
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Endocrinol. , 25 March 2025
Sec. Cardiovascular Endocrinology
Volume 16 - 2025 | https://doi.org/10.3389/fendo.2025.1526991
 Jie Men1*†
Jie Men1*† Chengrui Zhao1†
Chengrui Zhao1† Chenmin Xiang1†
Chenmin Xiang1† Guoyu Zhu1†
Guoyu Zhu1† Zhengyang Yu1
Zhengyang Yu1 Pengbo Wang1
Pengbo Wang1 Simin Wu1
Simin Wu1 Yuxi Zhang1
Yuxi Zhang1 Yishan Li1
Yishan Li1 Liuliu Wang1
Liuliu Wang1 Xueyan Gong2
Xueyan Gong2 Xiang Yang1
Xiang Yang1 Shuangling Zou1
Shuangling Zou1 Jia Ma1
Jia Ma1 Chenglong Cui1
Chenglong Cui1 Hao Li1
Hao Li1 Xuedi Ma1
Xuedi Ma1 Wenjie Wu1
Wenjie Wu1 Yaoming Wang1
Yaoming Wang1Background: Despite the growing attention towards the efficacy of high-intensity interval training (HIIT) on older adult health, a consensus regarding the pleiotropic effects of HIIT in this population is yet to be reached. Previous studies have predominantly focused on specific outcomes or particular groups, lacking comprehensive analysis.
Objective: We aimed to conduct a systematic evaluation of the impact of HIIT on body composition, cardiopulmonary function, and metabolic parameters in older adults.
Methods: The databases searched included PubMed, Web of Science, Cochrane Library, Scopus, WanFang, and other relevant sources from the inception of the database until July 2023. Randomized controlled trials (RCTs) on the effects of HIIT on body shape, cardiopulmonary function, and metabolic parameters in the older adult were searched.
Results: A total of 87 RCTs meeting the criteria were included, involving 4,213 older adult people. Meta-analysis results showed that HIIT significantly improved body fat percentage (BF%) [MD: −1.63%, p = 0.005], maximal oxygen uptake (VO2max) [MD: 2.46 mL min−1 kg−1, p < 0.00001], maximal heart rate (HRmax) [MD: 2.83 beats min−1, p = 0.02], and high-density lipoprotein (HDL) levels [MD: 0.04 mmol L−1, p = 0.002]. However, for systolic blood pressure (SBP) [MD: 0.49 mmHg, p = 0.60], resting heart rate (HRrest) [MD: −0.95 BPM −1, p = 0.24], triglycerides (TG) [tendency for MD: −0.02 mmol L−1, p = 0.61], low-density lipoprotein (LDL) [MD: −0.04 mmol L−1, p = 0.27] had no significant effect. Sensitivity analysis found that HIIT significantly improved waist circumference (WC) [MD: −1.89 cm, p = 0.17], diastolic blood pressure (DBP) [MD: −0.63 mmHg, p = 0.23], respiratory exchange rate (RER) [MD: 0.01, p = 0.20], total cholesterol (TC) [MD: 0.10 mmol L−1, p = 0.14], and fasting plasma glucose (FPG) [MD:−0.20 mmol L−1, p = 0.08], but the results lacked robustness. There was no significant improvement in DBP [MD: −0.63 mmHg, p = 0.23] and body mass index (BMI) [MD: −0.36 kg m−2, p = 0.06].
Conclusions: HIIT has shown certain potential and advantages in improving the physical health of the older adult, especially in cardiopulmonary function. However, more high-quality studies are needed to confirm the effects of HIIT on the physical health of the older adult in the future. It also provides a reference for the clinical practice and family health management of HIIT in the older adult and the development of HIIT guidelines.
Systematic review registration: https://www.crd.york.ac.uk/PROSPERO/myprospero, identifier CRD42023460252.
The latest research predicts that the global older adult population aged 60 and above is projected to reach 2 billion by 2050 (1). With advancing age, there is a decline in physiological system functionality and an increased susceptibility to stress response (2). Older adults experience rapid deterioration in functional capacity in response to acute illness or major events (3–5). At the same time, inadequate physical activity can accelerate the aging process and contribute to adverse outcomes, such as an increased susceptibility to falls, hospitalization, premature mortality, and all-cause mortality (6); compared with safe physical activity, unsafe physical activity will also increase the same risk. Therefore, the older adult should observe strict risk assessment and standardized guidance when engaging in physical activity (7, 8). It is important to note that while the decline in systemic function among older individuals can be prevented and delayed, it may become irreversible once it manifests as an adverse event. Therefore, in addition to mitigating risk factors for adverse events, greater emphasis should be placed on preventing/delaying the deterioration of systemic functions to achieve healthy aging (9). Copenhagen Consensus Statement 2019 highlights that physical activity is a crucial determinant for maintaining health and the normal functioning of physiological systems (6), and it represents one of the primary strategies to delay age-related decline in physical function among older adults (10). Despite its significant role in reducing mortality risk (11), cardiovascular disease (CVD), and certain cancers (12), a majority of older adult individuals refrain from engaging in exercise or leisure physical activities (13), owing to concerns regarding potential risks associated with exercise. A cross-sectional population study noted that the annual incidence of exercise-related cardiac arrest in the older adult is extremely rare (14), which means that the benefits of exercise far outweigh the possible risks.
An increasing number of studies have demonstrated that exercise can enhance age-related body composition (15); ameliorate dyslipidemia; improve cardiopulmonary function (16); prevent the onset of type 2 diabetes, hypertension, CVD, and cancer (17); and reduce overall mortality rates (18). The health benefits of exercise have garnered consensus among experts (12, 19). Currently, three forms of physical activity exhibit significant potential in promoting the health of older adults. The first category encompasses non-exercise physical activities (low intensity), such as walking and household chores. Recent evidence unequivocally indicates that low-intensity physical activities also confer health advantages for older adults, including reduced all-cause mortality and protection against Alzheimer’s disease (20, 21). The second form is moderate-intensity aerobic exercise, which is supported by substantial evidence establishing a dose–response relationship with health outcomes and recommended by global guidelines (12). The pleasure brought by exercise is positively correlated with compliance, but has an inverted U-shaped curve with exercise time (22), which is the main reason that the expected compliance rate of aerobic exercise is 27% (23), but the actual compliance rate is less than 5% (24). It is also the main reason for exercise paradox (beneficial but difficult to promote) (25). High-intensity interval training (HIIT) presents itself as a promising alternative for promoting health among older adults. Intermittent training incorporates short bursts of high-intensity anaerobic exercise followed by low-intensity aerobic recovery, yielding comparable exercise effects to moderate-intensity aerobic exercise. Moreover, HIIT offers advantages in terms of time efficiency and cost-effectiveness (26). It holds great potential for widespread utilization in older adult health promotion endeavors. Importantly, unlike other pharmacological treatments that solely target a single outcome, exercise typically exerts positive effects on multiple physiological systems (27). Recent studies have demonstrated that engaging in 3 to 4 min of HIIT per day can significantly reduce the risk of all-cause and cancer mortality by 38% to 40%, as well as decrease the risk of CVD mortality by 48% to 49% (28). Furthermore, numerous meta-analyses have provided support for the role of HIIT in enhancing the health status of older individuals (26, 29–37). However, previous investigations on the health benefits of HIIT in older adults possess certain limitations. Firstly, most outcome measures were assessed within a single (26, 30, 31, 34, 36) or dual (29, 32, 33) physiological system. Secondly, only specific diseases (26, 29, 31) or particular risk factors (32, 33, 35, 36) were considered. Thirdly, the majority of studies had small sample sizes and included non-older adult participants within their populations (26). Additionally, a meta-analysis encompassing 10 studies incorporated 6 studies from a single research group (36). These limitations inevitably weaken the advantages of HIIT, limit the comprehensive interpretation of the health benefits of HIIT in older adults, and lead to limitations in generalizability. Furthermore, a significant number of included studies were deemed to be of low quality (29, 30, 32). While most research findings support the positive health effects of HIIT on older adult individuals, particularly about certain physiological systems, it is important to note that these systematic reviews or meta-analyses primarily focus on specific improvements within individual physiological systems or diseases. Considering that the human body functions as an integrated whole, our emphasis lies in examining the overall health impacts of HIIT and recognizing the essentiality of valid scientific evidence when formulating strategies to address global health issues among older populations.
Given the above considerations, a systematic review was conducted to evaluate the effects of HIIT on 14 outcomes of body shape, cardiopulmonary function, and metabolism in older adults. We also conducted subgroup analysis on the factors that may affect the conclusions, such as study type, intervention period, and disease type, in order to obtain more comprehensive data and provide evidence support for clinical or family health management and the development of HIIT guidelines/recommendations.
The present review adhered to the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA) (38). It was prospectively registered in the International Registry of Prospective Systematic Reviews (PROSPERO) database under registration number “CRD42023460252” on 5 September 2023. The following databases were electronically searched: PubMed, Web of Science, Cochrane Library, Scopus, and WanFang database. A comprehensive search was conducted for randomized controlled trials (RCTs) investigating the HIIT on body composition, cardiopulmonary function, and metabolic indicators in older adults. The search period ranged from the inception of each database to 1 July 2023. The search strategy is structured to include terms related to “aged,” “older adult,” “older,” “High-Intensity Interval Training,” “HIIT,” and “High-Intensity Intermittent Exercise.” Additionally, relevant literature and previously published systematic reviews were manually screened to identify any studies missed during the initial search process (search details in Additional File 1 in Supplementary Material).
The retrieved literature was imported into NoteExpress, a literature management software. Two researchers independently conducted a comprehensive literature review, eliminating duplicate and irrelevant studies, extracting relevant data, and cross-verifying the selected literature. In case of any discrepancies, they consulted with each other or sought input from a third party for discussion. A screening flowchart is presented in Figure 1. The research object of this paper is the older adult, and subjects were not restricted by country, race, or sex, or physical condition, except athletes. The experimental group was HIIT, and the control group was blank control or low-intensity exercise control. Articles in Chinese or English reporting at least one outcome measure were also included. The outcome measures included body mass index (BMI), body fat percentage (BF%), waist circumference (WC), maximal oxygen uptake (VO2max), systolic blood pressure (SBP), diastolic blood pressure (DBP), resting heart rate (HRrest), maximal heart rate (HRmax), respiratory exchange rate (RER), total cholesterol (TC), triglycerides (TG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), and fasting plasma glucose (FPG).
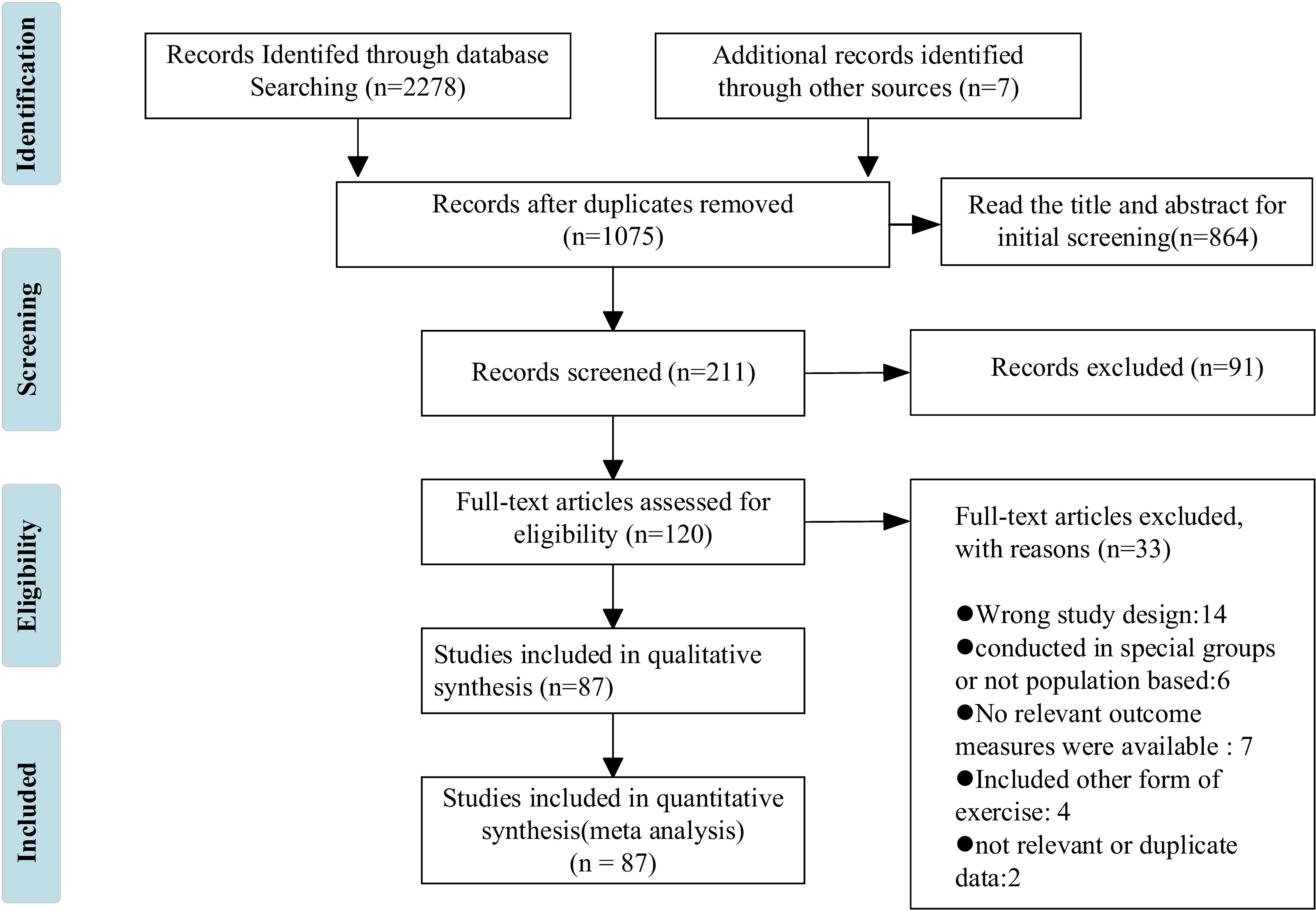
Figure 1. Study selection. The number of documents retrieved from the database is as follows: PubMed (n = 40), Embase (n = 694), Cochrane Library (n = 801), CNKI (n = 3), Scopus (n = 738), and WanFang Data (n = 2).
The data extraction encompassed the following components: basic information of the included literature (first author’s name, title, and publication year), characteristics of study subjects (age, gender, and number of subjects), training variables (intensity, form, frequency, and intervention period), main results, and key elements of risk of bias assessment. To calculate effect sizes for physical fitness measures in the intervention and control groups, baseline and follow-up mean as well as standard deviations (SDs) were extracted. In case any required data were missing, we contacted the corresponding author to obtain them. If relevant data could not be provided by the author, they were excluded from the analysis. The characteristics of the included studies are presented in Supplementary Table 1 (in Additional File 2).
RCTs were analyzed using the Cochrane Risk of Bias Tool 2.0 (39). There are three levels: low risk, high risk, and uncertain. Two researchers used ReviewManager 5.4.1 software to rigorously evaluate seven aspects of randomized allocation methods, allocation concealment of randomized methods, blinding of research subjects and interveners, blinding of outcome evaluators, integrity of outcome data, possibility of selective reporting, and other sources of bias. The risk of biased judgment in each domain was interpreted as low risk, moderate risk, severe risk, borderline risk, or no information (40). Two reviewers independently assessed the risk of bias, and any disagreements were resolved through a third party. In addition, the risk of publication bias was assessed using funnel plots when the meta-analysis included ≥10 studies.
Review Manager 5.3 (RevMan) software was used for statistical analysis. Because the literature had the same continuous outcome variable and the same unit of measurement, mean differences (MDs) and SDs along with 95% confidence intervals (95% CIs) were used to estimate the effect sizes observed in the studies. The random-effects model (overall effect p-value < 0.05) and fixed-effects model (overall effect p-value > 0.05 under the random-effects model) were the two main models used to synthesize all the results of forest plots. I2 was used to evaluate the degree of heterogeneity: above 25%, 50%, and 75% were low, medium, and high heterogeneity, respectively (41), and the level of meta-analysis was set as α = 0.05. When I2 ≤ 25%, the fixed-effects model was used for combined analysis. If I2 > 25%, the random-effects model was used for combined analysis. In order to increase the stability of the results, sensitivity analysis was performed by one-by-one elimination. These tests focused on significant heterogeneity associated with HIIT, in which individual studies were systematically removed from the meta-analysis and pooled effect estimates were recalculated to assess the impact of individual studies on the meta-analysis. As the objective of this study was to investigate the effects of HIIT on body shape, cardiopulmonary function, and metabolic parameters in the older adult, subgroup analyses were conducted according to intervention duration (≤12 weeks and >12 weeks) and disease type (CVD, diabetes, hypertension, cancer, and others) when I2 ≥ 50%. The possible publication bias was evaluated by drawing a funnel plot. Further analysis was performed when more than five studies were included in the subgroup.
Figure 1 describes the PRISMA process in detail. A total of 2,278 studies were retrieved from the database, and 7 studies were obtained from other sources. After the strict screening, 87 (42–128) studies were finally included, with a total of 4,213 older adult people. There were 2,099 subjects in the HIIT group, aged 66.8 ± 18.7 years, and 2,114 subjects in the control group, aged 63.1 ± 20 years. The exercise frequency ranged from 1 to 21 times per week, the duration ranged from 7 to 180 min per time, and the cycle ranged from 2 to 48 weeks. The measurement methods of outcome indicators were also reported. Among the included studies, 25 studies described medical supervision in detail, 29 did not describe it in detail, and 33 did not describe it. In addition, 24 studies reported a total of 196 people withdrawing due to family, subjective will, and other factors, accounting for 4.65% of the total number. There were 46 cases of adverse events, and the incidence of adverse events was 1.09%. Of note, adverse events were not attributed to HIIT. Figure 2 shows the geographical and sample distribution of the included studies, with 65.16% of the studies in Europe, 14.62% in North America and South America,14.24% in Asia, 5.03% in Australia, and 0.95% in Africa, involving 21 countries and regions. Of these, 658 were from Norway, 543 were from Switzerland, 537 were from Belgium, 191 were from Denmark, 182 were from Spain, 200 were from the United Kingdom, 148 were from Sweden, 144 were from Germany, 116 were from Italy, 26 were from France, 244 were from Canada, 222 were from the United States, 150 were from Brazil, 254 were from China, 133 were from Iran, 88 were from Japan, 72 were from South Korea, 29 were from Thailand, 24 were from Indonesia, 212 were from Australia, and 40 were from Egypt, for a total of 4,213 individuals.
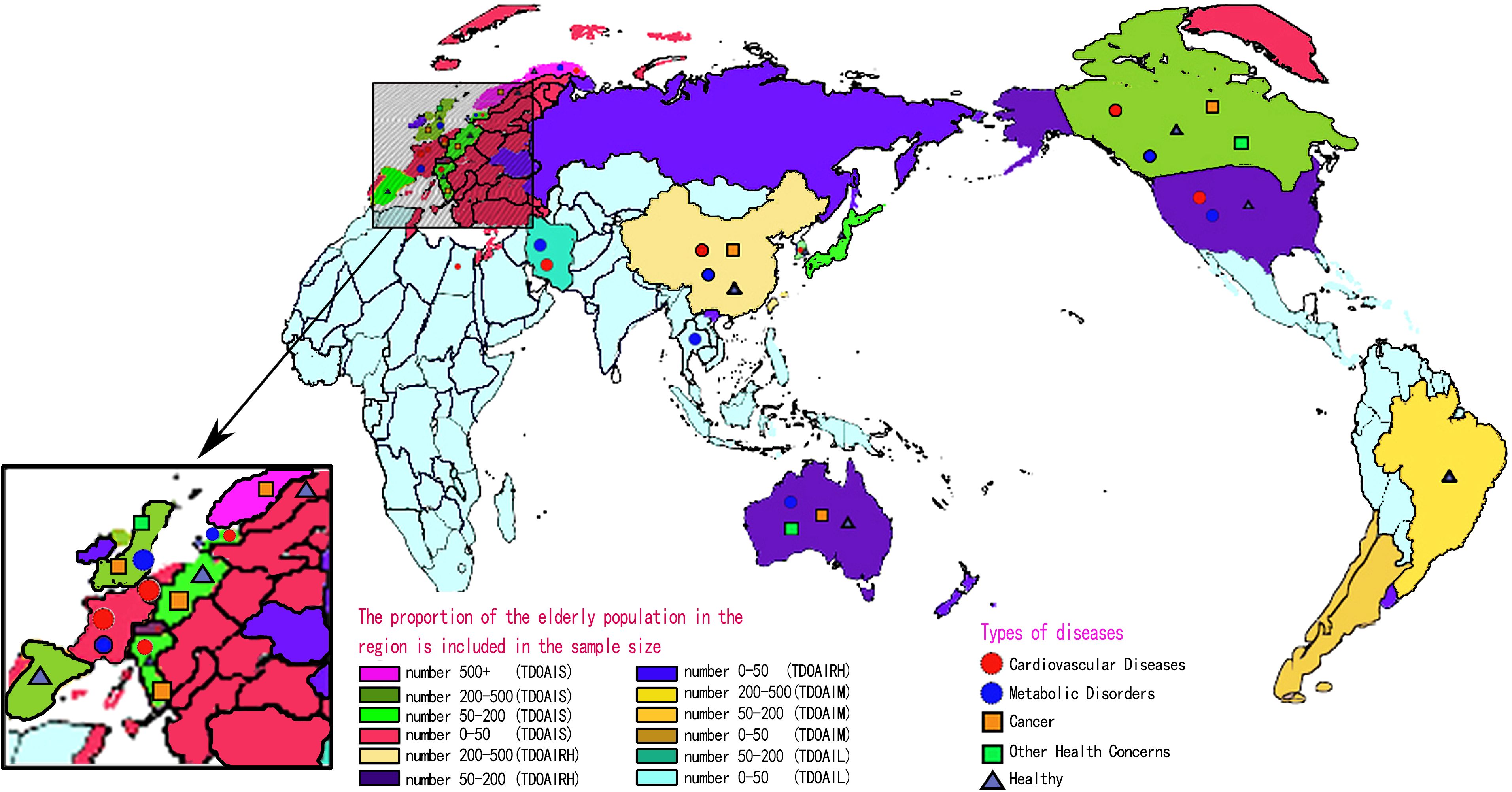
Figure 2. Contributing data source across world regions. TDOAIS, the degree of aging is serious; TDOAIRH, the degree of aging is relatively heavy; TDOAIM, the degree of aging is moderate; TDOAIL, the degree of aging is low. When the disease mark appears in a certain area, it means that the subjects included in the area have the disease or are in a healthy state. The proportion of the older adult population: TDOAIS > 17.91, 17.91 > TDOAIRH > 13.15, 13.15 > TDOAIM > 9.02, and TDOAIL < 9.02.
A total of 87 RCTs were included, and the overall risk of bias was low. Of these RCTs, 84/87 evaluated using randomized sequence generation, 84/87 assessments used allocation concealment, 77/87 subjects and staff were blinded, 77/87 were blinded to the outcome data evaluation, 84/87 had complete data, 86/87 reported no selectivity, and 39/87 had no other bias among the included (the results are shown in Additional File 3 in Supplementary Material).
The studies on the physical effects of the older adult can be divided into three aspects: body shape, cardiopulmonary function, and metabolic function. A total of 14 indicators were involved, and the detailed results of all indicators are recorded in Table 1.
BMI: A total of 27 (42, 43, 47, 50, 52, 53, 60, 67, 70–72, 75, 77–80, 84, 85, 88, 91, 100, 108, 109, 116, 123, 126, 127) studies were included. Compared with the control group, HIIT did not significantly improve BMI (MD: −0.36 kg m−2, p = 0.06) in the older adult, with statistical heterogeneity (I2 = 70%) (Figure 3). Sensitivity analysis showed that the results of this study were robust. Funnel plots were basically symmetric, and there was no evidence of publication bias.
BF%: A total of 16 (47, 50, 60, 73, 81, 87, 92, 103–105, 108, 111, 116, 118, 122, 123) studies were included. Compared with the control group, HIIT significantly improved BF% (MD: −1.63%, p = 0.05) without significant heterogeneity (I2 = 31%) (Figure 4). Sensitivity analysis showed that the results of this study were robust. Funnel plots were basically symmetric, and there was no evidence of publication bias.
WC: A total of 16 (49, 50, 54, 58, 61, 73, 91, 103–105, 108, 111, 115–118) studies were included. Compared with the control group, HIIT had no significant effect on WC in the older adult (MD: −1.89 cm, p = 0.17), but there was statistical heterogeneity (I2 = 80%) (Figure 5). After excluding the study by Tjønna et al. (117), I2 decreased to 48%, which was not statistically significant (MD: −0.53 cm, p = 0.58). After excluding the study by Madssen et al. (91), there was no significant change in I2, but HIIT significantly improved WC in the older adult (MD: −2.69 cm, p = 0.04). Sensitivity analysis suggested that the results were not robust. Funnel plots were basically symmetric, and there was no evidence of publication.
VO2max: A total of 56 (43, 44, 46, 49–51, 53, 54, 56–58, 60–63, 67, 68, 73–76, 78, 79, 83–85, 87, 91, 93–96, 98–104, 106–111, 113, 116, 117, 119–124, 126, 127) studies were included. Compared with the control group, HIIT had a significant effect on VO2max in the older adult (MD: 2.46 mL min−1 kg−1, p < 0.00001), but there was statistical heterogeneity (I2 = 79%) (Figure 6). Sensitivity analysis results suggested that the results of this study were robust. Funnel plots were basically symmetric, and there was no evidence of publication bias. Subgroup analysis showed that HIIT had a significant improvement in both the healthy and the sick older adult (p < 0.00001), but the improvement was greater in the sick older adult. There was no significant difference between subgroups (p > 0.05).
SBP: A total of 38 (44, 45, 49, 51, 52, 54–58, 64, 65, 68, 69, 73–75, 78, 82, 84, 85, 91, 92, 97, 101, 103–105, 107, 110–112, 117–119, 124, 126, 128) studies were included. Compared with the control group, HIIT had no significant effect on SBP in the older adult (MD: −0.49 mmHg, p = 0.60) (Figure 7). Sensitivity analysis suggested that the results were robust. Funnel plots were basically symmetric, and there was no evidence of publication bias.
DBP: A total of 37 (44, 45, 49, 51, 52, 54–58, 64, 65, 68, 69, 73, 75, 78, 82, 84, 85, 91, 92, 97, 101, 103–105, 107, 110–112, 117–119, 124, 126, 128) studies were included. Compared with the control group, HIIT had no significant effect on DBP in the older adult (MD: −0.63 mmHg, p = 0.23) (Figure 8). Sensitivity analysis suggested that the results were robust. Funnel plots were basically symmetric, and there was no evidence of publication bias.
HRrest: A total of 29 (43, 45, 54–57, 61, 65, 68, 73, 78, 79, 82–85, 91–93, 95, 97, 101, 103, 104, 107, 111, 118, 121, 126) studies were included. Compared with the control group, HIIT had no significant effect on HRrest in the older adult (MD: −0.95 BPM−1, p = 0.24) (Figure 9). Sensitivity analyses suggested that the results were robust. Funnel plots were basically symmetric, and there was no evidence of publication bias.
HRmax: A total of 39 (43, 44, 46, 51, 54, 56, 57, 63, 64, 68, 69, 73, 74, 76, 78, 79, 82–85, 91, 93–95, 97, 100–103, 107, 111, 117, 119–121, 123–125, 127) studies were included. Compared with the control group, HIIT significantly improved HRmax in the older adult (MD: 2.84 BPM−1, p = 0.02), but there was statistical heterogeneity (I2 = 79%) (Figure 10) (see Table 1). Sensitivity analyses suggested that the results were robust. Funnel plots were basically symmetric, and there was no evidence of publication bias. Differences between subgroups suggest that disease type may be a source of heterogeneity (p < 0.0001).
RER: A total of 26 (44, 46, 51, 54, 56, 57, 63, 74–76, 79, 82, 83, 91, 93–95, 99, 101, 102, 107, 114, 119, 120, 124, 127) studies were included. Compared with the control group, HIIT had no significant effect on RER in the older adult (MD: 0.01, p = 0.20) (Figure 11). After excluding studies by Villelabeitia-Jaureguizar et al. (124). I2 was reduced to 33% and RER was significantly improved (MD: 0.01, p = 0.04). Sensitivity analysis results suggested that the results of this study lacked robustness. Funnel plots were basically symmetric, and there was no evidence of publication bias.
TC: A total of 20 (42, 45, 49, 52, 54, 68, 73, 75, 76, 84, 86, 91, 92, 101, 104, 108, 112, 116, 119, 126) studies were included. Compared with the control group, HIIT had no significant effect on TC in the older adult (MD: 0.10 mmol L−1, p = 0.14), but there was statistical heterogeneity (I2 = 34%) (Figure 12). After excluding the study of Gjellesvik et al. (119), the heterogeneity decreased to 22% and significantly improved TC (MD: 0.13 mmol L−1, p = 0.006). Sensitivity analysis showed that the results of this study were not robust. Funnel plots were basically symmetric, and there was no evidence of publication bias.
TG: A total of 26 (42, 45, 49, 52, 54, 58, 68, 73, 75, 76, 83, 84, 91–95, 101, 104, 108, 112, 116, 117, 119, 122, 126) studies were included. Compared with the control group, HIIT had no significant effect on TG (MD: −0.02 mmol L−1, p = 0.61) (Figure 13). Sensitivity analyses suggested that the results were robust. Funnel plots were basically symmetric, and there was no evidence of publication bias.
HDL: A total of 25 (42, 45, 49, 54, 58, 68, 73, 75, 76, 83, 84, 86, 91–95, 101, 104, 108, 112, 116, 119, 122, 126) studies were included. Compared with the control group, HIIT significantly improved HDL (MD: 0.04 mmol L−1, p = 0.002) in the older adult, with low statistical heterogeneity (I2 = 21%) (Figure 14). Sensitivity analysis showed that the results of this study were robust. Funnel plots were basically symmetric, and there was no evidence of publication bias. There was no significant difference between subgroups (p > 0.05).
LDL: A total of 22 (42, 45, 49, 54, 58, 68, 73, 75, 76, 83, 84, 91–93, 101, 104, 108, 112, 116, 119, 122, 126) studies were included. HIIT had no significant effect on LDL compared with the control group (MD: −0.04 mmol L−1, p = 0.27) (Figure 15). Sensitivity analyses suggested that the results were robust. Funnel plots were basically symmetric, and there was no evidence of publication bias.
FPG: A total of 23 (47, 49, 52, 54, 58, 61, 68, 73, 75, 76, 86, 88, 91–95, 101, 108, 116, 117, 122, 126) studies were included. Compared with the control group, HIIT had no significant effect on FPG in the older adult (MD: −0.20 mmol L−1, p = 0.08) (Figure 16). After excluding the study by Gjellesvik et al. (119), the heterogeneity decreased to 22% and significantly improved FPG (MD: −0.13 mmol L−1, p = 0.006). The results of sensitivity analysis suggested that the results of this study lacked robustness. Funnel plots were basically symmetric, and there was no evidence of publication bias.
This meta-analysis examined the overall impact of HIIT on older adult health by analyzing 14 clinical indicators across three categories: body composition, cardiopulmonary function, and metabolism. To our knowledge, this study is the largest in the world to evaluate HIIT’s effects on older adult health, encompassing 87 eligible RCT studies involving 4,213 participants from various populations including healthy individuals as well as those with hypertension, CVD, diabetes, cancer, and other conditions; it would be pertinent to note that most of this population resides in the Northern Hemisphere. Our findings support the universal efficacy of HIIT in improving BF%, VO2max, HRmax, and HDL levels among older adults; however, it does not affect BMI, SBP, DBP, HRrest, TG, and LDL levels. The results for WC, RER, TC, and FPG should be interpreted with caution due to their lack of robustness, which may be attributed to differences in disease type. These findings provide evidence for the use of HIIT in clinical or home health management as well as guidelines/recommendations for the older adult.
Owing to the intricate relationship between body shape and health as well as disease (129), it serves as a primary indicator for assessing the health status of older individuals. Two extensive cohort studies have demonstrated that both high BMI and BF% along with low BMI elevate the risk of mortality (130, 131). Therefore, it is crucial for older adult individuals to effectively manage their BMI and reasonably reduce their BF%. This meta-analysis aims to elucidate the impact of HIIT on three clinical parameters related to body shape in older adults. The results of the meta-analysis demonstrated that HIIT effectively improved BF% in older adults, which is consistent with the findings reported by Donghai et al. (132) and McLeod et al. (33), but contradicts the conclusions drawn from other studies (26, 29–32, 34–37). Unlike previous studies that primarily included individuals with normal weight as baseline data, the older adults we enrolled were mostly overweight or obese (BMI ≥25), although we did not restrict inclusion and exclusion criteria. HIIT stimulates lipolysis by increasing catecholamines and growth hormone (133). Meanwhile, HIIT improved BF%, including body composition and lipid profile, by increasing HDL in human and animal models (134–137), a mechanism consistent with our meta results for biochemical metrics. The meta-analysis results indicated insufficient evidence regarding the impact of HIIT on WC and BMI in older adults, aligning with most previous systematic reviews (26, 29–32, 34–37). To further assess the stability of these combined results, a sensitivity analysis was performed. The pooled results for BMI were stable, and for WC after excluding the study conducted by Tjønna et al. (117), I2 decreased from 80% to 48%, This change may be related to two aspects. First, the difference in the included population: Tjønna et al. (117) included the older adult with metabolic syndrome, while other studies included healthy people or people with one disease. Second, unlike BMI, WC directly reflects the characteristics of fat distribution. Given the relationship between the pathological mechanism of metabolic syndrome and visceral fat function, the effect size may be nonlinear with other populations (138). These factors may exacerbate heterogeneity. It is worth noting that, excluding the study by Madssen et al. (91), there was no significant change in I2; however, WC exhibited a reversal (p = 0.04). Further analysis revealed that the exercise supervision implementation rate in the study was only 1/3, and this low compliance resulted in an increase in WC after HIIT intervention. This finding may further elucidate the insufficient effect of HIIT on WC combined results. Therefore, caution should be exercised when interpreting whether HIIT can improve WC in the older adult due to the lack of robustness of these results. HIIT did not significantly improve BMI in the older adult. The results of the study by Donghai et al. (132) suggested that although HIIT decreased BF% in the older adult, the increase in lean body mass did not change their total body weight significantly, resulting in no significant difference in BMI before and after exercise. This lack of significant difference in measurement results due to adaptive changes in the body is not related to the study hypothesis.
The pleiotropic effects of maintaining a high level of cardiopulmonary function on the health of the older adult have been widely recognized (139), and it has been identified as a priority for promoting the health of the older adult by the World Health Organization (WHO) (12). A total of six cardiopulmonary function indicators were included in this meta-analysis to evaluate the effects of HIIT on cardiopulmonary function in the older adult. The existing evidence shows that HIIT can effectively improve VO2max and HRmax in the older adult. The analysis of 55 studies showed that the increase of VO2max by 2.46 mL/kg/min is very significant, and VO2max is recognized as the gold standard of cardiopulmonary function. A 44-year follow-up study showed that VO2max was inversely associated with the risk of cancer, cardiovascular events, and all-cause mortality (140), and each 1-MET increase reduced cardiovascular events by 15% and all-cause mortality by 13% (141). Subgroup analysis found that the effect of HIIT on VO2max in the older adult was generalized and was independent of the intervention period and the health of the subjects. In addition, HRmax is also one of the important indexes to evaluate the cardiopulmonary function of the older adult, and it is positively correlated with VO2max. The improvement of HRmax is consistent with VO2max, but the improvement of HRmax is less extensive than VO2max. Although the aim of this study was not to investigate the underlying physiological mechanisms of HIIT, based on the current evidence, we highlighted some mechanisms related to the obtained results, such as high physiological stress caused by HIIT, high recruitment pattern of type I and type II fibers, and intense muscle contraction during exercise, leading to imbalance of the ATP/ADP relationship, increased PGC1-α activation and, thus, increased PGC1-α activation, and strong body VO2max (134, 142, 143). The results of this study show that there is insufficient evidence for HIIT to improve blood pressure in the older adult, which is consistent with the results of previous studies (30–37, 132), but in sharp contrast to the study by Du et al. (29), the difference in conclusions may be related to the training method [isometric exercise has a better effect on blood pressure change than other exercise methods (144)] and the sensitivity of the subjects to HIIT. Therefore, the effects of aging on HIIT-induced fitness may be interesting to investigate in the future. Blood pressure is the most important modifiable risk factor for all-cause morbidity and mortality; however, our evidence found that HIIT had no significant effect on SBP and DBP, and neither changed significantly after individual removal, possibly because the majority of non-hypertensive people we included undermined this effect. After the study by Villelabeitia-Jaureguizar et al. (124) was excluded by RER, the results were reversed (p = 0.04). Since this study compared the improvement difference between moderate-intensity exercise and HIIT, the effect of HIIT was reduced. The results for RER need to be interpreted with caution, given the lack of robustness of the sensitivity analyses. Unfortunately, there is insufficient evidence on the effect of HIIT on HRrest in the older adult; thus, it was omitted from this meta-analysis.
Metabolic abnormalities can cause a variety of chronic diseases, including obesity, CVD, diabetes, and cancer, which bring huge public health problems and medical burdens (145). Metabolic disorders are often more serious in the older adult. Priority should be given to reducing the related risks in the older adult at this stage while preventing them. A total of five metabolic indicators were included in this meta-analysis, and the existing evidence showed that HIIT effectively improved HDL, which was in sharp contrast with previous studies (26, 29, 31) probably because previous studies were based on comparing the differences between HIIT and moderate-intensity exercise, while most of our work was based on the comparison between HIIT and blank control. Because of the strong association between dyslipidemia and CVD, HIIT is of great significance for the improvement of HDL levels. However, HIIT had no significant effect on TG and LDL, which is consistent with previous studies (33, 37). The results of TC and FPG were reversed after excluding the study of Gjellesvik et al. (119), which may be due to the differences in the baseline levels of our included studies, two types of exercise in some studies, and the use of drugs to treat metabolic abnormalities or affect metabolic indicators. These factors may reduce the improvement effect of HIIT on metabolic indicators. It also indicates the lack of robustness of the TC and FPG results, and this result needs to be interpreted with caution.
Among the included studies, 25 studies provided comprehensive descriptions of medical supervision, while 29 studies lacked detailed information in this regard, and 33 studies did not mention it at all. Additionally, withdrawal from the intervention was reported in 24 studies due to reasons such as familial obligations, personal preferences, and other factors, involving a total of 196 participants, accounting for approximately 4.65% of the overall sample size. Adverse events were documented in 46 cases with an incidence rate of approximately 1.09%. Importantly, none of these adverse events were attributed to HIIT.
The large sample size and high heterogeneity observed in our study were expected due to differences in methodology and study subjects, as the range of studies included all older adults except those with contraindications to exercise. Therefore, subgroup analyses and sensitivity points were conducted to assess the role and stability of the pooled results. Sensitivity analysis revealed that WC, RER, TC, and FPG lacked robustness. Despite implementing a rigorous search strategy, language bias was inevitable as only Chinese and English literature was retrieved within our constraints. Furthermore, variations in exercise equipment, interval time, and intervention duration among the HIIT studies included prevented us from conducting subgroup analysis; thus, specific exercise doses cannot be recommended at this stage. Lastly, our included studies did not assess medication use or baseline/daily physical activity, which are critical factors for evaluating the hypothesis that HIIT improves health in older adults. Additionally, we lacked data on other influencing factors such as gender and exercise capacity. Finally, it should be emphasized that most of the included samples were from the Northern Hemisphere, and the unbalanced sample distribution may limit the promotion of HIIT in the Southern Hemisphere.
Current evidence suggests that HIIT is associated with improvements in BF%, VO2max, HRmax, and HDL, but not in BMI, SBP, DBP, HRrest, TG, and LDL in the older adult. In addition, owing to the differences in subjects, baseline, exercise dose, and sample distribution, the results of WC, RER, TC, and FPG lack robustness, and the conclusions need to be further studied and discussed. These findings provide data support for a comprehensive interpretation of the role of HIIT in promoting physical health in the older adult. In view of the significant clinical benefits of HIIT in improving the VO2max of the older adult, HIIT can be used as an effective means to improve the cardiopulmonary function of the older adult under the premise of medical supervision. However, the optimal exercise dose of HIIT is still uncertain, which requires future multi-center, large-scale, and high-quality studies as well as long-term prospective investigations to verify these results, and also needs to consider the global distribution of samples.
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
JMe: Formal Analysis, Funding acquisition, Methodology, Project administration, Resources, Supervision, Writing – review & editing. CZ: Funding acquisition, Project administration, Resources, Writing – review & editing. CX: Conceptualization, Formal Analysis, Funding acquisition, Investigation, Methodology, Writing – original draft. GZ: Conceptualization, Data curation, Funding acquisition, Resources, Software, Visualization, Writing – original draft. ZY: Data curation, Methodology, Software, Visualization, Writing – review & editing. PW: Data curation, Methodology, Software, Validation, Writing – review & editing. SW: Data curation, Investigation, Methodology, Software, Writing – review & editing. YZ: Data curation, Methodology, Software, Visualization, Writing – review & editing. YL: Data curation, Formal Analysis, Methodology, Software, Writing – review & editing. LW: Data curation, Methodology, Software, Writing – review & editing. XG: Conceptualization, Data curation, Methodology, Software, Writing – review & editing. XY: Data curation, Methodology, Writing – review & editing. SZ: Data curation, Formal Analysis, Methodology, Software, Writing – review & editing. JMa: Data curation, Software, Validation, Writing – review & editing. CC: Investigation, Software, Validation, Writing – review & editing. HL: Formal Analysis, Validation, Visualization, Writing – review & editing. XM: Data curation, Software, Supervision, Validation, Writing – review & editing. WW: Data curation, Investigation, Validation, Writing – review & editing. YW: Conceptualization, Supervision, Validation, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This paper is supported by the Institute of Metabolic Cardiovascular Diseases Key Laboratory Cultivation Base of Shanxi Province (TSGJ001), the Teaching Reform and Innovation Project of Higher Education in Shanxi Province (J2021967), the Key Discipline of Fenyang College of Shanxi Medical University (Physiology), the Teaching Reform Project of Fenyang College of Shanxi Medical University (FJ202013), the Talent Introduction Start-up Fund of Fenyang College of Shanxi Medical University (2022A01), the Innovation and Entrepreneurship Training Program for College Students in Shanxi Province (No. 20231800), and the Fenyang College Students’ Innovation and Entrepreneurship Program of Shanxi Medical University (FDC202104).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fendo.2025.1526991/full#supplementary-material
1. Asier Mañasa BP, Del-Pozo-Cruzc B, Rodríguez-Gómeza I, Losa-Reyna José, Júdicee PB, Sardinha LB, et al. Breaking sedentary time predicts future frailty in inactive older adults: A cross-lagged panel model. J GERONTOL A-BIOL. (2021) 76:839–900. doi: 10.1093/gerona/glaa159
2. Dent E, Martin FC, Bergman H. Management of frailty: opportunities, challenges, and future directions. LANCET. (2019) 394:1376–86. doi: 10.1016/S0140-6736(19)31785-4
3. Kim DH, Rockwood K. Frailty in older adults. New Engl J Med. (2024) 391:538–48. doi: 10.1056/NEJMra2301292
4. Junius-Walker U, Onder G, Soleymani D, Wiese B, Albaina O, Bernabei R, et al. The essence of frailty: A systematic review and qualitative synthesis on frailty concepts and definitions. Eur J Intern Med. (2018) 56:3–10. doi: 10.1016/j.ejim.2018.04.023
5. Yang X, Lupón J, Vidán MT, Ferguson C, Gastelurrutia P, Newton PJ, et al. Impact of frailty on mortality and hospitalization in chronic heart failure: A systematic review and meta-analysis. J Am Heart Assoc. (2018) 7:e008251. doi: 10.1161/JAHA.117.008251
6. Bangsbo J, Blackwell J, Boraxbekk CJ, Caserotti P, Dela F, Evans AB, et al. Copenhagen Consensus statement 2019: physical activity and ageing. BRIT J SPORT Med. (2019) 53:856–8. doi: 10.1136/bjsports-2018-100451
7. Murray CJL, Collaborators G. Findings from the global burden of disease study 2021. LANCET. (2024) 10440:2259–62. doi: 10.1016/S0140-6736(24)00769-4
8. Sherrington C, Fairhall NJ, Wallbank GK. Exercise for preventing falls in older people living in the community. Cochrane DB Syst Rev. (2019) 1:12424. doi: 10.1002/14651858.CD012424.pub2
9. Beard JRD, Officer AM, de Carvalho IAM, Sadana RS, Pot AMP, Michel JM, et al. The World report on ageing and health: a policy framework for healthy ageing. Lancet (British edition). (2016) 387:2145–54. doi: 10.1016/S0140-6736(15)00516-4
10. Angulo J, El Assar M, Álvarez-Bustos A, Rodríguez-Mañas L. Physical activity and exercise: Strategies to manage frailty. Redox Biol. (2020) 35:101513. doi: 10.1016/j.redox.2020.101513
11. Kraus WE, Powell KE, Haskell WL, Janz KF, Campbell WW, Jakicic JM, et al. Physical activity, all-cause and cardiovascular mortality, and cardiovascular disease. Med Sci SPORT EXER. (2019) 51:1270–81. doi: 10.1249/MSS.0000000000001939
12. Bull FC, Al-Ansari SS, Biddle S, Borodulin K, Buman MP, Cardon G, et al. World Health Organization 2020 guidelines on physical activity and sedentary behaviour. BRIT J SPORT Med. (2020) 54:1451–62. doi: 10.1136/bjsports-2020-102955
13. Moore SC, Lee IM, Weiderpass E, Campbell PT, Sampson JN, Kitahara CM, et al. Association of leisure-time physical activity with risk of 26 types of cancer in 1.44 million adults. JAMA Intern Med. (2016) 176:816–25. doi: 10.1001/jamainternmed.2016.1548
14. Holmstrom L, Chugh HS, Uy-Evanado A, Sargsyan A, Sorenson C, Salmasi S, et al. Sudden cardiac arrest during sports activity in older adults. JACC: Clin Electrophysiol. (2023) 9:893–903. doi: 10.1016/j.jacep.2022.10.033
15. Liao CD, Tsauo JY, Wu YT, Cheng CP, Chen HC, Huang YC, et al. Effects of protein supplementation combined with resistance exercise on body composition and physical function in older adults: a systematic review and meta-analysis. Am J Clin Nutr. (2017) 106:1078–91. doi: 10.3945/ajcn.116.143594
16. Edwards JJ, Griffiths M, Deenmamode AHP, O Driscoll JM. High-intensity interval training and cardiometabolic health in the general population: A systematic review and meta-analysis of randomised controlled trials. Sports Med (Auckland). (2023) 53:1753–63. doi: 10.1007/s40279-023-01863-8
17. Piercy KL, Troiano RP, Ballard RM, Carlson SA, Fulton JE, Galuska DA, et al. The physical activity guidelines for americans. JAMA-J Am Med Assoc. (2018) 320:2020–8. doi: 10.1001/jama.2018.14854
18. Zhao H, Zhang X, Shi Z, Yin L, Zhang W, He K, et al. Association of level of leisure-time physical activity with risks of all-cause mortality and cardiovascular disease in an elderly Chinese population: a prospective cohort study. J geriatric cardiology: JGC. (2020) 17:628–37. doi: 10.11909/j.issn.1671-5411.2020.10.003
19. Bowden Davies KA, Pickles S, Sprung VS, Kemp GJ, Alam U, Moore DR, et al. Reduced physical activity in young and older adults: metabolic and musculoskeletal implications. Ther Adv Endocrinol. (2019) 10:1956867858. doi: 10.1177/2042018819888824
20. Inoue K, Tsugawa Y, Mayeda ER, Ritz B. Association of daily step patterns with mortality in US adults. JAMA Netw Open. (2023) 6:e235174. doi: 10.1001/jamanetworkopen.2023.5174
21. De la Rosa A, Olaso-Gonzalez G, Arc-Chagnaud C, Millan F, Salvador-Pascual A, García-Lucerga C, et al. Physical exercise in the prevention and treatment of Alzheimer’s disease. J SPORT Health Sci. (2020) 9:394–404. doi: 10.1016/j.jshs.2020.01.004
22. Tavares VDDO, Schuch FB, Tempest G, Parfitt G, Oliveira Neto L, Galvão-Coelho NL, et al. Exercisers’ Affective and enjoyment responses: A meta-analytic and meta-regression review. Percept MOTOR SKILL. (2021) 128:2211–36. doi: 10.1177/00315125211024212
23. Guthold R, Stevens GA, Riley LM, Bull FC. Worldwide trends in insufficient physical activity from 2001 to 2016: a pooled analysis of 358 population-based surveys with 1.9 million participants. Lancet GLOB Health. (2018) 6:e1077–86. doi: 10.1016/S2214-109X(18)30357-7
24. Troiano RP, Berrigan D, Dodd KW, Masse LC, Tilert T, McDowell M. Physical activity in the United States measured by accelerometer. Med Sci SPORT EXER. (2008) 40:181–8. doi: 10.1249/mss.0b013e31815a51b3
25. Ekkekakis P, Parfitt G, Petruzzello SJ. The Pleasure and Displeasure People Feel When they Exercise at Different Intensities. Sports Med (Auckland). (2011) 41:641–71. doi: 10.2165/11590680-000000000-00000
26. Leal JM, Galliano LM, Del Vecchio FB. Effectiveness of high-intensity interval training versus moderate-intensity continuous training in hypertensive patients: a systematic review and meta-analysis. Curr Hypertens Rep. (2020) 22:26. doi: 10.1007/s11906-020-1030-z
27. Izquierdo M, Merchant RA, Morley JE, Anker SD, Aprahamian I, Arai H, et al. International exercise recommendations in older adults (ICFSR): expert consensus guidelines. J Nutr Health AGING. (2021) 25:824–53. doi: 10.1007/s12603-021-1665-8
28. Stamatakis E, Ahmadi MN, Gill JMR, Thøgersen-Ntoumani C, Gibala MJ, Doherty A, et al. Association of wearable device-measured vigorous intermittent lifestyle physical activity with mortality. Nat Med. (2022) 28:2521–9. doi: 10.1038/s41591-022-02100-x
29. Du L, Zhang X, Chen K, Ren X, Chen S, He Q. Effect of high-intensity interval training on physical health in coronary artery disease patients: A meta-analysis of randomized controlled trials. J Cardiovasc Dev Dis. (2021) 8:158. doi: 10.3390/jcdd8110158
30. Carpes L, Costa R, Schaarschmidt B, Reichert T, Ferrari R. High-intensity interval training reduces blood pressure in older adults: A systematic review and meta-analysis. Exp GERONTOL. (2022) 158:111657. doi: 10.1016/j.exger.2021.111657
31. Costa EC, Hay JL, Kehler DS, Boreskie KF, Arora RC, Umpierre D, et al. Effects of high-intensity interval training versus moderate-intensity continuous training on blood pressure in adults with pre- to established hypertension: A systematic review and meta-analysis of randomized trials. SPORTS Med. (2018) 48:2127–42. doi: 10.1007/s40279-018-0944-y
32. De Nardi AT, Tolves T, Lenzi TL, Signori LU, Silva A. High-intensity interval training versus continuous training on physiological and metabolic variables in prediabetes and type 2 diabetes: A meta-analysis. Diabetes Res Clin PR. (2018) 137:149–59. doi: 10.1016/j.diabres.2017.12.017
33. McLeod KA, Jones MD, Thom JM, Parmenter BJ. Resistance training and high-intensity interval training improve cardiometabolic health in high risk older adults: A systematic review and meta-anaylsis. Int J SPORTS Med. (2022) 43:206. doi: 10.1055/a-1560-6183
34. Poon ET, Wongpipit W, Ho RS, Wong SH. Interval training versus moderate-intensity continuous training for cardiorespiratory fitness improvements in middle-aged and older adults: a systematic review and meta-analysis. J SPORT Sci. (2021) 39:1996–2005. doi: 10.1080/02640414.2021.1912453
35. Ramos JS, Dalleck LC, Tjonna AE, Beetham KS, Coombes JS. The impact of high-intensity interval training versus moderate-intensity continuous training on vascular function: a systematic review and meta-analysis. Sports Med (Auckland). (2015) 45:679–92. doi: 10.1007/s40279-015-0321-z
36. Weston KS, Wisloff U, Coombes JS. High-intensity interval training in patients with lifestyle-induced cardiometabolic disease: a systematic review and meta-analysis. BRIT J SPORT Med. (2014) 48:1227–34. doi: 10.1136/bjsports-2013-092576
37. Wu ZJ, Wang ZY, Gao HE, Zhou XF, Li FH. Impact of high-intensity interval training on cardiorespiratory fitness, body composition, physical fitness, and metabolic parameters in older adults: A meta-analysis of randomized controlled trials. Exp GERONTOL. (2021) 150:111345. doi: 10.1016/j.exger.2021.111345
38. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ-BRIT Med J. (2021) 134:178–89. doi: 10.1136/bmj.n71
39. Sterne J, Savovic J, Page MJ, Elbers RG, Blencowe NS, Boutron I, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ-BRIT Med J. (2019) 366:l4898. doi: 10.1136/bmj.l4898
40. Sterne JA, Hernán MA, Reeves BC, Savović J, Berkman ND, Viswanathan M, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ (Online). (2016) 355:i4919. doi: 10.1136/bmj.i4919
41. Julian PT, Higgins SGTJ. Cochrane Reviews have recently started including the quantity I 2 to help readers assess the consistency of the results of studies in meta-analyses. What does this new quantity mean, and why is assessment of heterogeneity so important to clinical practice? BMJ-BRIT Med J. (2003) 327:557–60. doi: 10.1136/bmj.327.7414.557
42. Abdelhalem AM, Shabana AM, Onsy AM, Gaafar AE. High intensity interval training exercise as a novel protocol for cardiac rehabilitation program in ischemic Egyptian patients with mild left ventricular dysfunction. Egyptian Heart J. (2018) 70:287–94. doi: 10.1016/j.ehj.2018.07.008
43. Amundsen BH, Rognmo Ø, Hatlen-Rebhan G, Slørdahl SA. High-intensity aerobic exercise improves diastolic function in coronary artery disease. Scandinavian Cardiovasc journal: SCJ. (2008) 42:110–7. doi: 10.1080/14017430701744477
44. Angadi SS, Mookadam F, Lee CD, Tucker WJ, Haykowsky MJ, Gaesser GA. High-intensity interval training vs. moderate-intensity continuous exercise training in heart failure with preserved ejection fraction: a pilot study. J Appl Physiol. (1985) 2015:753–8. doi: 10.1152/japplphysiol.00518.2014
45. Ballin M, Lundberg E, Sorlen N, Nordstrom P, Hult A, Nordstrom A. Effects of interval training on quality of life and cardiometabolic risk markers in older adults: a randomized controlled trial. Clin INTERV AGING. (2019) 14:1589–99. doi: 10.2147/CIA.S213133
46. Banerjee S, Manley K, Shaw B, Lewis L, Cucato G, Mills R, et al. Vigorous intensity aerobic interval exercise in bladder cancer patients prior to radical cystectomy: a feasibility randomised controlled trial. SUPPORT Care CANCER. (2018) 26:1515–23. doi: 10.1007/s00520-017-3991-2
47. Banitalebi E, Mohammad F, Nasiri S. High-intensity interval training versus moderate intensity combined training (Resistance and aerobic) for improving insulin-related adipokines in type 2 diabeticWomen. Zahedan Journal of Research in Medical Sciences. (2018) 20:. doi: 10.5812/zjrms.68793
48. Bhatia C, Kayser B. Preoperative high-intensity interval training is effective and safe in deconditioned patients with lung cancer: A randomized clinical trial. J Rehabil Med. (2019) 51:712–8. doi: 10.2340/16501977-2592
49. Boukabous I, Marcotte-Chenard A, Amamou T, Boulay P, Brochu M, Tessier D, et al. Low-volume high-intensity interval training (HIIT) versus moderate-intensity continuous training on body composition, cardiometabolic profile and physical capacity in older women. J Aging Phys ACTIV. (2019) 27:879–89. doi: 10.1123/japa.2018-0309
50. Bruseghini P, Tam E, Calabria E, Milanese C, Capelli C, Galvani C. High intensity interval training does not have compensatory effects on physical activity levels in older adults. Int J Env Res PUB HE. (2020) 17:1083. doi: 10.3390/ijerph17031083
51. Cardozo GG, Oliveira RB, Farinatti PT. Effects of high intensity interval versus moderate continuous training on markers of ventilatory and cardiac efficiency in coronary heart disease patients. ScientificWorldJournal. (2015) 2015:192479. doi: 10.1155/tswj.v2015.1
52. Cassidy S, Thoma C, Hallsworth K, Parikh J, Hollingsworth KG, Taylor R, et al. High intensity intermittent exercise improves cardiac structure and function and reduces liver fat in patients with type 2 diabetes: a randomised controlled trial. DIABETOLOGIA. (2016) 59:56–66. doi: 10.1007/s00125-015-3741-2
53. Choi H, Han H, Choi J, Jung H, Joa K. Superior effects of high-intensity interval training compared to conventional therapy on cardiovascular and psychological aspects in myocardial infarction. Ann Rehabil Med. (2018) 42:145–53. doi: 10.5535/arm.2018.42.1.145
54. Conraads VM, Pattyn N, De Maeyer C, Beckers PJ, Coeckelberghs E, Cornelissen VA, et al. Aerobic interval training and continuous training equally improve aerobic exercise capacity in patients with coronary artery disease: the SAINTEX-CAD study. Int J Cardiol. (2015) 179:203–10. doi: 10.1016/j.ijcard.2014.10.155
55. Coswig VS, Barbalho M, Raiol R, Del VF, Ramirez-Campillo R, Gentil P. Effects of high vs moderate-intensity intermittent training on functionality, resting heart rate and blood pressure of elderly women. J Transl Med. (2020) 18:88. doi: 10.1186/s12967-020-02261-8
56. Currie KD, Rosen LM, Millar PJ, McKelvie RS, MacDonald MJ. Heart rate recovery and heart rate variability are unchanged in patients with coronary artery disease following 12 weeks of high-intensity interval and moderate-intensity endurance exercise training. Appl Physiol Nutr ME. (2013) 38:644–50. doi: 10.1139/apnm-2012-0354
57. Currie KD, Dubberley JB, Mckelvie RS, Macdonald MJ. Low-volume, high-intensity interval training in patients with CAD. Med Sci sports exercise. (2013) 45:1436–42. doi: 10.1249/MSS.0b013e31828bbbd4
58. Deiseroth A, Streese L, Kochli S, Wust RS, Infanger D, Schmidt-Trucksass A, et al. Exercise and arterial stiffness in the elderly: A combined cross-sectional and randomized controlled trial (EXAMIN AGE). Front Physiol. (2019) 10:1119. doi: 10.3389/fphys.2019.01119
59. Devin JL, Jenkins DG, Sax AT, Hughes GI, Aitken JF, Chambers SK, et al. Cardiorespiratory fitness and body composition responses to different intensities and frequencies of exercise training in colorectal cancer survivors. Clin COLORECTAL CANC. (2018) 17:e269–79. doi: 10.1016/j.clcc.2018.01.004
60. Devin JL, Sax AT, Hughes GI, Jenkins DG, Aitken JF, Chambers SK, et al. The influence of high-intensity compared with moderate-intensity exercise training on cardiorespiratory fitness and body composition in colorectal cancer survivors: a randomised controlled trial. J Cancer SURVIV. (2016) 10:467–79. doi: 10.1007/s11764-015-0490-7
61. Dolan LB, Campbell K, Gelmon K, Neil-Sztramko S, Holmes D, McKenzie DC. Interval versus continuous aerobic exercise training in breast cancer survivors–a pilot RCT. SUPPORT Care CANCER. (2016) 24:119–27. doi: 10.1007/s00520-015-2749-y
62. Dunne DF, Jack S, Jones RP, Jones L, Lythgoe DT, Malik HZ, et al. Randomized clinical trial of prehabilitation before planned liver resection. BRIT J Surg. (2016) 103:504–12. doi: 10.1002/bjs.10096
63. Edvardsen E, Skjonsberg OH, Holme I, Nordsletten L, Borchsenius F, Anderssen SA. High-intensity training following lung cancer surgery: a randomised controlled trial. THORAX. (2015) 70:244–50. doi: 10.1136/thoraxjnl-2014-205944
64. Eser P, Jaeger E, Marcin T, Herzig D, Trachsel LD, Wilhelm M. Acute and chronic effects of high-intensity interval and moderate-intensity continuous exercise on heart rate and its variability after recent myocardial infarction: A randomized controlled trial. Ann Phys Rehabil Med. (2022) 65:101444. doi: 10.1016/j.rehab.2020.09.008
65. Fernandes B, Barbieri FA, Arthuso FZ, Silva FA, Moretto GF, Imaizumi L, et al. High-intensity interval versus moderate-intensity continuous training in individuals with parkinson’s disease: hemodynamic and functional adaptation. J Phys ACT Health. (2020) 17:85–91. doi: 10.1123/jpah.2018-0588
66. Freyssin C, Verkindt C, Prieur F, Benaich P, Maunier S, Blanc P. Cardiac rehabilitation in chronic heart failure: effect of an 8-week, high-intensity interval training versus continuous training. Arch Phys Med REHAB. (2012) 93:1359–64. doi: 10.1016/j.apmr.2012.03.007
67. Gao Z, Ji P, Xia Y. Effects of different intensity aerobic exercise on cardiac function and exercise endurance in patients after percutaneous coronary intervention. Chin J Rehabil. (2015) 30:344–8. doi: 10.3969/j.issn.1001-1242.2015.04.007
68. Ghardashi AA, Izadi MR, Rakhshan K, Mafi F, Biglari S, Gandomkar BH. Improved brachial artery shear patterns and increased flow-mediated dilatation after low-volume high-intensity interval training in type 2 diabetes. Exp Physiol. (2018) 103:1264–76. doi: 10.1113/EP087005
69. Ghardashi-Afousi A, Holisaz MT, Shirvani H, Pishgoo B. The effects of low-volume high-intensity interval versus moderate intensity continuous training on heart rate variability, and hemodynamic and echocardiography indices in men after coronary artery bypass grafting: A randomized clinical trial study. ARYA ATHEROSCLER. (2018) 14:260–71. doi: 10.22122/arya.v14i6.1781
70. Guoliang F, Li Z, Tianyu H, Xu Z, Han Z, Xi L, et al. Effects of high intensity interval training on congnitive function in older adults. China SPORT Sci AND Technol. (2020) 56:32–7. doi: 10.16470/j.csst.2020130
71. Herrod P, Blackwell J, Boereboom CL, Atherton PJ, Williams JP, Lund JN, et al. The time course of physiological adaptations to high-intensity interval training in older adults. Aging Med (Milton). (2020) 3:245–51. doi: 10.1002/agm2.12127
72. Hurst C, Weston KL, Weston M. The effect of 12 weeks of combined upper- and lower-body high-intensity interval training on muscular and cardiorespiratory fitness in older adults. Aging Clin Exp Res. (2019) 31:661–71. doi: 10.1007/s40520-018-1015-9
73. Hwang CL, Lim J, Yoo JK, Kim HK, Hwang MH, Handberg EM, et al. Effect of all-extremity high-intensity interval training vs. moderate-intensity continuous training on aerobic fitness in middle-aged and older adults with type 2 diabetes: A randomized controlled trial. Exp GERONTOL. (2019) 116:46–53. doi: 10.1016/j.exger.2018.12.013
74. Hwang CL, Yu CJ, Shih JY, Yang PC, Wu YT. Effects of exercise training on exercise capacity in patients with non-small cell lung cancer receiving targeted therapy. SUPPORT Care CANCER. (2012) 20:3169–77. doi: 10.1007/s00520-012-1452-5
75. Iellamo F, Caminiti G, Sposato B, Vitale C, Massaro M, Rosano G, et al. Effect of High-Intensity interval training versus moderate continuous training on 24-h blood pressure profile and insulin resistance in patients with chronic heart failure. Intern Emerg Med. (2014) 9:547–52. doi: 10.1007/s11739-013-0980-4
76. Iellamo F, Manzi V, Caminiti G, Vitale C, Castagna C, Massaro M, et al. Matched dose interval and continuous exercise training induce similar cardiorespiratory and metabolic adaptations in patients with heart failure. Int J Cardiol. (2013) 167:2561–5. doi: 10.1016/j.ijcard.2012.06.057
77. Iellamo F, Caminiti G, Montano M, Manzi V, Franchini A, Mancuso A, et al. Prolonged post-exercise hypotension: effects of different exercise modalities and training statuses in elderly patients with hypertension. Int J Environ Res Public Health. (2021) 18:3229. doi: 10.3390/ijerph18063229
78. Izadi MR, Ghardashi Afousi A, Asvadi Fard M, Babaee Bigi MA. High-intensity interval training lowers blood pressure and improves apelin and NOx plasma levels in older treated hypertensive individuals. J Physiol Biochem. (2018) 74:47–55. doi: 10.1007/s13105-017-0602-0
79. Jaureguizar KV, Vicente-Campos D, Bautista LR, de la Peña CH, Gómez MJA, Rueda MJC, et al. Effect of high-intensity interval versus continuous exercise training on functional capacity and quality of life in patients with coronary artery disease: A RANDOMIZED CLINICAL TRIAL. J CARDIOPULM REHABIL. (2016) 36:96–105. doi: 10.1097/HCR.0000000000000156
80. Karenovics W, Licker M, Ellenberger C, Christodoulou M, Diaper J, Bhatia C, et al. Short-term preoperative exercise therapy does not improve long-term outcome after lung cancer surgery: a randomized controlled study. Eur J CARDIO-THORAC. (2017) 52:47–54. doi: 10.1093/ejcts/ezx030
81. Keogh JW, Grigg J, Vertullo CJ. Is high-intensity interval cycling feasible and more beneficial than continuous cycling for knee osteoarthritic patients? Results of a randomised control feasibility trial. PEERJ. (2018) 6:e4738. doi: 10.7717/peerj.4738
82. Keteyian SJ, Hibner BA, Bronsteen K, Kerrigan D, Aldred HA, Reasons LM, et al. Greater improvement in cardiorespiratory fitness using higher-intensity interval training in the standard cardiac rehabilitation setting. J CARDIOPULM REHABIL. (2014) 34:98–105. doi: 10.1097/HCR.0000000000000049
83. Kim C, Choi HE, Lim MH. Effect of high interval training in acute myocardial infarction patients with drug-eluting stent. Am J Phys Med REHAB. (2015) 94:879–86. doi: 10.1097/PHM.0000000000000290
84. Kim HK, Hwang CL, Yoo JK, Hwang MH, Handberg EM, Petersen JW, et al. All-extremity exercise training improves arterial stiffness in older adults. Med Sci SPORT EXER. (2017) 49:1404–11. doi: 10.1249/MSS.0000000000001229
85. Klonizakis M, Moss J, Gilbert S, Broom D, Foster J, Tew GA. Low-volume high-intensity interval training rapidly improves cardiopulmonary function in postmenopausal women. MENOPAUSE. (2014) 21:1099–105. doi: 10.1097/GME.0000000000000208
86. Koh HE, Ørtenblad N, Winding KM, Hellsten Y, Mortensen SP, Nielsen J. High-intensity interval, but not endurance, training induces muscle fiber type-specific subsarcolemmal lipid droplet size reduction in type 2 diabetic patients. Am J physiol: Endocrinol Metab. (2018) 315:E872–84. doi: 10.1152/ajpendo.00161.2018
87. Li X, Han T, Zou X, Zhang H, Feng W, Wang H, et al. Long-term high-intensity interval training increases serum neurotrophic factors in elderly overweight and obese Chinese adults. Eur J Appl Physiol. (2021) 121:2773–85. doi: 10.1007/s00421-021-04746-w
88. Li X. Effect of high-intensity interval training on blood glucose control, adipocytokine secretion and oxidative stress response in patients with T2DM. J Hainan Med University(English version). (2018) 24:14–8. doi: 10.13210/j.cnki.jhmu.20180223.002
89. Licker M, Karenovics W, Diaper J, Fresard I, Triponez F, Ellenberger C, et al. Short-term preoperative high-intensity interval training in patients awaiting lung cancer surgery: A randomized controlled trial. J Thorac Oncol. (2017) 12:323–33. doi: 10.1016/j.jtho.2016.09.125
90. Lohman T, Bains G, Cole S, Gharibvand L, Berk L, Lohman E. High-Intensity interval training reduces transcriptomic age: A randomized controlled trial. Aging Cell. (2023) 22:e13841. doi: 10.1111/acel.13841
91. Madssen E, Arbo I, Granoien I, Walderhaug L, Moholdt T. Peak oxygen uptake after cardiac rehabilitation: a randomized controlled trial of a 12-month maintenance program versus usual care. PloS One. (2014) 9:e107924. doi: 10.1371/journal.pone.0107924
92. Mitranun W, Deerochanawong C, Tanaka H, Suksom D. Continuous vs interval training on glycemic control and macro- and microvascular reactivity in type 2 diabetic patients. Scand J Med Sci SPOR. (2014) 24:e69–76. doi: 10.1111/sms.2014.24.issue-2
93. Moholdt TT, Amundsen BH, Rustad LA, Wahba A, Lovo KT, Gullikstad LR, et al. Aerobic interval training versus continuous moderate exercise after coronary artery bypass surgery: a randomized study of cardiovascular effects and quality of life. Am Heart J. (2009) 158:1031–7. doi: 10.1016/j.ahj.2009.10.003
94. Moholdt T, Aamot IL, Granoien I, Gjerde L, Myklebust G, Walderhaug L, et al. Long-term follow-up after cardiac rehabilitation: a randomized study of usual care exercise training versus aerobic interval training after myocardial infarction. Int J Cardiol. (2011) 152:388–90. doi: 10.1016/j.ijcard.2011.08.025
95. Molmen-Hansen HE, Stolen T, Tjonna AE, Aamot IL, Ekeberg IS, Tyldum GA, et al. Aerobic interval training reduces blood pressure and improves myocardial function in hypertensive patients. Eur J Prev Cardiol. (2012) 19:151–60. doi: 10.1177/1741826711400512
96. Mueller S, Winzer EB, Duvinage A, Gevaert AB, Edelmann F, Haller B, et al. Effect of high-intensity interval training, moderate continuous training, or guideline-based physical activity advice on peak oxygen consumption in patients with heart failure with preserved ejection fraction: A randomized clinical trial. JAMA: J Am Med Assoc. (2021) 325:542–51. doi: 10.1001/jama.2020.26812
97. Nemoto K, Gen-no H, Masuki S, Okazaki K, Nose H. Effects of high-intensity interval walking training on physical fitness and blood pressure in middle-aged and older people. MAYO Clin Proc. (2007) 82:803–11. doi: 10.4065/82.7.803
98. Northey JM, Pumpa KL, Quinlan C, Ikin A, Toohey K, Smee DJ, et al. Cognition in breast cancer survivors: A pilot study of interval and continuous exercise. J Sci Med SPORT. (2019) 22:580–5. doi: 10.1016/j.jsams.2018.11.026
99. Østerås H, Hoff J, Helgerud J. Effects of high-intensity endurance training on maximal oxygen consumption in healthy elderly people. J Appl GERONTOL. (2005) 24:377–87. doi: 10.1177/0733464804273185
100. Pattyn N, Beckers PJ, Cornelissen VA, Coeckelberghs E, De Maeyer C, Frederix G, et al. The effect of aerobic interval training and continuous training on exercise capacity and its determinants. Acta Cardiol. (2017) 72:328–40. doi: 10.1080/00015385.2017.1304712
101. Pattyn N, Vanhees L, Cornelissen VA, Coeckelberghs E, De Maeyer C, Goetschalckx K, et al. The long-term effects of a randomized trial comparing aerobic interval versus continuous training in coronary artery disease patients: 1-year data from the SAINTEX-CAD study. Eur J Prev Cardiol. (2016) 23:1154–64. doi: 10.1177/2047487316631200
102. Prado DML, Rocco EA, Silva AG, Rocco DF, Pacheco MT, Silva PF, et al. Effects of continuous vs interval exercise training on oxygen uptake efficiency slope in patients with coronary artery disease. Braz J Med Biol Res. (2016) 49:e4890. doi: 10.1590/1414-431X20154890
103. Ramos JS, Dalleck LC, Borrani F, Beetham KS, Mielke GI, Dias KA, et al. High-intensity interval training and cardiac autonomic control in individuals with metabolic syndrome: A randomised trial. Int J Cardiol. (2017) 245:245–52. doi: 10.1016/j.ijcard.2017.07.063
104. Ramosa JS, Lance CD, Maximiano VR, Fabio BELR, Gomersalla S, Kassia SB, et al. 12min/week of high-intensity interval training reduces aortic reservoir pressure in individuals withmetabolic syndrome: a randomized trial. J Hypertens. (2016) 34:1977–87. doi: 10.1097/HJH.00000000000001034
105. Reed JL, Terada T, Cotie LM, Tulloch HE, Leenen FH, Mistura M, et al. The effects of high-intensity interval training, Nordic walking and moderate-to-vigorous intensity continuous training on functional capacity, depression and quality of life in patients with coronary artery disease enrolled in cardiac rehabilitation: A randomized controlled trial (CRX study). Prog Cardiovasc Dis. (2022) 70:73–83. doi: 10.1016/j.pcad.2021.07.002
106. Rocco EA, Prado DML, Silva AG, Lazzari JMA, Bortz PC, Rocco DFM, et al. Effect of continuous and interval exercise training on the PETCO2 response during a graded exercise test in patients with coronary artery disease. Clinics (São Paulo Brazil). (2012) 67:623–7. doi: 10.6061/clinics/2012(06)13
107. Rognmo O, Hetland E, Helgerud J, Hoff J, Slordahl SA. High intensity aerobic interval exercise is superior to moderate intensity exercise for increasing aerobic capacity in patients with coronary artery disease. Eur J Cardiovasc Prev Rehabil. (2004) 11:216–22. doi: 10.1097/01.hjr.0000131677.96762.0c
108. Rohmansyah NA, Ka PR, Phanpheng Y, Hiruntrakul A. High-intensity interval training versus moderate-intensity continuous training for improving physical health in elderly women. INQUIRY. (2023) 60:1428200698. doi: 10.1177/00469580231172870
109. Schmitt J, Lindner N, Reuss-Borst M, Holmberg HC, Sperlich B. A 3-week multimodal intervention involving high-intensity interval training in female cancer survivors: a randomized controlled trial. Physiol Rep. (2016) 4:e12693. doi: 10.14814/phy2.12693
110. Simonsson E, Levik SS, Hedlund M, Holmberg H, Johansson B, Lindelof N, et al. Effects of controlled supramaximal high-intensity interval training on cardiorespiratory fitness and global cognitive function in older adults: the umea HIT study-A randomized controlled trial. J GERONTOL A-BIOL. (2023) 78:1581–90. doi: 10.1093/gerona/glad070
111. Siri M, Hollekim-Strand M, Bjørgaas MR, Grethe Albrektsen P, Arnt E. Tjønna P, Ulrik Wisløff P, et al. High-intensity interval exercise effectively improves cardiac function in patients with type 2 diabetes mellitus and diastolic dysfunction. J Am Coll Cardiol. (2014) 64:1758–62. doi: 10.1016/j.jacc.2014.07.971
112. Steen Krawcyk R, Vinther A, Petersen NC, Faber J, Iversen HK, Christensen T, et al. Effect of home-based high-intensity interval training in patients with lacunar stroke: A randomized controlled trial. Front Neurol. (2019) 10:664. doi: 10.3389/fneur.2019.00664
113. Stefanelli F, Meoli I, Cobuccio R, Curcio C, Amore D, Casazza D, et al. High-intensity training and cardiopulmonary exercise testing in patients with chronic obstructive pulmonary disease and non-small-cell lung cancer undergoing lobectomy. Eur J CARDIO-THORAC. (2013) 44:e260–5. doi: 10.1093/ejcts/ezt375
114. Suryanegara J, Cassidy S, Ninkovic V, Popovic D, Grbovic M, Okwose N, et al. High intensity interval training protects the heart during increased metabolic demand in patients with type 2 diabetes: a randomised controlled trial. Acta Diabetol. (2019) 56:321–9. doi: 10.1007/s00592-018-1245-5
115. Taylor JL, Holland DJ, Mielke GI, Bailey TG, Johnson NA, Leveritt MD, et al. Effect of high-intensity interval training on visceral and liver fat in cardiac rehabilitation: A randomized controlled trial. OBESITY. (2020) 28:1245–53. doi: 10.1002/oby.22833
116. Terada T, Friesen A, Chahal BS, Bell GJ, McCargar LJ, Boulé NG. Feasibility and preliminary efficacy of high intensity interval training in type 2 diabetes. Diabetes Res Clin PR. (2013) 99:120–9. doi: 10.1016/j.diabres.2012.10.019
117. Tjønna AE, Lee SJ, Kemi OJ, Najjar SM, Wisløff U, Rognmo Ø, et al. Aerobic interval training versus continuous moderate exercise as a treatment for the metabolic syndrome: A pilot study. Circ (New York N.Y.). (2008) 118:346–54. doi: 10.1161/CIRCULATIONAHA.108.772822
118. Toohey K, Pumpa KL, Arnolda L, Cooke J, Yip D, Craft PS, et al. A pilot study examining the effects of low-volume high-intensity interval training and continuous low to moderate intensity training on quality of life, functional capacity and cardiovascular risk factors in cancer survivors. PEERJ. (2016) 4:e2613. doi: 10.7717/peerj.2613
119. Gjellesvik TI, Becker F, Nilsen H, Lydersen S. Effects of High-Intensity Interval Training after Stroke (The HIIT-Stroke study) – A Multicenter Randomized Controlled Trial. Arch Phys Med REHAB. (2020) 101:939–47. doi: 10.1016/j.apmr.2020.02.006
120. Trachsel LD, Nigam A, Fortier A, Lalonge J, Juneau M, Gayda M. Moderate-intensity continuous exercise is superior to high-intensity interval training in the proportion of VO(2peak) responders after ACS. Rev Esp Cardiol (Engl Ed). (2020) 73:725–33. doi: 10.1016/j.recesp.2019.09.024
121. Trine Moholdt ILA, Granøien I, Lisbeth G, Gitte M, Walderhaug L, Line B, et al. Aerobic interval training increases peak oxygen uptake more than usual care exercise training in myocardial infarction patients: a randomized controlled study. Clin Rehabil. (2011) 1:33–44. doi: 10.1177/0269215511405229
122. Twerenbold S, Hauser C, Gander J, Carrard J, Gugleta K, Hinrichs T, et al. Short-term high-intensity interval training improves micro- but not macrovascular function in hypertensive patients. Scand J Med Sci SPOR. (2023) 33:1231–41. doi: 10.1111/sms.14343
123. Victoria L, Wyckelsma IL, Robyn MM, Aaron CP, Ben DP, Hedges CP, et al. Intense interval training in healthy older adults increases skeletal muscle [3H]ouabain-binding site content and elevates Na+,K+-ATPase a2 isoform abundance in Type II fibers. Physiol Rep. (2017) 5:e13219. doi: 10.14814/phy2.13219
124. Villelabeitia-Jaureguizar K, Vicente-Campos D, Berenguel Senen A, Hernández Jiménez V, Ruiz Bautista L, Barrios-Garrido-Lestache ME, et al. Mechanical efficiency of high versus moderate intensity aerobic exercise in coronary heart disease patients: A randomized clinical trial. Cardiol J. (2019) 26:130–7. doi: 10.5603/CJ.a2018.0052
125. Warburton DER, McKenzie DC, Haykowsky MJ, Taylor A, Shoemaker P, Ignaszewski AP, et al. Effectiveness of high-intensity interval training for the rehabilitation of patients with coronary artery disease. Am J Cardiol. (2005) 95:1080–4. doi: 10.1016/j.amjcard.2004.12.063
126. Winding KM, Munch GW, Iepsen UW, Van Hall G, Pedersen BK, Mortensen SP. The effect on glycaemic control of low-volume high-intensity interval training versus endurance training in individuals with type 2 diabetes. Diabetes Obes Metab. (2018) 20:1131–9. doi: 10.1111/dom.2018.20.issue-5
127. Wisløff U, Støylen A, Videm V, Bye A, Smith GL, Najjar SM, et al. Superior cardiovascular effect of aerobic interval training versus moderate continuous training in heart failure patients: A randomized study. Circ (New York N.Y.). (2007) 115:3086–94. doi: 10.1161/CIRCULATIONAHA.106.675041
128. Xianghui LIU JL. Effects of high intensity interval exercise on tei index and BNP in hypertensive patients. J Shaoyang University(Natural Sci Edition). (2018) 15:108–16.
129. Arnlov J, Ingelsson E, Sundstrom J, Lind L. Impact of body mass index and the metabolic syndrome on the risk of cardiovascular disease and death in middle-aged men. CIRCULATION. (2010) 121:230–6. doi: 10.1161/CIRCULATIONAHA.109.887521
130. Bhaskaran K, Dos-Santos-Silva I, Leon DA, Douglas IJ, Smeeth L. Association of BMI with overall and cause-specific mortality: a population-based cohort study of 3.6 million adults in the UK. Lancet Diabetes ENDO. (2018) 6:944–53. doi: 10.1016/S2213-8587(18)30288-2
131. Raj Padwal MM, William D, Leslie MM, Lisa M, Lix P, Sumit R, et al. Relationship among body fat percentage, body mass index, and all-cause mortality. Ann Intern Med. (2016) 164:532–41. doi: 10.7326/M15-1181
132. Wang Donghai YSJC. A meta - analysis of the effects of high intensity intermittent training on fitness, body composition and Risk factors of metabolic diseases in the elderly. J Harbin Institute Phys Educ. (2022) 40:80–9.
133. Su L, Fu J, Sun S, Zhao G, Cheng W, Dou C, et al. Effects of HIIT and MICT on cardiovascular risk factors in adults with overweight and/or obesity: A meta-analysis. PloS One. (2019) 14:e0210644. doi: 10.1371/journal.pone.0210644
134. Gibala MJ, Little JP, MacDonald MJ, Hawley JA. Physiological adaptations to low-volume, high-intensity interval training in health and disease. J Physiol. (2012) 590:1077–84. doi: 10.1113/jphysiol.2011.224725
135. Parolin ML, Chesley A, Matsos MP, Spriet LL, Jones NL, Heigenhauser GJF. Regulation of skeletal muscle glycogen phosphorylase and PDH during maximal intermittent exercise. Am J PHYSIOL-ENDOC M. (1999) 277:E890–900. doi: 10.1152/ajpendo.1999.277.5.E890
136. Aktaş H, Uzun YE, Kutlu O, Pençe HH, Özçelik F, Çil EÖ, et al. The effects of high intensity-interval training on vaspin, adiponectin and leptin levels in women with polycystic ovary syndrome. Arch Physiol Biochem. (2022) 128:37–42. doi: 10.1080/13813455.2019.1662450
137. Coll-Risco I, Aparicio VA, Nebot E, Camiletti-Moirón D, Martínez R, Kapravelou G, et al. Effects of interval aerobic training combined with strength exercise on body composition, glycaemic and lipid profile and aerobic capacity of obese rats. J SPORT Sci. (2015) 34:1452–60. doi: 10.1080/02640414.2015.1119296
138. Neeland IJ, Ross R, Després J, Matsuzawa Y, Yamashita S, Shai I, et al. Visceral and ectopic fat, atherosclerosis, and cardiometabolic disease: a position statement. Lancet Diabetes Endocrinol. (2019) 7:715–25. doi: 10.1016/S2213-8587(19)30084-1
139. Dallas KB, Rogo-Gupta L, Elliott CS. What impacts the all cause risk of reoperation after pelvic organ prolapse repair? A comparison of mesh and native tissue approaches in 110,329 women. J UROL. (2018) 200:389–96. doi: 10.1016/j.juro.2018.02.3093
140. Jensen MT, Holtermann A, Bay H, Gyntelberg F. Cardiorespiratory fitness and death from cancer: a 42-year follow-up from the Copenhagen Male Study. BRIT J SPORT Med. (2017) 51:1364–9. doi: 10.1136/bjsports-2016-096860
141. Kodama S. Cardiorespiratory fitness as a quantitative predictor of all-cause mortality and cardiovascular events in healthy men and women. JAMA-J Am Med Assoc. (2009) 301:2024. doi: 10.1001/jama.2009.681
142. Niklas P, Li W, Jens W, Michail T, Kent S. Mitochondrial gene expression in elite cyclists: effects of high-intensity interval exercise. Eur J Appl Physiol. (2010) 110:597–606. doi: 10.1007/s00421-010-1544-1
143. Poelkens F, Hopman MTE, Tack CC, Letter by Poelkens, et al. Regarding article, “Aerobic interval training versus continuous moderate exercise as a treatment for the metabolic syndrome: A pilot study. CIRCULATION. (2009) 119:e225. doi: 10.1161/CIRCULATIONAHA.108.818765
144. Carlson DJ, Dieberg G, Hess NC, Millar PJ, Smart NA. Isometric exercise training for blood pressure management: A systematic review and meta-analysis. MAYO Clin Proc. (2014) 89:327–34. doi: 10.1016/j.mayocp.2013.10.030
Keywords: high-intensity interval training, older adults, physical morphology, cardiopulmonary function, metabolic indicators
Citation: Men J, Zhao C, Xiang C, Zhu G, Yu Z, Wang P, Wu S, Zhang Y, Li Y, Wang L, Gong X, Yang X, Zou S, Ma J, Cui C, Li H, Ma X, Wu W and Wang Y (2025) Effects of high-intensity interval training on physical morphology, cardiopulmonary function, and metabolic indicators in older adults: a systematic review and meta-analysis. Front. Endocrinol. 16:1526991. doi: 10.3389/fendo.2025.1526991
Received: 20 November 2024; Accepted: 19 February 2025;
Published: 25 March 2025.
Edited by:
Leonardo Vidal Andreato, University of the State of Amazonas, BrazilReviewed by:
Inês Amanda Streit, Federal University of Amazonas, BrazilCopyright © 2025 Men, Zhao, Xiang, Zhu, Yu, Wang, Wu, Zhang, Li, Wang, Gong, Yang, Zou, Ma, Cui, Li, Ma, Wu and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jie Men, bWVuamllMjAyMEAxMjYuY29t
†These authors have contributed equally to this work and share first authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.