
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Cardiovasc. Med. , 13 January 2022
Sec. Lipids in Cardiovascular Disease
Volume 8 - 2021 | https://doi.org/10.3389/fcvm.2021.778961
This article is part of the Research Topic Case Reports in Lipids in Cardiovascular Disease: 2022 View all 6 articles
 Alena S. Limonova1*
Alena S. Limonova1* Alexandra I. Ershova1
Alexandra I. Ershova1 Alexey N. Meshkov2
Alexey N. Meshkov2 Anna V. Kiseleva2
Anna V. Kiseleva2 Mikhail G. Divashuk2,3
Mikhail G. Divashuk2,3 Marina V. Kurkina4
Marina V. Kurkina4 Oxana M. Drapkina5
Oxana M. Drapkina5We reported a case of sitosterolemia, which is a rare genetic disease, characterized by increased plant sterol absorption and great heterogeneity of clinical manifestations. Our patient was initially referred to the lipid clinic due to high cholesterol levels and premature cardiovascular disease. Diagnosis of familial hypercholesterolemia was established in accordance with the Dutch Lipid Clinic Network criteria. Next-generation sequencing was later performed, which revealed a nonsense mutation in the ABCG8 gene, which led to the diagnosis of sitosterolemia. The aim of our report is to demonstrate, how genetic testing helped to make the correct diagnosis and to explain many of the patient's health problems, which etiology remained unclear for many years.
Despite almost the same consumption of cholesterol and plant sterols with the western diet, in comparison with cholesterol, only trace amounts (~5%) of plant sterols are absorbed, with further quick excretion into the bile from hepatocytes (1). Disruption of these mechanisms by defective ABCG5/ABCG8 leads to the accumulation of plant sterols in body tissues. This genetic disorder, sitosterolemia (MIM 210250), is a rare autosomal recessive disease, first described in 1974 (2). It is caused by mutations in either of ATP binding cassette (ABC) transporter genes (ABCG5 and ABCG8). Although recent data suggest that not only homozygous but also heterozygous carriers of ABCG5 loss-of-function variants exhibit increased plant sterols and LDL-C levels and have increased risk of coronary artery disease (CAD), compared with non-carriers (3). These proteins are expressed only in hepatocytes, gallbladder epithelium, and enterocytes and function as obligate heterodimers responsible for excretion of sterols, with plant sterols preferred over cholesterol (4, 5). Thus, they perform a basic physiological process for the prevention of the accumulation of dietary plant sterols. Deficiency of ABCG5 or ABCG8 in sitosterolemia impairs excretion of plant sterol into the intestinal lumen from enterocytes and into bile in the liver, thereby resulting in a severe accumulation of plant sterols in plasma and tissues.
In this report, we presented a case of a patient with sitosterolemia and discuss mechanisms of different clinical manifestations of the disease, present in our patient. We aimed to demonstrate how next-generation sequencing (NGS) helped to make the correct diagnosis, which explained all the symptoms, present in this patient and which etiology remained unclear for many years.
A 41-year-old woman was referred to the lipid clinic due to her elevated cholesterol level. On examination, she had tendinous xanthomas of the Achilles tendons. Her body weight was 59 kg, and her height was 1.67 m, with a body mass index of 21.2 kg/m2. She had exertional dyspnea since adolescence. Hypercholesterolemia was revealed at the age of 34 years old (total cholesterol – 10.12 mmol/L, LDL-C – 8.8 mmol/L, HDL-C – not available). Two years later, due to the onset of angina and ischemic changes on ECG during physical activity, coronary arteriography was performed. It revealed 80% stenosis of the left main coronary artery, 80% stenosis of the anterior descending artery, occlusions of the proximal right coronary artery, the diagonal branch, and the obtuse marginal branch. At that time the duplex sonography revealed no atherosclerotic manifestations of other vascular territories (carotids and lower extremity arteries). Coronary artery bypass grafting (CABG) was performed. During the first 2 days after CABG, the patient had hemopericardium and hemothorax. She was diagnosed with intracerebral hemorrhage 10 days later.
On rosuvastatin 20 mg and ezetimibe 10 mg, which were initiated 1 year before the current presentation, her total cholesterol was 6.7 mmol/L, LDL-C - 3.95 mmol/L, HDL-C – 2.3 mmol/L. During follow-up with a cardiologist paroxysmal atrial fibrillation was diagnosed. Three years after CABG, the stress testing was performed with positive results. One year later, the CT-angiography revealed subtotal stenosis of the left main coronary artery, with normally functioning bypass grafts to the left anterior descending artery and right coronary artery. Duplex sonography demonstrated stenosis of carotid bifurcations and proximal internal carotid arteries up to 30% at the right and 35% at the left, and 35% stenosis of the left vertebral artery.
Her medical history was also significant for macrothrombocytopenia (with current PLT 113 x 10*9/L and MPV 14.7 fL), arthralgia of knee joints, and past recurrent serositis (pericarditis and pleurisy). The latter was treated with fluoroquinolones and prednisolone. Cardiolipin antibodies and anti-dsDNA test results were negative. No definite diagnosis was established. Due to the polycystic ovary syndrome, the resection of ovaries was fulfilled at the age of 23. Her gynecologic history was also noteworthy for frozen pregnancy of unknown etiology, weak labor activity which led to caesarian section (she has one child, a boy with no clinical manifestations), and early menopause at 39 years old.
Her family history was unremarkable with no information about premature CAD.
According to the Dutch Lipid Clinic Network criteria (16 points) heterozygous familial hypercholesterolemia (HeFH) was diagnosed. Maximum statin dose, ezetimibe, and proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors were prescribed and cascade screening was recommended.
Later on, the NGS was performed using the Ion S5 (Thermo Fisher Scientific, USA). Ampliseq libraries were prepared on the Ion Chef System (Thermo Fisher Scientific, USA) using the custom panel, created with the Ion AmpliSeq Designer (Thermo Fisher Scientific, USA), which consists of 25 genes (ABCA1, ABCG5, ABCG8, ANGPTL3, APOA1, APOA5, APOB, APOC2, APOC3, APOE, CETP, GPD1, GPIHBP1, LCAT, LDLR, LDLRAP1, LIPC, LIPI, LMF1, LPL, MTTP, PCSK9, SAR1B, STAP1, USF1) and 280 variants responsible for the lipid metabolism.
Variants with total frequency <0.01 identified in the patient are listed in Table 1. Missense variants in heterozygous state in the cholesteryl ester transfer protein (CETP) (NM_000078.2: c.708G>C, p.Gly236Arg) and apolipoprotein B (ApoB) (NM_000384.2: c12382G>A, p.Val4128Met) genes were revealed, wherein both were classified as variants of uncertain significance according to the American College of Medical Genetics and Genomics guidelines (ACMG2015). Pathogenic or likely pathogenic variants in LDLR, PCSK9, APOB, and LDLRAP1 were not detected. The patient was found to be homozygous for a nonsense mutation in the ABCG8 gene (NM_022437.2: c.1083G > A, p.Trp361Ter), leading to premature termination of signal codon 361 (6). According to ACMG2015 this variant is classified as pathogenic (criteria PVS1, PP5, PM2, PP3) (7). These variants were confirmed by Sanger sequencing on 3,500 DNA Analyzer (Thermo Fisher Scientific, USA). A multiplex sitosterol assay for diagnosis of sitosterolemia was performed by gas chromatography-mass spectrometry (GC-MS) TQ-8050 (Shimadzu, Japan) with autosampler AOC-20i, HP-5MS. Sample preparation and analysis conditions were performed with GC-MS according to Joon Hee Lee et al. (8). Her plant sterol levels in blood were markedly elevated: sitosterol 54.4 μmol/L, ref. 0.4 – 3.4 μmol/L; campesterol 18.8 μmol/L, ref.0.1 – 3.1 μmol/L (Figure 1), confirming the diagnosis. The limitation of the current work is no genetic and biochemical data being available from the relatives (with them residing too far away) and lack of follow-up data from the patients, as she refused further medical evaluation because of the COVID-19 pandemic.
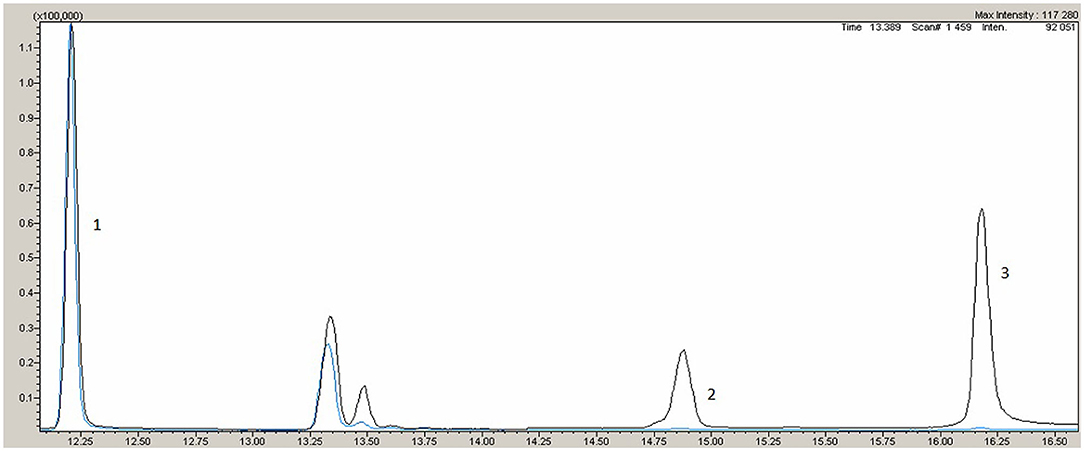
Figure 1. Gas chromatography-mass spectrometry. Black line - patient, blue line – normal reference. 1- internal standart, 2 - campresterol, 3 – sitosterol.
According to current clinical guidelines, for very high cardiovascular risk patients with atherosclerotic CAD target LDL-C level is <1.4 mmol/L, thus the patient was prescribed with maximum statin dose, ezetimibe, PCSK9 inhibitors. Plant sterol-free diet was recommended with restriction of the consumption of shellfish, margarine, vegetable oils, nuts, seeds, avocados, chocolate, whole grains, olives. Genetic testing of the patient's son was recommended. As it was revealed during the follow-up visit, the patient took statin and ezetimibe, with poor adherence to the sterol-free diet.
Sitosterolemia is a rare genetic disease. To date, approximately 110 homozygous individuals with sitosterolemia have been reported (9). For rare diseases, an average pathway to correct diagnosis takes 7.3 years mean, with 7 physicians and 2–3 misdiagnoses during the patient's diagnostic odyssey (https://globalgenes.org/rare-disease-facts/). This odyssey is, undoubtedly, a real burden for patients suffering from misdiagnoses and inappropriate treatment and for the healthcare system. For example, as described by Wang et al. in their series of cases, the mean ages of symptoms onset were 15.2 years, and the mean years of delay between symptom onset and diagnosis were 28.8 (range 15–49 years) (10). For our patient, it lasted for 7 years.
However, sitosterolemia may be not so rare, as previously believed. Evidence from recent research demonstrates, that misdiagnosis of sitosterolemia with familial hypercholesterolemia may be prevalent (11, 12), which is also the case of our patient. The great variety of possible manifestations means that physicians of different specialties, like cardiologists, hematologists, and rheumatologists can deal with this disease. But even in the case of a competent specialist who will have a suspicion of this rare disorder, the diagnosis needs confirmation. We experienced a real challenge in search of GC-MS laboratory, as it is usually used in research but not clinical ones. In the case of our patient, NGS helped to make the correct diagnosis.
Sitosterolemia is characterized by great clinical heterogeneity. This could be explained by a large variety of xenosterols present in human diets which may differ in their biological activity, but only a few sterols are commonly detected in the studies [details discussed in (13)]. Furthermore, we would like to discuss clinical manifestations present in our patients in order to demonstrate how the correct diagnosis, revealed by NGS, could at once explain many of the patient's different conditions, which could not be correctly diagnosed and treated previously.
Patients with sitosterolemia may present with normal cholesterol levels, as in the first described cases of the disease (2). However, some sitosterolemic patients have elevated cholesterol levels, as in this case and some previously described ones (14, 15). One of the possible explanations is that deletion in ABCG8 reduces biliary cholesterol secretion (16), and excretion of sterols into the bile seems pivotal for their accumulation, as liver transplantation in the patient with sitosterolemia led to complete normalization of the plant sterols levels (17). The turnover rates of plasma cholesterol and plant sterols in patients with sitosterolemia were similar, meaning increased retention and reduced elimination of both plant sterols and cholesterol, as was demonstrated by Salen et al. (18) with the usage of in vivo radiolabeled isotopic techniques. Some bias may come from the inability of conventional clinical methods to discriminate between cholesterol and plant sterols as these methods depend on the C-5 double bond or the presence of the 3β hydroxyl group, both of which are present in cholesterol and plant sterols (19). High cholesterol levels are more characteristic for children with their further tendency to decrease. For instance, Mymin et al. (20) revealed an inverse association between age and plasma cholesterol. However, for understanding the precise mechanisms of hypercholesterolemia further research is needed, as till now, there is more evidence for normal/reduced cholesterol levels. Thus, in hepatic tissues of affected individuals increased LDL receptors activity alongside reduced activity of the rate-limiting enzyme for cholesterol biosynthesis (3 -hydroxy-3- methyl-glutaryl-CoA reductase) was demonstrated (21). Yu et al. (22) showed that ABCG5/ABCG8-deficient mice had higher plant sterols levels in their plasma and liver, and had reduced cholesterol synthesis relative to wild-type mice. Results of the studies addressing the molecular mechanisms, by which plant sterols influence cholesterol homeostasis, proposed that plant sterols may not only compete with dietary and biliary cholesterol for intestinal absorption in mixed micelles but also interfere with different steps of cholesterol metabolism, i.e., cholesterol esterification and lipoprotein assembly, cholesterol internalization, cholesterol synthesis and removal of apoB100-containing lipoproteins [details are reviewed by Calpe-Berdiel et al. (23)].
The missense variant in the CETP gene revealed in our patient (NM_000078.2: c.708G>C, p.Gly236Arg) is not described in gnomAD, where another substitution (c.708G>A, p.Gly236Ser) was previously described with total frequency <0.01. SNPs, reported at http://www.hgmd.cf.ac.uk/ as associated with decreased CETP activity/increased HDL-C, are often located at beta strands within the protein sequence. Two hundred and thirty six amino acid position is also located at the beta-strand region. Thus, the patient's phenotype (HDL-C - 2.3 mmol/L) could be explained by this missense variant.
Clinical data regarding atherosclerosis development vary from early myocardial infarction and sudden cardiac death at the age of 5 (24) to no clinical manifestations (15), even among family members of an affected individual (20). Premature CAD may develop even in the case of normal cholesterol levels (25, 26).
The same clinical heterogeneity is true about xanthomas (20, 27). Our patient manifested with xanthomas of the Achilles tendons and premature premature CAD (marked atherosclerotic damage of coronary arteries diagnosed at the age of 36).
From enterocytes plant sterols, as well as cholesterol esters, proceed to chylomicrons which are further metabolized in the liver. Lipoprotein-derived cholesterol and plant sterols can further penetrate the artery wall and lead to inflammation and atherosclerotic plaque progression (28). In case of their high level in the blood, plant sterols can be found in atherosclerotic lesions and xanthomas (2, 29). Enhanced accumulation of sterols (including plant sterols) in homozygous individuals can accelerate foam cells formation (30). Plant sterols seem to have more cytotoxic effects, than cholesterol (31). However, different plant sterols demonstrated opposite results on inflammatory cytokine secretion in cultured macrophage foam cells (32). Bao et al. (33) proposed one of the possible explanations of how plant sterols can promote atherosclerosis progression. Authors demonstrated that macrophages incubated with sitosterol-containing lipoproteins undergo death in an accelerated manner, compared with free-cholesterol-induced macrophages death. As macrophage death is a key event for plaque disruption (34, 35), such data provide at least one mechanism for accelerated atherothrombosis in patients with very high levels of plant sterols (33). Plant sterols are more susceptible to oxidative processes than cholesterol, though further research is needed to demonstrate the health effects of their oxidized forms (36).
Similar mechanisms could be involved in xanthomas formation, as early stages of their formation resemble early stages of atherogenesis (37). Moreover, a lower plasma level of plant sterols is needed for xanthomas formation in comparison with cholesterol level (38).
In a recent genetic study, based on data from biobanks of Iceland, Denmark, and UK Biobank, authors evaluated the association of variants in ABCG5/ABCG8 with non-HDL cholesterol, a plant sterols, and the risk of CAD. They demonstrated that the degree of CAD risk conferred by ABCG5/ABCG8 variants is greater than predicted by their effect on non-HDL cholesterol levels only. The authors concluded that plant sterols may contribute to atherogenesis directly, irrespective of non-HDL cholesterol (39). Koeijvoets et al. (40) studied the association between two polymorphisms in the ABCG8 gene and CAD in 2012 patients with heterozygous familial hypercholesterolemia. They demonstrated that individuals carrying the risk genotype for both ABCG8 variants had an increased risk of cardiovascular disease (RR 1.57, 95% CI 1.13–2.18; p = 0.01) and coronary heart disease (RR 1.72, 95% CI 1.23–2.41; p = 0.002). Wu et al. (41) investigated the association between four ABCG5 and ABCG8 SNPs and CAD. Binary logistic regression analysis demonstrated that the homozygous C allele at Thr400LysC>A (rs4148217) resulted in a more than 2-fold greater risk of developing CAD, compared with those who carried the A allele in a dominant model. The multivariate analysis supported that such an effect was independent of several other risk factors, including age, gender, history of diabetes mellitus, and HDL-C level.
Our patient has a compromised gynecological anamnesis. As complete medical records of that period were unavailable, we can't definitely conclude, whether or not her gynecological anamnesis could be related to her main disease or some other problem. Anyway, we suppose that this aspect of her health problems is worth mentioning and discussing in light of high plant sterol levels.
Pieces of evidence from some animal studies and case reports of this disease suggest that high plant sterol levels may interfere with endocrine function and fertility. For instance, a family including three siblings, homozygous for sitosterolaemia, with adrenal insufficiency and ovarian failure was described previously (42). Solca et al. (43) demonstrated, that ABCG5/ABCG8 knock-out mice fed with a diet high in plant sterols were infertile. Exclusion of plant sterols from diet or blockage of their absorption (with ezetimibe) could restore fertility. No structural abnormalities of the ovaries were found. Oxidation products of plant sterols have been reported to modulate the action of 17β-estradiol in vitro and two human cell lines (44). In addition, plant sterols lowered plasma estrogen levels in mice (45). In double knock-out ABCG5/ABCG8 mice adrenal glands had a 91% reduction in cholesterol content, treatment with ezetimibe returned adrenal cholesterol levels to near-normal levels, however, the response of the glands to ACTH was unimpaired (46).
Our patient has both macrothrombocytopenia and bleeding abnormalities. The association of stomatocytic hemolysis and macrothrombocytopenia with sitosterolemia was revealed by Rees et al. In five pedigrees with these hematologic abnormalities they demonstrated the presence of both elevated levels of plant sterols in blood and mutations in ABCG5 and ABCG8, previously linked to sitosterolemia (47). Sitosterolemia may manifest solely with hematologic problems (48). In vitro experiments showed changes in shape and osmotic fragility of red blood cells when incubated in the presence of sitosterol (48).
Thrombocytopenia caused by sitosterolemia may be confused with other causes, including autoimmune disorders, which leads to unnecessary diagnostic steps and inappropriate steroid treatment (49). And in our patient autoimmune disorders were also ruled out previously. In one recent observational study among seven patients with unexplained long-standing thrombocytopenia, whole-exome sequencing helped to reveal sitosterolemia in one of them. It remains unclear why only some individuals develop hematologic manifestations of the disease. Further research is needed to find out whether other genetic/environmental factors contribute. But among the variants that have been reported to cause sitosterolemia, only 24 have been associated with macrothrombocytopenia with the variant found in our patient among the most prevalent ones (50).
Since ABCG5 and ABCG8 are not present on the surfaces of red blood cells and platelets, insertion of plant sterols into their membranes is supposed to be the most likely explanation. Kruit et al. (51) in experiments with ABCG5-deficient mice demonstrated, that they still had macrothrombocytopenia after transplantation of bone marrow from wild-type mice. At the same time, the bone marrow transplantation from ABCG5-deficient mice to wild-type mice did not result in platelet changes. Research by Kanaji et al. (52) improved our understanding of mechanisms, contributing to bleeding abnormalities and macrothrombocytopenia. It was demonstrated, that accumulation of plant sterols in platelet plasma membranes leads to their hyperactivity and that internalization of the αIIbβ3 complex and filamin A degradation cause macrothrombocytopenia and bleeding phenotype (52).
Arthralgia is another possible clinical manifestation of sitosterolemia (10, 53), with subsiding severity of the symptom with treatment (53). Arthralgia of knee joints is also present in our patient. To our knowledge, data regarding the mechanism of this clinical symptom seem to be missing in the literature. Togo et al. (53) proposed, that the accumulation of plant sterols in immune cells in sitosterolemia patients modulates the immune system.
Sitosterolemia is a rare genetic disease with great heterogeneity of clinical manifestations. Many of them were present in our patient but their real etiology could not be diagnosed correctly until NGS was performed. This emphasizes the promising role of NGS in real practice, as a powerful tool of timely diagnosis instead of the long-lasting diagnostic odyssey of such patients.
The datasets presented in this study can be found in online repository: https://www.ncbi.nlm.nih.gov/sra/?term=SRR16916455.
Ethical review and approval was not required for the study on human participants in accordance with the local legislation and institutional requirements. The patients/participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
AL was treating the patient, performing her follow-ups, and writing this paper. AE was treating the patient, supervising all parts of this paper's preparation, and edited this paper. AM supervised the genetic testing of the patient and provided valuable comments on this case. AK performed the NGS and edited this paper. MD performed the Sanger sequencing. OD is the chief of the center who provided valuable comments on this case. MK performed the development of a method for the determination of plant sterols and performed determination of their concentration of phytosterols. All authors contributed to the article and approved the submitted version.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Williams K, Segard A, Graf GA. Sitosterolemia: twenty years of discovery of the function of abcg5 abcg8. Int J Mol Sci. (2021) 22:1–14. doi: 10.3390/ijms22052641
2. Bhattacharyya AK, Connor WE. β Sitosterolemia and xanthomatosis. a newly described lipid storage disease in two sisters. J Clin Invest. (1974) 53:1033–43. doi: 10.1172/JCI107640
3. Nomura A, Emdin CA, Won HH, Peloso GM, Natarajan P, Ardissino Di, et al. Heterozygous ABCG5 gene deficiency and risk of coronary artery disease. Circ Genomic Precis Med. (2020) 13:417–23. doi: 10.1161/CIRCGEN.119.002871
4. Patel SB, Graf GA, Temel RE. ABCG5 and ABCG8: more than a defense against xenosterols. J Lipid Res. (2018) 59:1103–13. doi: 10.1194/jlr.R084244
5. Graf G, Yu L, Li W, Gerard R, Tuma P, Cohen J, et al. ABCG5 and ABCG8 are obligate heterodimers for protein trafficking and biliary cholesterol excretion. J Biol Chem. (2003) 278:48275–82. doi: 10.1074/jbc.M310223200
6. Berge KE, Tian H, Graf GA, Yu L, Grishin N V, Schultz J, et al. Accumulation of dietary cholesterol in sitosterolemia caused by mutations in adjacent ABC transporters. Science. (2000) 290:1771–5. doi: 10.1126/science.290.5497.1771
7. Richards S, Aziz N, Bale S, Bick D, Das S, Gastier-Foster J, et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American college of medical genetics and genomics and the association for molecular pathology. Genet Med. (2015) 17:405–24. doi: 10.1038/gim.2015.30
8. Lee JH, Lee K, Jun S-H, Song SH, Shin CH, Song J. A multiplex phytosterol assay utilizing gas chromatography-mass spectrometry for diagnosis of inherited lipid storage disorders. Ann Lab Med. (2019) 39:411–3. doi: 10.3343/alm.2019.39.4.411
9. Myrie SB, Steiner RD, Mymin D. Sitosterolemia. GeneReviews® (2020). Available online at: https://www.ncbi.nlm.nih.gov/books/NBK131810/ (accessed July 21, 2021).
10. Wang Z, Cao L, Su Y, Wang G, Wang R, Yu Z, et al. Specific macrothrombocytopenia/hemolytic anemia associated with sitosterolemia. Am J Hematol. (2014) 89:320–4. doi: 10.1002/ajh.23619
11. Lee J, Song D, Jun S, Song S, Shin C, Ki C, et al. High prevalence of increased sitosterol levels in hypercholesterolemic children suggest underestimation of sitosterolemia incidence. PLoS ONE. (2020) 15:e0238079. doi: 10.1371/journal.pone.0238079
12. Tada H, Okada H, Nomura A, Yashiro S, Nohara A, Ishigaki Y, et al. Rare and deleterious mutations in ABCG5/ABCG8 genes contribute to mimicking and worsening of familial hypercholesterolemia phenotype. Circ J. (2019) 83:1917–24. doi: 10.1253/circj.CJ-19-0317
13. Weingärtner O, Teupser D, Patel SB. The atherogenicity of plant sterols: the evidence from genetics to clinical trials. J AOAC Int. (2015) 98:742–9. doi: 10.5740/jaoacint.SGEWeingartner
14. Kawamura R, Saiki H, Tada H, Hata A. Acute myocardial infarction in a 25-year-old woman with sitosterolemia. J Clin Lipidol. (2018) 12:246–9. doi: 10.1016/j.jacl.2017.10.017
15. Hansel B, Carrié A, Brun-Druc N, Leclert G, Chantepie S, Coiffard A, et al. E B. Premature atherosclerosis is not systematic in phytosterolemic patients: severe hypercholesterolemia as a confounding factor in five subjects. Atherosclerosis. (2014) 234:162–8. doi: 10.1016/j.atherosclerosis.2014.02.030
16. Wang H, Patel S, Carey M, Wang D. Quantifying anomalous intestinal sterol uptake, lymphatic transport, and biliary secretion in Abcg8(-/-) mice. Hepatology. (2007) 45:998–1006. doi: 10.1002/hep.21579
17. Miettinen T, Klett E, Gylling H, Isoniemi H, Patel S. Liver transplantation in a patient with sitosterolemia and cirrhosis. Gastroenterology. (2006) 130:542–7. doi: 10.1053/j.gastro.2005.10.022
18. Salen, G, Shore, V, Tint, G, Forte, T, Shefer, S, Horak, I, . Increased Sitosterol Absorption, Decreased Removal, And Expanded Body Pools Compensate For Reduced Cholesterol Synthesis In Sitosterolemia With Xanthomatosis - PubMed. Available online at: https://pubmed.ncbi.nlm.nih.gov/2600539/ (accessed August 4, 2021).
19. Moghadasian MH, Frohlich JJ, Scudamore CH. Specificity of the commonly used enzymatic assay for plasma cholesterol determination. J Clin Pathol. (2002) 55:859. doi: 10.1136/jcp.55.11.859
20. Mymin D, Salen G, Triggs-Raine B, Waggoner D, Dembinski T, Hatch G. The natural history of phytosterolemia: observations on its homeostasis. Atherosclerosis. (2018) 269:122–8. doi: 10.1016/j.atherosclerosis.2017.12.024
21. Patel SB, Honda A, Salen G. Sitosterolemia: exclusion of genes involved in reduced cholesterol biosynthesis. J Lipid Res. (1998) 39:1055–61. doi: 10.1016/S0022-2275(20)33874-8
22. Yu L, von Bergmann K, Lutjohann D, Hobbs H, Cohen J. Selective sterol accumulation in ABCG5/ABCG8-deficient mice. J Lipid Res. (2004) 45:301–7. doi: 10.1194/jlr.M300377-JLR200
23. Calpe-Berdiel L, Escolà-Gil J, Blanco-Vaca F. New insights into the molecular actions of plant sterols and stanols in cholesterol metabolism. Atherosclerosis. (2009) 203:18–31. doi: 10.1016/j.atherosclerosis.2008.06.026
24. Mymin D, Wang J, Frohlich J, Hegele R. Image in cardiovascular medicine. Aortic xanthomatosis with coronary ostial occlusion in a child homozygous for a nonsense mutation in ABCG8. Circulation. (2003) 107:791. doi: 10.1161/01.CIR.0000050545.21826.AD
25. Kolovou G, Voudris V, Dogari E, Palatianos G, Cokkinos D. Coronary bypass grafts in a young girl with sitosterolemia. Eur Heart J. (1996) 17:965–6. doi: 10.1093/oxfordjournals.eurheartj.a014983
26. Miettinen T. Phytosterolaemia, xanthomatosis and premature atherosclerotic arterial disease: a case with high plant sterol absorption, impaired sterol elimination and low cholesterol synthesis. Eur J Clin Invest. (1980) 10:27–35. doi: 10.1111/j.1365-2362.1980.tb00006.x
27. Kiss S, Lee JY, Pitt J, MacGregor D, Wallace J, Marty M, et al. Dig deeper when it does not make sense: Juvenile xanthomas due to sitosterolemia. JIMD Rep. (2020) 56:34–9. doi: 10.1002/jmd2.12161
28. Salen G, Horak I, Rothkopf M, Cohen JL, Speck J, Tint GS, et al. Lethal atherosclerosis associated with abnormal plasma and tissue sterol composition in sitosterolemia with xanthomatosis. J Lipid Res. (1985) 26:1126–33. doi: 10.1016/S0022-2275(20)34286-3
29. Webb TR, Erdmann J, Stirrups KE, Stitziel NO, Masca NGD, Jansen H, et al. Systematic evaluation of pleiotropy identifies 6 further loci associated with coronary artery disease. J Am Coll Cardiol. (2017) 69:823–36. doi: 10.1016/j.jacc.2016.11.056
30. Nguyen L, Salen G, Shefer S, Tint G, Ruiz F. Macrophage 3-hydroxy-3-methylglutaryl coenzyme a reductase activity in sitosterolemia: effects of increased cellular cholesterol and sitosterol concentrations. Metabolism. (2001) 50:1224–9. doi: 10.1053/meta.2001.26707
31. Rubis B, Paszel A, Kaczmarek M, Rudzinska M, Jelen H, Rybczynska M. Beneficial or harmful influence of phytosterols on human cells? Br J Nutr. (2008) 100:1183–91. doi: 10.1017/S0007114508981423
32. Sabeva N, McPhaul C, Li X, Cory T, Feola D, Graf G. Phytosterols differentially influence ABC transporter expression, cholesterol efflux and inflammatory cytokine secretion in macrophage foam cells. J Nutr Biochem. (2011) 22:777–83. doi: 10.1016/j.jnutbio.2010.07.002
33. Bao L, Li Y, Deng S, Landry D, Tabas I. Sitosterol-containing lipoproteins trigger free sterol-induced caspase-independent death in ACAT-competent macrophages. J Biol Chem. (2006) 281:33635–49. doi: 10.1074/jbc.M606339200
34. Ball R, Stowers E, Burton J, Cary N, Skepper J, Mitchinson M. Evidence that the death of macrophage foam cells contributes to the lipid core of atheroma. Atherosclerosis. (1995) 114:45–54. doi: 10.1016/0021-9150(94)05463-S
35. Falk E, Shah P, Fuster V. Coronary plaque disruption. Circulation. (1995) 92:657–71. doi: 10.1161/01.CIR.92.3.657
36. Plat J, Brzezinka H, Lütjohann D, Mensink RP, Von Bergmann K. Oxidized plant sterols in human serum and lipid infusions as measured by combined gas-liquid chromatography-mass spectrometry. J Lipid Res. (2001) 42:2030–8. doi: 10.1016/S0022-2275(20)31532-7
37. Zak A, Zeman M, Slaby A, Vecka M. Xanthomas: clinical and pathophysiological relations. Biomed Pap Med Fac Univ Palacky Olomouc Czech Repub. (2014) 158:181–8. doi: 10.5507/bp.2014.016
38. Kidamb S, Patel S. Sitosterolaemia: pathophysiology, clinical presentation and laboratory diagnosis. J Clin Pathol. (2008) 61:588–94. doi: 10.1136/jcp.2007.049775
39. Helgadottir A, Thorleifsson G, Alexandersson KF, Tragante V, Thorsteinsdottir M, Eiriksson FF, et al. Genetic variability in the absorption of dietary sterols affects the risk of coronary artery disease. Eur Heart J. (2020) 41:2618. doi: 10.2139/ssrn.3335863
40. Koeijvoets KCMC, van der Net JB, Dallinga-Thie GM, Steyerberg EW, Mensink RP, Kastelein JJP, et al. ABCG8 gene polymorphisms, plasma cholesterol concentrations, and risk of cardiovascular disease in familial hypercholesterolemia. Atherosclerosis. (2009) 204:453–8. doi: 10.1016/j.atherosclerosis.2008.09.018
41. Wu GE, Li G-B, Yao M, Zhang DQ, Dai B, Ju CJ, et al. ABCG5/8 variants are associated with susceptibility to coronary heart disease. Mol Med Rep. (2014) 9:2512–20. doi: 10.3892/mmr.2014.2098
42. Mushtaq T, Wales J, Wright N. Adrenal insufficiency in phytosterolaemia. Eur J Endocrinol. (2007) 157:S61–5. doi: 10.1530/EJE-07-0222
43. Solca C, Tint G, Patel S. Dietary xenosterols lead to infertility and loss of abdominal adipose tissue in sterolin-deficient mice. J Lipid Res. (2013) 54:397–409. doi: 10.1194/jlr.M031476
44. Newill H, Loske R, Wagner J, Johannes C, Lorenz R, Lehmann L. Oxidation products of stigmasterol interfere with the action of the female sex hormone 17beta-estradiol in cultured human breast and endometrium cell lines. Mol Nutr Food Res. (2007) 51:888–98. doi: 10.1002/mnfr.200700025
45. Ju Y, Clausen L, Allred K, Almada A, Helferich W. beta-Sitosterol, beta-sitosterol glucoside, and a mixture of beta-sitosterol and beta-sitosterol glucoside modulate the growth of estrogen-responsive breast cancer cells in vitro and in ovariectomized athymic mice. J Nutr. (2004) 134:1145–51. doi: 10.1093/jn/134.5.1145
46. Yang C, Yu L, Li W, Xu F, Cohen J, Hobbs H. Disruption of cholesterol homeostasis by plant sterols. J Clin Invest. (2004) 114:813–22. doi: 10.1172/JCI22186
47. Rees D, Iolascon A, Carella M, O'marcaigh A, Kendra J, Jowitt S, et al. Stomatocytic haemolysis and macrothrombocytopenia (mediterranean stomatocytosis/macrothrombocytopenia) is the haematological presentation of phytosterolaemia. Br J Haematol. (2005) 130:297–309. doi: 10.1111/j.1365-2141.2005.05599.x
48. Wang G, Cao L, Wang Z, Jiang M, Sun X, Bai X, et al. Macrothrombocytopenia/stomatocytosis specially associated with phytosterolemia. Clin Appl Thromb Hemost. (2012) 18:582–7. doi: 10.1177/1076029611435090
49. Neff A. Sitosterolemia's stomatocytosis and macrothrombocytopenia. Blood. (2012) 120:4283. doi: 10.1182/blood-2012-06-429449
50. Bastida JM, Benito R, González-Porras JR, Rivera J. ABCG5 and ABCG8 gene variations associated with sitosterolemia and platelet dysfunction. Platelets. (2021) 32:573–7. doi: 10.1080/09537104.2020.1779926
51. Kruit J, Drayer A, Bloks V, Blom N, Olthof S, Sauer P, et al. Plant sterols cause macrothrombocytopenia in a mouse model of sitosterolemia. J Biol Chem. (2008) 283:6281–7. doi: 10.1074/jbc.M706689200
52. Kanaji T, Kanaji S, Montgomery R, Patel S, Newman P. Platelet hyperreactivity explains the bleeding abnormality and macrothrombocytopenia in a murine model of sitosterolemia. Blood. (2013) 122:2732–42. doi: 10.1182/blood-2013-06-510461
Keywords: genetic testing, next generation sequencing, ABCG8, sitosterolemia, macrothrombocytopenia, premature coronary artery disease
Citation: Limonova AS, Ershova AI, Meshkov AN, Kiseleva AV, Divashuk MG, Kurkina MV and Drapkina OM (2022) Case Report: Next Generation Sequencing in Clinical Practice–A Real Tool for Ending the Protracted Diagnostic Odyssey. Front. Cardiovasc. Med. 8:778961. doi: 10.3389/fcvm.2021.778961
Received: 17 September 2021; Accepted: 30 November 2021;
Published: 13 January 2022.
Edited by:
Michael J. Thomas, Medical College of Wisconsin, United StatesReviewed by:
Josep Julve, Institut de Recerca de l'Hospital de la Santa Creu i Sant Pau, SpainCopyright © 2022 Limonova, Ershova, Meshkov, Kiseleva, Divashuk, Kurkina and Drapkina. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Alena S. Limonova, bGltb25vdmEtYWxlbmFAeWFuZGV4LnJ1
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.