- 1Department of Medicine, Division of Cardiology, Baylor Scott & White Health, Round Rock, TX, United States
- 2Department of Medicine, Division of Cardiology, Baylor Scott & White Health, Temple, TX, United States
Atrial fibrillation (AF) represents the most common cardiac arrhythmia with significant morbidity and mortality implications. It is a common cause of hospital admissions, significantly impacts quality of life, increases morbidity and decreases life expectancy. Despite advancements in treatment options, prevalence of AF remains exceptionally high. AF is a challenging disease to manage, not just clinically but also financially. Evidence suggests lifestyle modification, including dietary changes, plays a significant role in the treatment of AF. This review aims to analyze the existing literature on the effects of dietary modifications on the incidence, progression, and outcomes of atrial fibrillation. It examines various dietary components, including alcohol, caffeine, omega-3 polyunsaturated fatty acids and minerals, and their impact on AF incidence, progression, and outcomes. The evidence surrounding the effects of dietary patterns, such as the Mediterranean and low carbohydrate diets, on AF is also evaluated. Overall, this review underscores the importance of dietary interventions as part of a comprehensive approach to AF management and highlights the need for further research in this emerging field.
Introduction
Atrial fibrillation is the most common cardiac arrhythmia characterized by irregular and often rapid heartbeat (1). It is a common cause of hospital admissions with high readmission rates and significantly impacts quality of life (2). Patients with atrial fibrillation have an average reduction in their life expectancy and significant morbidity (3). Despite advancements in pharmacological therapy and catheter-based procedures, the prevalence of AF remains exceptionally high. There are many factors associated with AF, and the chance of spontaneous conversion back to sinus rhythm is lower with the existence of one or more of these risk factors, which include, but are not limited to, heart failure, left atrial size, and duration of the patient in AF (4, 5) As these risk factors are commonly present in the patients of modern times, the prevalence of AF remains high. It is a challenging disease to manage, not just clinically but also financially, as it places a tremendous burden on healthcare globally (2). Previous studies and emerging evidence suggest lifestyle changes, including dietary changes, play a significant role in preventing and managing AF (6) Its recurrent nature, associated symptoms, increased risk of stroke, heart failure, and overall cardiovascular morbidity and mortality demands significant attention and development of more effective management strategies. The current therapeutic approach primarily focuses on symptom and heart rate management via rate and/or rhythm control, in addition to stroke prevention with the implementation of anticoagulation or mechanical exclusion of the left atrial appendage. A growing body of evidence supports restoration and maintenance of sinus rhythm as the preferred management strategy (7). Limited success maintaining sinus rhythm long term, toxicity of the antiarrhythmic drugs, progressive nature of associated co-morbidities and limited understanding of the atrial pathophysiology involved in the natural history of AF, has made it difficult to halt or slow its progression. However, the association of alcohol intake, obesity, diabetes, and autonomic imbalance with the incidence, recurrence and progression of AF has been well-recognized for quite some time (6). More recently, there has been a growing body of research supporting the adoption of risk factor modification as the fourth pillar in the management of AF, as proposed in the latest revision of the clinical practice guidelines published by the American College of Cardiology and American Heart Association Joint Committee (8). Lifestyle modifications have demonstrated a significant impact on the prevention and recurrence of AF. Dietary changes are a significant part of these lifestyle modifications. This review aims to provide a thorough overview of the evidence supporting specific dietary modifications in managing AF.
Methods
Databases and search strategy
A comprehensive and systematic literature search was conducted in two major electronic databases: PubMed and the Cochrane Library. These databases were selected due to their extensive collections of medical and clinical research articles. The aim was to identify studies published up to 2024 that examined the relationship between dietary factors and atrial fibrillation.
Keywords and search terms
The search strategy utilized a combination of keywords to ensure a broad and thorough search. Key terms included “Atrial fibrillation”, “Diet”, “Nutrition”, “Dietary interventions”. These terms were chosen to capture a wide range of studies that could provide insights into how diet and nutrition impact AF.
Inclusion criteria
To ensure relevance and quality, studies had to meet the following inclusion criteria:
Population: Studies involved human participants diagnosed with AF or those at risk of developing the condition.
Publication: Only studies published in peer-reviewed journals were considered.
Screening and selection process
The selection process involved several steps to ensure rigorous filtering and inclusion of relevant studies:
Initial Screening: Three independent reviewers screened the titles and abstracts of all identified articles. This initial step was crucial for excluding studies that were clearly irrelevant based on the title and abstract alone.
Full-Text Assessment: Articles that passed the initial screening were then subjected to a full-text assessment and the content of each of these studies was further investigated.
Discrepancy Resolution: Any discrepancies or disagreements among the reviewers were resolved through discussion and consensus, ensuring that the selection process was both thorough and unbiased.
Data extraction and synthesis
Given the anticipated diversity in study designs, populations, dietary interventions, and outcomes, a narrative synthesis approach was adopted. This method involves summarizing and interpreting the findings of the included studies in a descriptive manner rather than relying solely on statistical analysis. The narrative synthesis allowed for identification of common themes and highlighting consistent findings across different studies, contextual analysis which helped in understanding how different dietary factors may influence AF in various contexts and integration of diverse evidence by combining results from studies with varying methodologies to provide a comprehensive overview.
Summary of findings
The narrative synthesis aimed to collate and summarize the evidence regarding the impact of dietary interventions on atrial fibrillation. Key aspects included:
Types of dietary interventions: Examination of specific diets (e.g., Mediterranean diet), individual nutrients (e.g., omega-3 fatty acids), and other dietary components.
Outcomes: Analysis of outcomes such as AF incidence, symptom severity, and recurrence rates.
Population characteristics: Consideration of how different populations (e.g., age groups, comorbid conditions) respond to dietary changes.
By employing this systematic and structured approach, the review aimed to provide a detailed and comprehensive understanding of the current evidence on dietary interventions and their effects on atrial fibrillation.
Diets, nutrients and atrial fibrillation
Omega-3 fatty acids
The potential role of omega-3 fatty acids, primarily derived from fish consumption or supplementation, in influencing AF incidence has been extensively studied. Several lines of evidence from observational studies and randomized trials shed light on the complex relationship between omega-3 fatty acids and AF risk. Observational studies initially suggested a preventive effect of fish consumption against new-onset AF, particularly among elderly adults (9). Specifically, greater intake of broiled/baked fish, notably tuna, has been associated with a lower risk of AF. The protective effect is attributed to the beneficial impact of long-chain omega-3 fatty acids in fatty fish. However, fried fish or fish sandwiches did not confer the same protective effect, underscoring the importance of preparation methods (9). In contrast, randomized trials exploring marine omega-3 fatty acid supplementation for primary AF prevention have yielded discouraging results (10). A trial over 5.3 years found no significant effect in AF incidence compared to placebo, challenging the efficacy of omega-3 fatty acid supplements (10). In the other hand, a large cohort study involving over 54,000 participants over 13 years found that in vivo levels of omega-3 fatty acids had no association to the risk of incident AF, supporting their safety to regards to AF risk (11). A recent meta-analysis of prospective studies involving more than 200,000 participants and up to 12,000 cases of AF found no significant association between higher fish consumption or intake of omega-3 polyunsaturated fatty acids (PUFAs) and the development of AF (12). While fish and omega-3 PUFAs have been inversely associated with various cardiovascular diseases, including stroke and coronary heart disease, no significant inverse association was found with AF in this meta-analysis. The conflicting outcomes of clinical trials and observational studies may be attributed to various factors, including the heterogeneity of patient populations, differences in fish preparation methods, and the complex interplay between dietary components and cardiovascular health. Furthermore, the electrophysiological effects of omega-3 PUFAs, particularly EPA and DHA found in fish oil, on cardiac ion channels and membrane properties add another layer of complexity to the understanding of their potential role in arrhythmia prevention (13, 14). It is hypothesized that fatty acids may influence parameters crucial for generating and maintaining arrhythmias, but their impact on AF recurrence or postoperative AF, remains unclear. While omega-3 fatty acids derived from fish consumption or supplementation have been associated with various cardiovascular benefits, including potential anti-arrhythmic effects, currently they have no role in AF prevention or management. Further research and a higher level of evidence is necessary to better understand the association between omega-3 fatty acids, their modality of consumption, and cardiovascular health before their inclusion in strategies to treat or prevent AF.
Mediterranean diet
The Mediterranean Diet (Med-Diet) characterized by a high consumption of fruits, vegetables, whole grains, legumes, nuts, seeds, olive oil, a modest intake of fish and poultry, and a low consumption of red meat and sweets (15) has emerged as a robust preventative strategy against the development of AF. Numerous studies have demonstrated that adherence to the Mediterranean diet reduces the risk of AF and other manifestations of cardiovascular disease (16). Paradoxically and despite the lack of definitive evidence in support of omega 3 fatty acids supplements, it has been postulated that the Mediterranean diet's emphasis on foods rich in omega-3 fatty acids may contribute to decrease inflammation and improve cardiac function, lowering the risk of AF. Another proposed mechanism is the Mediterranean diet's high intake of fruits, vegetables, and olive oil which provide ample antioxidants and polyphenols, which have anti-inflammatory and cardioprotective effects (16). Hence the benefit could be attributable to the latter or their combination. The PREDIMAR study investigated the efficacy of a remotely delivered Med-Diet-based nutritional intervention in preventing atrial tachyarrhythmia recurrence post-catheter ablation in AF patients (17). The intervention, utilizing phone contacts, web-based tools, and resource access, enhanced adherence to the Med-Diet, notably resulting in positive dietary habit changes but failed to demonstrate an effect on AF recurrence post ablation. By recognizing diet as a crucial element of lifestyle modification, tools to optimize adherence could play a pivotal role in reducing AF risk. The study underlines the benefits of the predominantly plant-based traditional Mediterranean diet, focusing on fish, olive oil, nuts, fruits, and vegetables. Intriguingly in this study, the fish component of the Med-diet, rich in n-3 fatty acids, exhibits a “U” shaped curve, highlighting the delicate balance for optimal AF protection. A secondary analysis of the PREDIMED trial reveals significant AF protection with a Med-Diet supplemented with extra virgin olive oil (EVOO), emphasizing its anti-inflammatory and antioxidant properties (18). Other studies have evaluated the Med-Diet's intersection with metabolites in the tryptophan-kynurenine pathway, which are associated with heart failure and AF risk (19) The Med-Diet, especially when supplemented with EVOO, demonstrates potential counteraction against these metabolites, emphasizing its role in regulating inflammation (18). The comprehensive PREDIMED study showed a 30% reduction in cardiovascular events, improved blood pressure, insulin resistance, and lipid profiles, emphasizing its multifaceted impact (20). Additionally, Med-Diet has a positive impact in weight loss, triglycerides, blood pressure, and diabetes mellitus, presenting a holistic approach to cardiovascular health and hence offering a dietary formula to decreasing the various risk factors involved in the development of AF. A complementary study from 2014 further supports the cardiovascular benefits of the Med-Diet, highlighting its association with reduced platelet activation and thromboxane A2 production in AF patients (21). This study, focusing on elderly AF patients at high risk of atherosclerosis and thromboembolism, establishes a link between higher Med-Diet adherence and diminished platelet activation, supported by a reduction on thromboxane B2 biosynthesis (22). Notably, wine and olive oil consumption, integral to the Med-Diet by some definitions, were independently associated with lower platelet activation levels, suggesting a beneficial effect. While the study's observational nature and limited sample size pose constraints, it introduces a novel biological explanation for the cardiovascular advantages of Med-Diet, particularly when enriched with extra virgin olive oil. Alcohol consumption, nevertheless, has been associated with higher incidence of AF as discussed later in this review and should probably be excluded for now from the Med-Diet recommendations for patients with AF. In summary, the Mediterranean Diet has been described as a nutritional powerhouse, rich in antioxidants and displaying favorable metabolic effects. Its protective role in preventing AF still needs to be further evaluated but evidence supports that the diet addresses risk factors associated with metabolic syndrome which underscores the overall value of dietary interventions in promoting cardiovascular health. The nuanced relationships between specific dietary components, remote nutritional interventions, and metabolites further emphasize the need for additional research, including larger and better controlled interventional studies, to explore the clinical impact of the Med-Diet in AF patients and its potential role in reducing cardiovascular events and disease progression. Funding and scale for such studies remains a challenge. Currently, its potential benefits and lack of evidence for harm supports a cautious recommendation in favor of the Mediterranean Diet, excluding regular alcohol intake, as a healthy alternative for patients at risk or with AF. Future research may help optimize the elements of the diet, confirm, reject or identify specific populations more likely to benefit from this recommendation.
DASH diet
Another dietary intervention that has shown positive effect on the management of hypertension and statistical association with improved survival is the DASH diet but an effect on incidence, recurrence or progression of AF has not been demonstrated. In the Dietary Approaches to Stop Hypertension (DASH) study (23), a diet rich in fruits and vegetables combined with low-fat dairy foods in addition to reduced total and saturated fat resulted in significant blood pressure lowering effect and good adherence. Subsequent large population studies have shown an association between DASH and similar diets with improved cardiovascular outcomes and decreased mortality (24) but once corrected for other lifestyle factors have not shown a definitive effect on AF (25). Considering the effects on blood pressure, cardiovascular health, mortality and weight, it is reasonable to consider DASH as a healthy dietary alternative to recommend for patients with AF particularly those with associated hypertension and diabetes.
Low-carbohydrate diet
Diabetes mellitus, a complex metabolic disorder, constitutes a significant contributor to the heightened risk of AF, with studies indicating a 40% increase in AF risk associated with diabetes (26). The risk escalates with higher hemoglobin A1c levels and prolonged diabetes duration, establishing a direct correlation between diabetes severity and AF susceptibility. The intricate pathophysiologic mechanisms associated to diabetes exert their effect not only directly but also indirectly, intertwining with other AF comorbidities like obesity and dietary habits. Aggressive diabetes control is recommended as part of the comprehensive lifestyle modification intervention that proved successful in the management of AF in the ARREST AF trial (27). Surprisingly and complicating our understanding, a large prospective cohort study (ARIC study) spanning over two decades unravels a novel association between dietary choices and AF incidence (28). The research identifies a higher risk of incident AF linked to a low-carbohydrate intake as a percentage of energy. This marks a departure from traditional dietary assessments primarily focused on factors like omega-3 fatty acids. The study's robust design, involving a large community-based cohort with extensive follow-up and thorough statistical adjustments, underscores the reliability of its findings. The inverse relationship between carbohydrate intake and incident AF prompts considerations about the potential mechanisms at play. The study posits that low-carbohydrate diets lead to reduced intake of anti-inflammatory foods, trigger oxidative stress, and potentially elevate the risk of other cardiovascular diseases, all of which are established risk factors for AF. However, the study also acknowledges limitations inherent in its observational nature, including potential measurement errors in dietary assessments and challenges in accurately classifying AF types. Despite these caveats, the findings highlight the cautious evaluation of low-carbohydrate diets and their implications on arrhythmia. The call for additional research, including randomized controlled trials, echoes the need to delve deeper into the intricate relationship between dietary choices, metabolic conditions like diabetes, and the multifaceted landscape of AF risk factors. Further exploration is essential to guide recommendations and interventions for primary AF prevention in the complex interplay between metabolic health and dietary patterns.
Alcohol
Despite moderate alcohol intake being considered in the past an element of the Mediterranean Diet and a potential protective intervention against coronary artery disease, a meta-analysis conducted in 2010 consistently showed a clear association between alcohol consumption and the risk of AF onset across different settings, with varying strengths of association (29). A dose-response relationship between daily alcohol intake and AF risk was observed, suggesting a potential threshold under which the increased risk of AF may not be significant. Temporal analyses and interventions indicate the reversibility of AF following changes in alcohol consumption. The proposed mechanism is the effect of alcohol on the atrial tissue and its electrical properties, leading to abnormalities such as decreased conduction velocity and shortened refractory periods, promoting the development of atrial re-entry and the various pathophysiologic mechanism underlying AF (30). Another proposed pathophysiology mechanism involves affecting histamine levels and cytosolic sulfotransferases. Additionally, alcohol-related hypertension may contribute to atrial remodeling, further increasing the risk of AF onset (31). Both clinical and pathophysiological evidence strongly suggest that regular alcohol consumption may cause AF. A multicenter randomized controlled trial aimed to investigate the impact of reducing alcohol consumption on AF recurrence among regular drinkers with symptomatic AF found that substantial reduction in alcohol intake was associated with a decrease in AF recurrence and a reduced proportion of time spent in AF (32). Previous studies have shown a dose-related increased risk of incident AF with alcohol consumption, even with low levels of intake. However, limitations include relying on patient-reported alcohol quantities subject to recall bias, potential confounding factors, and challenges assessing secondary outcomes such as cardiovascular events. Gender variations are evident regarding the link between moderate alcohol intake and AF, with males showing a more pronounced increase in risk (33). A Danish study found that increasing alcohol intake over five years correlated with higher AF risk, but reducing intake did not significantly reduce risk (29). Overall, it has been recognized that regular alcohol consumption is a modifiable risk factor for AF and reducing alcohol intake might lead to a reduction in AF burden and recurrence. Prospective data on alcohol changes and AF risk is sparse. Limitations include self-reported alcohol intake, possible selection bias, and lack of consideration for binge drinking and sleep apnea. Observational data cannot establish causality, and AF assessment has frequently relied on diagnosis codes, possibly underestimating incidence. Based on current evidence alcohol abstinence is recommended to prevent AF and AF recurrences.
Gluten
Celiac disease (CD) is a chronic gastrointestinal inflammatory disorder characterized by malabsorption in individuals sensitive to gluten-containing grains. While its global prevalence in the general population is around 1%, it is notably higher in patients with autoimmune disorders, reaching 8%–20% (34). CD has been linked to a significantly higher risk of major adverse cardiovascular events, including cardiac arrhythmias (35). Cardiovascular disease is the most common cause of death among these patients (36). Theoretically, inflammation and fibrosis play a significant role in developing AF (37). Studies have shown a slightly elevated risk of AF in patients with CD, both before and after the diagnosis; however, the risk was higher around the time of the diagnosis, suggesting a role of increased amount of inflammation aggravating both CD as well as AF (38). Studies have found a link to various inflammatory markers like high-sensitivity C-reactive protein, sICAM-1, and fibrinogen (38, 39). Furthermore, cases of ventricular arrhythmia in CD patients with autoimmune myocarditis improved with a gluten-free diet, indicating a potential link between CD and arrhythmias in general (40). Our group has reported a small case series of patients with gluten sensitive arrhythmias including AF and idiopathic premature ventricular contractions with near resolution of symptoms after adopting a gluten free diet (41). In summary, there is a link between autoimmune diseases and the risk of development of AF, particularly in patients with CD; however, once the confounding factors are adjusted, the risk remains only small at around 30% (38). An evaluation to rule out sub-clinical or undiagnosed CD as well as an empiric trial of gluten avoidance in patients with idiopathic or “lone” AF may be reasonable based on its simplicity and lack of harm.
Caffeine
Contrary to common believe and the sporadic anecdotal association of caffeine intake to cardiac arrhythmias, a 2016 cohort study found that coffee ingestion and total caffeine intake were associated with a reduced risk of developing AF across various risk groups (42). While caffeine has been extensively studied, coffee contains numerous compounds besides caffeine that may contribute to health effects (43). Some studies suggest that compounds in coffee may counterbalance adverse effects of caffeine, with potential benefits for cardiovascular health. However, the specific compounds responsible for these benefits remain unidentified (43, 44). In a meta-analysis, participants consuming higher levels of coffee showed a lower risk of AF, consistent with prior findings (44). However, the study had limitations, such as potential residual confounding and the inability to distinguish between caffeinated and decaffeinated coffee intake. Despite these limitations, the study's strengths included a large sample size, detailed data collection, and almost complete adherence to follow-up. A 2018 meta-analysis concluded again that caffeine does not increase the risk of AF (45). It demonstrated an association between higher caffeine intake and a lower incidence of AF. A Potential mechanism for caffeine's protective effect against AF includes its lack of acute arrhythmogenicity in healthy individuals (44). Other studies have also found favorable outcomes associated with caffeine intake or coffee consumption, including reduced risk of death from various causes and no relationship between chronic caffeine consumption and ventricular ectopy (46). In another prospective study involving healthy middle-aged women, caffeine use was not associated with an increased risk of AF (47). Women in the highest quintile of caffeine intake had a similar AF risk to those in the lowest quintile, with minimal changes after multivariable adjustment. Interestingly, women in the third quintile of caffeine consumption were found to have lower risk of AF, suggesting the potential benefits of moderate caffeine intake. These findings, as mentioned earlier, suggest that increased caffeine consumption does not contribute to the increasing burden of AF in the general population, and moderate caffeine intake may even have a protective effect. Further studies are needed to elucidate coffee's cardioprotective effects beyond caffeine and determine potential differences in sensitivity between coffee and pure caffeine intake. The current body of evidence supports the conclusion that caffeine consumption does not increase AF incidence and could even reduce it, particularly with moderate coffee consumption. Nevertheless, most of this favorable data derive from large population studies. It remains unclear if a sub-group of individuals could be particularly sensitive to caffeine, subjects among whom caffeine intake could lead to AF or AF recurrences, acting perhaps as a trigger. In clinical practice, few patients report this clinical association and among them it remains reasonable to recommend avoidance of caffeine once identified as their reproducible trigger, regardless of the mechanism involved.
Chocolate
Chocolate consumption in its association with AF has yielded inconsistent findings across various studies (48). A Danish population-based cohort study involving 55,502 participants over an average of 13.5 years revealed a significant association between chocolate intake and reduced AF risk (49). However, two prior studies, the Women's Health Study and a cohort study of US male physicians, found no statistically significant associations between chocolate consumption and AF risk (50, 51). The Women's Health Study, with more than 33,000 female participants followed for over 14 years, showed non-significant hazard ratios across quintiles of chocolate consumption, except for the third quintile (50). Similarly, the Physicians' Health Study, following 18,819 US male physicians for approximately nine years, revealed non-significant hazard ratios for various levels of chocolate consumption (51). Data from two cohort studies and a meta-analysis, including 180,454 participants, also found no evidence of an association between chocolate consumption and AF risk (50). In an analysis of two prospective multicenter Swiss AF cohort studies (Swiss-AF) and (BEAT-AF), chocolate consumption was found to have no association with major adverse cardiac events such as ischemic stroke, myocardial infarction or cardiovascular death in a patient population with AF (52). While some studies have shown a beneficial link between moderate chocolate consumption and other cardiovascular diseases like ischemic heart disease, heart failure, and stroke, the impact on AF appears to be neutral (53). This discrepancy could be due to the cardiovascular effects of cocoa products, such as improved endothelial function and modest reductions in blood pressure and insulin resistance, which may have less influence on AF than atherosclerosis-related cardiovascular diseases (54). Strengths of the Swedish cohort studies included their large sample sizes, adjustment for major potential confounders, and reliance on objective data from the Swedish Patient Register. However, limitations such as inevitable misclassification of chocolate consumption, lack of information on milk chocolate vs. dark chocolate consumption, and the observational design should be noted. Despite the large sample size in the meta-analysis, no association between chocolate consumption and AF risk was observed. No definitive recommendation can be made regarding chocolate consumption in relationship to AF.
Salt
In a large-scale prospective observational study involving 473,080 adults, the relationship between estimated daily salt intake and the risk of new-onset AF was investigated (55). The study utilized urinary sodium excretion as a proxy for dietary salt intake, revealing a U-shaped association between sodium intake and AF risk among men; very low and high estimated daily sodium intakes were associated with elevated AF risk. Among women, while there was initially a tendency for a J-shaped association between sodium excretion and AF risk, this trend vanished after adjusting for established cardiovascular risk factors. A Finnish study, albeit smaller in scale, supported the association between high salt intake and increased AF risk but did not report the association between low sodium intake and AF risk among men (56). Other investigations have provided additional insights into the complex interplay between sodium intake and cardiovascular health, including its potential role on the pathophysiology of atrial tachycardia and atrial fibrillation, not solely from hypertension but also by elevating intracellular calcium levels within cardiac tissue via the sodium/calcium exchange mechanism. This elevation subsequently influences intracellular calcium release from the sarcoplasmic reticulum, consequently contributing to arrhythmias. Moreover, sodium can alter the mechano-electrical dynamics of the myocardium, potentially precipitating arrhythmias due to modifications in cell length or tension (57, 58). On the contrary, a meta-analysis of more than 1.4 million participants in 2021 indicated that salt intake does not correlate with a heightened risk of developing new-onset AF and that factors beyond salt intake itself may exert a more significant influence on the occurrence of new-onset AF (59). However, this study had some significant limitations. It combined studies with different designs, such as Mendelian randomization and cohort studies, which could lead to methodological heterogeneity. Additionally, observational studies, including Mendelian randomization studies, may suffer from selection bias as they typically recruit survivors, potentially missing individuals who have died due to cardiovascular disease related to salt intake. This could lead to a false null association between salt intake and AF risk due to competing risks rather than indicating no relationship between salt intake and AF risk. To summarize, most studies have found a strong link between salt intake and AF risk; low sodium intake is also associated with better blood pressure control and better cardiovascular outcomes which in turn may result in lower risk of AF. However, further research is warranted to elucidate causality and better understand sodium intake's role in AF development, prevention and management.
Antioxidants and micronutrients
Carotenoids
Low plasma concentrations of lutein and zeaxanthin are associated with a nearly twofold increased risk of AF (60). However, carotenoids such as b-cryptoxanthin, lycopene, a-carotene, b-carotene, and total carotenoid deficiency did not significantly correlate with AF risk. Recurrent AF was used in the risk analysis, with a high incidence observed despite antiarrhythmic therapy. It emphasizes the role of inflammation and oxidative stress in AF development and suggests that carotenoids, known for their antioxidant properties, may mitigate these factors. The anti-inflammatory and antioxidant effects of carotenoids may contribute to the remodeling of atrial myocytes, reducing the risk of AF. Carotenoids, in combination with different antioxidants found in fruits and vegetables, may have synergistic effects, providing more significant health benefits than individual antioxidants. With their reported interactions in the human body by scavenging free radicals, carotenoids may independently contribute to a decreased risk of AF. In conclusion, the low plasma levels of lutein and zeaxanthin are associated with an elevated risk of AF in the elderly. It supports the idea that consuming foods rich in carotenoids like fruits and vegetables as emphasized in the Mediterranean and DASH diets may be protective against AF.
Flavonoids
The postulated potential benefits of flavonoids in reducing AF risk remain inconsistent. In a large Danish cohort study spanning 23 years, higher habitual intake of total flavonoids was not significantly associated with lower incident AF risk overall. However, intriguingly, flavonoid intake showed a protective effect in smokers and heavy alcohol consumers (61). Flavonoids have been implicated in mitigating cardiovascular disease markers through anti-inflammatory and anti-thrombogenic pathways, but their role in AF prevention remains uncertain (61). Notably, the observed association between flavonoid intake and reduced AF risk in specific subgroups suggests potential protective effects mediated through inflammatory and oxidative stress pathways rather than direct antiarrhythmic properties (62). While studies have provided valuable insights, limitations inherent to observational research, such as unmeasured confounders and potential changes in dietary habits over time, warrant cautious interpretation of the results. Further research is needed to authenticate these findings and explore the potential benefits of flavonoid-rich foods, particularly in high-risk populations such as smokers and heavy alcohol consumers, for AF prevention.
Magnesium
A study involving 3,530 participants from the Framingham Offspring Study investigated the association between serum magnesium levels at baseline and the risk of atrial fibrillation in individuals free of cardiovascular disease (63). Using Cox proportional hazard regression analysis and adjusting for various factors, including conventional AF risk factors, the researchers found that lower serum magnesium levels were moderately associated with a higher risk of developing AF over a follow-up of up to 20 years. Individuals in the lower quartile of serum magnesium were approximately 50% more likely to develop AF than those in the upper quartiles. The results remained consistent even after excluding individuals on diuretics from the analysis. Given the common occurrence of hypomagnesemia in the general population, this research has potential clinical implications. Additionally, in critically ill patients, where post-operative AF is common, magnesium supplementation has shown promise in preventing AF development, likely through its anti-inflammatory and anti-arrhythmic effects (64). However, limitations such as small sample sizes, high risk of bias in some studies, and heterogeneity in outcomes should be considered when interpreting the findings. Further research is needed to validate these results, especially in critically ill populations, to improve patient outcomes and reduce healthcare utilization costs associated with AF.
Potassium
Hypokalemia has been associated with higher risk of AF (65, 66), cardiovascular events, stroke and mortality in the general population (67, 68) and among patients with hypertension (69). It is a recognized risk factor for AF after cardiac surgery (70). Low potassium has been shown to reduce sinoatrial node activity and increase pulmonary vein firing in animal models (71). Potassium-rich foods seem to counteract the negative effect of sodium by promoting diuresis and reducing aldosterone secretion (72). Surprisingly, potassium supplementation has failed to demonstrate a reduction in the incidence of AF after cardiac surgery (73). On the other hand, the use of potassium chloride supplementation and combination of sodium and potassium chloride as dietary substitutes for sodium has been shown to have a positive effect on hypertension, cardiovascular events and death (74–77). Direct evidence on the effects of potassium supplementation in AF is still lacking. Extrapolating from its beneficial effects on hypertension and cardiovascular outcomes and evidence in favor of the Mediterranean and DASH diet, it is reasonable to consider the recommendation of potassium-based salt substitutes for patients with or at risk of AF. Caution and close monitoring must be considered amongst patients with renal function impairment and at risk for hyperkalemia.
Miscellaneous micronutrients
Deficiencies and levels of certain other micronutrients, such as selenium (Se) and iron (Fe), have been linked to new-onset atrial fibrillation in a large community-based cohort. After adjusting for potential confounders, Se deficiency, similar to Mg deficiency, was associated with an increased risk of new-onset AF, particularly in non-smoking participants (78). Se deficiency also showed a significant association with non-smoking participants experiencing over a 65% increased risk. Mechanistically, it may contribute to mitochondrial dysfunction and oxidative stress, two factors involved in the pathogenesis of AF and heart failure. However, Fe deficiency did not show a significant association with new-onset AF, although it was observed that older men had a slightly higher risk (78). Histamine, derived from histidine, plays a role in immune responses and cardiovascular regulation. Elevated levels are associated with arrhythmogenic potential, stimulating H2 receptors to accelerate heart rate and trigger diastolic depolarization, potentially leading to atrial tachycardia (79). Histamine-induced depolarization of Purkinje fibers may promote ventricular tachycardia (79). Rare cases link hyper-histaminemia to cardiac arrests and AF (80). Due to its wide range of pro-arrhythmic properties, a pilot study suggested elevated levels in 21.2% of AF patients, possibly due to histamine-containing foods, allergies, infections, or immune disorders (81). However, prospective data on histamine's role in AF are limited. These findings suggest that nutritional imbalances may represent modifiable risk factors for AF, independent of heart failure development, highlighting potential avenues and research for AF prevention.
Obesity and atrial fibrillation
Obesity is prevalent in the US, where both obesity and AF burdens are high. The Long-Term Effect of Goal-Directed Weight Management in an Atrial Fibrillation Cohort: A Long-Term Follow-Up Study (LEGACY) study, focusing on 355 overweight AF patients, demonstrated the remarkable impact of modest weight loss (82). During the waiting period for their AF ablation procedure, 38% successfully lost around 36 pounds, resulting in significant AF reduction. Nearly half of the patients achieved complete remission solely through weight loss, eliminating the need for antiarrhythmics or ablation. Moreover, improvements were observed in systolic blood pressure, C-reactive protein, diabetes remission, LDL levels, triglycerides, echocardiographic abnormalities, and overall sense of well-being. Obesity's intricate relationship with AF involves complex mechanisms, including inflammation and oxidative stress, with weight loss showing promise in reducing AF development and recurrence (83). Similar reductions in AF have been reported among patients undergoing bariatric surgery (84). Challenges remain in understanding variations in AF risk associated with different fat types and the impact of obesity on permanent AF. Despite the mainstream belief in weight loss benefits, a post hoc analysis of the Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) study demonstrated an “obesity paradox” with better clinical outcomes amongst obese patients with AF (85). On the other hand, early-life obesity has been recognized as a predictor of AF risk. Epicardial fat, a key player in left atrial remodeling, poses imaging challenges when quantifying it to establish its relationship with AF severity. Based on the overall benefits of weight reduction in cardiovascular health, blood pressure and the results of the LEGACY (82) and similar clinical studies, weight reduction is recommended for reduction in the burden of AF and as co-adjuvant to antiarrhythmic drug therapy and catheter-based ablation.
Bacteriome, diet and atrial fibrillation
Multiple studies have demonstrated a strong association between alterations in the gut microbiota and the risk of cardiovascular disease and AF. It is described that patients with AF develop dysbiotic gut microbiota with higher microbial diversity and specific composition patterns. It is recognized that diet and drugs could be important determinants in the composition of the gut bacteriome (22). Bacteria metabolites like gut-derived lipopolysaccharide, Trimethylamine N-Oxide, secondary bile acids (86–89) are found elevated in patients with AF and linked to atrial inflammation and adverse electrical remodeling. Meanwhile the gut microbiota changes accompanying AF are also associated with disrupted production of the rather protective short chain fatty acids (90). Fecal transplantion studies in animals has demonstrated that transplanting dysbiotic microbiota or microbiota from aged animals to healthy subjects results in higher susceptibility to AF, increased levels of circulating lipopolysaccharide and evidence of inflammation and fibrosis in the atrium (91, 92). Despite the current body of evidence supporting an association between AF, the gut microbiota and its derived metabolites, it is not clear if they have causal or modulation effect. Even less understood is the potential opportunity to intervene via diet, pharmacotherapy or fecal transplantation in the prevention or treatment of AF (93).
Proposed mechanisms
AF is a complex arrhythmia with multiple underlying mechanisms. Several dietary factors and micronutrients studied for their potential role in the pathophysiology of AF affect various of these mechanisms, as described in this article. There is an overlap between the different mechanisms outlined here, but it is important to recognize the contrast between these separate processes and their respective significance in the complex pathophysiology of this arrythmia. Following is a brief overview of the various proposed mechanisms of action mediating the interactions between diet and the pathophysiology, clinical manifestation and progression of AF.
Inflammation and oxidative stress
Oxidative stress, caused by an imbalance between reactive oxygen species (ROS) production and the body's antioxidant defenses, is significantly implicated in AF development and remodeling (94). Studies have demonstrated that increased oxidative stress in atrial tissue leads to protein modifications and calcium accumulation, contributing to AF by reducing antioxidant levels like vitamin C and glutathione (95). Key sources of ROS in AF include NADPH oxidase, xanthine oxidase, nitric oxide synthase uncoupling, myeloperoxidase, and monoamine oxidase. These enzymes, particularly NADPH oxidase, are activated by conditions such as hypertension and hyperglycemia, leading to fibrosis and atrial remodeling. Experimental evidence also points to mitochondrial dysfunction and ROS production causing calcium leaks that trigger AF. Myeloperoxidase and monoamine oxidase further contribute to atrial fibrosis and oxidative damage, exacerbating AF risk. Ultimately, excessive ROS alter ionic currents and cellular signaling, prolonging action potentials, reducing cardiac conduction, and promoting re-entry and focal activity, which are central to AF pathogenesis. Diets rich in antioxidants, like fruits, vegetables, and whole grains, may help mitigate inflammation and oxidative stress. Micronutrients like vitamins C and E and polyphenols found in foods like berries and green tea have antioxidant properties and may reduce AF risk by combating oxidative damage (62). As previously mentioned in this review, the Mediterranean diet and a diet rich in carotenoids may mitigate the development and progression of AF through their antioxidant properties. Coffee has also been linked to decrease inflammation as it contains high levels of antioxidants such as cafestol, polyphenol, trigonelline, chlorogenic acid, and quinine. Moderate coffee consumption, through the aforementioned mechanism, has been linked to decrease incidence and risk of developing atrial fibrillation (44).
Electrical and structural remodeling
AF is associated with electrical as well as structural remodeling of the atria, characterized by alteration in ion channel function and atrial conduction. Omega-3 fatty acids found in fatty fish, like salmon and mackerel, have been shown to modulate ion channel function and stabilize atrial electrical activity, potentially reducing susceptibility to AF (96). This mechanism may be shared with other nutrients that prevent atrial inflammation, mechanical stress and cellular uncoupling. Structural changes in the atria, such as fibrosis and hypertrophy, contribute to AF substrate formation. Diets low in sodium help mitigate atrial fibrosis by reducing inflammation, hypertension and oxidative stress (97). Hyperglycemia and foods rich in sodium have been linked to atrial fibrosis, left atrial enlargement, and cause electrical and autonomic remodeling which can lead to inter and intra-atrial conduction delays. Potassium-rich foods like bananas, potatoes, and avocados may counteract the pro-fibrotic effects of sodium by promoting diuresis and reducing aldosterone secretion (72).
Autonomic nervous system dysfunction
Imbalances in the autonomic nervous system, both sympathetic and parasympathetic overactivity, can promote AF initiation and maintenance (98). In patients with established AF or structural heart abnormalities, sympathetic stimulation may be the driving factor in AF initiation (99). Vagal stimuli such as eating, sleeping, relaxation, and alcohol consumption have been identified as triggers for AF, particularly in younger individuals or those with a family history of AF (100, 101). Alcohol has been linked to shortened effective refractory period and low alcohol consumption may help reduce sympathetic tone and lower AF risk (33). Also, alcohol consumption has been linked to increased vagal stimulation and increasing the incidence of AF (101). This was more commonly reported in younger individuals and with beer and red wine consumption. Additionally, magnesium, present in foods like nuts, seeds, and leafy greens, plays a role in regulating autonomic function and may have antiarrhythmic effects with low magnesium levels been linked to increased automaticity (63). There have been multiple reports of vagal AF induced by ingestion of cold beverages as well (102). Vagal triggers for AF, such as eating or occurrences solely at night without adrenergic triggers, have been reported by a significant proportion of patients with paroxysmal AF (103).
Cell membrane stability
Deficiencies in certain micronutrients, such as magnesium and potassium, have been implicated in AF pathogenesis. By its effect on the slow-activating delayed rectifier K channel (IKs) and calcium channels (L-type) in the atria, Magnesium can stabilize the cardiac cell membrane and play a protective role against AF (104). PUFAs stabilize cardiomyocyte membranes and their integration into the cell membrane's phospholipids alters ionic currents, such as the sodium and calcium channels, leading to antiarrhythmic effects (105). Chronic administration of PUFAs results in membrane incorporation that modifies ion channel behaviors and reduces arrhythmogenic activity. By inhibiting atrial-specific potassium currents and decreasing Na+ -Ca2+ exchange current, it reduces delayed after depolarizations and arrhythmia risk. Balanced diets containing adequate amounts of these nutrients, along with supplementation, when necessary, may help maintain cardiac electrical stability and reduce AF susceptibility (64).
In summary, various dietary factors and micronutrients can influence multiple mechanisms involved in AF pathogenesis, including inflammation, oxidative stress, electrical and structural remodeling, autonomic dysfunction, and metabolic abnormalities (Figure 1). Adopting a balanced and nutrient-rich diet may reduce AF risk and improve outcomes. However, individual dietary strategies should be tailored based on specific patient characteristics and influenced by potentially identifiable individual underlying mechanisms contributing to AF.

Figure 1. Graphic representation of recognized positive and negative dietary risk factors and their interaction with individual non dietary risk factors. The poorly understood contribution of some dietary components, triggers, genetic predisposition and gut bacteriome is represented with question marks.
Highlighted studies
Future directions and clinical implications
Future directions and clinical implications of exploring diets and micronutrients in their relationship with AF encompass several key areas, focusing on both preventive strategies and therapeutic interventions. Here are some potential future directions and their clinical implications:
Precision nutrition approaches
Future research may delve into personalized nutrition strategies tailored to individuals based on their genetic predispositions, comorbidities, and lifestyle factors. Precision nutrition aims to optimize dietary interventions for AF prevention and management by considering individual variations in metabolism, gut microbiota composition, and dietary preferences. Clinical implementation of precision nutrition in AF management could involve comprehensive dietary assessments, genetic testing, and targeted nutritional interventions tailored to each patient's specific needs.
Clinical trials of dietary interventions
Large-scale randomized controlled trials investigating the efficacy of specific dietary interventions in preventing or managing AF are needed. These trials should evaluate various dietary patterns (e.g., Mediterranean diet, DASH diet), individual nutrients (e.g., omega-3 fatty acids, magnesium), and dietary supplements (e.g., antioxidants, vitamin D) in diverse patient populations with AF. Clinical trials should assess the effects of dietary interventions on AF recurrence and their impact on clinical outcomes, such as AF burden, progression, stroke risk, hospitalizations, heart failure and mortality.
Longitudinal cohort studies
Prospective longitudinal cohort studies with extended follow-up duration are essential for elucidating the long-term effects of dietary habits and nutrient intake on AF incidence and progression. To accurately capture dietary exposures, these studies should incorporate comprehensive dietary assessments, including food frequency questionnaires, 24-h dietary recalls, and biomarker measurements. Longitudinal cohorts can provide valuable insights into the temporal relationship between dietary factors and AF risk, identify potential dietary biomarkers of AF susceptibility, and elucidate the mechanisms underlying diet-AF associations. Modern tools like artificial intelligence and personal digital devices will facilitate collection, management and analysis of more detailed and accurate data points amongst larger populations.
Mechanistic studies
Further mechanistic studies are warranted to better understand the precise pathways through which specific diets and micronutrients influence AF pathogenesis. Mechanistic investigations should explore the effects of dietary components on inflammation, oxidative stress, ion channel function, autonomic tone, cardiac remodeling and other vital mechanisms underlying AF initiation and maintenance. Advanced experimental techniques, such as cellular electrophysiology, tissue engineering, and omics approaches, can help unravel the molecular mechanisms mediating the effects of diet on cardiac electrophysiology and structure.
Integration of nutritional counseling into AF management
Incorporating nutritional counseling and lifestyle modifications into routine AF management can optimize patient care and improve clinical outcomes. Healthcare providers should assess patients’ dietary habits, provide personalized recommendations based on evidence-based guidelines, and support behavior change. Multidisciplinary care teams, including dietitians, nurses, and pharmacists, can collaborate to deliver comprehensive nutritional interventions tailored to each patient's unique needs and preferences. Adequate monitoring and ongoing reinforcement are also recommended.
Telehealth and digital health solutions
Leveraging telehealth and digital health platforms can facilitate not only research but the delivery of dietary counseling and monitoring for patients with AF, especially in remote or underserved areas. Telehealth platforms can enable real-time dietary tracking, remote consultations with healthcare providers, virtual support groups, and nutrition and AF management educational resources. Integrating digital health solutions into routine clinical practice can enhance patient engagement, adherence to dietary recommendations, and self-management of AF.
Future directions in exploring dietary interventions and micronutrients in AF development encompass a multifaceted approach involving precision nutrition, clinical trials, longitudinal cohort studies, mechanistic research, nutritional counseling, and digital health solutions. By advancing our understanding of the role of diet in AF pathophysiology and implementing evidence-based nutritional interventions, healthcare providers can empower patients to adopt healthy dietary habits and reduce their risk of AF-related complications.
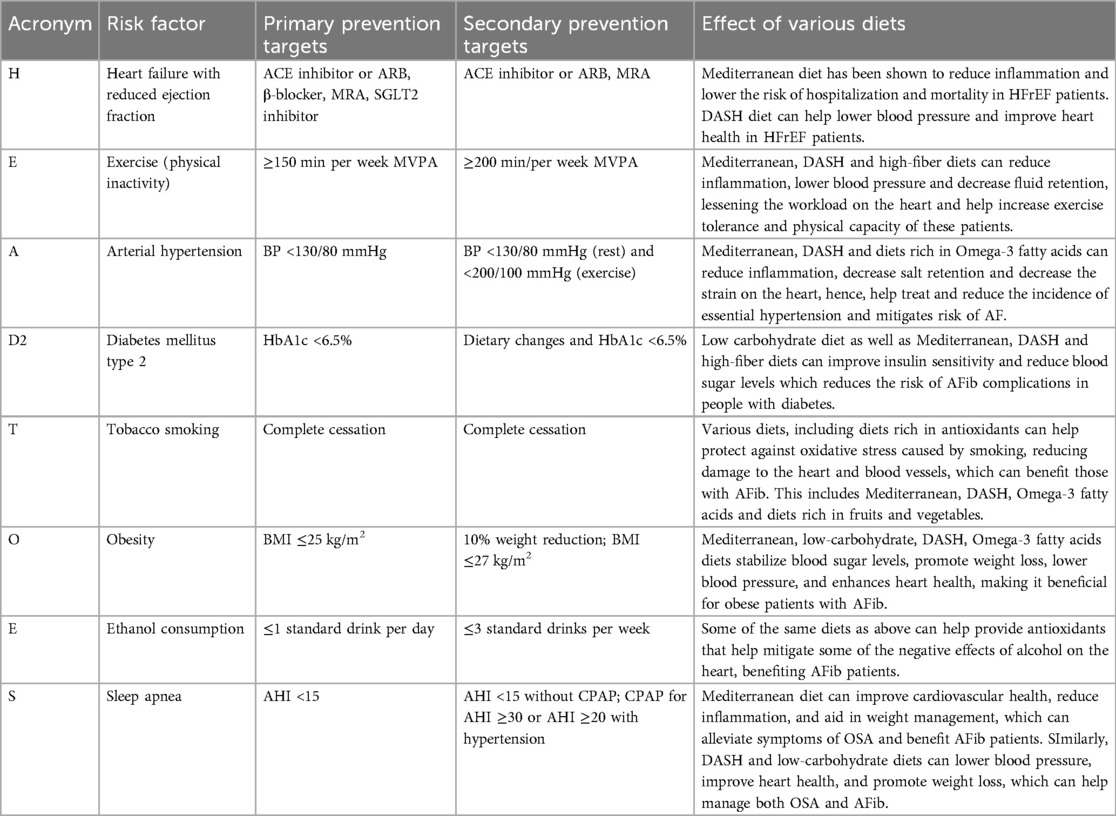
The HEAD-2-TOES scheme
The HEAD-2-TOES scheme was recently proposed as a comprehensive method to assist clinicians in managing and controlling specific risk factors associated with the development of atrial fibrillation (106). Controlling these modifiable risk factors is particularly crucial in the context of primary prevention. This scheme identifies and addresses various determinants, detailed in the table below, and provides an overview of both primary and secondary prevention targets. Additionally, it discusses the impact of various diets on these risk factors, highlighting how dietary choices can influence the effectiveness of prevention strategies (Table 2).
Conclusions
In conclusion, dietary interventions represent a promising avenue in managing AF. This review highlights the growing body of evidence supporting the role of diet in modulating AF risk and progression. A comprehensive analysis of existing literature has elucidated the potential mechanisms by which various dietary patterns, nutrients, and supplements may influence AF pathophysiology. From adopting heart-healthy dietary patterns such as the Mediterranean diet and DASH diet to incorporating specific nutrients like omega-3 fatty acids, magnesium, and antioxidants, dietary interventions offer a holistic approach to AF management. Moreover, emerging research underscores the importance of personalized nutrition strategies tailored to individual patient profiles, genetic predispositions, and lifestyle factors. Large clinical trials, longitudinal cohort studies, and mechanistic research are needed to elucidate further dietary interventions' efficacy, safety, and mechanisms in AF prevention and treatment. By integrating nutritional counseling, lifestyle modifications, and digital health solutions into routine clinical practice, healthcare providers could empower patients to optimize their dietary habits and improve their AF outcomes. Overall, dietary interventions hold promise as adjunctive therapies in the comprehensive management of AF and warrant further exploration in future research endeavors
Important take-home points
• Dietary interventions offer a promising approach for managing atrial fibrillation, with potential in modulating AF risk and progression.
• Adopting heart-healthy dietary patterns such as the Mediterranean diet and DASH diet is particularly beneficial for AF management.
• Incorporating specific nutrients like omega-3 fatty acids, magnesium, and antioxidants can positively impact AF management.
• Emerging research underscores the importance of personalized nutrition strategies tailored to individual patient profiles, genetic predispositions, and lifestyle factors.
• Large clinical trials, longitudinal cohort studies, and mechanistic research are needed to further elucidate the efficacy, safety, and mechanisms of dietary interventions in AF prevention and treatment.
• Integrating nutritional counseling, lifestyle modifications, and digital health solutions into routine clinical practice can empower patients to optimize their dietary habits and improve their AF outcomes.
Data availability statement
Requests to access the datasets should be directed tobXVoYW1tYWQubmFiaWxAYnN3aGVhbHRoLm9yZw==.
Author contributions
MN: Data curation, Investigation, Writing – original draft, Writing – review & editing. LR: Data curation, Investigation, Writing – original draft. AN: Data curation, Investigation, Writing – original draft. PC: Conceptualization, Investigation, Supervision, Writing – review & editing. GO: Conceptualization, Writing – review & editing. JM: Conceptualization, Writing – review & editing. AT: Conceptualization, Writing – review & editing. S-SH: Data curation, Investigation, Writing – original draft. JB: Conceptualization, Supervision, Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Conflict of interest
PC and AT have ownership interest in AlfredAI Inc.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Wyndham CR. Atrial fibrillation: the most common arrhythmia. Tex Heart Inst J. (2000) 27(3):257–67.11093410
2. Ball J, Carrington MJ, McMurray JJ, Stewart S. Atrial fibrillation: profile and burden of an evolving epidemic in the 21st century. Int J Cardiol. (2013) 167(5):1807–24. doi: 10.1016/j.ijcard.2012.12.093
3. Benjamin EJ, Wolf PA, D'Agostino RB, Silbershatz H, Kannel WB, Levy D. Impact of atrial fibrillation on the risk of death: the framingham heart study. Circulation. (1998) 98(10):946–52. doi: 10.1161/01.cir.98.10.946
4. Mariani MV, Pierucci N, Piro A, Trivigno S, Chimenti C, Galardo G, et al. Incidence and determinants of spontaneous cardioversion of early onset symptomatic atrial fibrillation. Medicina (Kaunas). (2022) 58(11):1513. doi: 10.3390/medicina58111513
5. Mariani MV, Pierucci N, Trivigno S, Cipollone P, Piro A, Chimenti C, et al. Probability score to predict spontaneous conversion to sinus rhythm in patients with symptomatic atrial fibrillation when less could be more? J Clin Med. (2024) 13(5):1470. doi: 10.3390/jcm13051470
6. Menezes AR, Lavie CJ, De Schutter A, Milani RV, O’Keefe J, DiNicolantonio JJ, et al. Lifestyle modification in the prevention and treatment of atrial fibrillation. Prog Cardiovasc Dis. (2015) 58(2):117–25, ISSN 0033-0620. doi: 10.1016/j.pcad.2015.07.001
7. Han S, Jia R, Cen Z, Guo R, Zhao S, Bai Y, et al. Early rhythm control vs. rate control in atrial fibrillation: a systematic review and meta-analysis. Front Cardiovasc Med. (2023) 10:978637. doi: 10.3389/fcvm.2023.978637
8. Joglar JA, Chung MK, Armbruster AL, Benjamin EJ, Chyou JY, Cronin EM, et al. 2023 ACC/AHA/ACCP/HRS guideline for the diagnosis and management of atrial fibrillation: a report of the American College of Cardiology/American Heart Association joint committee on clinical practice guidelines. Circulation. (2024) 149(1):e1–156. doi: 10.1161/CIR.0000000000001193. Erratum in: Circulation. 2024 149(1):e167. doi: 10.1161/CIR.0000000000001207. Erratum in: Circulation. 2024 149(9):e936. doi: 10.1161/CIR.0000000000001218. Erratum in: Circulation. 2024 149(24):e1413. doi: 10.1161/CIR.000000000000126338033089
9. Mozaffarian D, Psaty BM, Rimm EB, Lemaitre RN, Burke GL, Lyles MF, et al. Fish intake and risk of incident atrial fibrillation. Circulation. (2004) 110(4):368–73. doi: 10.1161/01.CIR.0000138154.00779.A5
10. Albert CM, Cook NR, Pester J, Moorthy MV, Ridge C, Danik JS, et al. Effect of marine omega-3 fatty acid and vitamin D supplementation on incident atrial fibrillation: a randomized clinical trial. JAMA. (2021) 325(11):1061–73. doi: 10.1001/jama.2021.1489
11. Qian F, Tintle N, Jensen PN, Lemaitre RN, Imamura F, Feldreich TR, et al. Omega-3 fatty acid biomarkers and incident atrial fibrillation. J Am Coll Cardiol. (2023) 82(4):336–49. doi: 10.1016/j.jacc.2023.05.024
12. Li FR, Chen GC, Qin J, Wu X. Dietaryfish and long-chain n-3 polyunsaturated fatty acids intake and risk of atrial fibrillation: a meta-analysis. Nutrients. (2017) 9(9):955. doi: 10.3390/nu9090955
13. Siscovick DS, Raghunathan TE, King I, Weinmann S, Wicklund KG, Albright J, et al. Dietary intake and cell membrane levels of long-chain n-3 polyunsaturated fatty acids and the risk of primary cardiac arrest. JAMA. (1995) 274(17):1363–7. doi: 10.1001/jama.1995.03530170043030
14. Calò L, Martino A, Tota C. The anti-arrhythmic effects of n-3 PUFAs. Int J Cardiol. (2013) 170(2 Suppl 1):S21–7. doi: 10.1016/j.ijcard.2013.06.043
15. Widmer RJ, Flammer AJ, Lerman LO, Lerman A. The Mediterranean diet, its components, and cardiovascular disease. Am J Med. (2015) 128(3):229–38. doi: 10.1016/j.amjmed.2014.10.014
16. Neumann FA, Jagemann B, Makarova N, Börschel CS, Aarabi G, Gutmann F, et al. Mediterranean diet and atrial fibrillation: lessons learned from the AFHRI case-control study. Nutrients. (2022) 14(17):3615. doi: 10.3390/nu14173615
17. Barrio-Lopez MT, Ruiz-Canela M, Ramos P, Tercedor L, Ibañez Criado JL, Ortiz M, et al. PREvention of recurrent arrhythmias with Mediterranean diet (PREDIMAR) study in patients with atrial fibrillation: rationale, design and methods. Am Heart J. (2020) 220:127–36. doi: 10.1016/j.ahj.2019.10.009
18. Martínez-González MÁ, Toledo E, Arós F, Fiol M, Corella D, Salas-Salvadó J, et al. Extravirgin olive oil consumption reduces risk of atrial fibrillation: the PREDIMED (prevención con dieta mediterránea) trial. Circulation. (2014) 130(1):18–26. doi: 10.1161/CIRCULATIONAHA.113.006921. Erratum in: Circulation. 2019 140(24):e939.
19. Razquin C, Ruiz-Canela M, Toledo E, Hernández-Alonso P, Clish CB, Guasch-Ferré M, et al. Metabolomics of the tryptophan-kynurenine degradation pathway and risk of atrial fibrillation and heart failure: potential modification effect of Mediterranean diet. Am J Clin Nutr. (2021) 114(5):1646–54. doi: 10.1093/ajcn/nqab238. Erratum in: Am J Clin Nutr. 2022 115(1):310.34291275
20. Goni L, de la O V, Barrio-López MT, Ramos P, Tercedor L, Ibañez-Criado JL, et al. A remote nutritional intervention to change the dietary habits of patients undergoing ablation of atrial fibrillation: randomized controlled trial. J Med Internet Res. (2020) 22(12):e21436. doi: 10.2196/21436
21. Pignatelli P, Pastori D, Farcomeni A, Nocella C, Bartimoccia S, Vicario T, et al. Mediterranean diet reduces thromboxane A2 production in atrial fibrillation patients. Clin Nutr. (2015) 34(5):899–903. doi: 10.1016/j.clnu.2014.09.011
22. Pastori D, Carnevale R, Nocella C, Novo M, Santulli M, Cammisotto V, et al. Gut-derived serum lipopolysaccharide is associated with enhanced risk of major adverse cardiovascular events in atrial fibrillation: effect of adherence to Mediterranean diet. J Am Heart Assoc. (2017) 6(6):e005784. doi: 10.1161/JAHA.117.005784
23. Appel LJ, Moore TJ, Obarzanek E, Vollmer WM, Svetkey LP, Sacks FM, et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH collaborative research group. N Engl J Med. (1997) 336(16):1117–24. doi: 10.1056/NEJM199704173361601
24. Patel YR, Robbins JM, Gaziano JM, Djoussé L. Mediterranean, DASH, and alternate healthy eating Index dietary patterns and risk of death in the physicians’ health study. Nutrients. (2021) 13(6):1893. doi: 10.3390/nu13061893
25. Tu SJ, Gallagher C, Elliott AD, Bradbury KE, Marcus GM, Linz D, et al. Associations of dietary patterns, ultra-processed food and nutrient intake with incident atrial fibrillation. Heart. (2023) 109(22):1683–9. doi: 10.1136/heartjnl-2023-322412
26. Leopoulou M, Theofilis P, Kordalis A, Papageorgiou N, Sagris M, Oikonomou E, et al. Diabetes mellitus and atrial fibrillation-from pathophysiology to treatment. World J Diabetes. (2023) 14(5):512–27. doi: 10.4239/wjd.v14.i5.512
27. Pathak RK, Middeldorp ME, Lau DH, Mehta AB, Mahajan R, Twomey D, et al. Aggressive risk factor reduction study for atrial fibrillation and implications for the outcome of ablation: the ARREST-AF cohort study. J Am Coll Cardiol. (2014) 64(21):2222–31. doi: 10.1016/j.jacc.2014.09.028
28. Zhang S, Zhuang X, Lin X, Zhong X, Zhou H, Sun X, et al. Low-carbohydrate diets and risk of incident atrial fibrillation: a prospective cohort study. J Am Heart Assoc. (2019) 8(9):e011955. doi: 10.1161/JAHA.119.011955
29. Frederiksen TC, Christiansen MK, Benjamin EJ, Overvad K, Olsen A, Dahm CC, et al. Five-year changes in alcohol intake and risk of atrial fibrillation: a Danish cohort study. Eur J Prev Cardiol. (2023) 30(11):1046–53. doi: 10.1093/eurjpc/zwac293
30. Voskoboinik A, Prabhu S, Ling LH, Kalman JM, Kistler PM. Alcohol and atrial fibrillation: a sobering review. J Am Coll Cardiol. (2016) 68(23):2567–76. doi: 10.1016/j.jacc.2016.08.074
31. Samokhvalov AV, Irving HM, Rehm J. Alcohol consumption as a risk factor for atrial fibrillation: a systematic review and meta-analysis. Eur J Cardiovasc Prev Rehabil. (2010) 17(6):706–12. doi: 10.1097/HJR.0b013e32833a1947
32. Voskoboinik A, Kalman JM, De Silva A, Nicholls T, Costello B, Nanayakkara S, et al. Alcohol abstinence in drinkers with atrial fibrillation. N Engl J Med. (2020) 382(1):20–8. doi: 10.1056/NEJMoa1817591
33. Gallagher C, Hendriks JML, Elliott AD, Wong CX, Rangnekar G, Middeldorp ME, et al. Alcohol and incident atrial fibrillation—a systematic review and meta-analysis. Int J Cardiol. (2017) 246:46–52. doi: 10.1016/j.ijcard.2017.05.133
34. Green PH, Cellier C. Celiac disease. N Engl J Med. (2007) 357(17):1731–43. doi: 10.1056/NEJMra071600
35. Emilsson L, Andersson B, Elfström P, Green PH, Ludvigsson JF. Risk of idiopathic dilated cardiomyopathy in 29,000 patients with celiac disease. J Am Heart Assoc. (2012) 1(3):e001594. doi: 10.1161/JAHA.112.001594
36. Ludvigsson JF, Montgomery SM, Ekbom A, Brandt L, Granath F. Small-intestinal histopathology and mortality risk in celiac disease. JAMA. (2009) 302(11):1171–8. doi: 10.1001/jama.2009.1320
37. Burstein B, Nattel S. Atrial fibrosis: mechanisms and clinical relevance in atrial fibrillation. J Am Coll Cardiol. (2008) 51(8):802–9. doi: 10.1016/j.jacc.2007.09.064
38. Emilsson L, Smith JG, West J, Melander O, Ludvigsson JF. Increased risk of atrial fibrillation in patients with coeliac disease: a nationwide cohort study. Eur Heart J. (2011) 32(19):2430–7. doi: 10.1093/eurheartj/ehr167
39. Conen D, Ridker PM, Everett BM, Tedrow UB, Rose L, Cook NR, et al. A multimarker approach to assess the influence of inflammation on the incidence of atrial fibrillation in women. Eur Heart J. (2010) 31(14):1730–6. doi: 10.1093/eurheartj/ehq146
40. Wang Y, Chen B, Ciaccio EJ, Jneid H, Virani SS, Lavie CJ, et al. Celiac disease and the risk of cardiovascular diseases. Int J Mol Sci. (2023) 24(12):9974. doi: 10.3390/ijms24129974
41. Banchs JE, Suarez K, Baquero G, LaPine S, Samii S, Wolbrette D, et al. Clinical response to dietary gluten restriction among patients with cardiac arrhythmias. European cardiac arrhythmia society (ECAS). J Interv Card Electrophysiol. (2017) 48(Suppl 1):S127–8. doi: 10.1007/s10840-017-0231-0
42. Mostofsky E, Johansen MB, Lundbye-Christensen S, Tjønneland A, Mittleman MA, Overvad K. Risk of atrial fibrillation associated with coffee intake: findings from the Danish diet, cancer, and health study. Eur J Prev Cardiol. (2016) 23(9):922–30. doi: 10.1177/2047487315624524
43. van Dam RM. Coffee consumption and coronary heart disease: paradoxical effects on biological risk factors versus disease incidence. Clin Chem. (2008) 54(9):1418–20. doi: 10.1373/clinchem.2008.111542
44. Mendoza MF, Sulague RM, Posas-Mendoza T, Lavie CJ. Impact of coffee consumption on cardiovascular health. Ochsner J. (2023) 23(2):152–8. doi: 10.31486/toj.22.0073
45. Abdelfattah R, Kamran H, Lazar J, Kassotis J. Does caffeine consumption increase the risk of new-onset atrial fibrillation? Cardiology. (2018) 140(2):106–14. doi: 10.1159/000489843
46. Graboys TB, Blatt CM, Lown B. The effect of caffeine on ventricular ectopic activity in patients with malignant ventricular arrhythmia. Arch Intern Med. (1989) 149(3):637–9. doi: 10.1001/archinte.1989.00390030105020
47. Conen D, Chiuve SE, Everett BM, Zhang SM, Buring JE, Albert CM. Caffeine consumption and incident atrial fibrillation in women. Am J Clin Nutr. (2010) 92(3):509–14. doi: 10.3945/ajcn.2010.29627
48. Klein A, Shenasa M, Baranchuk A. Social risk factors and atrial fibrillation. Card Electrophysiol Clin. (2021) 13(1):165–72. doi: 10.1016/j.ccep.2020.10.008
49. Mostofsky E, Berg Johansen M, Tjønneland A, Chahal HS, Mittleman MA, Overvad K. Chocolate intake and risk of clinically apparent atrial fibrillation: the Danish diet, cancer, and health study. Heart. (2017) 103(15):1163–7. doi: 10.1136/heartjnl-2016-310357
50. Larsson SC, Drca N, Jensen-Urstad M, Wolk A. Chocolate consumption and risk of atrial fibrillation: two cohort studies and a meta-analysis. Am Heart J. (2018) 195:86–90. doi: 10.1016/j.ahj.2017.09.013
51. Khawaja O, Petrone AB, Kanjwal Y, Gaziano JM, Djoussé L. Chocolate consumption and risk of atrial fibrillation (from the physicians’ health study). Am J Cardiol. (2015) 116(4):563–6. doi: 10.1016/j.amjcard.2015.05.009
52. Stauber A, Müller A, Rommers N, Aeschbacher S, Rodondi N, Bonati LH, et al. Association of chocolate consumption with neurological and cardiovascular outcomes in atrial fibrillation: data from two Swiss atrial fibrillation cohort studies (Swiss-AF and BEAT-AF). Swiss Med Wkly. (2023) 153:40109. doi: 10.57187/smw.2023.40109
53. Yuan S, Li X, Jin Y, Lu J. Chocolate consumption and risk of coronary heart disease, stroke, and diabetes: a meta-analysis of prospective studies. Nutrients. (2017) 9(7):688. doi: 10.3390/nu9070688
54. Faridi Z, Njike VY, Dutta S, Ali A, Katz DL. Acute dark chocolate and cocoa ingestion and endothelial function: a randomized controlled crossover trial. Am J Clin Nutr. (2008) 88(1):58–63. doi: 10.1093/ajcn/88.1.58
55. Wuopio J, Orho-Melander M, Ärnlöv J, Nowak C. Estimated salt intake and risk of atrial fibrillation in a prospective community-based cohort. J Intern Med. (2021) 289(5):700–8. doi: 10.1111/joim.13194
56. Pääkkö TJW, Perkiömäki JS, Silaste ML, Bloigu R, Huikuri HV, Antero Kesäniemi Y, et al. Dietary sodium intake is associated with long-term risk of new-onset atrial fibrillation. Ann Med. (2018) 50(8):694–703. doi: 10.1080/07853890.2018.1546054
57. Goddard J, Speights CJ, Borganelli M. Salt as a trigger for atrial tachycardia/fibrillation. Cureus. (2022) 14(6):e26168. doi: 10.7759/cureus.26168
58. Welsh CE, Welsh P, Jhund P, Delles C, Celis-Morales C, Lewsey JD, et al. Urinary sodium excretion, blood pressure, and risk of future cardiovascular disease and mortality in subjects without prior cardiovascular disease. Hypertension. (2019) 73(6):1202–9. doi: 10.1161/HYPERTENSIONAHA.119.12726
59. Bhagavathula AS, Rahmani J. Salt intake and new-onset of atrial fibrillation: a meta-analysis of over 1.4 million participants. Clin Nutr. (2021) 40(5):2600–1. doi: 10.1016/j.clnu.2021.04.009
60. Karppi J, Kurl S, Mäkikallio TH, Ronkainen K, Laukkanen JA. Low levels of plasma carotenoids are associated with an increased risk of atrial fibrillation. Eur J Epidemiol. (2013) 28(1):45–53. doi: 10.1007/s10654-012-9753-z
61. Parmenter BH, Dalgaard F, Murray K, Marquis-Gravel G, Cassidy A, Bondonno CP, et al. Intake of dietary flavonoids and incidence of ischemic heart disease in the Danish diet, cancer, and health cohort. Eur J Clin Nutr. (2023) 77(2):270–7. doi: 10.1038/s41430-022-01226-y
62. Santhakumar AB, Bulmer AC, Singh I. A review of the mechanisms and effectiveness of dietary polyphenols in reducing oxidative stress and thrombotic risk. J Hum Nutr Diet. (2014) 27(1):1–21. doi: 10.1111/jhn.12177
63. Khan AM, Lubitz SA, Sullivan LM, Sun JX, Levy D, Vasan RS, et al. Low serum magnesium and the development of atrial fibrillation in the community: the Framingham heart study. Circulation. (2013) 127(1):33–8. doi: 10.1161/CIRCULATIONAHA.111.082511
64. Miller S, Crystal E, Garfinkle M, Lau C, Lashevsky I, Connolly SJ. Effects of magnesium on atrial fibrillation after cardiac surgery: a meta-analysis. Heart. (2005) 91(5):618–23. doi: 10.1136/hrt.2004.033811
65. Krijthe BP, Heeringa J, Kors JA, Hofman A, Franco OH, Witteman JC, et al. Serum potassium levels and the risk of atrial fibrillation: the Rotterdam study. Int J Cardiol. (2013) 168(6):5411–5. doi: 10.1016/j.ijcard.2013.08.048
66. Wu Y, Kong XJ, Ji YY, Fan J, Ji CC, Chen XM, et al. Serum electrolyte concentrations and risk of atrial fibrillation: an observational and Mendelian randomization study. BMC Genomics. (2024) 25(1):280. doi: 10.1186/s12864-024-10197-2
67. Mattsson N, Nielsen OW, Johnson L, Prescott E, Schnohr P, Jensen GB, et al. Prognostic impact of mild hypokalemia in terms of death and stroke in the general population-A prospective population study. Am J Med. (2018) 131(3):318.e9–19. doi: 10.1016/j.amjmed.2017.09.026
68. Green DM, Ropper AH, Kronmal RA, Psaty BM, Burke GL, Cardiovascular Health Study. Serum potassium level and dietary potassium intake as risk factors for stroke. Neurology. (2002) 59(3):314–20. doi: 10.1212/wnl.59.3.314
69. Franse LV, Pahor M, Di Bari M, Somes GW, Cushman WC, Applegate WB. Hypokalemia associated with diuretic use and cardiovascular events in the systolic hypertension in the elderly program. Hypertension. (2000) 35(5):1025–30. doi: 10.1161/01.hyp.35.5.1025
70. Kievišas M, Keturakis V, Vaitiekūnas E, Dambrauskas L, Jankauskienė L, Kinduris Š. Prognostic factors of atrial fibrillation following coronary artery bypass graft surgery. Gen Thorac Cardiovasc Surg. (2017) 65(10):566–74. doi: 10.1007/s11748-017-0797-6
71. Lu YY, Cheng CC, Chen YC, Lin YK, Chen SA, Chen YJ. Electrolyte disturbances differentially regulate sinoatrial node and pulmonary vein electrical activity: a contribution to hypokalemia- or hyponatremia-induced atrial fibrillation. Heart Rhythm. (2016) 13(3):781–8. doi: 10.1016/j.hrthm.2015.12.005
72. Gallen IW, Rosa RM, Esparaz DY, Young JB, Robertson GL, Batlle D, et al. On the mechanism of the effects of potassium restriction on blood pressure and renal sodium retention. Am J Kidney Dis. (1998) 31(1):19–27. doi: 10.1053/ajkd.1998.v31.pm9428447
73. Lancaster TS, Schill MR, Greenberg JW, Moon MR, Schuessler RB, Damiano RJ Jr, et al. Potassium and magnesium supplementation do not protect against atrial fibrillation after cardiac operation: a time-matched analysis. Ann Thorac Surg. (2016) 102(4):1181–8. doi: 10.1016/j.athoracsur.2016.06.066
74. Tsai YC, Tsao YP, Huang CJ, Tai YH, Su YC, Chiang CE, et al. Effectiveness of salt substitute on cardiovascular outcomes: a systematic review and meta-analysis. J Clin Hypertens (Greenwich). (2022) 24(9):1147–60. doi: 10.1111/jch.14562
75. Filippini T, Naska A, Kasdagli M-I, Torres D, Lopes C, Carvalho C, et al. Potassium intake and blood pressure: a dose-response meta-analysis of randomized controlled trials. J Am Heart Assoc. (2020) 9(12):e015719. doi: 10.1161/JAHA.119.015719
76. Greer RC, Marklund M, Anderson CAM, Cobb LK, Dalcin AT, Henry M, et al. Potassium-enriched salt substitutes as a means to lower blood pressure: benefits and risks. Hypertension. (2020) 75(2):266–74. doi: 10.1161/HYPERTENSIONAHA.119.13241
77. Neal B, Wu Y, Feng X, Zhang R, Zhang Y, Shi J, et al. Effect of salt substitution on cardiovascular events and death. N Engl J Med. (2021) 385(12):1067–77. doi: 10.1056/NEJMoa2105675
78. Al-Mubarak AA, Grote Beverborg N, Zwartkruis V, van Deutekom C, de Borst MH, Gansevoort RT, et al. Micronutrient deficiencies and new-onset atrial fibrillation in a community-based cohort: data from PREVEND. Clin Res Cardiol. (2023) 112:968–78. doi: 10.1007/s00392-023-02276-3
79. Wolff AA, Levi R. Histamine and cardiac arrhythmias. Circ Res. (1986) 58(1):1–16. doi: 10.1161/01.res.58.1.1
80. Rohr SM, Rich MW, Silver KH. Shortness of breath, syncope, and cardiac arrest caused by systemic mastocytosis. Ann Emerg Med. (2005) 45(6):592–4. doi: 10.1016/j.annemergmed.2005.02.002
81. Layritz CM, Hagel AF, Graf V, Reiser C, Klinghammer L, Ropers D, et al. Histamine in atrial fibrillation (AF)–is there any connection? Results from an unselected population. Int J Cardiol. (2014) 172(3):e432–3. doi: 10.1016/j.ijcard.2013.12.185
82. Pathak RK, Middeldorp ME, Meredith M, Mehta AB, Mahajan R, Wong CX, et al. Long-term effect of goal-directed weight management in an atrial fibrillation cohort: a long-term follow-up study (LEGACY). J Am Coll Cardiol. (2015) 65(20):2159–69. doi: 10.1016/j.jacc.2015.03.002
83. Shu H, Cheng J, Li N, Zhang Z, Nie J, Peng Y, et al. Obesity and atrial fibrillation: a narrative review from arrhythmogenic mechanisms to clinical significance. Cardiovasc Diabetol. (2023) 22(1):192. doi: 10.1186/s12933-023-01913-5
84. Jamaly S, Carlsson L, Peltonen M, Jacobson P, Sjöström L, Karason K. Bariatric surgery and the risk of new-onset atrial fibrillation in Swedish obese subjects. J Am Coll Cardiol. (2016) 68(23):2497–504. doi: 10.1016/j.jacc.2016.09.940
85. Badheka AO, Rathod A, Kizilbash MA, Garg N, Mohamad T, Afonso L, et al. Influence of obesity on outcomes in atrial fibrillation: yet another obesity paradox. Am J Med. (2010) 123(7):646–51. doi: 10.1016/j.amjmed.2009.11.026
86. Yu L, Meng G, Huang B, Zhou X, Stavrakis S, Wang M, et al. A potential relationship between gut microbes and atrial fibrillation: trimethylamine N-oxide, a gut microbe-derived metabolite, facilitates the progression of atrial fibrillation. Int J Cardiol. (2018) 255:92–8. doi: 10.1016/j.ijcard.2017.11.071
87. Yang WT, Yang R, Zhao Q, Li XD, Wang YT. A systematic review and meta-analysis of the gut microbiota-dependent metabolite trimethylamine N-oxide with the incidence of atrial fibrillation. Ann Palliat Med. (2021) 10(11):11512–23. doi: 10.21037/apm-21-2763
88. Zuo K, Yin X, Li K, Zhang J, Wang P, Jiao J, et al. Different types of atrial fibrillation share patterns of gut Microbiota dysbiosis. mSphere. (2020) 5(2):e00071–20. doi: 10.1128/mSphere.00071-20
89. Alonso A, Yu B, Sun YV, Chen LY, Loehr LR, O’Neal WT, et al. Serum metabolomics and incidence of atrial fibrillation (from the atherosclerosis risk in communities study). Am J Cardiol. (2019) 123:1955–61. doi: 10.1016/j.amjcard.2019.03.017
90. Fang C, Zuo K, Liu Z, Liu Y, Liu L, Wang Y, et al. Disordered gut microbiota promotes atrial fibrillation by aggravated conduction disturbance and unbalanced linoleic acid/SIRT1 signaling. Biochem Pharmacol. (2023) 213:115599. doi: 10.1016/j.bcp.2023.115599
91. Zhang Y, Zhang S, Li B, Luo Y, Gong Y, Jin X, et al. Gut microbiota dysbiosis promotes age-related atrial fibrillation by lipopolysaccharide and glucose-induced activation of NLRP3-inflammasome. Cardiovasc Res. (2022) 118(3):785–97. doi: 10.1093/cvr/cvab114
92. Lu D, Zou X, Zhang H. The relationship between atrial fibrillation and intestinal flora with its metabolites. Front Cardiovasc Med. (2022) 9:948755. doi: 10.3389/fcvm.2022.948755
93. Korantzopoulos P, Letsas K, Fragakis N, Tse G, Liu T. Oxidative stress and atrial fibrillation: an update. Free Radical Res. (2018) 52(11-12):1199–209. doi: 10.1080/10715762.2018.1500696
94. Zhang Q, Guo Y, Li M, Yang R, Yao Y, Zhao Y, et al. Excessive dietary sodium intake augments long-term risk of atrial fibrillation in older adults with hyperglycemia: a community-based prospective cohort study. Diabetes Metab. (2023) 49(5):101475. doi: 10.1016/j.diabet.2023.101475
95. Iwasaki YK, Nishida K, Kato T, Nattel S. Atrial fibrillation pathophysiology: implications for management. Circulation. (2011) 124(20):2264–74. doi: 10.1161/CIRCULATIONAHA.111.019893
96. Rajagopalan B, Shah Z, Narasimha D, Bhatia A, Kim CH, Switzer DF, et al. Efficacy of intravenous magnesium in facilitating cardioversion of atrial fibrillation. Circ Arrhythm Electrophysiol. (2016) 9(9):e003968. doi: 10.1161/CIRCEP.116.003968
97. Carnes CA, Janssen PM, Ruehr ML, Nakayama H, Nakayama T, Haase H, et al. Atrial glutathione content, calcium current, and contractility. J Biol Chem. (2007) 282(38):28063–73. doi: 10.1074/jbc.M704893200
98. Chen PS, Chen LS, Fishbein MC, Lin SF, Nattel S. Role of the autonomic nervous system in atrial fibrillation: pathophysiology and therapy. Circ Res. (2014) 114(9):1500–15. doi: 10.1161/CIRCRESAHA.114.303772
99. Coumel P. Paroxysmal atrial fibrillation: a disorder of autonomic tone? Eur Heart J. (1994) 15(Suppl A):9–16. doi: 10.1093/eurheartj/15.suppl_a.9
100. Coumel P, Attuel P, Lavallée J, Flammang D, Leclercq JF, Slama R. Syndrome d'arythmie auriculaire d'origine vagale [the atrial arrhythmia syndrome of vagal origin]. Arch Mal Coeur Vaiss. (1978) 71(6):645–56. French.28709
101. Mandyam MC, Vedantham V, Scheinman MM, Tseng ZH, Badhwar N, Lee BK, et al. Alcohol and vagal tone as triggers for paroxysmal atrial fibrillation. Am J Cardiol. (2012) 110(3):364–8. doi: 10.1016/j.amjcard.2012.03.033
102. Routledge HC, Chowdhary S, Coote JH, Townend JN. Cardiac vagal response to water ingestion in normal human subjects. Clin Sci (Lond). (2002) 103(2):157–62. doi: 10.1042/cs1030157
103. Zhang J, Zuo K, Fang C, Yin X, Liu X, Zhong J, et al. Altered synthesis of genes associated with short-chain fatty acids in the gut of patients with atrial fibrillation. BMC Genomics. (2021) 22(1):634. doi: 10.1186/s12864-021-07944-0
104. Salvador-Montañés O, Gómez-Gallanti A, Garofalo D, Noujaim SF, Peinado R, Filgueiras-Rama D. Polyunsaturated fatty acids in atrial fibrillation: looking for the proper candidates. Front Physiol. (2012) 3:370. doi: 10.3389/fphys.2012.00370
105. Gawałko M, Agbaedeng TA, Saljic A, Müller DN, Wilck N, Schnabel R, et al. Gut microbiota, dysbiosis and atrial fibrillation. Arrhythmogenic mechanisms and potential clinical implications. Cardiovasc Res. (2022) 118(11):2415–27. doi: 10.1093/cvr/cvab292
Keywords: atrial fibrillation, diet, nutrients, alcohol, caffeine, prevention
Citation: Nabil MA, Rychlik L, Nicholson A, Cheung P, Olsovsky GD, Molden J, Tripuraneni A, Hajivandi S-S and Banchs JE (2024) Dietary interventions in the management of atrial fibrillation. Front. Cardiovasc. Med. 11: 1418059. doi: 10.3389/fcvm.2024.1418059
Received: 16 April 2024; Accepted: 22 July 2024;
Published: 1 August 2024.
Edited by:
Rui Providencia, University College London, United KingdomReviewed by:
Carlo Lavalle, Sapienza University of Rome, ItalyGermanas Marinskis, Vilnius University, Lithuania
© 2024 Nabil, Rychlik, Nicholson, Cheung, Olsovsky, Molden, Tripuraneni, Hajivandi and Banchs. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Muhammad Ahad Nabil, TXVoYW1tYWQubmFiaWxAQlNXSGVhbHRoLm9yZw==
 Muhammad Ahad Nabil
Muhammad Ahad Nabil Leanne Rychlik
Leanne Rychlik Ajay Tripuraneni
Ajay Tripuraneni Javier E. Banchs
Javier E. Banchs