
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Endocrinol. , 28 February 2025
Sec. Cardiovascular Endocrinology
Volume 16 - 2025 | https://doi.org/10.3389/fendo.2025.1405833
 Bahareh Amirkalali1
Bahareh Amirkalali1 Parvin Hassanzadeh1
Parvin Hassanzadeh1 Fatemeh Sheikholmolooki2
Fatemeh Sheikholmolooki2 Esmaeel Gholizadeh1
Esmaeel Gholizadeh1 Azam Doustmohammadian1
Azam Doustmohammadian1 Fahimeh Safarnezhad Tameshkel1
Fahimeh Safarnezhad Tameshkel1 Nima Motamed3
Nima Motamed3 Mansooreh Maadi1
Mansooreh Maadi1 Masoudreza Sohrabi1
Masoudreza Sohrabi1 Elham Sobhrakhshankhah1
Elham Sobhrakhshankhah1 Farhad Zamani1*†
Farhad Zamani1*† Hossein Ajdarkosh1*†
Hossein Ajdarkosh1*†Introduction: This study investigates the subclasses of metabolic syndrome (Mets) and their relationship with non-alcoholic fatty liver (NAFLD) and the probable predictive role of serum vitamin D and CRP levels.
Methods: This community-based, cross-sectional study was performed on adults in the framework of the Amol cohort prospective study (AmolCPS). Mets was defined as Adult Treatment Panel III criteria (ATP III) and ultrasound was used to diagnose NAFLD. Anthropometric and blood pressure measurements were conducted, and biochemical measurements were assessed after fasting. Data analysis included Latent class analysis, two-tailed χ2 statistics, one-way analysis of variance, and logistic regression using Mplus (version 7.4) and spss (version 26) softwares.
Results: The study involved 2308 participants, with a mean age of 43.17 ± 12.30 years. Mets prevalence was 25.64%, with three identified classes: Mets with Hypertension (HTN), Mets without HTN (Non-HTN), and Low Risk. Mets with HTN had a high probability of at least four components, particularly high SBP. Non-HTN had at least three high probable components, especially high TG and low HDL but not high SBP and DBP. The low-risk class had a low probability of all components except low HDL in women. Serum vitamin D and CRP levels did not significantly predict Mets classes in men, while CRP level significantly predicted the HTN class in women (OR:1.03, CI:1.004-1.067). Both HTN, and Non-HTN Mets classes significantly increased the odds of NAFLD compared to the low risk class, especially in women (HTN class OR: 4.20 vs 2.94; non-HTN class OR: 5.60 vs 3.12 in women and men respectively).
Conclusion: The latent class analysis in northern Iran identified three Mets classes: HTN, Non-HTN, and low-risk, with hypertension playing a crucial role in determining these classes. These classes were stronger predictors of NAFLD in women. Serum CRP and vitamin D levels did not emerge as significant predictors of the classes, except for serum CRP in the HTN class among women.
Metabolic syndrome is characterized by a cluster of interconnected metabolic abnormalities that elevate the risk of stroke, cardiovascular diseases (CVD), type 2 diabetes (T2D), non-alcoholic fatty liver disease (NAFLD), and various other health complications (1–3). The prevalence of metabolic syndrome in the global adult population is approximately 20-25% (4–6) and it continues to rise, posing a significant public health concern worldwide (1, 7). This upward trend has also been observed in Iran. In 2018, the prevalence of metabolic syndrome was estimated at 25.5% in women and 17.16% in men (8), which further increased to 34% in women and 22% in men by 2020 (9).
Metabolic syndrome has been described in various manners over the past decades, however, the metabolic abnormalities typically considered for its diagnosis consist of abdominal obesity (characterized by a large waist circumference or high waist-to-hip ratio), elevated fasting blood glucose, increased fasting serum insulin, reduced serum high-density cholesterol (HDL), elevated serum triglyceride levels (TG), and high blood pressure (10–13). In most definitions, the simultaneous presence of three or more of these components in an individual indicates the presence of metabolic syndrome, regardless of which specific three components are present. Previous research suggests that there may be distinct subgroups within this classification, with each subgroup potentially representing a different pathophysiologic state and carrying varying levels of risk for chronic diseases (14, 15).
Latent class analysis is a statistical method that enables the identification of distinct subgroups of metabolic syndrome (16) and reveals the alignment of metabolic syndrome components within a population (17). Latent class analysis utilizes a model-based approach to cluster individuals into different groups based on their responses to observed categorical variables, while also estimating latent variables from observed indicator variables (17). In Iran, several studies have utilized the Latent class analysis to investigate metabolic syndrome and its components (17–20). However, it is important to note that the prevalence of metabolic syndrome, its components, and its risk factors vary across geographical regions (21–23). Therefore, the identification of latent classes of metabolic syndrome within each specific area can greatly assist doctors, healthcare providers, and policymakers from the Health Ministry in developing tailored preventive programs for the residents of that particular region (15). Previous research has primarily focused on the relationship between latent classes of metabolic syndrome and CVD (14, 19, 24, 25) However, to the best of our knowledge, no studies have explored the association between latent classes of metabolic syndrome and NAFLD. This study also takes into consideration the role of serum vitamin D (26–29) and C-reactive protein (CRP) levels (30–32), two important factors associated with NAFLD, in determining the classification of individuals within the different metabolic syndrome subclasses. So, this study aims to investigate: 1) whether there are any subclasses of people with different profiles of metabolic syndrome components in the north of Iran, 2) whether there is a relationship between individual characteristics (such as age, BMI, serum vitamin D, and CRP level) and the placement of individuals in any of the subclasses, and 3) whether there is an association between the identified subclasses and NAFLD.
This community-based cross-sectional study was conducted in the framework of the Amol cohort prospective study (AmolCPS), the second phase. A detailed description of AmolCPS was explained in another article (33) but briefly, this study was conducted in Amol, a city in the northern region of Iran, in two different periods: 2009-2010 (phase 1) and 2016-2017 (phase 2). Study subjects were selected from 25 rural and 16 urban healthcare centers between the ages of 10 to 90. Then, the subjects were divided into 16 groups based on gender and age with ten-year intervals, 10-19, 20-29, 30-39, 40-49, 50-59, 60-69, 70-79, and 80-89. The selection of studies in each stratum was done using a simple randomization method, which was proportional to the population of each stratum.
The present study included all adults aged 18 to less than 65 years who had complete abdominal ultrasound and blood test results in the second phase of the study. Participants who followed a specific diet or exercise routine, had a history of liver disorders such as Wilson’s disease, autoimmune liver disease, hemochromatosis, viral infections, alcoholic fatty liver, malignancies, thyroid problems, autoimmune disorders, high alcohol consumption (more than 30 g/day in men and more than 20 g/day in women) (34, 35), were pregnant or breastfeeding were excluded from the study. Ultimately, 2308 individuals, comprising 1135 men and 1173 women, were included for analysis. The mean imputation method (replacing the missing values with the mean of the relevant variables) was used to address a total of 13 missing data points for weight and height.
This study has been approved by the ethics committee of Iran University of Medical Sciences by No. IR.IUMS.REC.1400.982 and was conducted according to the Helsinki Declaration. The participants entered the study after signing the informed consent form with full knowledge of the study objectives.
In AmolCPS, medical, pharmaceutical, and demographic information was collected by a standard questionnaire after signing the informed consent form (33). Data on physical activity were collected using a validated International Physical Activity Questionnaire (IPAQ), which was expressed as metabolic equivalent minutes per minute per week (MET-min/week) (36) Anthropometric measurements (weight (kg), height (m), and waist circumference (cm)) were done based on the standard protocol (37) and Body mass index (BMI) was calculated as dividing weight in kilograms by the square of height in meters. Blood pressure was measured in a quiet room after 15 minutes of rest with a mercury sphygmomanometer. The average of 2 times of measurement with an interval of 1 minute was considered as the systolic and diastolic blood pressure of the person.
Then, a venous blood sample was taken after 12 hours of fasting for biochemical measurements (including fasting plasma glucose (FPG), serum triglycerides (TG), high-density lipoprotein (HDL), C-reactive protein (CRP), 25(OH) vitamin D, Creatinine (Cr), Hemoglobin A1C (HbA1c)). According to the protocol using the BS200 Auto analyzer (Mindray, China), FPG, TG, Cr, and HDL were assessed enzymatically. The HbA1c level was measured by a Variant machine (Bio-Rad, Hercules, CA, United States). Serum concentration of 25 (OH) vitamin D was measured using an ELISA Kit (Pishtaz Teb Zaman Diagnostics, Tehran, Iran), and serum CRP level was quantitatively measured using a Bionic CRP kit, Tehran, Iran.
Each of the metabolic syndrome components was determined based on the definition of National Cholesterol Education Program Adult Treatment Panel III criteria (ATP III) as follows (38):
● FPG ≥100 mg/dl or drug treatment for elevated blood glucose
● Waist Circumference (WC) >102 cm in men or > 88 cm in women
● Serum TG ≥150 mg/dl or drug treatment for elevated triglycerides
● Serum HDL < 40 mg/dl in men or < 50 mg/dl in women or drug treatment for low HDL
● Blood Pressure ≥130/or ≥85 mm Hg or drug treatment for elevated blood pressure
Ultrasound was used to diagnose NAFLD. Sagittal, longitudinal, lateral, and intercostal views were obtained with a 3-5 MHz transducer (Esaote SpA, Genova, Italy). The normal liver was defined when the liver consistency was homogeneous, displayed fine-level echoes, and was minimally hyperechoic or even isoechoic in contrast to a regular renal cortex. Steatosis, on the other hand, was identified as a mild to severe increase in liver echogenicity, with severe cases exhibiting limited penetration of the posterior segment from the right hepatic lobe and poor or no visual images of hepatic vessels and diaphragm, in those without a history of excess alcohol consumption, drug-induced steatosis or viral and hereditary steatogenic hepatic conditions (39, 40). Ultrasound examinations were performed by a radiologist who was completely blind to the study protocol while the subjects were fasting.
The latent class analysis was used to analyze data. In this statistical procedure, different subgroups are qualitatively identified in a population that have some visible characteristics in common [in this study, six dichotomous observable variables of metabolic syndrome including high WC, high FPG, low HDL, high TG, High SBP, and high DBP, based on specific thresholds for metabolic syndrome definition (38)]. These subgroups are called latent classes (41). In the first step, for determination of the number of classes, the model is fitted with one class, and then in the next steps, another class is added to the model in each run. With the help of indices and tests (such as Akaike’s Information Criterion (AIC) and Bayesian Information Criterion (BIC), Le Mandel Rubin’s likelihood ratio test (LMR LR test), and entropy (Entropy)), the model with the appropriate number of classes is selected. In our study, individuals were assigned to latent classes based on their highest posterior probability of membership. Specifically, we used a threshold of 0.50 for class assignment, meaning that individuals with a probability greater than or equal to 0.50 were assigned to the corresponding class. Then we employed the BIC and LMR test as primary criteria for selecting the optimal number of classes. The BIC is a widely used criterion in model selection that balances model fit with complexity, penalizing models with more parameters to avoid overfitting. A lower BIC value indicates a better-fitting model. The LMR test, on the other hand, compares a k-class model to a k-1 class model by assessing whether the added complexity of a new class significantly improves the fit of the model. A significant result (p-value < 0.05) indicates that the k-class model provides a better fit than the k-1 class model. During our analysis, we examined multiple models ranging from one to five classes. For each class, the BIC was calculated. The BICs of the classes were compared and finally the class with the lowest BIC to avoid overfitting and underfitting and significant LMR LR test p-value (p-value < 0.05) was selected as the model. We considered gender differences in WC and serum HDL cutoffs in the definition of metabolic syndrome but used the same metrics, such as BIC, AIC, entropy, and the Lo-Mendell-Rubin test, for both sexes to be consistent in our analytical approach.
The Shapiro-Wilks test was utilized to assess the normality of continuous variables, and the assumption of normality for each variable included in the parametric analyses was specifically evaluated. Two-tailed χ2 statistics were utilized to explore differences for categorical variables. One-way analysis of variance (ANOVA) and student’s t-tests were applied to assess differences in continuous variables with a normal distribution, while non-parametric Kruskall-Wallis and Mann-Whitney tests were used to investigate differences for continuous variables without normal distribution. Additionally, Bonferroni post hoc tests were used for pairwise comparisons in the ANOVA Test, and adjusted Bonferroni post hoc tests were used in the Kruskal Wallis test. The association between subclasses of metabolic syndrome and other risk factors such as age, BMI, serum CRP, and 25(OH) vitamin D were evaluated using Multinomial Logistic Regression. Finally, logistic regression was used to analyze the relationship between metabolic syndrome subclasses and NAFLD adjusted for age, diabetes diagnosis, serum vitamin D, CRP, and Cr level. The low-risk class served as the reference group in regressions. Mplus (version 7.4) and spss (version 26) software were used for data analysis.
Figure 1 displays a flowchart outlining the criteria for inclusion and exclusion. This study included 2308 participants (men=49.17%) with a mean age of 43.17± 12.30 years. According to ATPIII criteria, the prevalence of metabolic syndrome was 25.64% in the study population with a significantly higher prevalence among women (33.3% vs. 17.7%, p<0.001). Women were significantly younger (42.58 ± 12.28 vs 43.78 ± 12.30, p=0.019), with higher BMI (29.68 ± 5.74 vs 26.54 ± 4.24, p<0.001), and higher prevalence of elevated WC (46.1% vs 8%, p<0.00) and SBP (17% vs 13.9%, p=0.043). Women also had significantly higher serum CRP levels (2.00 (1.00-4.90) vs 1.00 (0.40-2.50), p<0.001) and lower serum vitamin D (14.70 (8.70-22.67) vs 19.34 (14.00-24.40), p=0.001) and Cr (0.96 (0.87-1.04) vs 1.13 (1.04-1.25), p<0.001) levels. Low serum HDL level was significantly more prevalent in men (52.4% vs 29.8%, p<0.001). They also displayed a significantly higher level of physical activity and smoking habits but had a lower rate of diabetes diagnosis. HbA1c and other components of metabolic syndrome including high serum TG, FPG, and DBP were not significantly different between the two sexes (Table 1).
The number of latent classes, best fitted in the study population, was determined using the BIC value and LMR LR test p-value in each sex separately. In both sexes, the BIC value decreased by adding each class to the previous one until class 3. By adding class 4, the BIC value started increasing again in both sexes. LMR LR test p-values were also significant in class 3 in both men and women. According to the indicators and test results, the model with 3 classes had a better fit than other models for both sexes. The fit indices of the model are summarized in Table 2.
All participants were assigned to the class where they were most likely to have similar characteristics. One of the classes was named “metabolic syndrome with Hypertension (HTN)”, with a prevalence of 11.6% in men and 19.4% in women, showing a high probability for at least 4 components of metabolic syndrome, especially high SBP (men: 82.4%, women: 78.1%). The other class was called “ metabolic syndrome without HTN (Non-HTN)” which had a prevalence of 19.4% in men and 21.2% in women. In this class, at least 3 components of metabolic syndrome had high probability but high SBP and high DBP were not among those components. The two major prevalent components of metabolic syndrome in this group were high TG and low HDL in men and High WC and low HDL in women. In the “Low Risk” class, which included 68.9% of men and 59.3% of women, the probability of all components of metabolic syndrome were low except for low HDL in women. (Table 3) (Figure 2).
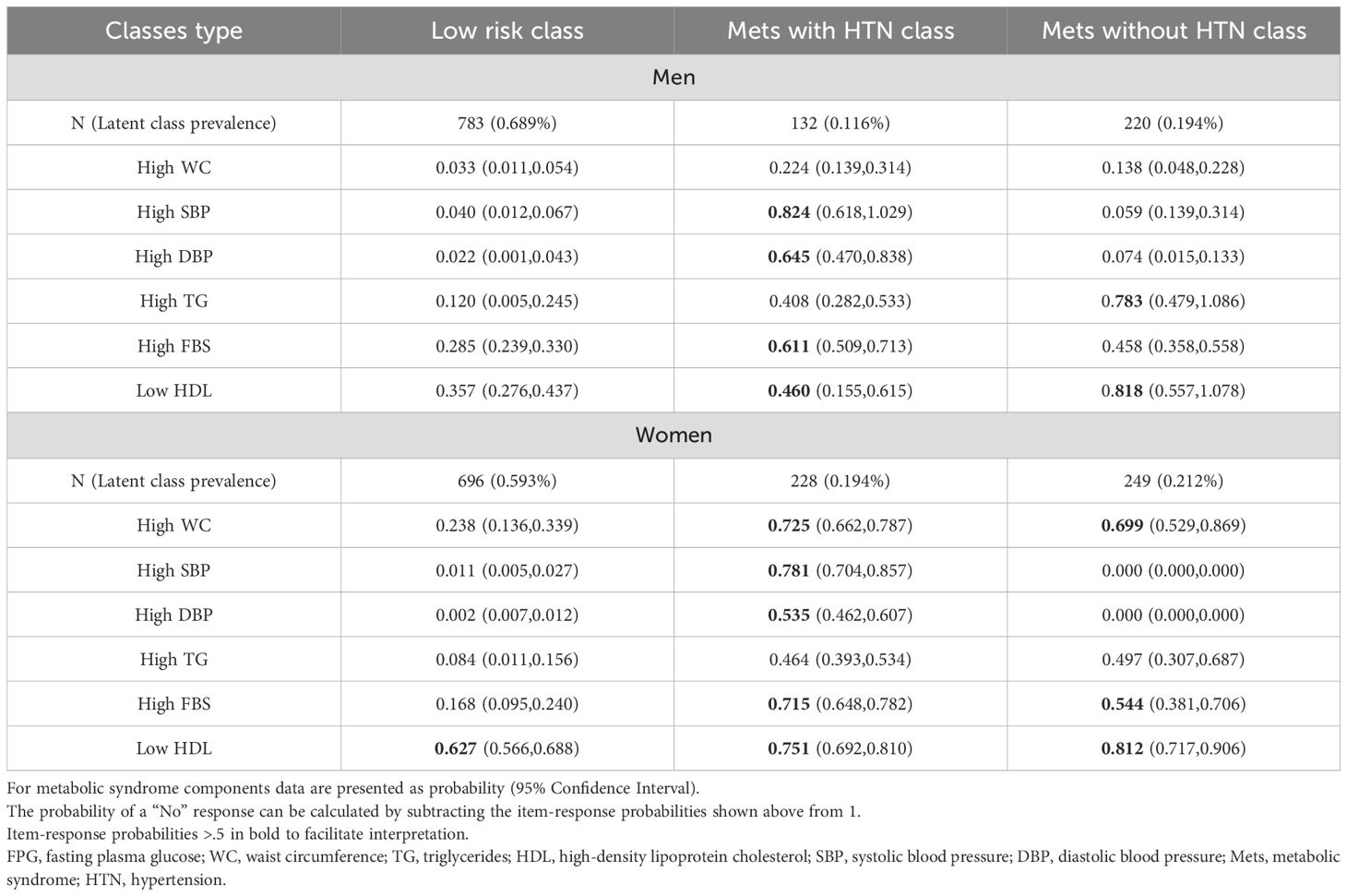
Table 3. The prevalence of latent classes and the estimated probability of observed metabolic syndrome components for each class type.
Table 4 presents the characteristics of participants in each latent class of metabolic syndrome. In Men, age was significantly different between all 3 classes. HTN class was the oldest and the low-risk class was the youngest. BMI was not significantly different between the two classes of metabolic syndrome but the low-risk group had a significantly lower BMI compared to the other classes. Men in the HTN class also had a significantly higher incidence of diabetes and were simultaneously more physically active. Mean WC, SBP, DBP, FPG, and serum TG were significantly different between all the 3 classes. HTN class had the highest mean SBP, DBP, FPG, and WC and Non-HTN class had the highest mean serum TG. Serum HDL was significantly lower only in the non-HTN class compared to the other two classes. In comparison to the low-risk class, both the HTN and Non-HTN classes showed significantly higher levels of HbA1c, serum CRP, and Cr. Nevertheless, the difference between the two groups was not considered statistically significant. Serum vitamin D levels were not significantly different among the 3 classes in men.
In women mean age and BMI were significantly different between all 3 classes. HTN class was the oldest and had the highest BMI while the low-risk class was the youngest with the lowest BMI. Women in the HTN group also had a significantly higher incidence of diabetes and were simultaneously more physically active. Mean serum SBP, DBP, and TG were significantly different between all three classes with the HTN class having the highest SBP and DBP and Non-HTN class having the highest mean serum TG level. The mean serum HDL level was significantly lower in the non-HTN class compared to the other classes. In comparison to the low-risk class, both the HTN and Non-HTN classes showed significantly higher levels of FPG, WC, HbA1c, serum vitamin D, CRP, and Cr. Nevertheless, the difference between the two groups was not considered statistically significant. (Table 4).
In men, using the “Low Risk” class as the reference class, BMI significantly predicted both classes of Mets (OR:1.26, CI:1.19-1.32, and OR:1.99, CI:1.15-1.24, for HTN and non-HTN classes respectively), while age only significantly predicted the HTN class. HbA1c (OR:1.42, CI:1.19-1.70, and OR:1.31, CI:1.12-1.54) and serum Cr (OR:2.93, CI:1.06-8.09, and OR:2.50, CI:1.06-5.88) were significant predictors of both HTN and non-HTN classes respectively, while serum vitamin D and CRP level were no significant predictors of metabolic syndrome classes.
In women, age (OR:1.16, CI:1.130-1.183, and OR:1.08, CI:1.065-1.103), BMI (OR:1.22, CI:1.171-1.268, OR:1.16, CI:1.124-1.207), HbA1c (OR:1.91, CI:1.60-2.29 and OR:1.75, CI:1.47-2.09), and serum Cr (OR:4.61, CI:1.72-12.34, and OR:3.23, CI:1.22-8.55) were significant predictors of both HTN and non-HTN Mets classes respectively, while serum CRP level only significantly predicted the HTN class (OR:1.03, CI:1.004-1.067). Serum vitamin D was not a significant predictor of metabolic syndrome classes (Table 5).
Using the “Low Risk” class as the reference class, both classes of metabolic syndrome significantly increased the OR for NAFLD in both men and women. After combining data from both sexes, latent class analysis revealed the same pattern across the population. In women, both HTN and Non-HTN classes resulted in a considerably higher OR for NAFLD compared to men even after adjustments for other covariates (age, serum vitamin D, serum c-reactive protein, diabetes, physical activity, and smoking status) (HTN class OR: 4.20 vs 2.94; non-HTN class OR: 5.60 vs 3.12 in women and men respectively) (Table 6).
Latent class analysis has proven to be instrumental in uncovering various patterns and subtypes of metabolic syndrome, thereby greatly enhancing our comprehension of the diversity within metabolic syndrome and its associated health consequences. Consequently, it serves as a valuable instrument for facilitating a more sophisticated comprehension of the condition and potentially guiding personalized treatment approaches.
In this particular study, the utilization of the latent class analysis allowed us to identify three distinct classes: HTN, non-HTN, and low-risk classes. The primary distinguishing factor of the HTN class was the presence of hypertension, indicated by high SBP or DBP. Additionally, hyperglycemia played a significant role in this class. On the other hand, the non-HTN class was characterized by dyslipidemia, specifically high serum TG and/or low HDL, with a very low likelihood of hypertension.
The occurrence of subclasses of metabolic syndrome in this investigation aligns with numerous prior investigations (17–20, 42, 43). Ghahramanloo et al. conducted a study in Qom, Iran, which identified three distinct latent classes among urban adult men (non-Mets, 55.1%; at risk, 21.3%; and Mets, 23.6%). Consistent with our findings in men, one of these classes exhibited a higher prevalence of high TG and low HDL, while the other class had a higher prevalence of hypertension. Additionally, the group with hypertension had a higher likelihood of hyperglycemia (20). Another study by the same author in Tehran, identified four latent classes of metabolic syndrome: non-Mets, low risk, high risk, and MetS. Similar to our findings, hypertension and hyperlipidemia were the primary components of the last two classes, but in contrast to our results, hyperglycemia was more closely associated with hyperlipidemia (17). In another study by Galvão et al, researchers used latent class analysis to identify different patterns of metabolic syndrome among women in the ELSA-Brasil cohort. Similar to our own findings, they identified three patterns that one them was characterized by high serum TG and low HDL, while the other pattern was associated with central obesity, hyperglycemia, and hypertension. The third pattern had low probabilities of all abnormalities, except for central obesity (44). Another study analyzed data from the Multi-Ethnic Study of Atherosclerosis (MESA) to identify patterns of metabolic syndrome components. This study also highlighted the importance of hypertension, low HDL, and hyperglycemia in defining different subgroups of metabolic syndrome (45).
The existence of a HTN latent subgroup within the population diagnosed with metabolic syndrome where hypertension and hyperglycemia are the two most important components can be elucidated through various mechanisms. Prior studies suggest that insulin resistance and endothelial dysfunction could link hypertension and hyperglycemia. Insulin resistance impairs the ability of insulin to promote glucose uptake in tissues, leading to hyperglycemia. Meanwhile Insulin has vasodilatory effects. When cells are resistant to insulin, this vasodilatory effect is diminished, contributing to increased vascular resistance and hypertension (46). High blood glucose levels can also damage the endothelium. This damage impairs the production of nitric oxide (NO), a molecule that helps blood vessels relax (47). Additionally, hyperglycemia can lead to the formation of advanced glycation end-products (AGEs), which further damage the endothelium and promote inflammation, contributing to hypertension (47, 48). Insulin resistance and hyperglycemia can also activate the sympathetic nervous system, which increases heart rate and constricts blood vessels, leading to higher blood pressure (49).
Furthermore, research suggests that the impact of insulin resistance on endothelial function may vary depending on gender. One potential explanation for this is the presence of hormonal disparities. Estrogen is known to have a protective effect on endothelial function, whereas testosterone’s relationship with endothelial function is more intricate (50). Another factor to consider is the inflammatory response. Insulin resistance often coincides with an inflammatory state, and the inflammatory reaction may differ between males and females. Some studies propose that males might be more vulnerable to the adverse effects of inflammation on endothelial function (51). This phenomenon could clarify why men in the present study displayed elevated systolic blood pressure and hyperglycemia even in the absence of abdominal obesity (low prevalence of increased waist circumference), unlike women. Boyko et al. also suggest that this connection between hypertension and hyperglycemia may not be influenced by central adiposity (as was noted in men in the present study) (15).
Impairment in renal function could potentially be the other link connecting hyperglycemia and hypertension. Previous studies have shown that prediabetes can worsen kidney function, while chronic kidney disease can exacerbate hypertension by affecting the body’s fluid and electrolyte balance (52, 53). In contrast, hypertension can further advance chronic kidney disease by putting stress on the kidneys, causing damage to blood vessels, and reducing their ability to effectively filter waste products (54, 55). In the current study, both HbA1c and serum Cr were found to be significant predictors of metabolic syndrome classes, particularly the HTN class. This finding strengthens the connection between kidney function impairment, hyperglycemia, and hypertension. Endothelial dysfunction caused in all the above mechanisms could be a key factor in the adverse effects of hypertension and hyperglycemia, even in the absence of diabetes, leading to a decline in function, vascular issues, and Alzheimer’s disease (56–58). Therefore, the presence of both high blood pressure and hyperglycemia, as seen in the HTN latent class identified in this study, highlights the urgent need to address these conditions to prevent organ damage and associated complications.
In the current study, the presence of non-HTN latent subgroup that was characterized by dyslipidemia, specifically high serum TG and/or low HDL, with a very low likelihood of hypertension, could be explained through mechanisms related to lipoprotein metabolism and genetic factors that do not necessarily involve hypertension such as hepatic overproduction of VLDL (59), impaired lipoprotein lipase activity or lipoprotein metabolism (60, 61). Understanding these mechanisms is crucial for developing targeted interventions for metabolic syndrome subclasses.
Serum CRP and vitamin D levels were not significant predictors of metabolic syndrome classes in the present study, except for serum CRP for HTN class in women. CRP is a protein produced by the liver in response to inflammation, primarily triggered by interleukin-6 (IL-6) and other inflammatory cytokines (62). Research suggests that adipose tissue can produce inflammatory cytokines, leading to increased levels of CRP (63–65). In the current research women exhibit significantly higher BMI, waist circumference, and double serum CRP levels, indicating that the association between serum CRP level and HTN class in women might indicate a heightened inflammatory state in women, which can contribute to the pathophysiology of hypertension (66). Studies have also shown that CRP levels are higher in individuals with specific components of metabolic syndrome, such as abdominal obesity, insulin resistance, and hypertension (67–72).
Regarding the association between serum vitamin D level and metabolic syndrome, different studies have reported inconsistent results. While some research has indicated a higher likelihood of developing metabolic syndrome and its components with low serum vitamin D levels (73, 74), others have not found such a connection (75, 76). These results imply that the association between serum vitamin D levels and metabolic syndrome may vary among different populations and age groups. In Iran, factors like limited sunlight exposure, low intake of vitamin D-rich foods (77), cultural norms regarding clothing that limits sun exposure (78), economic constraints affecting access to nutrition and healthcare (77), low physical activity, and air pollution (79, 80) all contribute to a high prevalence of vitamin D deficiency. The mean serum vitamin D level of 17.30 ng/mL in this study further underscores the prevalence of Vitamin D deficiency in Iran. This deficiency may complicate the ability to identify associations between vitamin D levels and metabolic syndrome classes, as low levels of serum vitamin D are commonly found in all classes, potentially masking differences that would otherwise be noticeable.
Understanding these factors can provide valuable context for interpreting the lack of association between vitamin D status and metabolic syndrome classes in our study, highlighting the complexity of this relationship within specific demographic groups.
Concerning the correlation between the identified metabolic syndrome classes and NAFLD, both the HTN and non-HTN classes exhibited a similar increase in the OR of NAFLD for each gender. However, the ORs of both classes were significantly higher in women compared to men, approximately three times higher. This notable difference in ORs can potentially be attributed to the higher prevalence of visceral adiposity in women within both metabolic syndrome classes. According to our finding 71.1% and 88.0% of women in HTN and Non-HTN classes had high waist circumference. In men, the frequencies were 35.2% and 38.5% for the HTN and Non-HTN classes, respectively (Supplementary Table 1). Visceral adiposity is a crucial risk factor for NAFLD, and its higher occurrence in women may explain the considerably higher ORs of both HTN and non-HTN classes for NAFLD in women compared to men. Our findings align with the study conducted by Ahanchi et al, which reported that the relationship between subclasses of metabolic syndrome and incident CVD varied by gender. They suggested that the etiology of metabolic syndrome involves multiple pathways, and it is necessary to reconsider the equal weighting of each component or the use of the same cut-off values in both genders (19).
Our findings indicate that there may be distinct latent classes within the defined metabolic syndrome that exhibit varying pathophysiology or different levels of risk for non-communicable diseases. The main finding in this study was the significance of hypertension in the classification of metabolic syndrome. Our study demonstrates a conditional probability of approximately 0 for SBP and DBP in both men and women in the “non-HTN” class, indicating excellent classification and high specificity of the SBP and DBP components for these classes. Conversely, there is a simultaneous high probability of hyperglycemia in the HTN class, suggesting a shared underlying mechanism for these two components, separate from dyslipidemia. Another noteworthy observation is the presence of elevated visceral adiposity in both classes among women, but not men, which may indicate a disparity in the role of visceral adiposity in the pathophysiological mechanism of metabolic syndrome between the two sexes.
Comprehension of the diversity within metabolic syndrome and its related health consequences helps in better identifying individuals who may be at a higher or lower risk of developing related conditions like NAFLD. Precise risk categorization allows for focused interventions and more frequent monitoring of those at high risk. Customizing interventions according to the specific metabolic syndrome subclass can maximize the effectiveness of lifestyle changes, medication, and monitoring methods.
The study’s notable strength lies in its utilization of a substantial population-based cohort study, thereby enhancing the credibility and reliability of our findings. Furthermore, we performed latent class analysis separately for each gender, thereby presenting distinct patterns for both males and females. This study also offers important information on the subclasses of metabolic syndrome and their connection to NAFLD. Nevertheless, it is crucial to recognize the limitations of this research. It is vital to acknowledge the potential impact of unobserved factors such as socioeconomic status, genetic traits, and lifestyle variables influencing the results. Being cross-sectional and observational is another limitation to consider when interpreting the results. Furthermore, although we have adjusted the association between latent classes of metabolic syndrome and NAFLD we suggest future-tailored research with higher sample sizes that can assess the associations in covariate-based stratified groups in each sex to address the unique health needs of both male and female participants based on the gender-specific insights from our study. Overcoming these limitations in future studies will improve our comprehension of the intricate relationships between NAFLD and metabolic well-being.
In the northern region of Iran, the latent class analysis revealed the presence of three distinct classes of metabolic syndrome: HTN, Non-HTN, and low-risk classes. Hypertension played a crucial role in determining these classes. Furthermore, both HTN and Non-HTN classes exhibited a higher prevalence of visceral adiposity and served as stronger predictors of NAFLD in women. Notably, serum CRP and vitamin D levels did not emerge as significant predictors of metabolic syndrome latent classes, except for serum CRP in the HTN class among women.
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
The studies involving humans were approved by ethics committee of Iran University of Medical Sciences by No. IR.IUMS.REC.1400.982. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
BA: Conceptualization, Data curation, Writing – original draft, Writing – review & editing. PH: Formal analysis, Methodology, Writing – review & editing. FS: Writing – original draft. EG: Data curation, Writing – review & editing, Investigation. AD: Writing – review & editing. FST: Writing – review & editing, Conceptualization. NM: Writing – review & editing, Formal analysis. MM: Investigation, Writing – review & editing. MS: Writing – review & editing, Investigation. ES: Writing – review & editing. FZ: Supervision, Writing – review & editing. HA: Supervision, Writing – review & editing.
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This research was conducted by a grant (1400-2-30-21394) from Iran University of Medical Sciences (IUMS).
The authors would like to thank the staff of all local health centers in Amol for their help throughout the completion of this research.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fendo.2025.1405833/full#supplementary-material
Mets, Metabolic syndrome; NAFLD, non-alcoholic fatty liver disease; LCA, Latent class analysis; HTN, Mets with Hypertension; Non-HTN, Mets without HTN; ATP III, Adult Treatment Panel III criteria.
1. Eckel RH, Alberti KG, Grundy SM, Zimmet PZ. The metabolic syndrome. Lancet. (2010) 375:181–3. doi: 10.1016/S0140-6736(09)61794-3
2. Esser N, Legrand-Poels S, Piette J, Scheen AJ, Paquot N. Inflammation as a link between obesity, metabolic syndrome and type 2 diabetes. Diabetes Res Clin Pract. (2014) 105:141–50. doi: 10.1016/j.diabres.2014.04.006
3. Kim LJ, Nalls MA, Eiriksdottir G, Sigurdsson S, Launer LJ, Koster A, et al. Associations of visceral and liver fat with the metabolic syndrome across the spectrum of obesity: the AGES-Reykjavik study. Obesity. (2011) 19:1265–71. doi: 10.1038/oby.2010.291
4. Scuteri A, Laurent S, Cucca F, Cockcroft J, Cunha PG, Mañas LR, et al. Metabolic syndrome across Europe: different clusters of risk factors. Eur J Prev Cardiol. (2015) 22:486–91. doi: 10.1177/2047487314525529
5. Harikrishnan S, Sarma S, Sanjay G, Jeemon P, Krishnan M, Venugopal K, et al. Prevalence of metabolic syndrome and its risk factors in Kerala, South India: Analysis of a community based cross-sectional study. PloS One. (2018) 13:e0192372. doi: 10.1371/journal.pone.0192372
6. Aljohani NJ. Metabolic syndrome: Risk factors among adults in Kingdom of Saudi Arabia. J Family Community Med. (2014) 21:170. doi: 10.4103/2230-8229.142971
7. Saklayen MG. The global epidemic of the metabolic syndrome. Curr Hypertens Rep. (2018) 20:12. doi: 10.1007/s11906-018-0812-z
8. Mazloomzadeh S, Rashidi Khazaghi Z, Mousavinasab N. The prevalence of metabolic syndrome in Iran: A systematic review and meta-analysis. Iran J Public Health. (2018) 47:473–80.
9. Fatahi A, Doosti-Irani A, Cheraghi Z. Prevalence and incidence of metabolic syndrome in Iran: a systematic review and meta-analysis. Int J Prev Med. (2020) 11:64–73. doi: 10.4103/ijpvm.IJPVM_489_18
10. Alberti KGMM, Zimmet PZ. Definition, diagnosis and classification of diabetes mellitus and its complications. Part 1: diagnosis and classification of diabetes mellitus. Provisional report of a WHO consultation. Diabetes Med. (1998) 15:539–53. doi: 10.1002/(SICI)1096-9136(199807)15:7<539::AID-DIA668>3.0.CO;2-S
11. Borges RL, Ribeiro AB, Zanella MT, Batista MC. Uric acid as a factor in the metabolic syndrome. Curr Hypertens Rep. (2010) 12:113–9. doi: 10.1007/s11906-010-0098-2
12. Alberti KGM, Zimmet P, Shaw J. The metabolic syndrome—a new worldwide definition. Lancet. (2005) 366:1059–62. doi: 10.1016/S0140-6736(05)67402-8
13. Association AH. Third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (adult treatment panel III). Circulation. (2002) 106:3143–373.
14. Riahi SM, Moamer S, Namdari M, Mokhayeri Y, Pourhoseingholi MA, Hashemi-Nazari SS. Patterns of clustering of the metabolic syndrome components and its association with coronary heart disease in the Multi-Ethnic Study of Atherosclerosis (MESA): A latent class analysis. Int J Cardiol. (2018) 271:13–8. doi: 10.1016/j.ijcard.2018.05.080
15. Boyko EJ, Doheny RA, McNeely MJ, Kahn SE, Leonetti DL, Fujimoto WY. Latent class analysis of the metabolic syndrome. Diabetes Res Clin Pract. (2010) 89:88–93. doi: 10.1016/j.diabres.2010.02.013
16. Abbasi-Ghahramanloo A, Janani L, Malakouti SK, Rabetian M, Rimaz S. Risk taking behaviors in relation to Attention Deficit and Hyperactivity Disorder in Iranian male workers: a latent class analysis. Neuropsychiatr Dis Treat. (2019) 15:2513–20. doi: 10.2147/NDT.S221858
17. Abbasi-Ghahramanloo A, Soltani S, Gholami A, Erfani M, Yosaee S. Clustering and combining pattern of metabolic syndrome components among Iranian population with latent class analysis. Med J Islam Repub Iran. (2016) 30:445.
18. Ahanchi NS, Hadaegh F, Alipour A, Ghanbarian A, Azizi F, Khalili D. Application of latent class analysis to identify metabolic syndrome components patterns in adults: Tehran Lipid and Glucose Study. Sci Rep. (2019) 9:1572. doi: 10.1038/s41598-018-38095-0
19. Ahanchi NS, Hadaegh F, Azizi F, Khalili D. Sex-specific clustering of metabolic syndrome components and incidence of cardiovascular disease: A latent class analysis in a population-based cohort study. J Diabetes Complicat. (2021) 35:107942. doi: 10.1016/j.jdiacomp.2021.107942
20. Abbasi-Ghahramanloo A, Moshiri E, Afrashteh S, Gholami A, Safiri S, Mohammadbeigi A, et al. Comorbidity of metabolic syndrome components in a population-based screening program: A latent class analysis. Med J Islam Repub Iran. (2020) 34:69. doi: 10.34171/mjiri.34.69
21. Wang C-C, Jhu J-J. On the application of clustering and classification techniques to analyze metabolic syndrome severity distribution area and critical factors. Int J Environ Res Public Health. (2019) 16:1575. doi: 10.3390/ijerph16091575
22. Gurka MJ, Filipp SL, DeBoer MD. Geographical variation in the prevalence of obesity, metabolic syndrome, and diabetes among US adults. Nutr Diabetes. (2018) 8:14. doi: 10.1038/s41387-018-0024-2
23. Mohamud WNW, Ismail A-S, Sharifuddin A, Ismail IS, Musa KI, Kadir KA, et al. Prevalence of metabolic syndrome and its risk factors in adult Malaysians: results of a nationwide survey. Diabetes Res Clin Pract. (2011) 91:239–45. doi: 10.1016/j.diabres.2010.11.025
24. Arguelles W, Llabre MM, Sacco RL, Penedo FJ, Carnethon M, Gallo LC, et al. Characterization of metabolic syndrome among diverse Hispanics/Latinos living in the United States: Latent class analysis from the Hispanic Community Health Study/Study of Latinos (HCHS/SOL). Int J Cardiol. (2015) 184:373–9. doi: 10.1016/j.ijcard.2015.02.100
25. Lin CS, Lee WJ, Lin SY, Lin HP, Chen RC, Lin CH, et al. Subtypes of premorbid metabolic syndrome and associated clinical outcomes in older adults. Front Med (Lausanne). (2021) 8:698728. doi: 10.3389/fmed.2021.698728
26. Wang X, Li W, Zhang Y, Yang Y, Qin G. Association between vitamin D and non-alcoholic fatty liver disease/non-alcoholic steatohepatitis: results from a meta-analysis. Int J Clin Exp Med. (2015) 8:17221–34.
27. Barchetta I, Cimini FA, Cavallo MG. Vitamin D supplementation and non-alcoholic fatty liver disease: present and future. Nutrients. (2017) 9:1015–23. doi: 10.3390/nu9091015
28. Sangouni AA, Ghavamzadeh S, Jamalzehi A. A narrative review on effects of vitamin D on main risk factors and severity of Non-Alcoholic Fatty Liver Disease. Diabetes Metab Syndr. (2019) 13:2260–5. doi: 10.1016/j.dsx.2019.05.013
29. Al Kady MS, Alegaily HS, El Kholy HR, Abd Elshafy SZ, Qayed AA. Serum vitamin D in patients with non alcoholic fatty liver disease. Benha Medical J. (2021) 38(2):712–24.
30. Oruç N, Ozutemiz O, Yuce G, Akarca US, Ersoz G, Gunsar F, et al. Serum procalcitonin and CRP levels in non-alcoholic fatty liver disease: a case control study. BMC Gastroenterol. (2009) 9:16–. doi: 10.1186/1471-230X-9-16
31. Foroughi M, Maghsoudi Z, Khayyatzadeh S, Ghiasvand R, Askari G, Iraj B. Relationship between non-alcoholic fatty liver disease and inflammation in patients with non-alcoholic fatty liver. Adv Biomed Res. (2016) 5:28–32. doi: 10.4103/2277-9175.176368
32. Velivala AP, Madhav V, PradeepBabu K. C-reactive protein as a marker for insulin resistance in non-alcoholic fatty liver disease in non-obese South Indian population. IOSR J Dental Med Sci (IOSRJDMS). (2013) 6(2):44–8.
33. Motamed N, Faraji AH, Khonsari MR, Maadi M, Tameshkel FS, Keyvani H, et al. Fatty liver index (FLI) and prediction of new cases of non-alcoholic fatty liver disease: A population-based study of northern Iran. Clin Nutr. (2020) 39:468–74. doi: 10.1016/j.clnu.2019.02.024
34. Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, et al. The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of Liver Diseases. Hepatology. (2018) 67:328–57. doi: 10.1002/hep.29367
35. Sberna A, Bouillet B, Rouland A, Brindisi M, Nguyen A, Mouillot T, et al. European Association for the Study of the Liver (EASL), European Association for the Study of Diabetes (EASD) and European Association for the Study of Obesity (EASO) clinical practice recommendations for the management of non-alcoholic fatty liver disease: evaluation of their application in people with Type 2 diabetes. Diabetes Med. (2018) 35:368–75. doi: 10.1111/dme.2018.35.issue-3
36. Hagströmer M, Oja P, Sjöström M. The International Physical Activity Questionnaire (IPAQ): a study of concurrent and construct validity. Public Health Nutr. (2006) 9:755–62. doi: 10.1079/PHN2005898
38. Alberti K, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, et al. Harmonizing the metabolic syndrome: a joint interim statement of the international diabetes federation task force on epidemiology and prevention; national heart, lung, and blood institute; American heart association; world heart federation; international atherosclerosis society; and international association for the study of obesity. Circulation. (2009) 120:1640–5. doi: 10.1161/CIRCULATIONAHA.109.192644
39. Hernaez R, Lazo M, Bonekamp S, Kamel I, Brancati FL, Guallar E, et al. Diagnostic accuracy and reliability of ultrasonography for the detection of fatty liver: a meta-analysis. Hepatology. (2011) 54:1082–90. doi: 10.1002/hep.24452
40. Lee SS, Park SH, Kim HJ, Kim SY, Kim M-Y, Kim DY, et al. Non-invasive assessment of hepatic steatosis: prospective comparison of the accuracy of imaging examinations. J Hepatol. (2010) 52:579–85. doi: 10.1016/j.jhep.2010.01.008
41. Weller BE, Bowen NK, Faubert SJ. Latent class analysis: a guide to best practice. J Black Psychol. (2020) 46:287–311. doi: 10.1177/0095798420930932
42. Esteghamati A, Zandieh A, Khalilzadeh O, Meysamie A, Ashraf H. Clustering of metabolic syndrome components in a Middle Eastern diabetic and non-diabetic population. Diabetol Metab Syndr. (2010) 2:36. doi: 10.1186/1758-5996-2-36
43. Oh J-Y, Hong YS, Sung Y-A, Barrett-Connor E. Prevalence and factor analysis of metabolic syndrome in an urban Korean population. Diabetes Care. (2004) 27:2027–32. doi: 10.2337/diacare.27.8.2027
44. Galvão NMS, Matos S, Almeida M, Gabrielli L, Barreto SM, Aquino EM, et al. Patterns of metabolic syndrome and associated factors in women from the ELSA-Brasil: a latent class analysis approach. Cad Saude Publica. (2023) 39:e00039923. doi: 10.1590/0102-311xen039923
45. Riahi SM, Fanoodi A, Shetty S, Hashemi-Nazari SS. Critical assessment of the metabolic syndrome definitions in the adult general population of the United States-the Multi-Ethnic Study of Atherosclerosis (MESA). J Diabetes Metab Disord. (2023) 22:851–9. doi: 10.1007/s40200-023-01213-5
46. Muniyappa R, Montagnani M, Koh KK, Quon MJ. Cardiovascular actions of insulin. Endocr Rev. (2007) 28:463–91. doi: 10.1210/er.2007-0006
47. Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature. (2001) 414:813–20. doi: 10.1038/414813a
48. Schalkwijk CG, Stehouwer CD. Vascular complications in diabetes mellitus: the role of endothelial dysfunction. Clin Sci. (2005) 109:143–59. doi: 10.1042/CS20050025
49. Grassi G, Seravalle G, Mancia G. Sympathetic activation in cardiovascular disease: evidence, clinical impact and therapeutic implications. Eur J Clin Invest. (2015) 45:1367–75. doi: 10.1111/eci.12553
50. Usselman CW, Stachenfeld NS, Bender JR. The molecular actions of estrogen in the regulation of vascular health. Exp Physiol. (2016) 101:356. doi: 10.1113/EP085148
51. Moss ME, Lu Q, Iyer SL, Engelbertsen D, Marzolla V, Caprio M, et al. Endothelial mineralocorticoid receptors contribute to vascular inflammation in atherosclerosis in a sex-specific manner. Arterioscler Thromb Vasc Biol. (2019) 39:1588–601. doi: 10.1161/ATVBAHA.119.312954
52. Lamprou S, Koletsos N, Mintziori G, Anyfanti P, Trakatelli C, Kotsis V, et al. Microvascular and endothelial dysfunction in prediabetes. Life. (2023) 13:644. doi: 10.3390/life13030644
53. Chen C, Liu G, Yu X, Yu Y, Liu G. Association between prediabetes and renal dysfunction from a community-based prospective study. Int J Med Sci. (2020) 17:1515. doi: 10.7150/ijms.46477
54. Burnier M, Damianaki A. Hypertension as cardiovascular risk factor in chronic kidney disease. Circ Res. (2023) 132:1050–63. doi: 10.1161/CIRCRESAHA.122.321762
55. Blin P, Joubert M, Jourdain P, Zaoui P, Guiard E, Sakr D, et al. Cardiovascular and renal diseases in type 2 diabetes patients: 5-year cumulative incidence of the first occurred manifestation and hospitalization cost: a cohort within the French SNDS nationwide claims database. Cardiovasc Diabetol. (2024) 23:22. doi: 10.1186/s12933-023-02101-1
56. Santulli G, Visco V, Ciccarelli M, Ferrante MNV, De Masi P, Pansini A, et al. Frail hypertensive older adults with prediabetes and chronic kidney disease: insights on organ damage and cognitive performance-preliminary results from the CARYATID study. Cardiovasc Diabetol. (2024) 23:125. doi: 10.1186/s12933-024-02218-x
57. Pansini A, Lombardi A, Morgante M, Frullone S, Marro A, Rizzo M, et al. Hyperglycemia and physical impairment in frail hypertensive older adults. Front Endocrinol (Lausanne). (2022) 13:831556. doi: 10.3389/fendo.2022.831556
58. Mone P, De Gennaro S, Moriello D, Frullone S, D’Amelio R, Ferrante MNV, et al. Insulin resistance drives cognitive impairment in hypertensive pre-diabetic frail elders: the CENTENNIAL study. Eur J Prev Cardiol. (2023) 30:1283–8. doi: 10.1093/eurjpc/zwad173
59. Adiels M, Taskinen M-R, Packard C, Caslake MJ, Soro-Paavonen A, Westerbacka J, et al. Overproduction of large VLDL particles is driven by increased liver fat content in man. Diabetologia. (2006) 49:755–65. doi: 10.1007/s00125-005-0125-z
60. Burnett JD, Deeb SS. Familial lipoprotein lipase deficiency, apo C-II deficiency, and hepatic lipase deficiency. The Online Metabolic and Molecular Bases of Inherited Disease. Eds. Valle DL, et al. McGraw-Hill Education (2009). https://ommbid.mhmedical.com/content.aspx?bookid=2709§ionid=225539482
61. Brahm AJ, Hegele RA. Chylomicronaemia—current diagnosis and future therapies. Nat Rev Endocrinol. (2015) 11:352–62. doi: 10.1038/nrendo.2015.26
62. Black S, Kushner I, Samols D. C-reactive protein. J Biol Chem. (2004) 279:48487–90. doi: 10.1074/jbc.R400025200
63. Lakoski SG, Herrington DM, Siscovick DM, Hulley SB. C-reactive protein concentration and incident hypertension in young adults: the CARDIA study. Arch Intern Med. (2006) 166:345–9. doi: 10.1001/archinte.166.3.345
64. Soltani Z, Washco V, Morse S, Reisin E. The impacts of obesity on the cardiovascular and renal systems: cascade of events and therapeutic approaches. Curr Hypertens Rep. (2015) 17:7. doi: 10.1007/s11906-014-0520-2
65. Sesso HD, Wang L, Buring JE, Ridker PM, Gaziano JM. Comparison of interleukin-6 and C-reactive protein for the risk of developing hypertension in women. Hypertension. (2007) 49:304–10. doi: 10.1161/01.HYP.0000252664.24294.ff
66. Ebong IA, Schreiner P, Lewis CE, Appiah D, Ghelani A, Wellons M. The association between high-sensitivity C-reactive protein and hypertension in women of the CARDIA study. Menopause. (2016) 23:662–8. doi: 10.1097/GME.0000000000000609
67. Florez HJ, Castillo-Florez S, Mendez AJ, Casanova-Romero P, Larreal-Urdaneta C, Lee DJ, et al. C-reactive protein is elevated in obese patients with the metabolic syndrome. Diabetes Res Clin Pract. (2006) 71 1:92–100. doi: 10.1016/j.diabres.2005.05.003
68. Lee W-Y, Park JS, Noh S, Rhee E-J, Sung KC, Kim BS, et al. C-reactive protein concentrations are related to insulin resistance and metabolic syndrome as defined by the ATP III report. Int J Cardiol. (2004) 97 1:101–6. doi: 10.1016/j.ijcard.2003.08.016
69. González AS, Guerrero DB, Soto MB, Diaz S, Martínez-Olmos MA, Vidal O. Metabolic syndrome, insulin resistance and the inflammation markers C-reactive protein and ferritin. Eur J Clin Nutr. (2006) 60:802–9. doi: 10.1038/sj.ejcn.1602384
70. Lemieux IG, Pascot A, Prud’homme D, Alméras N, Bogaty PM, Nadeau A, et al. Elevated C-reactive protein: another component of the atherothrombotic profile of abdominal obesity. Arteriosc Thromb Vasc Biol: J Am Heart Assoc. (2001) 21:961–7. doi: 10.1161/01.ATV.21.6.961
71. Chowta MN, Adhikari P, Sinha R, Acharya SD, Hn G, Ramapuram JT. Highly sensitive C reactive protein in patients with metabolic syndrome and cardiovascular disease. Ann Trop Med Public Health. (2012) 5:98–102. doi: 10.4103/1755-6783.95960
72. Santos AC, Lopes C, Guimarães JT, Barros H. Central obesity as a major determinant of increased high-sensitivity C-reactive protein in metabolic syndrome. Int J Obes. (2005) 29:1452–6. doi: 10.1038/sj.ijo.0803035
73. Chon SJ, Yun BH, Jung YS, Cho S, Choi YS, Kim S-Y, et al. Association between vitamin D status and risk of metabolic syndrome among Korean postmenopausal women. PloS One. (2014) 9:e89725–31. doi: 10.1371/journal.pone.0089721
74. Jeenduang N, Plyduang T, Horpet D. Association of 25-hydroxyvitamin D levels and metabolic syndrome in Thai postmenopausal women. Diabetes Metab Syndr. (2020) 14:1585–90. doi: 10.1016/j.dsx.2020.08.018
75. Abuhijleh H, Alkhatib DH, Ganji V. Hypovitaminosis D and metabolic syndrome in postmenopausal women. Healthcare. (2022) 10:2026–36. doi: 10.3390/healthcare10102026
76. Amirbaigloo A, Hosseinpanah F, Sarvghadi F, Tohidi M, Eskandary PS, Azizi F. Absence of association between vitamin D deficiency and incident metabolic syndrome: Tehran Lipid and Glucose Study. Metab Syndr Relat Disord. (2013) 11:236–42. doi: 10.1089/met.2012.0121
77. Aghapour B, Kheirouri S, Alizadeh M, Khodayari-Zarnaq R. Vitamin D deficiency prevention policies in Iran: a retrospective policy analysis. Front Nutr. (2023) 10:1249402. doi: 10.3389/fnut.2023.1249402
78. Rabbani A, Alavian S-M, Motlagh ME, Ashtiani MT, Ardalan G, Salavati A, et al. Vitamin D insufficiency among children and adolescents living in Tehran, Iran. J Trop Pediatr. (2009) 55:189–91. doi: 10.1093/tropej/fmn078
79. Hoseinzadeh E, Taha P, Wei C, Godini H, Ashraf GM, Taghavi M, et al. The impact of air pollutants, UV exposure and geographic location on vitamin D deficiency. Food Chem Toxicol. (2018) 113:241–54. doi: 10.1016/j.fct.2018.01.052
Keywords: latent class analysis, metabolic syndrome, Iran, non-alcoholic fatty liver, hypertension
Citation: Amirkalali B, Hassanzadeh P, Sheikholmolooki F, Gholizadeh E, Doustmohammadian A, Safarnezhad Tameshkel F, Motamed N, Maadi M, Sohrabi M, Sobhrakhshankhah E, Zamani F and Ajdarkosh H (2025) The crucial role of hypertension in determining latent classes of metabolic syndrome in northern Iran and predictive power of these classes in non-alcoholic fatty liver: a gender-based insight. Front. Endocrinol. 16:1405833. doi: 10.3389/fendo.2025.1405833
Received: 06 April 2024; Accepted: 22 January 2025;
Published: 28 February 2025.
Edited by:
Rodrigo O. Maranon, CCT CONICET Tucuman, ArgentinaReviewed by:
Ahmed Bendary, Benha University, EgyptCopyright © 2025 Amirkalali, Hassanzadeh, Sheikholmolooki, Gholizadeh, Doustmohammadian, Safarnezhad Tameshkel, Motamed, Maadi, Sohrabi, Sobhrakhshankhah, Zamani and Ajdarkosh. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Farhad Zamani, emFtYW5pLmZhcmhhZEBnbWFpbC5jb20=; Hossein Ajdarkosh, YWpkYXJrb3NoLmhAaXVtcy5hYy5pcg==
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.