
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Glob. Womens Health , 31 March 2025
Sec. Sex and Gender Differences in Disease
Volume 6 - 2025 | https://doi.org/10.3389/fgwh.2025.1544832
This article is part of the Research Topic The Impact of Sex and Gender in Disease Diagnostics in Global Health View all 5 articles
 Aijan Taalaibekova1*†
Aijan Taalaibekova1*† Michelle Meyer2,†
Michelle Meyer2,† Stefanie Ulrich2
Stefanie Ulrich2 Gulzada Mirzalieva1
Gulzada Mirzalieva1 Maamed Mademilov1
Maamed Mademilov1 Mona Lichtblau2
Mona Lichtblau2 Cornelia Betschart3
Cornelia Betschart3 Talant M. Sooronbaev1
Talant M. Sooronbaev1 Silvia Ulrich2
Silvia Ulrich2 Konrad E. Bloch2
Konrad E. Bloch2 Michael Furian2,4
Michael Furian2,4
Background: The susceptibility to acute mountain sickness (AMS) in relation to sexual hormones in women remains elusive, partly because hormones could not be conveniently measured. We evaluated a novel kit for self-monitoring sexual hormones in women and recorded AMS incidence during high-altitude sojourns.
Methods: Two groups of healthy, premenopausal women, mean ± SD age 23.1 ± 2.3 years, residing <1,000 m underwent baseline evaluations at 760 m before travelling to and staying for 2 days and nights (48 h) at 3,100 m or 3,600 m, respectively. Participants self-monitored morning urine sexual hormone concentrations (estrone-1-glucuronide, E1G, pregnanediol-3-alpha-glucuronide, PdG, and luteinizing hormone, LH) daily for 30d including altitude sojourns using the simple “Proov” kit (MFB Fertility Inc). Follicular and luteal menstrual cycle phases detected by LH peak, altitude-related adverse health effects (ARAHE), AMS [Lake Louise score 2018 (LLS) ≥ 3 points including headache] and pulse oximetry (SpO2) were assessed.
Results: 1,172 of 1,250 (93.8%) hormone measurements were successful, 78 of 1,250 (6.2%) failed due to nonadherence or technical failure. At 3,600 m, mean differences in urinary PdG concentration were 3.8 mcg/ml (95% CI, 0.6–7.1) between luteal and follicular cycle phases. At 3,100 m, corresponding difference was 8.5 mcg/ml (95% CI, 5.0–12.0). At 3,100 m, 9 of 21 (43%) women were diagnosed with AMS with SpO2 of 93.0 ± 1.6% and LLS of 0.3 ± 1.4 in the morning after the first night. At 3,600 m, 12 of 21 (57%) women had AMS (p = 0.355 vs. 3,100 m) with SpO2 of 86.8 ± 1.8% (p < 0.05 vs. 3,100 m) and LLS of 1.9 ± 1.4 (p < 0.05 vs. 3,100 m).
Conclusion: Self-monitoring female sexual hormones during high-altitude field studies with the employed kit is feasible and provides physiologically plausible trends of hormone levels over the menstrual cycle. Our data provide a valuable basis for designing further studies to evaluate AMS susceptibility in women.
To date, there is no reliable, validated, and simple tool for monitoring hormonal concentrations or reliably identifying menstrual cycle phases in women during altitude sojourns. The validation of a reliable tool is particularly important since the pathophysiology of altitude-related adverse health effects (ARAHE), particularly acute mountain sickness (AMS), and factors for individual susceptibility remain elusive (1). Importantly, it has not yet been quantitatively assessed whether women are more, equally or less prone to develop AMS than men (2). The unresolved issue might originate from underrepresentation of women in high altitude studies, lack of powered sex-specific analyses or the poor characterization of premenopausal women in regard of the menstrual cycle phase, sex hormone concentrations, in particular estrogen, luteinizing hormone and progesterone, and cycle day while staying at high altitude. Hormonal changes related to the menstrual cycle phases can alter breathing control, which may result in different altitude-related adaptations. Progesterone, which is up-regulated during the luteal phase and also during stress situations (3), is a well-known respiratory stimulant associated with elevated minute ventilation and decreased PaCO2 during the luteal phase compared to the follicular phase (4, 5). Despite these observations at low altitude, previous studies found no relationship between menstrual cycle phase or sex hormones and AMS susceptibility, however, most studies did not quantify and assess sex hormones and cycle phase at high altitude. In contrast, a small study conducted in 1997 and 1998 assessed sex hormone levels at high altitude and found a significant difference of 15% in AMS incidence between menstrual cycle phases, though it concluded that the difference was not clinically relevant (6). Another study found sexual hormone changes in lowland women going to high altitudes but have not analyzed them in regard to AMS (7). Based on these previous reports and technological advancements, the primary purpose of this prospective pilot study was to evaluate the feasibility, functionality, and tolerability of using commercially available fertility self-monitoring kits to measure urinary sex hormone concentrations and to determine the menstrual cycle phases and cycle days when travelling to high altitude. Furthermore, to facilitate sample size estimations and provide preliminary insights into potential differences in clinical and physiological outcomes between the luteal and follicular menstrual cycle phases, additional measures—such as AMS, nocturnal pulse oximetry, subjective sleep quality and vital signs—were assessed.
Therefore, this pilot study examined the hypotheses that commercially available self-monitor fertility devices are suitable for urinary hormone assessments at high altitudes and that the resulting hormone profiles allow an accurate determination of menstrual cycle phases and cycle days in females staying at high altitudes.
This prospective, non-randomized pilot study was conducted at the National Center of Cardiology and Internal Medicine, Respiratory Medicine Department, Bishkek (760 m), Kyrgyzstan; at the Swiss-Kyrgyz High-Altitude Clinic in Tuja Ashu (3,100 m), Kyrgyzstan, which is a 4 h bus ride away from the first location and where participants spent 2 days and nights (48 h); and at the High Altitude Kumtor Gold Mine operation facility (3,600 m), Kumtor, Kyrgyzstan, 8 h by bus, where participants also spent 2 days and nights (48 h). The ascent to 3,100 m (Expedition 1, n = 21) was performed in July 2022; the ascent to 3,600 m (Expedition 2, n = 21) in September 2022 and both altitude ascents were scheduled independently of the menstrual cycle phases or cycle days of the participants. Sixteen participants from Expedition 1 participated also in Expedition 2 and 5 new participants had to be recruited (Figure 1).
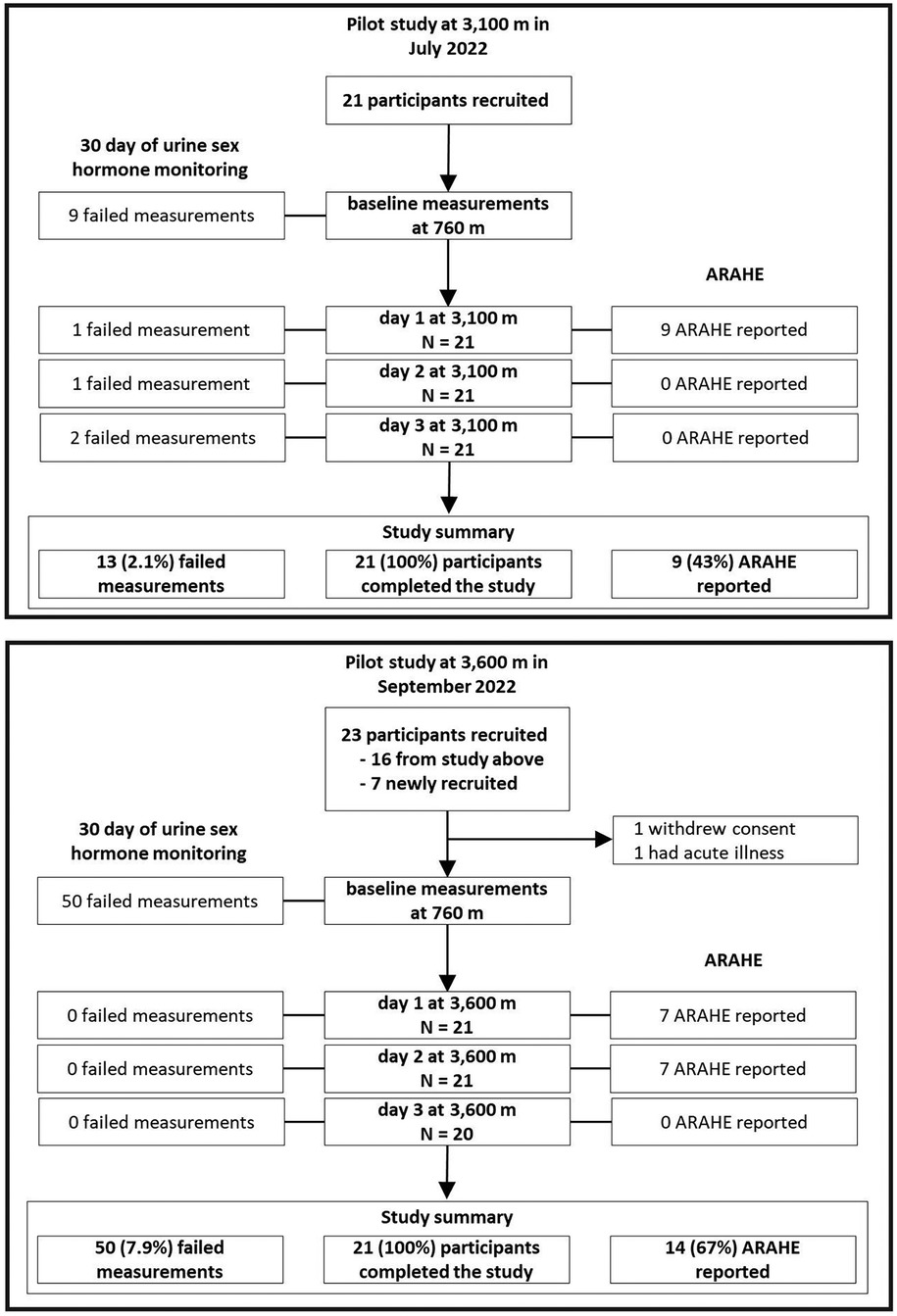
Figure 1. Study design of the pilot study. A pilot study validating the commercially available fertility monitor Proov to monitor hormones and menstrual cycle phases before, during and after an altitude sojourn. Women monitored their hormone for 30 consecutive days. During the hormone monitoring they ascended and stayed for 2-day and nights (48 h) at either 3,100 (Expedition 1) or 3,600 m (Expedition 2), respectively.
Participants in this study were recruited through advertisements at different universities around Bishkek. Included were premenopausal, naturally cycling, eumenorrheic, non-smoking, healthy women with a BMI >18 kg/m2 and <30 kg/m2, aged 18–44 years, living at altitudes under 1,000 m. Exclusion criteria were any pre-existing disease, regular intake of medication (including oral contraception), other types of hormonal contraceptives (hormonal intrauterine device, vaginal ring, subcutaneous injections or implants), pregnancy or nursing, and any altitude trip <4 weeks prior to the study. The study was approved by the Ethic Committee of the National Center of Cardiology and Internal Medicine named after academician M. Mirrakhimov (Protocol No. 07 in the year of 2022) and participants provided written informed consent.
For 30 consecutive days, beginning on the day of baseline measurements at 760 m, all participants monitored their urine metabolite concentrations of estrogen, luteinizing hormone, and progesterone using their first urine sample of the day, collected before any food or fluid intake. The hormone assessments were performed using two different sex hormone monitoring devices, described in detail below.
This device was used during both Expedition 1 & 2. The procedure requires a hormone test strip into a fresh morning urine sample for 10 –20 s, collected before any food or fluid intake. The Proov multi-hormone test strip lateral flow assay uses gold nanoparticles and buffered sample pads to adjust for pH and hydration levels, filter unwanted particulates and bind contaminants in urine that may interfere with test accuracy. The strip contains three test lines and one control line, corresponding to estrone-1-glucuronide (E1G), pregnanediol-3-alpha-glucuronide (PdG), and luteinizing hormone (LH) (beta subunit). These urinary metabolites can be used to determine the follicular phase, ovulation, and the luteal menstrual cycle phase in naturally cycling women (8). After 10 min, the participant takes a photo of the urine test strip with the smartphone and uploads it onto the company server using the Proov app. The application server employs machine learning algorithms specifically tailored to analyze photographed images of the test strip, accounting for variations in camera, lighting, and operating system, while also verifying input and output regularities and mathematically deriving the associated hormone levels (9). In the case of internet connection loss, the company can conduct offline analysis if the time, day and picture of the test strip of the participant are provided. In this study, daily concentrations of sex hormones E1G, PdG and LH were extracted from the Proov app.
This device was used during Expedition 2. Identical to the Proov testing procedure, Mira analysis requires to dip a hormone test strip for 10 –20 s into a fresh urine sample collected in the morning before any fluid or food intake. Thereafter, the strip will be inserted into the Mira device to initiate the quantitative analysis of estrogen-3-glucuronide (E3G), LH and PdG. After approximately 21 min the results will be synchronized with the Mira smartphone app through Bluetooth. In case of an analysis error, the strip cannot be re-analyzed.
Together with the information regarding the first day of the participant's last menstrual bleeding, the measured sex hormones were visually plotted. Based on the visually confirmed LH peak and increment of PdG, the follicular (first day of menstrual bleeding to LH peak) and luteal menstrual cycle phases (LH peak to end of cycle) were identified. To ensure a high-quality hormone data collection, all participants attended a one-time instructional session on the proper use of the hormone monitoring kits and the accompanying app. The apps were installed and setup under the guidance of the investigators. The following morning, all participants performed their first hormone assessment in the hospital, allowing them to discuss questions related to the procedure. Thereafter, daily reminders regarding hormone monitoring were sent to the participants. Additionally, they were encouraged to submit their hormone values each day or, if no values were available, to provide a reason for the missing data.
A medical history has been obtained, including information on menstrual cycle length and the starting and ending days of the participants' last menstruation. Physical examination included measurements of weight, height, blood pressure, heart rate, and cardiac and pulmonary auscultation. At altitudes, adverse events and AMS incidences were assessed, using both the revised and original Lake Louise questionnaires (LLQ), as well as the Environmental Symptom Questionnaire cerebral score (AMSc) (10, 11). Primary definition of AMS incidence was a 2018 LLQ score of ≥3 points including headache, over the two-day period at high altitude; secondary AMS definitions included a 1993 LLQ score of ≥3 points and an AMSc of ≥0.7. Since previous studies in both sick and healthy persons traveling to high altitudes reported the occurrence of non-AMS related ARAHE (12), this pilot study additionally assessed the incidence of ARAHE at high altitudes. ARAHE was defined as any medical health condition (including AMS) that occurred at high altitude and required medical intervention or premature descent.
Nocturnal pulse oximetry was performed at 760 m and at 3,600 m to assess nocturnal oxygenation. Sleepiness was assessed with the Karolinska Sleepiness Scale and subjective sleep quality with a 100-mm visual analogue scale ranging from 0 (extremely poor) to 100 (excellent) (13).
The main outcomes included the proportion and comparison of successful applications of the Proov and Mira hormone test kits, their failure rates, and reasons for failure. Additional outcomes included daily hormone concentrations throughout the menstrual cycle, differences in estrogen and progesterone concentrations between the follicular and luteal menstrual cycle phases, and the timing of the LH peak, defined as the day with the highest median value of 3 consecutive daily concentrations. The incidence of AMS and other ARAHE, nocturnal pulse oximetry, and subjective sleep quality as well as physiological variables in the different menstrual cycle phases were determined.
No sample size estimation was performed for this pilot study. A convenience sample of 20 participants per expedition to 3,100 m and 3,600 m were recruited. The feasibility of hormone monitoring at low and high altitudes, as well as during experimental field expeditions, was quantified using descriptive statistics on successful instances of hormone monitoring, hormone concentrations, and failure rates associated with the use of the Proov or Mira devices, respectively. Based on the findings of the two devices, menstrual cycle phases, ovulation day, and failure rates were compared using Chi-square test statistics and Pearson correlation. AMS incidences and physiological variables were then compared between menstrual cycle phases and between altitudes using mixed linear regressions to compute mean differences and 95% confidence intervals. Continuous outcomes are presented as means ± SE and binary outcomes as numbers and proportions. Two exploratory regression models were used to evaluate (1) the associations between sex hormone concentrations and SpO2 (mixed linear regression model); (2) the associations between menstrual cycle phases and the incidence of AMS (logistic regression model). Due to the small sample size, the parameters for the mixed models were predefined. Therefore, in the model for SpO2, sex hormones (E1G, LH and PdG) and assessment altitudes (760 m, 3,100 m and 3,600 m) were used as predictors. A two-sided p-value of 0.05 was considered statistically significant. Statistical analyses were performed in STATA version 15 (StataCorp LLC, College Station, Texas, USA).
The study flow chart is presented in Figure 1. Out of 26 participants with a mean ± SD age of 23.5 ± 2.5 years, a total of 21 (80.7%) went to 3,100 m; 21 (80.7%) went to 3,600 m and 16 (61.5%) participants completed both expeditions (Table 1). Based on the assessed hormone concentrations, 52.4% of participants were in their follicular, 40.5% in their luteal and 7.1% unknown menstrual cycle phase when staying at high altitudes. The menstrual cycle length assessed with the Proov app was 29 ± 2 days and almost identical with what participants reported during the screening visit (30 ± 2 days). The maximum deviation between the assessed and reported menstrual cycle length was 9 days.
When using the Proov hormone test kit, the daily urinary hormone measurement error rates were 4.5% (95% CI of 3.0–6.5%) in the group travelling to 3,100 m, 7.9% (95% CI of 5.9–10.3%) in those travelling to 3,600 m, and 6.2% overall (95% CI of 5.0–7.7%) (Table 1). At 3,600 m, the corresponding error rate when using the Mira hormone test kit was 11.4% (95% CI of 9.8–15.0%, P < 0.001 between error rates of Proov vs. Mira at 3,600 m). The urinary hormone monitoring and the assignment of the participants to a specific menstrual cycle phase and cycle day while staying at altitude were successful and are illustrated in Figure 2, Upper Panel. Figure 2, Lower Panel shows the proportions of participants suffering from AMS during the follicular compared to the luteal phase, however, exploratory mixed logistic regression analysis showed no association between menstrual cycle phases and the incidence of AMS defined by the 2018 LLQ criteria (Odds ratio of 0.50, 95% CI 0.09–2.8, p = 0.430). Table 2 shows the comparison of the menstrual cycle phases and the mean differences (95% CI) in other physiological and clinical outcomes between the luteal and follicular phases. Both progesterone and estrogen were higher during the luteal compared to the follicular phase. During the nights at 3,600 m, participants in the luteal phase showed a trend toward faster SpO2 acclimatization between nights 1 and 2, with a mean difference (95% CI) of 2.1% (−0.3 to 4.6, p = 0.085), compared to participants in the follicular phase (Table 3). These findings align with the results from mixed linear regression analysis on daytime SpO2, showing a positive correlation with PdG concentration and a negative correlation with E1G and absolute altitude of measurement (Supplement Table S2).
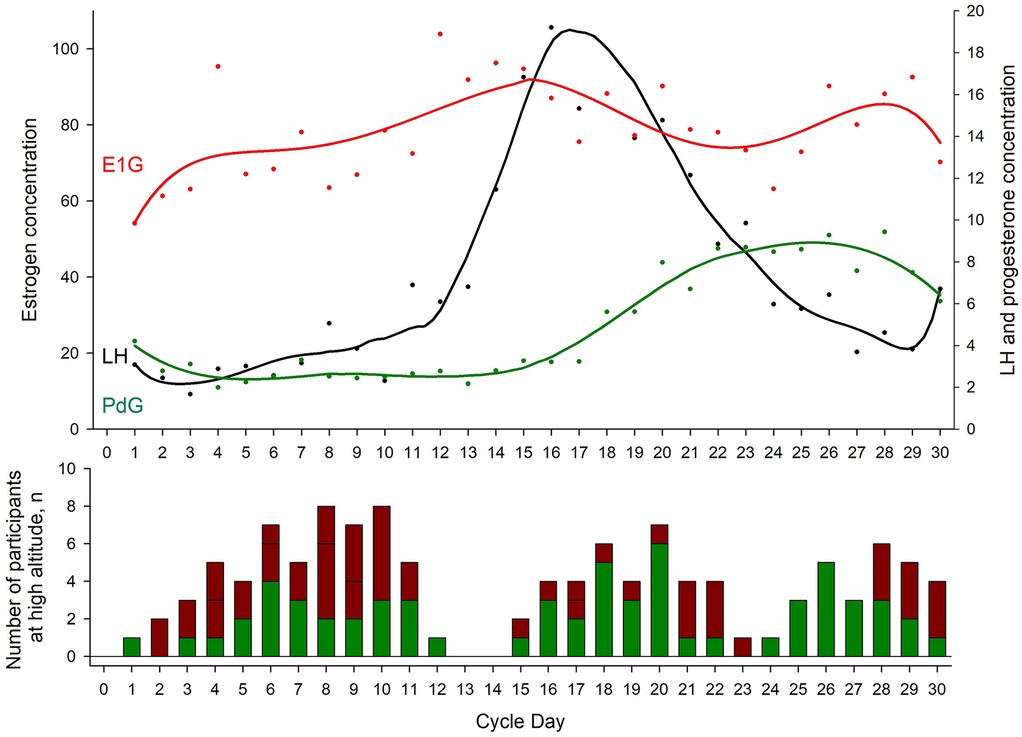
Figure 2. Hormone profile and acute mountain sickness incidence. Upper panel shows the mean hormone concentrations assessed by Proov for each menstrual cycle in coloured dots. The connecting line represents a smoother of the corresponding hormone. This figure indicates a commonly seen hormone profile of oestrogen (E1G), progesterone (PdG) and luteinizing hormone (LH) and confirms the feasibility of assessing hormones by urine stripes. The follicular phase was defined as the time from the first day of menstrual bleeding to the LH peak (Cycle Day 16 in the figure), while the luteal phase was defined as the time from the LH peak to the day before the next menstrual bleeding. Lower panel shows the absolute number of participants staying at altitude, stratified by menstrual cycle days. The green coloured proportion of the bars represent the number of participants without acute mountain sickness (acute mountain sickness was defined as a 2018 Lake Louise questionnaire score of ≥3 points including headache.); the red proportion of the bars represent the number of participants with, or the days after the diagnosis of acute mountain sickness. For example, when a participant experienced acute mountain sickness on day 2 at 3,600 m, this person was medically treated and, if feasible, descended to low altitude on day 3, however, day 2 and 3 were marked as AMS-positive in this figure.
During the altitude sojourns, 9 of 21 (43%) participants at 3,100 m and 12 of 21 (57%) at 3,600 m developed AMS (Table 4, p = 0.355 between altitudes). At 3,600 m, 24% of participants experienced other ARAHEs not related to AMS, including fainting (N = 1) and fever (N = 1) on the day of arrival; panic attack (N = 1) during the first night and abdominal pain (N = 1) and diarrhea (N = 1) over the time course of the 2 days and nights (Table 4). As shown in Figure 3, a high proportion of participants who experienced an ARAHE or AMS at 3,100 m also experienced one at 3,600 m.
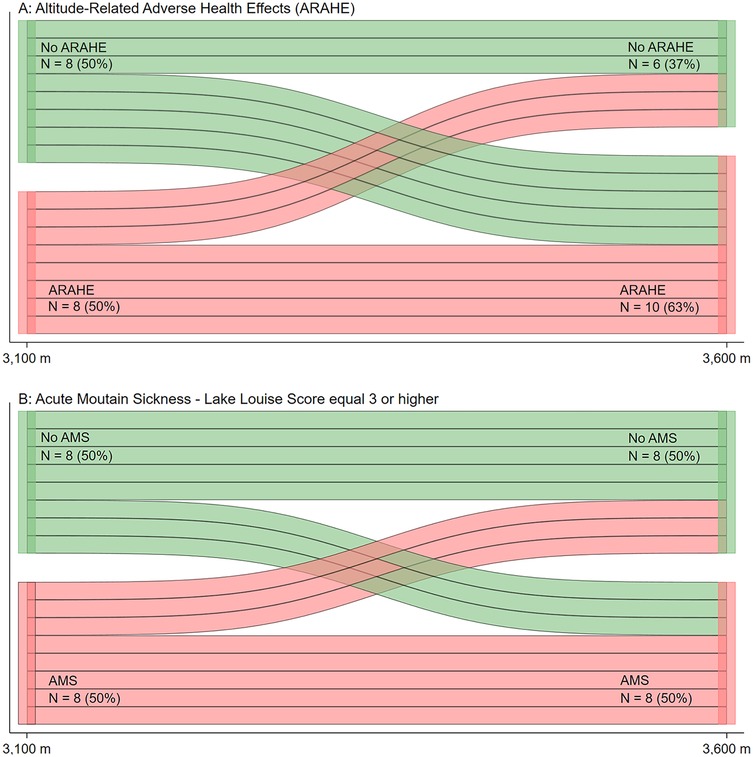
Figure 3. Incidences of acute mountain sickness (AMS) and altitude-related adverse health effects (ARAHE) in participants travelling to 3,100 and 3,600 m (N = 16). The data from 16 participants who completed both expeditions were analyzed. Green bars indicate participants free of AMS (B) or ARAHE (A) while red bars indicate participants suffering from AMS or ARAHE at the respective altitude. Acute mountain sickness was defined as a 2018 Lake Louise questionnaire score of ≥3 points including headache.
On the morning of day 2 at 3,100 m, no significant differences were observed in vital parameters nor subjective sleep quality compared to 760 m, except for an expected decrease in SpO2 (Table 5). In contrast, at 3,600 m, a significant decrease in SpO2 and subjective sleep quality as well as a significant increase in diastolic blood pressure, heart rate and AMS severity were observed (Table 5). At 3,600 m, nocturnal SpO2 dropped and oxygen desaturation index increased during the first night compared to 760 m, but significantly improved during the second night (Supplement Table S1).
This pilot study confirms that daily urinary sex hormone concentration monitoring using the commercially available fertility apps Proov and Mira is feasible for several consecutive days at high altitudes of up to 3,600 m, with the Proov device associated with lower error rates compared to Mira. Nevertheless, both hormone testing kits provide physiologically plausible urinary sex hormone concentrations to determine menstrual cycle phases and cycle days before, during, and after a high-altitude sojourn and can be considered as tools for future studies in premenopausal women evaluating susceptibility to AMS and its dependence on menstrual cycle phases. An important finding was that during the 2-day sojourns at high altitudes, the incidence of AMS was 43% at 3,100 m and 57% at 3,600 m. Moreover, other ARAHEs in the absence of AMS were observed at 3,600 m, encompassing fainting, fever, panic attacks, abdominal pain and diarrhoea. This underscores the diverse array of hypoxia-related, clinically relevant conditions in women that have rarely been reported.
The comparably high incidence of AMS observed in our study is confirmed by generally accepted data. In a study by Li et al., during a two-day stay at an altitude of 3,270 m, women had a higher incidence of AMS than men (50% vs. 10%, p < 0.05). Furthermore, multivariable regression analysis showed that not only the incidence, but also the severity of AMS, was significantly higher in women than in men (14). In contrast, the 2023 UIAA statement on AMS in women suggests that there is no sex difference in AMS susceptibility; however, this statement has not been quantitatively confirmed by any meta-analysis examining the influence of sex on AMS. This situation, together with the revised LLS definition of AMS in 2018 and the lack of any recent prospective studies on AMS in women and men, does not allow evidence-based conclusions on sex-differences in AMS susceptibility.
Underlying sex-differences potentially contributing to the higher AMS susceptibility in women could be related to the lower hypoxic ventilatory response compared to men. In a study by Camacho-Cardenosa et al. (15), it was shown that, during the first hours of hypoxia, SpO2 decreased more markedly in women than in men, probably due to an initially lower and/or less efficient respiratory response to moderate hypoxia. In addition, there is evidence that women experience headaches more frequently at low altitudes (16), indicating a potential susceptibility to AMS-like symptoms even before climbing to high altitude. This and other pre-existing conditions may contribute to the development of AMS (17, 18).
Adding even further variability, studies have reported differences in ventilation rates throughout the menstrual cycle (19–21). In particular, PdG, acting on both peripheral and central chemoreceptors, is thought to be a potent respiratory stimulant (22) enhancing minute ventilation during the mid-luteal phase compared to the follicular menstrual cycle phase (23, 24). Our exploratory analysis showed the same correlation, indicating that higher PdG concentrations are associated with higher daytime SpO2 values (Supplement Table S2), independent of location. In contrast, a recent study by Gardner et al. has not confirmed this relationship between PdG and AMS (25).
Nevertheless, several previous studies and recommendations concluded that there are no sex-related differences in AMS susceptibility (2). The controversies will remain until we obtain robust findings from prospective studies specifically designed and powered to investigate and detect sex-related differences in AMS (defined by the revised LLS). With the emergence of wearables and modern technologies, this challenge became solvable, as demonstrated in our study that monitored hormones and determined menstrual cycle phases and cycle days of women staying at high altitudes. Our findings in premenopausal women are promising and important since they highlight the high adherence of participants monitoring urine hormones with Proov.
This study was designed as a pilot study to test the feasibility of monitoring female sex hormones at high altitudes. Therefore, no sample size estimation related to AMS incidence differences between menstrual cycle phases and altitudes were performed. Moreover, the lack of a male control group limits the interpretability of AMS found in this pilot study. We applied a 30-day hormone-monitoring regimen; however fewer consecutive days of urine hormone monitoring might be sufficient to determine the menstrual cycle phases in participants staying at altitude. Despite the long duration of hormone monitoring, the observed error rate remained minimal, and adherence of the participants high, suggesting that this approach is feasible in future studies. Absence of internet access may hamper immediate analysis of urinary hormone levels with the Proov kit. However, by being aware of such conditions, taking a photograph of the test strip noting the date and time allows for offline analysis at a convenient later time, which significantly reduces the risk of data loss.
This pilot study in healthy, premenopausal women demonstrated good adherence and feasibility in monitoring female sexual hormones using the commercially available fertility monitoring kits and apps Proov and Mira during a sojourn at high altitudes. Therefore, future studies can apply this sexual hormone monitoring technique to enable in-depth analyses of the influence of sex, menstrual cycle phase, and hormone-related differences on AMS susceptibility. These insights allow better interpretability and comparability of AMS-related findings across studies and regions worldwide. In this pilot study, premenopausal women showed a high AMS incidence at both 3,100 m and 3,600 m, and these AMS incidences allow for evidence-based sample size estimations for future studies investigating sex-related differences in AMS incidence and the efficacy of preventive AMS measures, while monitoring and controlling for potential confounders, such as sex hormones and menstrual cycle phases. For example, in a randomized, placebo-controlled parallel trial at 3,600 m investigating whether acetazolamide can reduce the incidence of AMS from 57% by 50% to 28.5%, a total of 94 women (48 per group), taking either acetazolamide or placebo, would be required for the final analysis, with 80% power and a two-sided alpha level of 5%.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving humans were approved by Ethic Committee of the National Center of Cardiology and Internal Medicine named after academician M. Mirrakhimov (Protocol No. 07 in the year of 2022). The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
AT: Conceptualization, Data curation, Investigation, Methodology, Resources, Visualization, Writing – original draft, Writing – review & editing. MM: Conceptualization, Data curation, Formal analysis, Resources, Visualization, Writing – original draft, Writing – review & editing. SU: Conceptualization, Data curation, Investigation, Methodology, Validation, Writing – original draft, Writing – review & editing. GM: Data curation, Investigation, Methodology, Writing – original draft, Writing – review & editing. MM: Data curation, Investigation, Writing – original draft, Writing – review & editing. ML: Conceptualization, Validation, Writing – original draft, Writing – review & editing. CB: Conceptualization, Data curation, Methodology, Validation, Writing – original draft, Writing – review & editing. TS: Conceptualization, Investigation, Methodology, Supervision, Writing – original draft, Writing – review & editing. SU: Writing – review & editing, Conceptualization, Data curation, Methodology, Supervision, Validation, Writing – original draft. KB: Funding acquisition, Methodology, Supervision, Validation, Writing – original draft, Writing – review & editing. MF: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This study was partly funded by the Swiss National Sciences Foundation (210361).
We gratefully acknowledge all participants for taking part in this study.
The evaluation of the Proov Full Cycle Hormone Insights Kit and the Mira Fertility Tracking Kit was conducted solely for research purposes to assess a potential method for monitoring sex hormones at altitude.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fgwh.2025.1544832/full#supplementary-material
AMS, acute mountain sickness; E1G, etrone-1-glucuronide; PdG, pregnanediol-3-alpha-glucuronide; LH, luteinizing hormone; ARAHE, altitude-related adverse health effects; LLS, Lake Louise score; SpO2, oxygen saturation; PaCO2, partial pressure of carbon dioxide; BMI, body mass index; LLQ, Lake Louise questionnaires; UIAA, International Climbing and Mountaineering Federation.
1. Imray C, Wright A, Subudhi A, Roach R. Acute mountain sickness: pathophysiology, prevention, and treatment. Prog Cardiovasc Dis. (2010) 52(6):467–84. doi: 10.1016/j.pcad.2010.02.003
2. Derstine M, Jean D, Beidleman BA, Pichler Hefti J, Hillebrandt D, Horakova L, et al. Acute mountain sickness and high altitude cerebral edema in women: a scoping review—UIAA medical commission recommendations. High Alt Med Biol. (2023) 24(4):259–67. doi: 10.1089/ham.2023.0043
3. Herrera AY, Nielsen SE, Mather M. Stress-induced increases in progesterone and cortisol in naturally cycling women. Neurobiol Stress. (2016) 3:96–104. doi: 10.1016/j.ynstr.2016.02.006
4. Behan M, Wenninger JM. Sex steroidal hormones and respiratory control. Respir Physiol Neurobiol. (2008) 164(1–2):213–21. doi: 10.1016/j.resp.2008.06.006
5. Richalet JP, Lhuissier F, Jean D. Ventilatory response to hypoxia and tolerance to high altitude in women: influence of menstrual cycle, oral contraception, and menopause. High Alt Med Biol. (2020) 21(1):12–9. doi: 10.1089/ham.2019.0063
6. El-Migdadi F, Nusier M, Bashir N. Seasonal pattern of leutinizing, follicle-stimulating hormone, testosterone and progesterone in adult population of both sexes in the Jordan valley. Endocr Res. (2000) 26(1):41–8. doi: 10.1080/07435800009040144
7. Verratti V, Ietta F, Paulesu L, Romagnoli R, Ceccarelli I, Doria C, et al. Physiological effects of high-altitude trekking on gonadal, thyroid hormones and macrophage migration inhibitory factor (MIF) responses in young lowlander women. Physiol Rep. (2017) 5(20):e13400. doi: 10.14814/phy2.13400
8. D’Souza AC, Wageh M, Williams JS, Colenso-Semple LM, McCarthy DG, McKay AKA, et al. Menstrual cycle hormones and oral contraceptives: a multimethod systems physiology-based review of their impact on key aspects of female physiology. J Appl Physiol (1985). (2023) 135(6):1284–99. doi: 10.1152/japplphysiol.00346.2023
9. Wegrzynowicz AK, Beckley A, Eyvazzadeh A, Levy G, Park J, Klein J. Complete cycle mapping using a quantitative at-home hormone monitoring system in prediction of Fertile days, confirmation of ovulation, and screening for ovulation issues preventing conception. Medicina (Kaunas). (2022) 58(12):1853. doi: 10.3390/medicina58121853
10. Roach RC, Hackett PH, Oelz O, Bärtsch P, Luks AM, MacInnis MJ, et al. The 2018 lake louise acute mountain sickness score. High Alt Med Biol. (2018) 19(1):4–6. doi: 10.1089/ham.2017.0164
11. Sampson JB, Cymerman A, Burse RL, Maher JT, Rock PB. Procedures for the measurement of acute mountain sickness. Aviat Space Environ Med. (1983) 54(12 Pt 1):1063–73.6661120
12. Furian M, Mademilov M, Buergin A, Scheiwiller PM, Mayer L, Schneider S, et al. Acetazolamide To prevent adverse altitude effects in COPD and healthy adults. NEJM Evidence. (2022) 1(1):EVIDoa2100006. doi: 10.1056/EVIDoa2100006
13. Kaida K, Takahashi M, Åkerstedt T, Nakata A, Otsuka Y, Haratani T, et al. Validation of the karolinska sleepiness scale against performance and EEG variables. Clin Neurophysiol. (2006) 117(7):1574–81. doi: 10.1016/j.clinph.2006.03.011
14. Li T, Tan L, Furian M, Zhang Y, Luo L, Lei F, et al. Sex-specific difference in the effect of altitude on sleep and nocturnal breathing in young healthy volunteers. J Clin Med. (2022) 11(10):2869. doi: 10.3390/jcm11102869
15. Camacho-Cardenosa A, Camacho-Cardenosa M, Tomas-Carus P, Timón R, Olcina G, Burtscher M. Acute physiological response to a normobaric hypoxic exposure: sex differences. Int J Biometeorol. (2022) 66(7):1495–504. doi: 10.1007/s00484-022-02298-y
16. Li XY, Yang CH, Lv JJ, Liu H, Zhang LY, Yin MY, et al. Global, regional, and national epidemiology of migraine and tension-type headache in youths and young adults aged 15–39 years from 1990 to 2019: findings from the global burden of disease study 2019. J Headache Pain. (2023) 24(1):126. doi: 10.1186/s10194-023-01659-1
17. MacGregor EA, Frith A, Ellis J, Aspinall L, Hackshaw A. Incidence of migraine relative to menstrual cycle phases of rising and falling estrogen. Neurology. (2006) 67(12):2154–8. doi: 10.1212/01.wnl.0000233888.18228.19
18. Alcantara-Zapata DE, Lucero N, De Gregorio N, Astudillo Cornejo P, Ibarra Villanueva C, Baltodano-Calle MJ, et al. Women’s mood at high altitude. Sexual dimorphism in hypoxic stress modulation by the tryptophan–melatonin axis. Front Physiol. (2023) 13:1099276. doi: 10.3389/fphys.2022.1099276
19. Loeppky JA, Scotto P, Charlton GC, Gates L, Icenogle M, Roach RC. Ventilation is greater in women than men, but the increase during acute altitude hypoxia is the same. Respir Physiol. (2001) 125(3):225–37. doi: 10.1016/S0034-5687(00)00221-8
20. Rael B, Alfaro-Magallanes VM, Romero-Parra N, Castro EA, Cupeiro R, de Jonge XAK J, et al. Menstrual cycle phases influence on cardiorespiratory response to exercise in endurance-trained females. Int J Environ Res Public Health. (2021) 18(3):860. doi: 10.3390/ijerph18030860
21. da Silva SB, de Sousa Ramalho Viana E, de Sousa MBC. Changes in peak expiratory flow and respiratory strength during the menstrual cycle. Respir Physiol Neurobiol. (2006) 150(2):211–9. doi: 10.1016/j.resp.2005.03.001
22. Bairam A, Uppari N, Mubayed S, Joseph V. An overview on the respiratory stimulant effects of caffeine and progesterone on response to hypoxia and apnea frequency in developing rats. Adv Exp Med Biol. (2015) 860:211–20. doi: 10.1007/978-3-319-18440-1_23
23. Bayliss DA, Millhorn DE. Central neural mechanisms of progesterone action: application to the respiratory system. J Appl Physiol. (1992) 73(2):393–404. doi: 10.1152/jappl.1992.73.2.393
24. Beidleman BA, Rock PB, Muza SR, Fulco CS, Forte VA, Cymerman A. Exercise v˙e and physical performance at altitude are not affected by menstrual cycle phase. J Appl Physiol. (1999) 86(5):1519–26. doi: 10.1152/jappl.1999.86.5.1519
Keywords: high-altitude (HA), hormone/reproduction/sexual, AMS, self-monitoring, menstrual cycle phase
Citation: Taalaibekova A, Meyer M, Ulrich S, Mirzalieva G, Mademilov M, Lichtblau M, Betschart C, Sooronbaev TM, Ulrich S, Bloch KE and Furian M (2025) Monitoring sexual hormones in women going to high altitude—a pilot study. Front. Glob. Women's Health 6:1544832. doi: 10.3389/fgwh.2025.1544832
Received: 13 December 2024; Accepted: 17 March 2025;
Published: 31 March 2025.
Edited by:
Elena Marbán-Castro, Women in Global Health Spain, SpainReviewed by:
Nazareno Paolocci, Johns Hopkins University, United StatesCopyright: © 2025 Taalaibekova, Meyer, Ulrich, Mirzalieva, Mademilov, Lichtblau, Betschart, Sooronbaev, Ulrich, Bloch and Furian. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Aijan Taalaibekova, QWlqYW5fdGFhbGFpYmVrb3ZhQGJrLnJ1
†These authors share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.