
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Endocrinol. , 13 March 2025
Sec. Reproduction
Volume 16 - 2025 | https://doi.org/10.3389/fendo.2025.1542534
 Chun-I. Lee1,2,3†
Chun-I. Lee1,2,3† Yu-Jen Lee4,5†
Yu-Jen Lee4,5† Tsung-Hsien Lee1,3,6
Tsung-Hsien Lee1,3,6 Chi-Ying Lee4,7
Chi-Ying Lee4,7 Hui-Mei Tsao4
Hui-Mei Tsao4 En-Hui Cheng4,5
En-Hui Cheng4,5 Chun-Chia Huang1
Chun-Chia Huang1 Shun-Fa Yang6
Shun-Fa Yang6 Maw-Sheng Lee1,3,6*
Maw-Sheng Lee1,3,6*Background: Matrix metalloproteinases (MMPs) and tissue inhibitors of metalloproteinases (TIMPs) are critical regulators of extracellular matrix (ECM) proteolysis and play a pivotal role in trophoblast invasion during embryo implantation. This study aimed to investigate the effects of single-nucleotide polymorphisms (SNPs) in MMP and TIMP genes on clinical outcomes in women undergoing in vitro fertilization (IVF).
Methods: This retroprospective study included 1014 women undergoing their first fresh IVF cycle without donor eggs at Lee Women’s Hospital between January 2014 and December 2015. Peripheral blood samples were collected from all participants for DNA extraction and SNP genotyping using real-time polymerase chain reaction. The study focused on three SNPs: TIMP1 (rs4898 C/T), TIMP2 (rs2277698 C/T), and MMP2 (rs243865 C/T). Associations between these SNPs and IVF outcomes, including clinical pregnancy, embryo implantation, abortion, and live birth rates, were analyzed.
Results: Among 560 patients analyzed, no significant differences were observed in baseline characteristics between the live birth and non-live birth groups. However, the minor alleles (CT+TT) of MMP2 (rs243865) and TIMP2 (rs2277698) were significantly more frequent in the non-live birth group (MMP2: 24.4% vs. 17.7%, p = 0.044; TIMP2: 48.1% vs. 34.4%, p = 0.001). In contrast, no significant differences in the genotype distribution of TIMP1 (rs4898) were noted between the groups. Logistic regression analysis identified the minor T allele of TIMP2 as a significant predictor of non-live birth (adjusted odds ratio: 1.725; 95% CI: 1.217–2.445; p = 0.002). Combined genotypes of MMP2/TIMP2, such as CC/CT+TT and CT+TT/CT+TT, were associated with an increased risk of non-live birth, even after adjusting for covariates.
Conclusions: The study demonstrates that the minor T allele of TIMP2 (rs2277698 C/T) is associated with poor IVF outcomes, particularly non-live birth. This finding highlights the potential role of genetic variations in TIMP2 in influencing clinical outcomes of IVF. Further research is warranted to elucidate the underlying mechanisms in larger and more diverse populations.
Successful pregnancy relies on the proper development and implantation of the embryo. Embryo implantation involves apposition, adhesion, and invasion into the maternal endometrial matrix (1, 2). Extensive degradation and remodeling of the endometrial extracellular matrix (ECM) are crucial for this process, regulated by interactions between ECM components and the matrix metalloproteinase (MMP) family (3). Recent studies continue to highlight the essential role of ECM and its regulators in early pregnancy, with particular focus on novel therapeutic targets for implantation issues (4).
The MMP family consists of structurally related zinc-dependent endopeptidases that degrade various ECM proteins, thereby participating in ECM remodeling (5). These enzymes are involved in multiple physiological processes, including embryo implantation, trophoblast invasion and migration, and endometrial decidualization (6). MMP2, secreted by the embryo during the blastocyst stage, serves as the primary gelatinase in early pregnancy (6 to 8 weeks) and is a key regulator of embryonic invasion. The balance between MMP2 and tissue inhibitors of metalloproteinases (TIMPs) is essential for maintaining normal pregnancy (7, 8).
The MMP2/TIMP2 complex plays a critical role in endometrial remodeling during early pregnancy. Elevated levels of MMP2 and TIMP2 have been observed in patients with spontaneous abortion (9). Singh et al. demonstrated that dysregulation of MMP2 can result in excessive degradation of the endometrial ECM, leading to recurrent spontaneous abortion (RSA) (10). These findings highlight the importance of maintaining the balance between MMP and TIMP expression levels for successful embryo implantation, as an imbalance may contribute to implantation failure (8, 11). A meta-analysis investigating the association between MMP gene polymorphisms and RSA identified a significant correlation between the MMP2 (-735C>T) T allele and RSA risk (12).
To explore these findings further, we selected three specific single-nucleotide polymorphisms (SNPs): TIMP1 rs4898, TIMP2 rs2277698, and MMP2 rs243865. TIMP1 rs4898 and TIMP2 rs2277698 are located within the genes and play key roles in regulating MMP activity, which is crucial for endometrial remodeling and embryo implantation. MMP2 rs243865, located in the promoter region, affects MMP2 expression and alters transcription factor binding sites. Previous studies have shown that these SNPs are associated with recurrent spontaneous abortion or implantation failure (10, 12). Based on this evidence, we chose these loci to investigate their potential impact on IVF outcomes.
Most studies exploring the link between MMPs and infertility have focused on women experiencing RSA. This study investigates the effects of single-nucleotide polymorphisms (SNPs) in MMP and TIMP genes on clinical outcomes in women undergoing their first in vitro fertilization (IVF) cycle.
This retrospective study was conducted at Lee Women’s Hospital (Taichung, Taiwan) from January 2014 to December 2015. We included Han Chinese women under 45 years of age who had undergone their first IVF or intracytoplasmic sperm injection (ICSI) cycle with fresh blastocyst transfer during the study period. Women with autoimmune disorders, inflammatory diseases, genetic abnormalities, or other systemic diseases, as well as those who received donated eggs, were excluded. The study was approved by the Institutional Review Board of Chung Shan Medical University Hospital (CS13194).
Our objective was to identify associations of single-nucleotide polymorphisms (SNPs) in the MMP2 rs243865, TIMP1 rs4898, and TIMP2 rs2277698 genes with IVF clinical outcomes. The primary outcomes assessed were clinical pregnancy, implantation, abortion, and live birth rates. Clinical pregnancy was defined as the detection of an intrauterine gestational sac on ultrasonography. The implantation rate was calculated as the number of observed gestational sacs divided by the number of transferred embryos. Abortion was defined as the natural loss of a pregnancy before the 20th gestational week. Live birth was defined as the delivery of a baby showing signs of life.
All patients underwent ovarian stimulation followed by oocyte retrieval using the gonadotropin-releasing hormone (GnRH) antagonist protocol, as previously described (13). The same protocol was applied to all patients to avoid bias. The procedure began with daily injections of 0.5 mg leuprolide acetate (Lupron; Takeda Pharmaceutics, Konstanz, Germany) starting on day 21 of the previous menstrual cycle. On day 2 or 3 of the cycle, recombinant follicle-stimulating hormone (FSH; Gonal-F; Merck-Serono, Darmstadt, Germany) or highly purified FSH (Menopur; Ferring Pharmaceuticals, Kiel, Germany) was administered in flexible daily doses to promote follicular growth. Human chorionic gonadotropin (hCG; 10,000 IU; Profasi, Serono, Norwell, MA, USA) was used to trigger final oocyte maturation. Oocyte retrieval was performed 36 to 38 hours later. Fertilization was achieved through either conventional insemination or ICSI, depending on semen parameters. Fresh blastocyst transfer was performed within the study period.
Venous blood samples were collected on the day of oocyte retrieval for DNA extraction and subsequent genotyping. Genomic DNA was extracted from ethylenediaminetetraacetate (EDTA)-treated venous blood using the QIAamp DNA Blood Mini Kit (Qiagen, Valencia, USA) according to the manufacturer’s instructions (14). The extracted DNA was dissolved in Tris-EDTA buffer (10 mM Tris, 1 mM EDTA; pH 7.8). DNA quality was assessed by measuring optical density at 260 nm. The final DNA solution was stored at −20°C. This DNA served as the template for polymerase chain reaction (PCR). The evaluated SNPs included MMP2 rs243865, TIMP1 rs4898, and TIMP2 rs2277698, using a TaqMan allelic discrimination assay (TaqMan SNP). Allelic discrimination was performed with the ABI StepOne Real-Time PCR System (Applied Biosystems, Foster City, CA, USA) (15). Primer sequences for each genotype are listed in Table 1.
To analyze the effects of SNPs on live birth, we performed propensity score matching (PSM) to control for factors influencing live birth. Covariates used for calculating propensity scores included maternal age, duration of infertility, anti-Müllerian hormone (AMH) levels, and endometrial thickness on the day of embryo transfer. We employed the nearest neighbor matching algorithm with caliper matching, setting the standard deviation of the logit of the propensity score at 0.02. We matched 270 women who experienced non-live birth with 270 women who experienced live birth (control group) in a 1:1 ratio. Figure 1 presents a flowchart illustrating the group allocation.
Continuous variables were compared using Student’s t-test and are presented as mean ± SD values. Categorical variables were compared using the chi-square test and are presented as numbers (percentages). Associations between the tested SNPs and live birth were investigated using different genetic models: codominant (AA vs. Aa vs. aa), dominant (AA+Aa vs. aa), and allelic (A vs. a) models, employing the chi-square test. To enhance accuracy, both unadjusted (univariate) and adjusted (multivariate) analyses were performed using logistic regression, conducted through a generalized estimating equation model. Data analysis was performed using SPSS Statistics (version 22.0) for Windows (IBM Corporation, Armonk, NY, USA). A p-value of <0.05 was considered statistically significant.
The patient cohort in this study comprised 1014 women. Table 2 presents the baseline characteristics of the study groups both before and after propensity score matching (PSM). Initially, we identified 270 women who had experienced live birth and 744 women who had experienced non-live birth. Before PSM, significant differences between the groups were observed in maternal age (p < 0.001), duration of infertility (p = 0.045), AMH levels (p < 0.001), basal FSH levels (p = 0.018), and endometrial thickness on the day of embryo transfer (p = 0.005). After PSM, these differences were no longer significant, indicating that comparable cohorts had been established.
Subsequently, we examined the clinical outcomes of the two groups after PSM, as outlined in Table 3. Significant differences were observed between the groups in the rates of clinical pregnancy, implantation, and miscarriage (p < 0.001). The non-live birth group exhibited a substantially high miscarriage rate (79.2%), even among individuals who initially achieved pregnancy. Based on these findings, we hypothesized that differences in genotype distribution might influence the clinical outcomes between the two groups.
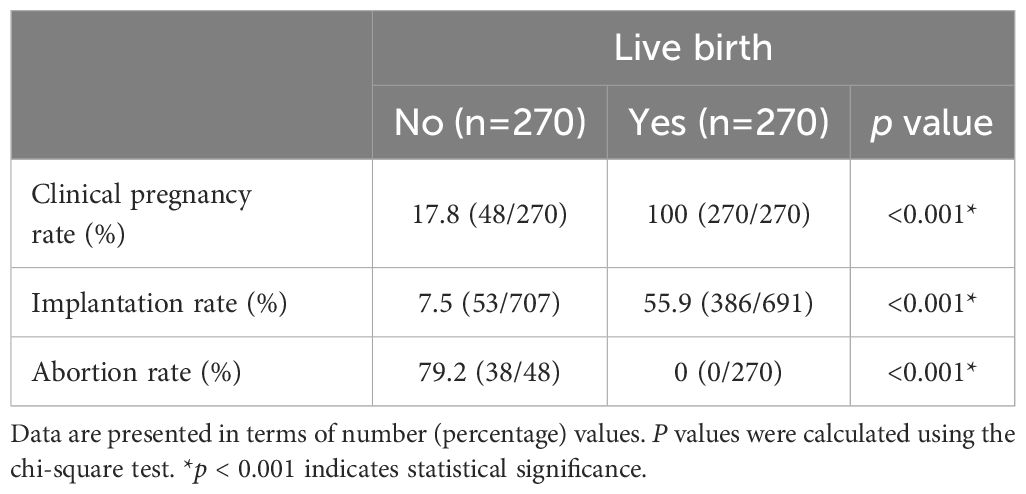
Table 3. Clinical outcomes in the non-live birth and live birth groups after propensity score matching.
The genotype distribution and allele frequencies of MMP and TIMP polymorphisms between the study groups are presented in Table 4. For the MMP2 C>T polymorphism (rs243865), the T allele exhibited a higher frequency in the non-live birth group than in the live birth group (13.1% vs. 9.1%; p = 0.033). When genotypes carrying the minor allele were combined (CT+TT), the allele frequency was significantly higher in the non-live birth group compared to the live birth group (24.4% vs. 17.7%; p = 0.044). Similarly, the T allele of the TIMP2 C>T SNP (rs2277698) displayed a higher frequency in the non-live birth group (27.2% vs. 18.7%; p = 0.001). Genotypes carrying the minor alleles exhibited significantly increased frequencies in both codominant and dominant models (CT: 41.9% vs. 31.5%; TT: 6.3% vs. 2.9%; CT+TT: 48.1% vs. 34.4%; p = 0.001). However, no significant between-group differences were observed in the allele frequency or genotype distribution of the TIMP1 T>C SNP (rs4898).
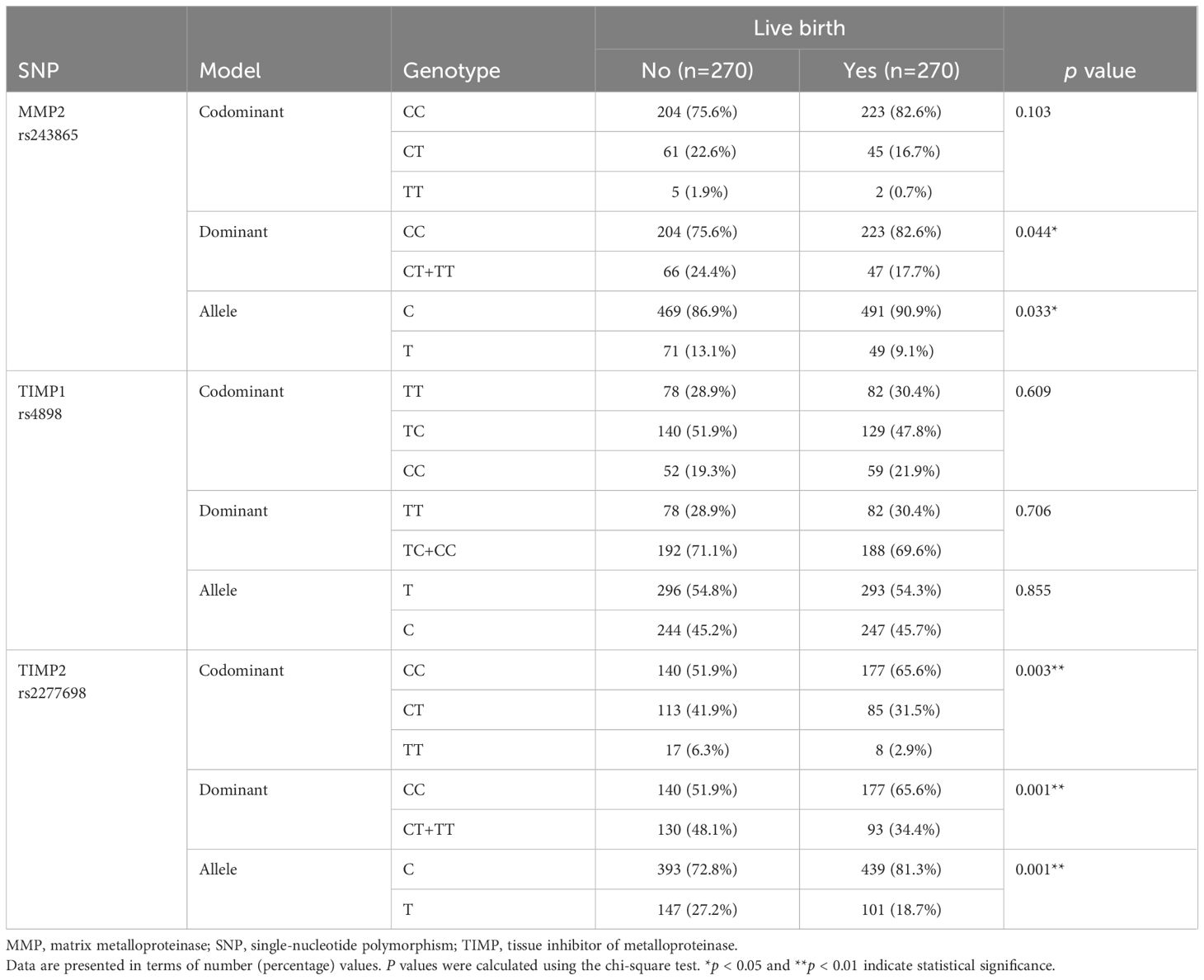
Table 4. Genotype distribution and allele frequencies of MMP2, TIMP1 and TIMP2 SNP in the non-live birth group and live birth group.
Table 5 presents the odds ratios (ORs) for the association between gene polymorphisms and non-live birth. A logistic regression analysis was conducted to investigate genotypes as predictors of non-live birth among women undergoing IVF. The univariate analysis revealed that genotypes containing the minor allele (CT+TT) of MMP2 or TIMP2 were significant predictors of non-live birth (unadjusted OR for MMP2: 1.535, p = 0.045; unadjusted OR for TIMP2: 1.767, p = 0.001). After adjusting for covariates, only the genotype containing the minor allele of TIMP2 remained a significant predictor (adjusted OR: 1.725; 95% CI: 1.217 to 2.445; p = 0.002).
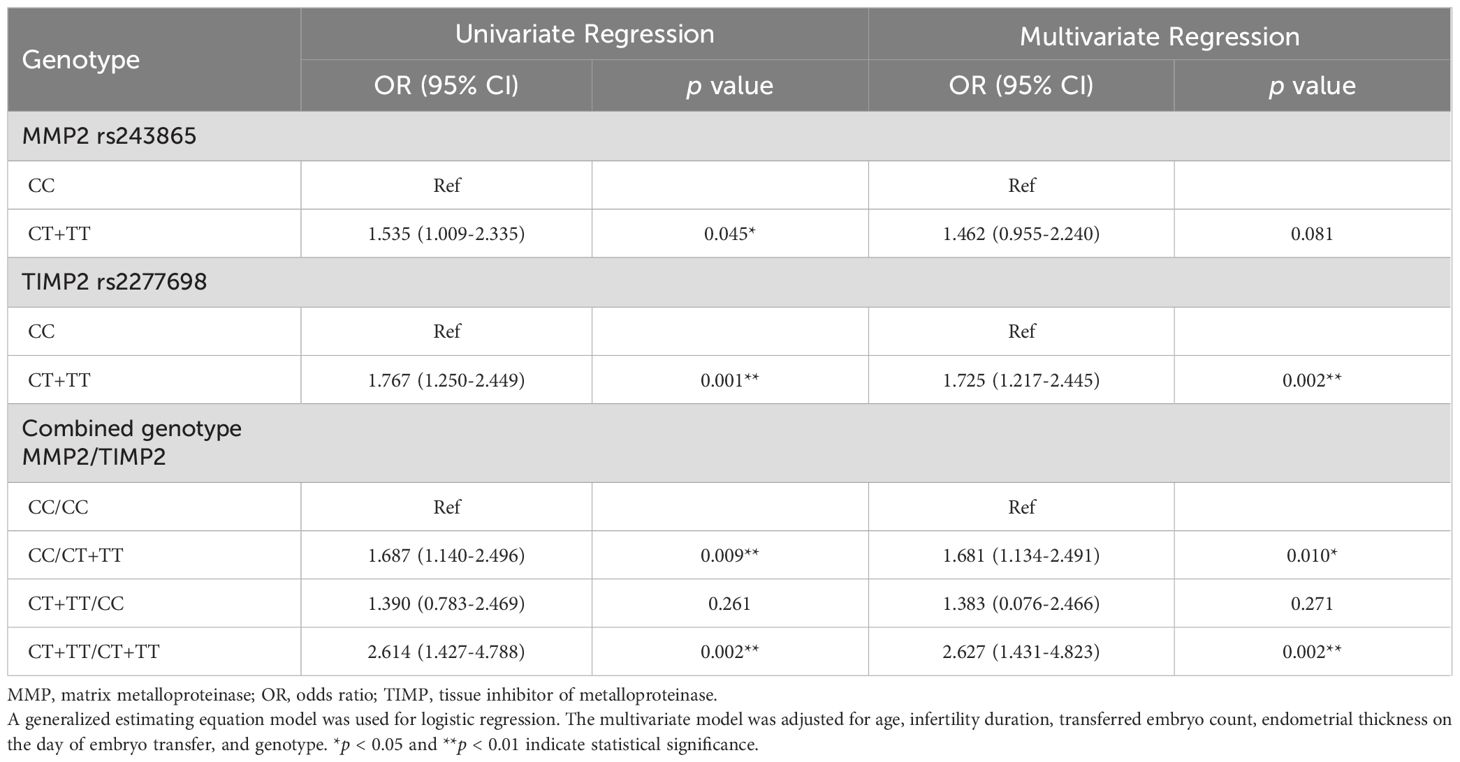
Table 5. Odds ratios for association between gene polymorphisms and non-live birth (dominant model).
A combined analysis was performed using the dominant models for MMP2 (CC) and TIMP2 (CT+TT). Compared with the reference genotype, the combined MMP2/TIMP2 genotypes CC/CT+TT and CT+TT/CT+TT were associated with 1.687- and 2.614-fold increases in the risk of non-live birth (unadjusted OR for CC/CT+TT: 1.687, p = 0.009; unadjusted OR for CT+TT/CT+TT: 2.614, p = 0.002). After adjusting for covariates, these combined genotypes remained significant predictors (adjusted OR for CC/CT+TT: 1.681, p = 0.010; adjusted OR for CT+TT/CT+TT: 2.627, p = 0.002). However, no significant association was observed between the CT+TT/CC genotype of MMP2/TIMP2 and non-live birth.
These findings indicate that the TIMP2 genotype containing the minor allele (T allele) is a key predictor of the risk of non-live birth among women undergoing IVF.
MMP2 rs243865 polymorphism is a point mutation located in the promoter region (–1306), where a C→T transition disrupts the binding of specificity protein 1 (SP1), significantly reducing promoter activity and downregulating MMP2 expression (16–18). Studies have demonstrated that MMP2 expression in carriers of the variant genotypes (CT+TT) is significantly lower than in those with the wild-type genotype (CC) (19). In Chinese women, the MMP2 rs243865 T allele is closely associated with the risk of recurrent spontaneous abortion (RSA) (20). Our study further supports these findings, revealing that the clinical pregnancy and implantation rates in the non-live birth group were significantly lower than those in the live birth group, while the miscarriage rate was notably higher. Moreover, the distribution of the variant genotypes (CT+TT) differed significantly between groups, suggesting that the MMP2 rs243865 T allele may contribute to adverse pregnancy outcomes by influencing extracellular matrix (ECM) regulation.
As the primary endogenous regulator of MMP2, TIMP2 maintains ECM balance by forming TIMP2/MMP2 complexes (21). TIMP2 exerts a concentration-dependent dual effect on MMP2 activity: low concentrations enhance MMP2 activity, whereas high concentrations inhibit it (22–24). TIMP2 rs2277698 polymorphism involves a C→T transition at position 303, resulting in a synonymous mutation (Ser101). Although the precise impact of this polymorphism on TIMP2 expression remains unclear, bioinformatic analyses suggest its potential role in splicing regulation and alterations in transcriptional activity. Moreover, its strong linkage disequilibrium with other SNPs, such as rs9889410 and rs11654470, may further modulate TIMP2 expression (25–27), emphasizing the complexity of genetic variations in the regulation of the extracellular matrix (ECM).
Evidence from studies on recurrent pregnancy loss (RPL) indicates that TIMP2 is overexpressed in both murine models and women with a history of RPL, suggesting its potential as a predictive marker for RPL (6, 28, 29). Excessive TIMP2 expression disrupts the fine-tuned MMP2/TIMP2 balance, leading to abnormal ECM degradation in the endometrium, which interferes with embryo implantation and normal placental development, potentially causing spontaneous miscarriage (6, 9). In our study, the proportion of carriers of the TIMP2 rs2277698 T allele genotype was significantly higher in the non-live birth group compared to the live birth group. This association was validated under codominant and dominant genetic models. Logistic regression analysis further identified the T allele as an independent predictor of non-live birth, underscoring its potential impact on pregnancy outcomes.
While our study establishes a significant association between TIMP2 rs2277698 polymorphism and adverse pregnancy outcomes, certain limitations must be acknowledged. The primary limitation is the lack of functional experiments to elucidate how the TIMP2 rs2277698 polymorphism influences gene expression and its downstream effects on IVF outcomes. Previous studies have suggested that synonymous mutations, such as rs2277698, may affect mRNA splicing efficiency and stability, thereby altering protein expression (25, 27). To address this gap, future studies should include gene expression analyses using qRT-PCR and Western blot to quantify TIMP2 levels in carriers of different genotypes. Furthermore, luciferase reporter assays could provide mechanistic insights into the impact of this polymorphism on transcriptional activity.
Another limitation is the absence of long-term follow-up data to assess the effects of identified SNPs on offspring health and developmental milestones. Existing evidence suggests that dysregulation of the MMP2/TIMP2 axis during pregnancy may impair placental function, thereby affecting fetal development (28, 29). Longitudinal studies linking maternal genotypes to offspring outcomes, such as growth, cognitive development, and metabolic health, are critical for understanding the broader clinical implications of these genetic associations.
Furthermore, our study population was limited to Han Chinese women undergoing their first IVF cycle, which may introduce selection bias and restrict the generalizability of the findings to other populations and natural pregnancies. To mitigate this limitation, large-scale studies in diverse populations are warranted to validate and extend our findings.
Lastly, the therapeutic potential of targeting the TIMP2/MMP2 balance remains an area for exploration. Preclinical studies suggest that small-molecule inhibitors or recombinant TIMP2 proteins may restore ECM balance under pathological conditions (21, 22). Additionally, antioxidant supplementation, such as resveratrol, has shown promise in modulating MMP activity (24). Experimental studies investigating these therapeutic approaches in IVF patients carrying the TIMP2 rs2277698 T allele could provide novel strategies to improve reproductive outcomes.
In conclusion, our study highlights the association between TIMP2 rs2277698 polymorphism and adverse clinical outcomes in IVF. These findings emphasize the critical role of maintaining ECM remodeling balance and advocate for further research into functional mechanisms, long-term offspring outcomes, and therapeutic interventions to enhance reproductive success.
All elements of this study involving human participants were reviewed and approved by the Institutional Review Board of Chung Shan Medical University, Taichung, Taiwan (CS13194). The ethics committee waived the requirement for written informed consent for participation.
The raw data supporting the conclusions of this article will be made available by the authors without undue reservation.
This study was approved by the Institutional Review Board of Chung Shan Medical University, Taichung, Taiwan, and met all ethical standards (Approval No.: CS13194). The study was conducted in accordance with local regulations and institutional requirements. The Institutional Review Board waived the requirement for written informed consent for this study, no written consent forms were obtained. Instead, participants were verbally informed and provided their consent.
C-IL: Conceptualization, Data curation, Supervision, Validation, Writing – review & editing. Y-JL: Data curation, Investigation, Methodology, Software, Validation, Writing – original draft, Writing – review & editing. T-HL: Supervision, Validation, Writing – review & editing. C-YL: Methodology, Software, Validation, Writing – review & editing. H-MT: Data curation, Methodology, Writing – review & editing. E-HC: Conceptualization, Supervision, Writing – review & editing. C-CH: Methodology, Supervision, Writing – review & editing. S-FY: Data curation, Methodology, Validation, Writing – review & editing. M-SL: Conceptualization, Supervision, Writing – review & editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
We thank Chung-I Chen, Chien-Hong Chen, and Hsiu-Hui Chen for their assistance with laboratory techniques in ART cycles. This manuscript was edited by Wallace Academic Editing.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
AMH, anti-Müllerian hormone; ECM, extracellular matrix; E2, estradiol; FSH, follicle-stimulating hormone; GnRH, gonadotropin-releasing hormone; hCG, human chorionic gonadotropin; ICSI, intracytoplasmic sperm injection; IVF, in vitro fertilization; LH, luteinizing hormone; MMP, matrix metalloproteinase; OR, odds ratio; PCR, polymerase chain reaction; PSM, propensity score matching; RPL, recurrent pregnancy loss; RSA, recurrent spontaneous abortion; SNP, single-nucleotide polymorphism; TIMP, tissue inhibitor of metalloproteinase.
1. Ochoa-Bernal MA, Fazleabas AT. Physiologic events of embryo implantation and decidualization in human and non-human primates. Int J Mol Sci. (2020) 21:1973. doi: 10.3390/ijms21061973
2. Kim SM, Kim JS. A review of mechanisms of implantation. Dev Reprod. (2017) 21:351–9. doi: 10.12717/DR.2017.21.4.351
3. Stamenkovic I. Extracellular matrix remodelling: the role of matrix metalloproteinases. J Pathol. (2003) 200:448–64. doi: 10.1002/path.1400
4. Jing MY, Chen X, Qiu HX, He WH, Zhou Y, Li D, et al. Insights into the immunomodulatory regulation of matrix metalloproteinase at the maternal-fetal interface during early pregnancy and pregnancy-related diseases. Front Immunol. (2023) 13:1067661. doi: 10.3389/fimmu.2022.1067661
5. Verma RP, Hansch C. Matrix metalloproteinases (MMPs): chemical-biological functions and (Q)SARs. Bioorg Med Chem. (2007) 15:2223–68. doi: 10.1016/j.bmc.2007.01.011
6. Cabral-Pacheco GA, Garza-Veloz I, Castruita-De-la-Rosa C, Ramirez-Acuña JM, Perez-Romero BA, Guerrero-Rodriguez JA, et al. The roles of matrix metalloproteinases and their inhibitors in human diseases. Int J Mol Sci. (2020) 21:9739. doi: 10.3390/ijms21249739
7. Kanca H, Walter I, Miller I, Schäfer-Somi S, Izgur H, Aslan S. Expression and activity of matrix metalloproteinases in the uterus of bitches after spontaneous and induced abortion. Reprod Domest Anim. (2011) 46:197–204. doi: 10.1111/j.1439-0531.2010.01619.x
8. Benkhalifa M, Zayani Y, Bach V, Copin H, Feki M, Benkhalifa M, et al. Does the dysregulation of matrix metalloproteinases contribute to recurrent implantation failure? Expert Rev Proteomics. (2018) 15:311–23. doi: 10.1080/14789450.2018.1464915
9. Nissi R, Talvensaari-Mattila A, Kotila V, Niinimäki M, Järvelä I, Turpeenniemi-Hujanen T. Circulating matrix metalloproteinase MMP-9 and MMP-2/TIMP-2 complex are associated with spontaneous early pregnancy failure. Reprod Biol Endocrinol. (2013) 15:11. doi: 10.1186/1477-7827-11-2
10. Singh N, Prasad P, Das B, Rastogi S. Involvement of matrix metalloproteinases and their inhibitors in endometrial extracellular matrix turnover in Chlamydia trachomatis-infected recurrent spontaneous aborters. Pathog Dis. (2017) 75(1):ftx007. doi: 10.1093/femspd/ftx007
11. Rahat B, Sharma R, Bagga R, Hamid A, Kaur J. Imbalance between matrix metalloproteinases and their tissue inhibitors in preeclampsia and gestational trophoblastic diseases. Reproduction. (2016) 152:11–22. doi: 10.1530/REP-16-0060
12. Yan Y, Fang LL, Li YX, Yu YP, Li YR, Cheng JC, et al. Association of MMP2 and MMP9 gene polymorphisms with the recurrent spontaneous abortion: A meta-analysis. Gene. (2021) 767:145173. doi: 10.1016/j.gene.2020.145173
13. Wu CH, Yang SF, Tsao HM, Chang YJ, Lee TH, Lee MS. Anti-müllerian hormone gene polymorphism is associated with clinical pregnancy of fresh IVF cycles. Int J Environ Res Public Health. (2019) 16:841. doi: 10.3390/ijerph16050841
14. Chung TT, Pan MS, Kuo CL, Wong RH, Lin CW, Chen MK, et al. Impact of RECK gene polymorphisms and environmental factors on oral cancer susceptibility and clinicopathologic characteristics in Taiwan. Carcinogenesis. (2011) 32:1063–8. doi: 10.1093/carcin/bgr083
15. Weng SL, Tzeng SL, Lee CI, Liu CH, Huang CC, Yang SF, et al. Association between gnRH receptor polymorphisms and luteinizing hormone levels for low ovarian reserve infertile women. Int J Environ Res Public Health. (2021) 18:7006. doi: 10.3390/ijerph18137006
16. Banday MZ, Sameer AS, Mir AH, Mokhdomi TA, Chowdri NA, Haq E. Matrix metalloproteinase (MMP) -2, -7 and -9 promoter polymorphisms in colorectal cancer in ethnic Kashmiri population - A case-control study and a mini review. Gene. (2016) 589:81–9. doi: 10.1016/j.gene.2016.05.028
17. Price SJ, Greaves DR, Watkins H. Identification of novel, functional genetic variants in the human matrix metalloproteinase-2 gene: Role of Sp1 in allele-specific transcriptional regulation. J Biol Chem. (2001) 276:7549–58. doi: 10.1074/jbc.M010242200
18. Cui Y, Zhu JJ, Ma CB, Cui K, Wang F, Ni SH, et al. Genetic polymorphisms in MMP 2, 3 and 9 genes and the susceptibility of osteosarcoma in a Chinese Han population. Biomarkers. (2016) 21:160–3. doi: 10.3109/1354750X.2015.1118550
19. Singh N, Hussain S, Sharma U, Suri V, Nijhawan R, Bharadwaj M, et al. The protective role of the -1306C>T functional polymorphism in matrix metalloproteinase-2 gene is associated with cervical cancer: implication of human papillomavirus infection. Tumour Biol. (2016) 37:5295–303. doi: 10.1007/s13277-015-4378-y
20. Li L, Liu J, Qin S, Li R. The association of polymorphisms in promoter region of MMP2 and MMP9 with recurrent spontaneous abortion risk in Chinese population. Med (Baltimore). (2018) 97:e12561. doi: 10.1097/MD.0000000000012561
21. Visse R, Nagase H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ Res. (2003) 92:827–39. doi: 10.1161/01.RES.0000070112.80711.3D
22. Caterina JJ, Yamada S, Caterina NC, Longenecker G, Holmback K, Shi J, et al. Inactivating mutation of the mouse tissue inhibitor of metalloproteinases-2 (Timp-2) gene alters proMMP-2 activation. J Biol Chem. (2000) 275:26416–22. doi: 10.1074/jbc.M001271200
23. Worley JR, Thompkins PB, Lee MH, Hutton M, Soloway P, Edwards DR, et al. Sequence motifs of tissue inhibitor of metalloproteinases 2 (TIMP-2) determining progelatinase A (proMMP-2) binding and activation by membrane- type metalloproteinase 1 (MT1-MMP). Biochem J. (2003) 372:799–809. doi: 10.1042/BJ20021573
24. Arpino V, Brock M, Gill SE. The role of TIMPs in regulation of extracellular matrix proteolysis. Matrix Biol. (2015) 44-46:247–54. doi: 10.1016/j.matbio.2015.03.005
25. Lee PH, Shatkay H. F-SNP: computationally predicted functional SNPs for disease association studies. Nucleic Acids Res. (2008) 36:D820–4. doi: 10.1093/nar/gkm904
26. Kukkonen MK, Tiili E, Vehmas T, Oksa P, Piirilä P, Hirvonen A. Association of genes of protease-antiprotease balance pathway to lung function and emphysema subtypes. BMC Pulm Med. (2013) 13:36. doi: 10.1186/1471-2466-13-36
27. Wang K, Wang GY, Huang SK, Luo AQ, Jing X, Li G, et al. Association between TIMP-2 gene polymorphism and breast cancer in Han Chinese women. BMC Cancer. (2019) 19:446. doi: 10.1186/s12885-019-5655-8
28. Dixon ME, Chien EK, Osol G, Callas PW, Bonney EA. Failure of decidual arteriolar remodeling in the CBA/J x DBA/2 murine model of recurrent pregnancy loss is linked to increased expression of tissue inhibitor of metalloproteinase 2 (TIMP-2). Am J Obstet Gynecol. (2006) 194:113–9. doi: 10.1016/j.ajog.2005.06.063
Keywords: TIMP2, rs2277698, single nucleotides polymorphisms, clinical outcomes, in vitro fertilization
Citation: Lee C-I, Lee Y-J, Lee T-H, Lee C-Y, Tsao H-M, Cheng E-H, Huang C-C, Yang S-F and Lee M-S (2025) TIMP2 rs2277698 polymorphism associated with adverse IVF outcomes in Han Chinese women. Front. Endocrinol. 16:1542534. doi: 10.3389/fendo.2025.1542534
Received: 10 December 2024; Accepted: 19 February 2025;
Published: 13 March 2025.
Edited by:
Kuan-Hao Tsui, Kaohsiung Veterans General Hospital, TaiwanReviewed by:
Li-Te Lin, Kaohsiung Veterans General Hospital, TaiwanCopyright © 2025 Lee, Lee, Lee, Lee, Tsao, Cheng, Huang, Yang and Lee. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Maw-Sheng Lee, bXNsZWVwaGRAY3NtdS5lZHUudHc=
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.