
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Chem. , 02 April 2025
Sec. Analytical Chemistry
Volume 13 - 2025 | https://doi.org/10.3389/fchem.2025.1549927
As an important inflammatory cytokine, interleukin-6 (IL-6) can mediate the entire pathological process of periodontitis and is closely associated with the degree of inflammation. Therefore, it is critical to develop convenient quantitative methods for monitoring IL-6 quantity in gingival crevicular fluid. In this study, methylene blue (MB)-decorated reduced graphene oxide (rGO) is employed as signal probe to further support the antibody-enabling specific recognition of IL-6. Due to π–π stacking and electrostatic interactions, rGO-MB nanocomposites can be stably obtained. rGO with good conductivity and large surface area characteristics promotes the redox signals of MB on the glassy carbon electrode (GCE). In addition, through the simple in situ self-polymerization of dopamine, the polydopamine (PDA) obtained can be not only directly used as a biological crosslinking agent for covalent immobilization of anti-IL-6 antibody but can also be regarded as a protective layer to enhance the stability of rGO-MB on the GCE surface. Such a designed PDA/rGO-MB/GCE-based immunosensor enables specific binding with IL-6 and produces a decreased electrochemical signal for MB, realizing the selective and sensitive quantitative measurement of IL-6. Consequently, our fabricated PDA/rGO-MB/GCE-based electrochemical immunosensor has an excellent linear relationship with IL-6 ranging from 1 pg/mL to 100 ng/mL, with a limit of detection as low as 0.48 pg/mL. Moreover, our as-prepared sensing strategy shows accurate monitoring of the IL-6 quantity in gingival crevicular fluid samples.
Periodontal disease is a chronic nonspecific condition with a 70%–85% prevalence in China, making it a major cause of tooth loss in middle-aged and older adults. Clinical diagnosis mainly relies on periodontal examination indices and digital subtraction radiography. However, this diagnostic approach has certain delays, making it difficult to assess periodontal risk early and evaluate the activity of periodontal disease in a timely manner. With the continuous development of molecular biology and biological detection technologies, many researchers have begun to focus on the study of biomarkers in gingival crevicular fluid during the active phase of periodontitis. Interleukin-6 (IL-6) is a kind of important inflammatory cytokine which can mediate the entire pathological process of periodontitis and is closely associated with the degree of inflammation (Noh et al., 2013; Kardeşler et al., 2011). Therefore, developing simple, accurate and cost-effective analytical techniques to meet the demand of point-of-care testing in clinical detection is important.
Currently, enzyme-linked immunosorbent assay is the standard technique for the quantitative monitoring of IL-6 in clinical studies (Kingsmore, 2006). However, there are several disadvantages to this method—it is time-consuming and requires a large sample volume and complex operation. Electrochemical sensors offer a powerful analytical tool with numerous merits, including rapid response, high selectivity and sensitivity, cost-effectiveness, portability, facile operation, and easy miniaturization (Su et al., 2022; Huang L. et al., 2023; Zeng et al., 2023). These advantages make them invaluable for a wide range of applications in diverse fields (Zhou et al., 2024a; Wu et al., 2025; Fan et al., 2024). There have been many studies on electrochemical biosensors for the sensitive determination of IL-6 (McCrae et al., 2023; Zhang W. et al., 2023; Zhu et al., 2025). Recognition elements can be categorized as aptamer, antibody, and IL-6 receptor. Electrochemical immunosensors based on the high specific affinity between antibody and IL-6 have received growing attention, and it is highly desirable to design convenient electrochemical immunosensors for the sensitive determination of IL-6.
Electrochemical sensors based on immobilized electroactive probes on the surface of the sensing interface provides an attractive analytical strategy thanks to the reagent saving and simplified assay procedure, indicating it as a versatile biosensing platform for practical applications (Ma et al., 2024; Zhang et al., 2024; Li et al., 2023). Several organic dyes (e.g., methylene blue (MB) (Krishnan et al., 2023) and thionine (Ding et al., 2022)) or transition-metal complex ions (e.g., tris(hexa-ammineruthenium (III) (Huang Z. et al., 2023), potassium ferricyanide (Yan et al., 2022a; Zhang T. et al., 2023; Guo et al., 2023), 1,10-phenanthroline)iron (II) (Duan et al., 2024; Huang et al., 2024), tris(2,2-bipyridine)ruthenium (II) (Yang et al., 2022; Gao et al., 2025), and tris(1,10-phenanthroline)ruthenium (II) (Chen et al., 2022)) serving as redox indicators have been applied in the field of electroanalytical determination. Among these, MB molecules with positive charges possess water solubility, inherent redox property, and good reversibility. Moreover, the formal potential of MB molecules far exceeds that of common co-existing substances (e.g., uric acid and ascorbic acid) in biological samples, and it is suitable for practical sample analysis application. In comparison with MB molecules in solution, reagentless electrochemical immunosensors with MB molecules immobilized on the electrode surface show merits of sparing reagents and cost, convenient detection procedure, and reusability (Zhou et al., 2023; Chen et al., 2023). Graphene and its derivatives have shown great potential in the field of biosensors due to their unique properties such as high surface area, high catalytic activity, excellent electrical conductivity, and chemical stability (Zhang C. et al., 2023; Zou et al., 2022; Zhou et al., 2024b; Zhao et al., 2025; Zhou et al., 2022; Deng et al., 2021; Zhu et al., 2022). To increase electrode conductivity, reduced graphene oxide (rGO) (Chen et al., 2013) and carbon nanotubes (Zhang et al., 2022) have been employed to hybridize with MB molecules to accelerate the electron transfer procedure of MB on the sensing interfaces.
This study reported a simple electrochemical immunosensor for the sensitive determination of IL-6 based on the rGO-MB nanocomposite. The rGO-MB nanocomposite was obtained by simply mixing rGO and MB solution via π–π stacking and electrostatic adsorption interactions. Polydopamine self-polymerized from dopamine not only forms a uniform protective layer but also offers functional sites (dopaquinone) for grafting species bearing −SH or −NH2 moieties (Alfieri et al., 2022). Arising from the dual-functional property of PDA, rGO-MB nanocomposite can be stably modified on the supporting electrode surface (glassy carbon electrode—GCE), and antibody corresponding to IL-6 can then be covalently immobilized on the electrode. Such obtained rGO-MB/GCE-based immunosensor integrates the electrochemical probe (MB), electron transfer accelerator (rGO), and biological recognition element (antibody) into the sensing interface, realizing the reagentless electrochemical measurement of IL-6. Moreover, analysis of IL-6 in gingival crevicular fluid was tested by our fabricated immunosensor, offering a universal platform to detect a series of important biomarkers.
Monolayered graphene oxide (GO) aqueous dispersion was provided from Hang-zhou Gaoxi Technology Co., Ltd. (Hangzhou, China) at a 1 mg/g concentration. Several inflammatory factors, including interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), matrix metalloproteinase-9 (MMP-9), interferon-γ (IFN-γ), c-reactive protein (CRP), interleukin-6 (IL-6), and IL-specific antibody, were ordered from Beijing KEY-BIO Biotech Co., Ltd. (Beijing, China). Methylene blue trihydrate (MB), glucose (Glu, 100%), uric acid (UA), ascorbic acid (AA), 3-hydroxytyramine hydrochloride (DA), bovine serum albumin (BSA), potassium ferrocyanide (K4Fe(CN)6, 99.5%), potassium ferricyanide (K3Fe(CN)6, 99.5%), sodium hydroxide (NaOH, 97%), sodium chloride (NaCl), disodium hydrogen phosphate dodecahydrate (Na2HPO4·12H2O, 99%), potassium chloride (KCl, 99.5%), and sodium dihydrogen phosphate dihydrate (NaH2PO4·2H2O, 99%) were bought from Shanghai Aladdin Bio-Chem Technology Co., Ltd. (Shanghai, China). Hydrazine hydrate (80%) was obtained from Hangzhou Shuanglin Chemical Reagent Co., Ltd. (Hangzhou, China). Ammonia solution (25.0–28.0 wt%) was bought from Hangzhou Gaojing Fine Chemical Co., Ltd. (Hangzhou, China). Phosphate-buffered saline (PBS) was obtained by mixing NaH2PO4·2H2O and Na2HPO4·12H2O. Gingival crevicular fluid samples provided by a healthy man were received from the Beijing Hospital of Integrated Traditional Chinese and Western Medicine (Beijing, China). All aqueous solutions were prepared by ultrapure water (Mill-Q Sys-tems, Millipore, 18.2 MΩ cm) throughout the study.
Ultraviolet–Vis (UV-Vis) absorption spectra were recorded on a UV-2450 UV-Vis spectrometer (Shimadzu, Japan). Scanning electron microscopy (SEM) measurements were carried out with a GeminiSEM 360 (Germany, ZEISS) scanning emission electron microscope. X-ray photoelectron spectroscopy (XPS) analyses were performed on a PHI5300 electron spectrometer (PE Ltd., United States) with Mg Kα radiation of 250 W and 14 kV.
A PGSTAT302N Autolab electrochemical workstation (Metrohm Autolab, Switzerland) was used to conduct cyclic voltammetry (CV), electrochemical impedance spectroscopy (EIS), and differential pulse voltammetry (DPV) measurements. Bare or modified GCE, an Ag/AgCl, and a platinum wire were regarded as the working, reference, and auxiliary electrodes, respectively. The experimental parameters for EIS measurements are: frequency range (0.1 Hz–100 kHz) and perturbation amplitude (5 mV).
Reduced graphene oxide (abbreviated as rGO) nanosheets were synthesized as per Li et al. (2008). Briefly, 32 mL H2O, hydrazine solution (7.0 μL, 80 wt%), and 120 μL ammonia solution (25–28 wt%) were added to a round-bottom flask containing GO dispersion solution (8 mL, 1 mg/mL) and then stirred in a water bath at 85°C for 3 h. Subsequently, stable rGO dispersion was obtained after centrifugation at 3,000 rpm for 30 min.
MB-decorated rGO nanocomposite (MB-rGO) was synthesized by mixing rGO (850 μL, 0.2 mg/mL) and MB (150 μL, 0.2 mg/mL), and the dispersion was then subjected to ultrasonic treatment for 30 min. After centrifugation at 12,000 rpm three times for 10 min each time to exclude the supernatant, the precipitate was collected—rGO-MB hybrid. Finally, the precipitate was redispersed in 500 μL ultrapure water to obtain an rGO-MB solution.
GCE was polished using different specifications of alumina powder (0.5 μm, 0.3 μm, and 0.05 μm) in sequence. Next, GCE was immersed in anhydrous ethanol and ultrapure water alternatively under ultrasonication. After cleaning with a large amount of water, GCE was coated by rGO-MB complex solution (10 μL) and further dried at 60°C to finally achieve GCE modified with rGO-MB—“rGO-MB/GCE”.
The rGO-MB/GCE modified with a polydopamine (PDA) layer (PDA/rGO-MB/GCE) was obtained by immersing rGO-MB/GCE in a 0.1 M PBS (pH = 8.5) containing 0.38 mg/mL DA solution was then washed with ultrapure water to exclude the redundant DA on the sensing interface. We then coated 10 μL antibody possessing high affinity to IL-6 (10 μg/mL, dispersed in a 0.01 M PBS (pH 7.4)) on the PDA/rGO-MB/GCE for incubation at 4°C for 60 min. Subsequently, the electrode obtained was rinsed with 0.01 M PBS (pH = 7.4) several times and coated with 1 wt% BSA at 4°C for 30 min to avoid nonspecific adsorption. Ultimately, the immunosensing interface was designated as the “BSA/Ab/PDA/rGO-MB/GCE”, which was used to quantitatively analyze IL-6 amounts.
IL-6 at various concentrations was subsequently coated on the BSA/Ab/PDA/rGO-MB/GCE surface and incubated at 4°C for 60 min. Then, BSA/Ab/PDA/rGO-MB/GCE after binding with target IL-6 was placed into a 0.1 M PBS (pH = 7.4) solution and tested by electrochemical techniques. For the analysis of IL-6 in gingival crevicular fluid samples, the samples directly received were diluted with 0.01 M PBS (pH = 7.4, dilution ratio is 100) and spiked with several known concentrations of IL-6. Our fabricated BSA/Ab/PDA/rGO-MB/GCE sensor was employed to test the above real samples, and the test results were compared with the known concentrations of IL-6.
Scheme 1 illustrates the fabricated procedure of the BSA/Ab/PDA/rGO-MB/GCE immunosensor and its mechanism for the electrochemical determination of IL-6. Both rGO and MB bear the π-conjugated structure and carry the opposite charges (negative charges for rGO and positive charges for MB; the pKa of MB is 11.6) in the tested condition (pH 7.4). rGO-MB nanocomposite can be prepared by simply mixing rGO dispersion solution with MB solution via π–π stacking and electrostatic interactions, which is subsequently dropped onto the clean GCE surface to obtain rGO-MB/GCE. rGO with the characteristics of good conductivity and large surface area promotes the redox signals of MB on the GCE surface. Then, PDA self-polymerized from DA under weak alkaline conditions can directly form a dense and adherent layer on the surface of rGO-MB/GCE, which not only acts as the protective layer for avoiding the leakage of the inner rGO-MB layer but also provides functional sites for the further modification of anti-IL-6 antibody. Note that −SH or −NH2 groups on the anti-IL-6 antibody can react with many functional groups (e.g., catechol, amine) on PDA through Schiff-based or Michael addition reactions, which enable the development of highly specific and sensitive biosensors. After avoiding non-specific adsorption using BSA, the electrochemical immunosensing interface is achieved, denoted “BSA/Ab/PDA/rGO-MB/GCE”. Arising from the specific immunoaffinity towards target IL-6, IL-6 can be captured at the BSA/Ab/PDA/rGO-MB/GCE and hinders the electron transfer procedure of immobilized MB molecules, resulting in a decline in electrochemical signals. Therefore, the decreased electrochemical signals exhibit a quantitative relationship with the IL-6 concentration, enabling the accurate monitoring of IL-6 quantity.
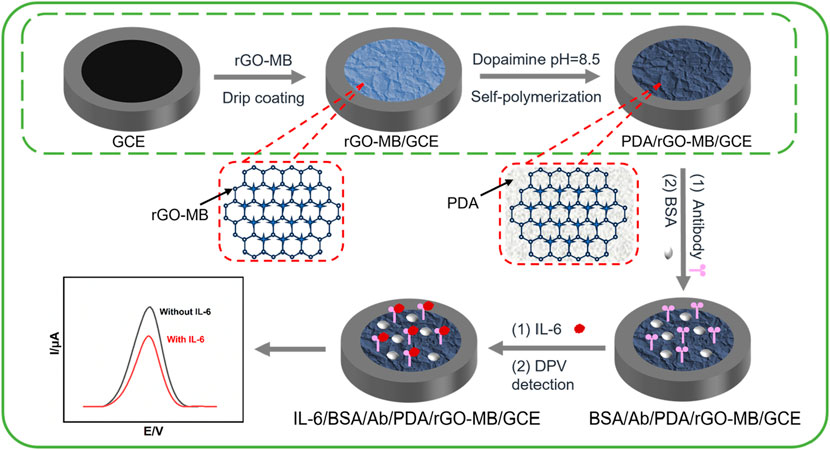
Scheme 1. Fabrication procedure of the BSA/Ab/PDA/rGO-MB/GCE immunosensor and the electrochemical determination of IL-6.
To confirm the successful preparation of rGO-MB nanocomposite, techniques including scanning electron microscopy (SEM), UV–visible spectroscopy (UV-vis), and X-ray photoelectron spectroscopy (XPS) were performed. As shown in Figures 1A and B, rGO has some wrinkles, and the introduction of MB does not affect the structure of rGO. As shown in Figure 1C, rGO has a weak adsorption peak at 253 nm in UV–vis spectrum, which is attributed to the π–π* transition of C–C/C=C bonds in the aromatic structure of rGO (red curve) (Chang et al., 2022; Yan et al., 2022b). The strong adsorption peak at 664 nm shown in MB arises from the π–π* transition of aromatic structure in the MB monomer (black curve). In contrast, rGO-MB nanocomposite remains the adsorption peak at 253 nm of rGO and shifts the prominent absorption peak at 664 nm of MB to 685 nm, implying that the MB and rGO planes are close and further verifying the successful preparation of rGO-MB nanocomposite. By comparing the C1s XPS spectra of GO and rGO-MB nanocomposite (Figures 1D, E), the intensities of oxygen-containing groups (C–O, C=O and O–C=O), remarkably decline, suggesting the presence of rGO in the rGO-MB nanocomposite (Lv et al., 2022; Cui et al., 2023; Zheng et al., 2022). In addition, rGO-MB shows the typical characteristic peak of S 2p (Figure 1F), which is the S element in MB molecules. Based on the above data, the hybrid material consisting of rGO and MB (rGO-MB) nanocomposite was successfully prepared.
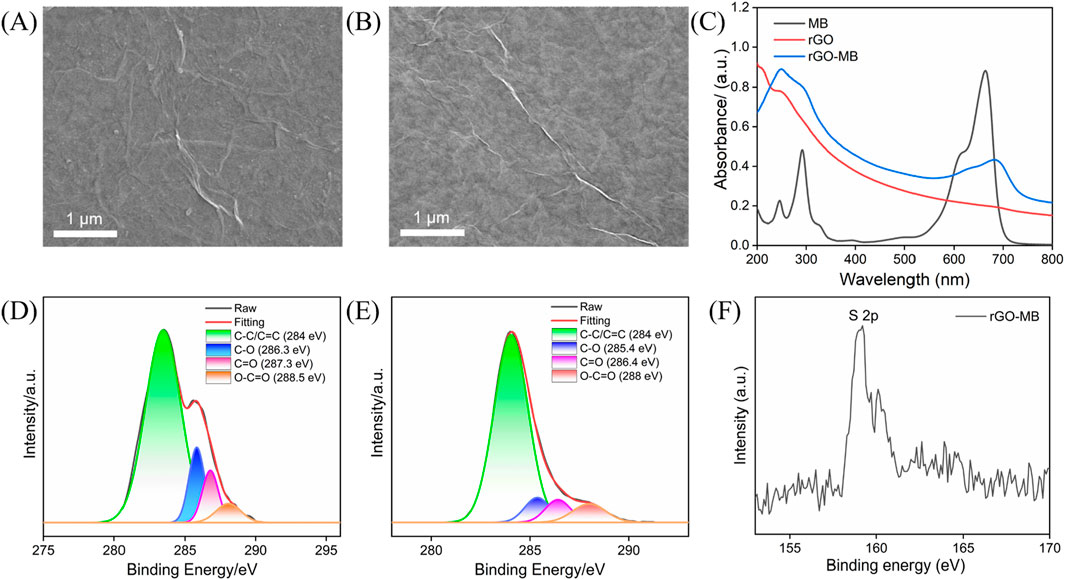
Figure 1. Characterizations of rGO-MB: (A,B) SEM, rGO (A) and rGO-MB nanocomposite (B); (C) UV–Vis; (D–F) XPS, C1s spectra of GO (D) and rGO-MB (E), and S 2p XPS spectrum of r GO-MB (F).
After dropping the rGO-MB nanocomposite onto the electrode surface, the electrochemical behavior of the rGO-MB/GCE surface was studied by cyclic voltammetry (CV). Figure 2A depicts the CV curves of bare GCE, rGO/GCE, MB/GCE, and rGO-MB/GCE in buffer solution. No redox peak was found at either the bare GCE or rGO/GCE. Moreover, the capacity current of rGO/GCE is apparently larger than that of bare GCE, which is due to the larger active surface area of rGO. A pair of redox peaks is displayed at the GCE modified with MB molecules (MB/GCE), with a formal potential of −0.183 V (anodic peak potential (Epa) at −0.148 V and cathodic peak potential (Epc) at −0.217 V), which is assigned to the electrochemical reaction procedure of MB molecules. In contrast, the co-existing rGO with MB is able to accelerate the electron transfer of MB on the GCE, giving rise to a couple of well-defined redox peaks at the MB-rGO/GCE and decreased formal potential (−0.289 V, Epa at −0.250 V and Epc at −0.328 V). To verify the stabilization effect of the PDA layer for inner rGO-MB nanocomposite, rGO/GCE and PDA/rGO-MB/GCE were successively scanned in buffer solution for ten cycles. As shown in Figures 2B and C, both the anodic (Ipa) and cathodic (Ipc) peak current of MB molecules at the rGO-MB/GCE gradually decrease with the increasing scanning cycles, whereas those at the PDA/rGO-MB/GCE remain unchanged, demonstrating the excellent stability of PDA/rGO-MB/GCE due to the PDA layer. In addition, the effect of the scan rate on the redox peak currents at the PDA/rGO-MB/GCE was recorded by CV, with the results displayed in Figure 3. It can be determined that redox peak currents of MB molecules at the PDA/rGO-MB/GCE are enhanced with an increase of scan rate in the range of 20 mV/s to 160 mV/s. The obtained Ipa or Ipc displays good linear relationship with scan rate (v), yielding two linear fitting equations of Ipa (μA) = 0.120 v (mV/s) −0.608 (R2 = 0.997) and Ipc (μA) = −0.161 v (mV/s) + 1.77 (R2 = 0.997). This indicates that the electrochemical reaction of immobilized MB at the PDA/rGO-MB/GCE is surface-controlled.
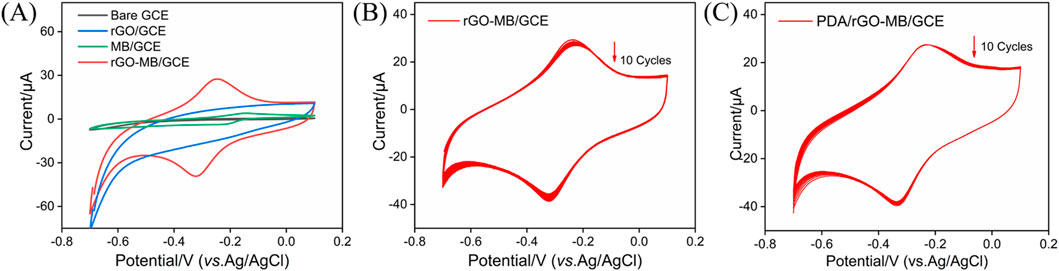
Figure 2. (A) CV plots of bare GCE, rGO/GCE, MB/GCE, and rGO-MB/GCE in 0.1 M PBS (pH = 7.4). Continuous CV responses of rGO-MB/GCE (B) and PDA/rGO-MB/GCE (C) in 0.1 M PBS (pH = 7.4). Scan rate: 50 mV/s.
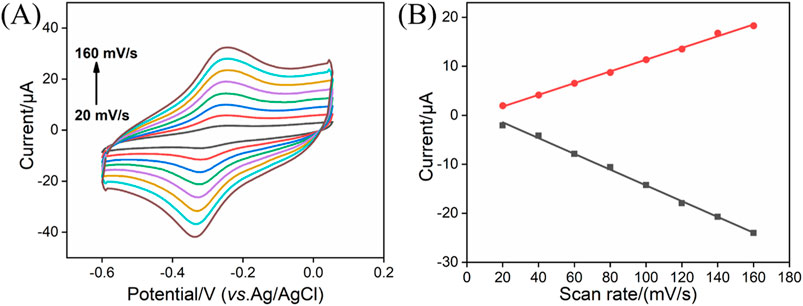
Figure 3. (A) CV curves of PDA/rGO-MB/GCE in 0.1 M PBS (pH = 7.4) at different scan rates (20–160 mV/s). (B) Relationship between redox peak currents and scan rate.
Stepwise fabrication of the BSA/Ab/PDA/rGO-MB/GCE immunosensor and its feasibility for IL-6 detection was studied by CV, DPV, and EIS. Figures 4A and B show the CV (a) and DPV (b) of rGO-MB/GCE, PDA/rGO-MB/GCE, Ab/PDA/rGO-MB/GCE, and BSA/Ab/PDA/rGO-MB/GCE before and after testing 1 ng/mL IL-6 in detection solution. As is evident, electrochemical responses at the rGO-MB/GCE decrease after consecutive modification of PDA, anti-IL-6 antibody (Ab), and BSA thanks to their poor conductivity impeding electron transfer at the sensing interface. Upon incubation with 1 ng/mL target IL-6, a remarkably reduced electrochemical signal is observed at the BSA/Ab/PDA/rGO-MB/GCE sensor, suggesting the potential of our developed biosensor for monitoring IL-6 amounts. In addition, the constructed immunosensor was verified by EIS in Figure 4C. As seen, the semicircle diameter in the high-frequency region corresponding to the charge transfer resistance at the electrode/electrolyte interface progressively increases with the stepwise modification procedure and IL-6 detection.
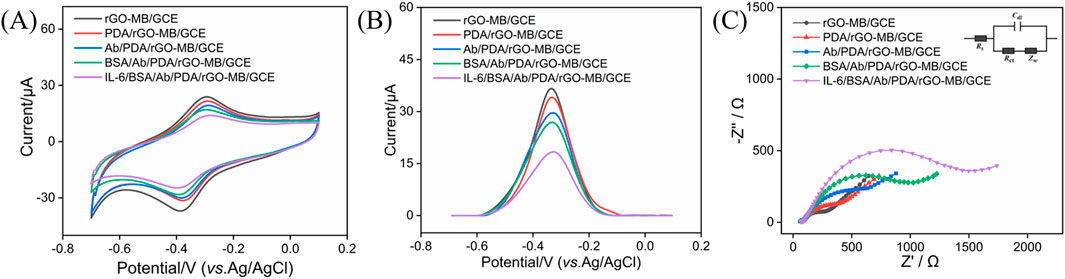
Figure 4. CV (A) and DPV plots (B) of rGO-MB/GCE, PDA/rGO-MB/GCE, Ab/PDA/rGO-MB/GCE, and BSA/Ab/PDA/rGO-MB/GCE before and after testing with 1 ng/mL IL-6 in 0.1 M PBS (pH = 7.4). (C) EIS curves of different electrodes tested in 0.1 M KCl containing 2.5 mM Fe(CN)63−/4−. The inset in (C) is an equivalent circuit, including electron transfer resistance (Rct), two-layer capacitance (Cdl), solution resistance (Rs), and Warburg impedance (Zw).
Prior to the IL-6 detection, the incubation times of anti-IL-6 antibody and target IL-6 were studied, with the results shown in Figures 5A and B. As is evident, electrochemical response can be unchanged after 60 and 90 min, respectively. Therefore, 60 and 90 min were selected as the optimal incubation time for anti-IL-6 antibody and target IL-6, respectively. Figure 5C shows the representative DPV curves of the BSA/Ab/PDA/rGO-MB/GCE immunosensor before and after incubation with various concentrations of IL-6 (1 pg/mL, 10 pg/mL, 100 pg/mL, 1 ng/mL, 10 ng/mL, and 100 ng/mL). As is evident, the obtained anodic peak current (I) decreases gradually with the increasing concentration of IL-6 (CIL-6). According to the DPV results shown in Figure 5C, a good fitting linear relationship between the I and the logarithm of IL-6 concentration was achieved, yielding a regression equation of I (μA) = −2.26 log CIL-6 (ng/mL) + 19.1 with R2 of 0.997 (Figure 5D). The limit of detection (LOD) is calculated by substituting the minimum acceptable signal (XL) into the above linear equation. XL can be obtained using XL = Xb + 3Sb, where Xb denotes the average response signal to the blank solution, with Sb as the standard deviation of its 11 time-measured signals. Based on the above method, LOD achieved at the BSA/Ab/PDA/rGO-MB/GCE sensor is 0.48 pg/mL. Table 1 compares the analytical performance—including electrochemical technique, linear range, and LOD of our fabricated BSA/Ab/PDA/rGO-MB/GCE immunosensor—with other electrochemical immunosensors. As seen, our fabricated BSA/Ab/PDA/rGO-MB/GCE immunosensor has a comparable wide linear range and low LOD. In addition, our as-prepared immunosensor integrates the electroactive probe (MB) into the sensing interface, which spares the addition of a probe into the detection solution and demonstrates the convenience of the operation.
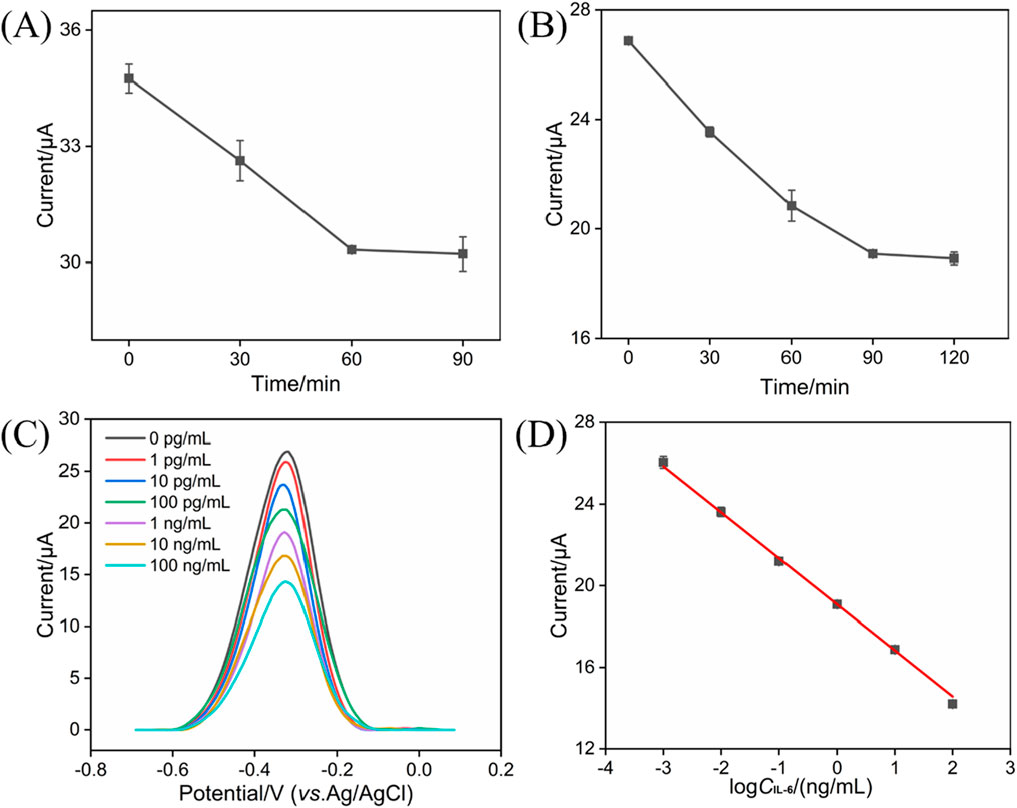
Figure 5. (A) Effect of incubation time between IL-6-specific antibody and the PDA/rGO-MB/GCE on the current response. The concentration of anti-IL-6 antibody is 10 μg/mL. (B) Effect of incubation time between the BSA/Ab/PDA/rGO-MB/GCE and target IL-6 on the obtained current response. The concentration of IL-6 is 1 ng/mL. (C) DPV curves of the BSA/Ab/PDA/rGO-MB/GCE immunosensor to various concentrations of IL-6 (1 pg/mL, 10 pg/mL, 100 pg/mL, 1 ng/mL, 10 ng/mL, and 100 ng/mL). (D) Corresponding calibration curve of IL-6 detection. Error bars in (A, B, D) are standard deviations (SDs) of three measurements.
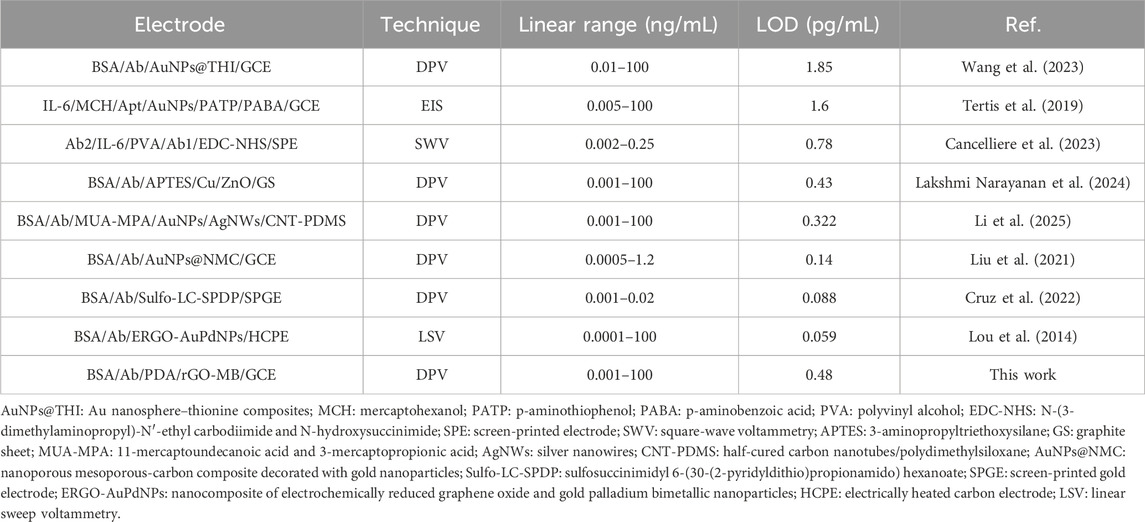
Table 1. Comparison of detection performance of different reported electrochemical strategies based on the immunocomplex reaction for monitoring IL-6 amounts.
Parameters such as selectivity, repeatability, and stability are very important for evaluating electrochemical sensors. Several co-existing substances which may interfere with IL-6 detection in real samples (e.g., gingival crevicular fluid) were incubated with the BSA/Ab/PDA/rGO-MB/GCE immunosensor and tested by the DPV technique. As shown in Figure 6A, glucose (Glu), K+, Na+, c-reactive protein (CRP), interferon-γ (IFN-γ), interleukin-1β (IL-1β), tumor necrosis factor-α (TNF-α), and matrix metalloproteinase-9 (MMP-9) at the ten-fold concentration of IL-6 show negligible influence on the anodic peak current variation, suggesting the excellent selectivity of the immunosensor design. In addition, a mixture of the above interfering species and target IL-6 results in the comparable electrochemical signal variation with IL-6 at the BSA/Ab/PDA/rGO-MB/GCE immunosensor, demonstrating the satisfactory anti-interference ability of our proposed immunosensor. Figure 6B shows the anodic peak currents obtained at the seven BSA/Ab/PDA/rGO-MB/GCE immunosensors fabricated in parallel after incubation with 1 ng/mL IL-6. As shown, a rather low RSD of 1.2% is achieved, indicating the good repeatability of our proposed immunosensor. The stability of the fabricated BSA/Ab/PDA/rGO-MB/GCE immunosensor was evaluated by testing the anodic peak current of BSA/Ab/PDA/rGO-MB/GCE immunosensor after incubation with 1 ng/mL IL-6 over a 9-day storage at 4 °C. As seen in Figure 6C, the rather stable current response with an RSD value of 0.9% implies the excellent stability of the fabricated immunosensor.
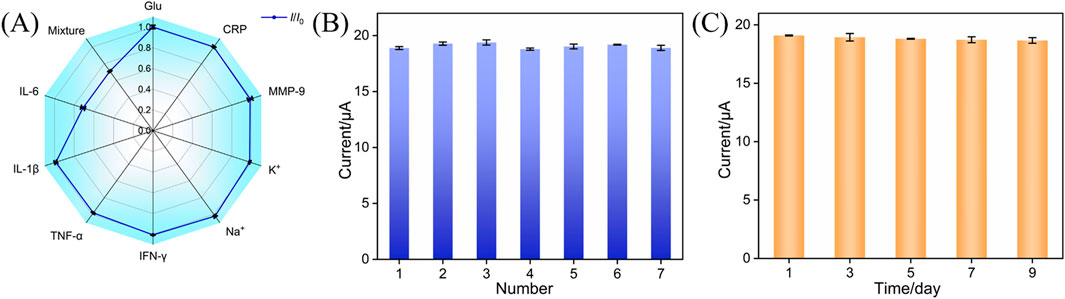
Figure 6. (A) Anodic peak currents tested at the proposed BSA/Ab/PDA/rGO-MB/GCE immunosensor before (I0) and after (I) testing with ten-fold concentration of Glu, CRP, MMP-9, K+, Na+, IFN-γ, TNF-α, IL-1β, IL-6 (1 ng/mL), or their mixture. (B) Anodic peak currents obtained on different BSA/Ab/PDA/rGO-MB/GCE immunosensors fabricated in parallel after incubation with 1 ng/mL IL-6. (C) Anodic peak currents obtained at the developed BSA/Ab/PDA/rGO-MB/GCE immunosensor to 1 ng/mL IL-6 after storage for different days. The error bars in (A–C) are the SD of three measurements and the tested solution is 0.1 M PBS (pH = 7.4).
Table 2 lists the tested results in gingival crevicular fluid samples by our as-prepared BSA/Ab/PDA/rGO-MB/GCE and the known spiked concentrations of IL-6. It can be found that the addition of standard concentrations of IL-6 (0.0100 ng/mL, 1.00 ng/mL and 100 ng/mL) in gingival crevicular fluid samples of a healthy human can be accurately detected by the BSA/Ab/PDA/rGO-MB/GCE with acceptable RSD values (<1.3%) and recoveries (97.3–99.3%). The above data indicate the capacity of BSA/Ab/PDA/rGO-MB/GCE for monitoring IL-6 amount in gingival crevicular fluid samples.
We developed a simple electrochemical immunosensor for the highly sensitive determination of IL-6 detection based on the electrochemical signal of immobilized MB-rGO nanocomposite and the immunoaffinity of anti-IL-6 antibody. The MB-rGO nanocomposite can be obtained by simply mixing MB molecules and rGO solution via π–π stacking and electrostatic interactions. The large surface area and good conductivity of rGO promote the redox signal of MB molecules, leading to an amplified electrochemical signal of immobilized MB-rGO nanocomposite on the GCE surface. Functionalization of PDA at the designed MB-rGO/GCE not only improves the stability of the electrochemical signal but also provides active sites for further immobilizing the anti-IL-6 antibody. When an MB-rGO/GCE-based immunosensor is incubated with target IL-6, an immunocomplex is formed at the sensing interface and leads to the decreased electrochemical signal of immobilized MB molecules, enabling the sensitive determination of IL-6. The proposed MB-rGO/GCE-based immunosensor with excellent selectivity, repeatability, and stability is capable of detecting IL-6 in gingival crevicular fluid samples, providing a universal probe-integrated electrochemical immunoplatform for a broad range of important biomarkers.
The original contributions presented in the study are included in the article/supplementary material; further inquiries can be directed to the corresponding author.
CZ: data curation, formal analysis, methodology, and writing–original draft. HW: data curation, formal analysis, investigation, methodology, and writing–original draft. JL: conceptualization, supervision, and writing–review and editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the National Natural Science Foundation of China (22374131).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Alfieri, M. L., Weil, T., Ng, D. Y. W., and Ball, V. (2022). Polydopamine at biological interfaces. Adv. Colloid Interface Sci. 305, 102689. doi:10.1016/j.cis.2022.102689
Cancelliere, R., Cosio, T., Campione, E., Corvino, M., D’Amico, M. P., Micheli, L., et al. (2023). Label-free electrochemical immunosensor as a reliable point-of-care device for the detection of Interleukin-6 in serum samples from patients with psoriasis. Front. Chem. 11, 1251360. doi:10.3389/fchem.2023.1251360
Chang, Q., Huang, J., He, L., and Xi, F. (2022). Simple immunosensor for ultrasensitive electrochemical determination of biomarker of the bone metabolism in human serum. Front. Chem. 10, 940795. doi:10.3389/fchem.2022.940795
Chen, D., Luo, X., and Xi, F. (2023). Probe-integrated electrochemical immunosensor based on electrostatic nanocage array for reagentless and sensitive detection of tumor biomarker. Front. Chem. 11, 1121450. doi:10.3389/fchem.2023.1121450
Chen, H., Huang, J., Zhang, R., and Yan, F. (2022). Dual-mode electrochemiluminescence and electrochemical sensor for alpha-fetoprotein detection in human serum based on vertically ordered mesoporous silica films. Front. Chem. 10, 1023998. doi:10.3389/fchem.2022.1023998
Chen, J. R., Jiao, X. X., Luo, H. Q., and Li, N. B. (2013). Probe-label-free electrochemical aptasensor based on methylene blue-anchored graphene oxide amplification. J. Mater. Chem. B 1 (6), 861–864. doi:10.1039/c2tb00267a
Cruz, A., Vieira, M., Mesquita, A. R., Sampaio, A., Mendes-Pinto, I., Soares, I., et al. (2022). Non-invasive diagnosis and monitoring tool of children’s mental health: a point-of-care immunosensor for IL-6 quantification in saliva samples. Front. Neurosci. 16, 919551. doi:10.3389/fnins.2022.919551
Cui, Y., Zhang, S., Zhou, X., Yan, F., and Hu, W. (2023). Silica nanochannel array on co-electrodeposited graphene-carbon nanotubes 3D composite film for antifouling detection of uric acid in human serum and urine samples. Microchem. J. 190, 108632. doi:10.1016/j.microc.2023.108632
Deng, X., Zhao, J., Ding, Y., Tang, H., and Xi, F. (2021). Iron and nitrogen co-doped graphene quantum dots as highly active peroxidases for the sensitive detection of l-cysteine. New J. Chem. 45, 19056–19064. doi:10.1039/d1nj03559b
Ding, M., Niu, H., Zhang, N., Hou, T., Guan, P., and Hu, X. (2022). Facile fabrication of electrochemically reduced graphene oxide/polythionine-methylene blue and its use as a platform for detection of nicotinamide adenine dinucleotide in the artificial urine sample. Electrochim. Acta 425, 140715. doi:10.1016/j.electacta.2022.140715
Duan, L., Zhang, C., Xi, F., Su, D., and Zhang, W. (2024). Direct and sensitive electrochemical determination of total antioxidant capacity in foods using nanochannel-based enrichment of redox probes. Molecules 29, 2423. doi:10.3390/molecules29112423
Fan, X., Wu, J., Zhang, T., and Liu, J. (2024). Electrochemical/electrochemiluminescence sensors based on vertically-ordered mesoporous silica films for biomedical analytical applications. ChemBioChem 25, e202400320. doi:10.1002/cbic.202400320
Gao, J., Zhang, S., and Xi, F. (2025). Homogeneous aptasensor with electrochemical and electrochemiluminescence dual detection channels enabled by nanochannel-based probe enrichment and DNase I cleavage for tumor biomarker detection. Molecules 30, 746. doi:10.3390/molecules30030746
Guo, Q., Fan, X., Yan, F., and Wang, Y. (2023). Highly sensitive electrochemical immunosensor based on electrodeposited platinum nanostructures confined in silica nanochannels for the detection of the carcinoembryonic antigen. Front. Chem. 11, 1271556. doi:10.3389/fchem.2023.1271556
Huang, J., Fan, X., Yan, F., and Liu, J. (2024). Vertical silica nanochannels and o-phenanthroline chelator for the detection of trace Fe(II). ACS Appl. Nano Mater. 7, 7743–7752. doi:10.1021/acsanm.4c00385
Huang, L., Su, R., and Xi, F. (2023a). Sensitive detection of noradrenaline in human whole blood based on Au nanoparticles embedded vertically-ordered silica nanochannels modified pre-activated glassy carbon electrodes. Front. Chem. 11, 1126213. doi:10.3389/fchem.2023.1126213
Huang, Z., Luo, X., Yan, F., and Zhou, B. (2023b). Homogeneous electrochemical aptasensor for sensitive detection of zearalenone using nanocomposite probe and silica nanochannel film. Molecules 28 (21), 7241. doi:10.3390/molecules28217241
Kardeşler, L., Buduneli, N., Çetinkalp, Ş., Lappin, D., and Kinane, D. F. (2011). Gingival crevicular fluid IL-6, tPA, PAI-2, albumin levels following initial periodontal treatment in chronic periodontitis patients with or without type 2 diabetes. Inflamm. Res. 60 (2), 143–151. doi:10.1007/s00011-010-0248-7
Kingsmore, S. F. (2006). Multiplexed protein measurement: technologies and applications of protein and antibody arrays. Nat. Rev. Drug Discov. 5 (4), 310–321. doi:10.1038/nrd2006
Krishnan, V., Gunasekaran, E., Prabhakaran, C., Kanagavalli, P., Ananth, V., and Veerapandian, M. (2023). Electropolymerized methylene blue on graphene oxide framework for the direct voltammetric detection of gallic acid. Mater. Chem. Phys. 295, 127071. doi:10.1016/j.matchemphys.2022.127071
Lakshmi Narayanan, M., Prabhu, K., Ponpandian, N., and Viswanathan, C. (2024). Cu encrusted RF sputtered ZnO thin film based electrochemical immunosensor for highly sensitive detection of IL-6 in human blood serum. Microchem. J. 199, 110061. doi:10.1016/j.microc.2024.110061
Li, D., Müller, M. B., Gilje, S., Kaner, R. B., and Wallace, G. G. (2008). Processable aqueous dispersions of graphene nanosheets. Nat. Nanotech. 3, 101–105. doi:10.1038/nnano.2007.451
Li, F., Han, Q., and Xi, F. (2023). The fabrication of a probe-integrated electrochemiluminescence aptasensor based on double-layered nanochannel array with opposite charges for the sensitive determination of C-reactive protein. Molecules 28, 7867. doi:10.3390/molecules28237867
Li, H., Li, X., Meng, Q., Han, J., Zhao, W., Chen, J., et al. (2025). Electric field-induced alignment of Ag/Au nanowires for ultrasensitive in situ detection of Interleukin-6. Biosens. Bioelectron. 271, 117033. doi:10.1016/j.bios.2024.117033
Liu, Z., Huang, Q., Chen, J., Yao, J., Jin, M., Wang, X., et al. (2021). Nanoparticle-assisted sacrificial synthesis of hierarchical porous carbon composite for rapid sample enrichment and ultrasensitive label-free immunosensing of interleukin-6 biomarker. J. Electroanal.Chem. 883, 115068. doi:10.1016/j.jelechem.2021.115068
Lou, Y., He, T., Jiang, F., Shi, J.-J., and Zhu, J.-J. (2014). A competitive electrochemical immunosensor for the detection of human interleukin-6 based on the electrically heated carbon electrode and silver nanoparticles functionalized labels. Talanta 122, 135–139. doi:10.1016/j.talanta.2014.01.016
Lv, N., Qiu, X., Han, Q., Xi, F., Wang, Y., and Chen, J. (2022). Anti-biofouling electrochemical sensor based on the binary nanocomposite of silica nanochannel array and graphene for doxorubicin detection in human serum and urine samples. Molecules 27 (24), 8640. doi:10.3390/molecules27248640
Ma, X., Zhang, Z., Zheng, Y., and Liu, J. (2024). Solid-phase electrochemiluminescence enzyme electrodes based on nanocage arrays for highly sensitive detection of cholesterol. Biosensors 14, 403. doi:10.3390/bios14080403
McCrae, L. E., Ting, W.-T., and Howlader, M. M. R. (2023). Advancing electrochemical biosensors for interleukin-6 detection. Biosens. Bioelectron. X 13, 100288. doi:10.1016/j.biosx.2022.100288
Noh, M. K., Jung, M., Kim, S. H., Lee, S. R., Park, K. H., Kim, D. H., et al. (2013). Assessment of IL-6, IL-8 and TNF-α levels in the gingival tissue of patients with periodontitis. Exp. Ther. Med. 6 (3), 847–851. doi:10.3892/etm.2013.1222
Su, R., Tang, H., and Xi, F. (2022). Sensitive electrochemical detection of p-nitrophenol by pre-activated glassy carbon electrode integrated with silica nanochannel array film. Front. Chem. 10, 954748. doi:10.3389/fchem.2022.954748
Tertis, M., Leva, P. I., Bogdan, D., Suciu, M., Graur, F., and Cristea, C. (2019). Impedimetric aptasensor for the label-free and selective detection of Interleukin-6 for colorectal cancer screening. Biosens. Bioelectron. 137, 123–132. doi:10.1016/j.bios.2019.05.012
Wang, M., Gao, F., Ni, S., Song, Y., Wang, C., Li, Q., et al. (2023). Electrochemical immunosensor based on gold-thionine for detection of subarachnoid hemorrhage biomarker. Front. Bioeng. Biotech. 11, 11153987. doi:10.3389/fbioe.2023.1153987
Wu, J., Wang, L., Wang, H., Gu, X., Zhou, Y., and Xi, F. (2025). Enhanced electrochemiluminescence of luminol at neutral medium using nanochannel-confined Co3O4 nanozyme for highly sensitive detection of tumor biomarker. Microchem. J. 209, 112903. doi:10.1016/j.microc.2025.112903
Yan, L., Xu, S., and Xi, F. (2022a). Disposal immunosensor for sensitive electrochemical detection of prostate-specific antigen Based on amino-rich nanochannels array-modified patterned indium tin oxide electrode. Nanomaterials 12, 3810. doi:10.3390/nano12213810
Yan, L., Zhang, C., and Xi, F. (2022b). Disposable amperometric label-free immunosensor on chitosan–graphene-modified patterned ITO electrodes for prostate specific antigen. Molecules 27, 5895. doi:10.3390/molecules27185895
Yang, L., Zhang, T., Zhou, H., Yan, F., and Liu, Y. (2022). Silica nanochannels boosting Ru(bpy)32+-mediated electrochemical sensor for the detection of guanine in beer and pharmaceutical samples. Front. Nutr. 9, 987442. doi:10.3389/fnut.2022.987442
Zeng, Z., Zhao, Y., Yang, L., Xi, F., and Su, D. (2023). Vertically ordered mesoporous silica film-assisted electrochemical cytosensor for the sensitive detection of HeLa cells. Front. Chem. 11, 1222067. doi:10.3389/fchem.2023.1222067
Zhang, C., Zhou, X., Yan, F., and Lin, J. (2023c). N-doped graphene quantum dots confined within silica nanochannels for enhanced electrochemical detection of doxorubicin. Molecules 28, 6443. doi:10.3390/molecules28186443
Zhang, J., Yang, L., Pei, J., Tian, Y., and Liu, J. (2022). A reagentless electrochemical immunosensor for sensitive detection of carcinoembryonic antigen based on the interface with redox probe-modified electron transfer wires and effectively immobilized antibody. Front. Chem. 10, 939736. doi:10.3389/fchem.2022.939736
Zhang, T., Gong, J., Han, Q., Hu, W., Yan, F., and Liu, J. (2024). Nanogold amplified electrochemiluminescence/electrochemistry in bipolar silica nanochannel array for ultrasensitive detection of SARS-CoV-2 pseudoviruses. Talanta 277, 126319. doi:10.1016/j.talanta.2024.126319
Zhang, T., Yang, L., Yan, F., and Wang, K. (2023b). Vertically-ordered mesoporous silica film based electrochemical aptasensor for highly sensitive detection of alpha-fetoprotein in human serum. Biosensors 13 (6), 628. doi:10.3390/bios13060628
Zhang, W., Du, J., Wang, K., Li, Y., Chen, C., Yang, L., et al. (2023a). Integrated dual-channel electrochemical immunosensor for early diagnosis and monitoring of periodontitis by detecting multiple biomarkers in saliva. Anal. Chim. Acta 1247, 340878. doi:10.1016/j.aca.2023.340878
Zhao, J., Shi, Z., Chen, M., and Xi, F. (2025). Highly active nanozyme based on nitrogen-doped graphene quantum dots and iron ion nanocomposite for selective colorimetric detection of hydroquinone. Talanta 281, 126817. doi:10.1016/j.talanta.2024.126817
Zheng, W., Su, R., Yu, G., Liu, L., and Yan, F. (2022). Highly sensitive electrochemical detection of paraquat in environmental water samples using a vertically ordered mesoporous silica film and a nanocarbon composite. Nanomaterials 12 (20), 3632. doi:10.3390/nano12203632
Zhou, H., Ding, Y., Su, R., Lu, D., Tang, H., and Xi, F. (2022). Silica nanochannel array film supported by ß-cyclodextrin-functionalized graphene modified gold film electrode for sensitive and direct electroanalysis of acetaminophen. Front. Chem. 9, 812086. doi:10.3389/fchem.2021.812086
Zhou, X., Gu, X., Zhang, S., Zou, Y., and Yan, F. (2024b). Magnetic graphene oxide and vertically-ordered mesoporous silica film for universal and sensitive homogeneous electrochemiluminescence aptasensor platform. Microchem. J. 200, 110315. doi:10.1016/j.microc.2024.110315
Zhou, X., Zou, Y., Ru, H., Yan, F., and Liu, J. (2024a). Silica nanochannels as nanoreactors for the confined synthesis of Ag NPs to boost electrochemical stripping chemiluminescence of the luminol-O2 system for the sensitive aptasensor. Anal. Chem. 96, 10264–10273. doi:10.1021/acs.analchem.4c01033
Zhou, Y., Luo, X., Yan, F., and Mou, Y. (2023). Electrostatic nanocage-confined probe for electrochemical detection of CA19-9 in human serum. ACS Omega 8, 48491–48498. doi:10.1021/acsomega.3c08370
Zhu, C., Zhao, Y., and Liu, J. (2025). Sensitive detection of biomarker in gingival crevicular fluid based on enhanced electrochemiluminescence by nanochannel-confined Co3O4 nanocatalyst. Biosensors 15, 63. doi:10.3390/bios15010063
Zhu, X., Xuan, L., Gong, J., Liu, J., Wang, X., Xi, F., et al. (2022). Three-dimensional macroscopic graphene supported vertically-ordered mesoporous silica-nanochannel film for direct and ultrasensitive detection of uric acid in serum. Talanta 238, 123027. doi:10.1016/j.talanta.2021.123027
Keywords: methylene blue, reduced graphene oxide, polydopamine, electrochemical immunosensor, IL-6
Citation: Zhu C, Wang H and Liu J (2025) Highly sensitive electrochemical immunosensor based on methylene blue-reduced graphene oxide nanocomposites as signal probes for IL-6 detection in gingival crevicular fluid samples. Front. Chem. 13:1549927. doi: 10.3389/fchem.2025.1549927
Received: 22 December 2024; Accepted: 03 March 2025;
Published: 02 April 2025.
Edited by:
Maria Rachele Guascito, University of Salento, ItalyReviewed by:
Habibulla Imran, Chonbuk National University, Republic of KoreaCopyright © 2025 Zhu, Wang and Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jiyang Liu, bGl1anlAenN0dS5lZHUuY24=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.