- 1Department of Respiratory Medicine, The First Hospital of Jilin University, Changchun, China
- 2College of Biological and Food Engineering, Jilin Engineering Normal University, Changchun, China
Staphylococcus aureus sortase A can anchor virulence proteins, which are responsible for bacterial adhesion, biofilm formation, and inflammation, to the cell membrane surface. The ability of β-lactam antibiotics to combat S. aureus infections is limited by the presence of β-lactamases in this pathogen. In this study, we determined that epicatechin gallate (ECG) and its analogues inhibited the transpeptidase activity of sortase A by interacting with it directly, and the biofilm formation and adhesion abilities of the bacterium decreased after treatment with ECG and its analogues. Additionally, ECG bound to β-lactamase and reduced its ability to hydrolyze nitrocefin. Furthermore, ECG synergized with ampicillin (Amp), enhancing its bactericidal effects and inhibiting the formation of persisters. ECG did not affect the expression of sortase A or β-lactamase but significantly alleviated the cytotoxicity of S. aureus USA300. ECG alone or combined with Amp in vivo improved the survival of mice infected with S. aureus USA300, alleviated pathological tissue damage and pulmonary edema, and reduced the extent of inflammation and level of colonization. The results of this study indicate that the active ingredients of green tea, especially ECG, have the potential to be developed as anti-S. aureus infection agents.
Introduction
For successful infection, Staphylococcus aureus (S. aureus) must adhere to and colonize the host (Ronner et al., 2014; Foster, 2019), and this process is aided by adhesins covalently anchored to the cell wall by sortase A (Foster et al., 2014; Foster, 2019; Alharthi et al., 2021). Sortase A inhibitors significantly reduce bacterial adherence to cells (Liu et al., 2015; Bozhkova et al., 2022; Shulga and Kudryavtsev, 2022). In addition, sortase A can anchor many other surface proteins to the cell wall, which could improve bacterial pathogenicity (Jonsson et al., 2003; Weiss et al., 2004) through mechanisms such as affecting biofilm formation (Kim et al., 2018; Xu et al., 2024) and promoting S. aureus evasion of the host immune system (Weiss et al., 2004). Some inhibitors targeting sortase A have been reported to reduce bacterial adherence to host cells and biofilm formation, thereby increasing the survival rate of model animals with S. aureus infection (Song et al., 2022; Su et al., 2022; Jiang et al., 2023). The number of virulence proteins anchored to the bacterial cell wall decreases significantly upon sortase A inactivation, which results in a reduction in S. aureus pathogenicity. Therefore, sortase A is an ideal target for developing inhibitors of S. aureus infection (Cascioferro et al., 2014; Cascioferro et al., 2015; Alharthi et al., 2021).
S. aureus β-lactamase can hydrolyze almost all β-lactam antibiotics, including the recently developed cephalosporins and all carbapenem antibiotics, known as the “last antibiotics”, which imposes a considerable challenge for the clinical treatment of S. aureus infection (Mossman et al., 2022; George et al., 2023); however, the development of new antibiotics is slow. Therefore, restoring bacterial sensitivity to current antibiotics is an effective strategy to alleviate resistance. Several compounds have been shown to restore bacterial susceptibility to antibiotics by targeting β-lactamases, indicating that restoring bacterial sensitivity to this class of antibiotics is feasible by inhibiting β-lactamase activity.
Catechins are active phenolic substances that are mainly found in green tea, including the main active components epicatechin gallate (ECG), catechin (C), epicatechin (EC), epigallocatechin (EGC), and epigallocatechin gallate (EGCG) (Kochman et al., 2020; Musial et al., 2020). These compounds have considerable biological functions, such as antiviral, antitumoral, antioxidant, and antithrombotic effects (Ide et al., 2016; Chen et al., 2021; Chen et al., 2022). EGCG has been reported to affect Streptococcus pneumoniae virulence by inactivating sortase A (Song et al., 2017), but compounds that act against both β-lactamase and sortase A simultaneously to suppress S. aureus virulence have not been reported. In this study, we discovered that the abovementioned components of green tea inhibited S. aureus USA300 infection by interacting with β-lactamase and sortase A. On the one hand, these compounds inhibited biofilm formation and bacterial adhesion by inhibiting sortase A. On the other hand, ECG, as a representative, restored the susceptibility of S. aureus USA300 to β-lactam antibiotics and protected mice from S. aureus USA300 infection.
Materials and methods
Reagents, strains, and growth conditions
The small molecules used in this study were obtained from Chengdu Purechem-Standard Co., Ltd. Penicillin G (PG), ampicillin (Amp), ceftriaxone sodium (Cef), kanamycin, and nitrocefin were purchased from Dalian Meilun Biotechnology Co., Ltd. Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), trypsin (0.25%), and penicillin−streptomycin solution were purchased from Beijing Solarbio Science & Technology Co., Ltd. The substrate peptide of sortase A (Dabcyl-QALPETGEE-Edans) was purchased from GL Biochem (Shanghai), Ltd. S. aureus USA300, which expresses β-lactamase, was stored in our laboratory and cultured in Luria−Bertani (LB) media at 37°C with shaking or under static conditions.
Protein preparation
The S. aureus sortase A gene was cloned and inserted into the pET-28a (+) vector, which was subsequently transformed into Escherichia coli BL21 (DE3) (Beyotime, Shanghai, China). The E. coli was cultured in LB medium supplemented with 0.2 mM isopropyl-beta-D-thiogalactopyranoside (Beyotime, Shanghai, China) at 18°C for 18 h. Then, the bacteria were harvested and lysed to obtain the supernatant. The supernatant was incubated with Ni-NTA agarose resin, and the purified protein was obtained after elution with 250 mM imidazole. To construct the sortase A mutants, primers designed for specific mutations (V168A, K173A, and L169A) were obtained using the QuikChange Site-Directed Mutagenesis Kit. The pET28a plasmid carrying the sortase A gene was used as a template, and the polymerase chain reaction (PCR) products were introduced into E. coli BL21 (DE3) after treatment with DpnI (Beyotime, Shanghai, China). The mutated proteins were obtained via the same purification method described above. Cloning, expression, and purification of β-lactamase and its mutants (Y96A, I158A, and I230A) were performed via the same methods. The mutants of sortase A and β-lactamase were used to identify the residues critical for interaction with the compounds. The primers used for this assay are shown in Table 1.
Inhibition of sortase A activity
The sortase A protein (10 µg) was coincubated with various concentrations of ECG and its analogues (0, 8, 16, and 32 µg/mL) at 37°C for 0.5 h. The substrate peptide (10 µg, final volume of 100 µL) was subsequently added, and the samples were incubated for 1 h in the dark. Then, the fluorescence intensity was detected by using a microplate reader (excitation 490 nm, emission 520 nm), and the inhibitory effects of ECG and its analogues against sortase A were evaluated via a previously described method (Song et al., 2017). The inhibitory effects of ECG against the mutants of sortase A (V168A, K173A and L169A) were determined via the same method.
Computational biology
AutoDock Vina software (Trott and Olson, 2010) was used to perform docking calculations after the compounds (set as the ligands) were docked into proteins (β-lactamase PDB ID: 6WGR; sortase A PDB ID: 6R1V) with AutoDock Tools on the basis of methods described previously (Gao et al., 2020). Molecular dynamics simulations of the candidates were carried out using GROMACS 2020.6 and previously reported methods (Liu TT. et al., 2019; Gao et al., 2020). Briefly, RESP charges were assigned to the ligands using Gaussian and amber, and topology files of the ligands were obtained by using AmberTools. The Amber 14sb force field and SPC/E water model were also used. The root mean square deviation (RMSD) was used to evaluate the equilibrium of the system, and key protein residues involved in the interactions with the ligands were identified via the molecular mechanics Poisson−Boltzmann surface area (MMPBSA) method.
Biofilm inhibition
Different concentrations of ECG and its analogues (0, 8, and 32 µg/mL) were used to treat S. aureus USA300, which were seeded into 96-well plates (5 × 106 CFUs/well). The samples were cultured at 37°C until static. At 24 h later, the medium was removed, and the samples were washed with sterile phosphate-buffered saline (PBS) and stained with 0.1% crystal violet. The optical density at 570 nm (OD570) of each well was measured after adding 33% glacial acetic acid to dissolve the solids to evaluate the biofilm formation inhibitory effects of the compounds (Grossman et al., 2021). The free LB medium was defined as negative control (NC).
Inhibition of β-lactamase activity
S. aureus USA300 β-lactamase protein activity inhibition assays were performed according to methods reported previously (Zhou et al., 2020). Briefly, different concentrations of ECG and its derivatives (0, 32, 64, and 128 µg/mL) were used to treat the purified β-lactamase protein (5 µg) for 30 min at 37°C, and nitrocefin (5 µg) was added for the final 10 min of incubation. The OD492 of each well was subsequently measured to determine whether these compounds affect the activity of β-lactamase (Zhou et al., 2020). The inhibitory effects of ECG against the β-lactamase mutants (Y96A, I158A, and I230A) were determined via the same method.
Analysis of the antimicrobial properties
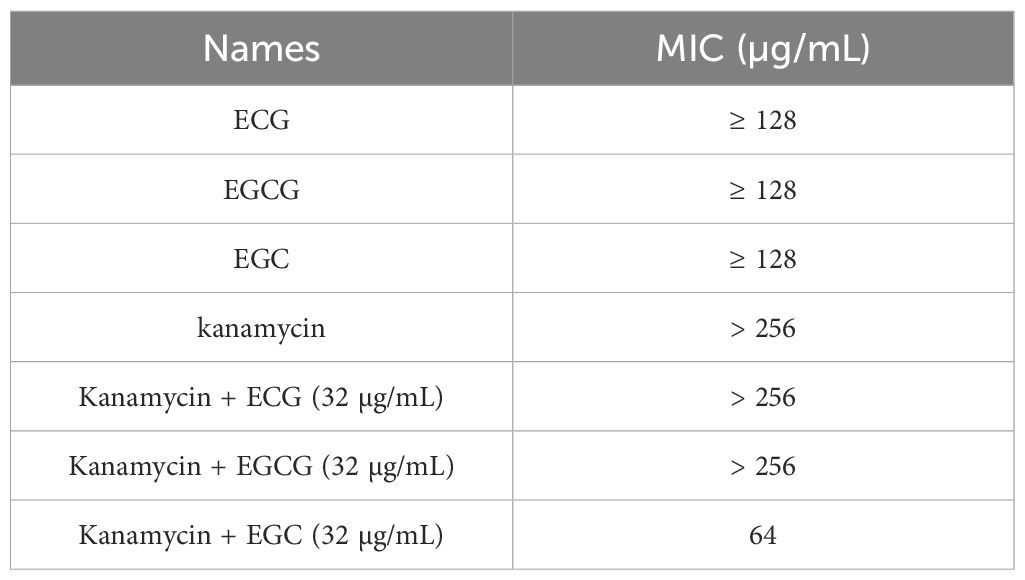
The minimum inhibitory concentrations (MICs) of PG, Amp, Cef, and kanamycin combined with or without the test compounds were determined on the basis of Clinical and Laboratory Standards Institute (CLSI) methods. Briefly, LB culture medium containing a series of concentrations of PG, Cef, Amp, or kanamycin (0–256 µg/mL) and ECG derivatives (0–32 µg/mL) was added to a 96-well plate, followed by the addition of S. aureus USA300 strains (5 × 105 CFUs/mL). Samples were cultured at 37°C for 24 h, and the MIC values were defined as the concentration of compound at which there was no visible bacteria growth. The antimicrobial properties of the ECG and its analogues and their synergistic effects with PG, Cef, Amp, and kanamycin were analyzed on the basis of the MIC values (Guo et al., 2024).
Time-dependent bacterial killing effects and analysis of persisters
On the basis of the synergistic effect between the ECG and its analogues and the tested antibiotics, Amp combined with ECG (32 µg/mL) was used to carry out a time-dependent bacterial killing assay. S. aureus USA300 in the logarithmic growth phase (approximately 2 × 106 CFUs/mL) was treated with ECG (32 µg/mL), Amp (64 µg/mL), or their combination and cultured with shaking. Samples from each group were collected every 2 h and plated on LB agar medium after dilution. The colonies were analyzed to evaluate the ability of ECG to improve the bactericidal ability of Amp (Liu S. et al., 2019). The persister assay was performed on the basis of a previously described method with some modifications (Keren et al., 2004). Briefly, S. aureus USA300 was treated with 10 µg/mL gentamicin, then ECG (32 µg/mL) was added, and the mixture was cultured with shaking. Finally, bacterial colonies were harvested at the indicated time points to analyze the formation of persisters (Keren et al., 2004).
Cell culture and assays
Mouse mononuclear macrophages (RAW264.7 cells) and human lung cancer epithelial cells (A549 cells) were cultured in DMEM supplemented with 10% FBS at 37°C with 5% CO2. The RAW264.7 cells seeded in 96-well plates (2 × 104 cells/well) were treated with S. aureus USA300 at a multiplicity of infection (MOI) of 60, after different concentrations of ECG (0, 16, or 32 µg/mL) were added, and the samples were cocultured for 5 h. Then, the samples were centrifuged (1,000 rpm for 10 min), the supernatant was added to an equal volume of lactate dehydrogenase (LDH) reagent (Solarbio, Beijing, China), and the mixture was incubated in the dark for 30 min. The level of LDH in each sample was determined by measuring the OD490. After having been washed with sterile PBS, the cells were treated with ethidium bromide (EB, 50 µg/mL) to observe the inhibitory effect of ECG on the toxicity of S. aureus USA300 by using fluorescence microscopy (OLYMPUS, IX83). The cytotoxicities of the tested compounds were determined by detecting the LDH levels. For the adhesion assay, A549 cells in 24-well plates (1 × 105 cells/well) were treated with S. aureus USA300 (MOI = 30) containing ECG (0, 8, or 32 µg/mL). At 1 h later, the medium was discarded, the cells were washed with sterile PBS, and the samples were plated onto LB agar medium after dilution. The number of colonies was used to assess the inhibitory effect of ECG on bacterial adhesion.
Protein expression and secretion analysis
S. aureus USA300 was cocultured with different concentrations of ECG (0, 16, or 32 µg/mL) for 8 h. The bacteria and supernatants were subsequently collected separately after centrifugation (12,000 rpm for 5 min). The expression levels of sortase A and β-lactamase in the bacteria were detected by western blotting. The secretion of β-lactamase into the supernatant was evaluated via Coomassie brilliant blue staining. The antibodies used in these experiments were stored in our laboratory (mouse sortase A, 1:1,000; mouse β-lactamase, 1:1,000; goat anti-mouse secondary antibodies, 1:10,000).
Animal infection model
Male C57BL/6J mice (weighing approximately 20 g) were purchased from Liaoning Changsheng Biotechnology Co., Ltd. All animal experiments were performed in accordance with the requirements of Jilin University. The mice had free access to food and water. The mice were intranasally inoculated with 30 μL of S. aureus USA300 suspension (5 × 108 CFUs) to establish a pneumonia model. At 2 h later, ECG (50 mg/kg), Amp (50 mg/kg), or their combination was administered to the infected mice. Mice in the positive control (PC) group were infected with S. aureus USA300 and administered an equal volume of solvent instead of compound, and the mice in the negative control (NC) group were administered an equal volume of sterile PBS only. A total of 10 mice were assigned to each group for the survival assay, and the survival status of the mice was observed every 12 h. For other analyses, each group contained eight mice. After 48 hours of treatment, the mice were anaesthetized by injection of pentobarbital sodium (50 mg/kg) and then euthanized by cervical dislocation. The lung tissues of the mice were harvested, and pathological damage was observed after haematoxylin−eosin staining. Additionally, the tissue bacterial load was analyzed after homogenization and culture on LB agar, the ratio of the weight of each lung before and after drying was analyzed, and the levels of inflammatory factors in the alveolar lavage fluid was detected via enzyme-linked immunosorbent assays (ELISAs; Sangon Biotech, Shanghai, China).
Statistical analysis
The data are presented as the means with the standard deviations (SDs) or standard errors of the means (SEMs) from three independent experiments. Statistical analysis was performed via the unpaired t test in GraphPad Prism 9.5.0. Statistical significance was defined as p ≤ 0.05.
Results
ECG and its analogues inhibit the activity of sortase A
The relative activity of sortase A was defined as 100% at a test compound concentration of 0 µg/mL. Sortase A activity was reduced to 79.11%, 64.14% and 44.52% upon treatment with 8, 16 and 32 µg/mL ECG, respectively (Figure 1A). Similarly, reductions in sortase A activity were observed after treatment with 8, 16 and 32 µg/mL EGCG (13.68%, 36.35% and 61.22%, respectively) (Figure 1B), EGC (28.23%, 29.87% and 48.07%, respectively) (Figure 1C), C (10%, 14.32% and 30.88%, respectively) (Figure 1D) and EC (2.94%, 16.12% and 40.14%, respectively) (Figure 1E). These results showed that ECG and its analogues could significantly inhibit the activity of sortase A and that the gallic acid moiety played a role in this activity.
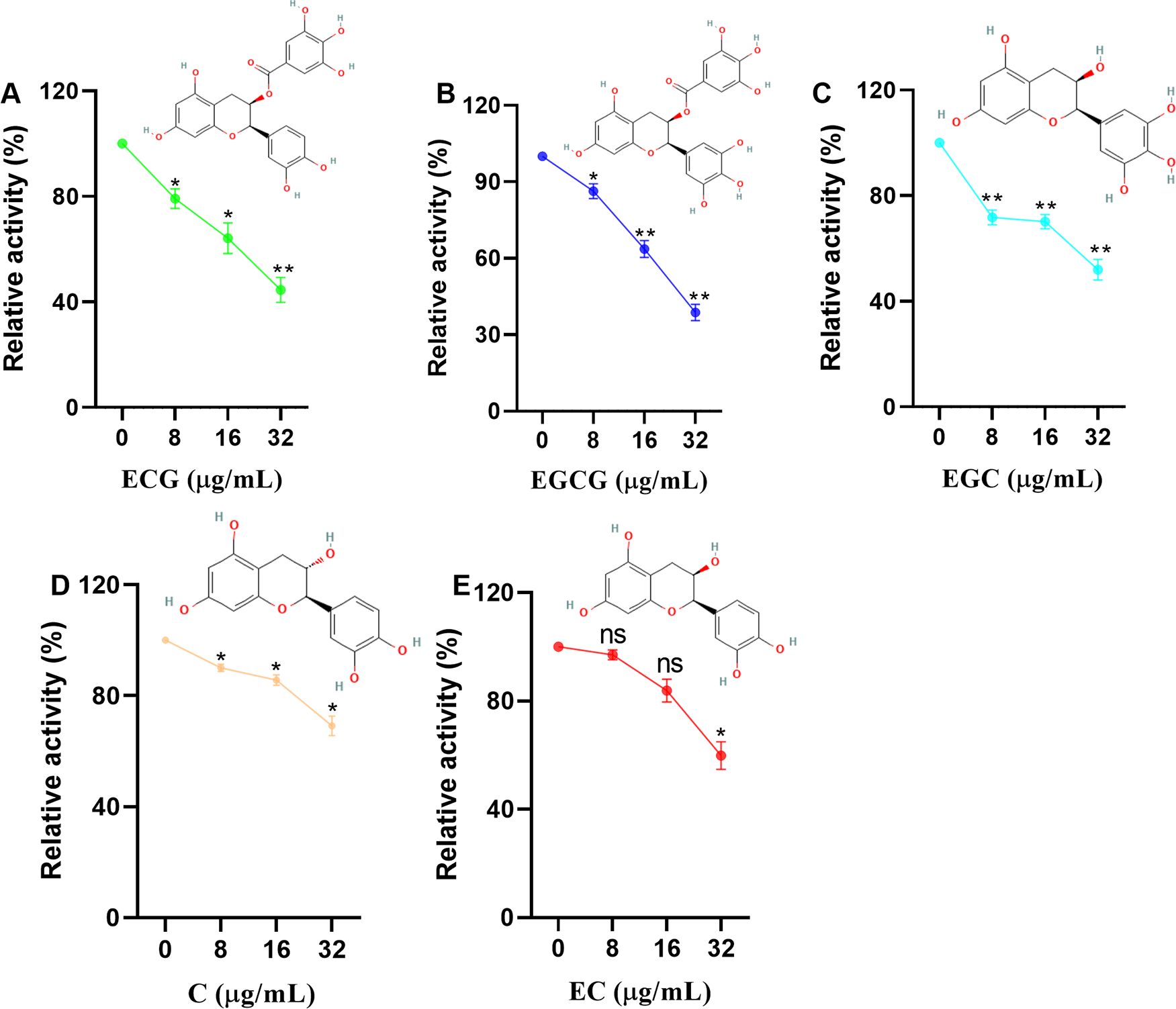
Figure 1. ECG and its analogues inhibit the activity of sortase A. Relative activities of sortase A after treatment with different concentrations of ECG (A), EGCG (B), EGC (C), C (D), and EC (E). The sortase A protein was coincubated with different concentrations of ECG and its analogues at 37°C for 30 min, and then the substrate peptide was added for an additional 1 h of incubation. The fluorescence intensity (excitation 490 nm, emission 520 nm) was measured by using a microplate reader. The relative activity of sortase A was calculated by using the following formula: (S1/S0) × 100, where S1 or S0 represents the fluorescence value of samples treated with or without tested compounds. Data are shown as means with SEMs, n = 3; ns indicates not significant, * indicates p ≤ 0.05, and ** indicates p ≤ 0.01.
ECG and its analogues show excellent affinity for S. aureus sortase A
Molecular docking was carried out, and these compounds were found to bind to the catalytic pocket of sortase A (Figure 2A). The binding energies of ECG, EGCG, EGC, EC, C to sortase A were -7.2, -7.0, -6.7, -6.97 and -6.9 kcal/mol, respectively. Analysis of the sortase A binding site revealed that Thr180, Ser116, Glu108, Asn107 and Asn114 are involved in the binding of EC to sortase A (Figure 2B); that Gln178 and Asn107 of sortase A interact with C (Figure 2C); that EGCG interacts with residues Gln178, Leu169, Asp170 and Asn107 of sortase A (Figure 2D); and that residues Gln178, Asn114, Leu169, Thr180, Asp170 (Figure 2E), Asn107 and Gln178 are involved in the interaction between EGC and sortase A (Figure 2F). These results indicate that the residues involved in the binding of each compound were not exactly the same even though all of the interacting residues were located in the same catalytic pocket.
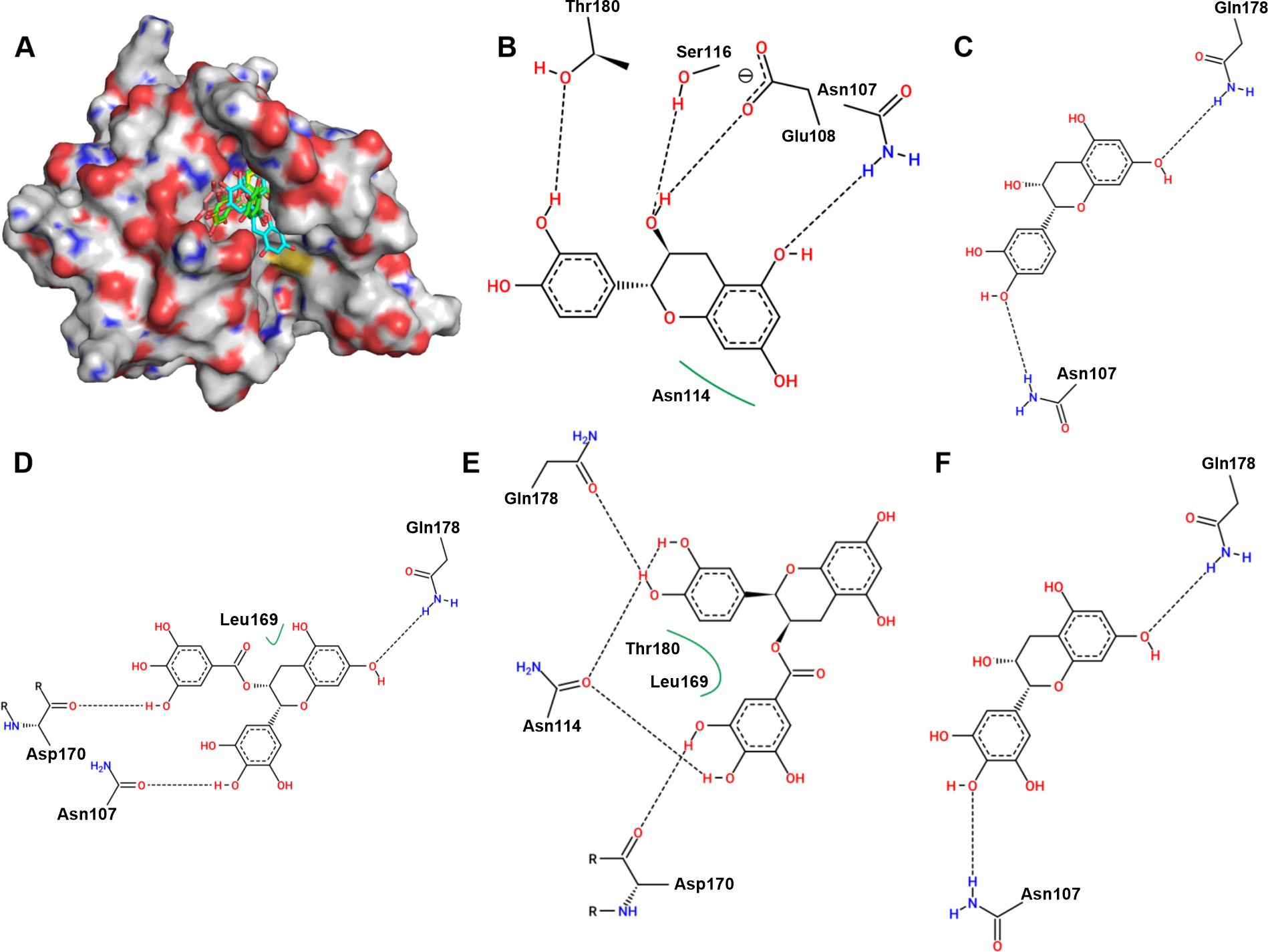
Figure 2. Binding mode of ECG and its analogues to sortase A and analysis of the potential binding sites. (A) Binding mode of ECG and its analogues to sortase A. Potential interaction sites of sortase A with C (B), EC (C), EGCG (D), ECG (E), and EGC (F). Hydrogen atoms and charges were added to sortase A (PDB ID: 6R1V) and ECG and its analogues using AutoDock Tools, and AutoDock Vina was used to perform docking. Potential binding sites were analyzed using Proteins Plus.
Residues Val168, Lys173, and Leu169 are critical for compound binding
The RMSD values of sortase A and ECG fluctuated around 0.38 nm and 0.1 nm, indicating that sortase A and the ECG complex reached an equilibrium state during the molecular dynamics simulations (Figure 3A). Analysis of the binding free energies from the molecular dynamics simulations revealed that electrostatic interactions (ele), van der Waals forces (vdw) and solvation energy (sol) participated in compound binding (Figure 3B). Residue energy decomposition analysis revealed that Val168, Leu169, Ala104, Thr180, Asp165, Ile182, Asp170 and Lys173 have greater contributions to the binding energy (Figure 3C), which was confirmed by the closer proximity of these residues to ECG (Figure 3D). Site-directed mutagenesis was performed to construct the V168A, K173A and L169A mutants, and the inhibitory effects of ECG on these mutants were significantly reduced compared with inhibition of the WT protein (Figure 3E), indicating that these residues are important for ECG and sortase A binding.
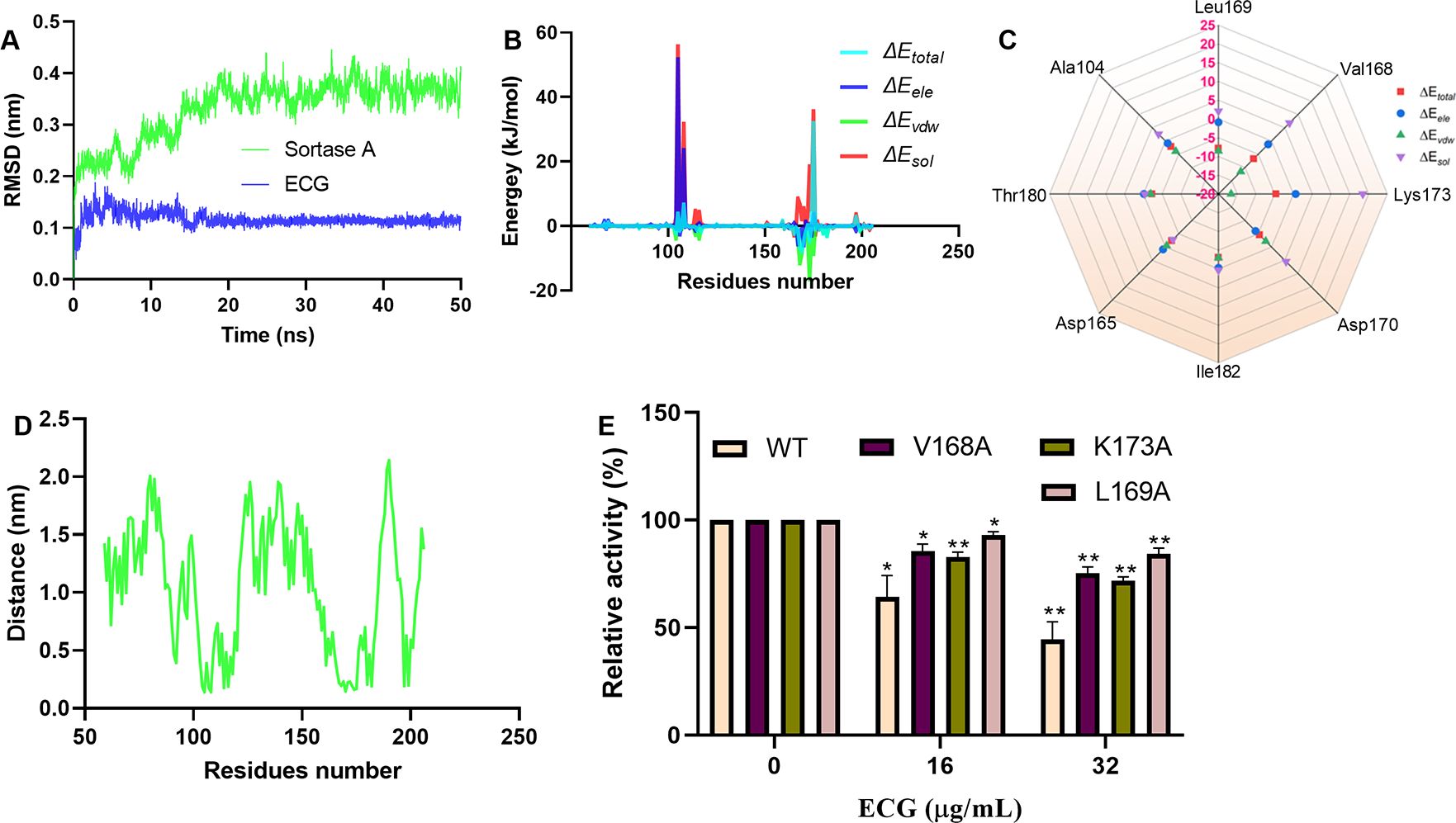
Figure 3. Identification of critical residues involved in ECG binding to sortase A. (A) RMSD values from the simulations. Contribution of each residue to the binding energy (B) and the residues with greater contributions (C). (D) Distances between sortase A residues and ECG. (E) Inhibitory effects of ECG against sortase A and its mutants. GROMACS version 2020.6 was used to perform molecular dynamics simulations using the amber 14SB force field and SPC/E water model. The MMPBSA method was used to calculate the binding free energies. Data are shown as means with SDs, n = 3; * indicates p ≤ 0.05 and ** indicates p ≤ 0.01.
ECG and its analogues inhibit S. aureus USA300 biofilm formation
sortase A is closely related to biofilm formation. Here, we quantitatively analyzed the effects of ECG and its analogues on S. aureus USA300 biofilm formation via crystal violet staining. S. aureus USA300 biofilm formation was defined as 100% in the absence of test compounds. When the bacteria were treated with 8 or 32 µg/mL C, biofilm formation was 103.40% and 85.84%, respectively (Figure 4A). Similarly, biofilm formation was reduced to 43.48% and 34.83% when the bacteria were treated with 8 and 32 µg/mL EGC, respectively (Figure 4B), and these values were 33.83% and 18.44%, respectively, upon treatment with ECG (Figure 4C); 13.26% and 10.92%, respectively, upon treatment with EGCG (Figure 4D); and 82.33% and 44.19%, respectively, upon treatment with EC (Figure 4E). These results showed that ECG and its analogues inhibited S. aureus USA300 biofilm formation to different degrees.
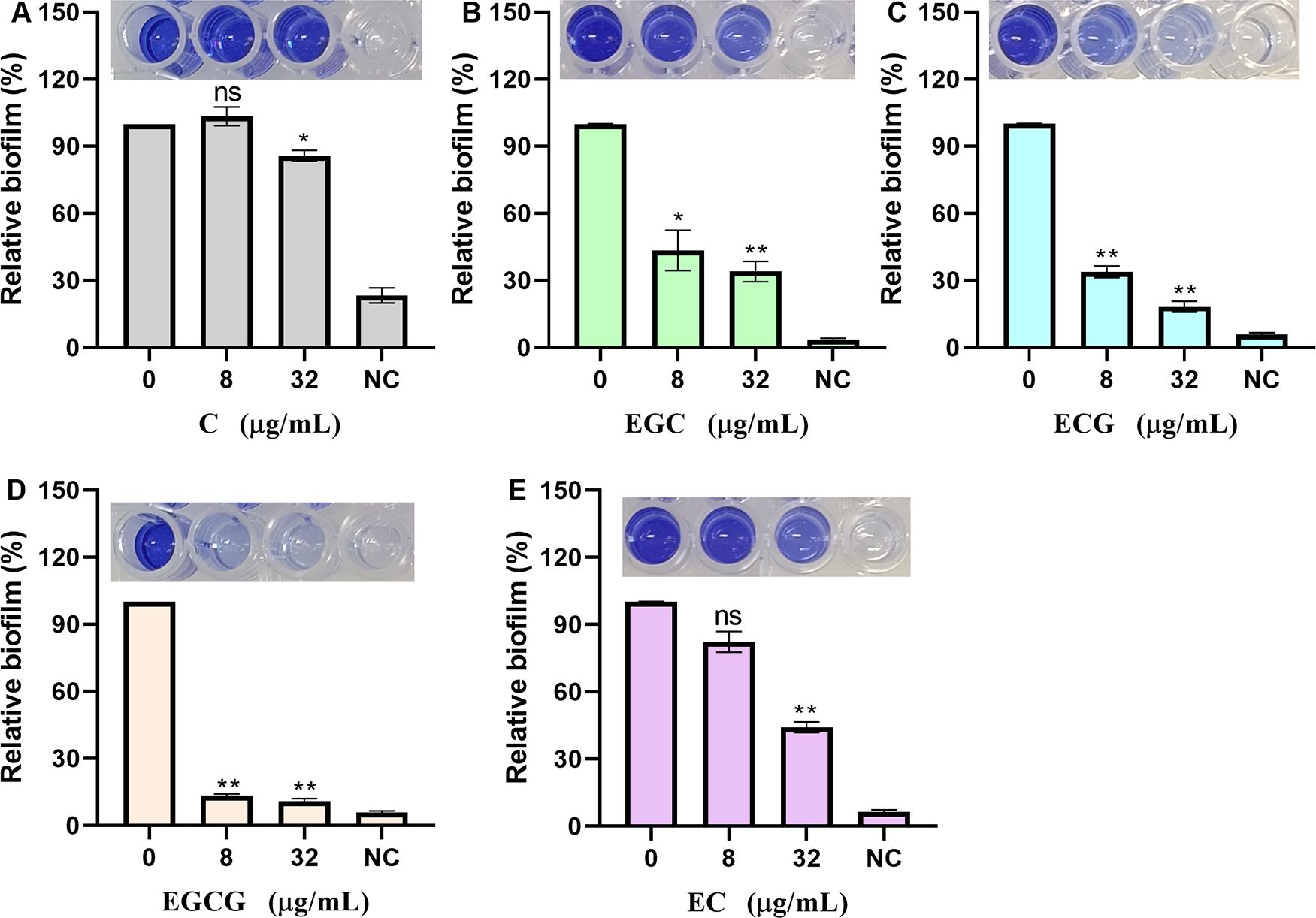
Figure 4. ECG and its analogues inhibit the biofilm formation of S. aureus USA300. Relative biofilm formation of S. aureus USA300 treated with different concentrations of C (A), EGC (B), ECG (C), EGCG (D), and EC (E). S. aureus USA300 was cocultured with different concentrations of ECG and its analogues at 37°C for 24 h, after which the samples were stained with 0.1% crystal violet, washed, and fixed, and the OD570 was finally measured after treatment with 33% glacial acetic acid. NC indicates negative control. Data are shown as means with SEMs, n = 3; ns indicates not significant, * indicates p ≤ 0.05, and ** indicates p ≤ 0.01.
ECG and its analogues reduce the adhesion of S. aureus USA300 to lung epithelial cells
Adhesion to host cells is the first step in S. aureus infection. The virulence factor anchored by sortase A to bacterial cell walls is closely related to bacterial adhesion. Therefore, in this study, the effects of ECG and its analogues on the adhesion of S. aureus USA300 to human lung epithelial cells were analyzed. The number of S. aureus USA300 colonies resulting from the treatment of A549 cells in the absence of test compounds was defined as 100%. When S. aureus USA300-treated A549 cells received 32 µg/mL of the test compounds, the relative adhesion of the bacteria was 54.67% for C (Figure 5A), 14.89% for EGCG (Figure 5B), 46.90% for EGC (Figure 5C), 17.83% for ECG (Figure 5D) and 49.91% for EC (Figure 5E). ECG and EGCG showed significantly better antiadhesion effects than the other compounds, which is consistent with the above results.
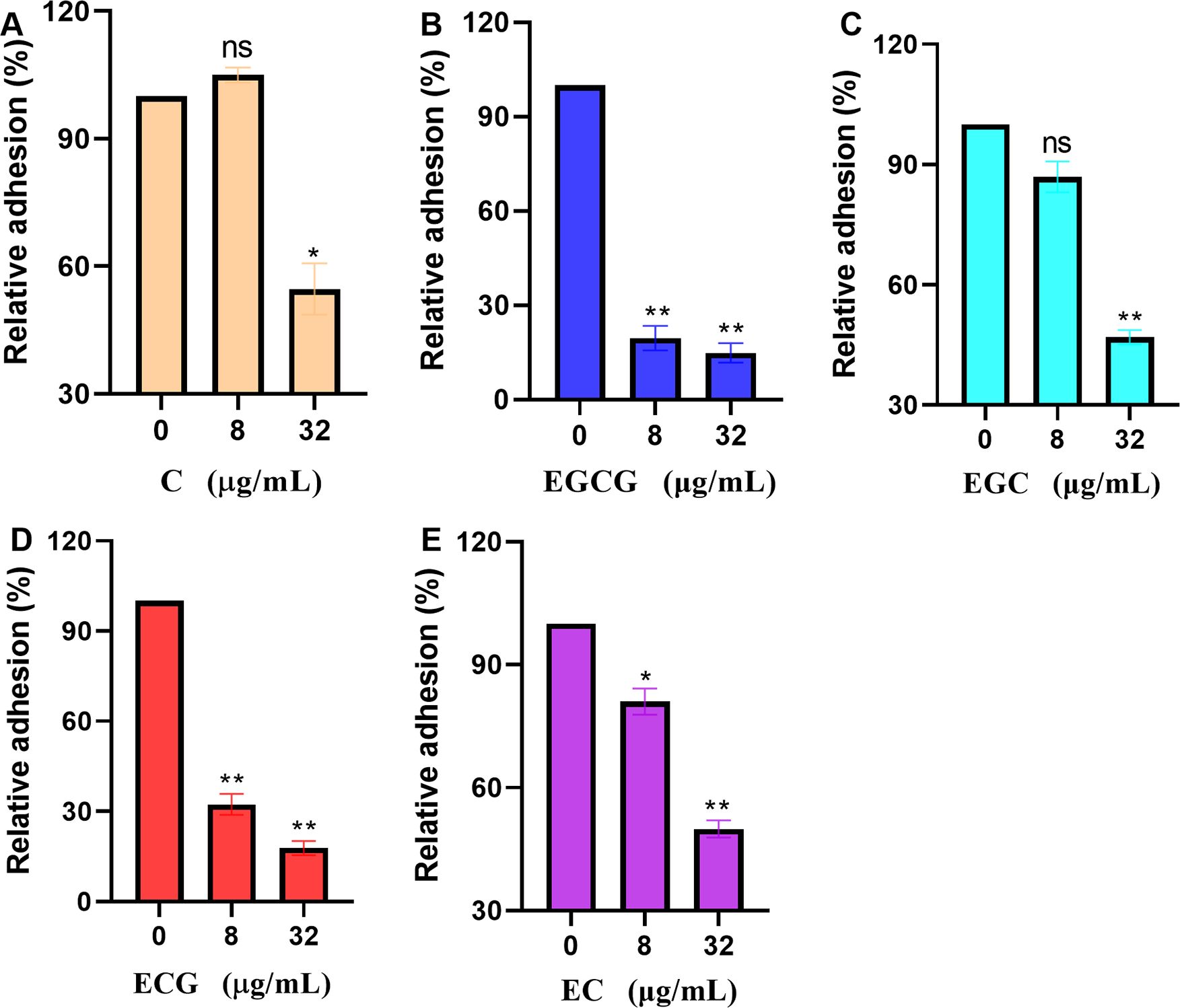
Figure 5. ECG and its analogues reduce the adhesion of S. aureus USA300 to lung epithelial cells. Relative adhesion of S. aureus USA300 to A549 cells after treatment with different concentrations of C (A), EGCG (B), EGC (C), ECG (D), and EC (E). Human lung cancer epithelial A549 cells in 24-well plates (1 × 105 cells/well) were treated with S. aureus USA300 (MOI = 30) and different concentrations of the test compounds for 1 (h). Then, the cells were harvested after discarding the medium, washed with sterile PBS, diluted, plated onto LB agar in equal volumes, and cultured overnight at 37°C. Data are shown as means with SEMs, n = 3; ns indicates not significant, * indicates p ≤ 0.05, and ** indicates p ≤ 0.01.
ECG and its analogues inhibit β-lactamase activity
The β-lactamase secreted by S. aureus can decompose β-lactam antibiotics and induce bacterial tolerance. Nitrocefin is a kind of cephalosporin that is usually used to detect the activity of β-lactamases (Zhou et al., 2020), almost all β-lactamases can hydrolyze the amide bond between the carbonyl carbon and the nitrogen group on the β-lactam ring of nitrocefin, which could be judged by the color changing or detecting the OD492 to analyze the inhibitory effect of inhibitors on β-lactamase activity. In this assay, the samples that were not treated with ECG or its analogues were defined as 100%. When treated with 128 µg/mL test compounds, β-lactamase activity was reduced to 29.01% for ECG (Figure 6A), 40.85% for EGCG (Figure 6B), 73.24% for EGC (Figure 6C), 63.94% for C (Figure 6D) and 69.58% for EC (Figure 6E), indicating that ECG and its analogues inhibited β-lactamase activity.
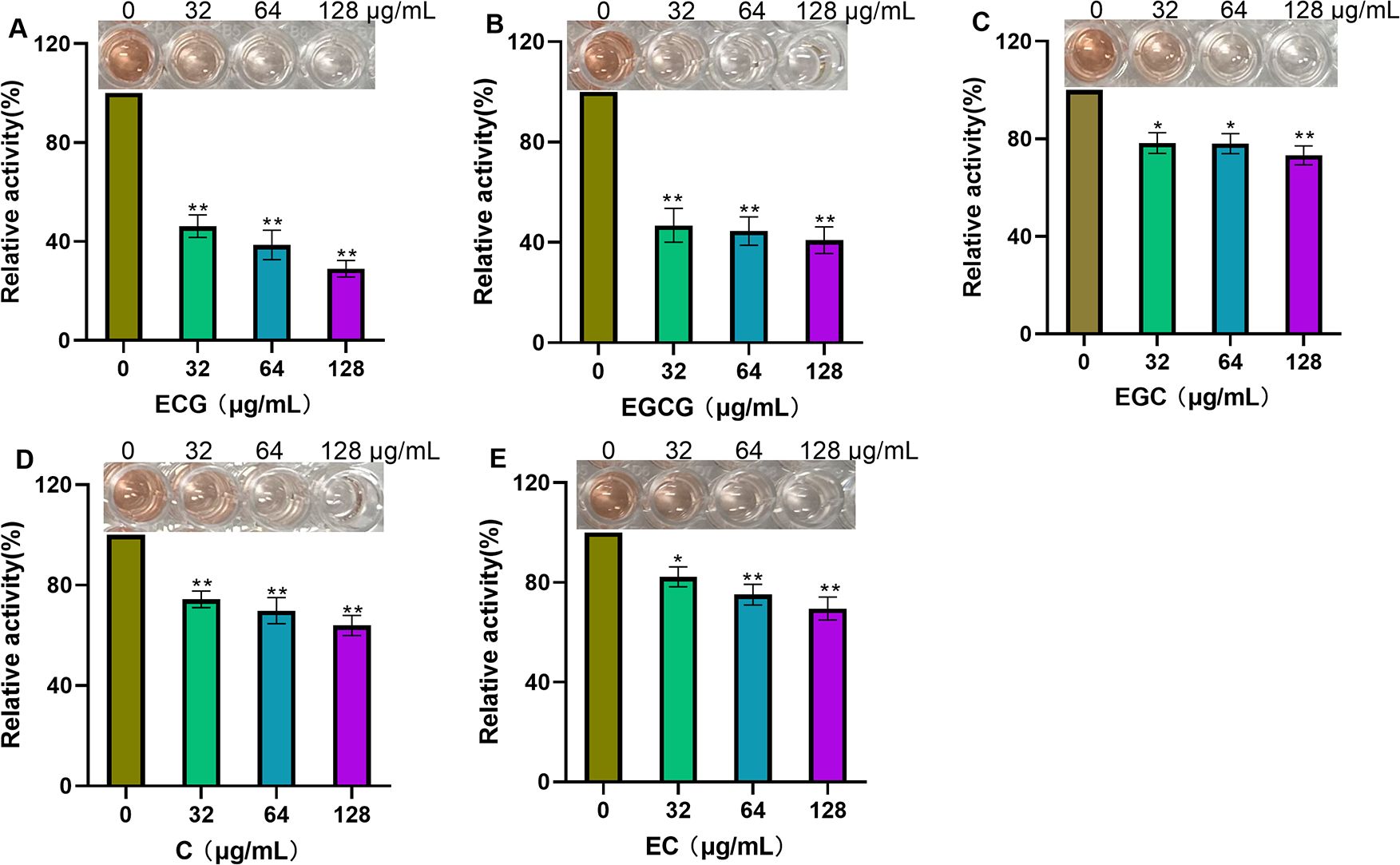
Figure 6. ECG and its analogues inhibit β-lactamase activity. Relative activities of β-lactamase after treatment with different concentrations of ECG (A), EGCG (B), EGC (C), C (D), and EC (E). The β-lactamase protein was treated with various concentrations of the test compounds at 37°C for 30 min, and then nitrocefin was added for an additional 10 min of incubation. The OD492 values were measured to evaluate the inhibitory effects of these compounds on β-lactamase activity. Data are shown as means with SDs, n = 3; * indicates p ≤ 0.05; ** indicates p ≤ 0.01.
ECG enhances the bactericidal ability of Amp and inhibits persister formation synergistically
This study evaluated the effects of ECG and its analogues on the tolerance of S. aureus USA300 to PG, Cef, Amp and kanamycin. When the concentrations of EGC, ECG and EGCG were 4 µg/mL, the MICs of PG and Cef were reduced by 2-, 8- and 2-fold, respectively (Figures 7A, B). The MIC values of Amp against S. aureus USA300 decreased by 16- and 2-fold when it was combined with ECG and EGCG (4 µg/mL), respectively, but cotreatment with EGC (4 µg/mL) did not change the MIC value of Amp (Figure 7C). The MIC value of kanamycin against S. aureus USA300 was greater than 256 µg/mL when kanamycin and ECG, EGCG, or EGC (32 µg/mL) was coadministered to S. aureus USA300 and the MIC value did not change significantly (Table 2), suggesting that these compounds did not synergize with kanamycin. These results indicate that ECG has excellent potential for use in combination with β-lactam antibiotics against infections caused by S. aureus USA300. For the synergistic sterilization assay, S. aureus USA300 showed similar growth trends with and without ECG treatment, but in the Amp and Amp combined with ECG treatment groups, the bacterial density gradually decreased over time. In the combination group, a more rapid downward trend was observed, and bacteria were not detected in the medium after 10 h (Figure 7D). The persister formation assay revealed that the bacterial density in the gentamicin treatment group decreased sharply, suggesting that the bacteria were killed quickly, but the downward trend gradually flattened, indicating that some bacteria survived as persisters; however, bacteria were not detected in the combination group after 2 h of treatment (Figure 7E). Taken together, these results indicate that ECG enhanced the bactericidal ability of Amp and inhibited persister formation synergistically.
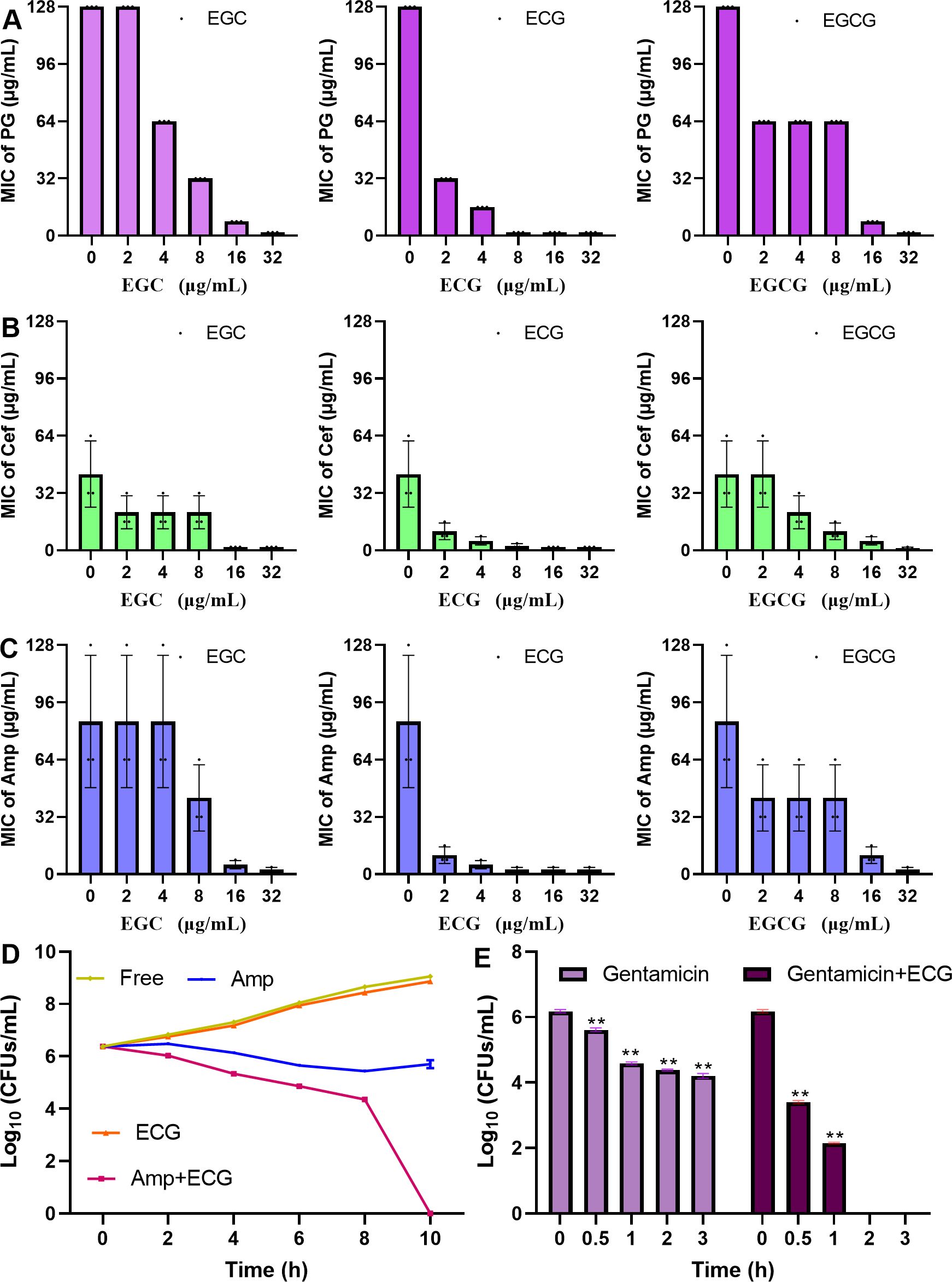
Figure 7. ECG enhances the bactericidal ability of Amp and inhibits persister formation synergistically. Changes in the MICs of PG (A), Cef (B), and Amp (C) upon cotreatment with EGC, ECG, and EGCG. (D) Logarithmic values of S. aureus USA300 density after treatment with ECG, Amp, and their combination. (E) Logarithmic values of S. aureus USA300 density after treatment with gentamicin with or without ECG. The MIC values of the tested antibiotics without or with ECG and its analogues were determined on the basis of CLSI methods. The S. aureus USA300 strain was treated with Amp, ECG, or their combination and then cultured with shaking. Samples were taken at specific time points to determine the number of clones. For the persister assay, S. aureus USA300 cultured overnight was treated with gentamicin with or without ECG, and colony numbers from the different samples were determined at the indicated time points. Data are shown as means with SDs, n = 3; ** indicates p ≤ 0.01.
ECG reduces S. aureus USA300-mediated cytotoxicity but does not affect the expression of its target proteins
S. aureus induces cytotoxic effects that promote the development of infection. In this study, ECG was used to evaluate the cytotoxicity mediated by S. aureus USA300. Red fluorescence was abundant in the S. aureus USA300 treatment group, indicating that most of the cells died, but the intensity of red fluorescence decreased gradually upon treatment with 16 or 32 µg/mL ECG (Figure 8A). In the LDH assay, the amount of LDH secreted by RAW264.7 cells treated with S. aureus USA300 alone was defined as 100%. When 16 or 32 µg/mL ECG was added to the samples, the amount of LDH decreased to 69.14% and 44.48%, respectively (Figure 8B), indicating that ECG significantly alleviated the toxicity mediated by S. aureus USA300 to the cells. The amounts of LDH secreted from cells treated with 32 or 64 µg/mL ECG analogues were similar to that in the DMEM treatment group (Supplementary Table S1), suggesting that these analogues did not exhibit cytotoxicity. Furthermore, the expression levels of sortase A and β-lactamase did not change significantly when the samples were treated with 16 or 32 µg/mL ECG (Figures 8C, D), and the secretion of β-lactamases in the bacterial culture supernatant was similar (Figure 8E), indicating that ECG did not affect the expression or secretion of β-lactamase or sortase A.
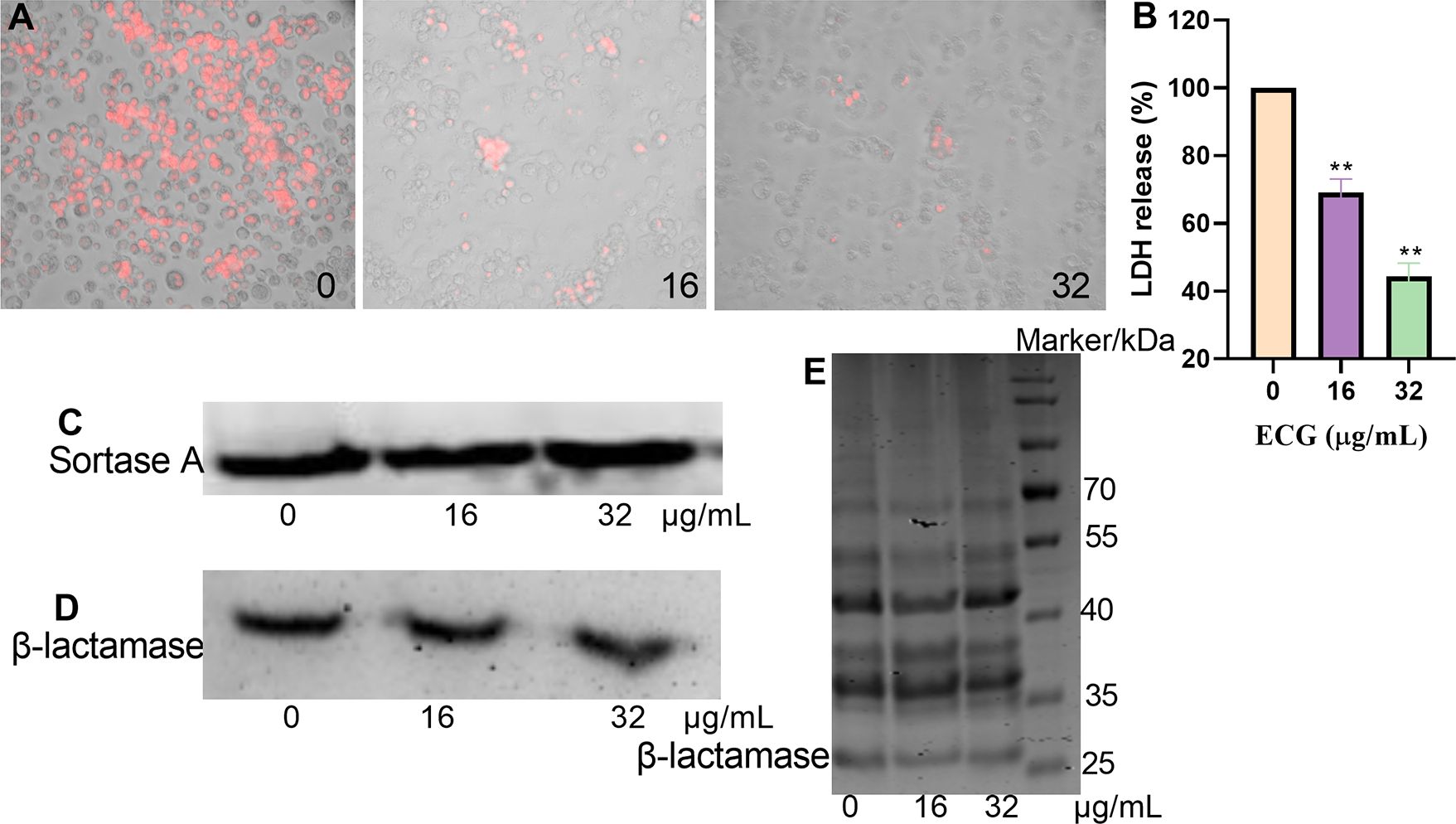
Figure 8. ECG reduces S. aureus USA300-mediated cytotoxicity but does not affect the expression of its target proteins. (A) Cell survival after treatment with different concentrations of ECG detected by EB staining. (B) LDH levels in the cell culture supernatant. RAW264.7 cells were seeded in a 96-well plate (2 × 104 cells/well) and treated with S. aureus USA300 (MOI = 60) with or without ECG for 5 (h). The samples were centrifuged, and the supernatant was obtained. Then, an equal volume of LDH reagent was added to each well, and the mixture was incubated for 20 min in the dark. LDH levels were determined by measuring the OD490. The cells were stained with 50 µg/mL EB to observe their survival status. Data are shown as means with SDs, n = 3; ** indicates p ≤ 0.01. Expression levels of sortase A (C) and β-lactamase (D) and the secretion of β-lactamase into the bacterial culture supernatant (E). S. aureus USA300 was treated with different concentrations of ECG at 37°C with shaking for 8 (h). Following centrifugation, the bacteria and supernatant were obtained, and the expression of sortase A and β-lactamase was measured via western blotting. The secretion of β-lactamase in the supernatant was evaluated via Coomassie brilliant blue staining.
Tyr96, Ile158, and Ile230 of β-lactamase are important for ECG binding
The β-lactamase–ECG complex was determined to be in equilibrium when the RMSD fluctuated around approximately 0.2 and 0.1 nm (Figure 9A). In this complex, ECG was located in the active center of the enzyme (Figure 9B), and binding site analysis revealed that Ile158, Ile230, Ala229, Pro265, Ala95, and Tyr96 of β-lactamase interacted with ECG, and Asn123 and Gln228 of β-lactamase formed H-bonds with ECG (Figure 9C). The binding free energy results revealed that Tyr96, Ile230, and Ile158 contributed more energy to this interaction, and the total free energy contributions of these residues were -8.73, -7.81, and -4.03 kJ/mol, respectively (Figure 9D). To identify key residues involved in ECG binding, site-directed mutagenesis of β-lactamase was performed, and the results revealed that the inhibitory effects of ECG against the Y96A, I158A, and I230A mutants were much lower than that against the WT protein (Figure 9E), suggesting that these residues are critical for ECG binding.
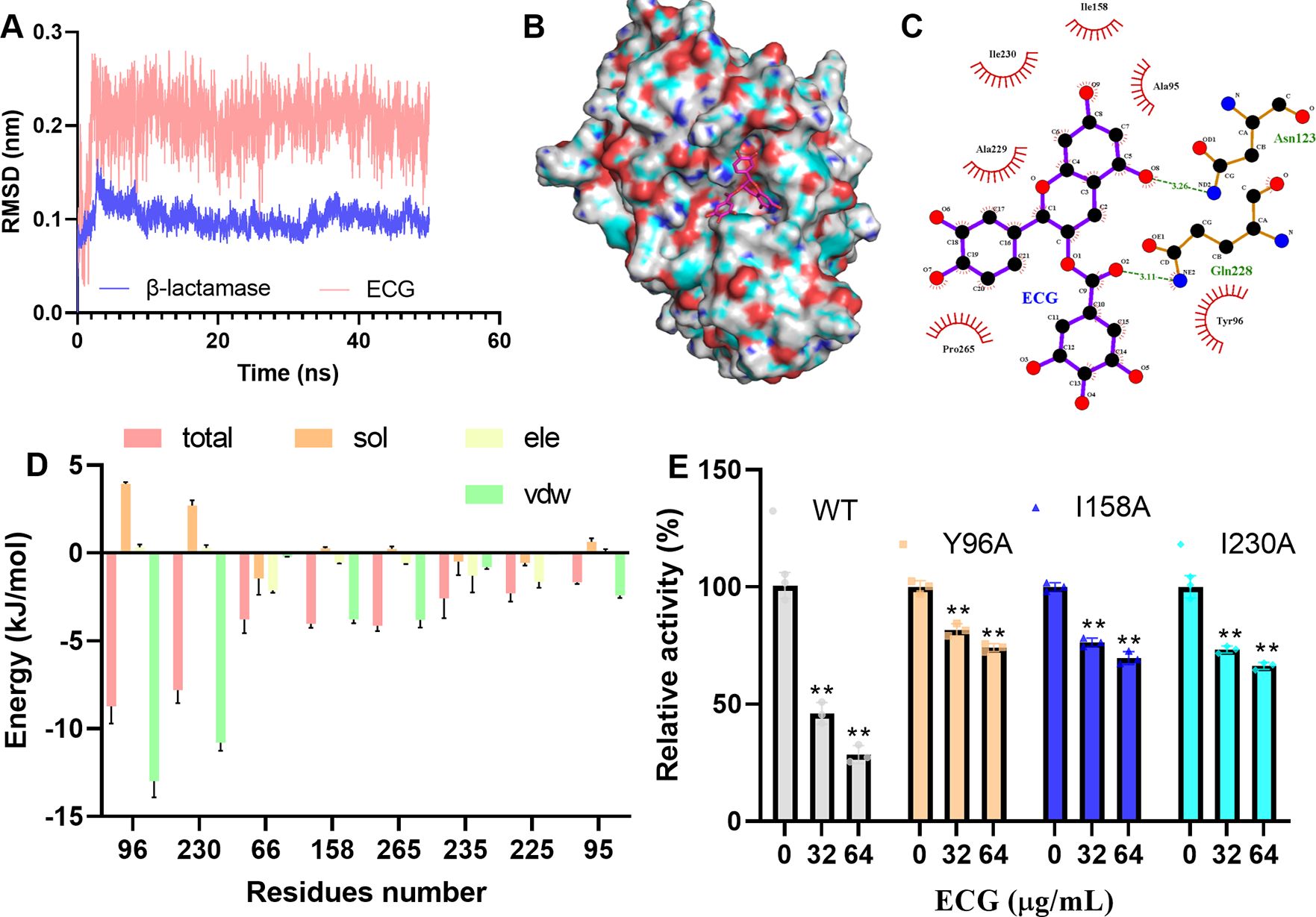
Figure 9. Tyr96, Ile158, and Ile230 are important for ECG binding to β-lactamase. (A) Fluctuations in the RMSDs during the experiment. Binding mode of ECG to β-lactamase (B) and the interacting residues (C). (D) Energy contributions of the residues involved in binding. (E) Activities of β-lactamase and its mutants after treatment with ECG. Molecular simulations were performed by using GROMACS 2020.6, the binding free energy between β-lactamase and ECG was measured via the MMPBSA method, and the interacting residues were analyzed using LigPlus. Data are shown as means with SDs, n = 3; ** indicates p ≤ 0.01.
ECG combined with Amp protects mice from S. aureus USA300 infection
Mice in the PC and Amp treatment groups died after 12 h, and the survival ratios of the two groups were 6.67% and 13.33%, respectively. However, in the ECG and combination groups, the mice died at 24 or 36 h, and the survival ratios reached 46.67% and 76.67%, respectively (Figure 10A). In addition, the number of S. aureus USA300 colonies in the ECG and ECG combined with the Amp treatment groups was significantly lower than that in the PC and Amp treatment groups (Figure 10B). Additionally, the lung tissues from the PC and Amp treatment groups exhibited inflammatory factor infiltration and the alveolar structures were destroyed compared with those in the NC group, whereas these manifestations were alleviated in the samples from the ECG and ECG combined with Amp groups (Figure 10C). Compared with that in the PC group, the wet/dry lung weight ratios from the ECG and ECG combined with Amp treatment groups were significantly lower, but Amp treatment alone did not affect this ratio (Figure 10D). As important indicators of inflammation level, TNF-α and IL-1β are often used to quantitatively evaluate the inflammation level of cells or tissue (Li et al., 2019; Wang et al., 2022). S. aureus infection is usually accompanied by an excessive inflammatory response, accompanied by an excessive release of TNF-α and IL-1β (Ghosh et al., 2023; Ghosh and Bishayi, 2024); the levels of TNF-α and IL-1β here in the samples from the ECG and ECG with Amp groups were significantly lower than those in the other groups, and Amp treatment alone slightly reduced the levels of these cytokines (Figures 10E, F). These results suggest that ECG alone or in combination with Amp could delay mouse death, improve mouse survival, weaken the colonization ability of bacteria, and relieve edema and inflammation in the lungs.
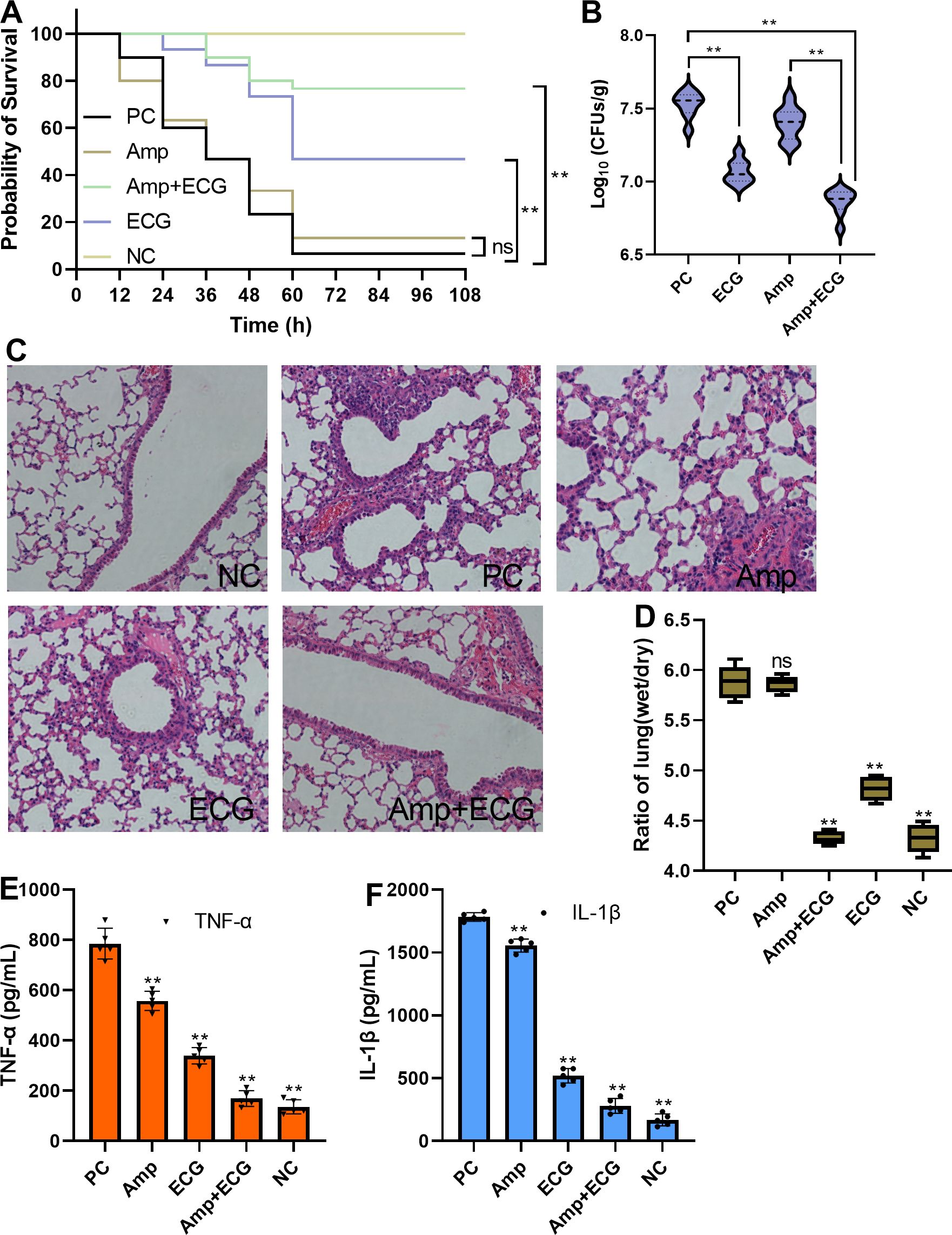
Figure 10. ECG combined with Amp protects mice from S. aureus USA300 infection. (A) Survival of the mice in the different treatment groups, n = 30. (B) Bacterial loads in the lungs of the mice in the different treatment groups, n = 8. (C) Degree of pathological injury in each of the different treatment groups. (D) Lung weight ratio and level of inflammation in the lungs (E, F) in different treatment groups. C57BL/6J mice were intranasally inoculated with S. aureus USA300 (5 × 108 CFUs/mouse) to establish a pneumonia model. The mice were treated with 50 mg/kg ECG, Amp, or their combination every 12 (h). To evaluate the survival rate, the mice were monitored at the specified time points. To evaluate the other indicators, lung tissues were obtained after the mice were euthanized. The bacterial loads of the lung tissues from the different groups were evaluated by analyzing the numbers of clones, and inflammation, pulmonary edema, and tissue damage were assessed by detecting the levels of cytokines, the weight ratio, and HE staining, respectively. Data are shown as means with SDs; n = 5; ns indicates not significant; ** indicates p ≤ 0.01.
Discussion
Sortase A is the most important member of the sortase family and anchors more than 20 different surface proteins to the bacterial cell wall (Geoghegan and Foster, 2017). The biological functions of some of these surface proteins, such as fibronectin-binding proteins that influence adhesion and biofilm formation, have been characterized (Kim et al., 2018; Foster, 2019; Xu et al., 2024). Chalcone has been reported to reduce the pathogenicity of Listeria monocytogenes by inhibiting sortase A activity by occupying the active center of sortase A (Li et al., 2016). Astilbin and trans-chalcone have been shown to inactivate Streptococcus mutans sortase A and reduce their formation of biofilms (Wallock-Richards et al., 2015; Wang et al., 2019). Wogonin was found to attenuate the pathogenicity of Streptococcus pneumoniae by inhibiting pneumolysin and sortase A, but the mechanism has not been elucidated (Gu et al., 2023). Here we found that ECG and its analogues bind directly to S. aureus sortase A and significantly reduce its activity; consequently, the S. aureus biofilm formation and adhesive abilities were inhibited, possibly resulting from the inactivation of sortase A, although other potential mechanisms maybe exist.
The crystal structure of sortase A and its substrate complex was reported in 2004 (PDB ID: 1T2W) (Zong et al., 2004). The substrate LPETG is located in the enzyme’s catalytic center, with its N-terminus located around hydrophobic residues Ile158, Ile199, Val201, Val 168, and Leu169 and its C-terminus pointing to the narrow opening of the active site, wherein the key active site residues His120, Cys184, and Arg197 are positioned around the substrate peptide (Zong et al., 2004). In this study, the crystal structure of sortase A (PDB ID: 6R1V) was used for molecular docking, and it was found that ECG binds to the active pocket and that Leu169, Val168, Lys173, Asp170, Ile182, and Thr180 in sortase A had greater contributions to the binding free energy. Additionally, the inhibitory effects of ECG against the V168A, L169A, and K173A mutants decreased significantly, as these residues were located on the active center of the enzyme.
β-lactam antibiotics have played an important role in combating bacterial infections since the application of penicillin in the treatment of S. aureus infection. Unfortunately, the emergence and diversification of β-lactamases have posed challenges to the control of bacterial infections (Chen and Herzberg, 2001; Mlynarczyk-Bonikowska et al., 2022), limiting the clinical efficacy of all currently available β-lactam antibiotics. Therefore, it is important to identify antibiotic adjuvants, especially natural compounds with no or little antibacterial activity, to improve the sustainability of these types of antibiotics in clinical applications. To date, some natural compounds used as adjuvants have been shown to reduce pathogenic bacterial virulence, such as plumbagin, which binds to the catalytic pocket of monooxygenase to reduce the resistance of tet(X3)/tet(X4)-positive bacteria to tetracycline (Xu et al., 2022); thymol nanoemulsion, which reverses colistin resistance in Salmonella enterica serovar Typhimurium (Sheng et al., 2023); and fisetin, which attenuates meropenem resistance in NDM-1-producing E. coli by targeting metallo-β-lactamases (Guo et al., 2022). This study revealed that ECG and its analogues restored the sensitivity of bacteria to Amp, Cef, and PG by inhibiting β-lactamase activity. ECG improved the bactericidal ability of Amp and inhibited the formation of persisters. ECG alone or in combination with Amp in vivo had a systemic protective effect in the S. aureus pneumonia model. These results suggest that ECG has the potential to be developed as an adjuvant for β-lactam antibiotics in the future to improve the sustainability of antibiotic applications.
O Herzberg reported the crystal structure of the S. aureus β-lactamase (PDB ID: 3BLM), which is composed of two domains, and the catalytic center is located at the interface between the two domains. Many residues are found in the catalytic pocket, including but not limited to Tyr105, Glu166, Leu169, Asn170, Lys73, Ser70, Ile239, Ser130, Lys234, and Tyr165 (Herzberg, 1991). Crystal structures of β-lactamases with their ligands were subsequently reported (PDB IDs: 1BLC and 6WGR), and both ligands were located in the catalytic center (Chen and Herzberg, 1992). Here the β-lactamase crystal structure 6WGR was used to perform molecular docking, and it was found that ECG bound to the catalytic pocket of β-lactamase. The calculated binding free energy and mutagenesis analysis results revealed that Tyr96, Ile158, and Ile230 are important for ECG binding. We aligned the 6WGR and 3BLM structures and found that Tyr96, Ile158, and Ile230 in 6WGR corresponded to Tyr105, Ile167, and Ile239 in 3BLM, which are the constituent residues of the active pocket.
Conclusion
This study revealed that green tea bioactive ingredients, especially ECG, target S. aureus sortase A and β-lactamase simultaneously to alleviate the pathogenicity of S. aureus. By interacting with sortase A, ECG decreased S. aureus biofilm formation and the adhesion of this bacteria to host cells. ECG also bound to β-lactamase to restore the susceptibility of this bacteria to β-lactam antibiotics, improve the bactericidal ability of Amp, and reduce the formation of persisters. ECG alleviated the cytotoxicity mediated by S. aureus USA300 without affecting the expression of sortase A or β-lactamase and protected mice from S. aureus USA300 infection comprehensively. These results support the potential of the development of ECG or its analogues as anti-S. aureus infection agents.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The animal study was approved by Animal Ethics Committee of Jilin University. The study was conducted in accordance with the local legislation and institutional requirements.
Author contributions
FT: Data curation, Investigation, Writing – original draft. LW: Methodology, Writing – original draft, Investigation. JW: Investigation, Visualization, Writing – original draft. ZT: Investigation, Visualization, Writing – original draft. GW: Conceptualization, Funding acquisition, Software, Writing – review & editing. LP: Conceptualization, Funding acquisition, Supervision, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by grant from the National Natural Science Foundation of China (Grant No.81870030 and No. 82070043) and the Scientific Research Project of Jilin province (JJKH20240246KJ).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fcimb.2025.1537564/full#supplementary-material
References
Alharthi, S., Alavi, S. E., Moyle, P. M., Ziora, Z. M. (2021). Sortase A (SrtA) inhibitors as an alternative treatment for superbug infections. Drug Discovery Today 26, 2164–2172. doi: 10.1016/j.drudis.2021.03.019
Bozhkova, S. A., Gordina, E. M., Labutin, D. V., Kudryavtsev, K. V. (2022). Oligopeptide sortase inhibitor modulates staphylococcus aureus cell adhesion and biofilm formation. Antibiotics (Basel) 11, 1836. doi: 10.3390/antibiotics11121836
Cascioferro, S., Raffa, D., Maggio, B., Raimondi, M. V., Schillaci, D., Daidone, G. (2015). Sortase A inhibitors: recent advances and future perspectives. J. Med. Chem. 58, 9108–9123. doi: 10.1021/acs.jmedchem.5b00779
Cascioferro, S., Totsika, M., Schillaci, D. (2014). Sortase A: an ideal target for anti-virulence drug development. Microb. Pathog. 77, 105–112. doi: 10.1016/j.micpath.2014.10.007
Chen, G., Cheng, K., Niu, Y., Zhu, L., Wang, X. (2022). (-)-Epicatechin gallate prevents inflammatory response in hypoxia-activated microglia and cerebral edema by inhibiting NF-κB signaling. Arch. Biochem. Biophys. 729, 109393. doi: 10.1016/j.abb.2022.109393
Chen, L., Guo, Y., Wu, Z., Zhao, S., Zhang, Z., Zheng, F., et al. (2021). Epicatechin gallate prevents the de novo synthesis of fatty acid and the migration of prostate cancer cells. Acta Biochim. Biophys. Sin. (Shanghai) 53, 1662–1669. doi: 10.1093/abbs/gmab144
Chen, C. C., Herzberg, O. (1992). Inhibition of beta-lactamase by clavulanate. Trapped intermediates in cryocrystallographic studies. J. Mol. Biol. 224, 1103–1113. doi: 10.1016/0022-2836(92)90472-V
Chen, C. C., Herzberg, O. (2001). Structures of the acyl-enzyme complexes of the Staphylococcus aureus beta-lactamase mutant Glu166Asp: Asn170Gln with benzylpenicillin and cephaloridine. Biochemistry 40, 2351–2358. doi: 10.1021/bi002277h
Foster, T. J. (2019). Surface proteins of staphylococcus aureus. Microbiol. Spectr. 7. doi: 10.1128/microbiolspec.GPP3-0046-2018
Foster, T. J., Geoghegan, J. A., Ganesh, V. K., Höök, M. (2014). Adhesion, invasion and evasion: the many functions of the surface proteins of Staphylococcus aureus. Nat. Rev. Microbiol. 12, 49–62. doi: 10.1038/nrmicro3161
Gao, Y., Wang, G., Wang, X., Yang, Y., Niu, X. (2020). Structure-Activity relationship of MDSA and its derivatives against Staphylococcus aureus Ser/Thr phosphatase Stp1. Comput. Biol. Chem. 85, 107230. doi: 10.1016/j.compbiolchem.2020.107230
Geoghegan, J. A., Foster, T. J. (2017). Cell wall-anchored surface proteins of staphylococcus aureus: many proteins, multiple functions. Curr. Top. Microbiol. Immunol. 409, 95–120. doi: 10.1016/j.tim.2017.11.008
George, C. R. R., Lahra, M. M., Nguyen, T., Gatus, B. (2023). Disc test for detecting staphylococcus aureus strains producing type A and type C β-lactamases. Microbiol. Spectr. 11, e0022023. doi: 10.1128/spectrum.00220-23
Ghosh, R., Bishayi, B. (2024). Endogenous blocking of TLR2 along with TNF-α and IL-1β ameliorates the severity of the S. aureus arthritis via modulating STAT3/SOCS3 expressions in tissue resident macrophages. Microb. Pathog. 187, 106518. doi: 10.1016/j.intimp.2024.112153
Ghosh, S., Ghosh, R., Sawoo, R., Dutta, P., Bishayi, B. (2023). Impact of dual neutralization of TNF-α and IL-1β along with Gentamicin treatment on the functions of blood and splenic neutrophils and its role on improvement of S. aureus induced septic arthritis. Int. Immunopharmacol 123, 110766. doi: 10.1016/j.intimp.2023.110766
Grossman, A. B., Burgin, D. J., Rice, K. C. (2021). Quantification of staphylococcus aureus biofilm formation by crystal violet and confocal microscopy. Methods Mol. Biol. 2341, 69–78. doi: 10.1007/978-1-0716-1550-8_9
Gu, K., Ding, L., Wang, Z., Sun, Y., Sun, X., Yang, W., et al. (2023). Wogonin attenuates the pathogenicity of Streptococcus pneumoniae by double-target inhibition of Pneumolysin and Sortase A. J. Cell Mol. Med. 27, 563–575. doi: 10.1111/jcmm.17684
Guo, Y., Liu, H., Yang, M., Ding, R., Gao, Y., Niu, X., et al. (2024). Novel metallo-β-lactamases inhibitors restore the susceptibility of carbapenems to New Delhi metallo-lactamase-1 (NDM-1)-harbouring bacteria. Br. J. Pharmacol. 181, 54–69. doi: 10.1111/bph.v181.1
Guo, Y., Yang, Y., Xu, X., Li, L., Zhou, Y., Jia, G., et al. (2022). Metallo-β-lactamases inhibitor fisetin attenuates meropenem resistance in NDM-1-producing Escherichia coli. Eur. J. Med. Chem. 231, 114108. doi: 10.1016/j.ejmech.2022.114108
Herzberg, O. (1991). Refined crystal structure of beta-lactamase from Staphylococcus aureus PC1 at 2.0 A resolution. J. Mol. Biol. 217, 701–719. doi: 10.1016/0022-2836(91)90527-D
Ide, K., Kawasaki, Y., Kawakami, K., Yamada, H. (2016). Anti-influenza virus effects of catechins: A molecular and clinical review. Curr. Med. Chem. 23, 4773–4783. doi: 10.2174/0929867324666161123091010
Jiang, T., Yuan, D., Wang, R., Zhao, C., Xu, Y., Liu, Y., et al. (2023). EChinacoside, a promising sortase A inhibitor, combined with vancomycin against murine models of MRSA-induced pneumonia. Med. Microbiol. Immunol. 212, 421–435. doi: 10.1007/s00430-023-00782-9
Jonsson, I. M., Mazmanian, S. K., Schneewind, O., Bremell, T., Tarkowski, A. (2003). The role of Staphylococcus aureus sortase A and sortase B in murine arthritis. Microbes Infect. 5, 775–780. doi: 10.1016/S1286-4579(03)00143-6
Keren, I., Kaldalu, N., Spoering, A., Wang, Y., Lewis, K. (2004). Persister cells and tolerance to antimicrobials. FEMS Microbiol. Lett. 230, 13–18. doi: 10.1016/S0378-1097(03)00856-5
Kim, S. J., Chang, J., Rimal, B., Yang, H., Schaefer, J. (2018). Surface proteins and the formation of biofilms by Staphylococcus aureus. Biochim. Biophys. Acta Biomembr 1860, 749–756. doi: 10.1016/j.bbamem.2017.12.003
Kochman, J., Jakubczyk, K., Antoniewicz, J., Mruk, H., Janda, K. (2020). Health benefits and chemical composition of matcha green tea: A review. Molecules 26. doi: 10.3390/molecules26010085
Li, H., Chen, Y., Zhang, B., Niu, X., Song, M., Luo, Z., et al. (2016). Inhibition of sortase A by chalcone prevents Listeria monocytogenes infection. Biochem. Pharmacol. 106, 19–29. doi: 10.1016/j.bcp.2016.01.018
Li, G., Shen, X., Wei, Y., Si, X., Deng, X., Wang, J. (2019). Quercetin reduces Streptococcus suis virulence by inhibiting suilysin activity and inflammation. Int. Immunopharmacol 69, 71–78. doi: 10.1016/j.intimp.2019.01.017
Liu, B., Chen, F., Bi, C., Wang, L., Zhong, X., Cai, H., et al. (2015). Quercitrin, an inhibitor of Sortase A, interferes with the adhesion of Staphylococcal aureus. Molecules 20, 6533–6543. doi: 10.3390/molecules20046533
Liu, T. T., Yang, T., Gao, M. N., Chen, K. X., Yang, S., Yu, K. Q., et al. (2019). The inhibitory mechanism of aurintricarboxylic acid targeting serine/threonine phosphatase Stp1 in Staphylococcus aureus: insights from molecular dynamics simulations [J. Acta Pharmacol. Sin. 40, 850–858. doi: 10.1038/s41401-019-0216-x
Liu, S., Zhang, J., Zhou, Y., et al. (2019). Pterostilbene restores carbapenem susceptibility in New Delhi metallo-β-lactamase-producing isolates by inhibiting the activity of New Delhi metallo-β-lactamases. Br. J. Pharmacol. 176, 4548–4557. doi: 10.1111/bph.v176.23
Mlynarczyk-Bonikowska, B., Kowalewski, C., Krolak-Ulinska, A., et al. (2022). Molecular mechanisms of drug resistance in staphylococcus aureus. Int. J. Mol. Sci. 23. doi: 10.3390/ijms23158088
Mossman, A. K., Svishchuk, J., Waddell, B. J. M., et al. (2022). Staphylococcus aureus in non-cystic fibrosis bronchiectasis: prevalence and genomic basis of high inoculum β-lactam resistance. Ann. Am. Thorac. Soc. 19, 1285–1293. doi: 10.1513/AnnalsATS.202108-965OC
Musial, C., Kuban-Jankowska, A., Gorska-Ponikowska, M. (2020). Beneficial properties of green tea catechins. Int. J. Mol. Sci. 21. doi: 10.3390/ijms21051744
Ronner, A. C., Curtin, J., Karami, N., et al. (2014). Adhesion of meticillin-resistant Staphylococcus aureus to DACC-coated dressings. J. Wound Care 23, 484, 6–484, 8. doi: 10.12968/jowc.2014.23.10.484
Sheng, Q., Wang, N., Zhou, Y., et al. (2023). A new function of thymol nanoemulsion for reversing colistin resistance in Salmonella enterica serovar Typhimurium infection. J. Antimicrob. Chemother. 78, 2983–2994. doi: 10.1093/jac/dkad342
Shulga, D. A., Kudryavtsev, K. V. (2022). Theoretical studies of leu-pro-arg-asp-ala pentapeptide (LPRDA) binding to sortase A of staphylococcus aureus. Molecules 27. doi: 10.3390/molecules27238182
Song, M., Teng, Z., Li, M., et al. (2017). Epigallocatechin gallate inhibits Streptococcus pneumoniae virulence by simultaneously targeting pneumolysin and sortase A. J. Cell Mol. Med. 21, 2586–2598. doi: 10.1111/jcmm.2017.21.issue-10
Song, W., Wang, L., Zhao, Y., et al. (2022). Hibifolin, a natural sortase A inhibitor, attenuates the pathogenicity of staphylococcus aureus and enhances the antibacterial activity of cefotaxime. Microbiol. Spectr. 10, e0095022. doi: 10.1128/spectrum.00950-22
Su, X., Yu, H., Wang, X., et al. (2022). Cyanidin chloride protects mice from methicillin-resistant Staphylococcus aureus-induced pneumonia by targeting Sortase A. Virulence 13, 1434–1445. doi: 10.1080/21505594.2022.2112831
Trott, O., Olson, A. J. (2010). AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J. Comput. Chem. 31, 455–461. doi: 10.1002/jcc.21334
Wallock-Richards, D. J., Marles-Wright, J., Clarke, D. J., et al. (2015). Molecular basis of Streptococcus mutans sortase A inhibition by the flavonoid natural product trans-chalcone. Chem. Commun. (Camb) 51, 10483–10485. doi: 10.1039/C5CC01816A
Wang, T., Liu, B., Zhang, C., et al. (2022). Kaempferol-driven inhibition of listeriolysin O pore formation and inflammation suppresses listeria monocytogenes infection. Microbiol. Spectr. 10, e0181022. doi: 10.1128/spectrum.01810-22
Wang, J., Shi, Y., Jing, S., et al. (2019). Astilbin inhibits the activity of sortase A from streptococcus mutans. Molecules 24. doi: 10.3390/molecules24030465
Weiss, W. J., Lenoy, E., Murphy, T., et al. (2004). Effect of srtA and srtB gene expression on the virulence of Staphylococcus aureus in animal models of infection. J. Antimicrob. Chemother. 53, 480–486. doi: 10.1093/jac/dkh078
Xu, Z., Li, Y., Xu, A., et al. (2024). Cell-wall-anchored proteins affect invasive host colonization and biofilm formation in Staphylococcus aureus. Microbiol. Res. 285. doi: 10.1016/j.micres.2024.127782
Xu, L., Zhou, Y., Niu, S., et al. (2022). A novel inhibitor of monooxygenase reversed the activity of tetracyclines against tet(X3)/tet(X4)-positive bacteria. EBioMedicine 78, 103943. doi: 10.1016/j.ebiom.2022.103943
Zhou, Y., Guo, Y., Sun, X., et al. (2020). Application of oleanolic acid and its analogues in combating pathogenic bacteria in vitro/vivo by a two-pronged strategy of β-lactamases and hemolysins. ACS Omega 5, 11424–11438. doi: 10.1021/acsomega.0c00460
Keywords: ECG, sortase A, β-lactamase, biofilm, persisters, Staphylococcus aureus
Citation: Teng F, Wang L, Wen J, Tian Z, Wang G and Peng L (2025) Epicatechin gallate and its analogues interact with sortase A and β-lactamase to suppress Staphylococcus aureus virulence. Front. Cell. Infect. Microbiol. 15:1537564. doi: 10.3389/fcimb.2025.1537564
Received: 01 December 2024; Accepted: 28 February 2025;
Published: 25 March 2025.
Edited by:
Sara María Soto, Instituto Salud Global Barcelona (ISGlobal), SpainReviewed by:
Mei-Hui Lin, Chang Gung University, TaiwanCurutiu Carmen, University of Bucharest, Romania
Copyright © 2025 Teng, Wang, Wen, Tian, Wang and Peng. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Guizhen Wang, d2FuZ2d6QGpsZW51LmVkdS5jbg==; Liping Peng, cGVuZ2xwQGpsdS5lZHUuY24=
†These authors have contributed equally to this work
 Fei Teng
Fei Teng Lihui Wang
Lihui Wang Jingyao Wen
Jingyao Wen Zizeng Tian
Zizeng Tian Guizhen Wang
Guizhen Wang Liping Peng
Liping Peng