
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
SYSTEMATIC REVIEW article
Front. Agron. , 25 March 2025
Sec. Pest Management
Volume 7 - 2025 | https://doi.org/10.3389/fagro.2025.1558395
Cotton is an important crop that significantly contributes to the economies of developing countries, providing income for farmers and driving economic growth in rural areas. However, cotton production in developing countries faces significant challenges due to insect pest infestations. The main impacts include yield losses and increased reliance on synthetic pesticides. Recent advances in pest management are constrained by the use of chemical pesticides that are harmful to the environment and less efficacy and stability of bio-based formulations, especially plant-based. Challenges are more significant in the developing countries where low technology, and reliance on synthetic adulterated products are experienced. To evaluate advances in sustainable pest management, 760 articles were collated and screened. A total of 39 qualified peer-reviewed articles were used to evaluate current research advancements in botanical nanoformulations for sustainable cotton insect pest management in developing countries and examined their efficacy on key cotton insect pests, formulation techniques, mode of action and environmental impact while identifying challenges such as nanoparticle stability and scalability. Results showed that botanical nanoformulations such as silver and zinc nanoparticles, nano-emulsions, and polymeric carriers enhance efficacy, stability, and environmental sustainability. About 85% of the studies were laboratory-based experiments, with only 15% being semi-field and/or field trials. Findings indicate that botanical nanoformulations are viable alternative for manging cotton insect pest management. However more researches are needed to optimize their stability and efficacy in real-world cotton insect pest management in developing countries context.
Cotton, Gossypium hirsutum L., is an important crop in many developing countries, serving as a critical source of income and livelihood for farmers (Malinga, 2021; Soumaré et al., 2021). Globally, in 2014, cotton generated $600 billion, from 25.6 million tons of lint harvested from 32 million hectares (Ateeq-ur-Rehman et al., 2020; Malinga, 2021). By 2018, cotton was cultivated in over 64 countries whereby India, United States, China, Brazil, and Pakistan produced over 75%, with India and China contributing 50% (Malinga, 2021). Other major producing countries include Australia, Uzbekistan, Turkey, Turkmenistan, Egypt and Burkina Faso, all of which play crucial roles in the global cotton market (Ateeq-ur-Rehman et al., 2020; Khawar and Singh, 2020). Africa contributes to 15% of lint exports, generating $2.1 billion in 2019.Cotton in West and Central Africa (WCA), where it is a significant economic driver, contributing between 3% and 15% to the gross domestic product (GDP) (Malinga, 2021; Soumaré et al., 2021).
Despite its economic significance, cotton production in developing countries faces substantial challenges, with pest infestations causing annual yield losses exceeding 50% in Africa and Asia (Sharma et al., 2017; Overton et al., 2021; Raju and Sharma, 2021; Ratto et al., 2022; Devi et al., 2023) (Overton et al., 2021; Ratto et al., 2022; Devi et al., 2023) (Sharma et al., 2017; Raju and Sharma, 2021). Cotton is susceptible to a wide range of insect pests which causes substantial damage to yield if left uncontrolled (Riaz et al., 2021), these insets include Helicoverpa armigera (Hubner), Earias insulana (Boisdual), E. vitella (Fabricius), Pectinophora gossypiella (Saunders), Spodoptera frugiperda (J. E. Smith), Spodoptera exigua (Hübner), Spodoptera littoralis (Boisd.), Amrasca biguttula (Ishida)/Amrasca devastans, Aphis gossypii (Glover), Thrips tabaci (Lind), Tetranychus cinnabarinus, Phenacoccus solenopsis and Bemisia tabaci.
A commonly used insect pest management option in developing countries is the application of chemical pesticides (Siddiqua et al., 2016; Akhter et al., 2023; Saini and Gupta, 2024). Despite their quick action and efficiency in the management of pests, chemical pesticides are reported to be unsustainable, create insect resurgence (Shahid et al., 2021), cause insect resistance to insecticides (Devi et al., 2014; Shahid et al., 2021; Kumar Sharma et al., 2022), kill natural enemies of insect pests and are often too expensive for smallholder farmers (Khanra et al., 2018; Nursal, 2019; Abdelaal et al., 2021). Chemical pesticides are also evidenced to have environmental contamination impacts (Sharma et al., 2020; Sarker et al., 2021). Additionally, most of the chemical pesticide applications are ineffectively or wrongly applied resulting in the loss of more than 70% of the given dose (Hou et al., 2021). In developing countries, smallholder farmers lack access to protective gear (Aïhounton et al., 2021) a scenario that exposes them to harmful pesticides (Overton et al., 2021; Soumaré et al., 2021). As a result, there is a pressing need for alternative pest management strategies that are effective, eco-friendly and sustainable (Isman, 2019; Acheuk et al., 2022; Srinivasan et al., 2022).
Botanical plant extracts containing protective secondary metabolites can effectively manage insect pests in storage and field conditions (Karani et al., 2017; Mkindi et al., 2019; Mkindi et al., 2020). They are made of biodegradable organic materials, have diverse bioactivity, and have low toxicity to non-target organisms (Bharani and Namasivayam, 2017; Prema et al., 2018). Some botanical formulations are contact, respiratory or stomach poisons (Sarwar and Sattar, 2016; Sarwar, 2017; Sarwar, 2023). They may also work as repellents. antifeedants or as phenological inhibitors (Sarwar and Sattar, 2016; Sarwar, 2023). However, the use of botanical pesticides is limited by physical stability, quick environmental degradation and poor water solubility (Abdelaal et al., 2021; Bae et al., 2022). Botanical nanoformulations have emerged as promising avenues to improve the efficacy, stability, solubility targeted and controlled release of these bioactive compounds against cotton insect pests (Khanra et al., 2018; Abdelaal et al., 2021; Metwally et al., 2022; Rafea et al., 2022; Alfaro-Corres et al., 2023). Botanical nanoformulations are defined as the process of creating nanoscale formulations derived from pesticidal plant materials for various applications with particular on cotton insect pests. Nanoformulations emerged from several processes including biogenic/biological synthesis, green synthesis, chemical synthesis, polymeric nanoparticles synthesis and nanoemulsions (Anees et al., 2022; Devi et al., 2023; Khaleel et al., 2023). Many different matrices can be used to produce nanostructured systems, including biodegradable polymers (Wani and Ali Khan, 2016; Sakban-Al-Tamimi et al., 2020; Anees et al., 2022; Qarachal et al., 2024), active compounds isolated from plants, metallic oxides as well as the essential oils (EOs) (de Oliveira et al., 2018). This paper aims to provide an overview of the use of botanical nanoformulations in cotton insect pest management in developing countries, focusing on the current research and advancements in development, enhancement of botanical efficacy, mode of action and challenges of botanical nanoformulations for cotton pest management in developing countries.
This review aimed to answer critical questions regarding the evidence of botanical nanoformulations in cotton insect pest management and their comparative effectiveness against conventional methods in terms of efficacy, mode of action, and implementation challenges in developing countries. The study employed the Population, Intervention, Comparison, and Outcome (PICO) framework to refine the search criteria for identifying relevant studies. Population (P): Cotton insect pests in developing countries, Intervention (I): Botanical nanoformulations for pest management, Comparison (C): Conventional insecticides and non-nano botanical insecticides, Outcome (O): Effectiveness of nanoformulations in controlling cotton insect pests. A structured literature search was conducted in Google Scholar and Scopus databases. These platforms were selected due to their extensive repositories of peer-reviewed articles and accessibility through the Nelson Mandela African Institution of Science and Technology. The search string consisted of major keywords and their related terms, including “Botanical nanoformulations,” “Cotton insect pests,” and “Pest management” within developing countries. The full search string is detailed in Table 1.
A systematic filtering approach was used to ensure the inclusion of relevant studies. The inclusion and exclusion criteria (Table 2) were designed to focus on peer-reviewed articles published between 2014 and 2024 in English, while excluding non-relevant and duplicate studies. Screening was performed in three stages. First; title screening, articles were selected based on keywords, research location, and scope. Second; abstract screening, abstracts were reviewed to assess the focus on pest management, formulation techniques, insect pest species, and research outcomes. Third; full-text review, studies were assessed to confirm their relevance to botanical nanoformulations insect pest management in cotton (Figure 1). This review collected data on nanoparticle types, plant species, lethal concentrations (LC50 and LC90), modes of action, and environmental effects. Data synthesis was performed using thematic categorization.
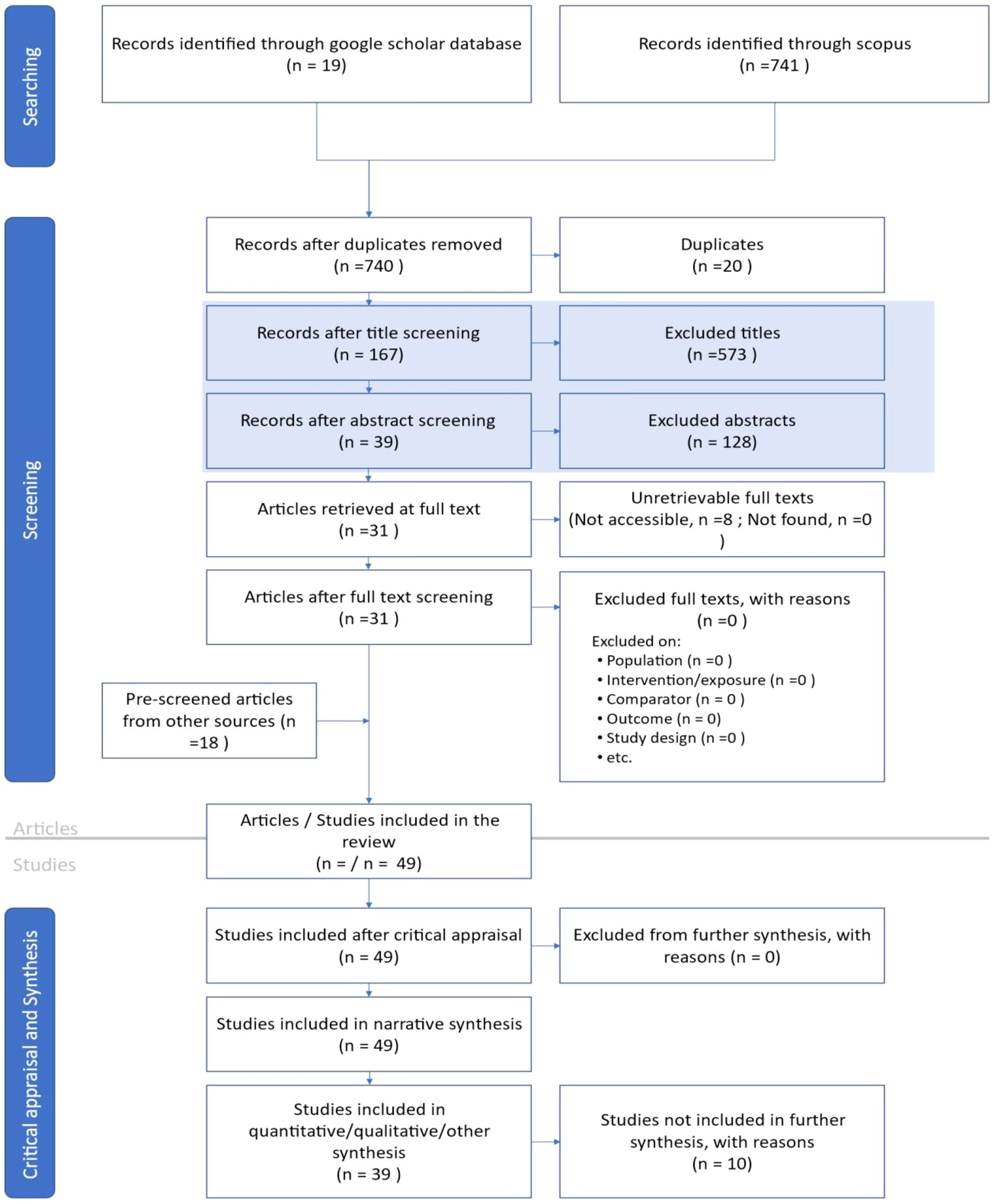
Figure 1. ROSES flow diagram for systematic reviews, version 1.0(1). Reproduced from Haddaway et al. (2017), licensed under CC BY 4.0.
The data extraction process followed established systematic review guidelines (PRISMA) (Page et al., 2021) capturing key study parameters, including nanomaterials used, nanoparticle synthesis methods, plant species and their derived compounds, targeted insect pests, and efficacy metrics. Relevant bibliographic details, such as author names, publication year, and study location, were also documented in excel sheet for structured analysis. The extracted data were organized to reflect the key theme and synthes relative to research question. Comparative assessment techniques identified the strengths and weaknesses of different formulations. Thematic analysis was also employed to examine trends in synthesis methods, formulation stability, and practical applicability in agricultural settings. The integration of findings allowed for an evidence-based discussion on the feasibility of botanical nanoformulations in sustainable cotton pest management.
Recognizing the predominance of laboratory-based studies 85% over field-based trials (15%), a limitation of this review is the potential overestimation of laboratory-reported efficacy. To mitigate this, studies were categorized based on experimental conditions (e.g., laboratory, semi-field, field) to facilitate a realistic interpretation of findings (Figure 2). Moreover, publication bias was minimized by including studies with both positive and negative results. The use of multiple databases and strict inclusion criteria further helped reduce selection bias.
The study mainly focused on articles from developing countries. Overall, India had more reported studies (Riaz et al., 2021), followed by Egypt and Brazil (six). Other developing countries with research articles included Iran, Côte d’Ivoire, Jordan, Iraq, Saudi Arabia, Indonesia, and Pakistan (Figure 3). The source of articles aligns with the significant role these countries play in global cotton production, with India, Brazil, and Pakistan ranking among the top producers. Egypt and Côte d’Ivoire, known for their high-quality long-staple cotton, contribute significantly to the textile industry, while countries like Iran, Iraq, Saudi Arabia, Jordan, and Indonesia are emerging players in cotton research, textile industry and pest management, Most of the reviewed research articles (85%) reported research conducted under laboratory conditions (Figure 2), where 33 of 39 reviewed articles were laboratory experiments, two were both laboratory and semi-field, one was both laboratory and field, three semi-field and one field experiment. This underlines the importance of optimizing formulation efficacy and collecting foundational data in controlled situations before scaling to more complicated field settings. While laboratory investigations allow for exact variable measurement and early assessment of performance, the limited number of field trials raises questions about the formulations’ practical application in a variety of agricultural circumstances. The findings were organized into thematic categories.
The review identified 45 plant species researched for nanoformulations for management of cotton insect pests, with Azadirachta indica most frequently mentioned (12%), followed by Punica granatum (5%). Calotropis procera, Origanum vulgare, Piper aduncum, Pongamia pinnata and Thymus vulgaris accounting for 3% each. Other plant species accounted for 2% each onthe reviewed articles as follows; Acacia catechu, Allium sativum, Annona squamosa, Artemisia herba-alba, Basilicum ocimum, Borago officinalis, Camptotheca acuminata, Capsicum chinense, Citrus limon, Cinnamomum camphora, Cnidium monnieri, Cuminum cyminum, Cymbopogon citratus, C. nardus, Euphorbia hirta, Ficus religiosa, F.benghalensis, Glandora prostrata, Heliotropium indicum, Ipomea carnea, Jatropha curcas, Justicia adhatoda, Lippia multiflora, Matricaria chamomilla, Myrtus communis, Ocimum basilicum, Olea europaea, O. majorana., P. hispidinervum, P. glabra, Phytolacca americana, Rosmarinus officinalis, Ruta graveolens, Sophora flavescens, Tagetes patula, T. daenensis, Ulex europaeus, and Zingiber officinaleas. These plants were reportedly rich in bioactive compounds which enhance nanoparticle efficacy through targeted pest disruption (Figure 4).
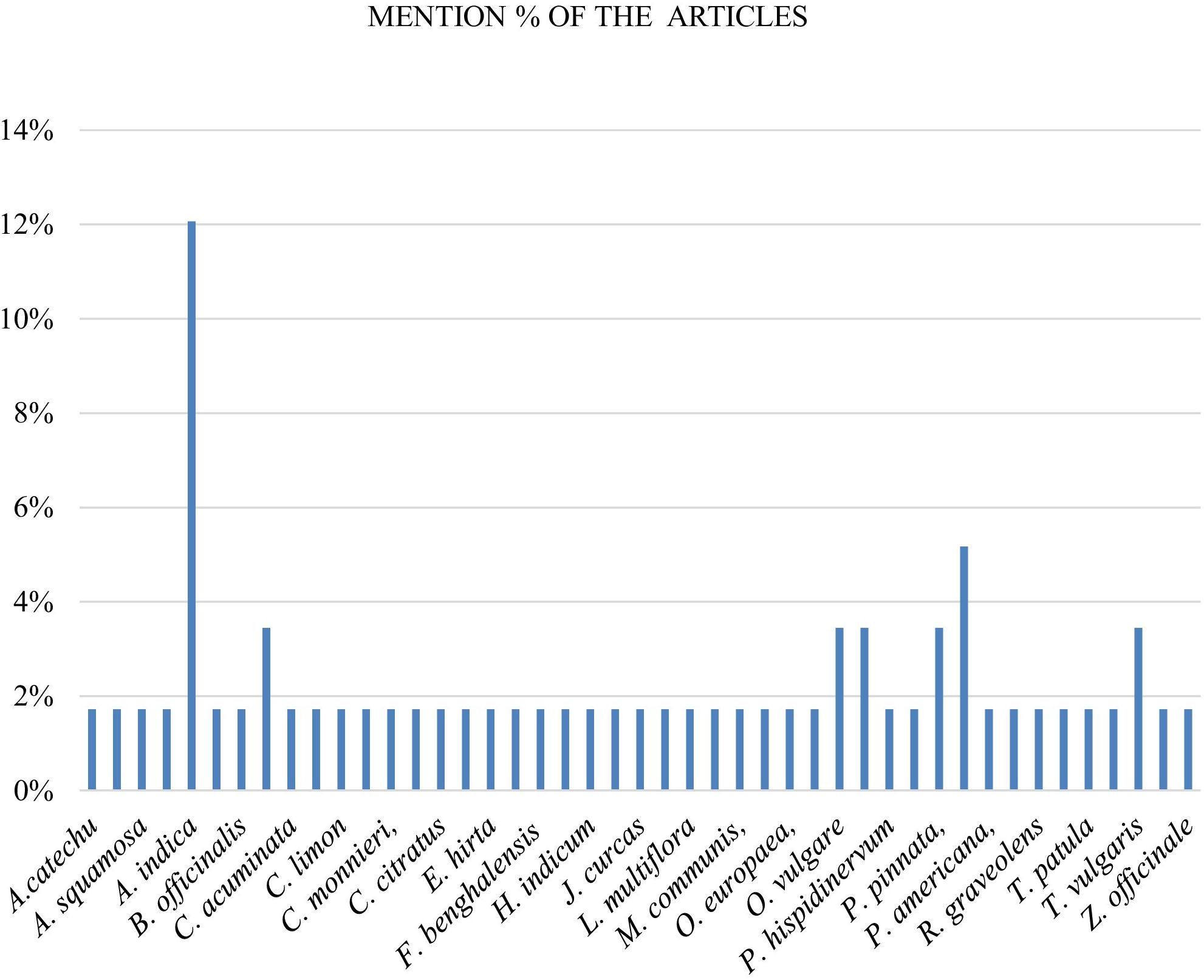
Figure 4. Percentage distribution of 45 pesticidal plant species researched for nanoformulations in cotton pest management.
The pesticidal plants are predominantly found within the Fabaceae, Lamiaceae, Asteraceae, Rutaceae, Apiaceae, Cucurbitaceae, Piperaceae, Solanaceae, Lythraceae, Caryophyllaceae, Convolvulaceae, Thymaceae, Phytolaccaceae, Rosaceae, Boraginaceae, Annonaceae, and Zingiberaceae plant families (Figure 5). The reviewed articles reveal that pesticidal compounds are found in various plant parts, including leaves, stems, roots, rhizomes, bark, flowers, fruits, cloves, and seeds (Bharani and Namasivayam, 2017; Khanra et al., 2018; Tia et al., 2021).
In the analysis of 39 reviewed articles on cotton pests tested against botanical nanoformulation, the most frequent studied insect was S. frugiperda cited in nine articles, followed closely by H. armigera cited in eight articles, Aphid species (A. gossypii, M. euphorbiae, B. brassicae) in five articles while S. litura and S. littoralis each appeared in four articles and P. solenopsis was referenced in three articles. Other insect pests like T. urticae and B. tabaci were mentioned in two articles each, while pests such as E. insulana, P. gossypiella and S. exigua appeared in one article for each (Table 3).
The botanical nanoformulation syntheses methods identified from the reviewed articles were five which include polymeric nanoparticles (30%), biogenic synthesis (25%), green synthesis (20%), chemical precipitation (10%), and nanoemulsification (15%). Polymeric carriers like chitosan and Poly-lactic-co-glycolic acid (PLGA) enhance stability and controlled release, while biogenic and green synthesis employ plant extracts for eco-friendly nanoparticle production. Chemical precipitation ensures precise nanoparticle size control, and nanoemulsification improves solubility and dispersion of essential oils (EOs). Nanomaterials identified from the 39 articles applied in botanical nanoformulations include metal-based nanomaterial such as silver nanoparticles (AgNPs), zinc oxide nanoparticle (ZnONPs), latex-fabricated gold nanoparticles (AuNPs), magnesium oxide nanoparticle (MgONPs) and copper oxide nanoparticles (CuONPs) and Polymeric nanomaterials like chitosan nanoparticles (ChNPs) and PLGA. Table 4 summarizes the nanomaterials used in the synthesis of botanical nanoformulations.
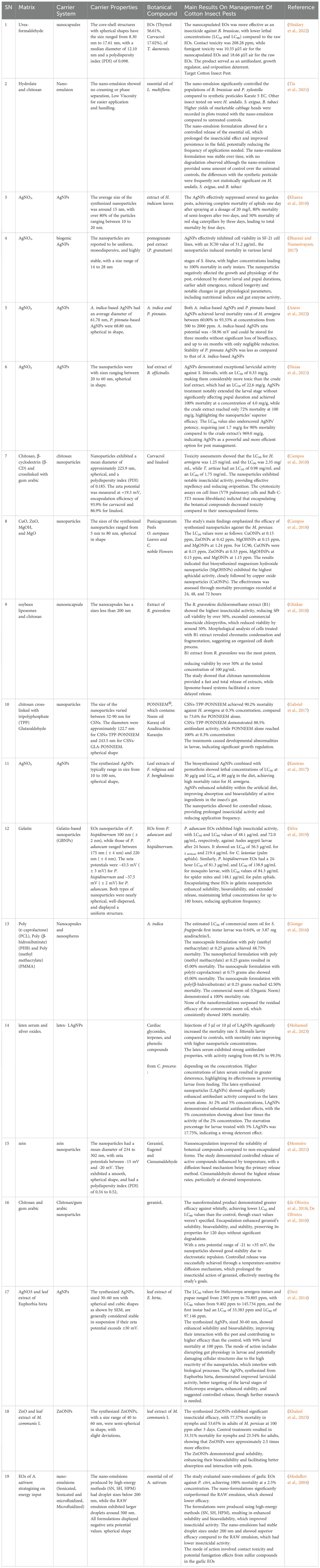
Table 4. Matrices to produce botanical nanoformulations and main results on management of cotton insect pest.
Evaluation of botanical nanoformulations from the reviewed articles showed diverse efficacy main reported on lethal concentration. Biosynthesis AgNPs using Oxymatrine had 0.5 ml/L as lethal concentration to kill 50% (LC50) and 1.5 ml/L lethal concentration to kill 90% (LC90) of A. gossypii, significantly increasing mortality compared to untreated controls (Sakban-Al-Tamimi et al., 2020). Biogenic AgNPs affected the developmental period of S. litura with an LC50 of 31.2 µg/mL showing improved efficacy over conventional insecticides (Bharani and Namasivayam, 2017). Silver nanoparticles synthesized from B. officinalis showed an LC50 of 0.33 mg/g and an LC90 of 1.7 mg/g against S. littoralis (Hazaa et al., 2021) indicating increased efficacy compared to traditional methods. Silver nanoparticles (A. indica) and (P. pinnata) against H. armigera, exhibited LC50 values of 500 ppm and 600 ppm with LC90 values reaching 2000 ppm and 1500 ppm respectively (Anees et al., 2022). In comparison, P. pinnata-based AgNPs demonstrated slightly higher LC50 and LC90 values, indicating a marginally reduced insecticidal activity as compared to A. indica based AgNPs. Green-synthesized silver nanoparticles from E. hirta demonstrated an LC50 of 2.905 ppm and an LC90 of 9.402 ppm against H. armigera (Devi et al., 2014), indicating significantly higher efficacy than conventional treatments. Silver nanoparticles from A. herba-alba exhibited LC50 values of 74.569 ml/ml (feeding) and 27.47 ml/ml (contact), LC90 values were 176.86 ml/ml (feeding) and 203 ml/ml (contact) (El-Ashmouny et al., 2022) signifying increased efficacy. Silver nanoparticles from A. catechu reported LC50 values of 71.04 mg/mL for S. litura and 85.33 mg/mL for H. armigera, LC90 for S. litura was 74.78 mg/mL and H. armigera was 88.91 mg/mL showing improved efficacy over traditional insecticides (Baranitharan et al., 2021). Toxicological effects of neem based AgNPs against H. armigera showed an LC50 of 114.67 ppm and LC90 of 202.71ppm (Asghar et al., 2022), indicating increased efficacy compared to standard treatments. Rosemary-AgNPs LC50 values for P. gossypiella and E. insulana were 18.655% and 16.75%, and their LC90 values were 143.29% and 64.39%, respectively (Kandil et al., 2024).
Screening of ZnONPs and CuONPs against S. frugiperda showed LC50 values of 520 ppm (ZnONPs) and 440 ppm (CuONPs) and LC90 for ZnONPs was 750 ppm and 500 ppm for CuONPs while neem alone LC50 was 500 ppm and LC90 was 1000 ppm indicating improved efficacy for nanoformulations (Mohammad et al., 2024). On the other toxicological effect of ZnONPs against H. armigera was LC50 127.79 ppm and LC90 242.97 ppm (Asghar et al., 2022).
Efficacy of polymeric nanoparticles were extracxted from the reviewed article and presented below. Nanocapsules loaded with T. daenensis EOs exhibited an LC50 of 208.28 ppm (nanocapsulated) and 702.57 ppm (raw EOs) and LC90 was 1026.44 ppm (nanocapsulated), LC90 2367.62 ppm (raw EOs) (Heidary et al., 2022) indicating increased efficacy with the nanocapsulated form. Carvacrol and linalool co-loaded in chitosan nanoparticles showed LC50 values of 1.25 mg/mL on H. armigera and 1.5 mg/mL on T. urticae, LC90 2.5 mg/mL for H. armigera and 3.0 mg/mL for T. urticae (Campos et al., 2018) demonstrating increased efficacy. Encapsulated EOs against A. aegypti, T. urticae, and C. lataniae reported LC50 values of 48.1 µg/mL, 56.5 µg/mL, and 148.1 µg/mL respectively (Silva et al., 2019). Neem-based nanoformulations against S. frugiperda had an LC50 of 0.64% (3.87 mg azadirachtin/L) (Giongo et al., 2016). The LC50 values of B. ocimum EOs nanoemulsions was 45 mg/L, and O. marjorana EOs nanoemulsions was 188 mg/L against A. craccivora while LC50 values of B. ocimum EOs was 992 mg/L and O. marjorana EOs was 3162 mg/L on same insect (Abdelaal et al., 2021).
In the reviewed articles botanical nanoformulations shown significant promise for sustainable cotton pest management by enhancing the efficacy of botanical extracts and EOs (de Oliveira et al., 2018; Ibrahim, 2019). Nanoemulsions systems reportedly to address limitations of EOs applications by preventing degradation, improving residue half-life, and enhancing solubility, delivery, and mobility (Campos et al., 2018; Abdelaal et al., 2021; Rafea et al., 2022). In research conducted by Abdelaal et al. (2021) demonstrated that nanoemulsified B. ocimum and O. marjorana EOs had significantly lower LC50 values against A. craccivora (45 mg/L and 188 mg/L, respectively) compared to their non-nanoemulsified counterparts (992 mg/L and 3162 mg/L, respectively). Similarly, nanoemulsions of L. multiflora EOs significantly reduced B. brassicae populations (28.48 ± 0.2%) (Manjesh et al., 2022) highlighting their potential to overcome limitations of traditional EOs applications cotton pest management.
Polymeric nanomaterials also exhibit enhanced pest management efficacy. Urea-formaldehyde nanocapsules demonstrate a lower LC50 of 208.28 ppm and LC90 1026.44 as compared to 702.57 ppm 2367.62 ppm of raw EOs against B. brassicae (Heidary et al., 2022). Chitosan nanoparticles co-loaded with carvacrol, and linalool have LC50 values of 1.25 mg/mL against H. armigera and 1.5 mg/mL against T. urticae (Campos et al., 2018). Gelatin nanoparticles yield LC50 values of 48.1 µg/mL for A. aegypti, 56.5 µg/mL for T. urticae, and 148.1 µg/mL for C. lataniae (Silva et al., 2019). The polymeric nanoparticles demonstrated significantly higher insecticidal activity compared to non-nanoparticle products. In research conducted by Gabriel et al. (2017) Chitosan nanoparticles (CSNs)- tripolyphosphate (TPP)-PONNEEM achieved a 90.2% mortality rate third instar of H. armigera at a 0.3% concentration surpassing neem oil’s 73.6% mortality at the same concentration (Gabriel et al., 2017). This improvement in effectiveness highlights the benefits of nano-encapsulation, which not only enhanced the insecticidal properties of the active ingredient, but also induces developmental abnormalities in pests, leading to malformed adults and smaller pupae (Gabriel et al., 2017). In research conducted by Bae et al. (2022) nanoencapsulated neem formulations using whey protein showed a significant reduction in LC50 values over time, dropping from 24.1 mg/mL on day one to 3.0 mg/mL by day 11, compared to bulk neem seed extract, which only decreased from 35.3 mg/mL to 8.5 mg/mL over the same period (Bae et al., 2022). Encapsulating azadirachtin in lignin microparticles accelerated mortality 100% faster in S. frugiperda and D. saccharalis, than non-encapsulated neem extracts (Costa et al., 2017) further highlighting the benefits of nanoencapsulation for cotton insect pest control. These results indicate that polymeric nanoformulations offer superior pest control efficacy at lower concentrations, making them a promising avenue for developing effective botanical nanopesticides.
Botanical nanoformulations utilizing metallic nanoparticles (AgNPs, MgONPs, CuONPs AuNPs and ZnONPs) enhanced the stability, efficacy, controlled release and targeted deliver. Biosynthesis AgNPs using Oxymatrine resulted in an LC50 of 0.5 ml/L and an LC90 of 1.5 ml/L against A. gossypii, significantly enhanced mortality compared to controls (Sakban-Al-Tamimi et al., 2020). Biogenic AgNPs showed an LC50 of 31.2 µg/mL against third instar of S. litura, outperforming conventional insecticides (Bharani and Namasivayam, 2017) while AgNPs from B. officinalis had LC50 and LC90 values of 0.33 mg/g and 1.7 mg/g against third instar of S. littoralis (Hazaa et al., 2021). Rosmarinus officinalis AgNPs showed LC50 values of 16.75% and 18.65% against E. insulana and P. gossypiella, respectively (Kandil et al., 2024). Siliver nanoformulations of A. indica and P. pinnata against second instar H. armigera exhibited LC50 values of 500 ppm and 600 ppm with LC90 values of 2000 ppm and 1500 ppm respectively (Anees et al., 2022). Research conducted by Asghar et al. (2022) stage-specific efficacy for neem AgNPs was observed where toxicity increased as larvae progressed to later instars, with LC90 values rising from 202.71 ppm in the third instar to 240.90 ppm in the fifth instar of H. armigera (Asghar et al., 2022). Zinc oxide nanoparticles (ZnO NPs) synthesized from neem extracts were also reported to enhance effective control of H. armigera where they exhibited LC50 values of 127.79 ppm, 114.069 ppm, and 155.38 ppm for the third, fourth, and fifth instars, respectively (Asghar et al., 2022). Green-synthesized nanoparticles from E. hirta showed an LC50 of 2.905 ppm against H. armigera (Devi et al., 2014). Silver nanoparticles from A. catechu reported LC50 values of 71.04 mg/mL for S. litura and 85.33 mg/mL for H. armigera (Baranitharan et al., 2021). Screening of zinc oxide and copper oxide nanoparticles against S. frugiperda yielded LC50 values of 520 ppm for ZnONPs and 440 ppm for CuONPs (Mohammad et al., 2024). Utilizing metallic nanoparticles demonstrates a broad spectrum of insecticidal activity and highlights the potential of botanical nanoformulation in creating targeted and efficient cotton insect pest management solutions although the long-term effects of metallic nanomaterials remain to be uncovered.
Botanical nanoformulations, encompassing nanoemulsions, polymeric nanoparticles, and metallic nanoparticles, represent a transformative approach in cotton insect pest management. Despite the promising results of botanical nanoformulations, some studies have pointed to limitations in efficacy compared to commercial botanical products. Out of 39 articles reviewed, one research article by Giongo et al. (2016) demonstrated that neem oil, when formulated as nanoemulsions, showed a significantly lower efficacy than commercial neem oil. The commercial neem oil achieved 100% mortality of third instar of S. frugiperda with an LC50 of 0.64% (3.87 mg azadirachtin/L), while the nanoformulations only reached a maximum mortality rate of 48.75% at a higher concentration (Giongo et al., 2016). This highlights that, while nanoformulations offer distinct advantages in terms of prolonged pest control and reduced environmental impact, they may not always provide immediate lethal effects on par with synthetic chemical insecticides.
Although the performance of botanical nanoformulations is evidently, the reviewed articles primarily focused on chewing insects (particularly Lepidoptera, such as H. armigera, S. frugiperda, S. littoralis and a few sucking insects (B. tabaci, P. solenopsis and Aphis species). The emerging serious cotton insect pests in developing countries in particular Tanzania include A. biguttula, T. cinnabarinus, E. insulana and T. tabaci, were less studied hence a gap in the current research landscape (Table 3) (Gabriel et al., 2017; De Oliveira et al., 2018; Ibrahim et al., 2022).
Botanical nanoformulations for cotton insect pest management in the reviewed articles utilized several synthesis methods, each with unique advantages (Bharani and Namasivayam, 2017; Gabriel et al., 2017; Tia et al., 2021; Asghar et al., 2022; Ibrahim et al., 2022; Khaleel et al., 2023)). The reviewed articles showed polymeric nanoparticle synthesis utilized biodegradable polymers like chitosan and PLGA for controlled release and stability (Gabriel et al., 2017; De Oliveira et al., 2018; Ibrahim et al., 2022) Biogenic synthesis used plant extracts and microorganisms to eco-friendly stabilize nanoparticles, producing AgNPs and biogenic ZnONPs (Bharani and Namasivayam, 2017). Green Synthesis applied plant-based agents for high biocompatibility, including neem-based formulations (Patil et al., 2016; Al Jabri et al., 2022)). Chemical precipitation involved reactions between metal salts and reducing agents for reproducible nanoparticles like ZnONPs (Asghar et al., 2022). Lastly, nanoemulsification stabilized EOs in nanoscale emulsions, enhanced solubility and bioavailability (Tia et al., 2021; Rafea et al., 2022).
The mode of action and synergism of these nanoformulations are influenced by nanomaterial used, synthesis method and the nature of the bioactive compounds encapsulated. In the reviewed article AgNPs were widely studied and about 29% of the studies applied silver oxide as the nanomaterial. Silver nanoparticles exhibited neurotoxic effects, leading to paralysis and death in pests such as A. gossypii and H. armigera (Sakban-Al-Tamimi et al., 2020; Heidary et al., 2022). Silver nanoparticles shown significant larvicidal activity, which include disruption of physiological processes and inhibition of gut protease activity (Bharani and Namasivayam, 2017; Hazaa et al., 2021). The R. officinalis AgNPs treated pests exhibited extended developmental periods E. insulana and P. gossypiella up to 34.9 and 33.83 days as compared to 24.3 and 21.9 days of untreated insect pests respectively (Kandil et al., 2024) and reduced survival rates, emphasizing the nanoparticles’ ability to disrupt pest growth and development. The synthesis of AgNPs from B. officinalis leaves enhanced larval mortality compared to crude extracts, showcasing their synergism and potent insecticidal properties (Bharani and Namasivayam, 2017; Huded et al., 2023).
Chitosan nanoparticles exhibited antifeedant and larvicidal activities against various pests (Gabriel et al., 2017; Campos et al., 2018; De Oliveira et al., 2018; Ibrahim et al., 2022). The reviewed studies demonstrated that chitosan nanoparticles enhanced stability and bioavailability of EOs, such as those from T. daenensis, leading to improved pest management (Heidary et al., 2022). Their ability to inhibit acetylcholinesterase further signifies their potential in disrupting neurophysiological functions in insects, providing a synergistic effect when used in conjunction with other biopesticides (Asghar et al., 2022; Devi et al., 2023). Gelatin nanoparticles and urea-formaldehyde nanocapsules are biopolymer that encapsulated EOs, botanical extracts and their stability and effectiveness were improved (Silva et al., 2019). In research conducted by Silva et al. (2019) demonstrated that gelatin-based nanoparticles effectively encapsulate EOs from P. aduncum and P. hispidinervum, achieving encapsulation efficiencies of 79.2% at 500 µg/mL and 84.5% at 1,000 µg/m. These nanoparticles maintain lethal concentrations for over 140 hours, enhancing insecticidal efficacy through controlled release and improved solubility (Giongo et al., 2016).
Zinc oxide nanoparticles and CuONPs act through cuticle penetration and metabolic disruption, effectively controlling pests like S. frugiperda (Al Jabri et al., 2022; Devi et al., 2023; Mohammad et al., 2024). Zinc oxide nanoparticles and CuONPs when used alongside neem-based formulations, their efficacy was significantly enhanced, demonstrating a synergistic approach to pest management (Mohammad et al., 2024). Gold nanoparticles was reported to enhance effective delivery systems, enhanced bioavailability of bioactive compounds, improved insecticidal activity while reduced environmental impact (Patil et al., 2016; Al Jabri et al., 2022; Asghar et al., 2022; Devi et al., 2023). Their modes of action involved disruption of digestive enzymes and metabolic processes (Patil et al., 2016).
The diverse nanomaterials (silver, chitosan, zinc oxide, copper oxide, and gold nanoparticles) employed in botanical nanoformulations for cotton insect pest management exhibited varied modes of action, including neurotoxicity, antifeedancy, cuticle penetration, metabolic disruption, and enhanced bioavailability of bioactive compounds, often exhibiting synergistic effects when combined with botanical insecticides altogether are reported to be safe for non-targeted organism like was highlited by Modafferi et al. (2004) that nanoemulsions of A. sativum EOs showed significant insecticidal activity against P. citri without harming non-target species, the Apis mellifera L.
(Baker et al., 2016; Siddiqua et al., 2016; Huded et al., 2023) The reviewed literature demonstrated that nanoformulations significantly enhanced the efficacy, and delivery of biopesticides by improving their solubility, stability, and controlled release properties (Giongo et al., 2016; Costa et al., 2017; Gabriel et al., 2017; Ghidan et al., 2018; Khanra et al., 2018; Silva et al., 2019; Lopes et al., 2020; Baranitharan et al., 2021; Tia et al., 2021; Asghar et al., 2022; Anees et al., 2022; Bae et al., 2022; Metwally et al., 2022; Rafea et al., 2022; Madasamy et al., 2023; Kandil et al., 2024; Mohammad et al., 2024).
The reviewed articles demonstrated that botanical nanoformulations enhanced the stability, bioavailability, and controlled release of plant extracts, thereby improving their cytotoxic effects and retention of bioactive compounds. In the research conducted by Alfaro-Corres et al. (2023) demonstrated that nanoparticles synthesized from C. chinense leaf extracts exhibit significant stability, with Zeta potential values approaching 200 mV for bimetallic nanoparticles (Cu/Mn and Zn/Cu), suggesting prolonged insecticidal properties compared to unformulated extracts (Alfaro-Corres et al., 2023). Silver encapsulated extracts of H. indicum characterized by a particle size of approximately 200 nm and low polydispersity, have demonstrated effective interaction with insect cell membranes, resulting in chromatin condensation while displaying reduced toxicity to human cells (PA1 cell line) (Khanra et al., 2018).
The encapsulation of camptothecin (CPT) was found to enhance its stability and targeted efficacy, particularly in the alkaline environments typical of lepidopteran midguts (Huang et al., 2024). In research conducted by Huang et al (Huang et al., 2024)demonstrated that using a star polycation (SPc) nanocarrier significantly boosts the efficacy and persistence of CPT. Additionally, research by Yan et al. (2021) revealed that the osthole/SPc complex does not negatively affect non-target predators, such as ladybird beetles, indicating that botanical nanoformulations can be designed to minimize harm to beneficial species in the ecosystem. Similarly, research conducted by Monteiro et al. (2021) showed that formulations containing limonene and carvacrol did not exhibit phytotoxic effects on Phaseolus vulgaris plants, suggesting a lower risk to beneficial plants in agricultural settings.
Chitosan nanoemulsions facilitated rapid release of active ingredients, whereas liposomal formulations provided delayed release, leading to greater effectiveness than non-encapsulated extracts (Lopes et al., 2020). These formulations maintain stability without phase separation and benefit from enhanced solubility. Controlled release mechanisms have been shown to prolong insecticidal effects and improve overall efficacy (Tia et al., 2021). Additionally, in research conducted by Kandil et al. (2024) reported that AgNPs maintain stability, effective targeting, and good solubility, thereby enhancing pest control and extending bioavailability against H. armigera (Gabriel et al., 2017).
Despite the potential of botanical nanoformulations, several challenges and limitations have been identified for their practical application.
A significant challenge across the studies is the variability in the size and stability of nanoparticles due to environmental factors, like temperature and light, which affect their effectiveness as insecticides (Sola et al., 2014; Chakraborty and Mondal, 2016; Attia, 2018; Reddy and Chowdary, 2021). For instance, the biosynthesis of nanoparticles may lead to inconsistencies in their characterization, impacting reliability in agricultural applications (Tia et al., 2021; Anees et al., 2022) (Tia et al., 2021). However, improving synthesis techniques using controlled biogenic and polymeric methods can standardize nanoparticle properties, ensuring consistent application and efficacy.
Most studies were confined to laboratory settings, with only 6% extending to semi-field and/or field trials. This presents an opportunity for scaling up field trials across diverse agroecological zones to validate real-world applicability and address variability in pest behavior under different conditions.
The long-term ecological impacts of nanoformulations in agriculture was not yet well identified from the articles reviewed. The potential toxicity associated with biogenic nanoparticles may adversely affect non-target organisms and beneficial insects. This raises ecological implications regarding the accumulation of nanoparticles in the environment, which can threaten biodiversity (Heidary et al., 2022). Additionally, ensuring uniform distribution and penetration of nanoparticles in plant tissues was shown as a challenge, as inconsistent applications can lead to variable pest control outcomes (Khanra et al., 2018). However, the fact that plants were known to have less toxicity encouraged an advancement in nanotechnology that enables the development of targeted formulations tailored to specific pests. Additionally biogenic nanoparticles, derived from natural sources, exhibit low toxicity to non-target species, making them an environmentally sustainable choice.
Regulatory hurdles and a lack of infrastructure for nano-formulation production and distribution pose significant barriers to their commercialization (Gupta et al., 2021). Addressing these challenges requires collaborative efforts between researchers, policymakers, and industry stakeholders to promote the development and deployment of sustainable insect pest management solutions (Ibrahim, 2019; El-Naby et al., 2020; Priyanka et al., 2020; Vichakshana et al., 2022). The increasing demand for eco-friendly pest control solutions presents a market opportunity for these formulations, which can enhance agricultural productivity while reducing health risks and environmental impacts.
To ensure the successful adoption of botanical nanoformulations, several key steps must be taken. Large-scale, multi-location field trials are essential to evaluate their performance under real-world conditions, providing robust data on efficacy and scalability. Farmer training programs should be developed to educate smallholder farmers on the benefits, application techniques, and safety protocols for these formulations. Collaboration with policymakers is crucial to establish regulatory frameworks that ensure the safe production, distribution, and use of botanical nanoformulated products while fostering trust among stakeholders. Efforts should also focus on cost optimization through research and innovation, making botanical nanoformulations affordable and accessible to resource-constrained farmers. Finally, these solutions must be seamlessly integrated into existing Integrated Pest Management (IPM) frameworks to enhance pest control strategies and promote sustainable cotton insect pest management practices.
Botanical nanoformulations represent a transformative step toward sustainable cotton pest management in developing countries. Their integration into farming practices can reduce dependency on synthetic pesticides in management of cotton insect pests in developing countries, promote biodiversity, and align with global efforts toward environmentally responsible agriculture. These eco-friendly alternatives to synthetic pesticides enhance efficacy, stability, targeted delivery and environmental compatibility, addressing critical challenges in cotton production sustainability. By advancing formulation techniques, expanding field trials, and fostering collaborations among stakeholders, these innovations can significantly reduce dependency on synthetic pesticides, transforming the cotton production systems in developing countries. Future research must focus on overcoming scalability barriers and integrating these formulations into comprehensive pest management frameworks to achieve long-term agricultural sustainability.
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
RL: Conceptualization, Data curation, Formal Analysis, Methodology, Resources, Writing – original draft, Writing – review & editing. AM: Conceptualization, Data curation, Funding acquisition, Methodology, Resources, Supervision, Validation, Visualization, Writing – review & editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This review was funded by the Organisation for Women in Science and Development (OWSD) under the project; Botanical Research and Service Center (BRESC). Grant number 4500501096 and Tanzania Cotton Board, Box 61, Mwanza.
The authors extend their gratitude to the Nelson Mandela African Institution of Science and Technology for providing access to research resources. Special appreciation is given to Juliana Godfrey for her technical assistance during the preparation of this manuscript.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that Generative AI was used in the creation of this manuscript.
Summarizing large articles and sentences phrasing.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abdelaal K., Essawy M., Quraytam A., Abdallah F., Mostafa H., Shoueir K., et al. (2021). Toxicity of Essential Oils Nanoemulsion against Aphis craccivora and their Inhibitory Activity On Insect enzymes. Processes 9, 624. doi: 10.3390/pr9040624
Acheuk F., Basiouni S., Shehata A. A., Dick K., Hajri H., Lasram S., et al. (2022). Status and prospects of botanical biopesticides in Europe and Mediterranean countries. Biomolecules. 12, 1–30. doi: 10.3390/biom12020311
Aïhounton G. B. D., Henningsen A., Trifkovic N. (2021).Pesticide handling and human health: conventional and organic cotton farming in Benin standard-nutzungsbedingungen. Available online at: https://hdl.handle.net/10419/235554 (Accessed May 17, 2024).
Akhter W., Shah F. M., Yang M., Freed S., Razaq M., Mkindi A. G., et al. (2023). Botanical biopesticides have an influence on tomato quality through pest control and are cost-effective for farmers in developing countries. PloS One 18. doi: 10.1371/journal.pone.0294775
Alfaro-Corres A. E., González-Mendoza D., Ruiz-Sánchez E., Ail-Catzin C., Valdez-Salas B., Gutiérrez-Miceli F., et al. (2023). Insecticidal activity and physicochemical characterization of nanoparticles from foliar extract of capsicum chinense. J. Renew Mater 11, 3933–3943. doi: 10.32604/jrm.2023.031129
Al Jabri H., Saleem M. H., Rizwan M., Hussain I., Usman K., Alsafran M. (2022). Zinc oxide nanoparticles and their biosynthesis: overview. Life 12, 594. doi: 10.3390/life12040594
Anees M. M., Patil S. B., Kambrekar D. N., Chandrashekhar S. S., Jahagirdar S. (2022). Biosynthesis, Characterization, Evaluation, and Shelf-Life Study of Silver Nanoparticles against Cotton Bollworm, Helicoverpa armigera (Hubner) (Noctuidae: Lepidoptera). Nanomaterials. 12, 3511. doi: 10.3390/nano12193511
Asghar M. S., Sarwar Z. M., Almadiy A. A., Shami A., Mohamed R. A. E. H., Ahmed N., et al. (2022). Toxicological effects of silver and zinc oxide nanoparticles on the biological and life table parameters of helicoverpa armigera (Noctuidae: lepidoptera). Agric. (Switzerland) 12, 1744. doi: 10.3390/agriculture12101744
Ateeq-ur-Rehman, Alam Bhatti M. M., Ummad-ud-din U., Hasan Naqvi S. A. (2020). Cotton Production and Uses: Agronomy, Crop Protection, and Postharvest Technologies (Singapore: Springer), 271–282 p.
Attia M. A. (2018). Toxicity Of Certain Essential Oils Loaded on Silica Nanoparticles against Tribolium castaneum (Coleoptera: Tenebrionidae) Adults Egyptian. J. Plant Prot. Res. Institute 1, 19–31. Available at: www.ejppri.eg.net (Accessed May 05, 2024).
Bae M., Lewis A., Liu S., Arcot Y., Lin Y. T., Bernal J. S., et al. (2022). Novel biopesticides based on nanoencapsulation of azadirachtin with whey protein to control fall armyworm. J. Agric. Food Chem. 70, 7900–7910. doi: 10.1021/acs.jafc.2c01558
Baker M. A., Makhdum A. H., Nasir M., Imran A., Ahmad A., Tufail F. (2016). Comparative Efficacy of Synthetic and Botanical Insecticides against Sucking Insect Pest and their Natural Enemies on Cotton Crop 1. World Wide Fund Pakistan for nature conservation, Rahim Yar Khan, Pakistan. 2. Sustainable Agriculture Programme, World Wide Fund-Pakistan (WWF-Pakistan). J. mt. area Res. 1, 1–4. Available at: http://journal.kiu.edu.pk/index.php/JMAR (Accessed May 19, 2024).
Baranitharan M., Alarifi S., Alkahtani S., Ali D., Elumalai K., Pandiyan J., et al. (2021). Phytochemical Analysis And Fabrication Of Silver Nanoparticles using Acacia catechu: An Efficacious and Ecofriendly Control Tool Against Selected Polyphagous Insect Pests. Saudi J. Biol. Sci. 28, 148–156. doi: 10.1016/j.sjbs.2020.09.024
Bharani R. A., Namasivayam S. K. R. (2017). Biogenic silver nanoparticles mediated stress on developmental period and gut physiology of major lepidopteran pest spodoptera litura (Fab.) (Lepidoptera: noctuidae) - an eco-friendly approach of insect pest control. J. Environ. Chem. Eng. 5, 453–467. doi: 10.1016/j.jece.2016.12.023
Campos E. V. R., Proença P. L. F., Oliveira J. L., Pereira A. E. S., De Morais Ribeiro L. N., Fernandes F. O., et al. (2018). Carvacrol and linalool co-loaded in B-cyclodextrin-grafted chitosan nanoparticles as sustainable biopesticide aiming pest control. Sci. Rep. 8, 7623. doi: 10.1038/s41598-018-26043-x
Candra Lina E., Sonia Febrianty Holeng H., Nelly N., Ernis G. (2023). Nanoemulsion of the Mixture of Citronella Grass Distillation Waste and Piper aduncum Essential Oil to Control Spodoptera frugiperda (Lepidoptera: Noctuidae). Philippine Journal of Science. 152 (3), 1131–1137. doi: 10.56899/152.03.30
Chakraborty K., Mondal E. (2016).Azadirachta indica-A Tree with Multifaceted Applications: An Overview. Available online at: https://www.researchgate.net/publication/305601232 (Accessed June 29, 2024).
Costa E. S., Perlatti B., Da Silva E. M., Matos A. P., Da Silva M. F. G. F., Fernandes J. B., et al. (2017). Use of lignins from sugarcane bagasse for assembling microparticles loaded with azadirachta indica extracts for use as neem-based organic insecticides. J. Braz. Chem. Soc. 28, 126–135. doi: 10.5935/0103-5053.20160155
da Costa Inácio G., Alves J. V. B., Santos M. F. C., Vacari A. M., Figueiredo G. P., Bernardes W. A., et al. (2020). Feeding Deterrence Towards Helicoverpa armigera by Tithonia diversifolia tagitinin C-Enriched Extract. Arabian J. Chem. 13, 5292–5298. doi: 10.1016/j.arabjc.2020.03.008
de Oliveira J. L., Campos E. V. R., Fraceto L. F. (2018). Recent developments and challenges for nanoscale formulation of botanical pesticides for use in sustainable agriculture. J. Agric. Food Chem. 66, 8898–8913. doi: 10.1021/acs.jafc.8b03183
De Oliveira J. L., Campos E. V. R., Pereira A. E. S., Nunes L. E. S., Da Silva C. C. L., Pasquoto T., et al. (2018). Geraniol encapsulated in chitosan/gum arabic nanoparticles: A promising system for pest management in sustainable agriculture. J. Agric. Food Chem. 66, 5325–5334. doi: 10.1021/acs.jafc.8b00331
Devi M. C., Jameela M. S., Asharaja A., Rajan G. R., Lyyadurai M., Reegan D. (2023). Effect of Calotropis procera (Aiton) Dryand.Based Zinc Oxide Nanoparticles on the Cotton Pest Spodoptera litura Fab. Indian J. Exp. Biol. 61, 224–229. doi: 10.56042/ijeb.v61i03.68093
Devi D. G., Murugan K., Panneer Selvam C. (2014). Green Synthesis of Silver Nanoparticles using Euphorbia hirta (Euphorbiaceae) Leaf Extract Crop Pest Of Cotton Bollworm, Helicoverpa armigera (Lepidoptera: Noctuidae). Euphorbia hirta Silver Nanopaticles JBiopest. 5, 54–66.
El-Ashmouny R. S., Rady M. H., Merdan B. A., El-Sheikh T. A. A., Hassan R. E., Gohary E. G. E. El. (2022). Larvicidal And Pathological Effects Of Green Synthesized Silver Nanoparticles from Artemisia herba-alba against Spodoptera littoralis Through Feeding and Contact Application. Egyptian J. Basic Appl. Sci. 9, 239–253. doi: 10.1080/2314808X.2022.2063012
El-Naby A., Shimaa, Mahmoud F. H. E., El-Habal N. A. M., Abdou M. S., Abdel-Rheim K. H. M. (2020). Efficacy of prepared Castor Oil Nanoemulsion Formulation against Rice Weevil Sitophilus oryzae on Stored Wheat Grain and Its Acute Toxic effect on Albino Rats. Egy Sci. J. Pestic 6, 33–44. Available at: www.esjpesticides.org.eg (Accessed April 04, 2024).
Gabriel P. M., Ignacimuthu S., Gandhi M. R., Shajahan A., Ganesan P., Packiam S. M., et al. (2017). Comparative studies of tripolyphosphate and glutaraldehyde cross-linked chitosan-botanical pesticide nanoparticles and their agricultural applications. Int. J. Biol. Macromol 104, 1813–1819. doi: 10.1016/j.ijbiomac.2017.06.043
Ghidan A. Y., Al-Antary T. M., Awwad A. M., Ghidan O. Y., Araj S. E. A., Ateyyat M. A. (2018). Comparison of different green synthesized nanomaterials on green peach aphid as aphicidal potential. Fresenius Environ. Bull. 27, 7009–7016.
Giongo A. M. M., Vendramim J. D., Forim M. R. (2016). Evaluation of neem-based nanoformulations as alternative to control fall armyworm. Ciencia e Agrotecnologia 40, 26–36. doi: 10.1590/S1413-70542016000100002
Gupta M. N., Khare S. K., Sinha R. (2021). Interfaces between nanomaterials and microbes. (Boca Raton: Taylor and Francis group), 1–16 p.
Haddaway N. R., Macura B., Whaley P., Pullin A. S. (2017). ROSES flow diagram for systematic reviews. Version 1.0. doi: 10.6084/m9.figshare.5897389
Hazaa M., Alm-Eldin M., Ibrahim A. E., Elbarky N., Salama M., Sayed R., et al. (2021). Biosynthesis of Silver Nanoparticles using Borago officinslis Leaf Extract, Characterization and Larvicidal Activity Against Cotton Leaf Worm, Spodoptera littoralis (Bosid). Int. J. Trop. Insect Sci. 41, 145–156. doi: 10.1007/s42690-020-00187-8
Heidary M., Karimzadeh J., Jafari S., Negahban M., Shakarami J. (2022). Aphicidal Activity of Urea–Formaldehyde Nanocapsules Loaded with the Thymus daenensis Celak Essential Oil on Brevicoryne brassicae L. Int. J. Trop. Insect Sci. 42, 1285–1296. doi: 10.1007/s42690-021-00646-w
Hou Q., Zhang H., Bao L., Song Z., Liu C., Jiang Z., et al. (2021). Ncs-delivered pesticides: A promising candidate in smart agriculture. Int. J. Mol. Sci. 22. doi: 10.3390/ijms222313043
Huang X., Ni X., Li H., Wei Y., Wang Z., Zhen C., et al. (2024). Synergistic mechanism of botanical pesticide camptothecin encapsulated in a nanocarrier against fall armyworm: enhanced stability and amplified growth suppression. Ecotoxicol Environ. Saf. 284, 116900. doi: 10.1016/j.ecoenv.2024.116900
Huded S., Savadatti E., Sharanappa C. H., Deb L., Borah A., Singh V., et al. (2023). Examining modern strategies for effective and sustainable agricultural plant protection techniques: A review. Int. J. Environ. Climate Change. 13, 1331–1343. doi: 10.9734/ijecc/2023/v13i113285
Ibrahim S. S. (2019). Essential oil nanoformulations as a novel method for insect pest control in horticulture. Horticulture. Available at: www.intechopen.com (Accessed May 05, 2024).
Ibrahim S. S., Abou-Elseoud W. S., Elbehery H. H., Hassan M. L. (2022). Chitosan-cellulose nanoencapsulation systems for enhancing the insecticidal activity of citronella essential oil against the cotton leafworm spodoptera littoralis. Ind. Crops Prod 184. doi: 10.1016/j.indcrop.2022.115089
Isman M. B. (2019). Botanical insecticides in the twenty-first century-fulfilling their promise? Annu Rev Entomol. 65, 233–249. doi: 10.1146/annurev-ento-011019-
Kandil M. A. A., El-Shennawy R. M., El-Medany W. A., Amer A. A., Abd-Elazeem E. M. (2024). Utilizing Rosmarinus officinalis Nanoparticles for Eco-friendly Control against Two Cotton Bollworms: Investigating Biological, Biochemical, and Histological Effects. Catrina 31, 41–54. doi: 10.21608/cat.2024.254614.1234
Kantrao S., Ravindra M. A., Akbar S. M. D., Jayanthi P. D. K., Venkataraman A. (2017). Effect of biosynthesized silver nanoparticles on growth and development of helicoverpa armigera (Lepidoptera: noctuidae): interaction with midgut protease. J. Asia Pac Entomol 20, 583–589. doi: 10.1016/j.aspen.2017.03.018
Karani A., Ndakidemi P., Mbega E. (2017). Botanical pesticides in management of common bean pests: importance and possibilities for adoption by small-scale farmers in Africa. J. Appl. Life Sci. Int. 12, 1–10. doi: 10.9734/JALSI/2017/32503
Khaleel A. I., Mohmed A. S., Al-Taey D. K. A., Kamaluddin Z. N. (2023). Green Synthesis of ZnO Nanoparticles Using Myrtus communis L. Extract and their Insecticidal Activity against Myzus persicae (Sulzer). Biopestic Int. 19, 143–148. doi: 10.59467/BI.2023.19.143
Khanra K., Choudhuri I., Bhattacharyya N. (2018). Bio-efficacy of nanoparticles in tea garden prepared from heliotropium indicum. Res. Crops 19, 320–324. doi: 10.5958/2348-7542.2018.00049.9
Kumar Sharma A., Kumar S., Singh M. (2022). Pesticides overburden in agriculture and its consequences-A review. J. Surv Fish Sci. 08, 394–397.
Lopes A. I. F., Monteiro M., Araújo A. R. L., Rodrigues A. R. O., Castanheira E. M. S., Pereira D. M., et al. (2020). Cytotoxic plant extracts towards insect cells: bioactivity and nanoencapsulation studies for application as biopesticides. Molecules 25, 5855. doi: 10.3390/molecules25245855
Madasamy M., Sahayaraj K., Sayed S. M., Al-Shuraym L. A., Selvaraj P., El-Arnaouty S. A., et al. (2023). Insecticidal Mechanism of Botanical Crude Extracts and their Silver Nanoliquids on Phenacoccus solenopsis. Toxics 11, 305. doi: 10.3390/toxics11040305
Malinga L. (2021). The importance and economic status of cotton production. Academia Lett. 2317, 1–8. doi: 10.20935/AL2317
Manjesh K., Kundu A., Dutta A., Saha S., Neelakanthaiah B. S. (2022). Bio-insecticidal nanoemulsions of essential oil and lipid-soluble fractions of pogostemon cablin. Front. Plant Sci. 13. doi: 10.3389/fpls.2022.874221
Metwally H. M., Ibrahim S. S., Sammour E. A. (2022). Aphicidal and biochemical effects of emulsifiable concentrate and nanoemulsion of two selected essential oils against black bean aphid, aphis fabae (Scop.). Egyptian Pharm. J. 21, 318–327. doi: 10.4103/epj.epj_40_22
Mkindi A. G., Tembo Y., Mbega E. R., Medvecky B., Kendal-smith A., Farrell I. W., et al. (2019). Phytochemical Analysis of Tephrosia vogelii Across East Africa Reveals three Chemotypes that Influence its Use as a Pesticidal Plant. Plants 8, 597. doi: 10.3390/plants8120597
Mkindi A. G., Tembo Y. L. B., Mbega E. R., Smith A. K., Farrell I. W., Ndakidemi P. A., et al. (2020). Extracts of common pesticidal plants increase plant growth and yield in common bean plants. Plants 9, 149. doi: 10.3390/plants9020149
Modafferi A., Giunti G., Urbaneja A., Laudani F., Latella I., Pérez-Hedo M., et al. (2004). High-Energy Emulsification of Allium sativum Essential Oil Boosts Insecticidal Activity against Planococcus citri with no risk to Honeybees. J. Pest Sci. 2024. doi: 10.1007/s10340-024-01800-2
Mohamed N. H., Ali A. M. H., Tawfik A. I., Ismail M. A., Abdel-Mageed W. M., Shoreit A. A. M., et al. (2023). Feeding Deterrence and Larvicidal Effects of Latex Serum and Latex-Synthesized Nanoparticles of Calotropis procera against the Cotton Leafworm, Spodoptera littoralis Open Access Full Length Research Article Advancements in Life Sciences-International Quarterly. J. Biol. Sci. 10 (4), 585–592. Available at: www.als-journal.com (Accessed October 27, 2024).
Mohammad A. M., Shaker A. M., Salem O. M. A., Elmashay A., Hassani R. (2024). Screening the larvicidal activity of zno, cuo nanoparticles, and neem seed oil extract against the full armyworm spodoptera frugiperda (J. E. Smith) (Lepidoptera, noctuidae). Emir J. Food Agric. 36, 1–9. doi: 10.3897/ejfa.2024.118380
Monteiro R. A., Camara M. C., de Oliveira J. L., Campos E. V. R., Carvalho L. B., Proença P. L. de F., et al. (2021). Zein based-nanoparticles loaded botanical pesticides in pest control: an enzyme stimuli-responsive approach aiming sustainable agriculture. J. Hazard Mater 417. doi: 10.1016/j.jhazmat.2021.126004
Natal C. M., Fernandes M. J. G., Pinto N. F. S., Pereira R. B., Vieira T. F., Rodrigues A. R. O., et al. (2021). New carvacrol and thymol derivatives as potential insecticides: synthesis, biological activity, computational studies and nanoencapsulation. RSC Adv. 11, 34024–34035. doi: 10.1039/D1RA05616F
Nursal I. S. (2019). “The effectiveness of botanical insecticides of four plant types and adjuvants on nutrition index of the fifth instar larvae of heliothis armigera hubner,” in The 4th International Conference on Biological Sciences and Biotechnology (ICBSB) Medan (Institute of Physics Publishing).
Overton K., Maino J. L., Day R., Umina P. A., Bett B., Carnovale D., et al. (2021). Global Crop Impacts, Yield Losses and Action Thresholds for Fall Armyworm (Spodoptera frugiperda): A review. Crop Prot. 145. doi: 10.1016/j.cropro.2021.105641
Page M. J., Moher D., Bossuyt P. M., Boutron I., Hoffmann T. C., Mulrow C. D., et al. (2021). PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. BMJ 372. doi: 10.1136/bmj.n160
Patil C. D., Borase H. P., Suryawanshi R. K., Patil. S. V. (2016). Trypsin inactivation by latex fabricated gold nanoparticles: A new strategy towards insect control. Enzyme Microb. Technol. 92, 18–25. doi: 10.1016/j.enzmictec.2016.06.005
Prema S., Ganapathy N., Renukadevi P., Mohankumar S., Kennedy J. S. (2018). Efficacy of Different Botanical Extracts on Thrips palmi in cotton. J. Pharmacognosy Phytochem. 7, 2824.
Priyanka P., Kumar D., Yadav A., Yadav K. (2020). “Nanobiotechnology and its application in agriculture and food production,” in Nanotechnology in the Life Sciences (Switzerland: Springer Science and Business Media B.V), 105–134.
Qarachal J. F., Sheidaee E., Bazyar P. (2024). The impact of various nanomaterials and nano-agrochemicals on agricultural systems. Journal of Engineering in Industrial Research 4 (4), 226–243. doi: 10.48309/jeires.2023.4.5
Rafea H. S., Ali S. H., El-Shiekh T. A., Youssef D. A., Amer H. M., El-Aasar A. E. M. (2022). Toxicological effect of camphor oil nanoemulsion on cotton leafworm and its safety evaluation on swiss albino mice. Egypt J. Chem. 65, 1301–1312. doi: 10.21608/EJCHEM.2022.139037.6140
Raju S. V. S., Sharma K. R. (2021). “Recent trends in insect pest management,” in Recent Trends in Insect Pest Management (AkiNik Publications).
Ratto F., Bruce T., Chipabika G., Mwamakamba S., Mkandawire R., Khan Z., et al. (2022). Biological control interventions reduce pest abundance and crop damage while maintaining natural enemies in sub-saharan Africa: A meta-analysis. Proc. R. Soc. B: Biol. Sci. 289. doi: 10.1098/rspb.2022.1695
Reddy D. S., Chowdary N. M. (2021). Botanical biopesticide combination concept—A viable option for pest management in organic farming. Egyptian J. Biol. Pest Control 31, 23. doi: 10.1186/s41938-021-00366-w
Riaz S., Johnson J. B., Ahmad M., Fitt G. P., Naiker M. (2021). A review on biological interactions and management of the cotton bollworm, helicoverpa armigera (Lepidoptera: noctuidae). J. Appl. Entomology 145, 467–498. doi: 10.1111/jen.v145.6
Saini P., Gupta K. K. (2024). Bioefficacy of botanicals with special emphasis on cassia fistula and nano-formulations on survival, growth, and development of insects: A sustainable approach of integrated pest management. Indian J. Nat. Prod Resour. 15 (2), 260–273. doi: 10.56042/ijnpr.v15i2.11503
Sakban-Al-Tamimi M., Zakaria Bakr S., Kh Mohammed J. (2020). The Effect Biosynthesis Of Silver Nanoparticles By Oxymatrine Botanical Insecticide against Aphis gossypii on Cucumber and their Predator Chrysoperla carnea. Euphrates J. Agric. Sci. 12, 586–597.
Sarker S., Akbor M. A., Nahar A., Hasan M., Islam A. R. M. T., Siddique M. A. B. (2021). Level of pesticides contamination in the major river systems: A review on south asian countries perspective. Heliyon 7, e07270. doi: 10.1016/j.heliyon.2021.e07270
Sarwar M. (2017). Biological Parameters of Pink Bollworm Pectinophora gossypiella (Saunders) (Lepidoptera: Gelechiidae): A looming Threat for Cotton and its Eradication Opportunity. Int. J. Res. Agric. Forestry. 4, 25–36.
Sarwar M. (2023). Botanic plant resources as insect pests administrator of field crops. J. Agric. Forestry Res. 2, 31–43. Available at: www.sarpo.net (Accessed March 20, 2023).
Sarwar M., Sattar M. (2016). An analysis of comparative efficacies of various insecticides on the densities of important insect pests and the natural enemies of cotton, gossypium hirsutum L. Pakistan J. Zool 48, 136.
Shahid M., Ullah U. N., Khan W. S., Saeed S., Razzaq K. (2021). Application of nanotechnology for insect pests management: A review. J. Innovative Sci. 7, 28–39. doi: 10.17582/journal.jis/2021/7.1.28.39
Sharma S., Kooner R., Arora R. (2017). “Insect pests and crop losses,” in Breeding Insect Resistant Crops for Sustainable Agriculture (Springer, Singapore), 45–66.
Sharma S., Loach N., Gupta S., Mohan L. (2020). Phyto-nanoemulsion: an emerging nano-insecticidal formulation. Environ. Nanotechnology Monit. Management. 14. doi: 10.1016/j.enmm.2020.100331
Siddiqua T. A., Miah M. R. U., Amin M. R., Yesmin K. (2016). Use of botanicals as environmentally safe management for cotton jassid. J. Environ. Sci. Natural Resources. 9, 131–134. doi: 10.3329/jesnr.v9i2.32183
Silva L. S., Mar J. M., Azevedo S. G., Rabelo M. S., Bezerra J. A., Campelo P. H., et al. (2019). Encapsulation of Piper aduncum and Piper hispidinervum Essential Oils in Gelatin Nanoparticles: A Possible Sustainable Control tool of Aedes aEgypti, Tetranychus urticae and Cerataphis lataniae. J. Sci. Food Agric. 99, 685–695. doi: 10.1002/jsfa.2019.99.issue-2
Sola P., Mvumi B. M., Ogendo J. O., Mponda O., Kamanula J. F., Nyirenda S. P., et al. (2014). Botanical pesticide production, trade and regulatory mechanisms in sub-saharan Africa: making A case for plant-based pesticidal products. Food Secur 6, 369–384. doi: 10.1007/s12571-014-0343-7
Soumaré M., Havard M., Bachelier B. (2021). Cotton in west and central Africa: from the agricultural revolution to the agro-ecological transition. Cahiers Agricultures. 30, 5. doi: 10.1051/cagri/2020044
Srinivasan R., Tamò M., Subramanian S. (2022). The case for integrated pest management in Africa: transition from a pesticide-based approach. doi: 10.1016/j.cois.2022.100970
Thakur P., Thakur S., Kumari P., Shandilya M., Sharma S., Poczai P., et al. (2022). Nano-Insecticide: Synthesis, Characterization, and Evaluation of Insecticidal Activity of ZnONPs against Spodoptera litura and Macrosiphum euphorbiae. Appl. Nanoscience (Switzerland) 12, 3835–3850. doi: 10.1007/s13204-022-02530-6
Tia V. E., Gueu S., Cisse M., Tuo Y., Gnago A. J., Konan E. (2021). Bio-insecticidal effects of essential oil nano-emulsion of lippia multiflora mold. On major cabbage pests. J. Plant Prot Res. 61, 103–109. doi: 10.24425/jppr.2021.136270
Vichakshana G. A. D., Young D. J., Choo W. S. (2022). Extraction, purification, food applications, and recent advances for enhancing the bioavailability of 6-gingerol from ginger – A review. Qual. Assur. Saf. Crops Foods 14, 67–83. doi: 10.15586/qas.v14i4.104367
Wani S. H., Ali Khan A. (2016).Nanotechnology and insecticidal formulations. Available online at: https://www.researchgate.net/publication/304482380 (Accessed June 29, 2024).
Keywords: botanical, nanoformulations, pesticidal plant extracts, sustainable cotton insect pest management, eco-friendly nanopesticides, metal nanocarriers, polymeric nanocarriers
Citation: Luneja RL and Mkindi AG (2025) Advances in botanical-based nanoformulations for sustainable cotton insect pest management in developing countries. Front. Agron. 7:1558395. doi: 10.3389/fagro.2025.1558395
Received: 10 January 2025; Accepted: 03 March 2025;
Published: 25 March 2025.
Edited by:
Oscar Liburd, University of Florida, United StatesReviewed by:
Philip Fanning, University of Maine, United StatesCopyright © 2025 Luneja and Mkindi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Renatus Laurent Luneja, bHVuZWphckBubS1haXN0LmFjLnR6
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.