- 1Diabetic Neuropathy Project, Tokyo Metropolitan Institute of Medical Science, Tokyo, Japan
- 2Laboratory of Molecular Neuroscience and Neurology, Tokyo University of Pharmacy and Life Sciences, Tokyo, Japan
Growing evidence suggests that hyperglycemia-related abnormalities in Schwann cells play a pivotal role in the development and progression of diabetic peripheral neuropathy (DPN). Several immortalized Schwann cell lines have been established in our laboratory and utilized for the study of DPN; IMS32 from normal ICR mice, 1970C3 from normal C57BL/6 mice, IWARS1 and IKARS1 from wild-type and aldose reductase-deficient C57BL/6 mice, and IFRS1 from normal Fischer 344 rats. These cell lines retain biological features of Schwann cells and display high proliferative activities that enable us to perform molecular and biochemical analyses. In addition, these cells have exhibited metabolic alterations under exposure to diabetes-associated conditions, such as hyperglycemia, dyslipidemia, glycative and oxidative stress load. Herein, recent studies with these cell lines regarding the pathogenic factors of DPN (augmentation of the polyol and other collateral glycolysis pathways, glycative and oxidative stress-induced cell injury, autophagic and proteostatic disturbances, etc.) and therapeutic strategies targeting these factors are introduced.
1 Introduction
As glial cells in the peripheral nervous system (PNS), Schwann cells are responsible for providing trophic support for the growth and maintenance of neurons and wrapping their axons in either a myelinating or an unmyelinating form. Following axonal injury, Schwann cells dedifferentiate into a ‘repair’ phenotype, contributing to axonal regeneration and functional recovery. They proliferate, migrate to the lesion site, eliminate axon debris alongside macrophages, promote the elongation of new axons, and finally remyelinate them (1, 2).
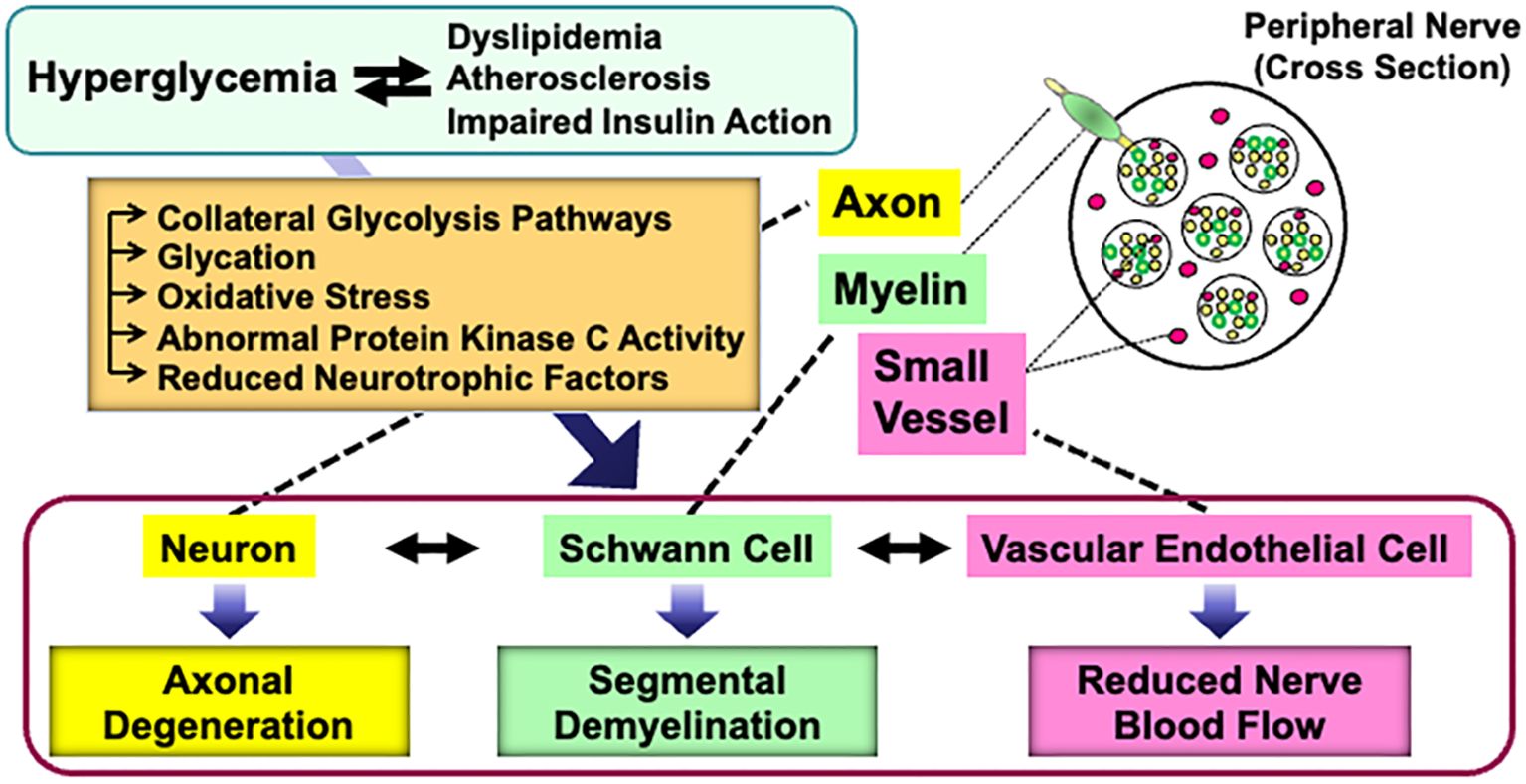
Abnormalities of Schwann cells and/or their crosstalk with neurons and other non-neuronal cells in the PNS can lead to various types of peripheral neuropathies. Diabetes Mellitus is one of the major causes of neuropathies, and diabetic peripheral neuropathy (DPN) is the most frequent and early-onset complication of type 1 and type 2 diabetes (3, 4). DPN is characterized by progressive, nerve-length-dependent loss of peripheral nerve fibers, leading to impaired sensory and autonomic function, pain, numbness, and eventually, complete loss of sensation. Although its pathogenesis remains largely unclear, hyperglycemic insults, along with dyslipidemia, atherosclerosis, and impaired insulin action (in the case of type 2 diabetes), cause a range of metabolic abnormalities in neurons, Schwann cells, and vascular endothelial cells. These changes result in axonal degeneration, demyelination, and reduced nerve blood flow, respectively (5). Furthermore, the abnormalities in each cell type can trigger dysfunction in others (6) (Figure 1).
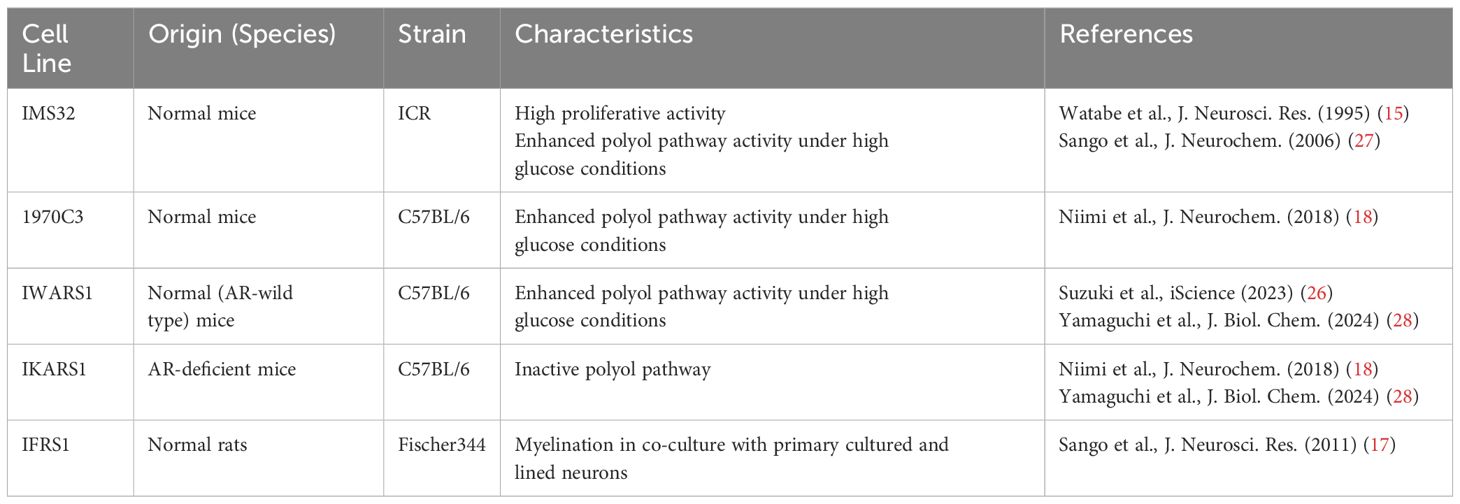
Cultured Schwann cells are valuable tools to explore the pathogenesis of DPN. Methods for primary culture of Schwann cells from both normal and diabetic animals have been established and widely used in DPN research (7–9). However, the primary cultures have several disadvantages, including time-consuming process, ethical concerns related to animal sacrifice, and an insufficient cell population for molecular and biochemical analyses. To address these issues, several Schwann cell lines derived from schwannoma tissues and long-term primary cultures have been developed as novel in vitro models for DPN (10, 11). Notably, spontaneously immortalized Schwann cell lines exhibit high proliferative activity while largely retaining the distinct phenotypes of Schwann cells, making them ideal for studying pathogenic mechanisms (12). Although the detailed mechanisms underlying cell immortalization remain unclear, this phenomenon is thought to represent an early step in the progression toward cellular transformation (13). Unlike primary cultured cells, which have a limited lifespan, immortalized cells can be passaged indefinitely and are not contact-inhibited when cultures reach confluence. However, most immortalized Schwann cells exhibit mitogenic responses to growth factors (e.g., transforming growth factor-β, platelet-derived growth factor, acidic and basic fibroblast growth factors, etc.) and secrete various neurotrophic factors in a manner similar to primary cultured Schwann cells (13–18). Additionally, both primary cultured and immortalized Schwann cells undergo differentiation (e.g., increased expression of myelin proteins and the Krox-20 transcription factor) and dedifferentiation (e.g., increased expression of SOX2 and c-Jun) in responses to genetic manipulation, chemical stimuli, and co-culture with neurons (19–23). These findings suggest that immortalized Schwann cells retain several characteristic features of primary cultured Schwann cells. We previously reported on immortalized Schwann cell lines established in our laboratory, such as IMS32 from ICR mice (15) and IFRS1 from Fischer 344 rats (17), which have proven to be useful tools in DPN research (24). Since then, numerous studies using these cell lines have been conducted by us and other researchers. Additionally, new Schwann cell lines have been established from both normal and aldose reductase (AR)-deficient C57BL/6 mice, including 1970C3 (18), IWARS1 (25, 26), and IKARS1 (18, 25) (Table 1). In this article, we briefly summarize the findings obtained from these cell lines, which are expected to contribute to the development of pathogenesis-based therapies for DPN.
2 IMS32 cells
IMS32 cells, one of the best-characterized immortalized Schwann cells, spontaneously arose from long-term primary cultures of adult ICR mouse dorsal root ganglia (DRG) and peripheral nerves. During the purification process, complement and anti-Thy1.2 antibody were used to eliminate fibroblasts from the culture (15). IMS32 cells display distinct Schwann cell phenotypes, including spindle-shaped morphology with immunoreactivity for glial cell markers, and synthesis and secretion of neurotrophic factors; however, there have been no evidence that the cells could myelinate neurites in co-culture with neurons. The high proliferative activity of IMS32 cells might disturb continuous and stable neuron-Schwann cell interactions. Despite this problem, IMS32 cells are recognized as a useful tool to study the action mechanisms of axonal regeneration-promoting factors (29–33), as well as the pathogenesis of neurodegenerative disorders (34–36) and glial cell-associated cancer metastasis (37).
As previously reported (24), IMS32 cells have been utilized for exploring the pathogenesis of DPN, such as polyol pathway hyperactivity (27), glycation (38), and reduced NGF secretion (39). Herein, several important studies published after 2011 will be introduced.
2.1 Schwann cell dedifferentiation
As stated in Introduction [1], Schwann cells undergo dedifferentiation following peripheral nerve injury as a prerequisite for successful axonal regeneration. In contrast, Schwann cell dedifferentiation is suggested to play a pathological role in peripheral neuropathies, including hereditary neuropathy (40), autoimmune neuritis (41), chemotherapy-induced neuropathy (42), and DPN. Neureguin-1/ErbB2 signaling, which promotes Schwann cell differentiation, was impaired in the peripheral nerves of diabetic mice (43). Additionally, hyperglycemic insults induced dedifferentiation of primary cultured and IMS32 Schwann cells, evidenced by reduced expression of myelin protein zero (MPZ) and enhanced expression of p75, a low-affinity neurotrophin receptor and marker of immature Schwann cells (44). Schwann cell dedifferentiation under diabetic conditions may lead to Schwann cell death and demyelination; however, the main pathology of DPN is believed to be axonal degeneration rather than demyelination, with the latter being evident in DPN patients only at advanced stages (45). Since the interaction between axons and Schwann cells is essential for maintaining peripheral nerve function, discordance arising from Schwann cell dedifferentiation might affect both myelinated and unmyelinated fibers in DPN. Conversely, the induction of Schwann cell differentiation could be a potential therapy for DPN (46). Transplantation of human mobilized mononuclear cells (hMNC) restored the amplitude of compound muscle action potentials and MPZ expression in the sciatic nerves of diabetic nude rats. Furthermore, co-cultured hMNC induced MPZ expression, along with morphologic maturation of IMS32 cells (47). These findings suggest that the ameliorating effects of hMNC on DPN can, at least partly, be attributed to Schwann cell differentiation.
2.2 Glucosamine toxicity
The enhanced AR activity and polyol pathway flux in the PNS under hyperglycemic conditions are believed to play a major role in the development of DPN (25). When AR-deficient mice were rendered diabetic through streptozotocin (STZ) treatment, they did not exhibit overt neurological symptoms 12 weeks after the onset of diabetes (48). However, reduced nerve conduction velocities were observed in both wild-type and AR-deficient diabetic mice 16 weeks after STZ injection (49). These findings suggest that other pathways, either downstream of or independent of the polyol pathway, contribute to the development of DPN in prolonged diabetes. Metabolomics analysis revealed elevated glucosamine levels in the sciatic nerves of both wild-type and AR-deficient mice exposed to 12 weeks of diabetes. Therefore, glucosamine accumulation might be a cause of DPN independent of AR and the polyol pathway. Supporting this hypothesis, glucosamine injection into normal mice induced neurological abnormalities resembling DPN, including reduced sensory and motor nerve conduction velocities, decreased intraepidermal nerve fiber density, diminished Na+-K+-ATPase activity, and lower ATP levels in sciatic nerves. Furthermore, exogenously applied glucosamine accelerated cell death in a concentration-dependent manner (1 mM < 2.5 mM < 5 mM < 10 mM) and impaired insulin signaling (downregulating the expression of phosphorylated AKT and S6 ribosomal protein) and ATP production in IMS32 cells under both normoglycemic (5.6 mM) and hyperglycemic (30 mM) conditions (49). While the mechanism of glucosamine elevation in the PNS under diabetic conditions remains unclear, it is possible that glucosamine contributes to the development of DPN by inhibiting insulin signaling and ATP production in Schwann cells. Glucosamine can be metabolized to glucosamine-6-phosphate, a key intermediate in the hexosamine biosynthetic pathway, which is the second collateral glycolysis pathway (50, 51). Although there is currently no direct evidence of the glucosamine-induced activation of the hexosamine pathway in Schwann cells or its role in the pathogenesis of DPN (Figure 2), our ongoing study using AR-deficient Schwann cells (IKARS1) may help clarify these issues (Yako et al., in preparation).
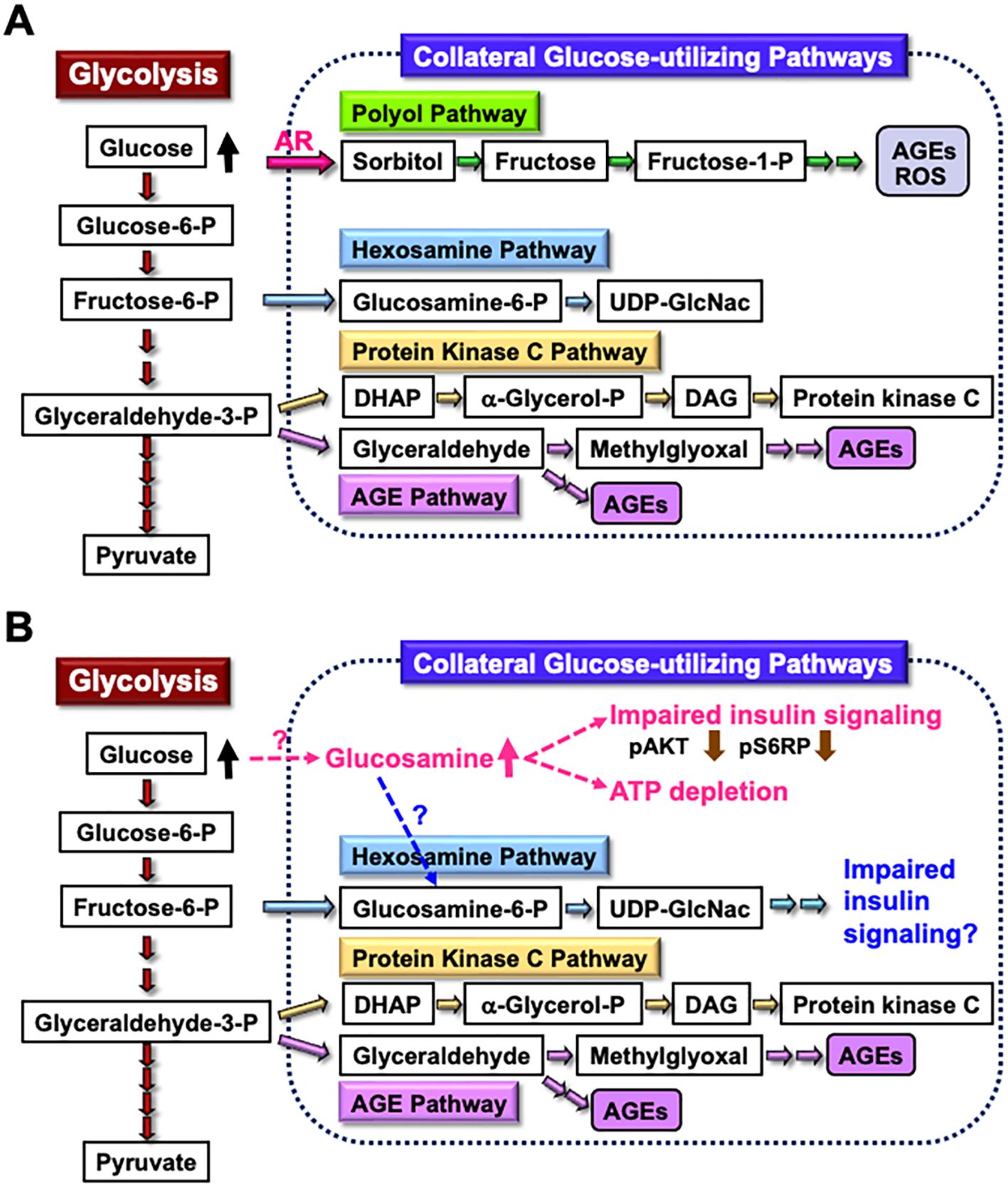
Figure 2. Glucosamine as a novel pathogenic factor in DPN. (A) Schematic representation of glycolysis and collateral glucose-utilizing pathways in Schwann cells. (B) Regardless of the presence or absence of polyol pathways, increased glucosamine uptake into Schwann cells under diabetic conditions, through a specific mechanism, may impair the insulin signaling pathway and reduce ATP synthesis. Additionally, glucosamine can be metabolized into glucosamine-6-phosphate (Glucosamine-6-P), a key intermediate in the hexosamine pathway, which may further contribute to impaired insulin signaling. These abnormalities may play a significant role in the development of DPN. Abbreviations: Glucose-6-P, glucose-6-phosphate; Fructose-6-P, fructose-6-phosphate; Glyceraldehyde-3-P, glyceraldehyde-3-phosphate; AR, aldose reductase; AGEs, advanced glycation endproducts; ROS, reactive oxygen species; UDP-GlcNAc, uridine diphosphate N-acetyl-D-glucosamine; DHAP, dihydroxyacetone phosphate; α-Glycerol-P, α-glycerol-phosphate; DAG, diacylglycerol; pS6RP, phospho-S6 ribosomal protein.
2.3 Lipotoxicity
In addition to hyperglycemia, dyslipidemia resulting from obesity and type 2 diabetes plays a pivotal role in the development and progression of DPN (52). Several studies have identified Schwann cell lipotoxicity as a pathogenic factor in DPN (53, 54). Supporting these findings, palmitate (PA), a representative saturated fatty acid, induces cell death with caspase-3 activation in IMS32 cells in a dose-dependent manner (55). PA also upregulates the protein level of CCAAT/enhancer-binding protein homologous protein (CHOP), a common marker of endoplasmic reticulum (ER) stress, in both IMS32 cells and primary cultured Schwann cells (55), as well as in rat Schwann cell line RSC96 (56, 57). These findings suggest that PA induces apoptosis via ER stress in Schwann cells. Additionally, PA is recognized as a potent ligand for Toll-like receptor 4 (TLR4), a key activator of the innate immune response (58). The TLR4 signaling pathway is suggested to contribute to PA-induced cytotoxicity associated with ER stress (59) or inflammatory responses (60). However, a previous study (55) did not find the evidence of TLR4 involvement in PA-induced apoptosis in IMS32 cells. In contrast, oxidized low-density lipoprotein (oxLDL) under high-glucose conditions triggered the cell death and upregulated TLR4 mRNA and protein expression in IMS32 cells (61). Moreover, pretreatment with TAK-242, a selective TLR4 inhibitor, attenuated oxLDL-dependent cell death and the apoptotic caspase-3 pathway under high-glucose conditions. These findings suggest that hyperactivation of TLR4 signaling by oxLDL contributes to apoptotic cell death in IMS32 cells under hyperglycemic conditions. Given that elevated LDL levels are a risk factor for DPN (52, 62), TLR4 signaling represents a potential therapeutic target for DPN.
2.4 Oxidative stress
Reactive oxygen species (ROS), highly reactive forms of molecular oxygen, have detrimental effects on cells and tissues by inducing DNA fragmentation and the oxidation of proteins and lipids. Oxidative stress is defined as an imbalance between ROS production and accumulation, and the antioxidant defense system’s ability to detoxify ROS (63). Under diabetic conditions, ROS production in the PNS is enhanced by several metabolic disorders, including glucose autooxidation, polyol pathway hyperactivity, advanced glycation endproducts (AGEs)−receptor for AGEs (RAGE) interactions, and abnormal protein kinase C activity. Additionally, polyol pathway hyperactivity contributes to the loss of endogenous antioxidants, such as taurine and reduced glutathione (GSH). Osmotic pressure from sorbitol accumulation inhibits taurine intake, while AR competes with glutathione reductase (GR) for nicotinamide adenosine dinucleotide phosphate (NADPH); excessive NADPH consumption by AR can lead to GR inhibition and GSH depletion (25, 64). We (27) and others (65) have demonstrated enhanced AR activity/expression in IMS32 cells exposed to high-glucose conditions. In the latter study, increased AR activity was accompanied by elevated O2- production, lipid peroxidation, and caspase 3 activity. These findings suggest causal relationships among the polyol pathway, oxidative stress, and apoptosis signaling.
Oxidative stress is a major therapeutic target for DPN, and α-lipoic acid (ALA) has been approved as an antioxidant treatment for DPN in several countries (66). In addition to ALA, omega-3 polyunsaturated fatty acids (ω-3 PUFAs), such as docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA), have demonstrated antioxidant and anti-inflammatory effects in diabetic conditions (67) and may be effective for DPN. Pretreatment with DHA and EPA alleviated IMS32 cell death caused by exposure to tert-butyl hydroperoxide (tBHP), an exogenous inducer of oxidative stress (68). The protective activities of DHA and EPA can be, at least in part, attributed to upregulation of endogenous antioxidant enzymes, such as heme oxygenase-1 and catalase. In another study, DHA protected PA-induced cell death in primary cultured rat Schwann cells through the activation of phosphatidyl inositol-3-kinase (PI3K)/AKT and mammalian target of rapamycin C2 kinase pathways (69); however, it remains unknown whether these pathways are involved in the DHA-induced upregulation of the antioxidant enzymes.
Stachybotrys microspora triprenyl phenols (SMTPs) are a family of triprenyl phenol metabolites derived from the fungus S. microspore. Among the SMTPs, SMTP-44D has been shown to exhibit antioxidant and anti-inflammatory effects on the nervous system (70). Administration of SMTP-44D ameliorated mechanical allodynia, thermal hyperalgesia, decreases in nerve conduction velocity and nerve blood flow, as well as increases in inflammatory molecules (e.g., tumor necrosis factor-α, interleukin (IL)-1β, IL-6, and malondialdehyde (MDA)) in the sciatic nerves of STZ-diabetic mice (71). In agreement with this study, SMTP-44D attenuated the upregulation of oxidative stress markers and inflammatory molecules, including NADPH oxidase-1, MDA, IL-6, and monocyte chemotactic protein 1, in IMS32 cells under hyperglycemic conditions (72). SMTP-44D also inhibited the enhanced activity of soluble epoxide hydrolase (sEH), which hydrolyzes epoxyeicosatrienoic acids (EETs) to dihydroxyeicosatrienoic acids (DHETs). Since EETs are potent endogenous signaling molecules associated with anti-inflammatory reactions, the protective effects of SMTP-44D in diabetic conditions may be mediated by its inhibition of sEH to sustain EET levels. These findings suggest the potential efficacy of SMTP-44D for DPN through its antioxidant and anti-inflammatory activities.
In addition to hyperglycemia, hypoglycemia due to intensive diabetes therapy and fluctuating glucose levels (glycemic variability) can trigger oxidative stress (73, 74). Recurrent short-term hyperglycemic and hypoglycemic conditions have been used as an in vitro model of glycemic variability, leading to enhanced oxidative stress and apoptosis in endothelial cells (75, 76), cardiomyocytes (77), and astrocytes (78), compared to constant hyperglycemic conditions. Similarly, intermittent short-term low and high glucose levels induced oxidative stress and apoptotic cell death in IMS32 cells (79). Since 4-phenyl butyric acid, an endoplasmic reticulum (ER) stress inhibitor, suppressed the cell death and oxidative stress induced by these conditions, glycemic variability-induced apoptosis and oxidative stress in Schwann cells may be mediated by ER stress responses (80). These findings suggest that good glycemic control to avoid hyperglycemia, hypoglycemia, and glucose fluctuation could prevent the onset and progression of DPN (81).
2.5 Pyruvate as a key molecule for ATP production under high glucose conditions
Endogenous pyruvate, produced from glucose through glycolysis, plays a key role in energy production, while exogenous pyruvate, taken up by cells via specific transporters, functions as a glycolysis accelerator and an antioxidant (82). However, the significance of pyruvate under diabetic conditions has remained unclear. In our study (83), exposure to high glucose (> 15 mM) in the absence of pyruvate led to rapid and extensive IMS32 cell death. Similarly, primary cultured adult rat DRG neurons, mouse mesangial MES13 cells, and human aortic endothelial cells underwent rapid cell death after exposure to the high-glucose conditions in the absence of pyruvate. Metabolome analysis revealed that the levels of pyruvate and certain TCA cycle intermediates, including 2-oxoglutarate, were significantly reduced in IMS32 cells under the high-glucose and pyruvate-starved conditions, and supplementation with these intermediates prevented cell death. Furthermore, exposure of IMS32 cells to these conditions resulted in a significant decrease in glycolytic flux and mitochondrial respiration, accompanied by enhanced flux through the polyol and other collateral glycolysis pathways. In our subsequent study using an inhibitor of poly-(ADP-ribose) polymerase (PARP), PARP activation under the high-glucose and pyruvate-starved conditions could contributes to the reduced glycolytic ATP production through the inhibition of glyceraldehyde-3-phosphate dehydrogenase (Figure 3). In contrast, PARP is unlikely to play a role in the impaired mitochondrial ATP production under those conditions (84). These findings suggest that exogenous pyruvate plays a crucial role in maintaining ATP production under high-glucose conditions through PARP-dependent glycolysis and PARP-independent TCA cycle in various cell types, including Schwann cells. Since supplementation of sodium pyruvate, an investigational drug for mitochondrial disease (85), ameliorated mechanical hypoalgesia and improved intraepidermal nerve fiber density in the lower limb of STZ-diabetic mice (Yako et al., in preparation), its potential therapeutic efficacy for DPN is promising.
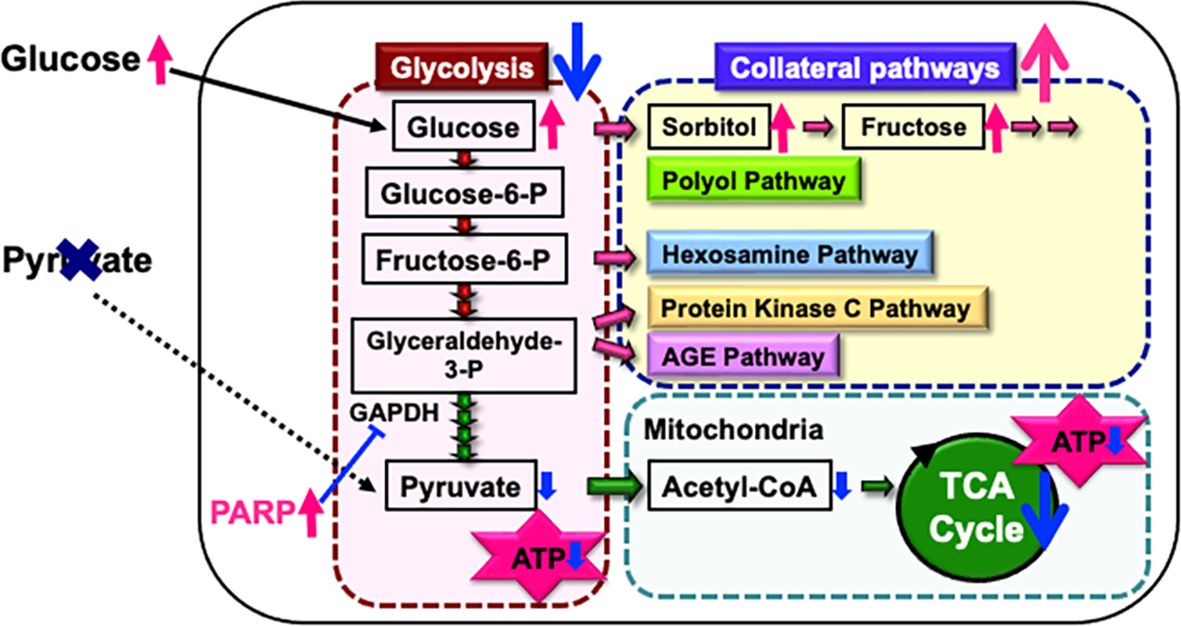
Figure 3. Possible mechanisms of IMS32 cell death under high-glucose and pyruvate-starved conditions. Deprivation of exogenous pyruvate reduces TCA cycle intermediates and mitochondrial ATP production, subsequently inhibiting glycolytic flux. Furthermore, enhanced activity of PARP under these conditions contributes to the suppression of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) activity. This cascade results in the inhibition of glycolysis and a metabolic shift toward collateral pathways.
3 IWARS1, IKARS1, and 1970C3 cells
IWARS1 and IKARS1 cells spontaneously arose from long-term primary cultures of adult wild-type and AR-deficient C57BL/6 mouse DRG and peripheral nerves, respectively (48). Schwann cell-enriched cultures were maintained under serum-free conditions in the presence of neuregulin-β, where fibroblasts ceased to grow. In the first attempt, IKARS1 cells were successfully established, but not IWASR1 cells. Therefore, 1970C3 cells, which were established from normal C57BL/6 mice, were used as a control for IKARS1 cells (18) until IWARS1cells were obtained in a second attempt. All the cell lines display distinct Schwann cell phenotypes, such as spindle-shaped morphology with intense immunoreactivity for glial cell markers and the synthesis and secretion of neurotrophic factors (18, 25). However, no studies have been conducted to determine whether these cells possess the capability to myelinate neurites in co-culture with neurons.
3.1 Physiological roles of AR
Enhanced AR activity and polyol pathway flux under hyperglycemic conditions have been implicated as a major cause of DPN and other diabetic complications. However, the physiological roles of the polyol pathway remain largely unclear. A recent study (86) suggests that the polyol pathway monitors intracellular glucose levels and regulates metabolic activities in response to glucose availability, but this function has not been verified in Schwann cells or the PNS. AR is a member of aldo-keto reductase (AKR) superfamily and participates in the detoxification of numerous aldehydic substances (87). Exposure to reactive aldehydes, including 3-deoxyglucosone, methylglyoxal (MG), and 4-hydroxynonenal (4HNE), significantly upregulated the mRNA expression of AKR1B7 and AKR1B8 in IKARS1 cells, but not in 1970C3 cells (18). Since no significant differences in viability between these two cells were observed after exposure to these aldehydes, aldehyde detoxification might be carried out by AKR1B7 and AKR1B8 in the absence of AR (aka AKR1B3). In addition to AKRs, the glyoxalase system is responsible for MG detoxification. Schwann cells deficient in glyoxalase 1 (GLO1) created using the CRISPR/Cas9 technique, did not show elevated MG concentrations. However, AR inhibition in GLO1-deficient Schwann cells increased intracellular MG levels and oxidative stress, suggesting that AR can compensate for the loss of GLO1 (88).
3.2 Glucoselysine as a novel AGE in the polyol pathway
In the second step of the polyol pathway, sorbitol is converted into fructose by sorbitol dehydrogenase (SDH). Fructose is further metabolized into dicarbonyl compounds such as 3-deoxyglucosone (3-DG) and MG, both recognized as potent glycating agents that contribute to the formation of AGEs. In addition to AGE-induced Schwann cell injury (89), 3-DG and MG have been shown to exert direct toxicity against Schwann cells (38, 90, 91). Recent studies have highlighted the AGEs produced from both exogenous (diet-derived) and endogenous (polyol pathway-derived) fructose as novel pathogenic factors in various diseases, including diabetic complications (92). Glucoselysine (GL) has been identified as a novel AGE primarily produced from fructose, and GL levels were found to increase in the eye lenses of STZ-diabetic rats in a time-dependent manner (93). Furthermore, exposure to high-glucose conditions increased both intracellular and extracellular GL levels in IWARS1 cells, but not in IKARS1 cells (28). Since the polyol pathway is absent in IKARS1 cells, it is likely that GL is produced via the polyol pathway and released from Schwann cells under diabetic conditions. A clinical investigation involving patients with type 2 diabetes and healthy participants revealed that serum GL levels were significantly higher in the diabetic patients. Moreover, GL levels in these patients were correlated with the duration of diabetes, as well as the presence of renal dysfunction and vascular complications (28). GL is expected to be a valuable biomarker for assessing the severity of DPN and other complications, as well as a potential therapeutic target in the polyol pathway-related pathogenesis.
3.3 Proteostatic disturbances
In addition to combined use with IKARS1 cells, IWARS1 cells can also be used individually as one of the mouse Schwann cell lines, similar to IMS32 cells. In our recent study (26), findings from a Drosophila model of DPN were further validated through proteome analyses using IWARS1 cells. High-sugar diet (HSD)-fed flies developed hyperglycemia and reduced insulin sensitivity, subsequently displaying DPN-like phenotypes, such as impaired noxious heat avoidance and atrophy of leg sensory neurons. Genetic screening of these flies identified the proteasome 26S subunit, non-ATPase 9 (PSMD9), as one of the modifier genes associated with impaired heat avoidance. PSMD9 gene polymorphisms have been linked to DPN risk (94), and PSMD9 is involved in proteasome activity (95). These findings suggest that proteasome activity via PSMD9 is linked to sensory dysfunction in HSD-fed flies. Supporting this hypothesis, glia-specific PSMD9 knockdown or proteasome inhibition suppressed the effects of HSD. Additionally, treatment with Ixazomib, an oral proteasome inhibitor, alleviated heat avoidance impairment and prevented atrophic changes in leg sensory neurons in HSD-fed flies. Subsequent proteome analyses using IWARS1 cells revealed that Ixasomib upregulated heat shock proteins (HSPs), including HSP40 and HSP70, suggesting that HSPs play a role in Ixazomib’s restorative effects. Furthermore, glia-specific knockdown of HSP40 or related genes negated the effects of Ixazomib. The glial proteasome is thus a promising therapeutic targets for DPN, and the exosomal secretion of HSPs from the glia may mediate the protective effect of proteasome inhibition (96).
4 IFRS1 cells
IFRS1 cells spontaneously arose from long-term primary cultures of adult Fischer 344 rat DRG and peripheral nerves under serum-free conditions in the presence of neuregulin-β and forskolin (17). IFRS1 cells display distinct Schwann cell phenotypes, including a spindle-shaped morphology with intense immunoreactivity for glial cell markers, as well as the synthesis and secretion of neurotrophic molecules (97, 98). Additionally, IFRS1 cells have been shown to myelinate neurites in co-culture with primary cultured adult rat DRG neurons (17), NGF-primed PC12 cells (21), rat neural stem cell-derived neurons, mouse embryonic stem cell-derived motor neurons (99), and NSC-34 motor neuron-like cells (100). Due to their capability to form myelin structure, IFRS1 cells are advantageous for studying the molecular mechanisms of myelination (101, 102) and demyelination (103, 104).
Unlike IMS32 cells, IFRS1 cells do not appear useful for studying the polyol pathway; exposure of IFRS1 cells to high-glucose conditions did not increase intracellular sorbitol and fructose levels (Sango et al., unpublished data). However, these cells have been utilized to explore other pathogenic factors in DPN, including impaired insulin signaling, glycation, and oxidative stress-induced autophagy.
4.1 Insulin signaling
Insulin plays a central role in regulating blood glucose levels, and impaired insulin signaling in muscle and adipose tissue is a known contributor to type 2 diabetes. It is important to note that insulin receptors are present not only in these tissues, where insulin regulates blood glucose uptake, but also in neurons and Schwann cells in the PNS (105, 106), where blood uptake occurs in an insulin-independent manner. These findings suggest that insulin may have neurotrophic and neuroprotective roles (107, 108), and that impaired insulin signaling in the PNS may contribute to the pathogenesis of DPN, as discussed in the context of glucosamine toxicity [2.2].
Insulin receptors have been identified in IFRS1 cells, where insulin application induces phosphorylation of AKT, mitogen-activated protein kinase kinase (MEK), and extracellular signal-regulated kinase (ERK) (22). Further studies using specific inhibitors for PI3K/AKT and MEK/ERK signaling pathways revealed that short-term insulin treatment promotes IFRS cell proliferation, likely through the PI3K/Akt pathway rather than the MEK/ERK pathway. In contrast, long-term insulin exposure upregulated the protein expression of MPZ via the MEK/ERK pathway and myelin basic protein (MBP) via the PI3K/AKT pathway. These findings indicate that the PI3K/AKT and MEK/ERK pathways are involved in insulin-induced proliferation, survival and differentiation of Schwann cells in distinct ways. Supporting this idea, MEK/ERK signaling has been shown to regulate Schwann cell mitosis (109), while disrupting insulin-PI3K/AKT signaling in Schwann cells leads to sensory neuropathy with impaired myelination (110). Targeting PI3K/AKT and MEK/ERK pathways in the PNS may thus represent a novel therapeutic strategy against DPN (111).
4.2 Glycation
Galectin-3 (GAL-3), a member of the β-galactoside-binding animal lectin family, is involved in various cell-to-cell and cell-to-matrix interactions. Recent studies have highlighted both physiological and pathological roles of GAL-3 in nervous tissue (112). Similar to RAGE, GAL-3 is recognized as an AGE-binding protein; however, the actions of these two proteins appear to be oppositional under diabetic conditions. While the AGEs-RAGE interaction can contribute to the development of DPN and other diabetic complications (113, 114), GAL-3 may act as a cytoprotective molecule by alleviating AGEs toxicity (115, 116). Exposure of IFRS1 cells to high glucose (30 mM) and 3-deoxyglucosone (3-DG; 0.2 mM), a precursor of AGEs, induced an upregulation of GAL-3 expression. Additionally, treatment with exogenous recombinant mouse GAL-3 (1 µg/mL) led to an upregulation of the anti-apoptotic marker Bcl-2 and a downregulation of the oxidative stress marker 4HNE in IFRS1 cells (91). These findings suggest that increased GAL-3 expression in Schwann cells under diabetes-mimicking conditions may play a pivotal role against DPN progression, although its precise mechanisms of action remain to be elucidated.
In addition to AGEs, their precursors−including MG, glyoxal, 3-DG, glyceraldehyde and glycolaldehyde (GA)−have shown detrimental effects on neurons and Schwann cells (38, 91, 117, 118). Among these, GA has been identified as the most harmful metabolite for IFRS1 cells and ND7/23 sensory neuron-like cells (118). Further analysis suggests that c-jun N-terminal kinase (JNK) and p-38 mitogen-activated kinase (p-38 MAPK) signaling pathways are involved in GA-induced ND7/23 cell death. It remains unclear whether GA toxicity against IFRS1 cells (118) and primary cultured rat Schwann cells (117) is associated with the activation of JNK and/or p-38 MAPK pathways. Nevertheless, GA and GA-induced intracellular AGEs accumulation may lead to ER stress, thereby being potentially contributing to DPN and other complications (117, 119).
4.3 Oxidative stress-induced autophagy
Autophagy is a catabolic process that maintains cellular homeostasis by eliminating damaged intracellular components through lysosomal degradation. Dysregulation of autophagy contributes to the progression of various diabetic complications, including DPN (120). It has been suggested that either excessive or impaired autophagy in Schwann cells under hyperglycemic conditions is associated with DPN pathogenesis (11, 121). Since autophagy is activated in response to increased ROS production (122), ROS accumulation induced by hyperglycemia in Schwann cells may enhance autophagic reactions.
The antioxidant activities of DHA toward IMS32 cells (68) and its potential efficacy in DPN has been described in the context of oxidative stress [2.4]. In a subsequent study (123), DHA pretreatment was shown to alleviate tBHP-induced oxidative stress, excessive autophagy, and cell death through the AMP-activated protein kinase-dependent signaling pathway in IFRS1 cells. In another study, melatonin reduced high glucose-induced ER stress and autophagy in RT4-D6P2T rat Schwann cells (124). These findings suggest that excessive autophagy induced by oxidative and/or ER stress in Schwann cells under diabetic conditions may be a viable therapeutic target for DPN. However, further evidence from animal and clinical studies is required to verify this hypothesis.
5 Translational findings in patients with DPN
This article summarizes the major findings on both classical and novel pathogenic factors of DPN identified using rodent Schwan cell lines established in our laboratory, including IMS32, 1970C3, IWARS1, IKARS1, and IFRS1. Additionally, we introduce potential therapeutic approaches targeting these factors. Some of these findings may have translational relevance for DPN in humans.
5.1 Schwann cell differentiation
Schwann cell dedifferentiation is considered a key pathogenic factor in peripheral nerve disorders, including DPN. Conversely, promoting Schwann cell differentiation may have therapeutic potential for DPN [2.1]. A promising candidate in this regard is ONO-2910 ((E)-3-(2-((5-(3-(phenylsulfonamido)phenyl)pent-4-en-1-yl)oxy)phenyl)propanoic acid, a novel Schwann cell differentiation enhancer developed by Ono Pharmaceutical Co., LTD, Osaka, Japan. Clinical trials for ONO-2910 in patients with DPN are currently underway.
5.2 Imeglimin as a promising antioxidant remedy for DPN
Imeglimin, a novel anti-hyperglycemic agent available in Japan, exerts a unique mechanism targeting mitochondrial dysfunction. Mitochondrial dysfunction is a key contributor to impaired glucose uptake in muscles, excessive gluconeogenesis in the liver, and increased pancreatic β-cell apoptosis. By protecting mitochondrial function and reducing ROS production, imeglimin improves glycemic control (125). Given that oxidative stress is a significant pathogenic factor for DPN and other diabetic complications [2.4], imeglimin may offer benefits beyond glycemic control, ameliorating these conditions. Our ongoing study demonstrates that imeglimin alleviates oxidative stress and apoptotic cell death in IMS32 cells exposed to high-glucose, low-glucose, and recurrent glucose fluctuation conditions (Kato et al., in preparation). Furthermore, a recent clinical study highlighted imeglimin’s favorable effects on body weight and lipoprotein profiles in type 2 diabetes patients (126). However, further studies are needed to evaluate its efficacy for chronic complications, including DPN.
5.3 The efficacy of GLP-1 receptor agonists for DPN
Exendin-4 (Ex-4), a glucagon-like peptide-1 receptor agonist (GLP-1RA), has demonstrated efficacy in ameliorating DPN in STZ-diabetic mice, independent of its blood glucose-lowering effects (127). Consistently, Ex-4 has been shown to enhance the survival and neurite outgrowth of rat DRG neurons (128), promote survival/proliferation and migration of IFRS1 Schwann cells, and myelination in DRG neurons-IFRS1 co-cultures (101). These findings suggest neuroprotective properties of Ex-4 and its potential role in targeting DPN. However, evidence supporting the efficacy of Ex-4 and other GLP-1RAs in humans remains controversial (129). A recent study associated Ex-4 therapy with improvements of nerve excitability in patients with type 2 diabetes (130). Additionally, GLP-1RA therapy has shown improvements in nerve conduction velocities and axonal excitability, and morphological abnormalities assessed using peripheral nerve ultrasonography in DPN patients (131). Nonetheless, further clinical research is required to establish the therapeutic efficacy of GLP-1RA for DPN.
6 Conclusion
Despite extensive research efforts, no FDA-approved disease-modifying therapies for DPN currently exist. We hope this article will aid researchers studying DPN in gaining a deeper understanding of the unique characteristics of the immortalized Schwann cells described here and facilitate their use in developing effective treatments (Figure 4).
Author contributions
KS: Funding acquisition, Project administration, Supervision, Writing – original draft, Writing – review & editing. HY: Writing – original draft, Writing – review & editing. NN: Writing – original draft, Writing – review & editing. ST: Writing – original draft, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. Our work reported in this review was supported by Grants-in-aid for Scientific Research from the Ministry of Education, Science, Sports and Culture of Japan (JSPS KAKENHI 24K10032) and was partly performed in the Cooperative Research Project Program of the Medical Institute of Bioregulation, Kyushu University.
Acknowledgments
We thank Drs. Sookja K. Chung, Koichi Kato, Keiko Naruse, Hiroki Mizukami, Junji Yamauchi, and Mari Suzuki for helpful suggestions on our experiments.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Generative AI statement
The author(s) declare that Generative AI was used in the creation of this manuscript. The whole manuscript was written by the authors, and CHAT GPT was used for English editing in some sections.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Bunge RP. Expanding roles for the Schwann cell: ensheathment, myelination, trophism and regeneration. Curr Opin Neurobiol. (1993) 3:805–9. doi: 10.1016/0959-4388(93)90157-t
2. Fornaro M, Marcus D, Rattin J, Goral J. Dynamic environmental physical cues activate mechanosensitive responses in the repair Schwann cell phenotype. Cells. (2021) 10:425. doi: 10.3390/cells10020425
3. Hanewinckel R, van Oijen M, Ikram MA, van Doorn PA. The epidemiology and risk factors of chronic polyneuropathy. Eur J Epidemiol. (2016) 31:5–20. doi: 10.1007/s10654-015-0094-6
4. Iqbal Z, Azmi S, Yadav R, Ferdousi M, Kumar M, Cuthbertson DJ, et al. Diabetic peripheral neuropathy: epidemiology, diagnosis, and pharmacotherapy. Clin Ther. (2018) 40:828–49. doi: 10.1016/j.clinthera
5. Sango K, Yako H, Takaku S, Niimi N. Pathogenic mechanisms of diabetic neuropathy. Brain Nerve. (2024) 76:671–80. doi: 10.11477/mf.1416202658
6. Guo G, Chen J, Shen Q, Chen Z. Single-cell transcriptome analysis reveals distinct cell populations in dorsal root ganglia and their potential roles in diabetic peripheral neuropathy. PloS One. (2024) 19:e0306424. doi: 10.1371/journal.pone.0306424
7. Scarpini E, Doronzo R, Baron P, Moggio M, Basellini A, Scarlato G. Phenotypic and proliferative properties of Schwann cells from nerves of diabetic patients. Int J Clin Pharmacol Res. (1992) 12:211–5.
8. Karihaloo AK, Joshi K, Chopra JS. Effect of sorbinil and ascorbic acid on myo-inositol transport in cultured rat Schwann cells exposed to elevated extracellular glucose. J Neurochem. (1997) 69:2011–8. doi: 10.1046/j.1471-4159.1997.69052011.x
9. Suzuki T, Mizuno K, Yashima S, Watanabe K, Taniko K, Yabe-Nishimura C. Characterization of polyol pathway in Schwann cells isolated from adult rat sciatic nerves. J Neurosci Res. (1999) 57:495–503. doi: 10.1002/(SICI)1097-4547(19990815)57:4<495::AID-JNR9>3.0.CO;2-Y
10. Kamiya H, Nakamura J, Hamada Y, Nakashima E, Naruse K, Kato K, et al. Polyol pathway and protein kinase C activity of rat Schwannoma cells. Diabetes Metab Res Rev. (2003) 19:131–9. doi: 10.1002/dmrr.354
11. Du W, Wang N, Li F, Jia K, An J, Liu Y, et al. STAT3 phosphorylation mediates high glucose-impaired cell autophagy in an HDAC1-dependent and -independent manner in Schwann cells of diabetic peripheral neuropathy. FASEB J. (2019) 33:8008–21. doi: 10.1096/fj.201900127R
12. De Vries GH, Boullerne AI. Glial cell lines: an overview. Neurochem Res. (2010) 35:1978–2000. doi: 10.1007/s11064-010-0318-9
13. Eccleston PA, Mirsky R, Jessen KR. Spontaneous immortalisation of Schwann cells in culture: short-term cultured Schwann cells secrete growth inhibitory activity. Development. (1991) 112:33–42. doi: 10.1242/dev.112.1.33
14. Bolin LM, Shooter EM. Characterization of a Schwann cell neurite-promoting activity that directs motoneuron axon outgrowth. J Neurosci Res. (1994) 37:23–35. doi: 10.1002/jnr.490370105
15. Watabe K, Fukuda T, Tanaka J, Honda H, Toyohara K, Sakai O. Spontaneously immortalized adult mouse Schwann cells secrete autocrine and paracrine growth-promoting activities. J Neurosci Res. (1995) 41:279–90. doi: 10.1002/jnr.490410215
16. Müller-Ostermeyer F, Claus P, Grothe C. Distinctive effects of rat fibroblast growth factor-2 isoforms on PC12 and Schwann cells. Growth Factors. (2001) 19:175–91. doi: 10.3109/08977190109001085
17. Sango K, Yanagisawa H, Kawakami E, Takaku S, Ajiki K, Watabe K. Spontaneously immortalized Schwann cells from adult Fischer rat as a valuable tool for exploring neuron-Schwann cell interactions. J Neurosci Res. (2011) 89:898–908. doi: 10.1002/jnr.22605
18. Niimi N, Yako H, Takaku S, Kato H, Matsumoto T, Nishito Y, et al. A spontaneously immortalized Schwann cell line from aldose reductase-deficient mice as a useful tool for studying polyol pathway and aldehyde metabolism. J Neurochem. (2018) 144:710–22. doi: 10.1111/jnc.14277
19. Bharucha VA, Peden KW, Tennekoon GI. SV40 large T antigen with c-Jun down-regulates myelin P0 gene expression: a mechanism for papovaviral T antigen-mediated demyelination. Neuron. (1994) 12:627–37. doi: 10.1016/0896-6273(94)90218-6
20. Thi AD, Evrard C, Rouget P. Proliferation and differentiation properties of permanent Schwann cell lines immortalized with a temperature-sensitive oncogene. J Exp Biol. (1998) 201:851–60. doi: 10.1242/jeb.201.6.851
21. Sango K, Kawakami E, Yanagisawa H, Takaku S, Tsukamoto M, Utsunomiya K, et al. Myelination in coculture of established neuronal and Schwann cell lines. Histochem Cell Biol. (2012) 137:829–39. doi: 10.1007/s00418-012-0934-3
22. Saiki T, Nakamura N, Miyabe M, Ito M, Minato T, Sango K, et al. The effects of insulin on immortalized rat Schwann cells, IFRS1. Int J Mol Sci. (2021) 22:5505. doi: 10.3390/ijms22115505
23. Murakami T, Ito Y, Sango K, Watabe K, Sunada Y. Human transthyretin gene expression is markedly increased in repair Schwann cells in an in vitro model of hereditary transthyretin amyloidosis. Neurochem Int. (2023) 164:105507. doi: 10.1016/j.neuint.2023.105507
24. Sango K, Yanagisawa H, Takaku S, Kawakami E, Watabe K. Immortalized adult rodent Schwann cells as in vitro models to study diabetic neuropathy. Exp Diabetes Res. (2011) 2011:374943. doi: 10.1155/2011/374943
25. Niimi N, Yako H, Takaku S, Chung SK, Sango K. Aldose reductase and the polyol pathway in Schwann cells: old and new problems. Int J Mol Sci. (2021) 22:1031. doi: 10.3390/ijms22031031
26. Suzuki M, Kuromi H, Shindo M, Sakata N, Niimi N, Fukui K, et al. A Drosophila model of diabetic neuropathy reveals a role of proteasome activity in the glia. iScience. (2023) 26:106997. doi: 10.1016/j.isci.2023.106997
27. Sango K, Suzuki T, Yanagisawa H, Takaku S, Hirooka H, Tamura M, et al. High glucose-induced activation of the polyol pathway and changes of gene expression profiles in immortalized adult mouse Schwann cells IMS32. J Neurochem. (2006) 98:446–58. doi: 10.1111/j.1471-4159.2006.03885.x
28. Yamaguchi H, Matsumura T, Sugawa H, Niimi N, Sango K, Nagai R. Glucoselysine, a unique advanced glycation end-product of the polyol pathway and its association with vascular complications in type 2 diabetes. J Biol Chem. (2024) 300:107479. doi: 10.1016/j.jbc.2024.107479
29. Fukuyama R, Ohta M, Ohta K, Saiwaki T, Fushiki S, Awaya A. A synthesized pyrimidine compound, MS-818, promotes walking function recovery from crush injury of the sciatic nerve through its indirect stimulation of Schwann cells. Restor Neurol Neurosci. (2000) 17:9–16. doi: 10.3233/RNN-2000-00135
30. Abe K, Namikawa K, Honma M, Iwata T, Matsuoka I, Watabe K, et al. Inhibition of Ras extracellular-signal-regulated kinase (ERK) mediated signaling promotes ciliary neurotrophic factor (CNTF) expression in Schwann cells. J Neurochem. (2001) 77:700–3. doi: 10.1046/j.1471-4159.2001.00286.x
31. Sango K, Tokashiki A, Ajiki K, Horie M, Kawano H, Watabe K, et al. Synthesis, localization and externalization of galectin-1 in mature dorsal root ganglion neurons and Schwann cells. Eur J Neurosci. (2004) 19:55–64. doi: 10.1046/j.1460-9568.2003.03102.x
32. Hashimoto M, Ishii K, Nakamura Y, Watabe K, Kohsaka S, Akazawa C. Neuroprotective effect of sonic hedgehog up-regulated in Schwann cells following sciatic nerve injury. J Neurochem. (2008) 107:918–27. doi: 10.1111/j.1471-4159.2008.05666.x
33. Klausmeyer A, Conrad R, Faissner A, Wiese S. Influence of glial-derived matrix molecules, especially chondroitin sulfates, on neurite growth and survival of cultured mouse embryonic motoneurons. J Neurosci Res. (2011) 89:127–41. doi: 10.1002/jnr.22531
34. Lee WC, Tsoi YK, Dickey CA, Delucia MW, Dickson DW, Eckman CB. Suppression of galactosylceramidase (GALC) expression in the twitcher mouse model of globoid cell leukodystrophy (GLD) is caused by nonsense-mediated mRNA decay (NMD). Neurobiol Dis. (2006) 23:273–80. doi: 10.1016/j.nbd.2006.03.005
35. Murakami T, Ohsawa Y, Zhenghua L, Yamamura K, Sunada Y. The transthyretin gene is expressed in Schwann cells of peripheral nerves. Brain Res. (2010) 1348:222–5. doi: 10.1016/j.brainres.2010.06.017
36. Inada R, Hirano M, Oka N, Samukawa M, Saigoh K, Suzuki H, et al. Phenotypic and molecular diversities of spinocerebellar ataxia type 2 in Japan. J Neurol. (2021) 268:2933–42. doi: 10.1007/s00415-021-10467-z
37. García-Reyes B, Kuzmanov I, Schneider R, Schneiker B, Efferz P, Kalff JC, et al. Glial cell-derived soluble factors increase the metastatic potential of pancreatic adenocarcinoma cells and induce epithelial-to-mesenchymal transition. J Cancer Res Clin Oncol. (2023) 149:14315–27. doi: 10.1007/s00432-023-05133-y
38. Ota K, Nakamura J, Li W, Kozakae M, Watarai A, Nakamura N, et al. Metformin prevents methylglyoxal-induced apoptosis of mouse Schwann cells. Biochem Biophys Res Commun. (2007) 357:270–5. doi: 10.1016/j.bbrc.2007.03.140
39. Tosaki T, Kamiya H, Yasuda Y, Naruse K, Kato K, Kozakae M, et al. Reduced NGF secretion by Schwann cells under the high glucose condition decreases neurite outgrowth of DRG neurons. Exp Neurol. (2008) 213:381–7. doi: 10.1016/j.expneurol.2008.06.017
40. Krauter D, Stausberg D, Hartmann TJ, Volkmann S, Kungl T, Rasche DA, et al. Targeting PI3K/Akt/mTOR signaling in rodent models of PMP22 gene-dosage diseases. EMBO Mol Med. (2024) 16:616–40. doi: 10.1038/s44321-023-00019-5
41. Szepanowski F, Winkelhausen M, Steubing RD, Mausberg AK, Kleinschnitz C, Stettner M. LPA1 signaling drives Schwann cell dedifferentiation in experimental autoimmune neuritis. J Neuroinflammation. (2021) 18:293. doi: 10.1186/s12974-021-02350-5
42. Koyanagi M, Imai S, Iwamitsu Y, Matsumoto M, Saigo M, Moriya A, et al. Cilostazol is an effective causal therapy for preventing paclitaxel-induced peripheral neuropathy by suppression of Schwann cell dedifferentiation. Neuropharmacology. (2021) 188:108514. doi: 10.1016/j.neuropharm.2021.108514
43. Pan P, Dobrowsky RT. Differential expression of neuregulin-1 isoforms and downregulation of erbin are associated with Erb B2 receptor activation in diabetic peripheral neuropathy. Acta Neuropathol Commun. (2013) 1:39. doi: 10.1186/2051-5960-1-39
44. Hao W, Tashiro S, Hasegawa T, Sato Y, Kobayashi T, Tando T, et al. Hyperglycemia promotes Schwann cell de-differentiation and de-myelination via sorbitol accumulation and Igf1 protein down-regulation. J Biol Chem. (2015) 290:17106–15. doi: 10.1074/jbc.M114.631291
45. Yagihashi S, Yamagishi S, Wada R. Pathology and pathogenetic mechanisms of diabetic neuropathy: correlation with clinical signs and symptoms. Diabetes Res Clin Pract. (2007) 77:S184–S189. doi: 10.1016/j.diabres.2007.01.054
46. Heinen A, Lehmann HC, Küry P. Negative regulators of Schwann cell differentiation-novel targets for peripheral nerve therapies? J Clin Immunol. (2013) 33:S18–26. doi: 10.1007/s10875-012-9786-9
47. Min SH, Kim JH, Kang YM, Lee SH, Oh BM, Han KS, et al. Transplantation of human mobilized mononuclear cells improved diabetic neuropathy. J Endocrinol. (2018) 239:277–87. doi: 10.1530/JOE-18-0516
48. Ho EC, Lam KS, Chen YS, Yip JC, Arvindakshan M, Yamagishi S, et al. Aldose reductase-deficient mice are protected from delayed motor nerve conduction velocity, increased c-Jun NH2-terminal kinase activation, depletion of reduced glutathione, increased superoxide accumulation, and DNA damage. Diabetes. (2006) 55:1946–53. doi: 10.2337/db05-1497
49. Mizukami H, Osonoi S, Takaku S, Yamagishi SI, Ogasawara S, Sango K, et al. Role of glucosamine in development of diabetic neuropathy independent of the aldose reductase pathway. Brain Commun. (2020) 2:fcaa168. doi: 10.1093/braincomms/fcaa168
50. Mizukami H, Osonoi S. Pathogenesis and molecular treatment strategies of diabetic neuropathy: collateral glucose-utilizing pathways in diabetic polyneuropathy. Int J Mol Sci. (2021) 22:94. doi: 10.3390/ijms22010094
51. Lv Y, Yao X, Li X, Ouyang Y, Fan C, Qian Y. Cell metabolism pathways involved in the pathophysiological changes of diabetic peripheral neuropathy. Neural Regen Res. (2024) 19:598–605. doi: 10.4103/1673-5374.380872
52. Stino AM, Rumora AE, Kim B, Feldman EL. Evolving concepts on the role of dyslipidemia, bioenergetics, and inflammation in the pathogenesis and treatment of diabetic peripheral neuropathy. J Peripher Nerv Syst. (2020) 25:76–84. doi: 10.1111/jns.12387
53. Padilla A, Descorbeth M, Almeyda AL, Payne K, De Leon M. Hyperglycemia magnifies Schwann cell dysfunction and cell death triggered by PA-induced lipotoxicity. Brain Res. (2011) 1370:64–79. doi: 10.1016/j.brainres.2010.11.013
54. Hinder LM, Figueroa-Romero C, Pacut C, Hong Y, Vivekanandan-Giri A, Pennathur S, et al. Long-chain acyl coenzyme A synthetase 1 overexpression in primary cultured Schwann cells prevents long chain fatty acid-induced oxidative stress and mitochondrial dysfunction. Antioxid Redox Signal. (2014) 21:588–600. doi: 10.1089/ars.2013.5248
55. Suzuki J, Akahane K, Nakamura J, Naruse K, Kamiya H, Himeno T, et al. Palmitate induces apoptosis in Schwann cells via both ceramide-dependent and independent pathways. Neuroscience. (2011) 176:188–98. doi: 10.1016/j.neuroscience.2010.11.035
56. Yang D, Xie J, Liang XC, Cui YZ, Wu QL. The synergistic effect of palmitic acid and glucose on inducing endoplasmic reticulum stress-associated apoptosis in rat Schwann cells. Eur Rev Med Pharmacol Sci. (2022) 26:148–57. doi: 10.26355/eurrev_202201_27761
57. Li J, Liu Q, Liu S, Xin H, Zhang X, Guo N. Maltol improves peripheral nerve function by inhibiting Schwann cell apoptosis via the PERK/eIF2α/CHOP pathway and MME upregulation in diabetic peripheral neuropathy. Pharm (Basel). (2024) 17:1139. doi: 10.3390/ph17091139
58. Lee JY, Sohn KH, Rhee SH, Hwang D. Saturated fatty acids, but not unsaturated fatty acids, induce the expression of cyclooxygenase-2 mediated through Toll-like receptor 4. J Biol Chem. (2001) 276:16683–9. doi: 10.1074/jbc.M011695200
59. Shen C, Ma W, Ding L, Li S, Dou X, Song Z. The TLR4-IRE1α pathway activation contributes to palmitate-elicited lipotoxicity in hepatocytes. J Cell Mol Med. (2018) 22:3572–81. doi: 10.1111/jcmm.13636
60. Xu D, Liang J, Cui M, Zhang L, Ren S, Zheng W, et al. Saturated fatty acids activate the inflammatory signalling pathway in Schwann cells: Implication in sciatic nerve injury. Scand J Immunol. (2020) 92:e12896. doi: 10.1111/sji.12896
61. Nihei W, Kato A, Himeno T, Kondo M, Nakamura J, Kamiya H, et al. Hyperglycaemia aggravates oxidised low-density lipoprotein-induced Schwann cell death via hyperactivation of Toll-like receptor 4. Neurol Int. (2024) 16:370–9. doi: 10.3390/neurolint16020027
62. Naqvi SSZH, Imani S, Hosseinifard H, Wen QL, Shahzad MN, Ijaz I, et al. Associations of serum low-density lipoprotein and systolic blood pressure levels with type 2 diabetic patients with and without peripheral neuropathy: systemic review, meta-analysis and meta-regression analysis of observational studies. BMC Endocr Disord. (2019) 19:125. doi: 10.1186/s12902-019-0453-5
63. Lushchak VI, Storey KB. Oxidative stress concept updated: Definitions, classifications, and regulatory pathways implicated. EXCLI J. (2021) 20:956–67. doi: 10.17179/excli2021-3596
64. Zhu J, Hu Z, Luo Y, Liu Y, Luo W, Du X, et al. Diabetic peripheral neuropathy: pathogenetic mechanisms and treatment. Front Endocrinol (Lausanne). (2024) 14:1265372. doi: 10.3389/fendo.2023.1265372
65. Cinci L, Corti F, Di Cesare Mannelli L, Micheli L, Zanardelli M, Ghelardini C. Oxidative, metabolic, and apoptotic responses of Schwann cells to high glucose levels. J Biochem Mol Toxicol. (2015) 29:274–9. doi: 10.1002/jbt.21695
66. Ziegler D. Pathogenetic treatments for diabetic peripheral neuropathy. Diabetes Res Clin Pract. (2023) 206:110764. doi: 10.1016/j.diabres.2023.110764
67. Mori TA, Woodman RJ, Burke V, Puddey IB, Croft KD, Beilin LJ. Effect of eicosapentaenoic acid and docosahexaenoic acid on oxidative stress and inflammatory markers in treated-hypertensive type 2 diabetic subjects. Free Radic Biol Med. (2003) 35:772–81. doi: 10.1016/s0891-5849(03)00407-6
68. Tatsumi Y, Kato A, Sango K, Himeno T, Kondo M, Kato Y, et al. Omega-3 polyunsaturated fatty acids exert anti-oxidant effects through the nuclear factor (erythroid-derived 2)-related factor 2 pathway in immortalized mouse Schwann cells. J Diabetes Investig. (2019) 10:602–12. doi: 10.1111/jdi.12931
69. Descorbeth M, Figueroa K, Serrano-Illán M, De León M. Protective effect of docosahexaenoic acid on lipotoxicity-mediated cell death in Schwann cells: Implication of PI3K/AKT and mTORC2 pathways. Brain Behav. (2018) 8:e01123. doi: 10.1002/brb3.1123
70. Shi X, Ohta Y, Shang J, Morihara R, Nakano Y, Fukui Y, et al. Neuroprotective effects of SMTP-44D in mice stroke model in relation to neurovascular unit and trophic coupling. J Neurosci Res. (2018) 96:1887–99. doi: 10.1002/jnr.24326
71. Shinouchi R, Shibata K, Hashimoto T, Jono S, Hasumi K, Nobe K. SMTP-44D improves diabetic neuropathy symptoms in mice through its antioxidant and anti-inflammatory activities. Pharmacol Res Perspect. (2020) 8:e00648. doi: 10.1002/prp2.648
72. Shinouchi R, Shibata K, Jono S, Hasumi K, Nobe K. SMTP-44D exerts antioxidant and anti-inflammatory effects through its soluble epoxide hydrolase inhibitory action in immortalized mouse Schwann cells upon high glucose treatment. Int J Mol Sci. (2022) 23:5187. doi: 10.3390/ijms23095187
73. Papachristoforou E, Lambadiari V, Maratou E, Makrilakis K. Association of glycemic indices (hyperglycemia, glucose variability, and hypoglycemia) with oxidative stress and diabetic complications. J Diabetes Res. (2020) 2020:7489795. doi: 10.1155/2020/7489795
74. Valente T, Arbex AK. Glycemic variability, oxidative stress, and impact on complications related to type 2 diabetes mellitus. Curr Diabetes Rev. (2021) 17:e071620183816. doi: 10.2174/1573399816666200716201550
75. Ge QM, Dong Y, Zhang HM, Su Q. Effects of intermittent high glucose on oxidative stress in endothelial cells. Acta Diabetol. (2010) 47:97–103. doi: 10.1007/s00592-009-0140-5
76. Risso A, Mercuri F, Quagliar L, Damante G, Ceriello A. Intermittent high glucose enhances apoptosis in human umbilical vein endothelial cells in culture. Am J Physiol Endocrinol Metab. (2001) 281(5):E924–30. doi: 10.1152/ajpendo.2001.281.5.E924
77. Chai Q, Miao J, Liu M, Zhang Z, Meng Z, Wu W. Knockdown of SGLT1 prevents the apoptosis of cardiomyocytes induced by glucose fluctuation via relieving oxidative stress and mitochondrial dysfunction. Biochem Cell Biol. (2021) 99:356–63. doi: 10.1139/bcb-2020-0491
78. Quincozes-Santos A, Bobermin LD, de Assis AM, Gonçalves CA, Souza DO. Fluctuations in glucose levels induce glial toxicity with glutamatergic, oxidative and inflammatory implications. Biochim Biophys Acta Mol Basis Dis. (2017) 1863:1–14. doi: 10.1016/j.bbadis.2016.09.013
79. Kato A, Tatsumi Y, Yako H, Sango K, Himeno T, Kondo M, et al. Recurrent short-term hypoglycemia and hyperglycemia induce apoptosis and oxidative stress via the ER stress response in immortalized adult mouse Schwann (IMS32) cells. Neurosci Res. (2019) 147:26–32. doi: 10.1016/j.neures.2018.11.004
80. Eftekharpour E, Fernyhough P. Oxidative stress and mitochondrial dysfunction associated with peripheral neuropathy in type 1 diabetes. Antioxid Redox Signal. (2022) 37:578–96. doi: 10.1089/ars.2021.0152
81. Jia Y, Long D, Yang Y, Wang Q, Wu Q, Zhang Q. Diabetic peripheral neuropathy and glycemic variability assessed by continuous glucose monitoring: A systematic review and meta-analysis. Diabetes Res Clin Pract. (2024) 213:111757. doi: 10.1016/j.diabres.2024.111757
82. Mallet RT. Pyruvate: metabolic protector of cardiac performance. Proc Soc Exp Biol Med. (2000) 223:136–48. doi: 10.1046/j.1525-1373.2000.22319.x
83. Yako H, Niimi N, Kato A, Takaku S, Tatsumi Y, Nishito Y, et al. Role of pyruvate in maintaining cell viability and energy production under high-glucose conditions. Sci Rep. (2021) 11:18910. doi: 10.1038/s41598-021-98082-w
84. Yako H, Niimi N, Takaku S, Kato A, Kato K, Sango K. Role of exogenous pyruvate in maintaining adenosine triphosphate production under high-glucose conditions through PARP-dependent glycolysis and PARP-independent tricarboxylic acid cycle. Int J Mol Sci. (2024) 25:11089. doi: 10.3390/ijms252011089
85. Koga Y, Povalko N, Inoue E, Nashiki K, Tanaka M. Biomarkers and clinical rating scales for sodium pyruvate therapy in patients with mitochondrial disease. Mitochondrion. (2019) 48:11–5. doi: 10.1016/j.mito.2019.02.001
86. Sano H, Nakamura A, Yamane M, Niwa H, Nishimura T, Araki K, et al. The polyol pathway is an evolutionarily conserved system for sensing glucose uptake. PloS Biol. (2022) 20:e3001678. doi: 10.1371/journal.pbio.3001678
87. Singh M, Kapoor A, Bhatnagar A. Physiological and Pathological Roles of Aldose Reductase. Metabolites. (2021) 11:655. doi: 10.3390/metabo11100655
88. Morgenstern J, Fleming T, Schumacher D, Eckstein V, Freichel M, Herzig S, et al. Loss of glyoxalase 1 induces compensatory mechanism to achieve dicarbonyl detoxification in mammalian Schwann cells. J Biol Chem. (2017) 292:3224–38. doi: 10.1074/jbc.M116.760132
89. Yu T, Li L, Chen T, Liu Z, Liu H, Li Z. Erythropoietin attenuates advanced glycation endproducts-induced toxicity of Schwann cells in vitro. Neurochem Res. (2015) 40:698–712. doi: 10.1007/s11064-015-1516-2
90. Fukunaga M, Miyata S, Liu BF, Miyazaki H, Hirota Y, Higo S, et al. Methylglyoxal induces apoptosis through activation of p38 MAPK in rat Schwann cells. Biochem Biophys Res Commun. (2004) 320:689–95. doi: 10.1016/j.bbrc.2004.06.011
91. Tsukamoto M, Sango K, Niimi N, Yanagisawa H, Watabe K, Utsunomiya K. Upregulation of galectin-3 in immortalized Schwann cells IFRS1 under diabetic conditions. Neurosci Res. (2015) 92:80–5. doi: 10.1016/j.neures.2014.11.008
92. Gugliucci A. Formation of fructose-mediated advanced glycation end products and their roles in metabolic and inflammatory diseases. Adv Nutr. (2017) 8:54–62. doi: 10.3945/an.116.013912
93. Ohno RI, Ichimaru K, Tanaka S, Sugawa H, Katsuta N, Sakake S, et al. Glucoselysine is derived from fructose and accumulates in the eye lens of diabetic rats. J Biol Chem. (2019) 294:17326–38. doi: 10.1074/jbc.RA119.010744
94. Gragnoli C. PSMD9 is linked to type 2 diabetes neuropathy. J Diabetes Complications. (2011) 25:329–31. doi: 10.1016/j.jdiacomp.2011.06.003
95. Sangith N, Srinivasaraghavan K, Sahu I, Desai A, Medipally S, Somavarappu AK, et al. Discovery of novel interacting partners of PSMD9, a proteasomal chaperone: Role of an atypical and versatile PDZ-domain motif interaction and identification of putative functional modules. FEBS Open Bio. (2014) 4:571–83. doi: 10.1016/j.fob.2014.05.005
96. Takeuchi T, Suzuki M, Fujikake N, Popiel HA, Kikuchi H, Futaki S, et al. Intercellular chaperone transmission via exosomes contributes to maintenance of protein homeostasis at the organismal level. Proc Natl Acad Sci USA. (2015) 112:E2497–2506. doi: 10.1073/pnas.1412651112
97. Takaku S, Yanagisawa H, Watabe K, Horie H, Kadoya T, Sakumi K, et al. GDNF promotes neurite outgrowth and upregulates galectin-1 through the RET/PI3K signaling in cultured adult rat dorsal root ganglion neurons. Neurochem Int. (2013) 62:330–9. doi: 10.1016/j.neuint.2013.01.008
98. Suttinont C, Maeno K, Yano M, Sato-Numata K, Numata T, Tsutsumi M. Role of Piezo2 in Schwann cell volume regulation and Its Impact on neurotrophic release regulation. Cell Physiol Biochem. (2024) 58:292–310. doi: 10.33594/000000713
99. Ishii T, Kawakami E, Endo K, Misawa H, Watabe K. Myelinating cocultures of rodent stem cell line-derived neurons and immortalized Schwann cells. Neuropathology. (2017) 37:475–81. doi: 10.1111/neup.12397
100. Takaku S, Yako H, Niimi N, Akamine T, Kawanami D, Utsunomiya K, et al. Establishment of a myelinating co-culture system with a motor neuron-like cell line NSC-34 and an adult rat Schwann cell line IFRS1. Histochem Cell Biol. (2018) 149:537–43. doi: 10.1007/s00418-018-1649-x
101. Takaku S, Tsukamoto M, Niimi N, Yako H, Sango K. Exendin-4 promotes Schwann cell survival/migration and myelination in vitro. Int J Mol Sci. (2021) 22:2971. doi: 10.3390/ijms22062971
102. Ali H, Morito K, Hasi RY, Aihara M, Hayashi J, Kawakami R, et al. Characterization of uptake and metabolism of very long-chain fatty acids in peroxisome-deficient CHO cells. Biochim Biophys Acta Mol Cell Biol Lipids. (2022) 1867:159088. doi: 10.1016/j.bbalip.2021.159088
103. Niimi N, Yako H, Tsukamoto M, Takaku S, Yamauchi J, Kawakami E, et al. Involvement of oxidative stress and impaired lysosomal degradation in amiodarone-induced schwannopathy. Eur J Neurosci. (2016) 44:1723–33. doi: 10.1111/ejn.13268
104. Takaku S, Sango K. Pretreatment with zonisamide mitigates oxaliplatin-induced toxicity in rat DRG neurons and DRG neuron-Schwann cell co-cultures. Int J Mol Sci. (2022) 23:9983. doi: 10.3390/ijms23179983
105. Sugimoto K, Murakawa Y, Zhang W, Xu G, Sima AA. Insulin receptor in rat peripheral nerve: its localization and alternatively spliced isoforms. Diabetes Metab Res Rev. (2000) 16:354–63. doi: 10.1002/1520-7560(200009/10)16:5<354::AID-DMRR149>3.0.CO;2-H
106. Sugimoto K, Murakawa Y, Sima AA. Expression and localization of insulin receptor in rat dorsal root ganglion and spinal cord. J Peripher Nerv Syst. (2002) 7:44–53. doi: 10.1046/j.1529-8027.2002.02005.x
107. Patel RA, Kurian P, Raizada MK, Crews FT. Insulin stimulates phosphatidylinositol 3-kinase activity in rat neuronal primary cultures. J Neurochem. (1993) 61:360–3. doi: 10.1111/j.1471-4159.1993.tb03578.x
108. Grote CW, Wright DE. A role for insulin in diabetic neuropathy. Front Neurosci. (2016) 10:581. doi: 10.3389/fnins.2016.00581
109. Chattopadhyay S, Shubayev VI. MMP-9 controls Schwann cell proliferation and phenotypic remodeling via IGF-1 and ErbB receptor-mediated activation of MEK/ERK pathway. Glia. (2009) 57:1316–25. doi: 10.1002/glia.20851
110. Hackett AR, Strickland A, Milbrandt J. Disrupting insulin signaling in Schwann cells impairs myelination and induces a sensory neuropathy. Glia. (2020) 68:963–78. doi: 10.1002/glia.23755
111. Pham VM. Targeting PI3K/AKT and MEK/ERK pathways for synergic effects on improving features of peripheral diabetic neuropathy. J Diabetes Investig. (2024) 15:1537–44. doi: 10.1111/jdi
112. Soares LC, Al-Dalahmah O, Hillis J, Young CC, Asbed I, Sakaguchi M, et al. Novel galectin-3 roles in neurogenesis, inflammation and neurological diseases. Cells. (2021) 10:3047. doi: 10.3390/cells10113047
113. Kang Q, Dai H, Jiang S, Yu L. Advanced glycation end products in diabetic retinopathy and phytochemical therapy. Front Nutr. (2022) 9:1037186. doi: 10.3389/fnut.2022.1037186
114. Osonoi S, Mizukami H, Takeuchi Y, Sugawa H, Ogasawara S, Takaku S, et al. RAGE activation in macrophages and development of experimental diabetic polyneuropathy. JCI Insight. (2022) 7:e160555. doi: 10.1172/jci.insight.160555
115. Pugliese G, Pricci F, Iacobini C, Leto G, Amadio L, Barsotti P, et al. Accelerated diabetic glomerulopathy in galectin-3/AGE receptor 3 knockout mice. FASEB J. (2001) 15:2471–9. doi: 10.1096/fj.01-0006com
116. Gallate ZS, D’Erminio DN, Nasser P, Laudier DM, Iatridis JC. Galectin-3 and RAGE differentially control advanced glycation endproduct-induced collagen damage in murine intervertebral disc organ culture. JOR Spine. (2023) 6:e1254. doi: 10.1002/jsp2.1254
117. Sato K, Tatsunami R, Yama K, Murao Y, Tampo Y. Glycolaldehyde induces endoplasmic reticulum stress and apoptosis in Schwann cells. Toxicol Rep. (2015) 2:1454–62. doi: 10.1016/j.toxrep.2015.10.014
118. Akamine T, Takaku S, Suzuki M, Niimi N, Yako H, Matoba K, et al. Glycolaldehyde induces sensory neuron death through activation of the c-Jun N-terminal kinase and p-38 MAP kinase pathways. Histochem Cell Biol. (2020) 153:111–9. doi: 10.1007/s00418-019-01830-3
119. Suzuki R, Fujiwara Y, Saito M, Arakawa S, Shirakawa JI, Yamanaka M, et al. Intracellular accumulation of advanced glycation end products induces osteoblast apoptosis via endoplasmic reticulum stress. J Bone Miner Res. (2020) 35:1992–2003. doi: 10.1002/jbmr.4053
120. Parmar UM, Jalgaonkar MP, Kulkarni YA, Oza MJ. Autophagy-nutrient sensing pathways in diabetic complications. Pharmacol Res. (2022) 184:106408. doi: 10.1016/j.phrs.2022.106408
121. Wang QQ, Zhai C, Wahafu A, Zhu YT, Liu YH, Sun LQ. Salvianolic acid B inhibits the development of diabetic peripheral neuropathy by suppressing autophagy and apoptosis. J Pharm Pharmacol. (2019) 71:417–28. doi: 10.1111/jphp.13044
122. Scherz-Shouval R, Shvets E, Fass E, Shorer H, Gil L, Elazar Z. Reactive oxygen species are essential for autophagy and specifically regulate the activity of Atg4. EMBO J. (2007) 26:1749–60. doi: 10.1038/sj.emboj.7601623
123. Tatsumi Y, Kato A, Niimi N, Yako H, Himeno T, Kondo M, et al. Docosahexaenoic acid suppresses oxidative stress-induced autophagy and cell death via the AMPK-dependent signaling pathway in immortalized Fischer rat Schwann cells 1. Int J Mol Sci. (2022) 23:4405. doi: 10.3390/ijms23084405
124. Salem HMA, Chok KC, Koh RY, Ng PY, Tiong YL, Chye SM. Melatonin ameliorates high glucose-induced autophagy in Schwann cells. Int J Biochem Mol Biol. (2023) 14:25–31.
125. Yaribeygi H, Maleki M, Sathyapalan T, Jamialahmadi T, Sahebkar A. Molecular mechanisms by which imeglimin improves glucose homeostasis. J Diabetes Res. (2020) 2020:8768954. doi: 10.1155/2020/8768954
126. Katsuyama H, Hakoshima M, Heshiki T, Iida S, Adachi H, Yanai H. Real-world effectiveness of imeglimin in patients with type 2 diabetes: A retrospective longitudinal study in Japan. Diabetes Res Clin Pract. (2024) 213:111752. doi: 10.1016/j.diabres.2024.111752
127. Himeno T, Kamiya H, Naruse K, Harada N, Ozaki N, Seino Y, et al. Beneficial effects of exendin-4 on experimental polyneuropathy in diabetic mice. Diabetes. (2011) 60:2397–406. doi: 10.2337/db10-1462
128. Tsukamoto M, Niimi N, Sango K, Takaku S, Kanazawa Y, Utsunomiya K. Neurotrophic and neuroprotective properties of exendin-4 in adult rat dorsal root ganglion neurons: involvement of insulin and RhoA. Histochem Cell Biol. (2015) 144:249–59. doi: 10.1007/s00418-015-1333-3
129. Mehta K, Behl T, Kumar A, Uddin MS, Zengin G, Arora S. Deciphering the neuroprotective role of glucagon-like peptide-1 agonists in diabetic neuropathy: current perspective and future directions. Curr Protein Pept Sci. (2021) 22:4–18. doi: 10.2174/1389203721999201208195901
130. Issar T, Kwai NCG, Poynten AM, Arnold R, Milner KL, Krishnan AV. Effect of exenatide on peripheral nerve excitability in type 2 diabetes. Clin Neurophysiol. (2021) 132:2532–9. doi: 10.1016/j.clinph.2021.05.033
Keywords: immortalized Schwann cells, diabetic peripheral neuropathy, polyol pathway, glycation, oxidative stress, autophagic and proteostatic disturbances
Citation: Sango K, Yako H, Niimi N and Takaku S (2025) Immortalized Schwann cell lines as useful tools for pathogenesis-based therapeutic approaches to diabetic peripheral neuropathy. Front. Endocrinol. 15:1531209. doi: 10.3389/fendo.2024.1531209
Received: 22 November 2024; Accepted: 24 December 2024;
Published: 21 January 2025.
Edited by:
Keiichiro Susuki, Wright State University, United StatesReviewed by:
Esma Nur Okatan, University of Istinye, TürkiyeTakashi Nishinaka, Kindai University, Japan
Copyright © 2025 Sango, Yako, Niimi and Takaku. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Kazunori Sango, sango-kz@igakuken.or.jp
 Kazunori Sango
Kazunori Sango