
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Genet., 16 September 2022
Sec. Stem Cell Research
Volume 13 - 2022 | https://doi.org/10.3389/fgene.2022.994163
This article is part of the Research TopicMusculoskeletal Disorders: from Mechanism to Translational Therapeutic StrategyView all 6 articles
 Wen-Bin Xu1
Wen-Bin Xu1 Vit Kotheeranurak2,3
Vit Kotheeranurak2,3 Huang-Lin Zhang4
Huang-Lin Zhang4 Jin-Yi Feng1
Jin-Yi Feng1 Jing-Wei Liu1
Jing-Wei Liu1 Chien-Min Chen5,6,7*†
Chien-Min Chen5,6,7*† Guang-Xun Lin1,4*†
Guang-Xun Lin1,4*† Gang Rui1,4*†
Gang Rui1,4*†Background: Osteoarthritis (OA) is a degenerative joint disease that seriously affects the quality of people. Unfortunately, the pathogenesis of OA has not been fully known. Therefore, this study aimed to construct a ceRNA regulatory network related to OA to explore the pathogenesis of OA.
Methods: Differentially expressed circRNAs (DEcircRNAs), microRNAs (DEmiRNAs), and mRNAs (DEmRNAs) were obtained from the Gene Expression Omnibus microarray data (GSE175959, GSE105027, and GSE169077). The miRNA response elements and target mRNAs were identified using bioinformatics approaches. Additionally, a circRNA–miRNA–mRNA network was established using Cytoscape version 3.8.0. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses of mRNAs in the network were conducted to explore the possible mechanisms underlying OA development. Protein–protein interaction (PPI) analysis was performed to determine the hub genes. Based on the hub genes, a sub network was constructed using Cytoscape 3.8.0 version. Finally, connectivity map (CMap) and drug–gene interaction database (DGIdb) analyses were performed to identify the potential therapeutic targets for OA.
Results: Altogether, five DEcircRNAs, 89 DEmiRNAs, and 345 DEmRNAs were identified. Moreover, a circRNA–miRNA–mRNA network was established using three circRNAs, seven miRNAs, and 37 mRNAs. GO and KEGG analyses demonstrated that the mRNAs in the network could be related to the occurrence and development of OA. PPI analysis was performed and six key genes, namely serpin family H member 1 [SERPINH1], collagen type VIII alpha 2 chain [COL8A2], collagen type XV alpha 1 chain [COL15A1], collagen type VI alpha 3 chain [COL6A3], collagen type V alpha 1 chain [COL5A1], and collagen type XI alpha 1 chain [COL11A1], were identified. Furthermore, a circRNA–miRNA–hub gene subnetwork was established in accordance with two circRNAs (hsa_circ_0075320 and hsa_circ_0051428), two miRNAs (hsa-miR-6124 and hsa-miR-1207-5p), and six hub genes (COL11A1, SERPINH1, COL6A3, COL5A1, COL8A2, and COL15A1). Finally, three chemicals (noscapine, diazepam, and TG100-115) based on CMap analysis and two drugs (collagenase Clostridium histolyticum and ocriplasmin) based on DGIdb were discovered as potential treatment options for OA.
Conclusion: This study presents novel perspectives on the pathogenesis and treatment of OA based on circRNA-related competitive endogenous RNA regulatory networks.
Osteoarthritis (OA) is a common arthrosis illness that may result in joint dysfunction, severely affecting the patient’s quality of life and increasing the social and economic burden (Cross et al., 2014; Kiadaliri et al., 2018). The prevalence of OA is high in middle-aged and elderly populations, and more than 50% of patients with knee pain are diagnosed with OA (Tang et al., 2016; Lespasio et al., 2017). However, the pathogenesis of OA is not yet fully understood. Therefore, understanding the pathogenesis of OA is essential to identify effective diagnostic and therapeutic targets.
Circular RNA (circRNAs) are neoteric noncoding RNAs (ncRNAs) with a circular form (Petkovic and Müller, 2015; Qu et al., 2015). In the absence of 5′ caps and 3′ tails, circRNAs are not influenced by exonucleases and become relatively stable (Li et al., 2015). CircRNAs, characterized by their specificity (Salzman et al., 2013) and high conservation (Vidal et al., 2016) are considered to play significant roles in various diseases (Kristensen et al., 2018).
Competing endogenous RNAs (ceRNAs) serve as transcripts for microRNA (miRNA) sponges and mutually regulate each other by binding to miRNAs (Qi et al., 2015). Currently, circRNAs are abundant in conserved miRNA response elements (MREs), which have emerged as novel targets in the ceRNA family (Zhong et al., 2018). Some circRNAs are involved in the occurrence and progression of various tumors via the ceRNA regulatory process (Wei et al., 2018; Xiong et al., 2018). However, whether circRNAs can also regulate the occurrence of OA via the ceRNA mechanism remains to be investigated.
In this study, some unknown circRNAs and their potential regulatory mechanisms in OA were analyzed using bioinformatic analysis. Figure 1 shows the flowchart of the entire procedure. First, differentially expressed circRNAs (DEcircRNAs), miRNAs (DEmiRNAs), and mRNAs (DEmRNAs) were obtained from the Gene Expression Omnibus (GEO) datasets. To characterize the role of DEcircRNAs as ceRNAs in OA, MREs and target mRNAs were predicted and a circRNA–miRNA–mRNA network was constructed. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) analyses were performed according to the mRNAs identified in the network to explore the possible mechanisms associated with OA. Subsequently, protein–protein interactions (PPIs) were used to identify related key mRNAs. Finally, connectivity map (CMap) and drug–gene interactions database (DGIdb) analyses were performed to identify potential compounds or drugs to treat OA.
Microarray data were extracted from the GEO database (Barrett et al., 2013). The circRNA expression profile was acquired from GSE175959 (three OA and three healthy knee tissues), the miRNA expression profile was obtained from GSE105027 (12 pairs of OA knee serum and healthy knee serum), and the mRNA expression profile was obtained from GSE169077 (five OA and six healthy knee tissues). Ethical approval was not required for our research because the data from GEO database are publicly available.
The specific process was as follows: 1) The platform files and series of matrix files of GSE175959, GSE105027, and GSE169077 were downloaded, and the corresponding probe names were transformed into international standard names (circRNA name, miRNA name, and gene symbol) by utilizing the practical extraction and report language (Perl); 2) The normalizeBetweenArrays package was used to normalize the data; 3) DEcircRNAs, DEmiRNAs, and DEmRNAs were selected using the Bioconductor Limma package, with the criteria as adjusted p-value < 0.05 and |log2(fold-change)| > 1.00(Ritchie et al., 2015); and 4) Heatmaps of DEcircRNAs, DEmiRNAs, and DEmRNAs were generated using the R package pheatmap.
The circBase (Glažar et al., 2014) and cancer-specific circRNAs database (CSCD) (Xia et al., 2018) were used to predict the target miRNA binding sites of DEcircRNAs. Furthermore, we analyzed the interactions of the target miRNAs of DEcircRNAs and DEmiRNAs, which were called as IDEmiRNAs.
IDEmiRNA–mRNA interactions were predicted according to the TargetScan (Agarwal et al., 2015) and miRDB databases (Wong and Wang, 2015). Only mRNAs that appeared in both databases were chosen and intersected with the DEmRNAs to obtain candidate target mRNAs, which were designated as IDEmRNAs.
circRNAs, IDEmiRNAs, and IDEmRNAs were selected to establish a circRNA–miRNA–mRNA regulatory network. We used Cytoscape 3.8.0 version to view this network (Su et al., 2014).
Before analysis, the gene symbols of mRNAs in the network were transformed into entrezIDs using R. Then, GO and KEGG analyses were implemented using the clusterProfiler package (Kanehisa and Goto, 2000; Yu et al., 2012). Both the p-value and q-values of GO analysis were <0.05, and the p-value of the KEGG analysis was <0.05.
CMap is a gene expression profiling database that uses the L1000 analysis platform to explore the network of interactions among drug/small-molecule compounds, genes, and disease states (Lamb et al., 2006). CMap analysis according to upregulated mRNAs in the ceRNA network was performed to identify candidate compounds for OA treatment. The connectivity scores ranged from –100 to 100, which was adapted to indicate closeness between the genes and compounds, with a positive score indicating a positive correlation with the uploaded genes and a negative score indicating a negative correlation with the uploaded genes.
According to the mRNAs discovered in the network, PPI analysis was performed using the Search Tool for the Retrieval of Interacting Genes/Proteins (STRING) (Szklarczyk et al., 2017). We then performed a visual analysis of the PPI network using Cytoscape 3.8.0 (Su et al., 2014). In addition, we also used the molecular complex detection (MCODE) (Bader and Hogue, 2003) plugin in Cytoscape software to defect essential modules and hub genes with the criterion of default parameters.
Correlation network analysis for hub genes was conducted using R software (igraph package). The correlation coefficient greater than 0.2 was considered to be relevant.
Drug–gene interaction analysis of hub genes was performed using DGIdb (https://dgidb.org/) (Griffith et al., 2013). DGIdb is an open-source project that provides information about genes associated with known or potential drugs. We uploaded the significant genes to DGIdb for matching with known drugs to identify the potential targets for OA treatment.
Three microarray datasets, GSE175959, GSE105027, and GSE169077, for circRNAs, miRNAs, and mRNAs, respectively, were used in our study. Based on the criteria set in advance [adjusted p-value < 0.05 and |log2(fold-change) | > 1.00], five DEcircRNAs (all upregulated) were identified in the GSE175959 dataset (Supplementary Figure S1A), 89 DEmiRNAs (all downregulated) were identified from the GSE105027 dataset (Supplementary Figure S1B), and 345 DEmRNAs (194 downregulated and 151 upregulated) were identified in the GSE169077 dataset (Supplementary Figure S1C). Supplementary Table S1 shows the basic information of these five DEcircRNAs, hsa_circ_0004662, hsa_circ_0051428, hsa_circ_0003312, hsa_circ_0008590, and hsa_circ_0075320. The circular structural styles are shown in Figure 2.
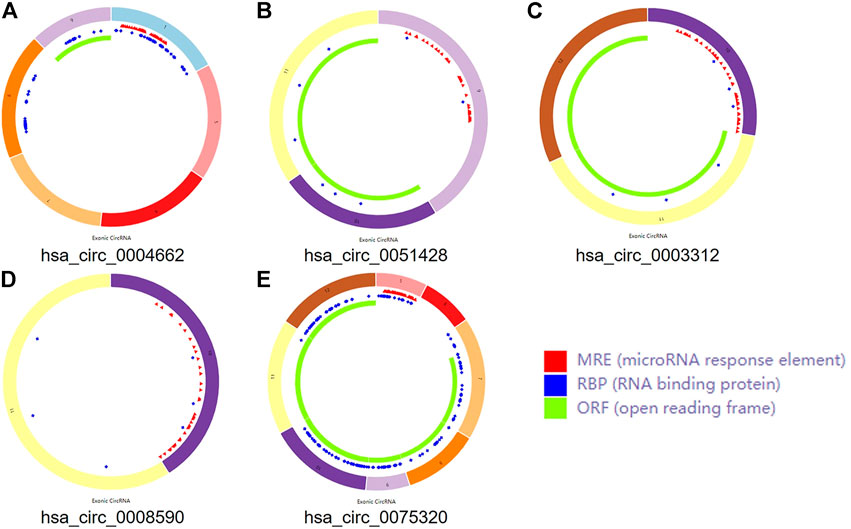
FIGURE 2. The structural patterns of the five circRNAs. (A) hsa_circ_0004662. (B) hsa_circ_0051428. (C) hsa_circ_0003312. (D) hsa_circ_0008590. (E) hsa_circ_0075320.
Some circRNAs act as sponges and play an important role in trapping miRNAs in non-tumorous tissues. To determine whether the five circRNAs showed similar functions in OA, MREs were predicted according to CSCD. A total of 239 MREs were identified after excluding duplicate target miRNAs. Ten significant IDEmiRNAs were obtained by intersecting 239 MREs with 89 DEmiRNAs (Supplementary Figure S2A).
Ten significant MREs associated with five circRNAs were identified. To better understand the mechanisms of miRNAs, the target mRNAs were predicted. In total, 3,902 target mRNAs were identified using the TargetScan and miRDB databases. Then, 83 overlapping IDEmRNAs were selected by the intersection of 345 DEmRNAs and 3,902 target mRNAs (Supplementary Figure S2B).
Five DEcircRNAs, 10 IDEmiRNAs, and 83 IDEmRNAs were identified by previous analysis. Based on the above results, a circRNA–miRNA–mRNA network was established, including three circRNAs (hsa_circ_0075320, hsa_circ_0003312, and hsa_circ_0051428), seven miRNAs (hsa-miR-6124, hsa-miR-6832-5p, hsa-miR-1207-5p, hsa-miR-4793-5p, hsa-miR-624-3p, hsa-miR-1225-5p, and hsa-miR-1229-5p), and 37 mRNAs (Figure 3). Heatmaps of circRNAs (Supplementary Figure S3A), miRNAs (Supplementary Figure S3B) and mRNAs (Supplementary Figure S3C) in the network are shown.
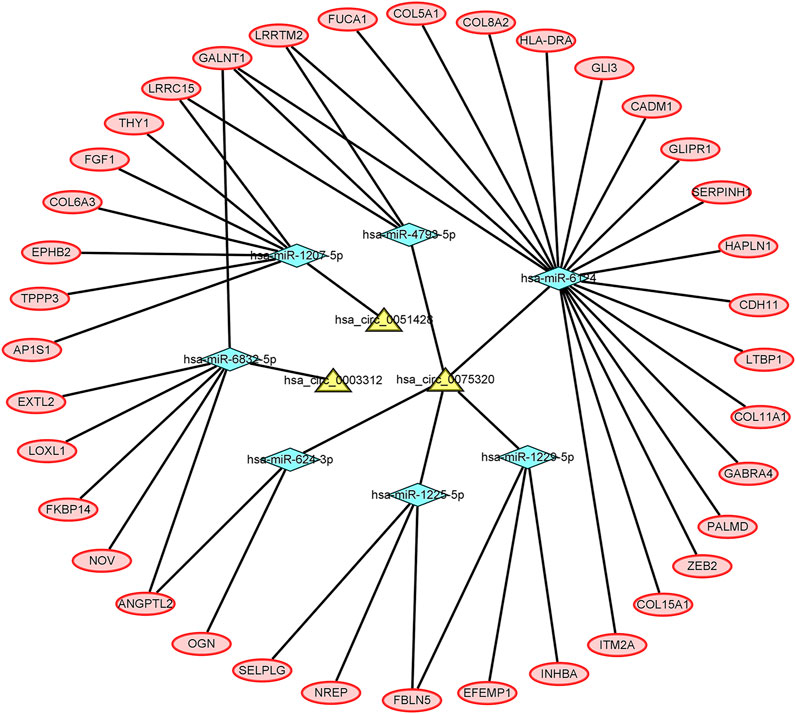
FIGURE 3. The circRNA–miRNA–mRNA network. The yellow nodes represent the circRNAs, the blue nodes represent the IDEmiRNAs, and the pink nodes represent the IDEmRNAs.
To better explore the potential mechanisms of mRNAs in the ceRNA regulatory network, GO enrichment analysis for the terms molecular function (MF), cellular component (CC), and biological process (BP), and KEGG pathway analysis were performed. The top 30 highly enriched GO and KEGG terms are shown in Figures 4, 5. In terms of MF, these genes were primarily clustered in the extracellular matrix structural constituent (n = 10), extracellular matrix structural constituent conferring tensile strength (n = 5), and glycosaminoglycan binding (n = 5) (Figure 4A). In terms of CC, these genes were primarily clustered in the collagen-containing extracellular matrix (n = 14) and endoplasmic reticulum lumen (n = 8) (Figure 4B). In terms of BP, these genes were primarily clustered in the extracellular matrix (n = 9) and extracellular structure (n = 9) organization (Figure 4C). According to the results of the KEGG analysis only five pathways, including protein digestion and absorption (n = 5), cell adhesion molecules (n = 3), transforming growth factor (TGF)-β signaling pathway (n = 2), Staphylococcus aureus infection (n = 2), lysosome, and other glycan degradation (n = 1), were enriched (Figure 5).
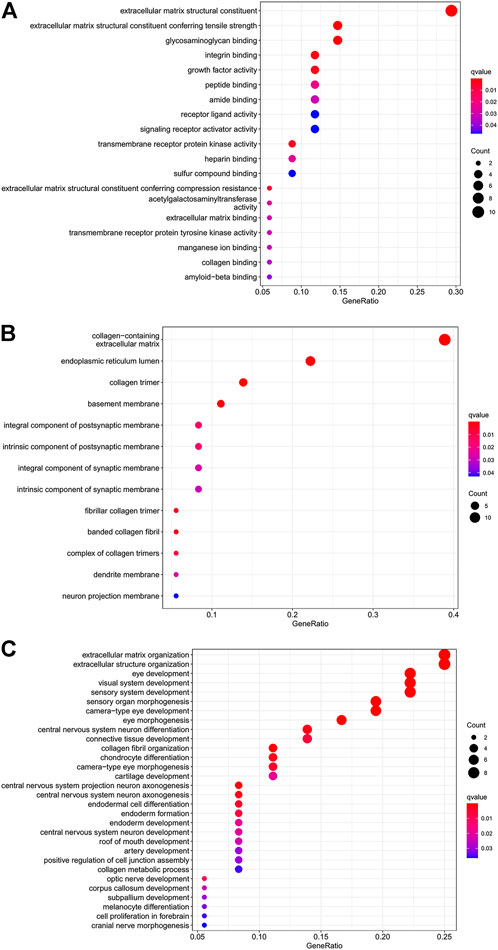
FIGURE 4. Dot plot of GO function enrichment analysis. (A) Molecular function analysis. (B) Cellular component analysis. (C) Biological process analysis.
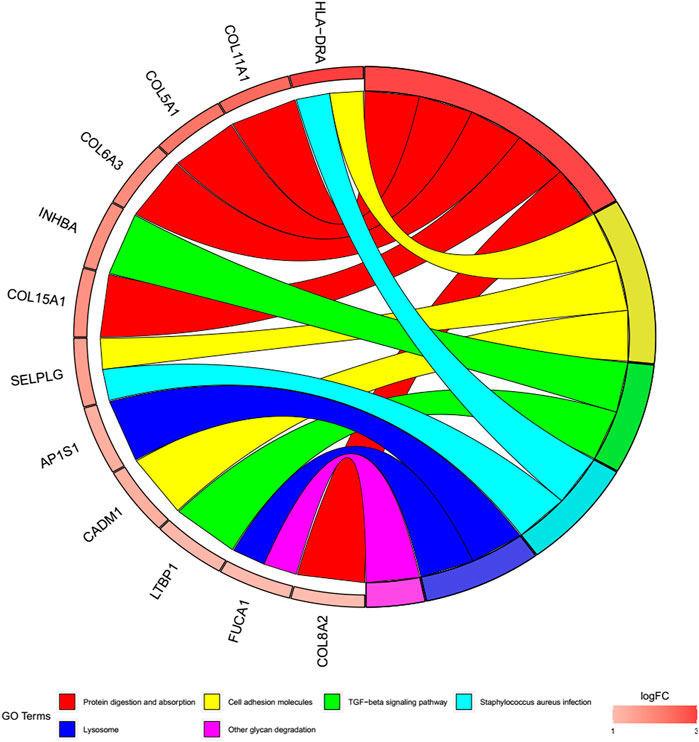
FIGURE 5. Cohort plot of KEGG pathways analysis shows that twelve genes are associated with six KEGG terms.
The mRNAs in the ceRNA network, including the 37 upregulated genes, were uploaded to the CMap website. After searching online, three chemicals (noscapine, diazepam, and TG100-115) with the highest negative enrichment scores were considered potential compounds for the treatment of OA (Supplementary Table S2). The chemical structures of these compounds are shown in Supplementary Figure S4.
To further explore the associations among mRNAs from the ceRNA network, we performed PPI analysis consisting of 18 nodes and 31 edges using the STRING database (Figure 6). Then, the MCODE app in Cytoscape 3.8.0 was used to screen the hub genes. Under the conditions mentioned in above methods, an important module (MCODE score: 5.0) including six nodes and 15 edges was found (Figure 7A), which demonstrates six significant hub genes (serpin family H member 1 [SERPINH1], collagen type VIII alpha 2 chain [COL8A2], collagen type XV alpha 1 chain [COL15A1], collagen type VI alpha 3 chain [COL6A3], collagen type V alpha 1 chain [COL5A1], and collagen type XI alpha one chain [COL11A1]) in OA. The network topological characteristics of six hub genes were showed in Supplementary Table S3. A circRNA-miRNA-hubgene subnetwork was established to identify the associations among the DEcircRNAs, IDEmiRNAs, and hub genes (Figure 7B), and the network included six regulatory axes (hsa_circ_0075320/hsa-miR-6124/COL11A1, hsa_circ_0075320/hsa-miR-6124/SERPINH1, hsa_circ_0075320/hsa-miR-6124/COL5A1, hsa_circ_0075320/hsa-miR-6124/COL8A2, hsa_circ_0075320/hsa-miR-6124/COL15A1, and hsa_circ_0051428/hsa-miR-1207-5p/COL6A3).
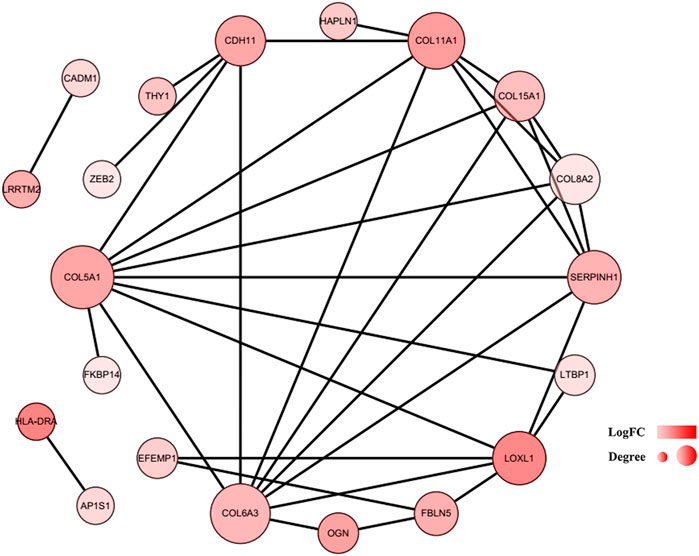
FIGURE 6. PPI network of 37 mRNAs. The color of a node in the PPI network reflects the log (FC) value of gene expression, and the size of node indicates the number of interacting proteins with the designated protein.
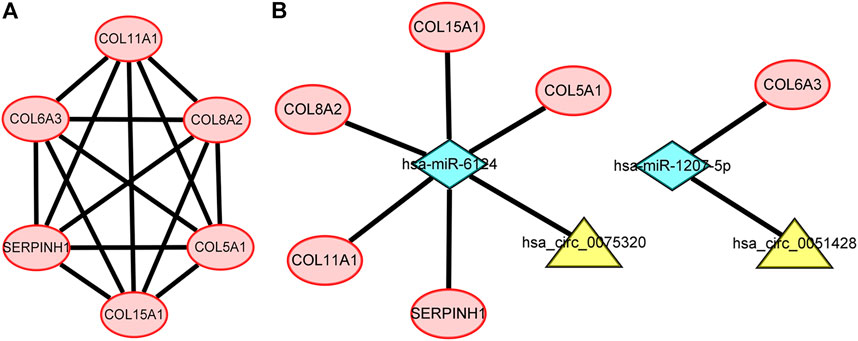
FIGURE 7. (A) PPI network consisting of the six hub genes. (B) A subnetwork of circRNAs–miRNAs–hub genes.
To further explore the sub network identified by MCODE, the correlation network analysis for the six hub genes was performed. The results demonstrated that all six hub genes showed positive correlation, especially among the COL5A1, COL8A2, and COL11A1, whose co-expression was the significant (Figure 8).
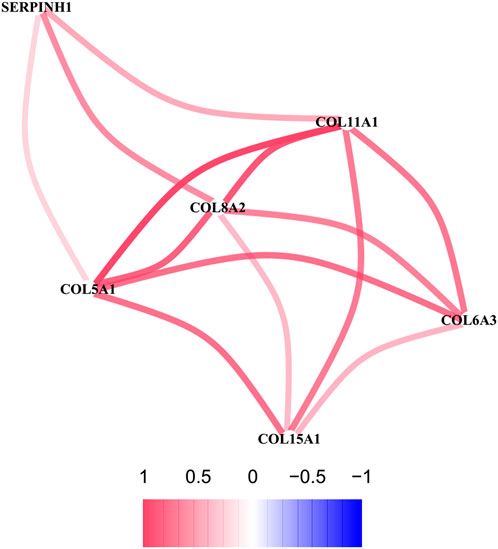
FIGURE 8. The correlation network of six hub genes. The red line represents a positive correlation between two genes, and the blue represents a negative correlation.
We utilized DGIdb to perform drug–gene interaction analysis for the six key genes identified in the PPI network. Finally, we found that two potential drugs, collagenase Clostridium histolyticum and ocriplasmin, may be effective in treating OA because they were found to be closely associated with the four hub genes (COL15A1, COL6A3, COL5A1, COL11A1).
OA is a common joints illness that leads to cartilage degeneration, synovial inflammation, and osteophyte formation (Xie and Chen, 2019). However, the pathogenesis of OA is not yet fully understood. Patients with OA usually miss the optimal treatment opportunity due to lack of early diagnostic indicators, leading to a poor prognosis. Therefore, it is important to identify early diagnostic indicators and potential therapeutic targets in patients with OA. In recent years, a bioinformatics-related method has been developed that can be used to predict the potential target genes of various diseases.
CircRNAs are stable ncRNAs that lack a 5′-cap or 3′-tail (Chen et al., 2019). Due to their circular structure, circRNAs identified as biomarkers for diagnosis and prognosis are not affected by RNA exonucleases and become relatively stable(Li et al., 2015; Zhang et al., 2018). Many complex diseases, including non-tumors, are influenced by circRNAs (Xiong et al., 2018; Huo et al., 2021). At present, an increasing number of researchers are focusing on the association between circRNAs and disease occurrence. Some evidence has suggested that circRNAs containing the MRE, RNA binding protein (RBP), and open reading frame (ORF) can combine with miRNAs, thus generally called as miRNA sponges, reducing the cytoplasmic levels of miRNAs and releasing their downstream target mRNAs (He et al., 2017; Bai et al., 2018; Wang et al., 2018). Although one study predicted that hsa_circ_0025119, hsa_circ_0025113, hsa_circ_0009897, and hsa_circ_0002447 exert significant effects on OA, the specific functions of circRNAs remain unknown (Wang et al., 2021). Therefore, to determine whether circRNAs function as ceRNAs in OA, we constructed a circRNA–miRNA–mRNA regulatory network based on biological forecasts, a PPI network based on STRING, and a sub network based on circRNAs, miRNAs, and hub genes.
In this study, five DEcircRNAs, 89 DEmiRNAs, and 345 DEmRNAs were first obtained from the GSE175959, GSE105027, and GSE169077 datasets, respectively. To date, these five DE circRNAs (hsa_circ_0004662, hsa_circ_0051428, hsa_circ_0003312, hsa_circ_0008590, and hsa_circ_0075320) have not been reported in previously published articles. circRNAs are ceRNAs that alter the gene expression by binding to miRNAs (Zhang et al., 2017; Zhong et al., 2018; Kim et al., 2021). To investigate whether the five circRNAs have a ceRNA role in OA, the circBase and CSCD databases were used to predict their MREs. To further explore the mechanisms of miRNAs and mRNAs of circRNAs downstream in OA, the miRDB and TargetScan databases were utilized to forecast target genes. Finally, a circRNAs-miRNA-mRNAs regulatory network was established based on three circRNAs (hsa_circ_0075320, hsa_circ_0003312, and hsa_circ_0051428), seven miRNAs (hsa-miR-6124, hsa-miR-6832-5p, hsa-miR-1207-5p, hsa-miR-4793-5p, hsa-miR-624-3p, hsa-miR-1225-5p, and hsa-miR-1229-5p), and 37 mRNAs.
We then conducted enrichment analysis based on the mRNAs in this network. The analysis demonstrated that the structural constituents of the extracellular matrix (ECM), such as collagen and glycosaminoglycan, are closely associated with the occurrence and progression of OA. Furthermore, type II collagen in cartilage ECM is disintegrated via increased expression of cartilage-degrading enzymes, ultimately resulting in the degeneration of articular cartilage (Glasson et al., 2005; Stanton et al., 2005). Aberrant crosslinking of collagen fibrils within the joint may result in OA (Hu et al., 2006; Wang et al., 2008). In this study, pathways detected by KEGG analysis were mainly related to protein digestion and absorption, cell adhesion molecules, TGF-β signaling pathway, S. aureus infection, lysosomes, and other glycan degradation. Cartilage damage is a major issue in OA. In recent years, there is moderate evidence showing that growth factors such as TGF-β have great potential in cartilage repair (Blaney Davidson et al., 2007; Wang et al., 2020). TGF-β can prevent the occurrence of osteoarthritis by maintaining the homeostasis of articular cartilage, and TGF-β supplementation can also enhance cartilage repair, which is a potential treatment method (Wang et al., 2020). According to the relevant research, some cell adhesion molecules can be used as predictors of OA (Schett et al., 2009). Therefore, the cell adhesion molecules are also closely relevant to the disease process for OA.
To further determine the key circRNAs involved in the ceRNA network, a PPI network was constructed, and six hub genes (SERPINH1, COL5A1, COL15A1, COL6A3, COL8A2, and COL11A1) were identified using the MCODE app. Bioinformatics analysis revealed that these hub genes are likely to play significant roles in the pathogenesis of OA. In previous studies, SERPINH1, a collagen-specific molecular chaperone critical for type I and type III collagen maturation,(Taguchi and Razzaque, 2007) was shown to be associated with OA via proteomic analysis (Tsolis et al., 2015). Consistent with our results, aberrant expression of COL5A1 was previously shown to be a target gene in synovial fibrosis therapy (Remst et al., 2014). COL15A1, whose expression was confirmed to be increased in OA, can encode a protein with significant expression in newly formed blood vessels, revealing a probable function in angiogenesis (Karlsson et al., 2010). COL6A3 is associated with cartilage degeneration, as verified by quantitative PCR analysis (Chou et al., 2013). COL8A2, a collagen-related gene, is more abundantly expressed in OA, but the exact mechanism is not fully understood (Karlsson et al., 2010). A meta-analysis demonstrated that common missense variants in COL11A1 are associated with OA (Styrkarsdottir et al., 2018). COL11A1 contributes to the development of OA due to the increased degradation of articular cartilage type II collagen caused by the type XI collagen mutation (Rodriguez et al., 2004). However, the mechanisms of action of the identified hub genes in OA need to be further explored.
Although the main functions of the six key genes have been described, their connections with circRNAs are yet to be explored. In this study, six circRNA–miRNA–hubgene subnetwork regulatory axes (hsa_circ_0075320/hsa-miR-6124/COL11A1, hsa_circ_0075320/hsa-miR-6124/SERPINH1, hsa_circ_0075320/hsa-miR-6124/COL5A1, hsa_circ_0075320/hsa-miR-6124/COL8A2, hsa_circ_0075320/hsa-miR-6124/COL15A1, hsa_circ_0051428/hsa-miR-1207-5p/COL6A3) were identified, suggesting competitive regulatory relationships between hsa_circ_0075320/hsa_circ_0051428 and the six hub genes. Nevertheless, considering that these outcomes were derived from bioinformatics analysis, additional experiments must be performed to validate the possible effects of these six axes.
Currently, pharmacotherapy is vital in treating OA, with most patients requiring short-term or long-term drug therapy. CMap analysis was performed to identify potential therapeutic agents. CMap analysis identified three compounds (noscapine, diazepam, and TG100-115) as candidate drugs, facilitating the exploration of their corresponding targets. Although noscapine is an alkaloid derived from opioids, it has been clinically used as an antitussive agent without any addictive or euphoric effects. The association between noscapine and OA has not yet been reported, but noscapine is suggested to act as an anti-inflammatory agent that remarkably decreases the levels of proinflammatory factors and shows antioxidant effects by inhibiting nitric oxide and reducing reactive oxygen species levels (Rahmanian-Devin et al., 2021). These therapeutic effects, combined with its low systemic toxicity, make noscapine a potential candidate for the treatment of various inflammatory diseases, including OA. Thomas et al. (1991) have demonstrated the effectiveness of diazepam in OA treatment. However, the long-term efficacy of diazepam in OA treatment requires further investigation and verification. Doukas et al. confirmed that TG100-115, a selective phosphatidylinositol 3-kinase inhibitor, can reduce infarct development and preserve myocardial function by effectively inhibiting edema and inflammation (Doukas et al., 2006). Although the link between TG100-115 and OA remains unclear, we believe that the occurrence and progression of OA can be controlled by effectively inhibiting edema and inflammation.
In addition to the CMap analysis described above, hub genes were found to be significant in OA. Hence, we conducted a drug–gene interaction analysis using the DGIdb online database and identified two related drugs, collagenase Clostridium histolyticum and ocriplasmin, for OA treatment. At present, ocriplasmin is mainly used to treat some ophthalmic diseases (Pirani et al., 2019), while collagenase Clostridium histolyticum is primarily used to treat Dupuytren’s contracture or Peyronie’s disease (Abdel Raheem et al., 2018; Fletcher et al., 2019). Nevertheless, whether such medications have satisfactory effects on OA needs to be further investigated.
In our study, six circRNA–miRNA–hubgene axes were ultimately identified, revealing competitive interactions between two circRNAs and six hub genes in OA. In the future, more experiments are needed to validate this probable ceRNA mechanism in OA.
However, there are several limitations to this research. Firstly, this study was done primarily using bioinformatics analysis; therefore, further experimental validation is needed. Secondly, the sample size was not large. Finally, further clinical studies should be performed to explore the correlation between these findings in this study and clinical practice.
In summary, a circRNA–miRNA–mRNA regulatory network was established, including three important circRNAs (hsa_circ_0075320, hsa_circ_0003312, and hsa_circ_0051428) via comprehensive bioinformatics analyses. We then constructed a circRNA–miRNA–hubgene regulatory subnetwork involving two vitally important circRNAs, hsa_circ_0075320, hsa_circ_0051428. Furthermore, three bioactive compounds, noscapine, diazepam, and TG100-115, according to the CMap analysis, and two related drugs, collagenase Clostridium histolyticum and ocriplasmin, according to the DGIdb database, were identified as potential treatment options for OA. This study reveals a unique perspective of the regulatory mechanisms and possible drugs for OA treatment from the ceRNA network view.
Publicly available datasets were analyzed in this study. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
W-BX, G-XL, C-MC, and GR designed the research study; W-BX wrote the original article; J-WL, J-YF, H-LZ performed the data analysis; VK, C-MC, G-XL participated in the conception of the study and revision of the manuscript. All authors approve of the final version to be published.
This research was funded by the Natural Science Foundation of Fujian Province (No. 2020J011244) and Natural Science Foundation of Fujian Province (No. 2021J05282); funded by the “Xiamen Health High-Level Talent Training Program.”
The author (G-XL) wishes to acknowledge the financial support of the “Xiamen Health High-Level Talent Training Program.”
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fgene.2022.994163/full#supplementary-material
SUPPLEMENTAY FIGURE S1 | Heatmaps of the differentially expressed circRNAs (DEcircRNAs), microRNAs (DEcircRNAs), and mRNAs (DEmRNAs).(A) Heatmap of the DEcircRNAs in the GSE175959 dataset. (B) Heatmap of the DEmiRNAs in the GSE105027 dataset. (C) Heatmap of the DEmRNAs in the GSE105027 dataset.
SUPPLEMENTAY FIGURE S2 | (A) Venn diagram of the 89 differentially expressed miRNAs (DEmiRNAs) obtained from GSE105027 and their overlap with the MREs. (B) Venn diagram of the 345 differentially expressed mRNAs (DEmRNAs) obtained from GSE169077 and their overlap with miRNA target genes.
SUPPLEMENTAY FIGURE S3 | (A) Heatmap of the three circular RNAs (circRNAs) in the competing endogenous RNA (ceRNA) network. (B) Heatmap of the seven miRNAs in the ceRNA network. (C) Heatmap of the 37 mRNAs in the ceRNA network.
SUPPLEMENTAY FIGURE S4 | Chemical structures of the three compounds found by the CMap analysis. (A) Noscapine. (B) Diazepam. (C) TG100-115.
Abdel Raheem, A., Johnson, M., Ralph, D., and Garaffa, G. (2018). Collagenase clostridium histolyticum: A novel medical treatment for Peyronie's disease. Minerva Urol. Nefrol. 70 (4), 380–385. doi:10.23736/s0393-2249.18.03118-1
Agarwal, V., Bell, G. W., Nam, J. W., and Bartel, D. P. (2015). Predicting effective microRNA target sites in mammalian mRNAs. Elife 4. doi:10.7554/eLife.05005
Bader, G. D., and Hogue, C. W. (2003). An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinforma. 4, 2. doi:10.1186/1471-2105-4-2
Bai, N., Peng, E., Qiu, X., Lyu, N., Zhang, Z., Tao, Y., et al. (2018). circFBLIM1 act as a ceRNA to promote hepatocellular cancer progression by sponging miR-346. J. Exp. Clin. Cancer Res. 37 (1), 172. doi:10.1186/s13046-018-0838-8
Barrett, T., Wilhite, S. E., Ledoux, P., Evangelista, C., Kim, I. F., Tomashevsky, M., et al. (2013). NCBI GEO: Archive for functional genomics data sets--update. Nucleic Acids Res. 41, D991–D995. Database issue). doi:10.1093/nar/gks1193
Blaney Davidson, E. N., van der Kraan, P. M., and van den Berg, W. B. (2007). TGF-beta and osteoarthritis. Osteoarthr. Cartil. 15 (6), 597–604. doi:10.1016/j.joca.2007.02.005
Chen, B. J., Huang, S., and Janitz, M. (2019). Changes in circular RNA expression patterns during human foetal brain development. Genomics 111 (4), 753–758. doi:10.1016/j.ygeno.2018.04.015
Chou, C. H., Lee, C. H., Lu, L. S., Song, I. W., Chuang, H. P., Kuo, S. Y., et al. (2013). Direct assessment of articular cartilage and underlying subchondral bone reveals a progressive gene expression change in human osteoarthritic knees. Osteoarthr. Cartil. 21 (3), 450–461. doi:10.1016/j.joca.2012.11.016
Cross, M., Smith, E., Hoy, D., Nolte, S., Ackerman, I., Fransen, M., et al. (2014). The global burden of hip and knee osteoarthritis: Estimates from the global burden of disease 2010 study. Ann. Rheum. Dis. 73 (7), 1323–1330. doi:10.1136/annrheumdis-2013-204763
Doukas, J., Wrasidlo, W., Noronha, G., Dneprovskaia, E., Fine, R., Weis, S., et al. (2006). Phosphoinositide 3-kinase gamma/delta inhibition limits infarct size after myocardial ischemia/reperfusion injury. Proc. Natl. Acad. Sci. U. S. A. 103 (52), 19866–19871. doi:10.1073/pnas.0606956103
Fletcher, J., Tan, E. S. L., Thomas, M., Taylor, F., Elliott, D., and Bindra, R. (2019). Collagenase injections for Dupuytren's contracture: Prospective cohort study in a public health setting. ANZ J. Surg. 89 (5), 573–577. doi:10.1111/ans.14988
Glasson, S. S., Askew, R., Sheppard, B., Carito, B., Blanchet, T., Ma, H. L., et al. (2005). Deletion of active ADAMTS5 prevents cartilage degradation in a murine model of osteoarthritis. Nature 434 (7033), 644–648. doi:10.1038/nature03369
Glažar, P., Papavasileiou, P., and Rajewsky, N. (2014). circBase: a database for circular RNAs. Rna 20 (11), 1666–1670. doi:10.1261/rna.043687.113
Griffith, M., Griffith, O. L., Coffman, A. C., Weible, J. V., McMichael, J. F., Spies, N. C., et al. (2013). DGIdb: Mining the druggable genome. Nat. Methods 10 (12), 1209–1210. doi:10.1038/nmeth.2689
He, R., Liu, P., Xie, X., Zhou, Y., Liao, Q., Xiong, W., et al. (2017). circGFRA1 and GFRA1 act as ceRNAs in triple negative breast cancer by regulating miR-34a. J. Exp. Clin. Cancer Res. 36 (1), 145. doi:10.1186/s13046-017-0614-1
Hu, K., Xu, L., Cao, L., Flahiff, C. M., Brussiau, J., Ho, K., et al. (2006). Pathogenesis of osteoarthritis-like changes in the joints of mice deficient in type IX collagen. Arthritis Rheum. 54 (9), 2891–2900. doi:10.1002/art.22040
Huo, Z., Li, H., Tian, L., Li, J., Zhang, K., Li, Z., et al. (2021). Construction of a potentially functional circRNA-miRNA-mRNA network in intervertebral disc degeneration by bioinformatics analysis. Biomed. Res. Int. 2021, 8352683. doi:10.1155/2021/8352683
Kanehisa, M., and Goto, S. (2000). Kegg: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 28 (1), 27–30. doi:10.1093/nar/28.1.27
Karlsson, C., Dehne, T., Lindahl, A., Brittberg, M., Pruss, A., Sittinger, M., et al. (2010). Genome-wide expression profiling reveals new candidate genes associated with osteoarthritis. Osteoarthr. Cartil. 18 (4), 581–592. doi:10.1016/j.joca.2009.12.002
Kiadaliri, A. A., Lohmander, L. S., Moradi-Lakeh, M., Petersson, I. F., and Englund, M. (2018). High and rising burden of hip and knee osteoarthritis in the Nordic region, 1990-2015. Acta Orthop. 89 (2), 177–183. doi:10.1080/17453674.2017.1404791
Kim, E., Kim, Y. K., and Lee, S. V. (2021). Emerging functions of circular RNA in aging. Trends Genet. 37 (9), 819–829. doi:10.1016/j.tig.2021.04.014
Kristensen, L. S., Hansen, T. B., Venø, M. T., and Kjems, J. (2018). Circular RNAs in cancer: Opportunities and challenges in the field. Oncogene 37 (5), 555–565. doi:10.1038/onc.2017.361
Lamb, J., Crawford, E. D., Peck, D., Modell, J. W., Blat, I. C., Wrobel, M. J., et al. (2006). The connectivity map: Using gene-expression signatures to connect small molecules, genes, and disease. Science 313 (5795), 1929–1935. doi:10.1126/science.1132939
Lespasio, M. J., Piuzzi, N. S., Husni, M. E., Muschler, G. F., Guarino, A., and Mont, M. A. (2017). Knee osteoarthritis: A primer. Perm. J. 21, 16–183. doi:10.7812/tpp/16-183
Li, J., Yang, J., Zhou, P., Le, Y., Zhou, C., Wang, S., et al. (2015a). Circular RNAs in cancer: Novel insights into origins, properties, functions and implications. Am. J. Cancer Res. 5 (2), 472–480.
Li, P., Chen, S., Chen, H., Mo, X., Li, T., Shao, Y., et al. (2015b). Using circular RNA as a novel type of biomarker in the screening of gastric cancer. Clin. Chim. Acta. 444, 132–136. doi:10.1016/j.cca.2015.02.018
Petkovic, S., and Müller, S. (2015). RNA circularization strategies in vivo and in vitro. Nucleic Acids Res. 43 (4), 2454–2465. doi:10.1093/nar/gkv045
Pirani, V., Pelliccioni, P., Cesari, C., Carrozzi, G., Cavallero, E., and Mariotti, C. (2019). Flare changes after intravitreal injection of ocriplasmin in symptomatic vitreomacular traction syndrome. Jpn. J. Ophthalmol. 63 (3), 255–261. doi:10.1007/s10384-019-00660-z
Qi, X., Zhang, D. H., Wu, N., Xiao, J. H., Wang, X., and Ma, W. (2015). ceRNA in cancer: possible functions and clinical implications. J. Med. Genet. 52 (10), 710–718. doi:10.1136/jmedgenet-2015-103334
Qu, S., Yang, X., Li, X., Wang, J., Gao, Y., Shang, R., et al. (2015). Circular RNA: A new star of noncoding RNAs. Cancer Lett. 365 (2), 141–148. doi:10.1016/j.canlet.2015.06.003
Rahmanian-Devin, P., Baradaran Rahimi, V., Jaafari, M. R., Golmohammadzadeh, S., Sanei-Far, Z., and Askari, V. R. (2021). Noscapine, an emerging medication for different diseases: A mechanistic review. Evid. Based. Complement. Altern. Med. 2021, 8402517. doi:10.1155/2021/8402517
Remst, D. F., Blom, A. B., Vitters, E. L., Bank, R. A., van den Berg, W. B., Blaney Davidson, E. N., et al. (2014). Gene expression analysis of murine and human osteoarthritis synovium reveals elevation of transforming growth factor β-responsive genes in osteoarthritis-related fibrosis. Arthritis Rheumatol. 66 (3), 647–656. doi:10.1002/art.38266
Ritchie, M. E., Phipson, B., Wu, D., Hu, Y., Law, C. W., Shi, W., et al. (2015). Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 43 (7), e47. doi:10.1093/nar/gkv007
Rodriguez, R. R., Seegmiller, R. E., Stark, M. R., and Bridgewater, L. C. (2004). A type XI collagen mutation leads to increased degradation of type II collagen in articular cartilage. Osteoarthr. Cartil. 12 (4), 314–320. doi:10.1016/j.joca.2003.12.002
Salzman, J., Chen, R. E., Olsen, M. N., Wang, P. L., and Brown, P. O. (2013). Cell-type specific features of circular RNA expression. PLoS Genet. 9 (9), e1003777. doi:10.1371/journal.pgen.1003777
Schett, G., Kiechl, S., Bonora, E., Zwerina, J., Mayr, A., Axmann, R., et al. (2009). Vascular cell adhesion molecule 1 as a predictor of severe osteoarthritis of the hip and knee joints. Arthritis Rheum. 60 (8), 2381–2389. doi:10.1002/art.24757
Stanton, H., Rogerson, F. M., East, C. J., Golub, S. B., Lawlor, K. E., Meeker, C. T., et al. (2005). ADAMTS5 is the major aggrecanase in mouse cartilage in vivo and in vitro. Nature 434 (7033), 648–652. doi:10.1038/nature03417
Styrkarsdottir, U., Lund, S. H., Thorleifsson, G., Zink, F., Stefansson, O. A., Sigurdsson, J. K., et al. (2018). Meta-analysis of Icelandic and UK data sets identifies missense variants in SMO, IL11, COL11A1 and 13 more new loci associated with osteoarthritis. Nat. Genet. 50 (12), 1681–1687. doi:10.1038/s41588-018-0247-0
Su, G., Morris, J. H., Demchak, B., and Bader, G. D. (2014). Biological network exploration with Cytoscape 3. Curr. Protoc. Bioinforma. 47, 811–1324. doi:10.1002/0471250953.bi0813s47
Szklarczyk, D., Morris, J. H., Cook, H., Kuhn, M., Wyder, S., Simonovic, M., et al. (2017). The STRING database in 2017: Quality-controlled protein-protein association networks, made broadly accessible. Nucleic Acids Res. 45 (D1), D362–d368. doi:10.1093/nar/gkw937
Taguchi, T., and Razzaque, M. S. (2007). The collagen-specific molecular chaperone HSP47: Is there a role in fibrosis? Trends Mol. Med. 13 (2), 45–53. doi:10.1016/j.molmed.2006.12.001
Tang, X., Wang, S., Zhan, S., Niu, J., Tao, K., Zhang, Y., et al. (2016). The prevalence of symptomatic knee osteoarthritis in China: Results from the China health and retirement longitudinal study. Arthritis Rheumatol. 68 (3), 648–653. doi:10.1002/art.39465
Thomas, M., Eriksson, S. V., and Lundeberg, T. (1991). A comparative study of diazepam and acupuncture in patients with osteoarthritis pain: A placebo controlled study. Am. J. Chin. Med. 19 (2), 95–100. doi:10.1142/s0192415x91000156
Tsolis, K. C., Bei, E. S., Papathanasiou, I., Kostopoulou, F., Gkretsi, V., Kalantzaki, K., et al. (2015). Comparative proteomic analysis of hypertrophic chondrocytes in osteoarthritis. Clin. Proteomics 12 (1), 12. doi:10.1186/s12014-015-9085-6
Vidal, A. F., Sandoval, G. T., Magalhães, L., Santos, S. E., and Ribeiro-dos-Santos, Â. (2016). Circular RNAs as a new field in gene regulation and their implications in translational research. Epigenomics 8 (4), 551–562. doi:10.2217/epi.16.3
Wang, B., Zhong, J. L., Xu, X. H., Wu, B., Shang, J., Jiang, N., et al. (2021). Gene expression profiling analysis to identify key genes and underlying mechanisms in meniscus of osteoarthritis patients. Comb. Chem. High. Throughput Screen. 24 (8), 1151–1167. doi:10.2174/1386207323666200902140656
Wang, C. J., Iida, K., Egusa, H., Hokugo, A., Jewett, A., and Nishimura, I. (2008). Trabecular bone deterioration in col9a1+/- mice associated with enlarged osteoclasts adhered to collagen IX-deficient bone. J. Bone Min. Res. 23 (6), 837–849. doi:10.1359/jbmr.080214
Wang, C., Shen, J., Ying, J., Xiao, D., and O'Keefe, R. J. (2020). FoxO1 is a crucial mediator of TGF-β/TAK1 signaling and protects against osteoarthritis by maintaining articular cartilage homeostasis. Proc. Natl. Acad. Sci. U. S. A. 117 (48), 30488–30497. doi:10.1073/pnas.2017056117
Wang, W. L., Yang, Z., Zhang, Y. J., Lu, P., Ni, Y. K., Sun, C. F., et al. (2018). Competing endogenous RNA analysis reveals the regulatory potency of circRNA_036186 in HNSCC. Int. J. Oncol. 53 (4), 1529–1543. doi:10.3892/ijo.2018.4499
Wei, H., Pan, L., Tao, D., and Li, R. (2018). Circular RNA circZFR contributes to papillary thyroid cancer cell proliferation and invasion by sponging miR-1261 and facilitating C8orf4 expression. Biochem. Biophys. Res. Commun. 503 (1), 56–61. doi:10.1016/j.bbrc.2018.05.174
Wong, N., and Wang, X. (2015). miRDB: an online resource for microRNA target prediction and functional annotations. Nucleic Acids Res. 43, D146–D152. doi:10.1093/nar/gku1104
Xia, S., Feng, J., Chen, K., Ma, Y., Gong, J., Cai, F., et al. (2018). Cscd: A database for cancer-specific circular RNAs. Nucleic Acids Res. 46 (D1), D925–d929. doi:10.1093/nar/gkx863
Xie, C., and Chen, Q. (2019). Adipokines: New therapeutic target for osteoarthritis? Curr. Rheumatol. Rep. 21 (12), 71. doi:10.1007/s11926-019-0868-z
Xiong, D. D., Dang, Y. W., Lin, P., Wen, D. Y., He, R. Q., Luo, D. Z., et al. (2018). A circRNA-miRNA-mRNA network identification for exploring underlying pathogenesis and therapy strategy of hepatocellular carcinoma. J. Transl. Med. 16 (1), 220. doi:10.1186/s12967-018-1593-5
Yu, G., Wang, L. G., Han, Y., and He, Q. Y. (2012). clusterProfiler: an R package for comparing biological themes among gene clusters. Omics 16 (5), 284–287. doi:10.1089/omi.2011.0118
Zhang, H. D., Jiang, L. H., Sun, D. W., Hou, J. C., and Ji, Z. L. (2018). CircRNA: A novel type of biomarker for cancer. Breast Cancer 25 (1), 1–7. doi:10.1007/s12282-017-0793-9
Zhang, Y., Liang, W., Zhang, P., Chen, J., Qian, H., Zhang, X., et al. (2017). Circular RNAs: Emerging cancer biomarkers and targets. J. Exp. Clin. Cancer Res. 36 (1), 152. doi:10.1186/s13046-017-0624-z
Keywords: osteoarthritis, circRNA, ceRNA, GEO, bioinformatics analysis
Citation: Xu W-B, Kotheeranurak V, Zhang H-L, Feng J-Y, Liu J-W, Chen C-M, Lin G-X and Rui G (2022) Identification of the circRNA–miRNA–mRNA regulatory network in osteoarthritis using bioinformatics analysis. Front. Genet. 13:994163. doi: 10.3389/fgene.2022.994163
Received: 14 July 2022; Accepted: 05 September 2022;
Published: 16 September 2022.
Edited by:
ZeYu Huang, Sichuan University, ChinaReviewed by:
Jun Cui, Hunan Normal University, ChinaCopyright © 2022 Xu, Kotheeranurak, Zhang, Feng, Liu, Chen, Lin and Rui. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chien-Min Chen, OTYwMTVAY2NoLm9yZy50dw==; Guang-Xun Lin, bGluZ3Vhbmd4dW5AaG90bWFpbC5jb20=; Gang Rui, cmVpZ2FuZ0AxNjMuY29t
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.