
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Bioeng. Biotechnol. , 07 March 2025
Sec. Biomaterials
Volume 13 - 2025 | https://doi.org/10.3389/fbioe.2025.1547343
This article is part of the Research Topic Advanced Functional Materials for Disease Diagnosis, Drug Delivery and Tissue Repair View all 16 articles
 Wenhe Guan1
Wenhe Guan1 Liang Zhang2*
Liang Zhang2*Diabetes is a widespread metabolic disorder that presents considerable challenges in its management. Recent advancements in biomaterial research have shed light on innovative approaches for the treatment of diabetes. This review examines the role of biomaterials in diabetes diagnosis and treatment, as well as their application in managing diabetic wounds. By evaluating recent research developments alongside future obstacles, the review highlights the promising potential of biomaterials in diabetes care, underscoring their importance in enhancing patient outcomes and refining treatment methodologies.
Diabetes mellitus is a chronic disease marked by prolonged hyperglycemia, which arises from defects in insulin secretion, insulin action, or a combination of both (American Diabetes Association, 2014; Chaudhury et al., 2017; Defronzo, 2009). The primary types of diabetes include Type 1 diabetes mellitus (T1DM) and Type 2 diabetes mellitus (T2DM). T1DM is mainly an autoimmune condition leading to the destruction of insulin-producing beta cells in the pancreas (Khaiz et al., 2025; Nyaga et al., 2018a; Nyaga et al., 2018b). In contrast, T2DM is often linked to insulin resistance, influenced by lifestyle factors and genetic predispositions (Dariya et al., 2019; Ghasemi and Norouzirad, 2019; Memon et al., 2022). The incidence of diabetes worldwide has been on a steady rise, resulting in significant public health implications, particularly as demographic trends lean towards aging populations and lifestyle changes, including increased obesity rates (Cano-Ibanez and Bueno-Cavanillas, 2024). The International Diabetes Federation reported that approximately 537 million adults were diagnosed with diabetes in 2021, with projections indicating a rise to 783 million individuals by 2045 (Klangjareonchai et al., 2021).
Conventional diabetes management approaches include pharmacological treatments such as insulin and oral hypoglycemic agents, as well as lifestyle changes encompassing diet and exercise (Deng et al., 2018). Nonetheless, achieving optimal glycemic control remains a challenge for numerous patients, often due to factors like medication adherence, the complexity of treatment protocols, and the psychosocial burdens associated with the disease (Al-Qerem et al., 2022; Summers-Gibson, 2021). These challenges highlight the urgent need for innovative strategies in diabetes management (Kalra et al., 2022).
In recent times, the application of biomaterials has surfaced as a promising pathway for the enhancement of diabetes treatment and management (Aldahish et al., 2024; Emad et al., 2024; Nemati et al., 2023). This review seeks to investigate the diverse applications of biomaterials within the realm of diabetes management, addressing their potential to mitigate the limitations of existing treatment methodologies while improving the quality of life for individuals with diabetes. The evolving role of biomaterials in diabetes management marks a significant advancement in addressing the complexities inherent to this chronic condition (Iqbal et al., 2023).
Biosensors have become essential instruments across various domains, particularly in healthcare. Within this sector, they provide rapid and precise monitoring of biological parameters (Kim et al., 2019; Li et al., 2023; Yoon et al., 2020). These sensors possess the capability to detect specific biological markers molecules, delivering crucial real-time information essential for the diagnosis, management, and prevention of diseases (Kong et al., 2024; Xing et al., 2024).
Conventional diagnostic approaches for diabetes, which largely rely on fasting plasma glucose (FPG), oral glucose tolerance tests (OGTT), and hemoglobin A1c (HbA1c) assessments, exhibit several shortcomings. These techniques are susceptible to various influences, such as stress, illness, and inconsistencies in laboratory procedures, which may result in misdiagnosis or delays in diagnosis (Young et al., 2023). For instance, HbA1c levels may not provide an accurate representation of glycemic control in specific populations, including those with hemoglobinopathies or individuals who have recently received blood transfusions (Bhatti et al., 2024). Traditional glucose testing methods, primarily based on blood glucose meters, encounter numerous challenges that hinder patient adherence and effective diabetes management. Ahmadian et al. conducted a comprehensive review of current technologies, comparing the benefits and drawbacks of both invasive and non-invasive glucose monitoring techniques (Ahmadian et al., 2023). Many of these methods necessitate finger-pricking, which can be painful and inconvenient, resulting in many patients opting to forgo regular testing (Burge, 2001). Furthermore, the precision of blood glucose meters can be influenced by several factors, including user error, calibration discrepancies, and environmental conditions, leading to variable readings (Tankasala and Linnes, 2019). Additionally, conventional testing methods typically offer only a snapshot of glucose levels at a single moment, failing to account for fluctuations that occur throughout the day. The psychological strain associated with diabetes management, including the stress from frequent monitoring and apprehension regarding complications, highlights the demand for reliable and minimally invasive glucose testing methods (Xie et al., 2023).
The significance of glucose monitoring sensors in diabetes management cannot be overstated. Recent advancements in biosensor technology have facilitated the development of non-invasive and continuous glucose monitoring systems that enhance patient adherence and improve health outcomes (Dua et al., 2024; Hina and Saadeh, 2020; Teymourian et al., 2020). The integration of biosensors with mobile technology and data analytics platforms has further increased their utility, allowing for continuous monitoring and remote health management (Arun et al., 2024; Bent et al., 2020).
Biomedical nanomaterials, particularly those engineered for glucose sensing, have demonstrated promising advancements in improving the sensitivity and specificity of diabetes diagnostics. For example, electrospun nanofibers have emerged as a novel category of functional nanocomposites exhibiting remarkable biosensing capabilities (Du et al., 2022). The incorporation of nanomaterials, such as gold nanoparticles and carbon nanotubes, has further enhanced the efficacy of biosensors, enabled the simultaneous detection of multiple analytes and accelerated response times (Nisar et al., 2024; Otero and Magner, 2020; Putzbach and Ronkainen, 2013). Moreover, when combined with organometallic compounds, these nanomaterials can significantly augment the performance of Raman spectroscopy, allowing for the detection of subtle spectral variations related to diabetes biomarkers. This synergistic approach not only improves detection sensitivity but also extends the range of potential applications in clinical diagnostics (Jagannathan et al., 2023).
The high surface area of these nanomaterials promotes increased loading of recognition elements, leading to enhanced detection capabilities. Enhanced performance of biosensors has been documented (Mousavi et al., 2022). Furthermore, nanomaterials can be tailored to respond to specific stimuli, facilitating the creation of intelligent biosensors capable of real-time monitoring of physiological variations (Scandurra et al., 2023). Recent innovations utilizing DNA nanostructures have demonstrated significant potential in biosensing applications, where they can be engineered for the selective binding of target molecules, thus improving detection specificity (Mohammad, 2024). Ongoing investigations in this field continue to reveal novel opportunities for the application of nanomaterials in biosensing, which may lead to the development of groundbreaking diagnostic tools for clinical use.
Raman spectroscopy operates on the principle of inelastic scattering of monochromatic light, typically emitted by a laser. When light interacts with the vibrations of molecules, it can scatter with a shift in energy that corresponds to the vibrational modes of those molecules. This characteristic renders Raman spectroscopy a versatile instrument for both qualitative and quantitative analyses across various applications, including the identification of biomarkers for diseases such as diabetes (Xie et al., 2023). A prominent example of this technique’s efficacy is its application in measuring urinary albumin levels, a critical biomarker for diabetic kidney disease. Research has illustrated that Raman spectroscopy can effectively identify specific spectral peaks linked to albumin concentrations in urine samples from individuals diagnosed with type 2 diabetes, indicating its potential for non-invasive monitoring of renal complications related to diabetes (Flores-Guerrero et al., 2020). Moreover, Raman spectroscopy has been employed to investigate retinal tissue for early indicators of diabetic retinopathy, offering insights into the biochemical alterations occurring in the retina due to prolonged hyperglycemia. The capacity of this technique to distinguish between healthy and diseased tissues through spectral analysis renders it an invaluable tool for early diagnosis and timely intervention in diabetic patients (Chen et al., 2021). Furthermore, advancements in machine learning algorithms applied to Raman spectral data have bolstered the precision of diabetes detection, highlighting the technology’s potential to transform diabetes management and enhance patient outcomes (Chen et al., 2024e).
The amalgamation of biomedical nanomaterials with Raman spectroscopy presents numerous advantages while also posing significant challenges (Oliveira et al., 2022). A primary advantage of this integration lies in the enhancement of diagnostic accuracy and sensitivity. For instance, a core-shell structure of Au nanorods@Raman tags@SiO2@Ag nanocomposite has been synthesized and employed for the surface-enhanced Raman scattering (SERS) detection of insulin and C-peptide in trace serum (Zhang et al., 2024b). This is illustrated in Figure 1.
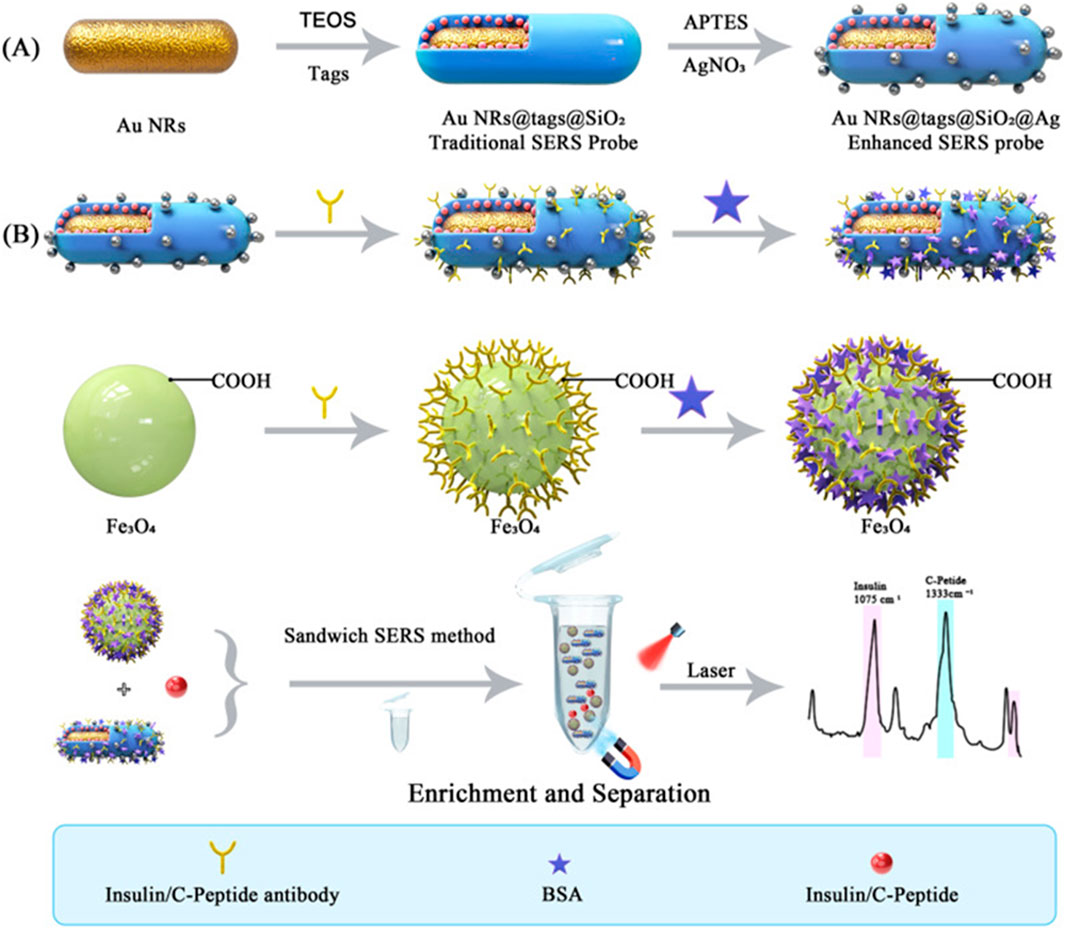
Figure 1. Depicts the schematic representation of (A) the synthesis of the SERS probe and (B) the SERS-based immunoassay utilized for the detection of insulin antibodies and C-peptide antibodies. Reproduced with permission from Zhang et al. (2024b).
Exhalation detection technology has attracted notable interest due to its non-invasive and convenient nature, particularly in the regulation of glucose levels, which is essential for managing conditions such as diabetes. In breath analysis, glucose is often detected indirectly through its metabolic byproducts, including acetone, which is produced during the metabolism of fatty acids when glucose levels are diminished (Galassetti et al., 2005; Hwang et al., 2021; Li et al., 2015). The well-established relationship between breath acetone and blood glucose levels provides a foundation for the development of sensors capable of measuring glucose levels via breath analysis (Kalidoss and Umapathy, 2019; Righettoni et al., 2013; Tanda et al., 2014). The accurate and prompt identification of acetone is vital for maintaining safety in industrial production and for the clinical assessment of diabetes. Consequently, the advancement of high-performance acetone sensors has become increasingly significant (Guan et al., 2025) (Table 1). Analyzing breath can facilitate real-time observation of metabolic alterations, enabling timely interventions to avert conditions such as hyperglycemia or hypoglycemia (Xie et al., 2023). Moreover, breath testing is characterized by its convenience and discretion, which enhances patient adherence and promotes ongoing monitoring during daily activities (Vajhadin et al., 2021).
However, environmental factors substantially influence the detection of glucose and its metabolites in exhaled breath, impacting both the collection and analytical processes. Elements such as humidity and the presence of competing volatile compounds can hinder gas sensor performance (Esteves et al., 2022; Xie et al., 2018). Therefore, it is essential to optimize the operating conditions of sensors to mitigate these environmental effects. Zhou et al. developed a self-designed condensation device for exhaled breath, which allowed for the condensation and collection of human exhaled breath, enabling the analysis of glucose in the collected condensate via ion chromatography using a pulsed amperometric instrument (Zhou et al., 2022). For instance, custom-built exhaled breath collection devices that regulate temperature and humidity have demonstrated potential for enhancing the reproducibility of glucose measurements in breath samples (Desai et al., 2025). A noninvasive blood glucose detection apparatus that utilizes acetone sensing in exhaled breath employs an α-Fe2O3-multiwalled carbon nanotube (MWCNT) nanocomposite to accurately measure acetone levels, even in high humidity conditions (Ansari et al., 2024). Furthermore, the incorporation of nanostructured materials and composite sensors has been shown to improve sensitivity and selectivity, enabling more precise glucose detection in the presence of interfering substances found in exhaled breath (K et al., 2025). Notably, an ultrasensitive acetone gas sensor based on a K/Sn-Co3O4 porous microsphere can accurately differentiate between diabetic patients and healthy individuals based on variations in acetone concentrations without the need to eliminate water vapor from exhaled breath, highlighting its substantial potential for diabetes diagnosis (Na et al., 2024).
Wearable sensors made from biomaterials designed for sweat glucose detection have garnered significant interest due to their capacity for continuous monitoring without the discomfort of finger-prick tests. Zhou et al. conducted a thorough review of the principles and advancements in electrochemical glucose sensors, compiling findings on various innovative nanomaterials suitable for continuous glucose monitoring (CGM) (Zhou et al., 2024b). The work illustrated the applications and construction strategies of diverse nanomaterials, including precious metals, nanometals, their compounds, and nonmetallic nanomaterials. Figure 2 in their study encapsulates these insights on CGM technology, while Figure 3 traces the evolution of biosensor development for wearables up to 2021.
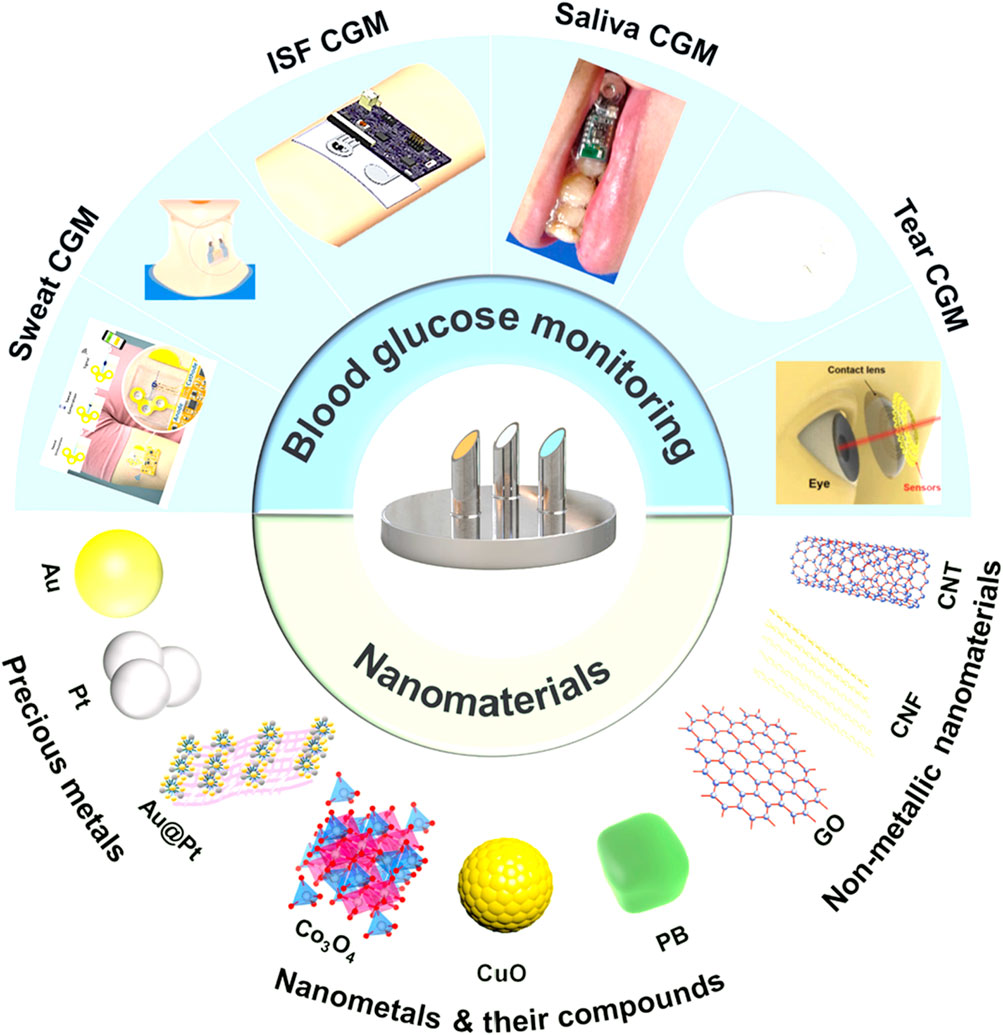
Figure 2. Illustrates the continuous glucose monitoring (CGM) sensors employed for the assessment of various biological fluids and the nanomaterials developed for tear glucose analysis in recent years, reproduced with permission from Zhou et al. (2024b).
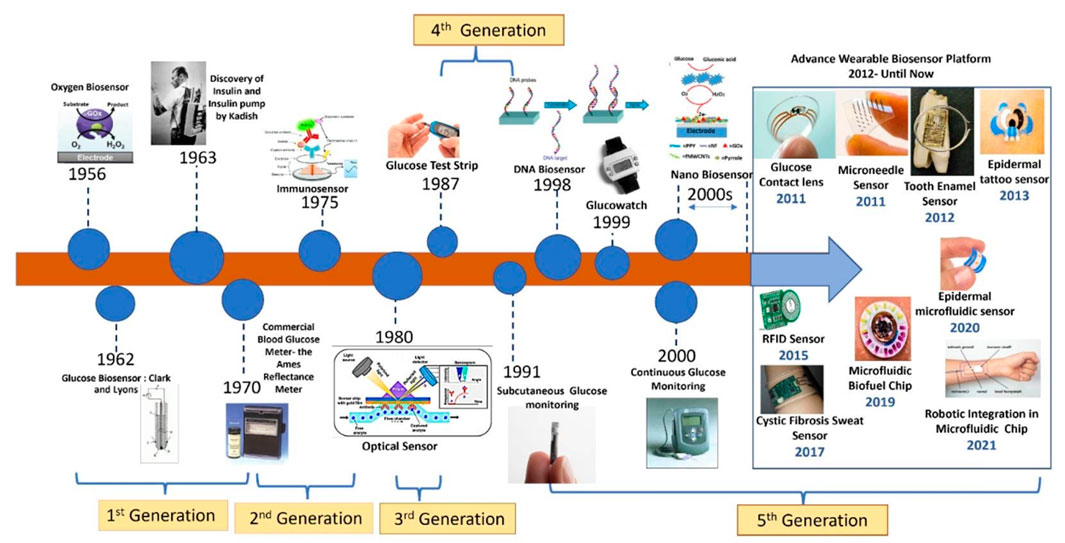
Figure 3. Depicts the historical progression of biosensor development for wearable technology up to 2021, reproduced with permission from Zafar et al. (2022).
These sensors utilize cutting-edge materials and designs to enhance sensitivity and selectivity, thereby enabling precise real-time glucose detection (Dervisevic et al., 2022; Zafar et al., 2022; Zhou et al., 2023). The hyaluronate (HA)-modified Au@Pt bimetallic electrodes have been validated through animal trials for their capacity to provide long-term, accurate, and robust CGMs in smart contact lenses, paving the way for continuous blood glucose monitoring (Han et al., 2023). In 2023, Zhang et al. summarized the metallic nanomaterials employed in wearable non-invasive glucose sensors, encompassing zero-dimensional (0D), one-dimensional (1D), and two-dimensional (2D) monometallic nanomaterials, as well as bimetallic configurations (Zhang et al., 2023b). In addition, Govindaraj et al. provided a thorough summary of various categories of non-enzymatic glucose sensor materials, which encompass composites, non-precious transition metals along with their respective metal oxides and hydroxides, precious metals and their alloys, carbon-based materials, conducting polymers, metal-organic framework (MOF)-based electrocatalysts, as well as glucose sensors designed for wearable devices (Govindaraj et al., 2023). Furthermore, enzyme-free nanoparticle-based glucose sensors signify a noteworthy advancement, presenting a more straightforward and cost-efficient alternative for glucose monitoring (Boucheta et al., 2024). Additionally, microfluidic devices have been engineered to assess the performance of these sensors, thereby ensuring their reliability in clinical environments (Yunos et al., 2021). Zhang et al. introduced a handheld biosensor capable of detecting acetone through fluorescence, utilizing the enzymatic reaction of secondary alcohol dehydrogenase (S-ADH) in conjunction with β-nicotinamide adenine dinucleotide (NADH, λex = 340 nm, λem = 490 nm). This device, characterized by its portability and high sensitivity and selectivity, is anticipated to see extensive application in clinical diagnostics as well as in the realm of wearable biochemical sensors in the forthcoming years (Zhang G. et al., 2025). As advancements in these technologies continue, they hold the potential to revolutionize diabetes management, enabling patients to achieve optimal glucose levels with enhanced convenience.
Insulin plays a pivotal role in the management of diabetes, necessitating effective delivery mechanisms. The utilization of biomaterials, known for their exceptional biocompatibility, degradability, and distinctive functional properties, is essential in this context. Such materials significantly enhance insulin stability, modulate its release kinetics, and facilitate targeted delivery, thereby offering a safer and more efficient therapeutic option for individuals with diabetes. Novel biomaterial carriers can transport antidiabetic drugs to address different types of diabetes (Table 2).
Insulin is indispensable for managing T1DM and is often required in numerous instances of T2DM. The engineered characteristics of nanoparticles, such as toxicity control, stability, and drug release mechanisms, allow for the delivery of higher drug concentrations to targeted sites (Zaric et al., 2019). The capacity of nanoparticle systems to improve insulin delivery through targeted and controlled release mechanisms has attracted significant attention (Cheng et al., 2021; Karimi et al., 2016; Zhang et al., 2022). Nanocarriers present an innovative strategy by offering advantages such as enhanced drug stability and absorption, targeted delivery to specific tissues or cells, controlled or stimuli-responsive drug release, increased bioavailability, minimized side effects, and improved patient compliance (Figure 4). Sarkhel et al. have encapsulated the diverse applications of nanomaterials in diabetes management, emphasizing the distinctive attributes of nano-based drug delivery systems and intelligent drug delivery techniques (Sarkhel et al., 2024). These nanoparticles can be customized to react to physiological conditions, such as fluctuating glucose levels, thereby permitting a more personalized approach to insulin administration (Karimi et al., 2016; Sharmah et al., 2024). MSN-based nanocomposites have been used to deliver therapeutic molecules like insulin, GLP-1, exenatide, DPP-4 inhibitor and plasmid-containing GLP-1 genes for managing diabetes mellitus for the last decade (Sarkar et al., 2023). For instance, innovative systems have emerged that leverage glucose-responsive nanoparticles to release insulin during hyperglycemic episodes, thereby effectively imitating the pancreas’s physiological insulin secretion mechanism (Jeong et al., 2022; Volpatti et al., 2021).

Figure 4. Demonstrates the various advantages of employing nanotechnology in diabetes management compared to traditional treatment methodologies. Reproduced with permission from Sarkhel et al. (2024).
Moreover, the inclusion of biocompatible materials in the formulation of nanoparticles ensures safety and efficacy in clinical applications (Tutty et al., 2022). Research has illustrated that nanoparticles can successfully encapsulate insulin, providing protection against degradation within the gastrointestinal tract during oral administration (Ren et al., 2023). This pioneering strategy not only enhances the stability of insulin but also promotes its absorption, yielding improved glycemic control in diabetic individuals. The integration of nanoparticles into insulin delivery systems indicates substantial potential for the development of more effective and patient-friendly diabetes treatments.
Tissue engineering has emerged as a groundbreaking technique in diabetes management, particularly in addressing the complications associated with the disease (Kaviani and Azarpira, 2016; Woo et al., 2023). This interdisciplinary domain merges biological, mechanical, and engineering principles to restore or enhance the functionality of damaged tissues and organs. Considering the increasing prevalence of diabetes and its complications, innovative strategies such as tissue engineering provide promising avenues for regeneration and repair, particularly in pancreatic and cellular contexts. Advancements within this field possess the potential to significantly enhance patient outcomes and offer alternatives to traditional therapies like insulin administration and organ transplantation.
The domain of pancreatic tissue engineering is primarily focused on the creation of functional pancreatic tissues or bioartificial organs designed to restore insulin secretion in diabetic patients (Figure 5). Recent investigations have underscored the encouraging role of decellularized pancreatic scaffolds, which maintain the extracellular matrix (ECM) architecture and critical biochemical signals necessary for cell attachment and functionality. The application of decellularized pig pancreas has shown promise in establishing an optimal environment for insulin-producing cells, thereby addressing the impairment of beta-cell function in T1DM (Hao L. et al., 2024; Lim et al., 2023). Research indicates that these bioengineered tissues can effectively replicate the intrinsic architecture of the pancreas, which may enhance both the survival rates and functionality of transplanted islet cells (Lim et al., 2023). Furthermore, advancements in 3D bioprinting technology have enabled the fabrication of complex pancreatic structures, thereby improved vascularization and facilitating the delivery of essential nutrients required for maintaining cell viability (Soetedjo et al., 2021). Additionally, the incorporation of bioactive materials, such as silver nanoparticles, has demonstrated improved biocompatibility of these scaffolds, further supporting their clinical application (Qiu et al., 2022). In summary, pancreatic tissue engineering holds significant promise in the advancement of regenerative therapies for diabetes.
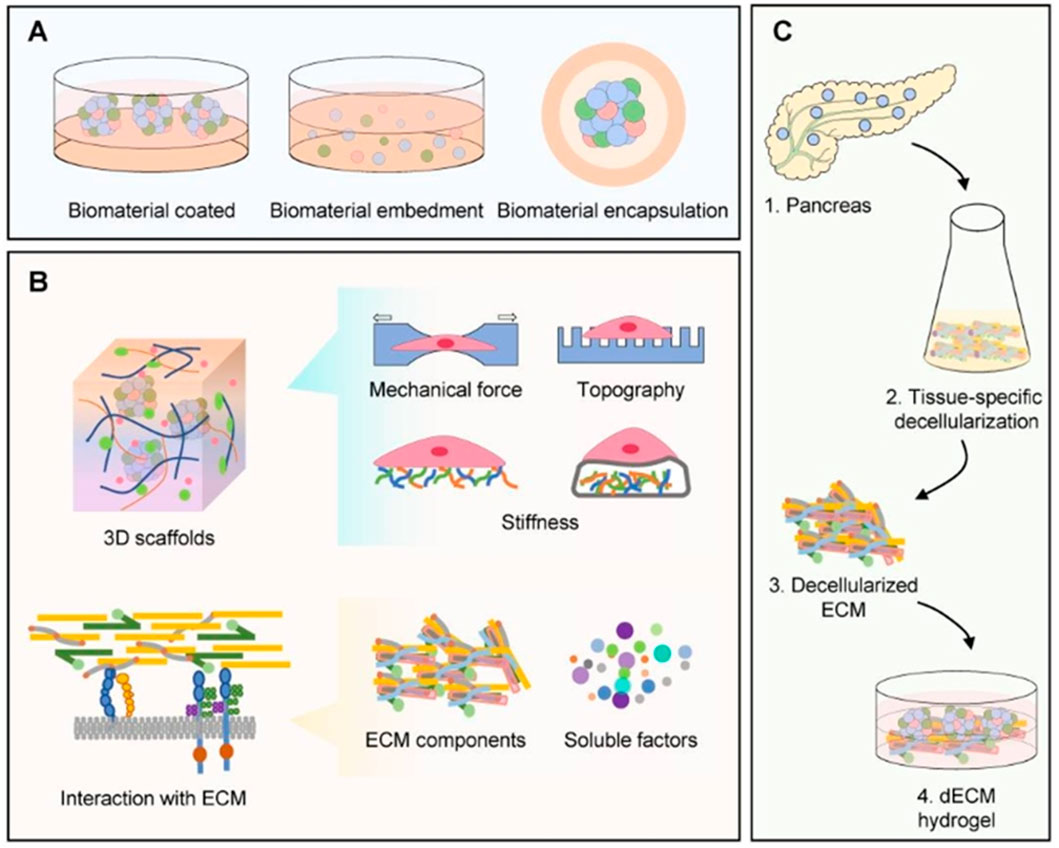
Figure 5. Illustrates a diagrammatic representation highlighting the application of materials in human islet organoids. (A) Applications of materials for production of human islet organoids, including strategies such as biomaterial coating, embedding, and encapsulation, plays a critical role in the advancement of diabetes treatments. (B) Biomaterials serve as three-dimensional scaffolds that replicate the native interactions with the extracellular matrix (ECM) essential for the generation of islet organoids. These scaffolds provide key factors such as mechanical forces, topographical features, stiffness, and signaling from ECM components and soluble factors. (C) The manufacturing process of decellularized ECM (dECM) materials is highlighted. This content is reproduced with permission from Jiang et al. (2022).
Cell transplantation, particularly the transplantation of islet cells, remains a fundamental aspect of T1DM management, with the primary objective of reinstating endogenous insulin production (Loretelli et al., 2020; Ramesh et al., 2013). However, barriers such as a limited supply of donors and the risk of immune rejection have hindered broader implementation. Recent advancements in tissue engineering have introduced innovative strategies aimed at enhancing the success rates of cell transplantation. For example, the application of interconnected toroidal hydrogels for islet encapsulation has proven effective in protecting transplanted cells from immune attacks. While still facilitating nutrient exchange (Ernst et al., 2019).
Additionally, the engineering of pluripotent stem cells into insulin-producing cells stands as a groundbreaking approach to generate a continual supply of functional cells for transplantation (Carvalho et al., 2022; Kasputis et al., 2018; Pagliuca et al., 2014). Further research has examined the potential of regulatory T cells that have been modified with insulin-specific chimeric antigen receptors to promote tolerance and reduce the risk of rejection during islet transplantation (Azad et al., 2024). These advancements in cell transplantation methodologies, when integrated with the principles of tissue engineering, hold the promise of significantly enhancing both the effectiveness and accessibility of diabetes treatments.
The fabrication of biomaterial scaffolds constitutes a crucial aspect of tissue engineering within the framework of diabetes therapy, as they provide vital structural support for cellular growth and tissue regeneration. These scaffolds emulate the ECM and promote a three-dimensional structure that is conducive to cell proliferation, differentiation, and development. They may also be employed in the management of diabetic wounds, a common complication associated with diabetes (Tallapaneni et al., 2021). Scaffolds can be categorized into two main types based on their origin: natural and synthetic polymer-based scaffolds.
Natural biomaterials have garnered substantial interest in the field of tissue engineering because of their intrinsic biocompatibility and their capacity to facilitate cellular activities that are crucial for tissue regeneration (Bagheri et al., 2020; Mei et al., 2023). These materials, sourced from biological origins, include collagen, gelatine, chitosan, and alginate, which replicate the ECM of native tissues, thus fostering cellular interactions and enhancing healing processes (Naranda et al., 2021; Sonmezer et al., 2023). This is illustrated in Figure 6. For instance, collagen scaffolds are particularly recognized for their excellent properties regarding cell adhesion and biodegradability, rendering them suitable for applications in wound healing and regenerative medicine (Chu et al., 2018; Larijani et al., 2024). Chitosan, a natural polysaccharide, exhibits remarkable biocompatibility, biodegradability, and antimicrobial capabilities, positioning it as a promising candidate for wound healing and tissue engineering applications (Wang J. et al., 2024).
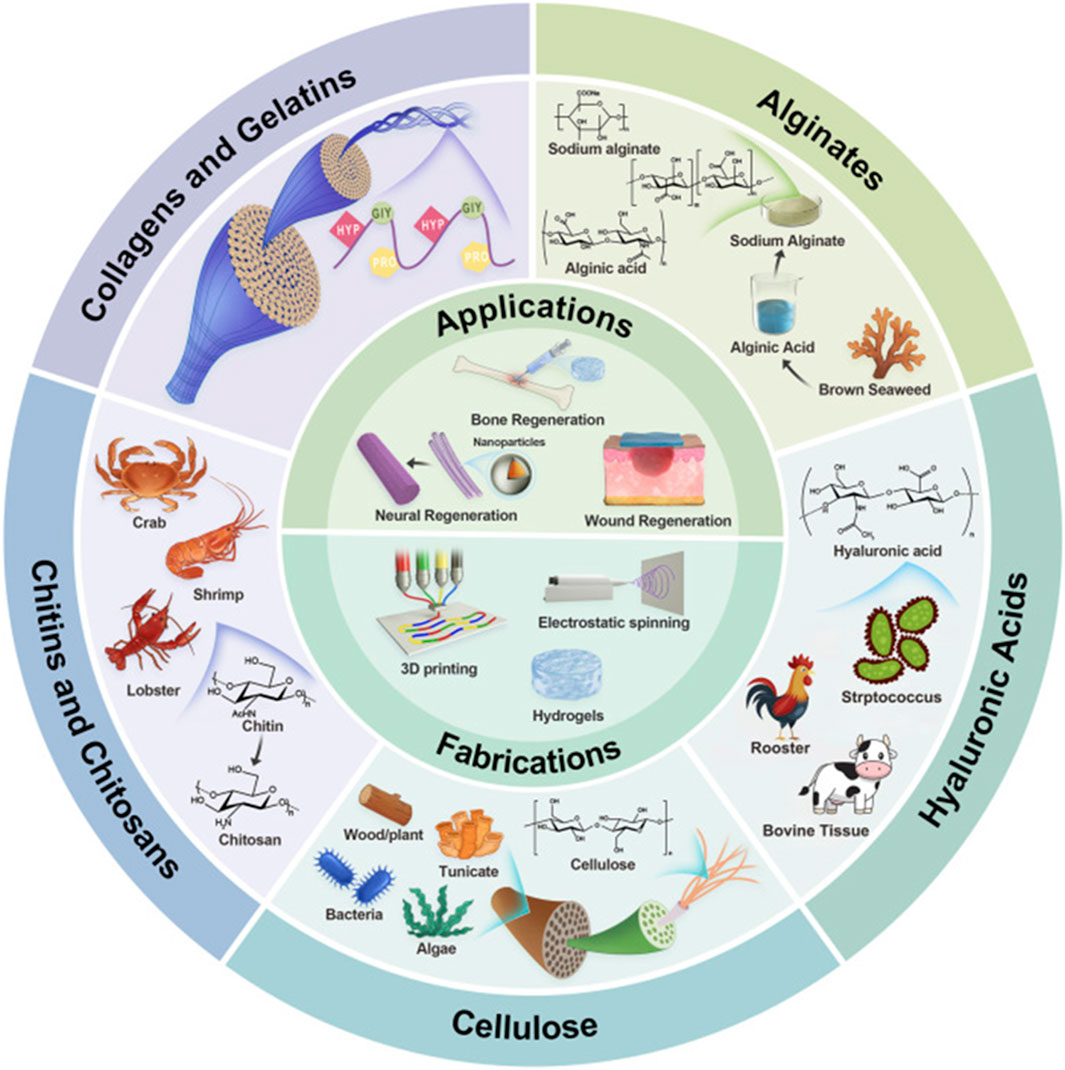
Figure 6. Illustrative schematic of naturally derived polymers:origin, structures, fabrications, and applications. Reproduced with permission from Hu et al. (2024a).
Moreover, the incorporation of bioactive molecules, such as growth factors and peptides, into natural materials can significantly enhance their regenerative capacity, leading to improved results in tissue repair and regeneration (Ravoor et al., 2021). Additionally, natural scaffolds can undergo modifications to improve their mechanical strength and degradation rates, thus allowing for customization tailored to specific applications. The inherent bioactive characteristics of natural biomaterials are further validated by their capacity to promote angiogenesis and facilitate tissue integration, both of which are essential for achieving favourable outcomes in tissue engineering (Goonoo, 2022).
The degradation behavior of these natural materials serves as a pivotal aspect concerning their application in biomedical contexts, significantly affecting their longevity, biocompatibility, and overall efficacy in tissue regeneration (Hu T. et al., 2024). Generally, natural materials are preferred due to their ability to undergo in vivo degradation, which permits a gradual replacement by newly synthesized tissue. Specifically, chitosan-based hydrogels have demonstrated a degradation process primarily governed by hydrolytic mechanisms, with degradation rates that can be modulated by varying the degree of crosslinking and the molecular weight (Lv et al., 2023). This characteristic proves particularly beneficial in scenarios such as drug delivery, where the establishment of controlled release profiles is vital for achieving therapeutic effectiveness.
The degradation byproducts of natural materials are frequently non-toxic and can be metabolically processed by the body, thereby minimizing the likelihood of adverse reactions (Xu et al., 2022). The capacity to engineer natural materials with specific degradation kinetics enhances their applicability across a range of uses, including bone regeneration, where it is optimal for scaffolds to degrade in synchrony with the formation of new bone (Koh et al., 2022). In summary, the degradation characteristics of natural materials not only contribute to their biocompatibility but also are integral to their functionality and efficacy in the field of regenerative medicine.
Synthetic biomaterials, such as polycaprolactone (PCL), polylactic acid (PLA), and polyvinyl alcohol (PVA), have been engineered to address certain limitations associated with their natural counterparts (Deng et al., 2022). Research indicates that PCL scaffolds can effectively support the proliferation of mesenchymal stem cells and promote wound healing in models of diabetes (Abdollahi et al., 2024b). These synthetic materials provide customizable mechanical properties, controllable degradation rates, and can be fabricated into various forms, including fibers, films, and hydrogels (Lim et al., 2023; Li et al., 2020). This flexibility enables the optimization of material properties to better align with the mechanical characteristics of natural tissues, which is critical for applications involving implants and wound dressings. Investigations have shown that by adjusting the cross-linking density and the composition of the polymer network, researchers can develop hydrogels with tailored mechanical properties that are conducive to enhancing cell adhesion and proliferation in tissue engineering (Huang et al., 2023).
The adaptability of synthetic biomaterials facilitates the integration of bioactive agents, including growth factors or therapeutic drugs, allowing for their controlled release to foster healing and tissue regeneration (Guo et al., 2022b). Furthermore, the incorporation of nanomaterials into synthetic polymers has significantly improved their mechanical attributes, yielding materials that not only exhibit enhanced strength and durability but also demonstrate bioactivity that supports healing and integration with surrounding tissues (Abdollahi et al., 2024b). The integration of conductive materials within scaffolds has been explored to enhance the functional capacity of engineered tissues through improved electrical signalling, which is particularly important for insulin secretion in pancreatic cells (Wang and Jin, 2024). Moreover, advancements in three-dimensional printing technologies have facilitated the creation of intricate scaffold architectures that accurately replicate the structure of native tissues, thereby further augmenting the effectiveness of these biomaterials. The enhancement of integration and functionality in biomaterials has been highlighted by (Metwally et al., 2023). The adaptability of these mechanical properties is crucial for the effective incorporation of synthetic materials in clinical applications, as it enables the design of substances capable of enduring physiological stresses while supporting biological activities.
By amalgamating various functionalities within a single biomaterial, researchers are equipped to tackle diverse therapeutic challenges simultaneously. Such biomaterials can facilitate the controlled release of therapeutic agents, thereby promoting localized healing and reducing systemic side effects (Heidari et al., 2023). Additionally, these multifunctional materials can embed antibacterial characteristics to mitigate infections, which commonly arise in chronic wounds (Renuka et al., 2022).
Despite the considerable benefits offered by synthetic materials, significant concerns regarding their degradation and biocompatibility persist as critical hurdles in their utilization. For instance, materials engineered for temporary implants must degrade in synchronization with tissue healing to prevent complications linked to either premature breakdown or prolonged presence in the organism (Li et al., 2022). Moreover, ensuring the biocompatibility of synthetic materials is vital, as those that provoke adverse immune responses can incite chronic inflammation and result in implant failure (Ciatti et al., 2024; Kzhyshkowska et al., 2015). Recent progress has concentrated on the creation of biodegradable polymers that preserve their mechanical strength while systematically decomposing into non-toxic byproducts (Guo et al., 2022b). Addressing these concerns surrounding degradation and biocompatibility is essential for the successful transition of synthetic materials from laboratory settings to clinical implementations, guaranteeing that they offer safe and effective solutions for patients.
Microneedle technology has emerged as a groundbreaking approach for insulin delivery, providing a minimally invasive alternative to conventional injection techniques (Bigham et al., 2025; Hong et al., 2022; Zong et al., 2022). The mechanism of insulin release from microneedles is depicted in Figure 7. These micro-scaled needles, which typically range in length from 25 to 1,000 μm (Figure 8), can penetrate the outer layer of the skin while circumventing nerve endings, thus minimizing discomfort and pain for patients (Chen et al., 2019; Luo et al., 2023; Wang et al., 2020). Recent advancements in the manufacturing technologies for microneedles, including 3D printing and mold-based methods, have enabled the creation of arrays capable of delivering precise dosages of insulin (Razzaghi et al., 2024). Evidence suggests that these microneedle arrays achieve bioavailability levels that are comparable to those obtained from traditional subcutaneous injections while significantly enhancing patient adherence due to their ease of use and reduced pain perception (Li et al., 2022; Queiroz et al., 2020). Furthermore, the incorporation of biodegradable materials in microneedle design has allowed for sustained insulin release, presenting a viable solution for long-term diabetes management (Chakraborty et al., 2023; Rajput et al., 2021). A nanoparticle-loaded microneedle (MN) patch, designed for transdermal drug delivery, aims to achieve blood glucose control and reactive oxygen species (ROS) scavenging for the synergistic treatment of diabetic nephropathy, thereby enhancing the efficiency of transdermal drug delivery while extending the duration of insulin action (Zheng et al., 2025). In summary, microneedle technology stands as a promising strategy for advancing insulin delivery systems and subsequently enhancing the quality of life for individuals with diabetes.
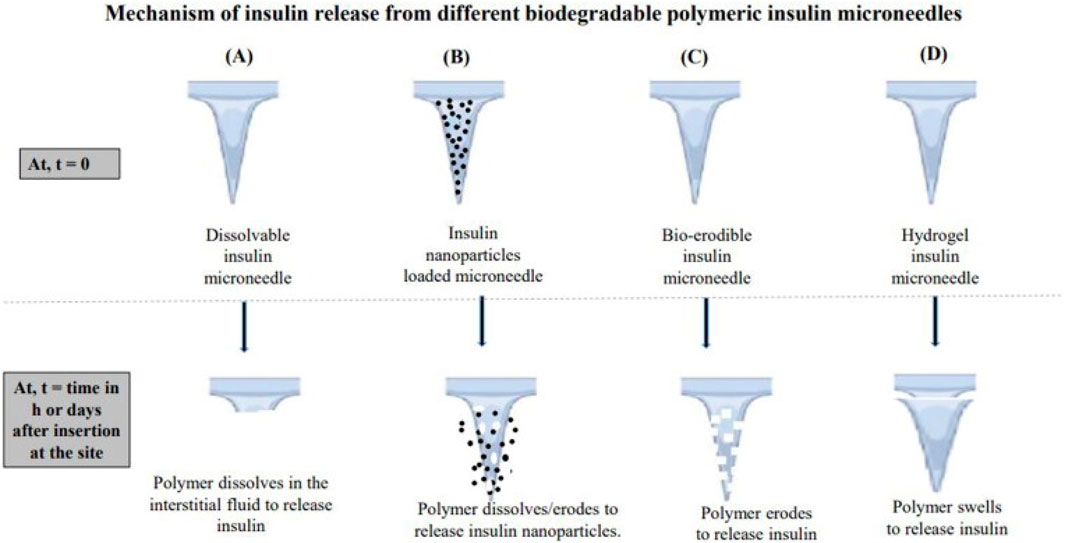
Figure 7. Mechanism of insulin release from the microneedles. (A) shows a soluble insulin microneedle that releases insulin through polymer dissolution. (B) shows a microneedle loaded with insulin nanoparticles, and insulin is released through the biodegradation of the shell or matrix. (C) shows a biodegradable insulin microneedle, and insulin is released after enzymatic hydrolysis. (D) shows a hydrogel insulin microneedle that continuously releases insulin after entering the dermis. Reproduced with permission from Starlin et al. (2024).

Figure 8. Hollow microneedles created from silicon and polymers. (A, B) Hollow microneedles with a tapered shape. Mukerjee et al. (2015), Wilke et al. (2005) (C) Hollow silicon microneedles with sharp tips Ma et al. (2006). (D) cylindrical microneedles with a side-opening orifice Zhang et al. (2009) (E) Hollow silicon microneedles with sharp tips Baron et al. (2008) (F) Hollow microneedles by exposing X-ray through a mask onto PolyMethylMetaAcrylic. Moon et al. (2005) (G) A micro-gear pump Amirouche et al. (2009) (H) Microneedles with on-board fluid pumps Lin and Pisano (1999) (I) Flow of liquid through glass hollow microneedles controlled by CO2 gas pressure Martanto et al. (2006) (J) An electrical microneedle applicator Verbaan et al. (2008) (K) Flow of liquid through hollow microneedles controlled by a syringe pump Gupta et al. (2009). Reproduced with permission from Kim et al. (2012).
The Smart delivery systems represent the cutting edge of insulin administration technology, merging innovative biomaterials with responsive mechanisms to develop dynamic delivery platforms. These systems are engineered to release insulin in a controlled manner, guided by real-time blood glucose monitoring, thereby providing a customized approach to managing T1DM (Condren et al., 2019; Latham, 2019; Moser et al., 2025; Renard, 2023). For example, hydrogels that expand or contract in response to changes in glucose concentrations have been developed, enabling on-demand insulin release as required (Ali et al., 2022; Annicchiarico et al., 2024). Furthermore, the incorporation of wearable technology within these smart delivery systems facilitates continuous glucose monitoring, which allows for automatic insulin administration in reaction to fluctuations in glucose levels (Renzu et al., 2024). This heightened level of responsiveness not only improves glycemic control but also reduces the risk of hypoglycemia, a prevalent issue in diabetes management. As research progresses, the potential for intelligent delivery systems to transform insulin therapy becomes increasingly evident, paving the way for more effective and user-friendly diabetes care solutions.
Current practices in managing diabetic wounds are based on four essential principles: (1) debridement, (2) infection control, (3) offloading, and (4) revascularization (Hu et al., 2022). In the context of diabetic wounds, particularly foot ulcers, the primary factor contributing to delayed healing is the diminished synthesis of collagen. This reduction adversely affects the solubility of the extracellular matrix (ECM) and provokes an exaggerated inflammatory response (Nirenjen et al., 2023). The inflammatory phase is marked by the secretion of pro-inflammatory cytokines such as IL-1, IL-6, and TNF-α. The subsequent proliferative phase is characterized by impaired angiogenesis and vasculogenesis, whereas in the remodeling phase, an increase in matrix metalloproteinases (MMPs) results in further degradation of the ECM, thereby exacerbating the challenges associated with wound healing (Figure 9). These factors present considerable hurdles for clinical management. Although traditional dressings have historically been essential in wound care, their effectiveness in treating diabetic wounds is significantly limited (Saco et al., 2016; Wang et al., 2024).
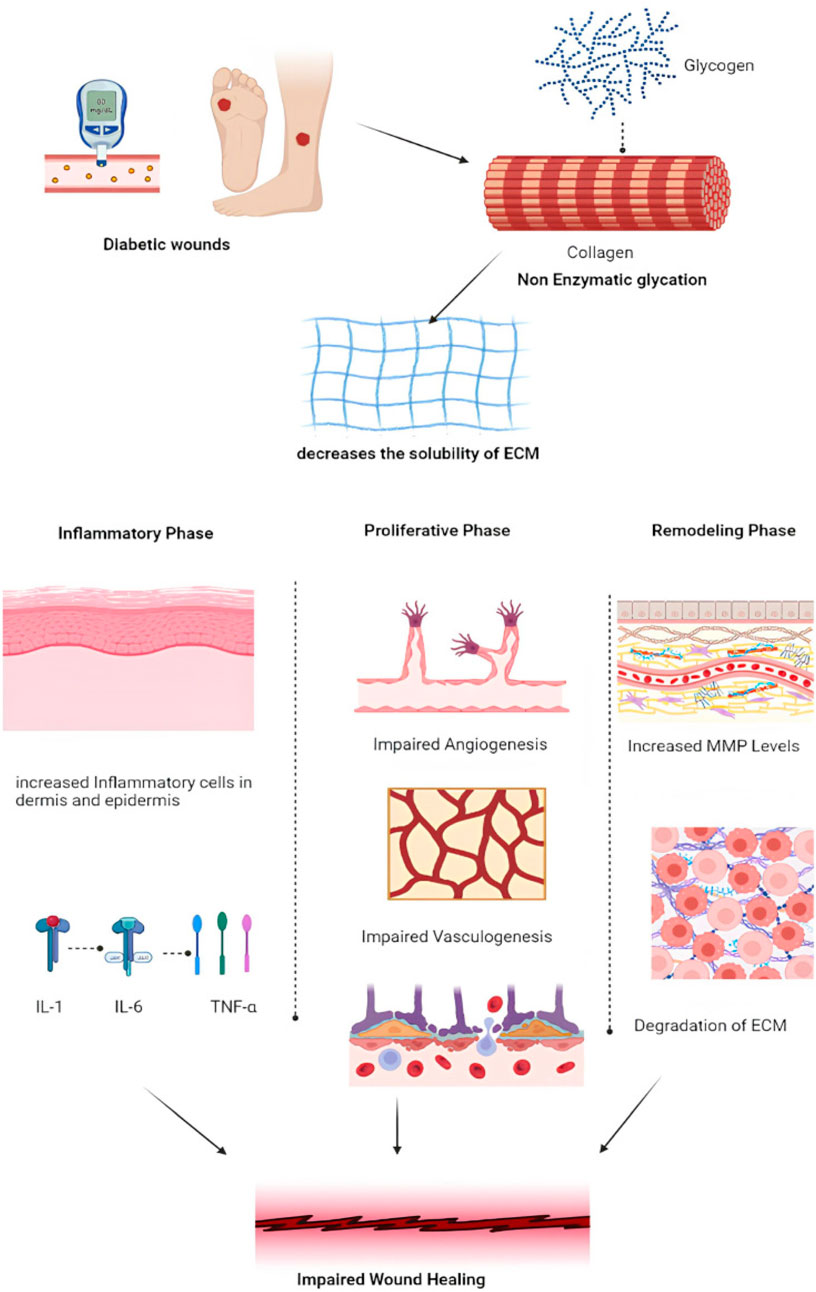
Figure 9. Schematic diagram of diabetic wound repair. Reproduced with permission from Aldahish et al. (2024).
Diabetic wounds exhibit a complex pathophysiological profile that includes impaired angiogenesis, a weakened immune response, and an increased vulnerability to infections (Rodriguez-Rodriguez et al., 2022). A significant limitation of conventional dressings, including gauze and hydrogels, is their singular functionality, which fails to adequately address the diverse challenges associated with diabetic wounds (Venkatesan and Rangasamy, 2023; Zhang et al., 2023a). These traditional dressings often lack the incorporation of bioactive agents that could facilitate healing, and their capacity to prevent bacterial proliferation is insufficient, leading to a heightened risk of infection (Zhou et al., 2024a). Furthermore, issues with adherence and retention of these dressings can necessitate frequent changes, which may disrupt the healing process and inflict additional pain and discomfort on patients (Jiang et al., 2023).
Moreover, the healing duration associated with conventional dressings can be extended, raising concerns for diabetic individuals who are predisposed to complications such as foot ulcers and potential amputations (Andrews et al., 2015; Sahu et al., 2018). The absence of advanced features in these dressings means they do not support critical physiological processes, such as angiogenesis and collagen deposition, which are essential for effective wound repair (Zhang et al., 2024). Consequently, there exists a pressing need for the formulation of more effective wound care solutions that integrate bioactive materials along with multifunctional attributes to enhance the healing of diabetic wounds (Cai F. et al., 2023). Various types of dressings, including conventional, bioactive, and interactive dressings, as well as skin substitutes, are being employed to treat wounds (Alven et al., 2020) (Figure 10).

Figure 10. Classification of wound dressings. Reproduced with permission from Alven et al. (2020).
In conclusion, while traditional dressings have played a crucial role in wound management, their inadequacies in addressing diabetic wounds underscore the necessity for a transition towards more innovative treatment strategies that can effectively tackle the distinct challenges they present. The diabetes patients can benefit significantly from the incorporation of sophisticated biomaterials and innovative technologies, which may prove instrumental in addressing existing challenges and enhancing patient outcomes in the management of diabetic wounds (refer to Table 3).
Recent innovations in wound dressing technologies have culminated in the creation of multifunctional dressings that incorporate biocompatible materials along with bioactive agents (Figure 11). According to a systematic review conducted by Vargas et al., bioactive glass (BG)-based materials show promise in expediting all phases of diabetic wound healing and improving the overall quality of wound recovery (Vargas et al., 2024). For example, electrospun nanofibers and hydrogels are employed to fabricate dressings that not only provide a protective barrier but also deliver therapeutic agents directly to the wound site (Fahimirad and Ajalloueian, 2019; Hong et al., 2023; Yang and Xu, 2023). Furthermore, the integration of electrical stimulation within wound dressings has revealed potential for enhancing healing rates by fostering cellular activities and optimizing blood circulation to the affected regions (Asadi and Torkaman, 2014; Fan et al., 2024; Hu Y. W. et al., 2024). Additionally, the incorporation of antimicrobial additives into wound dressings is increasingly gaining traction, providing an additional layer of defense against infections, which are a common complication in diabetic wounds (Chen et al., 2024d; Firoozbahr et al., 2023; Li et al., 2024; Rozman et al., 2020). In summary, advancements in wound dressing technologies signify a considerable leap forward in the effective management of diabetic wounds, offering tailored solutions.
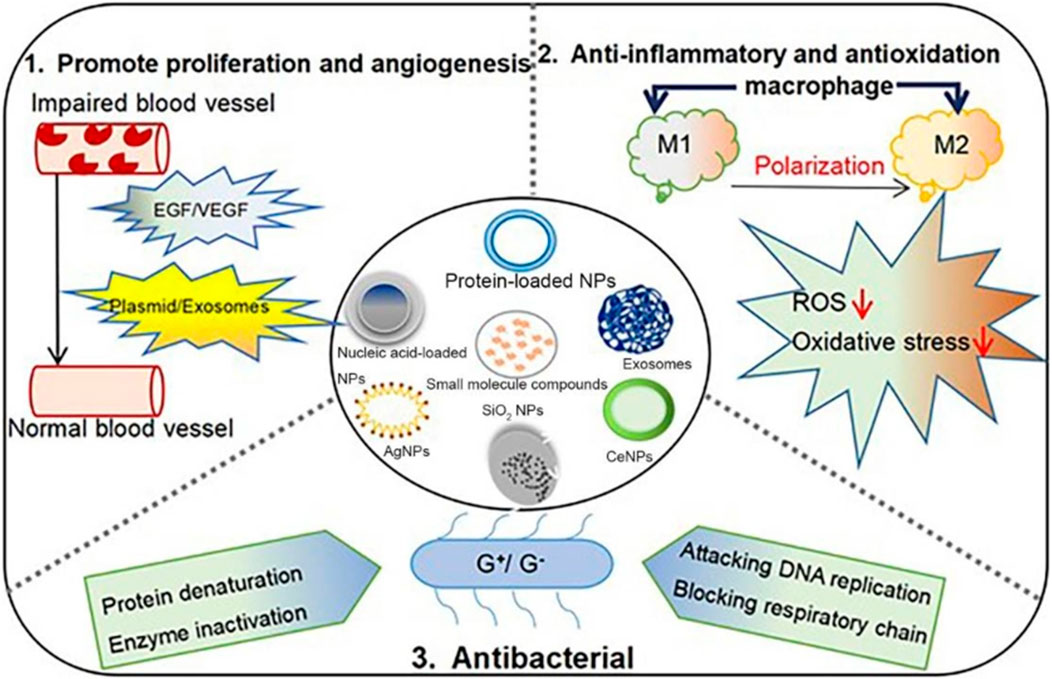
Figure 11. Presents a diagrammatic representation illustrating the various classifications and therapeutic mechanisms associated with biomaterials utilized in the management of diabetic wounds. This illustration is reproduced with permission from Qin et al. (2022).
The biocompatibility of materials is a crucial factor in the development of biomaterials for biomedical applications, particularly in the context of diabetic wound healing (Nandhakumar et al., 2022; Ren et al., 2022; Xu et al., 2023). These materials are specifically designed to interact positively with biological systems, thereby reducing adverse reactions while facilitating healing processes (Chan et al., 2023; Naahidi et al., 2017). Recent studies have underscored the promising potential of various biocompatible materials, such as chitosan, alginate, and hyaluronic acid, which have shown encouraging outcomes in promoting the healing of diabetic wounds (Peng et al., 2022). Although clinical trials remain limited, chitosan has emerged as a highly effective alternative for modulating local inflammatory responses and promoting wound healing, especially in patients with comorbid conditions that hinder typical skin healing processes, such as diabetes and vascular insufficiency (Maita et al., 2022). Chitosan-based biomaterials have gained recognition for their efficacy in wound healing, characterized by their antibacterial properties and ability to enhance cellular proliferation, rendering them suitable candidates for applications in wound care (Cai and Li, 2020; Rajinikanth et al., 2024). Systematic reviews and meta-analyses have established that, relative to the standard of care (SOC), patients receiving placenta-derived biomaterial treatments demonstrate a superior rate of complete wound healing in cases of diabetic foot ulcers (DFUs) (Ruiz-Munoz et al., 2024). Chen et al. corroborated that placenta-based tissue products exhibited the highest likelihood of wound healing (p-score = 0.90), followed by living cell skin substitutes (p-score = 0.70), acellular skin substitutes (p-score = 0.56), and advanced topical dressings (p-score = 0.34) when measured against standard DFU care (Chen L. et al., 2024).
Angiogenesis and cellular migration are fundamental processes in wound healing, which are frequently disrupted in diabetic wounds (Yang et al., 2024). Consequently, an optimal biomaterial should facilitate the development of new blood vessels to enhance blood flow and oxygen supply at the wound site. Achieving these characteristics necessitates the engineering of physico-chemical properties at both chemical and molecular levels, ensuring alignment with the required bioactivity for wound healing in diabetic conditions (Sharma and Kishen, 2024) (Figure 12). This necessity highlights the importance of comprehending the structure–function relationship within biopolymers.
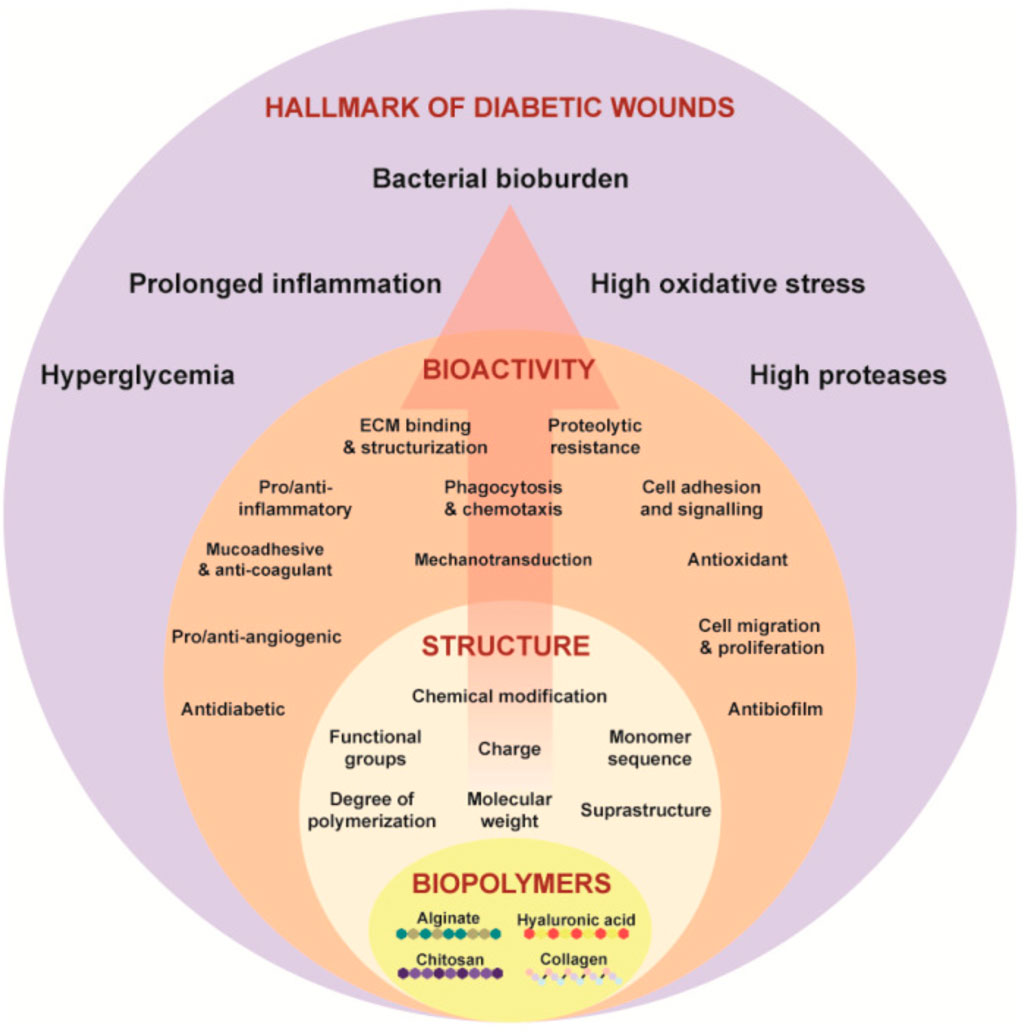
Figure 12. Illustrates the structure–function paradigm as represented in Equation concerning biopolymers such as alginate, chitosan, hyaluronic acid, and collagen, which target the critical features of chronic wounds. Reproduced with permission from Sharma and Kishen (2024).
Moreover, integrating natural compounds into these materials can enhance their biocompatibility and therapeutic efficacy, as demonstrated by the incorporation of honey and plant extracts in wound dressings (Prasathkumar and Sadhasivam, 2021; Yasin et al., 2023). The advancement of nanomaterials also presents novel opportunities for improving biocompatibility and functionality (Barhoum et al., 2022). Research has shown that these materials can enhance cellular responses and tissue integration (Bai et al., 2020). Overall, the creation of biocompatible materials is crucial for developing effective treatments for diabetic wounds, ensuring that they not only promote healing but also seamlessly integrate with the body’s biological systems.
Bioactive molecules play a critical role in the wound healing process, especially for individuals with diabetes, where natural healing mechanisms are often hindered (Moses et al., 2023; Oprita et al., 2023; Sultana et al., 2024). These molecules can be integrated into biomaterials to bolster their therapeutic effects. For instance, growth factors, cytokines, and antimicrobial peptides are currently being studied for their capacity to stimulate essential cellular activities, including migration, proliferation, and angiogenesis (Takahashi et al., 2021; Umehara et al., 2022; Yue et al., 2022). Recent research indicates that the incorporation of bioactive molecules into hydrogels and scaffolds can markedly improve healing outcomes for chronic wounds (Chen et al., 2024b; Rathna and Kulandhaivel, 2024; Yusuf and Adeleke, 2023). Figure 13 provides a schematic representation of various biomaterial dressings. Additionally, studies have highlighted the potential of metal nanoparticles as bioactive agents in diabetic wound therapy, offering antimicrobial properties while facilitating tissue regeneration (Zheng et al., 2024). The development of intelligent biomaterials capable of controlling the release of these bioactive molecules represents a promising research area, enabling targeted delivery and enhanced therapeutic effectiveness (Huang et al., 2023). Therefore, the strategic incorporation of bioactive molecules alongside biomaterials marks a significant advancement in diabetic wound treatment, fostering a more effective healing process.
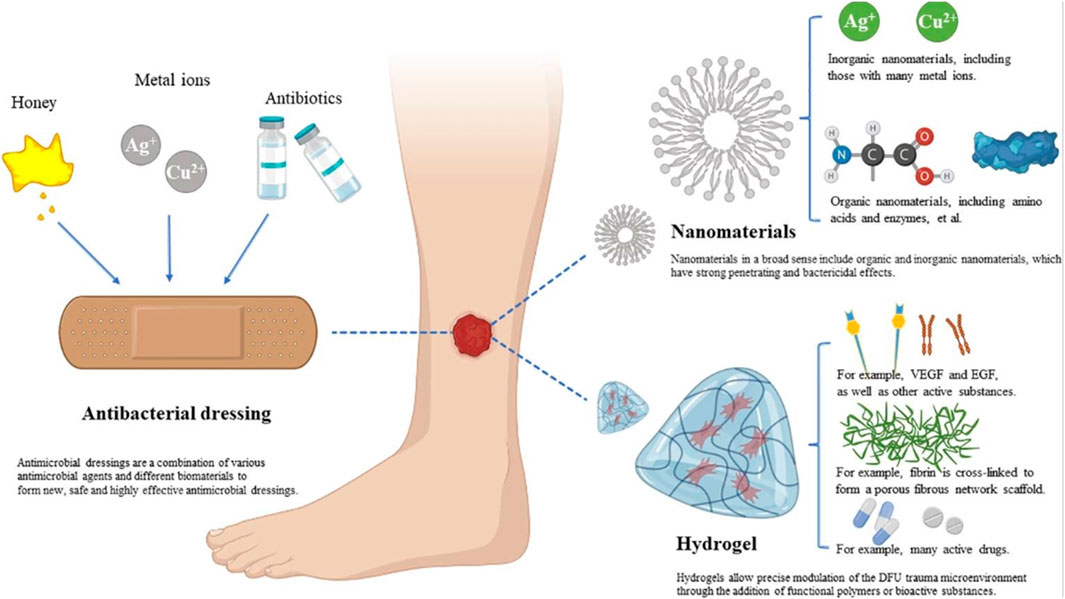
Figure 13. A visual representation illustrating antibacterial dressings, nanodressings, and hydrogel dressings. Reproduced with permission from Jiang et al. (2023).
The utilization of biomaterials in diabetes management represents a promising Frontier with significant potential, particularly in the domains of diabetes treatment and wound healing. Nonetheless, the deployment of biomaterials for diabetes management, especially in the context of wound healing, embodies a dual-edged sword characterized by both benefits and drawbacks.
From a positive perspective, biomaterials such as hydrogels, nanoparticles, and scaffolds present enhanced characteristics that can markedly improve outcomes in wound healing (Fadilah et al., 2022; Leng et al., 2022; Zhang Z. et al., 2024). These biomaterials can be meticulously engineered to facilitate controlled drug release, encourage angiogenesis, and amplify cellular responses, effectively addressing the complex, multifactorial nature of diabetic wounds. The integration of bioactive agents, including growth factors and exosomes derived from stem cells, into these materials has the potential to further stimulate tissue regeneration and enhance healing rates (Jing et al., 2023). Furthermore, biomaterials can be customized to exhibit antibacterial properties, thereby diminishing the risk of infection, a frequent complication associated with diabetic wounds (Zheng et al., 2024).
Conversely, the application of biomaterials is not devoid of challenges. A notable concern is the risk of immune rejection or adverse reactions, particularly in relation to synthetic materials (Tripathi et al., 2023). The biocompatibility of these materials is a critical aspect that necessitates thorough evaluation to prevent complications that could impede rather than promote healing (Zhao et al., 2023). Additionally, the intricate environment of diabetic wounds may hinder the effective performance of biomaterials. Elevated levels of reactive oxygen species (ROS) within diabetic wounds can undermine the efficacy of specific biomaterials, highlighting the need for the development of advanced formulations capable of alleviating oxidative stress (Cai et al., 2023a; He et al., 2023; Yao et al., 2019). Moreover, the cost and accessibility of sophisticated biomaterials may present an obstacle to their wide-scale adoption in clinical settings (Ansari and Darvishi, 2024; Chen and Liu, 2016). While these materials exhibit considerable promise, their incorporation into standard diabetes management requires a meticulous assessment of their long-term effects, potential complications, and overall cost-effectiveness.
In summary, although biomaterials present exciting prospects for improving diabetes management and wound healing, it remains imperative to weigh their benefits against potential drawbacks. Critical factors regarding biocompatibility, safety profiles, and long-term efficacy of biomaterials necessitate further exploration to guarantee their safe integration into clinical practice. Ongoing research and clinical trials will play a vital role in identifying the most effective and safe applications of biomaterials in this context, ultimately striving to enhance patient outcomes in diabetes care.
The safety and efficacy of biomaterials are of paramount concern as their use in clinical applications continues to expand (Kantak and Bharate, 2022). These materials must engage positively with biological systems, facilitating healing while minimizing adverse reactions (Knopf-Marques et al., 2016). The challenge lies in ensuring that these biomaterials do not provoke toxic responses or incite chronic inflammation, which could compromise their intended function. Advances in the understanding of the interaction between biomaterials and the immune system have paved the way for the design of materials capable of favorably modulating immune responses, thereby enhancing their therapeutic potential (Salthouse et al., 2023). Furthermore, the advancement of nanotoxicity evaluations is essential, as nanoparticles employed in biomaterials may pose risks distinct from their bulk forms (Akcan et al., 2020). As this discipline progresses, it is imperative for researchers to prioritize the creation of standardized protocols for assessing the safety of biomaterials to streamline regulatory approval processes and enhance clinical translation (Josyula et al., 2021).
In the last decade, a significant concentration of clinical research on biomaterials has emerged, closely linked to advances in fundamental research. Nonetheless, the findings derived from basic research may not necessarily translate directly to human applications (Socci et al., 2023). As previously noted in this manuscript, the biocompatibility and efficacy of certain established biodegradable biomaterials have been validated through clinical trials (Arrizabalaga and Nollert, 2018). The academic community broadly recognizes the potential for biomaterials to be integrated with agents such as stem cells and bioactive factors (Wilems et al., 2019). However, challenges such as ethical considerations and the variability in source materials hinder seamless clinical translation. Moreover, most animal models utilized in fundamental research are rodents, which, while advantageous due to their availability and established modeling techniques, present a significant limitation: their wound-healing mechanisms differ from those in humans (Nuutila et al., 2016). Several clinical trials have yet to achieve the anticipated outcomes in human subjects, causing stagnation in clinical translation efforts (Shamshad et al., 2023).
Consequently, it is crucial for basic research teams to foster close collaboration with clinical departments. By aligning with genuine clinical needs, they should conduct focused basic research aimed at facilitating clinical translation, thereby identifying safer and more effective biomaterials for application in clinical settings.
The capacity for real-time monitoring and data analysis marks a significant evolution in the domain of biosensors. With the progression of data analytics and machine learning, the interpretation of biosensor data has become increasingly sophisticated, enabling predictive insights and tailored healthcare solutions (Childs et al., 2024; Zhang et al., 2021; Schackart and Yoon, 2021). Real-time health monitoring systems can amalgamate data from various biosensors, offering a holistic view of a patient’s health status (Paganelli et al., 2022; Wu et al., 2023; Li et al., 2021). This integration allows for timely interventions and enhanced management of chronic ailments, including diabetes and cardiovascular conditions. Additionally, the emergence of mobile applications that connect with biosensors empowers patients to conveniently monitor their health metrics, thus promoting greater involvement in their own care (Gecili et al., 2020). The future of biosensors is poised to enhance patient outcomes and healthcare efficiency through the provision of actionable insights derived from real-time data analysis.
The shift towards personalized medicine signifies a groundbreaking approach within healthcare, particularly regarding biomaterials. Individual patients exhibit variability in their financial resources and a range of personal factors. A systematic analysis conducted by Maria et al. revealed no statistically significant differences in HbA1c values among patients with type 1, type 2, or gestational diabetes when utilizing different diabetes monitoring systems (DMS). Future endeavors in personalized medicine will necessitate more extensive research to assess the effectiveness, cost-effectiveness, and comparative efficacy of DMS, allowing for stratification into the most suitable subgroups of diabetic patients (Kamusheva et al., 2021). Table 4 lists some diabetes management systems that have obtained clinical approval. By customizing treatments to individual patient profiles, which include genetic, environmental, and lifestyle factors, healthcare providers can enhance therapeutic outcomes and reduce adverse effects (Kalra et al., 2022). This concept is illustrated in Figure 14. The incorporation of artificial intelligence and machine learning into the analysis of patient data can substantially improve the accuracy of personalized treatment strategies (Clinton and Cross, 2023).
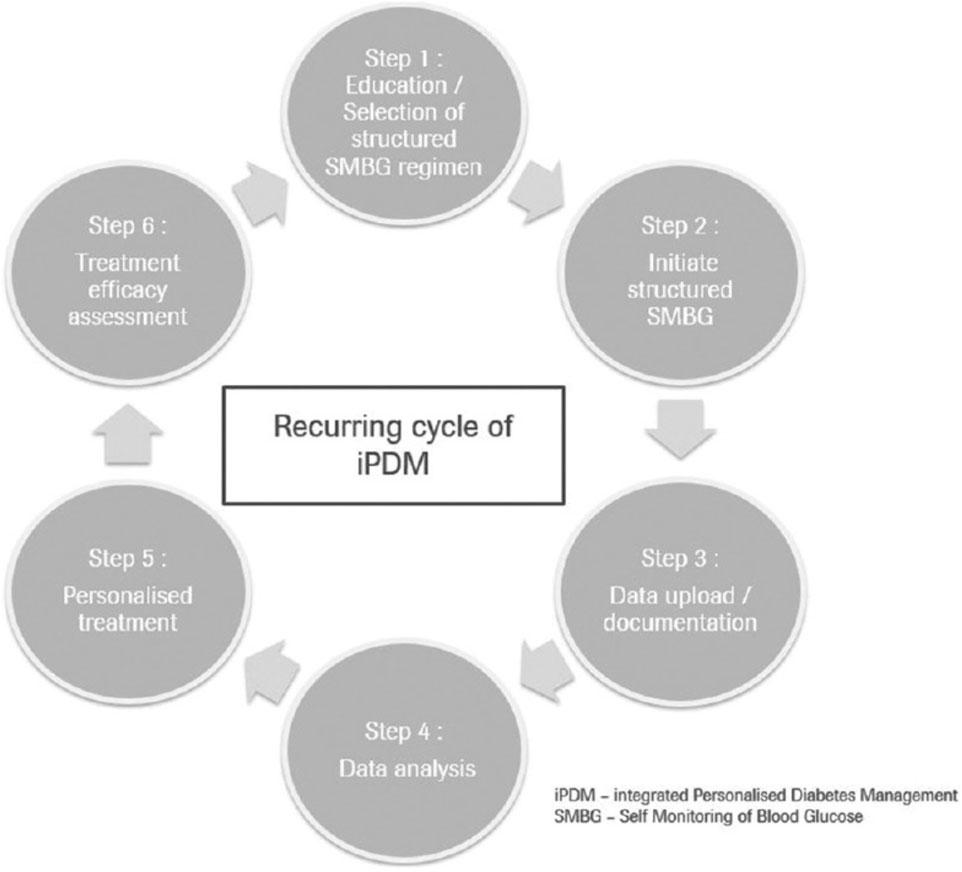
Figure 14. Depicts the Cycle of Integrated Personalized Diabetes Management, which comprises six iterative steps and forms a continuous revolving circle, applicable to each patient over differing timeframes. Reproduced with permission from Kalra et al. (2022).
Nevertheless, challenges still exist in terms of the accessibility of personalized therapies and the need for solid clinical evidence to support their efficacy across diverse populations (Varela-Moreno et al., 2021). Future research must concentrate on developing scalable models for personalized interventions that can be effectively implemented in clinical practice, ensuring that all patients can benefit from these advancements (Chen et al., 2024c; Huckvale et al., 2019; Lydiard and Nemeroff, 2019).
Multidisciplinary collaboration is essential for tackling the intricate challenges associated with the development and execution of biomaterials and personalized treatments. By uniting expertise from various fields, including engineering, biology, medicine, and data science, researchers can foster innovation and accelerate the application of scientific breakthroughs in clinical settings. Effective collaboration not only enhances research quality but also addresses the complex dimensions of health issues, leading to more holistic solutions (Errecaborde et al., 2019). For instance, collaborative initiatives in bioimage analysis have demonstrated the potential to enhance diagnostic precision and treatment planning (Schlaeppi et al., 2022). Furthermore, establishing standards for interprofessional collaboration can improve communication and cooperation among healthcare providers, ultimately leading to better patient outcomes (Bowman et al., 2021). As the healthcare landscape evolves, nurturing a culture of collaboration will be critical in overcoming obstacles and advancing the disciplines of biomaterials and personalized medicine.
The prevalence of diabetes, a prevalent metabolic disorder, is escalating globally. Conventional treatment modalities, such as pharmacological interventions and lifestyle modifications, often fall short of achieving optimal glycemic control due to issues like poor patient adherence and complex treatment protocols. There is an urgent need for innovative approaches.
The integration of multidisciplinary strategies will be vital for advancing biomedical research in the future. By merging perspectives from materials science, biomedical engineering, and clinical medicine, researchers can devise innovative solutions to tackle the multifaceted challenges posed by diabetes. Biomaterials encounter hurdles such as immune rejection, biocompatibility, and high costs in diabetes management applications. It is imperative to synthesize these findings through systematic reviews and meta-analyses, which can elucidate which materials and delivery systems are most likely to yield favorable outcomes for patients. This collaborative effort can facilitate the design of biomaterials that not only enhance insulin delivery and foster tissue regeneration but also prioritize patient safety and comfort.
In the future, it is necessary to strengthen research on the safety and effectiveness of biomaterials and establish standardized evaluation protocols; promote personalized treatment and formulate precise treatment plans according to individual differences of patients; strengthen multidisciplinary cooperation and promote the transformation of biomaterials from laboratory to clinic to improve the treatment effect and quality of life of diabetic patients.
WG: Writing–original draft. LZ: Writing–review and editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abozaid, O., El-Sonbaty, S. M., Hamam, N., Farrag, M. A., and Kodous, A. S. (2023). Chitosan-encapsulated nano-selenium targeting TCF7L2, PPARγ, and CAPN10 genes in diabetic rats. Biol. Trace Elem. Res. 201, 306–323. doi:10.1007/s12011-022-03140-7
Amirouche, F., Zhou, Y., and Johnson, T. (2009). Current micropump technologies and their biomedical applications. Micro. Techno. 15, 647–666. doi:10.1007/s00542-009-0804-7
Abdollahi, A., Aghayan, H.R., Mousivand, Z., Motasadizadeh, H., Maghsoudian, S., Abdorashidi, M., et al. (2024b). Chitosan based extruded nanofibrous bioscaffold for local delivery of mesenchymal stem cells to improve diabetic wound healing. Stem Cell. Res. Ther. 15, 262. doi:10.1186/s13287-024-03772-7
Ahmadian, N., Manickavasagan, A., and Ali, A. (2023). Comparative assessment of blood glucose monitoring techniques: a review. J. Med. Eng. Technol. 47, 121–130. doi:10.1080/03091902.2022.2100496
Akcan, R., Aydogan, H. C., Yildirim, M. S., Tastekin, B., and Saglam, N. (2020). Nanotoxicity: a challenge for future medicine. Turk J. Med. Sci. 50, 1180–1196. doi:10.3906/sag-1912-209
Aldahish, A., Shanmugasundaram, N., Vasudevan, R., Alqahtani, T., Alqahtani, S., Mohammad, A. A., et al. (2024). Silk fibroin nanofibers: advancements in bioactive dressings through electrospinning technology for diabetic wound healing. Pharm. (Basel) 17, 1305. doi:10.3390/ph17101305
Ali, A., Nouseen, S., Saroj, S., Shegane, M., Majumder, P., Puri, A., et al. (2022). Repurposing pinacol esters of boronic acids for tuning viscoelastic properties of glucose-responsive polymer hydrogels: effects on insulin release kinetics. J. Mater Chem. B 10, 7591–7599. doi:10.1039/d2tb00603k
Ali, A., Saroj, S., Saha, S., Gupta, S. K., Rakshit, T., and Pal, S. (2023). Glucose-responsive chitosan nanoparticle/poly (vinyl alcohol) hydrogels for sustained insulin release in vivo. Acs Appl. Mater Interfaces 15, 32240–32250. doi:10.1021/acsami.3c05031
Ali, H. S., York, P., and Blagden, N. (2009). Preparation of hydrocortisone nanosuspension through a bottom-up nanoprecipitation technique using microfluidic reactors. Int. J. Pharm. 375, 107–113. doi:10.1016/j.ijpharm.2009.03.029
Ali, Y., and Lehmussaari, K. (2006). Industrial perspective in ocular drug delivery. Adv. Drug Deliv. Rev. 58, 1258–1268. doi:10.1016/j.addr.2006.07.022
Al-Qerem, W., Jarab, A. S., Badinjki, M., Hammad, A., Ling, J., and Alasmari, F. (2022). Factors associated with glycemic control among patients with type 2 diabetes: a cross-sectional study. Eur. Rev. Med. Pharmacol. Sci. 26, 2415–2421. doi:10.26355/eurrev_202204_28475
Alven, S., Nqoro, X., and Aderibigbe, B. A. (2020). Polymer-based materials loaded with curcumin for wound healing applications. Polym. (Basel) 12, 2286. doi:10.3390/polym12102286
American Diabetes Association (2014). Diagnosis and classification of diabetes mellitus. Diabetes Care 37 (Suppl. 1), S81–S90. doi:10.2337/dc14-S081
Andrews, K. L., Houdek, M. T., and Kiemele, L. J. (2015). Wound management of chronic diabetic foot ulcers: from the basics to regenerative medicine. Prosthet. Orthot. Int. 39, 29–39. doi:10.1177/0309364614534296
Annicchiarico, A., Barile, B., Buccoliero, C., Nicchia, G. P., and Brunetti, G. (2024). Alternative therapeutic strategies in diabetes management. World J. Diabetes 15, 1142–1161. doi:10.4239/wjd.v15.i6.1142
Ansari, H. R., Kordrostami, Z., Mirzaei, A., and Kraft, M. (2024). Deep-learning-based blood glucose detection device using acetone exhaled breath sensing features of α-Fe2O3-MWCNT nanocomposites. Acs Appl. Mater Interfaces 16, 47973–47987. doi:10.1021/acsami.4c06855
Ansari, M., and Darvishi, A. (2024). A review of the current state of natural biomaterials in wound healing applications. Front. Bioeng. Biotechnol. 12, 1309541. doi:10.3389/fbioe.2024.1309541
Arrizabalaga, J. H., and Nollert, M. U. (2018). Human amniotic membrane: a versatile scaffold for tissue engineering. Acs Biomater. Sci. Eng. 4, 2226–2236. doi:10.1021/acsbiomaterials.8b00015
Arun, S., Sykes, E. R., and Tanbeer, S. (2024). Remotehealthconnect: innovating patient monitoring with wearable technology and custom visualization. Digit. Health 10, 20552076241300748. doi:10.1177/20552076241300748
Asadi, M. R., and Torkaman, G. (2014). Bacterial inhibition by electrical stimulation. Adv. Wound Care (New Rochelle) 3, 91–97. doi:10.1089/wound.2012.0410
Azad, A., Altunbas, H. A., and Manguoglu, A. E. (2024). From islet transplantation to beta-cell regeneration: an update on beta-cell-based therapeutic approaches in type 1 diabetes. Expert Rev. Endocrinol. Metab. 19, 217–227. doi:10.1080/17446651.2024.2347263
Baek, J. S., So, J. W., Shin, S. C., and Cho, C. W. (2012). Solid lipid nanoparticles of paclitaxel strengthened by hydroxypropyl-β-cyclodextrin as an oral delivery system. Int. J. Mol. Med. 30, 953–959. doi:10.3892/ijmm.2012.1086
Baron, N., Passave, J., Guichardaz, B., and Cabodevila, G. (2008). Investigations of development process of high hollow beveled microneedles using a combination of icp rie and dicing saw. Micro. Techno. 14, 1475–1480. doi:10.1007/s00542-008-0596-1
Bagheri, B., Zarrintaj, P., Samadi, A., Zarrintaj, R., Ganjali, M. R., Saeb, M. R., et al. (2020). Tissue engineering with electrospun electro-responsive chitosan-aniline oligomer/polyvinyl alcohol. Int. J. Biol. Macromol. 147, 160–169. doi:10.1016/j.ijbiomac.2019.12.264
Bai, Q., Han, K., Dong, K., Zheng, C., Zhang, Y., Long, Q., et al. (2020). Potential applications of nanomaterials and technology for diabetic wound healing. Int. J. Nanomedicine 15, 9717–9743. doi:10.2147/IJN.S276001
Barhoum, A., Garcia-Betancourt, M. L., Jeevanandam, J., Hussien, E. A., Mekkawy, S. A., Mostafa, M., et al. (2022). Review on natural, incidental, bioinspired, and engineered nanomaterials: history, definitions, classifications, synthesis, properties, market, toxicities, risks, and regulations. Nanomater. (Basel) 12, 177. doi:10.3390/nano12020177
Bent, B., Wang, K., Grzesiak, E., Jiang, C., Qi, Y., Jiang, Y., et al. (2020). The digital biomarker discovery pipeline: an open-source software platform for the development of digital biomarkers using mhealth and wearables data. J. Clin. Transl. Sci. 5, e19. doi:10.1017/cts.2020.511
Bhatti, P. K., Ghay, R., Singh, N. G., Thaman, V., and Narang, S. (2024). Estimation of hba1c levels in transfusion-dependent thalassemia patients in comparison with normal healthy individuals. Cureus 16, e73236. doi:10.7759/cureus.73236
Bigham, A., Zarepour, A., Khosravi, A., Iravani, S., and Zarrabi, A. (2025). Microneedle patches: a new vantage point for diabetic wound treatments. Biomater. Sci. 13, 379–407. doi:10.1039/d4bm01229a
Blagden, N., de Matas, M., Gavan, P. T., and York, P. (2007). Crystal engineering of active pharmaceutical ingredients to improve solubility and dissolution rates. Adv. Drug Deliv. Rev. 59, 617–630. doi:10.1016/j.addr.2007.05.011
Boucheta, H., Zouaoui, E., Ferkous, H., Madaci, A., Yadav, K. K., and Benguerba, Y. (2024). Advancing diabetes management: the future of enzyme-less nanoparticle-based glucose sensors-a review. J. Diabetes Sci. Technol. 570845427, 19322968241236211. doi:10.1177/19322968241236211
Boushra, M., Tous, S., Fetih, G., Xue, H. Y., Tran, N. T., and Wong, H. L. (2016). Methocel-lipid hybrid nanocarrier for efficient oral insulin delivery. J. Pharm. Sci. 105, 1733–1740. doi:10.1016/j.xphs.2016.02.018
Bowman, K. S., Suarez, V. D., and Weiss, M. J. (2021). Standards for interprofessional collaboration in the treatment of individuals with autism. Behav. Anal. Pract. 14, 1191–1208. doi:10.1007/s40617-021-00560-0
Burge, M. R. (2001). Lack of compliance with home blood glucose monitoring predicts hospitalization in diabetes. Diabetes Care 24, 1502–1503. doi:10.2337/diacare.24.8.1502
Cai, F., Chen, W., Zhao, R., and Liu, Y. (2023a). Mechanisms of Nrf2 and NF-κB pathways in diabetic wound and potential treatment strategies. Mol. Biol. Rep. 50, 5355–5367. doi:10.1007/s11033-023-08392-7
Cai, H., and Li, G. (2020). Efficacy of alginate-and chitosan-based scaffolds on the healing of diabetic skin wounds in animal experimental models and cell studies: a systematic review. Wound Repair Regen. 28, 751–771. doi:10.1111/wrr.12857
Cai, Y., Xu, X., Wu, M., Liu, J., Feng, J., and Zhang, J. (2023b). Multifunctional zwitterionic microneedle dressings for accelerated healing of chronic infected wounds in diabetic rat models. Biomater. Sci. 11, 2750–2758. doi:10.1039/d2bm02101c
Cano-Ibanez, N., and Bueno-Cavanillas, A. (2024). Lifestyle interventions in an aged population: challenges and opportunities from a public health perspective. Nutrients 16, 173. doi:10.3390/nu16010173
Carvalho, A. M., Nunes, R., and Sarmento, B. (2022). From pluripotent stem cells to bioengineered islets: a challenging journey to diabetes treatment. Eur. J. Pharm. Sci. 172, 106148. doi:10.1016/j.ejps.2022.106148
Chakraborty, C., Bhattacharya, M., and Lee, S. S. (2023). Currentstatus of microneedle array technology for therapeutic delivery: from bench to clinic. Mol. Biotechnol. 66, 3415–3437. doi:10.1007/s12033-023-00961-2
Chan, D., Maikawa, C. L., D'Aquino, A. I., Raghavan, S. S., Troxell, M. L., and Appel, E. A. (2023). Polyacrylamide-based hydrogel coatings improve biocompatibility of implanted pump devices. J. Biomed. Mater Res. A 111, 910–920. doi:10.1002/jbm.a.37521
Chaudhury, A., Duvoor, C., Reddy, D. V., Kraleti, S., Chada, A., Ravilla, R., et al. (2017). Clinical review of antidiabetic drugs: implications for type 2 diabetes mellitus management. Front. Endocrinol. (Lausanne) 8, 6. doi:10.3389/fendo.2017.00006
Chen, A. C., Lu, Y., Hsieh, C. Y., Chen, Y. S., Chang, K. C., and Chang, D. H. (2024a). Advanced biomaterials and topical medications for treating diabetic foot ulcers: a systematic review and network meta-analysis. Adv. Wound Care (New Rochelle) 13, 97–113. doi:10.1089/wound.2023.0024
Ciatti, C., Maniscalco, P., Bosio, S., Puma, P.C., Bianchi, G., and Quattrini, F. (2024). Pseudotumor from ceramic-on-ceramic total hip arthroplasty. Int. J. Surg. Case. Rep. 116, 109374. doi:10.1016/j.ijscr.2024.109374
Chen, X., Li, X., Xiao, X., Long, R., Chen, B., Lin, Y., et al. (2024d). Photothermal and antibacterial pda@ag/serma microneedles for promoting diabetic wound repair. Acs. Appl. Bio. Mater. 7, 6603–6616. doi:10.1021/acsabm.4c00793
Chen, F., Wu, P., Zhang, H., and Sun, G. (2024b). Signaling pathways triggering therapeutic hydrogels in promoting chronic wound healing. Macromol. Biosci. 24, e2300217. doi:10.1002/mabi.202300217
Chen, F. M., and Liu, X. (2016). Advancing biomaterials of human origin for tissue engineering. Prog. Polym. Sci. 53, 86–168. doi:10.1016/j.progpolymsci.2015.02.004
Chen, L., Sleeman, K. E., Bradshaw, A., Sakharang, W., Mo, Y., and Ellis-Smith, C. (2024c). The use of person-centered outcome measures to support integrated palliative care for older people: a systematic review. J. Am. Med. Dir. Assoc. 25, 105036. doi:10.1016/j.jamda.2024.105036
Chen, Q., Bao, K., Wang, X., and Chen, J. (2018). Recombinant fusion protein of lysozyme-antibacterial peptide for diabetic hand:clinical experience in one case. Chin. General Pract. 5, 620–622. doi:10.3969/j.issn.1007-9572.2017.00.211
Chen, S., Matsumoto, H., Moro-Oka, Y., Tanaka, M., Miyahara, Y., Suganami, T., et al. (2019). Smart microneedle fabricated with silk fibroin combined semi-interpenetrating network hydrogel for glucose-responsive insulin delivery. Acs Biomater. Sci. Eng. 5, 5781–5789. doi:10.1021/acsbiomaterials.9b00532
Chen, X., Li, X., Xiao, X., Long, R., Chen, B., Lin, Y., et al. (2024d). Photothermal and antibacterial pda@ag/serma microneedles for promoting diabetic wound repair. Acs Appl. Bio Mater 7, 6603–6616. doi:10.1021/acsabm.4c00793
Chen, X., Shen, J., Liu, C., Shi, X., Feng, W., Sun, H., et al. (2024e). Applications of data characteristic ai-assisted Raman spectroscopy in pathological classification. Anal. Chem. 96, 6158–6169. doi:10.1021/acs.analchem.3c04930
Chen, Y., Xie, T., Ye, M., Lai, Q., Wang, Y., Xu, Y., et al. (2021). Combination of pathological and spectroscopic characterization to promote diagnosis of retinal pigment epithelium-bruch's membrane complex in a diabetic rat model. Biomed. Opt. Express 12, 2221–2235. doi:10.1364/BOE.419716
Cheng, H., Guo, S., Cui, Z., Zhang, X., Huo, Y., Guan, J., et al. (2021). Design of folic acid decorated virus-mimicking nanoparticles for enhanced oral insulin delivery. Int. J. Pharm. 596, 120297. doi:10.1016/j.ijpharm.2021.120297
Chinese Society of Endocrinology, China Endocrinology and Metabolism Specialist Alliance (2024). Expert consensus on wound treatment of diabetic foot ulcer. Chin. J. Endocrinol. Metabolism 7, 565–569. doi:10.3760/cma.j.cn311282-20240625-00281
Chu, J., Shi, P., Yan, W., Fu, J., Yang, Z., He, C., et al. (2018). Pegylated graphene oxide-mediated quercetin-modified collagen hybrid scaffold for enhancement of mscs differentiation potential and diabetic wound healing. Nanoscale 10, 9547–9560. doi:10.1039/c8nr02538j
Childs, A., Mayol, B., Lasalde-Ramirez, J.A., Song, Y., Sempionatto, J.R., and Gao, W. (2024). Diving into sweat: advances, challenges, and future directions in wearable sweat sensing. Acs. Nano. 18, 24605–24616. doi:10.1021/acsnano.4c10344
Clinton, J. W., and Cross, R. K. (2023). Personalized treatment for crohn's disease: current approaches and future directions. Clin. Exp. Gastroenterol. 16, 249–276. doi:10.2147/CEG.S360248
Condren, M., Sabet, S., Chalmers, L. J., Saley, T., and Hopwood, J. (2019). Technology for augmenting type 1 diabetes mellitus management. J. Pediatr. Pharmacol. Ther. 24, 99–106. doi:10.5863/1551-6776-24.2.99
Dariya, B., Chalikonda, G., Srivani, G., Alam, A., and Nagaraju, G. P. (2019). Pathophysiology, etiology, epidemiology of type 1 diabetes and computational approaches for immune targets and therapy. Crit. Rev. Immunol. 39, 239–265. doi:10.1615/CritRevImmunol.2019033126
Das, P., Manna, S., Roy, S., Nandi, S. K., and Basak, P. (2023). Polymeric biomaterials-based tissue engineering for wound healing: a systemic review. Burns Trauma 11, tkac058. doi:10.1093/burnst/tkac058
Defronzo, R. A. (2009). From the triumvirate to the ominous octet: a new paradigm for the treatment of type 2 diabetes mellitus. Diabetes 58, 773–795. doi:10.2337/db09-9028
Deng, Z., Davis, J., Muniz-Rodriguez, F., and Richardson, F. (2018). Successful management of poorly controlled type 2 diabetes with multidisciplinary neurobehavioral rehabilitation: a case report and review. Diabetes Ther. 9, 1713–1718. doi:10.1007/s13300-018-0448-7
Deng, X., Gould, M., and Ali, M. A. (2022). A review of current advancements for wound healing: biomaterial applications and medical devices. J Biomed Mater Res B Appl Biomater 110, 2542–2573. doi:10.1002/jbm.b.35086
Dervisevic, M., Alba, M., Esser, L., Tabassum, N., Prieto-Simon, B., and Voelcker, N. H. (2022). Silicon micropillar array-based wearable sweat glucose sensor. Acs Appl. Mater Interfaces 14, 2401–2410. doi:10.1021/acsami.1c22383
Desai, A., Tankasala, D., Ng, G. P., Thakkar, P., Hoilett, O. S., Mather, K. J., et al. (2025). Selective collection of exhaled breath condensate for noninvasive screening of breath glucose. J. Diabetes Sci. Technol. 19, 161–164. doi:10.1177/19322968231179728
Du, Y., Zhang, X., Liu, P., Yu, D. G., and Ge, R. (2022). Electrospun nanofiber-based glucose sensors for glucose detection. Front. Chem. 10, 944428. doi:10.3389/fchem.2022.944428
Dua, A., Debnath, A., Kumar, K., Mazumder, R., Mazumder, A., Singh, R., et al. (2024). Advancements of glucose monitoring biosensor: current state, generations of technological progress, and innovation dynamics. Curr. Pharm. Biotechnol. 25. doi:10.2174/0113892010305386240625072535
Emad, N. A., Zai, I., Ahmad, S., Pandit, J., Khan, M. A., and Sultana, Y. (2024). Role of polyphenols, their nano-formulations, and biomaterials in diabetic wound healing. Endocr. Metab. Immune Disord. Drug Targets 24, 626–641. doi:10.2174/0118715303242310230927104709
Ernst, A.U., Wang, L.H., and Ma, M. (2019). Interconnected toroidal hydrogels for islet encapsulation. Adv Healthc Mater 8, e1900423. doi:10.1002/adhm.201900423
Errecaborde, K. M., Rist, C., Travis, D. A., Ragan, V., Potter, T., Pekol, A., et al. (2019). Evaluating one health: the role of team science in multisectoral collaboration. Rev. Sci. Tech. 38, 279–289. doi:10.20506/rst.38.1.2960
Esmaeili, A., Biazar, E., Ebrahimi, M., Heidari, K. S., Kheilnezhad, B., and Saeedi, L. F. (2023). Acellular fish skin for wound healing. Int. Wound J. 20, 2924–2941. doi:10.1111/iwj.14158
Esteves, C., Palma, S., Costa, H., Alves, C., Santos, G., Ramou, E., et al. (2022). Tackling humidity with designer ionic liquid-based gas sensing soft materials. Adv. Mater 34, e2107205. doi:10.1002/adma.202107205
Fadilah, N., Isa, I., Zaman, W., Tabata, Y., and Fauzi, M. B. (2022). The effect of nanoparticle-incorporated natural-based biomaterials towards cells on activated pathways: a systematic review. Polym. (Basel) 14, 476. doi:10.3390/polym14030476
Fahimirad, S., and Ajalloueian, F. (2019). Naturally-derived electrospun wound dressings for target delivery of bio-active agents. Int. J. Pharm. 566, 307–328. doi:10.1016/j.ijpharm.2019.05.053
Fan, W., Yang, X., Hu, X., Huang, R., Shi, H., and Liu, G. (2024). A novel conductive microtubule hydrogel for electrical stimulation of chronic wounds based on biological electrical wires. J. Nanobiotechnology 22, 258. doi:10.1186/s12951-024-02524-2
Firoozbahr, M., Kingshott, P., Palombo, E. A., and Zaferanloo, B. (2023). Recent advances in using natural antibacterial additives in bioactive wound dressings. Pharmaceutics 15, 644. doi:10.3390/pharmaceutics15020644
Flores-Guerrero, J. L., Munoz-Morales, A., Narea-Jimenez, F., Perez-Fuentes, R., Torres-Rasgado, E., Ruiz-Vivanco, G., et al. (2020). Novel assessment of urinary albumin excretion in type 2 diabetes patients by Raman spectroscopy. Diagn. (Basel) 10, 141. doi:10.3390/diagnostics10030141
Gupta, J., Felner, E. I., and Prausnitz, M. R. (2009). Minimally invasive insulin delivery in subjects with type 1 diabetes using hollow microneedles. Diabetes Technol Ther 11, 329–337. doi:10.1089/dia.2008.0103
Gecili, E., Huang, R., Khoury, J.C., King, E., Altaye, M., Bowers, K., et al. (2020). Functional data analysis and prediction tools for continuous glucose-monitoring studies. J. Clin. Transl. Sci. 5, e51. doi:10.1017/cts.2020.545
Galassetti, P. R., Novak, B., Nemet, D., Rose-Gottron, C., Cooper, D. M., Meinardi, S., et al. (2005). Breath ethanol and acetone as indicators of serum glucose levels: an initial report. Diabetes Technol. Ther. 7, 115–123. doi:10.1089/dia.2005.7.115
Ghasemi, A., and Norouzirad, R. (2019). Type 2 diabetes: an updated overview. Crit. Rev. Oncog. 24, 213–222. doi:10.1615/CritRevOncog.2019030976
Guo, L., Guan, N., Miao, W., Zhao, W., and Li, Q. (2022b). An electrospun scaffold loaded with an enteromorpha polysaccharide for accelerated wound healing in diabetic mice. Mar. Drugs. 20, 95. doi:10.3390/md20020095
Goonoo, N. (2022). Tunable biomaterials for myocardial tissue regeneration: promising new strategies for advanced biointerface control and improved therapeutic outcomes. Biomater. Sci. 10, 1626–1646. doi:10.1039/d1bm01641e
Govindaraj, M., Srivastava, A., Muthukumaran, M. K., Tsai, P. C., Lin, Y. C., Raja, B. K., et al. (2023). Current advancements and prospects of enzymatic and non-enzymatic electrochemical glucose sensors. Int. J. Biol. Macromol. 253, 126680. doi:10.1016/j.ijbiomac.2023.126680
Guan, Y., Li, X., Yang, H., Yang, Y., Du, Z., Hua, Z., et al. (2025). Pore-edge high active sites of 2d wo (3) nanosheets enhancing acetone sensing performance. Talanta 282, 127003. doi:10.1016/j.talanta.2024.127003
Han, H. H., Kim, S. K., Kim, S. J., Choi, I., Mok, J. W., Joo, C. K., et al. (2023). Long-term stable wireless smart contact lens for robust digital diabetes diagnosis. Biomaterials 302, 122315. doi:10.1016/j.biomaterials.2023.122315
Hao, L., Khajouei, F., Rodriguez, J., Kim, S., and Lee, E. (2024a). Unlocking the promise of decellularized pancreatic tissue: a novel approach to support angiogenesis in engineered tissue. Bioeng. (Basel) 11, 183. doi:10.3390/bioengineering11020183
Hao, X., Yu, T., Meng, X., Wei, C., Wang, Y., Sun, S., et al. (2024b). Efficient mixed-potential acetone sensor with yttria-stabilized zirconia and porous co (3)o(4) nanofoam sensing electrode for hazardous gas monitoring and breath analysis. J. Hazard Mater 478, 135462. doi:10.1016/j.jhazmat.2024.135462
He, Y., Liu, K., Guo, S., Chang, R., Zhang, C., Guan, F., et al. (2023). Multifunctional hydrogel with reactive oxygen species scavenging and photothermal antibacterial activity accelerates infected diabetic wound healing. Acta Biomater. 155, 199–217. doi:10.1016/j.actbio.2022.11.023
Hina, A., and Saadeh, W. (2020). A noninvasive glucose monitoring soc based on single wavelength photoplethysmography. Ieee Trans. Biomed. Circuits Syst. 14, 504–515. doi:10.1109/TBCAS.2020.2979514
Heidari, F., Yari, A., Teimourian, S., Joulai, V.S., and Nobakht, M. (2023). Effects of hair follicle stem cells coupled with polycaprolactone scaffold on cutaneous wound healing in diabetic male rats. J. Surg. Res. 281, 200–213. doi:10.1016/j.jss.2022.08.008
Hong, C., Chung, H., Lee, G., Kim, C., Kim, D., Oh, S. J., et al. (2023). Hydrogel/nanofiber composite wound dressing optimized for skin layer regeneration through the mechanotransduction-based microcellular environment. Acs Appl. Bio Mater 6, 1774–1786. doi:10.1021/acsabm.3c00014
Hong, Y., Yu, H., Wang, L., Chen, X., Huang, Y., Yang, J., et al. (2022). Transdermal insulin delivery and microneedles-based minimally invasive delivery systems. Curr. Pharm. Des. 28, 3175–3193. doi:10.2174/1381612828666220608130056
Hu, T., Fang, J., Shen, Y., Li, M., Wang, B., Xu, Z., et al. (2024a). Advances of naturally derived biomedical polymers in tissue engineering. Front. Chem. 12, 1469183. doi:10.3389/fchem.2024.1469183
Hu, Y., Xiong, Y., Tao, R., Xue, H., Chen, L., Lin, Z., et al. (2022). Advances and perspective on animal models and hydrogel biomaterials for diabetic wound healing. Biomater. Transl. 3, 188–200. doi:10.12336/biomatertransl.2022.03.003
Hu, Y. W., Wang, Y. H., Yang, F., Liu, D. X., Lu, G. H., Li, S. T., et al. (2024b). Flexible organic photovoltaic-powered hydrogel bioelectronic dressing with biomimetic electrical stimulation for healing infected diabetic wounds. Adv. Sci. (Weinh) 11, e2307746. doi:10.1002/advs.202307746
Huang, C., Yuan, W., Chen, J., Wu, L. P., and You, T. (2023). Construction of smart biomaterials for promoting diabetic wound healing. Molecules 28, 1110. doi:10.3390/molecules28031110
Huckvale, K., Venkatesh, S., and Christensen, H. (2019). Toward clinical digital phenotyping: a timely opportunity to consider purpose, quality, and safety. Npj Digit. Med. 2, 88. doi:10.1038/s41746-019-0166-1
Hwang, S. I., Chen, H. Y., Fenk, C., Rothfuss, M. A., Bocan, K. N., Franconi, N. G., et al. (2021). Breath acetone sensing based on single-walled carbon nanotube-titanium dioxide hybrids enabled by a custom-built dehumidifier. Acs Sens. 6, 871–880. doi:10.1021/acssensors.0c01973
Iitani, K., Ishizuki, N., Matsuhashi, Y., Yokota, K., Ichikawa, K., Toma, K., et al. (2024). Biofluorometric acetone gas sensor of sub-ppbv level sensitivity. Anal. Chem. 96, 20197–20203. doi:10.1021/acs.analchem.4c03816
Iqbal, S., Sohail, M., Fang, S., Ding, J., Shen, L., Chen, M., et al. (2023). Biomaterials evolution: from inert to instructive. Biomater. Sci. 11, 6109–6115. doi:10.1039/d3bm00322a
Jagannathan, M., Dhinasekaran, D., Rajendran, A. R., and Cho, S. (2023). A review of electroactive nanomaterials in the detection of nitrogen-containing organic compounds and future applications. Biosens. (Basel) 13, 989. doi:10.3390/bios13110989
Jarosinski, M. A., Dhayalan, B., Rege, N., Chatterjee, D., and Weiss, M. A. (2021). 'smart' insulin-delivery technologies and intrinsic glucose-responsive insulin analogues. Diabetologia 64, 1016–1029. doi:10.1007/s00125-021-05422-6
Jeong, G. J., Khan, S., Tabassum, N., Khan, F., and Kim, Y. M. (2022). Marine-bioinspired nanoparticles as potential drugs for multiple biological roles. Mar. Drugs 20, 527. doi:10.3390/md20080527
Jiang, L., Shen, Y., Liu, Y., Zhang, L., and Jiang, W. (2022). Making human pancreatic islet organoids: progresses on the cell origins, biomaterials and three-dimensional technologies. Theranostics 12, 1537–1556. doi:10.7150/thno.66670
Jiang, L., Wang, C., Fan, T., Lv, S., Pan, S., Sun, P., et al. (2024). Mixed potential type acetone sensor with ultralow detection limit for diabetic ketosis breath analysis. Acs Sens. 9, 464–473. doi:10.1021/acssensors.3c02253
Jiang, P., Li, Q., Luo, Y., Luo, F., Che, Q., Lu, Z., et al. (2023). Currentstatus and progress in research on dressing management for diabetic foot ulcer. Front. Endocrinol. (Lausanne) 14, 1221705. doi:10.3389/fendo.2023.1221705
Jin, X., Zhu, D. D., Chen, B. Z., Ashfaq, M., and Guo, X. D. (2018). Insulin delivery systems combined with microneedle technology. Adv. Drug Deliv. Rev. 127, 119–137. doi:10.1016/j.addr.2018.03.011
Jing, S., Li, H., and Xu, H. (2023). Mesenchymal stem cell derived exosomes therapy in diabetic wound repair. Int. J. Nanomedicine 18, 2707–2720. doi:10.2147/IJN.S411562
Josyula, A., Parikh, K. S., Pitha, I., and Ensign, L. M. (2021). Engineering biomaterials to prevent post-operative infection and fibrosis. Drug Deliv. Transl. Res. 11, 1675–1688. doi:10.1007/s13346-021-00955-0
K, S. K., Jothimani, D., Manoharan, M., Rela, M., Selvaraj, R., Seshadri, S., et al. (2025). Quartz-enhanced photoacoustic spectroscopy-based acetone and ammonia measurements from human breath near 8 μm wavelength band. Acs. Sens. doi:10.1021/acssensors.4c02336
Kalidoss, R., and Umapathy, S. (2019). An overview on the exponential growth of non-invasive diagnosis of diabetes mellitus from exhaled breath by nanostructured metal oxide chemi-resistive gas sensors and mu-preconcentrator. Biomed. Microdevices 22, 2. doi:10.1007/s10544-019-0448-z
Kalra, S., Bantwal, G., Sahay, R. K., Bhattacharya, S., Baruah, M. P., Sheikh, S., et al. (2022). Incorporating integrated personalised diabetes management (ipdm) in treatment strategy: a pragmatic approach. Indian J. Endocrinol. Metab. 26, 106–110. doi:10.4103/ijem.ijem_478_21
Kzhyshkowska, J., Gudima, A., Riabov, V., Dollinger, C., Lavalle, P., and Vrana, N.E. (2015). Macrophage responses to implants: prospects for personalized medicine. J. Leukoc. Biol. 98, 953–962. doi:10.1189/jlb.5VMR0415-166R
Kamusheva, M., Tachkov, K., Dimitrova, M., Mitkova, Z., Garcia-Saez, G., Hernando, M. E., et al. (2021). A systematic review of collective evidences investigating the effect of diabetes monitoring systems and their application in health care. Front. Endocrinol. (Lausanne) 12, 636959. doi:10.3389/fendo.2021.636959
Kantak, M. N., and Bharate, S. S. (2022). Analysis of clinical trials on biomaterial and therapeutic applications of chitosan: a review. Carbohydr. Polym. 278, 118999. doi:10.1016/j.carbpol.2021.118999
Karimi, M., Eslami, M., Sahandi-Zangabad, P., Mirab, F., Farajisafiloo, N., Shafaei, Z., et al. (2016). Ph-sensitive stimulus-responsive nanocarriers for targeted delivery of therapeutic agents. Wiley Interdiscip. Rev. Nanomed Nanobiotechnol 8, 696–716. doi:10.1002/wnan.1389
Kasputis, T., Clough, D., Noto, F., Rychel, K., Dye, B., and Shea, L. D. (2018). Microporous polymer scaffolds for the transplantation of embryonic stem cell derived pancreatic progenitors to a clinically translatable site for the treatment of type i diabetes. Acs Biomater. Sci. Eng. 4, 1770–1778. doi:10.1021/acsbiomaterials.7b00912
Kassem, M., Ali, A., Assal, M., and Badrawy, A. (2017). Formulation, characterization and in vivo application of oral insulin nanotechnology using different biodegradable polymers: advanced drug delivery system. Int. J. Adv. Res. (Indore) 5, 1028–1044. doi:10.21474/IJAR01/5863
Kaviani, M., and Azarpira, N. (2016). Insight into microenvironment remodeling in pancreatic endocrine tissue engineering: biological and biomaterial approaches. Tissue Eng. Regen. Med. 13, 475–484. doi:10.1007/s13770-016-0014-1
Khaiz, Y., Al, I. N., Bakkali, M., and Ahid, S. (2025). Association of the immunity genes with type 1 diabetes mellitus. Curr. Diabetes Rev. 21, 38–46. doi:10.2174/0115733998275617231218101116
Kim, J., Campbell, A. S., de Avila, B. E., and Wang, J. (2019). Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 37, 389–406. doi:10.1038/s41587-019-0045-y
Kim, Y. C., Park, J. H., and Prausnitz, M. R. (2012). Microneedles for drug and vaccine delivery. Adv. Drug Deliv. Rev. 64, 1547–1568. doi:10.1016/j.addr.2012.04.005
Klangjareonchai, T., Eguchi, N., Tantisattamo, E., Ferrey, A. J., Reddy, U., Dafoe, D. C., et al. (2021). Current pharmacological intervention and medical management for diabetic kidney transplant recipients. Pharmaceutics 13, 413. doi:10.3390/pharmaceutics13030413
Knopf-Marques, H., Pravda, M., Wolfova, L., Velebny, V., Schaaf, P., Vrana, N. E., et al. (2016). Hyaluronic acid and its derivatives in coating and delivery systems: applications in tissue engineering, regenerative medicine and immunomodulation. Adv. Healthc. Mater 5, 2841–2855. doi:10.1002/adhm.201600316
Koh, W. L., Foster, T., Mooranian, A., and Al-Salami, H. (2022). Applications of bile acids as biomaterials-based modulators, in biomedical science and microfluidics. Ther. Deliv. 13, 591–604. doi:10.4155/tde-2022-0044
Kong, C., Guo, Z., Teng, T., Yao, Q., Yu, J., Wang, M., et al. (2024). Electroactive nanomaterials for the prevention and treatment of heart failure: from materials and mechanisms to applications. Small 20, e2406206. doi:10.1002/smll.202406206
Krauland, A. H., and Alonso, M. J. (2007). Chitosan/cyclodextrin nanoparticles as macromolecular drug delivery system. Int. J. Pharm. 340, 134–142. doi:10.1016/j.ijpharm.2007.03.005
Larijani, G., Parivar, K., Hayati, R. N., Yaghmaei, P., and Amini, N. (2024). Fortified electrospun collagen utilizing biocompatible poly glycerol sebacate prepolymer (pgsp) and zink oxide nanoparticles (zno nps) for diabetics wound healing: physical, biological and animal studies. Regen. Ther. 26, 102–113. doi:10.1016/j.reth.2024.05.009
Latham, J. (2019). The artificial pancreas: what school nurses need to know. Nasn Sch. Nurse 34, 86–89. doi:10.1177/1942602X18804491
Leng, T., Wang, Y., Cheng, W., Wang, W., Qu, X., and Lei, B. (2022). Bioactive anti-inflammatory antibacterial metformin-contained hydrogel dressing accelerating wound healing. Biomater. Adv. 135, 212737. doi:10.1016/j.bioadv.2022.212737
Li, Z., Hu, J., Yang, L., Zhang, X., Liu, X., Wang, Z., and Li, Y. (2020). Integrated poss-dendrimer nanohybrid materials: current status and future perspective. Nanoscale 12, 11395–11415. doi:10.1039/d0nr02394a
Li, R., Wei, X., Xu, J., Chen, J., Li, B., Wu, Z., and Wang, Z. L. (2021). Smart wearable sensors based on triboelectric nanogenerator for personal healthcare monitoring. Micromachines (Basel) 12. doi:10.3390/mi12040352
Li, Y., Yang, L., Hou, Y., Zhang, Z., Chen, M., Wang, M., et al. (2022). Polydopamine-mediated graphene oxide and nanohydroxyapatite-incorporated conductive scaffold with an immunomodulatory ability accelerates periodontal bone regeneration in diabetes. Bioact Mater 18, 213–217. doi:10.1016/j.bioactmat.2022.03.021
Li, S., Li, H., Lu, Y., Zhou, M., Jiang, S., Du, X., et al. (2023). Advanced textile-based wearable biosensors for healthcare monitoring. Biosens. (Basel) 13, 909. doi:10.3390/bios13100909
Li, W., Liu, Y., Lu, X., Huang, Y., Liu, Y., Cheng, S., et al. (2015). A cross-sectional study of breath acetone based on diabetic metabolic disorders. J. Breath. Res. 9, 016005. doi:10.1088/1752-7155/9/1/016005
Li, X., Jiang, X., Gao, F., Zhou, L., Wang, G., Li, B., et al. (2024). Study and evaluation of a gelatin-silver oxide nanoparticles releasing nitric oxide production of wound healing dressing for diabetic ulcer. Plos One 19, e0298124. doi:10.1371/journal.pone.0298124
Lim, J., Goh, L., Otake, K. I., Goh, S. S., Loh, X. J., and Kitagawa, S. (2023). Biomedically-relevant metal organic framework-hydrogel composites. Biomater Sci. 11, 2661–2677. doi:10.1039/d2bm01906j
Lin, H., Liu, J., Hou, Y., Yu, Z., Hong, J., Yu, J., et al. (2025a). Microneedle patch with pure drug tips for delivery of liraglutide: pharmacokinetics in rats and minipigs. Drug Deliv. Transl. Res. 15, 216–230. doi:10.1007/s13346-024-01582-1
Lin, L., and Pisano, A. P. (1999). Silicon-processed microneedles. J. Microelectromech. Syst 8, 78–84. doi:10.1109/84.749406
Lin, Y., Wu, J., Zhuang, Z., Gong, X., Jin, Z., Lin, X., et al. (2025b). A ph-responsive microneedle patch for the transdermal delivery of biomineralized insulin nanoparticles to diabetes treatment. Int. J. Biol. Macromol. 284, 137955. doi:10.1016/j.ijbiomac.2024.137955
Litra, D., Chiriac, M., Ababii, N., and Lupan, O. (2024). Acetone sensors based on al-coated and ni-doped copper oxide nanocrystalline thin films. Sensors (Basel) 24, 6550. doi:10.3390/s24206550
Loretelli, C., Assi, E., Seelam, A. J., Ben, N. M., and Fiorina, P. (2020). Cell therapy for type 1 diabetes. Expert Opin. Biol. Ther. 20, 887–897. doi:10.1080/14712598.2020.1748596
Luo, X., Yu, Q., Yang, L., and Cui, Y. (2023). Wearable, sensing-controlled, ultrasound-based microneedle smart system for diabetes management. Acs Sens. 8, 1710–1722. doi:10.1021/acssensors.2c02863
Lupascu, F. G., Sava, A., Tatarusanu, S. M., Iacob, A. T., Dascalu, A., Profire, B. S., et al. (2024). New chitosan-based co-delivery nanosystem for diabetes mellitus therapy. Polym. (Basel) 16, 1825. doi:10.3390/polym16131825
Lv, S., Zhang, S., Zuo, J., Liang, S., Yang, J., Wang, J., et al. (2023). Progress in preparation and properties of chitosan-based hydrogels. Int. J. Biol. Macromol. 242, 124915. doi:10.1016/j.ijbiomac.2023.124915
Lydiard, J., and Nemeroff, C. B. (2019). Biomarker-guided tailored therapy. Adv. Exp. Med. Biol. 1192, 199–224. doi:10.1007/978-981-32-9721-0_10
Maita, K. C., Avila, F. R., Torres-Guzman, R. A., Garcia, J. P., Eldaly, A. S., Palmieri, L., et al. (2022). Local anti-inflammatory effect and immunomodulatory activity of chitosan-based dressing in skin wound healing: a systematic review. J. Clin. Transl. Res. 8, 488–498.
Martanto, W., Moore, J. S., Kashlan, O., Kamath, R., Wang, P. M., and O'Neal, J. M. (2006). Microinfusion using hollow microneedles. Pharm Res 23, 104–113. doi:10.1007/s11095-005-8498-8
Mei, S., Yang, Y., and Wang, J. (2023). Synthesis of decellularized cartilage extracellular matrix hydrogels. J. Vis. Exp. doi:10.3791/64797
Memon, B., Elsayed, A. K., Bettahi, I., Suleiman, N., Younis, I., Wehedy, E., et al. (2022). Ipscs derived from insulin resistant offspring of type 2 diabetic patients show increased oxidative stress and lactate secretion. Stem Cell Res. Ther. 13, 428. doi:10.1186/s13287-022-03123-4
Mirbagheri, M. S., Akhavan-Mahdavi, S., Hasan, A., Kharazmi, M. S., and Jafari, S. M. (2023). Chitosan-based electrospun nanofibers for diabetic foot ulcer management; Recent advances. Carbohydr. Polym. 313, 120512. doi:10.1016/j.carbpol.2022.120512
Mohamed, A. A., Khater, S. I., Hamed, A. A., Metwally, M., Mostafa-Hedeab, G., and El-Shetry, E. S. (2021). Chitosan-stabilized selenium nanoparticles alleviate cardio-hepatic damage in type 2 diabetes mellitus model via regulation of caspase, bax/bcl-2, and fas/fasl-pathway. Gene 768, 145288. doi:10.1016/j.gene.2020.145288
Mohammad, K. A. (2024). Principles and biomedical applications of self-assembled peptides: potential treatment of type 2 diabetes mellitus. Pharmaceutics 16, 1442. doi:10.3390/pharmaceutics16111442
Morcol, T., Nagappan, P., Nerenbaum, L., Mitchell, A., and Bell, S. J. (2004). Calcium phosphate-peg-insulin-casein (capic) particles as oral delivery systems for insulin. Int. J. Pharm. 277, 91–97. doi:10.1016/j.ijpharm.2003.07.015
Moser, O., Zaharieva, D. P., Adolfsson, P., Battelino, T., Bracken, R. M., Buckingham, B. A., et al. (2025). The use of automated insulin delivery around physical activity and exercise in type 1 diabetes: a position statement of the european association for the study of diabetes (easd) and the international society for pediatric and adolescent diabetes (ispad). Diabetologia 68, 255–280. doi:10.1007/s00125-024-06308-z
Moses, R. L., Prescott, T., Mas-Claret, E., Steadman, R., Moseley, R., and Sloan, A. J. (2023). Evidence for natural products as alternative wound-healing therapies. Biomolecules 13, 444. doi:10.3390/biom13030444
Moon, S. J., Lee, S. S., Lee, H. S., and Kwon, T. H. (2005). Fabrication of microneedle array using liga and hot embossing process. Micro. Techn. 11, 311–318. doi:10.1007/s00542-004-0446-8
Metwally, W.M., El-Habashy, S.E., El-Hosseiny, L.S., Essawy, M.M., Eltaher, H.M., El-Khordagui, L.K., et al. (2023). Bioinspired 3d-printed scaffold embedding ddab-nano zno/nanofibrous microspheres for regenerative diabetic wound healing. Biofabrication 16, 015001. doi:10.1088/1758-5090/acfd60
Mukerjee, E.V., Collins, S.D., Isseroff, R.R., and Smith, R.L. (2015). Microneedle array for transdermal biological fluid extraction and in situ analysis. Sensors & Actua. Phy. 114, 267–275. doi:10.1016/j.sna.2003.11.008
Ma, B., Liu, S., Gan, Z., Liu, G., Cai, X., Zhang, H., et al. (2006). A pzt insulin pump integrated with a silicon microneedle array for transdermal drug delivery. Microf. Nanoflu. 2, 417–423. doi:10.1007/s10404-006-0083-x
Mousavi, S. M., Behbudi, G., Gholami, A., Hashemi, S. A., Nejad, Z. M., Bahrani, S., et al. (2022). Shape-controlled synthesis of zinc nanostructures mediating macromolecules for biomedical applications. Biomater. Res. 26, 4. doi:10.1186/s40824-022-00252-y
Na, E., Tao, S., Wang, W., Li, J., Guo, Y., Gao, R., et al. (2024). Ultrasensitive acetone gas sensor based on a k/sn-co (3)o(4) porous microsphere for noninvasive diabetes diagnosis. Acs Sens. 9, 6148–6156. doi:10.1021/acssensors.4c02009
Naahidi, S., Jafari, M., Logan, M., Wang, Y., Yuan, Y., Bae, H., et al. (2017). Biocompatibility of hydrogel-based scaffolds for tissue engineering applications. Biotechnol. Adv. 35, 530–544. doi:10.1016/j.biotechadv.2017.05.006
Nandhakumar, M., Gosala, R., and Subramanian, B. (2022). Invigorating chronic wound healing by nanocomposites composed with bioactive materials: a comprehensive review. Biotechnol. Lett. 44, 1243–1261. doi:10.1007/s10529-022-03303-5
Naranda, J., Bracic, M., Vogrin, M., and Maver, U. (2021). Recent advancements in 3d printing of polysaccharide hydrogels in cartilage tissue engineering. Mater. (Basel) 14, 3977. doi:10.3390/ma14143977
Nemati, M., Ebrahimi, Z., Karbalaei, N., Dastghaib, S., Khakshournia, S., and Sargazi, M. (2023). In vitro and in vivo improvement of islet quality and transplantation successes following islet treatment with biomaterials in diabetic rats. J. Diabetes Res. 2023, 1–14. doi:10.1155/2023/1399917
Ni, T., Dong, Z., Xi, K., Lu, Y., Chang, K., Ge, C., et al. (2024). Nitrogen-doped carbon quantum dots activated dandelion-like hierarchical wo (3) for highly sensitive and selective mems sensors in diabetes detection. Acs Sens. doi:10.1021/acssensors.4c01840
Nirenjen, S., Narayanan, J., Tamilanban, T., Subramaniyan, V., Chitra, V., Fuloria, N. K., et al. (2023). Exploring the contribution of pro-inflammatory cytokines to impaired wound healing in diabetes. Front. Immunol. 14, 1216321. doi:10.3389/fimmu.2023.1216321
Nisar, S., Dastgeer, G., Shazad, Z. M., Zulfiqar, M. W., Rasheed, A., Iqbal, M. Z., et al. (2024). 2d materials in advanced electronic biosensors for point-of-care devices. Adv. Sci. (Weinh) 11, e2401386. doi:10.1002/advs.202401386
Nuutila, K., Singh, M., Kruse, C., Philip, J., Caterson, E. J., and Eriksson, E. (2016). Titanium wound chambers for wound healing research. Wound Repair Regen. 24, 1097–1102. doi:10.1111/wrr.12472
Nyaga, D. M., Vickers, M. H., Jefferies, C., Perry, J. K., and O'Sullivan, J. M. (2018a). The genetic architecture of type 1 diabetes mellitus. Mol. Cell Endocrinol. 477, 70–80. doi:10.1016/j.mce.2018.06.002
Nyaga, D. M., Vickers, M. H., Jefferies, C., Perry, J. K., and O'Sullivan, J. M. (2018b). Type 1 diabetes mellitus-associated genetic variants contribute to overlapping immune regulatory networks. Front. Genet. 9, 535. doi:10.3389/fgene.2018.00535
Oliveira, M. J., Dalot, A., Fortunato, E., Martins, R., Byrne, H. J., Franco, R., et al. (2022). Microfluidic sers devices: brightening the future of bioanalysis. Discov. Mater 2, 12. doi:10.1007/s43939-022-00033-3
Oprita, E. I., Iosageanu, A., and Craciunescu, O. (2023). Natural polymeric hydrogels encapsulating small molecules for diabetic wound healing. Gels 9, 867. doi:10.3390/gels9110867
Otero, F., and Magner, E. (2020). Biosensors-recent advances and future challenges in electrode materials. Sensors (Basel) 20, 3561. doi:10.3390/s20123561
Pagliuca, F. W., Millman, J. R., Gurtler, M., Segel, M., Van Dervort, A., Ryu, J. H., et al. (2014). Generation of functional human pancreatic β cells In Vitro. Cell 159, 428–439. doi:10.1016/j.cell.2014.09.040
Pei, J., Palanisamy, C. P., Alugoju, P., Anthikapalli, N., Natarajan, P. M., Umapathy, V. R., et al. (2023). A comprehensive review on bio-based materials for chronic diabetic wounds. Molecules 28, 604. doi:10.3390/molecules28020604
Peng, W., Li, D., Dai, K., Wang, Y., Song, P., Li, H., et al. (2022). Recent progress of collagen, chitosan, alginate and other hydrogels in skin repair and wound dressing applications. Int. J. Biol. Macromol. 208, 400–408. doi:10.1016/j.ijbiomac.2022.03.002
Paganelli, A.I., Mondejar, A.G., Da, S.A., Silva-Calpa, G., Teixeira, M.F., Carvalho, F., et al. (2022). Real-time data analysis in health monitoring systems: a comprehensive systematic literature review. J. Biomed. Inform. 127, 104009. doi:10.1016/j.jbi.2022.104009
Prasathkumar, M., and Sadhasivam, S. (2021). Chitosan/hyaluronic acid/alginate and an assorted polymers loaded with honey, plant, and marine compounds for progressive wound healing-know-how. Int. J. Biol. Macromol. 186, 656–685. doi:10.1016/j.ijbiomac.2021.07.067
Putzbach, W., and Ronkainen, N. J. (2013). Immobilization techniques in the fabrication of nanomaterial-based electrochemical biosensors: a review. Sensors (Basel) 13, 4811–4840. doi:10.3390/s130404811
Qin, W., Wu, Y., Liu, J., Yuan, X., and Gao, J. (2022). A comprehensive review of the application of nanoparticles in diabetic wound healing: therapeutic potential and future perspectives. Int. J. Nanomedicine 17, 6007–6029. doi:10.2147/IJN.S386585
Qiu, H., Zhang, L., Wang, D., and Miao, H. (2022). Silver nanoparticles improve the biocompatibility and reduce the immunogenicity of xenogeneic scaffolds derived from decellularized pancreas. Cell Reprogr. 24, 38–47. doi:10.1089/cell.2021.0071
Queiroz, M., Shanmugam, S., Santos, L., Campos, C. A., Santos, A. M., Batista, M. S., et al. (2020). Microneedles as an alternative technology for transdermal drug delivery systems: a patent review. Expert Opin. Ther. Pat. 30, 433–452. doi:10.1080/13543776.2020.1742324
Renuka, R.R., Julius, A., Yoganandham, S.T., Umapathy, D., Ramadoss, R., Samrot, A.V., et al. (2022). Diverse nanocomposites as a potential dressing for diabetic wound healing. Front. Endocrinol. (Lausanne) 13, 1074568. doi:10.3389/fendo.2022.1074568
Rajinikanth, B. S., Rajkumar, D., K, K., and Vijayaragavan, V. (2024). Chitosan-based biomaterial in wound healing: a review. Cureus 16, e55193. doi:10.7759/cureus.55193
Rajput, A., Kulkarni, M., Deshmukh, P., Pingale, P., Garkal, A., Gandhi, S., et al. (2021). A key role by polymers in microneedle technology: a new era. Drug Dev. Ind. Pharm. 47, 1713–1732. doi:10.1080/03639045.2022.2058531
Ramesh, A., Chhabra, P., and Brayman, K. L. (2013). Pancreatic islet transplantation in type 1 diabetes mellitus: an update on recent developments. Curr. Diabetes Rev. 9, 294–311. doi:10.2174/15733998113099990063
Rathna, R. P., and Kulandhaivel, M. (2024). Advancements in wound healing: integrating biomolecules, drug delivery carriers, and targeted therapeutics for enhanced tissue repair. Arch. Microbiol. 206, 199. doi:10.1007/s00203-024-03910-y
Ravoor, J., Thangavel, M., and Elsen, S. R. (2021). Comprehensive review on design and manufacturing of bio-scaffolds for bone reconstruction. Acs Appl. Bio Mater 4, 8129–8158. doi:10.1021/acsabm.1c00949
Razzaghi, M., Ninan, J. A., Azimzadeh, M., Askari, E., Najafabadi, A. H., Khademhosseini, A., et al. (2024). Remote-controlled sensing and drug delivery via 3d-printed hollow microneedles. Adv. Healthc. Mater 13, e2400881. doi:10.1002/adhm.202400881
Ren, C., Zhong, D., Qi, Y., Liu, C., Liu, X., Chen, S., et al. (2023). Bioinspired ph-responsive microalgal hydrogels for oral insulin delivery with both hypoglycemic and insulin sensitizing effects. Acs Nano 17, 14161–14175. doi:10.1021/acsnano.3c04897
Ren, S., Guo, S., Yang, L., and Wang, C. (2022). Effect of composite biodegradable biomaterials on wound healing in diabetes. Front. Bioeng. Biotechnol. 10, 1060026. doi:10.3389/fbioe.2022.1060026
Renard, E. (2023). Automated insulin delivery systems: from early research to routine care of type 1 diabetes. Acta Diabetol. 60, 151–161. doi:10.1007/s00592-022-01929-5
Renzu, M., Hubers, C., Conway, K., Gibatova, V., Mehta, V., and Taha, W. (2024). Emerging technologies in endocrine drug delivery: innovations for improved patient care. Cureus 16, e62324. doi:10.7759/cureus.62324
Righettoni, M., Schmid, A., Amann, A., and Pratsinis, S. E. (2013). Correlations between blood glucose and breath components from portable gas sensors and ptr-tof-ms. J. Breath. Res. 7, 037110. doi:10.1088/1752-7155/7/3/037110
Rodriguez-Rodriguez, N., Martinez-Jimenez, I., Garcia-Ojalvo, A., Mendoza-Mari, Y., Guillen-Nieto, G., Armstrong, D. G., et al. (2022). Wound chronicity, impaired immunity and infection in diabetic patients. Medicc Rev. 24, 44–58. doi:10.37757/MR2021.V23.N3.8
Rozman, N., Tong, W. Y., Leong, C. R., Anuar, M. R., Karim, S., Ong, S. K., et al. (2020). Homalomena pineodora essential oil nanoparticle inhibits diabetic wound pathogens. Sci. Rep. 10, 3307. doi:10.1038/s41598-020-60364-0
Ruiz-Munoz, M., Martinez-Barrios, F. J., and Lopezosa-Reca, E. (2024). Placenta-derived biomaterials vs. Standard care in chronic diabetic foot ulcer healing: a systematic review and meta-analysis. Diabetes Metab. Syndr. 19, 103170. doi:10.1016/j.dsx.2024.103170
Saco, M., Howe, N., Nathoo, R., and Cherpelis, B. (2016). Comparing the efficacies of alginate, foam, hydrocolloid, hydrofiber, and hydrogel dressings in the management of diabetic foot ulcers and venous leg ulcers: a systematic review and meta-analysis examining how to dress for success. Dermatol Online J. 22. doi:10.5070/d3228032089
Sahu, B., Prusty, A., and Tudu, B. (2018). Total contact casting versus traditional dressing in diabetic foot ulcers. J. Orthop. Surg. Hong. Kong 26, 2309499018802486. doi:10.1177/2309499018802486
Salthouse, D., Novakovic, K., Hilkens, C., and Ferreira, A. M. (2023). Interplay between biomaterials and the immune system: challenges and opportunities in regenerative medicine. Acta Biomater. 155, 1–18. doi:10.1016/j.actbio.2022.11.003
Sarkar, S., Ekbal, K. M., Kalita, J., and Manna, P. (2023). Mesoporous silica nanoparticles: drug delivery vehicles for antidiabetic molecules. Chembiochem 24, e202200672. doi:10.1002/cbic.202200672
Sarkhel, S., Shuvo, S. M., Ansari, M. A., Mondal, S., Kapat, P., Ghosh, A., et al. (2024). Nanotechnology-based approaches for the management of diabetes mellitus: an innovative solution to long-lasting challenges in antidiabetic drug delivery. Pharmaceutics 16, 1572. doi:10.3390/pharmaceutics16121572
Scandurra, A., Iacono, V., Boscarino, S., Scalese, S., Grimaldi, M. G., and Ruffino, F. (2023). Model of chronoamperometric response towards glucose sensing by arrays of gold nanostructures obtained by laser, thermal and wet processes. Nanomater. (Basel) 13, 1163. doi:10.3390/nano13071163
Schlaeppi, A., Adams, W., Haase, R., Huisken, J., MacDonald, R. B., Eliceiri, K. W., et al. (2022). Meeting in the middle: towards successful multidisciplinary bioimage analysis collaboration. Front. Bioinform 2, 889755. doi:10.3389/fbinf.2022.889755
Schackart, K.R., and Yoon, J.Y. (2021). Machine learning enhances the performance of bioreceptor-free biosensors. Sensors (Basel) 21, 5519. doi:10.3390/s21165519
Shamshad, A., Kang, C., Jenny, L. A., Persad-Paisley, E. M., and Tsang, S. H. (2023). Translatability barriers between preclinical and clinical trials of aav gene therapy in inherited retinal diseases. Vis. Res. 210, 108258. doi:10.1016/j.visres.2023.108258
Sharma, S., and Kishen, A. (2024). Bioarchitectural design of bioactive biopolymers: structure-function paradigm for diabetic wound healing. Biomimetics (Basel) 9, 275. doi:10.3390/biomimetics9050275
Sharmah, B., Afzal, N. U., Loying, R., Roy, A., Kalita, J., Das, J., et al. (2024). Glucose-responsive insulin delivery via surface-functionalized titanium dioxide nanoparticles: a promising theragnostic against diabetes mellitus. Acs Appl. Bio Mater 8, 475–487. doi:10.1021/acsabm.4c01426
Socci, M. C., Rodriguez, G., Oliva, E., Fushimi, S., Takabatake, K., Nagatsuka, H., et al. (2023). Polymeric materials, advances and applications in tissue engineering: a review. Bioeng. (Basel) 10, 218. doi:10.3390/bioengineering10020218
Soetedjo, A., Lee, J. M., Lau, H. H., Goh, G. L., An, J., Koh, Y., et al. (2021). Tissue engineering and 3d printing of bioartificial pancreas for regenerative medicine in diabetes. Trends Endocrinol. Metab. 32, 609–622. doi:10.1016/j.tem.2021.05.007
Sonmezer, D., Latifoglu, F., Toprak, G., and Baran, M. (2023). A native extracellular matrix material for tissue engineering applications: characterization of pericardial fluid. J. Biomed. Mater Res. B Appl. Biomater. 111, 1629–1639. doi:10.1002/jbm.b.35260
Starlin, C. M., Mahmood, S., Mohamed, S. Z., Wan, H. C., Sundarapandian, R., Ahamed, H. N., et al. (2024). Biodegradable polymeric insulin microneedles - a design and materials perspective review. Drug Deliv. 31, 2296350. doi:10.1080/10717544.2023.2296350
Sultana, A., Borgohain, R., Rayaji, A., Saha, D., and Kumar, D. B. (2024). Promising phytoconstituents in diabetes-related wounds: mechanistic insights and implications. Curr. Diabetes Rev. 21, e270224227477. doi:10.2174/0115733998279112240129074457
Summers-Gibson, L. (2021). The relationships between diabetes self-care, diabetes time management, and diabetes distress in women with type 2 diabetes mellitus. Sci. Diabetes Self Manag. Care 47, 245–254. doi:10.1177/26350106211014438
Takahashi, M., Umehara, Y., Yue, H., Trujillo-Paez, J. V., Peng, G., Nguyen, H., et al. (2021). The antimicrobial peptide human β-defensin-3 accelerates wound healing by promoting angiogenesis, cell migration, and proliferation through the FGFR/JAK2/STAT3 signaling pathway. Front. Immunol. 12, 712781. doi:10.3389/fimmu.2021.712781
Tallapaneni, V., Kalaivani, C., Pamu, D., Mude, L., Singh, S. K., and Karri, V. (2021). Acellular scaffolds as innovative biomaterial platforms for the management of diabetic wounds. Tissue Eng. Regen. Med. 18, 713–734. doi:10.1007/s13770-021-00344-1
Tanda, N., Hinokio, Y., Washio, J., Takahashi, N., and Koseki, T. (2014). Analysis of ketone bodies in exhaled breath and blood of ten healthy Japanese at ogtt using a portable gas chromatograph. J. Breath. Res. 8, 046008. doi:10.1088/1752-7155/8/4/046008
Tankasala, D., and Linnes, J. C. (2019). Noninvasive glucose detection in exhaled breath condensate. Transl. Res. 213, 1–22. doi:10.1016/j.trsl.2019.05.006
Teymourian, H., Barfidokht, A., and Wang, J. (2020). Electrochemical glucose sensors in diabetes management: an updated review (2010-2020). Chem. Soc. Rev. 49, 7671–7709. doi:10.1039/d0cs00304b
Tripathi, A. S., Zaki, M., Al-Hussain, S. A., Dubey, B. K., Singh, P., Rind, L., et al. (2023). Material matters: exploring the interplay between natural biomaterials and host immune system. Front. Immunol. 14, 1269960. doi:10.3389/fimmu.2023.1269960
Tutty, M. A., Vella, G., and Prina-Mello, A. (2022). Pre-clinical 2d and 3d toxicity response to a panel of nanomaterials; Comparative assessment of nbm-induced liver toxicity. Drug Deliv. Transl. Res. 12, 2157–2177. doi:10.1007/s13346-022-01170-1
Umehara, Y., Takahashi, M., Yue, H., Trujillo-Paez, J. V., Peng, G., Nguyen, H., et al. (2022). The antimicrobial peptides human β-defensins induce the secretion of angiogenin in human dermal fibroblasts. Int. J. Mol. Sci. 23, 8800. doi:10.3390/ijms23158800
Vajhadin, F., Mazloum-Ardakani, M., and Amini, A. (2021). Metal oxide-based gas sensors for the detection of exhaled breath markers. Med. Devices Sens. 4, e10161. doi:10.1002/mds3.10161
Varela-Moreno, E., Carreira, S. M., Guzman-Parra, J., Jodar-Sanchez, F., Mayoral-Cleries, F., and Anarte-Ortiz, M. T. (2021). Effectiveness of ehealth-based psychological interventions for depression treatment in patients with type 1 or type 2 diabetes mellitus: a systematic review. Front. Psychol. 12, 746217. doi:10.3389/fpsyg.2021.746217
Vargas, G. M., Aendekerk, F., de Boer, C., Geurts, J., Lucchesi, J., and Arts, J. (2024). Bioactive-glass-based materials with possible application in diabetic wound healing: a systematic review. Int. J. Mol. Sci. 25, 1152. doi:10.3390/ijms25021152
Verbaan, F. J., Bal, S. M., van den Berg, D. J., Dijksman, J. A., van Hecke, M., Verpoorten, H., et al. (2008). Improved piercing of microneedle arrays in dermatomed human skin by an impact insertion method. J. Control. Release. 128, 80–88. doi:10.1016/j.jconrel.2008.02.009
Venkatesan, V., and Rangasamy, J. (2023). Diabetic pedal osteomyelitis and its treatment. Chonnam Med. J. 59, 109–114. doi:10.4068/cmj.2023.59.2.109
Volpatti, L. R., Facklam, A. L., Cortinas, A. B., Lu, Y. C., Matranga, M. A., MacIsaac, C., et al. (2021). Microgel encapsulated nanoparticles for glucose-responsive insulin delivery. Biomaterials 267, 120458. doi:10.1016/j.biomaterials.2020.120458
Wang, J., and Jin, X. (2024). Strategies for decellularization, re-cellularization and crosslinking in liver bioengineering. Int J Artif Organs 47, 129–139. doi:10.1177/03913988231218566
Wang, J., Duan, X., Zhong, D., Zhang, M., Li, J., Hu, Z., et al. (2024). Pharmaceutical applications of chitosan in skin regeneration: a review. Int. J. Biol. Macromol. 261, 129064. doi:10.1016/j.ijbiomac.2023.129064
Wang, X., Yang, Y., Zhao, W., Zhu, Z., and Pei, X. (2024). Recent advances of hydrogels as smart dressings for diabetic wounds. J. Mater Chem. B 12, 1126–1148. doi:10.1039/d3tb02355a
Wang, Y., Wang, H., Zhu, X. X., Guan, Y., and Zhang, Y. (2020). Smart microneedle patches for rapid, and painless transdermal insulin delivery. J. Mater Chem. B 8, 9335–9342. doi:10.1039/d0tb01822h
Wilems, T., Vardhan, S., Wu, S., and Sakiyama-Elbert, S. (2019). The influence of microenvironment and extracellular matrix molecules in driving neural stem cell fate within biomaterials. Brain Res. Bull. 148, 25–33. doi:10.1016/j.brainresbull.2019.03.004
Woo, S. H., Choi, J. H., Mo, Y. J., Lee, Y. I., Jeon, W. B., and Lee, Y. S. (2023). Engineered elastin-like polypeptide improves the efficiency of adipose-derived stem cell-mediated cutaneous wound healing in type ii diabetes mellitus. Heliyon 9, e20201. doi:10.1016/j.heliyon.2023.e20201
Wilke, N., Hibert, C., Brien, J.O., and Morrissey, A. (2005). Silicon microneedle electrode array with temperature monitoring for electroporation. Sensors & Actuators a Physical 123, 319–325. doi:10.1016/j.sna.2005.05.017
Wu, X., Liu, C., Wang, L., and Bilal, M. (2023). Internet of things-enabled real-time health monitoring system using deep learning. Neural. Comput. Appl. 35, 14565–14576. doi:10.1007/s00521-021-06440-6
Xie, B., Wang, K., Li, B., Huang, R., Xu, Z., and Li, X. (2023). Biomaterial-mediated strategies for accurate and convenient diagnosis, and effective treatment of diabetes: advantages, current progress and future perspectives. J. Mater Chem. B 11, 3766–3786. doi:10.1039/d3tb00034f
Xie, Z., Ramakrishnam, R. M., Stewart, A. C., Nantz, M. H., and Fu, X. A. (2018). Imparting sensitivity and selectivity to a gold nanoparticle chemiresistor through thiol monolayer functionalization for sensing acetone. Rsc Adv. 8, 35618–35624. doi:10.1039/c8ra06137h
Xing, C., Luo, M., Sheng, Q., Zhu, Z., Yu, D., Huang, J., et al. (2024). Silk fabric functionalized by nanosilver enabling the wearable sensing for biomechanics and biomolecules. Acs Appl. Mater Interfaces 16, 51669–51678. doi:10.1021/acsami.4c10253
Xu, B., Liu, H., Yang, G., Zhang, S., Zhou, Z., and Gao, Y. (2024). Novel double-layered plga microparticles-dissolving microneedle (mps-dmn) system for peptide drugs sustained release by transdermal delivery. Int. J. Pharm. 670, 125128. doi:10.1016/j.ijpharm.2024.125128
Xu, G., Zhang, W., Du, J., Yuan, X., Zhang, W., Yan, W., et al. (2022). Biomass-derived porous carbon with high drug adsorption capacity undergoes enzymatic and chemical degradation. J. Colloid Interface Sci. 622, 87–96. doi:10.1016/j.jcis.2022.04.064
Xu, R., Fang, Y., Zhang, Z., Cao, Y., Yan, Y., Gan, L., et al. (2023). Recent advances in biodegradable and biocompatible synthetic polymers used in skin wound healing. Mater. (Basel) 16, 5459. doi:10.3390/ma16155459
Yang, J., and Xu, L. (2023). Electrospun nanofiber membranes with various structures for wound dressing. Mater. (Basel) 16, 6021. doi:10.3390/ma16176021
Yang, P., Li, S., Zhang, H., Ding, X., and Tan, Q. (2024). LRG1 accelerates wound healing in diabetic rats by promoting angiogenesis via the wnt/β-catenin signaling pathway. Int. J. Low. Extrem Wounds 23, 568–576. doi:10.1177/15347346221081610
Yao, Y., Zhang, H., Wang, Z., Ding, J., Wang, S., Huang, B., et al. (2019). Reactive oxygen species (ros)-responsive biomaterials mediate tissue microenvironments and tissue regeneration. J. Mater Chem. B 7, 5019–5037. doi:10.1039/c9tb00847k
Yasin, S., Said, Z., Halib, N., Rahman, Z. A., and Mokhzani, N. I. (2023). Polymer-based hydrogel loaded with honey in drug delivery system for wound healing applications. Polym. (Basel) 15, 3085. doi:10.3390/polym15143085
Yoon, J., Cho, H. Y., Shin, M., Choi, H. K., Lee, T., and Choi, J. W. (2020). Flexible electrochemical biosensors for healthcare monitoring. J. Mater Chem. B 8, 7303–7318. doi:10.1039/d0tb01325k
Young, K. G., McGovern, A. P., Barroso, I., Hattersley, A. T., Jones, A. G., Shields, B. M., et al. (2023). The impact of population-level hba(1c) screening on reducing diabetes diagnostic delay in middle-aged adults: a UK biobank analysis. Diabetologia 66, 300–309. doi:10.1007/s00125-022-05824-0
Yue, H., Song, P., Sutthammikorn, N., Umehara, Y., Trujillo-Paez, J. V., Nguyen, H., et al. (2022). Antimicrobial peptide derived from insulin-like growth factor-binding protein 5 improves diabetic wound healing. Wound Repair Regen. 30, 232–244. doi:10.1111/wrr.12997
Yunos, M., Manczak, R., Guines, C., Mansor, A., Mak, W. C., Khan, S., et al. (2021). Rf remote blood glucose sensor and a microfluidic vascular phantom for sensor validation. Biosens. (Basel) 11, 494. doi:10.3390/bios11120494
Yusuf, A. A., and Adeleke, O. A. (2023). Nanofibrous scaffolds for diabetic wound healing. Pharmaceutics 15, 986. doi:10.3390/pharmaceutics15030986
Zafar, H., Channa, A., Jeoti, V., and Stojanovic, G. M. (2022). Comprehensive review on wearable sweat-glucose sensors for continuous glucose monitoring. Sensors (Basel) 22, 638. doi:10.3390/s22020638
Zaric, B. L., Obradovic, M., Sudar-Milovanovic, E., Nedeljkovic, J., Lazic, V., and Isenovic, E. R. (2019). Drug delivery systems for diabetes treatment. Curr. Pharm. Des. 25, 166–173. doi:10.2174/1381612825666190306153838
Zhang, F., Pei, X., Peng, X., Gou, D., Fan, X., Zheng, X., et al. (2022). Dual crosslinking of folic acid-modified pectin nanoparticles for enhanced oral insulin delivery. Biomater. Adv. 135, 212746. doi:10.1016/j.bioadv.2022.212746
Zhang, G., Ichikawa, K., Iitani, K., Iwasaki, Y., and Mitsubayashi, K. (2025a). A handheld biofluorometric system for acetone detection in exhaled breath condensates. Analyst 150, 505–512. doi:10.1039/d4an01281j
Zhang, K., Zhang, C., Zhou, H., Yang, Y., Wen, Y., Jiao, X., et al. (2024a). Elastic nanofibrous dressings with mesenchymal stem cell-recruiting and protecting characteristics for promoting diabetic wound healing. Acs Appl. Mater Interfaces 16, 41869–41880. doi:10.1021/acsami.4c07369
Zhang, P., Dalton, C., and Jullien, G.A. (2009). Design and fabrication of mems-based microneedle arrays for medical applications. Micro. Techno. 15, 1073–1082. doi:10.1007/s00542-009-0883-5
Zhang, S., Ge, G., Qin, Y., Li, W., Dong, J., Mei, J., et al. (2023a). Recent advances in responsive hydrogels for diabetic wound healing. Mater Today Bio 18, 100508. doi:10.1016/j.mtbio.2022.100508
Zhang, S., Zhao, W., Zeng, J., He, Z., Wang, X., Zhu, Z., et al. (2023b). Wearable non-invasive glucose sensors based on metallic nanomaterials. Mater Today Bio 20, 100638. doi:10.1016/j.mtbio.2023.100638
Zhang, T., Wu, H., Qiu, C., Wang, M., Wang, H., Zhu, S., et al. (2024b). Ultrasensitive hierarchical aunrs@sio(2)@ag sers probes for enrichment and detection of insulin and c-peptide in serum. Int. J. Nanomedicine 19, 6281–6293. doi:10.2147/IJN.S462601
Zhang, X., Zhang, Y., Zheng, H., Yang, X., Zou, S., and Chen, J. (2025b). Design, fabrication, and evaluation of antimicrobial sponge microneedles for the transdermal delivery of insulin. Eur. J. Pharm. Biopharm. 206, 114586. doi:10.1016/j.ejpb.2024.114586
Zhang, Y., Sun, J., Liu, L., and Qiao, H. (2021). A review of biosensor technology and algorithms for glucose monitoring. J Diabetes Complications 35, 107929. doi:10.1016/j.jdiacomp.2021.107929
Zhang, Z., Xue, H., Xiong, Y., Geng, Y., Panayi, A. C., Knoedler, S., et al. (2024c). Copper incorporated biomaterial-based technologies for multifunctional wound repair. Theranostics 14, 547–570. doi:10.7150/thno.87193
Zhao, H., Li, J., She, X., Chen, Y., Wang, M., Wang, Y., et al. (2024a). Oxygen vacancy-rich bimetallic au@pt core-shell nanosphere-functionalized electrospun znfe(2)o(4) nanofibers for chemiresistive breath acetone detection. Acs Sens. 9, 2183–2193. doi:10.1021/acssensors.4c00382
Zhao, H., Li, Z., Wang, Y., Zhou, K., Li, H., Bi, S., et al. (2023). Bioengineered msc-derived exosomes in skin wound repair and regeneration. Front. Cell Dev. Biol. 11, 1029671. doi:10.3389/fcell.2023.1029671
Zhao, S., Lu, Z., Cai, R., Wang, H., Gao, S., Yang, C., et al. (2024b). A wearable osmotic microneedle patch provides high-capacity sustained drug delivery in animal models. Sci. Transl. Med. 16, eadp3611. doi:10.1126/scitranslmed.adp3611
Zheng, Q., Chen, C., Liu, Y., Gao, J., Li, L., Yin, C., et al. (2024). Metal nanoparticles: advanced and promising technology in diabetic wound therapy. Int. J. Nanomedicine 19, 965–992. doi:10.2147/IJN.S434693
Zheng, Y., Ma, Y., He, P., Yan, L., Cao, H., and Shao, F. (2025). Phycocyanin/hyaluronic acid microneedle patches loaded with celastrol nanoparticles for synergistic treatment of diabetic nephropathy. Acs Biomater. Sci. Eng. 11, 371–382. doi:10.1021/acsbiomaterials.4c01787
Zhong, Y., Song, B., He, D., Xia, Z., Wang, P., Wu, J., et al. (2020). Galactose-based polymer-containing phenylboronic acid as carriers for insulin delivery. Nanotechnology 31, 395601. doi:10.1088/1361-6528/ab9e26
Zhou, D., Zhang, S., Khan, A. U., Chen, L., and Ge, G. (2023). A wearable aunp enhanced metal-organic gel (au@mog) sensor for sweat glucose detection with ultrahigh sensitivity. Nanoscale 16, 163–170. doi:10.1039/d3nr05179j
Zhou, Y. Y., Chen, J. P., Gan, L., Xu, W., Liu, Y., Zhao, Y. G., et al. (2022). A non-invasive method for the detection of glucose in human exhaled breath by condensation collection coupled with ion chromatography. J. Chromatogr. 1685, 463564. doi:10.1016/j.chroma.2022.463564
Zhou, Y., Huang, H., Chen, G., Yuan, Q., Ren, J., Wu, J., et al. (2024a). Promoting the healing of diabetic wounds with an antimicrobial gel containing agnps with anti-infective and anti-inflammatory properties. J. Biomater. Sci. Polym. 35, 1236–1257. doi:10.1080/09205063.2024.2324494
Zhou, Y., Li, L., Tong, J., Chen, X., Deng, W., Chen, Z., et al. (2024b). Advanced nanomaterials for electrochemical sensors: application in wearable tear glucose sensing technology. J. Mater Chem. B 12, 6774–6804. doi:10.1039/d4tb00790e
Zhu, X., Cao, P., Li, P., Yu, Y., Guo, R., Li, Y., et al. (2024). Bimetallic ptau-decorated sno (2) nanospheres exhibiting enhanced gas sensitivity for ppb-level acetone detection. Nanomater. (Basel) 14, 1097. doi:10.3390/nano14131097
Keywords: biomaterials, diabetes, insulin delivery, tissue engineering, biosensors
Citation: Guan W and Zhang L (2025) Applications and prospects of biomaterials in diabetes management. Front. Bioeng. Biotechnol. 13:1547343. doi: 10.3389/fbioe.2025.1547343
Received: 18 December 2024; Accepted: 30 January 2025;
Published: 07 March 2025.
Edited by:
Xianzhi Zhang, Yale University, United StatesReviewed by:
Mahboubeh Nabavinia, The Research Institute at Nationwide Children’s Hospital, United StatesCopyright © 2025 Guan and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Liang Zhang, cnRqcHh6bEBzeW1jLmVkdS5jbg==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.