
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Genet. , 30 November 2021
Sec. Cancer Genetics and Oncogenomics
Volume 12 - 2021 | https://doi.org/10.3389/fgene.2021.674094
 Yifan Su1,2,3†
Yifan Su1,2,3† Qianlan Yao1,2,3†
Qianlan Yao1,2,3† Yuyin Xu1,2,3†
Yuyin Xu1,2,3† Chengli Yu1,2,3
Chengli Yu1,2,3 Jing Zhang1,2,3
Jing Zhang1,2,3 Qian Wang1,2,3
Qian Wang1,2,3 Jiwei Li1,2,3
Jiwei Li1,2,3 Di Shi1,2,3
Di Shi1,2,3 Baohua Yu1,2,3
Baohua Yu1,2,3 Yupeng Zeng1,2,3
Yupeng Zeng1,2,3 Xiaoli Zhu1,2,3
Xiaoli Zhu1,2,3 Qianming Bai1,2,3
Qianming Bai1,2,3 Xiaoyan Zhou1,2,3*
Xiaoyan Zhou1,2,3*Background: Expert consensus on BRCA1/2 genetic testing and clinical application in Chinese breast cancer patients recommends that BRCA1/2 testing should be performed in those with clinical risk factors, such as an early onset, triple-negative breast cancer (TNBC) or family history of cancer. With the increasing application of multigene panels, testing for genes beyond BRCA1/2 has become more prevalent. However, the non-BRCA mutation status of Chinese high-risk breast cancer patients has not been fully explored.
Methods: A total of 230 high-risk breast cancer patients from Fudan University Shanghai Cancer Center who had undergone peripheral blood germline 72 genes next-generation sequencing (NGS) from June 2018 to June 2020 were enrolled for retrospective analysis. The 72 genes include common hereditary breast cancer genes, such as homologous recombination repair (HRR) genes and other DNA damage repair genes. High-risk factors included: 1) TNBC; 2) male breast cancer; 3) primary bilateral breast cancer; 4) diagnosed with breast cancer at age less than or equal to 40 years; or 5) at least one first- and/or second-degree relative with BRCA-related cancer (breast or ovarian or prostate or pancreatic cancer).
Results: The germline pathogenic or likely pathogenic mutation rate was 29.6% (68/230) in high-risk breast cancer patients. Among them, 44 (19.1%, 44/230) were identified as harboring BRCA1/2 mutation, and 28 (12.2%, 28/230) patients carried non-BRCA germline variants. Variants were detected in 16 non-BRCA genes, including PALB2 (5, 2.2%), ATM (4, 1.7%), RAD51D (3, 1.3%), TP53 (3, 1.3%), CHEK2 (2, 0.9%), FANCA (2, 0.9%) and ATR, BARD1, BRIP1, ERCC3, HOXB13, MLH1, MRE11, PMS2, RAD51C, RAD54L (1, 0.4%). Besides, 22 (9.6%, 22/230) patients were non-BRCA HRR gene mutation (including ATM, ATR, BARD1, BRIP1, CHEK2, FANCA, MRE11, PALB2, RAD51C RAD51D and RAD54L) carriers. Among high-risk factors, family history showed a correlation with both BRCA (p = 0.005) and non-BRCA HRR gene mutation status (p = 0.036). In addition, TNBC showed a correlation with BRCA1 gene mutation status (p = 0.038). However, other high-risk factors have not shown significantly related to BRCA1/2, non-BRCA genes and non-BRCA HRR gene mutations (p > 0.05). In addition, 312 unique variants of uncertain significance (VUS) were identified among 175 (76.1%, 175/230) patients and 65 different genes.
Conclusions: Non-BRCA gene mutations are frequently identified in breast cancer patients with high risk factors. Family history showed a correlation with both BRCA (p = 0.005) and non-BRCA HRR gene mutation status (p = 0.036), so we strongly suggest that breast cancer patients with a BRCA-related family history receive comprehensive gene mutation testing in China, especially HRR genes, which are not only related to high risk of breast cancer, but also potentially related to poly ADP ribose polymerase inhibitor (PARPi) targeted therapy. The exact relationship of rare gene mutations to breast cancer predisposition and the pathogenicity of VUS need to be further investigated.
Breast cancer is considered the most common cancer among females worldwide. According to the epidemiologic analysis report of International Agency for Research on Cancer (IARC), breast cancer had the highest incidence among Chinese female malignant tumors in 2020, with 416,000 patients, accounting for 19.9% of all malignant tumors in women (World Health Organization, 2021). Due to pathogenic or likely pathogenic variants in some tumor suppressor genes, 5–10% of breast cancer cases are hereditary (Carroll et al., 2008), and 15–20% show familial aggregation. Compared to sporadic tumors, hereditary breast cancer is characterized by an early age of onset, male breast cancer, and multiple or bilateral primary tumors (Sung et al., 2017).
Mutations in BRCA1 and BRCA2 are closely related to increased susceptibility to breast cancer. Data for the Chinese population showed that the pathogenic or likely pathogenic mutations of BRCA1/2 were identified in 5.3% unselected Chinese breast cancer patients and in 18.1% familial breast cancer patients (Sun et al., 2017). Besides, the cumulative risk of developing breast cancer by the age of 70 is approximately 37.9% in BRCA1 mutation carriers and 36.5% in BRCA2 mutation carriers in China, corresponding to a 10-fold increase compared with the general population (Yao et al., 2016), which indicated that it is necessary to have BRCA1/2 testing for Chinese breast cancer patients. The Breast Cancer Precision Treatment Committee of the Chinese Medical Doctor Association has issued expert consensus on BRCA1/2 genetic testing and clinical application in Chinese breast cancer patients referring to the international guidelines issued by the National Comprehensive Cancer Network (NCCN) and proposed characteristics of breast cancer patients who should be recommended for BRCA1/2 testing based on clinical risk factors such as age of onset, hormone receptor status and personal or family history of cancer (Wang et al., 2018). For carriers of BRCA1/2 mutation or their families, there are well-established counseling strategies and management guidelines for early intervention or prevention, such as increased monitoring and consideration of risk-reducing interventions (Kwong et al., 2020). In addition, BRCA-related breast cancer is highly sensitive to platinum-based chemotherapy, and these patients can significantly benefit from poly ADP ribose polymerase inhibitor (PARPi) therapy. Nonetheless, more than 60% of breast cancer patients with genetic characteristics or family aggregation do not carry BRCA1/2 mutations (Telli et al., 2016; Tham et al., 2016; Kwong et al., 2020).
With the application of next-generation sequencing (NGS) over the past 10 years, many non-BRCA breast cancer susceptibility genes have been identified in succession, such as ATM, CHEK2, PALB2 and TP53. Multigene testing is of great significance for breast cancer risk prediction, molecular typing (especially for triple-negative breast cancer) and selection of precise treatment options. A multigene panel enables sequencing of a large number of genes simultaneously. Indeed, more than 200 multigene panels proposed by academic or commercial laboratories have been listed by the NCBI Genetic Test Registry (Thehandbook. Bethes, 2018). Chrystelle Colas et al. summarized the 26 genes most commonly included in the breast cancer multigene panels, including ATM, BARD1, BRCA1, BRCA2, BRIP1, CDH1, CHEK2, GEN1, MCPH1, MLH1, MRE11A, MSH2, MSH6, NBN, NF1, PALB2, PMS2, PTEN, RAD50, RAD51C, RAD51D, RINT1, SLX4, STK11, TP53 and XRCC2 (Colas et al., 2019). The prevalence and spectrum of germline mutations in breast cancer patients vary across ethnicities. However, only a few studies have reported the prevalence of non-BRCA gene mutations in the Chinese population. In addition, the association between clinical risk factors and non-BRCA genes in China remains uncertain, and the lack of authoritative guidelines for multigene testing restricts the discovery of more mutation carriers in clinical practice.
Indeed, most of the non-BRCA breast cancer susceptibility genes also participate in DNA homologous recombination repair (HRR) pathways, such as ATM, CHEK2 and PALB2. HRR is a high-fidelity DNA repair mechanism that is essential for maintaining the integrity of the genome. Evidence showed that pathogenic mutations in non-BRCA HRR genes could also help identify susceptibility to familial breast cancer and showed PARPi sensitivity similar to BRCA1/2 (Mirza et al., 2016; Castroviejo-Bermejo et al., 2018).
In our study, we collected clinicopathological data for 230 high-risk breast cancer patients who had undergone peripheral blood germline gene testing with a 72 multigene panel and retrospectively analyzed the association between risk factors and different gene groups. The aim of this study was to identify the prevalence and spectrum of germline mutations, especially non-BRCA and non-BRCA HRR genes mutations in high-risk Chinese breast cancer patients, clarify their clinicopathologic characteristics of mutation carriers and provide evidence for proposing the clinical recommendations for genetic testing in high-risk breast cancers.
All cases were collected from Fudan University Shanghai Cancer Center. Data for breast cancer patients who had undergone peripheral blood germline 72 multigene panel testing from June 2018 to June 2020 were collected for statistical analysis. High-risk breast cancer patients were recruited if they fulfilled any one of the following five criteria: 1) triple-negative breast cancer (TNBC); 2) male breast cancer; 3) primary bilateral breast cancer; 4) diagnosed with breast cancer at age less than or equal to 40 years; or 5) at least one first- and/or second-degree relative with BRCA-related cancer (breast or ovarian or prostate or pancreatic cancer). All samples were included in the study with approval from the independent ethical committee/institutional review board, and all participants signed informed consent forms. Genomic DNA extracted from peripheral blood were performed using QIAamp DNA blood MidiKit (QIAgen, Valencia, CA) according to manufacturer’s instructions. DNA concentration was measured using Qubit dsDNA assay. Clinicopathological parameters were electronically retrieved from the Hospital Information System (HIS) of Fudan University Shanghai Cancer Center.
Genome DNA was sheared using Covaris M220, followed by end repairing, phosphorylation and adaptor ligation. DNA fragments were captured using the 72-gene panel (Burning Rock Biotech Ltd.), which covering 370 kb of human genomic regions, and then purified beads (Agencourt AMPure XP Kit, Beckman Coulter, California, United States). Quality and fragment size of such DNA libraries were assessed by Bioanalyzer High Sensitivity DNA Analysis (Agilent). Then libraries were sequenced on Nextseq500 sequencer (Illumina, Inc., California, United States) with pair-end reads. The 72 genes included are AKT1, APC, AR, ATM, ATR, BAP1, BARD1, BRAF, BRCA1, BRCA2, BRIP1, CCND1, CDK12, CDKN1B, CDKN2A, CDKN2B, CHD1, CHEK1, CHEK2, CTNNB1, EMSY, EPCAM, ERCC2, ERCC3, ERCC4, ESR1, FAM175A, FANCA, FANCD2, FANCI, FANCL, FANCM, FOXA1, GEN1, HDAC2, HOXB13, MLH1, MLH3, MRE11, MSH2, MSH6, MUTYH, MYC, NBN, NCOR1, NCOR2, PALB2, PIK3CA, PIK3CB, PIK3R1, PMS2, POLE, PPP2R2A, PTEN, RAD50, RAD51, RAD51B, RAD51C, RAD51D, RAD52, RAD54L, RAF1, RB1, RNF43, RSP O 2, SPOP, STK11, TMPRSS2, TP53, XRCC2, ZBTB16 and ZNRF3. Among them, HRR genes include ATM, ATR, BARD1, BRCA1, BRCA2, BRIP1, CHEK1, CHEK2, FANCA, FANCI, MRE11, NBN, PALB2, RAD50, RAD51B, RAD51C, RAD51D and RAD54L.
Sequencing data were aligned to the human genome (hg19) using BWA aligner 0.7.10. Local alignment optimization, variant calling was performed using GATK 3.2, VarScan separately. Variants were filtered using the VarScan fpfilter pipeline, which locations with depth less than 100 were filtered. 5 reads were required for each INDEL alleles, while 8 reads for SNVs. According to allele frequency database (ExAC, 1,000 Genomes, ESP6500 et al.), variants with frequency over 1% were considered as genetic polymorphisms. Variants’ detail information was annotated by ANNOVAR and SnpEff v3.6. DNA translocation analysis was performed using both Tophat2 and Factera 1.4.3.
Minor allele frequency (MAF) of variant less than 1% was considered for further pathogenicity evaluation. Variants were classified as pathogenic, likely pathogenic, uncertain significance, likely benign and benign according to American College of Medical Genetics (ACMG) guideline (Richards et al., 2015). Pathogenic/likely pathogenic (P/LP) variants were regarded as deleterious mutations with clinical significance. The variants pathogenic determination referred to databases such as the BRCA Exchange database (https://brcaexchange.org/favicon.ico), LOVD database (https://databases.lovd.nl/shared/genes) and ClinVar (http://www.ncbi.nlm.nih.gov/clinvar/) and published papers. Bioinformatic tools including SIFT (http://sift.jcvi.org), Align GVGD (http://agvgd.iarc.fr/agvgd_input.php) and PolyPhen-2 (http://genetics.bwh.harvard.edu/pph2) were used as supplementary evidence to prove that a variant may affect normal function.
The χ2 test and Fisher’s exact test were employed to evaluate differences in gene mutation frequency across groups as well as the clinicopathological characteristics of mutation carriers. Statistical Product and Service Solutions (SPSS) Statistics 26.0 (IBM institute, Chicago, IL, United States) was used for all statistical analyses. All p values were two-sided, and p < 0.05 was considered statistically significant.
Of 230 breast cancer patients with at least one high-risk factor, the mean age at diagnosis was 37.5 years, with a range from 21 to 79 years; among them 170 (73.9%) had early-onset breast cancer. A total of 103 (44.8%) had a family history of BRCA-related cancer (breast or ovarian or prostate or pancreatic cancer) and 21 (9.1%) had a family history of other cancers (such as esophageal, gastric, gallbladder, nasopharyngeal or lung cancer). Two (0.9%) of the breast cancer patients were male. Most breast cancers were invasive ductal carcinoma (209, 90.9%) or ductal carcinoma in situ (19, 8.3%); 111 (48.3%) were luminal, 85 (37.0%) were TNBC, and 33 (14.3%) were HER2 positive (HER2+). A total of 5.7% patients were diagnosed with primary bilateral breast cancer. The patients’ clinicopathologic characteristics are summarized in Table 1.
Variants defined as pathogenic or likely pathogenic were selected for analysis. Of 230 high-risk patients, 68 (29.6%, 68/230) were pathogenic or likely pathogenic variant carriers. Table 1 shows the germline mutation frequency of different groups. Forty-four (19.1%, 44/230) high-risk patients were identified as harboring BRCA mutations: 33 (14.3%, 33/230) BRCA1 mutation carriers and 11 (4.8%, 11/230) BRCA2 mutation carriers. In addition to BRCA, 28 (12.2%, 28/230) patients carried non-BRCA gene germline variants. In addition, 27.4% (63/230) patients carried HRR gene mutations, of which 22 (9.6%, 22/230) patients were non-BRCA HRR gene variant carriers. The distribution and frequency of non-BRCA and non-BRCA HRR mutant genes are shown in Figure 1. It is worth noting that 5 patients carried more than one pathogenic or likely pathogenic variants simultaneously (Table 2).
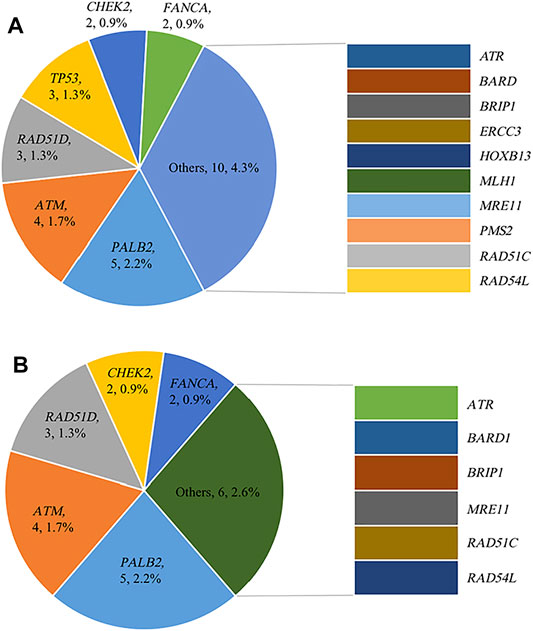
FIGURE 1. Distribution and frequency of germline mutations (A) Distribution and frequency of non-BRCA gene germline mutations; (B) Distribution and frequency of non-BRCA HRR gene germline mutations.
The BRCA mutation rate was 19.1% (44/230) in the high-risk groups of breast cancer patients. Among them, 33 (19.4%, 33/170) early-onset patients carried BRCA mutation, and 28.2% (29/103) patients with BRCA-related family history had BRCA mutations; 19 (22.4%) BRCA mutation carriers were identified among 85 TNBC patients and 5 (38.5%) carriers among primary bilateral breast cancer patients. However, no mutation was found in the 2 male patients. Family history showed a correlation with BRCA mutation (p = 0.005), with BRCA2 mutations having less impact than BRCA1 mutations. In addition, BRCA1 mutations showed a correlation with TNBC (p = 0.038) (Table 3).
When considering non-BRCA genes, the non-BRCA gene and non-BRCA HRR gene mutation rates were 12.2% (28/230) and 9.6% (22/230), respectively. Among them, 11.2% (19/170) early-onset patients had non-BRCA mutations; 18 (17.5%, 18/103) patients with BRCA-related family history were non-BRCA mutation carriers; besides, 8 (9.4%, 8/85) TNBC patients and 1 (7.7%, 1/13) primary bilateral breast cancer patients were identified. In addition, 15 (8.8%, 15/170) early-onset patients, 15 (14.6%, 15/103) patients with BRCA-related family history, 5 (5.9%, 5/85) TNBC patients and 1 (7.7%, 1/13) primary bilateral breast cancer patients carried non-BRCA HRR gene germline mutations. The relevance between non-BRCA HRR gene mutations and high-risk factors was lower than that between BRCA and high-risk factors. For each high-risk factor, only family history showed a correlation with the non-BRCA HRR gene mutation status (p = 0.036) (Table 4).
Sixty-eight patients were pathogenic or likely pathogenic mutation carriers. All the pathogenic or likely pathogenic variants were listed in Table 5. Among them, twenty-eight unique non-BRCA variants were identified, in which RAD51D variant c.270_271dup (p.Lys91fs) occurred twice. As mentioned above, PALB2 (n = 5), ATM (n = 4), RAD51D (n = 3) and TP53 (n = 3) were the top 4 genes among non-BRCA genes with the highest mutation rate. In addition, seven variants were identified in non-HRR genes: ERCC3 c.1854_1867del (p.Glu619fs); HOXB13 c.179del (p.Pro60fs); MLH1 c.1038G > C (p.Gln346His); PMS2 exon14-15cn_del; TP53 c.328dup (p.Arg110fs); TP53 c.637C > T (p.Arg213*) and TP53 c.733G > A (p.Gly245Ser).
Apart from pathogenic or likely pathogenic variants, 312 unique variants of uncertain significance (VUS) were identified among 175 (76.1%, 175/230) patients and 65 different genes. Of those, the most frequent genes were RAD54L (5.4%), followed by ATM (5.1%) and FANCA (4.6%). Table 6 summarizes all the VUSs identified in high penetrance breast cancer predisposition genes (BRCA1, BRCA2, CDH1, PALB2, PTEN and TP53).
The prevalence and spectrum of germline mutations in breast cancer patients vary across ethnicities (Hall et al., 2009; Han et al., 2011; Kwong et al., 2016). Compared with women of Western European descent (12.1%), women in Africa (15.6%) and Latin America (14.8%) had a higher incidence of BRCA1/2 pathogenic mutations (Hall et al., 2009). Data for the Asian population showed that the prevalence of BRCA1/2 germline mutations was 9.8% in South Korean non-familial high-risk breast cancer patients (Han et al., 2011) and 9.4% in Chinese hereditary breast-ovarian cancer (HBOC) families (Kwong et al., 2016). The application of NGS has enabled sequencing a large number of genes simultaneously, and thus, many other non-BRCA breast cancer susceptibility genes have been identified. The NCCN guideline list the susceptibility genes of hereditary breast cancer, including 6 high penetrance (BRCA1, BRCA2, CDH1, PALB2, PTEN and TP53) and 9 moderate to low penetrance (ATM, BARD1, BRIP1, CHEK2, NBN, NF1, RAD51C, RAD51D and STK11) susceptibility genes, most of which also participate in HRR pathway (Nielsen et al., 2016).
However, only a few studies to date report the prevalence of non-BRCA genes in the Chinese population. Samuel Guan Wei Ow et al. analyzed 419 Asian patients suspected to have hereditary breast cancer syndrome who underwent genetic testing and found that the frequency of detrimental mutations in non-BRCA genes varied from 0 to 13.3% due to differences in ethnicity (Ow et al., 2019). Using a 40 gene panel, another study found a non-BRCA gene mutation rate of 6.8% in a cohort of 937 Chinese breast cancer patients, with TP53 (1.9%), PALB2 (1.2%), CHEK2 (0.6%) and ATM (0.6%) being the major non-BRCA genes identified (Li et al., 2019). A recent study analyzed 1,338 Chinese high-risk breast cancer patients who tested mutation negative by a four-gene panel (BRCA1, BRCA2, PTEN and TP53) and found that pathogenic variants in cancer predisposition genes beyond BRCA1, BRCA2, PTEN and TP53 were detected in an additional 4.6% of patients using a multigene panel, with PALB2 (1.4%), RAD51D (0.8%) and ATM (0.8%) being the most commonly mutated genes (Kwong et al., 2020). Herein, we retrospectively analyze 230 high-risk breast cancer patients who had undergone peripheral blood germline 72 multigene panel testing, with a BRCA mutation rate of 19.1%; 12.2% of the patients carried non-BRCA mutations. Consistent with previous studies, the major mutated non-BRCA genes were PALB2 (2.2%), ATM (1.7%), RAD51D (1.3%) and TP53 (1.3%).
Apart from the high-frequency mutated genes, pathogenic variants were also detected in another 11 non-BRCA genes (ATR, BARD1, BRIP1, ERCC3, FANCA, HOXB13, MLH1, MRE11, RAD51C, RAD54L and PMS2). In addition to the hereditary breast cancer susceptibility genes listed by NCCN, MLH1 and PMS2 are risk genes recommended by NCCN for multigene testing. However, there is insufficient evidence to prove that MLH1 and PMS2 are related to breast cancer risk and mutation carriers need to be managed based on family history. The remaining 6 genes, ATR, ERCC3, FANCA, HOXB13, MRE11 and RAD54L, are not included in the risk genes recommended by NCCN guidelines. Among them, ATR, FANCA, MRE11 and RAD54L are HRR genes. MRE11, together with RAD50 and NBS1 (MRE11-RAD50-NBS1 complex), locates to the end of the double-strand breaks (DSBs) locus (Kinner et al., 2008; Wang et al., 2014) and activates ATM through the interaction between ATM and NBS1 (Bian et al., 2019), triggering ataxia telangiectasia and ATR activation. ATM and ATR then phosphorylate downstream targets, including BRCA1 and CHEK2. RAD54L binds double-strand DNA and induces a DNA topological change, which is thought to facilitate homologous DNA paring and stimulate DNA recombination. FANCA participates in the Fanconi anemia (FA) pathway, which is involved in DNA interstrand cross-link (ICL) damage repair and crucial for maintaining the integrity of the genome (Nakanishi et al., 2011). ERCC3 encodes an ATP-dependent DNA helicase that plays a role in the DNA nucleotide excision repair (NER) pathway. HOXB13 encodes a transcription factor that belongs to the homeobox gene family and plays a role in fetal skin development and cutaneous regeneration. HOXB13 gene mutations are often detected in prostate cancer patients. The correlation between these genes and the risk of hereditary breast cancer is not clear yet, and further studies are needed.
9.6% of high-risk breast cancer patients in our study carried non-BRCA HRR gene mutations. The current understanding of specific HRR genes is not comprehensive. Dana Sherill-Rofe and colleagues followed the coevolution of the HRR pathway across the eukaryotic life tree and defined a gold standard list of 79 well-established HRR genes. According to their function, these genes are divided into 6 parts: DSB recognition, end resection, FA pathway, regulation (DNA damage response), strand invasion and D-loop formation, synthesis and holiday junction processing (Pilié et al., 2019). Referring to the results of Dana Sherill-Rofe and taking intersection with the 72 multigene panel used in our study, we obtained 18 genes and defined these genes as HRR genes, including ATM, ATR, BARD1, BRCA1, BRCA2, BRIP1, CHEK1, CHEK2, FANCA, FANCI, MRE11A, NBN, PALB2, RAD50, RAD51B, RAD51C, RAD51D and RAD54L. Most patients with germline HRR gene mutations are not sporadic and have at least one first and/or second-degree relatives who also have a tumor, which indicates that the germline mutations of HRR genes are closely related to heredity (Li et al., 2008). In addition, mutations of non-BRCA HRR genes show similar PARPi sensitivity as BRCA1/2. PARPi can lead to cell death via a synergistic effect known as “synthetic lethality” with homologous recombination deficiency (HRD) (Zhang et al., 2012). The presence of pathogenic mutations in different non-BRCA HRR genes leads to difference responses to PARPi therapy. Abida et al. studied the response to the PARPi rucaparib in metastatic castration-resistant prostate cancer (mCRPC) patients with non-BRCA DNA damage repair pathogenic gene mutations and found that tumors with ATM, CDK12 and CHEK2 mutations had a limited response to rucaparib, while tumors with BRIP1, FANCA, PALB2 and RAD51B mutations benefitted from PARPis (Lin et al., 2016). In addition to PARP, ATM, ATR and CHEK1/2 may also be used for targeted therapy. The first ATR inhibitor, M6620, has been tested, and ATM inhibitors (such as M3541) are in clinical trials (Zhang et al., 2016; Lang et al., 2017).
In terms of each risk factor, our results suggest that the frequencies of BRCA mutations in early-onset breast cancer, familial breast cancer, TNBC and bilateral breast cancer (19.4–38.5%) are higher than those observed in other Chinese population studies (2.3–12.5%) (Gavande et al., 2016; Li et al., 2019; Sherill-Rofe et al., 2019; Abida et al., 2020; Hirsch et al., 2020; Foo et al., 2021) and the frequencies of non-BRCA mutations are also at a high level (7.7–17.5%). However, no pathologic variants were observed in the male patients in our study. In contrast, previous studies reported a high BRCA1/2 mutation frequency of 15.2–15.4% and a non-BRCA mutation rate of 15.4% in Chinese male breast cancer patients (Gavande et al., 2016; Li et al., 2019). Among high-risk factors, family history showed a correlation with both BRCA (p = 0.005) and non-BRCA HRR gene mutation status (p = 0.036). Besides, TNBC showed a correlation with BRCA1 gene mutation status (p = 0.038).
Almost all high-risk patients in our study met the standards of the expert consensus on the recommendation of BRCA genetic testing for breast cancer patients in China (Wang et al., 2018). The application of a multigene panel helped us to find 28 non-BRCA gene mutation carriers among 230 high-risk breast cancer patients. The latest consensus guidelines on genetic testing for hereditary breast cancer by the American Society of Breast Surgeons (ASBrS) recommends that all breast cancer patients should undergo multigene testing to assess whether they are at risk of hereditary cancer (Manahan et al., 2019). The Chinese Anti Cancer Association (CACA) breast cancer diagnosis and treatment guidelines (2019) recommends multigene testing for the following three situations: 1) patients with a personal or family history of tumor; 2) testing results can be fully explained; 3) testing results are helpful to clinical screening, diagnosis and treatment (Breast Cancer Professiona, 2019). However, in current clinical practice, multigene testing is mainly used in advanced breast cancer patients who have failed by multiline therapies in China. A recent study conducted multigene panel testing for breast cancer patients who had not undergone genetic testing and found that up to 50% of carriers of pathogenic or likely pathogenic mutations did not meet the NCCN guidelines (O’Leary et al., 2017; Beitsch et al., 2019; González-Santiago et al., 2020). All these indicate that the current standards for tumor polygene detection are still not perfect.
Among our cohort, 312 unique VUSs were identified among 175 (76.1%) patients and 65 different genes. However, the VUSs found in our cohort, including some rare variants, does not necessarily lead to the occurrence and development of tumors. The causes of tumors in these high-risk patients need to be further explored. On the one hand, reliable functional experiments are needed to reclassify the pathogenicity of VUSs. On the other hand, expanding genetic testing, such as whole-exome sequencing (WES) and whole genome sequencing (WGS), might help to reveal new breast cancer susceptibility genes.
Our research has some innovations and limitations. We focused on the prevalence and spectrum of non-BRCA, especially non-BRCA HRR gene germline mutations in the Chinese population, which is very rare in previous studies. Meanwhile, we analyzed the correlations between the high-risk factors and non-BRCA and non-BRCA HRR gene mutation status, which showed consistency with previous studies. However, as a retrospective study, the sample size for each risk factor was not controlled for, especially for the male and bilateral breast cancer groups, which limited the discovery of other possible variations.
In summary, 12.2% of high-risk breast cancer patients in our study carried non-BRCA gene mutations, with PALB2 (2.2%), ATM (1.7%), RAD51D (1.3%) and TP53 (1.3%) being the major non-BRCA genes mutated. As a BRCA-related family history is associated with HRR mutations, we strongly suggest that breast cancer patients with a BRCA-related family history receive comprehensive gene mutation testing, especially HRR genes, which are not only related to high risk of breast cancer, but also potentially related to PARPi targeted therapy in China. The exact relationship of rare gene mutations to breast cancer predisposition and the pathogenicity of VUSs need to be further investigated. With the widespread use of NGS technology, we are expecting to discover more breast cancer susceptibility genes. Guidelines for multigene testing and management of mutation carriers should be compiled to benefit Chinese breast cancer patients.
The original contributions presented in the study are publicly available. This data can be found here: https://www.biosino.org/node/project/detail/OEP002708
The studies involving human participants were reviewed and approved by Ethics Committee of Fudan University Shanghai Cancer Center. The patients/participants provided their written informed consent to participate in this study.
XZ was responsible for the revision of the manuscripts. YS completed the writing. JL, DS, YZ and BY were involved in the design of the manuscripts. QY, YX, CY, JZ, QW, XZ and QB were involved in the experiment design and operation. YS, QY and YX completed the documentation and contributed equally to this manuscript. All authors contributed to the article and approved the submitted version.
This work was supported by Innovation Group Project of Shanghai Municipal Health Commission (2019CXJQ03), Innovation Program of STCSM (20Z11900300), Shanghai Science and technology development fund (19MC1911000) and Shanghai Municipal Key Clinical Specialty (shslczdzk01301).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
We thank all the patients and their families for participation. We also appreciate the work of the investigators, study coordinators, operation staff, and the whole project team involved in this study.
Abida, W., Campbell, D., Patnaik, A., Shapiro, J. D., Sautois, B., Vogelzang, N. J., et al. (2020). Non-BRCA DNA Damage Repair Gene Alterations and Response to the PARP Inhibitor Rucaparib in Metastatic Castration-Resistant Prostate Cancer: Analysis from the Phase II TRITON2 Study. Clin. Cancer Res. 26 (11), 2487–2496. doi:10.1158/1078-0432.Ccr-20-0394
Beitsch, P. D., Whitworth, P. W., Hughes, K., Patel, R., Rosen, B., Compagnoni, G., et al. Underdiagnosis of Hereditary Breast Cancer: Are Genetic Testing Guidelines a Tool or an Obstacle? Jco (2019) 37(6):453, 460. doi:10.1200/jco.18.01631
Bian, L., Meng, Y., Zhang, M., and Li, D. (2019). MRE11-RAD50-NBS1 Complex Alterations and DNA Damage Response: Implications for Cancer Treatment. Mol. Cancer 18 (1), 169. doi:10.1186/s12943-019-1100-5
Breast Cancer Professional Committee of Chinese Anti-Cancer Association (2019). The Chinese Anti Cancer Association Breast Cancer Diagnosis and Treatment Guidelines (2019) (In Chinese). China Oncol. 29 (08), 609–680.
Carroll, J. C., Cremin, C., Allanson, J., Blaine, S. M., Dorman, H., Gibbons, C. A., et al. (2008). Hereditary Breast and Ovarian Cancers. Can. Fam. Physician 54 (12), 1691–1692.
Castroviejo‐Bermejo, M., Cruz, C., Llop‐Guevara, A., Gutiérrez‐Enríquez, S., Ducy, M., Ibrahim, Y. H., et al. (2018). A RAD 51 Assay Feasible in Routine Tumor Samples Calls PARP Inhibitor Response beyond BRCA Mutation. Embo Mol. Med. 10 (12). doi:10.15252/emmm.201809172
Colas, C., Golmard, L., de Pauw, A., Caputo, S. M., and Stoppa-Lyonnet, D. (2019). "Decoding Hereditary Breast Cancer" Benefits and Questions from Multigene Panel Testing. The Breast 45, 29–35. doi:10.1016/j.breast.2019.01.002
Foo, T., George, A., and Banerjee, S. (2021). PARP Inhibitors in Ovarian Cancer: An Overview of the Practice‐changing Trials. Genes Chromosomes Cancer 60, 385–397. Epub 2021/01/01. doi:10.1002/gcc.22935
Gavande, N. S., VanderVere-Carozza, P. S., Hinshaw, H. D., Jalal, S. I., Sears, C. R., Pawelczak, K. S., et al. (2016). DNA Repair Targeted Therapy: The Past or Future of Cancer Treatment? Pharmacol. Ther. 160, 65–83. Epub 2016/02/21. doi:10.1016/j.pharmthera.2016.02.003
González-Santiago, S., Cajal, T. R. Y., Ramón y Cajal, T., Aguirre, E., Alés-Martínez, J. E., Andrés, R., et al. (2020). SEOM Clinical Guidelines in Hereditary Breast and Ovarian Cancer (2019). Clin. Transl Oncol. 22 (2), 193–200. doi:10.1007/s12094-019-02262-0
Hall, M. J., Reid, J. E., Burbidge, L. A., Pruss, D., Deffenbaugh, A. M., Frye, C., et al. (2009). BRCA1andBRCA2mutations in Women of Different Ethnicities Undergoing Testing for Hereditary Breast-Ovarian Cancer. Cancer 115 (10), 2222–2233. Epub 2009/02/26. doi:10.1002/cncr.24200
Han, S. A., Park, S. K., Hyun Ahn, S., Hyuk Lee, M., Noh, D.-Y., Kim, L. S., et al. (2011). The Korean Hereditary Breast Cancer (KOHBRA) Study: Protocols and Interim Report. Clin. Oncol. 23 (7), 434–441. Epub 2011/04/19. doi:10.1016/j.clon.2010.11.007
Hirsch, S., Gieldon, L., Sutter, C., Dikow, N., and Schaaf, C. P. (2020). Germline Testing for Homologous Recombination Repair Genes-Opportunities and Challenges. Genes Chromosomes Cancer 60, 332–343. Epub 2020/10/21. doi:10.1002/gcc.22900
Kinner, A., Wu, W., Staudt, C., and Iliakis, G. (2008). -H2AX in Recognition and Signaling of DNA Double-Strand Breaks in the Context of Chromatin. Nucleic Acids Res. 36 (17), 5678–5694. doi:10.1093/nar/gkn550
Kwong, A., Shin, V. Y., Au, C. H., Law, F. B. F., Ho, D. N., Ip, B. K., et al. (2016). Detection of Germline Mutation in Hereditary Breast And/or Ovarian Cancers by Next-Generation Sequencing on a Four-Gene Panel. J. Mol. Diagn. 18 (4), 580–594. Epub 2016/05/10. doi:10.1016/j.jmoldx.2016.03.005
Kwong, A., Shin, V. Y., Chen, J., Cheuk, I. W. Y., Ho, C. Y. S., Au, C. H., et al. (2020). Germline Mutation in 1338 BRCA-Negative Chinese Hereditary Breast And/or Ovarian Cancer Patients. J. Mol. Diagn. 22 (4), 544–554. doi:10.1016/j.jmoldx.2020.01.013
Lang, G.-T., Shi, J.-X., Hu, X., Zhang, C.-H., Shan, L., Song, C.-G., et al. (2017). The Spectrum of BRCA Mutations and Characteristics of BRCA-Associated Breast Cancers in China: Screening of 2,991 Patients and 1,043 Controls by Next-Generation Sequencing. Int. J. Cancer 141 (1), 129–142. Epub 2017/03/16. doi:10.1002/ijc.30692
Li, J.-Y., Jing, R., Wei, H., Wang, M., Xiaowei, Q., Liu, H., et al. (2019). Germline Mutations in 40 Cancer Susceptibility Genes Among Chinese Patients with High Hereditary Risk Breast Cancer. Int. J. Cancer 144 (2), 281–289. doi:10.1002/ijc.31601
Li, W.-F., Hu, Z., Rao, N.-Y., Song, C.-G., Zhang, B., Cao, M.-Z., et al. (2008). The Prevalence of BRCA1 and BRCA2 Germline Mutations in High-Risk Breast Cancer Patients of Chinese Han Nationality: Two Recurrent Mutations Were Identified. Breast Cancer Res. Treat. 110 (1), 99–109. Epub 2007/09/14. doi:10.1007/s10549-007-9708-3
Lin, P.-H., Kuo, W.-H., Huang, A.-C., Lu, Y.-S., Lin, C.-H., Kuo, S.-H., et al. (2016). Multiple Gene Sequencing for Risk Assessment in Patients with Early-Onset or Familial Breast Cancer. Oncotarget 7 (7), 8310–8320. Epub 2016/01/30. doi:10.18632/oncotarget.7027
Manahan, E. R., Kuerer, H. M., Sebastian, M., Hughes, K. S., Boughey, J. C., Euhus, D. M., et al. (2019). Consensus Guidelines on Genetic` Testing for Hereditary Breast Cancer from the American Society of Breast Surgeons. Ann. Surg. Oncol. 26 (10), 3025–3031. Epub 2019/07/26. doi:10.1245/s10434-019-07549-8
Mirza, M. R., Monk, B. J., Herrstedt, J., Oza, A. M., Mahner, S., Redondo, A., et al. (2016). Niraparib Maintenance Therapy in Platinum-Sensitive, Recurrent Ovarian Cancer. N. Engl. J. Med. 375 (22), 2154–2164. doi:10.1056/NEJMoa1611310
Nakanishi, K., Cavallo, F., Perrouault, L., Giovannangeli, C., Moynahan, M. E., Barchi, M., et al. (2011). Homology-directed Fanconi Anemia Pathway Cross-Link Repair Is Dependent on DNA Replication. Nat. Struct. Mol. Biol. 18 (4), 500–503. doi:10.1038/nsmb.2029
The NCBI Handbook. Bethesda (MD): National Library of Medicine (US), National Center for Biotechnology Information. http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=Books.
Nielsen, F. C., van Overeem Hansen, T., and Sørensen, C. S. (2016). Hereditary Breast and Ovarian Cancer: New Genes in Confined Pathways. Nat. Rev. Cancer 16 (9), 599–612. Epub 2016/08/16. doi:10.1038/nrc.2016.72
O’Leary, E., Iacoboni, D., Holle, J., Michalski, S. T., Esplin, E. D., Yang, S., et al. (2017). Expanded Gene Panel Use for Women with Breast Cancer: Identification and Intervention beyond Breast Cancer Risk. Ann. Surg. Oncol. 24 (10), 3060–3066. doi:10.1245/s10434-017-5963-7
Ow, S. G. W., Ong, P. Y., and Lee, S.-C. (2019). Discoveries beyond BRCA1/2: Multigene Testing in an Asian Multi-Ethnic Cohort Suspected of Hereditary Breast Cancer Syndrome in the Real World. PLoS One 14 (3), e0213746. Epub 2019/03/16. doi:10.1371/journal.pone.0213746
Pilié, P. G., Tang, C., Mills, G. B., and Yap, T. A. (2019). State-of-the-art Strategies for Targeting the DNA Damage Response in Cancer. Nat. Rev. Clin. Oncol. 16 (2), 81–104. Epub 2018/10/26. doi:10.1038/s41571-018-0114-z
Richards, S., Aziz, N., Aziz, N., Bale, S., Bick, D., Das, S., et al. (2015). Standards and Guidelines for the Interpretation of Sequence Variants: a Joint Consensus Recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. 17 (5), 405–423. Epub 2015/03/06. doi:10.1038/gim.2015.30
Sherill-Rofe, D., Rahat, D., Findlay, S., Mellul, A., Guberman, I., Braun, M., et al. (2019). Mapping Global and Local Coevolution across 600 Species to Identify Novel Homologous Recombination Repair Genes. Genome Res. 29 (3), 439–448. doi:10.1101/gr.241414.118
Sun, J., Meng, H., Yao, L., Lv, M., Bai, J., Zhang, J., et al. (2017). Germline Mutations in Cancer Susceptibility Genes in a Large Series of Unselected Breast Cancer Patients. Clin. Cancer Res. 23 (20), 6113–6119. Epub 2017/07/21. doi:10.1158/1078-0432.CCR-16-3227
Sung, P.-L., Wen, K.-C., Chen, Y.-J., Chao, T.-C., Tsai, Y.-F., Tseng, L.-M., et al. (2017). The Frequency of Cancer Predisposition Gene Mutations in Hereditary Breast and Ovarian Cancer Patients in Taiwan: From BRCA1/2 to Multi-Gene Panels. PLoS One 12 (9), e0185615. Epub 2017/09/30. doi:10.1371/journal.pone.0185615
Telli, M. L., Timms, K. M., Reid, J., Hennessy, B., Mills, G. B., Jensen, K. C., et al. (2016). Homologous Recombination Deficiency (HRD) Score Predicts Response to Platinum-Containing Neoadjuvant Chemotherapy in Patients with Triple-Negative Breast Cancer. Clin. Cancer Res. 22 (15), 3764–3773. doi:10.1158/1078-0432.ccr-15-2477
Tham, K.-C., Kanaar, R., and Lebbink, J. H. G. (2016). Mismatch Repair and Homeologous Recombination. DNA Repair 38, 75–83. doi:10.1016/j.dnarep.2015.11.010
Wang, H. X., Sheng, Y., and Liu, Y. (2018). Expert Consensus on BRCA1/2 Genetic Testing and Clinical Application in Chinese Breast Cancer Patients (2018) (In Chinese). China Oncol. 28 (10), 787–800. doi:10.19401/j.cnki.1007-3639.2018.10.011
Wang, Q., Goldstein, M., Alexander, P., Wakeman, T. P., Sun, T., Feng, J., et al. (2014). Rad17 Recruits the MRE11-RAD50-NBS1 Complex to Regulate the Cellular Response to DNA Double-Strand Breaks. EMBO J. 33 (8), 862–877. doi:10.1002/embj.201386064
World Health Organization (2021). Global Cancer Observatory. Available at: https://gco.iarc.fr/today/online-analysis-pie?v=2020&mode=cancer&mode_population=continents&population=900&populations=160&key=total&sex=2&cancer=39&type=0&statistic=5&prevalence=0&population_group=0&ages_group%5B%5D=0&ages_group%5B%5D=17&nb_items=7&group_cancer=1&include_nmsc=1&include_nmsc_other=1&half_pie=0&donut=0.
Yao, L., Sun, J., Zhang, J., He, Y., Ouyang, T., Li, J., et al. (2016). Breast Cancer Risk in Chinese Women with BRCA1 or BRCA2 Mutations. Breast Cancer Res. Treat. 156 (3), 441–445. doi:10.1007/s10549-016-3766-3
Zhang, J., Pei, R., Pang, Z., Ouyang, T., Li, J., Wang, T., et al. (2012). Prevalence and Characterization of BRCA1 and BRCA2 Germline Mutations in Chinese Women with Familial Breast Cancer. Breast Cancer Res. Treat. 132 (2), 421–428. Epub 2011/05/27. doi:10.1007/s10549-011-1596-x
Keywords: breast cancer, high risk, non-BRCA genes, germline mutation, Chinese
Citation: Su Y, Yao Q, Xu Y, Yu C, Zhang J, Wang Q, Li J, Shi D, Yu B, Zeng Y, Zhu X, Bai Q and Zhou X (2021) Characteristics of Germline Non-BRCA Mutation Status of High-Risk Breast Cancer Patients in China and Correlation with High-Risk Factors and Multigene Testing Suggestions. Front. Genet. 12:674094. doi: 10.3389/fgene.2021.674094
Received: 20 April 2021; Accepted: 05 November 2021;
Published: 30 November 2021.
Edited by:
Tania Lee Slatter, University of Otago, New ZealandReviewed by:
Maria Del Pilar Estevez Diz, Universidade de São Paulo, BrazilCopyright © 2021 Su, Yao, Xu, Yu, Zhang, Wang, Li, Shi, Yu, Zeng, Zhu, Bai and Zhou. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiaoyan Zhou, eHl6aG91MTAwQDE2My5jb20=
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.