- Department of Pharmacy, The Third Xiangya Hospital, Central South University, Changsha, China
Background: Cephalosporins are an increasingly encountered cause of Kounis syndrome. The present study examined the clinical features of cephalosporin-induced Kounis syndrome and provided references for diagnosis, prevention, treatment, and prognosis.
Methods: We collected cephalosporin-induced Kounis syndrome case reports by searching Chinese and English databases from the establishment of the database to October 31, 2021.
Results: Twenty-five patients (17 males and eight females) were included, with a median age of 61 years (range 33–92). Cephalosporins were administered via oral, intravenous and intramuscular routes. All reactions occurred within 30 min, except in two patients. Fourteen patients experienced chest pain, 19 experienced hypotension, 16 had cutaneous reactions, 10 had respiratory symptoms, and seven had gastrointestinal symptoms. Thirteen patients had elevated troponin levels, and eight patients had elevated serum tryptase levels. The electrocardiogram showed ST-segment elevation in 13 patients, depression in four patients, and elevation and depression in six patients. Coronary angiography showed normal results in 12 patients and abnormal results in 13 patients. The skin prick test was positive for cephalosporin in three patients. Twenty-four of the 25 patients recovered after being given anti-allergic and acute coronary syndrome treatment, and there was one death.
Conclusions: Kounis syndrome is a serious adverse reaction to cephalosporin. Clinicians should consider Kounis syndrome in every patient receiving cephalosporin and presenting with acute chest pain or anaphylactic symptoms.
Introduction
Anaphylaxis is a severe, life-threatening, generalized or systemic hypersensitivity reaction (1). Cardiac tissue is susceptible to hypersensitivity processes (2). Myocardium, conduction system and coronary artery reactions to various allergens are well- established. The clinical condition of allergic angina syndrome was not described until 1991, as coronary spasm progresses to allergic acute myocardial infarction (3). Kounis syndrome (KS) is an acute coronary syndrome caused by an allergic reaction to foods, drugs, environmental exposures, and various conditions (4). KS is mediated by mast cells that interact with macrophages and T lymphocytes and results in the massive release of inflammatory mediators in cardiac tissue, coronary arteries and plaques (5). The main clinical signs and symptoms of KS are associated with allergic reactions accompanied by cardiac symptomatology. It is not a rare disease, but it is infrequently diagnosed. Although the incidence of KS is not clear, its special clinical manifestations and treatment have attracted clinical attention.
Antibiotics are the most common culprit for KS (6). Cephalosporins are commonly used antibiotics in hospitalized patients and outpatients. Hypersensitivity is the most common adverse reaction to cephalosporins (7). However, KS is a rare side effect of cephalosporins. Limited data are available for cephalosporin-induced KS. The present article collected relevant case reports to examine the cephalosporin types, clinical manifestations, electrocardiographic changes, laboratory abnormalities, and echocardiographic and angiographic findings of cephalosporin-induced KS. This research is of great significance to the diagnosis, treatment, prognosis and prevention of cephalosporin-induced KS. This research also provides a basis for clinicians to improve their understanding and diagnosis of KS.
Materials and Methods
Search Strategy
We searched databases from the establishment to October 31, 2021, including PubMed, Embase, The Cochrane Library, CNKI, VIP database and Wanfang database. The search method used a combination of subject words and free words, including Kounis syndrome, acute coronary syndrome, acute myocardial infarction, myocardial infarction, allergic angina syndrome, allergic angina, allergic myocardial infarction, vasospastic allergic angina, coronary artery disease, coronary spasm, coronary thrombosis, stent thrombosis, myocardial ischemia, chest pain, hypersensitivity, anaphylactic, anaphylactoid, antibiotics, cephalosporins (first-, second-, third-, fourth- and fifth-generation cephalosporins listed), beta-lactams, and adverse reactions.
Data Extraction
We used a self-designed table to extract relevant information of the patient, including country, sex, age, underlying disease, combined medication, cephalosporin administration, clinical manifestations, laboratory examinations, imaging examinations, treatment and prognosis.
Results
Basic Information
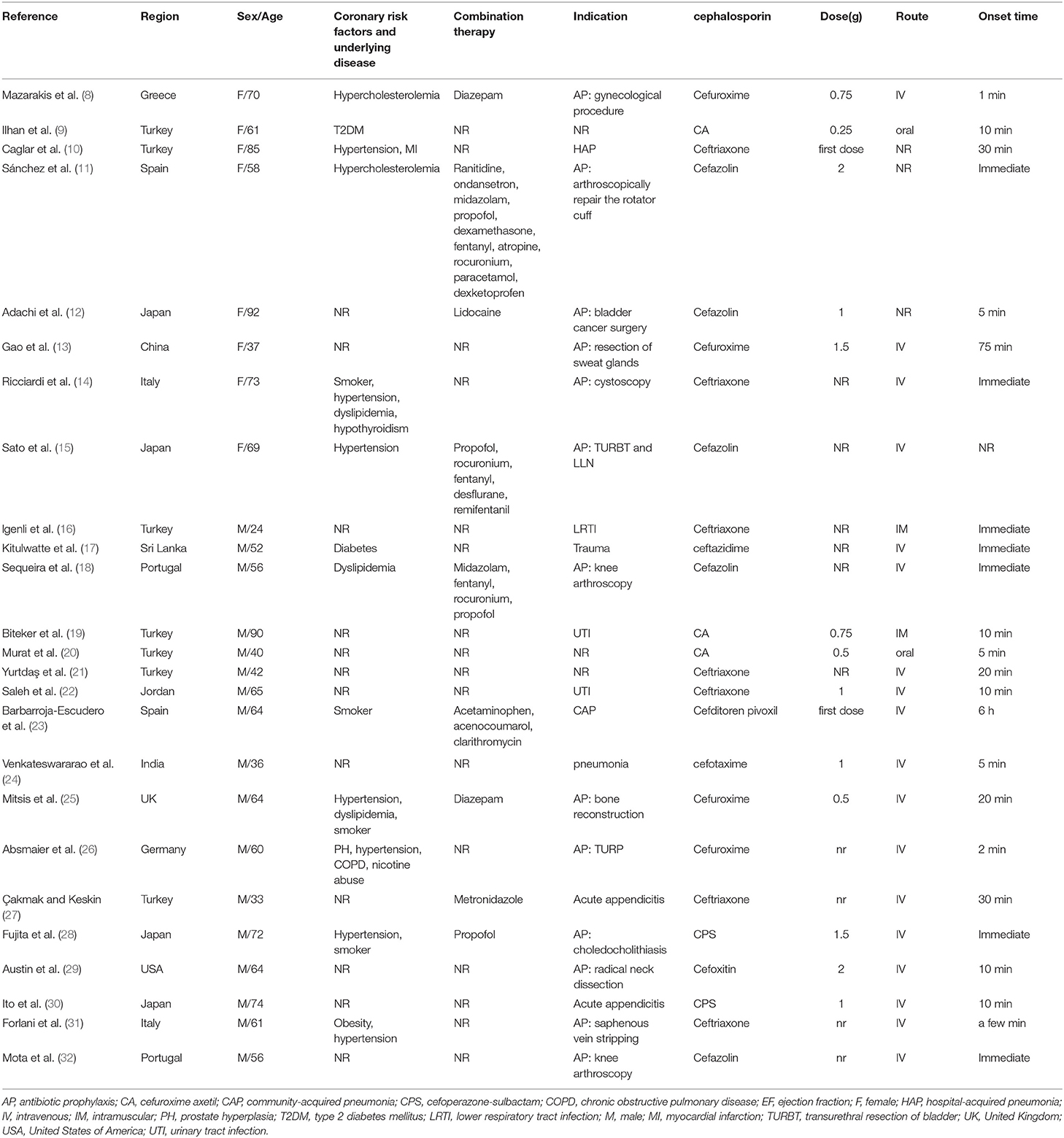
We initially identified 832 studies. Two hundred and ninety seven replicate studies were excluded. After an initial screening of titles and abstracts, a total of 467 articles were removed. Of the remaining 68 studies, a total of 25 articles were included after full-text screening (8–32). Patient information is summarized in Table 1. Twenty-five patients (17 males and eight females) were primarily from Europe (seven from Turkey), with a median age of 61 years (33–92 years). Cephalosporins were primarily used for perioperative antibiotic prophylaxis (13 patients) and infection treatment (nine patients). The cephalosporins included cefuroxime (seven patients), ceftriaxone (seven patients), cefazolin (five patients), cefoperazone-sulbactam (two patients), cefotaxime (one patient), cefoxitin (one patient), ceftazidime (one patient), and cefditoren (one patient). The route of administration included intravenous (18 patients), oral (two patients), intramuscular (two patients), and unknown (three patients). Thirteen patients had risk factors for KS, and 9 patients used other drugs simultaneously.
Clinical Manifestations
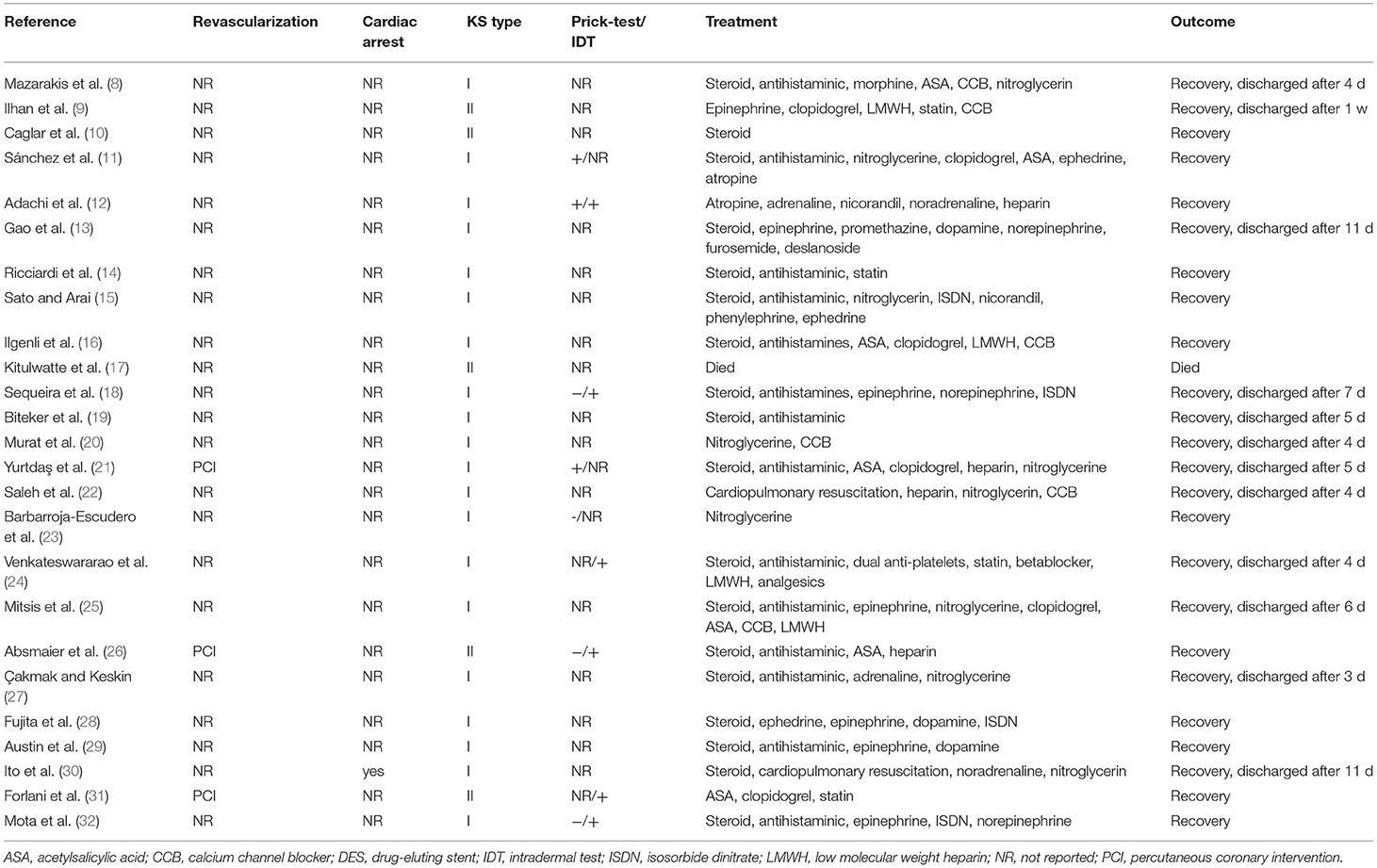
The clinical characteristics of the 25 included patients are summarized in Table 2. The time of administration and symptom onset varied from immediate to 6 h. Fourteen patients developed chest pain, and 19 patients developed hypotension. Allergic skin reactions occurred in 16 patients, including skin rash (15 patients) and itching (eight patients). Ten patients developed respiratory symptoms or signs, seven patients developed gastrointestinal symptoms, and nine patients developed neurological symptoms. One patient experienced cardiac arrest.
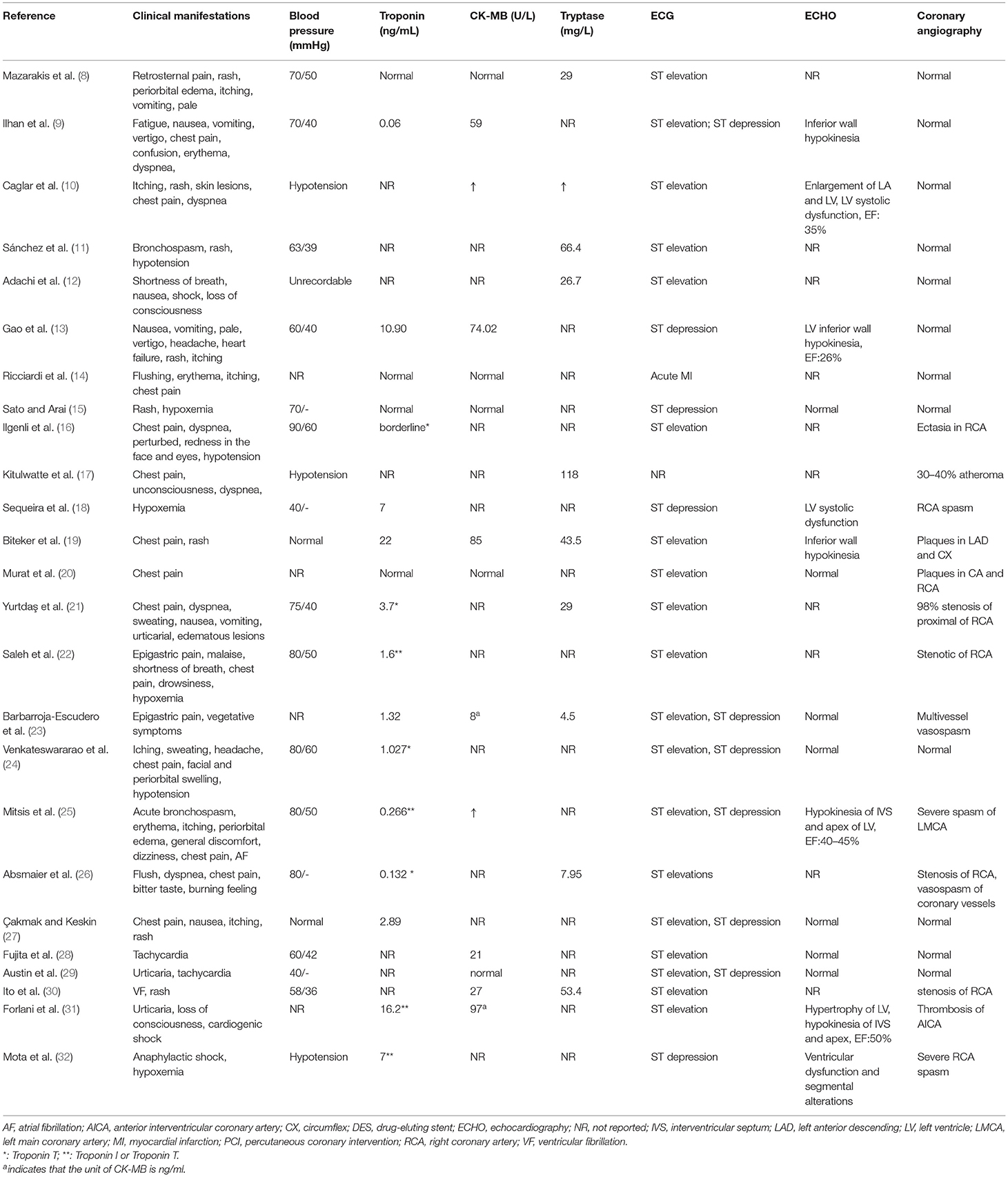
Table 2. Clinical manifestations and laboratory and imaging examinations of the 25 included patients.
Laboratory Examination
The laboratory test results are summarized in Table 2. Troponin levels were elevated in 13 of 18 patients, and creatine kinase-MB was elevated in nine of 14 patients. Serum tryptase was elevated in eight of 10 patients. The skin prick test was positive in three of seven patients. Intradermal tests were positive in six patients.
Imaging Examination
The imaging examination results are summarized in Table 2. Electrocardiograms (ECGs) primarily showed ST elevation (13 patients), ST depression (four patients), and ST elevation and ST depression (6 patients). Echocardiography in 15 patients showed hypokinesia (five patients), left ventricular systolic dysfunction (three patients), and reduced ejection fraction (four patients). Coronary angiography in 13 patients primarily showed spasm (five patients), stenosis (four patients) and plaque (three patients).
Treatment
The treatment and prognosis of the 25 included patients are summarized in Table 3. Drug treatment included corticosteroids (18 patients), antihistamines (15 patients), epinephrine (nine patients), vasodilators (16 patients), and antiplatelet drugs (nine patients). Two patients underwent cardiopulmonary resuscitation. Three patients underwent revascularization. Twenty-four patients eventually recovered, and one patient died. Twenty-one patients belonged to the type I variant, and 4 patients belonged to the type II variant.
Discussion
Three variants of KS have been described: type I variant (coronary artery spasm, no risk factors for coronary heart disease); type II variant (previous history of coronary atherosclerosis); and type III variant (previous history of coronary stent implantation). The clinical features of KS are the simultaneous appearance of acute myocardial ischemia and acute allergic reactions (4). KS should be suspected for acute coronary syndrome with chest pain symptoms accompanied by allergic reactions. ECG, coronary angiography, cardiac markers, and tryptase help identify this syndrome (2). Coronary stents are an important means of treating ischemic heart disease, including bare metal stents with platforms, drug-eluting stents and bioabsorbable stents (33). Previous reports have demonstrated that all stent components, namely the stent platform with their metals (e.g., nickel, chromium, titanium, manganese, and molybdenum), polymer coatings, and released drugs are strong allergens which apply continuous, repetitive, persistent and chronic allergic irritation to the coronary intima (4, 34). All these types of stents are accompanied by rare but worrying stent thrombosis. Thus, stent thrombosis is primarily a manifestation of KS.
KS has a geographical distribution (4). Cephalosporin-induced KS is primarily distributed in Europe and Asia, especially Turkey and Japan. Our study confirmed that cefuroxime, ceftriaxone, and cefazolin were the most frequently reported cephalosporin antibiotics. The time from exposure to trigger to onset of KS was within 30 min in 80% of cephalosporin-induced KS patients. Our analysis found that many cases occurred during the perioperative period. The high incidence of cephalosporin-induced KS in the perioperative period may be due to the relatively frequent use of antibiotics in the perioperative period. Potential cross-reactions may occur with different β-lactams. Cephalosporins consist of a β-lactam ring and hydrothiazide ring. Two side chains (R1 and R2) distinguish the different cephalosporins. The cephalosporins cefuroxime, cefotaxime, cefpodoxime proxetil and ceftriaxone have methoxyimino groups on the R1 side chain, which may cause cross-reactions (35). We also cannot exclude the possibility of KS caused by the use of other concomitant drugs, such as metronidazole and rocuronium (36, 37).
Most studies suggest that the mechanism of KS is similar to allergic reactions. The main inflammatory cells, mast cells, interact with macrophages and T lymphocytes to cause the release of inflammatory mediators, including histamine, neutral proteases, arachidonic acid products, platelet activating factor and heparin, which lead to coronary spasm and coronary atherosclerosis plaque rupture or thrombosis in coronary stents (38).
The treatment of KS is extremely challenging because the heart symptoms and allergic symptoms must be considered simultaneously. There is no standard of treatment for KS, and treatment recommendations are based on the experience of case reports. Treatment of the allergic events with corticosteroids or H1 and H2 blockers alone eliminates symptoms in patients with type I variant (39, 40). The administration of calcium channel blockers and nitrates eliminates allergy-induced coronary artery spasms (36). For patients with type II and III variants, treatment of the acute myocardial ischemia and acute allergies is required (41). Epinephrine is a first-line drug for the treatment of severe allergies, but it should be used with caution in KS because it can aggravate myocardial ischemia and cause coronary artery spasm (42). Beta-blockers may exacerbate coronary spasm due to the lack of antagonism of alpha-adrenergic receptors (42). Opioids, such as morphine, codeine and pethidine, relieve the acute chest pain (43).
The prognostic factors for KS include the type of KS, the presence of complications, and the presence of allergens. Most patients with KS can expect a full recovery with appropriate treatment (43). Patients with type I KS have the best prognosis (44). Our research showed that cephalosporin-induced KS had a better prognosis. However, serious complications related to KS may occur, such as cardiac arrest and death.
Conclusion
Clinicians and pharmacists should be aware of cephalosporin-induced KS to ensure the use appropriate therapeutic interventions and preventative measures. Antihistamines and steroids may be used to treat allergic reactions and nitrates and/or calcium channel blockers may be needed to treat coronary artery spasms.
Data Availability Statement
The original contributions presented in the study are included in the article, further inquiries can be directed to the corresponding author.
Author Contributions
WF and CW conceived the presented idea. WF, LS, ZD, WS, ZL, and CW wrote the manuscript. All authors discussed the results and contributed to the final manuscript.
Funding
This study was supported by research grants from the National Natural Science Foundation of China (81900344).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Johansson SG, Bieber T, Dahl R, Friedmann PS, Lanier BQ, Lockey RF, et al. Revised nomenclature for allergy for global use: report of the nomenclature review committee of the world allergy organization, October 2003. J Allergy Clin Immunol. (2004) 113:832–6. doi: 10.1016/j.jaci.2003.12.591
2. Abdelghany M, Subedi R, Shah S, Kozman H. Kounis syndrome: a review article on epidemiology, diagnostic findings, management and complications of allergic acute coronary syndrome. Int J Cardiol. (2017) 232:1–4. doi: 10.1016/j.ijcard.2017.01.124
3. Kounis NG, Zavras GM. Histamine-induced coronary artery spasm: the concept of allergic angina. Br J Clin Pract. (1991) 45:121–8.
4. Kounis NG. Kounis syndrome: an update on epidemiology, pathogenesis, diagnosis and therapeutic management. Clin Chem Lab Med. (2016) 54:1545–59. doi: 10.1515/cclm-2016-0010
5. Alblaihed L. Huis In 't Veld MA. Allergic acute coronary syndrome-Kounis syndrome. Emerg Med Clin North Am. (2022) 40:69–78. doi: 10.1016/j.emc.2021.08.010
6. Renda F, Landoni G, Trotta F, Piras D, Finco G, Felicetti P, et al. Kounis syndrome: an analysis of spontaneous reports from international pharmacovigilance database. Int J Cardiol. (2016) 203:217–20. doi: 10.1016/j.ijcard.2015.10.003
7. Khan DA, Banerji A, Bernstein JA, Bilgicer B, Blumenthal K, Castells M, et al. Cephalosporin allergy: current understanding and future challenges. J Allergy Clin Immunol Pract. (2019) 7:2105–14. doi: 10.1016/j.jaip.2019.06.001
8. Mazarakis A, Koutsojannis CM, Kounis NG, Alexopoulos D. Cefuroxime-induced coronary artery spasm manifesting as Kounis syndrome. Acta Cardiol. (2005) 60:341–5. doi: 10.2143/AC.60.3.2005015
9. Ilhan E, Güvenç TS, Poyraz E, Ayhan E, Soylu O. Kounis Syndrome secondary to cefuroxime axetil use in an asthmatic patient. Int J Cardiol. (2009) 137:e67–9. doi: 10.1016/j.ijcard.2009.04.026
10. Caglar FN, Caglar IM, Coskun U, Ugurlucan M, Okcun B. Kounis syndrome: myocardial infarction secondary to an allergic insult–a rare clinical entity. Acta Cardiol. (2011) 66:559–62. doi: 10.1080/ac.66.4.2126625
11. Sánchez VO, Roca LC, Moreno Adel P. Intraoperative “Kounis syndrome” that improved electrocardiography changes and hemodynamic situation after administering nitroglycerine. Braz J Anesthesiol. (2014) 64:281–5. doi: 10.1016/j.bjan.2013.06.016
12. Adachi H, Ihara M, Nojima Y, Kurimoto T, Nanto S. Kounis syndrome caused by anaphylaxis without skin manifestations after cefazolin administration. J Allergy Clin Immunol Pract. (2019) 7:317–9. doi: 10.1016/j.jaip.2018.05.030
13. Gao J, Gao Y, Ma J. Cefuroxime-associated Kounis syndrome with unique peculiarity in perioperative prophylaxis. J Infect Public Health. (2018) 11:889–92. doi: 10.1016/j.jiph.2018.02.009
14. Ricciardi L, Furci F, Casciaro M, Di Salvo E, Cristani M, Tigano V, et al. Drug induced Kounis syndrome: does oxidative stress play a role? Clin Mol Allergy. (2018) 16:21. doi: 10.1186/s12948-018-0099-2
15. Sato M, Arai T. A case of Kounis syndrome presenting as coronary artery spasm associated with cefazolin-induced anaphylaxis during general anesthesia. JA Clin Rep. (2019) 5:49. doi: 10.1186/s40981-019-0269-3
16. Ilgenli TF, Açikalin A, Türkmen S, Avci A, Akpinar O. ST elevation in inferior derivation, coronary ectasia, and slow coronary flow following ceftriaxone use. Am J Emerg Med. (2012) 30:1657. doi: 10.1016/j.ajem.2011.08.004
17. Kitulwatte I, Gangahawatte S, Perera U, Edirisinghe P. Death following ceftazidime-induced Kounis syndrome. Med Leg J. (2017) 85:215–8. doi: 10.1177/0025817217695904
18. Sequeira T, Gaspar Â, Mota I, Correia M, Chambel M, Morais-Almeida M. Kounis syndrome associated with selective anaphylaxis to cefazolin. J Investig Allergol Clin Immunol. (2018) 28:257–8. doi: 10.18176/jiaci.0248
19. Biteker M, Duran NE, Biteker FS, Gündüz S, Gökdeniz T, Kaya H, et al. Kounis syndrome secondary to cefuroxime-axetil use in an octogenarian. J Am Geriatr Soc. (2008) 56:1757–8. doi: 10.1111/j.1532-5415.2008.01912.x
20. Murat SN, Karasu BB, Ornek E, Akdemir R. Cefuroxime-axetil induced allergic angina: an insight into classification management of Kounis syndrome. Int J Cardiol. (2011) 151:e53–5. doi: 10.1016/j.ijcard.2010.04.086
21. Yurtdaş M, Aydin MK. A case of coronary spasm with resultant acute myocardial infarction: likely the result of an allergic reaction. Intern Med. (2012) 51:2161–4. doi: 10.2169/internalmedicine.51.7852
22. Saleh AA. Kounis syndrome: acute inferior myocardial infarction with atroventricular node block due to ceftriaxone: a first reported case. Ann Saudi Med. (2014) 34:250–3. doi: 10.5144/0256-4947.2014.250
23. Barbarroja-Escudero J, Sánchez-González MJ, Antolín-Amérigo D, Rodríguez-Rodríguez M, Salinas P, Fernández-Ortiz A, et al. Kounis syndrome induced by cefditoren pivoxil. Int J Cardiol. (2016) 207:112–4. doi: 10.1016/j.ijcard.2016.01.103
24. Venkateswararao S, Rajendiran G, Sundaram RS, Mounika G. Kounis syndrome secondary to intravenous cephalosporin administration. J Pharmacol Pharmacother. (2015) 6:225–7. doi: 10.4103/0976-500X.171877
25. Mitsis A, Christodoulou E, Georgiou P. Coronary spasm secondary to cefuroxime injection, complicated with cardiogenic shock - a manifestation of Kounis syndrome: case report and literature review. Eur Heart J Acute Cardiovasc Care. (2018) 7:624–30. doi: 10.1177/2048872617701885
26. Absmaier M, Biedermann T, Brockow K. Allergic myocardial infarction (Kounis syndrome) after cefuroxime with side-chain cross-reactivity. J Allergy Clin Immunol Pract. (2018) 6:1781–3. doi: 10.1016/j.jaip.2018.01.033
27. Çakmak A, Keskin G. Kounis syndrome: Is ceftriaxone or metronidazole responsible for acute myocardial infarction? A rare case. Anatol J Cardiol. (2021) 25:451–2. doi: 10.5152/AnatolJCardiol.2020.36422
28. Fujita Y, Chikamitsu M, Kimura M, Toriumi T, Endoh S, Sari A. An anaphylactic reaction possibly associated with an intraoperative coronary artery spasm during general anesthesia. J Clin Anesth. (2001) 13:221–6. doi: 10.1016/s0952-8180(01)00239-2
29. Austin SM, Barooah B, Kim CS. Reversible acute cardiac injury during cefoxitin-induced anaphylaxis in a patient with normal coronary arteries. Am J Med. (1984) 77:729–32. doi: 10.1016/0002-9343(84)90374-7
30. Ito K, Ito T, Fujita H., Hayashi K, Sugiura T, Seo Y, et al. Allergic acute coronary syndrome (Kounis syndrome) and sudden cardiac arrest due to cefoperazone-sulbactam in a patient with acute pancreatitis: A case report. Med: Case Reports Study Prot. (2020) 1:e0024. doi: 10.1097/MD9.0000000000000024
31. Forlani D, Scarano G, D'Alleva A, Di Marco M, Paloscia L, Gatta A, et al. Kounis syndrome as first manifestation of allergic sensitization. Case Rep Med. (2019) 2019:6317956. doi: 10.1155/2019/6317956
32. Mota I, Gaspar Â, Morais-Almeida M. Perioperative anaphylaxis including kounis syndrome due to selective cefazolin allergy. Int Arch Allergy Immunol. (2018) 177:269–73. doi: 10.1159/000490182
33. Nabel EG, Braunwald E. A tale of coronary artery disease and myocardial infarction. N Engl J Med. (2012) 366:54–63. doi: 10.1056/NEJMra1112570
34. Apostolos A, Drakopoulou M, Gregoriou S, Synetos A, Trantalis G, Tsivgoulis G, et al. Nickel hypersensitivity to atrial septal occluders: smoke without fire? Clin Rev Allergy Immunol. (2021). doi: 10.1007/s12016-021-08867-0
35. Romano A, Mayorga C, Torres MJ, Artesani MC, Suau R, Sánchez F, et al. Immediate allergic reactions to cephalosporins: cross-reactivity and selective responses. J Allergy Clin Immunol. (2000) 106:1177–83. doi: 10.1067/mai.2000.111147
36. Terlemez S, Eryilmaz U, Tokgöz Y, Uysal P, Coşan A, Bulut Y. Kounis syndrome caused by metronidazole–a case of 14 year-old boy. Int J Cardiol. (2015) 179:222–4. doi: 10.1016/j.ijcard.2014.11.049
37. Fagley RE, Woodbury A, Visuara A, Wall M. Rocuronium-induced coronary vasospasm–“Kounis syndrome”. Int J Cardiol. (2009) 137:e29–32. doi: 10.1016/j.ijcard.2008.05.052
38. Gangadharan V, Bhatheja S, Al Balbissi K. Kounis syndrome - an atopic monster for the heart. Cardiovasc Diagn Ther. (2013) 3:47–51. doi: 10.3978/j.issn.2223-3652.2013.02.04
39. Cevik C, Nugent K, Shome GP, Kounis NG. Treatment of Kounis syndrome. Int J Cardiol. (2010) 143:223–6. doi: 10.1016/j.ijcard.2010.02.040
40. Ioannidis TI, Mazarakis A, Notaras SP, Karpeta MZ, Tsintoni AC, Kounis GN, Rallis DG, Kounis NG. Hymenoptera sting-induced Kounis syndrome: effects of aspirin and beta-blocker administration. Int J Cardiol. (2007) 121:105–8. doi: 10.1016/j.ijcard.2006.08.039
41. Ibanez B, James S, Agewall S, Antunes MJ, Bucciarelli-Ducci C, Bueno H, et al. 2017 ESC Guidelines for the management of acute myocardial infarction in patients presenting with ST-segment elevation: the task force for the management of acute myocardial infarction in patients presenting with ST-segment elevation of the European society of cardiology (ESC). Eur Heart J. (2018) 39:119–77. doi: 10.1093/eurheartj/ehx393
42. Fassio F, Losappio L, Antolin-Amerigo D, Peveri S, Pala G, Preziosi D, et al. Kounis syndrome: a concise review with focus on management. Eur J Intern Med. (2016) 30:7–10. doi: 10.1016/j.ejim.2015.12.004
43. Kounis NG. Coronary hypersensitivity disorder: the Kounis syndrome. Clin Ther. (2013) 35:563–71. doi: 10.1016/j.clinthera.2013.02.022
Keywords: Kounis syndrome, coronary artery spasm, cephalosporin, chest pain, allergic angina
Citation: Fang W, Song L, Deng Z, Sun W, Li Z and Wang C (2022) Analysis of Clinical Features of Kounis Syndrome Induced by Cephalosporin. Front. Cardiovasc. Med. 9:885438. doi: 10.3389/fcvm.2022.885438
Received: 28 February 2022; Accepted: 31 March 2022;
Published: 26 April 2022.
Edited by:
Grigorios Tsigkas, University of Patras, GreeceReviewed by:
Anastasios Apostolos, National and Kapodistrian University of Athens, GreeceEleni-Evangelia Koufou, General University Hospital of Patras, Greece
Copyright © 2022 Fang, Song, Deng, Sun, Li and Wang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chunjiang Wang, d29uZ2NqQGNzdS5lZHUuY24=
 Weijin Fang
Weijin Fang Liying Song
Liying Song Zhenzhen Deng
Zhenzhen Deng Wei Sun
Wei Sun Zuojun Li
Zuojun Li Chunjiang Wang
Chunjiang Wang