
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Cell. Neurosci., 15 October 2024
Sec. Cellular Neurophysiology
Volume 18 - 2024 | https://doi.org/10.3389/fncel.2024.1475934
Ferroptosis represents an iron− and lipid peroxidation (LPO)-mediated form of regulated cell death (RCD). Recent evidence strongly suggests the involvement of ferroptosis in various neurodegenerative diseases (NDs), particularly Alzheimer’s disease (AD), Parkinson’s disease (PD), Huntington’s disease (HD), multiple sclerosis (MS), and amyotrophic lateral sclerosis (ALS), among others. The treatment of ferroptosis poses both opportunities and challenges in the context of ND. This review provides a comprehensive overview of characteristic features, induction and inhibition of ferroptosis, highlighting the ferroptosis inhibitor and the underlying mechanisms responsible for its occurrence. Moreover, the review explores how these mechanisms contribute to the pathogenesis and progression of major neurodegenerative disorders. Additionally, it presents novel insights into the role of ferroptosis in ND and summarizes recent advancements in the development of therapeutic approaches targeting ferroptosis. These insights and advancements hold potential to guide future strategies aimed at effectively managing these debilitating medical conditions.
Cells serve as the fundamental building blocks of life, embodying paramount significance in every facet thereof, owing to their indispensable roles in proliferation, differentiation, functional attributes, and ultimately cell death. Cells possess the inherent capability to experience two distinct modes of cell death: accidental cell death (ACD) and RCD (Tang et al., 2019; Mishra et al., 2018; Bedoui et al., 2020). ACD represents an uncontrolled and unrestrained biological process, whereas RCD encompasses meticulously orchestrated signaling cascades and well-defined effector mechanisms operating at the molecular level (Tang et al., 2019; Mishra et al., 2018; Bedoui et al., 2020). Recent scientific investigations have elucidated a burgeoning repertoire of groundbreaking non-apoptotic manifestations of RCD, garnering increasing recognition for their involvement in a myriad of human diseases. Among these RCD variants, apoptosis is the most extensively studied one, characterized by a reduction in cellular volume, chromatin condensation, and the emergence of apoptotic bodies (Xie et al., 2016a; Abou-Ghali and Stiban, 2015). Nevertheless, numerous alternative non-apoptotic modalities of RCD are currently under investigation, and among them, ferroptosis has emerged as a prominent research focus, which is an iron-dependent mechanism of cellular demise that executes independently of apoptosis (Bedoui et al., 2020).
Ferroptosis was first discovered as the mechanism by which erastin and RAS-selective lethal 3 (RSL3), synthetic lethal compounds, selectively kill oncogenic rat sarcoma (RAS) mutant cells (Dixon et al., 2012). It is characterized by its smaller mitochondria, reduced condition, increased membrane density, and mitochondrial membrane rupture. The differences between apoptosis and ferroptosis are shown in Table 1. However, the nucleus remains unaffected in ferroptosis. Ferroptosis can occur in various ways and under different conditions, including both physiological and pathological states. The physiological processes affected by ferroptosis can be categorized as follows: 1. Promotion of anti-viral immunity through selenium supplementation: When Cluster of Differentiation 4+ (CD4+) TFH cells are exposed to a selenium-rich diet, the expression of selenoprotein glutathione peroxidase 4 (GPX4) increases (Stockwell, 2022). This protein helps to suppress ferroptosis, leading to an increase in memory B cells and long-lasting viral immunity (Stockwell, 2022). 2. Role of ferroptosis in tumor suppression: A diet rich in polyunsaturated fatty acids (PUFA) promotes the production of polyunsaturated fatty acids-containing phospholipids (PUFA-PLs) (Stockwell, 2022). These compounds facilitate tumor ferroptosis. Additionally, Cluster of Differentiation 8+ (CD8+) T cells contribute to tumor cell ferroptosis by releasing Interferon-γ (IFNγ) and AA (Stockwell, 2022). 3. Ferroptosis associated with aging: As rats and mice age, they experience an increase in ferroptosis markers (Stockwell, 2022). 4. Involvement of ferroptosis in development: Nucleated erythrocytes undergo ferroptosis before enucleation and erythrocyte maturation (Stockwell, 2022). Ferroptosis that occurs under abnormal conditions often leads to diseases such as Sedaghatian-type spondylometaphyseal dysplasia (SSMD) and Systemic lupus erythematosus (SLE) (Stockwell, 2022). Furthermore, there have been numerous studies on ferroptosis in tumors. Inducing iron death can lead to the therapeutic death of tumor cells. For example, a diffuse large B cell lymphoma xenograft was sensitive to treatment with the system xc− inhibitor imidazole ketone erastin (IKE), which activated markers of ferroptosis in the xenografted tumors (Stockwell, 2022; Zhang et al., 2019; Badgley et al., 2020). Knockout of solute carrier family 7 member 11 (SLC7A11) provided significant benefit in a mouse genetic model of pancreatic cancer, without inducing markers of other types of cell death beyond ferroptosis (Stockwell, 2022; Zhang et al., 2019; Badgley et al., 2020). In addition to tumors, ferroptosis is also implicated in a variety of ND. Iron chelators and lipid-soluble antioxidants have been effective in suppressing ferroptosis, highlighting the involvement of iron dyshomeostasis and lipid peroxide accumulation in this process. In conclusion, the suppression of ferroptosis holds potential in preventing the initiation and progression of diseases.
ND is one of the leading causes of death in modern societies (Varela and Garcia-Rendueles, 2022), and has become an important public health burden, due to its increasing incidence and mortality and an associated rise in healthcare costs (GBD 2016 Neurology Collaborators, 2019; Yang et al., 2020). Therefore, it is urgent to seek an exact pathogenesis and treatment. ND is characterized by the gradual decline of motor and cognitive functions, resulting from the degeneration and loss of neurons in the central nervous system (CNS) (Bianchi et al., 2021). AD, PD, HD, MS and ALS are the most common ND (Wang et al., 2023a). Several notable features of ND, such as LPO and iron dyshomeostasis, align with the characteristics of ferroptosis, suggesting a potential involvement of ferroptosis in the progression of these diseases. Aging constitutes the primary risk factor for neurodegenerative ailments and is accompanied by cerebral iron accumulation (Buijs et al., 2017). Likewise, different ND have reported instances of iron accumulation in affected brain regions (Chen et al., 2024; Mohan et al., 2024; Rogers et al., 2019; Masaldan et al., 2019). Ferroptosis inhibitors have demonstrated their protective efficacy in cellular or animal AD, PD, and HD models (Skouta et al., 2014; Ding et al., 2023; Reichert et al., 2020). Hence, it is plausible that ferroptosis assumes a pivotal function in the development of diverse neurodegenerative disorders.
Herein, the main objective of this paper is to offer a comprehensive understanding of ferroptosis and its significance in ND. Additionally, it also aims to present some new insights into ferroptosis in ND.
Ferroptosis is an exclusive form of RCD that is distinguished by both morphological and mechanistic differences when compared to apoptosis and other types of RCD. In terms of morphology, cells undergoing ferroptosis do not exhibit the customary apoptotic characteristics such as chromatin condensation and apoptotic body formation. Instead, they are marked by contracted mitochondria and a reduction in the quantity of mitochondrial cristae. The lethal accumulation of lipid peroxides serves as a pivotal trait of ferroptosis and revolves around a competition between cellular activities that promote ferroptosis and the antioxidant-buffering capabilities offered by ferroptosis defense systems (Punziano et al., 2024). Ferroptosis occurs when the promoting activities of ferroptosis surpass the ability of defense systems to alleviate oxidative stress. It is this mechanism that distinguishes ferroptosis from other forms of RCD, which usually involve the initiation of specific cell death executioner proteins. Prominent examples of these proteins include caspases in apoptosis, mixed lineage kinase domain-like protein (MLKL) in necroptosis, and gasdermin D in pyroptosis (Tonnus et al., 2019; Aachoui et al., 2013; Evavold et al., 2018). Furthermore, cells undergoing ferroptosis exhibit distinctive profiles of oxidized phospholipids (PL), setting them apart from cells undergoing other types of cell death (Tonnus et al., 2019; Aachoui et al., 2013; Evavold et al., 2018). This further underscores the exceptional nature of ferroptosis as a RCD pathway.
The search for physiological markers associated with ferroptosis is essential. Various markers can be utilized, including LPO markers such as Thiobarbituric acid reactive substances (TBARS), C11-BODIPY, Liperfluo fluorescence, Liquid Chromatography-Mass Spectrometry (LC–MS)/MS lipidomics, Anti-melanoma differentiation-associated protein (anti-MDA) adduct antibody staining (Kobayashi et al., 2021; Yamada et al., 2001), and anti-4 Hydroxynonenal (anti-HNE) adduct antibody staining (Zheng et al., 2021). Additionally, markers of mitochondria, like Shrunken, dense mitochondria, and markers of TfR1 can be identified through anti-TfR1 antibody staining (Feng et al., 2020). During ferroptosis, several genes are induced, including ChaC glutathione specific gamma-glutamylcyclotransferase 1 (CHAC1), prostaglandin-endoperoxide synthase 2 (PTGS2), SLC7A11, and acyl-CoA synthetase long-chain family member 4 (ACSL4), while Regulator of G protein signaling 4 (RGS4) is downregulated. The altered expression of these genes can be detected using Realtime fluorescence quantitative PCR (qPCR), providing indicators of ferroptosis (Stockwell, 2022; Dou et al., 2022). Another article reports that over-oxidized stimulates iron death by inhibiting cystine uptake, which makes Peroxiredoxin-3 (PRDX3) peroxide also a specific marker for ferroptosis (Cui et al., 2023).
Ferroptosis is a cellular death mechanism that relies on the iron-induced oxidation of lipids (Stockwell, 2022; Yang et al., 2016; Kim et al., 2023), and the principle of ferroptosis is showed in Figure 1. The central process in ferroptosis execution is the iron-catalyzed peroxidation of PL that contain PUFA (Stockwell, 2022; Yang et al., 2016; Kim et al., 2023).
Under physiological conditions, lipids undergo constant peroxidation through non-enzymatic autoxidation or lipoxygenase (LOX)-mediated LPO, especially during cellular stress. However, the potentially harmful lipid hydroperoxides produced are continually monitored by the selenoenzyme GPX4. This unique enzyme directly reduces lipid hydroperoxides in the acyl chains of PL to lipid alcohols while oxidizing reduced glutathione (GSH) to form glutathione disulfide (Kim et al., 2023; Anthonymuthu et al., 2021). However, when GPX4 is deactivated or GSH levels are depleted in cells, phospholipid peroxides (PLOOH) accumulate, leading to the production of lipid radicals, such as lipid peroxyl radicals ((P)LOO∙) and alkyl radicals ((P)L∙), through iron-dependent free radical chain reactions. Uncontrolled LPO and the generation of reactive aldehydes due to LPO ultimately lead to cell membrane rupture and cell death through ferroptosis (Conrad and Pratt, 2019; Chu et al., 2019). Although lipid radicals are the primary contributors to ferroptosis, their origins are not yet well understood. Mitochondria are believed to be the main generators of reactive oxygen species (ROS) in a cell due to incomplete reduction of molecular oxygen (Shah et al., 2018; Zou et al., 2020; Yan et al., 2021). Therefore, mitochondrial ROS is essential for ferroptosis. Notably, blocking ferroptosis induced by GSH depletion can be achieved by inhibiting the electron transport chain, which releases electrons to generate superoxide anion (O2−∙) (Gan, 2021; Gao et al., 2019). Superoxide is primarily localized in the mitochondrial matrix and generated in the intermembrane space through complex III. It can then be transported from mitochondria to the cytoplasm through voltage-dependent anion channels (Schaar et al., 2015; Dan Dunn et al., 2015). However, the precise mechanisms by which mitochondrial superoxide is released into the cytoplasm or at a membrane to induce LPO are still unknown. Besides mitochondrial ROS, the rate of ferroptosis is also influenced by glutaminolysis and the tricarboxylic cycle (Gao et al., 2024). Disruption of oxidative phosphorylation by deleting cytochrome c oxidase assembly factor 10 leads to lysosomal and mitochondrial defects, resulting in LPO and ferroptosis of cardiac cells (Ahola et al., 2022). There are numerous pathways through which ferroptosis can be stimulated, involving diverse mechanisms.
Four main mechanisms have been identified for the induction of ferroptosis, including hindering system xc−, inhibiting, degrading, or inactivating GPX4, depleting reduced Coenzyme Q10 (CoQ10), and triggering LPO via peroxides, iron, or PUFA overload (Stockwell, 2022). There is ample evidence supporting that inhibiting system xc− is a potent mechanism for triggering ferroptosis. Various small molecule inhibitors of this antiporter, such as erastin, sulfasalazine, and glutamate, effectively block system xc− and thereby induce ferroptosis. Moreover, cystine depletion eliminates the extracellular substrate for system xc− and also promotes ferroptosis. Similarly, the genetic inactivation or small-molecule-mediated inhibition or degradation of GPX4 leads to ferroptosis in multiple cell types. In cases where GPX4 is absent, the inhibition of CoQ10 biosynthesis through the mevalonate pathway or the inactivation of CoQ10 reductases, such as Ferroptosis suppressor protein 1 (AIFM2/FSP1) or Dihydroorotate Dehydrogenase (DHODH), initiates ferroptosis. Finally, an excess of iron, PUFAs, or peroxides, such as Tert-butyl hydroperoxide (tBOOH) or FINO2, induces ferroptosis (Shah et al., 2018). All four of these mechanisms exhibit significant specificity in inducing ferroptosis compared to other forms of cell death. This specificity is evident as the lethal effects of these mechanisms are mostly or entirely neutralized by ferroptosis-specific inhibitors, and markers of alternative cell death mechanisms are not activated.
Biological processes exert control over ferroptosis by modulating the molecules involved in its promotion or surveillance, as well as redox and iron homeostasis, and cell metabolism. It is widely acknowledged that numerous signaling pathways dictate a cell’s susceptibility to ferroptosis in specific biological circumstances, supported by an ever-expanding body of evidence. According to the principle of ferroptosis, the inhibitors can be classified into three types: first, drugs that prevent iron accumulation at the source of ferroptosis; second, drugs that affect LPO by directly inhibiting ROS accumulation or indirectly inhibiting factors that influence the body, like enhancing the GPX4/GSH axis; and third, drugs that directly eliminate lipid free radicals to inhibit ferroptosis (Table 2) (Liang et al., 2023).
The main factor affecting iron accumulation is IRP, which is an RNA-binding protein that governs the translation of a cluster of mRNA molecules involved in iron homeostasis by binding to Inositol-requiring enzyme (IRE). Meanwhile, iron regulatory protein (IRP2) acts as the primary detector of labile iron in neurons. In situations where the cellular iron content is insufficient, IRP attaches to IRE located in the 5′ untranslated regions (5′ UTR) of mRNA molecules responsible for iron-responsive proteins, such as Ferritin L chain (FPN), β-amyloid precursor protein (APP), and α-synuclein, consequently inhibiting the translation of these iron-responsive proteins to reduce iron export and the storage of free iron. Conversely, when intracellular iron levels rise, the IRP and IRE dissociate, leading to an opposing effect characterized by reduced iron absorption and increased free iron storage and export. IRP2 plays an indispensable role in regulating iron within the brain, and its misregulation is closely associated with iron accumulation in ND. In the study of age-related auditory cortical neurodegeneration, it has been discovered that the upregulation of IRP2 augments intracellular iron levels, ultimately leading to ferroptosis (Yao et al., 2021).
Moreover, it has been observed that ferritinophagy promotes ferroptosis. The primary means of intracellular iron storage hinges predominantly on ferritin, which encompasses ferritin light chain polypeptide 1 (FTL1) and ferritin heavy chain polypeptide 1 (FTH1) (Nishizawa et al., 2020). Within the cell, ferritin serves as the premier protein complex for storing iron. Furthermore, iron is released from ferritin primarily through selective autophagy facilitated by nuclear receptor coactivator 4 (NCOA4), a process commonly recognized as ferritinophagy. The complex of ferritin combined with NCOA4 is transported to lysosomes for degradation, subsequently releasing iron for utilization in cellular physiological activities. It has been demonstrated that the intricacy of ferroptosis is affected by NCOA4-mediated ferritinophagy, with studies revealing that the levels of NCOA4 influence the sensitivity to ferroptosis. Suppression of NCOA4 hampers ferritin degradation and curtails ferroptosis, whereas its overexpression heightens the labile iron pool (LIP) (Fang et al., 2021), thereby promoting the accumulation of ROS and the initiation of ferroptosis.
The main ROS related factor is GPX4. Evidence indicates that the activity and stability of GPX4, a vital regulator of ferroptosis, undergo regulation. For instance, persistent oxidative stress and deficiency in glutathione can impair the GSH-dependent reduction of GPX4’s active site selenocysteine, leading to irreversible inactivation through the formation of a redox-dead dehydroalanine, also known as ß-cleavage (Orian et al., 2015). However, the formation of a selenylamide between the selenenic acid and an adjacent amino acid may safeguard the enzyme against irreversible inactivation. Further investigation is required to ascertain if any of these mechanisms are involved in pathological conditions such as ischemia/reperfusion injuries (IRI), although studies have demonstrated that intestinal IRI is associated with reduced GPX4 levels (Li et al., 2019). Additionally, certain substances that induce ferroptosis, including RSL3, can deplete GPX4 through covalent inhibition of its active site selenocysteine, impairment of mevalonate metabolism, or induction of general iron-dependent oxidative stress (Shimada et al., 2016; Gaschler et al., 2018; Chen et al., 2020).
Another significant suppressor of ferroptosis is FSP1, which is regulated by transcription factors Nuclear factor erythroid 2-related factor 2 (NRF2) (Chorley et al., 2012), CCAAT/enhancer-binding protein (CEBP) (Nguyen et al., 2020), and Peroxisome proliferator-activated receptor alpha (PPARα) (Venkatesh et al., 2020). Apart from transcriptional regulation, little is known about the regulation of FSP1’s oxidoreductase activity and how its subcellular localization influences its role in different physiological and pathophysiological processes (Nguyen et al., 2020; Bersuker et al., 2019; Wu et al., 2002; Doll et al., 2019). However, the ability of FSP1 to interact with both reducing and oxidizing substrates (such as Nicotinamide adenine dinucleotide (NADH), nicotinamide adenine dinucleotide phosphate (NADPH), CoQ10, and α-tocopherol) suggests that its regulation is intricate. In particular, it has recently been found that membrane-bound O-acyltransferase domain-containing 1 and 2 (MBOAT1/2) attenuates ferroptosis by bolstering the levels of MUFA present on the cellular membrane (Liang et al., 2023). More importantly, the potential of MBOAT2 to hinder ferroptosis without reliance on GPX4 and FSP1 through a mechanism involving PL remodeling-mediated surveillance has been identified (Liang et al., 2023). Nevertheless, the existing literature on this subject is limited, and additional research is warranted to gain a comprehensive understanding.
ND are a heterogeneous group of complex diseases characterized by neuronal loss and progressive degeneration of different areas of the nervous system. AD, PD, HD, MS, and ALS represent the prevailing neurodegenerative disorders in current medical research. The existence of LPO and iron dyshomeostasis in neurodegenerative disorders exhibits striking parallels to the distinctive traits observed in ferroptosis (Wang et al., 2023a). This suggests that ferroptosis might exert a pivotal role in the advancement of above-mentioned diseases. As might be expected, more recent studies have also looked at the effects of ferroptosis in ND.
AD is a chronic, progressive, and irreversible neurodegenerative condition characterized by memory impairment, language disturbances, severe behavioral abnormalities, and deficits in learning abilities (Bao et al., 2021). Ferroptosis, appears to play a significant role in AD by affecting various pathways related to iron metabolism, glutamate excitotoxicity, and the accumulation of ROS in lipids. In the context of AD, disruptions in the balance of iron within the brain have been observed, and higher levels of brain iron have been found to be positively correlated with disease progression and cognitive decline. Interactions among iron, beta-amyloid (Aβ) protein, and tau protein can lead to the production of ROS, which may contribute to the activation of ferroptotic cell death pathways. Moreover, the overexpression of Fpn, responsible for storing and regulating iron elements, playing a crucial role in the storage, release, and regulation of iron within cells (Jakaria et al., 2021), has been shown to partially improve memory impairment and reduce ferroptosis in mouse models of AD, providing further evidence for the crucial role of Fpn in the disease (Jakaria et al., 2021; Wang F. et al., 2022) (see Figure 2).
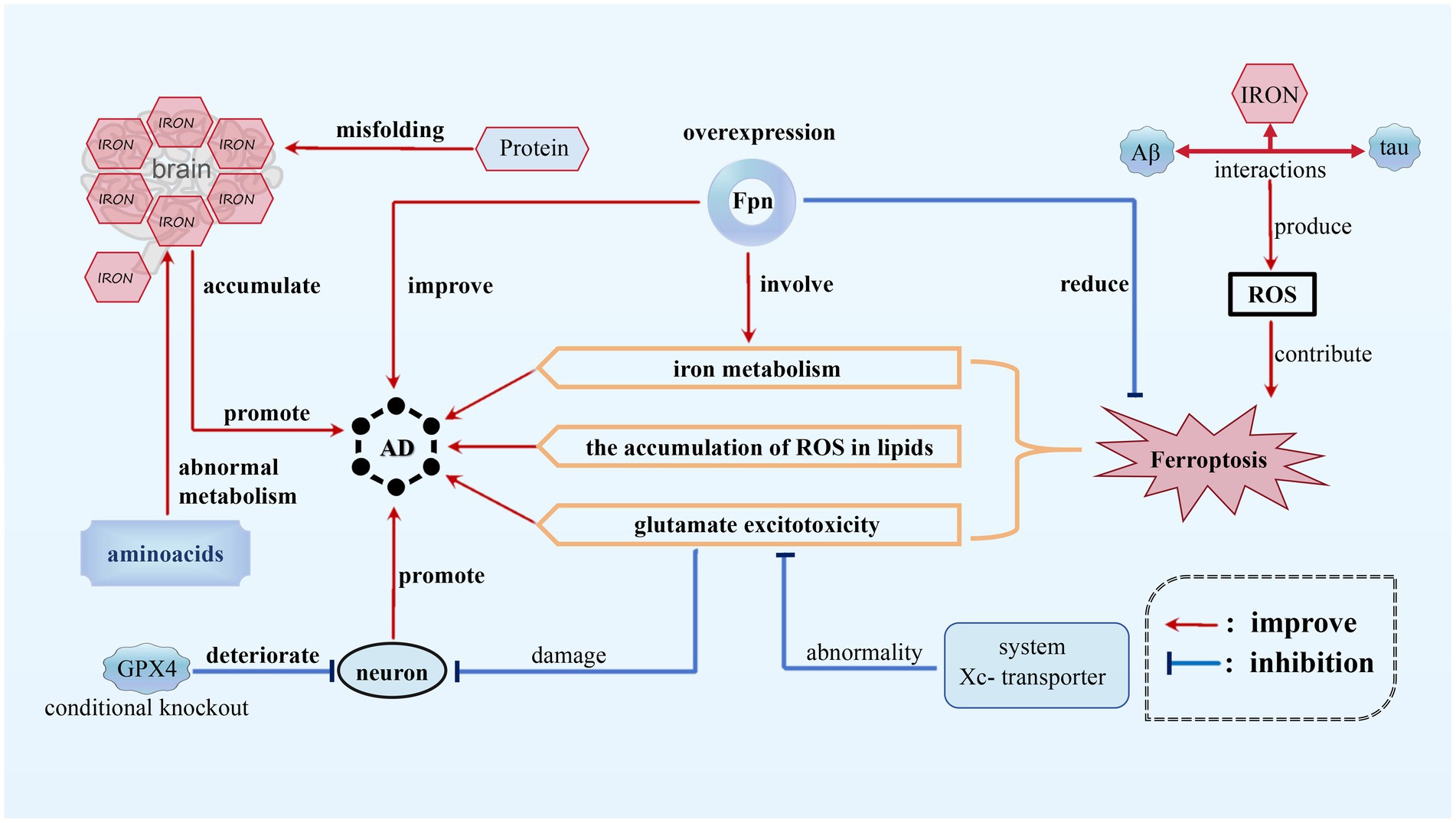
Figure 2. Ferroptosis in AD. In AD, disruptions in the balance of iron within the brain have been observed and higher iron level promotes the progression of AD. Interactions among iron, beta-amyloid (Aβ) protein, and tau protein can provoke ROS, contributing to ferroptosis of neurons. In addition, the abnormality of system Xc-transporter induces glutamate excitotoxicity, which also promotes AD progression. However, FPN overexpression can inhibit ferroptosis, thus alleviating AD.
Furthermore, neuronal damage in AD can be attributed to glutamate excitotoxicity, which is caused by abnormalities in the function of the cystine/glutamate antiporter system (system Xc−) transporter. Increased oxidative stress, as indicated by elevated levels of oxidative damage, also exists in AD patients. Protein misfolding and abnormal metabolism of amino acids may further exacerbate iron accumulation, implicating ferroptosis as a potential mechanism in the progression of the disease (Jakaria et al., 2021). As one of the most important anti-oxidases, the conditional knockout of GPX4 in mice caused degeneration of hippocampal neurons, suggesting a possible link between ferroptosis and neurodegeneration in AD models (Park et al., 2021).
Although the precise role of ferroptosis in AD is still being investigated, the growing body of evidence strongly suggests its involvement in regulating the pathology of the disease, garnering significant attention from researchers in the field. Various strategies can potentially mitigate ferroptosis in AD, such as antioxidant therapy using compounds like vitamin E or Lip-1 to reduce neurodegeneration, iron chelation therapy to control iron levels and limit the generation of ROS, modulation of glutamate signaling pathways to address excitotoxicity, regulation of lipid metabolism to prevent LPO, and activation of GPX4 to safeguard cells against LPO and ferroptosis (Park et al., 2021). These approaches collectively offer promising avenues for intervening in the progression of AD and attenuating the neurodegenerative processes.
PD is the second most prevalent chronic neurodegenerative disorder (Cannon and Greenamyre, 2013; Tufail and Hassan, 2020; Yang et al., 2017). It is distinguished by the formation of Lewy bodies and loss of dopaminergic neurons in the substantia nigra (SN) (Kosaka, 2014; Dickson, 2018; Han et al., 2023). The involvement of ferroptosisin PD has gained widespread recognition. PD patients exhibit anomalous iron buildup in the SN, accompanied by decreased GSH levels and increased lipid peroxides, which align with the biochemical characteristics of ferroptosis (Dong-Chen et al., 2023; Zeng et al., 2024; Yang et al., 2023). This suggests that ferroptosis contributes to the neurotoxicity observed in PD. Disruptions in iron homeostasis pathways are also implicated in PD. Recent data in the 6-Hydroxydopamine (6-OHDA) model of PD demonstrate that FTH1 links ferritinophagy and ferroptosis, offering a new perspective and potential target for pharmacological intervention (Tian et al., 2020). Alterations in iron regulatory proteins result in imbalances in iron storage and importation. Changes in ferritin levels, divalentmetal-ion transporter-1 (DMT1) expression, ceruloplasmin function, and ferroportin1 expression contribute to elevated cellular iron levels, potentially exacerbating the effects of ferroptosis on dopaminergic neurons.
Targeting ferroptosis pathways has been identified as a promising therapeutic strategy for PD. Inhibiting ferroptosis using compounds like Ferrostatin-1 (Fer-1) has demonstrated the ability to safeguard dopaminergic neurons against toxic substances and prevent neurodegeneration (Xia et al., 2024; Sun et al., 2020). Disparities in the expression of ferroptosis-related genes between normal individuals and PD patients, especially in the SN region, underscore the potential for developing novel treatments that regulate ferroptosis to address the mechanisms of dopaminergic neuron degeneration in PD (see Figure 3).

Figure 3. Ferroptosis in PD. The main pathological features of PD are the significant increase in Lewy bodies within the gray matter and the reduction in dopaminergic neurons. The destruction of dopaminergic neurons can be caused by neurotoxins produced by ferroptosis. This process disrupts iron homeostasis pathways and alters the function of ceruloplasmin. Furthermore, it increases the expression of ferroportin1 and DMT1, as well as ferritin levels. These changes result in abnormal iron accumulation in the substantia nigra (SN), ultimately leading to ferroptosis and creating a vicious circle. However, the compound Fer-1 has the ability to protect dopaminergic neurons and inhibit ferroptosis, thereby reducing the severity of PD.
To potentially diminish the impact of ferroptosis in PD, a multifaceted approach can be considered. Strategies such as iron chelation therapy with agents like deferoxamine, the utilization of ferroptosis inhibitors like Fer-1, regulation of iron homeostasis through targeting iron regulatory proteins, bolstering antioxidant defenses to counteract oxidative stress, and gene therapy to modulate the expression of ferroptosis-related genes could collectively offer potential avenues to alleviate the impact of ferroptosis on dopaminergic neurons in PD. These measures aim to reduce abnormal iron accumulation, mitigate LPO, and enhance cellular resilience against ferroptosis. Furthermore, FTH1 links ferritinophagy and ferroptosis in the 6-OHDA model of PD (Tian et al., 2020; Li et al., 2021), which provides a new perspective for a pharmacological target in PD.
HD is a hereditary, lethal neurodegenerative disorder caused by abnormal Coronary angiography (CAG) repeats in the gene responsible for encoding the protein Huntingtin, which expresses ubiquitin (Tabrizi et al., 2020). The pathophysiology of HD involves the accumulation of toxic iron in neurons and an increase in oxidative stress, potentially contributing to the neurodegenerative process. Ferroptosis, which is a regulated form of cell death characterized by iron-dependent LPO, appears to play a significant role in the progression of HD. Experimental studies utilizing HD mouse models have demonstrated that inhibition of ferroptosis can prevent degeneration of spinal motor neurons and enhance motor function, thereby establishing a direct connection between the occurrence of ferroptosis and the advancement of HD (Chen et al., 2015). Moreover, the disruption of mitochondrial fission and fusion balance in HD leads to elevated levels of ROS and oxidative stress (Mi et al., 2019). The presence of mutant huntingtin protein (mHTT) further intensifies oxidative stress, thereby disrupting the delicate equilibrium of cellular stability (Wyttenbach et al., 2002). Subsequently, this oxidative environment fosters LPO while reducing glutathione levels, exacerbating the disruption of cellular homeostasis and potentially triggering the occurrence of ferroptosis. Empirical evidence indicates that individuals afflicted with HD demonstrate increased levels of LPO and diminished plasma GSH levels, thereby emphasizing the intricate association between oxidative stress, ferroptosis, and the pathophysiological mechanism of HD (Klepac et al., 2007; Yuan et al., 2017) (see Figure 4).
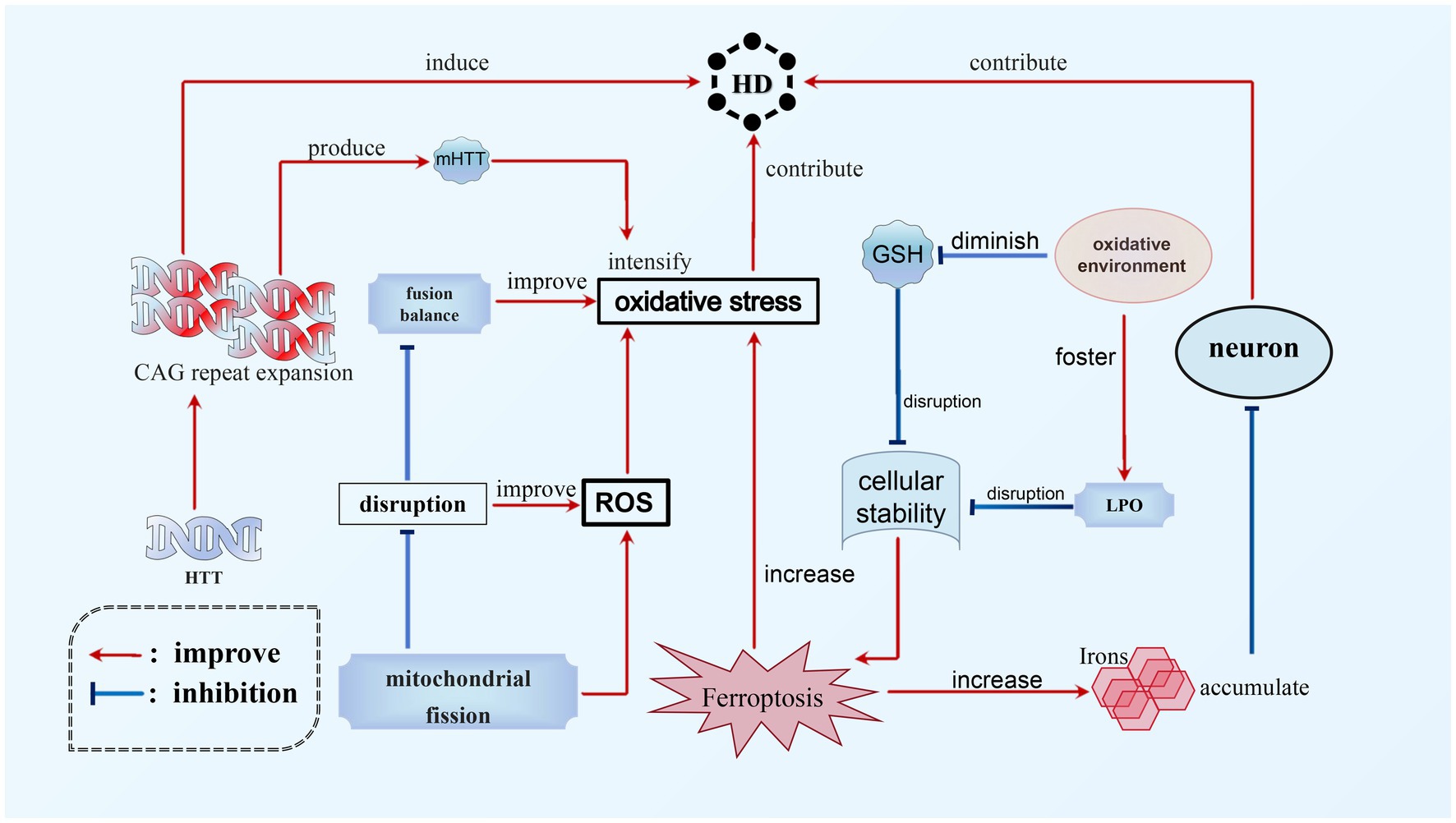
Figure 4. Ferroptosis in HD. The main cause of HD is the expansion of CAG repeats in HTT. This expansion not only directly leads to the occurrence of HD, but also contributes to the progression of the disease by worsening mHTT. Additionally, ferroptosis is another significant factor in the development of HD. There is a complex relationship between oxidative stress, ferroptosis, and HD. The accumulation of toxic iron during ferroptosis can promote the neurodegenerative process and worsen HD. Additionally, HD can increase levels of ROS, which, combined with mHTT, can exacerbate oxidative stress and further aggravate ferroptosis. This creates a vicious circle between these interconnected processes.
Multiple strategies present potential for mitigating ferroptosis in HD. These approaches encompass iron chelation for reducing iron levels, antioxidant therapy to counteract oxidative stress, activation of the Nfr2 pathway to fortify antioxidant defenses, modulation of mitochondrial function to diminish ROS production, and the development of medications that specifically target ferroptosis pathways, such as GPX4 inhibition. A combination of these strategies may hold the key to inhibiting ferroptosis in HD, offering promising avenues for managing the neurodegenerative processes associated with the disease and potentially decelerating its progression.
MS is an autoimmune inflammatory disorder that primarily affects the white matter of the CNS. The exact cause of the disease is still unknown (Reich et al., 2018), but it is characterized by neurodegeneration, inflammatory demyelination, primary demyelinating lesions, varying levels of axonal loss, and the proliferation of astrocytes and microglia (Reich et al., 2018; Kuhlmann et al., 2017). Through investigation, it has been discovered that the genetic composition responsible for a crucial protein involved in ferroptosis, known as acyl-CoA synthetase ACSL4, undergoes significant modifications in MS patients. These modifications correspond with ferroptosis, as observed in existing genomic databases of MS patients. In animal models of MS, such as the experimental autoimmune encephalomyelitis (EAE) mouse model, the accumulation of ROS and mitochondrial shrinkage have been observed. Magnetic resonance imaging (MRI) and histological analysis have also detected increased iron levels in both the white and gray matter of MS patients’ brains, with iron accumulation found in the white matter and macrophages of the basal ganglia (Dusek et al., 2016). These phenomena align with the characteristics of ferroptosis, indicating the presence of biochemical alterations related to ferroptosis in MS. (Luoqian et al., 2022) Furthermore, evidence from experiments suggests that neurons undergoing ferroptosis can stimulate T cell activation through the T cell receptor (TCR) signaling pathway in the EAE mouse model, worsening the progression of MS. Treatment with the iron-chelating agent Downstream Facing Port (DFP) in the EAE model has shown promising results, reducing Transferrin receptor protein 1 (TfR1) content, reversing iron overload, and inhibiting demyelination. By stabilizing inflammatory cells and suppressing the inflammatory response, DFP effectively inhibits ferroptosis in MS, making it a potential therapeutic option (Rayatpour et al., 2022) (see Figure 5).
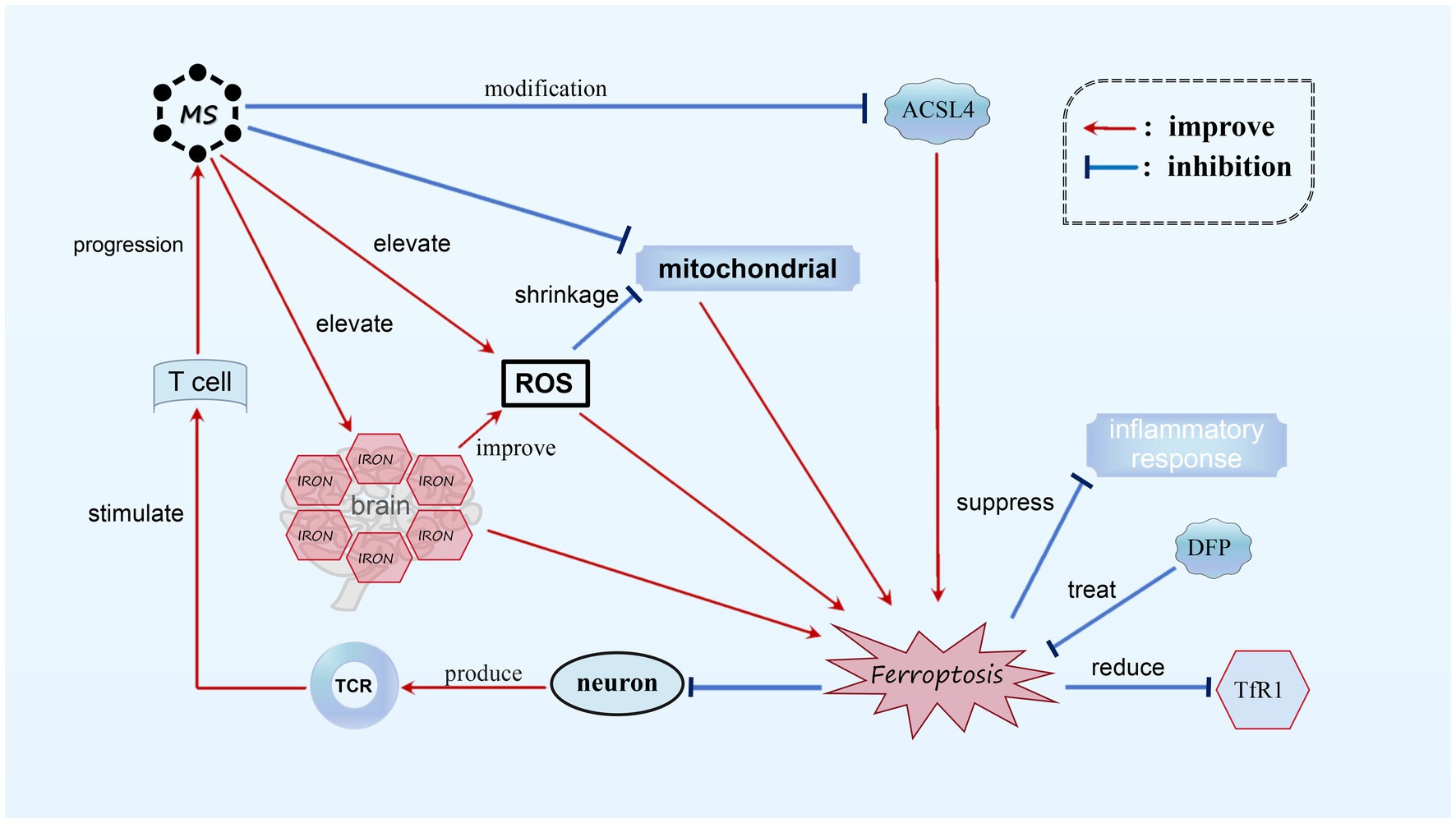
Figure 5. Ferroptosis in MS. In MS, we observed noticeable alterations in ACSL4, mitochondrial atrophy, and the accumulation of a substantial amount of ROS. Additionally, there is an abnormal increase in iron content within the brains of MS patients. These biochemical changes play a role in the occurrence of ferroptosis. Neurons undergoing ferroptosis can activate T cells through TCR stimulation, resulting in further deterioration of MS. It has been found that DFP is capable of reducing TfR1 content and suppressing the inflammatory response, thereby effectively treating MS by diminishing ferroptosis.
Although the exact cause of MS remains unclear, it is apparent that iron death plays a role in MS, with ferroptosis potentially contributing to its development. More research is needed to understand the genetic and chemical mechanisms involved in MS. Analyzing experimental data has shown a potential relationship between ferroptosis and MS, with both factors influencing each other. In future studies, exploring the inhibition of ferroptosis could slow down the progression of MS, while reducing ferroptosis-related characteristics in affected areas could further validate the impact of ferroptosis on MS.
ALS, also referred to as motor neuron disease (MND), is a progressive neurodegenerative disorder that impacts both upper and lower motor neurons, resulting in muscle weakness and ultimately respiratory failure and mortality within a few years of symptom onset (Wang T. et al., 2022). Anomalous accumulation of iron has been observed in the spinal cord and cerebral regions of individuals with ALS and animal models (Devos et al., 2020). This dysregulation of iron is associated with heightened LPO and oxidative stress, contributing to neuronal damage. Research has demonstrated increased levels of markers of ferroptosis, such as 4-HNE, in individuals with ALS. The suppression of crucial pathways involved in safeguarding cells against ferroptosis, such as the GSH/GPX4 axis, has been associated with degeneration of motor neurons and progression of the disease in ALS. Moreover, the FSP1-CoQH2 system and the GTP cyclohydrolase I-BH4 (GCH1-BH4) system have been identified as potential targets in ALS (Wang et al., 2023a,b; Wang T. et al., 2022). Modulating these systems could potentially offer therapeutic benefits by mitigating ferroptosis and oxidative stress in individuals with ALS.
Few studies have been conducted on ALS and ferroptosis, and the precise relationship between them remains unclear. Further exploration is necessary to understand this connection in more detail.
In this review, we present a concise overview of the current knowledge regarding the characterization, induction, and inhibition of ferroptosis (specifically focusing on iron death inhibitors), as well as its role in regulating the occurrence and progression of multiple neurodegenerative diseases, namely AD, PD, HD, MS and ALS. The current study indicates that ferroptosis plays a pivotal role in neurodegenerative diseases and has significant potential as a target for therapeutic interventions. However, the exploration of ferroptosis still faces formidable challenges. Firstly, the investigation of ferroptosis in cognitive dysfunction-related diseases is still in its nascent stage, and its underlying molecular mechanisms remain elusive. Hence, the identification of specific markers of ferroptosis is crucial to comprehensively and extensively study its intricate process. Secondly, the majority of the available data is derived from experimental studies, necessitating the inclusion of more clinical approaches. For instance, experimental employment of iron chelators and antioxidants has proven an effective means to disrupt the ferroptotic process in neurodegenerative diseases. Nonetheless, their efficacy is reduced when tested in human subjects, highlighting the need to explore signaling molecules implicated in alternative pathways triggering iron accumulation. Furthermore, ferroptosis serves not only as a detrimental process but also as a crucial physiological defense mechanism. By inducing cell death in cancer cells, ferroptosis effectively hampers tumour progression and metastasis (Stockwell, 2022; Jiang et al., 2021; Tang D. et al., 2021). In other words, ferroptosis inhibitors used to treat neurodegenerative diseases may facilitate the growth of tumours, such as butylated hydroxytoluene (Faraji et al., 2024). Consequently, prior to endorsing the clinical utilization of ferroptosis inhibitors in treating neurodegenerative diseases or other ailments, more comprehensive mechanistic investigations regarding the potential adverse effects of this compound are imperative. Additionally, the relationship between ferroptosis and other forms of cell death mechanisms remains inadequately understood, and there is a dearth of research on the combination of drugs. Therefore, future efforts should focus on elucidating the intricate associations and mechanisms linking ferroptosis to other well-known cell death pathways, as well as conducting experiments involving the concurrent administration of various agents. These endeavors hold the potential to illuminate innovative therapeutic interventions and strategies.
YF: Conceptualization, Funding acquisition, Writing – original draft, Writing – review & editing. YD: Funding acquisition, Visualization, Writing – review & editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Dr. Yang has our heartfelt gratitude for helping us. She has also been our tireless advocate as we worked through various setbacks on the way to completing our research.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Aachoui, Y., Leaf, I. A., Hagar, J. A., Fontana, M. F., Campos, C. G., Zak, D. E., et al. (2013). Caspase-11 protects against bacteria that escape the vacuole. Science 339, 975–978. doi: 10.1126/science.1230751
Abou-Ghali, M., and Stiban, J. (2015). Regulation of ceramide channel formation and disassembly: insights on the initiation of apoptosis. Saudi J. Biol. Sci. 22, 760–772. doi: 10.1016/j.sjbs.2015.03.005
Ahola, S., Rivera Mejías, P., Hermans, S., Chandragiri, S., Giavalisco, P., Nolte, H., et al. (2022). OMA1-mediated integrated stress response protects against ferroptosis in mitochondrial cardiomyopathy. Cell Metab. 34, 1875–1891.e7. doi: 10.1016/j.cmet.2022.08.017
Anthonymuthu, T. S., Tyurina, Y. Y., Sun, W. Y., Mikulska-Ruminska, K., Shrivastava, I. H., Tyurin, V. A., et al. (2021). Resolving the paradox of ferroptotic cell death: Ferrostatin-1 binds to 15LOX/PEBP1 complex, suppresses generation of peroxidized ETE-PE, and protects against ferroptosis. Redox Biol. 38:101744. doi: 10.1016/j.redox.2020.101744
Badgley, M. A., Kremer, D. M., Maurer, H. C., Del Giorno, K. E., Lee, H. J., Purohit, V., et al. (2020). Cysteine depletion induces pancreatic tumor ferroptosis in mice. Science 368, 85–89. doi: 10.1126/science.aaw9872
Bao, W.-D., Pei, P., Zhou Xiao-Ting, H., Fan, X. W., Kai, C., Jing, W., et al. (2021). Loss of ferroportin induces memory impairment by promoting ferroptosis in Alzheimer's disease. Cell Death Differ. 28, 1548–1562. doi: 10.1038/s41418-020-00685-9
Bedoui, S., Herold, M. J., and Strasser, A. (2020). Emerging connectivity of programmed cell death pathways and its physiological implications. Nat. Rev. Mol. Cell Biol. 21, 678–695. doi: 10.1038/s41580-020-0270-8
Bersuker, K., Hendricks, J. M., Li, Z., Magtanong, L., Ford, B., Tang, P. H., et al. (2019). The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature 575, 688–692. doi: 10.1038/s41586-019-1705-2
Bianchi, V. E., Herrera, P. F., and Laura, R. (2021). Effect of nutrition on neurodegenerative diseases. A systematic review. Nutr. Neurosci. 24, 810–834. doi: 10.1080/1028415X.2019.1681088
Buijs, M., Doan, N. T., van Rooden, S., Versluis, M. J., van Lew, B., Milles, J., et al. (2017). In vivo assessment of iron content of the cerebral cortex in healthy aging using 7-tesla T2*-weighted phase imaging. Neurobiol. Aging 53, 20–26. doi: 10.1016/j.neurobiolaging.2016.09.005
Cannon, J. R., and Greenamyre, J. T. (2013). Gene-environment interactions in Parkinson's disease: specific evidence in humans and mammalian models. Neurobiol. Dis. 57, 38–46. doi: 10.1016/j.nbd.2012.06.025
Cao, Y., Li, Y., He, C., Yan, F., Li, J. R., Xu, H. Z., et al. (2021). Selective Ferroptosis inhibitor Liproxstatin-1 attenuates neurological deficits and Neuroinflammation after subarachnoid hemorrhage. Neurosci. Bull. 37, 535–549. doi: 10.1007/s12264-020-00620-5
Chen, G. Q., Benthani, F. A., Wu, J., Liang, D., Bian, Z. X., and Jiang, X. (2020). Artemisinin compounds sensitize cancer cells to ferroptosis by regulating iron homeostasis. Cell Death Differ. 27, 242–254. doi: 10.1038/s41418-019-0352-3
Chen, L., Hambright, W. S., Na, R., and Ran, Q. (2015). Ablation of the Ferroptosis inhibitor glutathione peroxidase 4 in neurons results in rapid motor neuron degeneration and paralysis. J. Biol. Chem. 290, 28097–28106. doi: 10.1074/jbc.M115.680090
Chen, B., Li, X., Liu, J., Li, Y., Dai, W., Chen, Y., et al. (2021a). Ferroptosis-inhibitory effect and possible mechanisms of Ellagitannin Geraniin. ChemistryOpen. 10, 737–739. doi: 10.1002/open.202000255
Chen, B., Li, X., Ouyang, X., Liu, J., Liu, Y., and Chen, D. (2021b). Comparison of Ferroptosis-inhibitory mechanisms between Ferrostatin-1 and dietary stilbenes (Piceatannol and Astringin). Molecules 26, 975–978. doi: 10.3390/molecules26041092
Chen, Y., Wu, Z., Li, S., Chen, Q., Wang, L., Qi, X., et al. (2024). Mapping the research of Ferroptosis in Parkinson's disease from 2013 to 2023: a Scientometric review. Drug Des. Devel. Ther. 18, 1053–1081. doi: 10.2147/DDDT.S458026
Chen, Y., Yi, X., Huo, B., He, Y., Guo, X., Zhang, Z., et al. (2022). BRD4770 functions as a novel ferroptosis inhibitor to protect against aortic dissection. Pharmacol. Res. 177:106122. doi: 10.1016/j.phrs.2022.106122
Chen, H., Zhu, J., Le, Y., Pan, J., Liu, Y., Liu, Z., et al. (2022). Salidroside inhibits doxorubicin-induced cardiomyopathy by modulating a ferroptosis-dependent pathway. Phytomedicine 99:153964. doi: 10.1016/j.phymed.2022.153964
Cheng, J., Xu, T., Xun, C., Guo, H., Cao, R., Gao, S., et al. (2021). Carnosic acid protects against ferroptosis in PC12 cells exposed to erastin through activation of Nrf2 pathway. Life Sci. 266:118905. doi: 10.1016/j.lfs.2020.118905
Chorley, B. N., Campbell, M. R., Wang, X., Karaca, M., Sambandan, D., Bangura, F., et al. (2012). Identification of novel NRF2-regulated genes by ChIP-Seq: influence on retinoid X receptor alpha. Nucleic Acids Res. 40, 7416–7429. doi: 10.1093/nar/gks409
Chu, B., Kon, N., Chen, D., Li, T., Liu, T., Jiang, L., et al. (2019). ALOX12 is required for p53-mediated tumour suppression through a distinct ferroptosis pathway. Nat. Cell Biol. 21, 579–591. doi: 10.1038/s41556-019-0305-6
Conrad, M., and Pratt, D. A. (2019). The chemical basis of ferroptosis. Nat. Chem. Biol. 15, 1137–1147. doi: 10.1038/s41589-019-0408-1
Cui, S., Ghai, A., Deng, Y., Li, S., Zhang, R., Egbulefu, C., et al. (2023). Identification of hyperoxidized PRDX3 as a ferroptosis marker reveals ferroptotic damage in chronic liver diseases. Mol. Cell 83, 3931–3939.e5. doi: 10.1016/j.molcel.2023.09.025
Dan Dunn, J., Alvarez, L. A., Zhang, X., and Soldati, T. (2015). Reactive oxygen species and mitochondria: a nexus of cellular homeostasis. Redox Biol. 6, 472–485. doi: 10.1016/j.redox.2015.09.005
Devos, D., Cabantchik, Z. I., Moreau, C., Danel, V., Mahoney-Sanchez, L., Bouchaoui, H., et al. (2020). Conservative iron chelation for neurodegenerative diseases such as Parkinson's disease and amyotrophic lateral sclerosis. J. Neural Transm. (Vienna) 127, 189–203. doi: 10.1007/s00702-019-02138-1
Dickson, D. W. (2018). Neuropathology of Parkinson disease. Parkinsonism Relat. Disord. 46, S30–S33. doi: 10.1016/j.parkreldis.2017.07.033
Ding, X. S., Gao, L., Han, Z., Eleuteri, S., Shi, W., Shen, Y., et al. (2023). Ferroptosis in Parkinson's disease: molecular mechanisms and therapeutic potential. Ageing Res. Rev. 91:102077. doi: 10.1016/j.arr.2023.102077
Dixon, S. J., Lemberg, K. M., Lamprecht, M. R., Skouta, R., Zaitsev, E. M., Gleason, C. E., et al. (2012). Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell 149, 1060–1072. doi: 10.1016/j.cell.2012.03.042
Doll, S., Freitas, F. P., Shah, R., Aldrovandi, M., da Silva, M. C., Ingold, I., et al. (2019). FSP1 is a glutathione-independent ferroptosis suppressor. Nature 575, 693–698. doi: 10.1038/s41586-019-1707-0
Dong-Chen, X., Yong, C., Yang, X., Chen-Yu, S., and Li-Hua, P. (2023). Signaling pathways in Parkinson's disease: molecular mechanisms and therapeutic interventions. Signal Transduct. Target. Ther. 8:73. doi: 10.1038/s41392-023-01353-3
Dou, J., Liu, X., Yang, L., Huang, D., and Tan, X. (2022). Ferroptosis interaction with inflammatory microenvironments: mechanism, biology, and treatment. Biomed. Pharmacother. 155:113711. doi: 10.1016/j.biopha.2022.113711
Dusek, P., Schneider, S. A., and Aaseth, J. (2016). Iron chelation in the treatment of neurodegenerative diseases. J. Trace Elem. Med. Biol. 38, 81–92. doi: 10.1016/j.jtemb.2016.03.010
Evavold, C. L., Ruan, J., Tan, Y., Xia, S., Wu, H., and Kagan, J. C. (2018). The pore-forming protein Gasdermin D regulates Interleukin-1 secretion from living macrophages. Immunity 48, 35–44.e6. doi: 10.1016/j.immuni.2017.11.013
Fan, Z., Cai, L., Wang, S., Wang, J., and Chen, B. (2021). Baicalin prevents myocardial ischemia/reperfusion injury through inhibiting ACSL4 mediated Ferroptosis. Front. Pharmacol. 12:628988. doi: 10.3389/fphar.2021.628988
Fang, Y., Chen, X., Tan, Q., Zhou, H., Xu, J., and Gu, Q. (2021). Inhibiting Ferroptosis through disrupting the NCOA4-FTH1 interaction: a new mechanism of action. ACS Cent Sci. 7, 980–989. doi: 10.1021/acscentsci.0c01592
Faraji, P., Borchert, A., Ahmadian, S., and Kuhn, H. (2024). Butylated Hydroxytoluene (BHT) protects SH-SY5Y Neuroblastoma cells from Ferroptotic cell death: insights from in vitro and in vivo studies. Antioxidants (Basel). 13, 242–262. doi: 10.3390/antiox13020242
Feng, H., Schorpp, K., Jin, J., Yozwiak, C. E., Hoffstrom, B. G., Decker, A. M., et al. (2020). Transferrin receptor is a specific Ferroptosis marker. Cell Rep. 30, 3411–3423.e7. doi: 10.1016/j.celrep.2020.02.049
Fischer, W., Currais, A., Liang, Z., Pinto, A., and Maher, P. (2019). Old age-associated phenotypic screening for Alzheimer's disease drug candidates identifies sterubin as a potent neuroprotective compound from yerba santa. Redox Biol. 21:101089. doi: 10.1016/j.redox.2018.101089
Gan, B. (2021). Mitochondrial regulation of ferroptosis. J. Cell Biol. 220:e202105043. doi: 10.1083/jcb.202105043
Gao, Y., Tao, W., Wang, S., Duan, R., and Zhang, Z. (2024). AKR1C3 silencing inhibits autophagy-dependent glycolysis in thyroid cancer cells by inactivating ERK signaling. Drug Dev. Res. 85:e22142. doi: 10.1002/ddr.22142
Gao, M., Yi, J., Zhu, J., Minikes, A. M., Monian, P., Thompson, C. B., et al. (2019). Role of mitochondria in Ferroptosis. Mol. Cell 73, 354–363.e3. doi: 10.1016/j.molcel.2018.10.042
Gaschler, M. M., Andia, A. A., Liu, H., Csuka, J. M., Hurlocker, B., Vaiana, C. A., et al. (2018). FINO2 initiates ferroptosis through GPX4 inactivation and iron oxidation. Nat. Chem. Biol. 14, 507–515. doi: 10.1038/s41589-018-0031-6
GBD 2016 Neurology Collaborators (2019).. Global, regional, and national burden of neurological disorders, 1990-2016: a systematic analysis for the global burden of disease study 2016. Lancet Neurol. 18, 459–480. doi: 10.1016/S1474-4422(18)30499-X
Ge, M. H., Tian, H., Mao, L., Li, D. Y., Lin, J. Q., Hu, H. S., et al. (2021). Zinc attenuates ferroptosis and promotes functional recovery in contusion spinal cord injury by activating Nrf2/GPX4 defense pathway. CNS Neurosci. Ther. 27, 1023–1040. doi: 10.1111/cns.13657
Gong, Z., Wang, Y., Li, L., Li, X., Qiu, B., and Hu, Y. (2023). Cardamonin alleviates chondrocytes inflammation and cartilage degradation of osteoarthritis by inhibiting ferroptosis via p53 pathway. Food Chem. Toxicol. 174:113644. doi: 10.1016/j.fct.2023.113644
Guan, X., Li, Z., Zhu, S., Cheng, M., Ju, Y., Ren, L., et al. (2021). Galangin attenuated cerebral ischemia-reperfusion injury by inhibition of ferroptosis through activating the SLC7A11/GPX4 axis in gerbils. Life Sci. 264:118660. doi: 10.1016/j.lfs.2020.118660
Guerrero-Hue, M., García-Caballero, C., Palomino-Antolín, A., Rubio-Navarro, A., Vázquez-Carballo, C., Herencia, C., et al. (2019). Curcumin reduces renal damage associated with rhabdomyolysis by decreasing ferroptosis-mediated cell death. FASEB J. 33, 8961–8975. doi: 10.1096/fj.201900077R
Guo, Z., Lin, J., Sun, K., Guo, J., Yao, X., Wang, G., et al. (2022). Deferoxamine alleviates osteoarthritis by inhibiting chondrocyte Ferroptosis and activating the Nrf2 pathway. Front. Pharmacol. 13:791376. doi: 10.3389/fphar.2022.791376
Guo, H., Zhu, L., Tang, P., Chen, D., Li, Y., Li, J., et al. (2021). Carthamin yellow improves cerebral ischemia-reperfusion injury by attenuating inflammation and ferroptosis in rats. Int. J. Mol. Med. 47, 52–62. doi: 10.3892/ijmm.2021.4885
Han, J. Y., Lee, E. H., Kim, S. M., and Park, C. H. (2023). Efficient generation of dopaminergic neurons from mouse ventral midbrain astrocytes. Biomol. Ther. (Seoul) 31, 264–275. doi: 10.4062/biomolther.2022.140
He, H., Wang, L., Qiao, Y., Yang, B., Yin, D., and He, M. (2021). Epigallocatechin-3-gallate pretreatment alleviates doxorubicin-induced ferroptosis and cardiotoxicity by upregulating AMPKα2 and activating adaptive autophagy. Redox Biol. 48:102185. doi: 10.1016/j.redox.2021.102185
He, Y., Xi, J., Fang, J., Zhang, B., and Cai, W. (2023). Aloe-emodin alleviates doxorubicin-induced cardiotoxicity via inhibition of ferroptosis. Free Radic. Biol. Med. 206, 13–21. doi: 10.1016/j.freeradbiomed.2023.06.025
Hu, Q., Zuo, T., Deng, L., Chen, S., Yu, W., Liu, S., et al. (2022). β-Caryophyllene suppresses ferroptosis induced by cerebral ischemia reperfusion via activation of the NRF2/HO-1 signaling pathway in MCAO/R rats. Phytomedicine 102:154112. doi: 10.1016/j.phymed.2022.154112
Huang, Z., Chen, G., Wu, H., Huang, X., Xu, R., Deng, F., et al. (2023). Ebselen restores peri-implantitis-induced osteogenic inhibition via suppressing BMSCs ferroptosis. Exp. Cell Res. 427:113612. doi: 10.1016/j.yexcr.2023.113612
Jakaria, M., Belaidi, A. A., Bush, A. I., and Ayton, S. (2021). Ferroptosis as a mechanism of neurodegeneration in Alzheimer's disease. J. Neurochem. 159, 804–825. doi: 10.1111/jnc.15519
Jakaria, M., Belaidi, A. A., Bush, A. I., and Ayton, S. (2023). Vitamin a metabolites inhibit ferroptosis. Biomed. Pharmacother. 164:114930. doi: 10.1016/j.biopha.2023.114930
Jang, S., Chapa-Dubocq, X. R., Tyurina, Y. Y., St Croix, C. M., Kapralov, A. A., Tyurin, V. A., et al. (2021). Elucidating the contribution of mitochondrial glutathione to ferroptosis in cardiomyocytes. Redox Biol. 45:102021. doi: 10.1016/j.redox.2021.102021
Jiang, T., Chu, J., Chen, H., Cheng, H., Su, J., Wang, X., et al. (2020). Gastrodin inhibits H2O2-induced Ferroptosis through its Antioxidative effect in rat glioma cell line C6. Biol. Pharm. Bull. 43, 480–487. doi: 10.1248/bpb.b19-00824
Jiang, X., Stockwell, B. R., and Conrad, M. (2021). Ferroptosis: mechanisms, biology and role in disease. Nat. Rev. Mol. Cell Biol. 22, 266–282. doi: 10.1038/s41580-020-00324-8
Kenny, E. M., Fidan, E., Yang, Q., Anthonymuthu, T. S., New, L. A., Meyer, E. A., et al. (2019). Ferroptosis contributes to neuronal death and functional outcome after traumatic brain injury. Crit. Care Med. 47, 410–418. doi: 10.1097/CCM.0000000000003555
Kim, J. W., Lee, J. Y., Oh, M., and Lee, E. W. (2023). An integrated view of lipid metabolism in ferroptosis revisited via lipidomic analysis. Exp. Mol. Med. 55, 1620–1631. doi: 10.1038/s12276-023-01077-y
Klepac, N., Relja, M., Klepac, R., Hećimović, S., Babić, T., and Trkulja, V. (2007). Oxidative stress parameters in plasma of Huntington's disease patients, asymptomatic Huntington's disease gene carriers and healthy subjects: a cross-sectional study. J. Neurol. 254, 1676–1683. doi: 10.1007/s00415-007-0611-y
Kobayashi, S., Harada, Y., Homma, T., Yokoyama, C., and Fujii, J. (2021). Characterization of a rat monoclonal antibody raised against ferroptotic cells. J. Immunol. Methods 489:112912. doi: 10.1016/j.jim.2020.112912
Kosaka, K. (2014). Lewy body disease and dementia with Lewy bodies. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 90, 301–306. doi: 10.2183/pjab.90.301
Kose, T., Vera-Aviles, M., Sharp, P. A., and Latunde-Dada, G. O. (2019). Curcumin and (−)- Epigallocatechin-3-Gallate protect murine MIN6 pancreatic Beta-cells against Iron toxicity and Erastin-induced Ferroptosis. Pharmaceuticals (Basel) 12, 26–37. doi: 10.3390/ph12010026
Kuhlmann, T., Ludwin, S., Prat, A., Antel, J., Brück, W., and Lassmann, H. (2017). An updated histological classification system for multiple sclerosis lesions. Acta Neuropathol. 133, 13–24. doi: 10.1007/s00401-016-1653-y
Li, Y., Feng, D., Wang, Z., Zhao, Y., Sun, R., Tian, D., et al. (2019). Ischemia-induced ACSL4 activation contributes to ferroptosis-mediated tissue injury in intestinal ischemia/reperfusion. Cell Death Differ. 26, 2284–2299. doi: 10.1038/s41418-019-0299-4
Li, X., Si, W., Li, Z., Tian, Y., Liu, X., Ye, S., et al. (2021). miR-335 promotes ferroptosis by targeting ferritin heavy chain 1 in in vivo and in vitro models of Parkinson's disease. Int. J. Mol. Med. 47, 61–73. doi: 10.3892/ijmm.2021.4894
Li, T., Tan, Y., Ouyang, S., He, J., and Liu, L. (2022). Resveratrol protects against myocardial ischemia-reperfusion injury via attenuating ferroptosis. Gene 808:145968. doi: 10.1016/j.gene.2021.145968
Li, S., Zhen, L., Xiaomei, Y., Meng, Z., Yonghao, H., Di, W., et al. (2022). Propofol protects myocardium from ischemia/reperfusion injury by inhibiting Ferroptosis through the AKT/p53 signaling pathway. Front. Pharmacol. 13:841410. doi: 10.3389/fphar.2022.841410
Liang, D., Feng, Y., Zandkarimi, F., Wang, H., Zhang, Z., Kim, J., et al. (2023). Ferroptosis surveillance independent of GPX4 and differentially regulated by sex hormones. Cell. 186, 2748–2764.e22. doi: 10.1016/j.cell.2023.05.003
Lin, C. H., Tseng, H. F., Hsieh, P. C., Chiu, V., Lin, T. Y., Lan, C. C., et al. (2021). Nephroprotective role of Chrysophanol in hypoxia/Reoxygenation-induced renal cell damage via apoptosis, ER stress, and Ferroptosis. Biomedicines. 9, 1283–1295. doi: 10.3390/biomedicines9091283
Lin, J. H., Yang, K. T., Ting, P. C., Luo, Y. P., Lin, D. J., Wang, Y. S., et al. (2021). Gossypol acetic acid attenuates cardiac ischemia/reperfusion injury in rats via an Antiferroptotic mechanism. Biomol. Ther. 11, 1667–1682. doi: 10.3390/biom11111667
Liu, P., Feng, Y., Li, H., Chen, X., Wang, G., Xu, S., et al. (2020). Ferrostatin-1 alleviates lipopolysaccharide-induced acute lung injury via inhibiting ferroptosis. Cell. Mol. Biol. Lett. 25, 10–24. doi: 10.1186/s11658-020-00205-0
Liu, J., Li, X., Cai, R., Ren, Z., Zhang, A., Deng, F., et al. (2020). Simultaneous study of anti-Ferroptosis and antioxidant mechanisms of Butein and (S)-Butin. Molecules 25, 674–692. doi: 10.3390/molecules25030674
Liu, X. J., Lv, Y. F., Cui, W. Z., Li, Y., Liu, Y., Xue, Y. T., et al. (2021). Icariin inhibits hypoxia/reoxygenation-induced ferroptosis of cardiomyocytes via regulation of the Nrf2/HO-1 signaling pathway. FEBS Open Bio. 11, 2966–2976. doi: 10.1002/2211-5463.13276
Liu, Y., Wang, W., Li, Y., Xiao, Y., Cheng, J., and Jia, J. (2015). The 5-lipoxygenase inhibitor Zileuton confers neuroprotection against glutamate oxidative damage by inhibiting Ferroptosis. Biol. Pharm. Bull. 38, 1234–1239. doi: 10.1248/bpb.b15-00048
Liu, B., Zhao, C., Li, H., Chen, X., Ding, Y., and Xu, S. (2018). Puerarin protects against heart failure induced by pressure overload through mitigation of ferroptosis. Biochem. Biophys. Res. Commun. 497, 233–240. doi: 10.1016/j.bbrc.2018.02.061
Luo, H., and Zhang, R. (2021). Icariin enhances cell survival in lipopolysaccharide-induced synoviocytes by suppressing ferroptosis via the xc-/GPX4 axis. Exp. Ther. Med. 21, 72–80. doi: 10.3892/etm.2020.9504
Luoqian, J., Yang, W., Ding, X., Tuo, Q. Z., Xiang, Z., Zheng, Z., et al. (2022). Ferroptosis promotes T-cell activation-induced neurodegeneration in multiple sclerosis. Cell. Mol. Immunol. 19, 913–924. doi: 10.1038/s41423-022-00883-0
Masaldan, S., Belaidi, A. A., Ayton, S., and Bush, A. I. (2019). Cellular senescence and Iron Dyshomeostasis in Alzheimer's disease. Pharmaceuticals (Basel) 12, 93–104. doi: 10.3390/ph12020093
Mi, Y., Gao, X., Xu, H., Cui, Y., Zhang, Y., and Gou, X. (2019). The emerging roles of Ferroptosis in Huntington's disease. NeuroMolecular Med. 21, 110–119. doi: 10.1007/s12017-018-8518-6
Mishra, A. P., Salehi, B., Sharifi-Rad, M., Pezzani, R., Kobarfard, F., Sharifi-Rad, J., et al. (2018). Programmed cell death, from a Cancer perspective: an overview. Mol. Diagn. Ther. 22, 281–295. doi: 10.1007/s40291-018-0329-9
Mohan, S., Alhazmi, H. A., Hassani, R., Khuwaja, G., Maheshkumar, V. P., Aldahish, A., et al. (2024). Role of ferroptosis pathways in neuroinflammation and neurological disorders: from pathogenesis to treatment. Heliyon. 10:e24786. doi: 10.1016/j.heliyon.2024.e24786
Nguyen, H. P., Yi, D., Lin, F., Viscarra, J. A., Tabuchi, C., Ngo, K., et al. (2020). Aifm2, a NADH oxidase, supports robust glycolysis and is required for cold- and diet-induced thermogenesis. Mol. Cell. 77. doi: 10.1016/j.molcel.2019.12.002
Nishizawa, H., Matsumoto, M., Shindo, T., Saigusa, D., Kato, H., Suzuki, K., et al. (2020). Ferroptosis is controlled by the coordinated transcriptional regulation of glutathione and labile iron metabolism by the transcription factor BACH1. J. Biol. Chem. 295, 69–82. doi: 10.1074/jbc.RA119.009548
Orian, L., Mauri, P., Roveri, A., Toppo, S., Benazzi, L., Bosello-Travain, V., et al. (2015). Selenocysteine oxidation in glutathione peroxidase catalysis: an MS-supported quantum mechanics study. Free Radic. Biol. Med. 87, 1–14. doi: 10.1016/j.freeradbiomed.2015.06.011
Park, M. W., Cha, H. W., Kim, J., Kim, J. H., Yang, H., Yoon, S., et al. (2021). NOX4 promotes ferroptosis of astrocytes by oxidative stress-induced LPO via the impairment of mitochondrial metabolism in Alzheimer's diseases. Redox Biol. 41:101947. doi: 10.1016/j.redox.2021.101947
Probst, L., Dächert, J., Schenk, B., and Fulda, S. (2017). Lipoxygenase inhibitors protect acute lymphoblastic leukemia cells from ferroptotic cell death. Biochem. Pharmacol. 140, 41–52. doi: 10.1016/j.bcp.2017.06.112
Punziano, C., Trombetti, S., Cesaro, E., Grosso, M., and Faraonio, R. (2024). Antioxidant systems as modulators of Ferroptosis: focus on transcription factors. Antioxidants (Basel). 13, 298–350. doi: 10.3390/antiox13030298
Qiongyue, Z., Xin, Y., Meng, P., Sulin, M., Yanlin, W., Xinyi, L., et al. (2022). Post-treatment with Irisin attenuates acute kidney injury in Sepsis mice through anti-Ferroptosis via the SIRT1/Nrf2 pathway. Front. Pharmacol. 13:857067. doi: 10.3389/fphar.2022.857067
Qiu, H., Huang, S., Liu, Y., Liu, L., Guo, F., Guo, Y., et al. (2024). Idebenone alleviates doxorubicin-induced cardiotoxicity by stabilizing FSP1 to inhibit ferroptosis. Acta Pharm. Sin. B 14, 2581–2597. doi: 10.1016/j.apsb.2024.03.015
Rayatpour, A., Foolad, F., Heibatollahi, M., Khajeh, K., and Javan, M. (2022). Ferroptosis inhibition by deferiprone, attenuates myelin damage and promotes neuroprotection in demyelinated optic nerve. Sci. Rep. 12:19630. doi: 10.1038/s41598-022-24152-2
Reich, D. S., Lucchinetti, C. F., and Calabresi, P. A. (2018). Multiple Sclerosis. N. Engl. J. Med. 378, 169–180. doi: 10.1056/NEJMra1401483
Reichert, C. O., de Freitas, F. A., Sampaio-Silva, J., Rokita-Rosa, L., Barros, P. L., Levy, D., et al. (2020). Ferroptosis mechanisms involved in neurodegenerative diseases. Int. J. Mol. Sci. 21, 8765–8792. doi: 10.3390/ijms21228765
Rogers, J. T., Xia, N., Wong, A., Bakshi, R., and Cahill, C. M. (2019). Targeting the Iron-response elements of the mRNAs for the Alzheimer's amyloid precursor protein and ferritin to treat acute Lead and manganese neurotoxicity. Int. J. Mol. Sci. 20, 994–1010. doi: 10.3390/ijms20040994
Schaar, C. E., Dues, D. J., Spielbauer, K. K., Machiela, E., Cooper, J. F., Senchuk, M., et al. (2015). Mitochondrial and cytoplasmic ROS have opposing effects on lifespan. PLoS Genet. 11:e1004972. doi: 10.1371/journal.pgen.1004972
Shah, R., Shchepinov, M. S., and Pratt, D. A. (2018). Resolving the role of lipoxygenases in the initiation and execution of Ferroptosis. ACS Cent Sci. 4, 387–396. doi: 10.1021/acscentsci.7b00589
Shan, X., Lv, Z. Y., Yin, M. J., Chen, J., Wang, J., and Wu, Q. N. (2021). The protective effect of Cyanidin-3-glucoside on myocardial ischemia-reperfusion injury through Ferroptosis. Oxidative Med. Cell. Longev. 2021:8880141. doi: 10.1155/2021/8880141
Shi, H., Song, L., Wu, Y., Shen, R., Zhang, C., Liao, X., et al. (2024). Edaravone alleviates traumatic brain injury by inhibition of Ferroptosis via FSP1 pathway. Mol. Neurobiol. s12035-024-04216-2. doi: 10.1007/s12035-024-04216-2
Shimada, K., Skouta, R., Kaplan, A., Yang, W. S., Hayano, M., Dixon, S. J., et al. (2016). Global survey of cell death mechanisms reveals metabolic regulation of ferroptosis. Nat. Chem. Biol. 12, 497–503. doi: 10.1038/nchembio.2079
Skouta, R., Dixon, S. J., Wang, J., Dunn, D. E., Orman, M., Shimada, K., et al. (2014). Ferrostatins inhibit oxidative lipid damage and cell death in diverse disease models. J. Am. Chem. Soc. 136, 4551–4556. doi: 10.1021/ja411006a
Stockwell, B. R. (2022). Ferroptosis turns 10: emerging mechanisms, physiological functions, and therapeutic applications. Cell 185, 2401–2421. doi: 10.1016/j.cell.2022.06.003
Sun, Y., He, L., Wang, T., Hua, W., Qin, H., Wang, J., et al. (2020). Activation of p62-Keap1-Nrf2 pathway protects 6-Hydroxydopamine-induced Ferroptosis in dopaminergic cells. Mol. Neurobiol. 57, 4628–4641. doi: 10.1007/s12035-020-02049-3
Tabrizi, S. J., Flower, M. D., Ross, C. A., and Wild, E. J. (2020). Huntington disease: new insights into molecular pathogenesis and therapeutic opportunities. Nat. Rev. Neurol. 16, 529–546. doi: 10.1038/s41582-020-0389-4
Takashima, M., Ichihara, K., and Hirata, Y. (2019). Neuroprotective effects of Brazilian green propolis on oxytosis/ferroptosis in mouse hippocampal HT22 cells. Food Chem. Toxicol. 132:110669. doi: 10.1016/j.fct.2019.110669
Tang, D., Chen, X., Kang, R., and Kroemer, G. (2021). Ferroptosis: molecular mechanisms and health implications. Cell Res. 31, 107–125. doi: 10.1038/s41422-020-00441-1
Tang, D., Kang, R., Berghe, T. V., Vandenabeele, P., and Kroemer, G. (2019). The molecular machinery of regulated cell death. Cell Res. 29, 347–364. doi: 10.1038/s41422-019-0164-5
Tang, X., Li, X., Zhang, D., and Han, W. (2022). Astragaloside-IV alleviates high glucose-induced ferroptosis in retinal pigment epithelial cells by disrupting the expression of miR-138-5p/Sirt1/Nrf2. Bioengineered. 13, 8238–8253. doi: 10.1080/21655979.2022.2049471
Tang, Y., Luo, H., Xiao, Q., Li, L., Zhong, X., Zhang, J., et al. (2021). Isoliquiritigenin attenuates septic acute kidney injury by regulating ferritinophagy-mediated ferroptosis. Ren. Fail. 43, 1551–1560. doi: 10.1080/0886022X.2021.2003208
Tian, Y., Lu, J., Hao, X., Li, H., Zhang, G., Liu, X., et al. (2020). FTH1 inhibits Ferroptosis through Ferritinophagy in the 6-OHDA model of Parkinson's disease. Neurotherapeutics 17, 1796–1812. doi: 10.1007/s13311-020-00929-z
Tonnus, W., Meyer, C., Paliege, A., Belavgeni, A., von Mässenhausen, A., Bornstein, S. R., et al. (2019). The pathological features of regulated necrosis. J. Pathol. 247, 697–707. doi: 10.1002/path.5248
Tufail, M., and Hassan, I. (2020). Influence of consanguinity and medication on the development of Parkinson's disease. Dement. Geriatr. Cogn. Disord. 49, 390–393. doi: 10.1159/000509586
Varela, L., and Garcia-Rendueles, M. E. R. (2022). Oncogenic pathways in neurodegenerative diseases. Int. J. Mol. Sci. 23, 3223–3245. doi: 10.3390/ijms23063223
Venkatesh, D., O'Brien, N. A., Zandkarimi, F., Tong, D. R., Stokes, M. E., Dunn, D. E., et al. (2020). MDM2 and MDMX promote ferroptosis by PPARα-mediated lipid remodeling. Genes Dev. 34, 526–543. doi: 10.1101/gad.334219.119
Wang, Y., Liao, S., Pan, Z., Jiang, S., Fan, J., Yu, S., et al. (2022). Hydrogen sulfide alleviates particulate matter-induced emphysema and airway inflammation by suppressing ferroptosis. Free Radic. Biol. Med. 186, 1–16. doi: 10.1016/j.freeradbiomed.2022.04.014
Wang, Y., Lv, M. N., and Zhao, W. J. (2023a). Research on ferroptosis as a therapeutic target for the treatment of neurodegenerative diseases. Ageing Res. Rev. 91:102035. doi: 10.1016/j.arr.2023.102035
Wang, D., Peng, Y., Xie, Y., Zhou, B., Sun, X., Kang, R., et al. (2016). Antiferroptotic activity of non-oxidative dopamine. Biochem. Biophys. Res. Commun. 480, 602–607. doi: 10.1016/j.bbrc.2016.10.099
Wang, Y., Quan, F., Cao, Q., Lin, Y., Yue, C., Bi, R., et al. (2020). Quercetin alleviates acute kidney injury by inhibiting ferroptosis. J. Adv. Res. 28, 231–243. doi: 10.1016/j.jare.2020.07.007
Wang, T., Tomas, D., Perera, N. D., Cuic, B., Luikinga, S., Viden, A., et al. (2022). Ferroptosis mediates selective motor neuron death in amyotrophic lateral sclerosis. Cell Death Differ. 29, 1187–1198. doi: 10.1038/s41418-021-00910-z
Wang, F., Wang, J., Shen, Y., Li, H., Rausch, W. D., and Huang, X. (2022). Iron Dyshomeostasis and Ferroptosis: a New Alzheimer's disease hypothesis? Front. Aging Neurosci. 14:830569. doi: 10.3389/fnagi.2022.830569
Wang, Y., Wu, S., Li, Q., Sun, H., and Wang, H. (2023b). Pharmacological inhibition of Ferroptosis as a therapeutic target for neurodegenerative diseases and strokes. Adv. Sci. (Weinh). 10:e2300325. doi: 10.1002/advs.202300325
Wang, Z., Yao, M., Jiang, L., Wang, L., Yang, Y., Wang, Q., et al. (2022). Dexmedetomidine attenuates myocardial ischemia/reperfusion-induced ferroptosis via AMPK/GSK-3β/Nrf2 axis. Biomed. Pharmacother. 154:113572. doi: 10.1016/j.biopha.2022.113572
Wang, C., Yuan, W., Hu, A., Lin, J., Xia, Z., Yang, C. F., et al. (2020). Dexmedetomidine alleviated sepsis-induced myocardial ferroptosis and septic heart injury. Mol. Med. Rep. 22, 175–184. doi: 10.3892/mmr.2020.11114
Wen, L., Shi, D., Zhou, T., Tu, J., He, M., Jiang, Y., et al. (2020). Identification of two novel prenylated flavonoids in mulberry leaf and their bioactivities. Food Chem. 315:126236. doi: 10.1016/j.foodchem.2020.126236
Wu, M., Xu, L. G., Li, X., Zhai, Z., and Shu, H. B. (2002). AMID, an apoptosis-inducing factor-homologous mitochondrion-associated protein, induces caspase-independent apoptosis. J. Biol. Chem. 277, 25617–25623. doi: 10.1074/jbc.M202285200
Wyttenbach, A., Sauvageot, O., Carmichael, J., Diaz-Latoud, C., Arrigo, A. P., and Rubinsztein, D. C. (2002). Heat shock protein 27 prevents cellular polyglutamine toxicity and suppresses the increase of reactive oxygen species caused by huntingtin. Hum. Mol. Genet. 11, 1137–1151. doi: 10.1093/hmg/11.9.1137
Xia, Y., Wang, H., Xie, Z., Liu, Z. H., and Wang, H. L. (2024). Inhibition of ferroptosis underlies EGCG mediated protection against Parkinson's disease in a Drosophila model. Free Radic. Biol. Med. 211, 63–76. doi: 10.1016/j.freeradbiomed.2023.12.005
Xie, Y., Hou, W., Song, X., Yu, Y., Huang, J., Sun, X., et al. (2016a). Ferroptosis: process and function. Cell Death Differ. 23, 369–379. doi: 10.1038/cdd.2015.158
Xie, Y., Song, X., Sun, X., Huang, J., Zhong, M., Lotze, M. T., et al. (2016b). Identification of baicalein as a ferroptosis inhibitor by natural product library screening. Biochem. Biophys. Res. Commun. 473, 775–780. doi: 10.1016/j.bbrc.2016.03.052
Xu, Y., Lin, H., Wang, H., Pang, J., and Zhou, Y. (2021). Fraxetin attenuates ferroptosis in myocardial infarction via AKT/Nrf2/HO-1 signaling. Am. J. Transl. Res. 13, 10315–10327
Xu, S., Wu, B., Zhong, B., Lin, L., Ding, Y., Jin, X., et al. (2021). Naringenin alleviates myocardial ischemia/reperfusion injury by regulating the nuclear factor-erythroid factor 2-related factor 2 (Nrf2)/system xc−/glutathione peroxidase 4 (GPX4) axis to inhibit ferroptosis. Bioengineered 12, 10924–10934. doi: 10.1080/21655979.2021.1995994
Yamada, S., Kumazawa, S., Ishii, T., Nakayama, T., Itakura, K., Shibata, N., et al. (2001). Immunochemical detection of a lipofuscin-like fluorophore derived from malondialdehyde and lysine. J. Lipid Res. 42, 1187–1196. doi: 10.1016/S0022-2275(20)31568-6
Yan, B., Ai, Y., Sun, Q., Ma, Y., Cao, Y., Wang, J., et al. (2021). Membrane damage during Ferroptosis is caused by oxidation of phospholipids catalyzed by the oxidoreductases POR and CYB5R1. Mol. Cell 81, 355–369.e10. doi: 10.1016/j.molcel.2020.11.024
Yang, L., Guo, C., Zhu, J., Feng, Y., Chen, W., Feng, Z., et al. (2017). Increased levels of pro-inflammatory and anti-inflammatory cellular responses in Parkinson's disease patients: search for a disease Indicator. Med. Sci. Monit. 23, 2972–2978. doi: 10.12659/msm.904240
Yang, W., Hamilton, J. L., Kopil, C., Beck, J. C., Tanner, C. M., Albin, R. L., et al. (2020). Current and projected future economic burden of Parkinson's disease in the U. S. NPJ Parkinsons Dis. 6:15. doi: 10.1038/s41531-020-0117-1
Yang, W. S., Kim, K. J., Gaschler, M. M., Patel, M., Shchepinov, M. S., and Stockwell, B. R. (2016). Peroxidation of PUFA by lipoxygenases drives ferroptosis. Proc. Natl. Acad. Sci. USA 113, E4966–E4975. doi: 10.1073/pnas.1603244113
Yang, L., Liu, Y., Zhang, W., Hua, Y., Chen, B., Wu, Q., et al. (2021). Ferroptosis-inhibitory difference between Chebulagic acid and Chebulinic acid indicates beneficial role of HHDP. Molecules 26, 4300–4313. doi: 10.3390/molecules26144300
Yang, K., Zeng, L., Zeng, J., Deng, Y., Wang, S., Xu, H., et al. (2023). Research progress in the molecular mechanism of ferroptosis in Parkinson's disease and regulation by natural plant products. Ageing Res. Rev. 91:102063. doi: 10.1016/j.arr.2023.102063
Yao, F., Cui, X., Zhang, Y., Bei, Z., Wang, H., Zhao, D., et al. (2021). Iron regulatory protein 1 promotes ferroptosis by sustaining cellular iron homeostasis in melanoma. Oncol. Lett. 22:657. doi: 10.3892/ol.2021.12918
Yao, X., Zhang, Y., Hao, J., Duan, H. Q., Zhao, C. X., Sun, C., et al. (2019). Deferoxamine promotes recovery of traumatic spinal cord injury by inhibiting ferroptosis. Neural Regen. Res. 14, 532–541. doi: 10.4103/1673-5374.245480
Yuan, Z., Miao, Z., Gong, X., Zhao, B., Zhang, Y., Ma, H., et al. (2017). Changes on LPO, enzymatic activities and gene expression in planarian (Dugesia japonica) following exposure to perfluorooctanoic acid. Ecotoxicol. Environ. Saf. 145, 564–568. doi: 10.1016/j.ecoenv.2017.08.008
Yuan, Y., Zhai, Y., Chen, J., Xu, X., and Wang, H. (2021). Kaempferol ameliorates oxygen-glucose deprivation/Reoxygenation-induced neuronal Ferroptosis by activating Nrf2/SLC7A11/GPX4 Axis. Biomol. Ther. 11, 923–937. doi: 10.3390/biom11070923
Zeng, W., Cai, J., Zhang, L., and Peng, Q. (2024). Iron deposition in Parkinson's disease: a Mini-review. Cell. Mol. Neurobiol. 44:26. doi: 10.1007/s10571-024-01459-4
Zhang, F., Lin, B., Huang, S., Wu, P., Zhou, M., Zhao, J., et al. (2023). Melatonin alleviates retinal ischemia-reperfusion injury by inhibiting p53-mediated Ferroptosis. Antioxidants (Basel). 12, 1173–1196. doi: 10.3390/antiox12061173
Zhang, Y., Tan, H., Daniels, J. D., Zandkarimi, F., Liu, H., Brown, L. M., et al. (2019). Imidazole ketone Erastin induces Ferroptosis and slows tumor growth in a mouse lymphoma model. Cell Chem. Biol. 26, 623–633.e9. doi: 10.1016/j.chembiol.2019.01.008
Zhang, Z., Tang, J., Song, J., Xie, M., Liu, Y., Dong, Z., et al. (2022). Elabela alleviates ferroptosis, myocardial remodeling, fibrosis and heart dysfunction in hypertensive mice by modulating the IL-6/STAT3/GPX4 signaling. Free Radic. Biol. Med. 181, 130–142. doi: 10.1016/j.freeradbiomed.2022.01.020
Zhang, J., Zhu, Q., Peng, Z., Li, X. J., Ding, P. F., Gao, S., et al. (2024). Menaquinone-4 attenuates ferroptosis by upregulating DHODH through activation of SIRT1 after subarachnoid hemorrhage. Free Radic. Biol. Med. 219, 180–183. doi: 10.1016/j.freeradbiomed.2024.04.227
Zheng, H., Jiang, L., Tsuduki, T., Conrad, M., and Toyokuni, S. (2021). Embryonal erythropoiesis and aging exploit ferroptosis. Redox Biol. 48:102175. doi: 10.1016/j.redox.2021.102175
Zhu, R., Kang, Y., Li, Q., Peng, K., Shi, X., Yin, Z., et al. (2024). Alpha-tocopherol inhibits ferroptosis and promotes neural function recovery in rats with spinal cord injury via downregulating Alox15. Biomed. Pharmacother. 175:116734. doi: 10.1016/j.biopha.2024.116734
Keywords: ferroptosis, regulated cell death, ferroptosis inhibitor, inhibitor, neurodegenerative diseases
Citation: Fei Y and Ding Y (2024) The role of ferroptosis in neurodegenerative diseases. Front. Cell. Neurosci. 18:1475934. doi: 10.3389/fncel.2024.1475934
Received: 04 August 2024; Accepted: 27 September 2024;
Published: 15 October 2024.
Edited by:
Lang Wang, Zhejiang University, ChinaReviewed by:
Guangtao Xu, Jiaxing University Medical College, ChinaCopyright © 2024 Fei and Ding. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yifan Fei, ZmVpeTFmQHFxLmNvbQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.