
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
BRIEF RESEARCH REPORT article
Front. Artif. Intell., 07 February 2024
Sec. Machine Learning and Artificial Intelligence
Volume 7 - 2024 | https://doi.org/10.3389/frai.2024.1301997
 Raissa Souza1,2,3,4*
Raissa Souza1,2,3,4* Emma A. M. Stanley1,2,3,4
Emma A. M. Stanley1,2,3,4 Milton Camacho1,2,3,4
Milton Camacho1,2,3,4 Richard Camicioli5
Richard Camicioli5 Oury Monchi1,2,6,7,8
Oury Monchi1,2,6,7,8 Zahinoor Ismail2,8,9,10
Zahinoor Ismail2,8,9,10 Matthias Wilms2,4,11,12†
Matthias Wilms2,4,11,12† Nils D. Forkert1,2,4,8†
Nils D. Forkert1,2,4,8†Distributed learning is a promising alternative to central learning for machine learning (ML) model training, overcoming data-sharing problems in healthcare. Previous studies exploring federated learning (FL) or the traveling model (TM) setup for medical image-based disease classification often relied on large databases with a limited number of centers or simulated artificial centers, raising doubts about real-world applicability. This study develops and evaluates a convolution neural network (CNN) for Parkinson's disease classification using data acquired by 83 diverse real centers around the world, mostly contributing small training samples. Our approach specifically makes use of the TM setup, which has proven effective in scenarios with limited data availability but has never been used for image-based disease classification. Our findings reveal that TM is effective for training CNN models, even in complex real-world scenarios with variable data distributions. After sufficient training cycles, the TM-trained CNN matches or slightly surpasses the performance of the centrally trained counterpart (AUROC of 83% vs. 80%). Our study highlights, for the first time, the effectiveness of TM in 3D medical image classification, especially in scenarios with limited training samples and heterogeneous distributed data. These insights are relevant for situations where ML models are supposed to be trained using data from small or remote medical centers, and rare diseases with sparse cases. The simplicity of this approach enables a broad application to many deep learning tasks, enhancing its clinical utility across various contexts and medical facilities.
Distributed learning, and especially its federated learning (FL) implementation, has emerged as a viable and promising alternative to central learning for training of machine learning (ML) models to address various patient privacy regulations and administrative barriers (Tuladhar et al., 2022). It provides a practical solution for accessing extensive and diverse datasets by facilitating ML model training in distributed environments. In the standard FL setup, each center receives a copy of a global model from a central server and local training takes place at each center for a pre-defined number of epochs using the data locally available. After local training, the learned model parameters are sent back to the server. Using an aggregation function, the server combines these parameters to update the global model, which is then sent back to the centers for additional training and model refinement. This iterative process is usually repeated over multiple rounds to improve the global model's performance (McMahan et al., 2016).
Prior studies that explored this standard FL setup for disease classification tasks based on medical images often employed large databases with a limited number of participating centers where the data were acquired, and/or used such data to generate artificial centers to simulate diverse data contributions. Artificial centers with imbalanced contributions are typically generated using the Dirichlet distribution, resulting in an overall distribution that exhibits exponential decay. This means that many centers will contribute many datasets (i.e., many medical images) while a few centers will contribute a small number of datasets. For instance, Yan et al. (2021) used a COVID-19 database consisting of 15,282 chest X-ray images and five artificially generated centers. Cetinkaya et al. (2021) employed a COVID-19 database containing 28,833 chest X-ray images and 20 artificial centers based on the Dirichlet distribution. Liu et al. (2023) evaluated their FL approach for COVID-19 (5,908 chest X-rays) and skin lesion detection (10,015 images) tasks by artificially creating centers (8, 9, 10, 11, 12 centers for COVID-19 and 3, 4, 5, 6, 7 centers for skin lesion detection) using the Dirichlet distribution. Additionally, Wicaksana et al. (2023) developed a FL model for skin lesion and intracranial hemorrhage classification using 23,247 dermatoscopy images and 67,969 brain CT images, respectively, with six centers for skin lesion detection and two artificial centers for intracranial hemorrhage classification. Jiang et al. (2022) developed and evaluated a FL model for breast cancer classification using 450,000 histology images from five centers, while Adnan et al. (2022) simulated data distributions across 4, 8, 16, and 32 centers for histopathology classification using 30,070 images. Li et al. (2020) evaluated their FL approach using 370 resting-state fMRI data from four centers. Lastly, Zhou et al. (2022) created 20 artificial centers for diabetic retinopathy classification using a FL system trained with a total of 3,662 images.
Although all the studies mentioned above offered important technical advancements and insights, their scope was inherently limited by the number of (real) centers participating in the distributed learning setup. Moreover, even though artificial centers with dissimilar contributions were simulated by sampling from a Dirichlet distribution, most of the centers still contributed a large number of datasets. However, such conditions may not effectively represent real-world scenarios for 3D imaging data where some centers may only have access to a very few datasets and the disease of interest may present differently across centers, raising concerns about the performance of FL with genuinely diverse and skewed data distributions that arise from the limited data available at medical facilities (Ng et al., 2021; Tuladhar et al., 2022).
The traveling model (TM) paradigm, also known as Cyclical Weight Transfer (CWT) (Chang et al., 2018; Balachandar et al., 2020), is an alternative approach to the standard FL setup for distributed learning. In an initial analysis by Souza et al. (2022b), TM has been shown to outperform FL for cases where limited datasets are available at each participating center. Although their examination is based on empirical evidence, the results suggest that TM holds promise as a potential alternative to FL in these particular scenarios. Briefly described, in the TM setup, a single model undergoes sequential training across various centers following a predetermined travel sequence that dictates the order of center visits. The model is initialized at a central server or the first center and undergoes training with the available data at that center. Subsequently, the updated model travels to the next center, where it continues training with the locally available data. This process continues until the final center is reached, completing one training cycle. Similar to FL, multiple cycles can be performed to improve the global model's performance. However, unlike in the standard FL setup, there is no need for an aggregation function as the same model is continuously improved by traveling from center to center.
The benefit of the TM for small local datasets stems from the iterative training of a single model, addressing the challenge of local models yielding suboptimal parameters due to overfitting, which often occurs when training ML models with very small sample sizes. Furthermore, this approach overcomes the challenge of aggregating multiple models without marginalizing centers with fewer datasets. However, in contrast to FL, the TM paradigm has seen limited exploration so far, with only one study specifically focusing on small sample sizes available in each center (Souza et al., 2022b). Moreover, the TM has not been used and evaluated for training convolution neural networks (CNNs) for disease classification using real distributed 3D imaging data where some centers provide only very few training samples. Instead, similar to FL studies, TM investigations (Chang et al., 2018; Balachandar et al., 2020; Souza et al., 2022b) often make use of large databases with simulated centers, prompting concerns regarding their genuine applicability to real-world scenarios.
Therefore, this work aims to develop and evaluate a Parkinson's disease (PD) classifier utilizing the TM approach. This traveling model classifier is developed and evaluated using a large database comprising 1,817 three-dimensional T1-weighted brain magnetic resonance imaging (MRI) scans acquired in 83 different real centers around the world. Each of these centers contributes distinct and unique information, encompassing biological (e.g., sex, age, and target labels) and non-biological (e.g., scanner types and the number of participants per center) factors. Our major contributions include: (1) the development and evaluation of a TM approach for training 3D CNNs for a disease classification purpose using 3D datasets, and (2) the first work to make use of a real-world data distribution with many centers providing only very few training samples, acquired using a wide selection of MRI scanners and acquisition protocols.
In this study, we developed and trained a CNN model for PD classification from T1-weighted brain MRI data using a TM approach and analyzed its performance using the largest multi-center PD database described in the literature.
All analyses conducted in this study utilized a distinct multi-center PD database, comprising 1,817 T1-weighted MRI scans acquired in 83 different healthcare centers around the world1, 2, 3 (Acharya et al., 2007; Jack et al., 2008; Hanganu et al., 2014; Sudlow et al., 2015; Badea et al., 2017; Wei et al., 2018; Duchesne et al., 2019; LaMontagne et al., 2019; Lang et al., 2019; Talai et al., 2021; Thibeau-Sutre et al., 2022). Each center received ethics approval from their local ethics board and received written informed consent from all the participants under the declaration of Helsinki. This database is exceptionally diverse, encompassing variations in participant demographics, center contributions, scanner vendors (Siemens, GE, and Phillips), scanner types (23 scanners were utilized), and magnetic field strengths (1.5T or 3.0T). Table 1 provides an overview of the database demographics.
Whenever possible, datasets from each center were divided into 80% for training and 20% for testing, resulting in 1,410 MRI scans for training and 407 MRI scans for testing. For centers providing a very small number of samples (e.g., <25 samples), the data were split into training and testing sets aiming to achieve an overall balanced representation in terms of sex and age, as shown in Table 2. All datasets were pre-processed as described in Souza et al. (2023), which included skull-stripping, resampling to an isotropic resolution of 1 mm, bias field correction, affine image registration to the PD25-T1-MPRAGE-1mm brain atlas (Xiao et al., 2017), and cropping to reduce irrelevant background information.
In this study, we utilized a state-of-the-art simple fully convolutional network (SFCN) (see Figure 1A), which achieved a high, state-of-the-art accuracy (78.8%) differentiating healthy participants and patients with PD using multi-center T1-weighted MRI scans in a centralized approach (Camacho et al., 2023), as the basis for all experiments. A grid search was conducted to optimize this centralized model on the data available for this work, considering various parameters such as learning rate, dropout layer, and learning rate decay. The best model was selected based on early stopping criteria, with a patience of 10 epochs, considering the lowest testing loss. The chosen model utilized the Adam optimizer with an initial learning rate of 0.001 and employed an exponential decay rate every epoch. The training was performed using a shuffled batch size of 5 and a dropout layer before the flattening layer, with a dropout rate of 20%.
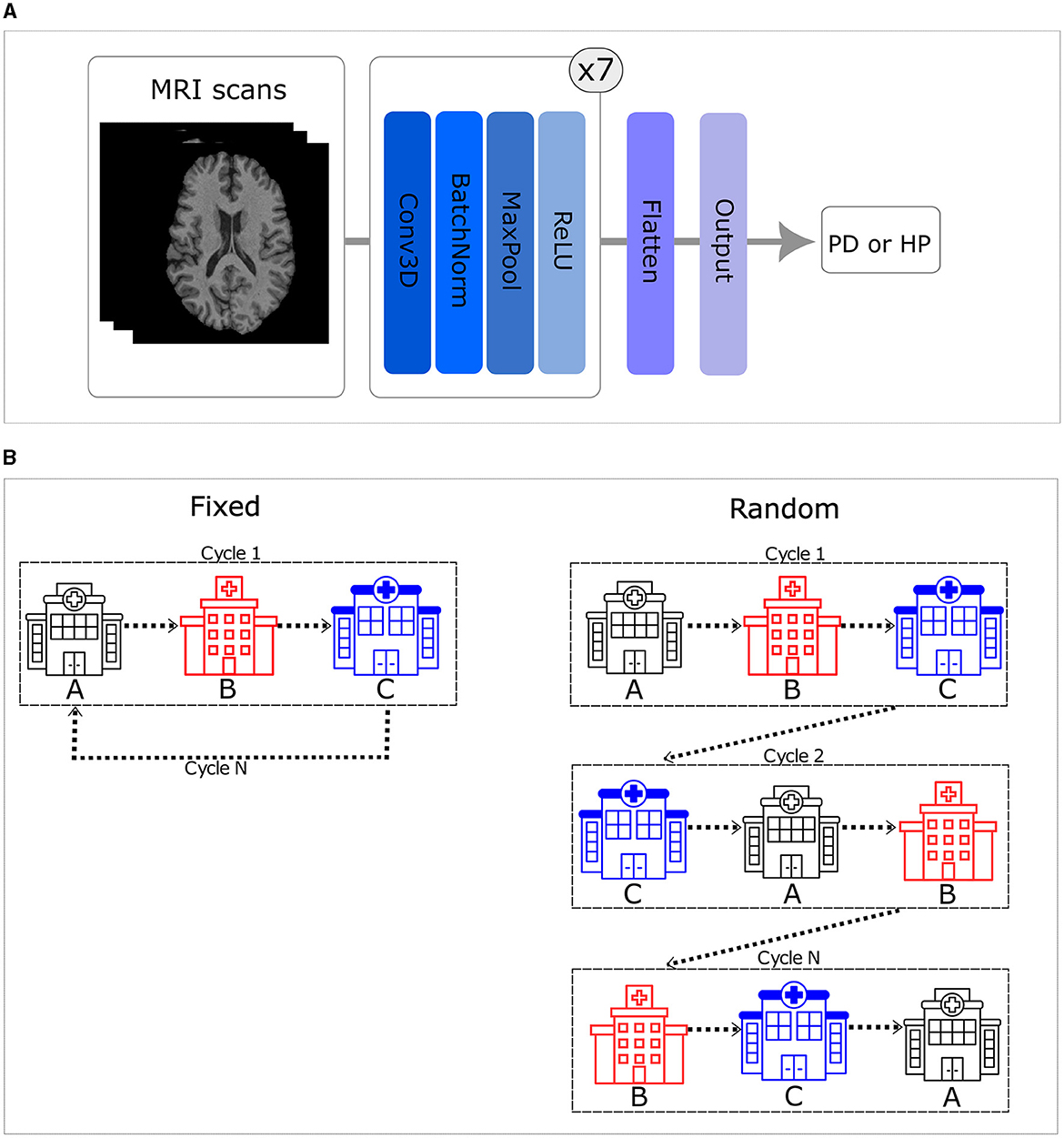
Figure 1. (A) Simple fully convolutional neural network (CNN) architecture. (B) Order of the training set. The left side illustrates that when trained in a fixed order, all centers are visited in a consistent sequence. In contrast, the right side shows that the sequence varies for each cycle when trained in a random order.
In this study, we implemented the first CNN model for PD classification from T1-weighted brain MRI data using a TM approach. Moreover, we explored six distinct configurations of the CNN trained using the TM approach. These configurations encompassed random and fixed traveling sequences, with variations in the number of local training epochs (one, two, and five) prior to moving to the next center. The fixed traveling order involves visiting every center in a consistent sequence throughout each cycle, which is defined, using a random seed equal to 42, once in the beginning. In contrast to that, the random order introduces cycle-to-cycle variability, using a different seed (i.e., adding 1 to the initial seed of 42 after each cycle), in the sequence visited by the model, practically emulating the batch shuffling process used in the centralized approach (see Figure 1B).
The performance of the traveling models was assessed for up to 30 cycles, ensuring consistency with the total number of epochs used to train the centralized model, where each epoch corresponds to a full cycle in the traveling setup. With respect to the optimizer and dropout layer, the only difference between the traveling models and the centralized model is the initial learning rate, which was set to 0.0001. This value was chosen after conducting a grid search and taking into account that several centers have fewer than five samples (4, 3, 2, or 1) available for local training, necessitating a smaller learning step. As a result, the traveling model experiments utilized a batch size of 5 or equal to the number of samples available at a local center, if this number was smaller than 5. Each sample in the batch corresponds to a unique participant T1-weighted MRI scan. Although the training was conducted on a single computer equipped with an NVIDIA GeForce RTX 3090 GPU, the training procedure adhered to the TM concept by fetching data from a single center at any given time. Nevertheless, the outcomes outlined in this study are expected to remain consistent and unaffected by the specific physical implementation (such as computer network and data transfer protocols) as long as each center employs the identical hardware and software configuration utilized in our training. Our code is available at https://github.com/RaissaSouza/pd-travelling-model.
For quantitative evaluation of our results, we measured the Area Under the Receiver Operating Characteristic Curve (AUC ROC), which provides a single scalar value that measures the overall threshold-independent performance of a binary classification model. More precisely, the AUC ROC score measures the model's capability to distinguish between positive (PD) and negative (healthy participants) classes across all possible thresholds. A higher AUC ROC score indicates a better predictive performance.
The results of this study show (see Figure 2) that the random traveling order consistently outperformed the fixed traveling order across all experimental setups. As seen in Figures 2A, B, models trained for one and two local epochs employing the random traveling order achieved AUC ROC results comparable to the centralized model (80.57%) when trained for 24 cycles or more. In contrast, Figure 2C reveals that models trained for five local epochs exhibited inferior performance compared to the centralized model, regardless of the traveling order (detailed metrics per cycle and training scheme are presented in Supplementary Table 2).
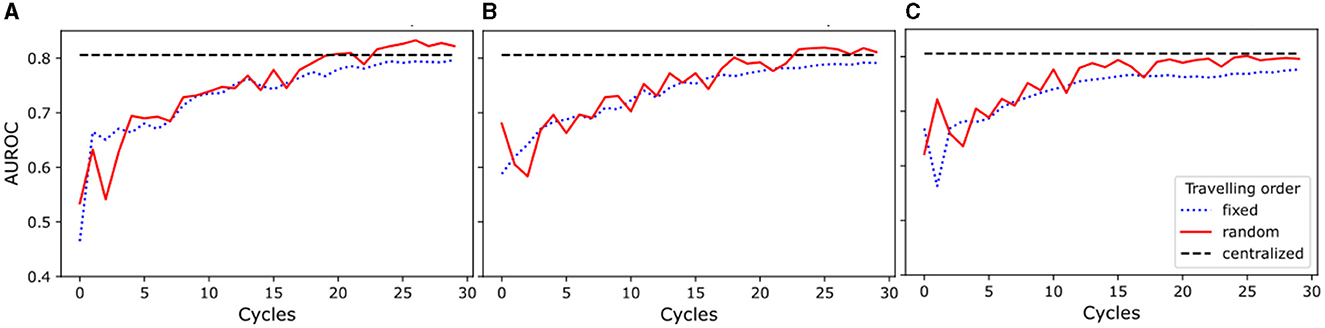
Figure 2. Area Under the Receiver Operating Characteristic Curve (AUROC) for experiments with random and fixed traveling order. The blue dotted line represents the results for the fixed traveling order, the red solid line represents the results for the random traveling order, and the black dashed line represents the best result for the centralized model AUROC after complete training. (A) Models trained for one local epoch. (B) Models trained for two local epochs. (C) Models trained for five local epochs.
Our results demonstrate that increasing the number of cycles improves the performance of the models for every setup investigated. Furthermore, our findings highlight that an increase in the number of local training epochs leads to greater instability (i.e., AUC ROC numbers vary between cycles) in the training process for the random traveling order, while conversely, it results in a smoother (i.e., less variability between cycles) trend for the fixed traveling order. Moreover, the model trained for a single local epoch and random traveling order displayed a more stable learning process when compared to the models that employed two and five local epochs and random traveling order. Additionally, Figure 2A shows a comparable level of stability in the training process for models trained with both fixed and random traveling orders for a single epoch.
The main finding of this work is that the traveling model is suitable and leads to good results when used for training CNN models for disease classification using 3D imaging data distributed across many medical centers and limited data availability at single centers. Most notably, when trained for an appropriate number of cycles, the model trained in this distributed way achieves comparable or slightly superior performance compared to a standard model that was trained in centralized fashion. Moreover, the effectiveness of the traveling CNN model extends beyond scenarios where centers contribute limited samples, as previously shown in Souza et al. (2022b) using tabulated data. It also performs exceptionally well in this real-world data context characterized by diverse forms of imaging data distribution imbalances, including variations in target labels, scanner types, and demographics, as evident in this unique and realistic PD database. Importantly, our findings affirm the practical applicability of the traveling model paradigm for image-based classification systems in real-world contexts, enhancing its reliability for clinical deployment across multiple medical facilities.
Our findings reveal that employing a random traveling order and a single local epoch constitutes the optimal configuration for PD classification using this particular database. This setup exhibits greater stability in performance as a function of cycles compared to scenarios involving two or five local epochs, suggesting that an excessive amount of local training could potentially lead to model overfitting to the data provided by individual centers. Souza et al. (2022b) reported similar findings in their analysis of the effects of local training per cycle. However, it is important to note that their study differed from ours as they utilized simulated artificial centers, identical data distribution, and tabular data, while we used image data acquired in 83 real-world centers with non-identical data distribution, and full 3D images instead of tabulated data. Similar outcomes were also demonstrated in Souza et al. (2022a,c), where brain age prediction and brain tumor segmentation were examined. Nevertheless, noteworthy distinctions should be emphasized. In one instance, contributions from centers were simulated using tabular data, while in the other case, a CNN for segmentation was utilized, which has access to numerous positive and negative cases (voxels) within a single image. As a result, this study provides novel and highly relevant contributions to the field, showcasing the suitability of the traveling models approach for image-based disease classification tasks using data acquired in 83 real-world centers.
The observed smoother performance trend of the fixed traveling order could be attributed to several factors. One potential explanation is that the model might memorize the sequence of centers and exploit it as a shortcut. Another possibility is that the model consistently encounters the same center toward the end of each cycle leading to overfitting. In contrast, using a random order helps mitigate (at least partially) the memorization of the center order or the last center's local samples by emulating the batch shuffling process employed in centralized training. Another perspective is that this smoother performance trend might relate to the phenomenon of catastrophic forgetting (French, 1999; Kirkpatrick et al., 2017), where the model loses knowledge of previously learned patterns from initial centers and becomes overly specialized toward later-center data. Lastly, the smoothness could imply that the model's weight updates become minimal in later cycles, leading to diminishing improvements. On the other hand, the increased instability seen in the random traveling order suggests more pronounced weight updates across cycles, likely contributing to the observed performance variability.
In essence, our study highlights the efficacy of the traveling model for 3D medical image classification applications, particularly when dealing with limited training samples. These insights carry important implications for scenarios in which small centers or remote medical facilities are meant to contribute data, cases involving rare diseases (Taruscio et al., 2018) with limited case numbers even at major centers, and situations where centers predominantly serve pediatric patients, with considerable developmental differences (Rahimzadeh et al., 2018). The simplicity of our approach makes it versatile and applicable to a wide array of deep learning tasks and databases, thereby enhancing its clinical utility across diverse contexts.
It is essential to highlight some of the limitations of this work. First, our work made exclusively use of a single established PD classifier model. Thus, it remains to be shown that the results hold true if different deep learning models or disease models are considered. Nevertheless, it is worth highlighting that the multi-center database utilized in this study is notably extensive and encompasses a considerably larger number of centers compared to datasets employed in numerous other federated learning and traveling model analyses thus far, which makes it likely that the results are generalizable. Second, our study solely employed T1-weighted MRI sequences, thereby leaving out the exploration of alternative image modalities. Third, this work only simulated the network for the traveling model pipeline using the multi-center database. Therefore, future work investigating how to create such a distributed computer network in practice and how to define transfer protocols to send the model to different locations to train in distinct computers is necessary. Nevertheless, the results presented in this work should hold true if every center trains the model using the same hard- and software that we used. Lastly, the establishment of a metric to investigate catastrophic forgetting is necessary to determine the underlying cause of the varying stability observed in our model's training process.
This work explored and systematically investigated the applicability of the traveling CNN model paradigm for distributed training of a PD classifier using data acquired in 83 real centers around the world, exhibiting considerable heterogeneity in the data distribution per center, with the majority of centers contributing only a limited number of imaging samples. To the best of our knowledge, this is the first work making use of a large database of 3D images from real centers with limited local data to train an image-based disease classifier in a distributed way. Moreover, this is the first description of a novel distributed learning approach, specifically designed and evaluated for PD classification. Our results demonstrated that the traveling CNN model can achieve results similar to central learning. Thus, the traveling model provides a new opportunity to apply machine learning models to diverse and skewed data distributions as a result of limited data availability at medical facilities.
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding author.
Each center received ethics approval from their local ethics board and received written informed consent from all the participants under the declaration of Helsinki. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation was not required from the participants or the participants' legal guardians/next of kin in accordance with the national legislation and institutional requirements.
RS: Conceptualization, Formal analysis, Funding acquisition, Investigation, Methodology, Visualization, Writing—original draft, Writing—review & editing. ES: Formal analysis, Writing—review & editing. MC: Data curation, Writing—review & editing. RC: Data curation, Writing—review & editing. OM: Data curation, Writing—review & editing. ZI: Data curation, Writing—review & editing. MW: Conceptualization, Formal analysis, Funding acquisition, Writing—review & editing. NF: Conceptualization, Formal analysis, Funding acquisition, Writing—review & editing.
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was supported by the Parkinson's Association of Alberta, the Hotchkiss Brain Institute, the Canadian Consortium on Neurodegeneration in Aging (CCNA), the Canadian Open Neuroscience Platform (CONP), the Natural Sciences and Engineering Research Council of Canada (NSERC), the Discovery Grant, the Canada Research Chairs program, the River Fund at Calgary Foundation, the Canadian Institutes for Health Research, the Tourmaline Chair in Parkinson's disease, and the Institut de valorisation des données (IVADO).
Data used in preparation of this article were obtained from the Alzheimer's Disease Neuroimaging Initiative (ADNI) database (adni.loni.usc.edu). As such, the investigators within the ADNI contributed to the design and implementation of ADNI and/or provided data but did not participate in analysis or writing of this report. A complete listing of ADNI investigators can be found at: http://adni.loni.usc.edu/wp-content/uploads/how_to_apply/ADNI_Acknowledgement_List.pdf.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/frai.2024.1301997/full#supplementary-material
Acharya, H. J., Bouchard, T. P., Emery, D. J., and Camicioli, R. M. (2007). Axial signs and magnetic resonance imaging correlates in Parkinson's disease. Can. J. Neurol. Sci. 34, 56–61. doi: 10.1017/S0317167100005795
Adnan, M., Kalra, S., Cresswell, J. C., Taylor, G. W., and Tizhoosh, H. R. (2022). Federated learning and differential privacy for medical image analysis. Sci. Rep. 12, 1953. doi: 10.1038/s41598-022-05539-7
Badea, L., Onu, M., Wu, T., Roceanu, A., and Bajenaru, O. (2017). Exploring the reproducibility of functional connectivity alterations in Parkinson's disease. PLoS ONE 12:e0188196. doi: 10.1371/journal.pone.0188196
Balachandar, N., Chang, K., Kalpathy-Cramer, J., and Rubin, D. L. (2020). Accounting for data variability in multi-institutional distributed deep learning for medical imaging. J. Am. Med. Inform. Assoc. 27, 700–708. doi: 10.1093/jamia/ocaa017
Camacho, M., Wilms, M., Mouches, P., Almgren, H., Souza, R., Camicioli, R., et al. (2023). Explainable classification of Parkinson's disease using deep learning trained on a large multi-center database of t1-weighted MRI datasets. Neuroimage Clin. 38:103405. doi: 10.1016/j.nicl.2023.103405
Cetinkaya, A. E., Akin, M., and Sagiroglu, S. (2021). “Improving performance of federated learning based medical image analysis in non-iid settings using image augmentation,” in 2021 International Conference on Information Security and Cryptology (ISCTURKEY) (Ankara: IEEE), 69–74. doi: 10.1109/ISCTURKEY53027.2021.9654356
Chang, K., Balachandar, N., Lam, C., Yi, D., Brown, J., Beers, A., et al. (2018). Distributed deep learning networks among institutions for medical imaging. J. Am. Med. Inform. Assoc. 25, 945–954. doi: 10.1093/jamia/ocy017
Duchesne, S., Chouinard, I., Potvin, O., Fonov, V. S., Khademi, A., Bartha, R., et al. (2019). The canadian dementia imaging protocol: harmonizing national cohorts. J. Magn. Reson. Imaging 49, 456–465. doi: 10.1002/jmri.26197
French, R. (1999). Catastrophic forgetting in connectionist networks. Trends Cogn. Sci. 3, 128–135. doi: 10.1016/S1364-6613(99)01294-2
Hanganu, A., Bedetti, C., Degroot, C., Mejia-Constain, B., Lafontaine, A.-L., Soland, V., et al. (2014). Mild cognitive impairment is linked with faster rate of cortical thinning in patients with Parkinson's disease longitudinally. Brain 137, 1120–1129. doi: 10.1093/brain/awu036
Jack, C. R., Bernstein, M. A., Fox, N. C., Thompson, P., Alexander, G., Harvey, D., et al. (2008). The Alzheimer's disease neuroimaging initiative (ADNI): MRI methods. J. Magn. Reson. Imaging 27, 685–691. doi: 10.1002/jmri.21049
Jiang, M., Wang, Z., and Dou, Q. (2022). “Harmofl: harmonizing local and global drifts in federated learning on heterogeneous medical images,” in Proceedings of the AAAI Conference on Artificial Intelligence (Palo Alto, CA: AAAI Press), 1087–1095. doi: 10.1609/aaai.v36i1.19993
Kirkpatrick, J., Pascanu, R., Rabinowitz, N., Veness, J., Desjardins, G., Rusu, A. A., et al. (2017). Overcoming catastrophic forgetting in neural networks. Proc. Natl. Acad. Sci. 114, 3521–3526. doi: 10.1073/pnas.1611835114
LaMontagne, P. J., Benzinger, T. L., Morris, J. C., Keefe, S., Hornbeck, R., Xiong, C., et al. (2019). Oasis-3: longitudinal neuroimaging, clinical, and cognitive dataset for normal aging and Alzheimer disease. medRxiv. doi: 10.1101/2019.12.13.19014902
Lang, S., Hanganu, A., Gan, L. S., Kibreab, M., Auclair-Ouellet, N., Alrazi, T., et al. (2019). Network basis of the dysexecutive and posterior cortical cognitive profiles in Parkinson's disease. Mov. Disord. 34, 893–902. doi: 10.1002/mds.27674
Li, X., Gu, Y., Dvornek, N., Staib, L. H., Ventola, P., Duncan, J. S., et al. (2020). Multi-site fMRI analysis using privacy-preserving federated learning and domain adaptation: abide results. Med. Image Anal. 65:101765. doi: 10.1016/j.media.2020.101765
Liu, Z., Wu, F., Wang, Y., Yang, M., and Pan, X. (2023). FEDCL: federated contrastive learning for multi-center medical image classification. Pattern Recognit. 143:109739. doi: 10.1016/j.patcog.2023.109739
McMahan, H. B., Moore, E., Ramage, D., Hampson, S., and Arcas, B. A. (2016). “Communication-efficient learning of deep networks from decentralized data,” in Proceedings of the 20th International Conference on Artificial Intelligence and Statistics, AISTATS 2017 (PMLR).
Ng, D., Lan, X., Yao, M. M.-S., Chan, W. P., and Feng, M. (2021). Federated learning: a collaborative effort to achieve better medical imaging models for individual sites that have small labelled datasets. Quant. Imaging. Med. Surg. 11, 852–857. doi: 10.21037/qims-20-595
Rahimzadeh, V., Schickhardt, C., Knoppers, B. M., Sénécal, K., Vears, D. F., Fernandez, C. V., et al. (2018). Key implications of data sharing in pediatric genomics. JAMA Pediatr. 172:476. doi: 10.1001/jamapediatrics.2017.5500
Souza, R., Aulakh, A., Mouches, P., Tuladhar, A., Wilms, M., Langner, S., et al. (2022a). “A comparative analysis of the impact of data distribution on distributed learning with a traveling model for brain age prediction,” in Medical Imaging 2022: Imaging Informatics for Healthcare, Research, and Applications, Volume 12037 (SPIE), 1. doi: 10.1117/12.2612728
Souza, R., Mouches, P., Wilms, M., Tuladhar, A., Langner, S., Forkert, N. D., et al. (2022b). An analysis of the effects of limited training data in distributed learning scenarios for brain age prediction. J. Am. Med. Inform. Assoc. 30, 112–119. doi: 10.1093/jamia/ocac204
Souza, R., Tuladhar, A., Mouches, P., Wilms, M., Tyagi, L., Forkert, N. D., et al. (2022c). “Multi-institutional travelling model for tumor segmentation in MRI datasets,” in Lecture Notes in Computer Science (including subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics) (Berlin: Springer), 420–432. doi: 10.1007/978-3-031-09002-8_37
Souza, R., Wilms, M., Camacho, M., Pike, G. B., Camicioli, R., Monchi, O., et al. (2023). Image-encoded biological and non-biological variables may be used as shortcuts in deep learning models trained on multisite neuroimaging data. J. Am. Med. Inform. Assoc. 30, 1925–1933. doi: 10.1093/jamia/ocad171
Sudlow, C., Gallacher, J., Allen, N., Beral, V., Burton, P., Danesh, J., et al. (2015). UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 12:e1001779. doi: 10.1371/journal.pmed.1001779
Talai, A. S., Sedlacik, J., Boelmans, K., and Forkert, N. D. (2021). Utility of multi-modal MRI for differentiating of Parkinson's disease and progressive supranuclear palsy using machine learning. Front. Neurol. 12:648548. doi: 10.3389/fneur.2021.648548
Taruscio, D., Vittozzi, L., Rocchetti, A., Torreri, P., and Ferrari, L. (2018). The occurrence of 275 rare diseases and 47 rare disease groups in Italy. Results from the national registry of rare diseases. Int. J. Environ. Res. Public Health. 15:1470. doi: 10.3390/ijerph15071470
Thibeau-Sutre, E., Couvy-Duchesne, B., Dormont, D., Colliot, O., and Burgos, N. (2022). “MRI field strength predicts Alzheimer's disease: a case example of bias in the ADNI data set,” in 2022 IEEE 19th International Symposium on Biomedical Imaging (ISBI), Volume 2022 (Kolkata: IEEE), 1–4. doi: 10.1109/ISBI52829.2022.9761504
Tuladhar, A., Rajashekar, D., and Forkert, N. D. (2022). Distributed Learning in Healthcare. Cham: Springer. doi: 10.1007/978-3-031-11199-0_10
Wei, D., Zhuang, K., Chen, Q., Yang, W., Liu, W., Wang, K., et al. (2018). Structural and functional MRI from a cross-sectional Southwest University Adult Lifespan Dataset (SALD). bioRxiv. doi: 10.1101/177279
Wicaksana, J., Yan, Z., and Cheng, K.-T. (2023). FCA: Taming Long-tailed Federated Medical Image Classification by Classifier Anchoring. Available online at: http://arxiv.org/abs/2305.00738
Xiao, Y., Fonov, V., Chakravarty, M. M., Beriault, S., Subaie, F. A., Sadikot, A., et al. (2017). A dataset of multi-contrast population-averaged brain MRI atlases of a Parkinson's disease cohort. Data Brief 12, 370–379. doi: 10.1016/j.dib.2017.04.013
Yan, B., Wang, J., Cheng, J., Zhou, Y., Zhang, Y., Yang, Y., et al. (2021). Experiments of federated learning for COVID-19 chest X-ray images. Commun. Comput. Inf. Sci. 1423, 41–53. doi: 10.1007/978-3-030-78618-2_4
Keywords: traveling model, federated learning, distributed learning, Parkinson's disease, multi-center
Citation: Souza R, Stanley EAM, Camacho M, Camicioli R, Monchi O, Ismail Z, Wilms M and Forkert ND (2024) A multi-center distributed learning approach for Parkinson's disease classification using the traveling model paradigm. Front. Artif. Intell. 7:1301997. doi: 10.3389/frai.2024.1301997
Received: 25 September 2023; Accepted: 23 January 2024;
Published: 07 February 2024.
Edited by:
Giorgio Maria Di Nunzio, University of Padua, ItalyCopyright © 2024 Souza, Stanley, Camacho, Camicioli, Monchi, Ismail, Wilms and Forkert. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Raissa Souza, cmFpc3NhLnNvdXphZGVhbmRyYWRAdWNhbGdhcnkuY2E=
†These authors share last authorship
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.