- 1Pathological Diagnosis Center, Zhoushan Hospital, Zhoushan, Zhejiang, China
- 2Department of Laboratory, Zhoushan Hospital, Zhoushan, Zhejiang, China
- 3Laboratory of Cell Biology and Molecular Biology, Zhoushan Hospital, Zhoushan, Zhejiang, China
- 4Department of Hepatobiliary Surgery, Zhoushan Hospital, Zhoushan, Zhejiang, China
Background: Primary live cancer (PLC), including hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (ICC). This meta-analysis was conducted to evaluate the diagnostic efficacy of blood methylated septin 9 gene (mSEPT9) for PLC and to analyze its performance across various subgroups.
Methods: We conducted a comprehensive search across PubMed, the Cochrane Library, Embase, and China National Knowledge Infrastructure (CNKI), covering research up to May 10, 2024. The pooled sensitivity, specificity, diagnostic odds ratios, and area under the summary receiver operating characteristic (AUC) were calculated for the diagnostic performance of mSEPT9 for PLC. The quality of the studies was assessed using the QUADAS-2 tool, and the meta-analysis was performed using Stata16.0 software.
Results: Ten articles with 2,182 participants were included in the meta-analysis. The pooled sensitivity of mSEPT9 for detecting primary liver cancer was 0.51 (95% confidence interval [CI]: 0.37-0.65), and the pooled specificity was 0.93 (95% CI: 0.78-0.98). The pooled diagnostic odds ratio was 13 (95% CI: -58), and the area under the Summary Receiver Operator Characteristic Curve was 0.75 (95% CI: 0.71-0.79). Subgroup analyses showed that ICC, case-control studies, qPCR and Asian populations had higher specificities (0.99 [95% CI: 0.97–1.00], 0.93 [95% CI: 0.91-0.95], 0.90 [95% CI: 0.88-0.92] and 0.94 [95% CI: 0.92-0.96], respectively) and diagnostic odds ratios (62.04 [95% CI: 6.53-589.53], 17.62 [95% CI: 4.03-76.99], 13.03 [95% CI: 2.01-84.63] and 14.19 [95% CI: 2.42-83.11], respectively) compared to hepatocellular carcinoma, cohort Study, and Euramerican populations.
Conclusions: This study confirmed that mSEPT9 in blood has high specificity and moderate sensitivity for detecting primary liver cancer. The diagnostic performance of mSEPT9 varied across different subgroups, limiting its use as an independent screening tool and necessitating its use in conjunction with other methods for confirmatory diagnostics.
Systematic review registration: https://www.crd.york.ac.uk/prospero/, identifier CRD42024549669.
Introduction
Primary liver cancer (PLC), including hepatocellular carcinoma (HCC) and intrahepatic cholangiocarcinoma (ICC), represents a major public health challenge globally given that it is the sixth most prevalent cancer and the third cause of cancer-related deaths across the globe. It kills around 830K people a year (1). The geographic variability in the incidence of liver cancer is explained in part by the distribution of risk factors, such as chronic infection with hepatitis B or C viruses, aflatoxin exposure, alcohol drinking, and nonalcoholic fatty liver disease. Greater rates of incidence are observed in Eastern Asia and sub-Saharan Africa, due to the high prevalence of chronic hepatitis B infection in those regions. Liver cancer has a dismal prognosis despite advances in early diagnosis and treatment, with a five-year survival rate of <20% in most regions, highlighting the need for innovative diagnostic and therapeutic strategies (2).
PLC is usually diagnosed using imaging techniques (such as ultrasound, computed tomography (CT), or magnetic resonance imaging (MRI) scans and tests for levels of substances (such as alpha-fetoprotein, AFP) in the plasma and/or serum (3). Recently, the era of liquid biopsy opened new possibilities for the non-invasive diagnosis of liver cancer, with expected improvements in sensitivity and specificity. Liquid biopsies, however, provide a dynamic snapshot of the genetics of the tumor and the tumor microenvironment by analyzing circulating tumor cells (CTCs), cell-free DNA (cfDNA), and other biomarkers from a blood sample (4, 5). It has shown promise, particularly in early detection and response and progression monitoring of treatment, highlighted by its enhanced ability to detect molecular sub-steps of tumor genesis and heterogeneity compared to conventional strategies. Recent studies also show that liquid biopsies aid in an appropriate and accurate diagnosis, reducing unnecessary imaging methods and supporting the clinical utility of liquid biopsies for liver cancer (6, 7).
Methylated septin 9 gene (mSEPT9), initially known for its promise in non-invasively indicating the presence of colorectal cancer, has also been investigated for its relevance in multiple other cancers including liver cancer (8). The pattern of methylation adducts in the gene results in its abnormal silencing and is a compelling tumor suppressor gene critical in cellular cytoskeleton organization as well as cell division (9–11). Its use for diagnosing liver cancer shows promise, in part because in initial studies it has shown a high level of sensitivity and specificity. SEPT9 protein expression examined by immunohistochemical (IHC) was significantly different among various forms of hepatic nodules. We demonstrated that SEPT9 expression was significantly correlated with advanced tumor grade and concurrent SATB2 staining in HCC tissues, which supported the importance of SEPT9 in liver carcinogenesis (12). This may highlight how the septic protein is specific in cancer because there was no SEPT9 expression in dysplastic nodes, occasionally in hepatocellular adenoma, in contrast to the non-expression in benign hepatocytes (13). In addition, this approach allows early detection of disease states and may be used to track disease (re)occurrence and response to therapy, making it an ideal candidate for current and future diagnostic strategies (14, 15). However, despite the promising potential of mSEPT9 in liver cancer diagnosis, it should be noted that the clinical application of mSEPT9 testing in most malignancies has not been established. Even in colorectal cancer, where mSEPT9 testing has been used clinically, significant drawbacks were observed, including high operational costs and unsatisfactory sensitivity (16).
The evolving landscape of liver cancer research increasingly supports the use of molecular biomarkers that offer both specificity and non-invasiveness as emerging diagnostic solutions. The methylation of the SEPT9 gene is the most promising target of these markers since it has already shown important diagnostic performance for colorectal cancer and seems to be of potential relevance also for liver cancer. We conducted an exploratory meta-analysis to summarize the diagnostic accuracy of mSEPT9 in patients suspected of primary liver cancer and to analyze its performance across various subgroups, with the aim of identifying areas for further research and refinement.
Methods
Study design
This systematic review was conducted and reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist (17). This study was based on a secondary analysis of published literature and therefore does not require ethical review approval. The study has been registered on the international prospective register of systematic reviews under registration number CRD42024549669.
Information sources and search strategy
Two authors (DJ and ZY) independently performed the abstract screen and full-text review, with disagreements resolved by consensus. Literature search was carried out in electronic databases as follows: PubMed, Cochrane Library, Embase, and China National Knowledge Infrastructure (CNKI). The search strategy was aimed at retrieving the most relevant items published up to the search date of this review and these articles were only included if they were published in English and Chinese. Aforementioned, the databases were searched electronically, and reference lists of relevant articles and review papers were also hand-searched to reduce the chances of missing out on the identifiable potential studies during the electronic search. We established a comprehensive search strategy to search for the studies regarding the diagnostic accuracy of mSEPT9 for primary liver cancer. In particular, the following keywords and search strings were employed: “methylated SEPT9”, “SEPT9 methylation”, “SEPT9”, “Septin9”, “Septin 9”, “serum”, “plasma”, “blood”, “liver cancer”, “liver tumour”, “liver carcinoma”, “liver neoplasms”, “hepatocellular tumour”, “hepatocellular carcinoma”, “hepatoma”. These terms were then combined using Boolean operators (AND, OR). Search strings used for each database are provided in Supplementary Table S1.
Inclusion and exclusion criteria
To be eligible for inclusion in the systematic review and meta-analysis, studies had to meet the following criteria: 1) evaluate the diagnostic performance of mSEPT9 for primary liver cancer (PLC) in adult human populations; 2) provide sufficient data to estimate sensitivity, specificity, and other diagnostic accuracy parameters, including true positive, false positive, true negative, and false negative values; 3) adopt histopathological or imaging as standard reference standard for diagnosis; 4) be accessible in full text, and 5) be published in a scientific journal. Exclusion criteria included: 1) reviews, case reports, letters, conference abstracts, and non-human studies; 2) articles that did not present original research data (e.g., editorials, commentaries); 3) Studies with unclear or inconsistent cut-off thresholds for mSEPT9, as variations in cut-off points could significantly impact diagnostic performance and limit comparability; 4) studies with populations that did not match the intended patient profile (e.g., non-PLC conditions, pediatric populations, or patients with other advanced liver diseases).
Data extraction
Data extraction was conducted by two authors (LQ and JC), independently, based on a predefined excel sheet, including the first author name, publication year, country, study period, study design, total number of patients, patient population characteristics (with reference to specific pathology such as HCC or ICC), mean age, gender distribution, diagnostic methods, methods for detecting SEPT9 methylation, test cut-off values, and the number of SEPT9 positive cases. We also extracted the true positives (TP), false positives (FP), false negatives (FN), and true negatives (TN) to calculate sensitivity, specificity, positive and negative likelihood ratios (PLR and NLR), and diagnostic scores.
Quality assessment
Two reviewers (LQ and JC) independently assessed quality and resolved disagreements by discussion or by consulting a third reviewer. All studies included in this systematic review and meta-analysis were evaluated by the Quality Assessment of Diagnostic Accuracy Studies-2 (QUADAS-2) tool. QUADAS-2 is a major methodological tool of accepted standard specifically for risk of bias and applicability concerns in diagnostic accuracy studies. The evaluation was divided by four principle fields: patient selection, index test, reference standard, and flow and timing. Risk of bias was assessed for each domain, and the first three domains were also evaluated for concerns regarding applicability to the research question.
Statistical analysis
The identification accuracy of liver cancers was evaluated via the statistical software Stata 16.0 (Stata Corp LLC) in this study. The summary receiver operating characteristic (SROC) curve provided a global measure of test effectiveness. For each study, the diagnostic odds ratio (DOR) was calculated. Heterogeneity was assessed by the I2 statistic, with an I2 value of less than 25% indicating mild heterogeneity, I2 value from 25% to 50% indicating moderate heterogeneity, and an I2 value of more than 50% indicating significant heterogeneity. A fixed-effects model using the Mantel-Haenszel method was applied in cases of mild heterogeneity, while a random-effects model utilizing the DerSimonian and Laird method was employed for greater heterogeneity. Fagan’s nomogram was utilized to translate likelihood ratios into clinically relevant post-test probabilities. Publication bias was evaluated using Deeks’ funnel plot asymmetry test. Additionally, a distribution scatter diagram visualized the spread of diagnostic accuracies across studies, and subgroup analyses explored the sources of heterogeneity based on study characteristics such as pathology, design of the study, test method and nationality. To further ensure the robustness of our findings, we conducted a sensitivity analysis, systematically excluding each study to observe changes in the overall effect estimate.
Results
Study selection
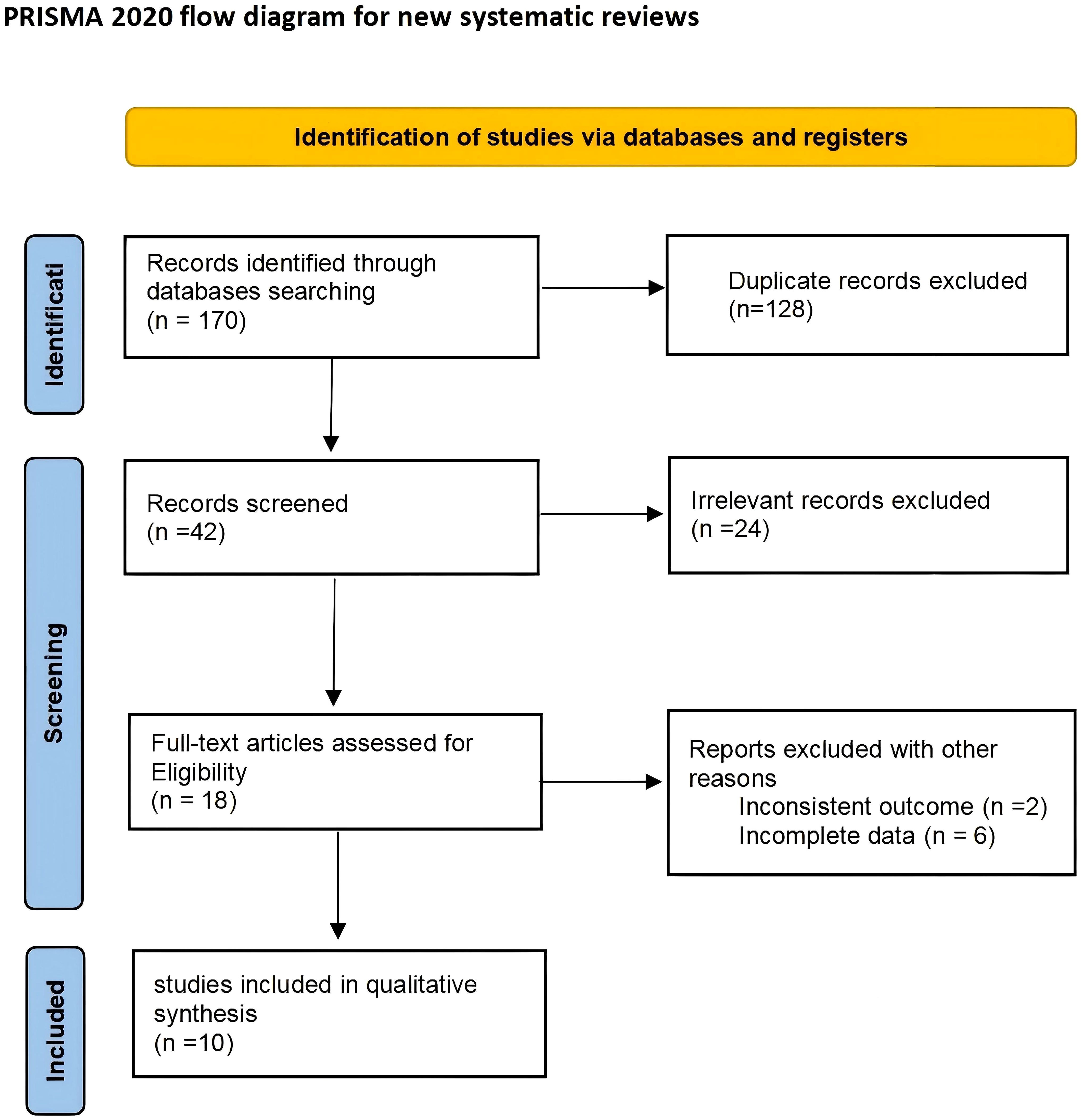
A total of 170 articles were identified in the initial search. 128 of these were excluded due to duplication or non-adherence to the inclusion criteria. Another search in the titles and abstracts provided 24 of the articles to be excluded. The full text of remaining 18 articles were further reviewed in which eight articles were excluded from the review as two studies failed to detail their diagnostic test and six did not have enough data to calculate the sensitivity and specificity. In total, 10 studies met the inclusion criteria for this meta-analysis (Figure 1).
Characteristics of included studies
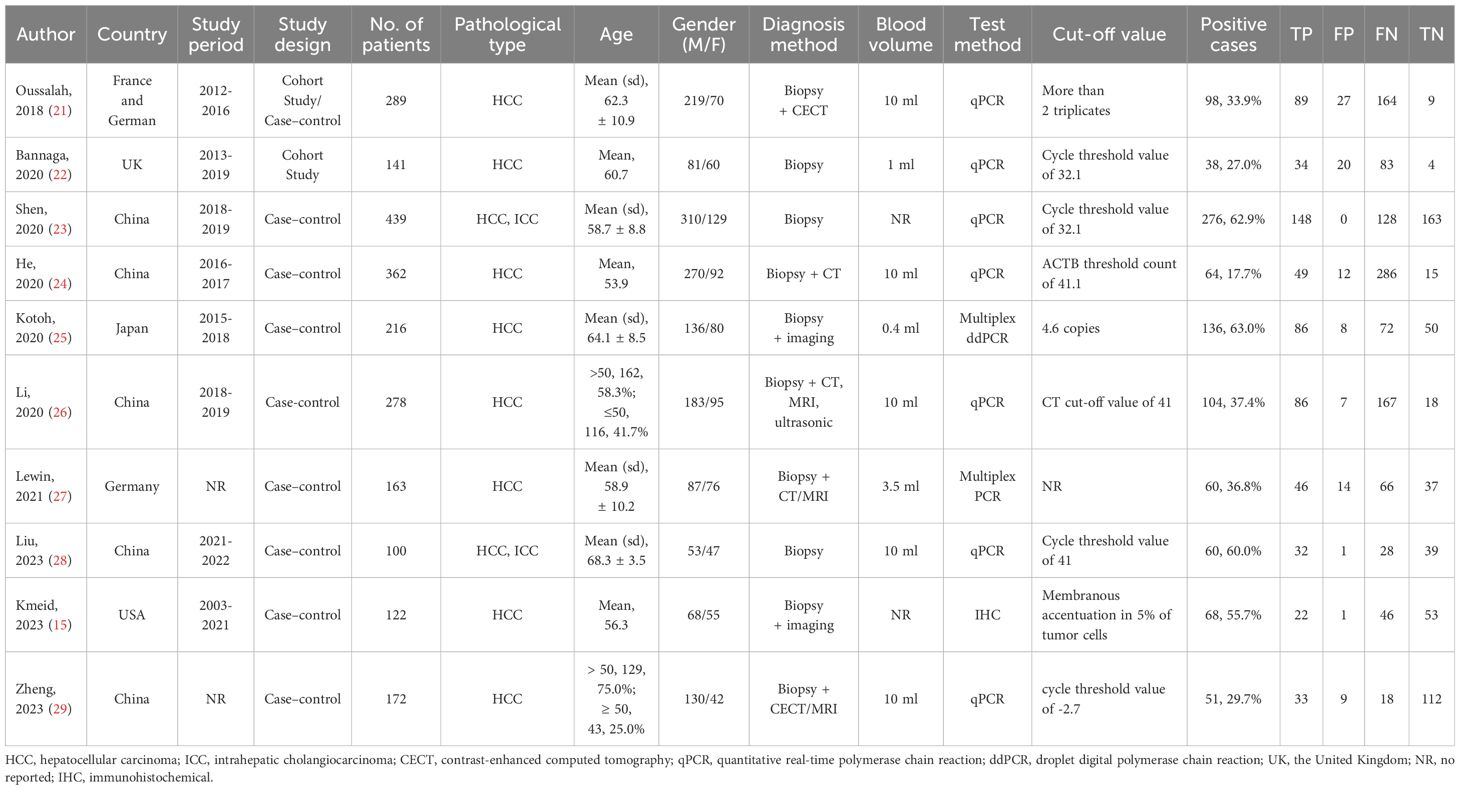
This systematic review covers 10 studies from 2012 to 2023 consisting of 2,182 participants with the majority derived from HCC and a smaller portion from ICC (13, 18–26). The dataset used in this study covered various countries, including France, Germany, the United Kingdom (UK), China, Japan, and the United States of America (USA). Among the included studies, 7 were case-control studies, 2 was a cohort study, and the remaining one study consisted of a cohort study (initial) and a case-control study (replication). The mean age of participants was from 53.9 to 68.3 years and gender distribution was also different between studies, most having a higher percentage of men. Main diagnostic methods of PLC include biopsy, CT, MRI, and ultrasound examination. The volume of blood samples collected from patients varies from 0.4 ml to 10 ml. In the detection of mSEPT9, seven studies employed quantitative polymerase chain reaction (qPCR), while the remaining three studies utilized multiplex droplet digital PCR (ddPCR), multiplex PCR, and IHC, respectively. There was wide variability in the cut-off values to define positive SEPT9 methylation cases, ranging from discernable cycle threshold values to the quantification of gene copies. The number and percentage of positive cases in each study varied from 38 to 276 and from 17.7% to 62.9%, which were demonstrated in Table 1.
Quality assessment
Supplementary Figure S1 displays the quality assessment results for all included studies. In the assessment of risk of bias, most studies exhibited low risk. However, some studies were unclear in the areas of “patient selection” (n=5), “index testing” (n=4), and “flow and time” (n=3). Meanwhile, applicability concerns were present for most studies toward the patient selection (n=8), index test (n=4), but not reference standard (n=8).
Overall diagnostic value of mSEPT9 for primary liver cancer
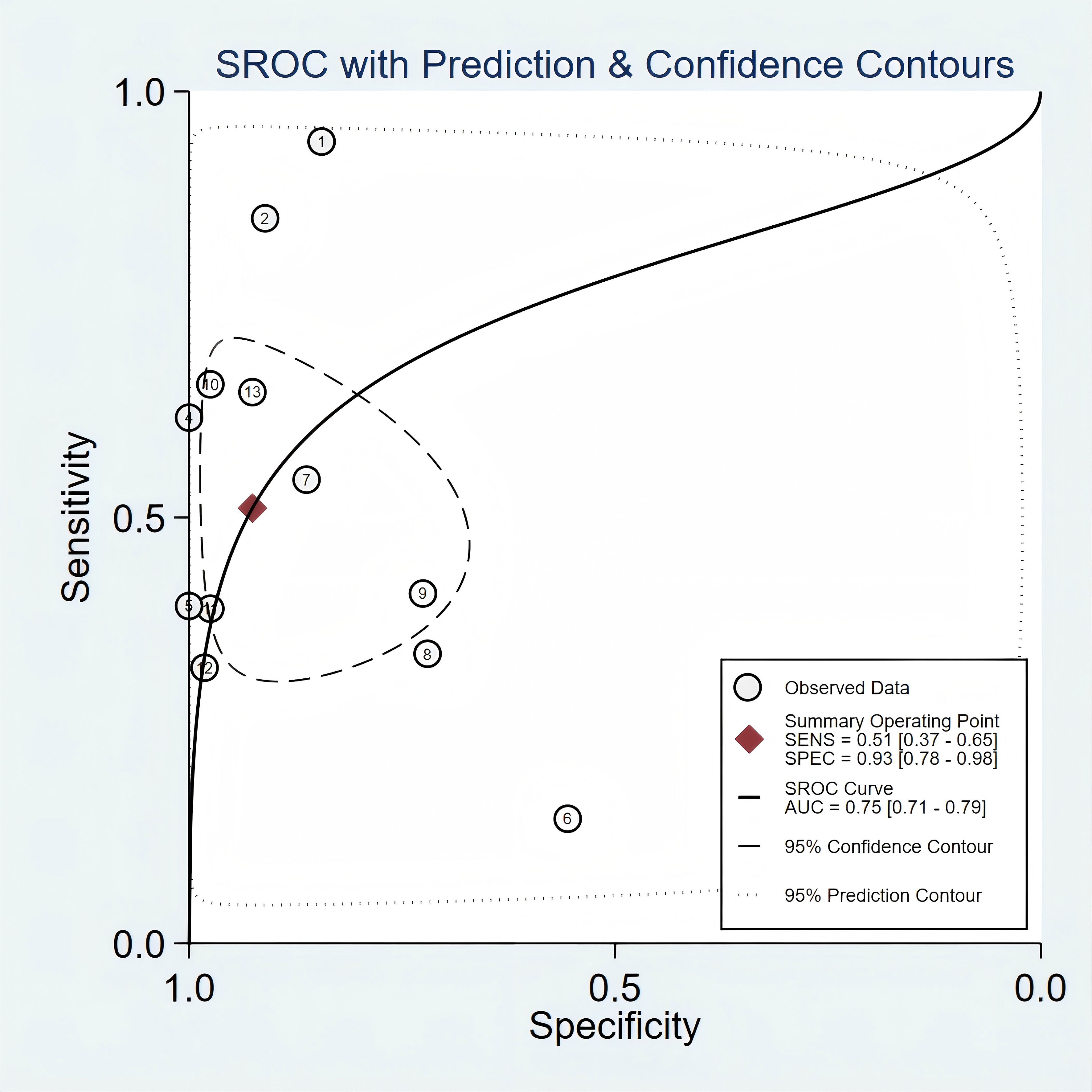
Figure 2 presented the forest plot depicting the pooled sensitivity and specificity of mSEPT9 in diagnosing primary liver cancer, which were 0.51 (95% confidence interval (CI): 0.37–0.65) and 0.93 (95% CI: 0.78–0.98), respectively. Significant heterogeneity was observed among the included studies, with I² values of 96.06 (95% CI: 94.81–97.32) for sensitivity and 94.70 (95% CI: 92.84–96.55) for specificity. Additionally, PLR, NLR, diagnostic score, and DOR were reported as 6.86 (95% CI: 1.96–24.03), 0.53 (95% CI: 0.38–0.74), 2.56 (95% CI: 1.07–4.06), and 12.97 (95% CI: 2.91–57.84), respectively, as illustrated in Figure 3. The area under the curve (AUC) for mSEPT9 and primary liver cancer was 0.75 (95% CI: 0.71–0.79), shown in Figure 4. The Spearman correlation coefficient of -0.311 (p = 0.301) indicated that there was no threshold effect in the analysis.
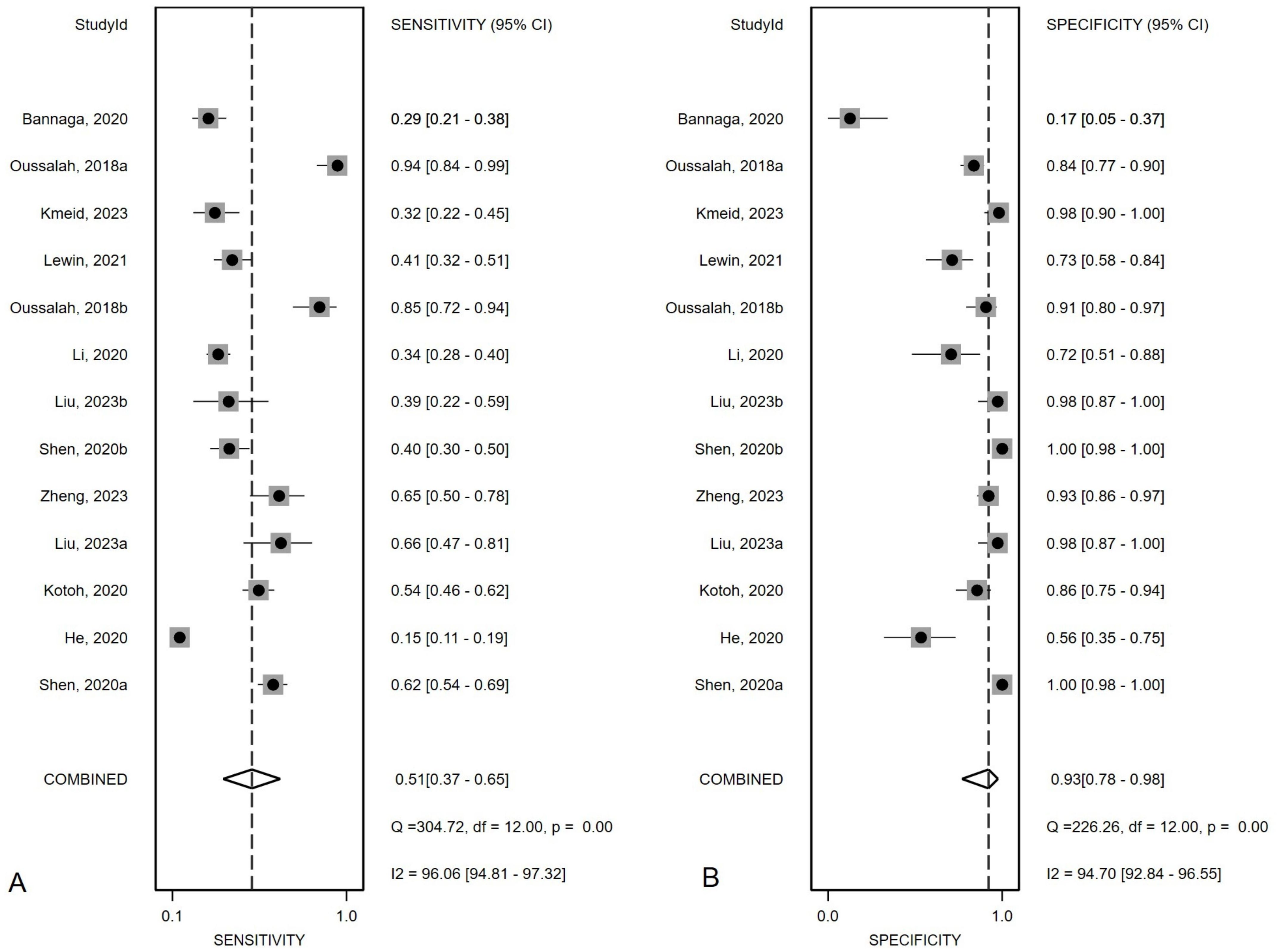
Figure 2. Summary sensitivity (A) and specificity (B) plotted on forest graphs for mSEPT9 in diagnosis for primary liver cancer.
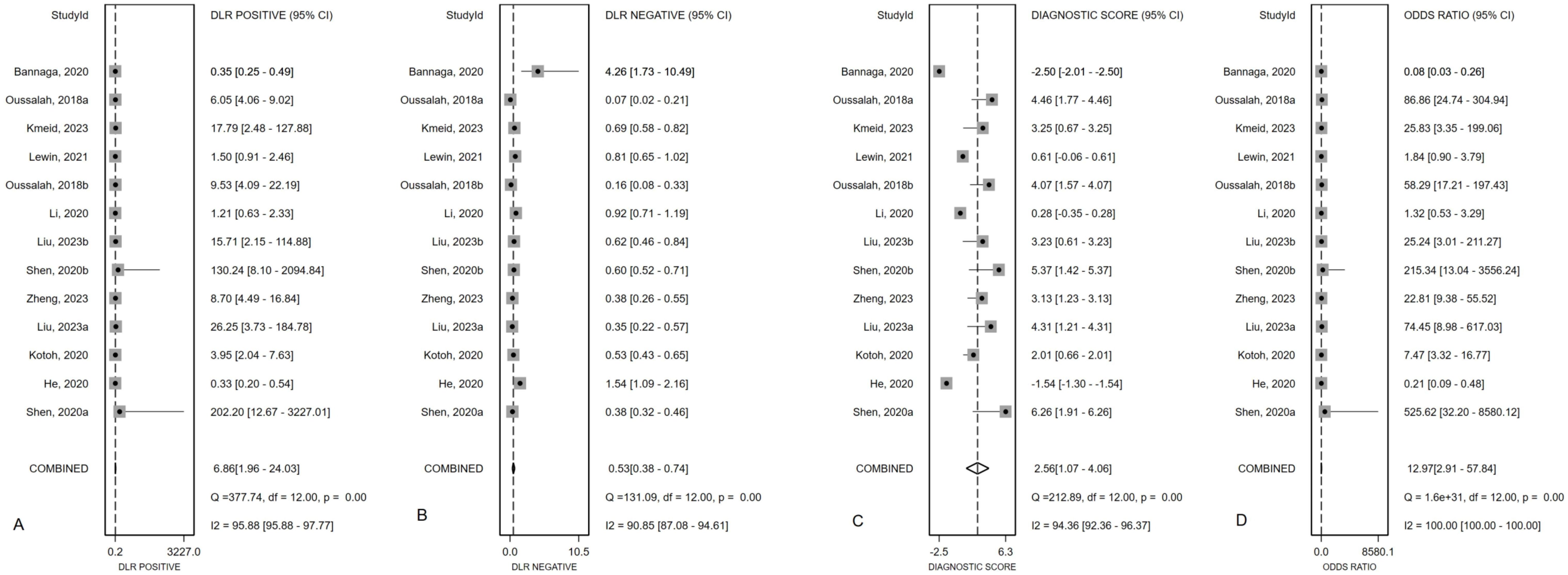
Figure 3. Summary PLR (A), NLR (B), diagnostic score (C) and DOR (D) plotted on forest graphs for mSEPT9 in diagnosis for primary liver cancer.
Subgroup analyses
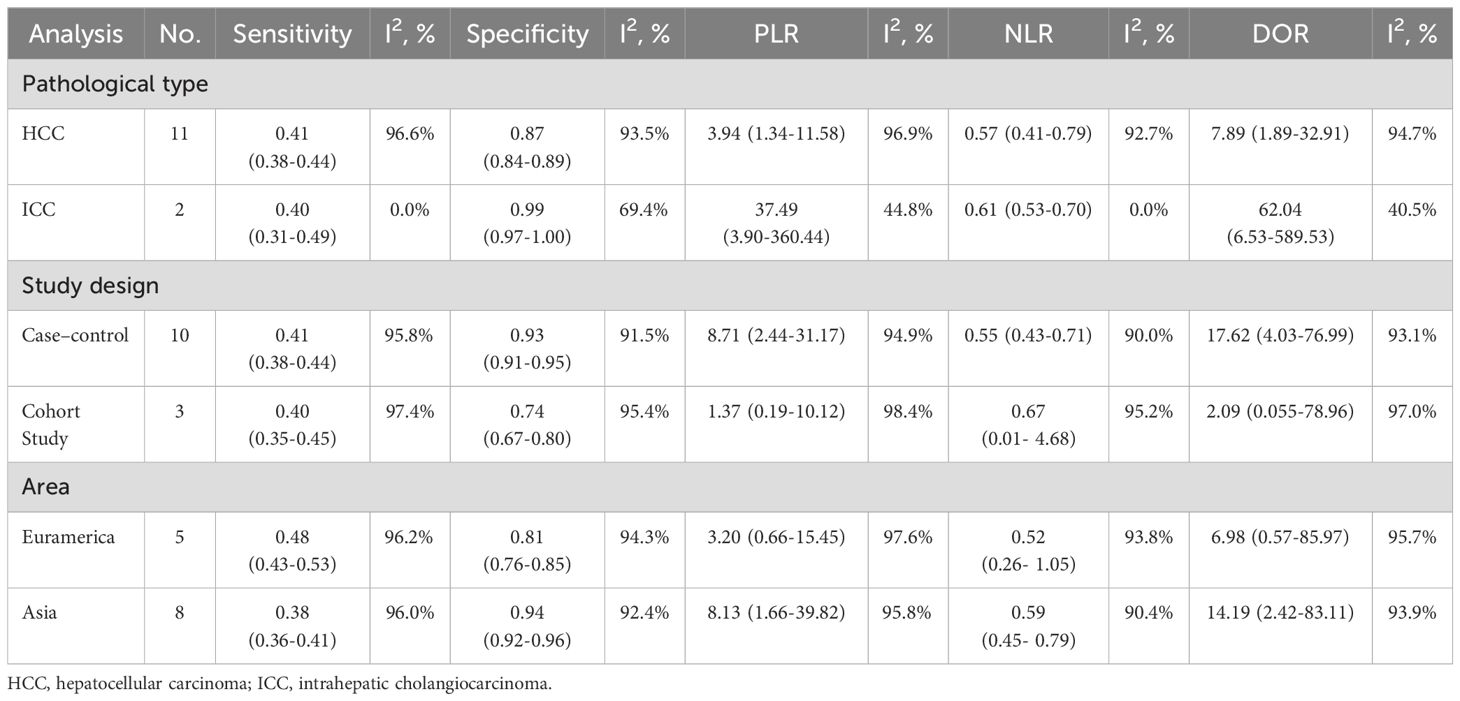
Subgroup analyses of diagnostic test performance metrics by pathological type, study design, geographic area, and test method (Table 2). For pathological types, mSEPT9 testing demonstrated distinct diagnostic performances in HCC and ICC. In HCC patients, mSEPT9 showed a sensitivity of 0.41 (95% CI: 0.38-0.44) and a specificity of 0.87 (95% CI: 0.84-0.89), with a DOR of 7.89. This performance is characterized by high heterogeneity (I² = 96.6%). On the other hand, ICC displayed a slightly lower sensitivity at 0.40 (95% CI: 0.31-0.49), but a notably higher specificity of 0.99 (95% CI: 0.97-1.00). The DOR for ICC was exceptionally high at 62.04, accompanied by substantially lower heterogeneity. Regarding study design, case-control studies showed a sensitivity of 0.41 (95% CI: 0.38-0.44) and a higher specificity of 0.93 (95% CI: 0.91-0.95), with a DOR of 17.62. Cohort studies displayed a lower sensitivity of 0.40 (95% CI: 0.35-0.45) and specificity of 0.74 (95% CI: 0.67-0.80), with a DOR of 2.09. Geographically, studies conducted in Asia demonstrated a sensitivity of 0.38 (95% CI: 0.36-0.41) and higher specificity of 0.94 (95% CI: 0.92-0.96), resulting in a DOR of 14.19. Conversely, studies from Euramerican regions showed a sensitivity of 0.48 (95% CI: 0.43-0.53) and a lower specificity of 0.81 (95% CI: 0.76-0.85), with a DOR of 6.98. For test method, qPCR has a sensitivity of 0.40 (0.37- 0.42) and a specificity of 90.90 (0.88- 0.92). Other methods reported a sensitivity of 0.46 (0.40- 0.51) and a specificity of 0.86 (0.80- 0.91), showing slightly higher sensitivity but lower specificity than qPCR. The DOR further underscores the effectiveness, with qPCR at 13.03, significantly higher than 5.64 for other methods.
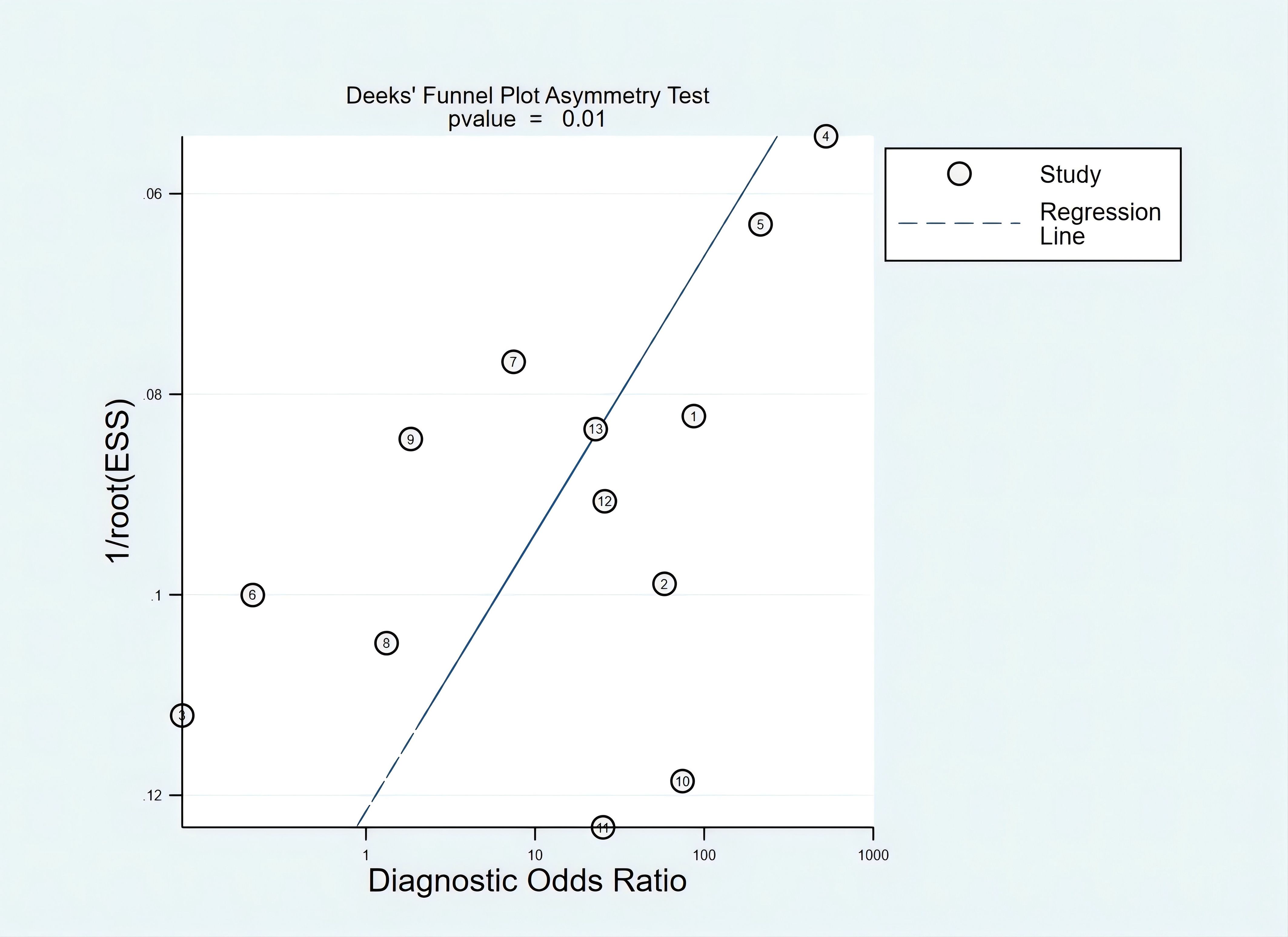
Publication bias
The asymmetry in the plot, as indicated by several studies deviating significantly from the regression line, was statistically significant, with a p-value of 0.01. This suggested the presence of publication bias (Figure 5).
Clinical diagnostic value
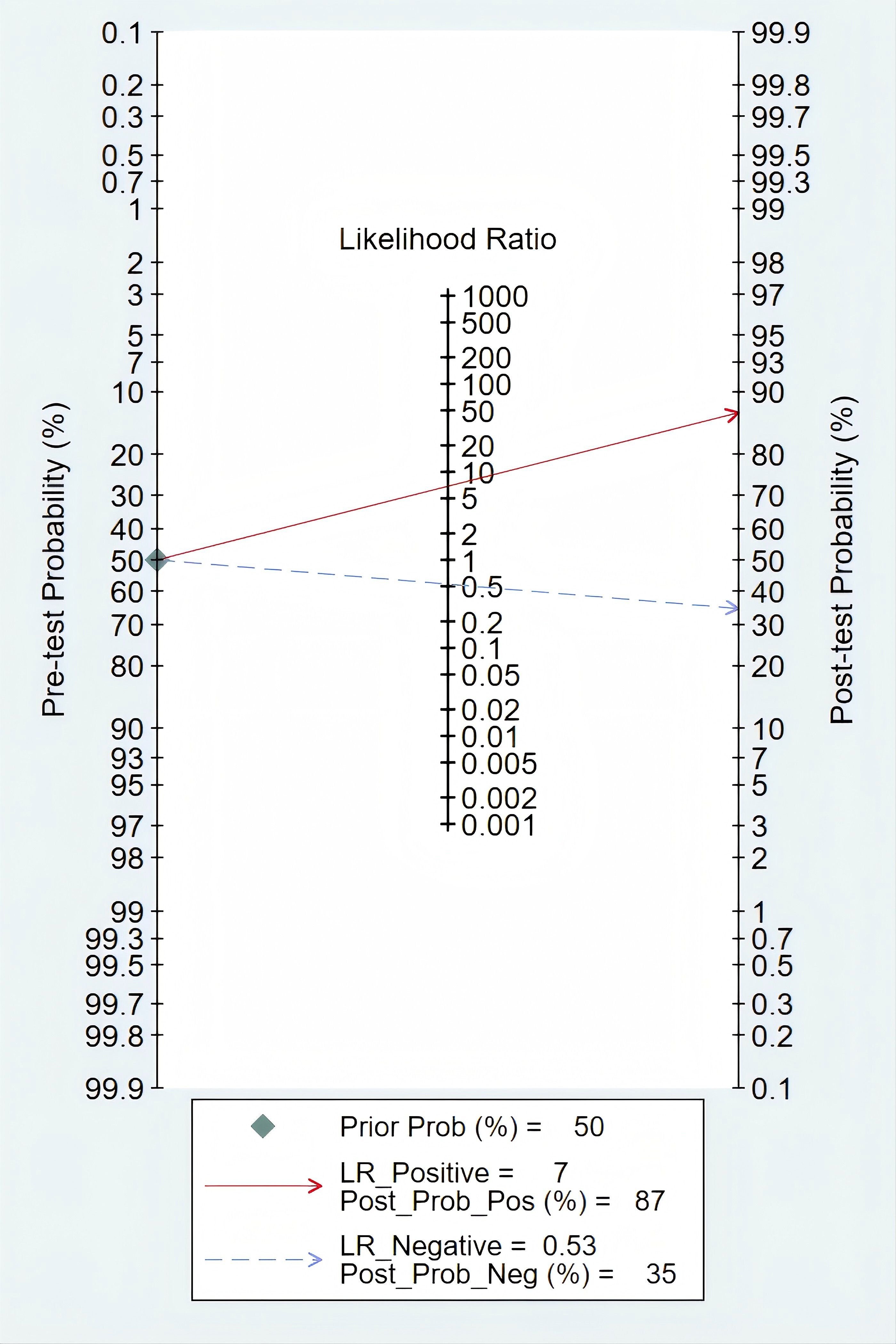
According to Fagan’s nomogram, our findings indicated that a positive mSEPT9 test result increased the post-test probability of an individual having PLC to approximately 87%, assuming a pre-test probability of 50%. This suggested that if a patient tested positive for PLC using mSEPT9, the likelihood that they truly had the disease was significantly elevated to 87%. Conversely, a negative test result reduced the post-test probability to around 35%, indicating that a negative mSEPT9 test could effectively rule out the disease with a high degree of certainty (Figure 6).
In addition, we constructed a likelihood ratio scattergram, evaluating the clinical applicability of mSEPT9 in the diagnosis of PLC (Figure 7). The summary likelihood ratios of the mSEPT9 test are located in the lower right quadrant, indicating that mSEPT9 did not reach the pathological standard for exclusion and confirmation, thus limiting its clinical utility.
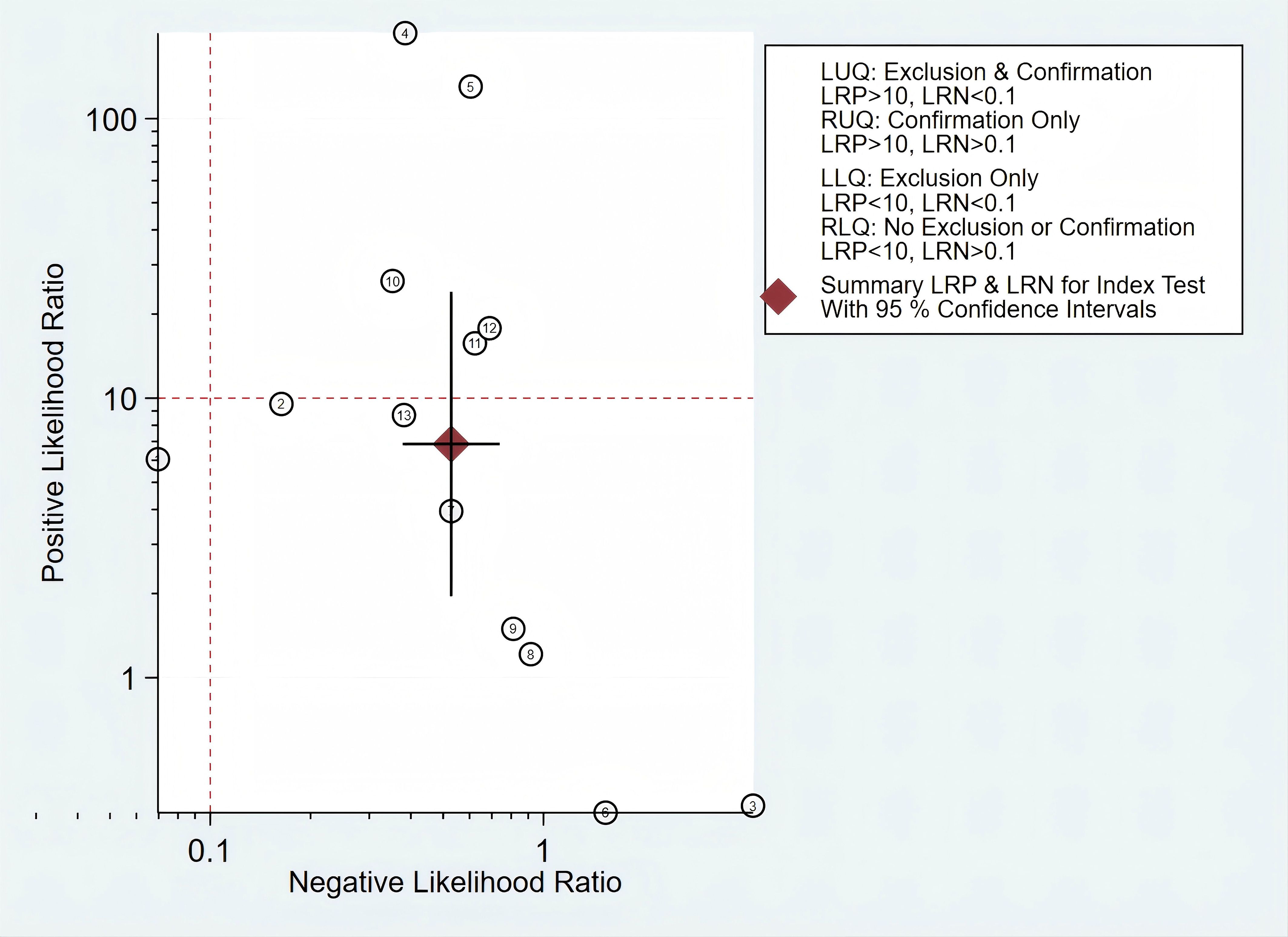
Figure 7. The likelihood ratio scattergram showing the different clinical significances of mSEPT9 in primary liver cancer.
Sensitivity analyses
Based on the sensitivity analysis, the overall effect estimates showed minimal changes when each study was sequentially excluded, indicating that the meta-analysis results are stable (Supplementary Figure S2).
Discussion
This analysis summarized the diagnostic performance of mSEPT9 for liver cancer, emphasizing its moderate sensitivity and high specificity. While mSEPT9 demonstrated strong performance in excluding the disease, it was not sufficient on its own to detect all positive cases within the patient cohort. Therefore, combining mSEPT9 with other diagnostic methods, such as imaging techniques or additional biomarkers, was essential to improve the detection rate of positive cases. We observed substantial heterogeneity in diagnostic metrics across studies, suggesting potential variability in test application or differences in study populations. The presence of publication bias, as suggested by Deeks’ funnel plot, indicated that smaller studies with potentially overestimated effects might have been more likely to be published. These results underscored the potential utility of mSEPT9 in liver cancer diagnostics but also emphasized the need for careful consideration of the test’s limitations and the contexts of its use.
DNA methylation is a critical component of the complex epigenetic transcriptional regulation system that is often altered in cancer, leading to hypermethylation in promoter sequences and hypomethylation across genomic sequences (27). SEPT9 gene methylation has emerged as a significant biomarker in oncology, primarily due to its role in regulating gene expression through epigenetic mechanisms (28). Some studies suggest that mSEPT9 demonstrates diagnostic potential in various types of cancer. A meta-analysis including 19 case-control studies assessed the diagnostic performance of mSEPT9 in early colorectal cancer (CRC) screening, finding that mSEPT9 testing has high specificity (92%) and moderate sensitivity (69%) (29). In recent years, researchers have also begun to focus on the diagnostic efficacy of mSEPT9 in breast cancer patients, finding that it has higher specificity and lower sensitivity, with diagnostic accuracy slightly superior to that of carcinoembryonic antigen, cancer antigen 153, and cytokeratin 19 (30). In addition, Powrózek et al. evaluated plasma mSEPT9 in 70 cancer patients and 100 healthy individuals, finding a sensitivity of 44.3%, specificity of 92.3%, and a positive predictive value of 91.2%. They observed higher methylation rates in non-small cell lung cancer and squamous cell carcinoma compared to small cell lung cancer (31). mSEPT9 has emerged as a promising biomarker for cancer diagnosis, and is associated with clinical features such as TNM staging, tumor size, and overall survival time (32).
However, despite its potential, the diagnostic performance of mSEPT9 for liver cancer remains unclear. SEPT9 is typically hypermethylated in liver cancer, which suppresses its expression. This gene plays a critical role in liver functions, such as the development of lipid droplets associated with cirrhosis and non-alcoholic steatohepatitis (NASH), as well as in cellular processes like apoptosis and hepatic stellate cell activation, which are crucial for liver fibrogenesis and carcinogenesiss (19). Liquid biopsy analysis, including circulating cfDNA analysis, offers the opportunity to detect DNA methylation markers such as SEPT9 in a non-invasive manner for patients with liver disease (33). Given that previous studies have highlighted unsatisfactory sensitivity, advancements in testing methodologies have led to the development of more sensitive assays, such as co-amplification at lower denaturation temperature-polymerase chain reaction (COLD-PCR), which improves detection efficiency without the need for bisulfite treatment of DNA and can detect extremely low levels of methylated DNA (34). Furthermore, the combination of mSEPT9 and other biomarkers (such as AFP) has been emphasized in recent research, not only to increase sensitivity for malignant tumor diagnosis but also to predict overall survival, microvascular invasion, and tumor proliferation (26). Importantly, a model to predict outcomes in HCC patients treated with molecular targeted agents, combining mSEPT9 with clinical parameters, demonstrated good predictive ability (35, 36). Previous studies have shown that mSEPT9, along with a panel including other epigenetic markers such as RASSF1A and other methylation gene profiles, was able to improve sensitivity and specificity (37, 38). We also observed a multiplex marker panel, including SEPT9 and multiple HCC-specific markers, as a training panel that can identify HCC-specific methylation patterns with high sensitivity using next-generation sequencing (24). Besides, the combination of multi-omics approaches incorporating genomics, epigenomics, transcriptomics, proteomics, and metabolomics has been highlighted as essential for advancing liver cancer screening. These comprehensive strategies may remedy the limitations of single-marker tests for HCC by considering the intricate molecular landscape of the disease, enabling a more detailed and precise diagnosis (39–41). Of note, some of the trials included in our study suggested that mSEPT9 performed better in detecting advanced liver cancer compared to early-stage detection (18, 21, 26). Clearly, these studies indicated the need to develop new techniques and methods for mSEPT9 detection, especially focusing on enhancing its performance in the early stages of cancer.
There were several limitations that should be noted. First, the studies included in this analysis had bias risks in certain areas, particularly in patient selection, which may have impacted the results. Second, the included studies exhibited variations in interventions, blood volume, and measuring method, which may limit the generalizability of the results. Third, not all studies in the meta-analysis provided the specific cutoffs used for classifying SEPT9 marker-based outcomes. Such heterogeneity in these measurements might be associated with the varying diagnostic accuracy as reported by these different studies, simply because the different cutoff values led to different sensitivity and specificity outcomes. Fourth, HCC and ICC have distinct pathological and molecular profiles, and their amalgamation might have obscured specific diagnostic insights that could be unique to each. Fifth, although SEPT9 methylation was a biomarker for liver cancer, it may have also been positive in other gastrointestinal cancers such as colorectal, gastric, and esophageal cancers. Studies often selected control groups without these cancers to avoid interference, which limited its real-world applicability. Finally, Deeks’ funnel plot suggested publication bias, indicating that smaller studies with potentially exaggerated effects were more likely to be published, which could have led to an overestimation of mSEPT9’s sensitivity; future larger, multicenter trials should prioritize publishing negative or inconclusive results to reduce this bias.
Conclusions
This meta-analysis showed that mSEPT9 testing has high specificity and moderate sensitivity for detecting primary liver cancer, with significant variability in performance metrics across different subgroups. This variability limited its use as an independent screening tool in clinical settings. Future research is urgently needed to enhance the sensitivity of mSEPT9 and to explore its integration with other diagnostic tools, aiming to improve its cancer screening efficacy.
Data availability statement
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
Author contributions
DJ: Methodology, Project administration, Validation, Writing – original draft, Data curation. LQ: Data curation, Formal analysis, Investigation, Writing – review & editing. JC: Software, Visualization, Writing – review & editing. ZY: Project administration, Supervision, Writing – review & editing. JD: Conceptualization, Funding acquisition, Writing – review & editing, Resources.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. Zhejiang Medical and Health Science and Technology Project (2023KY1298, 2024KY517). Zhoushan Medical and Health Science and Technology Project (2022RC01).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fendo.2025.1434174/full#supplementary-material
Supplementary Figure 1 | Quality assessment result of the included studies.
Supplementary Figure 2 | Results of sensitivity analysis.
References
1. Global Burden of Disease Cancer Collaboration, Fitzmaurice C, Abate D, Abbasi N, Abbastabar H, Abd-Allah F, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2017: A systematic analysis for the global burden of disease study. JAMA Oncol. (2019) 5:1749–68. doi: 10.1001/jamaoncol.2019.2996
2. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. (2018) 68:394–424. doi: 10.3322/caac.21492
3. Zhou J, Sun H, Wang Z, Cong W, Wang J, Zeng M, et al. Guidelines for the diagnosis and treatment of hepatocellular carcinoma (2019 edition). Liver Cancer. (2020) 9:682–720. doi: 10.1159/000509424
4. Maravelia P, Silva DN, Rovesti G, Chrobok M, Stål P, Lu YC, et al. Liquid biopsy in hepatocellular carcinoma: opportunities and challenges for immunotherapy. Cancers (Basel). (2021) 13:4334. doi: 10.3390/cancers13174334
5. Luo H, Wei W, Ye Z, Zheng J, Xu RH. Liquid biopsy of methylation biomarkers in cell-free DNA. Trends Mol Med. (2021) 27:482–500. doi: 10.1016/j.molmed.2020.12.011
6. Pelizzaro F, Cardin R, Penzo B, Pinto E, Vitale A, Cillo U, et al. Liquid biopsy in hepatocellular carcinoma: where are we now? Cancers (Basel). (2021) 13:2274. doi: 10.3390/cancers13092274
7. Lehrich BM, Zhang J, Monga SP, Dhanasekaran R. Battle of the biopsies: Role of tissue and liquid biopsy in hepatocellular carcinoma. J Hepatol. (2024) 80:515–30. doi: 10.1016/j.jhep.2023.11.030
8. Yan S, Liu Z, Yu S, Bao Y. Diagnostic value of methylated septin9 for colorectal cancer screening: A meta-analysis. Med Sci Monit. (2016) 22:3409–18. doi: 10.12659/MSM.900590
9. Tóth K, Sipos F, Kalmár A, Patai AV, Wichmann B, Stoehr R, et al. Detection of methylated SEPT9 in plasma is a reliable screening method for both left- and right-sided colon cancers. PLoS One. (2012) 7:e46000. doi: 10.1371/journal.pone.0046000
10. Bergheim J, Semaan A, Gevensleben H, Groening S, Knoblich A, Dietrich J, et al. Potential of quantitative SEPT9 and SHOX2 methylation in plasmatic circulating cell-free DNA as auxiliary staging parameter in colorectal cancer: a prospective observational cohort study. Br J Cancer. (2018) 118:1217–28. doi: 10.1038/s41416-018-0035-8
11. Song L, Chen Y, Gong Y, Wan J, Guo S, Liu H, et al. Opportunistic screening and survival prediction of digestive cancers by the combination of blood mSEPT9 with protein markers. Ther Adv Med Oncol. (2020) 12:1758835920962966. doi: 10.1177/1758835920962966
12. Lee W, Li X, Chandan VS. Hepatocellular carcinomas can be Special AT-rich sequence-binding protein 2 positive: an important diagnostic pitfall. Hum Pathol. (2020) 105:47–52. doi: 10.1016/j.humpath.2020.09.001
13. Kmeid M, Park YN, Chung T, Pacheco RR, Arslan ME, Lee H. SEPT9 expression in hepatic nodules: an immunohistochemical study of hepatocellular neoplasm and metastasis. Appl Immunohistochem Mol Morphol. (2023) 31:278–87. doi: 10.1097/PAI.0000000000001112
14. Long J, Chen P, Lin J, Bai Y, Yang X, Bian J, et al. DNA methylation-driven genes for constructing diagnostic, prognostic, and recurrence models for hepatocellular carcinoma. Theranostics. (2019) 9:7251–67. doi: 10.7150/thno.31155
15. Gonçalves E, Gonçalves-Reis M, Pereira-Leal JB, Cardoso J. DNA methylation fingerprint of hepatocellular carcinoma from tissue and liquid biopsies. Sci Rep. (2022) 12:11512. doi: 10.1038/s41598-022-15058-0
16. Peterse EFP, Meester RGS, de Jonge L, Omidvari AH, Alarid-Escudero F, Knudsen AB, et al. Comparing the Cost-Effectiveness of Innovative Colorectal Cancer Screening Tests [published correction appears in J Natl Cancer Inst. 2022 Jul 11;114(7):1050]. J Natl Cancer Inst. (2021) 113:154–61. doi: 10.1093/jnci/djaa103
17. Moher D, Liberati A, Tetzlaff J, Altman DG. PRISMA Group Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Br Med J. (2009) 339:b2535. doi: 10.1371/journal.pmed.1000097
18. Oussalah A, Rischer S, Bensenane M, Conroy G, Filhine-Tresarrieu P, Debard R, et al. Plasma mSEPT9: A novel circulating cell-free DNA-based epigenetic biomarker to diagnose hepatocellular carcinoma. EBioMedicine. (2018) 30:138–47. doi: 10.1016/j.ebiom.2018.03.029
19. Bannaga AS, Alvarez R, Zhou L, Petchey M, Noufaily A, Hitchins MP, et al. Role of methylated septin 9 as an adjunct diagnostic and prognostic biomarker in hepatocellular carcinoma. HPB (Oxford). (2021) 23:1595–606. doi: 10.1016/j.hpb.2021.03.015
20. Shen ZM, Feng J, Wei J, Yan GT. The clinical value of Septin9 methylation in the differential diagnosis of liver cancer. Int J Lab Med. (2020) 41:2831–8. doi: 10.3969/j.issn.1673-4130.2020.23.004
21. He N, Feng G, Zhang C, Wu F, Zhang T, Yang Y. Plasma levels of methylated septin 9 are capable of detecting hepatocellular carcinoma and hepatic cirrhosis. Mol Med Rep. (2020) 22:2705–14. doi: 10.3892/mmr.2020.11356
22. Kotoh Y, Suehiro Y, Saeki I, Hoshida T, Maeda M, Iwamoto T, et al. Novel liquid biopsy test based on a sensitive methylated SEPT9 assay for diagnosing hepatocellular carcinoma. Hepatol Commun. (2020) 4:461–70. doi: 10.1002/hep4.1469
23. Li B, Huang H, Huang R, Zhang W, Zhou G, Wu Z, et al. SEPT9 gene methylation as a noninvasive marker for hepatocellular carcinoma. Dis Markers. (2020) 2020:6289063. doi: 10.1155/2020/6289063
24. Lewin J, Kottwitz D, Aoyama J, deVos T, Garces J, Hasinger O, et al. Plasma cell free DNA methylation markers for hepatocellular carcinoma surveillance in patients with cirrhosis: a case control study. BMC Gastroenterol. (2021) 21:136. doi: 10.1186/s12876-021-01714-8
25. Liu H, Wei D, Yan Z, Cui B, Song Y, Yin H. Analyzing the role of septin9 gene methylation in the diagnosis and treatment of primary liver cancer in the elderly. Altern Ther Health Med. (2023) 29:194–9.
26. Zheng K, Dai L, Zhao Y, Li L, Li W, Zhang X, et al. Methylated SEPT9 combined with AFP and PIVKA-II is effective for the detection of HCC in high-risk population. BMC Gastroenterol. (2023) 23:260. doi: 10.1186/s12876-023-02900-6
27. Dawson MA, Kouzarides T. Cancer epigenetics: from mechanism to therapy. Cell. (2012) 150:12–27. doi: 10.1016/j.cell.2012.06.013
28. Sun J, Fei F, Zhang M, Li Y, Zhang X, Zhu S, et al. The role of mSEPT9 in screening, diagnosis, and recurrence monitoring of colorectal cancer. BMC Cancer. (2019) 19:450. doi: 10.1186/s12885-019-5663-8
29. Hariharan R, Jenkins M. Utility of the methylated SEPT9 test for the early detection of colorectal cancer: a systematic review and meta-analysis of diagnostic test accuracy. BMJ Open Gastroenterol. (2020) 7:e000355. doi: 10.1136/bmjgast-2019-000355
30. Wang C, Qi J, He T, Li J, Zhang CC. Value of peripheral blood septin9 gene methylation in the diagnosis of breast cancer and evaluation of chemotherapy efficacy. Chin J Health Care Med. (2021) 23:228–30. doi: 10.3969/j.issn.1674-3245.2021.03.006
31. Powrózek T, Krawczyk P, Kucharczyk T, Milanowski J. Septin 9 promoter region methylation in free circulating DNA-potential role in noninvasive diagnosis of lung cancer: preliminary report. Med Oncol. (2014) 31:917. doi: 10.1007/s12032-014-0917-4
32. Yang X, Xu ZJ, Chen X, Zeng SS, Qian L, Wei J, et al. Clinical value of preoperative methylated septin 9 in Chinese colorectal cancer patients. World J Gastroenterol. (2019) 25:2099–109. doi: 10.3748/wjg.v25.i17.2099
33. Arechederra M, Recalde M, Gárate-Rascón M, Fernández-Barrena MG, Ávila MA, Berasain C. Epigenetic biomarkers for the diagnosis and treatment of liver disease. Cancers (Basel). (2021) 13:1265. doi: 10.3390/cancers13061265
34. Rashid S, Sun Y, Ali Khan Saddozai U, Hayyat S, Munir MU, Akbar MU, et al. Circulating tumor DNA and its role in detection, prognosis and therapeutics of hepatocellular carcinoma. Chin J Cancer Res. (2024) 36:195–214. doi: 10.21147/j.issn.1000-9604.2024.02.07
35. Huang F, Yang G, Jiang H, Chen X, Yang Y, Yu Q, et al. Role of plasma methylated SEPT9 for predicting microvascular invasion and tumor proliferation in hepatocellular carcinoma. Technol Cancer Res Treat. (2022) 21:15330338221144510. doi: 10.1177/15330338221144510
36. Saeki I, Suehiro Y, Yamauchi Y, Hoshida T, Tanabe N, Oono T, et al. Methylated SEPT9 assay-based liquid biopsy as a biomarker in molecular targeted agent-treated hepatocellular carcinoma. Hepatol Int. (2023) 17:1289–99. doi: 10.1007/s12072-023-10488-y
37. Müller D, Győrffy B. DNA methylation-based diagnostic, prognostic, and predictive biomarkers in colorectal cancer. Biochim Biophys Acta Rev Cancer. (2022) 1877:188722. doi: 10.1016/j.bbcan.2022.188722
38. Qian T, Zhou Z, Zhang Q, Liou YL, Zhou H. SEPT9, H4C6, and RASSF1A methylation in nasopharyngeal swabs: A reflection of potential minimally invasive biomarkers for early screening of nasopharyngeal cancer. Med (Baltimore). (2023) 102:e36583. doi: 10.1097/MD.0000000000036583
39. Chen F, Wang J, Wu Y, Gao Q, Zhang S. Potential biomarkers for liver cancer diagnosis based on multi-omics strategy. Front Oncol. (2022) 12:822449. doi: 10.3389/fonc.2022.822449
40. Dai WC, Fan R, Sun AH, He FC, Hou JL. Multi-omics research contributes to early screening, diagnosis and treatment of liver cancer. Chin J hepatology. (2022) 30:793–6. doi: 10.3760/cma.j.cn501113-20220628-00357
Keywords: diagnostic performance, methylation, SEPT9, primary liver cancer, meta-analysis
Citation: Jin D, Qian L, Chen J, Yu Z and Dong J (2025) Diagnostic accuracy of methylated SEPT9 for primary liver cancer: a systematic review and meta-analysis. Front. Endocrinol. 16:1434174. doi: 10.3389/fendo.2025.1434174
Received: 31 May 2024; Accepted: 23 January 2025;
Published: 13 February 2025.
Edited by:
Lele Song, Eighth Medical Center of the General Hospital of the Chinese People’s Liberation Army, ChinaReviewed by:
Guodong Zhao, Zhejiang University of Technology, ChinaRazvan Iacob, Fundeni Clinical Institute, Romania
Copyright © 2025 Jin, Qian, Chen, Yu and Dong. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jinliang Dong, RG9uZy5qbEBvdXRsb29rLmNvbQ==
 Danwen Jin1
Danwen Jin1 Ze Yu
Ze Yu Jinliang Dong
Jinliang Dong