
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Immunol., 16 September 2020
Sec. Microbial Immunology
Volume 11 - 2020 | https://doi.org/10.3389/fimmu.2020.02195
This article is part of the Research TopicRecent Advances in the Immunology of Helminth Infection – Protection, Pathogenesis and PanaceasView all 21 articles
 Anuradha Rajamanickam1*
Anuradha Rajamanickam1* Saravanan Munisankar1
Saravanan Munisankar1 Kannan Thiruvengadam2
Kannan Thiruvengadam2 Pradeep A. Menon2
Pradeep A. Menon2 Chandrakumar Dolla2
Chandrakumar Dolla2 Thomas B. Nutman3
Thomas B. Nutman3 Subash Babu1,3,4
Subash Babu1,3,4Several epidemiological and immunological studies indicate a reciprocal association between obesity/metabolic syndrome and helminth infections. Numerous studies demonstrated that obesity is concomitant with chronic low-grade inflammation, which is marked by vital changes in cellular composition and function of adipose tissue. However, the effect of helminth infection on the homeostatic milieu in obesity is not well-understood. To determine the relationship between Strongyloides stercoralis (Ss) infection and obesity, we examined an array of parameters linked with obesity both before and at 6 months following anthelmintic treatment. To this end, we measured serum levels of pancreatic hormones, incretins, adipokines and Type-1, Type-2, Type-17, and other proinflammatory cytokines in those with non-diabetic obesity with (INF) or without Ss infection (UN). In INF individuals, we evaluated the levels of these parameters at 6 months following anthelmintic treatment. INF individuals revealed significantly lower levels of insulin, glucagon, C-peptide, and GLP-1 and significantly elevated levels of GIP compared to UN individuals. INF individuals also showed significantly lower levels of Type-1, Type-17 and other pro-inflammatory cytokines and significantly increased levels of Type-2 and regulatory cytokines in comparison to UN individuals. Most of these changes were significantly reversed following anthelmintic treatment. Ss infection is associated with a significant alteration of pancreatic hormones, incretins, adipokines, and cytokines in obese individuals and its partial reversal following anthelmintic treatment. Our data offer a possible biological mechanism for the protective effect of Ss infection on obesity.
Obesity and metabolic disorders are major public health problems because of their high prevalence worldwide. In 2016, ~1.9 billion adults were overweight and of these 650 million were obese (1). In India, more than 135 million people are suffering from obesity (2). Obesity is described as unequal body weight for height with an extra deposit of adipose tissue along with low-grade chronic and systemic inflammation (3). Obesity induced inflammation can lead to the development of type 2 diabetes, cardiovascular disease, liver disease, certain type of cancers and other pathological conditions (4, 5).
Obesity is linked with an abnormal expansion in adipose tissue mass and adiposity, as well as poorly regulated levels of adipokines and dysregulation of Type-1 and Type-2 cytokines (6). Adipose tissue aids in energy storage and secretes adipokines—adiponectin, leptin, tumor necrosis factor-α (TNF-α), resistin, and plasminogen-activator type 1 (PAI-1) (7). Obese adipose tissue primarily releases proinflammatory cytokines such as TNF-α, IL-6, leptin, visfatin, resistin, and plasminogen activator inhibitor-1. Obesity activates an immune response which incorporates a systemic elevation of inflammatory cytokines, the recruitment of immune cells to inflamed tissues, activation of leukocytes, and the generation of repair tissue responses (8).
Helminth infections affect approximately one-quarter of the world's population and are widespread in lower to middle-income countries (9). The occurrence of obesity is commonly predominant in urbanized countries where most helminth infections have been eliminated (10, 11). Recent data in both animal and human studies showed a reciprocal association between helminth infection and metabolic disorders, type-2 diabetes, insulin resistance and obesity (12–16), suggesting that helminths may have role in the prevention or delay of these diseases. In previous studies, we have shown that glycemic, hormonal, and cytokine factors in T2DM individuals are modulated by Strongyloides stercoralis infection and these changes are partially reversed following anthelmintic therapy (17). Additionally, we have also shown that T2DM with Ss infected individuals exhibited significantly lower systemic levels of cytokines and chemokines and dampens the pro-inflammatory milieu, an effect which is then reversed upon anthelmintic treatment (18). However, the mechanisms of how helminth infections mediate protection against non-diabetic obesity are unknown.
Therefore, in the current study, we wanted to examine the association among Ss infection and non-diabetic obesity and assessed the influence of Ss infection on factors essential in adipose tissue homeostasis. To this end, we estimated systemic levels of pancreatic hormones (insulin, glucagon and C-peptide), incretins (Ghrelin, GIP, GLP-1), adipokines (adiponectin, adipsin, resistin, leptin, visfatin and PAI-1) and a variety of cytokines, including Type-1 (IFN-γ, IL-2, and TNF-α), Type-17 (IL-17A and IL-22), Type-2/regulatory cytokines (IL-4, IL-5, IL-13, and IL-10) and other pro-inflammatory cytokines (IL-1α, IL-1β, IL-6, IL-12, and GM-CSF) in those with obesity with or without concomitant Ss infection. We also examined the consequence of anthelmintic therapy on the above-mentioned factors in Ss-infected individuals.
The study protocol was (12-I-073) permitted by Institutional Review Boards of the National Institute of Allergy and Infectious Diseases (USA) and the National Institute for Research in Tuberculosis (India) (approval no. NCT00375583 and NCT00001230). All individuals were screened as part of a natural history study protocol, and informed written consent was acquired from all individuals.
We enrolled 115 study participants comprising of 58 clinically asymptomatic Ss-infected study participants with obesity (hereafter INF), and 57 study participants with obesity and negative for Ss infection (hereafter UN) in Kanchipuram District, Tamil Nadu, South India (Table 1). None had previous anthelmintic treatment, a history of helminth infections or of HIV. Individuals with diabetes or prediabetes (HbA1c above 5.7%) were excluded. These individuals were different from the individuals recruited in our previous studies (17, 18).
Anthropometric measurements, including height, weight, waist circumference and biochemical parameters, including plasma glucose, lipid profiles and HbA1c were obtained using standardized techniques as detailed elsewhere (19).
The recombinant NIE antigen-ELISA detects IgG antibodies and was performed for the identification of Ss infection, as described previously (20, 21). Single stool specimens were collected at baseline from all screened individuals before anthelmintic therapy. Other intestinal helminth infections were identified by Stool microscopy and expelled from the study. Circulating filarial antigen tests were used for the diagnosis of filarial infection and those who were positive were excluded from the study. A single dose of ivermectin (12 mg) and albendazole (400 mg) were given to the INF study participants. Following 6 months of anthelmintic treatment, stool and blood samples were collected from the treated individuals to check the IgG antibody levels and to identify the presence of helminth infection. All INF individuals exhibited reductions in IgG titers at this time point and no helminths were detected by microscopy.
BMI more than or equal to 30 kg/m2 is described as obesity based on The American Association of Clinical Endocrinologists/American College of Endocrinology algorithm and American Diabetes Association guidelines. Height and body weight were measured using a digital scale.
Plasma levels of pancreatic hormones (insulin, glucagon, and C-peptide), incretins (Ghrelin, GIP, and GLP-1), adipokines (adiponectin, adipsin, resistin, leptin, visfatin, and PAI-1) and the levels of Type-1 cytokines: IFN-γ, IL-2, TNF-α, Type-17 cytokines: IL-17A, IL-22, Type-2 cytokines: IL-4, IL-5, IL-13, regulatory cytokine: IL-10 and the pro-inflammatory cytokines: IL-1α, IL-1β, IL-6, IL-12, and GM-CSF were measured using a Human Magnetic Luminex Assay kit (R&D Systems, USA) on the Bio-Rad Luminex platform (Luminex, USA), according to the manufacturer's instructions.
Central tendency was measured by using Geometric means (GM). Comparison between INF and UN were performed using Mann-Whitney U-tests with Holm's correction for multiple comparisons and the before and after treatment parameters were analyzed using Wilcoxon signed rank test. Backward stepwise methods in multiple logistic regression analysis was performed to identify factors that were influenced by Ss infection. Analyses were performed using Graph-Pad PRISM Version 8.0 (GraphPad, San Diego, CA) and Stata 15 (College Station, TX). JMP14 software was used to plot Principle Component Analysis (PCA).
The baseline demographic characteristics and biochemical parameters are shown in Table 1. There were no significant differences in age, sex, BMI or other biochemical parameters between the two groups.
To estimate the effect of Ss infection on pancreatic hormones (C-peptide, Insulin, and Glucagon), incretins (Ghrelin, GIP, and GLP-1) and adipokines (adiponectin, adipsin, resistin, leptin, visfatin, and PAI-1) in obesity, we assessed the levels of aforesaid parameters in INF and UN study participants. As illustrated in Figure 1A, the levels of insulin (GM of 17.09 pg/ml in INF compared to 30.45 pg/ml in UN; p = 0.0012), glucagon (GM of 178.7 pg/ml in INF compared to 225.4 pg/ml in UN; p = 0.0011), C-peptide (GM of 84.17 pg/ml in INF compared to 139.8 pg/ml in UN; p = 0.0070) and GLP-1 (GM of 63.69 pg/ml in INF compared to 76.71 pg/ml in UN; p = 0.0072) were significantly lower in INF than UN individuals. In contrast, GIP (GM of 20.39 pg/ml in INF compared to 12.07 pg/ml in UN; p = 0.0072) levels were significantly higher in INF compared to UN individuals. As shown in Figure 1B, resistin (GM of 0.503 ng/ml in INF compared to 0.613 ng/ml in UN; p = 0.0006), leptin (GM of 0.808 ng/ml in INF compared to 1.171 ng/ml in UN; p = 0.0048), visfatin (GM of 4.267 ng/ml in INF compared to 5.217 pg/ml in UN; p = 0.0215) and PAI-1 (GM of 2.51 ng/ml in INF compared to 3.66 ng/ml in UN; p = 0.0013) were significantly lower in INF than UN individuals. In contrast, adiponectin (GM of 29.41 ng/ml in INF compared to 9.54 ng/ml in UN; p = 0.0070) and adipsin (GM of 4.27 ng/ml in INF compared to 2.63 ng/ml in UN; p = 0.0055) were significantly higher in INF than UN individuals. Therefore, Ss infection is characterized by lower systemic levels of the pancreatic hormones, incretins and altered levels of adipokines in Ss-infected individuals with obesity.
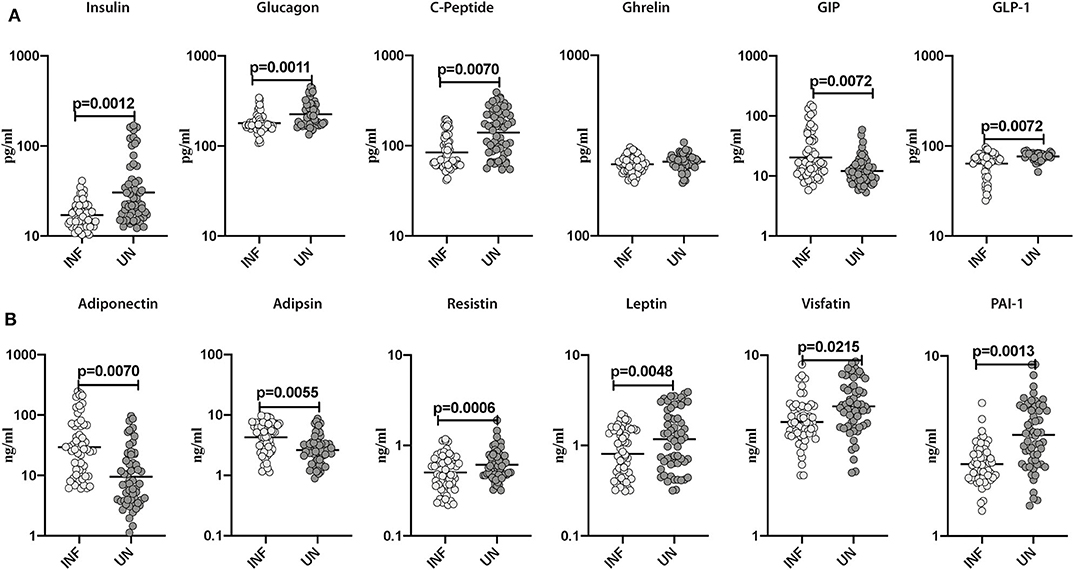
Figure 1. Lower systemic levels of pancreatic hormones and altered levels of incretins and adipokines in INF individuals. (A) Plasma levels of insulin, glucagon, C-peptide, ghrelin, GIP and GLP-1 in INF and UN individuals were measured. (B) Plasma levels of adiponectin, adipsin, resistin, leptin, visfatin and PAI-1 in INF and UN individuals were measured. Each dot is an individual subject with the bar representing the geometric mean (GM). Mann– Whitney U-test with Holms correction for multiple comparisons were done to calculate p-values.
Subsequently, we sought to examine the influence of Ss infection on the systemic levels of Type-1 (IFN-γ, TNF-α, and IL-2), Type-17 (IL-17A and IL-22), Type-2 (IL-4, IL-5, and IL-13), regulatory (IL-10) and pro-inflammatory cytokines (IL-1α, IL-1β, IL-6, IL-12, and GM-CSF) in INF and UN individuals. As illustrated in Figure 2A, INF individuals exhibited significantly lower levels of IFN-γ (GM of 176.2 pg/ml in INF compared to 631.6 pg/ml in UN; p = 0.0014), IL-2 (GM of 115.8 pg/ml compared to 150.9 pg/ml; p = 0.0013), TNF-α (GM of 432.2 pg/ml in INF compared to 768.8 pg/ml in UN; p = 0.0012), IL-17A (GM of 144.7 pg/ml in INF compared to 275.1 pg/ml in UN; p = 0.0087) in comparison with UN individuals. In contrast, INF individuals exhibited significantly higher levels of IL-22 (GM of 234.4 pg/ml in INF compared to 93.14 pg/ml in UN; p = 0.0010) when compared to UN individuals. As illustrated in Figure 2B, IL-4 (GM of 588.4 pg/ml in INF compared to 338.8 pg/ml in UN; p = 0.0009), IL-5 (GM of 72.71 pg/ml in INF compared to 37.79 pg/ml in UN; p = 0.0008), IL-13 (GM of 82.68 pg/ml in INF compared to 56 pg/ml in UN; p = 0.0006), and IL-10 (GM of 193.4 pg/ml in INF compared to 119.4 pg/ml in UN; p = 0.0007) levels were significantly lower in INF compared to UN individuals. As illustrated in Figure 2C, IL-1β (GM of 168.7 pg/ml in INF compared to 800 pg/ml in UN; p = 0.0009), IL-6 (GM of 56.35 pg/ml in INF compared to 82.19 pg/ml in UN; p = 0.0009) and IL-12 (GM of 50.39 pg/ml in INF compared to 61.89 pg/ml in UN; p = 0.0009) levels were significantly lower in INF than UN individuals. Thus, Ss infection with obesity is characterized by lower systemic levels of IL-17, Type-1 associated cytokines and pro-inflammatory cytokines and higher systemic levels of IL-22 and Type-2 associated cytokines.
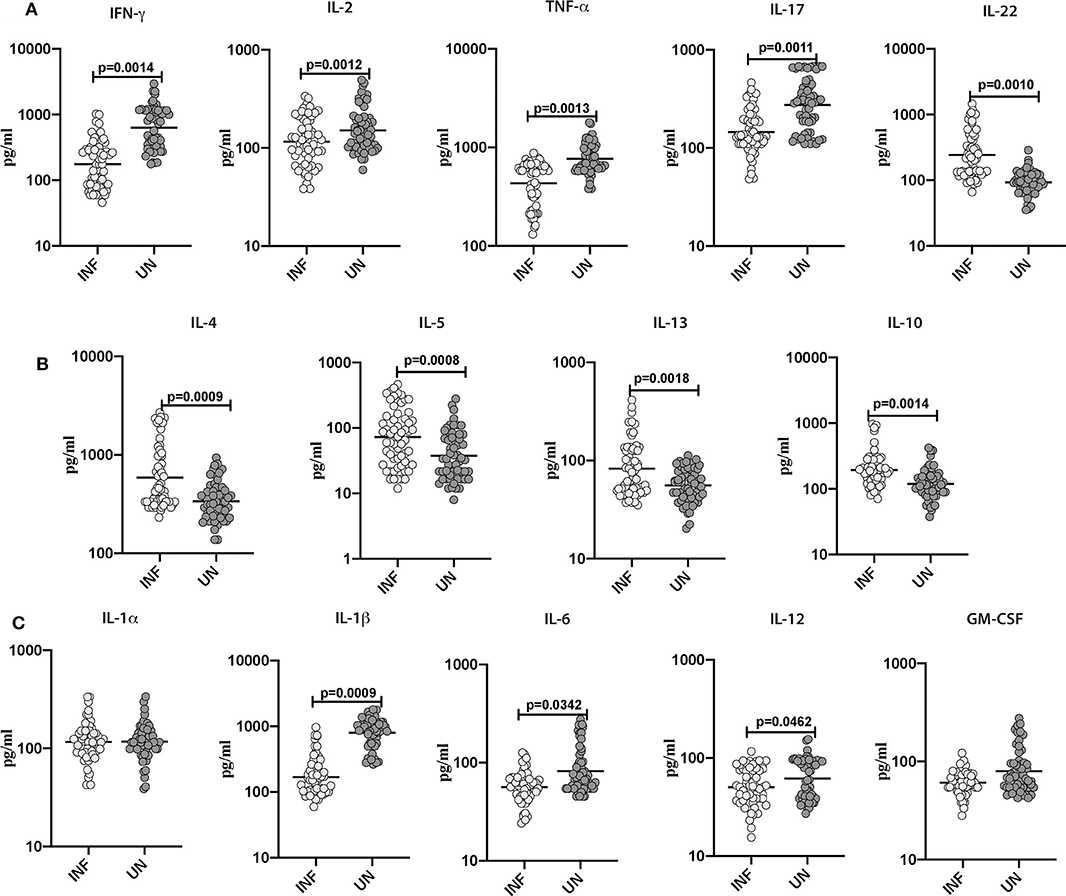
Figure 2. Lower systemic levels of Type-1 and Type-17 and other pro-inflammatory cytokines and higher levels of Type-2 cytokines in INF individuals. (A) Plasma levels of Type-1 (IFNγ, TNFα and IL-2)-, Type-17 (IL-17A and IL-22)- cytokines in INF and UN. (B) Plasma levels of Type-2 (IL-4, IL-5, and IL-13)- and regulatory (IL-10) cytokine in INF and UN individuals were measured. (C) Plasma levels of other pro-inflammatory (IL-1α, IL-1β, IL-6, IL-12, and GM-CSF) in INF and UN individuals were measured. Each dot is an individual subject with the bar representing the geometric mean (GM). Mann–Whitney U-test with Holms correction for multiple comparisons were done to calculate p-values.
Subsequently, we sought to examine the effect of anthelmintic therapy on the levels of pancreatic hormones, incretins and adipokines in INF individuals. For this purpose, we assessed the plasma levels of pancreatic hormones, incretins and adipokines in INF individuals following 6 months of anthelmintic therapy. As illustrated in Figure 3A, the post-treatment levels of insulin (percentage increase of 9%; p = 0.0009), C-peptide (percentage increase of 6%; p = 0.0012), GIP (percentage increase of 13%; p = 0.0011) and GLP-1 (percentage increase of 16%; p = 0.0183) were significantly increased than pre-treatment levels. As illustrated in Figure 3B, the post-treatment levels of adiponectin (percentage decrease of 11%; p = 0.0005) and adipsin (percentage decrease of 25%; p = 0.0003) were significantly decreased than the pre-treatment levels. In contrast, the post-treatment levels of resistin (percentage increase of 14%; p = 0.0006), leptin (percentage increase of 13%; p = 0.0004), and PAI-1 (percentage increase of 21%; p = 0.0055) were significantly increased than the pre-treatment levels. As illustrated in Supplementary Figure 1, the levels of pancreatic hormones, incretins and adipokines were not significantly different between the uninfected and post-treated individuals. Therefore, anthelmintic therapy is associated with significant alteration of pancreatic hormones, incretins and adipokines in obese individuals.
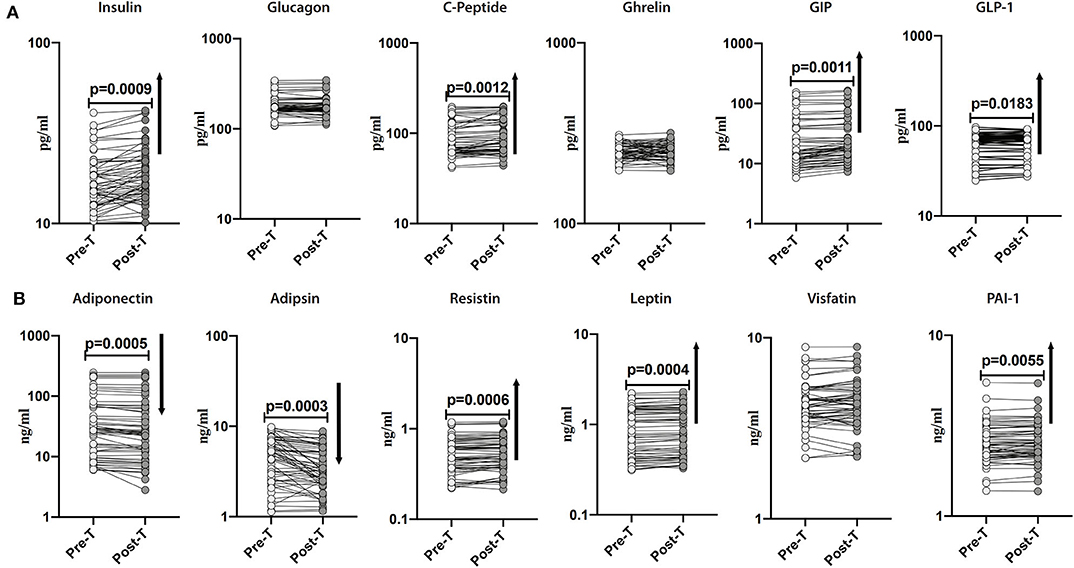
Figure 3. Anthelmintic treatment alters systemic levels of pancreatic hormones, incretins, and adipokines in INF individuals. (A) Plasma levels of insulin, glucagon, C-peptide, ghrelin, GIP, and GLP-1 in INF individuals pre-treatment [Pre-T] and 6 months following treatment [post-T] were measured. (B) Plasma levels of adiponectin, adipsin, resistin, leptin, visfatin, and PAI-1 in INF individuals pre-treatment [Pre-T] and 6 months following treatment [post-T] were measured. p-values were calculated using the Wilcoxon matched pair test.
To examine the impact of anthelmintic therapy on systemic levels of Type-1 (IFN-γ, TNF-α, and IL-2)- and Type-17 (IL-17A and IL-22)- associated cytokines, we assessed the cytokines in INF individuals at baseline and following 6 months of anthelmintic therapy. As illustrated in Figure 4A, following anthelmintic treatment, the levels of IFN-γ (percentage increase of 8.3%; p = 0.0014), IL-2 (percentage increase of 7.9%; p = 0.0012), TNF-α (percentage increase of 4.6%; p = 0.0013), IL-17A (percentage increase of 6.4%; p = 0.0011) were increased, whereas IL-22 (percentage decrease of 1.2%; p = 0.0010) levels were decreased.
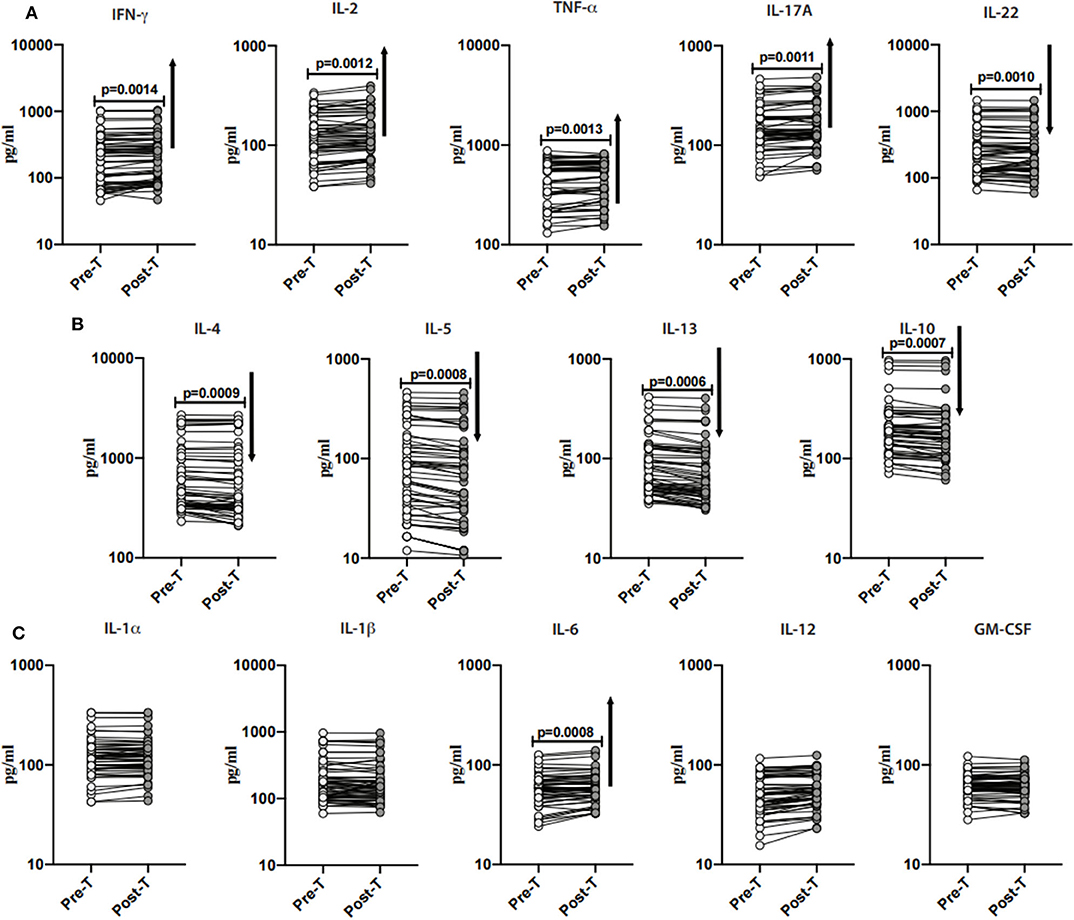
Figure 4. Anthelmintic treatment alters systemic levels of Type-1, Type-17, Type-2- cytokines, and IL-10 in INF individuals. (A) Plasma levels of Type-1 (IFNγ, TNFα, and IL-2)- and Type-17 (IL-17A and IL-22)- cytokines in INF individuals pre-treatment [Pre-T] and 6 months following treatment [post-T] were measured. (B) Plasma levels of Type-2 (IL-4, IL-5, and IL-13)- and regulatory (IL-10) cytokine in INF individuals pre-treatment [Pre-T] and 6 months following treatment [post-T] were measured. (C) Plasma levels of other pro-inflammatory (IL-1α, IL-1β, IL-6, IL-12, and GM-CSF) in INF individuals pre-treatment [Pre-T] and 6 months following treatment [post-T] were measured. p-values were calculated using the Wilcoxon matched pair test.
Next, to examine the impact of anthelmintic therapy on Type-2- and regulatory cytokines, we assessed the cytokines in INF individuals and baseline and following 6 months of anthelmintic therapy. As illustrated in Figure 4B, IL-4 (percentage decrease of 6.8%; p = 0.0009), IL-5 (percentage decease of 14.5%; p = 0.0008), IL-13 (percentage decrease of 12.7%; p = 0.0006) and IL-10 (percentage decrease of 8%; p = 0.0007) levels were decreased when compared to their pre-treatment levels.
Further, to examine the impact of anthelmintic treatment on pro-inflammatory cytokines, we measured the pro-inflammatory cytokines in INF individuals at 6 months following anthelmintic treatment. As shown in Figure 4C, IL-6 (percentage increase of 5.3%; p = 0.0008) levels were increased when compared to pre-treatment levels. Other pro-inflammatory cytokines did not show any significant difference when compared to their pre-treatment levels.
As shown in Supplementary Figure 2, the levels of Type-1, Type-17, and pro-inflammatory cytokines and Type-2 cytokines were not significantly different between the uninfected and post-treated individuals. Thus, anthelmintic treatment is associated with increased Type-1 and Type-17 associated cytokines, pro-inflammatory cytokine, IL-6, and decreased Type-2 associated cytokines in obese individuals.
PCA was used to visualize differences between the groups based on the entire data set. To determine the clustering pattern of pancreatic hormones, incretins, adipokines, and cytokines between INF (red circle) and UN (blue circle) individuals, we strategized PCA with diverse inputs. As illustrated in Figure 5A, PCA analysis exhibited that pancreatic hormones, incretins, adipokines cluster differently between INF and UN individuals. The score plot of the first two components revealed 23.2 and 16.7% of overall variance, correspondingly. As shown in Figure 5B, PCA analysis of cytokines exhibited two different clusters between INF and UN individuals. The score plot of the first two components revealed 25.5 and 16.9% of overall variance, correspondingly. Thus, PCA analysis reveals the overall influence of pancreatic hormones, incretins, adipokines, and cytokine of Ss infection on obesity.
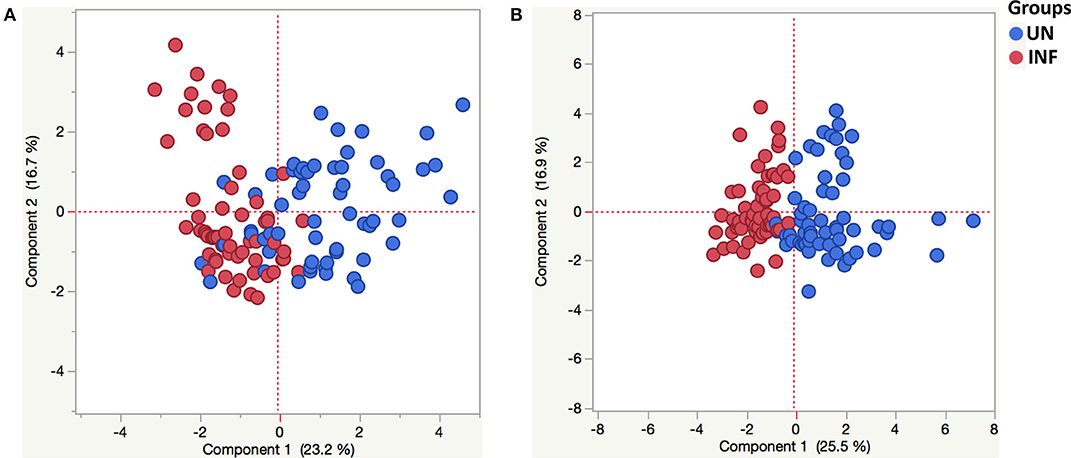
Figure 5. Principle component analysis reveals tendencies in pancreatic hormones, incretins, adipokines and cytokine milieu in helminth-obese individuals. Principal component analysis (PCA) was performed to show the distribution of data from the combination of two groups INF (red circles) and UN (blue circles). The PCA represents the two principal components of variation. (A) PCA for insulin, glucagon, C-peptide, ghrelin, GIP and GLP-1, adiponectin, adipsin, resistin, leptin, visfatin, and PAI-1in INF and UN individuals. (B) PCA for Type-1 (IFNγ, TNFα, and IL-2), Type-17 (IL-17A and IL-22), and other pro-inflammatory (IL-1α, IL-1β, IL-6, IL-12, and GM-CSF) cytokines of INF and UN individuals.
The influence of confounding variables on obese individuals with different analytes was evaluated in this study using multivariate regression analysis. As illustrated in Table 2, even after correcting for the influence of the age and sex, the levels of biochemical parameters such as AST, ALT and urea, RBG, HbA1c, diabetic parameters such as insulin, glucagon, C-peptide, GIP, GLP-1, adiponectin, adipsin, resistin, leptin, visfatin, and PAI-1; cytokines like IFN-γ, IL-2, TNF-α, IL-17, IL-22, IL-4, IL-5, IL-13, IL-10, IL-1β, IL-6, and IL-12 were all significantly manipulated by Ss infection. Therefore, our data corroborate that Ss infection has a great impact on numerous significant factors in obese individuals, including blood glucose levels, and the levels of the adipocytokines and the more conventional cytokines.
Helminth infections are known to limit harmful inflammatory responses and assist local and systemic metabolic homeostasis (22). Previously published reports revealed that helminths could limit the progression of metabolic diseases (23, 24), probably by altering host inflammatory responses (24). Thus, it has been proposed that a decrease in helminth infections could influence the incidence of inflammatory diseases, T2DM, obesity, insulin resistance and metabolic syndrome in many of the high income countries (25).
An earlier study showed that insulin and C-peptide levels were increased in obese individuals compared to lean controls and suggested obese subjects have an impaired glucose homeostasis and exhibit prediabetic factors, including hyperinsulinemia, and insulin resistance (26–29). Glucagon levels were elevated in obese individuals (26). Likewise, the present study has demonstrated that Ss infection was associated with lower systemic levels of insulin, glucagon and C-peptide levels when compared with obese without Ss infection and reversal following anthelmintic treatment. Recently, we have shown that Ss infection has a protective role on diabetes-related parameters and that Ss infected T2DM individuals showed decreased levels of insulin and glucagon that increased following therapy (17). Recent data also revealed that adiponectin reduces IFNγ and IL-17 responses by T cells in obese mice (30). Previously we have shown that adiponectin and adipsin levels were lowered in Ss infection with T2DM in comparison with Ss uninfected with T2DM individuals. In the present study, both adiponectin and adipsin are exhibited at elevated levels in the systemic circulation in INF individuals. These levels were significantly decreased after 6 months of anthelmintic therapy, indicating that Ss infection could modify the adipocytokine levels in INF individuals. Leptin induces pro-inflammatory immune responses and limits the proliferation of regulatory T cells, while adiponectin promotes the production of anti-inflammatory cytokines (31). The difference between the levels of these two adipokines has been shown to be linked with pro-inflammatory conditions and insulin resistance. A recent study revealed that leptin to adiponectin ratios in STH-infected individuals was increased following anthelmintic therapy which may in small part, lead to the moderate rise in insulin resistance (32). In our study, resistin, leptin, visfatin, PAI-1 levels were significantly lower in the INF group and increased following anthelmintic therapy. Helminths may have an impact on glucose homeostasis and insulin resistance in obesity through alternative mechanisms such as modulating the levels of adipocytokines (33, 34). This implies that adipokines have a key role in the facilitation of helminth-associated beneficial influence on insulin resistance.
Helminth infections have the ability to modulate immune responses (35). Previously, we have reported that Ss-infected individuals exhibited significantly decreased systemic levels of the pro-inflammatory cytokines and significantly heightened levels of the Type-2-related and regulatory cytokines (36). Type-1 related cytokines IL-1, IL-6, IL-8, IFN-γ, and MCP-1 are highly expressed in obese individuals (37). Another study showed that accumulation of inflammatory cells and adipocytes results in increased secretion of cytokines like tumor necrosis factor alpha (TNF-α) and interleukin-6 (IL-6) and lead to the progression of metabolic syndrome, consequently worsening the outcome of obesity (38). Previously, we have shown that Ss infection in T2DM showed significantly lower levels of Type-1 and Type-17 associated cytokines and pro-inflammatory cytokines with an increase following anthelmintic treatment (17, 18). In line with this in our study, Type-1 (IFN-γ, TNF-α, and IL-2)- and Type-17 (IL-17A)- cytokines were significantly decreased in INF individuals when compared to those without Ss infection. Hence, our data implies that helminth infection is characterized by the alteration of Type-1- and Type-17- cytokine responses. In our current study, IL-22 levels were increased in Ss-infected obese individuals and decreased following treatment. Since IL-22 is known to play a crucial role in modulating lipid metabolism and the IL-22 pathway is vital for preserving epithelial integrity, lowering chronic inflammation, and improving metabolic syndromes (39, 40), our data implies that IL-22 modulation might have a potential impact on lipid metabolism in obese individuals.
IL-4 and IL-13 are critically associated with the regulation of adipose tissue homeostasis, indicating that helminths may have an impact on metabolic status by altering adipose tissue function (41). An earlier study on Ss infection with T2DM showed significantly increased levels of Type-2 related cytokines (17). In our current study, an increased expression of the prototypical Type-2-associated cytokines IL-4, IL-5, IL-13, and IL-10 in Ss-infected obese individuals was observed. IL-10 has been shown to be able to improve inflammation in obese adipose tissue and insulin resistance induced by proinflammatory cytokines, TNF-α and IL-6 (1, 42). Various studies have determined that helminth infection and helminth- derived molecules, S.mansoni secreted SEA (43), Schistosoma mansoni egg-derived ω1 recombinant SEA (44), L. sigmodontis antigen (15) play an important role in metabolic disorders by stimulating a T helper 2 (Th2) response and increasing insulin sensitivity. Our data suggest that Ss infection could limit the inflammatory process (lower levels of pancreatic hormones, Type-I, Type-17, pro-inflammatory cytokines) by producing type-2 cytokines. This needs to be studied further mechanistically.
Taken together, our data clearly show the positive impacts of helminth infection on obesity related metabolic dysfunction. Our study has certain limitations including not determining insulin resistance directly, not having a non-obese control group and not having a placebo control group. Nevertheless, our study reveals that Ss infection could play a defensive role opposing the deleterious consequences of obesity by altering hormones, adipokines and the associated cytokine milieu. Certain parameters such as glucagon, visfatin and cytokines remain not significantly altered following treatment, which could perhaps reflect different kinetics in the alteration of these parameters or a lack of alteration. Our data suggest a significant association between helminth infection and the alteration of the homeostatic milieu in obesity and provide better knowledge of helminth-driven immune- and non-immune–mediated modulation of host metabolism. Our data also reinforce the possibility for helminths as a new class of biologics in alleviating inflammatory diseases and metabolic disorders. A more detailed characterization of the definitive immunomodulatory components of helminths could promote a more precise treatment approach against obesity and other inflammatory diseases.
All datasets generated for this study are included in the article/Supplementary Material.
The studies involving human participants were reviewed and approved by All participants were examined as part of a natural history study protocol (12-I-073) approved by Institutional Review Boards of the National Institute of Allergy and Infectious Diseases (USA) and the National Institute for Research in Tuberculosis (India) (approval no. NCT00375583 and NCT00001230), and informed written consent was obtained from all participants. The patients/participants provided their written informed consent to participate in this study.
SB and AR conceived and planned the experiments. AR and SM executed the experiments. AR, SB, and KT analyzed the data. PM and CD provided patient samples. TN contributed reagents, materials, and analysis tools. AR, SB, and TN wrote, reviewed, and edited the manuscript. All authors contributed to manuscript revision, read, and approved the submitted version.
This work was funded by the Division of Intramural Research, NIAID, NIH.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
We thank Dr. M. Satiswaran, Mrs. Suganthi, and Prabbu Balakrishnan for valuable assistance in collecting the clinical data for this study. We thank the staff of the Department of Epidemiology, NIRT, for valuable assistance in recruiting the patients for this study.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2020.02195/full#supplementary-material
Supplementary Figure 1. No significant differences in the systemic levels of pancreatic hormones, incretins, and adipokines between UN and Post treated individuals. (A) Plasma levels of insulin, glucagon, C-peptide, ghrelin, GIP and GLP-1 in UN, and Post-T individuals were measured. (B) Plasma levels of adiponectin, adipsin, resistin, leptin, visfatin and PAI-1 in UN and Post-T individuals were measured. Each dot is an individual subject with the bar representing the geometric mean (GM). Mann– Whitney U-test were done to calculate p-values.
Supplementary Figure 2. No significant differences in the systemic levels of Type-1 and Type-17 and other pro-inflammatory cytokines and Type-2 cytokines between UN and Post treated individuals. (A) Plasma levels of Type-1 (IFNγ, TNFα, and IL-2)-, Type-17 (IL-17A and IL-22)- cytokines in UN and Post-T individuals were measured. (B) Plasma levels of Type-2 (IL-4, IL-5, and IL-13)- and regulatory (IL-10) cytokine in UN and Post-T individuals were measured. (C) Plasma levels of other pro-inflammatory (IL-1α, IL-1β, IL-6, IL-12, and GM-CSF) in UN and Post-T individuals were measured. Each dot is an individual subject with the bar representing the geometric mean (GM). Mann– Whitney U-test were done to calculate p-values.
1. Mantovani A, Sica A, Sozzani S, Allavena P, Vecchi A, Locati M. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. (2004) 25:677–86. doi: 10.1016/j.it.2004.09.015
2. Ahirwar R, Mondal PR. Prevalence of obesity in India: a systematic review. Diabetes Metab Syndr. (2019) 13:318–21. doi: 10.1016/j.dsx.2018.08.032
3. Gonzalez-Muniesa P, Martinez-Gonzalez MA, Hu FB, Despres JP, Matsuzawa Y, Loos RJF, et al. Obesity. Nat Rev Dis Primers. (2017) 3:17034. doi: 10.1038/nrdp.2017.34
4. Gregor MF, Hotamisligil GS. Inflammatory mechanisms in obesity. Annu Rev Immunol. (2011) 29:415–45. doi: 10.1146/annurev-immunol-031210-101322
5. Williams EP, Mesidor M, Winters K, Dubbert PM, Wyatt SB. Overweight and obesity: prevalence, consequences, and causes of a growing public health problem. Curr Obes Rep. (2015) 4:363–70. doi: 10.1007/s13679-015-0169-4
6. Lee IS, Shin G, Choue R. Shifts in diet from high fat to high carbohydrate improved levels of adipokines and pro-inflammatory cytokines in mice fed a high-fat diet. Endocr J. (2010) 57:39–50. doi: 10.1507/endocrj.K09E-046
7. Garg MK, Dutta MK, Mahalle N. Adipokines (adiponectin and plasminogen activator inhhibitor-1) in metabolic syndrome. Indian J Endocrinol Metab. (2012) 16:116–23. doi: 10.4103/2230-8210.91206
8. Spencer M, Yao-Borengasser A, Unal R, Rasouli N, Gurley CM, Zhu B, et al. Adipose tissue macrophages in insulin-resistant subjects are associated with collagen VI and fibrosis and demonstrate alternative activation. Am J Physiol Endocrinol Metab. (2010) 299:E1016–27. doi: 10.1152/ajpendo.00329.2010
9. Hotez PJ, Alvarado M, Basanez MG, Bolliger I, Bourne R, Boussinesq M, et al. The global burden of disease study 2010: interpretation and implications for the neglected tropical diseases. PLoS Negl Trop Dis. (2014) 8:e2865. doi: 10.1371/journal.pntd.0002865
10. Bhurosy T, Jeewon R. Overweight and obesity epidemic in developing countries: a problem with diet, physical activity, or socioeconomic status? ScientificWorldJournal. (2014) 2014:964236. doi: 10.1155/2014/964236
11. Shimokawa C, Obi S, Shibata M, Olia A, Imai T, Suzue K, et al. Suppression of obesity by an intestinal helminth through interactions with intestinal microbiota. Infect Immun. (2019) 87:e00042–19. doi: 10.1128/IAI.00042-19
12. Aravindhan V, Mohan V, Surendar J, Muralidhara Rao M, Pavankumar N, Deepa M, et al. Decreased prevalence of lymphatic filariasis among diabetic subjects associated with a diminished pro-inflammatory cytokine response (CURES 83). PLoS Negl Trop Dis. (2010) 4:e707. doi: 10.1371/journal.pntd.0000707
13. Hays R, Esterman A, Giacomin P, Loukas A, Mcdermott R. Does strongyloides stercoralis infection protect against type 2 diabetes in humans? Evidence from australian aboriginal adults diabetes. Res Clin Pract. (2015) 107:355–61. doi: 10.1016/j.diabres.2015.01.012
14. Wiria AE, Hamid F, Wammes LJ, Prasetyani MA, Dekkers OM, May L, et al. Infection with soil-transmitted helminths is associated with increased insulin sensitivity. PLoS ONE. (2015) 10:e0127746. doi: 10.1371/journal.pone.0127746
15. Berbudi A, Surendar J, Ajendra J, Gondorf F, Schmidt D, Neumann AL, et al. Filarial infection or antigen administration improves glucose tolerance in diet-induced obese mice. J Innate Immun. (2016) 8:601–16. doi: 10.1159/000448401
16. Su CW, Chen CY, Li Y, Long SR, Massey W, Kumar DV, et al. Helminth infection protects against high fat diet-induced obesity via induction of alternatively activated macrophages. Sci Rep. (2018) 8:4607. doi: 10.1038/s41598-018-22920-7
17. Rajamanickam A, Munisankar S, Bhootra Y, Dolla C, Thiruvengadam K, Nutman TB, et al. Metabolic Consequences of Concomitant Strongyloides stercoralis Infection in Patients With Type 2 Diabetes Mellitus. Clin Infect Dis. (2019) 69:697–704. doi: 10.1093/cid/ciy935
18. Rajamanickam A, Munisankar S, Dolla C, Menon PA, Thiruvengadam K, Nutman TB, et al. Helminth infection modulates systemic pro-inflammatory cytokines and chemokines implicated in type 2 diabetes mellitus pathogenesis. PLoS Negl Trop Dis. (2020) 14:e0008101. doi: 10.1371/journal.pntd.0008101
19. Deepa M, Pradeepa R, Rema M, Mohan A, Deepa R, Shanthirani S, et al. The Chennai Urban Rural Epidemiology Study (CURES)–study design and methodology (urban component) (CURES-I). J Assoc Physicians India. (2003) 51:863–70.
20. Bisoffi Z, Buonfrate D, Sequi M, Mejia R, Cimino RO, Krolewiecki AJ, et al. Diagnostic accuracy of five serologic tests for Strongyloides stercoralis infection. PLoS Negl Trop Dis. (2014) 8:e2640. doi: 10.1371/journal.pntd.0002640
21. Buonfrate D, Formenti F, Perandin F, Bisoffi Z. Novel approaches to the diagnosis of Strongyloides stercoralis infection. Clin Microbiol Infect. (2015) 21:543–52. doi: 10.1016/j.cmi.2015.04.001
22. Mishra PK, Palma M, Bleich D, Loke P, Gause WC. Systemic impact of intestinal helminth infections. Mucosal Immunol. (2014) 7:753–62. doi: 10.1038/mi.2014.23
23. Shen SW, Lu Y, Li F, Shen ZH, Xu M, Yao WF, et al. The potential long-term effect of previous schistosome infection reduces the risk of metabolic syndrome among Chinese men. Parasite Immunol. (2015) 37:333–9. doi: 10.1111/pim.12187
24. Tracey EF, Mcdermott RA, Mcdonald MI. Do worms protect against the metabolic syndrome? A systematic review and meta-analysis Diabetes. Res Clin Pract. (2016) 120:209–20. doi: 10.1016/j.diabres.2016.08.014
25. Wiria AE, Djuardi Y, Supali T, Sartono E, Yazdanbakhsh M. Helminth infection in populations undergoing epidemiological transition: a friend or foe? Semin Immunopathol. (2012) 34:889–901. doi: 10.1007/s00281-012-0358-0
26. Meyer-Gerspach AC, Cajacob L, Riva D, Herzog R, Drewe J, Beglinger C, et al. Mechanisms regulating insulin response to intragastric glucose in lean and non-diabetic obese subjects: a randomized, double-blind, parallel-group trial. PLoS ONE. (2016) 11:e0150803. doi: 10.1371/journal.pone.0150803
27. Ladwa M, Hakim O, Amiel SA, Goff LM. A systematic review of beta cell function in adults of black african ethnicity. J Diabetes Res. (2019) 2019:7891359. doi: 10.1155/2019/7891359
28. Thomas DD, Corkey BE, Istfan NW, Apovian CM. Hyperinsulinemia: an early indicator of metabolic dysfunction. J Endocr Soc. (2019) 3:1727–47. doi: 10.1210/js.2019-00065
29. Kumar AA, Satheesh G, Vijayakumar G, Chandran M, Prabhu PR, Simon L, et al. Postprandial metabolism is impaired in overweight normoglycemic young adults without family history of diabetes. Sci Rep. (2020) 10:353. doi: 10.1038/s41598-019-57257-2
30. Surendar J, Frohberger SJ, Karunakaran I, Schmitt V, Stamminger W, Neumann AL, et al. Adiponectin Limits IFN-gamma and IL-17 producing CD4 T cells in obesity by restraining cell intrinsic glycolysis. Front Immunol. (2019) 10:2555. doi: 10.3389/fimmu.2019.02555
31. Carbone F, La Rocca C, Matarese G. Immunological functions of leptin and adiponectin. Biochimie. (2012) 94:2082–8. doi: 10.1016/j.biochi.2012.05.018
32. Tahapary DL, De Ruiter K, Martin I, Brienen E, a.T., Van Lieshout L, et al. Effect of anthelmintic treatment on leptin, adiponectin and leptin to adiponectin ratio: a randomized-controlled trial. Nutr Diabetes. (2017) 7:e289. doi: 10.1038/nutd.2017.37
33. Finucane FM, Luan J, Wareham NJ, Sharp SJ, O'rahilly S, Balkau B, et al. Correlation of the leptin:adiponectin ratio with measures of insulin resistance in non-diabetic individuals. Diabetologia. (2009) 52:2345–9. doi: 10.1007/s00125-009-1508-3
34. Chou HH, Hsu LA, Wu S, Teng MS, Sun YC, Ko YL. Leptin-to-adiponectin ratio is related to low grade inflammation and insulin resistance independent of obesity in non-diabetic taiwanese: a cross-sectional cohort study. Acta Cardiol Sin. (2014) 30:204–14.
35. Allen JE, Maizels RM. Diversity and dialogue in immunity to helminths. Nat Rev Immunol. (2011) 11:375–88. doi: 10.1038/nri2992
36. Anuradha R, Munisankar S, Bhootra Y, Jagannathan J, Dolla C, Kumaran P, et al. Systemic Cytokine Profiles in Strongyloides stercoralis Infection and alterations following Treatment. Infect Immun. (2016) 84:425–31. doi: 10.1128/IAI.01354-15
37. Tilg H, Moschen AR. Adipocytokines: mediators linking adipose tissue, inflammation and immunity. Nat Rev Immunol. (2006) 6:772–83. doi: 10.1038/nri1937
38. Olefsky JM, Glass CK. Macrophages, inflammation, and insulin resistance. Annu Rev Physiol. (2010) 72:219–46. doi: 10.1146/annurev-physiol-021909-135846
39. Yang L, Zhang Y, Wang L, Fan F, Zhu L, Li Z, et al. Amelioration of high fat diet induced liver lipogenesis and hepatic steatosis by interleukin-22. J Hepatol. (2010) 53:339–47. doi: 10.1016/j.jhep.2010.03.004
40. Wang X, Ota N, Manzanillo P, Kates L, Zavala-Solorio J, Eidenschenk C, et al. Interleukin-22 alleviates metabolic disorders and restores mucosal immunity in diabetes. Nature. (2014) 514:237–41. doi: 10.1038/nature13564
41. Moyat M, Coakley G, Harris NL. The interplay of type 2 immunity, helminth infection and the microbiota in regulating metabolism. Clin Transl Immunology. (2019) 8:e01089. doi: 10.1002/cti2.1089
42. Gordon S, Taylor PR. Monocyte and macrophage heterogeneity. Nat Rev Immunol. (2005) 5:953–64. doi: 10.1038/nri1733
43. Hussaarts L, Garcia-Tardon N, Van Beek L, Heemskerk MM, Haeberlein S, Van Der Zon GC, et al. Chronic helminth infection and helminth-derived egg antigens promote adipose tissue M2 macrophages and improve insulin sensitivity in obese mice. FASEB J. (2015) 29:3027–39. doi: 10.1096/fj.14-266239
Keywords: strongyloides stercoralis infection, obesity, adipocytokines, pancreatic hormones, cytokines
Citation: Rajamanickam A, Munisankar S, Thiruvengadam K, Menon PA, Dolla C, Nutman TB and Babu S (2020) Impact of Helminth Infection on Metabolic and Immune Homeostasis in Non-diabetic Obesity. Front. Immunol. 11:2195. doi: 10.3389/fimmu.2020.02195
Received: 25 June 2020; Accepted: 11 August 2020;
Published: 16 September 2020.
Edited by:
Mark C. Siracusa, Rutgers Biomedical and Health Sciences, United StatesReviewed by:
William Horsnell, University of Cape Town, South AfricaCopyright © 2020 Rajamanickam, Munisankar, Thiruvengadam, Menon, Dolla, Nutman and Babu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Anuradha Rajamanickam, YW51cmFkaGFAbmlydC5yZXMuaW4=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.