- 1U.S. Department of Agriculture, Animal and Plant Health Inspection Service, Wildlife Services, National Wildlife Research Center, Florida Field Station, Gainesville, FL, United States
- 2U.S. Fish and Wildlife Service, National Wildlife Refuge System, Southeast Inventory & Monitoring Branch, Atlanta, GA, United States
Green iguanas (Iguana iguana), large-bodied lizards native to South and Central America, have established multiple invasive populations worldwide. Where established, invasive green iguanas have negative impacts on private property, agriculture and horticulture, infrastructure and human safety, and the natural environment. Most attempts to formally quantify impacts of invasive green iguanas have been conducted in Puerto Rico, or are limited to singular reports. Green iguanas have been established in Florida since 1966 and currently pose a high management concern which must be informed by research on control methods. Impact data from Florida to justify these efforts are often anecdotal or out of date. Here we present a contemporary review of known and potential impacts of green iguanas to multiple sectors in Florida over time and space. We leverage data accessed from various stakeholders, including nuisance wildlife report data collected from the Florida Fish and Wildlife Commission’s Wildlife Impact Management Section, conflict reports from the USDA Wildlife Services Management Information System, wildlife strike reports from the Federal Aviation Administration, fault reports from energy companies, and observations from natural resource managers. We identify important knowledge gaps in quantifying the scope and magnitude of green iguana impacts necessary to guide future management actions.
1 Introduction
Green iguanas (Iguana iguana), large bodied lizards native to South and Central America, have been established in Florida since 1966 (King and Krakauer, 1966; Krysko et al., 2007; Meshaka et al., 2022). In Florida, they can reach densities of up to 626 individuals per hectare (Smith et al., 2007a), and are present in a range of habitats from urban to rural (Meshaka et al., 2004a; Krysko et al., 2007). Qualitatively, invasive green iguanas are known to impact agriculture and horticulture (Eilers, 2002; Kern, 2004; Krysko et al., 2007; López-Torres et al., 2012; Villanueva et al., 2022), infrastructure and human safety (Engeman et al., 2005a; Sementelli et al., 2008; López-Torres et al., 2012; Falcón et al., 2013), the natural environment (Krysko et al., 2007; Truglio et al., 2008; Burgos-Rodríguez et al., 2016), and private property (Kern, 2004; Krysko et al., 2007).
Based on these qualitative impacts, Florida wildlife officials have described iguanas as a species on which research is urgently needed in order to document their geographic distribution and understand their ecosystem impacts, particularly given how ubiquitous the species has become in south Florida (Ferriter et al., 2007). This need was further corroborated by a 2016 expert panel which ranked green iguana as a high management concern with need for research on control methods (Engeman and Avery, 2016), and again in 2021 in the U.S. Department of Agriculture Animal and Plant Health Inspection Service (USDA APHIS) Wildlife Services External Stakeholder Needs Assessment (Eckery et al., 2021). The sale and keeping of iguanas as pets was recently banned by the Florida Fish and Wildlife Conservation Commission (FWC), though breeding and sale of iguanas was permitted with certain exceptions until June 2024 (2021 FWC Rule 68-5.006, 68-5.007). While many control methods for green iguanas have been suggested (e.g. Kern, 2004; Sementelli et al., 2008), few have been empirically tested (Witmer et al., 2007; but see Krauss et al., 2014; Rivera-Milán and Haakonsson, 2020; Wasilewski et al., 2022). Part of the stagnation in research may stem from the lack of quantification of green iguana impacts in Florida to justify allocation of resources toward research and control efforts.
Iguanas cause different types of damage to different sectors; for example, the mere presence of iguanas is a strike hazard on airport runways (Engeman et al., 2005a; Falcón et al., 2013); burrowing behavior impacts canal infrastructure (Sementelli et al., 2008; López-Torres et al., 2012; Falcón et al., 2013; Miller, 2020) and private property (Kern, 2004; Krysko et al., 2007; López-Torres et al., 2012); climbing behaviors pose risk to electrical substations (Falcón et al., 2013; Alonso, 2022); herbivory can result in defoliation and decreased fitness of plants (Carlo Joglar and García Quijano, 2008). Iguanas negatively impact agriculture in Puerto Rico via consumption of crops (Villanueva et al., 2022), but impacts to agriculture in Florida have not been assessed beyond anecdotal reports of iguanas consuming ornamental and hobby garden plants (Kern, 2004; Krysko et al., 2007). Evaluating the impacts of non-native green iguanas coincident with the ban of green iguana in the pet trade provides an important baseline for which to evaluate the long-term impacts of this legal change on iguana impacts.
While green iguanas are not a new problem to Florida, research efforts on the species are lagging behind other problematic emergent invasive reptile species (e.g., Python bivittatus, Burmese pythons, Guzy et al., 2023; and Salvator merianae, Argentine Black and White Tegus, Engeman et al., 2019; Harvey et al., 2021). While a ban in trade will hopefully lessen the influx of new genetic diversity, green iguanas are already established with self-sustaining populations occurring throughout much of the state. Their invasive range will likely continue to expand with climate warming (Borden and Flory, 2021; Biber et al., 2023), associated expansion of suitable habitat (Bardou et al., 2023), and human-facilitated movements, whether intentional or accidental (Francis and Chadwick, 2015).
It is prudent to examine the scope of existing and potential green iguana conflicts and impacts to identify areas for further research to best inform management of this established and problematic invasive species. Herein we synthesize and add to existing reviews on green iguana impacts (e.g., Krysko et al., 2007; Falcon et al., 2013) by leveraging existing databases curated by the FWC, USDA Wildlife Services (WS) Operations, Federal Aviation Administration (FAA), and communicating with key stakeholders. The aim is to highlight areas of current research need. We recognize that conducting studies to fill those gaps will likely reveal additional stakeholders, impacts, and/or solutions that were not evident at the writing of this publication and that will be critical for management in the future. Herein, we review the current knowledge of iguana impacts to different sectors, including relevant methods, results, and discussion within each section for ease of interpretation.
1.1 Overview of green iguana general biology
The general biology of green iguanas is documented across several sources wherein the majority of information is based on studies in their native range (e.g. Jacobson, 2004; Krysko et al., 2007; Meshaka, 2011; Falcón et al., 2013; Bock et al., 2016). The gaps in knowledge of iguana biology (Burghardt, 2004), whether with respect to diet, behavior, or other traits, are important to achieve better management and control. For example, there is precedent for changes in vital rates in invasive reptile populations relative to their native counterparts (Fisher et al., 2021); this warrants research attention in green iguanas. Vital rates and other traits often differ based on density of the species; while we recognize that the degree of impacts to the ecosystem will vary based on iguana density, determining the density and abundance of green iguanas in Florida is beyond the scope of this piece.
A brief overview of green iguana biology and behavior is important to understanding the scope of impacts. Green iguanas are heliothermic lizards that bask to warm their body temperature (reviewed in Rodda, 2003), and have limited tolerance to cool temperatures (Meshaka, 2011; Falcón et al., 2012; Chappell, 2020). Typically they are found in tropical and sub-tropical semi-forested habitats at the edges of salt and freshwater aquatic features (Townsend et al., 2003; reviewed in Rodda, 2003). They are adept swimmers and climbers that spend the majority of their time in vegetation and tree canopies (Rodda, 2003), occasionally congregating in open areas (Sekscienski, 2012). Green iguanas are strictly herbivorous with few rare exceptions documented in the literature; they consume a broad variety of plants with some apparent preferences (reviewed in Baer, 2003). Green iguanas will dig burrows up to 4.5m long to use as shelter (Rodda and Burghardt, 1985). Iguana size (snout-to-vent length) spans 77.5 mm as hatchlings to 580 mm as adults (Meiri, 2024). Females lay clutches of up to 86 eggs in nests that are dug up to 1m deep and can include an additional 24m in tunnels stemming laterally from the main entrance (Mora, 1989; reviewed in Rodda, 2003; Meiri, 2024). Females sometimes display nest-guarding behaviors (reviewed in Iverson et al., 2004). During the breeding season, green iguanas can become territorial– females defend their nest sites (Iverson et al., 2004), while males defend their signaling territories from male rivals by tail lashing, biting, and chasing (reviewed in Rodda, 2003).
2 Materials and methods
2.1 Iguana impacts to private property
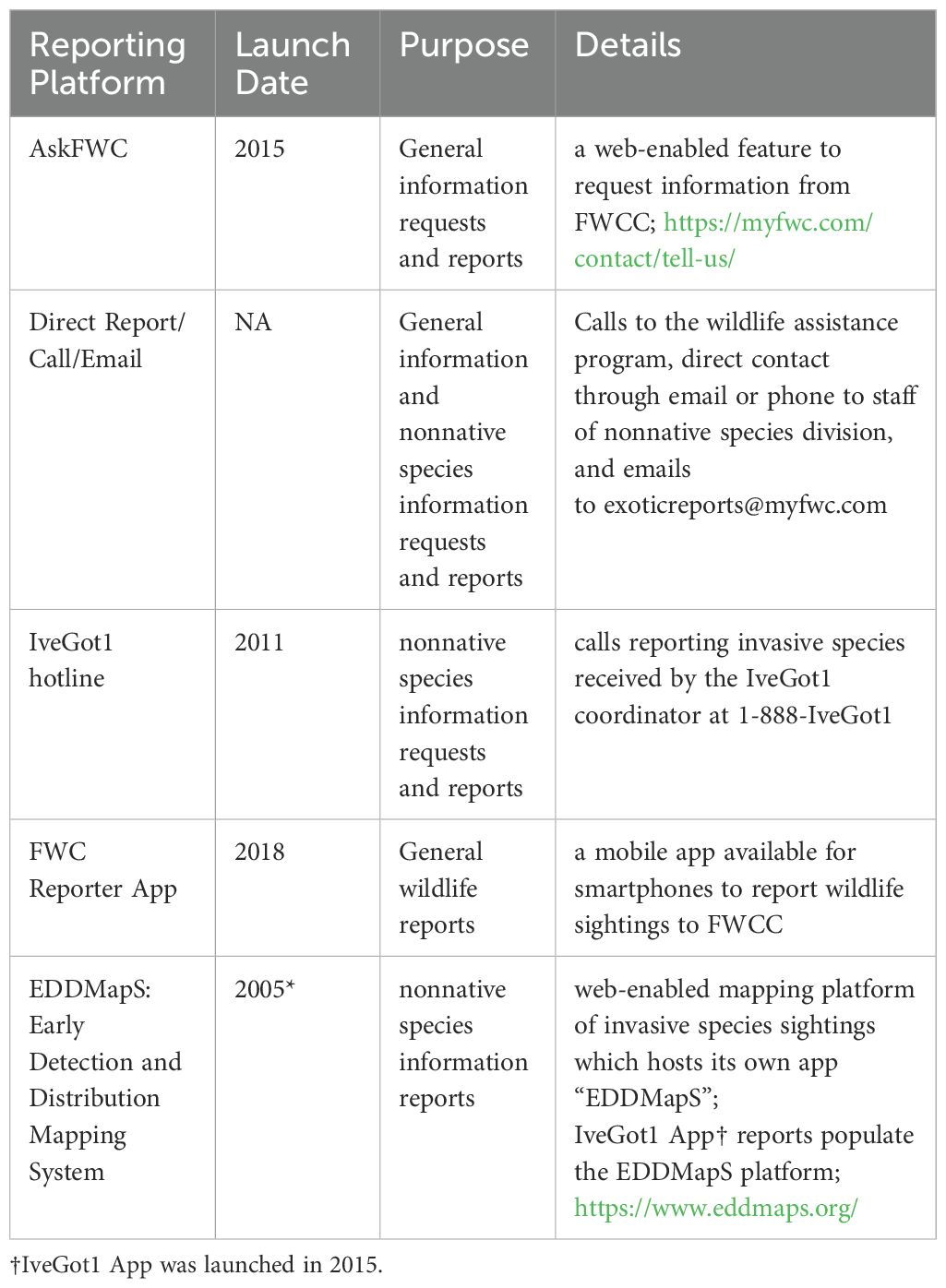
To determine iguana impacts in Florida, we queried the FWC Wildlife Impact Management Section (WIMS) database (Florida Fish and Wildlife Conservation Commission (FWC), 2023) which compiles records of sightings, captures, complaints, and requests for information about nuisance wildlife. Documents queried included reports received through AskFWC, direct reports via call or email, the IveGot1 hotline, the FWC Reporter App, and EDDMapS (Table 1; Diaz, personal communication, May 2024). This query omitted entries in the database that were retrieved from literature, data on iguana removals contracted by FWC, and data in biological surveys conducted in collaboration with FWC, the latter of which were geographically restricted in area. We then excluded observations categorized as “Unlikely” by FWC (N=7) as these observations generally included descriptions of other species of lizards. Data ranged from July 2012 to 11 September 2023, including a total of 3023 individual direct reports of green iguanas. We also communicated with two wildlife trappers specializing in iguana removals who are directly contacted by residents with existing iguana issues. While invaluable to this study, the information gleaned from the trappers is based on their personal experience and not tied to a curated database.
Using polynomial linear regression in R (R Core Team, 2023), we evaluated trends in total iguana reports over time. We used ANOVA to evaluate potential differences in iguana reports received among months and seasons (R Core Team, 2023). We excluded 2023 data from time and season related analyses to avoid biasing results because 2023 does not represent a full reporting year. For reports with coordinate information, we buffered by 5 km to account for local dispersal distance of green iguanas (NatureServe, 2024) and to protect reporter privacy.
Where possible, we categorized calls by impact type. We assigned an impact type when the “Description of Incident” and/or “Observation Details” contained text matching key words or descriptions for a given category (Supplementary Table S1). The categories included: consuming vegetation, defecation, digging in yards, digging into water barriers, other damage, trapped inside structures, safety concerns, interactions with pets, interactions with native wildlife, and deceased iguanas. Due to wide variation of descriptors that fit within certain categories (e.g. “using the bathroom”, “freaking out”) and spelling errors in the database (e.g. “dedication” instead of defecation; “borrow” instead of burrow), we read all description text manually and assigned a category based on context fitting within the key word distinctions.
2.2 Iguana impacts to agriculture and horticulture
Currently, there is no database available to analyze impacts of green iguanas on agriculture and horticulture in Florida. We conducted an informal literature review and spoke with several agricultural extension agents to determine the scope of impacts to agriculture. Our findings are presented within the discussion.
2.3 Iguana impacts to infrastructure
To assess the general scope of iguana conflicts reported to USDA APHIS WS, the primary federal agency responsible for mitigating wildlife conflict in the US, we queried the USDA WS Management Information System (MIS; United States Department of Agriculture Wildlife Services, 2023) for conflict reports of green iguanas in Florida. Conflicts were placed into the following categories: aviation, water structures (such as canals, dikes, and dams), soil erosion, general human safety, general property damage, gardens, and wildlife. We queried the FAA Airstrike database for reported iguana airstrikes in Florida and Puerto Rico (Federal aviation administration wildlife strike database, 2023). To evaluate areas at risk of iguana airstrike hazards not present in the FAA Airstrike database, we used ArcGIS Pro (Esri, 2023) to create a buffer of 5 km around all direct iguana reports in Florida to account for local dispersal distance of green iguanas (NatureServe, 2024) and protect reporter privacy (FWC WIMS) then extracted all airports that intersect this area (according to the FAA OPSNet Airport Operations Database, 2022). We additionally communicated with two Florida power companies to understand the scope of current conflicts green iguanas cause for energy infrastructure.
2.4 Iguana impacts to natural areas
We conducted an informal literature review of published information on green iguana diets, focusing on studies where direct observations took place. Several studies collected information from farmers and other residents of areas where iguanas live, asking them what they think iguanas eat whereas other studies collected empirical data while observing iguanas or examining gut contents. We excluded information compiled on opinions of iguana diet because it is difficult to ascertain whether opinions are based on direct observation or influenced by outside sources or belief (Swanson, 1950; García-Quijano et al., 2011). These diet data are available as Supplementary Material (Supplementary Dataset 1; Supplementary Material 3).
We conducted an overlay analysis using ArcGIS Pro software (Esri, 2023) to determine where green iguanas’ occurrences overlap with imperiled flowering plant and butterfly species, federally designated critical habitat, and Federal and State managed protected areas. Green iguana occurrences were compiled from FWC reports and research-grade observations available in the iNaturalist database (iNaturalist community, 2024). Iguana occurrences were buffered by 5 km to account for local dispersal distance of green iguanas (NatureServe, 2024) and stratified by USDA Climate Zones 9b – 12a. We did not include occurrences that coincided with cooler climate zones (9a and lower, winter average low temperature below -3.9 C or 25 F) due to these observations consisting primarily of incidental reports. The resulting iguana distribution polygon was used in the overlay analysis to identify areas where year-round iguana activity may impact imperiled flower plant and butterfly species, and potentially degrade critical habitat for federally listed species and federally and state managed natural areas.
3 Results
3.1 Iguana impacts to private property
Direct reports of iguanas to FWC per year have increased since 2012, peaking in 2019 (Figure 1; Supplementary Material 1). Green iguanas have been reported from 44 of 67 counties in Florida (Figure 2). The oldest reports are from Miami-Dade, Highlands, Duval, and Palm Beach counties. Reports from Miami-Dade spiked from fewer than 10 reports per year from 2012- 2015 to greater than 25 reports per year from 2018 onward. In Palm Beach reports after 2018 are consistently greater than 50 per year. In contrast, reports from Highlands and Duval counties have remained below 10 reports per year. Since 2020 six counties appeared in the direct report database for the first time (2020: Glades, Leon; 2021: Alachua, Liberty, Sumter; 2022: Nassau).

Figure 1. Total direct reports about green iguanas (Iguana iguana) to the Florida Fish and Wildlife Conservation Commission through 2022. (A) displays reports received by month; (B) displays reports received by different sources.
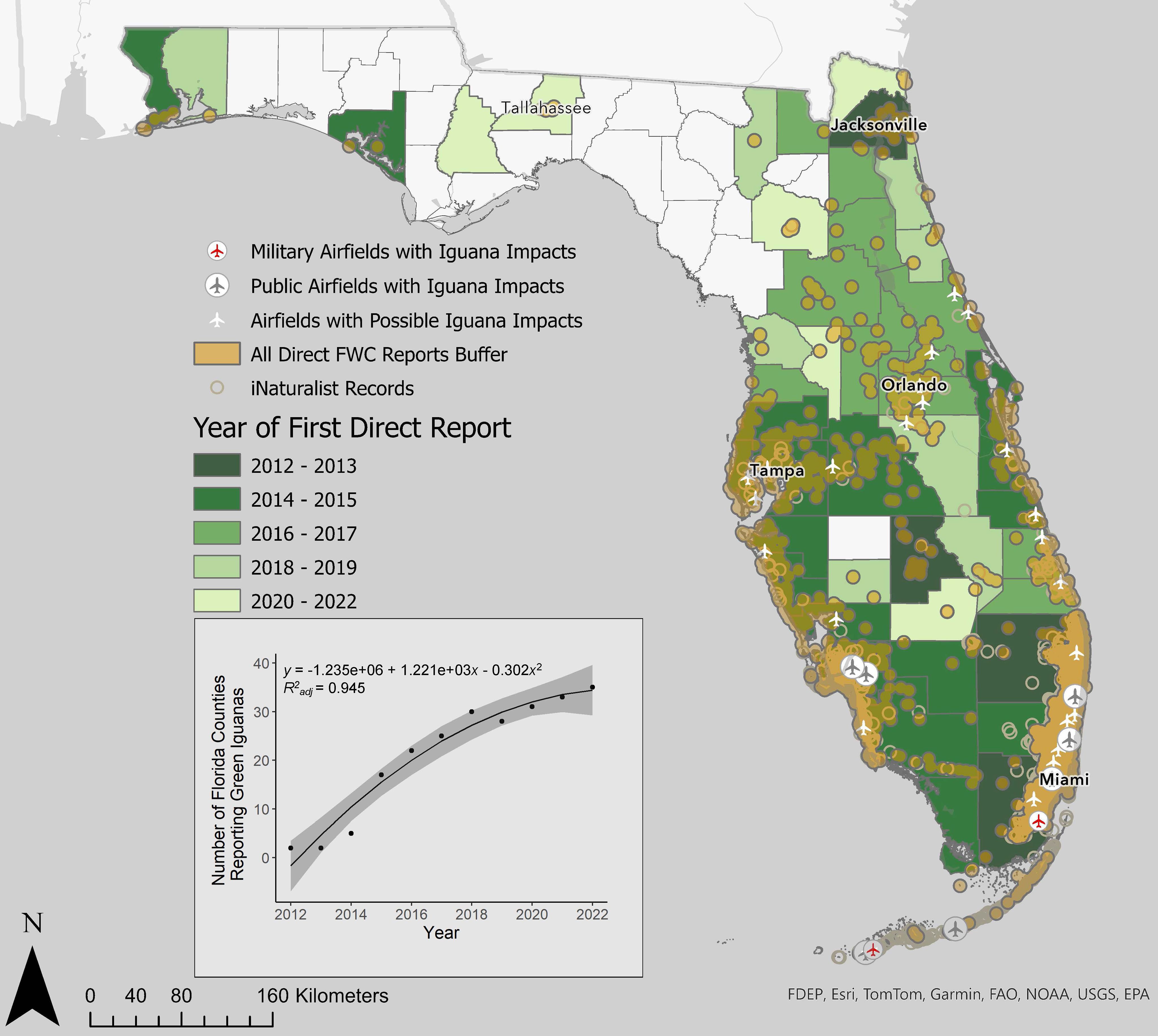
Figure 2. Geographic distribution of direct iguana reports in the Florida Fish and Wildlife Commission Wildlife Impact Management Section database through 2022 buffered by 5 km (orange) and records from iNaturalist (open circles). Airports with recorded iguana impacts are shown by airplanes in circles (gray=public, red=military). Airports with no reported iguana impacts, but intersecting the buffer are shown as white airplanes. Counties are colored by the year of first iguana report to FWC WIMS, with darker colors representing oldest reports. Inset figure displays the number of Florida counties reporting iguanas each year from 2012.
When considering all green iguana reports, there are no differences in the number of reports by month (F8,94 = 0.737, p=0.66), but the number of reports do differ by season (F3,94 = 4.71, p=0.004, Figure 1). This difference is driven by the higher number of reports in summer (June-August) than winter (December-February; p=0.002). We found no difference in the number of reports between either summer or winter when compared with spring and fall (Supplementary Material 2).
From the total number of iguana reports, 13% were assigned to at least one impact category. Seven percent of reports specified more than one impact type during a call. The most common impact reported (34%) was iguanas becoming trapped in structures (e.g. fences, houses, garages, lanais, pools), followed by eating plants (19.4% of categorized reports), posing a real or perceived safety concern (e.g. iguanas posing a roadstrike or falling hazard, or fear of iguanas; 15.1% of reports), digging in yards (11.3%), deceased bodies (7.3% of reports), digging in canal structures (6.8%), defecating (5%), posing a real or perceived threat to pets (4%), posing a real or perceived threat to wildlife (3.8%), and other impacts (2.3%; Figure 3).
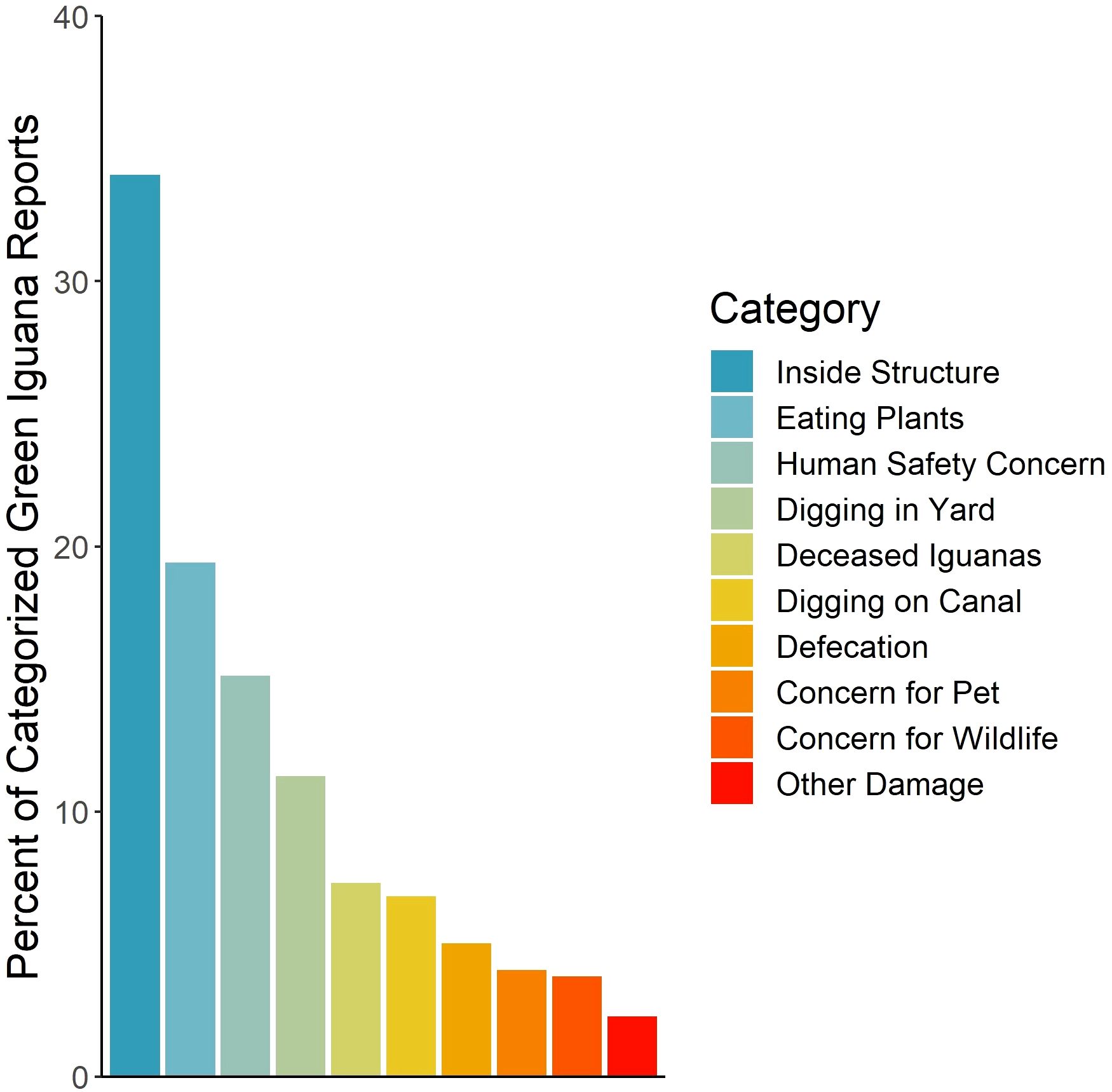
Figure 3. Reports about green iguanas (Iguana iguana) to the Florida Fish and Wildlife Conservation Commission categorized by impact type, accessed from the Wildlife Impact Management Section database through 11 September 2023.
3.2 Iguana impacts to infrastructure
The most common recorded conflicts with iguanas in USDA WS MIS are aviation related (Figure 4). The second most commonly reported green iguana conflict by WS MIS was water structure conflict. Since 2016, WS has maintained removal contracts with 2 military airfields (2009; 2012) and 1 international airport in Florida. By 2013, iguanas were documented strike hazards at Miami International Airport (MIA) and Florida Keys Marathon Airport (Falcón et al., 2013). In the 10 years following, five additional public airports in Florida and two in Puerto Rico documented iguanas as hazards (Figure 2; FAA Wildlife Strike Database). Our intersection analysis identified an additional 24 airports where iguana-associated airstrike impacts may be occurring or may occur in the future (Figure 2). Reports of iguana conflicts at airports include iguanas on tarmacs causing several strikes with landing gear and prompting go-around procedures to avoid strikes (FAA Wildlife Strike Database).
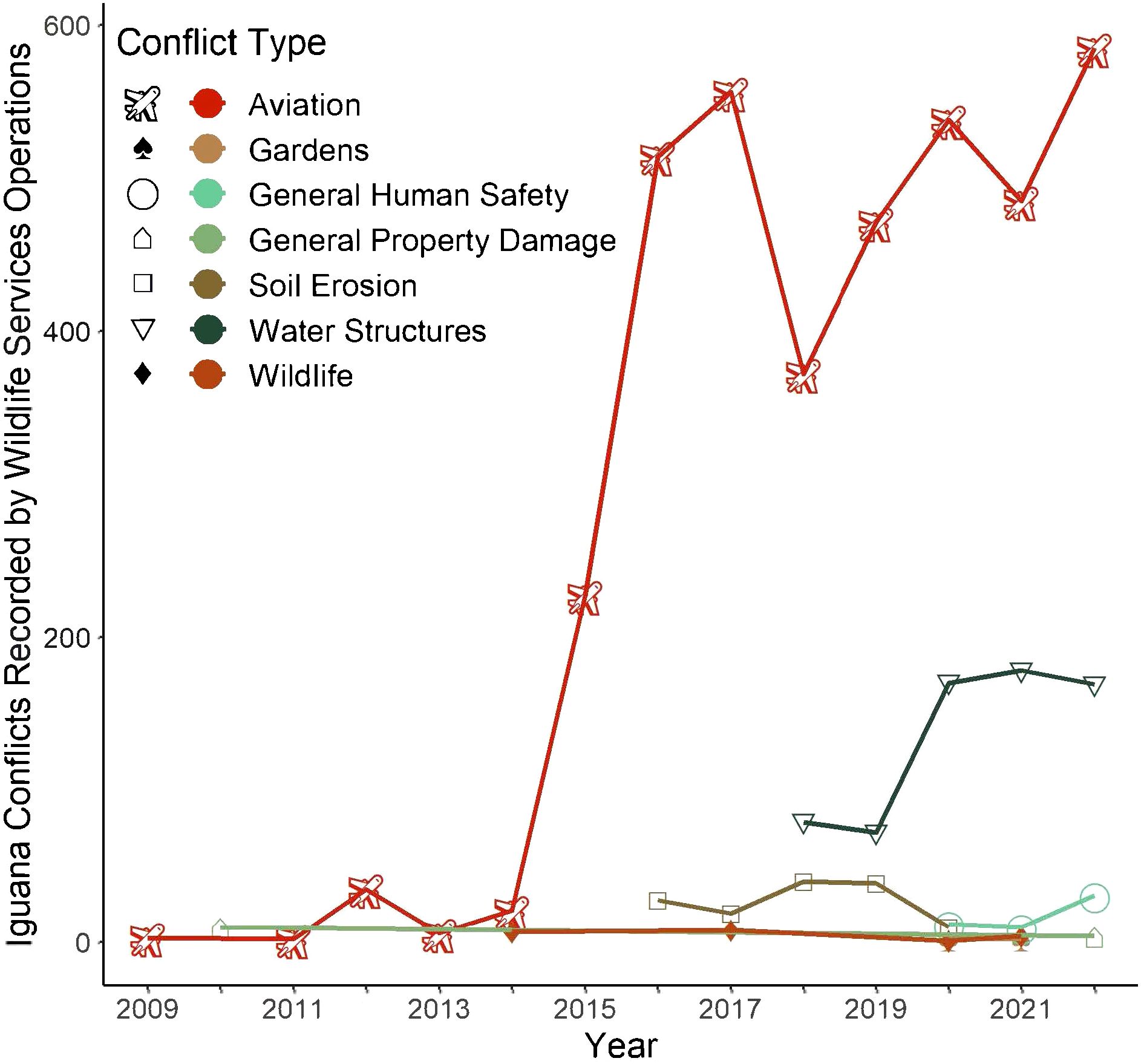
Figure 4. Summary of reported conflicts with green iguanas in Florida from USDA Wildlife Services Management Information Service from 2009 through 2022. Data reflect reports per site per day. There may be multiple concurrent conflicts reported at a site at a time.
Iguanas are responsible for 15-20 power outages a year and are currently the number one animal-related outage concern faced by Florida Power and Light (FPL), which services much of southeast Florida (Allen Khalili, personal communication, November 2023). Keys Energy, which services the lower Florida Keys, has reported an average of four iguana related outages per year, ranging from 94 to 29,550 customer-minutes without power per incident. The extent of damage caused by iguana-related power outages is generally variable as it depends on which component fails. The costs to FPL have ranged from $50,000 to $100,000 (Khalili, personal communication, November 2023). The estimates provided only represent incidents where an iguana body was found at the fault, and may underestimate the full scope of iguana damage given that iguanas have been seen escaping after a shock leading to an outage (DePhillips, personal communication, November 2023). In addition to directly causing power outages, iguanas congregating over equipment leads to accumulation of their feces and degradation of protective paint, increasing the frequency of repair schedules (DePhillips, personal communication, November 2023).
3.3 Iguana impacts to natural areas
Overall, green iguanas are documented consuming plants across 110 genera and 51 families in their native and invasive ranges (Supplementary Material 1). Plants consumed include species used as agricultural cultivars (e.g. sweet potato, Ipomoea sp.; wild grape Vitis sp.), and in horticulture (Hibiscus sp; firebush, Hamelia patens). 21 plants were noted by study authors as apparently not consumed by iguanas despite abundance and availability (Supplementary Dataset 1).
While the climate zones represented in the iguana distribution polygon range from 9b to 12a, the focal imperiled species occurring within the polygon were in climate zone ranges from 10a to 11b (Figures 5, 6). We identified 54 unique imperiled flowering plant (n=39) and butterfly (n=15) species occurring within the iguana distribution polygon. Additionally, we found that the iguana distribution polygon intersected with established critical habitat for 21 federally listed species and proposed critical habitat for 4 listed species in Florida (Supplementary Table S2). Of those 54 species, 45 occurred within protected areas (federal - n=21; state - n=24) that also overlap with the iguana distribution polygon (Figures 5, 6).
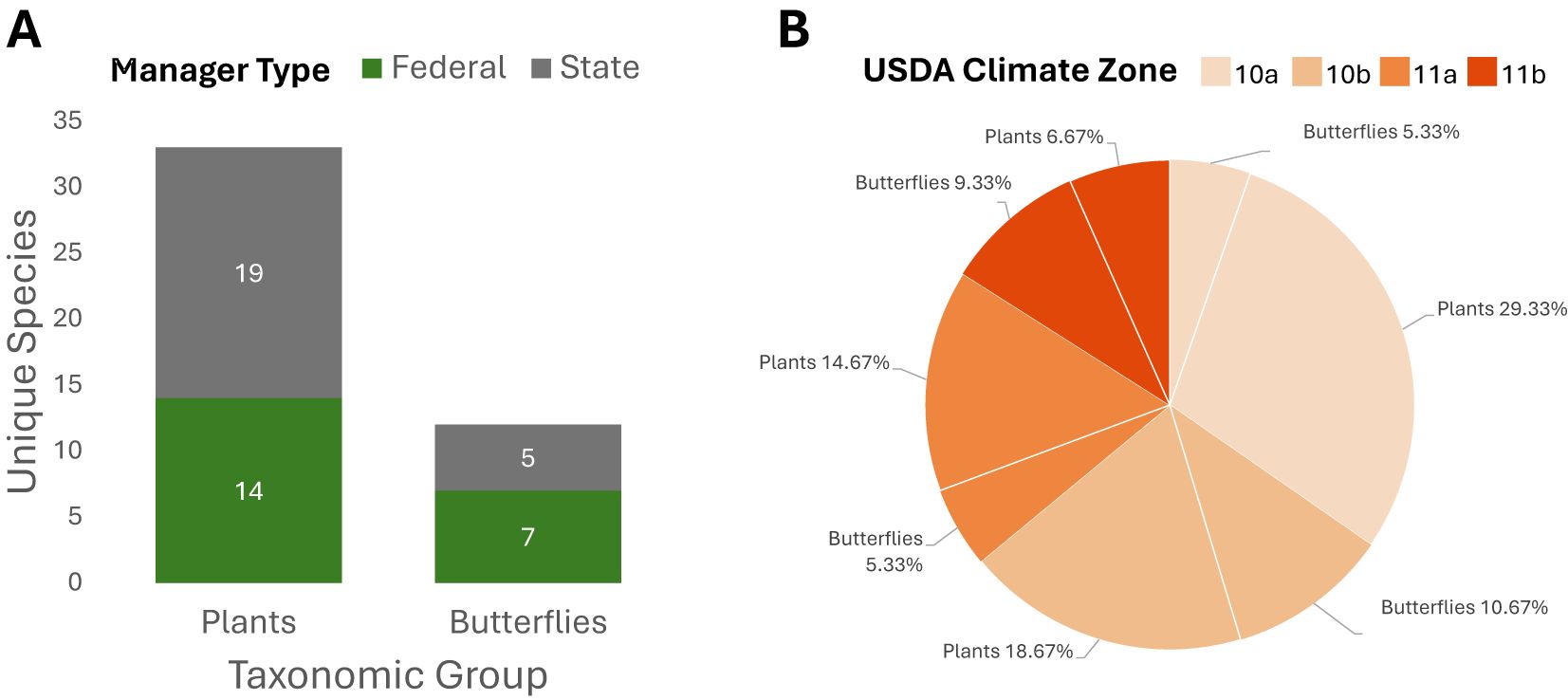
Figure 5. (A) Summary of unique focal species overlapping the iguana distribution polygon in state and federally managed protected areas in Florida. (B) Proportion of unique focal species intersecting the iguana distribution polygon in Florida, stratified by taxonomic group and USDA climate zone.
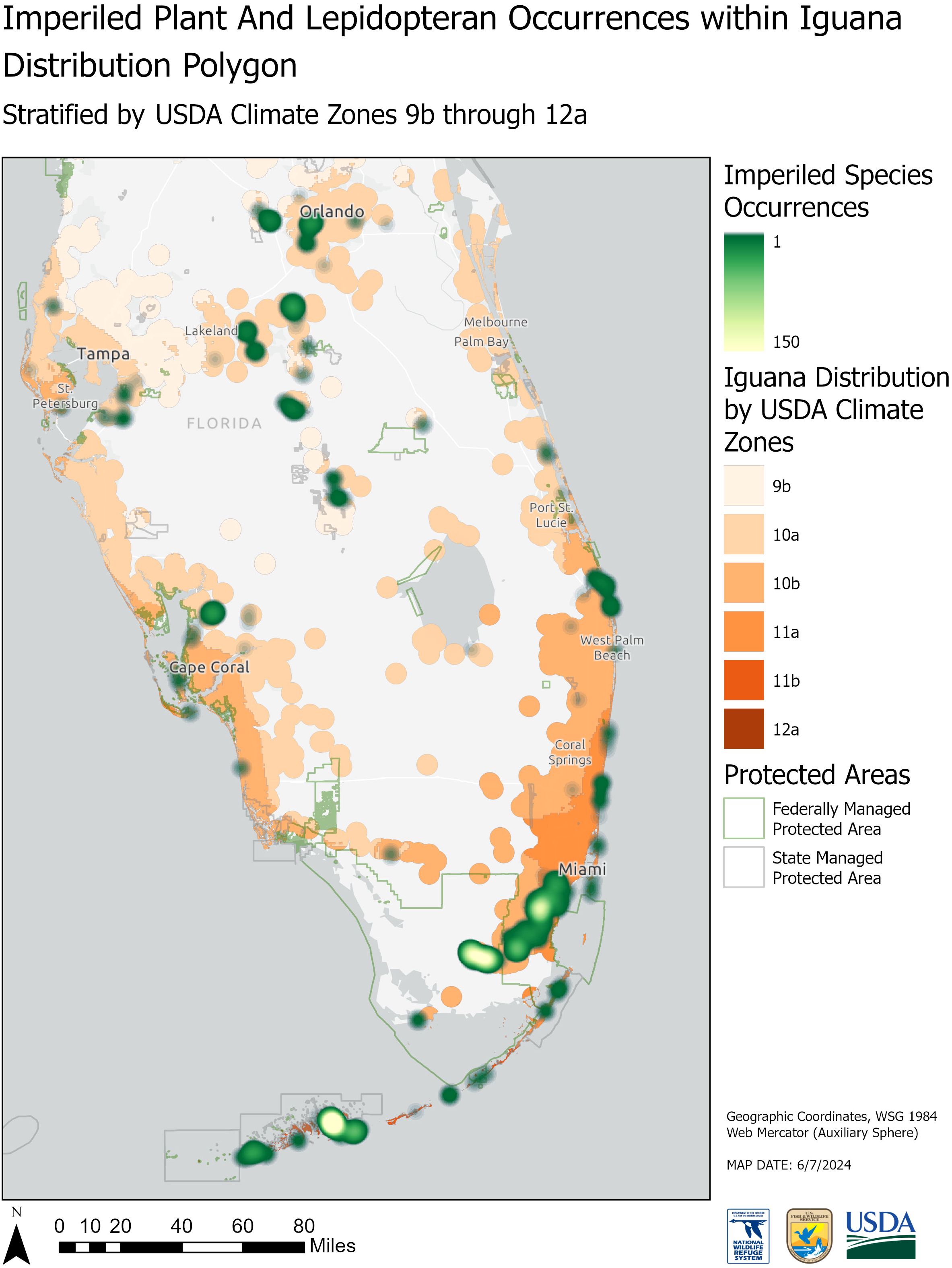
Figure 6. Map displaying overlap of iguana reports and Federally listed plant and lepidopteran species in Florida. Iguana reports are colored by USDA climate zones. Brighter colors represent greater numbers of listed species overlapping with iguana occurrences. Polygons enclose state (gray) and federal (green) protected areas. List of species available in Supplementary Material.
4 Discussion
4.1 Iguana impacts to private property
While reports of iguanas to FWC have increased over time, it is not yet clear whether the number of reports received are reaching an asymptote or trending downward. Reports must be interpreted with caution; they do not directly translate to actual iguana conflict, however the likelihood of a report being filed is influenced by several factors which are not likely constant across the data collection period. These include 1) the actual density of iguanas, 2) the number of conflicts actually occurring and 3) the propensity of an individual to report an iguana. The propensity of an individual to make an iguana report further depends on the availability of reporting mechanisms and their decision to make the report. The increase in reports from 2015 to 2019 appears to be driven by the public’s adoption of multiple new reporting platforms. AskFWC was launched in 2015, and the FWC Reporter App was launched in 2018 (Rachael Diaz, FWC Data Coordinator). The IveGot1 hotline and app were both introduced in 2011 (Everglades National Park, 2011); data from the IveGot1 app populates EDDMaPs reports and is reflected as EDDMaPs in the data presented here. There appears to have been an overall lag in public adoption of the IveGot1 platforms, as the increase in reports over time is reflected in total reports to the IveGot1 hotline (Rachael Diaz, FWC Data Coordinator). The likelihood of filing a report following an iguana observation or conflict may also have changed over the data collection period. For example, because FWC does not have the capacity to provide iguana control services to callers, instead referring callers to a list of commercial trappers, a plateau or downward trend in reports could reflect a shift in the public’s decision to contact FWC rather than a decrease in actual iguana conflicts. Further, individuals may be more likely to report iguana activity when iguanas are novel/recently established in a locality (Epanchin-Niell et al., 2021). Studies evaluating the number of actual conflicts and likelihood to report conflicts are needed to understand the nature of the report data.
It is important to note that the report data do not indicate areas of established populations of green iguanas and can reflect even one observation of a single iguana. Using these report data, we found green iguanas are reported from an additional 29 counties in Florida than the most recent assessment in 2019 (Meshaka et al., 2022). Consistent low numbers of reports from some areas (e.g. Duval county) may highlight areas of concern for long distance human-assisted dispersal events. For example, there were 8 reports that specifically mentioned finding green iguanas in northern Florida in accepted shipments from either southern Florida or Puerto Rico, or arriving with people traveling from those locations. A better understanding of iguana physiological tolerance to cold is necessary to predict the potential for establishment in northern areas where reports indicate a clear risk of introduction (human assisted or otherwise).
The recent first-time reports of iguanas are notable because they are coincident with recent restrictions prohibiting the keeping and sale of green iguanas (EO 20-19, Chapter 68-5, F.A.C; FWC Rule 68-5.006, 68-5.007). Executive order EO 20-19, Chapter 68-5, F.A.C took effect on July 1, 2020, prohibiting acquisition of green iguanas as pets and requiring owners prior to that date to apply for a permit to maintain possession of pet green iguanas. The 2021 FWC Rule 68-5.006, 68-5.007 went into effect April 29, 2021, accompanied by a grace period of 90-180 days for owners of captive green iguanas to come into compliance with updated requirements (caging and identification tags) or liquidate their inventory. The FWC rule allowed the breeding and sale of iguanas via limited exception commercial use permits through June 2024. While it is not possible to discern whether reports represent illegal releases of green iguanas in response to regulation, there is precedent (Maceda-Veiga et al., 2019), and the possibility should be considered (Engeman et al., 2022).
The increase in reports in summer versus winter could be due to several factors including: 1) iguanas are less likely to be active in the cooler, shorter days in winter and may seek shelter in burrows during cold spells (Rodda, 2003). Thus, because the opportunity window to cause damage is shorter in winter, it is possible that a decrease in reports does reflect a decrease in damage; 2) warmer weather and longer daylight hours in summer present more opportunity for diurnal lizards to be active, which may also coincide with hours when people are home or recreating outdoors. For example, people with typical 9 am – 5 pm work schedules may have more opportunity to observe iguanas causing damage on their properties during summer, when iguanas are active earlier and later in the day. Similarly, school-aged children are likelier to be outdoors and see an iguana when school is not in session (i.e., over the summer months); 3) there may be more frequent conflicts in summer than winter, including the use of swimming pools which may be higher by both humans and iguanas in summer; 4) iguana nests hatch in summertime (Meshaka, 2011), and hatchlings are a bright green color, thus iguanas may be more seasonally abundant and visible in summer (Meshaka et al., 2007), which could lead to an increase in reports.
Impacts iguanas pose to private property are multi-faceted. Green iguanas inside structures were not identified as a major issue in previous publications, though their use of pools was previously identified as a complaint (Krysko et al., 2007; Falcón et al., 2013). While we included iguanas’ use of pools (with or without complaints of defecation) in this category (trapped in structures), iguanas were also found in other structures such as garages, houses, lanais, fences, windows, and vehicles, thus posing issues to residents across socioeconomic statuses. Iguanas consuming plants is a common concern identified in previous reviews (Kern, 2004; Krysko et al., 2007; Falcón et al., 2013), and is corroborated by the FWC data as the second most prevalent impact type. We anticipated iguana consumption of plants as a major impact in Florida, and discuss the impacts of iguanas to plants in more detail in the following section (Horticulture and Agriculture).
We found that complaints of iguanas digging in yards were more common than digging in canals, likely due to the fact that there are fewer properties on canals than elsewhere. More attention has been given to iguana digging damage to canal infrastructure than to private property (Sementelli et al., 2008). We address the impacts to infrastructure below. Green iguanas were also identified as a real or perceived safety concern for people, pets, and wildlife. Reports noted iguanas as strike hazards for cars and bicycles, territorial and defensive iguanas cornered by pets or people, and iguanas entering burrows known to be inhabited by other wildlife (e.g. burrowing owls). There were several reports of people fearful of iguanas on their property. An iguana being handled may cause potential injury or health concern via defensive bite (Meshaka et al., 2022; Charruau et al., 2020). Even for those who do not intend to handle an iguana, the perceived danger of the large lizard may impact quality of life for the reporter (Spada et al., 2008) and should not be minimized.
There were many reports of deceased iguanas that did not all contain enough details to categorize as a human health concern. These may have been a result of previous vehicular strikes, others may have been abandoned by iguana hunters that did not collect and remove the bodies. At least one observation of a “deceased” iguana seems to reflect a cold-stun event, where the iguana was initially reported as dead, but the caller later reported the iguana alive. The deceased iguana reports were scattered geographically, so are not likely to represent a disease event, though these data could indicate such in the future.
While not a common complaint to FWC, iguanas defecating in and around pools and patios is a common complaint to iguana trappers (Portuallo, Iguana Control, personal communication, November 2023), and several dog owners have reported concern of their dogs consuming iguana feces (Kennamer, IggyTrap, personal communication, November 2023). In addition to being unsightly and a nuisance to clean, the feces may pose a health hazard to pets and humans (Charruau et al., 2020). A study on a cohort of captive green iguanas found strains of Escherichia coli capable of causing disease in humans (Bautista-Trujillo et al., 2020), and green iguanas are one of the top species indicated in reptile-associated salmonellosis (Pees et al., 2023). Accumulation of feces by wild iguanas may have similar capacity for disease transmission, though this has not been formally evaluated.
Control measures on private property exist but are piecemeal. Residents have the opportunity to report issues to FWC and/or contact a privately contracted iguana trapper to remove the nuisance animals. There are several companies in Florida that provide iguana removal services which directly remove the animals from a property. Private land owners must pay for this service and effective removal requires more than one visit to a property; even with removal of the majority of problem iguanas, single individuals have potential to continue to destroy residential landscaping or garden plants quickly. Cost of services range from $500 for a 3-week effort, $400 per month for scheduled one week a month removals, or $1900 for annual contract of on call removals (Portuallo, Kennamer, personal communication, November 2023). Iguana removal companies claim that the cost of providing services is a barrier to effective removal efforts, as some private property owners cannot afford the service even when sustaining property damage. Residences that do not partake in removal efforts can become “havens” for iguanas, even when they are removed from adjacent properties (Portuallo, Kennamer, personal communication, November 2023). An income-based subsidy to lower the cost of services, not unprecedented for other wildlife conflicts such as with flying fox damage in Australia (Mo et al., 2020), was suggested as a potential solution to overcome economic barriers to iguana removal. Some municipalities contract iguana trapping services for residents (Portuallo, Kennamer, personal communication, November 2023), though contracts vary widely and are not likely the case for most areas containing established iguanas.
In addition to removal services, owners of private property often take other measures to control iguana damage. Some individuals construct barriers to entry into private property by iguanas including slick wraps around tree trunks, dock pilings, or sea wall barriers, wire mesh to prevent digging, and fences with sloped tops to prevent iguanas from scaling over top. Planting “unpalatable plants” is another method used by some private property owners, however feeding studies are needed to more comprehensively and quantitatively determine palatability of plants to green iguanas. Other deterrent measures include the use of lasers, hanging reflective objects (e.g., CDs), sprinklers, and chemical deterrents, among others. These measures are extremely variable in cost and no published study to date has evaluated their efficacy for iguana control.
4.2 Iguana impacts to horticulture and agriculture
The first formal assessment of invasive green iguana damage to agriculture, published in 2022, reported iguana damage to at least 55 species of agriculturally important crops in Puerto Rico (Villanueva et al., 2022). Farmers in Puerto Rico reported single-season losses of $7,280 to $32,000 (Villanueva et al., 2022). The most commonly affected crop species included pumpkin, eggplant, and lettuce, all of which are also grown in Florida. In addition to direct damage to crops, farmers in Puerto Rico reported losses attributed to costs of constructing barriers and continuous active iguana removals. Farmers in the native range of green iguanas also report costs to buffering crops from iguanas to prevent losses (Eilers, 2002).
Commercial tropical fruit producers in Florida report iguanas eating leaves of fruit-bearing trees (e.g. papaya), basking in the trees, and defecating on produce to an extent to which the crop cannot be marketed due to food safety issues (J. Wasielewski, University of Florida Institute of Food and Agricultural Sciences (IFAS) Extension Collier County, personal communication, November 2023; Food and Drug Administration, 2015: Subparts I § 112.83 and K § 112.112). The extent of crop losses due to iguana fecal contamination has not yet been evaluated. A number of potential zoonotic pathogens can be present in iguana feces. Iguanas serve as hosts for Oxyuris and Trichostrongylus parasites, which can cause gastrointestinal disease in humans (Martínez Salazar et al., 2015) and Trichostrongylus parasites can cause disease in cattle, sheep, goats, and horses (Martínez Salazar et al., 2015; Nielsen, 2019). As noted above, green iguanas are capable of hosting a number of potentially pathogenic bacteria (Charruau et al., 2020), including documented human-pathogenic strains of Escherichia coli (Bautista-Trujillo et al., 2020) and Salmonella (Pees et al., 2023). Notably, contaminated food is an important source of salmonellosis in humans, and has been indicated in consumption of reptile meat or consumption of food contaminated with reptile excrement (Pees et al., 2023). The range of health risks to humans from iguana fecal contamination will require targeted study of iguana pathogens and parasites.
Direct damage to crops from consumption is not well documented in Florida. Producers of beans, cucumbers, and other leafy vegetables in the Everglades agricultural area have recently reported near-total crop losses due to green iguana herbivory, which is motivating some producers to consider selling the land to condominium developers (Keith Schneider UF IFAS, personal communication, November 2024). There is potential for iguanas to negatively impact other agricultural industries, including as yet unforeseen sectors, as they do in Puerto Rico and in their native range (Eilers, 2002; Villanueva et al., 2022). For example, captive iguanas have been reported to consume cannabis, however it is unclear if they consumed processed material or plants (Girling and Fraser, 2011). Their continued presence may present a barrier to the establishment of the hemp industry in Florida.
In Florida, IFAS Extension reports 20 garden and horticultural species consumed by iguanas (Gabel, 2005; Kern, 2004) and there are anecdotal reports of iguana damage to an additional 5 horticulturally important plants (Krysko et al., 2007). Extension agents in Miami-Dade county report complaints of iguanas eating vegetable and flower gardens, and fruits from privately owned trees where fruit are allowed to ripen until picking (Charpentier, Wasielewski, personal communication, November 2023). Lists of ornamental plants apparently avoided or preferred by iguanas have been curated based on experience of master gardeners, garden clubs, and other specialists and are available to homeowners through extension resources (Gabel, 2005; Kern, 2004). Employees of botanical gardens (e.g., Fairchild Botanical Garden) in South Florida have documented iguana herbivory on several ornamental plants which has prompted caging of several plants to exclude iguanas (Walsdorf, personal communication, March 2024). The plants on these lists have not been formally evaluated for iguana preference or herbivory rates.
Targeted studies identifying whether certain varieties of plants are more susceptible to iguana damage, for example, regarding color of leaves or flowers, will be useful to people managing beautification in areas where iguanas are abundant (e.g., private homes, golf courses, shopping centers). To our knowledge, however, no formal assessment has been conducted to document plant preference or to quantify agricultural and horticultural costs attributed to green iguana in Florida.
Measures to mitigate impacts to horticulture and agriculture from iguana herbivory are variable and may include placing slick wraps around tree trunks, fencing to prevent intrusion, hanging reflective materials such as CDs, deterrent sprays, sprinklers, etc. Villanueva et al. (2022) identified egg removal as a common strategy that farmers in Puerto Rico employ in an attempt to manage iguana populations, however, they noted that a related study indicated egg removal is not a viable strategy for population control (Rodríguez Gómez, 2013). Most measures have not yet been evaluated for efficacy; at least some industries would likely benefit from a quantitative examination of mitigation efforts and their success.
4.3 Iguana impacts to infrastructure
The majority of conflicts recorded by USDA WS MIS are aviation related. Green iguanas have caused activity to halt and have resulted in strikes by aircraft at Luis Muñoz Marín International Airport (SJU) in Puerto Rico (Engeman et al., 2005a). The main risk associated with iguanas at airports is not the direct damage they cause to aircraft on the tarmac, but rather the presence of iguana nests and iguanas basking on or near the runway which attract birds of prey. Large birds attracted to the iguanas and iguana eggs are more damaging than the iguanas themselves because they constitute airborne strike hazards (Leif Stephens, USDA WS state director AL,VI, PR, personal communication, February 2024). Due to continued active management, the risks of iguanas as direct and indirect strike hazards have been mitigated to within safe levels. The costs of this management vary: at SJU, iguana control efforts were estimated to cost $98,000 per year in 2011 (Falcón et al., 2013). Currently, USDA APHIS Wildlife Services Operations employs a biologist at San Juan Airport that spends at least 80% of their time on iguana management, amounting to $54,000 in salary alone for iguana control by a single biologist at one airport (estimated at GS9-step 5; Leif Stephens, WS state director AL,VI, PR, personal communication, February 2024) implying that the actual cost of iguana management is significantly higher.
Burrowing behavior constitutes another major source of iguana-associated infrastructure damage. Burrows near canals can reach up to 2,800 burrows per hectare (Sementelli et al., 2008). The cost estimated to repair damage from a single burrow in a canal wall in South Florida in 2008 was $400 (Sementelli et al., 2008). This amount is only a reactive cost that does not account for iguana removal or burrowing prevention efforts. In 2020, repairs to an iguana-damaged dam cost 1.8 million dollars in Palm Beach (Miller, 2020). Burrowing can also negatively affect roadways. In 2012, the cost of roadway repair in Puerto Rico at a single 316 acre reserve was estimated as $5,000 annually (López-Torres et al., 2012). In addition to burrowing damage to roadways, green iguanas are well-documented vehicular strike hazards in both their native and invasive ranges (Rodda, 1990; Krysko et al., 2007; Falcón et al., 2013). While we do not have current estimates for the scope of impacts to roadways in Florida, the Florida Department of Transportation is concerned about green iguanas as strike hazards and undermining roadway infrastructure (Hall, USDA WS State Director FL, personal communication, November 2023).
Iguanas congregate on transformers which emit heat that iguanas use as basking opportunity, and by touching terminals on both ends of the transformer, can short-circuit the system (DePhillips, personal communication, November 2023). Faults outside the transformer can destroy bushings and cause internal damage, requiring replacement of the entire transformer. Transformer replacement can cost up to $1 million to replace and would further be complicated by supply chain issues in securing a replacement (DePhillips, personal communication, November 2023). Economic assessments of the impact of iguana-caused power outages are not available. Estimates of costs for power outages caused by brown treesnakes in Guam were $375,000 per hour 30 years ago (Fritts, 2002). Based on costs incurred from iguana impacts to power infrastructure in Florida in the recent past, we anticipate that the economic toll that iguanas have on Florida infrastructure exceeds that of brown treesnakes on Guam and will continue to do so.
Mitigation tools for green iguanas are specific to the infrastructural needs and can vary in scope and cost. USDA APHIS WS is the primary federal agency carrying out iguana control measures. WS operations primarily use firearms to remove green iguanas, and also employ traps and nest destruction. Other entities employ methods including installing wire mesh around canal walls and banks to deter digging, constructing sloped and/or buried fences to prevent iguana entry to an area, and filling in iguana burrows with concrete (Sementelli et al., 2008). Current measures to mitigate iguanas at power stations include trapping, custom exclusion fencing, and proprietary equipment covers. Fencing upgrades are estimated to cost up to $250,000 and take upwards of 25 years to implement (Khalili, personal communication, November 2023). Equipment covers cost up to $10,000 per transformer and must be replaced every four to five years due to degradation in salt and UV exposure (DePhillips, personal communication, November 2023). To date, few equipment covers are rated for 69 kV, and none are rated for voltages as high as 138 kV (DePhillips, personal communication, November 2023), which leaves specialized equipment at risk of damage by iguanas short-circuiting the system.
4.4 Iguana impacts to natural areas
Iguanas are documented consuming a variety of plants, and overlap in areas with imperiled plant species. There is abundant evidence demonstrating that green iguanas can defoliate native trees, including mangroves (López-Torres et al., 2012), which provide vital ecosystem services via their buffering capacity during wind and flooding events (Lorenz, 2013). While preference studies have not yet been conducted, the majority of gut contents in a study of I. iguana in Puerto Rico were composed of black mangrove (A. germinans), suggesting a potential impact on mangrove forest structure and recruitment (Govender et al., 2012). Green iguanas are also known to eat mature fruit (see above) and thereby may also influence invasive and native seed germination and dispersal, although effects are likely species-specific (Burgos-Rodriguez et al., 2016). Another potential ecological impact of invasive iguanas is the alteration of nutrient transfer and microbial community, filling a previously empty niche as a large herbivorous lizard (Swierk and Langkilde, 2009). The information gathered likely does not represent a comprehensive assessment of iguana diet. This partly stems from the limited opportunity for behavioral observation given that iguanas spend only a small fraction of their waking hours feeding (Moberly, 1968; Rand et al., 1990; Sekscienski, 2012). Long term observations and increased numbers of diet samples may be necessary to gather a better understanding of iguana impacts on plants. The data on plants apparently not consumed should be treated with caution, as we note at least one discrepancy: Carlo Joglar and García Quijano (2008) noted that iguanas did not consume Pithecellobium dulce, but this species was confirmed in iguana diet contents in another study (Govender et al., 2012). This emphasizes the need for thorough diet content studies and targeted avoidance-preference studies to better understand the breadth and impacts of iguana foraging.
Juvenile iguanas may consume insects in captivity (Schumacher et al., 2003), but observations of green iguanas of any age consuming invertebrates in the wild is rare (Hirth, 1963; Townsend et al., 2005; Govender et al., 2012). In the majority of investigations, iguanas are found to be strictly herbivorous (Van Devender, 1982; Rand et al., 1990; Swanson, 1950; Troyer, 1984). While iguanas are unlikely to pose a direct threat to native invertebrates, they may pose an indirect threat through consumption of key host plants for imperiled insects and incidental ingestion of the insects themselves (Hunt personal communication, March 2024; Possley personal communication, March 2024; see below).
Like many herbivores, green iguanas may preferentially forage on tender, new growth of plants (van Marken Lichtenbelt, 1993; but see Troyer, 1984) which are less fibrous and more digestible (Kurokawa et al., 2022). Butterflies tend to oviposit on new growth of plants to facilitate foraging of first instar caterpillars which also rely on the soft, less fibrous new growth to feed on (Clissold et al., 2009). Given the concentration of herbivory on the newest, most tender growth, it is likely that while consuming the plant, adult green iguanas are incidentally consuming butterfly eggs and early instar caterpillars. For example, iguana herbivory on gray nickerbean (Guilandina bonduc) has been widely documented (Krysko et al., 2007). Nickerbean is a critical host plant for the small, endangered butterfly, the Miami Blue (Cyclargus thomasi bethunebakeri). While iguanas are one of the few visible threats potentially contributing to the decline of this endangered species (Hunt personal communication, March 2024), there have not been any targeted studies on iguana consumption of host plants to identify the scope of impact iguanas may have on butterfly recruitment. However researchers monitoring the Miami Blue butterfly have anecdotally noted a lack of recruitment of nickerbean at sites where green iguanas are common and have also noted iguanas feeding on young plants and preventing flowering/seed set of mature plants (by consuming flower-containing ends of stems; Cuni et al., 2022). Other iguana behavior, including burrowing, further imperils butterflies and other nectivorous species by disturbing soils and uprooting flowering plants that would otherwise bloom thus providing floral subsidies for butterflies and other insects (Hunt personal communication, March 2024). This is apparent in the Marquesas Keys, where both nectar sources and host plants, including Pithecellobium keyense, have been damaged or destroyed by iguanas (Hunt, personal communication, March 2024). Based on occurrence overlap, iguanas have the potential to impact several other imperiled species. Targeted thermal ecology research is necessary to determine where iguanas may establish across different climate zones to forecast current or future risk to species of concern.
Non-native iguanas can also cause conflict with vertebrates native to the invaded range. Florida hosts multiple native burrowing animals, and while burrows are not novel features in the landscape, they may be increased where iguanas occur. Green iguanas have been documented in both gopher tortoise (Gopherus polyphemus; Truglio et al., 2008) and burrowing owl burrows (Athene cunicularia; Krysko et al., 2007), which are both listed as Threatened in Florida. It is not clear if green iguana use of burrows of other animals leads to their displacement as in other iguana species (e.g. McKnight et al., 2024, 2023; Rivero and Durso, 2023). Green iguanas in their native range have been documented disturbing nests of American Crocodiles (Crocodylus acutus; Dugan et al., 1981; Bock and Rand, 1989; Charruau and Hénaut, 2012); there is potential for similar interactions in Florida, where American Crocodiles are designated as Threatened by the US Endangered Species Act. In Florida, iguanas are consumed by nonnative and native species, including Burmese python (Python bivittatus, Romagosa et al., 2022), domestic dog (Canis familiaris; Meshaka et al., 2004b), burrowing owl (A. cunicularia; McKie et al.., 2005); Yellow-crowned night heron (Nyctanassa violacea; Engeman et al., 2005b); raccoons (Procyon lotor; Smith et al., 2006a), and their eggs are consumed by gray fox (Urocyon cinereoargenteus, Smith et al., 2007b). Juvenile iguana and iguana eggs apparently supplement raccoon populations, as when high densities of raccoons are removed, iguana populations rebound (Meshaka et al., 2007). This presents a predicament for managing two species commonly reported for wildlife human conflict. The propensity for iguanas to bask in trees in wetland areas may put them at odds with nesting wading bird colonies. A camera study in Puerto Rico recorded iguanas walking in and amongst artificial nests baited with chicken eggs (Carlo Joglar and García Quijano, 2008). One iguana chewed on straw used to construct the nest, but no iguana depredation events were recorded (Carlo Joglar and García Quijano, 2008). Though green iguanas are not known to consume eggs (one report of scavenging of dropped eggshells; Schwartz and Henderson, 1991), iguanas may influence nest attendance and incubation by adult birds and affect recruitment.
While natural resource managers are employing methods described above, a removal method that may be well-suited for natural areas is targeting green iguanas at night on their open sleeping perches (Wasilewski et al., 2022). Removal of iguanas at night may be more effective than diurnal removals, because the iguanas are not as lucid and are easier to capture by hand or snare pole, or by net after knocking them off the perch with an extendable pole or drone (Wasilewski et al., 2022). This approach allowed for removal of nearly 2 iguanas per person-hour of effort. This and other methods, such as trapping, direct diurnal removals, egg removals, need to be evaluated for relative efficacy and impacts on population density.
4.5 Conclusion
Iguanas impact natural resource management, private property, agricultural operations, and human infrastructure due to their direct impacts and allocation of budget focused on their removals and mitigating damage. They may also impact ecosystem services via impacts on plant recruitment, pollinator recruitment, subsidizing mesopredator populations, and/or altering waterscape structure through burrowing. While costs of impacts to natural areas can be difficult to quantify (e.g. quantifying ecosystem services), it is possible to monetize these impacts by assessing costs of control efforts.
Impacts to horticulture and agriculture in Florida consist of anecdotal reports, and impacts in Puerto Rico have recently been documented via a combination of interviews, surveys, and a controlled experiment. Farming methods and crop types differ between Puerto Rico and Florida, thus targeted research is important to understand the site-specific problems in both locations. Interviewing producers and landowners is likely to reveal the scope of direct and indirect impacts and the types of produce and farming techniques affected by green iguanas in Florida. This can then be followed by research quantifying damage by iguanas, preferred or avoided crop types, farming methods most impacted by iguanas, and effectiveness of specific barrier and deterrent methods.
While there have been attempts to assign costs to invasive green iguana impacts in various aspects (e.g. Sementelli et al., 2008; López-Torres et al., 2012; Falcón et al., 2013; Villanueva et al., 2022), these generally only represent singular cases consisting of estimates from Puerto Rico. Gaps in data availability and inconsistencies in reporting impacts make it difficult to assign a range-wide cost estimate for green iguanas in Florida. Moving forward, it would be beneficial if documentation of impacts were well documented and curated. An updated assessment of green iguana should include efforts to monetize impacts which are known but have not been quantified. It will be important to calculate economic impacts to individuals as well as larger entities such as energy companies, departments of safety for water resource management as well as aviation and other transportation, and natural area land managers (e.g., state and national parks). Quantifying impacts will provide decision-makers with estimates to budget for cost-effective research and management options and shed light on the scope of impacts caused by the invasion of this lizard.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Ethics statement
Ethical approval was not required for the study involving animals in accordance with the local legislation and institutional requirements because Animal research presented represents a summary of previous research already published. No animals were handled for the new data presented herein, and no ethics approval was required.
Author contributions
NC: Conceptualization, Data curation, Formal Analysis, Investigation, Methodology, Project administration, Visualization, Writing – original draft, Writing – review & editing. PJ: Data curation, Formal Analysis, Investigation, Methodology, Software, Visualization, Writing – original draft, Writing – review & editing. EK: Data curation, Formal Analysis, Investigation, Methodology, Project administration, Software, Visualization, Writing – original draft, Writing – review & editing. BK: Conceptualization, Investigation, Project administration, Supervision, Writing – original draft, Writing – review & editing.
Author's note
The findings and conclusions in this publication are those of the author(s) and should not be construed to represent any official USDA or U.S. Government determination or policy. Any use of trade, firm, company, or product names is for descriptive purposes only and does not imply endorsement by the U.S. Government and does not guarantee nor warrant the standard of any product mentioned. Product and company names are mentioned solely to report factually on available data and to provide specific information.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported (in part) by the USDA APHIS WS.
Acknowledgments
We thank the many experts who shared their relevant green iguana observations for inclusion in this manuscript.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/famrs.2025.1529065/full#supplementary-material
References
Alonso M. (2022). ‘It’s a nightmare:’ Iguana causes power outage in Florida town for third time this year (CNN). Available online at: https://www.cnn.com/2022/12/18/us/florida-iguana-power-outage-trnd (Accessed June 3, 2024).
Baer D. J. (2003). “Nutrition in the wil,” in Biology, Husbandry, and Medicine of the Green Iguana. Ed. Jacobson E. R. (Krieger Publishing Company, Florida), 38–46.
Bardou R., Osland M. J., Scyphers S., Shepard C., Aerni K. E., Alemu J. B. I., et al. (2023). Rapidly changing range limits in a warming world: critical data limitations and knowledge gaps for advancing understanding of mangrove range dynamics in the southeastern USA. Estuar. Coast. 46, 1123–1140. doi: 10.1007/s12237-023-01209-7
Bautista-Trujillo G. U., Gutiérrez-Miceli F. A., Mandujano-García L., Oliva-Llaven M. A., Ibarra-Martínez C., Mendoza-Nazar P., et al. (2020). Captive green iguana carries diarrheagenic Escherichia coli pathotypes. Front. Vet. Sci. 7. doi: 10.3389/fvets.2020.00099
Biber M. F., Voskamp A., Hof C. (2023). Potential effects of future climate change on global reptile distributions and diversity. Glob. Ecol. Biogeogr. 32, 519–534. doi: 10.1111/geb.13646
Bock B. C., Páez V. P., Rand A. S., Burghardt G. M. (2016). Life table and stochastic matrix projection analysis for a population of green iguanas (Iguana iguana): implications for conservation and control. Herpetol. Conserv. 11, 47–60. doi: 10.2307/1445983
Bock B. C., Rand A. S. (1989). Factors influencing nesting synchrony and hatching success at a green iguana nesting aggregation in Panama. Copeia 4, 978–986. doi: 10.2307/1445983
Borden J. B., Flory S. L. (2021). Urban evolution of invasive species. Front. Ecol. Environ. 19, 184–191. doi: 10.1002/fee.2295
Burghardt G. M. (2004). “Iguana Research: Looking back and looking ahead,” in Iguanas: Biology and Conservation. Eds. Alberts A. C., Carter R. L., Hayes W. K., Martins E. P. (University of California Press, California), 1–12.
Burgos-Rodríguez J. A., Avilés-Rodríguez K. J., Kolbe J. J. (2016). Effects of invasive Green Iguanas (Iguana iguana) on seed germination and seed dispersal potential in southeastern Puerto Rico. Biol. Invasions 18, 2775–2782. doi: 10.1007/s10530-016-1190-6
Carlo Joglar T. A., García Quijano C. G. (2008). Assessing ecosystem and cultural impacts of the green iguana (Iguana iguana) invasion in the San Juan Bay Estuary (SJBE) in Puerto Rico (San Juan, Puerto Rico: Final Report to the San Juan Bay Estuary Program).
Chappell B. (2020). When winter hits Florida, iguanas fall from trees- and maybe onto the grill (NPR). Available online at: https://www.npr.org/2020/01/22/798630899/when-winter-hits-florida-iguanas-fall-from-trees-and-maybe-onto-the-grill (Accessed May 21, 2024).
Charruau P., Hénaut Y. (2012). Nest attendance and hatchling care in wild American crocodiles (Crocodylus acutus) in Quintana Roo, Mexico. Anim. Biol. 62, 29–51. doi: 10.1163/157075511X597629
Charruau P., Pérez-Juárez J. G., Medina M., Méndez de la Cruz F. R., Pérez-Flores J. (2020). Bacterial flora of wild black (Ctenosaura similis Gray 1831) and green (Iguana iguana Linneaus 1758) iguanas from a Mexican Caribbean atoll. Herpetol. Notes 13, 369–376.
Clissold F. J., Sanson G. D., Read J., Simpson S. J. (2009). Gross vs. net income: How plant toughness affects performance of an insect herbivore. Ecology 90, 3393–3405. doi: 10.1890/09-0130.1
Cuni L., Moore T., Possley J. (2022). Final Report for University of Florida (UF), Dr. Jaret Daniels Lab, McGuire Center for Lepidoptera & Biodiversity/Disney Conservation Fund.
Dugan B. A., Rand A. S., Burghardt G. M., Bock B. C. (1981). Interactions between nesting crocodiles and iguanas. J. Herpetol. 15, 409–414. doi: 10.2307/1563530
Eckery D. C., Algeo T. P., Alls S. A., Carlisle K. M., Chipman R. B., Keirn G. M., et al. (2021). Wildlife Services research needs assessment (external) (Ft. Collins, Colorado: USDA, APHIS, WS National Wildlife Research Center), 2021 1–79.
Eilers C. H. A. M. (2002). Iguana production: hope or scope? Wagenigen, The Netherlands: Wageningen Institute of Animal Sciences.
Engeman R., Avery M. (2016). Prioritizing management and research actions against invasive reptiles in Florida: A collaboration by an expert panel (Fort Collins, Colorado: USDA National Wildlife Research Center Staff Publications).
Engeman R., Goss P., Bessette E. (2022). How might a ban on the commercial breeding of select exotic reptiles promote unwanted releases: the curious case of Florida. Red Bellied Courier 9, 88–95.
Engeman R. M., Kaiser B. W., Osorio K. J. (2019). Evaluating methods to detect and monitor populations of a large invasive lizard: the Argentine giant tegu. Environ. Sci. pollut. Res. 26, 31717–31729. doi: 10.1007/s11356-019-06324-2
Engeman R., Smith H., Constantin B. (2005a). Invasive iguanas as airstrike hazards at Luis Muñoz Marin International Airport, San Juan, Puerto Rico. J. Aviat./Aerosp. Educ. Res. 14, 45–50. doi: 10.15394/jaaer.2005.1518
Engeman R. M., Sweet E. M., Smith. H. T. (2005b). Iguana iguana (Green iguana). Predation. Herpetol. Rev. 36, 320.
Epanchin-Niell R., Thompson A. L., Treakle T. (2021). Public contributions to early detection of new invasive pests. Conserv. Sci. Pract. 3, e422. doi: 10.1111/csp2.422
Everglades National Park (2011). “IveGot1” – Mobile App to Identify and track Invasive Exotics in Florida now Available! [News Release]. Available online at: https://www.nps.gov/ever/learn/news/ivegot1mobileapppr112911.htm (Accessed March 12, 2025).
Falcón W., Ackerman J. D., Daehler C. C. (2012). March of the green iguana: Non-native distribution and predicted geographic range of Iguana iguana in the Greater Caribbean Region. IRCF Reptiles Amphibians 19, 150–1160. doi: 10.17161/randa.v19i3.14532
Falcón W., Ackerman J. D., Recart W., Daehler C. C. (2013). Biology and impacts of pacific island invasive species. 10. Iguana iguana, the green iguana (Squamata: Iguanidae). Pac. Sci. 67, 157–186. doi: 10.2984/67.2.2
Federal aviation administration wildlife strike database. Available online at: https://wildlife.faa.gov/ (Accessed November 8, 2023).
Federal Aviation Operations Network (OPSNET) (2022). Airport operations: Florida. Available online at: https://aspm.faa.gov/opsnet/sys/main.asp (Accessed February 7, 2024).
Ferriter A., Doren B., Goodyear C., Thayer D., Burch J., Toth L., et al. (2007). Chapter 9: The status of nonindigenous species in the south Florida environment. 2006 South Florida Environmental Report. South Florida Water Management District. Available at: https://apps.sfwmd.gov/sfwmd/SFER/2009_SFER/v1/chapters/v1_ch9.pdf (Accessed March 12, 2025).
Fisher S., Fisher R. N., Alcaraz S. E., Gallo-Barneto R., Patino-Martínez C., López-Jurado L. F., et al. (2021). Reproductive plasticity as an advantage of snakes during island invasion. Conserv. Sci. Pract. 3, e554. doi: 10.1111/csp2.554
Florida Fish and Wildlife Conservation Commission (FWC). (2023). Nuisance Wildlife Database: Iguana iguana.
Food and Drug Administration (2015). eCFR:: 21 CFR Part 112 – Standards for the Growing, Harvesting, Packing, and Holding of Produce for Human Consumption (Accessed May 20, 2024).
Francis R. A., Chadwick M. A. (2015). Urban invasions: non-native and invasive species in cities. Geogr. 100, 144–151. doi: 10.1080/00167487.2015.12093969
Fritts T. H. (2002). Economic costs of electrical system instability and power outages caused by snakes on the island of Guam. Int. Biodeter. Biodegr. 49, 93–100. doi: 10.1016/S0964-8305(01)00108-1
García-Quijano C. G., Carlo T. A., Acre-Nazario J. (2011). Human ecology of a species introduction: interactions between humans and introduced green iguanas in a Puerto Rican urban estuary. Hum. Organ. 70, 164–178. doi: 10.17730/humo.70.2.p24755p02826h047
Girling S. J., Fraser M. A. (2011). Cannabis intoxication in three green iguanas (Iguana iguana). J. Small Anim. Pract. 52, 113–116. doi: 10.1111/j.17485827.2010.01017.x
Govender Y., Muñoz M. C., Ramírez Camejo L. A., Puente-Rolón A. R., Cuevas E., Sternberg L. (2012). An isotopic study of diet and muscles of the green iguana (Iguana iguana) in Puerto Rico. J.Herpetol. 46, 167–170. doi: 10.1670/11-004
Guzy J. C., Falk B. G., Smith B. J., Willson J. D., Reed R. N., Aumen N. G., et al. (2023). Burmese pythons in Florida: A synthesis of biology, impacts, and management tools. NeoBiota 80, 1–119. doi: 10.3897/neobiota.80.90439
Harvey R. G., Dalaba J. R., Ketterlin J., Roybal A., Quinn D., Mazzotti F. J. (2021). Growth and spread of the Argentine black and white tegu population in Florida. WEC437/UW482. EDIS 2021 (Gainesville, Florida, USA: University of Florida EDIS). doi: 10.32473/edis-UW482-2021
Hirth H. F. (1963). Some aspects of the natural history of Iguana iguana on a tropical strand. Ecology 44, 613–615. doi: 10.2307/1932553
iNaturalist community (2024). Observations of Iguana iguana from Florida, USA observed on/between 1997-2023. Available online at: https://www.inaturalist.org (Accessed February 5, 2024).
Iverson J. B., Hines K. N., Valiulis J. M. (2004). The nesting ecology of the Allen Cays rock iguana Cyclura Cyclura inornata in the Bahamas. Herpetol.Monogr. 18, 1–36. Available at: https://www.jstor.org/stable/4093019 (Accessed March 12, 2025).
Jacobson E. R. (2004). Biology, husbandry, and medicine of the green iguana (Florida: Krieger Publishing Company), 1–188.
Kern W. (2004). Dealing with iguanas in the south Florida landscape (Gainesville, Florida: Entomology and Nematology, Florida Cooperative Extension Service, Institute of Food and Agricultural Science). doi: 10.32473/edis-in528-2004
King W., Krakauer T. (1966). The exotic herpetofauna of southeast Florida. Q. J. Fla. Acad. Sci. 29, 144–154.
Krauss U., Isidore L., Mitchel N., Seely L., Prospere A., Ramessar A., et al. (2014). “Assessment of control methods for invasive alien iguanas in Saint Lucia,” in Closing Symposium for GEF project Mitigating the Threat of Invasive Alien Species in the Insular Caribbean, Port of Spain (Trinidad) March 2014. Port of Spain, Trinidad. 30. 1–36.
Krysko K. L., Enge K. M., Donlan E. M., Seitz J. C., Golden E. A. (2007). Distribution, natural history, and impacts of the introduced green iguana (Iguana iguana) in Florida. Iguana 14, 142–151. Available at: https://journals.ku.edu/iguana/article/view/17706 (Accessed March 12, 2025).
Kurokawa H., Oguro M., Takayanagi S., Aiba M., Shibata R., Mimura M., et al. (2022). Plant characteristics drive ontogenetic changes in herbivory damage in a temperate forest. J. Ecol. 110, 2772–2784. doi: 10.1111/1365-2745.13990
López-Torres A. L., Claudio-Hernández H. J., Rodríguez-Gómez C. A., Longo A. V., Joglar R. L. (2012). Green iguanas (Iguana iguana) in Puerto Rico: Is it time for management? Biol. Invasions 14, 35–45. doi: 10.1007/s10530-011-0057-0
Lorenz J. (2013). Shoreline Habitat: Mangroves. Report. MARES- MARine and Estuarine goal Setting for South Florida. National Oceanic and Atmospheric Administration (NOAA) Atlantic Oceanographic & Meteorological Laboratory. Available online at: https://www.aoml.noaa.gov/ocd/ocdweb/docs/MARES/MARES_SEFC_ICEM__20131001_Appendix_Mangroves.pdf (Accessed March 12, 2025).
Maceda-Veiga A., Escribano-Alacid J., Martínez-Silvestre A., Verdaguer I., Mac Nally R. (2019). What’s next? The release of exotic pets continues virtually unabated 7 years after enforcement of new legislation for managing invasive species. Biol. Invasions 21, 2933–2947. doi: 10.1007/s10530-019-02023-8
Martínez Salazar M., Arcos García J. L., Veléz Hernández L., Mendoza Martínez G. D., López Pozos R. (2015). La iguana verde (Iguana iguana) y sus parásitos en una unidad de manejo intensivo en la costa de Oaxaca. Temas Ciencia y Tecnología 19, 43–52.
McKie A. C., Hammond J. E., Smith H. T., Meshaka W. E. (2005). Invasive green iguana interactions in a burrowing owl colony in Florida. Fla. Field Nat. 33, 125–127.
McKnight S. L., Johnson S. A., MacKenzie D., Acevedo M. A., Kluever B. M. (2024). Management of invasive iguanas mitigates deleterious impacts on an imperiled tortoise. Manage. Biol. Invasion 15, 471–492. doi: 10.3391/mbi.2024.15.4.01
McKnight S. L., Kluever B. M., Hall P., Acevedo M. A., Johnson S. A. (2023). History, status, impacts, and management of invasive black spiny-tailed iguanas (Ctenosaura similis) on Gasparilla island, Florida: WEC459/UW512. EDIS 2023. (Gainesville, Florida, USA: University of Florida EDIS). doi: 10.32473/edis-uw512-2023
Meiri S. (2024). SquamBase-A database of squamate (Reptilia: Squamata) traits. Glob. Ecol. Biogeog. 33:e13812. doi: 10.1111/geb.13812
Meshaka W. E. Jr. (2011). A runaway train in the making: the exotic amphibians, reptiles, turtles, and crocodilians of Florida. Monograph 1. Herpetol. Conserv. Biol. 6, 1–101.
Meshaka W. E. Jr., Bartlett R. D., Smith H. T. (2004a). Colonization success by green iguanas in Florida. Iguana 11, 154–161.
Meshaka W. E. Jr., Butterfield B. P., Hauge J. B. (2004b). The exotic amphibians and reptiles of Florida (Malabar, Florida, USA: Krieger Publishing Company).
Meshaka W. E. Jr., Collins S. L., Bury R. B., McCallum M. L. (2022). Exotic Amphibians and Reptiles of the United States (Gainesville Florida, USA: University Press of Florida).
Meshaka W. E., Smith H. T., Golden E., Moore J. A., Fitchett S., Cowan E. M., et al. (2007). Green iguanas (Iguana iguana): the unintended consequence of sound wildlife management practices in a south Florida park. Herpetol. Conserv. Bio. 2, 149–156. Available at: https://digitalcommons.unl.edu/icwdm_usdanwrc/693 (Accessed March 12, 2025).
Miller K. (2020). Invasive iguanas: burrowing reptiles blamed for $1.8M of damages to palm beach dam (Palm Beach Post). Available at: https://www.tcpalm.com/story/news/local/florida/2020/01/21/iguanas-not-just-nuisance-these-lizards-contributed-1-8-m-repair-bill-west-palm/4530432002/ (Accessed March 12, 2025).
Mo M., Roach M., Demers M.-C. A. (2020). Reducing human-wildlife conflict through subsidizing mitigation equipment and services: helping communities living with the gray-headed flying-fox. Hum. Dimens. Wildl. 25, 387–397. doi: 10.1080/10871209.2020.1735580
Moberly W. R. (1968). The metabolic responses of the common iguana, Iguana iguana, to activity under restraining. Comp. Biochem. Physiol. 27, 1–20. doi: 10.1016/0010-406X(68)90749-4
Mora J. M. (1989). Eco-behavioral aspects of two communally nesting iguanines and the structure of their shared nesting burrows. Herpetologica 45, 293–298. Available at: https://www.jstor.org/stable/3892884 (Accessed March 12, 2025).
NatureServe (2024). NatureServe Network Biodiversity Location Data accessed through NatureServe Explorer (Arlington, Virginia: NatureServe). Available online at: https://explorer.natureserve.org/Taxon/ELEMENT_GLOBAL.2.103640/Iguana_iguana (Accessed June 01, 2024).
Nielsen M. K. (2019).Trichostrongylus axei infection in horses. In: Merck manual veterinary manual professional version (Merck & Co., Inc). Available online at: https://www.merckvetmanual.com/digestive-system/gastrointestinal-parasites-of-horses/trichostrongylus-axei-infection-in-horses (Accessed April 22, 2024).
Pees M., Brockmann M., Steiner N., Marschand R. E. (2023). Salmonella in reptiles: a review of occurrence, interactions, shedding and risk factors for human infections. Front. Cell Dev. Biol. 11. doi: 10.3389/fcell.2023.1251036
Rand A. S., Dugan B. A., Monteza H., Vianda D. (1990). The diet of a generalized folivore: Iguana iguana in Panama. J. Herpetol. 24, 211–243. doi: 10.2307/1564235
R Core Team (2023). R: A language and environment for statistical computing (Vienna, Austria: R Foundation for Statistical Computing). Available at: https://www.R-project.org/ (Accessed October 1, 2023).
Rivera-Milán F. F., Haakonsson J. (2020). Monitoring, modeling and harvest management of non-native invasive green iguanas on Grand Cayman, Cayman Islands. Biol. Invasions 22, 1879–1888. doi: 10.1007/s10530-020-02233-5
Rivero J., Durso A. M. (2023). Black spiny-tailed iguana (Ctenosaura similis) use of gopher tortoise (Gopherus polyphemus) burrows in Buckingham, Florida. Fla. Field Nat. 51, 58–73. Available at: https://digitalcommons.usf.edu/ffn/vol51/iss3/2 (Accessed March 12, 2025).
Rodda G. H. (1990). Highway madness revisited: Roadkilled Iguana iguana in the llanos of Venezuela. J. Herpetol. 24, 209–211. doi: 10.2307/1564234
Rodda G. H. (2003). “Biology and reproduction in the wild,” in Biology, Husbandry, and Medicine of the Green Iguana. Ed. Jacobson E. R. (Krieger Publishing Company, Florida), 1–27.
Rodda G. H., Burghardt G. M. (1985). Iguana iguana (Green iguana). Terrestriality Herpetol. Rev. 16, 112.
Rodríguez Gómez C. A. (2013). A distribution model, nest temperatures, predators and results for a five year management plan of Iguana iguana in Puerto Rico. University of Puerto Rico, Rio Piedras, Puerto Rico 164 pp.
Romagosa C. M., Mazzotti F. J., Snow R. W., Bartoszek I., Dove C. J., Diego-Juárez-Sánchez A., et al. (2022). Diet of Invasive Burmese Pythons (Python molurus bivittatus) in Southern Florida 1995-2020: (Fort Collins, Colorado: U.S Geological Survey Data Release). doi: 10.5066/P9A0V89V
Schumacher J., Kohler G., Maxwell L. K., Antonio F. B., Ehrig R. W. (2003). “Husbandry and management,” in Biology, Husbandry, and Medicine of the Green Iguana. Ed. Jacobson E. R. (Krieger Publishing Company, Florida), 75–95.
Schwartz A., Henderson R. W. (1991). Amphibians and Reptiles of the West Indies: Descriptions, Distributions, and Natural History (Florida: University Press of Florida), 1–720.
Sekscienski S. R. (2012). Activity patterns of green iguana (Iguana iguana) at Hugh Taylor Birch State Park, Fort Lauderdale, Florida. Boca Raton, Florida: Florida Atlantic University.
Sementelli A., Smith H., Meshaka W., Engeman R. (2008). Just green iguanas?: The associated costs and policy implications of exotic invasive wildlife in south Florida. Public Works Manage. Policy 12, 599–606. Available at: https://digitalcommons.unl.edu/icwdm_usdanwrc/1036 (Accessed March 12, 2025).
Smith H., Golden E., Meshaka W. (2007a). Population density estimates for a green iguana (Iguana iguana) colony in a Florida state park. J. Kansas Herpetol. 21, 19–20.
Smith H. T., Meshaka W. E. Jr., Busch G., Cowan. E. M. (2007b). Gray Fox predation of nests as a potential limiting factor in the colonization success of the Green Iguana in southern Florida. J. Kansas Herpetol. 23, 7–8.
Smith H. T., Meshaka W. E. Jr., Engeman R. M., Crossett S. M., Foley M. E., Busch G. (2006). Raccoon predation as a potential limiting factor in the success of the Green Iguana in southern Florida. J. Kansas Herpetol. 20, 7–8.
Spada M. M., Nikčević A. V., Moneta G. B., Wells A. (2008). Metacognition, perceived stress, and negative emotion. Pers. Individ. Differ. 44, 1172–1181. doi: 10.1016/j.paid.2007.11.010
Swanson P. L. (1950). The Iguana Iguana iguana iguana (L). Herpetologica 6, 187–194. Available at: https://www.jstor.org/stable/3890004 (Accessed March 12, 2025).
Swierk L. N., Langkilde T. (2009). Micronutrient input into a mangrove ecosystem in Jobos Bay, Puerto Rico, by the exotic green iguana Iguana iguana. Curr. Zool. 55, 435–438. doi: 10.1093/czoolo/55.6.435
Townsend J. H., Krysko K. L., Enge. K. M. (2003). Introduced iguanas in southern Florida: a history of more than 35 years. Iguana 10, 111–118.
Townsend J. H., Slapcinsky J., Krysko K. L., Donlan E. M., Golden E. A. (2005). Predation of a tree snail Drymaeus multilineatus (Gastropoda: Bulimulidae) by Iguana iguana (Reptilia: Iguanidae) on Key Biscayne, Florida. Southeast. Nat. 4, 361–364. Available at: https://www.jstor.org/stable/3877970 (Accessed March 12, 2025).
Troyer K. (1984). Diet selection and digestion in Iguana iguana: the importance of age and nutrient requirements. Oecologia 61, 201–207. doi: 10.1007/BF00396761
Truglio M., Smith H., Meshaka W. (2008). Use of gopher tortoise burrows (Gopherus polyphemus) by the exotic green iguana (Iguana iguana) in southern Florida. J. Kansas Herpetol. 25, 22–23.
United States Department of Agriculture Wildlife Services (USDA WS). (2023). Management Information System (MIS) 2000.
Van Devender R. W. (1982). “Growth and ecology of spiny-tailed and green iguanas in Costa Rica, with comments on the evolution of herbivory and large body size,” in Iguanas of the World. Eds. Burghardt G. M., Rand A. S. (Noyes Publications, New Jersey), 162–183.
van Marken Lichtenbelt W. D. (1993). Optimal foraging of a herbivorous lizard, the green iguana in a seasonal environment. Oecologica 95, 246–256. doi: 10.1007/BF00323497
Villanueva C. N. D., Prado G. P. M., Gould W., García-Quijano C., Kolbe J. J. (2022). Interviews with farmers suggest negative direct and indirect effects of the invasive green iguana (Iguana iguana) on agriculture in Puerto Rico. Manage. Biol. Invasions 13, 781–797. doi: 10.3391/mbi.2022.13.4.13
Wasilewski J. A., Krysko K. L., Wasilewski N., Metzger E. F. III (2022). Efficient nocturnal capture methods for controlling a diurnal and arboreal invasive lizard species. Herpetol. Conserv. Bio. 17, 37–42.
Witmer G. W., Burke P. W., Pitt W. C., Avery M. L. (2007). “Management of invasive vertebrates in the United States: an overview,” in Managing Vertebrate Invasive Species: Proceedings of an International Symposium. Eds. Witmer G. W., Pitt W. C., Fagerstone K. A. (USDA, APHIS, WS National Wildlife Research Center, Collins, Colorado), 127–137.
Keywords: nonnative, lizard, squamate, reptile, damage
Citation: Claunch NM, Jones PM, Khazan ES and Kluever BM (2025) State of knowledge for invasive green iguanas in Florida reveals negative impacts and pervasive research needs. Front. Amphib. Reptile Sci. 3:1529065. doi: 10.3389/famrs.2025.1529065
Received: 15 November 2024; Accepted: 28 February 2025;
Published: 26 March 2025.
Edited by:
Thomas Fieldsend, King’s College London, United KingdomReviewed by:
Pierre Charruau, Colegio de la Frontera Sur (ECOSUR), MexicoWalter Meshaka, Pennsylvania Historical and Museum Commission, United States
Copyright © 2025 Claunch, Jones, Khazan and Kluever. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Natalie M. Claunch, bmF0YWxpZS5jbGF1bmNoQHVzZGEuZ292
 Natalie M. Claunch
Natalie M. Claunch Paulina M. Jones
Paulina M. Jones Emily S. Khazan2
Emily S. Khazan2