
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Genet. , 20 March 2025
Sec. Genomics of Plants and the Phytoecosystem
Volume 16 - 2025 | https://doi.org/10.3389/fgene.2025.1534894
This article is part of the Research Topic Survival Strategies and Biotechnological Applications of Plants in Extreme Environments View all articles
Introduction: In the context of climate change, the frequency and intensity of droughts in arid and semi-arid areas have shown a substantially increasing trend, which inevitably affects plant survival and growth. However, it is unclear what survival and growth strategies plants subjected to drought pretreatment in the early life stages adopt when facing subsequent drought stress.
Methods: Here, we conducted a field experiment and set up two treatments, control and drought pretreatment, to investigate the effects of drought pretreatment on the survival, phenology, morphology, biomass, and stoichiometric characteristics of the ephemeral plant Erodium oxyrhinchum in the Gurbantunggut Desert, China.
Results: The results showed that the leafing, flowering, and fruiting stages under drought pretreatment occurred markedly earlier than the control treatment by 5.25 ± 1.2 d, 3.13 ± 0.84 d, and 4.75 ± 1.63 d, respectively. The life history of E. oxyrhinchum decreased 5 ± 1.38 d under drought pretreatment. Drought pretreatment accelerated seedling mortality, leading to a faster and earlier decline in survival percentage. The survival percentage of E. oxyrhinchum under drought pretreatment at the full blooming stage was approximately 18.59%, which was 5.19% higher than that of the control treatment. In addition, a positive correlation was observed between morphological traits and individual biomass, and drought pretreatment substantially increased individual biomass and reproductive output. For example, the reproductive biomass under drought pretreatment was 1.41 times than that of the control treatment during the full fruiting stage, indicating that plants subjected to drought pretreatment exhibited an overcompensation effect. Finally, from the perspective of stoichiometric characteristics, plants subjected to drought pretreatment require more phosphorus to enhance their resistance to severe drought.
Conclusion: This study provides novel insights for the conservation and restoration of desert ecosystems in the context of climate change.
Drought is expected to increase in frequency, severity, and duration in the future because of global warming, which will pose serious threats to plant survival, growth, and regeneration (Lin et al., 2014; Huang et al., 2015a). Many studies have indicated that drought can affect multiple life history stages of plants, such as by decreasing germination percentage, increasing seedling mortality, and altering biomass allocation (Zia et al., 2021; Walters et al., 2023; Zhang et al., 2023; Brunet, et al., 2024). However, some researchers have found that plants can adopt various strategies to cope with drought exposure, including changes in root configuration, reduced leaf area, regulation of antioxidant accumulation, and ratios of C, N, and P (Guo et al., 2023; Fuente et al., 2023; Haghpanah et al., 2024). In particular, when plants are rewatered after an initial drought, they often show an overcompensation effect and improve their adaptability to drought stress (Hofer et al., 2017; Gao et al., 2024). Therefore, it is crucial to investigate the effects of drought pretreatment on plants in the context of global climate change.
Plants subjected to drought pretreatment may exhibit coping strategies compared to those that have experienced a single drought event (Haider et al., 2024). Consequently, numerous scholars have explored the effects of drought pretreatment on plants from multiple perspectives, including morphological changes, biomass allocation, and stoichiometry composition. For example, Gao et al. (2024) found that the root length of maize seedlings increased by 21.05% after drought rewatering, enhancing the ability of maize seedlings to adapt to drought stress following two cycles of drought rewatering treatments. Lv et al. (2024) also found that drought pretreatment not only increased the number of small vascular bundles, altered the shape and density of stomata, and enhanced water use efficiency in potatoes, but also increased the amount and rate of dry biomass transport, thereby reducing the adverse effects of drought stress on potato tubers. Another study from the perspective of the content and ratios of stoichiometry also found that the content of N in different organs and P in the fine roots of Pinus yunnanensis seedlings increased after rewatering with light drought, moderate drought, and severe drought treatments, while the C:N ratio decreased (Liu et al., 2024). These findings indicate that drought pretreatment can increase the root length of maize seedlings and the dry biomass of potatoes, altering the allocation of stoichiometric composition through an overcompensated effect. However, most studies have focused on crops or tree species, with relatively little research on the effects of drought pretreatment on desert herbs in arid environment.
The frequency and severity of drought events are increasingly influenced by global climate change, particularly in arid and semiarid regions (Ghannoum, 2009; Hosseinizadeh et al., 2015). In the temperate desert ecosystems of Central Asia, ephemeral plants play crucial roles in windbreaks and sand fixation, stabilising moving sand dunes, and protecting agricultural lands from sand encroachment (Sun et al., 2008; Mu et al., 2021; Lu et al., 2022). Moreover, these plants primarily rely on snowmelt and rainfall in early spring for rapid growth, completing their life history before the onset of hot and dry summers, and characterising them as typical drought-avoidant species (Mu et al., 2021). Consequently, ephemeral plants are considered ideal materials for studying plant responses to drought stress (Chen et al., 2019a; Mu et al., 2021). In particular, E. oxyrhinchum is the dominant ephemeral plant in the Gurbantunggut Desert (Mu et al., 2021). It germinates in March-April, flowers in April-May, and fruits in May-June, completing its life history in 2–3 months (Zhang and Chen, 2002; Wang and Zhang, 2010; Zhang et al., 2022a; Zhang et al., 2022b). This life strategy of E. oxyrhinchum enables it to evade extreme summer temperatures in arid regions. During the growing season of E. oxyrhinchum, soil moisture from snowmelt and spring rainfall exhibited a fluctuating downward trend, with soil dryness gradually increasing (Chen et al., 2019a). Studies have indicated that the soil water content in early spring typically remains above 10%, and by the end of its life history, it can drop to less than 4% (Chen et al., 2019a). Other studies have shown that plants often increase the allocation of belowground biomass and alter their leaf morphology, transitioning from deeply lobed to entirely palmate leaves under drought conditions (Chen et al., 2019b). Based on the drought-escaping characteristics of ephemeral plants and the dynamic trends of soil water content in the Gurbantunggut Desert, we propose the first hypothesis: E. oxyrhinchum reduces its survival percentage, enhances drought tolerance, and triggers compensatory effects on morphology and biomass accumulation.
Furthermore, Carbon (C), nitrogen (N), and phosphorus (P) are essential for plant growth and physiological activities (Lu et al., 2023). Plants adapt to environmental changes through coordination among the elements in their bodies, which reflects the relationship between plants and their environment (Jiang et al., 2023). Accordingly, we proposed the second hypothesis: E. oxyrhinchum subjected to drought pretreatment will exhibit significant increases in the concentration of C, N, and P in leaves. To validate these hypotheses, we conducted a pre-drought experiment in the Gurbantunggut Desert to assess the effect of drought pretreatment on survival, phenology, morphology, biomass, and stoichiometric characteristics of E. oxyrhinchum. Therefore, this study provides a theoretical and practical basis for the adaptive evolution and conservation of ephemeral plants in desert environments.
The study area is located on the southern edge of the Gurbantunggut Desert in Northwest China. The Gurbantunggut Desert, characterised by a typical temperate continental arid climate, is the largest stabilised and semi-stabilised desert in China (Chen et al., 2019c; Lu et al., 2022; Zhang et al., 2022b). The mean annual temperature varies from 6°C to 10°C, with extreme high temperatures over 40°C (Liu et al., 2020a). The annual potential evaporation can exceed 2,000 mm, and the annual precipitation in the central region of the desert is only 70–100 mm, which mainly occurs in winter and spring (Liu et al., 2020a; Lu et al., 2022). Natural conditions in the Gurbantunggut Desert are extremely harsh and susceptible to human activity and extreme climatic events. However, compared with other desert regions in China, herbaceous plants in the Gurbantunggut Desert are relatively abundant in spring, especially in ephemeral plant communities (Erfan et al., 2023). E. oxyrhinchum is a typical annual ephemeral plant found in the Gurbantunggut Desert. It contributes to more than 50% of the aboveground productivity of ephemeral plants, is the dominant species in the Gurbantunggut Desert, and plays a crucial role in the desert ecosystem of China (Zang et al., 2020).
The field experimental plot is located at the southern edge of the Gurbantunggut Desert (44°26′N, 87°54′E), which was established in September 2015. Firstly, in order to prevent interference from wildlife and humans, we have established reinforced bilateral fences. Secondly, we have installed PVC light-transmitting and rain-sheltering equipment, it can be used to block the interference of natural precipitation. Finally, a 15 m × 15 m plot with relatively flat terrain and no distribution of E. oxyrhinchum was selected within the aforementioned field plot. A randomized block design was used, with four large plots (5 m × 5 m) spaced 3 m apart. Within each large plot, 16 small plots (1 m × 1 m) were set up, with 8 plots designated for control treatment and 8 plots for drought pretreatment (Figures 1A, B). A 5 mm thick water-impermeable membrane was used between the large and small plots to prevent water seepage.
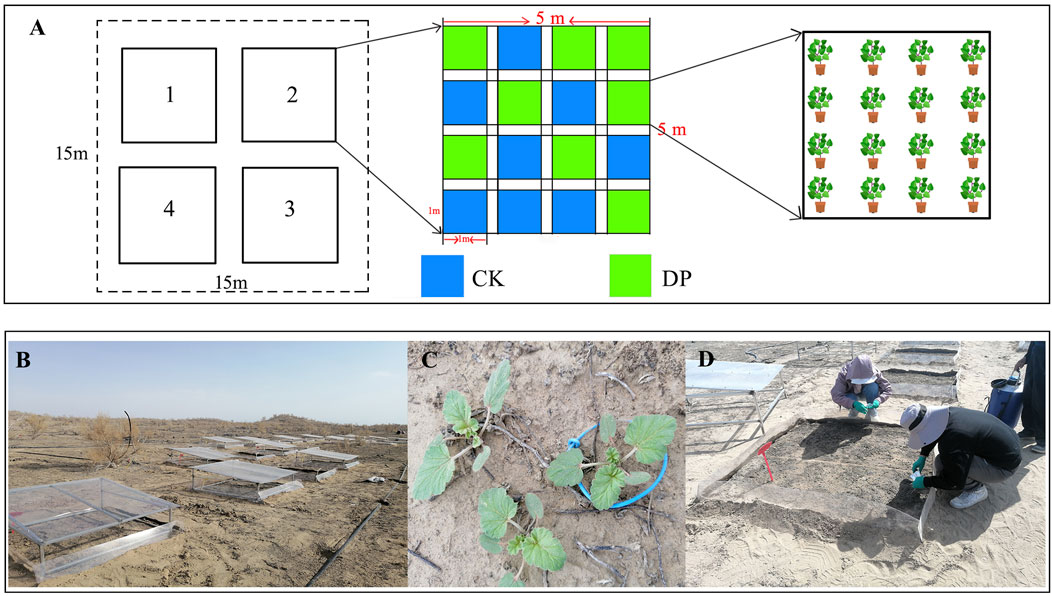
Figure 1. Experimental design (A), PVC light-transmitting and rain-sheltering equipment (B), Erodium oxyrhinchum seedlings (C) and sample collection (D) in the field. Note: Control treatment, CK; Drought pretreatment, DP.
Considering the average precipitation (102 mm) during the growth season of ephemeral plants in the Gurbantunggut desert over the past decade, we conducted an experiment of drought pretreatment by controlling watering frequency on the basis of complete rain cover. For the control treatment (CK): watering once every 2 days during the cotyledon, leaf expansion, and initial flowering stages, and once every 10 days during the full booming, fruiting and withering stages, with each watering amount of 8 mm (calculated based on the average watering amount of the life history and phenological transition time of E. oxyrhinchum). The cumulative watering volume for the control treatment was approximately 104 mm. For the drought pretreatment (DP): watering once every 4 days during the cotyledon stage to simulate mild drought, watering once every 7 days during the full leaf expansion stage to simulate moderate drought, and once every 10 days during the full blooming, fruiting and withering stages to simulate severe drought (Figure 2). And once every 2 days during other phenological stages consistent with the control treatment (Figure 2). The cumulative watering amount was about 56 mm.
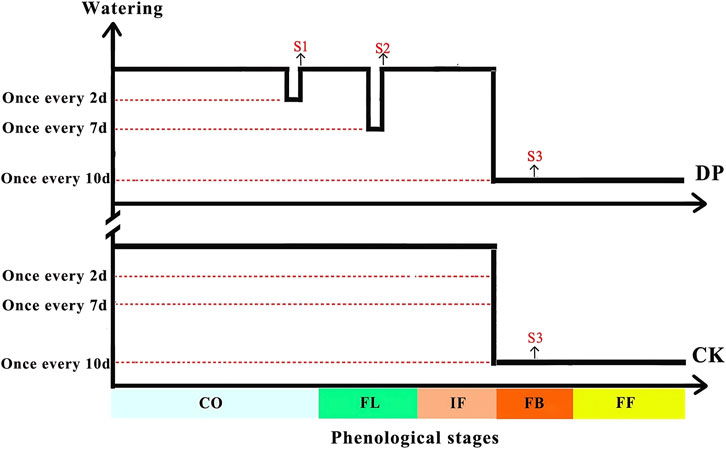
Figure 2. Experimental design of drought pretreatment for the desert ephemeral plant Erodium oxyrhinchum. Note: Control treatment, CK; Drought pretreatment, DP; Cotyledon stage, CO; Full leaf expansion stage, FL; Initial flowering stage, IF; Full blooming stage, FB; Full fruiting stage, FF; Mild drought, S1; Moderate drought, S2; Severe drought, S3.
Plant samples were collected at different phenological stages of E. oxyrhinchum: the cotyledon stage (29 March), initial leaf expansion stage (mid-April), full leaf expansion stage (late April), initial flowering stage (early May), full booming stage (mid-May), and full fruiting stage (late May). Whole plants were preserved in water bags and transported to the laboratory for analysis, and the leaves of E. oxyrhinchum were preserved in liquid nitrogen to determine the stoichiometric indicators (Figures 1C, D).
For each treatment, the number of days from 1 January 2023 to the germination, leafing, blooming, fruiting, and withering stages was calculated. These data served as the phenological metrics for subsequent analyses.
As the plants grew, their traits (plant height, leaf area, root length, and number of leaves) were measured. We measured the leaf area of fresh leaves using an LI-COR 3000 leaf area meter. After the plant traits were measured, the plants in all treatments were harvested and separated into roots, stems, leaves, and reproductive organs (flowers, fruits, and seeds). The roots were carefully washed free of soil. Plant samples were blanched in an oven at 105°C for 30 min before drying at 70°C to a consistent weight. The plant organs were weighed using an electronic balance (OHAUS, Pine Brook, NJ, United States, 0.0001 g). Individual biomass (IB) was determined by adding the aboveground biomass (AGB) and belowground biomass (BGB), and the root to shoot ratio (R/S) was computed as the ratio of belowground to aboveground biomass.
Mature leaves from the upper section of E. oxyrhinchum were dried and ground into powder using a ball mill for stoichiometric analysis. The C and N concentrations in the leaves were determined using an elemental analyser (EA3000, Italy), and the P concentration was determined using the molybdenum-antimony colorimetric method.
The survival percentage of E. oxyrhinchum was analysed using independent sample t-tests with a significance level of 0.05, and Levene’s test was used to confirm the homogeneity of variance. In addition, independent sample t-tests (consistent with the survival percentage analysis method) and two-way ANOVA were used to analyse plant height, root length, number of leaves, leaf area, individual biomass, reproductive biomass, root to shoot ratio, growth rate, and stoichiometric characteristics. The coefficient of variation (CV) was employed to examine the variability of leaf C, N, and P concentration and their ratios throughout the sampling period: 0≤CV<15% shows weak variability, 15%≤CV<35% indicates moderate variability, and CV ≥ 35% indicates high variability. Data analysis was conducted using SPSS27.0, and graphical representations were generated using R-4.3.2.
Statistical analysis revealed that the phenological stages of E. oxyrhinchum under drought pretreatment were significantly earlier than those under the control treatment (p < 0.001, Table 1). Specifically, under drought pretreatment, the full leaf expansion stage occurred 5.25 ± 1.2 days earlier, the initial flowering stage 3.13 ± 0.84 days earlier, the initial fruiting stage 4.75 ± 1.63 days earlier, and the withering stage 5 ± 1.38 days earlier than that of control treatment (Table 1). Notably, the entire life history of E. oxyrhinchum under drought pretreatment were also significant shorter than that of control treatment: DP (69.63 ± 0.75 days) < CK (74.63 ± 0.63 days).
On 20 March 2023 the seeds of E. oxyrhinchum exhibited an explosive germination strategy, with seedlings appearing uniform before entering the cotyledon stage (Figure 3). The survival percentages of E. oxyrhinchum in the control and drought treatments did not significantly decrease from the cotyledon stage to April 19. After the full leaf expansion stage, the survival percentage of E. oxyrhinchum under drought pretreatment showed an earlier and faster decline than that of the control treatment (Figure 3). By the end of May, plants exposed to the drought pretreatment rapidly entered the withering stage. By early June, E. oxyrhinchum had completed its reproductive output and ended its life history between the control treatment and drought pretreatment (Figure 3).
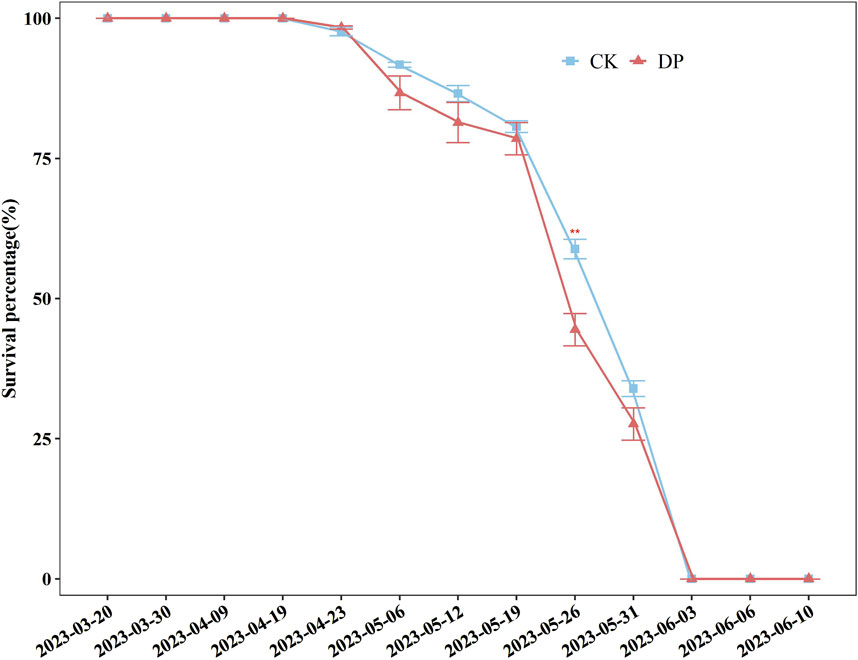
Figure 3. Effects of drought pretreatment on the survival percentage of Erodium oxyrhinchum. Note: Control treatment, CK; Drought pretreatment, DP; *p < 0.05, **p < 0.01, ***p < 0.001.
The results of the two-way ANOVA indicated significant differences in terms of plant height, root length, leaf area, and leaf number of E. oxyrhinchum at different phenological stages (p < 0.001, Table 2). There were significant differences between the control and drought treatments in terms of plant height and root length (p < 0.05, Table 2); however, no significant differences were observed in leaf number (p = 0.502, Table 2) or leaf area (p = 0.12, Table 2). Furthermore, there was a significant interaction effect between drought treatment and phenological stage on plant height and leaf number (p < 0.05, Table 2).
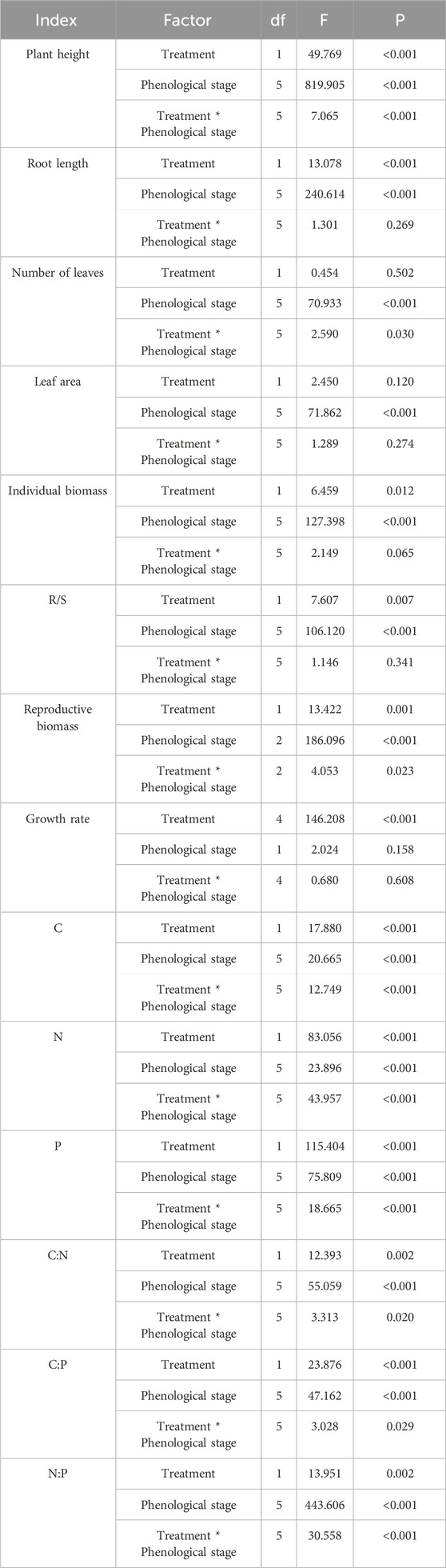
Table 2. Summary of a two-way ANOVA showing the effects of different drought treatments and phenological stages on the morphology, biomass and stoichiometric characteristics of Erodium oxyrhinchum.
The height, root length, and leaf area of E. oxyrhinchum in the control and drought treatments increased gradually with the phenological stage (Figure 4). Prior to the fruiting stage, the number of leaves increases. From the initial leaf expansion stage to the full fruiting stage, plant height under drought pretreatment was significantly higher than that of the control treatment (p < 0.05, Figure 4). During the initial leaf expansion stage, root length under drought pretreatment was significantly longer than that of the control treatment. At the initial leaf expansion and flowering stages, the number of leaves under the drought pretreatment was significantly higher than that of the control treatment, but the difference did not reach a significant level during the full blooming and fruiting stages.
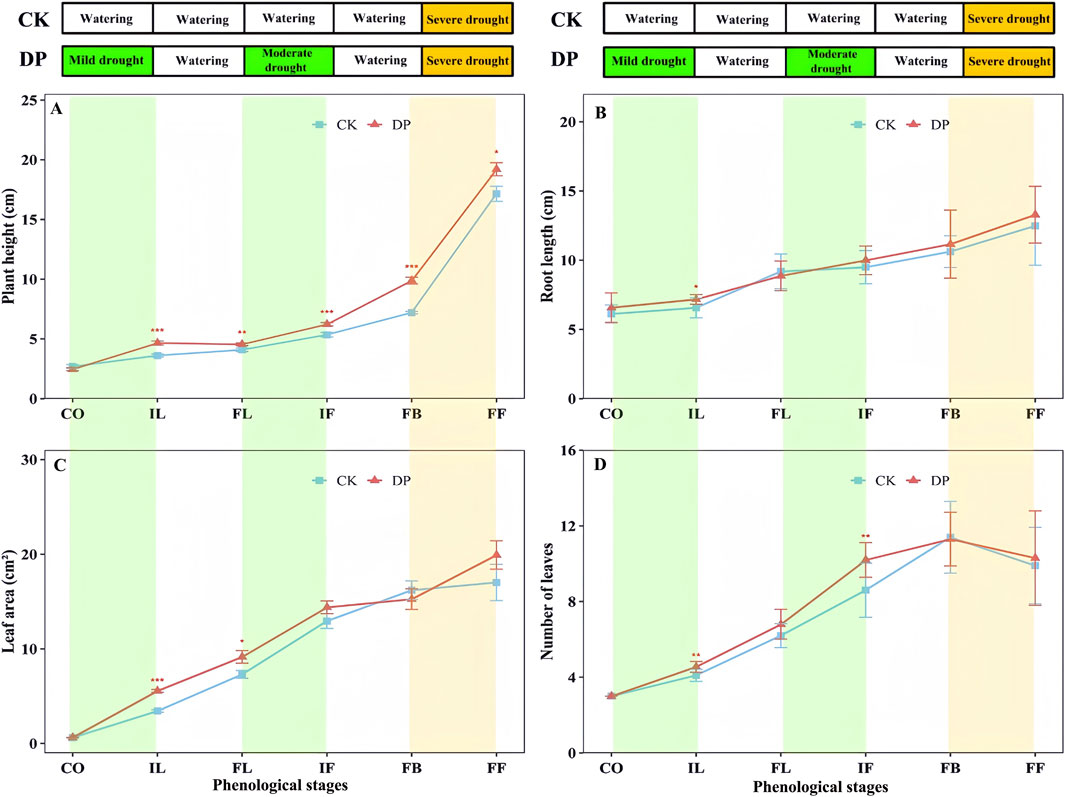
Figure 4. Effects of drought pretreatment on the plant height (A), root length (B), leaf area (C), and number of leaves (D) of Erodium oxyrhinchum. Note: Control treatment, CK; Drought pretreatment, DP; Cotyledon stage, CO; Initial leaf expansion stage, IL; Full leaf expansion stage, FL; Initial flowering stage, IF; Full blooming stage, FB; Full fruiting stage, FF; *p < 0.05, **p < 0.01, ***p < 0.001.
According to the results of the two-way ANOVA, the individual biomass, reproductive biomass, and growth rate of E. oxyrhinchum showed significantly increasing trends, but the R/S exhibited a significantly decreasing trend as the plants grew (p < 0.001, Table 2). Similarly, there were significant differences in the individual biomass, root to shoot ratio, and reproductive biomass of E. oxyrhinchum between the control and drought treatments (p < 0.05, Table 2). Throughout the reproductive season, the reproductive biomass under the drought pretreatment was consistently higher than that under the control treatment (Figure 5). Specifically, the reproductive biomass under drought pretreatment during the full fruiting stage was 1.41 times than that of the control. From the perspective of the entire life history, the individual biomass of E. oxyrhinchum under the drought pretreatment was greater than that of the control treatment (Figure 5). For example, during initial leaf expansion, the individual biomass of E. oxyrhinchum under the drought treatment was 1.29 times than that of the control treatment.
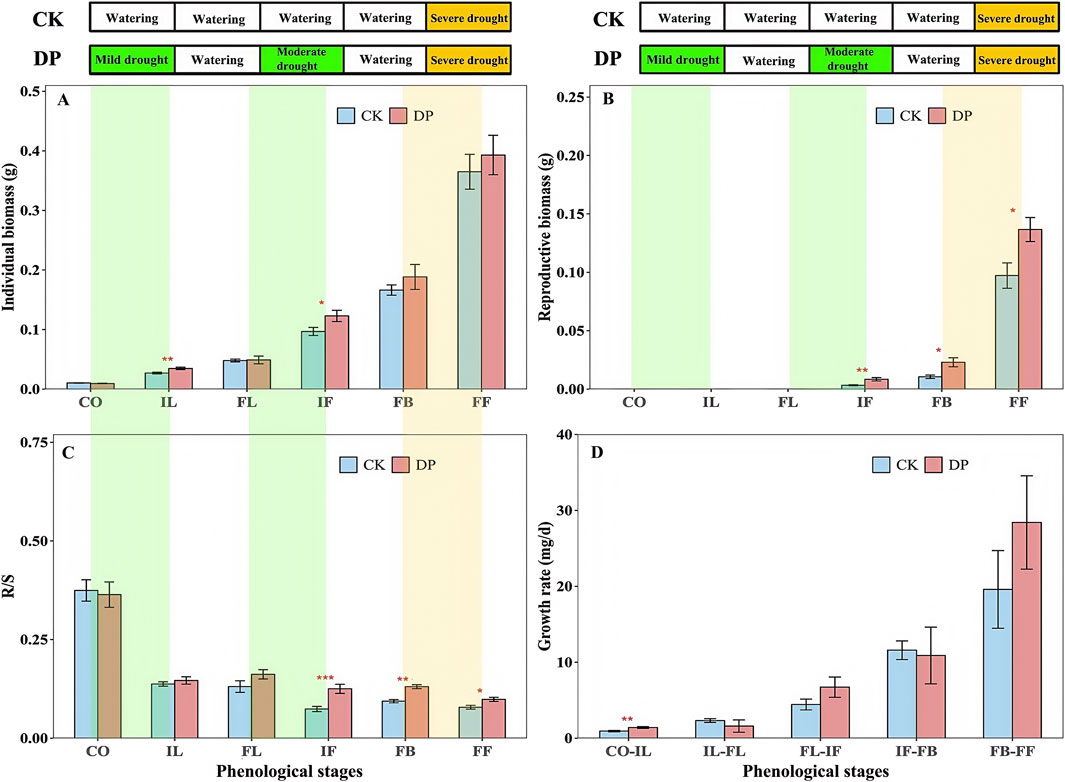
Figure 5. Effects of drought pretreatment on the individual biomass (A), reproductive biomass (B), R/S (C), growth rate (D) of Erodium oxyrhinchum. Note: Control treatment, CK; Drought pretreatment, DP; Cotyledon stage, CO; Initial leaf expansion stage, IL; Full leaf expansion stage, FL; Initial flowering stage, IF; Full blooming stage, FB; Full fruiting stage, FF; *p < 0.05, **p < 0.01, ***p < 0.001.
Two-way ANOVA revealed significant differences in the stoichiometric characteristics and ratios of C: N, and P in the leaves of E. oxyrhinchum between the control and drought treatments (p < 0.001, Table 2). Similarly, from the perspective of the entire life history, significant differences in the stoichiometric characteristics and C: N, and P in the leaves of E. oxyrhinchum were observed at different phenological stages (p < 0.01). From the initial leaf expansion stage to the full leaf expansion stage, the C concentration in the leaves of E. oxyrhinchum under drought pretreatment was significantly higher than that in the control treatment (Figure 6A). However, at the initial flowering and full blooming stages, the C concentration under drought pretreatment was significantly lower than that of the control treatment. In addition, the N concentration in the leaves of E. oxyrhinchum exhibited a trend of initially increased and then decreased (Figure 6C). Before the initial flowering stage, the N concentration in the leaves of E. oxyrhinchum under the drought pretreatment was significantly higher than that in the control treatment. Similarly, from the perspective of the entire life history, the P concentration in the leaves of E. oxyrhinchum under drought pretreatment was significantly higher than that in the control treatment (Figure 6E).
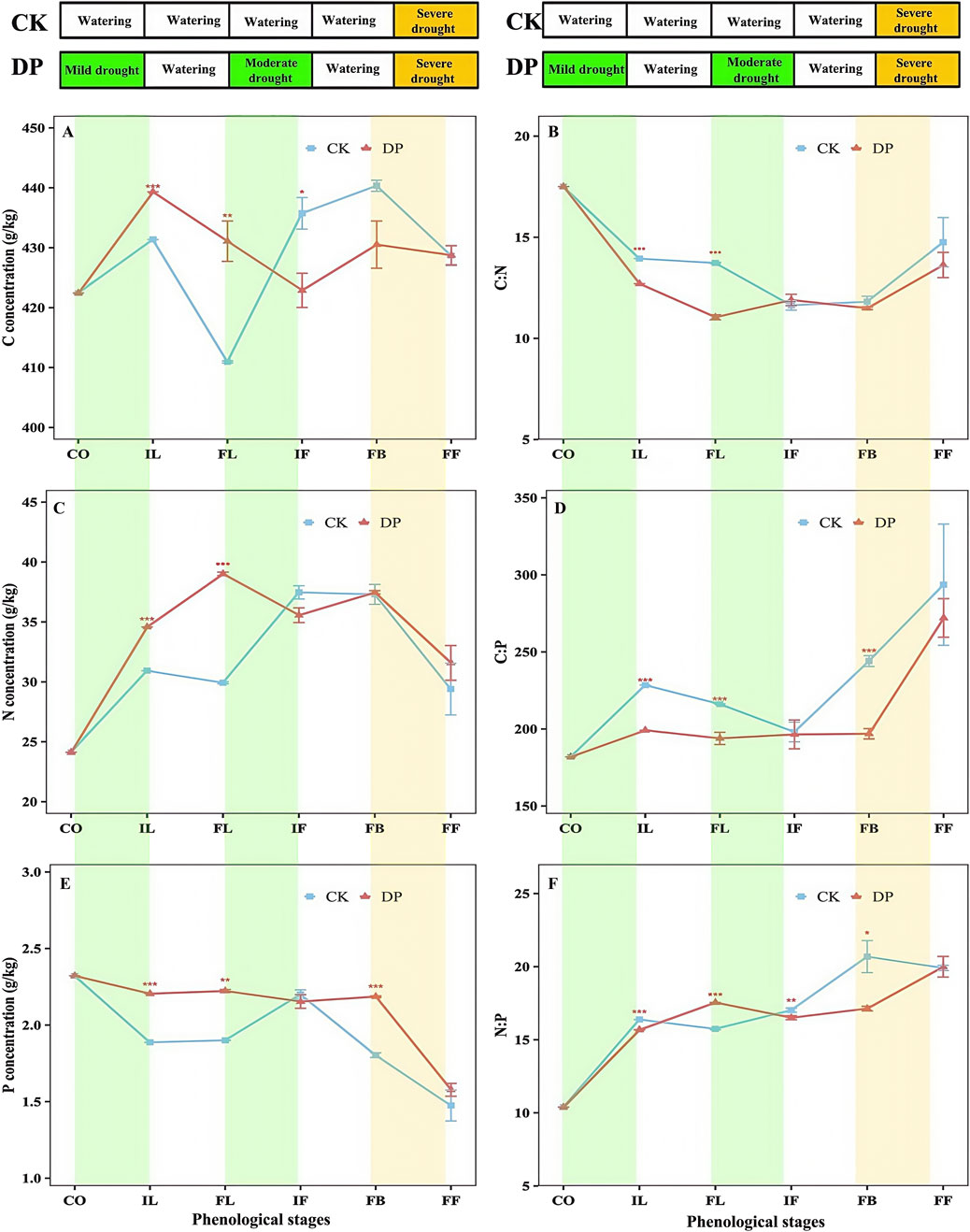
Figure 6. Effects of drought pretreatment on the C, N, and P stoichiometric characteristics of Erodium oxyrhinchum. Note: C concentration (A); C:N ratio (B); N concentration (C); C:P ratio (D); P concentration (E); N:P ratio (F); Control treatment, CK; Drought pretreatment, DP; Cotyledon stage, CO; Initial leaf expansion stage, IL; Full leaf expansion stage, FL; Initial flowering stage, IF; Full blooming stage, FB; Full fruiting stage, FF; *p < 0.05, **p < 0.01, ***p < 0.001.
The C: N ratio in the leaves of E. oxyrhinchum initially decreased and then increased as the phenological stage progressed, whereas the C:P and N:P exhibited an increasing trend. From the cotyledon stage to the full leaf expansion stage, the C: N in the leaves of E. oxyrhinchum under drought pretreatment was significantly lower than that in the control treatment (p < 0.001, Figure 6B). From the perspective of the entire life history, the C:P in the leaves of E. oxyrhinchum under drought pretreatment was also significantly lower than that in the control treatment (Figure 6D). Similarly, from the cotyledon stage to the full blooming stage, the N:P under the drought pretreatment was significantly lower than that under the control treatment (p < 0.05, Figure 6F).
From the perspective of the entire life history, the N and P concentration in the leaves of E. oxyrhinchum exhibited greater variation than the C concentration. Specifically, the coefficient of variation for C concentration in the leaves of E. oxyrhinchum between the control treatment and drought pretreatment was less than 5%, indicating weak variation (Table 3). However, from the full blooming stage to the full fruiting stage, the coefficient of variation for the N concentration under the control treatment and the P concentration under the drought pretreatment was greater than 15%, indicating moderate variation.
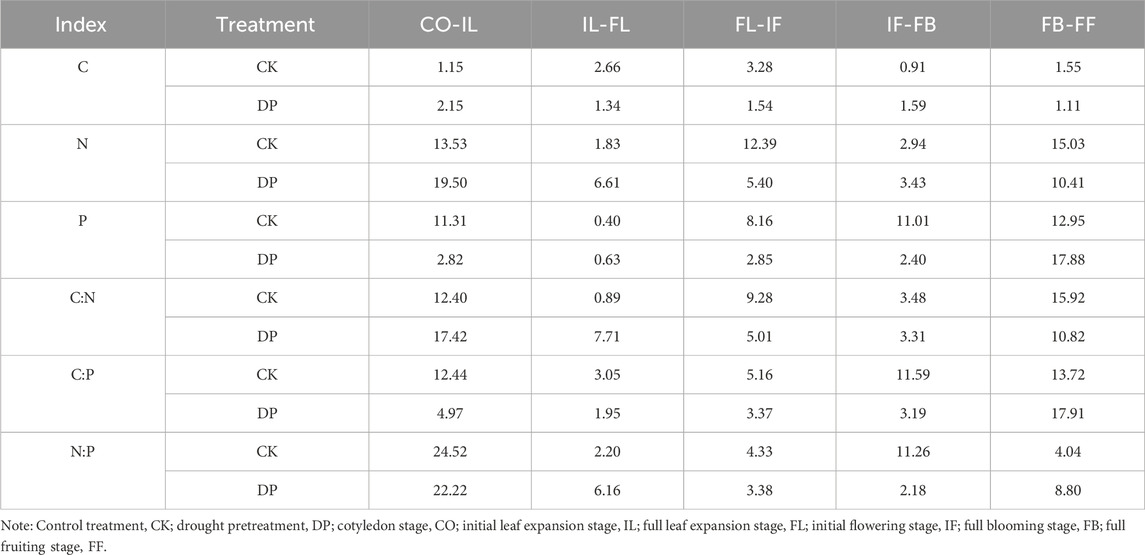
Table 3. Coefficient of variation of C, N, and P stoichiometric characteristics of Erodium oxyrhinchum at different phenological stages.
This study aimed to investigate the effects of drought pretreatment on the survival, phenology, morphology, biomass, and stoichiometric characteristics of E. oxyrhinchum in the Gurbantunggut Desert. The results indicated that E. oxyrhinchum exposed to drought pretreatment exhibited significantly earlier phenological stages and lower survival percentages, and the reproductive biomass and individual biomass were significantly greater than those of the control treatment, partially supporting the first hypothesis. Furthermore, plants exposed to drought pretreatment needed to absorb more phosphorus than those exposed to the control treatment, but the differences in C and N in leaves did not reach a significant level, thus partially confirming the second hypothesis.
Phenology represents cyclical changes in vegetation and is a sensitive biological indicator of climate change (Li et al., 2021). This study revealed that the phenological stages of E. oxyrhinchum subjected to drought pretreatment were markedly accelerated. For instance, the full leaf expansion stage under drought pretreatment was 5.25 ± 1.2 days earlier than that of the control treatment. Early leafing allows plants to initiate photosynthesis sooner, thereby enhancing biomass accumulation (Ganjurjav et al., 2021; Amini et al., 2023; Wu and Yang, 2024). Similarly, the onset of flowering and fruiting of E. oxyrhinchum under drought pretreatment also occurred significantly earlier than in the control treatment. This may be because earlier leafing under drought pretreatment induced plants to enter the reproductive stage. Previous studies also have indicated that desert ephemeral plants employ a “drought escape strategy” by accelerating phenological stages to complete life history (Franks, 2011; Ding et al., 2022). These findings not only highlight the positive response of ephemeral plants to drought stress but also demonstrate that drought pretreatment can accelerate the rapid transitions of phenological stages, thereby influencing the life history of ephemeral plants.
The survival percentage reflects the adaptability of plants to environmental stress and is a valuable indicator of drought resistance in seedlings (He et al., 2024; Chen et al., 2025). In this study, the survival percentage of E. oxyrhinchum from March 20 to April 19 did not show a significant decrease trend, but the survival percentage of E. oxyrhinchum significantly decline after the full leaf expansion stage. The survival percentage of E. oxyrhinchum under drought pretreatment exhibited a faster and earlier declining trend than that of the control treatment. With the rapid growth of E. oxyrhinchum, the difference in survival percentage may be related to the decrease in soil water content and intensification of intraspecific competition. Similarly, Guo et al. (2020) investigated the combined effects of drought and intraspecific competition on the growth of C. lanceolata, and the results indicated that intense competition imposed by neighbours was a great threat to the survival of young C. lanceolata plants under prolonged drought. Furthermore, the trend in soil water content revealed lower moisture levels under drought pretreatment (Supplementary Figure 1), further suggesting intensified intraspecific competition among plants. Therefore, ephemeral plants usually adopt a strategy of decreasing their survival percentage, reducing intraspecific competition, and shortening their life history to cope with intense drought stress.
Plants respond to environmental stress by adjusting their morphological traits and biomass allocation (Freschet et al., 2018; Geleta et al., 2024). Our study demonstrated that plants exposed to drought pretreatment showed more significant morphological changes than those in the control treatment (Figure 4). Specifically, from the initial leaf expansion to the full blooming stage, the plant height of E. oxyrhinchum under the drought pretreatment was significantly higher than that of the control treatment. Additionally, during the initial leaf expansion and flowering stages, the number of leaves under the drought pretreatment was significantly higher than that in the control treatment. The root length of E. oxyrhinchum under drought pretreatment was also significantly greater than that of the control treatment during the initial leaf expansion stage. This suggests that ephemeral plants under drought pretreatment could prioritise resource allocation to their roots, allowing them to extend deeper or wider soil layers to obtain more water and nutrients and improve their adaptability. Similarly, Xu et al. (2023) found that drought stress prompted Alhagi sparsifolia to allocate more resources for root growth, thereby enhancing its survival and adaptability in desert environments.
The positive correlation between morphological traits and biomass has been validated extensively (Lu et al., 2014; Chen et al., 2019b). In this study, correlation analysis also revealed a positive correlation between plant height, root length, leaf area, and leaf number, and the individual biomass of E. oxyrhinchum (Table 4). In addition, we found that the individual and reproductive biomass of E. oxyrhinchum under drought pretreatment was significantly higher than that of the control treatment. This suggests that moderate drought stress did not significantly decrease the biomass of E. oxyrhinchum but rather promoted its growth of E. oxyrhinchum, demonstrating an overcompensation effect. Similarly, in agricultural practices, managers often apply mild short-term drought stress to specific crop root zones, inducing a stress response through alternating partial root-zone irrigation, thereby promoting biomass accumulation and improving yield (Consoli et al., 2017; Fu et al., 2017; Liu et al., 2020b; Wang et al., 2024). The persistence of annual plant populations depends entirely on seed production (Lan and Zhang, 2008). In this study, drought pretreatment significantly increased the reproductive output of E. oxyrhinchum, which is likely related to the accelerated transition of ephemeral plants from vegetative to reproductive growth under drought stress. In the early spring in the Gurbantunggut Desert, soil moisture undergoes a gradual change from high to low, indicating that drought stress continues to intensify (Huang et al., 2015b; Chen et al., 2019d; Lu et al., 2022). To ensure population persistence, ephemeral plants must complete their reproductive output before the onset of extreme drought or hot summers (Mu et al., 2021; Lu et al., 2022). Therefore, the overcompensation effect in E. oxyrhynchum under drought stress is an important strategy for population persistence and community stability in the Gurbantunggut Desert.
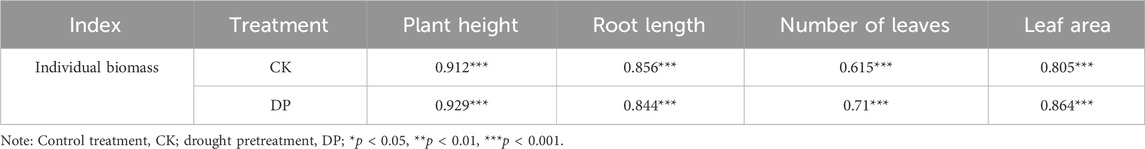
Table 4. The correlation analysis between individual biomass and morphological traits of Erodium oxyrhinchum under different drought treatments.
C, N, and P are essential nutrients that play critical roles in plant growth and development, influencing key physiological and ecological processes such as photosynthesis, transpiration, and reproductive growth (Zhang et al., 2021; Costa et al., 2024). Our study revealed that the leaf C concentration under drought pretreatment was significantly higher than that of the control treatment during the initial and full leaf expansion stages. This suggests that drought pretreatment increases C accumulation in the early stages of growth, which is likely because plants adjust their energy storage structure under drought conditions to maintain survival and growth. A study on tropical plant seedlings found that plants allocate limited carbon resources to nonstructural carbohydrates under drought stress, which is beneficial for maintaining normal metabolic requirements and rapid recovery after drought stress (O’Brien et al., 2014). Additionally, the coefficient of variation for leaf C concentration under the control and drought treatments was less than 5%. This may be because carbon is a fundamental element of plant structure, and the C concentration in leaves generally remains relatively stable. Our study showed that the leaf N concentration of E. oxyrhinchum in the control and drought treatments exhibited a trend of first increased and then decreased. This may be because the growth rate of the leaves reached its maximum and required higher nitrogen absorption to synthesise proteins before the flowering stage. However, as plants grow, more N is allocated to the reproductive organs, resulting in a relative decrease in the leaf N concentration. In addition, the leaf P concentration under drought pretreatment was significantly higher than that of the control treatment. This may be due to the enhanced resistance and overcompensation effects in plants exposed to drought stress, which require more phosphorus for rRNA synthesis to support plant growth (Elser et al., 2010).
The leaf N:P ratio can be used as an indicator to assess whether plant productivity is limited by nutrients (Yuan et al., 2011; Wang et al., 2014; Lu et al., 2023). Generally, it is accepted that when N:P < 14, plant growth is more limited by nitrogen, when 14 < N:P < 16, plant growth is simultaneously limited by nitrogen and phosphorus, and when N:P > 16, phosphorus becomes the primary limiting factor for plant growth (Güsewell and Koerselman, 2002; Sardans et al., 2012; Yang et al., 2018). Our study found that leaf N:P under drought pretreatment was less than 14 during the cotyledon stage, 14 < N:P < 16 during the initial leaf expansion stage, and N:P > 16 from the full leaf expansion stage to the full fruiting stage (Figure 6F). Similarly, the leaf N:P in the control treatment was greater than 16 from the leaf expansion stage to the full fruiting stage. These findings indicated that the growth of E. oxyrhinchum during the cotyledon stage was primarily limited by N. After the initial leaf expansion stage, N and P limit plant growth simultaneously. The effect of P on plant growth gradually increased during the full leaf expansion, flowering, and fruiting stages. Therefore, it is necessary to optimise ecological restoration measures based on the nutritional needs of plants and improve the effectiveness of restoration.
Plants are often exposed to various degrees of drought under natural conditions, which are likely to affect their growth and development. Our study revealed that E. oxyrhinchum exposed to drought stress significantly accelerated the transition of phenological stages and shortened its life history. Meanwhile, the survival percentage of E. oxyrhinchum under drought pretreatment significantly decreased but showed overcompensation effects on morphological traits and biomass accumulation. This indicates that ephemeral desert plants exhibit a trade-off between survival and growth when subjected to drought stress. In addition, from the perspective of stoichiometric characteristics, E. oxyrhinchum subjected to drought pretreatment required more phosphorus to enhance its resistance to severe drought in the Gurbantunggut desert. Therefore, this study provides novel insights into the restoration of desert ecosystems based on plant nutritional requirements in the context of climate change.
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
QL: Data curation, Formal Analysis, Software, Visualization, Writing–original draft, Conceptualization, Investigation, Writing–review and editing. HL: Conceptualization, Writing–original draft, Methodology, Project administration. CG: Conceptualization, Methodology, Writing–original draft, Formal Analysis, Resources. QZ: Conceptualization, Formal Analysis, Methodology, Writing–riginal draft, Validation. TP: Formal Analysis, Writing–riginal draft, Data curation, Investigation. ZC: Writing–original draft, Conceptualization, Methodology, Project administration. YC: Project administration, Writing–original draft, Data curation, Formal Analysis, Funding acquisition, Software, Validation, Visualization, Writing–review and editing.
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This work was funded by the National Natural Sciences Foundation of China (32101262), Shandong Provincial Colleges and Universities Youth Innovation Team Development Plan Project (2024KJG012), Key Laboratory of Lake and Watershed Science for Water Security (2024SKL017), and the visiting research fund for teachers of ordinary undergraduate universities in Shandong Province.
We would like to express our sincere gratitude to the members of our research team for their invaluable support and contribution in collecting samples for the field experiment. We also extend our deep appreciation to the editors and reviewers of Frontiers for their thoughtful handling of our manuscript and for providing constructive feedback that greatly improved the quality of our work.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fgene.2025.1534894/full#supplementary-material
Amini, A., Majidi, M. M., Mokhtari, N., and Ghanavati, M. (2023). Drought stress memory in a germplasm of synthetic and common wheat: antioxidant system, physiological and morphological consequences. Sci. Rep. 13 (1), 8569. doi:10.1038/s41598-023-35642-2
Brunet, J., Inouye, D. W., Rankin, E. E. W., and Giannini, T. C. (2024). Global change aggravates drought, with consequences for plant reproduction. Ann. Bot., mcae186. doi:10.1093/aob/mcae186
Chen, C. J., Bao, M. F., Wang, W. H., Shang, J. H., Zeng, Y. X., and Sha, X. D. (2025). Current situation and prospects for drought-resistance breeding in Medicago sativa. Acta Prataculturae Sin. 34 (3), 204. doi:10.11686/cyxb2024105
Chen, Y. F., Cao, Q. M., Li, D. X., Liu, H. L., and Zhang, D. Y. (2019d). Effects of temperature and light on seed germination of ephemeral plants in the Gurbantunggut Desert, China: implications for vegetation restoration. J. Arid Land 11, 916–927. doi:10.1007/s40333-019-0027-4
Chen, Y. F., Shi, X., Zhang, L. W., Baskin, J. M., Baskin, C. C., Liu, H. L., et al. (2019c). Effects of increased precipitation on the life history of spring- and autumn-germinated plants of the cold desert annual Erodium oxyrhynchum (Geraniaceae). AoB PLANTS 11 (2), plz004. doi:10.1093/aobpla/plz004
Chen, Y. F., Zhang, L. W., Shi, X., Ban, Y., Liu, H. L., and Zhang, D. Y. (2019b). Life history responses of spring-and autumn-germinated ephemeral plants to increased nitrogen and precipitation in the Gurbantunggut Desert. Sci. Total Environ. 659, 756–763. doi:10.1016/j.scitotenv.2018.12.368
Chen, Y. F., Zhang, L. W., Shi, X., Liu, H. L., and Zhang, D. Y. (2019a). Life history responses of two ephemeral plant species to increased precipitation and nitrogen in the Gurbantunggut Desert. PeerJ 7 (4), e6158. doi:10.7717/peerj.6158
Consoli, S., Stagno, F., Vanella, D., Boaga, J., Cassiani, G., and Roccuzzo, G. (2017). Partial root-zone drying irrigation in orange orchards: effects on water use and crop production characteristics. Eur. J. Agron. 82, 190–202. doi:10.1016/j.eja.2016.11.001
Costa, M. G., de Mello Prado, R., Palaretti, L. F., and de Souza Júnior, J. P. (2024). The effect of abiotic stresses on plant C: N: P homeostasis and their mitigation by silicon. Crop J. 12 (2), 340–353. doi:10.1016/j.cj.2023.11.012
Ding, S. J., Cheng, M. X., Xie, S. Q., Wang, X. S., Hao, X. M., and Zhuang, L. (2022). Response of growth characteristics and chlorophyll fluorescence characteristics of ephemeral plants to different soil types. Acta Agrestia Sin. 30 (12), 3308–3316. doi:10.11733/j.issn.1007-0435
Elser, J. J., Fagan, W. F., Kerkhoff, A. J., Swenson, N. G., and Enquist, B. J. (2010). Biological stoichiometry of plant production: metabolism, scaling and ecological response to global change. New Phytol. 186 (3), 593–608. doi:10.1111/j.1469-8137.2010.03214.x
Erfan, A., Li, J., Li, H., and Zhuang, W. W. (2023). Comparison of nodule characteristics of two astragalus species and their relationship with soil factors. Acta Agrestia Sin. 31 (7), 2177–2185. doi:10.11733/j.issn.1007-0435.2023.07.029
Franks, S. J. . (2011). Plasticity and evolution in drought avoidance and escape in the annual plant Brassica rapa. New Phytol. 190 (1), 249–257. doi:10.1111/j.1469-8137.2010.03603.x
Freschet, G. T., Violle, C., Bourget, M. Y., Scherer-Lorenzen, M., and Fort, F. (2018). Allocation, morphology, physiology, architecture: the multiple facets of plant above- and below-ground responses to resource stress. New Phytol. 219, 1338–1352. doi:10.1111/nph.15225
Fu, F. B., Li, F. S., and Kang, S. Z. (2017). Alternate partial root-zone drip irrigation improves water and nitrogen use efficiencies of sweet-waxy maize with nitrogen fertigation. Sci. Rep. 7 (1), 17256–17310. doi:10.1038/s41598-017-17560-2
Fuente, J. L. L., Zunzunegui, M., and Barradas, M. C. D. (2023). Physiological responses to water stress and stress memory in Argania spinosa. Plant Stress 7, 100133. doi:10.1016/j.stress.2023.100133
Ganjurjav, H., Gornish, E., Hu, G. Z., Wu, J. S., Wan, Y. F., Li, Y., et al. (2021). Phenological changes offset the warming effects on biomass production in an alpine meadow on the Qinghai-Tibetan Plateau. J. Ecol. 109 (2), 1014–1025. doi:10.1111/1365-2745.13531
Gao, C. K., Wu, P. N., Wang, Y. L., Wen, P. F., Guan, X. K., and Wang, T. C. (2024). Drought and rewatering practices improve adaptability of seedling maize to drought stress by a super-compensate effect. Heliyon 10 (20), e39602. doi:10.1016/j.heliyon.2024.e39602
Geleta, R. J., Roro, A. G., and Terfa, M. T. (2024). Phenotypic and yield responses of common bean (Phaseolus vulgaris l.) varieties to different soil moisture levels. BMC Plant Biol. 24 (1), 242. doi:10.1186/s12870-024-04856-5
Ghannoum, O. (2009). C4 photosynthesis and water stress. Ann. Bot. 4, 635–644. doi:10.1093/aob/mcn093
Guo, P. R., Wu, L. L., Wang, Y., Liu, D., and Li, J. A. (2023). Effects of Drought stress on the morphological structure and flower organ physiological characteristics of camellia oleifera flower buds. Plants 12 (13), 2585–7747. doi:10.3390/plants12132585
Guo, Q. X., Wu, X. Y., Korelainen, H., and Li, C. Y. (2020). Stronger intra-specific competition aggravates negative effects of drought on the growth of Cunninghamia lanceolata. Environ. Exp. Bot. 175, 104042. doi:10.1016/j.envexpbot.2020.104042
Güsewell, S., and Koerselman, W. (2002). Variation in nitrogen and phosphorus concentrations of wetland plants. Perspect. Plant Ecol. Evol. Syst. 5 (1), 37–61. doi:10.1078/1433-8319-0000022
Haghpanah, M., Hashemipetroudi, S., Arzani, A., and Araniti, F. (2024). Drought tolerance in plants: physiological and molecular responses. Plants 13 (21), 2962. doi:10.3390/plants13212962
Haider, S., Bibi, K., Munyaneza, V., Zhang, H., Zhang, W., Ali, A., et al. (2024). Drought-induced adaptive and ameliorative strategies in plants. Chemosphere 364, 143134. doi:10.1016/j.chemosphere.2024.143134
He, M., Jiang, Y. H., Zhu, C. F., Wang, Y. H., Wu, Z. X., and Zhao, J. X. (2024). Identification and screening of turnip germplasm resources for drought resistance. Chin. Cucurbits Veg. 37 (08), 100–108. doi:10.16861/j.cnki.zggc.202423.0721
Hofer, D., Suter, M., Buchmann, N., and Lüscher, A. (2017). Nitrogen status of functionally different forage species explains resistance to severe drought and post-drought overcompensation. Agric. Ecosyst. & Environ. 236, 312–322. doi:10.1016/j.agee.2016.11.022
Hosseinizadeh, A., SeyedKaboli, H., Zareie, H., Akhondali, A., and Farjad, B. (2015). Impact of climate change on the severity, duration, and frequency of drought in a semi-arid agricultural basin. Geoenvironmental Disasters 2, 23–29. doi:10.1186/s40677-015-0031-8
Huang, G., Li, Y., and Padilla, F. M. (2015b). Ephemeral plants mediate responses of ecosystem carbon exchange to increased precipitation in a temperate desert. Agric. For. Meteorology 201, 141–152. doi:10.1016/j.agrformet.2014.11.011
Huang, J. P., Yu, H. P., Guan, X. D., Guan, X., Wang, G. Y., and Guo, R. X. (2015a). Accelerated dryland expansion under climate change. Nat. Clim. Change 6, 166–171. doi:10.1038/nclimate2837
Jiang, P. P., Han, X. J., Liu, Z. Y., Fan, S. J., and Zhang, X. J. (2023). C: N: P stoichiometric variations of herbs and its relationships with soil properties and species relative abundance along the Xiaokai River irrigation in the Yellow River Delta, China. Front. Plant Sci. 14, 1130477. doi:10.3389/fpls.2023.1130477
Lan, H. Y., and Zhang, F. C. (2008). Reviews on special mechanisms of adaptability of early-spring ephemeral plants to desert habitats in Xinjiang. Acta Bot. Boreal-Occident Sin. 28 (7), 8. doi:10.3321/j.issn:1000-4025.2008.07.032
Li, P., Liu, Z. L., Zhou, X. L., Xie, B. G., Li, Z. W., Luo, Y. P., et al. (2021). Combined control of multiple extreme climate stressors on autumn vegetation phenology on the Tibetan Plateau under past and future climate change. Agric. For. Meteorology 308-309 (4), 108571. doi:10.1016/j.agrformet.2021.108571
Lin, R. P., Zhou, T. J., and Qian, Y. (2014). Evaluation of global monsoon precipitation changes based on five reanalysis datasets. J. Clim. 27 (3), 1271–1289. doi:10.1175/JCLI-D-13-00215.1
Liu, C. Y., Wu, J. W., Gu, J. Y., and Duan, H. J. (2024). Response of non-structural carbohydrates and carbon, nitrogen, and phosphorus stoichiometry in Pinus yunnanensis seedlings to drought Re-watering. Forests 15 (11), 1864. doi:10.3390/f15111864
Liu, J. G., Liu, W. G., Long, X. E., Chen, Y. G., Huang, T. W., Huo, J. S., et al. (2020a). Effects of nitrogen addition on C: N:P stoichiometry in moss crust-soil continuum in the N-limited Gurbantunggut Desert, Northwest China. Eur. J. Soil Biol. 98, 103174. doi:10.1016/j.ejsobi.2020.103174
Liu, R., Yang, Y., Wang, Y. S., Wang, X. C., Rengel, Z., Zhang, W. J., et al. (2020b). Alternate partial root-zone drip irrigation with nitrogen fertigation promoted tomato growth, water and fertilizer-nitrogen use efficiency. Agric. Water Manag. 233, 233106049–106049. doi:10.1016/j.agwat.2020.106049
Lu, J. J., Tan, D. Y., Baskin, J. M., and Baskin, C. C. (2014). Germination season and watering regime, but not seed morph, affect life history traits in a cold desert diaspore-heteromorphic annual. Plos One 9 (7), e102018. doi:10.1371/journal.pone.0102018
Lu, J. N., Zhao, X. Y., Wang, S. K., Feng, S., Ning, Z. Y., Wang, R. X., et al. (2023). Untangling the influence of abiotic and biotic factors on leaf C, N, and P stoichiometry along a desert-grassland transition zone in northern China. Sci. Total Environ. 884, 163902. doi:10.1016/j.scitotenv.2023.163902
Lu, Y. T., Liu, H. L., Chen, Y. F., Zhang, L., Kudusi, K., and Song, J. H. (2022). Effects of drought and salt stress on seed germination of ephemeral plants in desert of northwest China. Front. Ecol. Evol. 10, 1026095. doi:10.3389/fevo.2022.1026095
Lv, Z. Y., Zhang, H., Huang, Y., Zhu, L., Yang, X., Wu, L. F., et al. (2024). Drought priming at seedling stage improves photosynthetic performance and yield of potato exposed to a short-term drought stress. J. Plant Physiology 292, 154157. doi:10.1016/j.jplph.2023.154157
Mu, X. H., Huang, G., Li, Y., Zheng, X. J., Xu, G. Q., Wu, X., et al. (2021). Population dynamics and life history response to precipitation changes for a desert ephemeral plant with biseasonal germination. Front. Plant Sci. 12, 625475. doi:10.3389/fpls.2021.625475
O’Brien, M. J., Leuzinger, S., Philipson, C. D., Tay, J., and Hector, A. (2014). Drought survival of tropical tree seedlings enhanced by non-structural carbohydrate levels. Nat. Clim. Change 4 (8), 710–714. doi:10.1038/nclimate2281
Sardans, J., Rivas-Ubach, A., and Peñuelas, J. (2012). The elemental stoichiometry of aquatic and terrestrial ecosystems and its relationships with organismic lifestyle and ecosystem structure and function: a review and perspectives. Biogeochemistry 111 (1-3), 1–39. doi:10.1007/s10533-011-9640-9
Sun, Y., Li, X. L., and Feng, G. (2008). Effect of arbuscular mycorrhizal colonization on ecological functional traits of ephemerals in the Gurbantonggut desert. Symbiosis 46 (3), 121–127. doi:10.1634/stemcells.2008-0642
Walters, M. B., Kunkle, J. M., Kobe, R. K., and Farinosi, E. J. (2023). Seedling drought responses governed by root traits, site-soil moisture regimes and overstory competition-facilitation. For. Ecol. Manag. 544, 121159. doi:10.2139/ssrn.4305875
Wang, M., Murphy, M. T., and Moore, T. R. (2014). Nutrient resorption of two evergreen shrubs in response to long-term fertilization in a bog. Oecologia 174 (2), 365–377. doi:10.1007/s00442-013-2784-7
Wang, S. S., and Zhang, Y. M. (2010). Morphological characters of Erodium oxyrrhynchum diaspore. Chin. J. Ecol. 29 (5), 855–861. doi:10.3724/SP.J.1035.2010.01150
Wang, Y., Sun, Q. C., Zhang, S. Z., Jiang, Z. P., Wen, B. H., Ge, X. Y., et al. (2024). Research progress on the application of alternate partial root-zone irrigation in agricultural production. Acta Agrestia Sin. 32 (4), 995–1011. doi:10.11733/j.issn.1007-0435.2024.04.003
Wu, H., and Yang, Z. (2024). Effects of Drought Stress and postdrought rewatering on winter wheat: a meta-analysis. Agronomy 14 (2), 298. doi:10.3390/agronomy14020298
Xu, M. Q., Gao, Y. J., Zhang, Z. H., Huang, C., and Zeng, F. J. (2023). Effects of drought stress on growth and physiology of Alhagi sparsifolia seedlings. Arid Zone Res. 40 (2), 257–267. doi:10.13866/j.azr.2023.02.10
Yang, Y., Liu, B., and An, S. (2018). Ecological stoichiometry in leaves, roots, litters and soil among different plant communities in a desertified region of Northern China. Catena 166, 166328–166338. doi:10.1016/j.catena.2018.04.018
Yuan, Z. Y., Chen, H. Y. H., and Reich, P. B. (2011). Global-scale latitudinal patterns of plant fine-root nitrogen and phosphorus. Nat. Commun. 2 (1), 344. doi:10.1038/ncomms1346
Zang, Y. X., Min, X. J., Dios, V. R., Ma, J. Y., and Sun, W. (2020). Extreme drought affects the productivity, but not the composition, of a desert plant community in Central Asia differentially across microtopographies. Sci. Total Environ. 717, 137251. doi:10.1016/j.scitotenv.2020.137251
Zhang, J., Hao, F. H., Wu, Z. F., Li, M. W., Zhang, X., and Fu, Y. S. (2023). Response of vegetation phenology to extreme climate and its mechanism. Acta Geogr. Sin. 78 (9), 2241–2255. doi:10.11821/dlxb202309008
Zhang, J. H., Li, M. X., Xu, L., Zhu, J. X., Dai, G. H., and He, N. P. (2021). C: N: P stoichiometry in terrestrial ecosystems in China. Sci. Total Environ. 795, 148849. doi:10.1016/j.scitotenv.2021.148849
Zhang, L., Liu, H. L., Zhang, L. W., Chen, Y. F., and Baskin, C. C. (2022b). Effects of increased precipitation on C, N and P stoichiometry at different growth stages of a cold desert annual. Glob. Ecol. Conservation 37, e02158. doi:10.1016/j.gecco.2022.e02158
Zhang, L. Y., and Chen, C. D. (2002). On the general characteristics of plant diversity of Gurbantunggut sandy desert. Acta Ecol. Sin. 22 (11), 1923–1932.
Zhang, Y. Y., Meng, H. H., Zhou, X. B., Yin, B. F., Zhou, D. Q., and Tao, Y. (2022a). Biomass allocation patterns of an ephemeral species (Erodium oxyrhinchum) in different habitats and germination types in the Gurbantunggut Desert, China. Arid Zone Res. 39 (2), 10. doi:10.13866/j.azr.2022.02.21
Keywords: drought pretreatment, overcompensation effect, Erodium oxyrhinchum, ephemeral plant, the Gurbantunggut desert
Citation: Liu Q, Li H, Gong C, Zhang Q, Pan T, Cao Z and Chen Y (2025) Effects of drought pretreatment on the morphology traits, biomass, and stoichiometric characteristics of the desert ephemeral plant. Front. Genet. 16:1534894. doi: 10.3389/fgene.2025.1534894
Received: 26 November 2024; Accepted: 03 March 2025;
Published: 20 March 2025.
Edited by:
Yan Wang, Xinjiang University, ChinaReviewed by:
Ping Yue, Chinese Academy of Sciences (CAS), ChinaCopyright © 2025 Liu, Li, Gong, Zhang, Pan, Cao and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yanfeng Chen, Y2hlbnlhbmZlbmdAcWZudS5lZHUuY24=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.