- The First Affiliated Hospital, Wenzhou Medical University, Wenzhou, China
Embryonic lethal abnormal vision-like (ELAVL) proteins are RNA binding proteins that were originally discovered as indispensable regulators of the development and functioning of the nervous system. Subsequent studies have shown that ELAVL proteins not only exist in the nervous system, but also have regulatory effects in other tissues. ELAVL proteins have attracted attention as potential therapeutic targets because they stabilize multiple mRNAs by binding within the 3′-untranslated region and thus promote the development of tumors, including hepatocellular carcinoma, pancreatic cancer, ovarian cancer, breast cancer, colorectal carcinoma and lung cancer. Previous studies have focused on these important relationships with downstream mRNAs, but emerging studies suggest that ELAVL proteins also interact with non-coding RNAs. In this review, we will summarize the relationship of the ELAVL protein family with mRNA and non-coding RNA and the roles of ELAVL protein family members in a variety of physiological and pathological processes.
Introduction
The embryonic lethal abnormal vision-like (ELAVL) proteins in fish, frogs, and mammals are defined as RNA-binding proteins (RBPs), and they play important roles in post-transcriptional regulation (Campos et al., 1985). ELAVL proteins were first discovered in Drosophila due to their ability to interact with AU-rich element (ARE)-containing transcripts (Campos et al., 1985). Each member of the ELAVL protein family, which includes ELAVL1-4 (HuR HuB, HuC, HuD) (Figure 1), consists of three similar and conserved RNA recognition motifs (RRM) (Toba and White, 2008; Colombrita et al., 2013). The sequence of a hinge region between RRMs 2 and 3 differs among the four family members (Good, 1995), and its presence is key to the ability of these proteins to shuttle into and out of the nucleus (Fan and Steitz, 1998a).
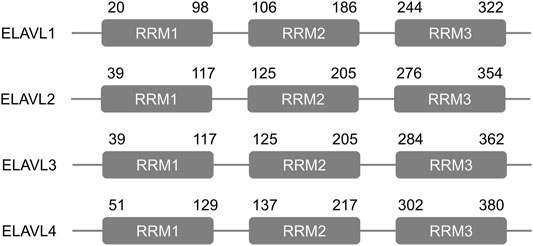
FIGURE 1. Structure of the human ELAVL proteins. Each ELAVL protein consists of three similar and conserved RNA recognition motifs (RRMs). The numbers indicate amino acid positions.
Biologically, ELAVL proteins were originally discovered as indispensable regulators of nervous system development and physiological function (Rogulja-Ortmann et al., 2014; Wang et al., 2019a; Zhao et al., 2020a). Interestingly, subsequent studies have shown that they not only exist in the nervous system, but also have regulatory effects in other tissues, including hepatocytes (Wang et al., 2021), fat cells (Siang et al., 2020), vascular smooth muscle cells (Liu et al., 2020a), and intestinal epithelial cells (Liu et al., 2019).
Previous studies have mainly focused on the relationship between ELAVL proteins and downstream mRNA transcripts, and less attention has been paid to interactions between ELAVLs and non-coding RNA (ncRNA) molecules. The regulation and metabolism of ncRNA is an emerging research topic, and ncRNAs have been shown to play important roles in a variety of fields, such as cancer (Deschenes-Furry et al., 2007; Schultz et al., 2020), inflammation (Wen et al., 2021), and cell differentiation (Chen et al., 2021a). This review aims to summarize relationships of the ELAVL family of proteins with mRNAs and ncRNAs and the regulation of ELAVL family proteins in various physiological and pathological processes.
Members of the ELAVL Family
ELAVL1, also known as human antigen R (HuR), has been established as a tumor-specific antigen in colorectal carcinoma (Wang et al., 2000) and tumors of the central nervous system (Nabors et al., 2001). This protein is a widely expressed RBP whose function in many cell types has been elucidated. The gene encoding human ELAVL1 is localized to chromosome 19p13.2, and while the protein is mainly localized to the nucleus, it will translocate to the cytoplasm following stimulation by a variety of extracellular stimuli (Chand et al., 2017).
The consequences for an mRNA after ELAVL1 binding to its ARE depend on the mRNA itself and the cell type. When the target ARE, which usually contains multiple AUUUA repeats, appears in the 3′-untranslated regions (UTRs) of an mRNA, ELAVL1 binding often results in mRNA instability (Fan et al., 1997), and thus selective binding of ELAVL1 to the ARE at the 3′ end of an mRNA can lead to rapid degradation (Myer et al., 1997). However, in most cases, ELAVL1 plays an important role in stabilizing the mRNA sequence. Competitive binding to mRNA in the 3′-UTR by ELAVL1 prevents exonuclease- or endonuclease-mediated degradation induced by other RBPs (Chen et al., 1995; Fan and Steitz, 1998b). In one specific case, ELAVL1 promotes neuronal movement by the stabilizing of profilin 1 mRNA (Chen et al., 1995). Similarly, ELAVL1 binding extends the half-life of CX43 mRNA in rat liver epithelial cells (Ale-Agha et al., 2009).
In addition to interacting with the 3′-UTR, ELAVL1 also has multiple effects in other mRNA regions. For example, ELAVL1 can reduce the activity of the internal ribosome entry site (IRES) in the initiation of translation by binding to the 5′-UTR of the mRNA that codes for the insulin like growth factor type 1 receptor (Meng et al., 2005). Conversely, ELAVL1 can stimulate the initiation of translation of X-linked inhibitor of apoptosis (XIAP) mRNA by binding to the IRES in the 5′-UTR of the XIAP mRNA (Durie et al., 2011).
Other members of the ELAVL protein family have not been studied to the extent that ELAVL1 has, but these other family members have been shown to be important in multiple physiological and pathological processes. ELAVL2 (also known as HuB or Hel-N1), ELAVL3 (HuC) and ELAVL4 (HuD) are mainly distributed in neuronal cells (Ripin et al., 2019). ELAVL2 plays a key role in several processes in the early stages of neuronal differentiation, such as cell cycle exit (Hambardzumyan et al., 2009). ELAVL3 has been shown to affect brain function in that low expression levels of ELAVL3 correlated with impaired spatial learning ability of mice and led to the down-regulation of expression of growth associated protein-43 (Quattrone et al., 2001). In addition, the levels of the ELAVL4 transcript and protein in the superior cervical ganglion were found to decrease after the severing of the axon (Deschenes-Furry et al., 2007). These results show that ELAVL2-4 are involved in the development and functioning of the nervous system. Interestingly, however, the functions of these three protein family members go far beyond the brain. They are also essential in the maintenance of physiological functions and the regulation of the occurrence and development of a variety of diseases (Mazan-Mamczarz et al., 2003; Casolaro et al., 2008; Beauchamp et al., 2010; Ahuja et al., 2016; Lee et al., 2018; Zhao et al., 2019).
The Regulation of ELAVL Proteins by Non-RNA Molecules
The regulation of ELAVL proteins by non-RNA molecules can be divided into three categories: regulation of protein expression level, regulation of nucleocytoplasmic shuttling, and regulation of the binding of ELAVL proteins to the transcripts in cytoplasm. In gastric tumorigenesis, the activation of AKT serine/threonine kinase promotes the binding of Nuclear Factor-kappa B to the ELAVL promoter, which enhances transcription and the stability of the transcripts (Kang et al., 2008). In addition, the esophageal cancer related gene 2 protein has been shown to increase ubiquitination and degradation of ELAVL1 in the colon cancer-derived RKO cell line and the breast cancer-derived MCF7 cell line, but this protein failed to produce similar effects on several non-ubiquitinable mutant forms of ELAVL1 (Lucchesi et al., 2016).
In response to proliferative signals, the phosphorylation of ELAVL1 protein by cyclin-dependent kinase 1 at S202 prevents its translocation to the cytoplasm, resulting in the inhibition of its pro-proliferation and anti-apoptotic effects (Kim et al., 2008). Upon cessation of the external signal, two mechanisms serve to promote relocalization to the cytoplasm. One mechanism involves polyADP-ribosylation of ELAVL1 by poly (ADP-ribose) polymerase 1 (Ke et al., 2017), and a second mechanism is mediated by p38 mitogen-activated protein kinase (Farooq et al., 2009). In another enzyme-controlled mechanism of regulation, sulfhydration by cystathionine γ lyase prevents the homodimerization of ELAVL1, which ultimately leads to decreases in activity to levels insufficient to increase the expression level of downstream genes in mouse endothelial cells (Bibli et al., 2019).
Interactions Between ELAVL Proteins and Various Classes of RNA Molecules
Interactions Between ELAVL Proteins and mRNAs
One of the main functions of the ELAVL protein family is to regulate the stability and half-life of downstream mRNA. Therefore, their regulatory effects on cells almost entirely depend on the function of downstream mRNAs and the direction of regulation. The most common binding sites where ELAVL proteins interact with mRNAs, AREs in the 3′-UTR, are found in up to 8% of human genome transcripts (Bakheet et al., 2006). These AREs are often regarded as regulatory elements that promote mRNA decay; most RBPs that bind to this region, such as tristetraprolin, butyrate response factor 1, AU-binding factor 1, and KH-type splicing regulatory protein, greatly reduce the half-life of the target RNA (Gherzi et al., 2004; Lykke-Andersen and Wagner, 2005; Gratacós and Brewer, 2010). In most cases, however, the effect of the binding of ELAVL proteins to a downstream mRNA supports stability (Barreau et al., 2005). Previously discovered mRNAs that have been found to bind to the ELAVL family are shown in Supplementary Table 1.
Interactions Between ELAVL Proteins and ncRNAs
Micro RNA (miRNA)
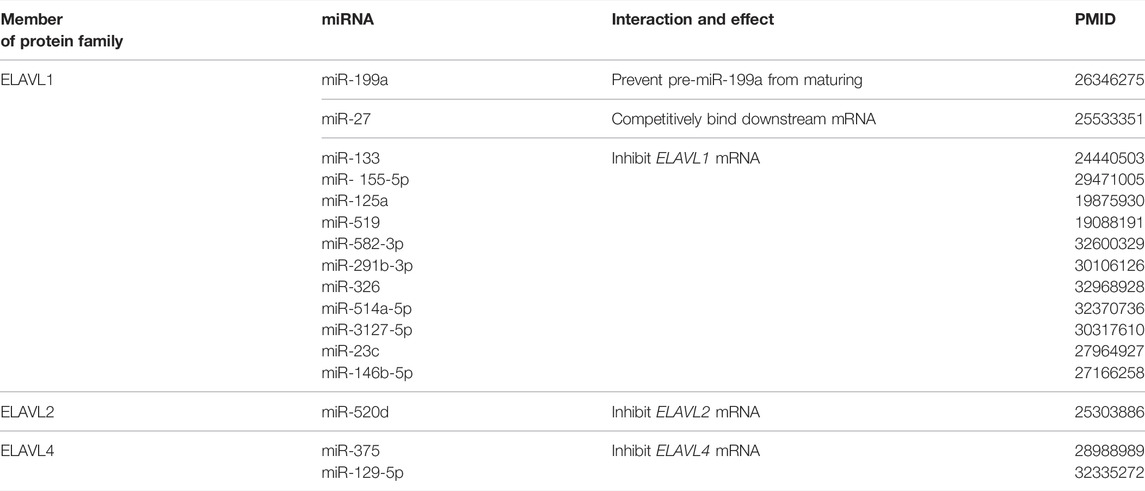
Various miRNA molecules can bind to the 3′-UTRs of mRNA to decrease the stability of the mRNA. Therefore, the regulation of miRNAs to the ELAVL protein family is mainly reflected in the stability of mRNAs of the latter (Table 1). For example, miR-133, which targets the ELAVL1 mRNA and is sponged by long intergenic non-protein coding RNA, muscle differentiation 1 (linc-MD1), regulates the expression of ELAVL1. The ELAVL1 protein in turn promotes the interaction between linc-MD1 and miR-133 in the early stages of myogenesis (Legnini et al., 2014). Through targeting AREs in ELAVL1 mRNA, miR-155-5p negatively regulates the protein level of ELAVL1 and thus the migration of tumor cells in colorectal cancer (Al-Haidari et al., 2018). In breast cancer, miR-125a inhibited cell proliferation and promoted apoptosis by downregulating ELAVL1 which was highly expressed in cancer cells, and this effect was partially rescued by ELAVL1 overexpression (Guo et al., 2009). In normal human dermal fibroblasts, the overexpression of miR-520d-5p has been shown to down-regulate ELAVL2 and restore cell proliferation; down-regulation of ELAVL2 with small interfering RNA alone achieved the same effect (Ishihara et al., 2014). In mutant motor neurons, decreased expression of miR-375 resulted in increased expression of its downstream targets, which include ELAVL4 as well as p53. These changes promoted the apoptosis and fragility of mutant motor neurons in amyotrophic lateral sclerosis (De Santis et al., 2017).
In turn, ELAVL proteins can regulate miRNAs maturation or co-regulate downstream with miRNAs. In another mode of regulation for miR-199a, hypoxia-induced expression of ELAVL1 prevents the maturation of pre-miR-199a, thereby promoting enhancement of glycolysis through impacts of miR-199a on hexokinase 2 and pyruvate kinase 2 expression in the tumor microenvironment (Zhang et al., 2015). It has been shown that miR-27 targets the mRNA encoding zinc finger protein 36 mRNA in macrophages, but ELAVL1 and miR-27 compete for binding to the 3′-UTR of this mRNA to regulate its stability (Lu et al., 2014). These examples indicate that there is significant crosstalk among RBPs, miRNAs and mRNAs, and that the regulation is not limited to a few isolated cases.
Long Non-coding RNA (lncRNA)
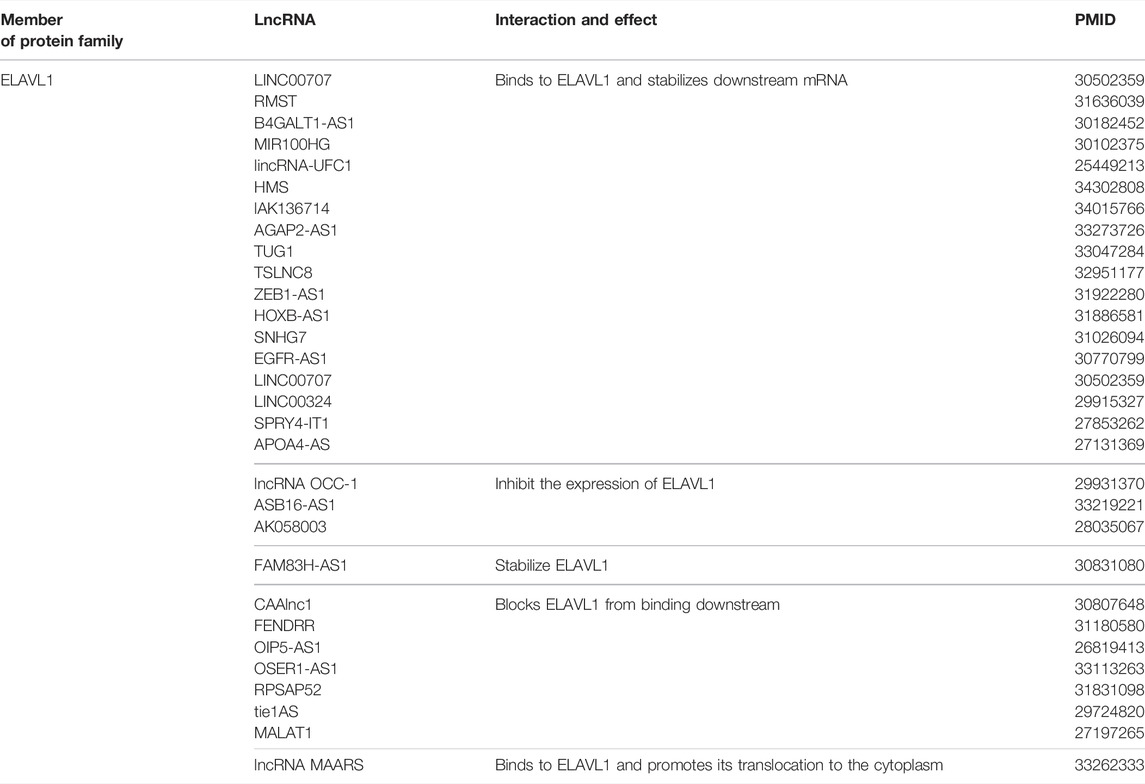
The mechanisms by which lncRNAs regulate mRNA activity through the ELAVL protein family, especially ELAVL1, include 1) direct binding to ELAVL1 and promotion of its binding to downstream mRNA, 2) inhibiting of the expression of ELAVL1, 3) stabilizing ELAVL1 protein, 4) direct binding to ELAVL1 and blocking of its binding to downstream mRNA, and 5) promoting the translocation of ELAVL1 from the nucleus to the cytoplasm (Table 2).
First, LINC00707 has been reported as a malignant factor in the progression of lung adenocarcinoma and gastric cancer. LINC00707, which is highly expressed in tumor tissues, has been shown to form a complex with ELAVL1 protein. This complex increases the expression of downstream proteins, such as vav guanine nucleotide exchange factor 3/F11 receptor, and ultimately leads to tumor progression and a poorer tumor prognosis (Xie et al., 2019). In a similar way, lncRNA RMST enhances the binding of ELAVL1 to the mRNA of the target gene DNMT3B, thereby increasing the expression of DNMT3B and global levels of DNA methylation (Peng et al., 2020).
Second, ELAVL1 interacts with lncRNA OCC-1, which acts as a protective factor in colorectal cancer, inhibits the growth of tumor cells in vivo and in vitro. This inhibition is achieved by sensitizing ELAVL1 to ubiquitination and making it prone to degradation (Lan et al., 2018). In the third type of regulation, up-regulated FAM83H-AS1 binds to ELAVL1 and stabilizes it, which can induce cell metastasis and resistance to radiotherapy in ovarian cancer (Dou et al., 2019). As described in the fourth mode of regulation, pull-down assays and RNA immunoprecipitation have confirmed the binding relationship between CAAlnc1 and ELAVL1, which blocked the binding of ELAVL1 to mRNAs associated with fat production (Shen et al., 2019).
According to multiple reports, lncRNA MAARS, which is positively correlated with the progression of atherosclerosis disease, interacts with ELAVL1 and reduces its cytoplasmic localization, which reduces the apoptosis of macrophages and delays the course of the disease (Simion et al., 2020).
Circular RNA (circRNA)
The class of circRNAs includes closed circular ncRNA molecules that are not easily degraded and have been shown to regulate the progression of various diseases (Li et al., 2015). For example, circRHOBTB3 is expressed at relatively low levels in hepatocellular carcinoma, and it has been reported that over-expression of circRHOBTB3 can lead to degradation of ELAVL1 and thus inhibition of the expression of the ELAVL1 target gene PTBP1 (Chen et al., 2021b). ELAVL1 also interacts competitively with circDLC1 to negatively regulate the expression of the gene encoding matrix metalloproteinase 1 (Liu et al., 2021); this mechanism occurs in a similar manner with regard to circPPM1F and its target gene PPM1F (Zhang et al., 2020) and circPABPN1 and its target gene ATG16L1 (Li et al., 2020).
The complex between circ-CCND1 and ELAVL1 promotes the expression of the gene encoding cyclin D1 and ultimately leads to the malignant proliferation of laryngeal squamous cell carcinoma (Zang et al., 2020). A similar mechanism has been found in the relationship among CircAGO2/ELAVL/AGO2 (Chen et al., 2019). Further studies have shown that circRNA-mediated positive regulation of downstream mRNAs may be achieved by promoting the cytoplasmic relocation of ELAVL1; thus, circBACH1 binds directly to ELAVL1 and mediates its translocation from the nucleus, thereby increasing its binding to the mRNA encoding p27 (Liu et al., 2020b). At the level of transcription, nuclear-localized circ-HuR derived from ELAVL1 can bind to the transcription factor cellular nucleic acid-binding protein to block this protein from binding to the ELAVL1 promoter, resulting in a decreased expression of ELAVL1 (Yang et al., 2019). In another regulatory mechanism, direct binding of Hsa_circ_00074854 to the ELAVL1 protein improves the stability of the protein, and ultimately promotes hepatocellular carcinoma migration, invasion and epithelial-mesenchymal transition (Wang et al., 2021). The interaction between ncRNA and ELAVL proteins is shown in Figure 2.
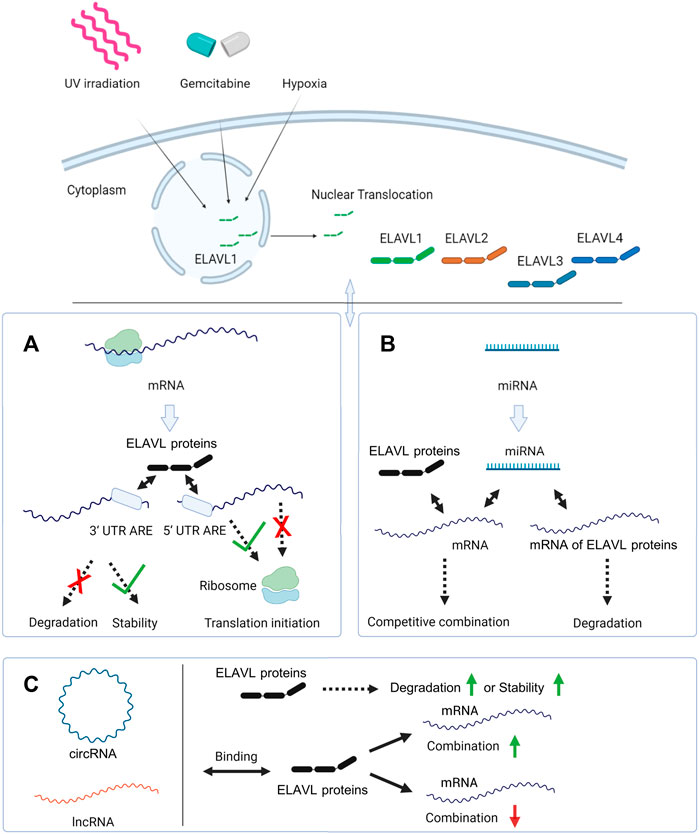
FIGURE 2. The interaction of ELAVL proteins with mRNAs and ncRNAs. In response to external stimuli like UV irradiation, gemcitabine or hypoxia, ELAVL1 is transported from the nucleus to the cytoplasm. And ELAVL2-4 are mainly localized in the cytoplasm. (A) ELAVL proteins inhibit mRNAs’ degradation and stabilize them by binding the 3′-UTR. In another regulation mode, ELAVL proteins can promote or inhibit mRNAs’ translation by binding the 5′-UTR; (B) miRNAs and ELAVL proteins co-regulate downstream mRNAs, and some miRNAs can regulate the expression of ELAVL proteins by binding their mRNAs. (C) circRNA and lncRNAs promote ELAVL proteins’ degradation or stabilize them by binding to them. And circRNAs and lncRNAs can also promote or inhibit the binding of ELAVL proteins to downstream mRNAs. Created with BioRender.com.
ELAVL Proteins in Pathological and Physiological Processes
ELAVL Proteins in Cancers
ELAVL1
ELAVL1 is indispensable to life. For example, the ELAVL1 gene is expressed during mouse embryonic development and growth cycle (Gouble and Morello, 2000), and the knockout of mouse ELAVL1 leads to hematopoietic failure, loss of intestinal villi, and death within 10 days (Ghosh et al., 2009). Thus, ELAVL1 protein is necessary in the maintenance of normal life processes. On the other hand, dysregulation of the expression of the ELAVL1 gene or the activity of the protein can also lead to aberrant cellular growth and cancer. A schematic of the relationships between ELAVL1 and cancer is shown in Figure 3.
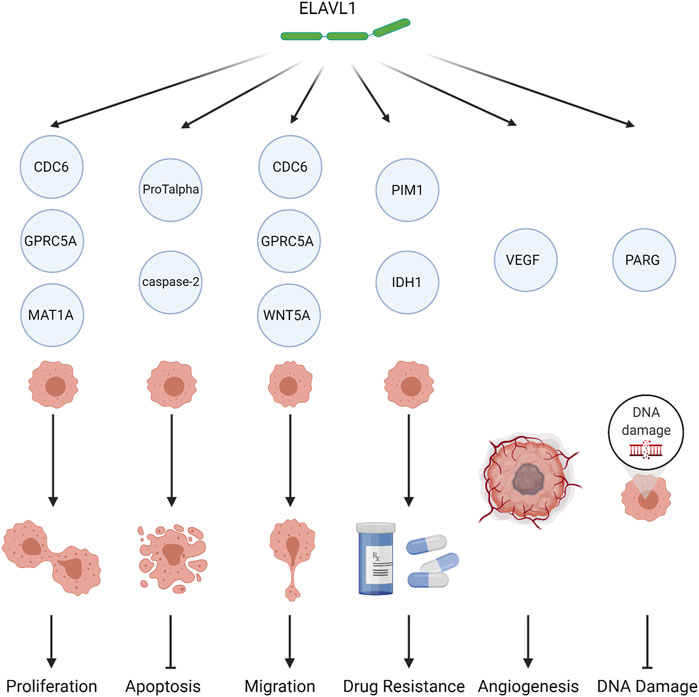
FIGURE 3. The role of ELAVL1 in tumors. ELAVL1 affects tumor biology by either (1) promoting proliferation, (2) inhibiting apoptosis, (3) promoting migration, (4) inducing drug resistance, (5) promoting angiogenesis, or (6) reducing DNA damage. Created with BioRender.com.
Colorectal Cancer
The functions of ELAVL1 in colorectal cancer have been studied extensively. Importantly, studies have connected ELAVL1 to cyclooxygenase 2 (COX-2), which has been shown through in vivo and in vitro studies to be a key factor in the malignant progression of colorectal cancer. As noted, ELAVL1 typically distributes mainly to the nucleus, but β-catenin can promote the cytoplasmic translocation of ELAVL1 (Lee and Jeong, 2006). Enrichment of ELAVL1 in the cytoplasm prolongs stabilization of the mRNA that encodes for COX-2 by binding with an ARE in the 3′-UTR, causing an increase in COX-2 protein levels (Dixon et al., 2001). Accordingly, the cytoplasmic localization of ELAVL1 has been shown to be significantly positively correlated with tumor stage (Denkert et al., 2006).
Interactions with the cell cycle also explain involvement of ELAVL1 in colorectal cancer. For example, ELAVL1 positively regulates the cell division 6 protein, which is highly expressed in colorectal cancer and which drives both the malignant behavior of colorectal cancer and its resistance to oxaliplatin (Cai et al., 2019). It has been reported that in the colorectal carcinoma RKO cell line, the levels of ELAVL1 in the cytoplasm increase during the late G1, S, and G2 phases of the cell cycle, and it binds to the 3′-UTR of mRNAs encoding cyclins A and B1, resulting in enhanced stability of these mRNAs and increased expression of the proteins, ultimately leading to increased cell proliferation (Wang et al., 2000). In animal experiments, overexpressing ELAVL1 in RKO cells results in increased tumor sizes upon injection into nude mice (de Silanes et al., 2003).
Ovarian Cancer
ELAVL1 can promote the expression level of COX-2 in ovarian cancer as well as in colorectal cancer, and COX-2 is also positively correlated with poor prognosis and high-grade of ovarian cancer. When the nuclear translocation of ELAVL1 is suppressed, the expression of COX-2 decreases in vitro (Erkinheimo et al., 2003). Accordingly, it has been found that levels of ELAVL1 in the cytoplasm of cells in ovarian cancer tissues was significantly increased relative to levels in borderline tumors or normal ovaries (Denkert et al., 2004). Another interaction with ovarian cancer involves miR-519, which targets the 3′-UTR of ELAVL1 mRNA, inhibiting its translation and slowing cell division of A2780 cells, a human ovarian cancer cell line, in vitro (Abdelmohsen et al., 2008). Also, in A2780 cells, glucose deprivation has been shown to enhance ELAVL1-dependent TUBB3 expression at the mRNA and protein levels, resulting in tumor invasion (Raspaglio et al., 2010).
Breast Cancer
ELAVL1 affects the development of breast cancer by regulating the mRNAs associated with a variety of proteins. Accordingly, among familial non-BRCA1/2 breast cancer patients, ELAVL1 can be used as an independent prognostic factor, associated with low survival rate and high tumor malignancy (Heinonen et al., 2007). Mechanistically, ELAVL1 promotes the expression of interleukin (IL)-8, which has clear connections with the progression of breast cancer, via binding to the 3′-UTR of the mRNA encoding IL-1β (Suswam et al., 2005). In addition, the abnormal expression of cyclin E1, Wnt-5a, thrombospondin 1 and the colony stimulating factor receptor is directly related to the increased expression of ELAVL1 in breast cancer models (Guo and Hartley, 2006; Leandersson et al., 2006; Mazan-Mamczarz et al., 2008; Woo et al., 2009). When ELAVL1 is silenced, the programmed death of tumor cells increases and invasion is inhibited (Heinonen et al., 2011). ELAVL1 is also regulated by upstream factors in breast cancer. Levels of miR-125 are negatively correlated with ELAVL1; miR-125 may thus act as an inhibitor of ELAVL1 to decrease translation by binding with its mRNA (Guo et al., 2009).
Pancreatic Cancer
The role of ELAVL1 in pancreatic cancer remain controversial. Overexpression of ELAVL1 in pancreatic cancer cells has been shown to increase the sensitivity of patients to gemcitabine treatment. The mechanism of this effect involves the binding of ELAVL1 to and the promotion effect of the mRNA that encodes deoxycytidine kinase whose products can activate gemcitabine (Costantino et al., 2009). In another report, ELAVL1 was also shown to be involved in the apoptosis of pancreatic cancer cells exposed to gemcitabine. ELAVL1 translocates to the cytoplasm after gemcitabine treatment, where it binds to the mRNA of retinoic acid-induced protein 3, which acts as an oncogene, leading to an increase of it at the initial stage of drug treatment (Zhou et al., 2016). Hypoxia in the tumor microenvironment can similarly induce nucleocytoplasmic shuttling of ELAVL1, which then promotes the expression of the PIM1 serine/threonine kinase, which leads to resistance to oxaliplatin (Blanco et al., 2016). In pancreatic ductal adenocarcinoma, ELAVL1 promotes the translation of poly (ADP-ribose) glycohydrolase mRNA, leading to enhanced DNA repair and resistance to the PARP inhibitor olaparib (Chand et al., 2017).
Other Types of Cancer
ELAVL1 has been found to play important roles in multiple kinds of tumors. For instance, ELAVL1 has been found to be highly expressed in prostate cancer, and it thus acts as an independent predictor positively correlating with tumor staging and metastasis. ELAVL1 promotes cell proliferation and migration of cells of the prostate cancer lines LNCaP and PC-3 by targeting vascular endothelial growth factors A and C and COX-2 (Barbisan et al., 2009; Mitsunari et al., 2016).
In hepatocellular carcinoma cells, the highly expressed lincRNA-UFC1 directly binds to ELAVL1, leading to an increase of β-catenin mRNA and protein and finally increased cell proliferation and decreased apoptosis (Cao et al., 2015). Similarly, hsa_circ_0074854 promotes the migration and invasion of hepatocellular carcinoma cells by stabilizing ELAVL1 (Wang et al., 2021). On another hand, ELAVL1 binds pre-miRNA-199a to prevent its maturation, leading to enhanced glycolytic metabolism in hepatocellular carcinoma cells in response to hypoxia (Zhang et al., 2015).
In glioblastoma multiforme and adjacent tissues, high expression of ELAVL1 can also be detected (Nabors et al., 2001). In glioblastoma, pyruvate kinase M2, which is up-regulated, binds to ELAVL1 and promotes its cytoplasmic localization, prompting tumor cells to enter a dividing state and promoting cell growth (Mukherjee et al., 2016). In a nude mouse model of glioblastoma, knockdown of ELAVL1 reduced tumor growth and proliferation, and prolonged survival time (Wang et al., 2019b).
Generally speaking, ELAVL1 often appears as a malignant factor. On the one hand, it is indispensable in life activities. On the other hand, the tumorigenic effects of high expression of ELAVL1 acts as an important contributor to the progression and invasion of many types of tumor through various pathways. Therefore, ELAVL1 may be a potential drug target with universal applicability.
ELAVL2-4
The three other family members, ELAVL2, ELAVL3 and ELAVL4, initially received much attention as neuroendocrine markers for small cell lung cancer (SCLC) (King, 1997; D'Alessandro et al., 2008). Among them, ELAVL4 received the most attention in SCLC, because it was found to be expressed in 100% of SCLC cells and more than 50% of neuroblastoma cells, and treatment targeting ELAVL4 can reduce tumor progression in nude mouse models (Ohwada et al., 1999; Ehrlich et al., 2014). As the antigen target of autoreactive CD4+ T cells, ELAVL4 may directly participate in cell-mediated anti-tumor immunity and nervous system damage (Benyahia et al., 1999).
At the level of post-transcriptional regulation, ELAVL4 regulates RNA as an RBP, but in tumorigenic neuroblasts, ELAVL4 also takes part in the nuclear processing and stability of the pre-mRNA of the proto-oncogenic transcription factor N-myc (Lazarova et al., 1999). ELAVL4 has also been shown to interact with both the 3′-UTR and 5′-UTR regions of the p27 mRNA to promote its translation leading to tumor suppression, but ELAVL4 and p27 levels are both reduced in pancreatic neuroendocrine tumors (Kim et al., 2018). Outside of SCLC, ELAVL2 has been shown to be an independent risk factor in esophageal squamous cell carcinoma, and it increases the resistance of these tumor cells to paclitaxel and cisplatin (Zhao et al., 2019).
There are also notable cancer-related interactions between members of the ELAVL family. A combination of ELAVL2 and ELAVL1 has been shown to localize to the nucleus and to be indispensable in the activation of several proto-oncogenes, including v-fos, v-ets, and v-myc (Hatanaka et al., 2019). Also, by binding with a structure containing an AU-rich sequence, ELAVL2 and ELAVL4 together inhibit the assembly of the core complex of telomerase to reduce its activity and cell growth in human neuroblastoma cells; notably, the activity of this complex antagonizes the function of ELAVL1 (Cheng et al., 2021). Surprisingly, considering the importance of the other ELAVL family members, few reports link ELAVL3 to tumor development or progression.
ELAVL Proteins in Disorders of the Nervous System
The ELAVL protein family was originally best known for its associations with the nervous system (Akamatsu et al., 1999). In the development of the neocortex, the deletion of ELAVL1 reduces the phosphorylation of eIF2a and eEF2 and the formation of polysomes, ultimately leading to the mis-localization of mRNAs. The lack of ELAVL1 reduces the stability of PFN1 mRNA and affects actin polymerization, resulting in the mis-localization of neurons in the neocortex (Kraushar et al., 2014; Zhao et al., 2020a). It also participates in cellular metabolism and protection from oxidation-induced neurodegeneration (Skliris et al., 2015).
With regard to various disease states, ELAVL1 has either protective or damaging effects, depending on the circumstances. In spinal muscle atrophy, ELAVL1 stabilizes survival motor neuron transcripts, which leads to accumulation of the protein products, thus alleviating the loss of alpha motor neurons that otherwise lead to progressive muscle atrophy (Farooq et al., 2009). In the occurrence and progress of neurodegenerative diseases, including age-related macular degeneration, ELAVL1 promotes the early elevation and accumulation of P62 in response to the early activation of autophagy, clearing protein multimers and alleviating neurodegenerative effects (Marchesi et al., 20182018).
On the other hand, ELAVL1 has been found to play deleterious roles in Huntington’s disease and amyotrophic lateral sclerosis. In Huntington’s disease, which is caused by mutations in the HTT gene and abnormal accumulation of the HTT protein, the HTT protein itself induces ELAVL1 to stabilize HTT mRNA, forming a positive feedback loop (Zhao et al., 2020b). Moreover, inhibition of ELAVL1 has been shown to block the chronic activation of microglia in amyotrophic lateral sclerosis and to delay the course of this disease (Matsye et al., 2017).
In neurons, ELAVL2, ELAVL3, and ELAVL4 function in stages. ELAVL2 protein is expressed in early neuron progenitor cells through to mature neurons, while ELAVL3 and 4 are expressed later than ELAVL2 and function mainly in cortical neuron development (Yano et al., 2016). Multiple studies have linked ELAVL4 to neuron development and plasticity (Bronicki et al., 2012; Loffreda et al., 2020). ELAVL4 is engaged in stabilization of tau microtubule-associated protein transcripts and maintains axon development in neuronal cells. Accordingly, the inhibition of ELAVL4 results in the cessation of axonal growth (Aranda-Abreu et al., 1999; Fukao et al., 2009; Hao le et al., 2017), and a deficiency of ELAVL4 in mice leads to transient impaired cranial nerve development during the embryonic period in vivo (Akamatsu et al., 2005).
Similar to ELAVL1, ELAVL4 is indispensable for establishing neocortex and hippocampal circuits and maintaining the function of these circuits (DeBoer et al., 2014). Furthermore, in the adult subventricular zone neural stem and progenitor cells, ELAVL4 promotes neuronal differentiation through special AT-rich sequence-binding protein 1 (SATB1). A lack of SATB1 affects the maturation of neuronal stem cells, and the overexpression of SATB1 in ELAVL4-suppressed cells can restore the neuronal differentiation phenotype (Wang et al., 2015). ELAVL2 promotes the exit from the cell cycle during the neuronal stem cell maturation, and overexpression of ELAVL2 restricts the proliferation of neuronal stem cells (Hambardzumyan et al., 2009). For ELAVL3, a low level of ELAVL3 leads to the impairment of spatial learning ability of mice with lowered expression of growth-associated protein 43 (Quattrone et al., 2001).
ELAVL Proteins in Other Physiological and Pathological Process
In addition to cancer and processes involving the nervous system, the ELAVL protein family is also involved in muscle differentiation (Beauchamp et al., 2010), aging (Lee et al., 2018), inflammation (Casolaro et al., 2008), stress events (Mazan-Mamczarz et al., 2003; Ahuja et al., 2016) and other processes. Figure 4 shows how tumors and other different diseases interact with ELAVL proteins.
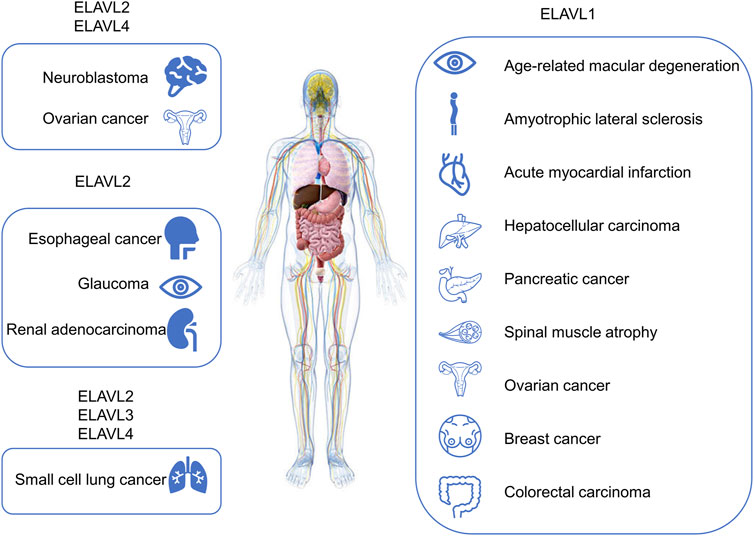
FIGURE 4. Diseases or pathophysiological processes related to ELAVL proteins. Diseases related to ELAVL proteins are included in the figure.
The impact on muscle differentiation involves a novel proteolytic cleavage of ELAVL1. When transferred to the cytoplasm in myoblasts, some ELAVL1 is cleaved into two fragments, cleavage products (CP) 1 and 2, of sizes 24 and 8 kDa, respectively. CP1 forms a complex with ELAVL1 import factor transportin-2 (TRN2), allowing uncleaved ELAVL1 to remain in the cytoplasm. The other fragment, CP2, promotes myogenesis (Beauchamp et al., 2010).
The known impact of ELAVL1 on aging involves interactions with the telomeric protein TIN2. ELAVL1 binding destabilizes TIN2 mRNA to decay quickly. Therefore, when ELAVL1 is inhibited, the expression of TIN2 protein in the mitochondria are increased. These factors correlate with increased levels of ROS, ultimately leading to cell senescence (Lee et al., 2018).
Inflammatory stimuli lead to poly ADP-ribosylation of D226 of ELAVL1. Modified ELAVL1 oligomerizes in the presence of PARP1, resulting in the protection of pro-inflammatory mRNA from degradation induced by miRNA and other factors. Therefore, treatments targeting ELAVL1 alleviate the lipopolysaccharide-induced accumulation of inflammatory cells in the airways of mice (Ke et al., 2021). In another inflammatory disease, pterygium, the activation of matrix metalloproteinase 9 by ELAVL1 amplifies the pro-inflammatory effect of IL-1β (Cui et al., 2020). The combination of ELAVL1 and matrix metalloproteinase 9 also appears in the inflammatory infiltration of cardiomyocytes in acute myocardial infarction. IL-10 can inhibit this combined effect to alleviate fibrosis and inflammation, and ultimately reduce damage to left ventricular function (Krishnamurthy et al., 2009).
Under the influence of different stressors, the function of ELAVL1 changes accordingly. Low-level UV irradiation induces the translocation of ELAVL1 to the cytoplasm, where it interacts with and preserves transcription products by binding with them after the formation of DNA damage. At the same time, the transcription process is inhibited to prevent the generation of false transcripts. In this mode, ELAVL1 is beneficial to cell survival (Mazan-Mamczarz et al., 2003). On the other hand, after ionizing radiation, ELAVL1 is separated from almost all mRNAs, including proliferation-related and apoptosis-related proteins. While this mechanism is different from that associated with UV irradiation, it similarly contributes to a better survival outcome for cells (Masuda et al., 2011). However, when cells are subjected to high pressure stress, ELAVL1 is translocated to the cytoplasm and where it is cleaved by caspases at A226, and this action amplifies apoptotic signals (Mazroui et al., 2008).
Inhibitors of ELAVL1
Research on the inhibitors of ELAVL proteins has mainly focused on ELAVL1. In view of the positive regulation of ELAVL1 in tumor promotion, the research and development of inhibitors is of significance. At present, the effects of inhibitors on ELAVL1 are mainly focused in four directions: inhibiting its nucleocytoplasmic trafficking, blocking its binding to mRNA, suppressing its dimerization/multimerization and downregulating its expression. As shown in Figure 5, MS-444 (Meisner et al., 2007), dehydromutactin (Meisner et al., 2007), okicenone (Meisner et al., 2007), eltrombopag (Zhu et al., 2020) and SRI-42127 (Filippova et al., 2021) inhibit the dimerization/multimerization of ELAVL1; 5-aza-2′-deoxycytidine (AZA) (Hostetter et al., 2008), trichostatin A (TSA) (Hostetter et al., 2008), pyrvinium pamoate (Guo et al., 2016) and Rottlerin (Latorre et al., 2012) inhibite ELAVL1’s nucleocytoplasmic trafficking; Dihydrotanshinone-I (Lal et al., 2017), azaphilone-9 (AZA-9) (Kaur et al., 2017), quercetin (Chae et al., 2009), b-40 (Chae et al., 2009), suramin (Kakuguchi et al., 2018), KH-3 (Wu et al., 2020) and CMLD1 (Wu et al., 2015) block ELAVL1’s binding to mRNA; CMLD2 downregulates ELAVL1’s expression (Muralidharan et al., 2017) and blocks its binding to mRNA (Wu et al., 2015). Among these inhibitors, MS-444, dehydromutactin, okicenone, SRI-42127, AZA-9, b-40, KH-3, CMLD1 and CMLD2 are specific inhibitors of ELAVL1, while others are not. For example, eltrombopag often acts as a thrombopoietin (TPO) receptor agonist (Bussel et al., 2019), AZA as a DNA methyltransferase inhibitor (Song et al., 2022) and TSA as a histone deacetylase inhibitor (He et al., 2022). Pyrvinium pamoate (Faheem et al., 2022), Rottlerin (Hufnagel et al., 2009), dihydrotanshinone-I (Sun et al., 2022), quercetin (Zaragozá et al., 2022) and suramin (Zhang et al., 2022) all have effects on other physiological or pathological processes.
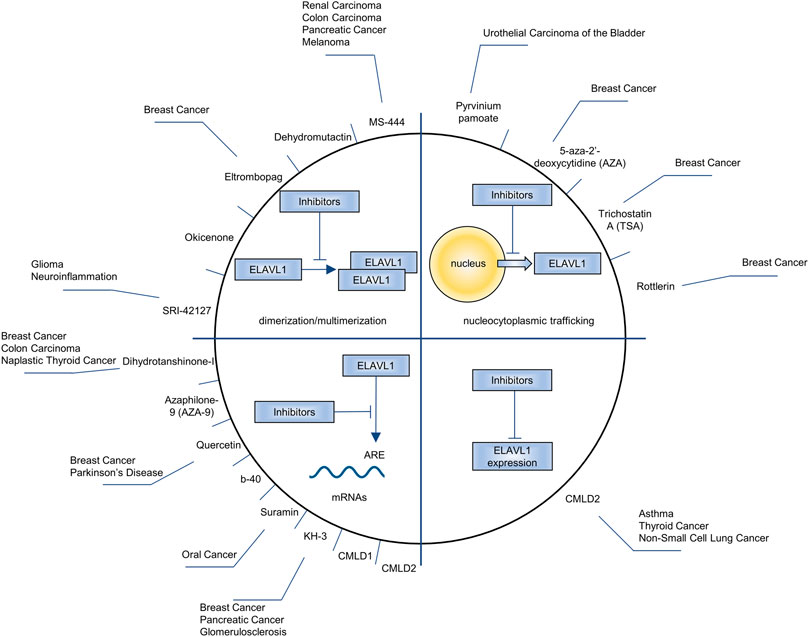
FIGURE 5. The drugs being used to inhibit ELAVL1, the processes how drugs affect ELAVL1 and the diseases the drugs aim to alleviate.
Conclusion and Perspective
This review summarizes the role of ELAVL proteins in various pathophysiological processes and in regulating mRNA and ncRNA. Given the ubiquity of ELAVL proteins existence and their role in a variety of diseases, it is logical to develop innovative small molecules. Based on the interaction mechanism between ncRNA and ELAVL proteins, the development of ncRNA analogs to promote the degradation of ELAVL proteins or inhibit the translation of their mRNAs is worth discussing. Developing chemically inactive analogs of ELAVL proteins to interfere with their synergy or competition with ncRNA in a competitive manner may also provide a new perspective to reduce the functionality of the protein family. Notably, although the ELAVL proteins have promising potential as a therapeutic target, many questions still need to be further explored. Interfering with ELAVL proteins seems to be a new strategy; however, considering that they are an integral part of life activities and interact with too many RNA molecules, whether their intervention will cause other unexpected side effects needs to be handled carefully. Is it better to use it alone or in combination with other drugs? According to previous reports, inhibition of ELAVL1 sensitized tumors to treatment with platinum-based drugs, including oxaliplatin and cisplatin (Young et al., 2009). What about drugs other than platinum-based drugs? In addition, considering ELAVL2-4 participating in many pathological processes and the similarity of the structure between ELAVL2-4 and ELAVL1, substantial study is imminent to fill the lacunae in the development of ELAVL2-4 inhibitors.
Author Contributions
HJC and DDZ collected literatures, prepared figures and tables, drafted the manuscript and edited the manuscript. YZY helped to arrange the manuscript. LHY, XYH and LXW conceived the idea and reviewed the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (82170061 and 82170062).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fcell.2022.847761/full#supplementary-material
References
Abdelmohsen, K., Srikantan, S., Kuwano, Y., and Gorospe, M. (2008). miR-519 Reduces Cell Proliferation by Lowering RNA-Binding Protein HuR Levels. Proc. Natl. Acad. Sci. U.S.A. 105, 20297–20302. doi:10.1073/pnas.0809376106
Ahuja, D., Goyal, A., and Ray, P. S. (2016). Interplay between RNA-Binding Protein HuR and microRNA-125b Regulates P53 mRNA Translation in Response to Genotoxic Stress. RNA Biol. 13, 1152–1165. doi:10.1080/15476286.2016.1229734
Akamatsu, W., Fujihara, H., Mitsuhashi, T., Yano, M., Shibata, S., Hayakawa, Y., et al. (2005). The RNA-Binding Protein HuD Regulates Neuronal Cell Identity and Maturation. Proc. Natl. Acad. Sci. U.S.A. 102, 4625–4630. doi:10.1073/pnas.0407523102
Akamatsu, W., Okano, H. J., Osumi, N., Inoue, T., Nakamura, S., Sakakibara, S.-I., et al. (1999). Mammalian ELAV-like Neuronal RNA-Binding Proteins HuB and HuC Promote Neuronal Development in Both the central and the Peripheral Nervous Systems. Proc. Natl. Acad. Sci. 96, 9885–9890. doi:10.1073/pnas.96.17.9885
Al-Haidari, A., Algaber, A., Madhi, R., Syk, I., and Thorlacius, H. (2018). MiR-155-5p Controls colon Cancer Cell Migration via post-transcriptional Regulation of Human Antigen R (HuR). Cancer Lett. 421, 145–151. doi:10.1016/j.canlet.2018.02.026
Ale-Agha, N., Galban, S., Sobieroy, C., Abdelmohsen, K., Gorospe, M., Sies, H., et al. (2009). HuR Regulates gap Junctional Intercellular Communication by Controlling β-catenin Levels and Adherens junction Integrity. Hepatology 50, 1567–1576. doi:10.1002/hep.23146
Aranda-Abreu, G. E., Behar, L., Chung, S., Furneaux, H., and Ginzburg, I. (1999). Embryonic Lethal Abnormal Vision-like RNA-Binding Proteins Regulate Neurite Outgrowth and Tau Expression in PC12 Cells. J. Neurosci. 19, 6907–6917. doi:10.1523/jneurosci.19-16-06907.1999
Bakheet, T., Williams, B. R., and Khabar, K. S. (2006). ARED 3.0: the Large and Diverse AU-Rich Transcriptome. Nucleic Acids Res. 34, D111–D114. doi:10.1093/nar/gkj052
Barbisan, F., Mazzucchelli, R., Santinelli, A., Lopez-Beltran, A., Cheng, L., Scarpelli, M., et al. (2009). Overexpression of ELAV-like Protein HuR Is Associated with Increased COX-2 Expression in Atrophy, High-Grade Prostatic Intraepithelial Neoplasia, and Incidental Prostate Cancer in Cystoprostatectomies. Eur. Urol. 56, 105–112. doi:10.1016/j.eururo.2008.04.043
Barreau, C., Paillard, L., and Osborne, H. B. (2005). AU-rich Elements and Associated Factors: Are There Unifying Principles? Nucleic Acids Res. 33, 7138–7150. doi:10.1093/nar/gki1012
Beauchamp, P., Nassif, C., Hillock, S., van der Giessen, K., von Roretz, C., Jasmin, B. J., et al. (2010). The Cleavage of HuR Interferes with its Transportin-2-Mediated Nuclear Import and Promotes Muscle Fiber Formation. Cell Death Differ 17, 1588–1599. doi:10.1038/cdd.2010.34
Benyahia, B., Liblau, R., Merle-Béral, H. l. n., Tourani, J.-M., Dalmau, J., and Delattre, J.-Y. (1999). Cell-mediated Autoimmunity in Paraneoplastic Neurological Syndromes with Anti-hu Antibodies. Ann. Neurol. 45, 162–167. doi:10.1002/1531-8249(199902)45:2<162::aid-ana5>3.0.co;2-r
Bibli, S.-I., Hu, J., Sigala, F., Wittig, I., Heidler, J., Zukunft, S., et al. (2019). Cystathionine γ Lyase Sulfhydrates the RNA Binding Protein Human Antigen R to Preserve Endothelial Cell Function and Delay Atherogenesis. Circulation 139, 101–114. doi:10.1161/circulationaha.118.034757
Blanco, F. F., Jimbo, M., Wulfkuhle, J., Gallagher, I., Deng, J., Enyenihi, L., et al. (2016). The mRNA-Binding Protein HuR Promotes Hypoxia-Induced Chemoresistance through Posttranscriptional Regulation of the Proto-Oncogene PIM1 in Pancreatic Cancer Cells. Oncogene 35, 2529–2541. doi:10.1038/onc.2015.325
Bronicki, L. M., Belanger, G., and Jasmin, B. J. (2012). Characterization of Multiple Exon 1 Variants in Mammalian HuD mRNA and Neuron-specific Transcriptional Control via Neurogenin 2. J. Neurosci. 32, 11164–11175. doi:10.1523/jneurosci.2247-12.2012
Bussel, J., Kulasekararaj, A., Cooper, N., Verma, A., Steidl, U., Semple, J. W., et al. (2019). Mechanisms and Therapeutic Prospects of Thrombopoietin Receptor Agonists. Semin. Hematol. 56, 262–278. doi:10.1053/j.seminhematol.2019.09.001
Cai, J., Wang, H., Jiao, X., Huang, R., Qin, Q., Zhang, J., et al. (2019). The RNA-Binding Protein HuR Confers Oxaliplatin Resistance of Colorectal Cancer by Upregulating CDC6. Mol. Cancer Ther. 18, 1243–1254. doi:10.1158/1535-7163.Mct-18-0945
Campos, A. R., And, D. G., and White, K. (1985). Mutant Alleles at the Locus Elav in Drosophila melanogaster lead to Nervous System Defects. A Developmental-Genetic Analysis. J. Neurogenet. 2, 197–218. doi:10.3109/01677068509100150
Cao, C., Sun, J., Zhang, D., Guo, X., Xie, L., Li, X., et al. (2015). The Long Intergenic Noncoding RNA UFC1, a Target of MicroRNA 34a, Interacts with the mRNA Stabilizing Protein HuR to Increase Levels of β-Catenin in HCC Cells. Gastroenterology 148, 415–426. doi:10.1053/j.gastro.2014.10.012
Casolaro, V., Fang, X., Tancowny, B., Fan, J., Wu, F., Srikantan, S., et al. (2008). Posttranscriptional Regulation of IL-13 in T Cells: Role of the RNA-Binding Protein HuR. J. Allergy Clin. Immunol. 121, 853–859. e854. doi:10.1016/j.jaci.2007.12.1166
Chae, M.-J., Sung, H. Y., Kim, E.-H., Lee, M., Kwak, H., Chae, C. H., et al. (2009). Chemical Inhibitors Destabilize HuR Binding to the AU-Rich Element of TNF-α mRNA. Exp. Mol. Med. 41, 824–831. doi:10.3858/emm.2009.41.11.088
Chand, S. N., Zarei, M., Schiewer, M. J., Kamath, A. R., Romeo, C., Lal, S., et al. (2017). Posttranscriptional Regulation of PARG mRNA by HuR Facilitates DNA Repair and Resistance to PARP Inhibitors. Cancer Res. 77, 5011–5025. doi:10.1158/0008-5472.Can-16-2704
Chen, C. Y., Xu, N., and Shyu, A. B. (1995). mRNA Decay Mediated by Two Distinct AU-Rich Elements from C-Fos and Granulocyte-Macrophage colony-stimulating Factor Transcripts: Different Deadenylation Kinetics and Uncoupling from Translation. Mol. Cell Biol 15, 5777–5788. doi:10.1128/mcb.15.10.5777
Chen, J., Wu, Y., Luo, X., Jin, D., Zhou, W., Ju, Z., et al. (2021). Circular RNA circRHOBTB3 Represses Metastasis by Regulating the HuR-Mediated mRNA Stability of PTBP1 in Colorectal Cancer. Theranostics 11, 7507–7526. doi:10.7150/thno.59546
Chen, R., Lei, S., She, Y., Zhou, S., Shi, H., Li, C., et al. (2021). Lnc-GD2H Promotes Proliferation by Forming a Feedback Loop with C-Myc and Enhances Differentiation through Interacting with NACA to Upregulate Myog in C2C12 Myoblasts. Front. Cell Dev. Biol. 9, 671857. doi:10.3389/fcell.2021.671857
Chen, Y., Yang, F., Fang, E., Xiao, W., Mei, H., Li, H., et al. (2019). Circular RNA circAGO2 Drives Cancer Progression through Facilitating HuR-Repressed Functions of AGO2-miRNA Complexes. Cell Death Differ 26, 1346–1364. doi:10.1038/s41418-018-0220-6
Cheng, X., Gu, X., Xia, T., Ma, Z., Yang, Z., Feng, H. L., et al. (2021). HuB and HuD Repress Telomerase Activity by Dissociating HuR from TERC. Nucleic Acids Res. 49, 2848–2858. doi:10.1093/nar/gkab062
Colombrita, C., Silani, V., and Ratti, A. (2013). ELAV Proteins along Evolution: Back to the Nucleus? Mol. Cell Neurosci. 56, 447–455. doi:10.1016/j.mcn.2013.02.003
Costantino, C. L., Witkiewicz, A. K., Kuwano, Y., Cozzitorto, J. A., Kennedy, E. P., Dasgupta, A., et al. (2009). The Role of HuR in Gemcitabine Efficacy in Pancreatic Cancer: HuR Up-Regulates the Expression of the Gemcitabine Metabolizing Enzyme Deoxycytidine Kinase. Cancer Res. 69, 4567–4572. doi:10.1158/0008-5472.Can-09-0371
Cui, Y. H., Feng, Q. Y., Liu, Q., Li, H. Y., Song, X. L., Hu, Z. X., et al. (2020). Posttranscriptional Regulation of MMP‐9 by HuR Contributes to IL‐1β‐induced Pterygium Fibroblast Migration and Invasion. J. Cell Physiol 235, 5130–5140. doi:10.1002/jcp.29387
D'Alessandro, V., Muscarella, L. A., Copetti, M., Zelante, L., Carella, M., and Vendemiale, G. (2008). Molecular Detection of Neuron-specific ELAV-Like-Positive Cells in the Peripheral Blood of Patients with Small-Cell Lung Cancer. Cell Oncol 30, 291–297. doi:10.3233/clo-2008-0424
De Santis, R., Santini, L., Colantoni, A., Peruzzi, G., de Turris, V., Alfano, V., et al. (2017). FUS Mutant Human Motoneurons Display Altered Transcriptome and microRNA Pathways with Implications for ALS Pathogenesis. Stem Cell Rep. 9, 1450–1462. doi:10.1016/j.stemcr.2017.09.004
de Silanes, I. L., Fan, J., Yang, X., Zonderman, A. B., Potapova, O., Pizer, E. S., et al. (2003). Role of the RNA-Binding Protein HuR in colon Carcinogenesis. Oncogene 22, 7146–7154. doi:10.1038/sj.onc.1206862
DeBoer, E. M., Azevedo, R., Vega, T. A., Brodkin, J., Akamatsu, W., Okano, H., et al. (2014). Prenatal Deletion of the RNA-Binding Protein HuD Disrupts Postnatal Cortical Circuit Maturation and Behavior. J. Neurosci. 34, 3674–3686. doi:10.1523/jneurosci.3703-13.2014
Denkert, C., Koch, I., von Keyserlingk, N., Noske, A., Niesporek, S., Dietel, M., et al. (2006). Expression of the ELAV-like Protein HuR in Human colon Cancer: Association with Tumor Stage and Cyclooxygenase-2. Mod. Pathol. 19, 1261–1269. doi:10.1038/modpathol.3800645
Denkert, C., Weichert, W., Pest, S., Koch, I., Licht, D., Köbel, M., et al. (2004). Overexpression of the Embryonic-Lethal Abnormal Vision-like Protein HuR in Ovarian Carcinoma Is a Prognostic Factor and Is Associated with Increased Cyclooxygenase 2 Expression. Cancer Res. 64, 189–195. doi:10.1158/0008-5472.can-03-1987
Deschenes-Furry, J., Mousavi, K., Bolognani, F., Neve, R. L., Parks, R. J., Perrone-Bizzozero, N. I., et al. (2007). The RNA-Binding Protein HuD Binds Acetylcholinesterase mRNA in Neurons and Regulates its Expression after Axotomy. J. Neurosci. 27, 665–675. doi:10.1523/jneurosci.4626-06.2007
Dixon, D. A., Tolley, N. D., King, P. H., Nabors, L. B., McIntyre, T. M., Zimmerman, G. A., et al. (2001). Altered Expression of the mRNA Stability Factor HuR Promotes Cyclooxygenase-2 Expression in colon Cancer Cells. J. Clin. Invest. 108, 1657–1665. doi:10.1172/jci12973
Dou, Q., Xu, Y., Zhu, Y., Hu, Y., Yan, Y., and Yan, H. (2019). LncRNA FAM83H-AS1 Contributes to the Radioresistance, Proliferation, and Metastasis in Ovarian Cancer through Stabilizing HuR Protein. Eur. J. Pharmacol. 852, 134–141. doi:10.1016/j.ejphar.2019.03.002
Durie, D., Lewis, S. M., Liwak, U., Kisilewicz, M., Gorospe, M., and Holcik, M. (2011). RNA-binding Protein HuR Mediates Cytoprotection through Stimulation of XIAP Translation. Oncogene 30, 1460–1469. doi:10.1038/onc.2010.527
Ehrlich, D., Wang, B., Lu, W., Dowling, P., and Yuan, R. (2014). Intratumoral Anti-HuD Immunotoxin Therapy for Small Cell Lung Cancer and Neuroblastoma. J. Hematol. Oncol. 7, 91. doi:10.1186/s13045-014-0091-3
Erkinheimo, T. L., Lassus, H., Sivula, A., Sengupta, S., Furneaux, H., Hla, T., et al. (2003). Cytoplasmic HuR Expression Correlates with Poor Outcome and with Cyclooxygenase 2 Expression in Serous Ovarian Carcinoma. Cancer Res. 63, 7591–7594. http://aacrjournals.org/cancerres/article-pdf/63/22/7591/2509724/zch02203007591.pdf.
Faheem, S. A., El- Sayed, N. M., Moustafa, Y. M., Saeed, N. M., and Hazem, R. M. (2022). Pyrvinium Pamoate Ameliorates Cyclosporin A- Induced Hepatotoxicity via the Modulation of Wnt/β-Catenin Signaling and Upregulation of PPAR-γ. Int. Immunopharmacology 104, 108538. doi:10.1016/j.intimp.2022.108538
Fan, X. C., Myer, V. E., and Steitz, J. A. (1997). AU-rich Elements Target Small Nuclear RNAs as Well as mRNAs for Rapid Degradation. Genes Dev. 11, 2557–2568. doi:10.1101/gad.11.19.2557
Fan, X. C., and Steitz, J. A. (1998). Overexpression of HuR, a Nuclear-Cytoplasmic Shuttling Protein, Increases the Invivo Stability of ARE-Containing mRNAs. Embo j 17, 3448–3460. doi:10.1093/emboj/17.12.3448
Fan, X. C., and Steitz, J. A. (1998). HNS, a Nuclear-Cytoplasmic Shuttling Sequence in HuR. Proc. Natl. Acad. Sci. 95, 15293–15298. doi:10.1073/pnas.95.26.15293
Farooq, F., Balabanian, S., Liu, X., Holcik, M., and MacKenzie, A. (2009). p38 Mitogen-Activated Protein Kinase Stabilizes SMN mRNA through RNA Binding Protein HuR. Hum. Mol. Genet. 18, 4035–4045. doi:10.1093/hmg/ddp352
Filippova, N., Yang, X., Ananthan, S., Calano, J., Pathak, V., Bratton, L., et al. (2021). Targeting the HuR Oncogenic Role with a New Class of Cytoplasmic Dimerization Inhibitors. Cancer Res. 81, 2220–2233. doi:10.1158/0008-5472.Can-20-2858
Fukao, A., Sasano, Y., Imataka, H., Inoue, K., Sakamoto, H., Sonenberg, N., et al. (2009). The ELAV Protein HuD Stimulates Cap-dependent Translation in a Poly(A)- and eIF4A-dependent Manner. Mol. Cell 36, 1007–1017. doi:10.1016/j.molcel.2009.11.013
Gherzi, R., Lee, K.-Y., Briata, P., Wegmüller, D., Moroni, C., Karin, M., et al. (2004). A KH Domain RNA Binding Protein, KSRP, Promotes ARE-Directed mRNA Turnover by Recruiting the Degradation Machinery. Mol. Cell 14, 571–583. doi:10.1016/j.molcel.2004.05.002
Ghosh, M., Aguila, H. L., Michaud, J., Ai, Y., Wu, M.-T., Hemmes, A., et al. (2009). Essential Role of the RNA-Binding Protein HuR in Progenitor Cell Survival in Mice. J. Clin. Invest. 119, 3530–3543. doi:10.1172/jci38263
Good, P. J. (1995). A Conserved Family of Elav-like Genes in Vertebrates. Proc. Natl. Acad. Sci. 92, 4557–4561. doi:10.1073/pnas.92.10.4557
Gouble, A., and Morello, D. (2000). Synchronous and Regulated Expression of Two AU-Binding Proteins, AUF1 and HuR, throughout Murine Development. Oncogene 19, 5377–5384. doi:10.1038/sj.onc.1203910
Gratacós, F. M., and Brewer, G. (2010). The Role of AUF1 in Regulated mRNA Decay. Wiley Interdiscip. Rev. RNA 1, 457–473. doi:10.1002/wrna.26
Guo, J., Lv, J., Chang, S., Chen, Z., Lu, W., Xu, C., et al. (2016). Inhibiting Cytoplasmic Accumulation of HuR Synergizes Genotoxic Agents in Urothelial Carcinoma of the Bladder. Oncotarget 7, 45249–45262. doi:10.18632/oncotarget.9932
Guo, X., and Hartley, R. S. (2006). HuR Contributes to Cyclin E1 Deregulation in MCF-7 Breast Cancer Cells. Cancer Res. 66, 7948–7956. doi:10.1158/0008-5472.Can-05-4362
Guo, X., Wu, Y., and Hartley, R. (2009). MicroRNA-125a Represses Cell Growth by Targeting HuR in Breast Cancer. RNA Biol. 6, 575–583. doi:10.4161/rna.6.5.10079
Hambardzumyan, D., Sergent-Tanguy, S., Thinard, R., Bonnamain, V., Masip, M., Fabre, A., et al. (2009). AUF1 and Hu Proteins in the Developing Rat Brain: Implication in the Proliferation and Differentiation of Neural Progenitors. J. Neurosci. Res. 87, 1296–1309. doi:10.1002/jnr.21957
Hao le, T., Duy, P. Q., An, M., Talbot, J., Iyer, C. C., Wolman, M., et al. (2017). HuD and the Survival Motor Neuron Protein Interact in Motoneurons and Are Essential for Motoneuron Development, Function, and mRNA Regulation. J. Neurosci. 37, 11559–11571. doi:10.1523/jneurosci.1528-17.2017
Hatanaka, T., Higashino, F., Tei, K., and Yasuda, M. (2019). The Neural ELAVL Protein HuB Enhances Endogenous Proto-Oncogene Activation. Biochem. Biophysical Res. Commun. 517, 330–337. doi:10.1016/j.bbrc.2019.07.089
He, X. B., Wu, Y., Huang, H., and Guo, F. (2022). A Novel Histone Deacetylase Inhibitor‐based Approach to Eliminate Microglia and Retain Astrocyte Properties in Glial Cell Culture. J. Neurochem. doi:10.1111/jnc.15581
Heinonen, M., Fagerholm, R., Aaltonen, K., Kilpivaara, O., Aittomäki, K., Blomqvist, C., et al. (2007). Prognostic Role of HuR in Hereditary Breast Cancer. Clin. Cancer Res. 13, 6959–6963. doi:10.1158/1078-0432.Ccr-07-1432
Heinonen, M., Hemmes, A., Salmenkivi, K., Abdelmohsen, K., Vilén, S.-T., Laakso, M., et al. (2011). Role of RNA Binding Protein HuR in Ductal Carcinoma In Situ of the Breast. J. Pathol. 224, 529–539. doi:10.1002/path.2889
Hostetter, C., Licata, L. A., Costantino, C. L., Witkiewicz, A., Yeo, C., Brody, J. R., et al. (2008). Cytoplasmic Accumulation of the RNA Binding Protein HuR Is central to Tamoxifen Resistance in Estrogen Receptor Positive Breast Cancer Cells. Cancer Biol. Ther. 7, 1496–1506. doi:10.4161/cbt.7.9.6490
Hufnagel, H., Hakim, P., Lima, A., and Hollfelder, F. (2009). Fluid Phase Endocytosis Contributes to Transfection of DNA by PEI-25. Mol. Ther. 17, 1411–1417. doi:10.1038/mt.2009.121
Ishihara, Y., Tsuno, S., Kuwamoto, S., Yamashita, T., Endo, Y., Hasegawa, J., et al. (2014). Hsa-miR-520d Converts Fibroblasts into CD105+ Populations. Drugs R. D 14, 253–264. doi:10.1007/s40268-014-0064-6
Kakuguchi, W., Nomura, T., Kitamura, T., Otsuguro, S., Matsushita, K., Sakaitani, M., et al. (2018). Suramin, Screened from an Approved Drug Library, Inhibits HuR Functions and Attenuates Malignant Phenotype of Oral Cancer Cells. Cancer Med. 7, 6269–6280. doi:10.1002/cam4.1877
Kang, M. J., Ryu, B. K., Lee, M. G., Han, J., Lee, J. H., Ha, T. K., et al. (2008). NF-κB Activates Transcription of the RNA-Binding Factor HuR, via PI3K-AKT Signaling, to Promote Gastric Tumorigenesis. Gastroenterology 135, 2030–2042. doi:10.1053/j.gastro.2008.08.009
Kaur, K., Wu, X., Fields, J. K., Johnson, D. K., Lan, L., Pratt, M., et al. (2017). The Fungal Natural Product Azaphilone-9 Binds to HuR and Inhibits HuR-RNA Interaction In Vitro. PLoS One 12, e0175471. doi:10.1371/journal.pone.0175471
Ke, Y., Han, Y., Guo, X., Wen, J., Wang, K., Jiang, X., et al. (2017). PARP1 Promotes Gene Expression at the post-transcriptional Level by Modulating the RNA-Binding Protein HuR. Nat. Commun. 8, 14632. doi:10.1038/ncomms14632
Ke, Y., Lv, X., Fu, X., Zhang, J., Bohio, A. A., Zeng, X., et al. (2021). Poly(ADP-ribosyl)ation Enhances HuR Oligomerization and Contributes to Pro-inflammatory Gene mRNA Stabilization. Cell. Mol. Life Sci. 78, 1817–1835. doi:10.1007/s00018-020-03618-4
Kim, C., Jeong, D. E., Heo, S., Ji, E., Rho, J. G., Jung, M., et al. (2018). Reduced Expression of the RNA-Binding Protein HuD in Pancreatic Neuroendocrine Tumors Correlates with Low p27Kip1 Levels and Poor Prognosis. J. Pathol. 246, 231–243. doi:10.1002/path.5135
Kim, H. H., Abdelmohsen, K., Lal, A., Pullmann, R., Yang, X., Galban, S., et al. (2008). Nuclear HuR Accumulation through Phosphorylation by Cdk1. Genes Dev. 22, 1804–1815. doi:10.1101/gad.1645808
King, P. H. (1997). Differential Expression of the Neuroendocrine genesHel-N1 and HuD in Small-Cell Lung Carcinoma: Evidence for Down-Regulation of HuD in the Variant Phenotype. Int. J. Cancer 74, 378–382. doi:10.1002/(sici)1097-0215(19970822)74:4<378::aid-ijc3>3.0.co;2-s
Kraushar, M. L., Thompson, K., Wijeratne, H. R. S., Viljetic, B., Sakers, K., Marson, J. W., et al. (2014). Temporally Defined Neocortical Translation and Polysome Assembly Are Determined by the RNA-Binding Protein Hu Antigen R. Proc. Natl. Acad. Sci. U.S.A. 111, E3815–E3824. doi:10.1073/pnas.1408305111
Krishnamurthy, P., Rajasingh, J., Lambers, E., Qin, G., Losordo, D. W., and Kishore, R. (2009). IL-10 Inhibits Inflammation and Attenuates Left Ventricular Remodeling after Myocardial Infarction via Activation of STAT3 and Suppression of HuR. Circ. Res. 104, e9–18. doi:10.1161/circresaha.108.188243
Lal, P., Cerofolini, L., D’Agostino, V. G., Zucal, C., Fuccio, C., Bonomo, I., et al. (2017). Regulation of HuR Structure and Function by Dihydrotanshinone-I. Nucleic Acids Res. 45, 9514–9527. doi:10.1093/nar/gkx623
Lan, Y., Xiao, X., He, Z., Luo, Y., Wu, C., Li, L., et al. (2018). Long Noncoding RNA OCC-1 Suppresses Cell Growth through Destabilizing HuR Protein in Colorectal Cancer. Nucleic Acids Res. 46, 5809–5821. doi:10.1093/nar/gky214
Latorre, E., Tebaldi, T., Viero, G., Spartà, A. M., Quattrone, A., and Provenzani, A. (2012). Downregulation of HuR as a New Mechanism of Doxorubicin Resistance in Breast Cancer Cells. Mol. Cancer 11, 13. doi:10.1186/1476-4598-11-13
Lazarova, D. L., Spengler, B. A., Biedler, J. L., and Ross, R. A. (1999). HuD, a Neuronal-specific RNA-Binding Protein, Is a Putative Regulator of N-Myc Pre-mRNA Processing/stability in Malignant Human Neuroblasts. Oncogene 18, 2703–2710. doi:10.1038/sj.onc.1202621
Leandersson, K., Riesbeck, K., and Andersson, T. (2006). Wnt-5a mRNA Translation Is Suppressed by the Elav-like Protein HuR in Human Breast Epithelial Cells. Nucleic Acids Res. 34, 3988–3999. doi:10.1093/nar/gkl571
Lee, H. K., and Jeong, S. (2006). β-Catenin Stabilizes Cyclooxygenase-2 mRNA by Interacting with AU-Rich Elements of 3′-UTR. Nucleic Acids Res. 34, 5705–5714. doi:10.1093/nar/gkl698
Lee, J. H., Jung, M., Hong, J., Kim, M. K., and Chung, I. K. (2018). Loss of RNA-Binding Protein HuR Facilitates Cellular Senescence through Posttranscriptional Regulation of TIN2 mRNA. Nucleic Acids Res. 46, 4271–4285. doi:10.1093/nar/gky223
Legnini, I., Morlando, M., Mangiavacchi, A., Fatica, A., and Bozzoni, I. (2014). A Feedforward Regulatory Loop between HuR and the Long Noncoding RNA linc-MD1 Controls Early Phases of Myogenesis. Mol. Cell 53, 506–514. doi:10.1016/j.molcel.2013.12.012
Li, X.-X., Xiao, L., Chung, H. K., Ma, X.-X., Liu, X., Song, J.-L., et al. (2020). Interaction between HuR and circPABPN1 Modulates Autophagy in the Intestinal Epithelium by Altering ATG16L1 Translation. Mol. Cell Biol 40 (6), e00492-19. doi:10.1128/mcb.00492-19
Li, Y., Zheng, Q., Bao, C., Li, S., Guo, W., Zhao, J., et al. (2015). Circular RNA Is Enriched and Stable in Exosomes: a Promising Biomarker for Cancer Diagnosis. Cell Res 25, 981–984. doi:10.1038/cr.2015.82
Liu, B., Yang, G., Wang, X., Liu, J., Lu, Z., Wang, Q., et al. (2020). CircBACH1 (Hsa_circ_0061395) Promotes Hepatocellular Carcinoma Growth by Regulating P27 Repression via HuR. J. Cell Physiol 235, 6929–6941. doi:10.1002/jcp.29589
Liu, H., Lan, T., Li, H., Xu, L., Chen, X., Liao, H., et al. (2021). Circular RNA circDLC1 Inhibits MMP1-Mediated Liver Cancer Progression via Interaction with HuR. Theranostics 11, 1396–1411. doi:10.7150/thno.53227
Liu, L., Xiao, L., Chung, H. K., Kwon, M. S., Li, X.-X., Wu, N., et al. (2019). RNA-binding Protein HuR Regulates Rac1 Nucleocytoplasmic Shuttling through Nucleophosmin in the Intestinal Epithelium. Cell Mol. Gastroenterol. Hepatol. 8, 475–486. doi:10.1016/j.jcmgh.2019.06.002
Liu, S., Jiang, X., Lu, H., Xing, M., Qiao, Y., Zhang, C., et al. (2020). HuR (Human Antigen R) Regulates the Contraction of Vascular Smooth Muscle and Maintains Blood Pressure. Atvb 40, 943–957. doi:10.1161/atvbaha.119.313897
Loffreda, A., Nizzardo, M., Arosio, A., Ruepp, M.-D., Calogero, R. A., Volinia, S., et al. (2020). miR-129-5p: A Key Factor and Therapeutic Target in Amyotrophic Lateral Sclerosis. Prog. Neurobiol. 190, 101803. doi:10.1016/j.pneurobio.2020.101803
Lu, Y.-C., Chang, S.-H., Hafner, M., Li, X., Tuschl, T., Elemento, O., et al. (2014). ELAVL1 Modulates Transcriptome-wide miRNA Binding in Murine Macrophages. Cell Rep. 9, 2330–2343. doi:10.1016/j.celrep.2014.11.030
Lucchesi, C., Sheikh, M. S., and Huang, Y. (2016). Negative Regulation of RNA-Binding Protein HuR by Tumor-Suppressor ECRG2. Oncogene 35, 2565–2573. doi:10.1038/onc.2015.339
Lykke-Andersen, J., and Wagner, E. (2005). Recruitment and Activation of mRNA Decay Enzymes by Two ARE-Mediated Decay Activation Domains in the Proteins TTP and BRF-1. Genes Dev. 19, 351–361. doi:10.1101/gad.1282305
Marchesi, N., Thongon, N., Pascale, A., Provenzani, A., Koskela, A., Korhonen, E., et al. (2018). Autophagy Stimulus Promotes Early HuR Protein Activation and p62/SQSTM1 Protein Synthesis in ARPE-19 Cells by Triggering Erk1/2, p38MAPK, and JNK Kinase Pathways. Oxidative Med. Cell. longevity 2018, 1–15. doi:10.1155/2018/4956080
Masuda, K., Abdelmohsen, K., Kim, M. M., Srikantan, S., Lee, E. K., Tominaga, K., et al. (2011). Global Dissociation of HuR-mRNA Complexes Promotes Cell Survival after Ionizing Radiation. Embo j 30, 1040–1053. doi:10.1038/emboj.2011.24
Matsye, P., Zheng, L., Si, Y., Kim, S., Luo, W., Crossman, D. K., et al. (2017). HuR Promotes the Molecular Signature and Phenotype of Activated Microglia: Implications for Amyotrophic Lateral Sclerosis and Other Neurodegenerative Diseases. Glia 65, 945–963. doi:10.1002/glia.23137
Mazan-Mamczarz, K., Galbán, S., de Silanes, I. L., Martindale, J. L., Atasoy, U., Keene, J. D., et al. (2003). RNA-binding Protein HuR Enhances P53 Translation in Response to Ultraviolet Light Irradiation. Proc. Natl. Acad. Sci. U.S.A. 100, 8354–8359. doi:10.1073/pnas.1432104100
Mazan-Mamczarz, K., Hagner, P. R., Corl, S., Srikantan, S., Wood, W. H., Becker, K. G., et al. (2008). Post-transcriptional Gene Regulation by HuR Promotes a More Tumorigenic Phenotype. Oncogene 27, 6151–6163. doi:10.1038/onc.2008.215
Mazroui, R., Di Marco, S., Clair, E., von Roretz, C., Tenenbaum, S. A., Keene, J. D., et al. (2008). Caspase-mediated Cleavage of HuR in the Cytoplasm Contributes to Pp32/PHAP-I Regulation of Apoptosis. J. Cell Biol 180, 113–127. doi:10.1083/jcb.200709030
Meisner, N.-C., Hintersteiner, M., Mueller, K., Bauer, R., Seifert, J.-M., Naegeli, H.-U., et al. (2007). Identification and Mechanistic Characterization of Low-Molecular-Weight Inhibitors for HuR. Nat. Chem. Biol. 3, 508–515. doi:10.1038/nchembio.2007.14
Meng, Z., King, P. H., Nabors, L. B., Jackson, N. L., Chen, C. Y., Emanuel, P. D., et al. (2005). The ELAV RNA-Stability Factor HuR Binds the 5'-untranslated Region of the Human IGF-IR Transcript and Differentially Represses Cap-dependent and IRES-Mediated Translation. Nucleic Acids Res. 33, 2962–2979. doi:10.1093/nar/gki603
Mitsunari, K., Miyata, Y., Asai, A., Matsuo, T., Shida, Y., Hakariya, T., et al. (2016). Human Antigen R Is Positively Associated with Malignant Aggressiveness via Upregulation of Cell Proliferation, Migration, and Vascular Endothelial Growth Factors and Cyclooxygenase-2 in Prostate Cancer. Translational Res. 175, 116–128. doi:10.1016/j.trsl.2016.04.002
Mukherjee, J., Ohba, S., See, W. L., Phillips, J. J., Molinaro, A. M., and Pieper, R. O. (2016). PKM2 Uses Control of HuR Localization to Regulate P27 and Cell Cycle Progression in Human Glioblastoma Cells. Int. J. Cancer 139, 99–111. doi:10.1002/ijc.30041
Muralidharan, R., Mehta, M., Ahmed, R., Roy, S., Xu, L., Aubé, J., et al. (2017). HuR-targeted Small Molecule Inhibitor Exhibits Cytotoxicity towards Human Lung Cancer Cells. Sci. Rep. 7, 9694. doi:10.1038/s41598-017-07787-4
Myer, V. E., Fan, X. C., and Steitz, J. A. (1997). Identification of HuR as a Protein Implicated in AUUUA-Mediated mRNA Decay. Embo j 16, 2130–2139. doi:10.1093/emboj/16.8.2130
Nabors, L. B., Gillespie, G. Y., Harkins, L., and King, P. H. (2001). HuR, a RNA Stability Factor, Is Expressed in Malignant Brain Tumors and Binds to Adenine- and Uridine-Rich Elements within the 3' Untranslated Regions of Cytokine and Angiogenic Factor mRNAs. Cancer Res. 61, 2154–2161. http://aacrjournals.org/cancerres/article-pdf/61/5/2154/2493096/ch050102154.pdf.
Ohwada, A., Nagaoka, I., Takahashi, F., Tominaga, S., and Fukuchi, Y. (1999). DNA Vaccination against HuD Antigen Elicits Antitumor Activity in a Small-Cell Lung Cancer Murine Model. Am. J. Respir. Cell Mol Biol 21, 37–43. doi:10.1165/ajrcmb.21.1.3625
Peng, W.-X., Koirala, P., Zhang, W., Ni, C., Wang, Z., Yang, L., et al. (2020). lncRNA RMST Enhances DNMT3 Expression through Interaction with HuR. Mol. Ther. 28, 9–18. doi:10.1016/j.ymthe.2019.09.024
Quattrone, A., Pascale, A., Nogues, X., Zhao, W., Gusev, P., Pacini, A., et al. (2001). Posttranscriptional Regulation of Gene Expression in Learning by the Neuronal ELAV-like mRNA-Stabilizing Proteins. Proc. Natl. Acad. Sci. 98, 11668–11673. doi:10.1073/pnas.191388398
Raspaglio, G., De Maria, I., Filippetti, F., Martinelli, E., Zannoni, G. F., Prislei, S., et al. (2010). HuR Regulates β-Tubulin Isotype Expression in Ovarian Cancer. Cancer Res. 70, 5891–5900. doi:10.1158/0008-5472.Can-09-4656
Ripin, N., Boudet, J., Duszczyk, M. M., Hinniger, A., Faller, M., Krepl, M., et al. (2019). Molecular Basis for AU-Rich Element Recognition and Dimerization by the HuR C-Terminal RRM. Proc. Natl. Acad. Sci. USA 116, 2935–2944. doi:10.1073/pnas.1808696116
Rogulja-Ortmann, A., Picao-Osorio, J., Villava, C., Patraquim, P., Lafuente, E., Aspden, J., et al. (2014). The RNA-Binding Protein ELAV Regulates Hox RNA Processing, Expression and Function within the Drosophila Nervous System. Development 141, 2046–2056. doi:10.1242/dev.101519
Schultz, C. W., Preet, R., Dhir, T., Dixon, D. A., and Brody, J. R. (2020). Understanding and Targeting the Disease‐related RNA Binding Protein Human Antigen R (HuR). Wiley Interdiscip. Rev. RNA 11, e1581. doi:10.1002/wrna.1581
Shen, L., Han, J., Wang, H., Meng, Q., Chen, L., Liu, Y., et al. (2019). Cachexia-related Long Noncoding RNA, CAAlnc1, Suppresses Adipogenesis by Blocking the Binding of HuR to Adipogenic Transcription Factor mRNAs. Int. J. Cancer 145, 1809–1821. doi:10.1002/ijc.32236
Siang, D. T. C., Lim, Y. C., Kyaw, A. M. M., Win, K. N., Chia, S. Y., Degirmenci, U., et al. (2020). The RNA-Binding Protein HuR Is a Negative Regulator in Adipogenesis. Nat. Commun. 11, 213. doi:10.1038/s41467-019-14001-8
Simion, V., Zhou, H., Haemmig, S., Pierce, J. B., Mendes, S., Tesmenitsky, Y., et al. (2020). A Macrophage-specific lncRNA Regulates Apoptosis and Atherosclerosis by Tethering HuR in the Nucleus. Nat. Commun. 11, 6135. doi:10.1038/s41467-020-19664-2
Skliris, A., Papadaki, O., Kafasla, P., Karakasiliotis, I., Hazapis, O., Reczko, M., et al. (2015). Neuroprotection Requires the Functions of the RNA-Binding Protein HuR. Cell Death Differ 22, 703–718. doi:10.1038/cdd.2014.158
Song, J., VanBuskirk, J. A., and Merbs, S. L. (2022). Regulation of Opsin Gene Expression by DNA Methylation and Histone Acetylation. Ijms 23, 1408. doi:10.3390/ijms23031408
Sun, C., Han, B., Zhai, Y., Zhao, H., Li, X., Qian, J., et al. (2022). Dihydrotanshinone I Inhibits Ovarian Tumor Growth by Activating Oxidative Stress through Keap1-Mediated Nrf2 Ubiquitination Degradation. Free Radic. Biol. Med. 180, 220–235. doi:10.1016/j.freeradbiomed.2022.01.015
Suswam, E. A., Nabors, L. B., Huang, Y., Yang, X., and King, P. H. (2005). IL-1? Induces Stabilization of IL-8 mRNA in Malignant Breast Cancer Cellsvia the 3? Untranslated Region: Involvement of Divergent RNA-Binding Factors HuR, KSRP and TIAR. Int. J. Cancer 113, 911–919. doi:10.1002/ijc.20675
Toba, G., and White, K. (2008). The Third RNA Recognition Motif of Drosophila ELAV Protein Has a Role in Multimerization. Nucleic Acids Res. 36, 1390–1399. doi:10.1093/nar/gkm1168
Wang, F., Tidei, J. J., Polich, E. D., Gao, Y., Zhao, H., Perrone-Bizzozero, N. I., et al. (2015). Positive Feedback between RNA-Binding Protein HuD and Transcription Factor SATB1 Promotes Neurogenesis. Proc. Natl. Acad. Sci. U.S.A. 112, E4995–E5004. doi:10.1073/pnas.1513780112
Wang, J., Leavenworth, J. W., Hjelmeland, A. B., Smith, R., Patel, N., Borg, B., et al. (2019). Deletion of the RNA Regulator HuR in Tumor‐associated Microglia and Macrophages Stimulates Anti‐tumor Immunity and Attenuates Glioma Growth. Glia 67, 2424–2439. doi:10.1002/glia.23696
Wang, W., Caldwell, M. C., Lin, S., Furneaux, H., and Gorospe, M. (2000). HuR Regulates Cyclin A and Cyclin B1 mRNA Stability during Cell Proliferation. Embo j 19, 2340–2350. doi:10.1093/emboj/19.10.2340
Wang, Y., Gao, R., Li, J., Tang, S., Li, S., Tong, Q., et al. (2021). Downregulation of Hsa_circ_0074854 Suppresses the Migration and Invasion in Hepatocellular Carcinoma via Interacting with HuR and via Suppressing Exosomes-Mediated Macrophage M2 Polarization. Ijn 16, 2803–2818. doi:10.2147/ijn.S284560
Wang, Y., Guo, Y., Tang, C., Han, X., Xu, M., Sun, J., et al. (2019). Developmental Cytoplasmic-To-Nuclear Translocation of RNA-Binding Protein HuR Is Required for Adult Neurogenesis. Cell Rep. 29, 3101–3117. e3107. doi:10.1016/j.celrep.2019.10.127
Wen, H., Chen, Z., Cui, Y., and Xu, Y. (2021). LncRNA NONHSAT009968 Inhibits the Osteogenic Differentiation of hBMMSCs in SA-Induced Inflammation via Wnt3a. Biochem. Biophysical Res. Commun. 577, 24–31. doi:10.1016/j.bbrc.2021.08.086
Woo, H.-H., Zhou, Y., Yi, X., David, C. L., Zheng, W., Gilmore-Hebert, M., et al. (2009). Regulation of Non-AU-rich Element Containing C-Fms Proto-Oncogene Expression by HuR in Breast Cancer. Oncogene 28, 1176–1186. doi:10.1038/onc.2008.469
Wu, X., Gardashova, G., Lan, L., Han, S., Zhong, C., Marquez, R. T., et al. (2020). Targeting the Interaction between RNA-Binding Protein HuR and FOXQ1 Suppresses Breast Cancer Invasion and Metastasis. Commun. Biol. 3, 193. doi:10.1038/s42003-020-0933-1
Wu, X., Lan, L., Wilson, D. M., Marquez, R. T., Tsao, W.-c., Gao, P., et al. (2015). Identification and Validation of Novel Small Molecule Disruptors of HuR-mRNA Interaction. ACS Chem. Biol. 10, 1476–1484. doi:10.1021/cb500851u
Xie, M., Ma, T., Xue, J., Ma, H., Sun, M., Zhang, Z., et al. (2019). The Long Intergenic Non-protein Coding RNA 707 Promotes Proliferation and Metastasis of Gastric Cancer by Interacting with mRNA Stabilizing Protein HuR. Cancer Lett. 443, 67–79. doi:10.1016/j.canlet.2018.11.032
Yang, F., Hu, A., Li, D., Wang, J., Guo, Y., Liu, Y., et al. (2019). Circ-HuR Suppresses HuR Expression and Gastric Cancer Progression by Inhibiting CNBP Transactivation. Mol. Cancer 18, 158. doi:10.1186/s12943-019-1094-z
Yano, M., Hayakawa‐Yano, Y., and Okano, H. (2016). RNA Regulation Went Wrong in Neurodevelopmental Disorders: The Example of Msi/Elavl RNA Binding Proteins. Int. J. Dev. Neurosci. 55, 124–130. doi:10.1016/j.ijdevneu.2016.01.002
Young, L. E., Sanduja, S., Bemis–Standoli, K., Pena, E. A., Price, R. L., and Dixon, D. A. (2009). The mRNA Binding Proteins HuR and Tristetraprolin Regulate Cyclooxygenase 2 Expression during colon Carcinogenesis. Gastroenterology 136, 1669–1679. doi:10.1053/j.gastro.2009.01.010
Zang, Y., Li, J., Wan, B., and Tai, Y. (2020). circRNA circ‐CCND1 Promotes the Proliferation of Laryngeal Squamous Cell Carcinoma through Elevating CCND1 Expression via Interacting with HuR and miR‐646. J. Cell Mol Med 24, 2423–2433. doi:10.1111/jcmm.14925
Zaragozá, C., Álvarez-Mon, M. Á., Zaragozá, F., and Villaescusa, L. (2022). Flavonoids: Antiplatelet Effect as Inhibitors of COX-1. Molecules 27, 1146. doi:10.3390/molecules27031146
Zhang, C., Han, X., Yang, L., Fu, J., Sun, C., Huang, S., et al. (2020). Circular RNA circPPM1F Modulates M1 Macrophage Activation and Pancreatic Islet Inflammation in Type 1 Diabetes Mellitus. Theranostics 10, 10908–10924. doi:10.7150/thno.48264
Zhang, L. F., Lou, J. T., Lu, M. H., Gao, C., Zhao, S., Li, B., et al. (2015). Suppression of miR‐199a Maturation by HuR Is Crucial for Hypoxia‐induced Glycolytic Switch in Hepatocellular Carcinoma. Embo j 34, 2671–2685. doi:10.15252/embj.201591803
Zhang, X., Lee, M. D., Buckley, C., Wilson, C., and McCarron, J. G. (2022). Mitochondria Regulate TRPV4‐mediated Release of ATP. Br. J Pharmacol. 179, 1017–1032. doi:10.1111/bph.15687
Zhao, Q., Li, C., Yu, M., Sun, Y., Wang, J., Ma, L., et al. (2020). HuR Stabilizes HTT mRNA via Interacting with its Exon 11 in a Mutant HTT-dependent Manner. RNA Biol. 17, 500–516. doi:10.1080/15476286.2020.1712894
Zhao, W. S., Yan, W. P., Chen, D. B., Dai, L., Yang, Y. B., Kang, X. Z., et al. (2019). Genome-scale CRISPR Activation Screening Identifies a Role of ELAVL2-Cdkn1a axis in Paclitaxel Resistance in Esophageal Squamous Cell Carcinoma. Am. J. Cancer Res. 9, 1183–1200. https://www.ncbi.nlm.nih.gov/labs/pmc/articles/PMC6610048/pdf/ajcr0009-1183.pdf.
Zhao, Y.-F., He, X.-X., Song, Z.-F., Guo, Y., Zhang, Y.-N., Yu, H.-L., et al. (2020). Human Antigen R-Regulated mRNA Metabolism Promotes the Cell Motility of Migrating Neurons. Development 147. doi:10.1242/dev.183509
Zhou, H., Telonis, A. G., Jing, Y., Xia, N. L., Biederman, L., Jimbo, M., et al. (2016). GPRC5A Is a Potential Oncogene in Pancreatic Ductal Adenocarcinoma Cells that Is Upregulated by Gemcitabine with Help from HuR. Cell Death Dis 7–e2294. doi:10.1038/cddis.2016.169
Zhu, Y., Yang, L., Xu, J., Yang, X., Luan, P., Cui, Q., et al. (2020). Discovery of the Anti-angiogenesis Effect of Eltrombopag in Breast Cancer through Targeting of HuR Protein. Acta Pharmaceutica Sinica B 10, 1414–1425. doi:10.1016/j.apsb.2020.02.007
Glossary
AGAP2 ArfGAP with GTPase domain, ankyrin repeat and PH domain 2
AGAP2-AS1 AGAP2 antisense RNA 1
AGO2 Argonaute 2
APOA4-AS Rho GTPase activating protein 21
ARE AU-rich element
ASB16 ankyrin repeat and SOCS box containing 16
ASB16-AS1 ASB16 antisense RNA 1
ATG16L1 Autophagy Related 16 Like 1
B4GALT1-AS1 B4GALT1 Antisense RNA 1
BRCA breast cancer;
CAAlnc1 cachexia-related anti-adipogenesis lncRNA 1
CircAGO2 circular RNA Argonaute 2
circBACH1 circular RNA BTB Domain and CNC Homolog 1
circ-CCND1 circular RNA Cyclin D1
circDLC1 circular RNA DLC1
circ-HuR circular RNA ELAV Like RNA Binding Protein 1
circRHOBTB3 circular RNA RHOBTB3
circRNA circular RNA
COX-2 cyclooxygenase 2
CP cleavage products
CX43 Connexin 43
DNMT3B DNA methyltransferase 3B
eEF2 Eukaryotic Translation Elongation Fa
EGFR epidermal growth factor receptor
EGFR-AS1 EGFR antisense RNA 1
eIF2a Eukaryotic Translation Initiation Factor 2A
ELAVL proteins Embryonic lethal abnormal vision-like proteins
FAM83H-AS1 family with sequence similarity 83 member H antisense RNA 1
FENDRR FOXF1 adjacent non-coding developmental regulatory RNA
HCT116 human colon cancer cells 116
Hel-N1 human elav-like neuronal protein 1
HMS HOXC10 mRNA stabilizing factor
HOXB homeobox B cluster
HOXB-AS1 HOXB cluster antisense RNA 1
HTT Huntingtin
HuB human antigen B
HuC human antigen C
HuD human antigen D
HuR human antigen R
IL interleukin
IRES internal ribosome entry site
lincRNA-UFC1 long intergenic noncoding RNA UFC1
lncRNA Long non-coding RNA
MAARS Macrophage-Associated Atherosclerosis
MALAT1 metastasis associated lung adenocarcinoma transcript 1
MD1 muscle differentiation 1
MIR100HG mir-100-let-7a-2-mir-125b-1 cluster host gene
miRNA micro RNA
mRNA message RNA
ncRNA non-coding RNA
NF-κB Nuclear Factor-kappa B
OCC-1 Overexpressed in colon carcinoma-1
OIP5 Opa interacting protein 5
OIP5-AS1 OIP5 antisense RNA 1
OSER1 oxidative stress responsive serine rich 1
OSER1-AS1 OSER1 antisense RNA 1
p27 protein 27
p53 protein 53
P62 protein 62
PARP1 poly(ADP-ribose) polymerase 1
PFN1 profilin 1
PIM1 Pim-1 Proto-Oncogene, Serine/Threonine Kinase
PPM1F Protein Phosphatase, Mg2+/Mn2+ Dependent 1F
PTBP1 Polypyrimidine Tract Binding Protein 1
RBP RNA-binding protein
RMST habdomyosarcoma 2-Associated Transcript
ROS reactive oxygen species
RPSAP52 ribosomal protein SA pseudogene 52
RRM RNA recognition motif
SATB1 sequence-binding protein 1
SCLC small cell lung cancer
SNHG7 small nucleolar RNA host gene 7
SPRY4 sprouty RTK signaling antagonist 4
SPRY4-IT1 SPRY4 intronic transcript 1
tie1AS tie1 locus-tie1 antisense
TIN2 TRF1-interacting nuclear protein 2
TRN2 transportin-2
TSLNC8 Tumor suppressor long noncoding RNA on chromosome 8p12
TUBB3 Tubulin Beta 3 Class III
TUG1 aurine up-regulated 1
UTR untranslated region
UV Ultraviolet Rays
Wnt-5a Wnt Family Member 5A
XIAP X-linked inhibitor of apoptosis
ZEB1 zinc finger E-box binding homeobox 1
ZEB1-AS1 ZEB1 antisense RNA 1.
Keywords: ELAVL proteins, RNA binding proteins, non-coding RNAs, post-transcriptional regulation, cancers
Citation: Cai H, Zheng D, Yao Y, Yang L, Huang X and Wang L (2022) Roles of Embryonic Lethal Abnormal Vision-Like RNA Binding Proteins in Cancer and Beyond. Front. Cell Dev. Biol. 10:847761. doi: 10.3389/fcell.2022.847761
Received: 03 January 2022; Accepted: 04 March 2022;
Published: 06 April 2022.
Edited by:
Alexander Brodsky, Lifespan, United StatesReviewed by:
Greco Hernández, National Institute of Cancerology (INCAN), MexicoIhab Younis, Carnegie Mellon University in Qatar, Qatar
Copyright © 2022 Cai, Zheng, Yao, Yang, Huang and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lehe Yang, eWFuZ2xlaGVAd211LmVkdS5jbg==; Xiaoying Huang, emp3emh4eUAxMjYuY29t; Liangxing Wang, d2FuZ2xpYW5neGluZ0B3emhvc3BpdGFsLmNu
†These authors have contributed equally to this work
 Haijian Cai†
Haijian Cai† Lehe Yang
Lehe Yang Xiaoying Huang
Xiaoying Huang Liangxing Wang
Liangxing Wang