
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Cell. Infect. Microbiol. , 01 March 2023
Sec. Antibiotic Resistance and New Antimicrobial drugs
Volume 13 - 2023 | https://doi.org/10.3389/fcimb.2023.1130333
This article is part of the Research Topic Molecular mechanisms of resistance to "last resort" antimicrobials in Enterobacterales View all 10 articles
 Qin Li1†
Qin Li1† Qiao Chen1†
Qiao Chen1† Shuang Liang2
Shuang Liang2 Wei Wang1
Wei Wang1 Bingying Zhang1
Bingying Zhang1 Alberto J. Martín-Rodríguez3
Alberto J. Martín-Rodríguez3 Qinghua Liang1
Qinghua Liang1 Feiyang Zhang1
Feiyang Zhang1 Ling Guo2
Ling Guo2 Xia Xiong4
Xia Xiong4 Renjing Hu5*
Renjing Hu5* Li Xiang1*
Li Xiang1* Yingshun Zhou1,6*
Yingshun Zhou1,6*Introduction: Tigecycline and carbapenems are considered the last line of defense against microbial infections. The co-occurrence of resistance genes conferring resistance to both tigecycline and carbapenems in Pseudomononas asiatica was not investigated.
Methods: P. asiatica A28 was isolated from hospital sewage. Antibiotic susceptibility testing showed resistance to carbapenem and tigecycline. WGS was performed to analyze the antimicrobial resistance genes and genetic characteristics. Plasmid transfer by conjugation was investigated. Plasmid fitness costs were evaluated in Pseudomonas aeruginosa transconjugants including a Galleria mellonella infection model.
Results: Meropenem and tigecycline resistant P. asiatica A28 carries a 199, 972 bp long plasmid PLA28.4 which harbors seven resistance genes. Sequence analysis showed that the 7113 bp transposon Tn7389 is made up of a class I integron without a 5’CS terminal and a complete tni module flanked by a pair of 25bp insertion repeats. Additionally, the Tn7493 transposon, 20.24 kp long, with a complete 38-bp Tn1403 IR and an incomplete 30-bp Tn1403 IR, is made up of partial skeleton of Tn1403, a class I integron harboring blaOXA-10, and a Tn5563a transposon. Moreover, one tnfxB3-tmexC3.2-tmexD3b-toprJ1b cluster was found in the plasmid and another one in the the chromosome. Furthermore, plasmid PLA28.4 could be conjugated to P. aeruginosa PAO1, with high fitness cost.
Discussion: A multidrug-resistant plasmid carrying tmexCD3-toprJ1b and two novel transposons carrying blaVIM-2 and blaOXA-10 -resistant genes was found in hospital sewage, increasing the risk of transmission of antibiotic-resistant genes. These finding highlight the necessary of controlling the development and spread of medication resistance requires continuous monitoring and management of resistant microorganisms in hospital sewage.
Tigecycline is one of the last lines of defense against carbapenem-resistant bacterial infections (Aghapour et al., 2019; Jo and Ko, 2021). The Resistance-Nodulation-Division (RND) MDR efflux pump gene cluster tmexCD1-toprJ1 or the variants such as the tnfxB3-tmexC3.2-tmexD3b-toprJ1b is one of the mechanisms which mediates the tigecycline resistance. Additionally, metallo-β-lactamases (MBLs) and carbapenemase coding genes like blaKPC are the main mechanisms mediating carbapenem resistance (Hu et al., 2021; Huang et al., 2022). Emergence of tigecycline and carbapenem resistant bacteria such as E. coli, Klebsiella spp. and the Pseudomonas spp. from the patients poses great challenges to infection control (Lv et al., 2020; Wang et al., 2021a; Wang et al., 2021b; Gao et al., 2022; Li et al., 2022).
The genes encoding resistance determinants such as MBLs are usually found in plasmids or are associated with integrons and transposons (Mann et al., 2022). Integrons are able to capture genes that are part of gene cassettes via a site-specific recombination event and transposons contribute significantly to the transfer and transmission of antibiotic resistance (AR) in bacterial populations (Alavi et al., 2011; Mann et al., 2022). It is commonly believed that the hospital sewage provides a significant platform for the generation of new transposons and many of the novel transposons have been reported from the sewage. Acinetobacter johnsonii M19 isolated from hospital sewage carries a novel transposon Tn6618 containing carbapenem resistant gene blaOXA-23, while Shewanella xiamenensis T17 carries the novel transposon Tn6297 encoding OXA-416 (Yousfi et al., 2017; Zong et al., 2020).
P. asiatica, a newly proposed unique species of the genus Pseudomonas, belongs to the Pseudomonas putida group, which is a potential human pathogen that can cause nosocomial illness (Tohya et al., 2019a). Moreover, the most prevalent carbapenem resistance gene in the genome of clinical isolates of P. aeruginosa is the blaVIM-2 Metal -β -lactamase (MβL) gene, which is usually present in part of the cassette repertoire of class 1 integrons/transposons (Botelho et al., 2018). The blaVIM-2 gene has been found in P. asiatica (Brovedan et al., 2021; Tohya et al., 2021), indicating that it is an important reservoir of this gene.
Here, we describe a novel plasmid that co-harbors the tigecycline association resistance gene tmexCD3-toprJ1b, a blaVIM-2-carrying novel transposon Tn7389, as well as blaOXA-10-carrying novel transposon Tn7493 from a Pseudomonas asiatica strain.
Wastewater samples were collected from a large tertiary hospital in Luzhou in August 2019. The sewage samples were collected from outflow of the sewage treatment stations of hospital. The samples were collected in sterile glass bottles (200ml) at a set time each time. Sewage samples were mixed and diluted with sterile water in a ratio of 1:10 and subsequently inoculated on a MacConkey agar plate at 37°C for 18-24h in the presence of antibiotics: meropenem (0.5 mg/L). One strain, named A28, was isolated and purified three times on Luria-Bertani (LB) broth agar medium following the repeated plate streaking method. The species was identified by detecting the 16S rRNA gene with universal primers 27F (5′-AGA GTT TGA TYM TGG CTC AG-3′) and 1492R (5′-GGY TAC CTT GTT ACG ACT T-3′), and further confirmed by WGS analysis (Smyth et al., 2020).
The minimal inhibitory concentrations (MICs) of A28 to antimicrobial agents were determined by broth microdilution method according to the recommendations of the CLSI 2021 breakpoints. Escherichia coli strain ATCC 25922 was used as quality control.
The whole genome of strain A28 isolate was sequenced using Oxford Nanopore Technologies. The species was identified using JSpecies (http://jspecies.ribohost.com/jspeciesws/#analyse). ARGs were identified using ResFinder v.4.1 (https://cge.cbs.dtu.dk/services/ResFinder). MLST (Multi-Locus Sequence Typing) v.2.0 (https://cge.cbs.dtu.dk/services/MLST/) was used to determine the STs of the strain. RAST server v.2.0 (https://rast.nmpdr.org/rast.cgi) was used for genome annotation. The circular map of plasmids was generated using the BLAST Ring Image Generator (BRIG) tool and compared to highly similar plasmids in the NCBI database. The Transposon Registry assigned a name to the novel transposon (https://transposon.lstmed.ac.uk/).
Conjugation assays were carried out using sodium azide-resistant E. coli J53, rifampicin-resistant E. coli EC600 (Rifr), and rifampicin-resistant P. aeruginosa PAO1 as recipients. Transconjugants were selected on LB agar plates containing meropenem (0.5 mg/L) and sodium azide (100 mg/L) or rifampicin (100 mg/L). The donor and recipient strains were mixed in ratios of 1:1, then cultured overnight on LB agar plates at 37°C.The resistance genes of blaVIM-2 in transconjugants were validated by PCR. A growth curve assay was used to calculate the fitness of the plasmid between P. aeruginosa transconjugants and P. aeruginosa PAO1 (Zhang et al., 2022). Overnight cultures were diluted 1:50 in LB without antibiotics and measured at OD600 every 15 minutes for 11 hours on a Synergy H1 (Labsystems) instrument, with each sample repeated three times. Student’s t-test was used for statistical analysis, with a significance threshold of 95% (P< 0.05).
The ability of the transconjugant and wild-type strain to generate biofilms was determined using crystal violet staining (Ding et al., 2021). The bacterial suspension was discarded and washed three times with sterile water after standing culture at 37°C for 24 hours. Crystal violet was dissolved in a 33% acetic acid solution, and its OD595 value was determined.
By using serial dilutions, the transconjugant PAO1-A28 and P. aeruginosa PAO1 were divided into two different amounts of bacterial suspensions ranging from 1×10 5 c.f.u. ml −1 to 1×10 6 c.f.u. ml −1. Using a microsyringe, 10µl of the prepared bacterial suspensions were injected into the body cavity of G. mellonella through the right hind foot. The control group was injected with 10 µl PBS buffer. Ten G. mellonella were injected with bacteria in each group and placed in a Petri dish at 37° C for 72 hours. At 12-hour intervals, G. mellonella was observed to survive.
Bacterium A28 was identified as P. asiatica and was resistant to meropenem, imipenem, tigecycline, gentamicin, ceftazidime, aztreonam, and ciprofloxacin, but susceptible to polymyxin and tetracycline (Table 1). The genome of P. asiatica strain A28 was assembled into two complete circularized contigs, one chromosome (5824126 bp, CP063456.1) with GC content 62.51% and one plasmid PLA28.4 (199972 bp, CP063457.1) with GC content 56.36%. Species identification with ANI analysis confirmed that the strain A28 belonged to P. asiatica, A28 and had a 98.75% identity (89.30% query coverage) to P. asiatica RYU5 strain (accession: SAMN05581751) (Tohya et al., 2019b). MLST analysis revealed that the ST of strain A28 was ST15.
Plasmid PLA28.4 is a 199,972 bp circular plasmid with 233 predicted open reading frames. PLA28.4 does, however, feature a putative replication initiator protein RepA (encoded by bp16,426 to 17,292) that has 100% cover and 93.43% amino acid sequence similarity to RepA from the IncP-7 plasmid pCAR1 (GenBank accession number AB088420.1) in P. resinovorans (Maeda et al., 2003). ParA (encoded by bp 18444 to 18923) and ParB (encoded by bp 19123 to 20256) are partitioning proteins that are 80.62% to 97.07% similar to the partition proteins of the IncP-7 plasmid pCAR1. Besides, plasmid PLA28.4 carried 7 resistance genes, including blaVIM-2, blaOXA-10, aac(6’)-Ib3, aph(3’)-I, sul1, aac(6’)-Ib-cr) and the RND-type efflux pump gene cluster tnfxB3-tmexC3.2-tmexD3b-toprJ1b (Figure 1).
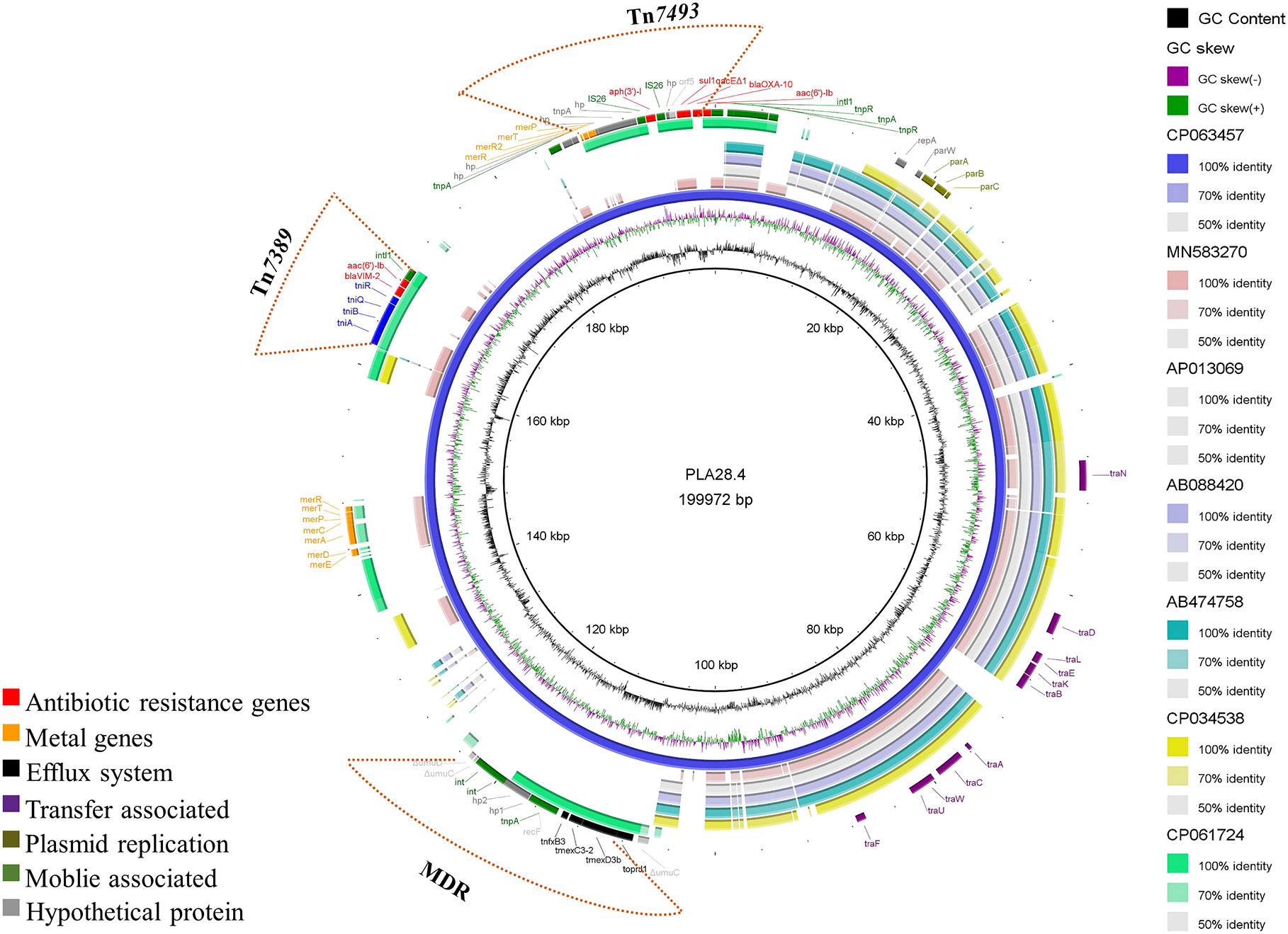
Figure 1 Comparative structural analysis of PLA28.4 with other similar plasmids available in the NCBI nr database. Starting from the center: (1) GC content of PLA28.4 with an average of 56.36%. (2) GC skew, with a positive GC skew toward the inside and a negative GC skew toward the outside. (3) The reference plasmid PLA28.4 plasmid sequence (CP063457). (4) Plasmid pNK546b (MN583270). (5) Plasmid pCAR1.3 (AP013069). (6) Plasmid pCAR1 (AB088420). (7) Plasmid pCAR1.2 (AB474758). (8) Plasmid unnamed (CP034538). (8) Plasmid pZXPA-20-602k (CP061724). (9) Gene annotation. The Figure was constructed using BRIG.
Sequence analysis showed that PLA28.4 was closely related to the IncP-7 plasmid Pnk546b (GenBank accession number MN583270.1) (Li et al., 2020) with a query coverage rate of 50% and identification rate of 84.74%. Additionally, PLA28.4 shares a similar plasmid backbone with the IncP-7-type plasmid pCAR1.3 (GenBank accession number AP013069.1) and pCAR1 (Shintani et al., 2006) from Pseudomonas resinovorans, and an unnamed plasmid (GenBank accession number CP034538.1) from Pseudomonas poae. Including the replication/partition region repA-parW-parA as well as the conjugative transfer system (consisting of traNDLEKBACWUF mobility genes), indicating that the plasmid PLA28.4 is conjugative.
Moreover, plasmid PLA28.4 had 23% sequence coverage and 99.27% identity with the megaplasmid pZXPA-20-602k (GenBank accession number: CP061724.1) from P. putida, which has both blaVIM-2 and multidrug resistance efflux pump TmexCD1-ToprJ1-like gene cluster (Li et al., 2021).
Tn7389 is a new transposon with a 7113 bp backbone and three accessory modules. A complete Tn402-like tni module showed 99.98% nucleotide sequence similarity with the genes for transposase (tniA), transposase helper proteins (tniB, tniQ) and decomposition enzymes (tniC) of Klebsiella aerogenes Tn5090 (Encoding a consistent sequence of corresponding proteins). The 5’ CS of Tn7389 is an incomplete class 1 integron that lacks the 3’ CS and contains the antibiotic resistance gene cassette(aacA4-blaVIM-2) and lacks the 3’ CS (Figure 2). Tn7389 differs from the In1701 gene cassettes found on P. aeruginosa DMC-27B, and their integrase is one base inconsistent (Jahan et al., 2020). Tn7389 has two resistance genes, blaVIM-2 and aacA4, but In1701 only has one carbapenem resistance gene, blaVIM-5. Tn402-like transposons Tn6635 and Tn6636 harboring blaVIM-2 were also discovered in two P. asiatica strains, and these two transposons carried the same entire Tn402-like tni module, but only the tniA gene had one base mutation (G409A) compared to Tn7389 (Brovedan et al., 2021). Tn7389 has the same structure as the Tn6017 transposon found in P. aeruginosa and P. putida isolated from a Spanish hospital (Juan et al., 2010). However, the similarity of tni modules is only 86.36%. Tn7389 displayed an inconsistent arrangement of resistance genes on the gene cassettes compared to the Tn402-like transposon on the plasmid of P. asiatica LD209 (Marchiaro et al., 2014). Compared to the megaplasmid PZXPA-20-602K, Tn7389’s variable region (VR) lacks the dhfrIIc gene, whereas the Tn5090-like transposon of PZXPA-20-602K has a complete type 1 integron 3’ CS region with a size of more than 46 kbp (Li et al., 2021).
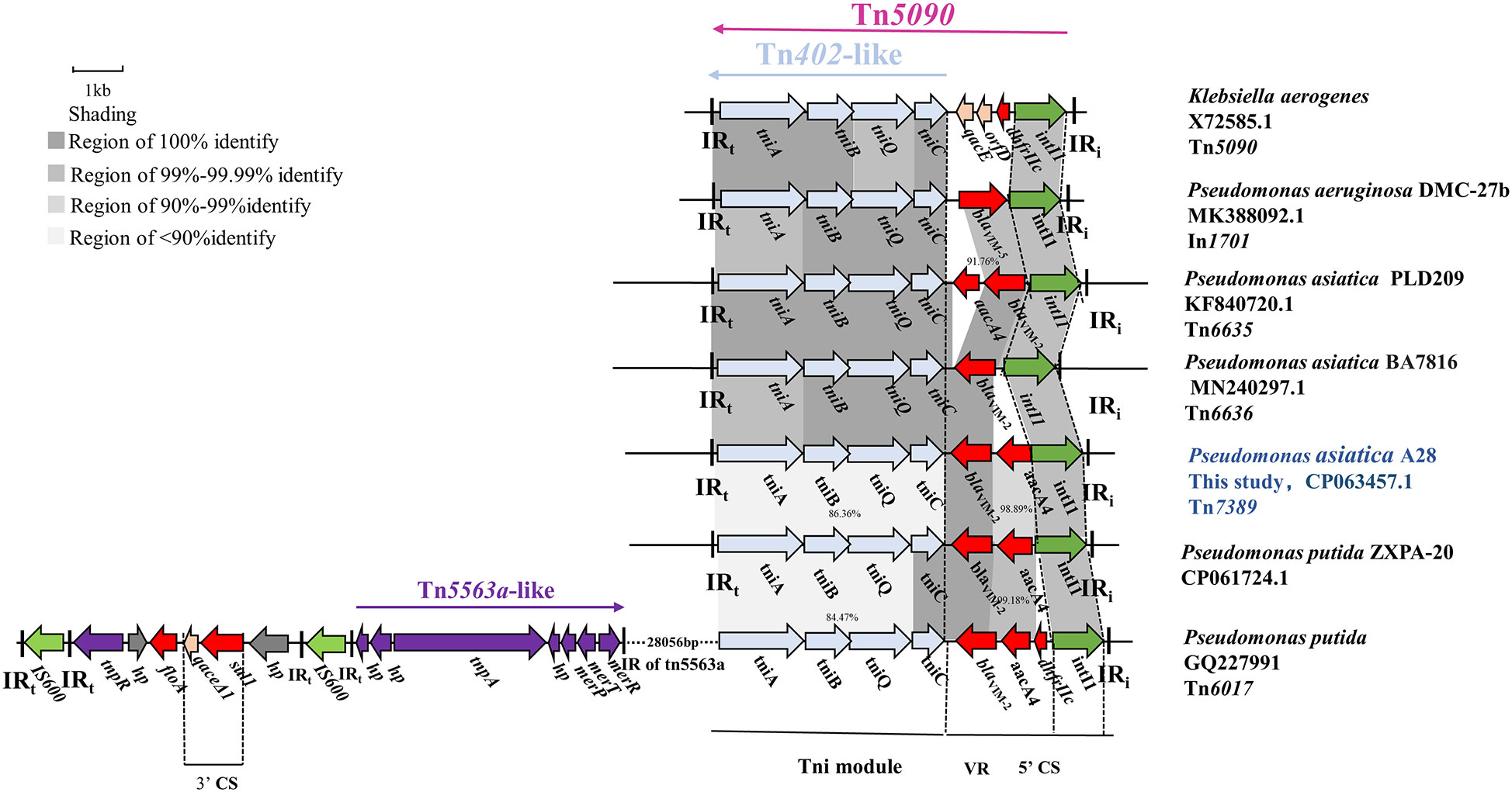
Figure 2 Genetic environment of the novel Tn402-like transposon Tn7389 in P. asiatica A28. The construction of sequence comparison was performed using BLAST (http://blast.ncbi.nlm.nih.gov). Green arrows, integrases of a class of integrons; Light blue arrow: Tn402 tni module; red arrows, antibiotic resistance genes; purple arrows, Tn5563a-like genes; gray arrows, hypothetical protein.
The blaOXA-10 gene locates within a compound Tn1403-like transposon of 20.24 kp length, flanked by a complete 38-bp IR of Tn1403 and an incomplete 30-bp IR of Tn1403, and was named Tn7493 (Figure 3). Two cassettes, aacA4-blaOXA-10, encoding resistance to aminoglycosides and oxacillinase, were found in the class 1 integron. Upstream gene cassettes were 5’ CS of intI1 and IRi, flanked by tnpAR and 38-bp IR of transposon Tn1403 (Stokes et al., 2007), and tnpR, 39-bp-long IRs of Tn5563a. Downstream of aacA4-blaOXA-10 was sul1-type 3’ -CS, orf5-hp, and IRt, almost identical to the transposon Tn6217 reported from P. aeruginosa (Xiong et al., 2013). On the flanks of IRt were two reverse insertion sequences, IS26, with an aph(3’)-I gene in the middle. The 3’ CS is a truncated transposon Tn5563a that contains a mercury resistance operon (merPTR) (Szuplewska et al., 2014), without the 3’ CS of Tn1403 and Tn5393.
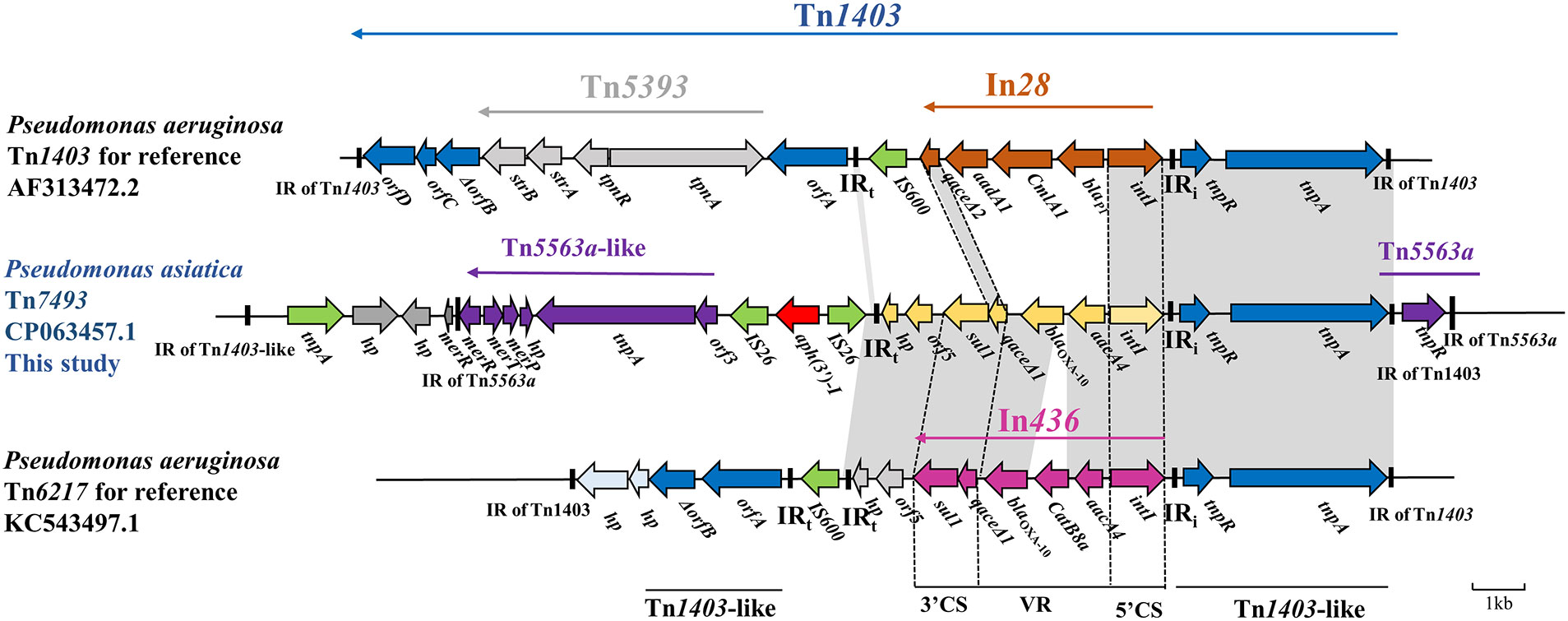
Figure 3 Genetic environment of the novel Tn1403-like transposon Tn7493 in P. asiatica A28. The extents and directions of genes are shown by arrows labeled with gene names. The construction of sequence comparison was performed using BLAST (http://blast.ncbi.nlm.nih.gov).
Two identical RND-type efflux pump fragments tnfxB2-tmexC3.2-tmexD3b-toprJ1b coexist in the chromosome and plasmid PLA28.4 of P. asiatica A28 (Figure 4). The tnfxB2-tmexC3.2- tmexD3-toprJ1b fragment was 100% identical to the cluster found in other six Pseudomonas spp. from Homo sapiens. (GenBank accession no. CP045554.1, CP039989.1, CP017073.1, CP064948.1, CP064945.1, CP062218.1) and 99.98% identical (one nucleotide substitution) to another cluster found in P. putida megaplasmid pZXPA-20-602k (GenBank accession number: CP061724.1) (from a migratory bird, Zhejiang, China) (Li et al., 2021). Like the tnfxB2-tmexCD1-toprJ1 cluster of K. pneumoniae AHM7C8I (Lv et al., 2020) (GenBank accession number: MK347425.1), tnfxB3-tmexC3.2-tmexD3b-toprJ1b is adjacent to recF (encoding AAA family ATPase), two hypothetical genes (hp1 and hp2), and two site-specific integrase genes (int1 and int2). Of these, recF has a single base substitution (A2283G), and hp2 has one base substitution (G1820T). P. asiatica A28 had 100% similarity with the tnfxB3-tmexC3.2-tmexD3b-toprJ1b-recF-hp1-hp2-int1-int2 structure of P. aeruginosa (GenBank accession no. CP039989.1) and P. putida (GenBank accession no. CP062218.1).

Figure 4 The genetic context of the multidrug resistant efflux pump tnfxB3-tmexC3.2-tmexD3b-toprJ1b. The extents and directions of genes are shown by arrows labeled with gene names. Black arrows, tnfxB1-tmexCD1-toprJ1-like gene clusters; pink arrows, int and int-like genes, predicted to encode site-specific integrases; blue arrows, umuC and umuD; green arrows, mobile related genes; red arrows, antibiotic resistance genes; yellow arrows, mercury resistance genes; gray arrows, hypothetical protein. Regions of homology between 96% and 100% are shaded.
The plasmid PLA28.4 could not be transferred to the recipient cell E. coli J53/C600 by conjugation but could be transferred to P. aeruginosa PAO1. The transfer frequency of PLA28.4 was (2.039±0.077) × 10-8 per recipient. Consequently, we evaluated the effect of acquiring resistance plasmids on biological fitness and observed significant differences in growth rate related to plasmid acquisition in P. aeruginosa PAO1 from 4h-12h (P <0.0001, Figure 5A). Biofilm formation was significantly reduced in the transconjugant strain (P<0.05) (Figure 5B). We examined the susceptibility of G. mellonella to the transconjugant PAO1-A28 and P. aeruginosa PAO1, which were injected with 1×10 5 c.f.u. ml −1 to 1×10 6 c.f.u. ml −1 of the strains and incubated in the dark at 37°C for up to 72 h. As shown in Figures 5C, D, compared with PAO1, the transconjugant PAO1-A28 showed significantly reduced virulence against G. mellonella (P<0.05). The decreased virulence of transconjugant to G. mellonella might be due to the adaptive cost of plasmids.
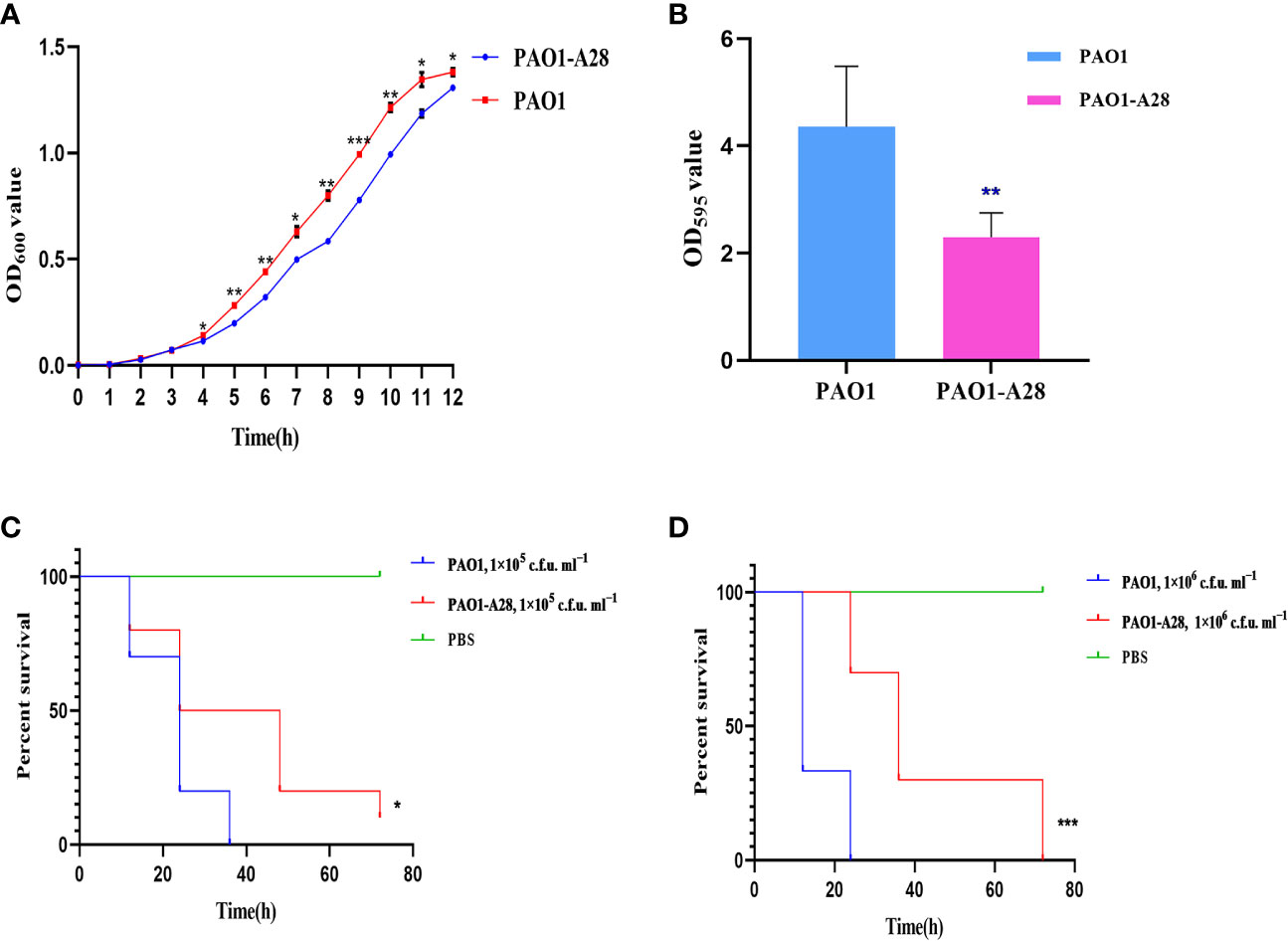
Figure 5 Fitness costs and stability of PLA28.4 in strain P. aeruginosa PAO1. (A) Growth curve of the transconjugant and recipient PAO1. (B) Biofilm formation of the transconjugant and recipient PAO1. (C, D) Survival of G. mellonella following infection with the transconjugant and recipient PAO1. *Statistically significant (p < 0.05), **statistically significant (p < 0.01), and ***statistically significant (p < 0.001).
As an important reservoir of ARB and ARG, hospital sewage is an important medium for ARG to spread to other environments. In this study, a tigecycline and carbapenem-resistant culture obtained from hospital sewage belonged to P. asiatica ST15, which is a newly proposed unique species of the genus Pseudomonas, belongs to the Pseudomonas putida group (Tohya et al., 2020). Sequencing analysis revealed that it coharboring carrying a tmexCD3-toprJ1b, a novel Tn5090-like transposon Tn7389 harboring blaVIM-2, and a Tn1403-like transposon Tn7493 harboring blaOXA-10. Tn5090 (also known as Tn402) was discovered on IncP-7 plasmid R751 from K. aerogenes in 1994 (Rådström et al., 1994). In Tn7389, two 25-bp initial reverse repeat (IRi) and terminal reverse repeat (IRt) of Tn5090/Tn402 transposon families were located 171 bp downstream of intI1 and 116bp upstream of tniA, respectively, suggesting that the blaVIM-2 could be mobilized using the tni machinery. The integrase and recombination sites containing class 1 integrons can be inserted and removed in the form of gene cassettes at attI1 (Toleman and Walsh, 2011). Multiple Tn5090-like transposons carrying blaVIM-2 have been found in Pseudomonas in a growing number of investigations, suggesting that Tn5090-like transposons are key mobile components of VIM-2 transmission in Pseudomonas (Santos et al., 2010). The blaVIM-2 gene could be mobile via the tni mechanism, which may promote its transmission among other pathogens in the hospital sewage environment and requires closer monitoring.
Tn1403 was discovered on RPL11 plasmids from clinical P. aeruginosa isolates expressing resistance to ampicillin, streptomycin, puromycin, and chloramphenicol (Vézina and Levesque, 1991). Tn1403-like transposons have been found primarily in Pseudomonas spp. and have been shown to carry diverse types of ARGs, suggesting that they may play an important role in ARG and metal resistance gene transmission in Pseudomonas. In addition, disinfectant-sulfanilamide resistance (qacEΔ1-sul1) genes cause bacterial resistance to chlorine-containing disinfectants and allows bacteria to survive in disinfected water, which poses a threat to health care systems.
Although there are different variants of the MDR efflux pumps tmexCD1-toprJ1, similar structures have also been found in Aeromonas caviae, Raoultella planticola, and Klebsiella quasipneumoniae, suggesting potential horizontal transfer mechanisms among various species (Wang et al., 2021a; Dong et al., 2022; Gao et al., 2022). The transfer of tnfxB2-tmexCD1-toprJ1 has previously been found to be mobilized by site-specific integrase (Lv et al., 2020). However, it could be linked to umuCD, a neighboring mutant DNA repair system, because integrase can accelerate the excision and integration of umuCD (Peng et al., 2021). The proximity of umuCD to the efflux pump structure in various bacteria revealed that it might help spread tmexCD1-TopRJ1-like gene clusters.
The IncP-7 plasmid is a conjugative transfer plasmid with a narrow host range (Shintani et al., 2010). Although most reports suggest that IncP-7 plasmids could only be transmitted in Pseudomonas (Xiong et al., 2013), pCAR1 was discovered to be transferable to Sterotrophomonas-like strains in natural water (Shintani et al., 2008). Moreover, the IncP-7 type plasmid pNK546b in P. aeruginosa NK546 also assisted the transmission of another resistant plasmid pNK546a that could not be self-transmissible (Li et al., 2020). In this study, the IncP-7 plasmid PLA28.4 of P. asiatica could be transferred to P. aeruginosa PAO1, suggesting PLA28.4 has the capacity to transmit numerous resistance genes in hospital sewage, according to this study. Collectively, plasmid fitness cost studies found that transferring the PLA28.4 plasmid into P. aeruginosa PAO1 resulted in a lower growth rate, less biofilm generation, and lower pathogenicity, demonstrating that transmission of the PLA28.4 plasmid caused bacteria to pay a cost of adaptation.
We discovered a P. asiatica carrying a plasmid containing the tmexCD1-toprJ1-like gene cluster, and two novel transposons carrying blaVIM-2 and blaOXA-10, respectively. Controlling the development and spread of medication resistance requires continuous monitoring and management of resistant microorganisms in hospital sewage.
We discovered a P. asiatica carrying a plasmid containing the tmexCD1-toprJ1-like gene cluster, and two novel transposon carrying blaVIM-2 and blaOXA-10, respectively. Controlling the development and spread of medication resistance requires continuous monitoring and management of resistant microorganisms in hospital sewage.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below: https://www.ncbi.nlm.nih.gov/genbank/, CP063456.1.
SL, WW, BYZ and QHL collected the data. FYZ and LG performed the bioinformatic analyses. QL, QC, AM-R wrote the initial draft of the manuscript. QL, RJH, LX and YSZ conceived the project, reviewed the articles and extracted the data. XX contributed to the revision of this article. All authors contributed to the article and approved the submitted version.
This research was supported by the Sichuan Province Science and Technology project [2020YJ0338, 2022YFS0631 and 2022YFS0632], Natural Science Foundation of Luzhou (2021-NYF-20, 2022-RCM-173) and Southwest Medical University Foundation (21YYJC0529), Wuxi Commission of Health Funding (T202134).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Aghapour, Z., Gholizadeh, P., Ganbarov, K., Bialvaei, A. Z., Mahmood, S. S., Tanomand, A., et al. (2019). Molecular mechanisms related to colistin resistance in enterobacteriaceae. Infection Drug resistance. 12, 965–975. doi: 10.2147/idr.S199844
Alavi, M. R., Antonic, V., Ravizee, A., Weina, P. J., Izadjoo, M., Stojadinovic, A. (2011). An enterobacter plasmid as a new genetic background for the transposon Tn1331. Infection Drug resistance 4, 209–213. doi: 10.2147/idr.S25408
Botelho, J., Roberts, A. P., León-Sampedro, R., Grosso, F., Peixe, L. (2018). Carbapenemases on the move: It's good to be on ICEs. Mobile DNA 9, 37. doi: 10.1186/s13100-018-0141-4
Brovedan, M. A., Marchiaro, P. M., Díaz, M. S., Faccone, D., Corso, A., Pasteran, F., et al. (2021). Pseudomonas putida group species as reservoirs of mobilizable Tn402-like class 1 integrons carrying bla(VIM-2) metallo-β-lactamase genes. Infection Genet. Evol. J. Mol. Epidemiol. evolutionary Genet. Infect. Dis. 96, 105131. doi: 10.1016/j.meegid.2021.105131
Ding, M., Shi, J., Ud Din, A., Liu, Y., Zhang, F., Yan, X., et al. (2021). Co-Infections of two carbapenemase-producing enterobacter hormaechei clinical strains isolated from the same diabetes individual in China. J. Med. Microbiol. 2021, 70(3). doi: 10.1099/jmm.0.001316
Dong, N., Zeng, Y., Wang, Y., Liu, C., Lu, J., Cai, C., et al. (2022). Distribution and spread of the mobilised RND efflux pump gene cluster tmexCD-toprJ in clinical gram-negative bacteria: A molecular epidemiological study. Lancet Microbe 2022. doi: 10.1016/s2666-5247(22)00221-x
Gao, X., Wang, C., Lv, L., He, X., Cai, Z., He, W., et al. (2022). Emergence of a novel plasmid-mediated tigecycline resistance gene cluster, tmexCD4-toprJ4, in klebsiella quasipneumoniae and enterobacter roggenkampii. Microbiol. Spectr. 10 (4), e0109422. doi: 10.1128/spectrum.01094-22
Hu, R., Li, Q., Zhang, F., Ding, M., Liu, J., Zhou, Y. (2021). Characterisation of bla(NDM-5) and bla(KPC-2) co-occurrence in K64-ST11 carbapenem-resistant klebsiella pneumoniae. J. Glob Antimicrob. Resist. 27, 63–66. doi: 10.1016/j.jgar.2021.08.009
Huang, J., Yi, M., Yuan, Y., Xia, P., Yang, B., Liao, J., et al. (2022). Emergence of a fatal ST11-KL64 tigecycline-resistant hypervirulent klebsiella pneumoniae clone cocarrying bla(NDM) and bla(KPC) in plasmids. Microbiol. Spectr. 2022, e0253922. doi: 10.1128/spectrum.02539-22
Jahan, M. I., Rahaman, M. M., Hossain, M. A., Sultana, M. (2020). Occurrence of intI1-associated VIM-5 carbapenemase and co-existence of all four classes of β-lactamase in carbapenem-resistant clinical pseudomonas aeruginosa DMC-27b. J. antimicrobial chemotherapy. 75 (1), 86–91. doi: 10.1093/jac/dkz426
Jo, J., Ko, K. S. (2021). Tigecycline heteroresistance and resistance mechanism in clinical isolates of acinetobacter baumannii. Microbiol. Spectr. 9 (2), e0101021. doi: 10.1128/Spectrum.01010-21
Juan, C., Zamorano, L., Mena, A., Albertí, S., Pérez, J. L., Oliver, A. (2010). Metallo-beta-lactamase-producing pseudomonas putida as a reservoir of multidrug resistance elements that can be transferred to successful pseudomonas aeruginosa clones. J. antimicrobial chemotherapy. 2010;65 (3), 474–478. doi: 10.1093/jac/dkp491
Li, Z., Cai, Z., Cai, Z., Zhang, Y., Fu, T., Jin, Y., et al. (2020). Molecular genetic analysis of an XDR pseudomonas aeruginosa ST664 clone carrying multiple conjugal plasmids. J. antimicrobial chemotherapy. 75 (6), 1443–1452. doi: 10.1093/jac/dkaa063
Li, R., Peng, K., Xiao, X., Liu, Y., Peng, D., Wang, Z. (2021). Emergence of a multidrug resistance efflux pump with carbapenem resistance gene blaVIM-2 in a pseudomonas putida megaplasmid of migratory bird origin. J. antimicrobial chemotherapy 76 (6), 1455–1458. doi: 10.1093/jac/dkab044
Li, Y., Qiu, Y., Gao, Y., Chen, W., Li, C., Dai, X., et al. (2022). Genetic and virulence characteristics of a raoultella planticola isolate resistant to carbapenem and tigecycline. Sci. Rep. 12 (1), 3858. doi: 10.1038/s41598-022-07778-0
Lv, L., Wan, M., Wang, C., Gao, X., Yang, Q., Partridge, S. R., et al. (2020). Emergence of a plasmid-encoded resistance-Nodulation-Division efflux pump conferring resistance to multiple drugs, including tigecycline, in klebsiella pneumoniae. mBio 2020, 11(2). doi: 10.1128/mBio.02930-19
Maeda, K., Nojiri, H., Shintani, M., Yoshida, T., Habe, H., Omori, T. (2003). Complete nucleotide sequence of carbazole/dioxin-degrading plasmid pCAR1 in pseudomonas resinovorans strain CA10 indicates its mosaicity and the presence of large catabolic transposon Tn4676. J. Mol. Biol. 326 (1), 21–33. doi: 10.1016/s0022-2836(02)01400-6
Mann, R., Rafei, R., Gunawan, C., Harmer, C. J., Hamidian, M. (2022). Variants of Tn6924, a novel Tn7 family transposon carrying the bla(NDM) metallo-β-Lactamase and 14 copies of the aphA6 amikacin resistance genes found in acinetobacter baumannii. Microbiol. Spectr. 10 (1), e0174521. doi: 10.1128/spectrum.01745-21
Marchiaro, P. M., Brambilla, L., Morán-Barrio, J., Revale, S., Pasteran, F., Vila, A. J., et al. (2014). The complete nucleotide sequence of the carbapenem resistance-conferring conjugative plasmid pLD209 from a pseudomonas putida clinical strain reveals a chimeric design formed by modules derived from both environmental and clinical bacteria. Antimicrobial Agents chemotherapy. 58 (3), 1816–1821. doi: 10.1128/aac.02494-13
Peng, K., Wang, Q., Yin, Y., Li, Y., Liu, Y., Wang, M., et al. (2021). Plasmids shape the current prevalence of tmexCD1-toprJ1 among klebsiella pneumoniae in food production chains. mSystems. 6 (5), e0070221. doi: 10.1128/mSystems.00702-21
Rådström, P., Sköld, O., Swedberg, G., Flensburg, J., Roy, P. H., Sundström, L. (1994). Transposon Tn5090 of plasmid R751, which carries an integron, is related to Tn7, mu, and the retroelements. J. bacteriology. 176 (11), 3257–3268. doi: 10.1128/jb.176.11.3257-3268.1994
Santos, C., Caetano, T., Ferreira, S., Mendo, S. (2010). Tn5090-like class 1 integron carrying bla(VIM-2) in a pseudomonas putida strain from Portugal. Clin. Microbiol. Infect. 16 (10), 1558–1561. doi: 10.1111/j.1469-0691.2010.03165.x
Shintani, M., Fukushima, N., Tezuka, M., Yamane, H., Nojiri, H. (2008). Conjugative transfer of the IncP-7 carbazole degradative plasmid, pCAR1, in river water samples. Biotechnol. letters. 2008;30 (1), 117–122. doi: 10.1007/s10529-007-9519-y
Shintani, M., Takahashi, Y., Yamane, H., Nojiri, H. (2010). The behavior and significance of degradative plasmids belonging to inc groups in pseudomonas within natural environments and microcosms. Microbes environments. 2010;25 (4), 253–265. doi: 10.1264/jsme2.me10155
Shintani, M., Yano, H., Habe, H., Omori, T., Yamane, H., Tsuda, M., et al. (2006). Characterization of the replication, maintenance, and transfer features of the IncP-7 plasmid pCAR1, which carries genes involved in carbazole and dioxin degradation. Appl. Environ. Microbiol. 72 (5), 3206–3216. doi: 10.1128/aem.72.5.3206-3216.2006
Smyth, C., O'Flaherty, A., Walsh, F., Do, T. T. (2020). Antibiotic resistant and extended-spectrum β-lactamase producing faecal coliforms in wastewater treatment plant effluent. Environ. pollut. (Barking Essex 1987) 262, 114244. doi: 10.1016/j.envpol.2020.114244
Stokes, H. W., Elbourne, L. D., Hall, R. M. (2007). Tn1403, a multiple-antibiotic resistance transposon made up of three distinct transposons. Antimicrobial Agents chemotherapy. 51 (5), 1827–1829. doi: 10.1128/aac.01279-06
Szuplewska, M., Ludwiczak, M., Lyzwa, K., Czarnecki, J., Bartosik, D. (2014). Mobility and generation of mosaic non-autonomous transposons by Tn3-derived inverted-repeat miniature elements (TIMEs). PloS One 9 (8), e105010. doi: 10.1371/journal.pone.0105010
Tohya, M., Tada, T., Watanabe, S., Kuwahara-Arai, K., Zin, K. N., Zaw, N. N., et al. (2019a). ) emergence of carbapenem-resistant pseudomonas asiatica producing NDM-1 and VIM-2 metallo-β-Lactamases in Myanmar. Antimicrobial Agents chemotherapy 2019, 63(8). doi: 10.1128/aac.00475-19
Tohya, M., Uechi, K., Tada, T., Hishinuma, T., Kinjo, T., Ohshiro, T., et al. (2021). Emergence of clinical isolates of pseudomonas asiatica and pseudomonas monteilii from Japan harbouring an acquired gene encoding a carbapenemase VIM-2. J. Med. Microbiol. 2021 70 (1). doi: 10.1099/jmm.0.001258
Tohya, M., Watanabe, S., Tada, T., Tin, H. H., Kirikae, T. (2020). Genome analysis-based reclassification of pseudomonas fuscovaginae and pseudomonas shirazica as later heterotypic synonyms of pseudomonas asplenii and pseudomonas asiatica, respectively. Int. J. systematic evolutionary Microbiol. 70 (5), 3547–3552. doi: 10.1099/ijsem.0.004199
Tohya, M., Watanabe, S., Teramoto, K., Uechi, K., Tada, T., Kuwahara-Arai, K., et al. (2019b). Pseudomonas asiatica sp. nov., isolated from hospitalized patients in Japan and Myanmar. Int. J. systematic evolutionary Microbiol. 69 (5), 1361–1368. doi: 10.1099/ijsem.0.003316
Toleman, M. A., Walsh, T. R. (2011). Combinatorial events of insertion sequences and ICE in gram-negative bacteria. FEMS Microbiol. Rev. 35 (5), 912–935. doi: 10.1111/j.1574-6976.2011.00294.x
Vézina, G., Levesque, R. C. (1991). Molecular characterization of the class II multiresistance transposable element Tn1403 from pseudomonas aeruginosa. Antimicrobial Agents chemotherapy. 35 (2), 313–321. doi: 10.1128/aac.35.2.313
Wang, C. Z., Gao, X., Yang, Q. W., Lv, L. C., Wan, M., Yang, J., et al. (2021). ) a novel transferable resistance-Nodulation-Division pump gene cluster, tmexCD2-toprJ2, confers tigecycline resistance in raoultella ornithinolytica. Antimicrobial Agents chemotherapy 2021, 65(4). doi: 10.1128/aac.02229-20
Wang, Q., Peng, K., Liu, Y., Xiao, X., Wang, Z., Li, R. (2021). Characterization of TMexCD3-TOprJ3, an RND-type efflux system conferring resistance to tigecycline in Proteus mirabilis, and its associated integrative conjugative element. Antimicrobial Agents chemotherapy 65 (7), e0271220. doi: 10.1128/aac.02712-20
Xiong, J., Alexander, D. C., Ma, J. H., Déraspe, M., Low, D. E., Jamieson, F. B., et al. (2013). Complete sequence of pOZ176, a 500-kilobase IncP-2 plasmid encoding IMP-9-mediated carbapenem resistance, from outbreak isolate pseudomonas aeruginosa 96. Antimicrobial Agents chemotherapy. 57 (8), 3775–3782. doi: 10.1128/aac.00423-13
Yousfi, K., Touati, A., Lefebvre, B., Fournier, É, Côté, J. C., Soualhine, H., et al. (2017). A novel plasmid, pSx1, harboring a new Tn1696 derivative from extensively drug-resistant shewanella xiamenensis encoding OXA-416. Microbial Drug resistance (Larchmont NY) 23 (4), 429–436. doi: 10.1089/mdr.2016.0025
Zhang, B., Hu, R., Liang, Q., Liang, S., Li, Q., Bai, J., et al. (2022). Comparison of two distinct subpopulations of klebsiella pneumoniae ST16 Co-occurring in a single patient. Microbiol. Spectr. 2022, e0262421. doi: 10.1128/spectrum.02624-21
Zong, G., Zhong, C., Fu, J., Zhang, Y., Zhang, P., Zhang, W., et al. (2020). The carbapenem resistance gene bla(OXA-23) is disseminated by a conjugative plasmid containing the novel transposon Tn6681 in acinetobacter johnsonii M19. Antimicrobial resistance infection control 9 (1), 182. doi: 10.1186/s13756-020-00832-4
Keywords: hospital sewage, blaVIM-2, transposon, tigecycline resistance gene cluster, Pseudomononas asiatica
Citation: Li Q, Chen Q, Liang S, Wang W, Zhang B, Martín-Rodríguez AJ, Liang Q, Zhang F, Guo L, Xiong X, Hu R, Xiang L and Zhou Y (2023) Coexistence of tmexCD3-toprJ1b tigecycline resistance genes with two novel blaVIM-2-carrying and blaOXA-10-carrying transposons in a Pseudomononas asiatica plasmid. Front. Cell. Infect. Microbiol. 13:1130333. doi: 10.3389/fcimb.2023.1130333
Received: 23 December 2022; Accepted: 06 February 2023;
Published: 01 March 2023.
Edited by:
Punyawee Dulyayangkul, Chulabhorn Research Institute, ThailandReviewed by:
Subhasree Roy, National Institute of Cholera and Enteric Diseases (ICMR), IndiaCopyright © 2023 Li, Chen, Liang, Wang, Zhang, Martín-Rodríguez, Liang, Zhang, Guo, Xiong, Hu, Xiang and Zhou. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yingshun Zhou, eWluZ3NodW56aG91QHN3bXUuZWR1LmNu; Renjing Hu, d2Vpd2VpaHVodTExMkAxNjMuY29t; Li Xiang, eGlhbmdsaTIwMTQwMzE0QDE2My5jb20=
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.