
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Immunol., 23 November 2021
Sec. Primary Immunodeficiencies
Volume 12 - 2021 | https://doi.org/10.3389/fimmu.2021.788278
This article is part of the Research TopicInborn Errors of the Immune System Associated with Atopy and EosinophilsView all 5 articles
 Henry Y. Lu1,2
Henry Y. Lu1,2 Robert Sertori3
Robert Sertori3 Alejandra V. Contreras3
Alejandra V. Contreras3 Mark Hamer4
Mark Hamer4 Melina Messing2,4
Melina Messing2,4 Kate L. Del Bel1
Kate L. Del Bel1 Elena Lopez-Rangel1
Elena Lopez-Rangel1 Edmond S. Chan1
Edmond S. Chan1 Wingfield Rehmus1
Wingfield Rehmus1 Joshua D. Milner5
Joshua D. Milner5 Kelly M. McNagny4,6
Kelly M. McNagny4,6 Anna Lehman6
Anna Lehman6 David L. Wiest3†
David L. Wiest3† Stuart E. Turvey1,2*† on behalf of CAUSES Study
Stuart E. Turvey1,2*† on behalf of CAUSES StudyB-cell lymphoma/leukemia 11B (BCL11B) is a C2H2 zinc finger transcription factor that is critically important for regulating the development and function of a variety of systems including the central nervous system, the skin, and the immune system. Germline heterozygous variants are associated with a spectrum of clinical disorders, including severe combined immunodeficiency as well as neurological, craniofacial, and dermal defects. Of these individuals, ~50% present with severe allergic disease. Here, we report the detailed clinical and laboratory workup of one of the most severe BCL11B-dependent atopic cases to date. Leveraging a zebrafish model, we were able to confirm a strong T-cell defect in the patient. Based on these data, we classify germline BCL11B-dependent atopic disease as a novel primary atopic disorder.
Primary atopic disorders (PADs) are a group of monogenic disorders that present with dysregulated allergic effector responses (1). Major clinical features include severe atopic dermatitis, food allergies, allergic asthma, urticaria, eosinophilia, and elevated IgE. Many PADs also have comorbid immunodeficiency and immune dysregulation such as systemic lupus erythematosus and autoimmune vasculitis as is the case in some patients with dominant-negative STAT3 variants (1). Due to the heterogeneous presentation of PADs, it can often be difficult to differentiate monogenic from polygenic etiologies. However, the increased clinical implementation of diagnostic next-generation sequencing has led to increased identification of PAD patients.
B-cell lymphoma/leukemia 11B (BCL11B) is a C2H2 zinc finger (ZF) transcription factor protein that is broadly expressed and is important in regulating the development of various tissues, including the central nervous system, T cells, skin, and teeth (2). It serves as both a transcriptional activator and repressor and binds GC-rich response elements (2). Germline heterozygous variants in BCL11B have been identified in ~17 patients thus far (extracted from publications written in English) and are associated with a range of clinical phenotypes. This includes severe combined immunodeficiency (SCID) with neurological, craniofacial, and dermal abnormalities, coronal suture synostosis, short stature, and intellectual disability, speech delay, dysmorphic facies, dental abnormalities, and T-cell and innate lymphoid cell defects (3–5). Notably, eight of 17 (~47%) of these patients presented with atopic disease, including asthma, eosinophilia, food allergies, and eczema (4). Here, we present the case of a female patient with severe atopic disease, neurodevelopmental abnormalities, and immune dysregulation, who carries a novel germline heterozygous variant in BCL11B. We propose heritable BCL11B-related atopic disease as a novel PAD.
All study participants and/or their parents/guardians provided written informed consent to participate. All individuals also provided consent to be published. Research study protocols were approved by The University of British Columbia Clinical Research Ethics Board.
The potentially pathogenic NM_138576:c.2487G>A;NP_612808:p.Cys826Tyr BCL11B variant was selected because (i) germline BCL11B variants have been associated with both neurodevelopmental disorders as well as atopic disease; (ii) it is absent in population databases (e.g., gnomAD); and (iii) it is predicted to be pathogenic by a variety of in silico pathogenicity prediction tools. Notably, VarCards, which incorporates 23 pathogenicity prediction algorithms, including SIFT, Polyphen-2, MutationTaster, MutationAssessor, FATHMM, and more, predicts the p.Cys826Tyr BCL11B variant to be pathogenic as determined by 21/23 algorithms. Furthermore, this variant has a combined annotation-dependent depletion (CADD) score of 23, which is far above its mutation significance cutoff (MSC) of 3.313.
To investigate the impact of the p.Cys826Tyr BCL11B variant on T-cell development, wild-type (WT) or p.Cys826Tyr human BCL11B was ectopically expressed in one-cell-stage zebrafish embryos for which the conserved zebrafish BCL11B orthologue bcl11ba had been knocked down, as previously described (3). Briefly, bcl11ba morpholino oligonucleotides were injected into one-cell embryos to block translation and splicing of zebrafish bcl11ba along with either WT or p.Cys826Tyr BCL11B in a heat-inducible pSGH2 vector. 30 h post-fertilization, embryos were heated at 37°C for 1 h to induce WT or p.Cys826Tyr BCL11B expression. To visualize the development of T-cell progenitors, 5 days post-fertilization, zebrafish embryos were subjected to whole-mount in situ hybridization (WISH) with a lck probe.
The patient is a 14-year-old Canadian girl born to non-consanguineous parents (Figure 1A) who presented with microcephaly, mild intellectual disability, severe and broad food allergies (Table 1), atopic dermatitis, asthma, elevated IgE (Figure 1B), eosinophilia (Figure 1C), alopecia totalis, brittle nails, prurigo nodularis, and antinuclear antibodies. In particular, the food allergy diagnosis was based on three converging lines of evidence: (1) history consistent with IgE-mediated immediate hypersensitivity reactions (i.e., urticaria, lip swelling, vomiting, diarrhea) following exposure to egg, peanut, hazelnut, sesame, sunflower seed, tuna, and liquid cow’s milk); (2) positive epicutaneous skin prick testing to cow’s milk, salmon, peanuts, treenuts, and seeds, as well as multiple environmental allergens, including tree and grass pollen, cat, dog, and dust mite; and (3) the presence of serum-specific IgE. A brain magnetic resonance imaging (MRI) scan was normal and did not reveal any evidence of corpus callosum agenesis as was reported in a previous case (Figure 1D) (3). Family history was unremarkable; the patient’s five siblings are healthy.
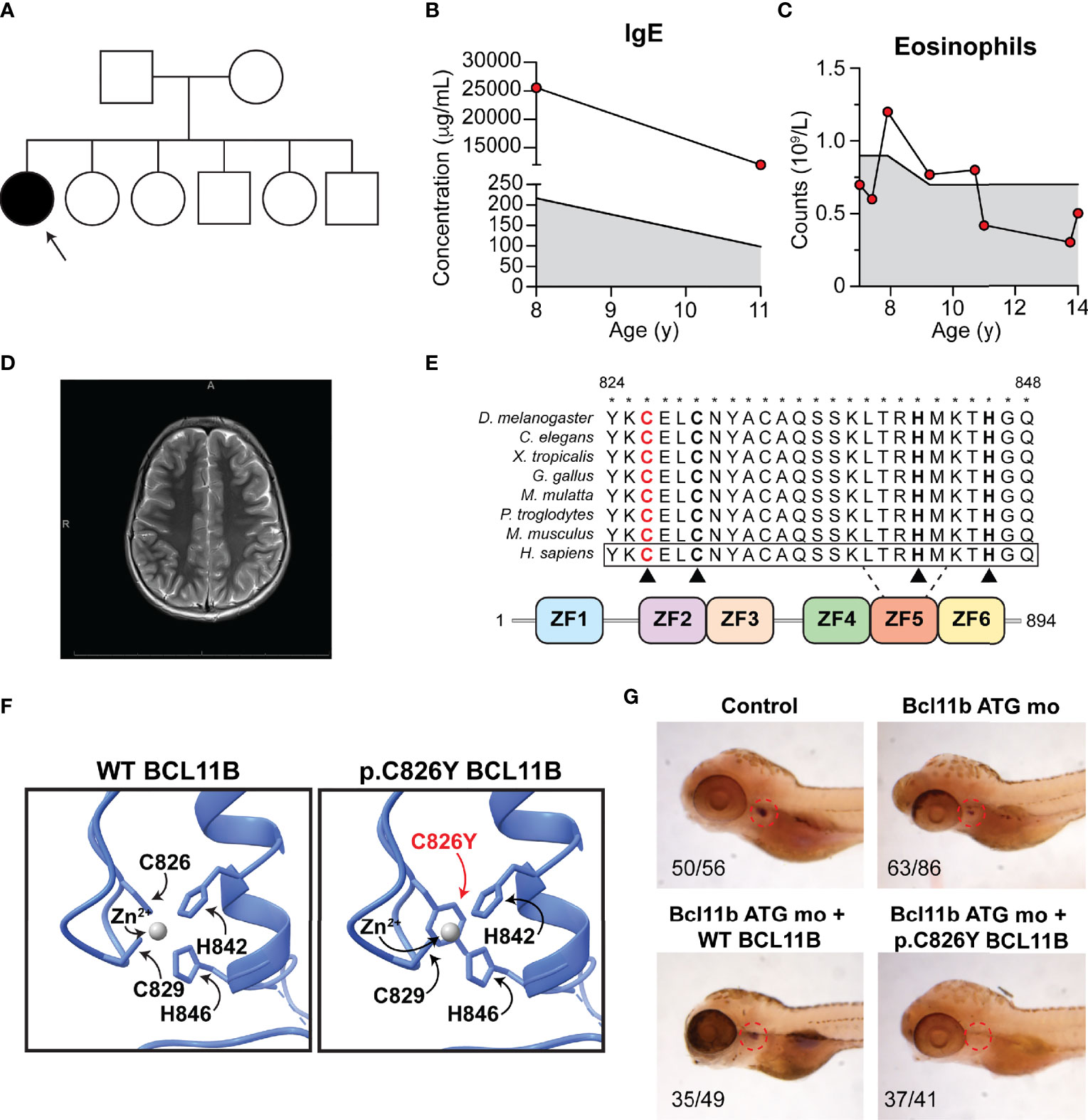
Figure 1 Patient phenotype and functional validation of a novel BCL11B variant. (A) Family pedigree. Arrow, proband. (B) Patient IgE at ages 8 and 11. (C) Patient eosinophil counts from ages 7 to 14. (B, C) Shaded region, reference range. (D) Patient brain magnetic resonance imaging. (E) Schematic of BCL11B protein. Red, substitution location. Asterisks, conservation. Black triangles, zinc coordinating residues. ZF, zinc finger. (F) Homology-based modeling of affected region. (G) Rescue of T-cell development visualized by lck probe post-bcl11ba knockdown with Bcl11b ATG mo and heat-inducible reexpression of WT (bottom left) or p.Cys826Tyr BCL11B (bottom right). Indicated are the number of individuals with observed phenotype.
Given the unique combination of clinical features, the patient underwent trio whole exome sequencing, which revealed a novel de novo missense NM_138576:c.2487G>A;NP_612808:p.Cys826Tyr BCL11B variant (Figure 1E). This variant has never been reported in any population databases and was predicted to be pathogenic by a large range of in silico pathogenicity prediction tools, including 21/23 algorithms used by VarCards (6). Notably, this variant had a CADD score of 23, where the suggested CADD cutoff for BCL11B according to MSC is 3.313 (7). Cys826 in the fifth zinc finger of the protein was highly conserved through evolution (Figure 1E). Homology-based structural modeling using the crystal structure of the related BCL11A transcription factor (8) revealed that the p.Cys826Tyr substitution likely perturbs the coordination of zinc ions, which is important for protein-protein interactions and the formation of protein complexes (2) (Figure 1F).
Since BCL11B is critically important for regulating T-cell development and function (2), we carried out detailed immunophenotyping of the patient. Surprisingly, we discovered that the patient had a modest B-cell developmental defect (elevated naïve B cells and decreased memory B cells) (Table 2). However, patient T-cell numbers were generally unremarkable. T-cell receptor excision circles (TRECs) were within the reference range, thymic output was normal, and CD4+ and CD8+ T-cell subsets (naïve, terminally differentiated, central memory, effector memory, Th17, Treg, etc.) were all comparable with controls (Table 2). Furthermore, mitogen proliferation responses were normal.
To evaluate whether the p.Cys826Tyr BCL11B variant is disease causing, we assessed the impact of this variant on T-cell development in zebrafish as previously described (3). Briefly, we knocked down the BCL11B zebrafish orthologue bcl11ba by injecting bcl11ba antisense morpholino oligonucleotides into zebrafish embryos and then ectopically expressed WT BCL11B or p.Cys826Tyr BCL11B to investigate whether T-cell development is rescued. Confirming the pathogenicity of the p.Cys826Tyr BCL11B variant, the mutant variant was unable to rescue T-cell development as zebrafish ectopically expressing the mutant variant had no detectable thymocytes as measured by a lck probe on WISH (Figure 1G).
Here, we report one of the most severe atopic cases of BCL11B deficiency to date and the first known Canadian patient. To our knowledge, this is also the first patient with germline BCL11B variants associated with immune dysregulation/autoimmunity. Based on the clinical features of this patient and the eight previous individuals with atopic features (summarized and contrasted in Table 3), we propose to classify germline BCL11B-related atopic disease as a novel PAD. This case further emphasizes the fact that PADs can have heterogeneous clinical presentations, which can often appear to be benign especially relative to inborn errors of immunity. Based on our data and ACMG/AMP guidelines, the p.Cys826Tyr BCL11B variant can now be classified as pathogenic as it satisfies PS2, PS3, PM2, and PP3 criteria.
Although our in-depth immunophenotyping of the patient revealed intact T-cell numbers, our zebrafish model demonstrated that the p.Cys826Tyr BCL11B variant was unable to rescue T-cell development. Despite this mismatch, patient T cells are clearly defective as the patient presented with alopecia and allergic disease, both of which are T-cell dependent (9, 10). In the future, it would be important to study how immune deficiency arose in this patient despite intact thymic development (normal TRECs and recent thymic emigrants), in contrast to the ablated T-cell development observed in zebrafish. For example, since BCL11B is essential for multiple T-cell developmental checkpoints in the thymus (11), one possibility is dysregulated thymic architecture. This could be investigated by performing patient-derived CD34+ cell xenotransplants or leveraging artificial thymic organoid systems (12).
Given the modestly impaired B-cell differentiation, a part of the immunodeficiency could be derived from impaired T helper function (11). It is possible that the p.Cys826Tyr BCL11B variant alters the binding affinity of WT BCL11B to its targets or causes protein complexes to bind to new targets, leading to a predisposition to developing allergic disease. For example, p.Cys826Tyr BCL11B could continuously induce GATA3 expression without silencing IL-4 production (13). However, since the C-terminal ZFs are thought to regulate a specific subset of BCL11B activities through as-yet-unidentified binding partners (2), further investigation is needed. This is complicated by the fact that BCL11B target genes in mature effector T cells are not well understood nor is it clear how or whether BCL11B has a role in promoting or restricting the plasticity of T helper subsets other than TH17 and Tregs (2). Future work should also focus on clarifying why atopic disease arises in some individuals with germline BCL11B variants but not others and how specific variants lead to atopy.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving human participants were reviewed and approved by the Clinical Research Ethics Board, The University of British Columbia. Written informed consent to participate in this study was provided by the participants’ legal guardian/next of kin. The animal study was reviewed and approved by the Institutional Animal Care and Use Committee, Fox Chase Cancer Center.
HL performed immunophenotyping, obtained clinical data, carried out the multiple sequence alignment, in silico pathogenicity prediction, and protein modeling, wrote the manuscript, and made the figures. RS, AC, and DW modeled the BCL11B variant in zebrafish. MH, MM, KD, and KM helped with writing and editing the manuscript. EL-R, EC, WR, JM, and AL collected clinical data and helped write and edit the manuscript. ST supervised all the research, provided funding, contributed to experimental design and analysis, and helped write and edit the manuscript. All authors contributed to the article and approved the submitted version.
This work was supported by grants from the Canadian Institutes of Health Research (ST), Genome British Columbia (SIP007) (ST), Rare Disease Foundation (2174) (HL and ST), and BC Children’s Hospital Foundation (CAUSES Study, HL, ST). ST holds a Tier 1 Canada Research Chair in Pediatric Precision Health and the Aubrey J. Tingle Professor of Pediatric Immunology. HL is supported by a Canadian Institutes of Health Research Frederick Banting and Charles Best Canada Graduate Scholarship (CGS-D), Killam Doctoral Scholarship, University of British Columbia Four Year Doctoral Fellowship, and a BC Children’s Hospital Research Institute Graduate Studentship. DW was supported by the National Institutes of Health grants P30CA006927 and P01AI138962.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Milner JD. Primary Atopic Disorders. Annu Rev Immunol (2020) 38:785–808. doi: 10.1146/annurev-immunol-042718-041553
2. Sidwell T, Rothenberg EV. Epigenetic Dynamics in the Function of T-Lineage Regulatory Factor Bcl11b. Front Immunol (2021) 12:669498. doi: 10.3389/fimmu.2021.669498
3. Punwani D, Zhang Y, Yu J, Cowan MJ, Rana S, Kwan A, et al. Multisystem Anomalies in Severe Combined Immunodeficiency With Mutant BCL11B. N Engl J Med (2016) 375:2165–76. doi: 10.1056/NEJMoa1509164
4. Lessel D, Gehbauer C, Bramswig NC, Schluth-Bolard C, Venkataramanappa S, van Gassen KLI, et al. BCL11B Mutations in Patients Affected by a Neurodevelopmental Disorder With Reduced Type 2 Innate Lymphoid Cells. Brain (2018) 141:2299–311. doi: 10.1093/brain/awy173
5. Daher MT, Bausero P, Agbulut O, Li Z, Parlakian A. Bcl11b/Ctip2 in Skin, Tooth, and Craniofacial System. Front Cell Dev Biol (2020) 8:581674. doi: 10.3389/fcell.2020.581674
6. Li J, Shi L, Zhang K, Zhang Y, Hu S, Zhao T, et al. VarCards: An Integrated Genetic and Clinical Database for Coding Variants in the Human Genome. Nucleic Acids Res (2018) 46:D1039–48. doi: 10.1093/nar/gkx1039
7. Itan Y, Shang L, Boisson B, Ciancanelli MJ, Markle JG, Martinez-Barricarte R, et al. The Mutation Significance Cutoff: Gene-Level Thresholds for Variant Predictions. Nat Methods (2016) 13:109–10. doi: 10.1038/nmeth.3739
8. Yang Y, Xu Z, He C, Zhang B, Shi Y, Li F. Structural Insights Into the Recognition of Gamma-Globin Gene Promoter by BCL11A. Cell Res (2019) 29:960–3. doi: 10.1038/s41422-019-0221-0
9. Xing L, Dai Z, Jabbari A, Cerise JE, Higgins CA, Gong W, et al. Alopecia Areata is Driven by Cytotoxic T Lymphocytes and is Reversed by JAK Inhibition. Nat Med (2014) 20:1043–9. doi: 10.1038/nm.3645
10. Woodfolk JA. T-Cell Responses to Allergens. J Allergy Clin Immunol (2007) 119:280–94. doi: 10.1016/j.jaci.2006.11.008
11. Avram D, Califano D. The Multifaceted Roles of Bcl11b in Thymic and Peripheral T Cells: Impact on Immune Diseases. J Immunol (2014) 193:2059–65. doi: 10.4049/jimmunol.1400930
12. Bosticardo M, Pala F, Calzoni E, Delmonte OM, Dobbs K, Gardner CL, et al. Artificial Thymic Organoids Represent a Reliable Tool to Study T-Cell Differentiation in Patients With Severe T-Cell Lymphopenia. Blood Adv (2020) 4:2611–6. doi: 10.1182/bloodadvances.2020001730
Keywords: primary atopic disorders, inborn errors of immunity, primary immunodeficiencies, hyper IgE, BCL11B
Citation: Lu HY, Sertori R, Contreras AV, Hamer M, Messing M, Del Bel KL, Lopez-Rangel E, Chan ES, Rehmus W, Milner JD, McNagny KM, Lehman A, Wiest DL and Turvey SE (2021) A Novel Germline Heterozygous BCL11B Variant Causing Severe Atopic Disease and Immune Dysregulation. Front. Immunol. 12:788278. doi: 10.3389/fimmu.2021.788278
Received: 01 October 2021; Accepted: 02 November 2021;
Published: 23 November 2021.
Edited by:
Amit Nahum, Soroka Medical Center, IsraelReviewed by:
Arnon Broides, Ben-Gurion University of the Negev, IsraelCopyright © 2021 Lu, Sertori, Contreras, Hamer, Messing, Del Bel, Lopez-Rangel, Chan, Rehmus, Milner, McNagny, Lehman, Wiest and Turvey. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Stuart E. Turvey, c3R1cnZleUBjdy5iYy5jYQ==
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.