
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Ecol. Evol. , 15 June 2023
Sec. Phylogenetics, Phylogenomics, and Systematics
Volume 11 - 2023 | https://doi.org/10.3389/fevo.2023.1177627
Theraphosinae is the most diverse subfamily of Theraphosidae spiders, but their evolutionary history remains unresolved to date. This problem is common in taxonomic groups with phylogenetic hypotheses that have often been based only on qualitative morphological characters and, rarely, on molecular analyses. Phylogenomics has significantly contributed to the understanding of the evolution of many non-model groups, such as spiders. Herein, we employed ultraconserved elements (UCEs) phylogenomics to propose a new hypothesis for a group of Theraphosinae genera, namely Lasiodoriforms: Vitalius, Lasiodora, Eupalaestrus, Pterinopelma, Proshapalopus, and Nhandu. We propose three genera and their respective morphological diagnoses are provided. Our phylogeny supports the transfer of species from the genus Vitalius to Pterinopelma and Proshapalopus and from Proshapalopus to Eupalaestrus. Finally, we describe a new species of Vitalius from Southern Brazil. Based on these three new generic descriptions and transferred species, the Lasiodoriform tarantulas comprise nine genera from Argentina, Brazil, Paraguay, and Uruguay, and the genus Vitalius now includes seven species.
The subfamily Theraphosinae is endemic to the Neotropics and is one of twelve currently included in Theraphosidae Thorell, 1896, the family that includes tarantulas (Guadanucci, 2014; Lüddecke et al., 2018; Turner et al., 2018; Foley et al., 2019). Theraphosinae comprises the greatest diversity within the family, currently including 69 genera and more than 500 species (World Spider Catalog, 2023), distributed mainly in the Neotropics (Central America, South America, southern Mexico, and the Caribbean), as well as in the southwestern United States and northern Mexico temperate regions (Hamilton et al., 2016b; Mendoza and Francke, 2017; Pérez-Miles and Perafán, 2020).
Defining species boundaries and identifying useful morphological characters are challenging tasks when dealing with theraphosids, as they present homogeneous morphology and continuous variation across genera and species (Bertani, 2001; Fukushima and Bertani, 2017). Phylogenetic reconstructions from morphological matrices often result in low-supported clades and trees with many homoplastic characters and, consequently, discordant evolutionary hypotheses. In addition, morphological similarities are even greater in females, making it difficult to identify genera and species (Bertani et al., 2011, 2012; Bertani and Leal, 2016; Hamilton et al., 2016b; Galleti-Lima and Guadanucci, 2018; Perafán and Valencia-Cuellar, 2018).
When taking a broader look at informative morphological characters, there have been a few hypotheses among Theraphosidae subfamilies that have shown that the monophyly of Theraphosinae is supported by the following synapomorphies: keels on the male palpal bulb, the subtegulum extended over the tegulum, and the presence of different types of urticating setae (types I, III, IV, V, VI, or VII) (Raven, 1985; Pérez-Miles et al., 1996; Bertani and Guadanucci, 2013; Guadanucci, 2014; Kaderka et al., 2019). This has also been shown as monophyletic in molecular analyses (Lüddecke et al., 2018; Foley et al., 2019). Additionally, there have been hypotheses based on the morphological features that proposed to explain the interrelationships of theraphosinae genera (Pérez-Miles et al., 1996; Bertani, 2001; Bertani et al., 2011; Bertani and Guadanucci, 2013; Perafán et al., 2015; Galleti-Lima and Guadanucci, 2018; Perafán and Valencia-Cuellar, 2018; Fabiano-Da-Silva et al., 2019), as well as one phylogenetic hypothesis using molecular data, based on a single mitochondrial gene region (Turner et al., 2018).
Despite disagreements between studies, some phylogenetic lineages within Theraphosinae have been recovered as monophyletic in both morphological and molecular hypotheses. For example, clades supported by the presence of different types of urticating setae (III + IV and III + I) (Pérez-Miles et al., 1996; Turner et al., 2018), the “spoon-like” embolus clade (Bertani, 2001), and a group supported by the presence of subapical and retrolateral keels in the male palpal bulb (Bertani, 2001; Turner et al., 2018). In their phylogenetic hypothesis, Turner et al. (2018) recommended the division of Theraphosinae into three tribes: Grammostolini, tarantulas with type IV urticating setae; Hapalopini, lineage where most genera possess only type III urticating setae also known as “dwarf tarantulas” and the sister lineage of Grammostolini; and Theraphosini, tarantulas with type I urticating setae, such as Vitalius Lucas et al., 1993, and its closely related genera.
The group of tarantulas that share the presence of subapical and retrolateral keels in the male palpal bulb include the following South American genera: Lasiodora Koch, 1850, Eupalaestrus Pocock, 1901, Pterinopelma Pocock, 1901, Proshapalopus Mello-Leitão, 1923, Nhandu Lucas, 1983, and Vitalius. It has been recovered as monophyletic in recent phylogenies (Bertani, 2001; Bertani et al., 2012; Galleti-Lima and Guadanucci, 2018; Perafán and Valencia-Cuellar, 2018; Turner et al., 2018). The first study to focus on these tarantulas, carried out by Bertani (2001), produced a phylogenetic hypothesis using morphological characteristics and a taxonomic revision of Eupalaestrus, Prohapalopus, Nhandu, and Vitalius. Intrageneric relationships within Vitalius have never been fully resolved, with most species forming a poorly solved polytomic clade (Bertani, 2001; Bertani et al., 2011; Galleti-Lima and Guadanucci, 2018; Perafán and Valencia-Cuellar, 2018), and in some topologies, the genus was not recovered as monophyletic (Perafán and Valencia-Cuellar, 2018).
More recently, Bertani et al. (2011) proposed the revalidation of Pterinopelma, with P. vitiosum (Keyserling, 1891) as the type-species, and provided a description of P. sazimai (Bertani et al., 2011). In the subsequent year, Bertani et al. (2012) proposed the transfer of Vitalius nondescriptus (Mello-Leitão, 1926), which is sympatric with P. sazimai, and in 2016, Bertani and Leal (2016) described Pterinopelma felipeleitei and the male of P. sazimai, which was considered as a misidentification of the male of P. felipeleitei by Bertani et al. (2011). In addition, the authors discussed that P. sazimai perhaps should not be included in Pterinopelma due to the morphology of the male palpal bulb. The monophyly of the genus Pterinopelma, its interrelationships, and species composition are key points for understanding the phylogeny of Vitalius and Nhandu. In a phylogenetic study that aimed to understand the evolution of stridulating setae in Theraphosinae spiders (Galleti-Lima and Guadanucci, 2018), Pterinopelma was not recovered as monophyletic, and the species P. sazimai was closely related to Nhandu and distantly related from other Pterinopelma species.
New molecular sequencing techniques can be used to access hundreds or thousands of loci for phylogenetic inference. Targeted enrichment approaches (e.g., Ultraconserved Elements and Anchored Hybrid Enrichment) (Faircloth et al., 2012; Lemmon and Lemmon, 2013) have been useful for inferring robust phylogenetic relationships of the non-model groups, such as Mygalomorphae spiders, such as Aphonopelma Pocock, 1901, Aptostichus Simon, 1891, Aliatypus Smith, 1908, Antrodiaetus Ausserer, 1871, Hexurella Gertsch and Platnick, 1979, Mecicobothrium Holmberg, 1882, Megahexura Kaston, 1972, Porrhothele Simon, 1892 (Faircloth et al., 2012; Hamilton et al., 2016b; Starrett et al., 2016; Yeates et al., 2016; Branstetter et al., 2017; Hedin et al., 2018, 2019; Derkarabetian et al., 2019; Zhang et al., 2019; Kulkarni et al., 2020; Xu et al., 2021). To date, there has been only one study using either of these phylogenomic approaches in tarantulas, the North American genus Aphonopelma (Hamilton et al., 2016a). Kulkarni et al. (2020) designed a spider-specific UCE probe set, which has been recovering a high number of loci, contributing to several questions in different phylogenetic studies.
Motivated by the need for a robust phylogenetic hypothesis that includes all diversity of Vitalius, Nhandu, and Pterinopelma to support taxonomic reclassifications, we present the first phylogenomic study of Theraphosinae spiders in South America using sequences of UCEs. In addition to the representatives of Lasiodora, Eupalaestrus, Pterinopelma, Proshapalopus, Nhandu, and Vitalius, we tested the monophyly of Vitalius, including all species, and propose reclassifications for some of these genera based on the resulting topology.
All Theraphosinae species used in this research are deposited in the Coleção Aracnológica Diamantina (CAD). A list with a voucher number of these specimens is assembled in Supplementary File S1. All type-species from genera belonging to the South American lineage that possesses bulbs with retrolateral and subapical keels were included in the analysis. Because we were seeking to resolve the polytomies of the past morphological phylogenetic hypotheses, all Vitalius species were included. For outgroup comparison, we used morphologically distinct theraphosine genera from ingroup taxa (i.e., without subapical and retrolateral keels), such as Aphonopelma hentzi (Girard, 1853), Cyrtopholis portoricae Chamberlin, 1917, an unidentified Sericopelma Ausserer, 1875, and Stichoplastoris Rudloff, 1997. All taxonomic decisions were made based on node supports (>90) and diagnosable morphological differences.
Images and measurements used in morphological descriptions were obtained with a Leica M205 C stereo microscope and the Leica Application Suite V4.12 software. In the description of the new species, body length measurements include the chelicerae, but not the spinnerets. Leg segment lengths were measured in the dorsal view between the joints. The carapace, eye tubercle, labium, and sternum were measured in length and width. We followed Bertani (2001) for the use of the terminologies of structures and spination.
The specimens used for images and descriptions are deposited in the following collections: CAD, Coleção Aracnológica Diamantina, Universidade Estadual Paulista, Rio Claro, Brazil—J. P. L. Guadanucci; IBSP, Instituto Butantan, São Paulo, Brazil—A. D. Brescovit; MCN, Museu de Ciências Naturais, Fundação Zoobotânica do Rio Grande do Sul, Porto Alegre, Brazil—R. Ott; MCTP, Museu de Ciências e Tecnologia, Pontifícia Universidade Católica do Rio Grande do Sul, Porto Alegre, Brazil—R. A. Teixeira; MZSP, Museu de Zoologia da Universidade de São Paulo, São Paulo, Brazil—R. Pinto da Rocha; and UFMG, Universidade Federal de Minas Gerais, Belo Horizonte, Brazil—A. Santos.
Genomic DNA was extracted from muscle tissues using the Qiagen DNeasy Tissue KitTM (Qiagen, Valencia, CA, USA) or OmniprepTM (G-Biosciences) and then quantitatively and qualitatively assessed on agarose gel electrophoresis and a Qubit 2.0 Fluorometer (Thermo Fisher Scientific). Library prep, UCE hybridization, and high-throughput Illumina sequencing were carried out at Rapid Genomics (Gainesville, FL). DNA was sheared to a mean fragment length of 500 bp, fragments were end-repaired and A-tailed, followed by the incorporation of unique dual-indexed Illumina adaptors and PCR enrichment. Samples were equimolar pooled and sequenced on an SP flow cell (2 × 250 bp) or an S4 flow cell (2 × 150 bp). The resulting data were processed using Phyluce version 1.7.1 (Faircloth, 2016) and a combined arachnid-spider hybrid probeset (Starrett et al., 2016; Kulkarni et al., 2020), where match settings for minimum identity and minimum coverage of 85 and 85 (respectively) were used. Loci were then aligned using MAFFT (Katoh and Standley, 2013), with alignments externally trimmed and then internally trimmed using gblocks with b1, b2, b3, and b4 and settings of 0.5, 0.5, 5, and 10, respectively, to remove poorly aligned blocks within the sequences. The data were then additionally cleaned using AMAS v 1.0 (Borowiec, 2016) and SPRUCEUP (Borowiec, 2019), which removes poorly aligned sequence fragments from individual sequences within alignments. Visual examination of distance distribution plots identified a setting of 0.9 as optimal for trimming, with a 50% occupancy matrix then being generated for use in subsequent analyses. A maximum likelihood-based phylogeny was inferred from 1,375 loci using IQTree2 (Nguyen et al., 2015; Minh et al., 2020), with nodal support determined by the ultrafast bootstrap and the SH-aLRT test (Anisimova et al., 2011) for 1,000 replicates each. All 1,375 loci were modeled independently (for details on each model used, see “brazil.0.9.iqtree.log” file attached in Supplementary File S2). All analyses were performed on the University of Idaho Research Computing and Data Services (RCDS) high-performance computing cluster. Tree edits were performed using FigTree V. 1.4.4 software (Rambaut, 2018) and then edited a image editor. DNA sequence alignments and associated phylogenetic trees and data matrices, accompanying tree files, and scripts have been deposited in Supplementary File S2.
The updated new names were used in the terminal taxa of the Lasiodoriforms phylogenetic tree. Consider the prefix of the specimens in the tree as referring to the genus to which the specimen previously belonged (e.g., PROS, Proshapalopus; EUPA, Eupalaestrus; LASI, Lasiodora; PTER, Pterinopelma; NHAN, Nhandu; VITA, Vitalius).
The dataset comprised 1,375 loci (1,194,888 base pairs) with 63 terminals (6 outgroup and 57 ingroup terminals). The resulting trees from the ML analysis are shown at the genera level (Figure 1) and species level (Figure 2). The clade named herein as Lasiodoriforms was recovered and well-supported (>90) by both the SH-aLRT test and ultra-fast bootstrapping. The support values in the nodes of each genus were also high in both tests, except for the node referring to Proshapalopus multicuspidatus + Vitalius nondescriptus. It is important to note that long branches of certain lineages (e.g., P. multicuspidatus and P. felipeleitei) are because of data quality issues (i.e., gaps and missing data). The accentuated branch length is not likely to be a true representation—though the phylogenetic placement of these lineages do not appear to change following significant data cleaning. Our concatenated analysis indicated Eupalaestrus (including P. amazonicus) as a sister group of the remaining Lasiodoriforms. The node at the evolutionary divergence between P. multicuspidatus + V. nondescriptus and the remaining lineages also presented high support values for both tests. Unfortunately, the placement of P. anomalus is still uncertain, due to the low support value (bootstrapping = 86). The three morphologically distinct taxa [Pterinopelma felipeleitei (Lasiodora + Pterinopelma sazimai)] were recovered as monophyletic, where two of them would be considered monotypic genera: P. felipeleitei and P. sazimai. Vitalius wacketi was recovered as the sister group to Nhandu (Pterinopelma + Vitalius). Finally, the monophyletic relationship of Pterinopelma and Vitalius is not well-supported, with low SH-aLRT and bootstrap values (SH-aLRT = 80.5 and bootstrapping = 82).
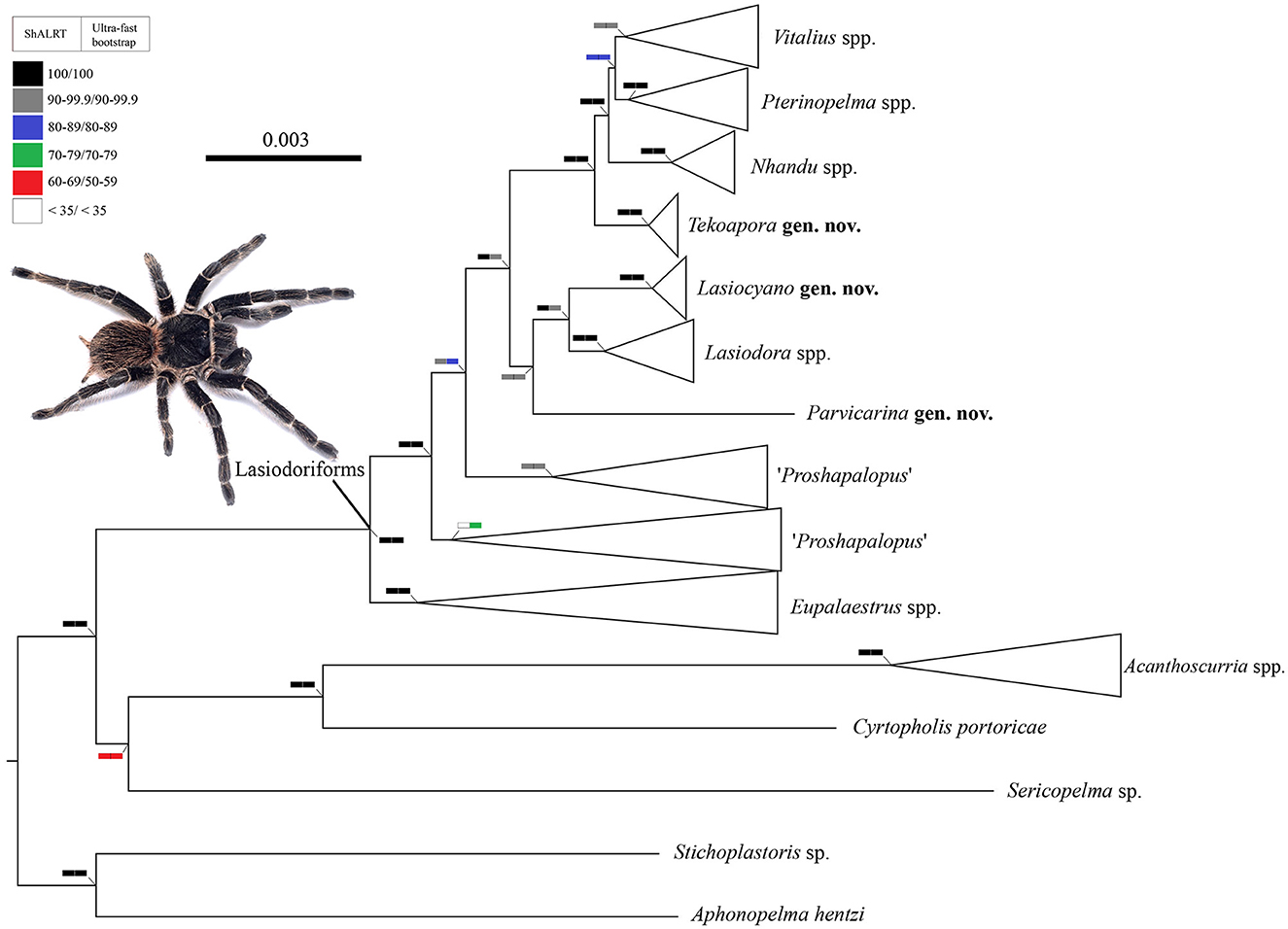
Figure 1. A cladogram summarizing the phylogenetic relationships among Lasiodoriform genera and out-group lineages. Boxes on the nodes correspond to the SH-aLRT test and Ultrafast Bootstrapping support values. Live specimen: Lasiodora sp. ♀. Photo credits: Wolf Moeller.
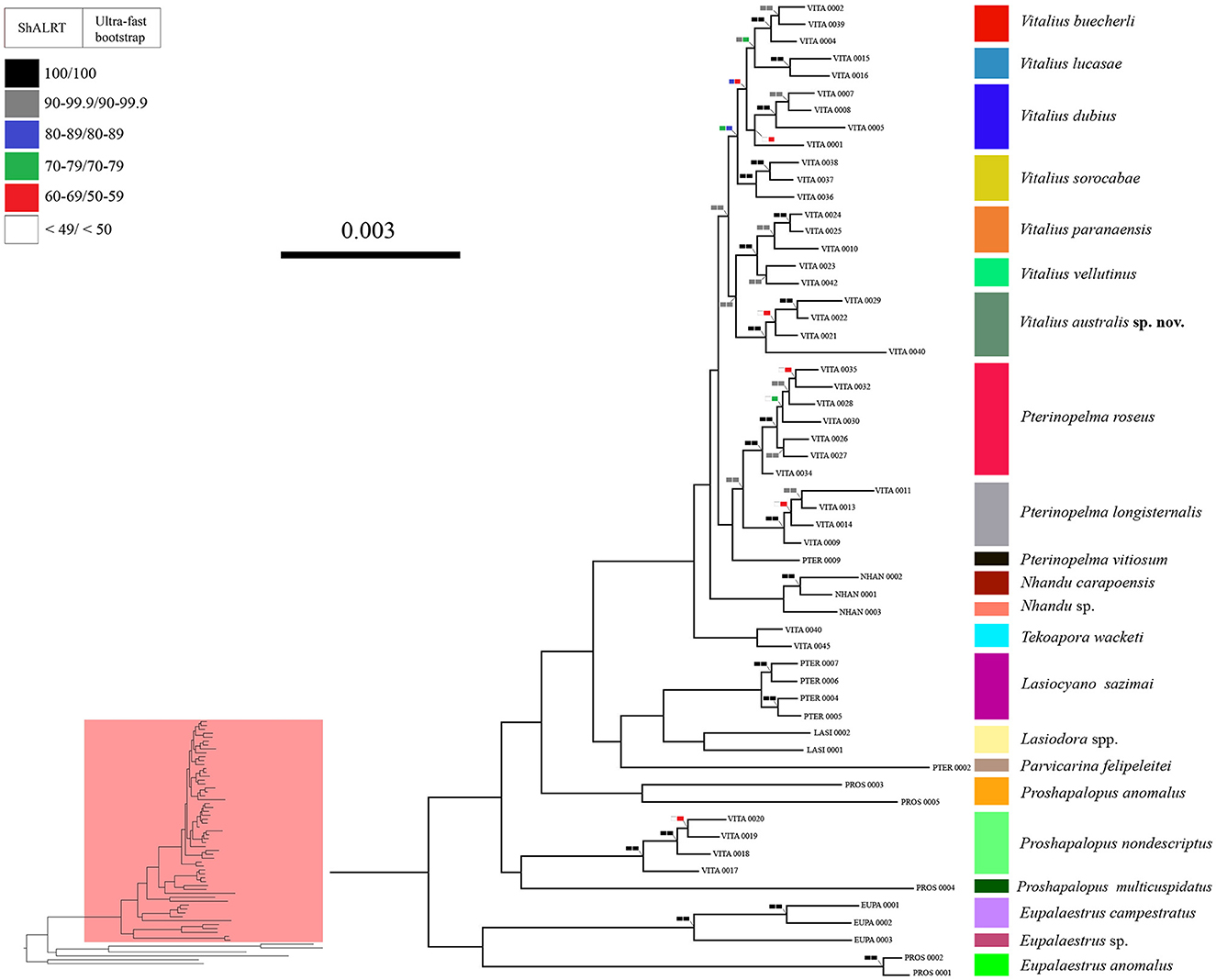
Figure 2. A phylogeny showing the relationships of Lasiodoriform genera. Boxes on the nodes correspond to the SH-aLRT test and Ultrafast Bootstrapping support values. Lasiodoriform lineages are highlighted in pink.
The genus Vitalius, as defined hitherto, was not recovered as monophyletic (Figure 2). To recover the monophyly of the group, it is necessary to transfer four species to other genera (see Taxonomy). Within the genus Vitalius (sensu stricto), two clades were recovered with high support values (SH-aLRT = 99.1 and bootstrapping = 90), the first comprising the type-species V. sorocabae sister group of V. dubius (V. lucasae + V. buecherli) and the group with Vitalius australis sp. nov. emerging as the sister group of V. paranaensis + V. vellutinus.
Our results, in addition to not recovering the monophyly of Vitalius, also showed that the genera Proshapalopus and Pterinopelma, in their current conformation, are paraphyletic. To achieve taxonomic stability, we propose a series of changes in the composition of the genera Pterinopelma and Vitalius, as well as the description of three new genera and a new species (see Taxonomy below). However, owing to the low support in the node that joins Proshapalopus multicuspidatus and Vitalius nondescriptus (see Figure 2), as well as the distant positioning of the type species of the genus Proshapalopus anomalus, we chose to keep the genus Proshapalopus paraphyletic.
Taxonomic changes are proposed based on the resulting phylogeny (Figures 1, 2) and morphological differences. New morphological descriptions for all Lasiodoriform genera (Figures 3–10) and the new species, including the taxonomic changes, are found in Supplementary File S3. In this study, we propose the following taxonomic acts: (1) transfer of Proshapalopus amazonicus to Eupalaestrus; (2) transfer of Pterinopelma felipeleitei to Parvicarina gen. nov. and of Pterinopelma sazimai to Lasiocyano gen. nov.; and (3) transfer of Vitalius nondescriptus to Proshapalopus, Vitalius wacketi to Tekoapora gen. nov., and Vitalius roseus and Vitalius longisternalis to Pterinopelma.

Figure 3. Some Lasiodoriform live specimens. (A) Eupalaestrus campestratus ♀; (B) Eupalaestrus anomalus ♀; (C) Lasiodora sp. ♀; (D) Nhandu carapoensis ♀; (E) Proshapalopus anomalus ♀; and (F) Vitalius sorocabae ♀. White arrows indicate the thickened tibia IV. Photo credits: (D) Wolf Moeller. (E) Rafael P. Indicatti.
Below, we have provided information for each genus belonging to the Lasiodoriforms, in particular, type species, species included in the genus, diagnoses and distribution, and a diagnosis for Vitalius australis sp. nov.
Theraphosidae Thorell, 1869
Subfamily Theraphosinae
Eupalaestrus Pocock, 1901
Type-species: Eupalaestrus pugilator Pocock, 1901, by original designation.
Contents: Eupalaestrus campestratus (Simon, 1891), Eupalaestrus crassimetatarsis Borges et al., 2021, Eupalaestrus larae Ferretti and Barneche, 2012, Eupalaestrus roccoi Borges et al., 2021, Eupalaestrus spinosissimus Mello-Leitão, 1923 and Eupalaestrus weijenberghi (Thorell, 1894), and Eupalaestrus anomalus (Mello-Leitão, 1923) new comb., nomen rest.
Emended diagnosis: Eupalaestrus males (except E. anomalus) differ from those of all other theraphosine genera by the presence of a denticulate row on the subapical area of the male palpal bulb embolus (see Figures 50, 51 in Bertani, 2001) in conjunction with the lack of stridulatory setae on the palpus trochanter. E. anomalus males differ from other theraphosine genera by the thickened tibia IV (see Figure 20 in Bertani, 2001). Eupalaestrus females differ from those of all other theraphosine genera, except Vitalius lucasae, by having the tibia IV thickened (Figure 3A), in conjunction with scopula on the retrolateral face of femur IV and the absence of stridulatory setae on the retrolateral face of the trochanter of the palpus. They differ from V. lucasae by the presence of type III urticating setae (except E. anomalus) or by the presence of stiff setae on metatarsus IV (E. spinosissimus). Females of E. anomalus differ from V. lucasae by their distinct geographic distribution (E. anomalus occurs in the Amazon forest, while V. lucasae occurs in the southern region of Brazil).
Distribution: Except for E. anomalus (from Brazilian Amazon, state of Mato Grosso) and E. spinosissimus (Brazilian Atlantic Forest, state of São Paulo and Rio de Janeiro), the genus is predominantly found in open areas, occurring in Brazilian Cerrado (state of Mato Grosso do Sul), Chaco (Argentina, Brazil, and Paraguay), Pampa (Argentina, Brazil, and Uruguay), and Atlantic Forest (Argentina, Brazil, and Paraguay).
Remarks: Eupalaestrus anomalus is restored into the genus. Previously, Lucas et al. (1993) transferred this species from Pocock, 1901 to Eupalaestrus anomalus. Later, Bertani (2001) transferred the species to Proshapalopus based on the morphological phylogenetic analysis, necessitating replacing the name with Proshapalopus amazonicus. We transferred the species to Eupalaestrus because it appears as a sister group to two other species in our phylogeny. However, its distribution (Brazilian Amazon) is far from the range of the other species of the genus (southern Brazil, Paraguay, northern Argentina, and Uruguay).
Proshapalopus (Mello-Leitão, 1923)
Type species: Proshapalopus anomalus (Mello-Leitão, 1923)
Contents: P. anomalus, P. multicuspidatus (Mello-Leitão, 1929), and Proshapalopus nondescriptus (Mello-Leitão, 1926) new comb.
Diagnosis: Males can be distinguished from other Theraphosinae (except Eupalaestrus anomalus and Vitalius lucasae) by the presence of an accentuated male palpal bulb ventral median depression (see Figures 68, 69 in Bertani, 2001; Figures 4B, E). It differs from Eupalaestrus anomalus by the presence of non-thickened tibia IV (Figure 3B), from Vitalius lucasae by the presence of an accessory keel under the prolateral inferior keel on the male palpal bulb (P. anomalus and P. multicuspidatus) (see Figure 73 in Bertani, 2001; Figure 4E), bifid tibial spur with very narrow branches, and the presence of long brown hairs on metatarsus IV (P. nondescriptus) (see Figure 3 in Bertani et al., 2012; Figure 4A). Females can be distinguished from other Theraphosinae by the presence of type I urticating setae with region “a” being shorter than region “b” (see Figure 9 in Bertani et al., 2012).

Figure 4. “Proshapalopus” spp. (A) Proshapalopus nondescriptus ♂ live specimen. (B) Proshapalopus nondescriptus male palpal bulb, prolateral view. (C) Proshapalopus nondescriptus male palpal bulb, retrolateral view. (D) Proshapalopus multicuspidatus (male juvenile) live specimen. (E) Proshapalopus multicuspidatus male palpal bulb, prolateral view. (F) Proshapalopus multicuspidatus, retrolateral view. An arrow showing the dense tuft of brown setae in the apical region of tarsus IV. A, Apical keel; AC, Accessory keel; D, Ventral median depression; PI, Prolateral inferior keel; PS, Prolateral superior; R, Retrolateral keel; SA, Subapical keel. Scale bar: 1 mm. Photo credits: (A, C) Wolf Moeller.
Distribution: BRAZIL: Atlantic Forest, from extreme southeast of the state of Minas Gerais to the states of Rio de Janeiro and Espírito Santo, south of Rio Doce River (P. anomalus), Atlantic Forest, from the state of Paraiba to south of the state of Bahia and Chapada Diamantina (portion from Bahia) (P. multicuspidatus) and from the state of Minas Gerais, in the highlands (up 1,100 m a.s.l.) of Espinhaço, Itacolomi, Caraça, and Gandarela (P. nondescriptus) (Bertani, 2001; Bertani et al., 2012).
Remarks: Bertani et al. (2012) redescribed and transferred the holotype Hapalopus nondescriptus Mello-Leitão, 1926 to Vitalius nondescriptus (Mello-Leitão, 1926), based on the presence of a subapical keel. Furthermore, the authors described the female for the first time, emphasizing only the presence of type I urticating setae, with region “a” being shorter than region “b” (typical of Proshapalopus species). We decided to transfer this species to the genus Proshapalopus based on our resulting phylogenetic topology and morphological characteristics shared by the species.
Parvicarina Galleti-Lima, Hamilton, Borges and Guadanucci, new genus
ZooBank registration: urn:lsid:zoobank.org:act:D50EDC50-41CC-4DFD-886F-2F0D2C3FD7CC.
Type species: Pterinopelma felipeleitei Bertani and Leal, 2016, by monotypy.
Contents: Parvicarina felipeleitei (Bertani and Leal, 2016) new comb.
Etymology: Generic name from the composition of the Parvi (=small) and carina (=keel), in reference to the small keels present on the male palpal bulb of the type-species. The genus name is feminine.
Diagnosis: Males of Parvicarina felipeleitei can be distinguished from other theraphosine genera by the following combination of characters: weakly developed palpal bulb keels, almost imperceptible (Figures 5C–F), and strongly curved metatarsus I (see Figure 12 in Bertani and Leal, 2016) touching the apex of the retrolateral spur when flexed (Figure 5G). Bifid tibial spur with the apical region of the retrolateral branch curved prolaterally (Figure 5E; see Figures 11, 12 in Bertani and Leal, 2016). Females can be distinguished by the spermathecae separated by a sclerotized area, which come up to half of the poorly sclerotized spermathecal stalks (Figure 5B); absence of stridulatory setae on prolateral face of coxa I; absence of long setae on the carapace (Figure 5A); absence of type III urticating setae; sternum as long as wide (see Figure 9 in Bertani and Leal, 2016); and carapace and legs black with conspicuous white rings on distal femora, patellae, tibiae, and metatarsi (Figure 5A; see Figures 20, 21 in Bertani and Leal, 2016).
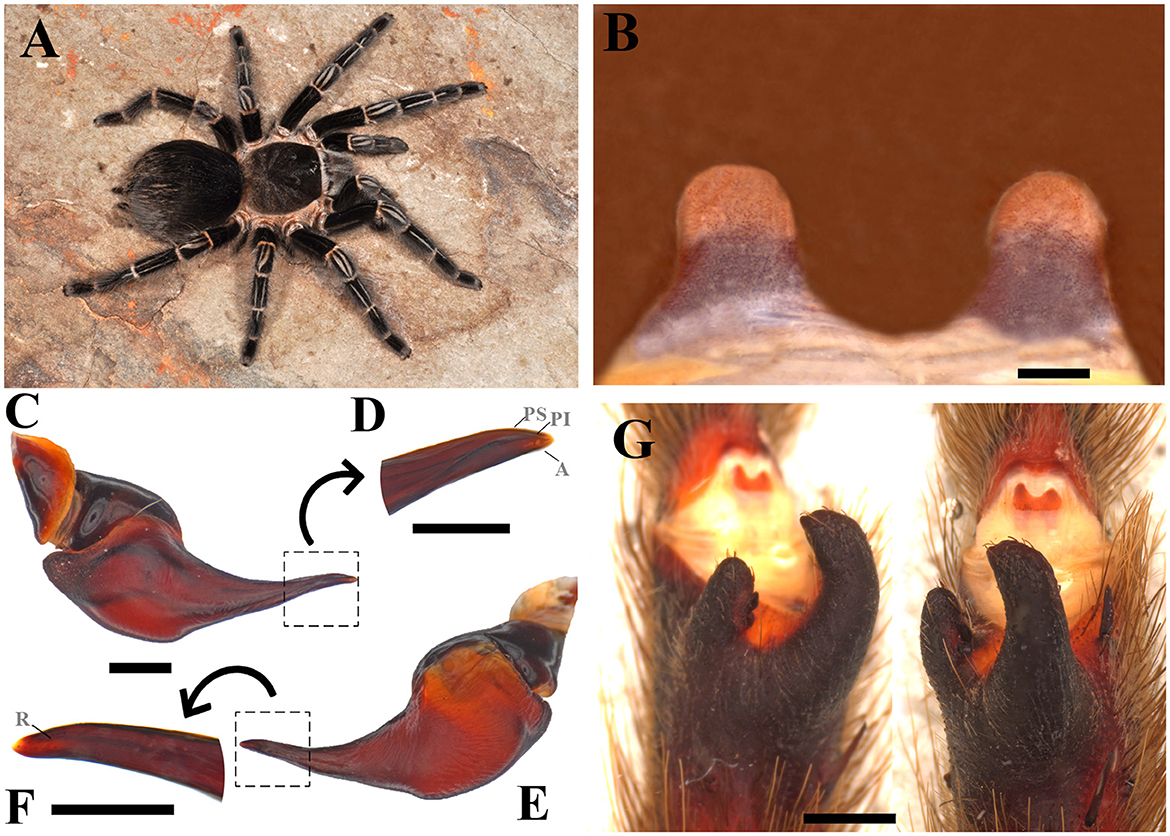
Figure 5. Parvicarina felipeleitei. (A) Parvicarina felipeleitei ♀ live specimen. (B) Ventral face of spermathecae. (C) Parvicarina felipeleitei male palpal bulb, prolateral view. (D) Prolateral view of the embolus in detail. (E) Parvicarina felipeleitei male palpal bulb, retrolateral view. (F) A retrolateral view of the embolus in detail. (G) Parvicarina felipeleitei tibial apophysis and flexion of the metatarsal I on the tibial apophysis. A, Apical keel; PI, Prolateral inferior keel; PS, Prolateral superior; R, Retrolateral keel. Scale bar: (A–G) = 1 mm; (D, F) = 0.5 mm. Photo credits: (A) Pedro H. Martins.
Distribution: BRAZIL: Serra do Arrependido Condado (Parque Estadual Pico do Itambé, municipality of Serro) and PARNA Serra do Cipó, which includes the municipalities of Morro do Pilar and Santana do Riacho. It also has records from the municipalities of Gouveia and Diamantina. The sites where individuals of the species were found belong to the Espinhaço Mountain Range and are located in the central portion of the state of Minas Gerais in Brazil (Bertani and Leal, 2016) (Figure 11A).
Lasiocyano Galleti-Lima, Hamilton, Borges, and Guadanucci, new genus
ZooBank registration: urn:lsid:zoobank.org:act:653A6545-E873-4071-B9EC-42146430C462.
Type species: Pterinopelma sazimai Bertani et al., 2011, by monotypy.
Contents: Lasiocyano sazimai (Bertani et al., 2011) new comb.
Etymology: Generic name from the composition of Lasio (=hairy) and Cyano (=blue), in reference to the blue iridescent setae of the type-species. The genus name is neuter.
Diagnosis: Males of Lasiocyano sazimai can be distinguished from all theraphosine genera by the palpal bulb with well-developed prolateral superior, prolateral inferior, retrolateral, subapical, and apical keels (Figures 6C, D; see Figures 6, 7 in Bertani and Leal, 2016); embolus slender at its distal portion (Figures 6C, D; see Figures 6, 7 in Bertani and Leal, 2016); the absence of stridulatory setae on prolateral face of coxa I; bifid tibial spur with apical region of retrolateral branch curved prolaterally (Figure 6E; see Figure 5 in Bertani and Leal, 2016); and metatarsus I touching the apex of retrolateral tibial spur when flexed (Figure 6E). Females of this species can be distinguished by having the following combination of traits: small spermatheca with spermathecal stalks proportional to the size of the spermathecal receptacles (Figure 6B); the absence of stridulatory setae on the prolateral face of coxa I; the absence of long setae on the carapace (Figure 6A); and the presence of type III urticating setae; Additionally, both males and females have carapace, chelicerae, and legs covered with blue or purple iridescent setae, which can vary to darker shades (Figure 6A; see Figures 1, 19 in Bertani and Leal, 2016).

Figure 6. Lasiocyano sazimai. (A) Lasiocyano sazimai ♀ live specimen. (B) Ventral face of spermathecae. (C) Lasiocyano sazimai male palpal bulb, prolateral view. (D) Lasiocyano sazimai male palpal bulb, retrolateral view. (E) Lasiocyano sazimai tibial apophysis and flexion of the metatarsal I on the tibial apophysis. A, Apical keel; PI, Prolateral inferior keel; PS, Prolateral superior; R, Retrolateral keel; SA, Subapical keel. Scale bar: 1 mm. Photo credits: (A) Wolf Moeller.
Distribution: BRAZIL: Highlands (up 1,100 m a.s.l.) of the Espinhaço Mountain range and States of Minas Gerais and Bahia, Brazil (Bertani et al., 2011; Bertani and Leal, 2016) (Figure 11B).
Tekoapora Galleti-Lima, Hamilton, Borges, and Guadanucci, new genus
ZooBank registration: urn:lsid:zoobank.org:act:BE01E016-45A9-48A1-A0B2-7D80D04507BE.
Type species: Vitalius wacketi (Mello-Leitão, 1923), by monotypy.
Contents: Tekoapora wacketi (Mello-Leitão, 1923) new comb.
Etymology: Generic name is derived from the way that the original people from the Serra do Mar region (Mbyá) referred to this geological formation (“tekoá porã”, means “good land”) (Azanha and Ladeira, 1988). The genus name is masculine.
Diagnosis: Males differ from other theraphosine species by having a palpal bulb with a long embolus, strongly pronounced keels (Figures 7C, D), and male spurs with a prolateral branch, which is smaller and more thickened than the retrolateral branch (Figure 7E). Females can be distinguished by having tibiae IV not thickened (Figure 7A); sternum slightly longer than wide; carapace not bordered by long hairs pointing to the center carapace (Figure 7A); big spermathecae with spermathecal receptacles not enlarged (Figure 7B); femora with many long ventral setae; and sternum and coxae black, covered by short hairs and the presence of leg rings and longitudinal leg stripes being easily distinct.

Figure 7. Tekoapora wacketi. (A) Tekoapora wacketi ♀ live specimen. (B) Ventral face of spermathecae. (C) Tekoapora wacketi male palpal bulb, prolateral view. (D) Tekoapora wacketi male palpal bulb, retrolateral view. (E) Tekoapora wacketi tibial apophysis and flexion of the metatarsal I on the tibial apophysis. A, Apical keel; PI, Prolateral inferior keel; PS, Prolateral superior; R, Retrolateral keel; SA, Subapical keel. Scale bar: 1 mm. Photo credits: (A) Rafael P. Indicatti.
Distribution: BRAZIL: Tropical latifoliated (broadleaf) forest of “Serra do Mar” and coastal region, including many islands, from south of the state of Rio de Janeiro and southwards to states of São Paulo, Paraná, and Santa Catarina (Bertani, 2001) (Figure 11C).
Pterinopelma Pocock, 1901
Type-species: Pterinopelma vitiosum (Keyserling, 1891), by original designation.
Contents: P. vitiosum, Pterinopelma longisternalis (Bertani, 2001) new comb. and Pterinopelma roseus (Mello-Leitão, 1923) new comb.
Diagnosis: Pterinopelma shares with Lasiodora, Parvicarina gen. nov., Vitalius, and Nhandu the absence of accessory prolateral keels, as well as a ventral median region depression on the palpal bulb and the presence of prolateral superior, prolateral inferior, retrolateral, subapical and apical palpal keels in male bulb (Figures 9A, B, D, E, G, H); Females differ from all other genera in this study by the shape of the spermathecae with receptacles separated by a small central depression and spermathecal stalk with a slight constriction at the base of the spermathecal bulb (Figures 8B, D, F). Both sexes can be distinguished from Lasiodora by the absence of the stridulatory setae on the prolateral coxae. Pterinopelma males are distinguished from Nhandu and Vitalius by the palpal bulb shape that narrows abruptly from its median region forward, a long embolus, denticles in the prolateral inferior keel (P. vitiosum) (Figure 9A), and remarkable subapical keel (P. longisternalis and P. roseus) (Figures 9D, E, G, H). Females of Pterinopelma can be distinguished from Nhandu by the absence of long setae on the carapace (Figures 8A, C, E) and from Vitalius by having a spermathecal stalk narrower than the spermathecal bulb, giving it a rounded shape (Figures 8B, D, F).

Figure 8. Pterinopelma. (A) Pterinopelma vitiosum ♀ live specimen. (B) The ventral face of spermathecae of P. vitiosum. (C) Pterinopelma longisternalis ♀ live specimen. (D) The ventral face of spermathecae of P. longisternalis. (E) Pterinopelma roseus ♀ live specimen. (F) Ventral face of spermathecae of P. roseus. Scale bar: 1 mm. Photo credits: (C) Rafael P. Indicatti.
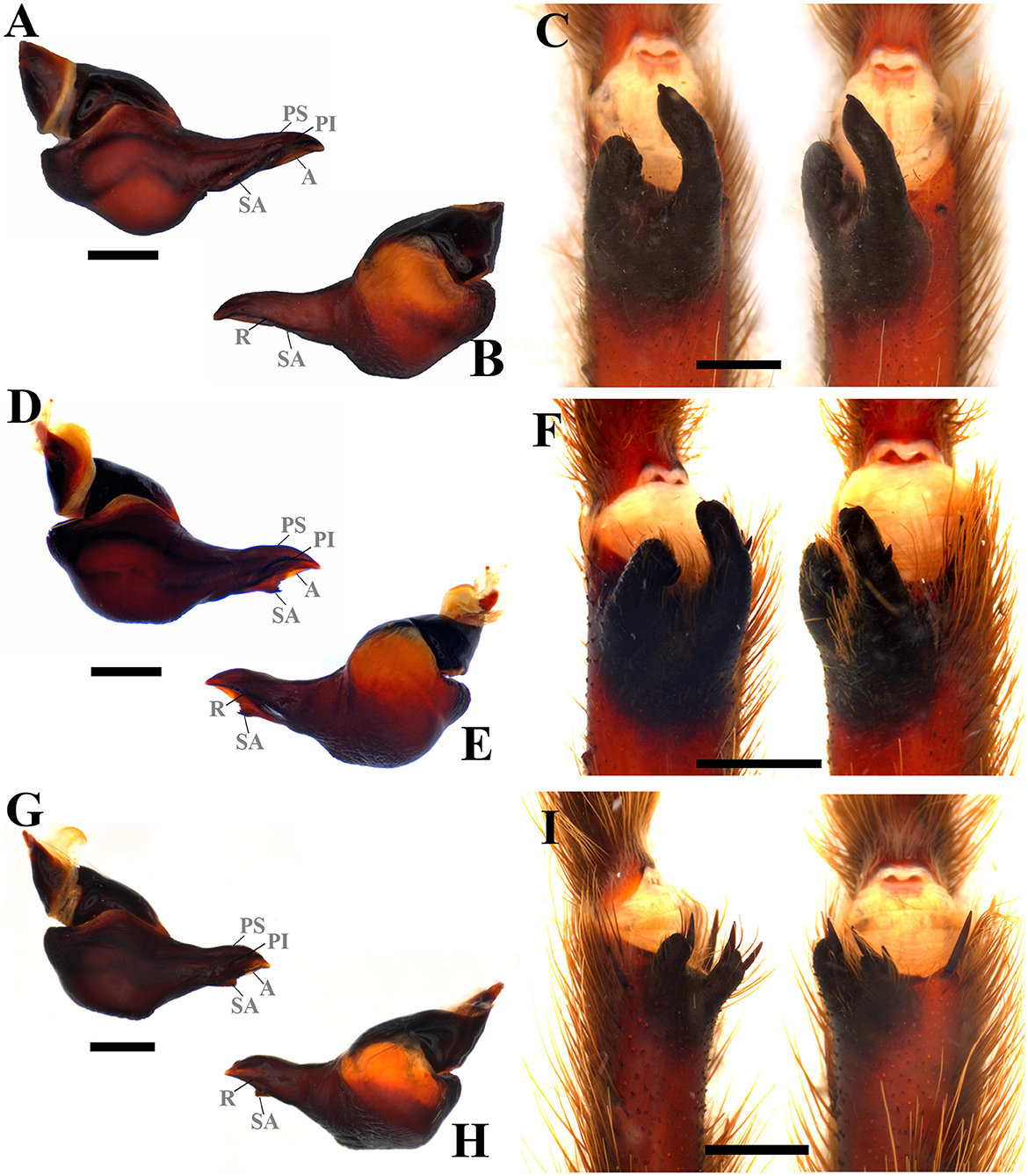
Figure 9. Male sex characteristics of Pterinopelma species. (A) P. vitiosum male palpal bulb, prolateral view. (B) P. vitiosum male palpal bulb, retrolateral view. (C) P. vitiosum tibial apophysis and flexion of the metatarsal I on the tibial apophysis. (D) P. longisternalis male palpal bulb, prolateral view. (E) P. longisternalis male palpal bulb, retrolateral view. (F) P. longisternalis tibial apophysis and flexion of the metatarsal I on the tibial apophysis. (G) P. roseus male palpal bulb, prolateral view. (H) P. roseus male palpal bulb, retrolateral view. (I) P. roseus tibial apophysis and flexion of the metatarsal I on the tibial apophysis. A, Apical keel; PI, Prolateral inferior keel; PS, Prolateral superior; R, Retrolateral keel; SA, Subapical keel. Scale bar: 1 mm.
Distribution: ARGENTINA: Araucaria forests northeastern of Misiones province (P. longisternalis) and southeastern of Misiones province (P. roseus). BRAZIL: northern of the state of Rio Grande do Sul, where vegetation comprises Subcaducifolious Atlantic Forest with Araucaria angustifolia and patches of “campo” (P. vitiosum), subtropical subcaducifolious forest of states of Paraná and Santa Catarina, west of “Serra do Mar” (P. longisternalis), and subtropical subcaducifolious forest of northwest and central state of Rio Grande do Sui (P. roseus) (Bertani, 2001; Bertani et al., 2011; Ferretti et al., 2014, 2015) (Figure 11D).
Vitalius Lucas et al., 1993
Type-species: Vitalius sorocabae (Mello-Leitão, 1923), by original designation.
Contents: V. sorocabae, V. buecherli, V. dubius, V. lucasae, V. paranaensis, V. vellutinus, and Vitalius australis new species
Emended diagnosis: Males can be distinguished from species of other theraphosine genera except Proshapalopus, Lasiodora, Lasiocyano gen. nov., Tekoapora gen. nov., Nhandu, and Pterinopelma by the presence of a triangular subapical keel on the male palpal bulb (see Figures 74–79, 91–96, 101–104, 119, 120, 123, 124, 127, 128 in Bertani, 2001; Figures 10C, D). They can be distinguished, from Lasiodora, by the absence of a velvet stridulating setae on the coxae; from P. anomalus and P. multicuspidatus, by the absence of an acessory keel in the male palpal bulb; from P. nondescriptus, except for V. lucasae, by the absence of accentuated ventral median depression on the palpal bulb; from Nhandu, except for V. lucasae, by the male spur with converging branches originating from a common base, tapering distally, with the prolateral branch thickened or by small, almost vestigial, male spurs (V. vellutinus) (see Figures 108–111 in Bertani, 2001). Moreover, V. lucasae can be distinguished from P. nondescriptus and Nhandu by the thickened tibiae IV. Males of Vitalius differ from Lasiocyano gen. nov. by the absence of blue color patter and the apical region of retrolateral branch not curved prolaterally (see Figures 80, 97, 108–111, 117, 121, 125 in Bertani, 2001; Figure 10E); from Tekoapora gen. nov. by the not thickened both tibial aphophysis branches (see Figures 80, 97, 108–111, 117, 121, 125 in Bertani, 2001; Figure 10E); and from Pterinopelma by the palpal bulb shape that not narrows abruptly from its median region forward (see Figures 74–79, 91–96, 101–104, 119, 120, 123, 124, 127, 128 in Bertani, 2001; Figures 10C, D). Females can be distinguished from Lasiodora by the absence of a velvet stridulating setae on the coxae, from Proshapalopus by the Type I urticating setae with region “A” longer or equal to region “B”; from Lasiocyano gen. nov. by the absence of blue color and Type III urticating setae; from Nhandu by the absence of many long, curly, scattered hairs over the carapace; from Tekoapora gen. nov. by the small spermathecae; and from all Pterinopelma species by the spermathecae without a small central depression separating the receptacles (see Figures 81, 82, 98–100, 107, 118, 122, 126 in Bertani, 2001; Figure 10B).
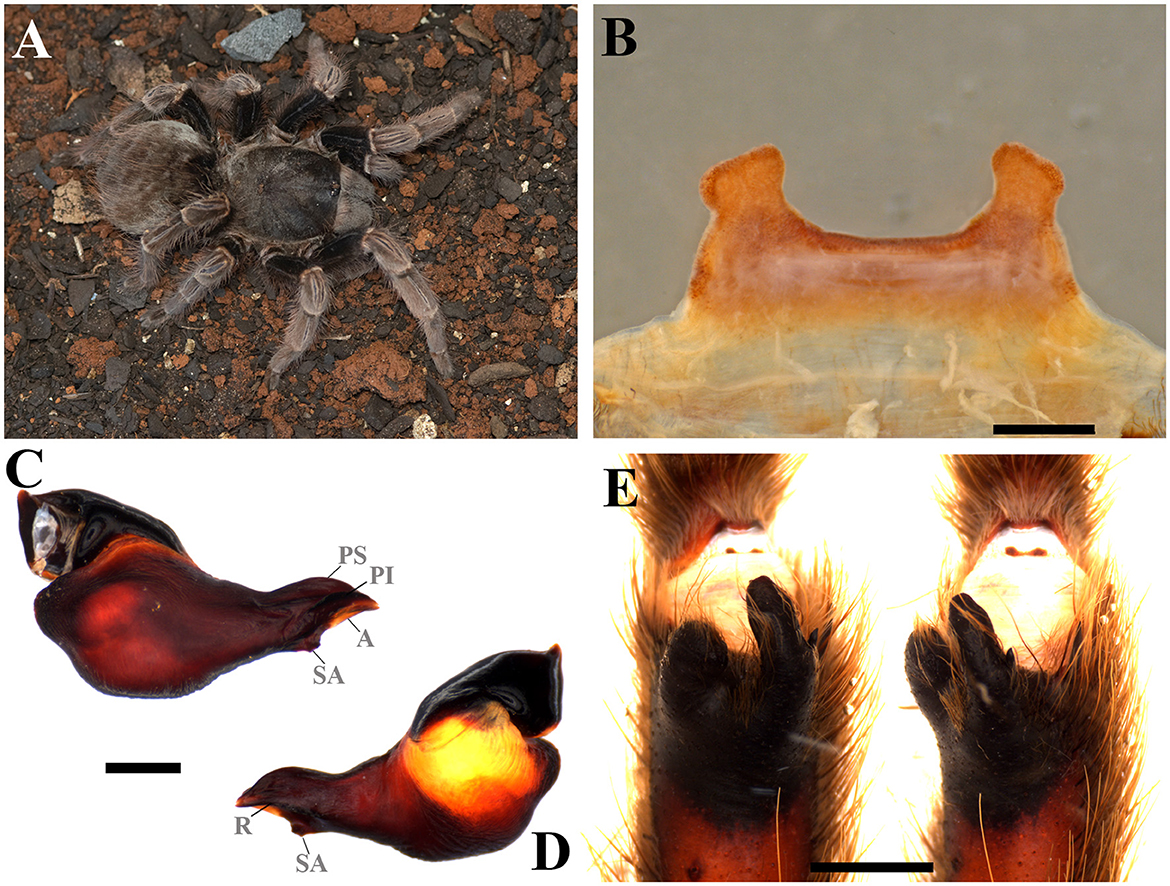
Figure 10. Vitalius australis sp. nov. (A) Vitalius australis sp. nov. ♀ live specimen. (B) Ventral face of spermathecae. (C) Vitalius australis sp. nov. male palpal bulb, prolateral view. (D) Vitalius australis sp. nov. male palpal bulb, retrolateral view. (E) Vitalius australis sp. nov. tibial apophysis and flexion of the metatarsal I on the tibial apophysis. A, Apical keel; PI, Prolateral inferior keel; PS, Prolateral superior; R, Retrolateral keel; SA, Subapical keel. Scale bar: 1 mm. Photo credits: (A) Wolf Moeller.
Distribution: ARGENTINA: Misiones province; BRAZIL: States of São Paulo, Paraná, and Rio Grande do Sul (Figures 11E, F).
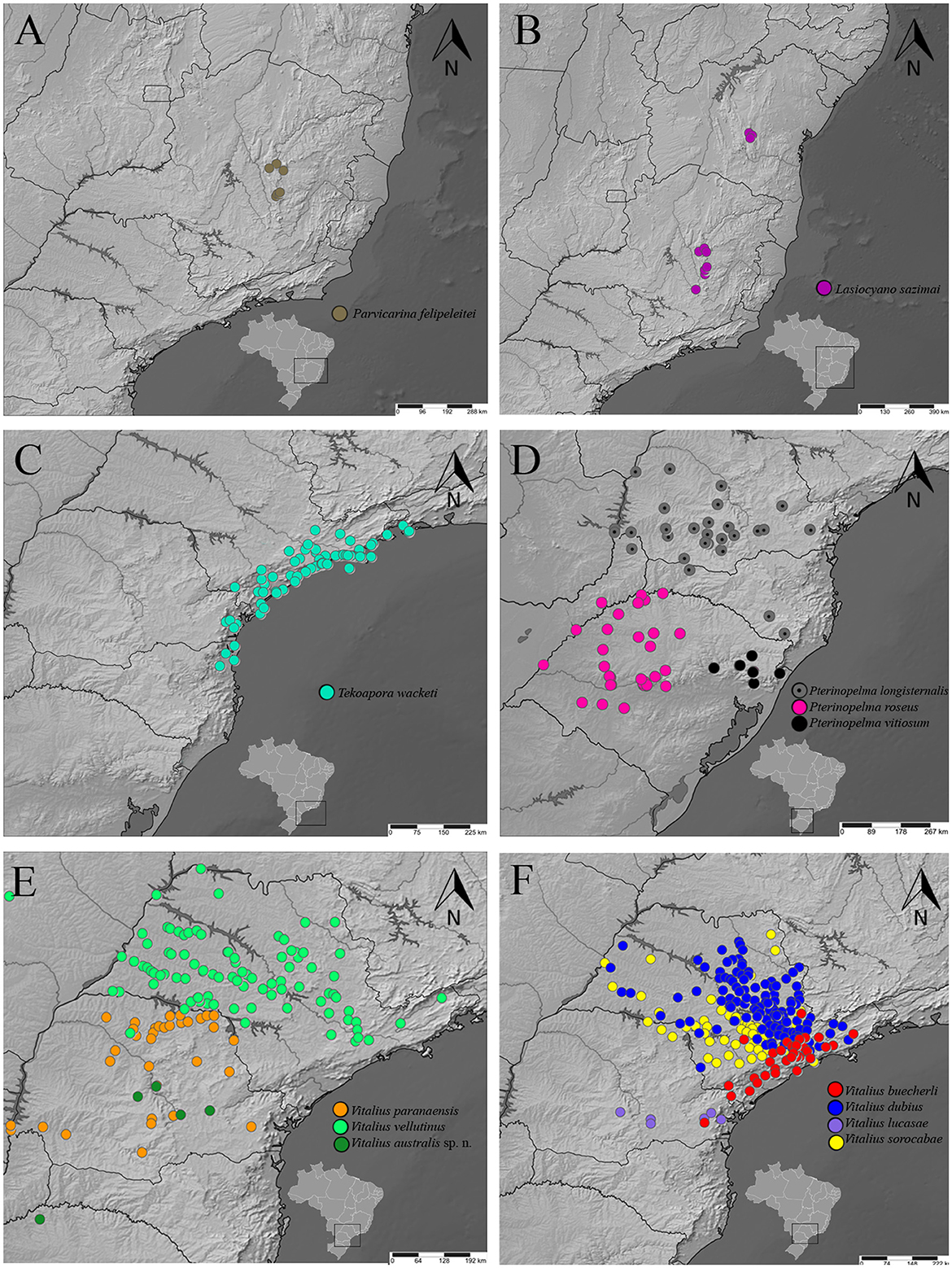
Figure 11. A distribution map of rearranged Lasiodoriform species according to the topology. (A) Parvicarina felipeleitei. (B) Lasiocyano sazimai. (C) Takoapora wacketi. (D) Pterinopelma spp. (E) V. paranaensis, V. vellutinus, and Vitalius australis sp. nov. (F) V. buecherli, V. dubius. V. lucasae, and V. sorocabae.
Vitalius australis Galleti-Lima, Hamilton, Borges, and Guadanucci, new species
ZooBank registration: urn:lsid:zoobank.org:act:57A4852C-84CA-425E-94DF-340656BE3142.
Type material: Holotype: (CAD 1244). Paratypes: (CAD 1210, CAD 1243).
Etymology: Generic name in reference to the region where the species is found (Southern Region of Brazil). The genus name is male.
Diagnosis: Males can be distinguished from congeners by the long embolus, very pronounced prolateral superior and inferior keels, and a well-developed male spur, with the retrolateral branch tapering distally (Figures 10C, D). Females differ from the other species by the short and homogeneously sclerotized spermatheca, with a spermathecal stalk with a strong constriction at the base of the spermathecal bulb that does not have a rounded shape, and also by the elevated central region (Figure 10B).
Distribution: Probably from the Seasonal Deciduous Forest in the north of Rio Grande do Sul to the Araucaria Forest region in the interior of Paraná (Figure 11E).
The presence of keels on the male palpal bulb is a synapomorphic character of Theraphosinae (Pérez-Miles et al., 1996). These keels were first proposed to have taxonomic value for tarantulas by Bücherl (1957) and later, Bertani (2000) proposed terminology, homologies, and detailed descriptions of palpal bulb keels of Theraphosinae genera, regarding their morphology, position, and distribution among this genera. In a morphological, phylogenetic, and biogeographic study of the genera Vitalius, Nhandu, and Proshapalopus, Bertani (2001) suggested the presence of a retrolateral keel and a male palpal bulb with the embolus apex slightly laterally flattened, characterized by concave areas above and under this retrolateral keel, as synapomorphies of Eupalaestrus, Lasiodora, Proshapalopus, Nhandu, and Vitalius clades. Bertani (2001) also found that Lasiodora, Proshapalopus, Nhandu, and Vitalius formed a monophyletic group supported by the presence of a subapical keel and males with metatarsus I that touches the retrolateral branch of the tibial apophysis when flexed (except in Vitalius species), and the genus Eupalaestrus, as sister-group. Our analysis supports the monophyly of the group, and we propose the name Lasiodoriform to include: Eupalaestrus, “Proshapalopus”, Parvicarina gen. nov., Lasiodora, Lasiocyano gen. nov., Tekoapora gen. nov., Nhandu, Pterinopelma, and Vitalius. The genus Lasiodora is the oldest described genus among the taxa of this group, and all the genera share sexual characteristics similar to Lasiodora, such as bulbs with subapical and retrolateral keels (except for the male bulbs of some species of Eupalaestrus and Parvicarina gen. nov.), male tibial apophysis with two branches (except for some species of Nhandu, who had a secondary loss of the tibial aphophysis), and spermathecae fused in a small central area.
The monophyly of the genus Eupalaestrus (Figures 3A, B) is supported by the presence of a denticulate row (see Figures 50, 51 in Bertani, 2001), which is found in the same position as the subapical keel but is morphologically distinct. Bertani (2000, 2001) suggested that the denticulate row of Eupalaestrus is a homolog feature to the subapical keel of Lasiodora, Proshapalopus, Nhandu, and Vitalius. In this study, we assume that the genus Eupalaestrus belongs in the Lasiodoriform lineage. Eupalaestrus anomalus (Figure 3B), which emerged as a sister lineage to the other species of the genus, has a palpal bulb with a small and triangular subapical keel, which is almost inconspicuous (see Figures 64, 65 in Bertani, 2001). This finding putatively corroborates that the denticulate row of Eupalaestrus is a primary homolog of the subapical keel, as proposed by Bertani (2000, 2001), making this characteristic a synapomorphy of the genus that is absent in E. anomalus. All species of Eupalaestrus have males and females with thickened tibia IV, making this an important synapomorphy of the genus.
Proshapalopus multicuspidatus is related to P. nondescriptus, previously included in Vitalius. P. nondescriptus was previously included in Vitalius because females lack type III urticating setae and males have the palpal bulb with a well-pronounced subapical keel (Bertani et al., 2012). Here, we present morphological characteristics that support the relationship between P. multicuspidatus and P. nondescriptus, such as the presence of a ventral median depression (D) on a male palpal bulb (Figures 4B, C, E, F) and females with type I urticating setae with region “a” shorter than region “b” (see Figure 3 in Bertani, 2001 and Figure 9 in Bertani et al., 2012). Females of P. anomalus also present this same variation of type I urticating setae, in addition to males with palpal bulbs possessing an accessory keel (AC), a character shared with males of P. multicuspidatus. One potential issue is that the relation of P. multicuspidatus + P. nondescriptus has low support in both tests (SH-aLRT and bootstrapping). We expected to find P. anomalus, P. multicuspidatus and P. nondescriptus in the same clade because they share these characteristics; however, we did not recover these species as monophyletic in our topology. To restore the monophyly of Vitalius (the focus of this study), we propose here the transfer P. nondescriptus to Proshapalopus, which now includes P. anomalus, P. multicuspidatus, and P. nondescriptus, keeping the genus Proshapalopus paraphyletic (see topology in Figures 1, 2). We presumed that the relationship between P. multicuspidatus, P. nodescriptus, and P. anomalus can be recovered with additional analyses that include more representatives of these taxa.
We must highlight the absence of the recently described genus Cymbiapophysa (Gabriel and Sherwood, 2020), from Ecuador, Colombia, and possibly Peru (Gabriel and Sherwood, 2020; Sherwood et al., 2021). The genus was described based on the holotype of Metriopelma velox Pocock, 1903 and comprises four species, including Cymbiapophysa marimbai (Perafán and Valencia-Cuellar, 2018), which was previously described in the genus Proshapalopus, based on its position in a morphological phylogeny, and supported by morphological characteristic diagnostic of Proshapalopus (Perafán and Valencia-Cuellar, 2018). It is worth noting that the authors emphasized the disruptive distribution of this taxa in comparison to other species of Proshapalopus and discussed the hypothesis with which this taxon could form a new taxonomic group. Based on the morphology of the male palpal bulb, tibial apophysis, and female spermathecae, Sherwood et al. (2021) transferred this species to Cymbiapophysa and questioned the designation of the keels and palpal bulb structures of the species by Perafán and Valencia-Cuellar (2018), proposing a new terminology. The support to include this species in Cymbiapophysa is weak, given that C. marimbai lacks cymbial apophysis (found in other species of the genus), presenting only a sclerotized portion of cymbium, as noted by Perafán and Valencia-Cuellar (2018), but no phylogeny was presented to test the monophyly of the genus. Sherwood et al. (2021) also observed a similar cymbial apophysis of other Cymbiapophysa species on P. amazonicus (here transferred to Eupalaestrus). We were unable to insert any Cymbiapophysa (including C. marimbai) in our analysis to clarify its phylogenetic position. However, we agree with the hypothesis presented by Perafán and Valencia-Cuellar (2018) that C. marimbai may be related to an undescribed lineage, but new taxonomic and phylogenetic studies would be necessary to test the hypothesis and clear the genus positioning.
The monophyly of this group (Figures 3C, 5A, 6A) is well-supported in the phylogenomic analysis and can be further supported by the morphology of the male tibial apophysis with the apical region of the retrolateral branch curved prolaterally (Figures 5E, 6E; see Figure 58 in Bertani, 2001). Considering the topology of the tree, two options are possible: (1) if the whole clade can be considered Lasiodora, then the diagnoses of the genus should be amended to include two extra species; or (2) keep Lasiodora as it is, diagnosed by the presence of a stridulating setae on coxae I and II above the suture, and propose two new monotypic genera. The synonymy between these genera was not considered here because the three taxa have other distinct and exclusive characters, those of which warrant generic recognition (see diagnoses in Taxonomy). Because of this, we chose the second option, a decision that agrees with the repeatedly supported monophyly of Lasiodora (Bertani, 2001; Bertani et al., 2011; Galleti-Lima and Guadanucci, 2018; Perafán and Valencia-Cuellar, 2018). Moreover, the two novel genera can be diagnosed by the absence of a subapical keel, as well as poorly-developed keels for Parvicarina gen. nov. and by the body color with iridescent blue setae on the carapace, chelicerae, and legs in Lasiocyano gen. nov.
According to records in the literature (Bertani et al., 2011; Bertani and Leal, 2016; Galleti-Lima and Guadanucci, 2018) and scientific collections, Lasiocyano sazimai has exclusive distribution of rocky fields of the Espinhaço Mountain Range of the states of Bahia and Minas Gerais, a region that has been heavily impacted by anthropogenic action over the years (ICMBio, 2018). Another important factor to note is the illegal trade in wild animals (ICMBio, 2018), which, along with habitat loss, has resulted in the Lasiocyano sazimai being listed as endangered on the official list of Brazilian terrestrial invertebrate animals threatened with extinction (see list in Leite, 2021).
All past morphological phylogenies that focused on Lasiodoriforms have recovered the sister group relation Nhandu + Vitalius (Bertani, 2001; Bertani et al., 2011; Galleti-Lima and Guadanucci, 2018; Perafán and Valencia-Cuellar, 2018). Although the revalidation of Pterinopelma was based on phylogenetic inference, two of the three species (P. sazimai and P. felipeleitei) were not analyzed in a phylogenetic context after misidentifications were pointed out (Bertani and Leal, 2016). A recent morphology-based phylogenetic analysis that included Pterinopelma species (Galleti-Lima and Guadanucci, 2018) challenged the monophyly of Nhandu + Vitalius, where P. sazimai appeared as a sister group to the genus, Nhandu. Females of P. vitiosum share morphological similarities with these other two genera, especially Vitalius, due to the absence of type III urticating setae in females and their color pattern. These similarities are so evident that P. longisternalis and P. roseus were placed in Vitalius by Bertani (2001), and only with the use of phylogenomics was it possible to verify the proximity of these two species to P. vitiosum. We observed high morphological similarity between Vitalius, and the species included herein is Pterinopelma (P. roseus, P. longisternalis, and P. vitiosum). Owing to these findings, we propose a set of morphological characters distinguishing Vitalius from Pterinopelma and other Lasiodoriforms (see diagnosis for these genera in Taxonomy).
The genera that constitute the Tekoapora gen. nov., Nhandu, Pterinopelma, and Vitalius clade have subtle morphological diagnoses that are difficult to be recognized. Despite this, we decided to keep the three genera due to the support of the nodes forming monophyletic groups and the placement of the type species in each of these groups. Tekoapora gen. nov., a sister-group of the other genera, has a combination of characteristics that appeared multiple times and individually along the other related genera, such as the presence of a long embolus maintained only in Pterinopelma or the strongly pronounced keels in the male palpal bulb, observed in Nhandu but not in other genera. Furthermore, Tekoapora gen. nov. appears to be endemic to the Serra do Mar, a mountain system that extends from the coastal region of Espírito Santo to the south of Santa Catarina, and which was raised by the tectonic action during the post-Cretaceous (Ross, 1996; Bertani, 2001). Bertani (2001) suggested that this geological event could be related to the distribution of Vitalius species (extrapolating here to all genera of this clade, since the vast majority are sympatric).
Among the Vitalius species, there are two recognizable groups: one formed by V. buecherli, V. lucasae, V. dubius, and V. sorocabae and another formed by V. paranaensis, V. vellutinus, and Vitalius australis sp. nov. The proximity between the species V. dubius and V. buecherli has been discussed by Bertani (2001), who pointed to the similarity of the palpal bulb of these species (see Figures 91–96, 123, 124 in Bertani, 2001). Furthermore, the position of V. lucasae as the sister lineage to V. buecherli differs from the morphological phylogenetic hypotheses, where the species was the sister group to all other Vitalius species. The distribution of these species appears to be geographically continuous, extending through the Atlantic Plateau, with a broadleaf tropical forest vegetation. This was suggested by Bertani (2001) as a factor of sympatry between V. dubius and V. buecherli. Additionally, Bertani (2001) suggested that the areas of occurrence of V. dubius and V. buecherli were also interconnected with V. lucasae through the distribution area of V. sorocabae, which here emerged as a sister group to all these species.
Within the other group (V. paranaensis, V. vellutinus and Vitalius australis sp. nov.), V. paranaensis is sister to V. vellutinus, a relationship similar to what was discovered by Bertani (2001), but without the proposition of well-defined morphological diagnostic characters to distinguish between females. Bertani (2001) proposed that females of these two taxa could only be recognized by geographic distribution, with V. paranaensis restricted to the state of Paraná and the province of Misiones, Argentina, while V. vellutinus is distributed across the west of the state of São Paulo. We recovered the relationship of two individuals identified as V. vellutinus, one collected in the state of São Paulo and the other in the state of Paraná, highlighting the need for further investigation toward new diagnostic delimitation for these females. In addition, the males of the new species (Vitalius australis sp. nov.) present morphological characteristics similar to the males of V. paranaensis and can only be differentiated due to their phylogenetic relationship with a morphologically distinct female individual. From this, it was possible to determine small morphological differences in the tibial apophyses and male palpal bulb.
The original contributions presented in the study are publicly available. This data can be found here: ZooBank, urn:lsid:zoobank.org:act:653A6545-E873-4071-B9EC-42146430C462, urn:lsid:zoobank.org:act:D50EDC50-41CC-4DFD-886F-2F0D2C3FD7CC, urn:lsid:zoobank.org:act:BE01E016-45A9-48A1-A0B2-7D80D04507BE, and urn:lsid:zoobank.org:act:57A4852C-84CA-425E-94DF-340656BE3142.
Ethical review and approval was not required for the study on animals in accordance with the local legislation and institutional requirements.
AG-L and JG conceived the research that resulted in this paper. CH developed and funded all molecular analyses, in addition to collaborating on the manuscript. LB assisted with specimen collection, taxonomic and morphological discussions, and collaborated with the manuscript. The text was revised by all authors. All authors contributed to the article and approved the submitted version.
This research was funded by the Conselho Nacional de Desenvolvimento Científico e Tecnológico—CNPq (Process No. 168493/2017-9) for AG-L, Fundação de Amparo à Pesquisa do Estado de São Paulo—FAPESP (Process No. 2017/11985-9) for JG, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES (Process No. 88881.709394/2022-01), (Process No. 88882.428068/2019-01) for LB, and University of Idaho to CH.
We would like to express our gratitude to all curators who provided access to the materials studied in this research. We thank the technician (Andrea Noble Stuen) and graduate students (Karina Silvestre Bringas and Erik Ciaccio) from the Department of Entomology, Plant Pathology and Nematology in the University of Idaho who were responsible for the sample extractions. We are also grateful to the Instituto Chico Mendes de Conservação da Biodiversidade—ICMBio (64478-1 and 68145-1) and the Comissão Técnica Científica do Instituto Florestal— COTEC (260108-004.071/2018), for granting authorizations for specimen collection. We thank Pró-Reitoria de Pesquisa of Unesp for the financial support to publish this article (Edital 01/2023 - PROPe). We thank Leornado Carvalho, Pedro Taucce, Nelson Ferretti, Facundo Labarque, Guilherme Azevedo and Eduardo Sotto for their suggestions and coments. We also thank Everton Trova, Hector Manuel Ozório Gonzalez-Filho, Ian Meireles, Rafael Fonseca-Ferreira, and Victor Morais Ghirotto for assisting us in the field. We would also like to thank Pedro Henrique Martins and Wolf Moeller for providing photos of live specimens and helping in fieldwork. We are indebted to Rafael Prezzi Indicatti for providing images of live specimens, assisting with fieldwork, and providing taxonomic advice and to Andressa Paladini for her providing help in revising the manuscript. We also thank the reviewers for their insightful comments and suggestions on the manuscript.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fevo.2023.1177627/full#supplementary-material
Anisimova, M., Gil, M., Dufayard, J. F., Dessimoz, C., and Gascuel, O. (2011). Survey of branch support methods demonstrates accuracy, power, and robustness of fast likelihood-based approximation schemes. System. Biol. 60, 685–699. doi: 10.1093/sysbio/syr041
Ausserer, A. (1871). Beiträge zur Kenntniss der Arachniden-Familie der Territelariae Thorell (Mygalidae Autor). Verhandlungen der Kaiserlich-Königlichen Zoologisch-Botanischen Gesellschaft in Wien. 21, 117–224
Ausserer, A. (1875). Zweiter Beitrag zur Kenntniss der Arachniden-Familie der Territelariae Thorell: (Mygalidae Autor). Verhandlungen der Kaiserlich-Königlichen Zoologisch-Botanischen Gesellschaft in Wien. 25, 125–206.
Azanha, G., and Ladeira, M. I. (1988). Os índios da Serra do Mar: a presença Mbyá-Guarani em São Paulo. São Paulo: CTI & Nova Stella Ed.
Bertani, R. (2000). Male palpal bulbs and homologous features in Theraphosinae (Araneae, Theraphosidae). J. Arachnol. 28, 29–42. doi: 10.1636/0161-8202(2000)0280029:MPBAHF2.0.CO;2
Bertani, R. (2001). Revision, cladistic analysis, and zoogeography of Vitalius, Nhandu, and Proshapalopus; with notes on other theraphosine genera (Araneae, Theraphosidae). Arquivos Zoologia 36, 265–356.
Bertani, R., and Guadanucci, J. P. L. (2013). Morphology, evolution and usage of urticating setae by tarantulas (Araneae: Theraphosidae). Zoologia 30, 403–418. doi: 10.1590/S1984-46702013000400006
Bertani, R., and Leal, F. (2016). A new species of Pterinopelma (Araneae: Theraphosidae) from the highlands of the state of Minas Gerais, Brazil and description of the male of P. sazimai. Zoologia 33, 190. doi: 10.1590/S1984-4689zool-20150190
Bertani, R., Nagahama, R. H., and Fukushima, C. S. (2011). Revalidation of Pterinopelma Pocock 1901 with description of a new species and the female of Pterinopelma vitiosum (Keyserling 1891) (Araneae: Theraphosidae: Theraphosinae). Zootaxa 2814, 1–18. doi: 10.11646/zootaxa.2814.1.1
Bertani, R., Nagahama, R. H., and Fukushima, C. S. (2012). Vitalius nondescriptus comb. nov. (Araneae: Theraphosidae: Theraphosinae): an example of theraphosid taxonomic chaos. Zoologia 29, 467–473. doi: 10.1590/S1984-46702012000500011
Borges, L. M., Paladini, A., and Bertani, R. (2021). Two new species of Eupalaestrus Pocock, 1901 (Araneae: Theraphosidae) from southern Brazil and northeastern Argentina. J. Natural Commun. Hist. 55, 889–908. doi: 10.1080/00222933.2021.1925767
Borowiec, M. L. (2016). AMAS: a fast tool for alignment manipulation and computing of summary statistics. PeerJ 4, e1660. doi: 10.7717/peerj.1660
Borowiec, M. L. (2019). Spruceup: fast and flexible identification, visualization, and removal of outliers from large multiple sequence alignments. J. Open Sour. Softw. 4, 1635. doi: 10.21105/joss.01635
Branstetter, M. G., Longino, J. T., Ward, P. S., and Faircloth, B. C. (2017). Enriching the ant tree of life: Enhanced UCE bait set for genome-scale phylogenetics of ants and other Hymenoptera. Methods Ecol. Evol. 8, 768–776. doi: 10.1111/2041-210X.12742
Bücherl, W. (1957). Sobre a importância dos bulbos copuladores e das apófises tibiais dos machos na sistemática das aranhas caranguejeiras (Orthognatha). Anais da Academia Brasileira de Ciências. 29, 377–416.
Chamberlin, R. V. (1917). New spiders of the family Aviculariidae. Bull. Museum Compar. Zool. 61, 25–75.
Derkarabetian, S., Benavides, L. R., and Giribet, G. (2019). Sequence capture phylogenomics of historical ethanol-preserved museum specimens: Unlocking the rest of the vault. Mol. Ecol. Resour. 19, 1531–1544. doi: 10.1111/1755-0998.13072
Fabiano-Da-Silva, W., Guadanucci, J. P. L., and da Silva, M. B. (2019). Taxonomy and phylogenetics of Tmesiphantes Simon, 1892 (Araneae, Theraphosidae). System. Biodiver. 17, 650–668. doi: 10.1080/14772000.2019.1685021
Faircloth, B. C. (2016). PHYLUCE is a software package for the analysis of conserved genomic loci. Bioinformatics 32, 786–788. doi: 10.1093/bioinformatics/btv646
Faircloth, B. C., McCormack, J. E., Crawford, N. G., Harvey, M. G., Brumfield, R. T., and Glenn, T. C. (2012). Ultraconserved elements anchor thousands of genetic markers spanning multiple evolutionary timescales. System. Biol. 61, 717–726. doi: 10.1093/sysbio/sys004
Ferretti, N., and Barneche, J. (2012). New species of Eupalaestrus from Argentina (Araneae, Theraphosidae, Theraphosinae. Iheringia Série Zoologia 102, 327–330. doi: 10.1590/S0073-47212012000300012
Ferretti, N., Copperi, S., Schwerdt, L., and Pompozzi, G. (2014). First record of Vitalius roseus (Mello-Leitão, 1923)(Araneae: Theraphosidae: Theraphosinae) in Argentina: distribution map, natural history, and sexual behaviour. Arachnology 16, 241–243. doi: 10.13156/arac.2015.16.7.241
Ferretti, N., Copperi, S., Schwerdt, L., and Pompozzi, G. (2015). First record of Vitalius longisternalis Bertani, 2001 (Araneae, Theraphosidae) in Argentina and notes on its natural history in Misiones province. Check List 11, 1748–1748. doi: 10.15560/11.5.1748
Foley, S., Lüddecke, T., Cheng, D. Q., Krehenwinkel, H., Künzel, S., Longhorn, S. J., et al. (2019). Tarantula phylogenomics: a robust phylogeny of deep theraphosid clades inferred from transcriptome data sheds light on the prickly issue of urticating setae evolution. Mol. Phylogen. Evol. 140, 106573. doi: 10.1016/j.ympev.2019.106573
Fukushima, C. S., and Bertani, R. (2017). Taxonomic revision and cladistic analysis of Avicularia Lamarck, 1818 (Araneae, Theraphosidae, Aviculariinae) with description of three new aviculariine genera. ZooKeys 659, 1–185. doi: 10.3897/zookeys.659.10717
Gabriel, R., and Sherwood, D. (2020). Revised taxonomic placement of Pseudhapalopus Strand, 1907, with notes on some related taxa (Araneae: Theraphosidae). Arachnology 18, 301–316. doi: 10.13156/arac.2020.18.4.301
Galleti-Lima, A., and Guadanucci, J. P. L. (2018). Morphology of setae on the coxae and trochanters of theraphosine spiders (Mygalomorphae: Theraphosidae). J. Arachnol. 46, 214–225. doi: 10.1636/JoA-S-17-021.1
Gertsch, W. J., and Platnick, N. I. (1979). A revision of the spider family Mecicobothriidae (Araneae, Mygalomorphae). American Museum Novitates. 2687, 1–32.
Girard, C. (1853). “Arachnidians,” in Natural History of the Red River of Louisiana, Washington, 32nd Congress, 2nd Session, Senate Appendix F (Zoology), eds R. Marcy, 262–271. [reprinted twice in 1854: 33rd Congress, 1st Session, House of Representatives, 233–242; and 33rd Congress, 1st Session, Senate, 251–261].
Guadanucci, J. P. L. (2014). Theraphosidae phylogeny: relationships of the ‘Ischnocolinae'genera (Araneae, Mygalomorphae). Zool. Scripta 43, 508–518. doi: 10.1111/zsc.12065
Hamilton, C. A., Hendrixson, B. E., and Bond, J. E. (2016a). Taxonomic revision of the tarantula genus Aphonopelma Pocock, 1901(Araneae, Mygalomorphae, Theraphosidae) within the United States. Zookeys 560, 1–340. doi: 10.3897/zookeys.560.6264
Hamilton, C. A., Lemmon, A. R., Lemmon, E. M., and Bond, J. E. (2016b). Expanding anchored hybrid enrichment to resolve both deep and shallow relationships within the spider tree of life. BMC Evolut. Biol. 16, 1–20. doi: 10.1186/s12862-016-0769-y
Hedin, M., Derkarabetian, S., Alfaro, A., Ramírez, M. J., and Bond, J. E. (2019). Phylogenomic analysis and revised classification of atypoid mygalomorph spiders (Araneae, Mygalomorphae), with notes on arachnid ultraconserved element loci. PeerJ 7, e6864. doi: 10.7717/peerj.6864
Hedin, M., Derkarabetian, S., Ramírez, M. J., Vink, C., and Bond, J. E. (2018). Phylogenomic reclassification of the world's most venomous spiders (Mygalomorphae, Atracinae), with implications for venom evolution. Sci. Rep. 8, 1636. doi: 10.1038/s41598-018-19946-2
Holmberg, E. L. (1882). Observations à propos du sous-ordre des araignées territélaires (Territelariae), spécialement du genre nordaméricain Catadysas Hentz et de la sous-famille Mecicobothrioidae. Holmberg. Boletin de la Academia Nacional de Ciencias en Córdoba. 4, 153–174.
Kaderka, R., Bulantová, J., Heneberg, P., and Rezáč, M. (2019). Urticating setae of tarantulas (Araneae: Theraphosidae): morphology, revision of typology and terminology and implications for taxonomy. PLoS ONE 14, e0224384. doi: 10.1371/journal.pone.0224384
Katoh, K., and Standley, D. M. (2013). MAFFT multiple sequence alignment software version 7: improvements in performance and usability. Mol. Biol. Evolut. 30, 772–780. doi: 10.1093/molbev/mst010
Keyserling, E. (1891). Die Spinnen Amerikas. Brasilianische Spinnen. Bauer and Raspe, Nürnberg. 3, 1–278.
Kulkarni, S., Wood, H., Lloyd, M., and Hormiga, G. (2020). Spider-specific probe set for ultraconserved elements offers new perspectives on the evolutionary history of spiders (Arachnida, Araneae). Mol. Ecol. Resour. 20, 185–203. doi: 10.1111/1755-0998.13099
Lemmon, E. M., and Lemmon, A. R. (2013). High-Throughput genomic data in systematics and phylogenetics. Ann. Rev. Ecol. Evol. System. 44, 99–121. doi: 10.1146/annurev-ecolsys-110512-135822
Lucas, S. (1983). Descrição de gênero e espécie novos da subfamília Theraphosinae (Araneae, Ortoghatha, Theraphosidae. Memórias do Instituto Butantan. 44/45, 157–160.
Lucas, S., da Silva, P. I., and Jr. Bertani, R. (1993). Vitalius a new genus of the subfamily Theraphosinae Thorell, 1870 from Brazil (Araneae, Theraphosidae). Spixiana. 16, 241–245.
Lüddecke, T., Krehenwinkel, H., Canning, G., Glaw, F., Longhorn, S. J., Tänzler, R., et al. (2018). Discovering the silk road: Nuclear and mitochondrial sequence data resolve the phylogenetic relationships among theraphosid spider subfamilies. Mol. Phylogen. Evol. 119, 63–70. doi: 10.1016/j.ympev.2017.10.015
Mello-Leitão, C. F. (1926). Algumas Theraphosoideas novas do Brasil. Revista do Museu Paulista. 14, 307–324.
Mello-Leitão, C. F. (1929). Aranhas do Pernambuco, colhidas por D. Bento Pickel. Anais da Academia Brasileira de Ciências. 1, 91–112.
Mendoza, J., and Francke, O. (2017). Systematic revision of Brachypelma red-kneed tarantulas (Araneae: Theraphosidae), and the use of DNA barcodes to assist in the identification and conservation of CITES-listed species. Invertebrate System. 31, 157–179. doi: 10.1071/IS16023
Minh, B. Q., Schmidt, H. A., Chernomor, O., Schrempf, D., Woodhams, M. D., Von Haeseler, A., et al. (2020). IQ-TREE 2: new models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evolut. 37, 1530–1534. doi: 10.1093/molbev/msaa015
Nguyen, L. T., Schmidt, H. A., Von Haeseler, A., and Minh, B. Q. (2015). IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol. Biol. Evol. 32, 268–274. doi: 10.1093/molbev/msu300
Perafán, C., Cifuentes, Y., and Estrada-Gomez, S. (2015). Aguapanela, a new tarantula genus from the Colombian Andes (Araneae, Theraphosidae). Zootaxa 4033, 529–542. doi: 10.11646/zootaxa.4033.4.4
Perafán, C., and Valencia-Cuellar, D. (2018). Proshapalopus marimbai, a new tarantula species (Mygalomorphae, Theraphosidae) and first genus record from Colombia. Trop. Zool. 31, 200–213. doi: 10.1080/03946975.2018.1493181
Pérez-Miles, F., Lucas, S. M., da Silva, P. I. Jr., and Bertani, R. (1996). Systematic revision and cladistic analysis of Theraphosinae (Araneae: Theraphosidae). Mygalomorph 1, 33–68.
Pérez-Miles, F., and Perafán, C. (2020). “Theraphosinae,” in New World Tarantulas (Cham: Springer), 121–151. doi: 10.1007/978-3-030-48644-0_5
Pocock, R. I. (1901). Some new and old genera of South American Avicularidae. Ann. Magaz. Nat. History 8, 540–555. doi: 10.1080/03745480109443359
Pocock, R. I. (1903). On some genera and species of South-American Aviculariidae. Ann. Magazine Natural Hist. 11, 81–115. doi: 10.1080/00222930308678729
Rambaut, A. (2018). FigTree, Version 1.4. 4. Program Distributed By the Author. Institute of Evolutionary Biology, University of Edinburgh.
Raven, R. J. (1985). The spider infraorder Mygalomorphae (Araneae): cladistics and systematics. Bull. AMNH 182, 1.
Rudloff, J. P. (1997). Revision der Gattung Holothele Karsch, 1879 nebst Aufstellung einer neuen Gattung Stichoplastoris gen. nov. (Araneae, Theraphosidae) und Wiedereinsetzung einiger weiterer Gattungen der Mygalomorphae. Arachnol. Magazin 5, 1–19.
Sherwood, D., Gabriel, R., Brescovit, A. D., and Lucas, S. M. (2021). A new species of Cymbiapophysa Gabriel and Sherwood, 2020 from Colombia and a transfer from Proshapalopus Mello-Leitão, 1923 (Araneae: Theraphosidae). Arachnology 18, 838–843. doi: 10.13156/arac.2021.18.8.838
Simon, E. (1891). Etudes arachnologiques. 23e Mémoire. XXXVIII. Descriptions d'espèces et de genres nouveaux de la famille des Aviculariidae. Annal. Soc. Entomol. France 60, 300–312.
Simon, E. (1892). Histoire naturelle des araignées. Deuxième édition, tome premier. Paris: Roret. p. 1–256.
Smith, C. P. (1908). A preliminary study of the Araneae Theraphosidae of California. Ann. Entomol. Soc. Am. 1, 207–236. doi: 10.1093/aesa/1.4.207
Starrett, J., Derkarabetian, S., Hedin, M., Bryson, R. W. Jr., McCormack, J. E., and Faircloth, B. C. (2016). High phylogenetic utility of an ultraconserved element probe set designed for Arachnida. Mol. Ecol. Resour. 17, 812–823. doi: 10.1101/065201
Thorell, T. (1869). On European spiders. Part I. Review of the European genera of spiders, preceded by some observations on zoological nomenclature. Nova Acta Regiae Societatis Scientiarum Upsaliensis 7, 1–108.
Thorell, T. (1894). Förteckning öfver arachnider från Java och närgrändsande öar, insamlade af Carl Aurivillius; jemte beskrifningar å några sydasiatiska och sydamerikanska spindlar. Bihang till Kongliga Svenska Vetenskaps-Akademiens Handlingar 20, 1–63.
Turner, S. P., Longhorn, S. J., Hamilton, C. A., Gabriel, R., Pérez-Miles, F., and Vogler, A. P. (2018). Re-evaluating conservation priorities of New World tarantulas (Araneae: Theraphosidae) in a molecular framework indicates non-monophyly of the genera, Aphonopelma and Brachypelma. System. Biodiver. 16, 89–107. doi: 10.1080/14772000.2017.1346719
World Spider Catalog (2023). World Spider Catalog. Version 24. Natural History Museum Bern. Available online at: http://wsc.nmbe.ch (accessed February 6, 2023).
Xu, X., Su, Y. C., Ho, S. Y., Kuntner, M., Ono, H., Liu, F., et al. (2021). Phylogenomic analysis of ultraconserved elements resolves the evolutionary and biogeographic history of segmented trapdoor spiders. System. Biol. 70, 1110–1122. doi: 10.1093/sysbio/syaa098
Yeates, D. K., Zwick, A., and Mikheyev, A. S. (2016). Museums are biobanks: unlocking the genetic potential of the three billion specimens in the world's biological collections. Curr. Opin. Insect Sci. 18, 83–88. doi: 10.1016/j.cois.2016.09.009
Keywords: Mygalomorphae, Theraphosidae, new world species, phylogenomics, Taxonomy
Citation: Galleti-Lima A, Hamilton CA, Borges LM and Guadanucci JPL (2023) Phylogenomics of Lasiodoriforms: reclassification of the South American genus Vitalius Lucas, Silva and Bertani and allied genera (Araneae: Theraphosidae). Front. Ecol. Evol. 11:1177627. doi: 10.3389/fevo.2023.1177627
Received: 01 March 2023; Accepted: 24 May 2023;
Published: 15 June 2023.
Edited by:
Felipe Gobbi Grazziotin, Instituto Butantan, BrazilReviewed by:
Marc Domènech, University of Bristol, United KingdomCopyright © 2023 Galleti-Lima, Hamilton, Borges and Guadanucci. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Arthur Galleti-Lima, Z2FsbGV0aS5saW1hQHVuZXNwLmJy
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.