
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Endocrinol. , 20 March 2025
Sec. Reproduction
Volume 16 - 2025 | https://doi.org/10.3389/fendo.2025.1473228
Background: A newly developed technique, Atherogenic Index of Plasma (AIP), is linked to numerous metabolic disorders. Prior researches have indicated strong correlation between AIP and waist circumference (WC), as well as between WC and infertility. Yet no investigation has examined link involving the AIP and infertility, as well as the potential mediating role of WC in this relationship.
Methods: The study included 1,322 women from the 2013–2018 NHANES. Infertility was the outcome variable. Moreover, mediation analysis explored the mediating role of WC in the above relationships.
Results: There were 1,163 controls and 159 infertile participants among the 1,322 participants. The study demonstrated increased WC and elevated AIP among infertile women. Also, the AIP demonstrated an independent correlation with a higher likelihood of infertility, regardless of adjustments for confounding factors. Subgroup analysis indicated the AIP was related to the prevalence of infertility even among women aged 35 years or younger with no history of cardiovascular disease (CVD), pelvic infections, or use of female hormones. Finally, WC had a substantial mediating effect on correlation between AIP and infertility, accounting for 54.49% of the association. Yet, it appears that the various IR surrogates did not demonstrate variability in their predictive ability for infertility [AIP: 0.642 (95% CI: 0.599, 0.683) vs. WC 0.658 (95% CI: 0.618, 0.705) vs. HOMA-IR 0.637 (95% CI: 0.593, 0.686)].
Conclusion: A notable positive correlation exists between AIP and female infertility. It provides the first evidence to demonstrate the mediating role of WC in the above relationship. Managing abdominal obesity and monitoring AIP levels may contribute to reduce the likelihood of infertility.
An estimated 8 to 12% of couples worldwide experience infertility, which is characterized as the inability to conceive after 12 months of regular, unprotected intercourse (1–3). The infertility rate among women in the US aged 18 to 45 has been rising, increasing from 5.8% in the period 2006-2010 to 8.1% in 2017-2019 (4). Beyond its emotional toll, infertility precipitates significant socio-economic challenges and familial strain, positioning it as a pressing public health issue. Consequently, the U.S. Centers for Disease Control and Prevention (CDC) underscores the importance of prioritizing the diagnosis and treatment of infertility to mitigate its impact (5).
An expanding body of both animal and clinical research underscores a notable correlation between infertility and insulin resistance (IR) (6–8). A widely utilized and straightforward way to measure IR is the Homeostasis Model Assessment of Insulin Resistance (HOMA-IR), yet it is challenging to implement in the vast majority of areas that are developing. AIP, a novel, straightforward, and dependable marker for atherosclerosis and cardiovascular disease risk prediction, is derived from the logarithmic transformation of the triglyceride (TG) to high-density lipoprotein cholesterol (HDL-C) ratio (9–15). Recent investigations have established a robust correlation between AIP and conditions such as IR, prediabetes, and diabetes, highlighting its potential as a significant predictor of metabolic disorders (16–20). Given the pronounced link between AIP and IR, exploring the potential connection between AIP and infertility emerges as a crucial step towards identifying more convenient and accessible markers for screening infertility in women of reproductive age. Yet, to date, an investigation into the relationship between AIP and the prevalence of infertility among reproductive-aged females remains unexplored.
Previous studies have identified WC as a contributing factor to infertility (21–23). Additionally, prior researches have indicated strong correlations between AIP and WC (24–26). Nevertheless, the interaction between them remains thoroughly unexplored. Exploring the mediating role of WC could offer valuable insights into the mechanisms by which AIP impacts the risk of infertility, potentially uncovering new avenues for intervention. This could lead to more targeted interventions, ultimately improving infertility prevention.
Thus, we endeavor to illuminate the correlation between the AIP and infertility in the US by conducting a cross-sectional investigation of information from the NHANES covering the years 2013 to 2018.
The NHANES was conducted under the oversight of the NCHS Research Ethics Review Board. This investigation utilized data from the 2013–2018 NHANES, encompassing a cohort of 29,400 participants. The study’s methodology is depicted in Figure 1. The exclusion criteria were rigorously applied as follows: (1) 14451 male participants were excluded; (2) 10,625 individuals beyond the age of 45 years or younger than 18 years were omitted; (3) those without available AIP values were excluded (N=2,574); (4) subjects with incomplete fertility data were removed (N=194); (5) participants missing either the outcome of interest or necessary covariate information were also excluded (N=234). Following this meticulous screening process, a cohort of 1,322 eligible subjects was retained for subsequent analysis.
Using self-reports and the reproductive health questionnaire, infertility was characterized. Individuals who affirmed experiencing infertility were classified into the infertility group; otherwise, they were categorized as not having infertility. To maintain the integrity and precision of the study’s outcomes, participants who either abstained from answering or expressed uncertainty about their fertility status were systematically removed from the analysis. This approach ensured that the analysis was conducted with the most accurate and reliable data available.
The formula for calculating AIP which is an exposure variable, is log [TG (mg/dL)/HDL-C(mg/dL)]. Utilizing subsequent definitions of the quartiles, subjects were divided into different categories relying on their AIP values: Q1 (AIP < -0.077), Q2 (AIP -0.077 to <0.127), Q3 (AIP -0.127 to < 0.348), and Q4 (AIP ≥ 0.348) (27).
All waist circumference is taken at the mobile examination center (MEC). HOMA-IR is equal to fasting insulin (µU/mL) ×fasting glucose (mmol/L)/22.5 (28).
In this study, covariates were meticulously selected to encompass both continuous variables, such as age and poverty income ratio (PIR), and categorical variables. The categorical variables included race, smoking status, education level, BMI status, CVD, leisure time physical activity (LTPA), daily sitting time, history of treatment for pelvic inflammatory disease (PID), usage of female hormones, and history of taking birth control pills in accordance with prior characterized or clinical expertise (21–23).
The categorizations of participants’ responses regarding PID, whether they have ever used birth control pills, and whether they have ever used female hormones are straightforwardly determined based on participants’ responses, categorized as either “yes” or “no”. CVD encompassed conditions that were clinically diagnosed, including coronary heart disease, congestive heart failure, stroke, and angina. Self-reported data on daily sitting time and weekly LTPA were collected through respondent-level interviews. Based on their sedentary time, participants were classified into four groups (29). For physical activity levels, participants were divided into three categories: inactive (individuals not engaging in any LTPA), insufficiently active (0 - 150 min LTPA/week), and sufficiently active (>150 min LTPA/week) (30).
The statistical analyses were conducted using R 4.4.2 software and EmpowerStats. The threshold for determining statistical significance was established at P < 0.05. Characteristics across groups were contrasted utilizing t-tests and chi-square tests in Table 1. Levels of AIP were stratified into four groups for analysis. This analysis involved the calculation of odds ratios (ORs) and 95% confidence intervals (CIs) to quantify the strength and precision of these associations. No factor was altered by Model 1. Model 2 brought into aspects including age, racial background, levels of education, and PIR. Model 3 was adjusted for smoked status, pelvic infection, ever taken birth control pills, ever use female hormones, CVD, daily sitting time, LTPA, building upon the adjustments made in Model 2. Subsequently, the effects of AIP on infertility were evaluated through subgroup analyses and interaction tests, focusing on groups characterized by age, smoked status, daily sitting time, history of pelvic infection, usage of female hormones, history of taking birth control pills, CVD and LTPA.
In addition, the non-linear connections between AIP and infertility were researched utilizing a Generalized Additive Model (GAM). Furthermore, mediation analysis was carried out to investigate whether waist circumference mediated the relation between AIP and infertility in reproductive-aged women.
There were 1,163 controls and 159 infertile participants among the 1,322 participants. Table 1 reveals the features of the subjects categorized by AIP quartiles. AIP levels failed to indicate any meaningful correlations with CVD, daily sitting duration, history of pelvic infection, usage of female hormones, or history of taking birth control pills (P > 0.05). In contrast, AIP were correlations with to age, race, education level, PIR, WC, smokers, LTPA and individuals with infertility (all P-values < 0.05). Participants with increased AIP tended to be older with a larger WC, lower PIR, and education levels compared to those in the lower AIP quartile groups.
The results demonstrated a considerably greater likelihood of WC and elevated AIP among infertile women in the US, across all models, regardless of adjustments for confounding factors in Table 2. After adjusting for all confounding variables (model 3), each 1-unit increase in AIP was linked with an 94% increase in the odds of infertility. Furthermore, in Model 3, which adjusted for all relevant covariates, participants in the highest AIP quartile had an OR of 1.97(95% CI: 1.13–3.44), indicating an 97% higher likelihood of infertility in comparison with others in the lower AIP quartile groups.
The smooth curve fitting demonstrated a non-linear link between AIP and infertile participants (Figure 2). Further calculations determined the inflection point to be 0.16 (Table 3). AIP and female infertility exhibited an intensely favorable relationship before the inflection point, with an OR of 11.13(95% CI: 2.83–43.72). This indicates a strong relationship between higher AIP levels and increased infertility risk up to the inflection point. However, beyond the inflection point, the link between AIP and female infertile participants was not significant, with an OR of 0.71(95% CI: 0.28–1.78), indicating a significant threshold effect of AIP and infertility.
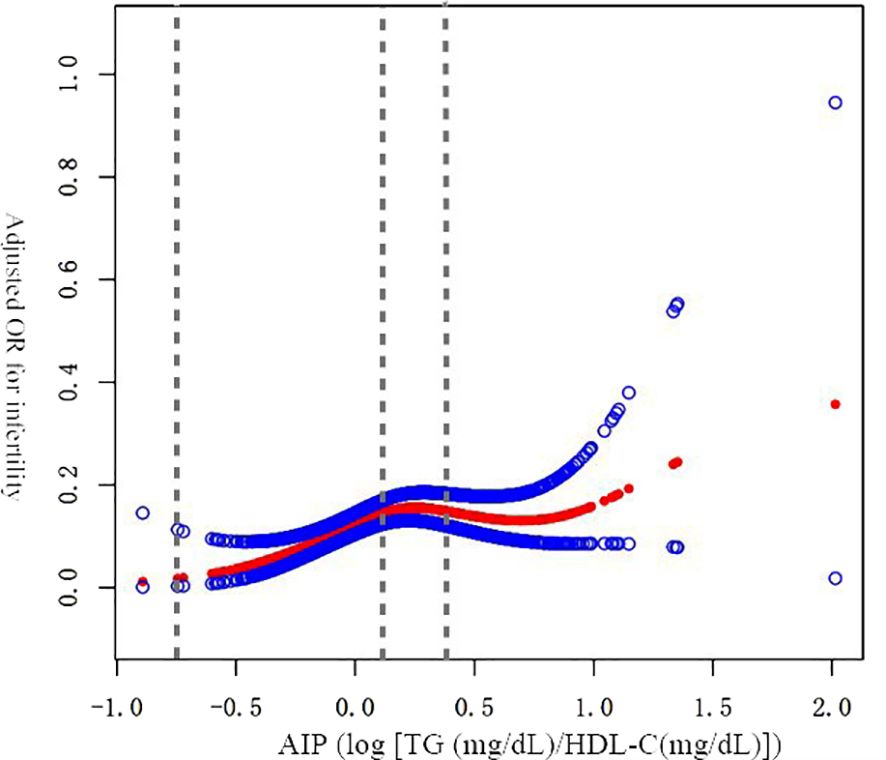
Figure 2. Non-linear link between the AIP and infertile participants. adjusted for all relevant covariates including adjusted for age, racial background, levels of education, PIR, smoked status, PID, ever taken birth control pills, ever use female hormones, CVD, daily sitting time, LTPA.
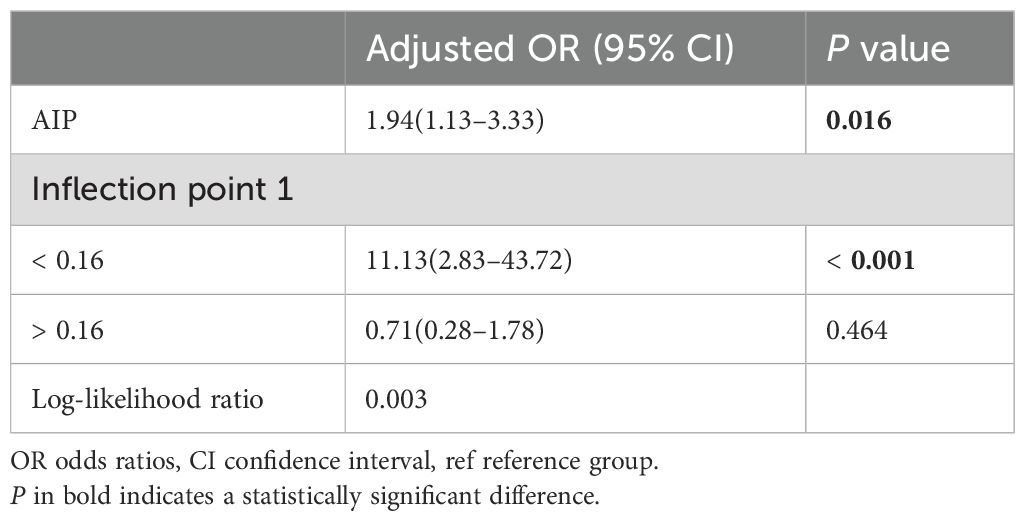
Table 3. Threshold effect analysis of AIP on female infertility using a two-piecewise linear regression model.
To further investigate the factors influencing the link between AIP and the likelihood of female infertility, we conducted stratified analyses and interaction tests based on age, smoked status, daily sitting time, history of pelvic infection, usage of female hormones, history of taking birth control pills, CVD and LTPA (Table 4). Significant interactions were detected for age, smoked status, daily sitting time, history of pelvic infection, ever taken birth control pills, ever use female hormones, CVD and LTPA, with all P-values for interaction being < 0.05. A positive link between AIP and female infertile participants persisted in specific subgroups, including women aged 35 years or younger, smokers, people whether have CVD, those without a history of pelvic infections, users of female hormones, and individuals who have ever taken birth control pills. Notably, the subgroup analysis indicated that even among women aged 35 years or younger with no history of cardiovascular disease, pelvic infections, or use of female hormones, an elevated AIP still correlates with the gradually relative odds of infertility.
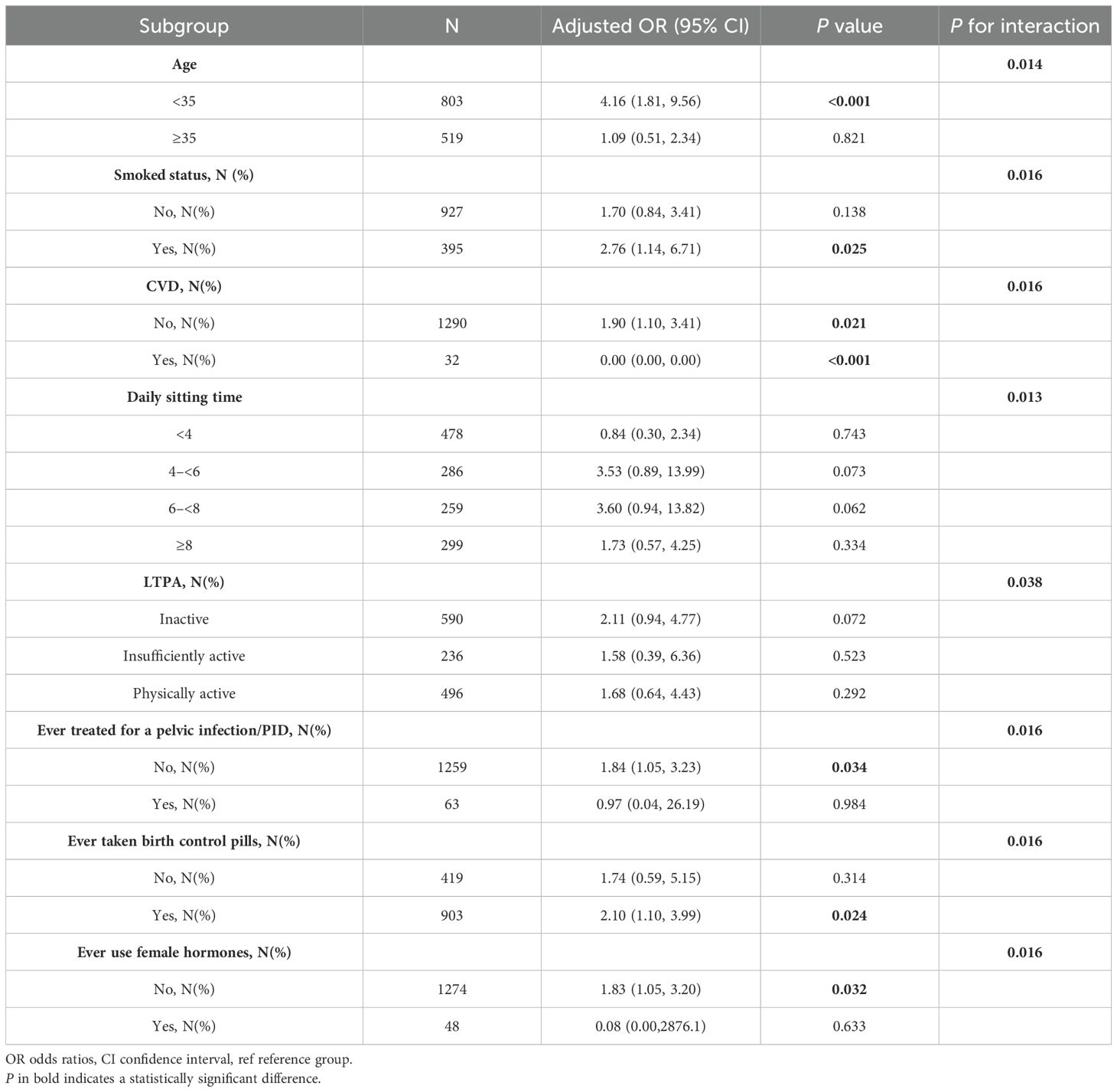
Table 4. Stratified associations between AIP and female infertility according to baseline characteristics.
All of the models in Table 5 demonstrated an important positive correlation between WC and female infertility [model 1: OR (95% CI) =1.020(1.011–1.028); model 2: OR (95% CI) = 1.017(1.008–1.027); model 3: OR (95% CI) = 1.018 (1.009–1.028)].
An increased AIP level was associated with an elevated risk of WC across all models [model 1: β (95% CI) = 16.14(20.38–26.02); model 2: β (95% CI) = 16.37(20.56–26.15); model 3: β (95% CI) = 5.60 (19.79–25.48)] as shown in Table 6. In Model 3, the WC of the highest quartile was 14.6cm higher than that of the lowest quartile.
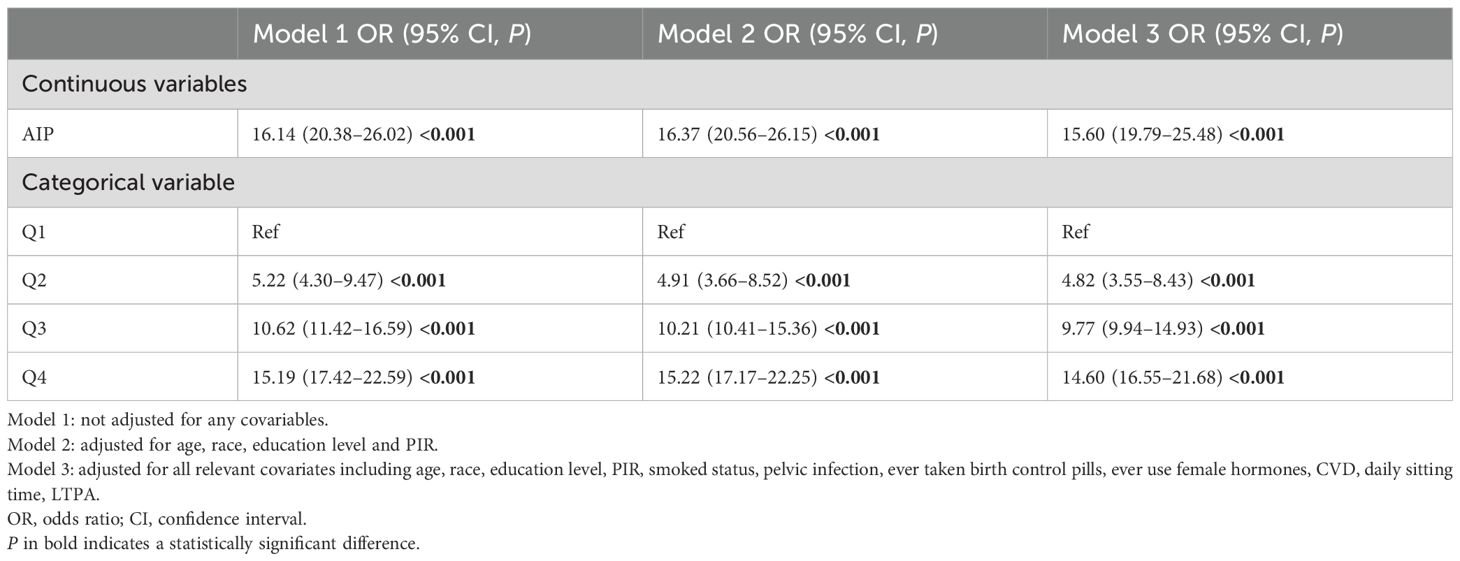
Table 6. Relative odds of waist circumference according to AIP in different models among all participants.
As depicted in Figure 3, WC demonstrated a strong link with infertility [total effect (95% CI, P): 0.030(0.009,0.051), 0.012]. Additionally, there was a substantial moderating effect for WC in the link between AIP and infertility [mediation effect (95% CI, P): 0.016(0.005–0.026), 0.004], accounting for 54.49% of the association. While there was no direct correlation between AIP and infertility [direct effect (95% CI, P): 0.014 (-0.011, 0.037), 0.337]. These findings indicate that WC served as a significant complete mediator in the link between AIP and infertility.
The results of the ROC curves were presented in Table 7; Figure 4. Regarding infertility, the AUCs of AIP, WC and the HOMA-IR index were 0.642 (95% CI: 0.599, 0.683) vs. 0.658 (95% CI: 0.618, 0.705) vs. 0.637 (95% CI: 0.593, 0.686). Although the AUC of WC was higher, there was no statistically significant difference between the AUCs of the diverse surrogates (P > 0.05). It appears that the various IR surrogates did not demonstrate variability in their predictive ability for infertility.
The study involving 1,322 women aged 18–45 years, demonstrated a positive correlation between AIP and experiencing infertility regardless of adjustments for confounding factors. A nonlinear correlation between AIP and infertility was observed, with different relationships detected on either side of the breakpoint (AIP = 0.16). Specifically, AIP was positively associated with the likelihood of infertility on the left side of the breakpoint, while the association on the right side was not statistically significant, indicating a significant threshold effect of AIP on female infertility. Significant associations between AIP and infertility were more likely to occur among women aged 35 years or younger with no history of cardiovascular disease, pelvic infections, or use of female hormones. Additionally, this research demonstrated that WC is a contributing factor to infertility, and high levels of AIP are positively correlated with WC. Notably, WC was a significant complete mediator of the relationship between AIP and infertility. Yet, it appears that the various IR surrogates did not demonstrate variability in their predictive ability for infertility.
Studies have shown that polycystic ovary syndrome (PCOS), endometriosis, and endometritis are linked to infertility. As of now, there have been no studies that have demonstrated a potential link between AIP and infertile participants. Yet Previous investigations demonstrate that lipid metabolism are associated with an increased risk of infertility. For instance, Essah et al. found that American women with PCOS had higher mean TG levels and lower mean serum HDL cholesterol levels (31). Similarly, Fatma et al. demonstrated that elevated TG and decreased HDL-C were linked to the exacerbation of endometriosis in women (32). Consistent with prior investigations, our study identified that elevated AIP have a correlation with a higher likelihood of infertility. Additionally, subgroup analyses showed that the above correlation was more pronounced in women aged 35 years or younger. This finding is intriguing as it challenges the common belief that advanced age is the primary factor contributing to reproductive dysfunction in women, but our study indicated otherwise. This may be related to estrogen in females, lowering TG level and influencing AIP. A separate study revealed that the serum estradiol levels were elevated in older women compared to younger women (21–35 years) (33, 34). The specific mechanism needs to be further studied.
Prior research has indicated that obesity has a negative impact on reproductive health, particularly in relation to infertility (35–39). As the prevalence of abdominal obesity increases, WC is crucial for obesity surveillance (40). Many research has demonstrated a correlation between WC and infertility. Li et al. demonstrated a negative relationship between WC and infertile women (21). The smaller WC was connected with a higher likelihood of becoming pregnant, according to research by Moran et al. (41). Furthermore, in a cross-sectional observational study of 3239 women in American, Jierong et al. observed that WC, was relevant in predicting female infertility outcomes (22). In our study, AIP was not directly associated with infertility but rather might be mediated by WC. The connection between AIP and infertility, as well as the heightened risk of infertility due to WC, can be accounted for by several potential biological mechanisms. First, AIP is a biomarker of dyslipidemia. Abnormal blood lipid levels can induce IR through mechanisms involving inflammation and oxidative stress (42, 43). Dyslipidemia and IR is significantly related to WC (44, 45). IR can negatively affect oocyte quality by impairing mitochondrial function, which is crucial for maintaining oocyte health (8). Many participants with PCOS experience oxidative stress, where excess reactive oxygen species (ROS) disrupt mitochondrial function and activate inflammatory factors such as TNF-α, interleukin 1β (IL-1β), and interleukin 6 (IL-6). These inflammatory responses can impair endometrial receptivity by reducing insulin sensitivity, thereby affecting female reproductive function (46–49). Therefore, the potential mechanisms underlying the associations of AIP with WC and WC with infertility likely involve IR, dyslipidemia, oxidative stress, and inflammatory factors.
As far as we are aware, this study presents the first evidence of a stable positive link between AIP and an elevated likelihood of infertility utilizing information collected by NHANES. In light of the prospective significance of this study for future research on the link and mechanism between AIP and infertility, we think it is a valuable beginning for further study of this association. The outcomes highlighted the clinical utility of AIP as an indicator, including its capacity to replace the present, costly standard tests for diagnosing infertility and to predict the prognosis of infertility as well as the impacts of the use of assisted reproductive technologies. There should be more research done on the underlying mechanism of the link between AIP and infertility. Since these parameters are affordable, simple to use, and highly linked with the prevalence of infertility. Moreover, through mediation analysis, it is the first to confirm that WC may indeed act as a mediator in this relationship. Hence, female who attempt to conceive may enhance their likelihood of achieving pregnancy by reducing their waist circumference.
However, this study also has some limitations. First, this study was a cross-sectional study, limiting the ability to determine a causal relationship between AIP and infertility. Second, infertility outcomes were based on self-reported data, which may introduce information and recall bias and represent only the subject’s past disease state; current disease state outcomes are unknown, and these may have influenced the results. Finally, confounding factors that may have influenced the observed associations cannot be excluded.
The study presents the first evidence of a stable positive association between AIP and an increased risk of infertility. Additionally, it is the first to demonstrate the mediating role of waist circumference in the link between AIP and infertility. It implies that maintaining AIP within a lower range and prioritizing WC regulation is imperative to mitigate the likelihood of infertility.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below: https://www.cdc.gov/nchs/nhanes/index.htm.
The studies involving humans were approved by NCHS Research Ethics Review Board. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
XW: Conceptualization, Methodology, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. DL: Data curation, Formal Analysis, Investigation, Methodology, Project administration, Software, Writing – original draft, Writing – review & editing.
The author(s) declare that no financial support was received for the research and/or publication of this article.
The authors express their gratitude to all staff and participants who contributed to the NHANES database.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Vander Borght M, Wyns C. Fertility and infertility: Definition and epidemiology. Clin Biochem. (2018) 62:2–10. doi: 10.1016/j.clinbiochem.2018.03.012
2. Nichols AR, Rifas-Shiman SL, Switkowski KM, Zhang M, Young JG, Hivert M-F, et al. History of infertility and midlife cardiovascular health in female individuals. JAMA Netw Open. (2024) 7:e2350424. doi: 10.1001/jamanetworkopen.2023.50424
3. Alan P, Kristin B, Tommaso F, Karl H, Micah H, William H, et al. Definitions of infertility and recurrent pregnancy loss: a committee opinion. Fertil Steril. (2020) 113:533–5. doi: 10.1016/j.fertnstert.2019.11.025
4. Snow M, Vranich TM, Perin J, Trent M. Estimates of infertility in the United States: 1995–2019. Fertil Steril. (2022) 118:560–7. doi: 10.1016/j.fertnstert.2022.05.018
5. Macaluso M, Wright-Schnapp TJ, Chandra A, Johnson R, Satterwhite CL, Pulver A, et al. A public health focus on infertility prevention, detection, and management. Fertil Steril. (2010) 93:16.e1–16.e10. doi: 10.1016/j.fertnstert.2008.09.046
6. Ou X-H, Li S, Wang Z-B, Li M, Quan S, Xing F, et al. Maternal insulin resistance causes oxidative stress and mitochondrial dysfunction in mouse oocytes. Hum Reprod. (2012) 27:2130–45. doi: 10.1093/humrep/des137
7. Wu S, Divall S, Nwaopara A, Radovick S, Wondisford F, Ko C, et al. Obesity-induced infertility and hyperandrogenism are corrected by deletion of the insulin receptor in the ovarian theca cell. Diabetes. (2014) 63:1270–82. doi: 10.2337/db13-1514
8. Hansda SR, Haldar C. Uterine anomalies in cell proliferation, energy homeostasis and oxidative stress in PCOS hamsters, M. auratus: Therapeutic potentials of melatonin. Life Sci. (2021) 281:119755. doi: 10.1016/j.lfs.2021.119755
9. Wu T-T, Zheng Y-Y, Yang Y-N, Li X-M, Ma Y-T, Xie X. Age, sex, and cardiovascular risk attributable to lipoprotein cholesterol among chinese individuals with coronary artery disease: A case–control study. Metab Syndrome Related Disord. (2019) 17:223–31. doi: 10.1089/met.2018.0067
10. Dobiásová M. AIP–atherogenic index of plasma as a significant predictor of cardiovascular risk: from research to practice. Vnitr Lek. (2006) 52:64–71.
11. Wu T-T, Gao Y, Zheng Y-Y, Ma Y-T, Xie X. Atherogenic index of plasma (AIP): a novel predictive indicator for the coronary artery disease in postmenopausal women. Lipids Health Dis. (2018) 17:197. doi: 10.1186/s12944-018-0828-z
12. Zheng Y, Li C, Yang J, Seery S, Qi Y, Wang W, et al. Atherogenic index of plasma for non-diabetic, coronary artery disease patients after percutaneous coronary intervention: a prospective study of the long-term outcomes in China. Cardiovasc Diabetol. (2022) 21:29. doi: 10.1186/s12933-022-01459-y
13. Fernández-Macías JC, Ochoa-Martínez AC, Varela-Silva JA, Pérez-Maldonado IN. Atherogenic index of plasma: novel predictive biomarker for cardiovascular illnesses. Arch Med Res. (2019) 50:285–94. doi: 10.1016/j.arcmed.2019.08.009
14. Kim SH, Cho YK, Kim Y-J, Jung CH, Lee WJ, Park J-Y, et al. Association of the atherogenic index of plasma with cardiovascular risk beyond the traditional risk factors: a nationwide population-based cohort study. Cardiovasc Diabetol. (2022) 21:81. doi: 10.1186/s12933-022-01522-8
15. Sadeghi M, Heshmat-Ghahdarijani K, Talaei M, Safaei A, Sarrafzadegan N, Roohafza H. The predictive value of atherogenic index of plasma in the prediction of cardiovascular events; a fifteen-year cohort study. Adv Med Sci. (2021) 66:418–23. doi: 10.1016/j.advms.2021.09.003
16. Fu L, Zhou Y, Sun J, Zhu Z, Xing Z, Zhou S, et al. Atherogenic index of plasma is associated with major adverse cardiovascular events in patients with type 2 diabetes mellitus. Cardiovasc Diabetol. (2021) 20:201. doi: 10.1186/s12933-021-01393-5
17. Yin B, Wu Z, Xia Y, Xiao S, Chen L, Li Y. Non-linear association of atherogenic index of plasma with insulin resistance and type 2 diabetes: a cross-sectional study. Cardiovasc Diabetol. (2023) 22:157. doi: 10.1186/s12933-023-01886-5
18. Onat A, Can G, Kaya H, Hergenç G. Atherogenic index of plasma” (log10 triglyceride/high-density lipoprotein–cholesterol) predicts high blood pressure, diabetes, and vascular events. J Clin Lipidol. (2010) 4:89–98. doi: 10.1016/j.jacl.2010.02.005
19. Vatcheva KP, Fisher-Hoch SP, Reininger BM, McCormick JB. Sex and age differences in prevalence and risk factors for prediabetes in Mexican-Americans. Diabetes Res Clin Pract. (2020) 159:107950. doi: 10.1016/j.diabres.2019.107950
20. Rahimlou M, Mirzaei K, Keshavarz SA, Hossein-nezhad A. Association of circulating adipokines with metabolic dyslipidemia in obese versus non-obese individuals. Diabetes Metab Syndrome: Clin Res Rev. (2016) 10:S60–5. doi: 10.1016/j.dsx.2015.09.015
21. Li M-C, Mínguez-Alarcón L, Arvizu M, Chiu Y-H, Ford JB, Williams PL, et al. Waist circumference in relation to outcomes of infertility treatment with assisted reproductive technologies. Am J Obstet Gynecol. (2019) 220:578.e1–578.e13. doi: 10.1016/j.ajog.2019.02.013
22. Ke J, Feng Y, Chen Z. Association between waist circumference and female infertility in the United States. PloS One. (2023) 18:e0295360. doi: 10.1371/journal.pone.0295360
23. Yin Y-H, Zhou S-Y, Lu D-F, Chen X-P, Liu B, Lu S, et al. Higher waist circumference is associated with increased likelihood of female infertility: NHANES 2017-2020 results. Front Endocrinol. (2023) 14:1216413. doi: 10.3389/fendo.2023.1216413
24. Shin HR, Song S, Cho JA, Ly SY. Atherogenic index of plasma and its association with risk factors of coronary artery disease and nutrient intake in korean adult men: the 2013–2014 KNHANES. Nutrients. (2022) 14:1071. doi: 10.3390/nu14051071
25. Lioy B, Webb RJ, Amirabdollahian F. The association between the atherogenic index of plasma and cardiometabolic risk factors: A review. Healthcare. (2023) 11:966. doi: 10.3390/healthcare11070966
26. Niroumand S, Khajedaluee M, Khadem-Rezaiyan M, Abrishami M, Juya M, Khodaee G, et al. Atherogenic Index of Plasma (AIP): A marker of cardiovascular disease. Med J Islam Repub Iran. (2015) 29:240.
27. Dobiásová M, Frohlich J. The plasma parameter log (TG/HDL-C) as an atherogenic index: correlation with lipoprotein particle size and esterification rate in apoB-lipoprotein-depleted plasma (FER(HDL)). Clin Biochem. (2001) 34:583–8. doi: 10.1016/s0009-9120(01)00263-6
28. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and?-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. (1985) 28:412–9. doi: 10.1007/BF00280883
29. Li Y, Di X, Liu M, Wei J, Li T, Liao B. Association between daily sitting time and kidney stones based on the National Health and Nutrition Examination Survey (NHANES) 2007-2016: A cross-sectional study. Int J Surg. (2024) 110(8):4624–32. doi: 10.1097/JS9.0000000000001560
30. Piercy KL, Troiano RP, Ballard RM, Carlson SA, Fulton JE, Galuska DA, et al. The physical activity guidelines for americans. JAMA. (2018) 320:2020–8. doi: 10.1001/jama.2018.14854
31. Essah PA, Nestler JE, Carmina E. Differences in dyslipidemia between American and Italian women with polycystic ovary syndrome. J Endocrinol Invest. (2008) 31:35–41. doi: 10.1007/BF03345564
32. Verit FF, Erel O, Celik N. Serum paraoxonase-1 activity in women with endometriosis and its relationship with the stage of the disease. Hum Reprod. (2008) 23:100–4. doi: 10.1093/humrep/dem340
33. Homma H, Kurachi H, Nishio Y, Takeda T, Yamamoto T, Adachi K, et al. Estrogen suppresses transcription of lipoprotein lipase gene. J Biol Chem. (2000) 275:11404–11. doi: 10.1074/jbc.275.15.11404
34. Burger HG, Hale GE, Dennerstein L, Robertson DM. Cycle and hormone changes during perimenopause: the key role of ovarian function. Menopause. (2008) 15:603–12. doi: 10.1097/gme.0b013e318174ea4d
35. Catalano PM, Shankar K. Obesity and pregnancy: mechanisms of short term and long term adverse consequences for mother and child. BMJ. (2017) 356:j1. doi: 10.1136/bmj.j1
36. Poston L, Caleyachetty R, Cnattingius S, Corvalán C, Uauy R, Herring S, et al. Preconceptional and maternal obesity: epidemiology and health consequences. Lancet Diabetes Endocrinol. (2016) 4:1025–36. doi: 10.1016/S2213-8587(16)30217-0
37. Pasquali R. Obesity and reproductive disorders in women. Hum Reprod Update. (2003) 9:359–72. doi: 10.1093/humupd/dmg024
38. Snider AP, Wood JR. Obesity induces ovarian inflammation and reduces oocyte quality. Reproduction. (2019) 158:R79–90. doi: 10.1530/REP-18-0583
39. Glueck CJ, Goldenberg N. Characteristics of obesity in polycystic ovary syndrome: Etiology, treatment, and genetics. Metabolism. (2019) 92:108–20. doi: 10.1016/j.metabol.2018.11.002
40. Ross R, Neeland IJ, Yamashita S, Shai I, Seidell J, Magni P, et al. Waist circumference as a vital sign in clinical practice: a Consensus Statement from the IAS and ICCR Working Group on Visceral Obesity. Nat Rev Endocrinol. (2020) 16:177–89. doi: 10.1038/s41574-019-0310-7
41. Moran L, Tsagareli V, Norman R, Noakes M. Diet and IVF pilot study: Short-term weight loss improves pregnancy rates in overweight/obese women undertaking IVF: Weight loss and IVF. Aust New Z J Obstet Gynaecol. (2011) 51:455–9. doi: 10.1111/j.1479-828X.2011.01343.x
42. Goodpaster BH, Kelley DE. Skeletal muscle triglyceride: Marker or mediator of obesity-induced insulin resistance in type 2 diabetes mellitus? Curr Diabetes Rep. (2002) 2:216–22. doi: 10.1007/s11892-002-0086-2
43. Li N, Fu J, Koonen DP, Kuivenhoven JA, Snieder H, Hofker MH. Are hypertriglyceridemia and low HDL causal factors in the development of insulin. Curr Diabetes Rep. (2014) 233(1):130–138. doi: 10.1016/j.atherosclerosis.2013.12.013
44. Iwani NAKZ, Jalaludin MY, Zin RMWM, Fuziah MZ, Hong JYH, Abqariyah Y, et al. Triglyceride to HDL-C ratio is associated with insulin resistance in overweight and obese children. Sci Rep. (2017) 7:40055. doi: 10.1038/srep40055
45. Liu J, Jin X, Feng Z, Huang J. Using anthropometric parameters to predict insulin resistance among patients without diabetes mellitus. Sci Rep. (2024) 14:21407. doi: 10.1038/s41598-024-57020-2
46. Özer A, Bakacak M, Kıran H, Ercan Ö, Köstü B, Kanat-Pektaş M, et al. Increased oxidative stress is associated with insulin resistance and infertility in polycystic ovary syndrome. Ginekol Pol. (2016) 87:733–8. doi: 10.5603/GP.2016.0079
47. Zeng X, Huang Q, Long SL, Zhong Q, Mo Z. Mitochondrial dysfunction in polycystic ovary syndrome. DNA Cell Biol. (2020) 39:1401–9. doi: 10.1089/dna.2019.5172
48. Kim J, Wei Y, Sowers JR. Role of mitochondrial dysfunction in insulin resistance. Circ Res. (2008) 102:401–14. doi: 10.1161/CIRCRESAHA.107.165472
Keywords: atherogenic index of plasma, infertility, NHANES, waist circumference, cross-sectional study
Citation: Wei X and Liu D (2025) Waist circumference mediates the relationship between atherogenic index of plasma and infertility. Front. Endocrinol. 16:1473228. doi: 10.3389/fendo.2025.1473228
Received: 30 July 2024; Accepted: 03 March 2025;
Published: 20 March 2025.
Edited by:
Fred Sinowatz, Ludwig Maximilian University of Munich, GermanyReviewed by:
William J. Massey, Cleveland Clinic, United StatesCopyright © 2025 Wei and Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Dandan Liu, bGl1YXRoeWRAMTYzLmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.