
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CASE REPORT article
Front. Anesthesiol. , 28 March 2025
Sec. Perioperative Medicine
Volume 4 - 2025 | https://doi.org/10.3389/fanes.2025.1535087
This article is part of the Research Topic Advancements and Challenges in Perioperative Medicine: 2023 View all 9 articles
 Brian A. Williams1,2*
Brian A. Williams1,2* Marsha E. Ritter1,2
Marsha E. Ritter1,2 Patrick J. Kennedy1,2
Patrick J. Kennedy1,2 Alissa M. Kmatz2
Alissa M. Kmatz2 Leath Abdullah2
Leath Abdullah2 Rick L. Scanlan2
Rick L. Scanlan2 Nicholas J. McNiffe3
Nicholas J. McNiffe3 William M. Fowler4
William M. Fowler4 Michael S. Patzkowski3,5
Michael S. Patzkowski3,5
The four individual cases presented include three clinical scenarios in which the operating surgeon, on behalf of the surgical patient, desired focal “conduction analgesia” without motor block to facilitate postoperative physical examination and/or the immediate advancement of physical therapy/ambulation objectives during early convalescence. The authors at two centers opted for available pharmacologic motor-sparing nerve blocks (entailing motor–sensory nerve fiber analgesia without using typical local anesthetics) as opposed to commonplace anatomic motor-sparing nerve blocks (involving local anesthetics that block sodium channels but do not target motor nerve fibers). Three of the four cases involved an acute postoperative timeline, while the fourth entailed an outpatient physical examination 6 months postoperatively in a patient with chronic, persistent surgical pain that was subsequently diagnosed with complex regional pain syndrome (CRPS). The intervention consisted of a four-medication motor-sparing nerve block (midazolam–buprenorphine–clonidine–dexamethasone), which had been previously reported. All four patients were satisfied with the short-term analgesic outcomes, and the surgeons' physical therapy and related objectives were met through the avoidance of motor block. In the latter case, which was later diagnosed as CRPS, the surgeon determined through physical examination that bony and soft-tissue healing was appropriate, obviating the need for repeat surgical exploration, thus allowing for an earlier referral to a chronic pain consultant for (ultimately) CRPS management. The described four-medication, preservative-free perineural combination, previously shown to be non-toxic in vitro and in vivo, may have important clinical applications when motor-sparing analgesia is desired in varying contexts and in cases of extreme postsurgical pain. The presentation of these four cases is followed by an aggregate data summary (n = 305 cases) of analgesic duration (mean 30.5 h, 95% CI 28.5–32.5 h) and pre-/postintervention peak pain scores (pre: mean 7.8/10 [95% CI 7.5–8.0]; post: mean 6.3/10 [95% CI 6.0–6.6]) after the described four-drug nerve block intervention, derived from quality improvement data at one of the two contributing author centers.
Motor-sparing perineural analgesia after orthopedic and related surgery has been challenging to achieve. One common type of surgery where surgeons and physical therapists typically desire as much motor-sparing analgesia as possible is arthroscopic anterior cruciate ligament reconstruction (ACLR). ACLR is a prevalent knee procedure performed on an outpatient basis, which represents a significant shift in clinical practice, as the authors (BAW and MSP) have extensive experience performing nerve blocks (e.g., femoral, sciatic) that were not motor-sparing (1–12). In more recent years, a meta-analysis (13) addressing ACLR again described the difficulty in managing associated postoperative pain, concluding that “adductor canal block, local infiltration analgesia and systemic analgesia alone each perform poorly for acute pain management” following ACLR. Balancing patient satisfaction with pain control and orthopedists’ singularly focused requests for unimpaired quadriceps function after ACLR [and similar or more invasive (14) procedures] should also address fall risk via maintenance of gross motor function and proprioception to the extent possible. Herein, we provide perspectives on motor-sparing analgesic block (MSAB) options using midazolam–buprenorphine–clonidine–dexamethasone (MDZ-BCD) for a future prospective study on ACLR and present two other cases in which MSABs with MDZ-BCD proved valuable to our surgical colleagues (and their patients): one involving acute surgical care after profound lower-extremity trauma, and another case 6 months after prior foot/ankle surgery.
MSABs can be achieved through two different mechanisms: (i) anatomic [such as adductor canal blocks (ACBs) and other techniques involving local anesthetics/infiltration] and (ii) pharmacologic (particularly with MDZ-BCD, as explained in the following). We agree with the findings of meta-analyses that anatomic MSABs do not provide meaningful analgesia for ACLR (13) and other invasive knee surgeries (14) and that prioritizing “knee function” over meaningful analgesia risks the development of hyperalgesia and opioid tolerance with dose escalation, with each adverse condition promoting the other. In addition, with conventional, local anesthetic-based ACBs, motor preservation cannot be guaranteed due to variable effects on the vastus medialis and potentially other quadriceps muscles (14). Pharmacologic MSABs using MDZ-BCD, meanwhile, entail no local anesthetics. In considering combinations of potential perineural analgesic adjuvants, a detailed literature review can be useful to determine what enhances the effect of local anesthetic analgesia and duration; these adjuvants can then be combined (ideally first ensuring the safety of the combination) while excluding the local anesthetic. After a detailed review of such adjuvants (15), we selected the MDZ-BCD combination based on basic science studies demonstrating its safety in small animal models, both in vitro [2011 (16)] and in vivo [2015 (17)], with no meaningful blockade of sodium channels reported mechanistically (thus suggesting the potential for motor preservation). Other pharmacologic options (e.g., dexmedetomidine) have not been previously tested in vitro or in vivo in combination with local anesthetics, dexamethasone, and buprenorphine; therefore, multimodal perineural safety of a dexmedetomidine-based combination could not be assumed or implied. Further, all MDZ-BCD agents are available (inexpensively) as preservative-free formulations. Midazolam was demonstrated in 2005 to both prolong the duration of nerve blocks and improve analgesic effects when combined with local anesthetics (18), but neurotoxicity safety concerns were identified in vitro when midazolam was combined with local anesthetics (16)—concerns that did not recur once the local anesthetic was removed from the admixture (16).
For several years, author BAW has used MDZ-BCD for MSABs after demonstrating laboratory safety (19) and drug compatibility (17) for single injections. We have previously reported outcomes associated with MDZ-BCD (20, 21), without any apparent complications (22). Herein, we present the first two ACLR cases in male Veterans (authors BAW/MER/PJK), a third case (authors NJM/WMF/MSP) from a level 1 trauma military treatment facility using the same MSAB MDZ-BCD strategy following profound lower-extremity trauma, and one final case (authors BAW/RLS/AMK/LA) involving foot-ankle surgery 6 months prior, where the MSAB technique with MDZ-BCD (along with appropriate negative imaging and a satisfactory physical examination) (i) avoided the need for exploratory revision surgery and (ii) facilitated earlier referral for treatment of a subsequently diagnosed complex regional pain syndrome (CRPS). Thereafter (in Sections 3 and 4), we summarize aggregate quality improvement (QI) case series data collected beyond the scope of the four cases presented.
The first ACLR case involved a 40-year-old Caucasian male Veteran with a medical history of traumatic brain injury and obstructive sleep apnea, presenting for a hamstring tendon autograft. He had no other relevant medical or demographic history. His reported preoperative baseline pain with movement was 7/10 on the verbal pain scale (VPS). He received a preoperative femoral MSAB consisting of 1 mg of midazolam, 2 mg of dexamethasone, 40 µg of clonidine, and 300 µg of buprenorphine, diluted in saline to a volume of 22 mL. He received spinal anesthesia with 1.8 mL of 0.75% hyperbaric bupivacaine. He was discharged home the same day, ambulating with crutches with partial weight-bearing, indicating satisfactory resolution of the motor block from bupivacaine spinal. He was contacted 72 h postoperatively to discuss MSAB pain management. We were unable to determine how much, if any, oxycodone the patient took after discharge; however, he reported his worst pain with movement as 6/10 on the VPS (i.e., lower than his preoperative VPS) 48 h after receiving the MSAB with MDZ-BCD. Three years after this case, when agreeing to be presented in case report format to professional literature, he did not voice any concerns regarding long-term side effects from the MDZ-BCD block.
The second ACLR case involved a 33-year-old Caucasian male Veteran with a medical history of obesity, migraine headaches, and tobacco use, presenting for quadriceps tendon autograft. There was no other relevant medical or demographic history. With his migraine history possibly contributing to his postoperative nausea/vomiting (PONV) risk, we opted to use the MSAB with MDZ-BCD to minimize postoperative opioid burden (as a PONV trigger). Additionally, due to his obesity and the associated fall risk, we recommended the MDZ-BCD approach. Preoperatively, he reported his baseline pain with movement as 3/10 on the VPS. The patient received a femoral MSAB consisting of 1.5 mg of midazolam, 40 µg of clonidine, 2 mg of dexamethasone, and 300 µg of buprenorphine in 44 mL of saline. His spinal anesthetic consisted of 1.7 mL of 0.75% hyperbaric bupivacaine. As with Case 1, he was discharged home the same day, ambulating with crutches and partial weight-bearing, indicating satisfactory resolution of any motor block from bupivacaine spinal. During a telephone follow-up 72 h postoperatively, he reported that the MSAB lasted for 31 h, with his worst (rebound) pain with movement being 8/10 on the VPS at that time. Four years after this case when agreeing to be presented in case report format to professional literature, he did not voice any concerns regarding long-term side effects from the MDZ-BCD block.
Author BAW had frequently noted (during the two cases described previously and in the aggregate of n = 305 cases presented in the following) a tendency for patients to complain of a focal, mild burning sensation not typically observed in patients experiencing nerve blocks (while awake) with usual local anesthetics. The likely rationale for this phenomenon is the lack of local anesthetics in the described MDZ-BCD mixture, serving to “auto-numb” the injection site (whether the MDZ-BCD burning sensation originated from pH differences and/or from multi-milliliter volume-induced pressure sensations). Experience taught that early verbal instruction and reassurance, along with slightly more intravenous sedation at the time of the block, were typically useful mitigation strategies.
It is our collective experience that orthopedic surgeons are not concerned with a motor block during the surgery itself, explaining the popularity of spinal anesthesia in more invasive cases such as knee and hip arthroplasty. We have not encountered surgeons or physical therapists raising concerns about the motor function return after using spinal anesthesia that resolves only a few minutes or hours after the procedure is completed.
The third case involved a 20-year-old civilian man of Latino/Hispanic ethnicity, with no medical history, who was admitted through the trauma service following an accidental self-inflicted gunshot wound to the thigh. His alcohol and drug screens were positive. On visual examination, there was a 1-cm-diameter entrance wound on the anterior proximal thigh and a 1-cm-diameter exit wound on the posterior lateral thigh. A physical examination was otherwise unremarkable, with intact vasculature, motor, and neural function. Radiographic imaging revealed a severely comminuted subtrochanteric ballistic fracture of the femur. The patient was placed in traction in anticipation of intramedullary nail fixation. In the 12 h between hospital admission and the start of surgery, he received a total of 1,650 mg of oral acetaminophen, 800 mg of oral ibuprofen, 150 mcg of intravenous fentanyl, 1 mg of intravenous hydromorphone, and 5 mg of oral oxycodone. Despite these analgesics, his preoperative VPS scores consistently ranged from 6 to 8 out of 10. The staff orthopedic trauma surgeon declined a local anesthetic-based peripheral nerve block because of concerns about delayed postoperative mobilization; however, after further discussion, the surgical team was amenable to a pharmacologic MSAB (as an anatomic MSAB would have been distal to the site of injury). After general anesthesia was induced and before surgery commenced, the regional anesthesia service performed single injections of femoral (23 mL of injectate) and lateral femoral cutaneous nerve (7 mL of injectate) MSAB with MDZ-BCD, which were ultrasound-guided and injection-pressure-monitored. The MSAB drug mixture comprised of a preservative-free, saline-diluted 30 mL solution containing 1 mg of midazolam, 50 µg of clonidine, 200 µg of buprenorphine, and 2 mg of dexamethasone. The total surgical time was 3 h, and the patient showed no nociceptive tendencies that required intravenous dosing of additional analgesics/opioids intraoperatively after his nerve blocks. Following arrival to the postanesthesia care unit (PACU), the patient denied experiencing operative site pain and demonstrated no motor blockade. On command, he was able to perform an operative straight leg raise with full knee extension, elevating his heel >2 in. off the bed. The maximum reported pain score during the 2 days of his postoperative inpatient admission was 5/10 on the VPS. He confidently reported the duration of perineural analgesia lasting >24 h before complete resolution. The patient did not receive any postoperative analgesics until 8 h after the MSAB injections, at which time he received (as needed) 5 mg of oral oxycodone. He was able to independently ambulate with axillary crutches, meeting discharge criteria on postoperative day 2, with no complications. At his 6-week postdischarge follow-up, he reported no sensory deficits and was walking up to approximately 2 miles per day, with minimal quadriceps cramping or associated discomfort.
The fourth and final case, utilizing this time an outpatient MSAB, involved a 48-year-old Caucasian female Veteran. Her foot and ankle surgical history entailed hardware removal and revision of a talonavicular joint arthrodesis 6 months before the referent outpatient clinic visit, with new complaints of “aggressive” ankle spasms and twitching, which led to an inability to (i) concentrate during the daytime and (ii) sleep at night. The ankle twitches, per podiatrist coauthor RLS, were characterized as continual spasmodic motions involving the firing of both peroneus brevis and peroneus longus muscles. Following the MSAB with MDZ-BCD, the patient continued to experience some spasmodic motion of the ankle, but there was a decrease in pain in both the foot and ankle, with movement and during physical examination, which showed that the previously fused talonavicular joint had no motion (as expected) and there was some attenuation in the anterior tibial tendon along its distal course. After the MSAB procedure, during a physical examination by the attending podiatrist (the examination had not been possible earlier while in the podiatry clinic), the muscle strength of the plantar flexors was graded as 5+/5, while dorsiflexion/inversion/eversion strength was graded 4+/5.
The unusual spasms accentuated focal somatic (and other) pain manifestations in the involved lower extremity. Therefore, we recorded movement pain separately: during spasms and during weight-bearing activities, both before and after the MSAB procedure. The MSAB injection prepared (30 mL, diluted with normal saline) consisted of 1.5 mg of midazolam, 65 µg of clonidine, 4 mg of dexamethasone, and 600 µg of buprenorphine, with all described components being preservative-free. During the stimulator-guided injections, 22 mL was administered to the popliteal sciatic plexus (when both tibial and peroneal twitches were elicited) and 8 mL was injected into the saphenous at the groin (guided by a proxy twitch response to the vastus medialis in the absence of any twitch response from the vastus intermedius/rectus femoris entailing a patellar snap). The prepopliteal block pain scores of the patient were “11”/10 (during spasms), 10/10 (during “gas-pedal” weight-bearing activities), and 8/10 (at rest), while the immediate postpopliteal block pain scores were initially 8/10, 5/10, and 4/10, respectively. With the popliteal block (placed while the patient was in the prone position), followed by the subsequent saphenous/groin block performed (while the patient was supine), the final post-MSAB pain scores were 2/10, 2/10, and 1/10, respectively, after the saphenous procedure and after further “soaking” of the earlier popliteal block. The podiatrist's examination after the described MSAB procedures confirmed both prompt onset and sustained immediate effect: the patient rated her postblock pain as 1–2/10, even when including manual muscle testing activity (with only slight pain increases to 2–3/10). When contacted later, the patient estimated an analgesic duration of ∼26 h, after which the pain scores returned to a 9/10 at rest and during spasms. The patient was ultimately diagnosed with CRPS based on the Budapest criteria (23), supported by the following findings on history and physical examination. For sensory criteria, the patient reported allodynia, along with pain induced by range-of-motion testing of the affected ankle. For vasomotor criteria, examination revealed that the affected foot appeared more erythematous than the non-affected foot, and the patient endorsed periodic color and temperature changes. With regard to edema, although there was no visible or palpable edema on examination, the patient unequivocally endorsed periodic edema. Finally, for motor criteria, the patient endorsed both muscle weakness and occasional tremors (distinct from the overlying twitch response), and the range of motion was present on examination at the ankle and appeared unrelated to prior surgeries. After the CRPS diagnosis, the patient was referred for intermittent ketamine infusions (at the first center) as the chronic pain management plan; she has since completed six consecutive outpatient sessions 3–5 weeks apart (as of this writing, January 23, 2025), during which she has not reported any complaints regarding the CRPS-affected foot and ankle related to the MSAB MDZ-BCD block. Each ketamine encounter, with escalating lower doses up to a plateau dose of 500 mg/encounter, has had 2–4 weeks of successful analgesia/antihyperalgesia. With a further, partially unrelated medical history of alcohol misuse in remission and bulimia, we were grateful for the more rapid diagnosis and treatment plan (for CRPS) and the avoidance of a re-exploratory and/or revision foot and ankle surgery.
Importantly, the attending podiatrist was physically present during the MSAB procedure and was able to examine the patient's foot and ankle immediately, after previously being unable to examine the patient in the podiatry clinic physical plant space due to extreme withdrawal pain.
Following up on the first author's previous reports (20–22), we now present updated cumulative pharmacologic MSAB data addressing analgesic duration and peak pain scores for cases where motor-sparing analgesia was deemed desirable, in which conventional oral analgesics were deemed likely to be unsatisfactory or posed undue risks (e.g., past opioid use disorder and not wanting to re-expose patients to oxycodone). With regard to patient selection for the MSAB cumulative data analysis, MDZ-BCD block cases were extracted from the lead author's institutional QI dataset dating back to July 1, 2011, including case series previously published (20–22), after institutional approval of QI data analysis at the lead author's institution (author BAW). These pharmacologic motor-sparing blocks did not include local anesthetics; instead, they used MDZ-BCD. The types of blocks are specified in Table 1, along with baseline pain scores with movement, peak pain scores after perceived analgesic resolution, and the number of hours between block insertion and patient-estimated timing of the peak postoperative pain score (i.e., subtracting the latter date/time from the insertion date/time). Figure 1 illustrates the concepts of analgesic duration and peak pain score to facilitate an understanding of the data in Table 1. The methodology for preoperative and peak postblock pain measurements, as well as analgesic duration calculations, has been previously published elsewhere (20, 24) and can be summarized as follows. The time of the block was designated as “time zero”; prior to this, a baseline pain score (with movement) was elicited via routine patient queries and documented in the electronic medical record (EMR). At the lead center, outpatients discharged home on the same day after receiving any nerve blocks were routinely contacted a minimum of 2 days (3 days on a Friday-to-Monday discharge) after the block, to elicit peak pain score (with movement) at the declared time of perceived block dissipation from a pain perspective (since these were motor-sparing blocks). Continuing, lead center inpatients underwent an EMR review postoperatively for QI purposes, commonly by author BAW or a designee, and hospital-recorded pain scores were examined using a linear graph that was automated in the EMR. The peak pain scores were cross-correlated with initial opioid dosing from the EMR's medication administration record.
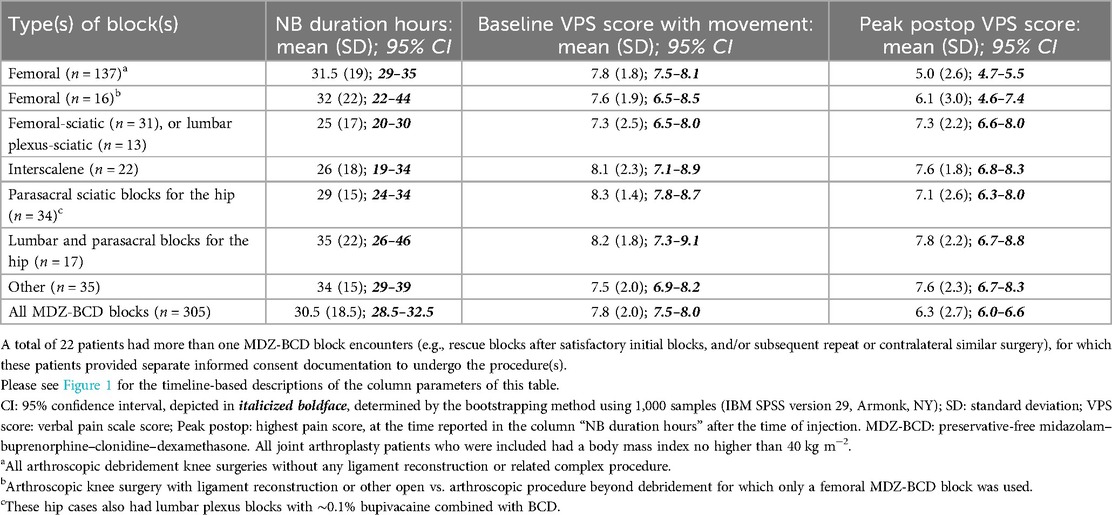
Table 1. Pharmacologic motor-sparing blocks with preservative-free midazolam–buprenorphine–clonidine–dexamethasone at the VA Pittsburgh Medical Center, United States (2011–2020, for whom complete data were available for all three listed parameters).
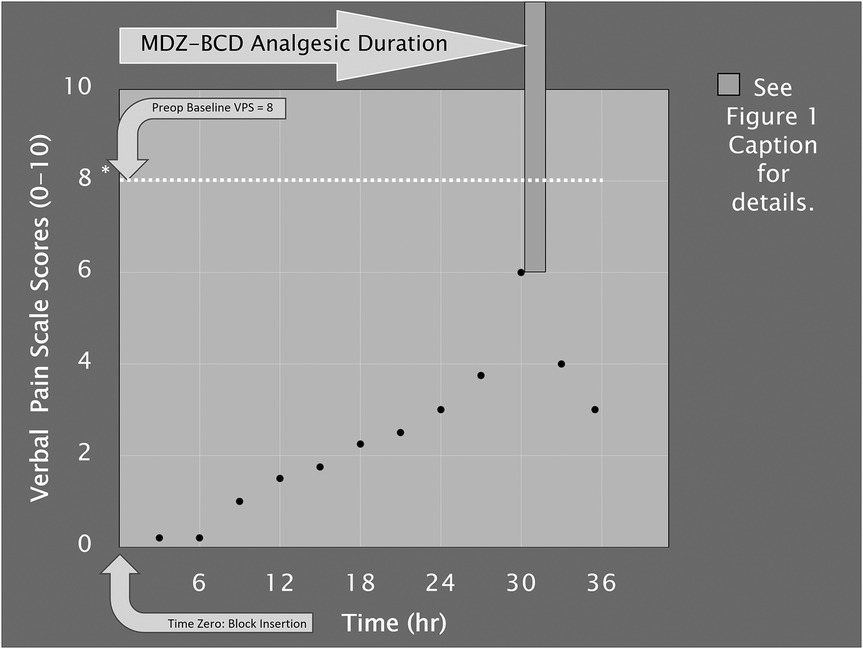
Figure 1. Schematic representation of analgesic duration (X-axis) of the motor-sparing analgesic block (MSAB) with combined perineural midazolam–buprenorphine–clonidine–dexamethasone (MDZ-BCD) in a single theoretical patient. The preoperative verbal pain scale (VPS, white dotted line, and upper-figure, curved, light-gray arrow) score for this patient is quantified as 8 out of 10 before the block and is documented on the electronic medical record (EMR). Time zero (lower-figure, curved, light-gray arrow) just below the X-axis represents the time of insertion of the MSAB MDZ-BCD block. The series of black dots represent interval VPS scores from the EMR (from 0 to 10, Y-axis) if the patient were an inpatient. Outpatients only reported their peak pain scores during a postoperative nursing phone call, which were then entered into the EMR, per the first center's clinical care routine. The vertical darker-gray bar projecting from the top of the figure to the 30-h timepoint represents two concepts. First, this marks the endpoint of meaningful analgesic duration, coinciding with the postoperative peak pain score. Next, by reaching down to the stated peak pain score (of 6 out of 10) and intersecting with the dotted line (at 8 out of 10), this represents the preoperative VPS reference score, from which the relative rebound pain (for peak VPS score = 6) calculates as 6 minus 8, or −2, being 2 points lower than the baseline VPS score. The MDZ-BCD block, therefore, has a calculated duration of 30 h, depicted with the straight, light-gray arrow. These concepts are helpful in the interpretation of Table 1, and this methodology has been used and published on several occasions. As a further nuanced explanation of this methodology, if there were two “tied” peak pain scores with an analgesic “trough” seen on the curve between the “two peaks,” with the first peak being followed by oral opioid and the next peak being followed by intravenous opioid, then the latter pain peak would be counted as the end-duration point. For most cases, both inpatients and outpatients (during the quality improvement query) had a progress note added to the medical record (typically by author BAW), replicating the methodology of block duration and rebound pain determinations previously reported (17, 18, 20, 23).
MDZ-BCD appeared to be associated with longer-duration analgesia than would be expected from plain local anesthetics such as ropivacaine or bupivacaine (31 h vs. 12–16, Table 1), but neither center had cases involving plain local anesthetics for incorporating case-matching comparisons. Surprisingly, the overall reported peak pain score at the first center [6.3 on a 0–10 scale; 95% confidence interval (CI) 6.0–6.6] appeared to be significantly lower than the baseline pain score with movement (7.8; 95% CI 7.5–8.0) before the MDZ-BCD MSAB was placed, as illustrated by the non-overlapping 95% CIs.
Treating pain associated with ACLR is challenging, and ACLR is an uncommon procedure in the lead author's Veterans hospital. Therefore, we presented updated QI data addressing analgesic duration and peak pain scores (Table 1) for cases in which the MSAB with MDZ-BCD was deemed desirable. Local anesthetic nerve blocks produce a motor blockade that orthopedic surgeons commonly wish to avoid, while anatomic motor-sparing blocks, in serial network meta-analyses, appeared to provide clinically inadequate analgesia (13, 14). It seems conceivable that if the ACB does not provide meaningful analgesia for ACLR (13), then the ACB would also not provide meaningful analgesia for knee arthroplasty, even when supplemented by infiltration of the interspace between the popliteal artery and the posterior capsule of the knee (iPACK), which indeed was confirmed by the same authors using a similar network meta-analysis methodology, published in the same year (14). MSABs appear to provide significant pharmacologic analgesic potential where anatomic motor-sparing blocks have repeatedly (13, 14) been shown to underperform. In short, some knee procedures generate more pain than others, depending on femoral vs. sciatic distribution and associated anatomic trespass (2, 25) during surgery. There may be value in pharmacologically addressing entire nerve distributions for motor-sparing blocks instead of hoping that using local anesthetics targeting sensory-only nerve compartments (such as the ACB and iPACK, while avoiding motor and sensorimotor nerve bundles and segments) are good enough despite repeated evidence (13, 14) to the contrary.
There are multiple proposed benefits to the use of the pharmacologic MSAB (e.g., with MDZ-BCD) in orthopedic surgery: most notably, the absence of a motor blockade that allows for immediate ambulation, which may also decrease fall risk while maintaining the ability to perform a postoperative neurologic (e.g., rule out compartment syndrome) and/or neuromuscular examination. This is particularly helpful in the following cases: examination of (i) the tibial nerve after posterior cruciate ligament reconstruction, (ii) the common peroneal nerve after posterolateral corner reconstruction, (iii) axillary and musculocutaneous nerves after open shoulder surgery (Latarjet, in particular), and (iv) the posterior interosseous nerve after distal biceps repair. In each of these surgeries, the aforementioned nerves are in close proximity to the surgical procedure, and nerve damage remains a commonly cited risk of the surgery itself. For many orthopedic surgeons, it is the inability to examine the motor function of these nerves after surgery that supports a conviction to decline regional anesthesia, at the acknowledged detriment of meaningful postoperative analgesia.
MSABs with MDZ-BCD offer a promising analgesic option for injuries and surgeries that carry a high risk of compartment syndrome. This manuscript's second center has used such blocks, for example, for major trauma and arthritis settings such as tibial plateau fracture, tibial tubercle avulsion fracture (and its repair), both-bone forearm fracture (i.e., radius-ulna), high tibial osteotomy, distal femoral osteotomy, and tibial tubercle osteotomy. These surgeries themselves are considered among the most painful in orthopedics, and the lack of regional anesthesia may lead surgeons to increasingly rely on largely inadequate and risk-laden opioid-focused pain strategies.
In MDZ-BCD MSAB cases from the first center, the analgesic duration (31 h, Table 1) exceeded what would be expected from plain local anesthetics (i.e., ropivacaine or bupivacaine, 12–16 h). The overall reported peak VPS score (∼6 on a 0–10 scale) was lower than baseline VPS scores with movement (∼8 on the same 0–10 scale). The extended analgesia provided by low-concentration midazolam (as part of MDZ-BCD, without local anesthetics) may be related to selective C-fiber inhibition (26); at these concentrations, midazolam does not affect sodium channels, yielding minimal motor effects (26). We encourage the use of quantitative sensory and motor testing, following MDZ-BCD MSAB administration as a substitute for local anesthetics, to determine the potential for significantly reduced (if any) motor weakness. Authors from the first center have previously reported favorable same-day ambulation outcomes after lower-extremity joint arthroplasty when using the MSAB with MDZ-BCD (21).
Clonidine, buprenorphine, and dexamethasone have long-standing and well-documented histories of their use as adjuvants to local anesthetic nerve blocks (Table 2) (20–22, 24, 27–33); however, midazolam's history is more limited when it comes to published studies (34). Midazolam, even in low concentrations, leads to a significant increase in neurotoxicity when combined with local anesthetics, but it does not appear to be neurotoxic on its own at the described concentrations (16). Midazolam can work as an analgesic on its own and (perineurally) is relatively selective for C-fibers, with a smaller, short-acting portion affecting A-fibers (35). This attenuation of compound action potentials occurs at clinically relevant concentrations, with no influence on potency/efficacy by the combination of clonidine, buprenorphine, or dexamethasone (26). Midazolam alone provides a clinically motor-sparing neuromodulatory effect by reducing nociceptor transmission.
Although the lead author's original (36) concerns later forecasting (19) MDZ-BCD not being sufficiently potent for major analgesia in lower-extremity orthopedic procedures, our third case presented herein (from our second center of interest in pharmacologic MSABs) indeed illustrated the potential of the MDZ-BCD combination in maximizing the sensory-specific neuromodulation while still providing clinically meaningful analgesia.
Further research is warranted, and many avenues are worthy of clinical investigation. Primarily, confirmation of the MDZ-BCD motor-sparing profile beyond subjective physical examinations, objective motor function testing of velocity, position, and torque using a validated device, such as a dynamometer, is needed and would significantly facilitate surgeon acceptance of pharmacologic MSABs, when early motion is imperative. Additionally, further work is needed to describe the magnitude of analgesia provided for different levels of tissue disruption: (i) skin and subcutaneous tissue (such as in varicose vein and arteriovenous graft surgery); (ii) tendon, ligament, and muscle tissue (such as in rotator cuff repair and knee ligament reconstruction surgery); and (iii) osseous tissue (such as in arthroplasty, osteotomy, and fracture fixation surgery). This would inform the proper selection of local anesthetic vs. pharmacologic MSAB regional anesthesia techniques to achieve balanced analgesic and motion outcomes. Uniquely and relevant to orthopedic trauma patients is the impact on patients who are at high risk from compartment syndrome, whether from injury or subsequent surgical trespass; such patients are often denied regional analgesia/anesthesia for fear of masking diagnostic symptoms. The MDZ-BCD combination may provide potential answers for surgical procedures or patient populations that would either benefit from or outright require motor-sparing analgesia and is therefore ripe for further clinical investigation.
This case series has several acknowledged limitations. First, while the aggregated cases (n = 305) span different clinical situations over several years, only four cases were discussed in detail, perhaps limiting generalizability. Additionally, at the lead center, we were (and are) unable to compare MDZ-BCD with plain local anesthetics or with motor-sparing techniques (such as ACB) in a controlled setting due to case mix and other factors. Next, while the absence of motor block was emphasized, potential side effects of the four-drug combination were not obtainable (other than expected, although rare, periprocedural sedation from either midazolam, buprenorphine, or clonidine combined with the subsequent anesthetic encounter, and these patients emerging from anesthesia with less pain than would otherwise be expected, possibly augmenting sedative effects, resulting in corresponding respiratory depression and/or delayed extubation, when applicable). Further, long-term outcomes beyond immediate analgesic effects were not extensively discussed other than in the four presented detailed cases. One case (not presented in the four-case narrative), more than a decade before this current writing, encountered surgical foot–ankle neuralgia after a podiatry procedure (not involving MDZ-BCD); after conventional therapies failed over the course of 1 year, author RLS referred the patient to author BAW, who administered a series of four MDZ-BCD popliteal sciatic injections, spread out over the latter half of the following year, over a 5-month period. This patient appeared to relocate his home after these encounters, but he did return for three encounters after the initial rescue effort. Finally, we acknowledge the absence of a case-matched control group receiving usual opioid-based care without any involvement of regional anesthesia procedures.
To conclude, MSABs with MDZ-BCD emphasize pharmacologic over anatomic motor-sparing principles. The described and presented pharmacologic analgesic non-motor blocks, encompassing the entire applicable distribution of a nerve or plexus, may prove to satisfy all stakeholders compared with limited sensory nerve compartments that do not specifically carry motor or sensorimotor nerve bundles, perhaps providing meaningful opioid-sparing analgesia after surgery of the extremities.
The original contributions presented in the study are included in the article and further inquiries can be directed to the corresponding author.
Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
BW: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Supervision, Validation, Writing – original draft, Writing – review & editing. MR: Investigation, Supervision, Writing – original draft, Writing – review & editing. PK: Data curation, Investigation, Methodology, Writing – review & editing. AK: Data curation, Investigation, Writing – review & editing. LA: Data curation, Investigation, Methodology, Writing – review & editing. RS: Data curation, Investigation, Methodology, Writing – review & editing. NM: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Writing – original draft, Writing – review & editing. WF: Data curation, Formal analysis, Investigation, Methodology, Writing – review & editing. MP: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Resources, Supervision, Writing – original draft, Writing – review & editing.
The authors declare that no financial support was received for the research and/or publication of this article.
The authors are grateful to our Veterans and Active Military for their service and for their understanding of the described maneuvers as a means to offset the risks of the opioid epidemic, such that these issues sufficiently resonated to receive the described procedures as part of our local recommended standards of care. The authors are also grateful for the manuscript review and orthopedic surgery-specific insights of (and comments from) Jeanne Patzkowski, MD (LTC, MC, USA), Chief, Department of Orthopaedic Surgery, Brooke Army Medical Center, Fort Sam Houston, TX, USA.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. Patzkowski MS, Scott SS, Patzkowski JC, Highland KB. Femoral nerve blockade does not lead to subjective functional deficits after anterior cruciate ligament reconstruction. Mil Med. (2022) 187:e644–8. doi: 10.1093/milmed/usab269
2. Williams BA, Kentor ML, Vogt MT, Williams JP, Chelly JE, Valalik S, et al. Femoral-sciatic nerve blocks for complex outpatient knee surgery are associated with less postoperative pain before same-day discharge: a review of 1200 consecutive cases from the period 1996–1999. Anesthesiology. (2003) 98:1206–13. doi: 10.1097/00000542-200305000-00024
3. Williams BA, Kentor ML, Vogt MT, Vogt WB, Coley KC, Williams JP, et al. The economics of nerve block pain management after anterior cruciate ligament reconstruction: significant hospital cost savings via associated PACU bypass and same-day discharge. Anesthesiology. (2004) 100:697–706. doi: 10.1097/00000542-200403000-00034
4. Williams BA, Kentor ML, Williams JP, Vogt MT, DaPos SV, Harner CD, et al. PACU bypass after outpatient knee surgery is associated with fewer unplanned hospital admissions but more phase II nursing interventions. Anesthesiology. (2002) 97:981–8. doi: 10.1097/00000542-200210000-00034
5. Williams BA, Kentor ML, Williams JP, Figallo CM, Sigl JC, Anders JW, et al. Process analysis in outpatient knee surgery: effects of regional and general anesthesia on anesthesia-controlled time. Anesthesiology. (2000) 93:529–38. doi: 10.1097/00000542-200008000-00033
6. Williams BA, Kentor ML, Vogt MT, Irrgang JJ, Bottegal MT, West RV, et al. Reduction of verbal pain scores after anterior cruciate ligament reconstruction with two-day continuous femoral nerve block: a randomized clinical trial. Anesthesiology. (2006) 104:315–27. doi: 10.1097/00000542-200602000-00018
7. Williams BA, Vogt MT, Kentor ML, Figallo CM, Kelly MD, Williams JP. Nausea and vomiting after outpatient ACL reconstruction with regional anesthesia: are lumbar plexus blocks a risk factor? J Clin Anesth. (2004) 16:276–81. doi: 10.1016/j.jclinane.2003.09.008
8. Williams BA, Kentor ML, Irrgang JJ, Bottegal MT, Williams JP. Nausea, vomiting, sleep, and restfulness upon discharge home after outpatient anterior cruciate ligament reconstruction with regional anesthesia and multimodal analgesia/antiemesis. Reg Anesth Pain Med. (2007) 32:193–202. doi: 10.1016/j.rapm.2006.12.002
9. Williams BA, Kentor ML, Bottegal MT. The incidence of falls at home in patients with perineural femoral catheters: a retrospective summary of a randomized clinical trial. Anesth Analg. (2007) 104:1002. doi: 10.1213/01.ane.0000256006.46703.7f
10. Williams BA, Dang Q, Bost JE, Irrgang JJ, Orebaugh SL, Bottegal MT, et al. General health and knee function outcomes from 7 days to 12 weeks after spinal anesthesia and multimodal analgesia for anterior cruciate ligament reconstruction. Anesth Analg. (2009) 108:1296–302. doi: 10.1213/ane.0b013e318198d46e
11. Williams BA, Bottegal MT, Kentor ML, Irrgang JJ, Williams JP. Rebound pain scores as a function of femoral nerve block duration after anterior cruciate ligament reconstruction: retrospective analysis of a prospective, randomized clinical trial. Reg Anesth Pain Med. (2007) 32:186–92. doi: 10.1016/j.rapm.2006.10.011
12. Mauro CS, Irrgang JJ, Williams BA, Harner CD. Loss of extension following anterior cruciate ligament reconstruction: analysis of incidence and etiology using IKDC criteria. Arthroscopy. (2008) 24:146–53. doi: 10.1016/j.arthro.2007.08.026
13. Hussain N, Brull R, Vannabouathong C, Speer J, Lagnese C, McCartney CJL, et al. Network meta-analysis of the analgesic effectiveness of regional anaesthesia techniques for anterior cruciate ligament reconstruction. Anaesthesia. (2023) 78(2):207–24. doi: 10.1111/anae.15873
14. Hussain N, Brull R, Vannabouathong C, Robinson C, Zhou S, D’Souza RS, et al. Analgesic effectiveness of motor-sparing nerve blocks for total knee arthroplasty: a network meta-analysis. Anesthesiology. (2023) 139:444–61. doi: 10.1097/ALN.0000000000004667
15. Williams BA, Murinson BB, Grable BR, Orebaugh SL. Future considerations for pharmacologic adjuvants in single-injection peripheral nerve blocks for patients with diabetes mellitus. Reg Anesth Pain Med. (2009) 34:445–57. doi: 10.1097/AAP.0b013e3181ac9e42
16. Williams BA, Hough KA, Tsui BY, Ibinson JW, Gold MS, Gebhart GF. Neurotoxicity of adjuvants used in perineural anesthesia and analgesia in comparison with ropivacaine. Reg Anesth Pain Med. (2011) 36:225–30. doi: 10.1097/AAP.0b013e3182176f70
17. Williams BA, Butt MT, Zeller JR, Coffee S, Pippi MA. Multimodal perineural analgesia with combined bupivacaine-clonidine-buprenorphine-dexamethasone: safe in vivo and chemically compatible in solution. Pain Med. (2015) 16:186–98. doi: 10.1111/pme.12592
18. Jarbo K, Batra YK, Panda NB. Brachial plexus block with midazolam and bupivacaine improves analgesia. Can J Anaesth. (2005) 52:822–6. doi: 10.1007/BF03021776
19. Knight JB, Schott NJ, Kentor ML, Williams BA. Neurotoxicity of common peripheral nerve block adjuvants. Curr Opin Anaesthesiol. (2015) 28:598–604. doi: 10.1097/ACO.0000000000000222
20. Williams BA, Ibinson JW, Mangione MP, Scanlan RL, Cohen PZ. Clinical benchmarks regarding multimodal peripheral nerve blocks for postoperative analgesia: observations regarding combined perineural midazolam-clonidine-buprenorphine-dexamethasone. Pain Med. (2015) 16:1–6. doi: 10.1111/pme.12599
21. Williams BA, Kennedy PJ, Ritter ME, Fourman MS, Nettrour JF. Pain and rehabilitation parameters after hip replacement using differing multimodal perineural analgesia strategies: a quality improvement series. Pain Med. (2021) 22:1455–8. doi: 10.1093/pm/pnaa384
22. Williams BA, Ibinson JW, Gould AJ, Mangione MP. The incidence of peripheral nerve injury after multimodal perineural anesthesia/analgesia does not appear to differ from that following single-drug nerve blocks (2011−2014). Pain Med. (2017) 18:628–36. doi: 10.1093/pm/pnv107
23. Harden R, Bruehl S. Diagnostic criteria: the statistical derivation of the four criterion factors. In: Wilson PR, Stanton-Hicks M, Harden R, editors. CRPS: Current Diagnosis and Therapy. Seattle, WA, USA: IASP Press (2005). p. 45–58.
24. Williams BA, Ibinson JW, Mangione MP, Modrak RT, Tonarelli EJ, Rakesh H, et al. Research priorities regarding multimodal peripheral nerve blocks for postoperative analgesia and anesthesia based on hospital quality data extracted from over 1,300 cases (2011−2014). Pain Med. (2015) 16:7–12. doi: 10.1111/pme.12609
25. Williams BA, Spratt D, Kentor ML. Continuous nerve blocks for outpatient knee surgery. Tech Reg Anesth Pain Manage. (2004) 8(2):76–84. doi: 10.1053/j.trap.2004.06.001
26. Yilmaz-Rastoder E, Gold MS, Hough KA, Gebhart GF, Williams BA. Effect of adjuvant drugs on the action of local anesthetics in isolated rat sciatic nerves. Reg Anesth Pain Med. (2012) 37:403–9. doi: 10.1097/AAP.0b013e3182485965
27. Williams BA, Ibinson JW, Ritter ME, Ezaru CS, Rakesh HR, Paiste HJ, et al. Extended perineural analgesia after hip and knee replacement when buprenorphine-clonidine-dexamethasone is added to bupivacaine: preliminary report from a randomized clinical trial. Pain Med. (2020) 21:2893–902. doi: 10.1093/pm/pnaa229
28. Williams BA, Dalessandro JH. Same-day pain and rehabilitation parameters after knee replacement with multimodal, mepivacaine-based, perineural analgesia-a case report of bilateral procedures one year apart. Pain Med. (2021) 22:765–6. doi: 10.1093/pm/pnaa257
29. Williams BA, Ibinson JW, Mikolic JM, Boudreaux-Kelly MY, Paiste HJ, Gilbert KL, et al. Day-one pain reductions after hip and knee replacement when buprenorphine-clonidine-dexamethasone is added to bupivacaine nerve/plexus blocks: a randomized clinical trial. Pain Med. (2022) 23:57–66. doi: 10.1093/pm/pnab325
30. YaDeau JT, Dines DM, Liu SS, Gordon MA, Goytizolo EA, Lin Y, et al. What pain levels do TSA patients experience when given a long-acting nerve block and multimodal analgesia? Clin Orthop Relat Res. (2019) 477:622–32. doi: 10.1097/CORR.0000000000000597
31. YaDeau JT, Paroli L, Fields KG, Kahn RL, LaSala VR, Jules-Elysee KM, et al. Addition of dexamethasone and buprenorphine to bupivacaine sciatic nerve block: a randomized controlled trial. Reg Anesth Pain Med. (2015) 40:321–9. doi: 10.1097/AAP.0000000000000254
32. YaDeau JT, Gordon MA, Goytizolo EA, Lin Y, Fields KG, Goon AK, et al. Buprenorphine, clonidine, dexamethasone, and ropivacaine for interscalene nerve blockade: a prospective, randomized, blinded, ropivacaine dose-response study. Pain Med. (2016) 17:940–60. doi: 10.1093/pm/pnv010
33. Turner JD, Dobson SW, Henshaw DS, Edwards CJ, Weller RS, Reynolds JW, et al. Single-injection adductor canal block with multiple adjuvants provides equivalent analgesia when compared with continuous adductor canal blockade for primary total knee arthroplasty: a double-blinded, randomized, controlled, equivalency trial. J Arthroplasty. (2018) 33:3160–6. doi: 10.1016/j.arth.2018.05.026
34. Edinoff AN, Houk GM, Patil S, Bangalore Siddaiah H, Kaye AJ, Iyengar PS, et al. Adjuvant drugs for peripheral nerve blocks: the role of alpha-2 agonists, dexamethasone, midazolam, and non-steroidal anti-inflammatory drugs. Anesth Pain Med. (2021) 11:e117197. doi: 10.5812/aapm.117197
35. Yilmaz E, Hough KA, Gebhart GF, Williams BA, Gold MS. Mechanisms underlying midazolam-induced peripheral nerve block and neurotoxicity. Reg Anesth Pain Med. (2014) 39:525–33. doi: 10.1097/AAP.0000000000000176
Keywords: nerve block, neuromodulation, physical therapy, acute pain, case report, midazolam, buprenorphine, dexamethasone
Citation: Williams BA, Ritter ME, Kennedy PJ, Kmatz AM, Abdullah L, Scanlan RL, McNiffe NJ, Fowler WM and Patzkowski MS (2025) Case Report: Two-center case series of motor-sparing nerve blocks comprised of preservative-free midazolam–buprenorphine–clonidine–dexamethasone. Front. Anesthesiol. 4:1535087. doi: 10.3389/fanes.2025.1535087
Received: 26 November 2024; Accepted: 5 March 2025;
Published: 28 March 2025.
Edited by:
Alparslan Turan, Cleveland Clinic, United StatesReviewed by:
Giuliano Lo Bianco, Fondazione Istituto G. Giglio, ItalyCopyright: © 2025 Williams, Ritter, Kennedy, Kmatz, Abdullah, Scanlan, McNiffe, Fowler and Patzkowski. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Brian A. Williams, d2lsbGlhbXNiYUBhbmVzLnVwbWMuZWR1; YnJpYW4ud2lsbGlhbXM2QHZhLmdvdg==; d2lsbGlhbXNiYUBnbWFpbC5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.