
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Genet. , 18 March 2025
Sec. Cancer Genetics and Oncogenomics
Volume 16 - 2025 | https://doi.org/10.3389/fgene.2025.1549602
This article is part of the Research Topic Genetic and Immunological Insights in Solid Tumors: Comprehensive Approaches to Treatment View all 5 articles
Background: Inflammatory biomarkers have shown prognostic value in Non-Small Cell Lung Cancer (NSCLC), but the inclusion of Adenocarcinoma In Situ (AIS) cases in previous studies may introduce bias. This study aims to evaluate the prognostic significance of inflammatory biomarkers in NSCLC while excluding AIS.
Methods: This study included patients who received surgery for lung carcinoma from August 2016 and August 2019. We collected demographic, clinical, laboratory, and outcome information. Inflammatory biomarkers were analyzed using receiver operating characteristic (ROC) curves, Kaplan-Meier survival analysis, and Cox regression to assess their prognostic value.
Results: Higher levels of inflammatory biomarkers correlated with poorer survival, with significant differences in overall survival (OS) between high- and low-expression groups. However, multivariate Cox regression identified age, tumor stage, and differentiation as independent prognostic factors, while biomarkers were not independently predictive.
Conclusion: Inflammatory biomarkers have short-term prognostic value in invasive NSCLC, but traditional clinical and pathological factors remain key for long-term outcomes.
Lung cancer remains one of the most common and lethal malignancies worldwide. According to global cancer statistics, approximately 1.8 million deaths from lung cancer occur annually (Leiter et al., 2023). Data from the National Cancer Center of China indicate that in 2022, 733,000 lung cancer-related deaths ranked first among cancer-related deaths in the country, surpassing the combined deaths from the second and third most common cancers (Han et al., 2024). Despite the widespread application of low-dose computed tomography (LDCT), which has improved early detection rates and increased surgical opportunities for lung cancer patients, the overall long-term survival rate of lung cancer patients remains unsatisfactory.
Inflammation plays a critical role in the tumor microenvironment (TME). Tumor-associated neutrophils secrete cytokines that promote tumor angiogenesis and growth, while tumor-associated macrophages derived from monocytes facilitate immune escape and systemic immune dysregulation. Thus, inflammation-related clinical biomarkers, such as the systemic immune-inflammation index (SII), neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), monocyte-to-lymphocyte ratio (MLR), and systemic inflammation response index (SIRI), may have prognostic value in cancer patients. Several clinical cohort studies have demonstrated the prognostic relevance of these inflammatory markers in various malignancies, including lung cancer (Templeton et al., 2014; Feng et al., 2020; Xie et al., 2023).
In China, non-small cell lung cancer (NSCLC) accounts for approximately 80% of all lung cancer cases, with adenocarcinoma and squamous cell carcinoma being the predominant histological subtypes. In recent years, the incidence of adenocarcinoma in situ (AIS), primarily characterized by ground-glass opacities (GGOs) on imaging, has increased significantly. As a malignancy without histological invasion, AIS has a highly favorable long-term prognosis (Inamura, 2018). However, many predictive models for lung cancer have not explicitly excluded AIS patients, potentially introducing confounding factors into their findings.
This study aims to explore prognostic factors for NSCLC patients after excluding AIS cases to construct a more precise inflammation-related survival prediction model. The findings may provide a reference for the rapid and convenient application of these markers in clinical practice.
This retrospective cohort study included NSCLC patients who underwent surgical treatment at Beijing Chaoyang Hospital between August 2016 and August 2019. Patients with in situ lung cancer were excluded to ensure a homogeneous study population. The final cohort consisted of 222 patients with complete clinical, pathological, and survival data. The selection of the study population is represented by a flowchart (Figure 1).
We collected the following data using an electronic medical record system: patient baseline information (age, gender, smoking history, and family history, etc.), laboratory test results (complete blood count, etc.), surgical-related data (surgery time, intraoperative bleeding volume, etc.) and pathological results (pathological type, degree of differentiation, etc.). All patients who were enrolled had venous blood samples collected within 24 h of admission and underwent a complete blood count analysis. All hematology was collected prior to the patient’s surgery. In this study, inflammatory biomarkers (MLR, PLR, NLR, SII, and SIRI) were derived from complete blood count (CBC) tests. All blood samples were collected within 24 h of patient admission. The samples were analyzed using an automated hematology analyzer. The biomarkers were calculated as follows: MLR (Monocyte-to-Lymphocyte Ratio) was calculated by dividing the monocyte count by the lymphocyte count; NLR (Neutrophil-to-Lymphocyte Ratio) was calculated by dividing the neutrophil count by the lymphocyte count; PLR (Platelet-to-Lymphocyte Ratio) was calculated by dividing the platelet count by the lymphocyte count; SII (Systemic Immune-Inflammation Index) was calculated by multiplying the neutrophil count by the platelet count and dividing by the lymphocyte count; SIRI (Systemic Inflammation Response Index) was calculated by multiplying the monocyte count by the neutrophil count and dividing by the lymphocyte count.
Continuous variables were compared between groups by either the Student’s t-test or the Mann-Whitney U-test. Categorical variables were compared between groups by Pearson’s chi-square test or Fisher’s exact test. Time-dependent receiver operating characteristic (ROC) curves were utilized to determine optimal cutoff values for the biomarkers in predicting 1,3 and 5-year overall survival (OS). Corresponding area under the curve (AUC) values were calculated, and patients were categorized into high- and low-expression groups based on these thresholds. OS was defined as the interval from surgery to death or last follow-up. Kaplan–Meier curves with the log-rank test were used to survival analysis. Univariate Cox regression identified potential prognostic factors. Variables with P < 0.05 were included in multivariate Cox regression. Statistical significance was defined as two-sided P < 0.05. All statistical analyses were performed using SPSS 26.0 and GraphPad Prism 8.0.
A total of 222 patients diagnosed with NSCLC who underwent surgical treatment were included in this study. The mean age of the cohort was 63 months, comprising 133 male and 89 female patients. The median follow-up duration was 77 months. Patients were stratified into groups based on survival outcomes.
Baseline characteristics are summarized in Table 1. Significant differences were observed between the survival and non-survival groups in terms of sex, age, and smoking history. However, no significant differences were found in BMI, hypertension, diabetes, or coronary artery disease. Regarding surgical variables, differences in hospitalization duration, surgical approach, and operation time were statistically significant. Pathological characteristics, including tumor differentiation, histological type, vascular invasion, and tumor thrombus, also showed significant differences between the groups. Among inflammatory markers, preoperative white blood cell, neutrophil, and monocyte counts exhibited significant differences, as did the derived markers NLR, MLR, PLR, SII, and SIRI.
Time-dependent ROC curves were employed to investigate the correlation between preoperative inflammatory biomarkers and postoperative prognosis (Figure 2). The optimal cutoff values for NLR, PLR, MLR, SII, and SIRI in predicting 1-year OS were determined, with corresponding AUCs of 0.741, 0.731, 0.756, 0.746, and 0.749, respectively. Based on these cutoff values, patients were divided into high- and low-expression groups, and Cox regression analysis was performed.
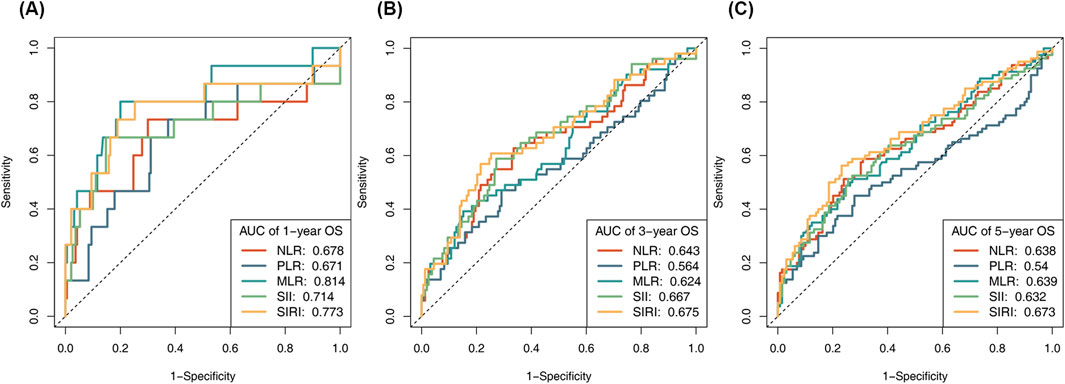
Figure 2. The receiver operating characteristic (ROC) curves explore the value of NLR, PLR, MLR, SII and SIRI in predicting (A) 1-year survival, (B) 3-year survival, and (C) 5-year survival outcomes of NSCLC patients.
Univariate Cox regression analysis results are presented in Table 2. Variables with statistical significance were included in multivariate Cox regression analysis, which revealed that age, operation time, tumor stage, and differentiation were independent factors associated with OS. Long-term prognosis was assessed using Kaplan-Meier survival analysis (Figure 3). Patients were stratified into high- and low-expression groups based on the five inflammatory biomarkers. The low-expression groups consistently demonstrated significantly higher OS rates compared to the high-expression groups, with all group differences being statistically significant (log-rank test, p < 0.001).
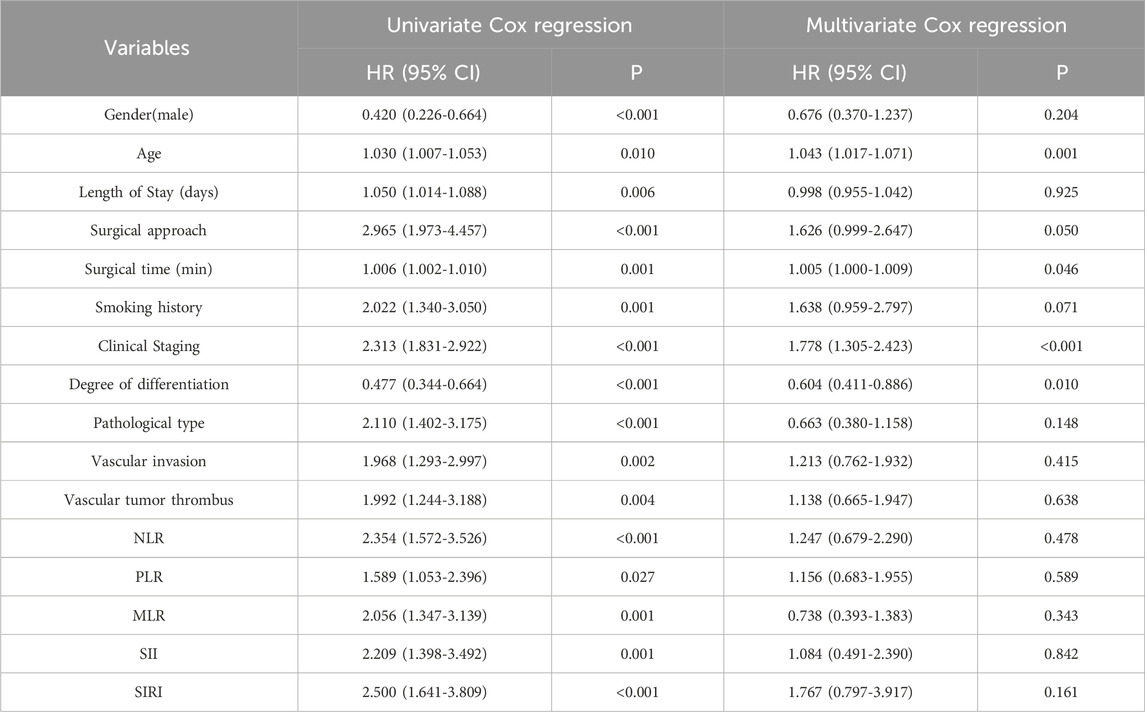
Table 2. Cox regression examination investigating the impact of clinicopathological variables on patients’ overall survival.
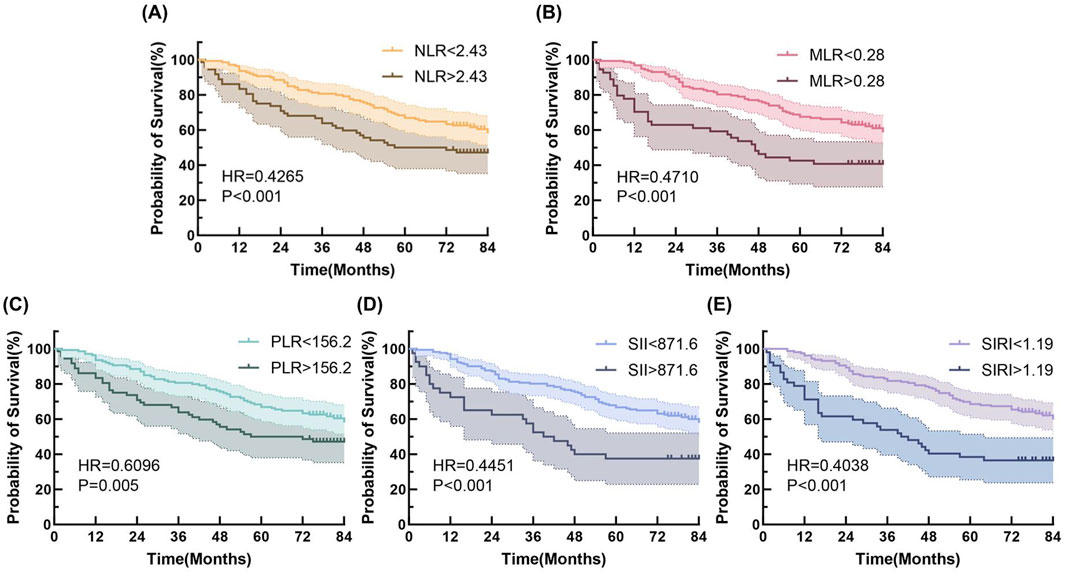
Figure 3. Kaplan-Meier curves for long time prognosis by (A) NLR, (B) MLR, (C) PLR, (D) SII and (E) SIRI.
Chronic inflammation has been widely recognized as a critical factor in cancer development and progression. Clinical studies examining inflammation-related prognostic biomarkers in lung cancer patients have proliferated in recent years. However, there remains a lack of long-term follow-up data from NSCLC cohorts that exclude AIS patients. In this retrospective study, we investigated prognostic factors in NSCLC patients after excluding AIS cases. Our findings confirmed significant differences in survival based on levels of various inflammatory biomarkers, with higher levels correlating with poorer long-term survival.
Inflammatory cells and their derived indices play pivotal roles in tumor initiation, progression, and metastasis (Inamura, 2018; Diakos et al., 2014). These indices reflect the complex interplay between pro-tumorigenic and immune surveillance dysregulation within the TME. For instance, tumor-associated neutrophils promote angiogenesis by secreting vascular endothelial growth factor (VEGF) and activating oncogenic pathways such as NF-κB via chronic reactive oxygen species (ROS) release. Tumor-associated macrophages (TAMs), particularly M2-polarized TAMs, secrete VEGF and matrix metalloproteinases (MMPs), fostering tumor angiogenesis and invasion (Zou et al., 2017; Swierczak et al., 2015; Barbetta et al., 2018; Cortese et al., 2020). Platelets contribute to tumor metastasis by facilitating epithelial-mesenchymal transition (EMT) and protecting circulating tumor cells (Żmigrodzka et al., 2020). These cellular processes are encapsulated by inflammatory indices such as NLR, PLR, MLR, SII, and SIRI (Qi et al., 2016). Numerous studies have demonstrated the prognostic value of these markers in various malignancies, including lung, liver, and breast cancers (Hu et al., 2014; Chen et al., 2020). For example, a follow-up study of 1,431 stage I lung adenocarcinoma patients found significant correlations between NLR, SII, SIRI, and cancer-specific survival (CSS) or disease-free survival (DFS) (Shen et al., 2021). Similarly, studies in advanced lung cancer cohorts have established associations between elevated SIRI levels and poorer prognosis, as well as similar findings for NLR and PLR (Jiang et al., 2021; Zuo et al., 2023).
However, our study found that while higher levels of these inflammatory markers were associated with poorer survival, they did not independently predict survival in multivariate Cox regression analyses. This is in contrast to some of the findings in the existing literature, where a number of studies have demonstrated the potential of various inflammatory markers to independently predict long-term survival in predicting lung cancer prognosis. For example, a summary evaluation of 86 meta-analyses on NLR and prognosis of malignant tumors showed that there is a strong association between elevated NLR and poor prognosis in cancer patients (Cupp et al., 2020). A study by Aguiar-Bujanda et al. found that SII had a high clinical value. Elevated SII was associated with advanced tumor stage and lymph node metastasis, and could be used as a prognostic predictor in patients with advanced tumors (Aguiar-Bujanda et al., 2018). This discrepancy may be attributed to potential interactions among these markers, diminishing their individual contributions in multivariate models. Additionally, inflammatory markers may primarily reflect short-term changes within the TME, while long-term prognostic factors such as tumor stage and pathological features overshadow their influence. Long-term prognosis is more influenced by the biological behavior of the tumor and postoperative treatment. Supporting this hypothesis, our time-dependent ROC analysis revealed higher diagnostic efficacy for 1-year survival compared to three- or 5-year survival endpoints. Kaplan-Meier survival analysis further demonstrated that patients with lower levels of NLR, PLR, MLR, SII, and SIRI exhibited significantly better survival outcomes. This underscores the potential utility of these markers in short-term prognosis, though their inclusion as independent factors in clinical prediction models remains limited.
Multivariate Cox regression identified age, operation time, tumor stage, and differentiation as independent prognostic factors for OS, consistent with previous studies. These findings emphasize the importance of clinical and pathological characteristics in NSCLC prognosis. Yotsukura’s team conducted clinical follow-up on 524 postoperative patients with pathological diagnosis of AIS, indicating that the tumor-related survival rate after surgery was nearly 100% (Jiang et al., 2021; Yotsukura et al., 2021). Our cohort excluded AIS patients, whose favorable prognosis and unique biological behavior differ markedly from invasive cancers. This exclusion enhanced the specificity of our results by directly linking prognosis to tumor-related characteristics rather than confounding factors such as localized inflammation or early-stage disease features.
Although this study systematically analyzed the prognostic impact of inflammatory biomarkers on NSCLC patients through long-term follow-up, certain limitations remain. First, as a single-center retrospective study, it may be subject to unavoidable selection bias. Second, the dynamic changes of inflammatory biomarkers were not included in the analysis, which might limit their predictive value in long-term follow-up. Despite these limitations, this study has notable clinical significance. By excluding AIS patients, we clarified the short-term prognostic value of inflammatory biomarkers in invasive NSCLC and reaffirmed the central role of traditional clinical and pathological features (such as tumor stage and differentiation) in long-term prognosis. While our study mainly focused on biomarkers, we recognize that inflammatory factors such as IL-6, TNF-α, and MCP-1 may also significantly influence the prognosis of NSCLC. IL-6 and TNF-α promote tumor progression by enhancing inflammation and creating an immunosuppressive microenvironment, while MCP-1 recruits monocytes and macrophages, potentially aiding immune evasion and metastasis. Although these factors were not measured in our study, we recommend that future research investigate their specific effects on tumor cells. Based on these findings, we propose that the application of inflammatory biomarkers in postoperative management of invasive lung cancer requires further confirmation through prospective studies.
Inflammatory markers, while valuable in short-term prognostic evaluation, cannot yet be considered independent risk factors. Traditional clinical and pathological features (e.g., age, operation time, tumor stage, and differentiation) have clear independent prognostic value in NSCLC. Close monitoring of patients with high inflammatory marker levels is warranted and may provide clinical benefit in improving long-term outcomes.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving humans were approved by Beijing chaoyang hospital ethics committee. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation was not required from the participants or the participantsandapos; legal guardians/next of kin in accordance with the national legislation and institutional requirements.
QZ: Conceptualization, Data curation, Software, Writing–original draft. SoC: Formal Analysis, Investigation, Methodology, Writing–original draft. BH: Project administration, Supervision, Writing–review and editing. ShC: Project administration, Resources, Supervision, Writing–review and editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Aguiar-Bujanda, D., Dueñas-Comino, A., Saura-Grau, S., Ros-Sanjuan, L., Blanco-Sanchez, M. J., Hernandez-Sosa, M., et al. (2018). Neutrophil to lymphocyte ratio as a prognostic factor in European patients with epidermal growth factor receptor-mutant non-small cell lung cancer treated with tyrosine kinase inhibitors. Oncol. Res. Treat. 41 (12), 755–761. doi:10.1159/000492344
Barbetta, A., Nobel, T. B., Sihag, S., Hsu, M., Tan, K. S., Bains, M. S., et al. (2018). Neutrophil to lymphocyte ratio as predictor of treatment response in esophageal squamous cell cancer. Ann. Thorac. Surg. 106 (3), 864–871. doi:10.1016/j.athoracsur.2018.04.007
Chen, L., Kong, X., Wang, Z., Wang, X., Fang, Y., and Wang, J. (2020). Pre-treatment systemic immune-inflammation index is a useful prognostic indicator in patients with breast cancer undergoing neoadjuvant chemotherapy. J. Cell. Mol. Med. 24 (5), 2993–3021. doi:10.1111/jcmm.14934
Cortese, N., Carriero, R., Laghi, L., Mantovani, A., and Marchesi, F. (2020). Prognostic significance of tumor-associated macrophages: past, present and future. Seminars Immunol. 48, 101408. doi:10.1016/j.smim.2020.101408
Cupp, M. A., Cariolou, M., Tzoulaki, I., Aune, D., Evangelou, E., and Berlanga-Taylor, A. J. (2020). Neutrophil to lymphocyte ratio and cancer prognosis: an umbrella review of systematic reviews and meta-analyses of observational studies. BMC Med. 18(1), 360. doi:10.1186/s12916-020-01817-1
Diakos, C. I., Charles, K. A., McMillan, D. C., and Clarke, S. J. (2014). Cancer-related inflammation and treatment effectiveness. Lancet Oncol. 15 (11), e493–e503. doi:10.1016/S1470-2045(14)70263-3
Feng, Y., Zhang, N., Wang, S., Zou, W., He, Y., Ma, J. A., et al. (2020). Systemic inflammation response index is a predictor of poor survival in locally advanced nasopharyngeal carcinoma: a propensity score matching study. Front. Oncol. 10, 575417. doi:10.3389/fonc.2020.575417
Han, B., Zheng, R., Zeng, H., Wang, S., Sun, K., Chen, R., et al. (2024). Cancer incidence and mortality in China, 2022. J. Natl. Cancer Cent. 4 (1), 47–53. doi:10.1016/j.jncc.2024.01.006
Hu, B., Yang, X. R., Xu, Y., Sun, Y. F., Sun, C., Guo, W., et al. (2014). Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma. Clin. Cancer Res. 20 (23), 6212–6222. doi:10.1158/1078-0432.CCR-14-0442
Inamura, K. (2018). Clinicopathological characteristics and mutations driving development of early lung adenocarcinoma: tumor initiation and progression. Int. J. Mol. Sci. 19 (4), 1259. doi:10.3390/ijms19041259
Jiang, S., Wang, S., Wang, Q., Deng, C., Feng, Y., Ma, F., et al. (2021). Systemic inflammation response index (SIRI) independently predicts survival in advanced lung adenocarcinoma patients treated with first-generation EGFR-TKIs. Cancer Manag. Res. 13, 1315–1322. doi:10.2147/CMAR.S287897
Leiter, A., Veluswamy, R. R., and Wisnivesky, J. P. (2023). The global burden of lung cancer: current status and future trends. Nat. Rev. Clin. Oncol. 20 (9), 624–639. doi:10.1038/s41571-023-00798-3
Qi, Q., Zhuang, L., Shen, Y., Geng, Y., Yu, S., Chen, H., et al. (2016). A novel systemic inflammation response index (SIRI) for predicting the survival of patients with pancreatic cancer after chemotherapy. Cancer 122 (14), 2158–2167. doi:10.1002/cncr.30057
Shen, Y. J., Qian, L. Q., Ding, Z. P., Luo, Q. Q., Zhao, H., Xia, W. Y., et al. (2021). Prognostic value of inflammatory biomarkers in patients with stage I lung adenocarcinoma treated with surgical dissection. Front. Oncol. 11, 711206. doi:10.3389/fonc.2021.711206
Swierczak, A., Mouchemore, K. A., Hamilton, J. A., and Anderson, R. L. (2015). Neutrophils: important contributors to tumor progression and metastasis. Cancer Metastasis Rev 34 (4), 735–51. doi:10.1007/s10555-015-9594-9
Templeton, A. J., McNamara, M. G., Šeruga, B., Vera-Badillo, F. E., Aneja, P., Ocaña, A., et al. (2014). Prognostic role of neutrophil-to-lymphocyte ratio in solid tumors: a systematic review and meta-analysis. J. Natl. Cancer Inst. 106 (6), dju124. doi:10.1093/jnci/dju124
Tomita, M., Shimizu, T., Ayabe, T., Yonei, A., and Onitsuka, T. (2011). Preoperative neutrophil to lymphocyte ratio as a prognostic predictor after curative resection for non-small cell lung cancer. Anticancer Res. 31 (9), 2995–2998.
Xie, H., Ruan, G., Wei, L., Deng, L., Zhang, Q., Ge, Y., et al. (2023). The inflammatory burden index is a superior systemic inflammation biomarker for the prognosis of non-small cell lung cancer. J. cachexia, sarcopenia muscle 14 (2), 869–878. doi:10.1002/jcsm.13199
Yotsukura, M., Asamura, H., Motoi, N., Kashima, J., Yoshida, Y., Nakagawa, K., et al. (2021). Long-term prognosis of patients with resected adenocarcinoma in situ and minimally invasive adenocarcinoma of the lung. J. Thorac. Oncol. 16 (8), 1312–1320. doi:10.1016/j.jtho.2021.04.007
Żmigrodzka, M., Witkowska-Piłaszewicz, O., and Winnicka, A. (2020). Platelets extracellular vesicles as regulators of cancer progression-an updated perspective. Int. J. Mol. Sci. 21 (15), 5195. doi:10.3390/ijms21155195
Zou, J. M., Qin, J., Li, Y. C., Wang, Y., Li, D., Shu, Y., et al. (2017). IL-35 induces N2 phenotype of neutrophils to promote tumor growth. Oncotarget 8 (20), 33501–33514. doi:10.18632/oncotarget.16819
Keywords: NSCLC, inflammatory, biomarker, prognostic biomarker, lung cancer
Citation: Zhao Q, Cui S, Hu B and Chen S (2025) Retrospective analysis of inflammatory biomarkers and prognosis in non-small cell lung cancer without adenocarcinoma in situ. Front. Genet. 16:1549602. doi: 10.3389/fgene.2025.1549602
Received: 21 December 2024; Accepted: 20 February 2025;
Published: 18 March 2025.
Edited by:
Zhijia Liu, Eighth Medical Center of the General Hospital of the Chinese People’s Liberation Army, ChinaCopyright © 2025 Zhao, Cui, Hu and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Bin Hu, aHViaW43MDVAYWxpeXVuLmNvbQ==; Shuo Chen, Y3l5eXh3a2NzQHNpbmEuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.