
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Genet. , 05 March 2025
Sec. Neurogenomics
Volume 16 - 2025 | https://doi.org/10.3389/fgene.2025.1551832
Background: Acute intermittent porphyria (AIP) is a rare metabolic disorder resulting from defects in the heme biosynthesis pathway, often presenting with non-specific symptoms such as abdominal pain, seizures, and neuropsychiatric disturbances. Diagnosis is challenging due to the overlap of symptoms with other conditions, and early recognition is critical for effective treatment.
Case Presentation: A 24-year-old female presented with a 6-day history of persistent lower abdominal pain and generalized tonic-clonic seizures, following the consumption of seafood. Neuroimaging revealed white matter hyperintensities, and urine analysis showed dark red discoloration, suggestive of porphyria. Genetic testing confirmed a novel c.499-1_514del mutation in the HMBS gene, diagnosing AIP. The patient was treated with intravenous glucose, heme arginate, and anticonvulsants. Symptom resolution was noted within days, and follow-up MRI showed significant improvement.
Conclusion: This case underscores the importance of early diagnosis and management in AIP. Genetic testing plays a crucial role in confirming the diagnosis, especially in atypical cases. Timely intervention with glucose and heme arginate, combined with supportive care, led to rapid symptom resolution, reinforcing the reversibility of AIP-associated neuroimaging changes. Clinicians should maintain a high index of suspicion for AIP in patients with unexplained abdominal and neurological symptoms to prevent long-term complications.
Acute intermittent porphyria (AIP) is the most common of the acute porphyrias and part of a group of metabolic disorders caused by defects in the heme biosynthesis pathway. AIP is specifically associated with mutations in the HMBS gene (hydroxymethylbilane synthase), leading to reduced enzyme activity and the accumulation of toxic precursors, primarily delta-aminolevulinic acid (ALA) and porphobilinogen (PBG) (Souza et al., 2021). The incidence of AIP varies globally, estimated at 1 in 20,000 in certain populations but remains underdiagnosed due to its non-specific presentation (Lefever et al., 2022; Muschalek et al., 2022). The pathophysiology of AIP involves a cascade of biochemical disturbances triggered by various factors, including medications, hormonal changes, dietary triggers, and stress (Phillips, 2019; Spiritos et al., 2019).
Clinical manifestations of AIP are diverse and can include severe abdominal pain, often misattributed to gastrointestinal or gynecological causes, and neuropsychiatric symptoms ranging from seizures and peripheral neuropathy to altered mental status (Kizilaslan et al., 2023). Neurological complications can be severe, resulting in long-term deficits if not promptly recognized and treated (Ramanujam and Anderson, 2015; Balwani et al., 2024). Neuroimaging findings in AIP may show changes consistent with metabolic encephalopathy, such as white matter hyperintensities on T2-weighted and FLAIR MRI sequences. The condition is also associated with posterior reversible encephalopathy syndrome (PRES), which reflects the reversible nature of the underlying pathophysiological changes when managed effectively (Yang et al., 2020; Jaramillo-Calle et al., 2019).
This case report explores a young female patient presenting with characteristic AIP symptoms and demonstrates how timely diagnosis, supported by genetic and neuroimaging studies, can lead to successful management and outcome.
A 24-year-old female presented with a 6-day history of persistent lower abdominal pain, which began following the consumption of a seafood buffet. Two days after the onset of abdominal pain, she developed recurrent limb convulsions, followed by generalized tonic-clonic seizures. Seizures were characterized by generalized tonic-clonic movements, including limb convulsions, eye deviation, frothing at the mouth, and postictal confusion with transient memory loss.
The patient had no prior medical history of seizures, neurological disorders, or metabolic conditions. There was no known family history of porphyria. Possible triggering factors included dietary intake, stress, and hormonal changes. She denied any recent medication use, alcohol consumption, or toxin exposure.
Upon admission, a thorough physical and neurological examination was performed. Mental status assessment revealed confusion and disorientation, particularly postictally, with mild amnesia. The patient was alert but had difficulty recalling recent events. Motor function showed no signs of hemiparesis or ataxia, but mild weakness in both upper limbs was noted after the seizures. Reflexes were normal, with no hyperreflexia or signs of pathological reflexes. Cranial nerve assessment was unremarkable, with no facial asymmetry or abnormalities in visual fields, pupillary reflexes, or extraocular movements.
Initial abdominal ultrasound and contrast-enhanced CT scans were inconclusive, revealing only a suspected right adnexal corpus luteum cyst. Given the persistence of neurological symptoms, a brain MRI was subsequently performed for further evaluation. Imaging revealed slightly prolonged T1 and T2 signals with high signals on FLAIR sequences. The lesions were predominantly located in the bilateral cerebral hemispheres and the right cerebellar hemisphere, displaying an asymmetric distribution and primarily affecting the white matter. Diffusion-weighted imaging (DWI) showed high signal intensity, while the apparent diffusion coefficient (ADC) map indicated high values. No significant low-signal areas were noted on susceptibility-weighted imaging (SWI). Post-contrast images displayed mild to moderate enhancement, while magnetic resonance spectroscopy (MRS), arterial spin labeling (ASL), magnetic resonance angiography (MRA), and magnetic resonance venography (MRV) all showed normal findings. Based on these imaging characteristics (Figure 1) and neurological symptoms, initial considerations included encephalitis, demyelinating processes, or metabolic/toxic encephalopathy.
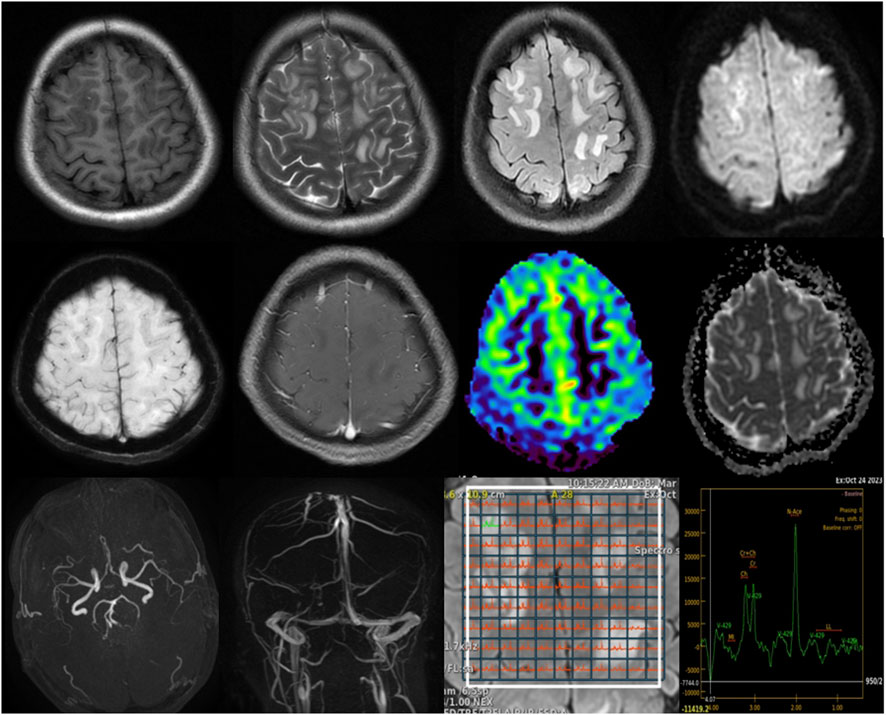
Figure 1. Brain MRI findings: T1-weighted images show slightly hypointense to isointense lesions, while T2-weighted and FLAIR sequences reveal hyperintense lesions predominantly involving the bilateral cerebral white matter with asymmetric distribution. DWI demonstrates high signal intensity, with corresponding high values on the apparent diffusion coefficient ADC map, indicating vasogenic edema. SWI shows no significant susceptibility artifacts. Post-contrast images reveal mild to moderate enhancement. MRS, MRA, and MRV findings are unremarkable, as shown.
Laboratory investigations revealed elevated creatine kinase (CK) levels at 14,538 U/L (normal range: 24–195 U/L), gamma-glutamyl transferase (GGT) at 370 U/L (normal range: 9–48 U/L), suggesting significant muscle injury and potential liver or biliary stress, respectively. Sodium was low at 130 mmol/L (normal range: 135–145 mmol/L), while potassium, calcium, magnesium, and phosphorus were within normal ranges, supporting the overall metabolic stability aside from mild hyponatremia. Additionally, inflammatory markers such as C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) were within normal ranges, suggesting the absence of a significant inflammatory response. Renal function was assessed, revealing a serum creatinine level of 71.3 μmol/L (normal range: 50–100 μmol/L) and a blood urea nitrogen level of 5.22 mmol/L (normal range: 2.5–7.1 mmol/L), both of which indicate stable kidney function without signs of impairment. Uric acid was slightly elevated at 419 μmol/L (normal range: 120–400 μmol/L), which may suggest a metabolic component, but does not directly indicate renal impairment. Urine analysis showed notable findings: urobilinogen 2+, while urine occult blood, glucose, ketones, bilirubin, and white blood cells were all negative. The presence of urobilinogen at this level may suggest mild liver involvement or hemolysis. Due to these findings, a multidisciplinary team discussion was held to consider differential diagnoses. Rhabdomyolysis was considered as a potential cause given the elevated CK levels. However, AIP could not be ruled out. Consequently, it was recommended to conduct specialized genetic testing for porphyria, which was sent out to an external laboratory for confirmation.
To explore the potential for AIP, urine analysis was performed, revealing a notable color change from light yellow to dark red upon standing for six hours—an indication of porphyrin precursor accumulation (Figure 2). However, levels of ALA and PBG in the urine were not measured in this case, as this test was not performed during the patient’s initial workup. Genetic testing was conducted by a certified external laboratory using next-generation sequencing (NGS), which identified the c.499-1_514del mutation in the HMBS gene. This finding, documented in the official diagnostic report, confirmed the diagnosis of AIP. Follow-up MRI 2 weeks post-treatment showed significant resolution of these hyperintensities, suggesting the reversible nature of AIP-associated cerebral changes (Figure 3).

Figure 2. Urine color changes in AIP: (A) Freshly collected urine sample, showing a typical yellow color. (B) Same urine sample after standing for 6 h, showing a characteristic dark red color due to porphyrin precursor accumulation.
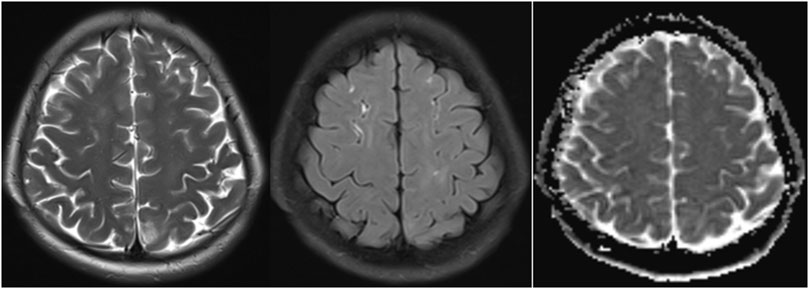
Figure 3. MRI findings after 2 weeks of treatment in AIP: Significant reduction in hyperintense lesions on T2-weighted, FLAIR sequences and ADC map.
The patient was treated with intravenous glucose and heme arginate to suppress hepatic ALA synthesis. Intravenous glucose was administered as a 10% dextrose infusion at a rate of 200–500 mL/h, titrated based on blood glucose levels to maintain euglycemia. Heme arginate was administered at a dose of 3 mg/kg body weight IV once daily for 4 consecutive days, diluted in 100 mL of 0.9% normal saline and infused over 30 min. Levetiracetam was administered as an anticonvulsant at a dose of 500 mg to 1,000 mg IV twice daily, adjusted based on the patient’s response and renal function.
In addition to the specific treatments, supportive treatment included correction of hyponatremia with intravenous sodium chloride solution (normal saline), as well as monitoring and adjusting electrolytes (potassium, calcium, and magnesium) to maintain metabolic stability. Fluid management was provided to ensure adequate hydration, and pain management was addressed with analgesics as necessary. The patient’s symptoms gradually improved, with limb seizures resolving within 2 days and abdominal pain significantly decreasing by day five. No further generalized seizures occurred, and the patient was discharged in stable condition. Follow-up MRI confirmed the near-complete resolution of the previously noted abnormalities, reinforcing the effectiveness of early intervention in reversing AIP-associated neuroimaging changes.
AIP is a rare metabolic disorder resulting from defects in the heme biosynthesis pathway, primarily due to mutations in the HMBS gene (Li et al., 2023; Bustad et al., 2021). This case report highlights several distinctive features, most notably the novel pathogenic mutation identified in the HMBS gene (c.499-1_514del variant). This mutation has not been previously reported in the literature, marking an important contribution to the genetic spectrum of AIP. The c.499-1_514del mutation is likely to lead to the loss of function of hydroxymethylbilane synthase, an enzyme involved in heme synthesis, based on its location in the HMBS gene and the known effects of similar mutations. As a result, the accumulation of porphyrin precursors such as ALA and PBG occurs, which is likely responsible for the neurotoxic symptoms seen in this patient. This discovery not only adds a new genetic variation to the AIP gene pool but also highlights the importance of genetic testing in diagnosing this rare condition, especially in atypical cases. The identification of this novel mutation has significant implications for family screening and early diagnosis, which could improve outcomes by facilitating early intervention and management (Wang et al., 2023).
In the differential diagnosis, we systematically ruled out several potential causes for the patient’s symptoms. Initially, encephalitis was suspected given the neurological symptoms; however, the MRI findings, which predominantly involved white matter lesions, did not support this diagnosis, as encephalitis usually affects gray matter regions (Guo et al., 2022; Qiao et al., 2020). Additionally, inflammatory markers were unremarkable, further ruling out this possibility. Demyelinating conditions such as multiple sclerosis were also considered, but the asymmetric distribution of lesions was atypical for such processes (Wattjes et al., 2021; Benedict et al., 2020). Metabolic or toxic encephalopathy was another differential, but the normal metabolic panel and the imaging pattern, which showed vasogenic rather than cytotoxic edema, helped exclude this possibility (de Oliveira et al., 2019). Rhabdomyolysis was initially considered due to elevated CK levels; however, the absence of myoglobinuria, muscle pain, and renal dysfunction argued against this diagnosis (Stahl et al., 2020).
Neuroimaging in AIP typically reveals white matter hyperintensities on T2-weighted and FLAIR MRI sequences (Kumar et al., 2023), as was observed in this case. These findings are often transient and can mimic conditions like PRES. The high signal intensity seen on DWI and high ADC values further supported the diagnosis of vasogenic edema, which is a reversible condition typically associated with endothelial dysfunction and disruption of the blood-brain barrier (Ando et al., 2022; Anderson et al., 2020). The absence of restricted diffusion on DWI and the normalization of ADC values after treatment provided additional evidence of the reversible nature of the cerebral lesions. The significant improvement in the patient’s MRI findings 2 weeks post-treatment with heme arginate and glucose infusion underscores the reversibility of the neuroimaging changes seen in AIP, emphasizing the importance of early diagnosis and treatment to prevent long-term neurological sequelae.
The clinical presentation of AIP can be challenging, as it manifests with a wide range of symptoms that often overlap with other conditions. In this case, the patient presented with severe, recurrent abdominal pain and generalized tonic-clonic seizures, both of which are hallmark symptoms of AIP. These symptoms are typically triggered by the accumulation of toxic porphyrin precursors, such as ALA and PBG, which affect the central nervous system. Seizures in AIP can be precipitated by electrolyte imbalances, particularly hyponatremia, as seen in this patient. The abnormal sodium levels (130 mmol/L) in the patient were likely caused by inappropriate antidiuretic hormone secretion (Yusuf et al., 2023), which is common in AIP and exacerbates the neurological symptoms. In this case, the initial diagnosis of a right adnexal corpus luteum cyst highlighted the diagnostic challenges posed by non-specific presentations.
Laboratory investigations play a crucial role in diagnosing AIP. Elevated CK levels, as observed in this patient, indicate significant muscle involvement, which may be related to the neurological effects of AIP. Hyponatremia is another common feature, often overlooked in AIP, but it can exacerbate the neurological symptoms and contribute to the onset of seizures. Urine analysis revealed a characteristic darkening of the urine upon standing, which is a classic diagnostic feature of AIP due to the oxidation of porphyrin precursors (Storjord et al., 2023). This visual clue is crucial in raising suspicion for AIP and prompting further genetic testing. The diagnosis was ultimately confirmed through genetic analysis, which identified the novel c.499-1_514del mutation in the HMBS gene. This mutation leads to a deficiency of hydroxymethylbilane synthase, impairing the heme biosynthesis pathway and contributing to the accumulation of porphyrin precursors.
The integration of clinical, biochemical, genetic, and imaging findings is essential for accurate diagnosis and management. In this case, treatment with intravenous glucose and heme arginate effectively suppressed the production of porphyrin precursors, stabilizing the patient’s condition. The follow-up MRI confirmed the near-complete resolution of the previously noted abnormalities, reinforcing the effectiveness of early intervention in reversing AIP-associated neuroimaging changes. The use of anticonvulsants, such as levetiracetam, helped prevent further seizures, and supportive care addressed electrolyte imbalances, particularly the hyponatremia.
This case underscores the complexity of diagnosing AIP, especially when clinical symptoms overlap with other more common conditions. The discovery of a novel mutation in the HMBS gene adds to the growing body of knowledge about the genetic underpinnings of AIP and highlights the importance of genetic testing in cases with atypical presentations. The rapid improvement in neuroimaging findings following treatment underscores the importance of early recognition and intervention in preventing long-term complications. Given the rarity of AIP and the potential for reversible neurological changes, it is crucial for clinicians to consider AIP in the differential diagnosis of young adults presenting with unexplained neurovisceral symptoms.
AIP remains a challenging diagnosis due to its non-specific clinical manifestations and the overlap of symptoms with other more common conditions. This case highlights the importance of early recognition and appropriate management of AIP, particularly in young adults presenting with unexplained neurovisceral symptoms such as abdominal pain and seizures. The identification of a novel pathogenic mutation in the HMBS gene further contributes to the expanding genetic spectrum of AIP and underscores the role of genetic testing in the diagnosis of atypical cases. Timely intervention with intravenous glucose and heme arginate, along with supportive care, resulted in a rapid resolution of both neurological and visceral symptoms, demonstrating the reversible nature of AIP-associated neuroimaging changes. This case reinforces the need for clinicians to maintain a high index of suspicion for AIP in patients with recurrent abdominal pain, neurological symptoms, and unexplained imaging findings, as early treatment can prevent long-term neurological sequelae and improve patient outcomes.
The original contributions presented in the study are included in the article/supplementary material, further inquiries can be directed to the corresponding authors.
The studies involving humans were approved by the ethics committee of the Jiangxi ProvincialPeople’s Hospital. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
WD: Conceptualization, Funding acquisition, Methodology, Project administration, Writing–original draft, Writing–review and editing. BZ: Investigation, Writing–original draft. XW: Data curation, Writing–original draft. RZ: Data curation, Writing–original draft. PH: Data curation, Writing–original draft. BF: Funding acquisition, Writing–review and editing. MY: Data curation, Validation, Writing–review and editing. ZL: Conceptualization, Validation, Writing–review and editing.
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This study received funding from the National Natural Science Foundation of China (Grants No. 82160335).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
AIP, Acute intermittent porphyria; ALA, delta-aminolevulinic acid; PBG, porphobilinogen; PRES, posterior reversible encephalopathy syndrome; DWI, Diffusion-weighted imaging; ADC, apparent diffusion coefficient; SWI, susceptibility-weighted imaging; MRS, magnetic resonance spectroscopy; ASL, arterial spin labeling; MRA, magnetic resonance angiography; MRV, magnetic resonance venography; CK, creatine kinase; GGT, gamma-glutamyl transferase; CRP, C-reactive protein; ESR, erythrocyte sedimentation rate; HMBS, hydroxymethylbilane synthase.
Anderson, R. C., Patel, V., Sheikh-Bahaei, N., Liu, C. S. J., Rajamohan, A. G., Shiroishi, M. S., et al. (2020). Posterior reversible encephalopathy syndrome (PRES): pathophysiology and neuro-imaging. Front. Neurol. 11, 463. doi:10.3389/fneur.2020.00463
Ando, Y., Ono, Y., Sano, A., Fujita, N., and Ono, S. (2022). Posterior reversible encephalopathy syndrome: a review of the literature. Intern Med. 61 (2), 135–141. doi:10.2169/internalmedicine.7520-21
Balwani, M., Keel, S., Meissner, P., Sonderup, M., Stein, P., and Yasuda, M. (2024). Case-based discussion of the acute hepatic porphyrias: Updates on pathogenesis, diagnosis and management. Liver Int. 45, e15924. doi:10.1111/liv.15924
Benedict, R. H. B., Amato, M. P., DeLuca, J., and Geurts, J. J. G. (2020). Cognitive impairment in multiple sclerosis: clinical management, MRI, and therapeutic avenues. Lancet Neurol. 19 (10), 860–871. doi:10.1016/S1474-4422(20)30277-5
Bustad, H. J., Kallio, J. P., Vorland, M., Fiorentino, V., Sandberg, S., Schmitt, C., et al. (2021). Acute intermittent porphyria: an overview of therapy developments and future perspectives focusing on stabilisation of HMBS and proteostasis regulators. Int. J. Mol. Sci. 22 (2), 675. doi:10.3390/ijms22020675
de Oliveira, A. M., Paulino, M. V., Vieira, A. P. F., McKinney, A. M., da Rocha, A. J., Dos Santos, G. T., et al. (2019). Imaging patterns of toxic and metabolic brain disorders. Radiographics 39 (6), 1672–1695. doi:10.1148/rg.2019190016
Guo, Y., Lv, X., Zhang, J., Li, C., Wei, L., Zhou, N., et al. (2022). Gray matter atrophy and corresponding impairments in connectivity in patients with anti-N-methyl-D-aspartate receptor encephalitis. Brain Imaging Behav. 16 (5), 2001–2010. doi:10.1007/s11682-022-00670-5
Jaramillo-Calle, D. A., Solano, J. M., Rabinstein, A. A., and Bonkovsky, H. L. (2019). Porphyria-induced posterior reversible encephalopathy syndrome and central nervous system dysfunction. Mol. Genet. Metab. 128 (3), 242–253. doi:10.1016/j.ymgme.2019.10.011
Kizilaslan, E. Z., Ghadge, N. M., Martinez, A., Bass, M., Winayak, R., Mathew, M., et al. (2023). Acute intermittent porphyria's symptoms and management: a narrative review. Cureus 15 (3), e36058. doi:10.7759/cureus.36058
Kumar, V., Lalawmpuia, C., Lodhi, P., and Guglani, B. (2023). Acute intermittent porphyria presenting with non-convulsive status epilepticus and posterior reversible encephalopathy syndrome. Neurol. India 71 (6), 1302–1303. doi:10.4103/0028-3886.391353
Lefever, S., Peersman, N., Meersseman, W., Cassiman, D., and Vermeersch, P. (2022). Development and validation of diagnostic algorithms for the laboratory diagnosis of porphyrias. J. Inherit. Metab. Dis. 45 (6), 1151–1162. doi:10.1002/jimd.12545
Li, S., Lei, J. J., Dong, B. X., Ren, Y., and Yang, J. (2023). HMBS gene mutations and hydroxymethylbilane synthase activity in acute intermittent porphyria: a systematic review. Med. Balt. 102 (39), e35144. doi:10.1097/MD.0000000000035144
Muschalek, W., Hermasch, M. A., Poblete-Gutiérrez, P., and Frank, J. (2022). The porphyrias. J. Dtsch. Dermatol Ges. 20 (3), 316–331. doi:10.1111/ddg.14743
Phillips, J. D. (2019). Heme biosynthesis and the porphyrias. Mol. Genet. Metab. 128 (3), 164–177. doi:10.1016/j.ymgme.2019.04.008
Qiao, J., Zhao, X., Wang, S., Li, A., Wang, Z., Cao, C., et al. (2020). Functional and Structural brain Alterations in encephalitis with LGI1 Antibodies. Front. Neurosci. 14, 304. doi:10.3389/fnins.2020.00304
Ramanujam, V. S., and Anderson, K. E. (2015). Porphyria diagnostics-Part 1: a Brief Overview of the porphyrias. Curr. Protoc. Hum. Genet. 86 (17), 1–17. doi:10.1002/0471142905.hg1720s86
Souza, P. V. S., Badia, B. M. L., Farias, I. B., Gonçalves, E. A., Pinto, WBVR, and Oliveira, A. S. B. (2021). Acute hepatic porphyrias for the neurologist: current concepts and perspectives. Arq. Neuropsiquiatr. 79 (1), 68–80. doi:10.1590/0004-282X20200096
Spiritos, Z., Salvador, S., Mosquera, D., and Wilder, J. (2019). Acute intermittent porphyria: current perspectives and case presentation. Ther. Clin. Risk Manag. 15, 1443–1451. doi:10.2147/TCRM.S180161
Stahl, K., Rastelli, E., and Schoser, B. (2020). A systematic review on the definition of rhabdomyolysis. J. Neurol. 267 (4), 877–882. doi:10.1007/s00415-019-09185-4
Storjord, E., Wahlin, S., Karlsen, B. O., Hardersen, R. I., Dickey, A. K., Ludviksen, J. K., et al. (2023). Potential Biomarkers for the earlier diagnosis of kidney and liver damage in acute intermittent porphyria. Life (Basel) 14 (1), 19. doi:10.3390/life14010019
Wang, B., Bonkovsky, H. L., Lim, J. K., and Balwani, M. (2023). AGA clinical Practice Update on diagnosis and management of acute hepatic porphyrias: expert review. Gastroenterology 164 (3), 484–491. doi:10.1053/j.gastro.2022.11.034
Wattjes, M. P., Ciccarelli, O., Reich, D. S., Banwell, B., de Stefano, N., Enzinger, C., et al. (2021). 2021 MAGNIMS-CMSC-NAIMS consensus recommendations on the use of MRI in patients with multiple sclerosis. Lancet Neurol. 20 (8), 653–670. doi:10.1016/S1474-4422(21)00095-8
Yang, Y., Chen, X., Wu, H., Peng, H., Sun, W., He, B., et al. (2020). A novel heterozygous mutation in the HMBS gene in a patient with acute intermittent porphyria and posterior reversible encephalopathy syndrome. Mol. Med. Rep. 22 (1), 516–524. doi:10.3892/mmr.2020.11117
Yusuf, A., Alhaj, O., Aldaheri, A., AlShamsi, A., AlMarshoodi, M., AlKindi, F., et al. (2023). Rare coexistence of acute intermittent porphyria with systemic lupus erythematous: case report and literature review. J. Investig. Med. High. Impact Case Rep. 11, 23247096231181856. doi:10.1177/23247096231181856
Keywords: HMBS gene mutation, abdominal pain, seizures, neuroimaging, acute intermittent porphyria
Citation: Dong W, Zeng B, Wang X, Zhang R, Huang P, Fan B, Yuan M and Li Z (2025) A novel HMBS gene mutation in acute intermittent porphyria: a case report of abdominal pain, seizures, and reversible neuroimaging findings. Front. Genet. 16:1551832. doi: 10.3389/fgene.2025.1551832
Received: 14 January 2025; Accepted: 18 February 2025;
Published: 05 March 2025.
Edited by:
Charlotte L. Alston, Wellcome Trust Centre for Mitochondrial Research (WT), United KingdomReviewed by:
Paulo Victor Sgobbi Souza, Federal University of São Paulo, BrazilCopyright © 2025 Dong, Zeng, Wang, Zhang, Huang, Fan, Yuan and Li. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zicong Li, cXF3ODgyMjc3QDE2My5jb20=; Min Yuan, eXVhbm1pbjIwMTMxNEBzaW5hLmNvbQ==
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.