Immunol (2007) 37(6):1562–74. doi: 10.1002/eji.200737068
62. Urban BC, Ferguson DJP, Pain A, Willcox N, Plebanski M, Austyn JM, et al. Plasmodium Falciparum-Infected Erythrocytes Modulate the Maturation of Dendritic Cells. Nature (1999) 400(6739):73–7. doi: 10.1038/21900
63. Wykes MN, Horne-Debets JM, Leow C-Y, Karunarathne DS. Malaria Drives T Cells to Exhaustion. Front Microbiol (2014) 5:249. doi: 10.3389/fmicb.2014.00249
64. Cockburn IA, Zavala F. Dendritic Cell Function and Antigen Presentation in Malaria. Curr Opin Immunol (2016) 40:1–6. doi: 10.1016/j.coi.2016.01.010
65. Dockrell DH. Apoptotic Cell Death in the Pathogenesis of Infectious Diseases. J Infect (2001) 42(4):227–34. doi: 10.1053/jinf.2001.0836
66. Urban BC, Willcox N, Roberts DJ. A Role for CD36 in the Regulation of Dendritic Cell Function. Proc Natl Acad Sci USA (2001) 98(15) 8750–5. doi: 10.1073/pnas.151028698
67. Lang KS, Roll B, Myssina S, Schittenhelm M, Scheel-Walter H-G, Kanz L, et al. Enhanced Erythrocyte Apoptosis in Sickle Cell Anemia, Thalassemia and Glucose-6-Phosphate Dehydrogenase Deficiency. Cell Physiol Biochem (2002) 12(5-6):365–72. doi: 10.1159/000067907
68. Gong L, Parikh S, Rosenthal PJ, Greenhouse B. Biochemical and Immunological Mechanisms by Which Sickle Cell Trait Protects Against Malaria. Malar J (2013) 12(317):1–9. doi: 10.1186/1475-2875-12-317
69. Felder KM, Hoelzle K, Ritzmann M, Kilchling T, Schiele D, Heinritzi K, et al. Hemotrophic Mycoplasmas Induce Programmed Cell Death in Red Blood Cells. Cell Physiol Biochem (2011) 27(5):557–64. doi: 10.1159/000329957
70. Aird WC. The Hematologic System as a Marker of Organ Dysfunction in Sepsis. Mayo Clin Proc (2003) 78(7):869–81. doi: 10.4065/78.7.869
71. Rivera-Correa J, Verdi J, Sherman J, Sternberg JM, Raper J, Rodriguez A. Autoimmunity to Phosphatidylserine and Anemia in African Trypanosome Infections. PloS Negl Trop Dis (2021) 15(9):e0009814. doi: 10.1371/journal.pntd.0009814
72. Vijay R, Guthmiller JJ, Sturtz AJ, Crooks S, Johnson JT, Li L, et al. Hemolysis-Associated Phosphatidylserine Exposure Promotes Polyclonal Plasmablast Differentiation. J Exp Med (2021) 218(6):1–13. doi: 10.1084/jem.20202359
73. Qadri SM, Donkor DA, Bhakta V, Eltringham-Smith LJ, Dwivedi DJ, Jane C, et al. Phosphatidylserine Externalization and Procoagulant Activation of Erythrocytes Induced by Pseudomonas Aeruginosa Virulence Factor Pyocyanin. J Cell Mol Med (2016) 20(4):710–20. doi: 10.1111/jcmm.12778
74. Holthoff JH, Wang Z, Seely KA, Gokden N, Mayeux PR. Resveratrol Improves Renal Microcirculation, Protects the Tubular Epithelium, and Prolongs Survival in a Mouse Model of Sepsis-Induced Acute Kidney Injury. Kidney Int (2012) 81(4):370–8. doi: 10.1038/ki.2011.347
75. Nicolay JP, Schneider J, Niemoeller OM, Artunc F, Portero-Otin M, Haik G Jr, et al. Stimulation of Suicidal Erythrocyte Death by Methylglyoxal. Cell Physiol Biochem (2006) 18(4-5):223–32. doi: 10.1159/000097669
76. Mohammedi K, Bellili-Muñoz N, Marklund SL, Driss F, Nagard HL, Patente TA, et al. Plasma Extracellular Superoxide Dismutase Concentration, Allelic Variations in the SOD3 Gene and Risk of Myocardial Infarction and All-Cause Mortality in People With Type 1 and Type 2 Diabetes. Cardiovasc Diabetol (2015) 14:845. doi: 10.1186/s12933-014-0163-2
Keywords: eryptosis, Plasmodium infection, malaria pathogenesis, immunopathology, severe malarial anemia
Citation: Scovino AM, Totino PRR and Morrot A (2022) Eryptosis as a New Insight in Malaria Pathogenesis. Front. Immunol. 13:855795. doi: 10.3389/fimmu.2022.855795
Received: 16 January 2022; Accepted: 04 April 2022;
Published: 13 May 2022.
Edited by:
Xinzhuan Su, National Institutes of Health (NIH), United StatesReviewed by:
Juan L. Rivera-Correa, NewYork-Presbyterian, United StatesCopyright © 2022 Scovino, Totino and Morrot. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Alexandre Morrot, YWxleGFuZHJlLm1vcnJvdEBpb2MuZmlvY3J1ei5icg==
 Aline Miranda Scovino
Aline Miranda Scovino Paulo Renato Rivas Totino
Paulo Renato Rivas Totino Alexandre Morrot
Alexandre Morrot