- 1Department of Laboratory Medicine, Hangzhou Medical College, Hangzhou, China
- 2Zhu’s Team, Guangdong Medical University, Zhanjiang, China
- 3Department of Biology, Chemistry, Pharmacy, Free University of Berlin, Berlin, Germany
- 4Guangdong Provincial Key Laboratory of Systems Biology and Synthetic Biology for Urogenital Tumors, Shenzhen Key Laboratory of Genitourinary Tumor, Department of Urology, The First Affiliated Hospital of Shenzhen University, Shenzhen Second People’s Hospital (Shenzhen Institute of Translational Medicine), Shenzhen, China
Optogenetic is a technique that combines optics and genetics to control specific neurons. This technique usually uses adenoviruses that encode photosensitive protein. The adenovirus may concentrate in a specific neural region. By shining light on the target nerve region, the photosensitive protein encoded by the adenovirus is controlled. Photosensitive proteins controlled by light can selectively allow ions inside and outside the cell membrane to pass through, resulting in inhibition or activation effects. Due to the high precision and minimally invasive, optogenetics has achieved good results in many fields, especially in the field of neuron functions and neural circuits. Significant advances have also been made in the study of many clinical diseases. This review focuses on the research of optogenetics in the field of neurobiology. These include how to use optogenetics to control nerve cells, study neural circuits, and treat diseases by changing the state of neurons. We hoped that this review will give a comprehensive understanding of the progress of optogenetics in the field of neurobiology.
Introduction
In 2005, optogenetics was born and appeared in the public (Boyden et al., 2005). Since the advent of optogenetics, many top medical journals have described it as a core technology for the future of humanity (Method of the year, 2010; News, 2010; Adamantidis et al., 2015). Optogenetics can be combined with molecular biology, viral biology and other methods to introduce foreign light-sensitive protein genes into living cells (Amitrano et al., 2021; Di Ventura and Weber, 2021). Therefore, optogenetics has made many achievements in the field of neurobiology. Such as exploring unknown neuron functions (Figure 1), the discovery of neural circuits (Figure 2), and treatment of neurological diseases (Figure 3). By using the technology of optogenetics, the research of many difficult diseases has been advanced greatly. Using optogenetics to induce the differentiation of neural progenitor cells, the researchers were able to treat stroke in mice (Yu et al., 2019; Peters et al., 2021; Vassalli et al., 2021). The mice were able to remove and reactivate a memory by changing the connections between related neurons in the brain with different frequencies of light (Nabavi et al., 2014). Blindness can be treated by using optogenetics to activate the photogene expression of the related photoactivation channels or pumps in retinal cells (Ostrovsky and Kirpichnikov, 2019). This technology have led us into a higher research field.
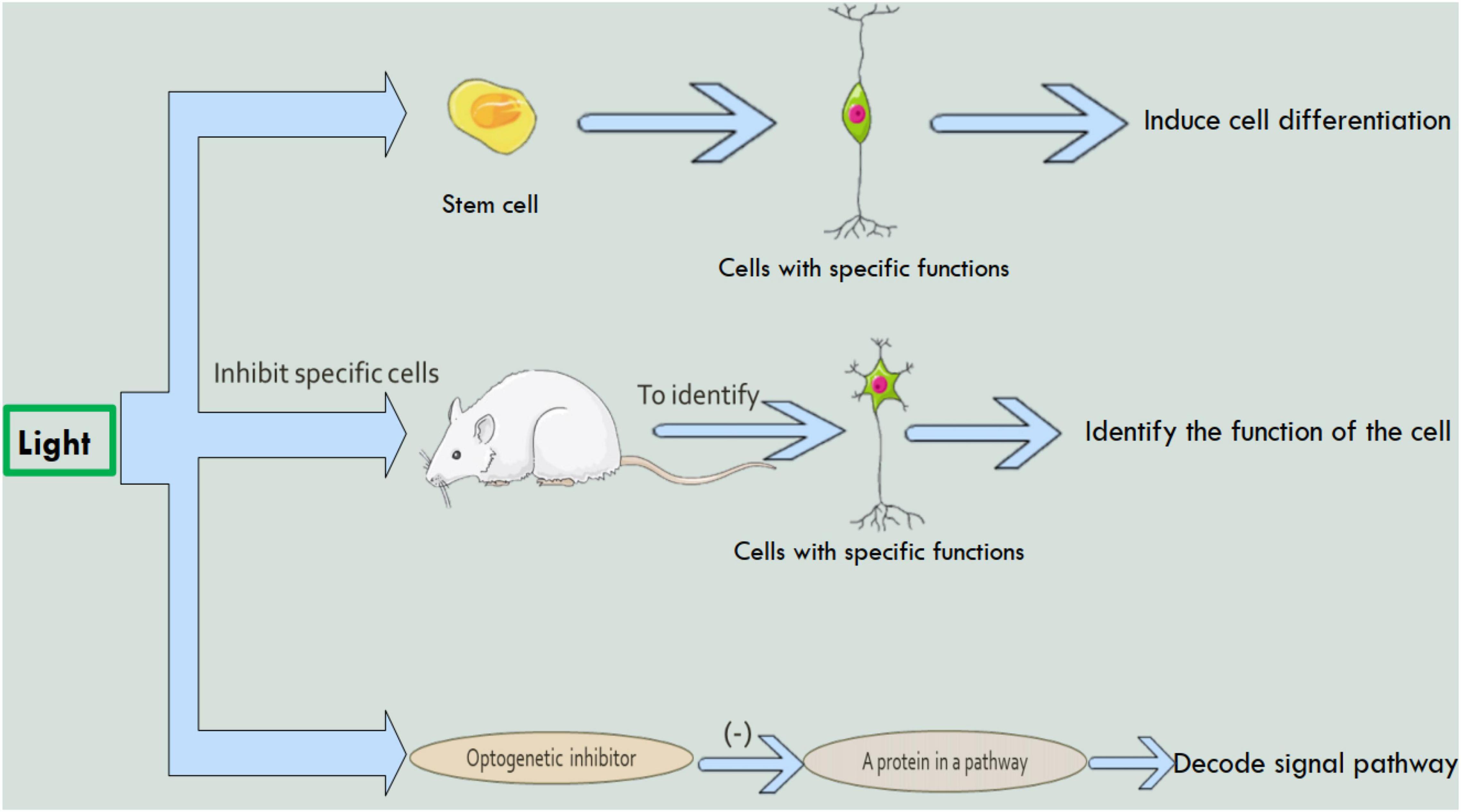
Figure 1. The study of optogenetics in the field of neurons. Using optogenetics, it is possible to induce stem cell differentiation, identify cell function, and decode intercellular signaling pathways.
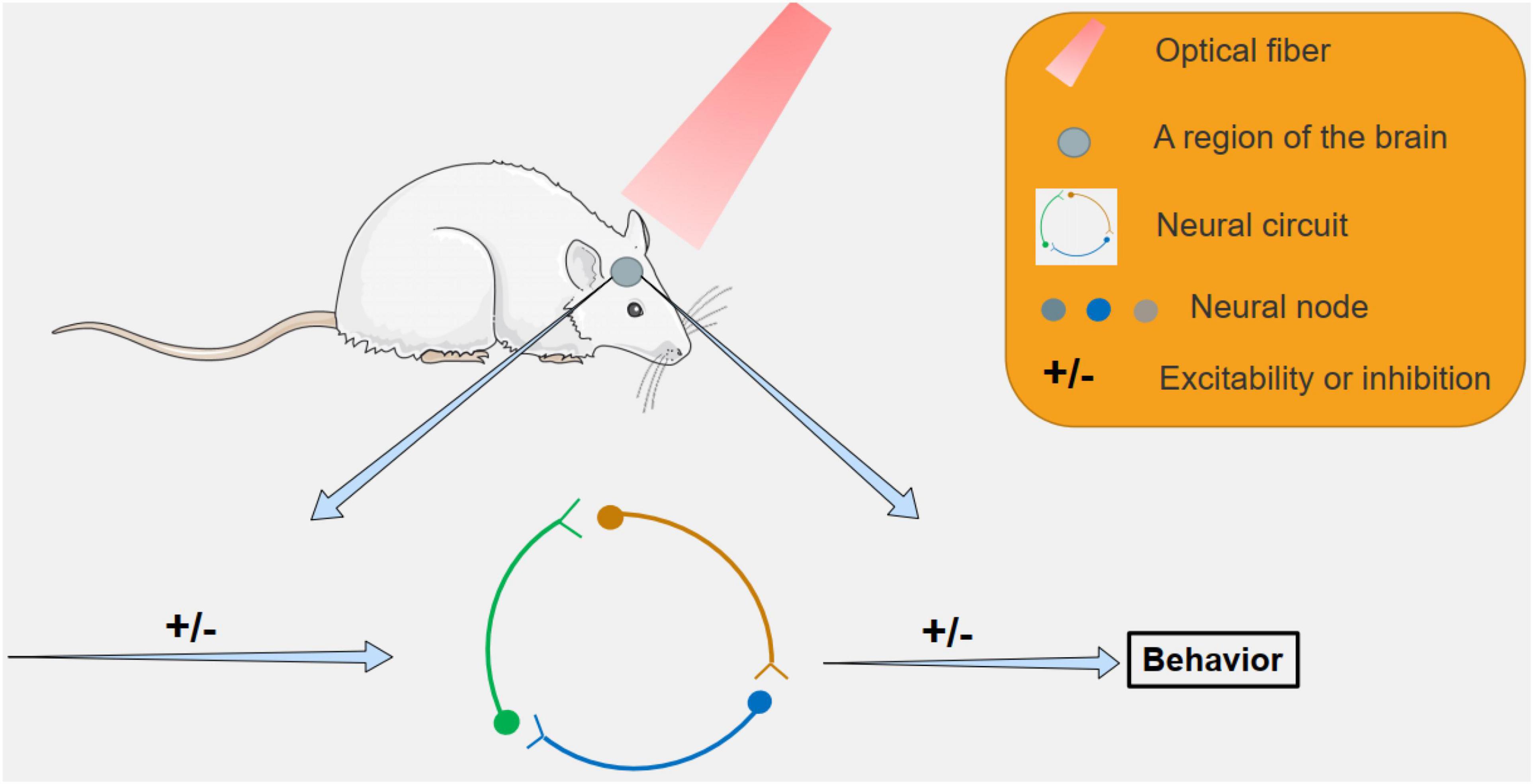
Figure 2. The study of neural circuits by optogenetic methods. By targeting specific areas with light, specific neural circuits can be inhibited or activated, leading to behavioral changes in mice, and related neural circuits can be studied.
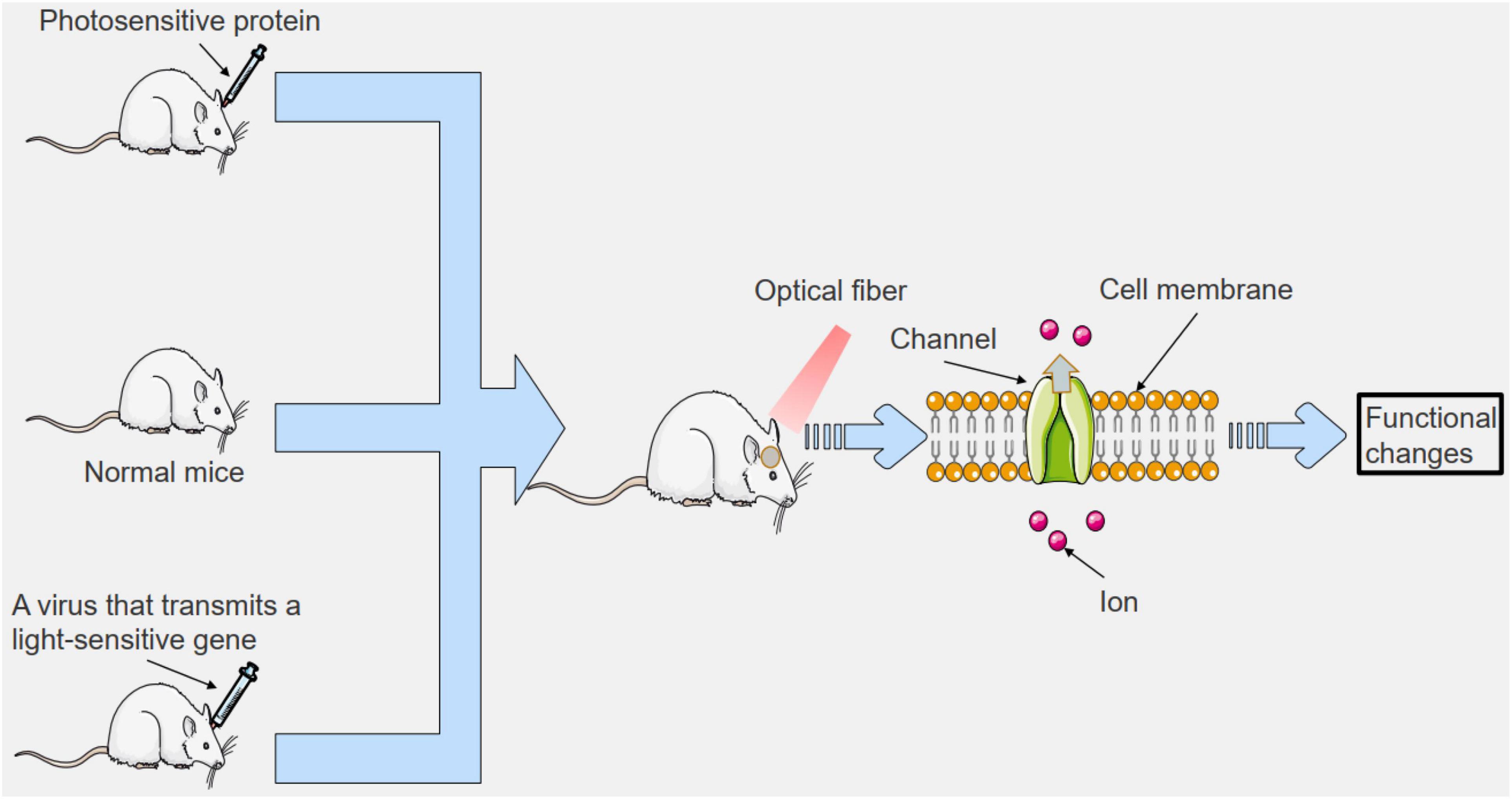
Figure 3. To study the mechanisms of clinical diseases using optogenetics. We can introduce photosensitive proteins outside the body, or we can introduce viruses that transmit photosensitive genes, or we can shine light directly on specific areas. This can lead to changes in cell membrane pathways in the irradiated area, which can affect cell function. The related functions of organisms can be altered to study clinical diseases.
Optogenetic Research Methods
Nerve Cells
Induce Neuronal Differentiation
In 2019, Luo et al. (2019) used photoelectric fullerene-bound photosensitive protein (HEBR) to reprogram and differentiate human fibroblasts. In this study, researchers transfected HEBR plastids into human fibroblasts using fullerene as a cell culture substrate (Figure 1). Previous research has shown that environmental stresses, such as acidity, can stimulate cell reprogramming. When the researchers illuminated HERB transfected fibroblasts with green light, the pH in and out of the transfected fibroblasts changed momentously, causing the fibroblasts to differentiate into neuron-like cells. This study has clinical significance in nerve repair.
In other studies, researchers combined a variety of technologies, such as optogenetics, synthetic biology (Padmanabhan et al., 2019; Hemmati et al., 2020; Yi et al., 2020; Zhang et al., 2020), for the first time to achieve far red light to control the expression of genomic genes. The far-red light-regulated CRISPR-dCas9 endogenous gene transcription activation device was developed for the first time (Gjaltema and Schulz, 2018), which successfully induced the pluripotent stem cells into functional neural cells (Shao et al., 2018). By combining BphS, which respond to red light proteins in rhodobacter, transcription factors BldD in streptococcus, and protein dCas9 in streptococcus pyogenes, this device can accurately realize the reversible activation of target genes inside and outside the organism, with high precision. Meanwhile, the frequency of light used in the study is in the physiological range and has no side effects on organisms. In theory, the results of this research can be widely used for precise epigenetic regulation (Hori and Kikuchi, 2019). And, in the future, this technology may be applied in the clinical field to treat diseases such as muscular dystrophy (Vajtay et al., 2019).
Control the Behavior of Nerve Cells
In earlier studies, optogenetics simply stimulated neurons (Nowak et al., 2010). Early applications of optogenetic control neurons were mainly two: driving proton pumps with light to charge mitochondria (Hallett et al., 2016), and polarizing or depolarizing neurons (Yao et al., 2012). By making a single neuron hyperpolarized, the function of this neuron can be studied (Aquili et al., 2014). In the latest study, researchers can decode and control signaling pathways in neurons (Figure 1; Melero-Fernandez de Mera et al., 2017). In this study, the intracellular signaling pathways of the organism can be controlled by light, using the main bioresonance effect of the organism. Using light to control specific signaling pathways that regulate the behavior of nerve cells, researchers can learn which neurons are involved in those pathways. In related studies, we can learn more about the JNK signaling pathway by inhibiting p38MAPK with OptoJNKI (a photosensitive substance that inhibits p38MAPK) (Melero-Fernandez de Mera et al., 2017). Moreira et al. (2019) managed to control the taste of fruit flies by shining different LEDs on different taste neurons. In this study, the researchers managed to alter feeding behavior in fruit flies by manipulating taste receptors. This technique can be used to study the progression of clinical diseases and to discover new therapies.
Study Neuronal Function
Studies have shown that people with schizophrenia and other psychiatric and neurological disorders have gamma oscillations in their brains (Gao et al., 2021a,b). But exactly how gamma oscillations are produced is not clear (Fan et al., 2020; Lu W. et al., 2020; Song et al., 2020). Cardin et al. (2009) discovered how the brain produces gamma oscillations by using optogenetics to manipulate the activity of nerve cells. By manipulating the interneurons’ related behavior with different frequencies of light, the researchers were able to observe the extent of the gamma oscillations produced by the interneurons. The research will contribute to a range of neurological disorders.
In 2014, researchers used optogenetics to inactivate cells in parts of the rat brain (Figure 1) to identify the neurons responsible for behavioral decision-making (Aquili et al., 2014). This study is the first to show that optogenetics inhibition of nucleus accumbens neurons during reward and false feedback can increase the behavioral complexity of individuals (Aquili et al., 2014). In the same year, another researcher used optogenetics to identify neurons that control aggression in the hypothalamus of mice (Lee et al., 2014). In 2017, scientists used optogenetics to find neurons in the brains of mice that control hunting behavior (Han et al., 2017). Because the hypothalamus in humans and mice is structurally similar, these findings are also useful for studying human behavior.
Neural Circuits
The Neural Circuits That Regulate Sodium Appetite
Sodium ions are important ions in the nervous system that regulate neurons. If sodium ion is not ingested for a long time, it will cause symptoms such as loss of appetite, weakness of limbs and dizziness. When a variety of animals are deficient in sodium ions, they will consume a large amount of salt rich in sodium ions, which is called sodium appetite (Thornton and Fitzsimons, 1995; Geerling and Loewy, 2008; Molnar and Labouesse, 2021). After a large intake of salt, the body will produce a sense of satisfaction to prevent further intake (Wolf et al., 1984). Previous studies on sodium appetite were flawed and did not conform to the single variable principle of the experiment. Until 2019, Lee et al. (2019) demonstrated that the pre-LCPDYN neurons are the core neurons in the regulation of sodium appetite by combining optogenetics with other techniques, and are regulated by the homeostatic and sense-related brain regions (Table 1 and Figure 2). In this study, researchers used optogenetic techniques to inhibit pre-LCPDYN neurons, and found that the pre-LCPDYN neurons are essential in the neural circuits that regulate sodium appetite. This experiment has revealed the mechanism of nerve circuits regulating sodium appetite and related conclusions.
The Nerve Basis of Compulsive Feeding
Globally, obesity (Hosseinpanah et al., 2019; Lu D. et al., 2020) and type 2 diabetes (Broder et al., 2014; Song et al., 2020; Liu Y. et al., 2021) are among the major diseases that endanger human health. Bad eating habits can bring about many diseases. Clinically, the treatment for severely obese patients is usually gastric bypass surgery. This approach is extremely traumatic. Optogenetics separates normal eating behavior from reward-seeking eating (Cardi et al., 2018), providing new ideas for the treatment of this disease.
Nieh et al. (2015) demonstrated that the hypothalamic-ventral tegmental pathway is involved in controlling feeding in starving mice by activating or inhibiting specific neurons using optogenetics (Jennings et al., 2015; Table 1 and Figure 2). In this study, the researchers introduced light-sensitive proteins that control the activity of neurons into the lateral hypothalami-ventral tegmental region (VTA) and activated the region with light, causing already satiated mice to take longer to eat.
In another study, Sternson et al. successfully distinguished appetitive behavior (Sternson and Atasoy, 2014) from neurons that satisfy behavior (Atasoy et al., 2012; Gatto and Goulding, 2018). In the study, the mice were given food freely or a reward for completing a task. Neuronal activity in the lateral hypothalamus of mice was also imaged. Based on this study, researchers were able to identify the neural basis of compulsive eating.
In 2021, the researchers used an AVV virus vector to deliver the ChR2 light-sensitive protein gene to a specific vagus nerve in the stomach (Kim et al., 2021; Table 1 and Figure 2). By using a tiny LED was inserted into the end of a flexible shaft in the stomach. Mice were successfully induced to feel full by external stimulation of specific gastric vagus nerve with remote radio frequency source. In 2022, researchers used optogenetics to inhibit neuropod cells in mice intestinal mucosa and found that mice consumed less sucrose (Buchanan et al., 2022). These studies suggest that optogenetics has a role to play in understanding the neural circuit of compulsive feeding.
Social Behavioral Neural Circuits
Social behavior is one of the characteristics of biology, but the social behavior neural circuits of biology is hardly understood. But through optogenetics, scientists are unraveling the mysteries of biological social behavior. Chiang and his colleagues developed an automatic laser tracking and optogenetic manipulation system (Hsiao et al., 1408; Wu et al., 2014) (ALTOMS) that can be used to study the social memory of fruit flies. After the expression of photosensitive proteins in neurons at specific sites, neurons involved in pain expression photosensitive pathways can be activated when laser irradiation is applied to specific sites (Karim et al., 2006; Table 1 and Figure 2). Using the system, the researchers were able to get certain males to quickly learn to avoid females, while other males continued to approach. At the same time, automated laser tracking and optogenetic manipulation systems (ALTOMS) (Hsiao et al., 1408; Wu et al., 2014) are expected to help identify the neural circuits responsible for specific drosophila behavior and understand the circuitry behind the ability to form memories based on social interaction learning.
In the latest study, researchers implanted tiny wireless optogenetic electronic devices into the brains of mice. The synchrony between brain neurons in the medial prefrontal cortex of mice induced social preference in mice (Yang Y. et al., 2021; Table 1 and Figure 2). The micro technique used in this study was less invasive and had less effect on the natural behavior of mice. Now, optogenetics not only controls the behavior of rodents but also primates (Rajalingham et al., 2021). These studies demonstrates the broad application of optogenetics in human social behavior circuits.
Body Temperature Regulating Neural Circuits
Thermoregulation is important for many life activities (Davison, 1976). Human beings have known for the last century that the thermoregulatory center is located in the hypothalamus (Hamilton and Ciaccia, 1971), but it is difficult to use traditional methods to analyze the thermoregulatory mechanism. In order to elucidate the neurons and neural circuits of hypothalamus involved in body temperature regulation, relevant researchers used optogenetics combined with physiological calcium signal recording (Tretyn, 1999) and other means to conduct experiments from the level of neural circuits on the hypothalamus of mice (Zhao et al., 2017). This study found neurons in the preoptic region of the hypothalamus that regulate thermally driven cooling behavior, as well as neurons in the dorsolateral part of the dorsolateral part of the hypothalamus responsible for the thermogenesis mechanism caused by cold stimulation. By using optogenetics to activate vLPO neurons, the researchers found that vLPO neurons are at the core neuron in thermoregulatory neural circuit (Table 1 and Figure 2; Zhao et al., 2017). In addition, a new marker for heat-sensitive neurons, brain-derived neurotrophic factor (BDNF), was identified (Ciszowski et al., 2016). This study provides new clues for physiological and pathological research based on thermoregulation.
Spatial Learning and Memory Circuits
Using techniques such as optogenetics, single synapse tracing (Spreafico et al., 1981) and in vivo multi-channel electrophysiological recording (Brozoski et al., 2006), the researchers found that the excitatory pyramidal cells of the entorhinal cortex (ECIIPN) (Shu et al., 2016) formed a single synaptic connection(eciipn-ca1pv synapses) with the inhibitory small abruption protein cells of the hippocampus CA1 region (CA1PV) (Shu et al., 2016; Yang et al., 2018). In the transgenic mouse model of Alzheimer’s disease, this memory loop is selectively damaged (Yang et al., 2018). Researchers used optogenetics stimulation therapy to repair eciipn-ca1pv synaptic degeneration damage and effectively treat memory loss in Alzheimer’s disease (Table 1 and Figure 2). This experiment proves that this loop is involved in regulating spatial learning and memory (Yang et al., 2018). In another study, researchers combined optogenetic technology with multi-channel synchronous optical stimulation and electrical recording technology, and found a circuit of emotional influence on spatial learning and memory in terms of structure and function (Zhang et al., 2017). Vahaba et al. (2017) used optogenetics to manipulate the NIF and HVC regions in the brains of zebra finches (Spierings and ten Cate, 2014). By controlling the interaction of these two regions, the researchers managed to encode the finches’ memories (Vahaba et al., 2017). Huang et al. (2021) (Table 1 and Figure 2), Huang et al. treated mice with phototherapy and recorded the potential changes in the hippocampal CA1 region of mice. The results showed that light therapy improved spatial memory and was associated with changes in the activity patterns of hippocampal neurons. These studies on spatial learning and memory could provide insights into the treatment of Alzheimer’s disease and some psychiatric disorders.
Neural Circuit Mechanisms That Activate Addictive Memory
Drug addiction (Martinez-Gonzalez et al., 2016; Xu et al., 2021) is an abnormal learning and memory process. Withdrawal scenarios can reactivate the addictive memory when the patient enters a scene that was previously associated with withdrawal symptoms (Hellemans et al., 2006). According to previous studies, the basolateral amygdala (BLA) plays an important role in inducing addictive memory retrieval (Wang et al., 2014; Khakpoor et al., 2016; Yi et al., 2021). However, its downstream neural circuits remain unknown. In this study, researchers combined neural tracer, optogenetics, chemical genetics and other methods. It was found that after activation of BLA -PrL loop, PrL was induced to transmit information back to BLA by projecting neurons, so as to activate the increase of Arc protein expression level in another group of BLA neurons and cause the recall of addictive memory (Table 1 and Figure 2; Alaghband et al., 2014). The study revealed the important role of the prefrontal cortex as a hub of the neural circuits in reactivating addictive memories in withdrawal scenarios, providing support for the treatment of drug addiction.
Nervous System-Based Clinical Research
Central Nervous System
Alzheimer’s Disease
Currently, none of the drugs used to improve cognitive function can fundamentally treat Alzheimer’s disease, but can only alleviate the symptoms. In 2016, Roy’s team successfully restored memory in mice using optogenetics (Table 1 and Figure 3; Roy et al., 2016). In the study, the researchers implanted light-sensitive proteins into the hippocampus of mice with memory loss. In response to light, memory cells in the hippocampus of the mice were activated. The next day, without light, the mice lost their memory again. As shown in the study, the hippocampus of the memory recovery mice established a strong connection with the entorhinal cortex (Meda et al., 2013) which is missing in Alzheimer’s patients (Liu and Zhang, 2019; Vallee et al., 2020). Another study used optogenetics stimulation therapy to repair eciipn-ca1pv synaptic degeneration damage and effectively treat memory damage caused by Alzheimer’s disease (Figure 3; Yang et al., 2018). At the same time, an important research achievement in Alzheimer’s disease is the treatment of cognitive dysfunction with near-infrared bioluminescence (Saltmarche et al., 2017; Arenas et al., 2020). Up to now, many studies have demonstrated that near-infrared bioluminescence therapy can improve cognitive function (Chao, 2019; Kinouchi et al., 2021). The above research may provide ideas for the radical cure of Alzheimer’s disease.
Parkinson’s Disease
Parkinson’s disease is a chronic disease with no clinical cure (Guo et al., 2019; Altinoz et al., 2020). However, Parkinson’s disease can be alleviated and treated with optogenetics. Ztaou et al. (2016) found that bilateral activation of indirect pathway MSNs by optogenetics can produce Parkinson’s-like presentation. However, activation of MSNs in the direct pathway alleviates symptoms such as freezing, bradykinesia, and difficulty in initiating movement (Kravitz et al., 2010; de Almeida and da Silva, 2021; Malaquias et al., 2021; Yang X. et al., 2021). Yang et al. (2018) found that the use of optogenetics combined with deep brain stimulation (DBS) (Castano-Candamil et al., 2019) to stimulate the afferent axons of the subthalamic nucleus region at high frequency can significantly treat Parkinson’s disease (Table 1 and Figure 3). Carter et al. (2010) used optogenetic targeting to control the LC-NE region of the cerebral cortex of mice, and were able to treat sleep disorders in mice with Parkinson’s disease (Figure 3). Steinbeck et al. (2015) induced rapid and reversible reactivation of motor defects in mice that had recovered from Parkinson’s motor defects induced by injury (Figure 3). And a recent study showed that photogenetic stimulation of the deep brain can relieve Parkinson’s disease in rats (Ingram et al., 2020; Yu et al., 2020). These studies suggest that optogenetics has great potential in the clinical treatment of Parkinson’s disease.
Epilepsy
More than 20% of epileptic patients develop stubborn resistance to epileptic drugs (Zhou et al., 2013; Murawiec et al., 2020), which eventually develops into refractory epilepsy (Martinez-Juarez et al., 2012; Tsai et al., 2021). The researchers injected green light-emitting nanoparticles into the hippocampus of mice (Roet et al., 2019), and irradiated the cranium surface with infrared light, and found that the epileptic neurons of mice were effectively silenced (Table 1 and Figure 3; Chen et al., 2018). The nanoparticles used in this experiment are stable, biocompatible and can be used for a long time. Lu et al. (2016) conducted in vivo and in vitro experiments, and the excitatory photosensitive protein was expressed in inhibitory neurons to inhibit epileptoid activity up to 70.0 and 82.4%, respectively. These studies indicate that optogenetic techniques are superior to other methods in the treatment of epilepsy.
Stroke
Cerebral apoplexy (Ng et al., 2019) is caused by the obstruction of blood flow to the brain tissues caused by vascular obstruction, which often occurs suddenly, with such symptoms as fainting, hemiplegia, and slant of the tongue, with a high mortality and disability rate. There is still a lack of effective treatment drugs, and transplantation of nerve progenitor cells is a good way to restore the function of nerve neurons in the brain (Cabral-Costa and Kowaltowski, 2020; Ostolaza et al., 2020; Roy-O’Reilly et al., 2020). Yu et al. (2019) provided luciferin CTZ to the brain of mice by intranasal administration (Table 1). When CTZ encounters luminescent proteins, it emits the required light. This study showed that survival rates for the growth and differentiation of neural progenitor cells increased significantly, more intact axons and nerve connections were produced, and better responses to electrical stimulation were achieved. The affected limb also showed better recovery. In young mice, stroke affected limb function was restored to normal levels, and even in older mice, stroke symptoms were partially recovered (Table 1 and Figure 3). The findings offer hope for an effective treatment for stroke.
Memory
A memory can bring either pleasure or fear (Kirmayer et al., 1995; Riksen and Netea, 2020). And relevant neurobiological studies have shown that an experience can cause changes in multiple brain areas, such as the cerebral cortex, hippocampus and amygdala, thus producing memory (Josselyn et al., 2015). In the following studies, optogenetics can manipulate memories and erase bad memories by manipulating neurons in the brain. In 2014, researchers successfully removed and reactivated a certain memory by changing the connections of related neurons in the brain of rats with different frequencies of light (Table 1 and Figure 3; Nabavi et al., 2014). In 2017, researchers used optogenetic technology in conjunction with electrophysiological technology and behavioral experiments (Ishikawa and Sakaguchi, 2013) to study the role of specific neural pathways in fear memory (Klavir et al., 2017; Yilmaz et al., 2020). In 2019, relevant studies for the first time found the subgroup of neurons that regulate the new memory of fear extinction, which improved people’s cognition of fear memory (Lacagnina et al., 2019). Also, in 2019, people used optogenetic methods to manipulate memory-related neurons to encode memory imprinting without experience for the first time (Table 1 and Figure 3; Vetere et al., 2019). The above research indicates that optogenetics plays an important role in the study of the mechanism of memory generation and memory-related diseases.
Peripheral Nervous System
Skeletal System
In the past, electric stimulation was often used for patients who lost motor function, and the electric stimulation treatment was prone to muscle fatigue and inaccurate discharge (Asakawa et al., 2021). However, optogenetics can stimulate a certain muscle fiber accurately and with low trauma, which can be used to study the treatment of motor system injury. In 2010, researchers started to apply optogenetics to the treatment of motor impairment (Table 1 and Figure 3). Llewellyn et al. (2010) utilized light stimulation of muscle fibers, and after 20 min of light stimulation of muscle fibers, the muscle still maintained a third of the maximum stress (Table 1 and Figure 3). Bryson et al. (2014) constructed a mouse model of muscle loss innervation and transplanted the embryoid containing ChR2 motor neurons into the mouse. By shining blue light on the transplant site, the researchers were able to restore leg muscle function (Table 1 and Figure 3). Srinivasan et al. (2018) used tiny LED lights to control light-sensitive proteins expressed in the legs of mice. The study succeeded in controlling ankle movement in mice. These studies suggest that optogenetics has great potential in controlling biological motor systems, especially in the treatment of paralysis and the treatment of muscle degeneration.
Urinary System
Related researchers developed a full closed-loop optogenetic control system (Mickle et al., 2019) and implanted it into female mice with drug-induced bladder dysfunction. This system can detect bladder filling, and the system can also irradiate the bladder for optogenetic control (Kessler et al., 2019). Shown in the study, photosensitive proteins are expressed in nerve cells of the bladder in mice by optogenetics technique, which makes the neurons in the bladder of mice in a hyperpolarized state. For 7 days after the system was implanted, the mice did well. Finally, the rats returned to normal bladder function (Table 1 and Figure 3; Mickle et al., 2019). Through further research and testing, this method is of clinical value.
Pain
Pain is one of the common clinical symptoms. Prolonged severe pain can seriously affect the patient’s quality of life. At present, photogenetic technology can solve the pain problem very well. In 2017, a new in-spinal optogenetics device was used for pain treatment and research (Table 1 and Figure 3; Samineni et al., 2017; Mai et al., 2020). Using this device, the researchers activated the afferent nerve of trpv1-chr2 channel protein, causing pain response behavior in mice (Jacob and Szerb, 1951; Zhu et al., 2020; Ji et al., 2021; Zhang et al., 2021). The researchers then ran a real-time comparison experiment, and the results were the same. Now that the device’s function is clear, it can be widely used in pain research. Another study used the selective silencing of related neurons by a wirelessly controlled electro-optical system to reduce ongoing pain and induced skin allergic reactions in mice under cystitis conditions (Table 1 and Figure 3; Samineni et al., 2017). And it had no bad effect on the mice. Hua et al. (2020) discovered a class of inhibitory neurons called “CeAga.” It turns off pain. Inhibition of the expression of CeAga neurons by optogenetics stopped the pain behavior in mice (Mai et al., 2020). Due to its high accuracy and low side effects, optogenetics may be widely used in the field of pain in the near future.
Vision
Special neurons in the retina react to light and transmit it to the brain to produce vision (Pozarickij et al., 2020; Creeden et al., 2021; Liu H. et al., 2021; Yu et al., 2021; Elkhalifa et al., 2022). When neurons in the retina stop working properly, the eye can’t work properly. Nowadays, optogenetic can be used to treat eye diseases such as color blindness (Table 1 and Figure 3; Cideciyan et al., 2016). The researchers sensitized the cells to light by the expression of light genes that encode light-activated channels or pumps in the remaining retinal cells (Ostrovsky and Kirpichnikov, 2019; Blomeier et al., 2021; He et al., 2021; Kramer et al., 2021; Mickoleit et al., 2021). Relevant research has achieved good results in recent years. In 2017, Russian scientists injected drugs with certain genetic structure into the blind eyes of experimental rodents, and the sight of experimental animals was partially restored (Aung et al., 2017). Berry et al. (2019) restored vision to blind mice (Table 1 and Figure 3). In 2021, the company of Bionic Sight successfully used optogenetics to enable patients with advanced retinitis pigmentosa to see light and motion (Harris and Gilbert, 2022).
Conclusion, Challenges and Perspectives
Since the advent of optogenetics, the technology has occupied the research field of neurobiology. Using optogenetics, researchers have decoded many neural circuits that cannot be decoded with other techniques. Such as social behavioral neural circuits, body temperature regulating neural circuits, spatial learning and memory circuits and so on. In view of the minimally invasive and high accuracy of optogenetics, optogenetics has a broad prospect in clinical treatment. Many irreversible diseases, especially neurodegenerative changes, can be solved by optogenetics. And the related research has entered the clinical trial stage.
Although optogenetics has achieved a lot in many fields, it still faces many challenges. Many scientists have proposed that exogenous light exposure causes neurons to respond in a non-physiological way, leading to incorrect physiological conclusions (Oh et al., 2021). And whether exogenous photosensitive proteins can have potential effects on nerve cells. Due to economic and other factors, optogenetics is mostly used in mouse experiments. Clinical trials of optogenetics are rare. Therefore, it will take a long time to prove that optogenetics can be widely applied to humans.
At present, optogenetics continues to flourish. Photogenetic treatments for retinal degeneration (Cehajic-Kapetanovic et al., 2019; Gilhooley et al., 2021) and pain (Harriott et al., 2021) are also in clinical trials. Researchers are also developing more precise (Adesnik and Abdeladim, 2021) and less invasive optogenetic devices, such as SOUL (Gong et al., 2020). At the same time, optogenetics has strong compatibility, it can be used to study a variety of diseases, such as diabetes (Li et al., 2021), inflammation (Baumschlager and Khammash, 2021; Bhat et al., 2021; Dos Santos et al., 2021; Jamaluddin et al., 2021; Michoud et al., 2021; Santos et al., 2021; Senok et al., 2021), tumors (Kim et al., 2017; Adampourezare et al., 2021; Esmaeili et al., 2022), depression (Hare et al., 2019), epilepsy (Zhang and Wang, 2021) and so on. And in 2020, 45 laboratories around the world integrated all optogenetics resources and create an optogenetics experimental database (Tremblay et al., 2020). According to the current progress, optogenetics has a broad prospect. It is believed that soon, optogenetics will become a major technique in neurobiology.
Author Contributions
XZ and ZL designed this study and supervised the research. WC wrote the manuscript. WL, YLi, ZZ, YX, YLiao, LY, QL, and MH discussed the manuscript. CL, ZL, and XZ edited the manuscript. All authors read and approved the final manuscript.
Funding
This work was supported partly by the National Natural Science Foundation of China (81972366), Guangdong Key Laboratory funds of Systems Biology and Synthetic Biology for Urogenital Tumors (2017B030301015), and its Open Grant (2021B030301015-3).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Adamantidis, A., Arber, S., Bains, J. S., Bamberg, E., Bonci, A., Buzsaki, G., et al. (2015). Optogenetics: 10 years after ChR2 in neurons–views from the community. Nat. Neurosci. 18, 1202–1212. doi: 10.1038/nn.4106
Adampourezare, M., Dehghan, G., Hasanzadeh, M., and Hosseinpoure Feizi, M. A. (2021). Application of lateral flow and microfluidic bio-assay and biosensing towards identification of DNA-methylation and cancer detection: recent progress and challenges in biomedicine. Biomed Pharmacother 141:111845. doi: 10.1016/j.biopha.2021.111845
Adesnik, H., and Abdeladim, L. (2021). Probing neural codes with two-photon holographic optogenetics. Nat Neurosci 24, 1356–1366. doi: 10.1038/s41593-021-00902-9
Alaghband, Y., O’Dell, S. J., Azarnia, S., Khalaj, A. J., Guzowski, J. F., and Marshall, J. F. (2014). Retrieval-induced NMDA receptor-dependent arc expression in two models of cocaine-cue memory. Neurobiol. Learn. Mem. 116, 79–89. doi: 10.1016/j.nlm.2014.09.001
Altinoz, M. A., Elmaci, I., Hacimuftuoglu, A., Ozpinar, A., Hacker, E., and Ozpinar, A. (2020). PPARdelta and its ligand erucic acid may act anti-tumoral, neuroprotective, and myelin protective in neuroblastoma, glioblastoma, and Parkinson’s disease. Mol. Aspects Med. 2020:100871. doi: 10.1016/j.mam.2020.100871
Amitrano, A. M., Berry, B. J., Lim, K., Kim, K. D., Waugh, R. E., Wojtovich, A. P., et al. (2021). Optical control of CD8(+) T cell metabolism and effector functions. Front. Immunol. 12:666231. doi: 10.3389/fimmu.2021.666231
Aquili, L., Liu, A. W., Shindou, M., Shindou, T., and Wickens, J. R. (2014). Behavioral flexibility is increased by optogenetic inhibition of neurons in the nucleus accumbens shell during specific time segments. Learn Mem. 21, 223–231. doi: 10.1101/lm.034199.113
Arenas, F., Castro, F., Nunez, S., Gay, G., Garcia-Ruiz, C., and Fernandez-Checa, J. C. (2020). STARD1 and NPC1 expression as pathological markers associated with astrogliosis in post-mortem brains from patients with Alzheimer’s disease and down syndrome. Aging 12, 571–592. doi: 10.18632/aging.102641
Asakawa, K., Handa, H., and Kawakami, K. (2021). Illuminating ALS motor neurons with optogenetics in zebrafish. Front. Cell Dev. Biol. 9:640414. doi: 10.3389/fcell.2021.640414
Atasoy, D., Betley, J. N., Su, H. H., and Sternson, S. M. (2012). Deconstruction of a neural circuit for hunger. Nature 488, 172–177. doi: 10.1038/nature11270
Aung, T., Ozaki, M., Lee, M. C., Schlotzer-Schrehardt, U., Thorleifsson, G., Mizoguchi, T., et al. (2017). Genetic association study of exfoliation syndrome identifies a protective rare variant at LOXL1 and five new susceptibility loci. Nat. Genet. 49, 993–1004. doi: 10.1038/ng.3875
Baumschlager, A., and Khammash, M. (2021). Synthetic biological approaches for optogenetics and tools for transcriptional light-control in bacteria. Adv. Biol. 5:e2000256. doi: 10.1002/adbi.202000256
Berry, M. H., Holt, A., Salari, A., Veit, J., Visel, M., Levitz, J., et al. (2019). Restoration of high-sensitivity and adapting vision with a cone opsin. Nat. Commun. 10:1221. doi: 10.1038/s41467-019-09124-x
Bhat, S. V., Price, J. D. W., and Dahms, T. E. S. (2021). AFM-based correlative microscopy illuminates human pathogens. Front. Cell Infect. Microbiol. 11:655501. doi: 10.3389/fcimb.2021.655501
Blomeier, T., Fischbach, P., Koch, L. A., Andres, J., Minambres, M., Beyer, H. M., et al. (2021). Blue light-operated CRISPR/Cas13b-mediated mRNA knockdown (lockdown). Adv. Biol. 5:e2000307. doi: 10.1002/adbi.202000307
Boyden, E. S., Zhang, F., Bamberg, E., Nagel, G., and Deisseroth, K. (2005). Millisecond-timescale, genetically targeted optical control of neural activity. Nat. Neurosci. 8, 1263–1268. doi: 10.1038/nn1525
Broder, H. L., Tormeti, D., Kurtz, A. L., Baah-Odoom, D., Hill, R. M., Hirsch, S. M., et al. (2014). Type II diabetes and oral health: perceptions among adults with diabetes and oral/health care providers in Ghana. Commun. Dent Health 31, 158–162.
Brozoski, T. J., Caspary, D. M., and Bauer, C. A. (2006). Marking multi-channel silicon-substrate electrode recording sites using radiofrequency lesions. J. Neurosci. Methods 150, 185–191. doi: 10.1016/j.jneumeth.2005.06.012
Bryson, J. B., Machado, C. B., Crossley, M., Stevenson, D., Bros-Facer, V., Burrone, J., et al. (2014). Optical control of muscle function by transplantation of stem cell-derived motor neurons in mice. Science 344, 94–97. doi: 10.1126/science.1248523
Buchanan, K. L., Rupprecht, L. E., Kaelberer, M. M., Sahasrabudhe, A., Klein, M. E., Villalobos, J. A., et al. (2022). The preference for sugar over sweetener depends on a gut sensor cell. Nat. Neurosci. 25, 191–200. doi: 10.1038/s41593-021-00982-7
Cabral-Costa, J. V., and Kowaltowski, A. J. (2020). Neurological disorders and mitochondria. Mol. Aspects Med. 71:100826. doi: 10.1016/j.mam.2019.10.003
Cardi, V., Mallorqui-Bague, N., Albano, G., Monteleone, A. M., Fernandez-Aranda, F., and Treasure, J. (2018). Social difficulties as risk and maintaining factors in anorexia nervosa: a mixed-method investigation. Front. Psychiatry 9:12. doi: 10.3389/fpsyt.2018.00012
Cardin, J. A., Carlen, M., Meletis, K., Knoblich, U., Zhang, F., Deisseroth, K., et al. (2009). Driving fast-spiking cells induces gamma rhythm and controls sensory responses. Nature 459, 663–667. doi: 10.1038/nature08002
Carter, M. E., Yizhar, O., Chikahisa, S., Nguyen, H., Adamantidis, A., Nishino, S., et al. (2010). Tuning arousal with optogenetic modulation of locus coeruleus neurons. Nat. Neurosci. 13, 1526–1533. doi: 10.1038/nn.2682
Castano-Candamil, S., Piroth, T., Reinacher, P., Sajonz, B., Coenen, V. A., and Tangermann, M. (2019). An easy-to-use and fast assessment of patient-specific DBS-induced changes in hand motor control in Parkinson’s disease. IEEE Trans. Neural. Syst. Rehabil. Eng. 27, 2155–2163. doi: 10.1109/TNSRE.2019.2941453
Cehajic-Kapetanovic, J., Birtel, J., McClements, M. E., Shanks, M. E., Clouston, P., Downes, S. M., et al. (2019). Clinical and molecular characterization of PROM1-related retinal degeneration. JAMA Netw Open 2:e195752. doi: 10.1001/jamanetworkopen.2019.5752
Chao, L. L. (2019). Effects of home photobiomodulation treatments on cognitive and behavioral function, cerebral perfusion, and resting-state functional connectivity in patients with dementia: a pilot trial. Photobiomodul Photomed Laser Surg 37, 133–141. doi: 10.1089/photob.2018.4555
Chen, S., Weitemier, A. Z., Zeng, X., He, L., Wang, X., Tao, Y., et al. (2018). Near-infrared deep brain stimulation via upconversion nanoparticle-mediated optogenetics. Science 359, 679–684. doi: 10.1126/science.aaq1144
Cideciyan, A. V., Roman, A. J., Jacobson, S. G., Yan, B., Pascolini, M., Charng, J., et al. (2016). Developing an outcome measure with high luminance for optogenetics treatment of severe retinal degenerations and for gene therapy of cone diseases. Invest. Ophthalmol. Vis. Sci. 57, 3211–3221. doi: 10.1167/iovs.16-19586
Ciszowski, K., Gomolka, E., Gawlikowski, T., Szpak, D., Potoczek, A., and Boba, M. (2016). [Brain-derived neurotrophic factor (BDNF) and nerve growth factor (NGF) blood levels in patients with acute carbon monoxide poisoning - a preliminary observations]. Przegl. Lek 73, 552–559.
Creeden, J. F., Imami, A. S., Eby, H. M., Gillman, C., Becker, K. N., Reigle, J., et al. (2021). Fluoxetine as an anti-inflammatory therapy in SARS-CoV-2 infection. Biomed. Pharmacother 138:111437. doi: 10.1016/j.biopha.2021.111437
Davison, J. (1976). Hydra hymanae: regulation of the life cycle by time and temperature. Science 194, 618–620. doi: 10.1126/science.982029
de Almeida, W. S., and da Silva, D. A. (2021). Does polysaccharide quaternization improve biological activity? Int. J. Biol. Macromol. 182, 1419–1436. doi: 10.1016/j.ijbiomac.2021.05.012
Di Ventura, B., and Weber, W. (2021). The rise of molecular optogenetics. Adv. Biol. 5:e2100776. doi: 10.1002/adbi.202100776
Dos Santos, A. G. A., da Silva, M. G. L., Carneiro, E. L., de Lima, L. L., Fernandes, A., Silveira, T. G. V., et al. (2021). A new target organ of leishmania (viannia) braziliensis chronic infection: the intestine. Front. Cell Infect. Microbiol. 11:687499. doi: 10.3389/fcimb.2021.687499
Elkhalifa, D., Rayan, M., Negmeldin, A. T., Elhissi, A., and Khalil, A. (2022). Chemically modified mRNA beyond COVID-19: Potential preventive and therapeutic applications for targeting chronic diseases. Biomed. Pharmacother 145:112385. doi: 10.1016/j.biopha.2021.112385
Esmaeili, Y., Khavani, M., Bigham, A., Sanati, A., Bidram, E., Shariati, L., et al. (2022). Mesoporous silica@chitosan@gold nanoparticles as “on/off” optical biosensor and pH-sensitive theranostic platform against cancer. Int. J. Biol. Macromol. 202, 241–255. doi: 10.1016/j.ijbiomac.2022.01.063
Fan, W., Mai, L., Zhu, X., Huang, F., and He, H. (2020). The role of microglia in perioperative neurocognitive disorders. Front. Cell Neurosci. 14:261.
Gao, G., Fan, C., Li, W., Liang, R., Wei, C., Chen, X., et al. (2021a). Mesenchymal stem cells: ideal seeds for treating diseases. Hum. Cell. 34, 1585–1600.
Gao, G., Li, C., Fan, W., Zhang, M., Li, X., Chen, W., et al. (2021b). Brilliant glycans and glycosylation: seq and ye shall find. Int. J. Biol. Macromol. 189, 279–291. doi: 10.1016/j.ijbiomac.2021.08.054
Gatto, G., and Goulding, M. (2018). Locomotion control: brainstem circuits satisfy the need for speed. Curr. Biol. 28, R256–R259. doi: 10.1016/j.cub.2018.01.068
Geerling, J. C., and Loewy, A. D. (2008). Central regulation of sodium appetite. Exp. Physiol. 93, 177–209. doi: 10.1113/expphysiol.2007.039891
Gilhooley, M. J., Hickey, D. G., Lindner, M., Palumaa, T., Hughes, S., Peirson, S. N., et al. (2021). ON-bipolar cell gene expression during retinal degeneration: implications for optogenetic visual restoration. Exp. Eye Res. 207:108553. doi: 10.1016/j.exer.2021.108553
Gjaltema, R. A. F., and Schulz, E. G. (2018). CRISPR/dCas9 switch systems for temporal transcriptional control. Methods Mol. Biol. 1767, 167–185. doi: 10.1007/978-1-4939-7774-1_8
Gong, X., Mendoza-Halliday, D., Ting, J. T., Kaiser, T., Sun, X., Bastos, A. M., et al. (2020). An ultra-sensitive step-function opsin for minimally invasive optogenetic stimulation in mice and macaques. Neuron 107:e8.
Guo, H. M., Zhou, Z. Y., Huang, Y. Q., Li, X., and Wang, X. J. (2019). Investigation of the roles of dysbindin-1 and SATB2 in the progression of Parkinson’s disease. Eur. Rev. Med. Pharmacol. Sci. 23, 7510–7516. doi: 10.26355/eurrev_201909_18865
Hallett, R. A., Zimmerman, S. P., Yumerefendi, H., Bear, J. E., and Kuhlman, B. (2016). Correlating in vitro and in vivo activities of light-inducible dimers: a cellular optogenetics guide. ACS Synth. Biol. 5, 53–64. doi: 10.1021/acssynbio.5b00119
Hamilton, C. L., and Ciaccia, P. J. (1971). Hypothalamus, temperature regulation, and feeding in the rat. Am. J. Physiol. 221, 800–807. doi: 10.1152/ajplegacy.1971.221.3.800
Han, W., Tellez, L. A., Rangel, M. J. Jr., Motta, S. C., Zhang, X., Perez, I. O., et al. (2017). Integrated control of predatory hunting by the central nucleus of the amygdala. Cell 168:e18. doi: 10.1016/j.cell.2016.12.027
Hare, B. D., Shinohara, R., Liu, R. J., Pothula, S., DiLeone, R. J., and Duman, R. S. (2019). Optogenetic stimulation of medial prefrontal cortex Drd1 neurons produces rapid and long-lasting antidepressant effects. Nat. Commun. 10:223. doi: 10.1038/s41467-018-08168-9
Harriott, A. M., Chung, D. Y., Uner, A., Bozdayi, R. O., Morais, A., Takizawa, T., et al. (2021). Optogenetic spreading depression elicits trigeminal pain and anxiety behavior. Ann. Neurol. 89, 99–110. doi: 10.1002/ana.25926
Harris, A. R., and Gilbert, F. (2022). Restoring vision using optogenetics without being blind to the risks. Graefes Arch. Clin. Exp. Ophthalmol. 260, 41–45. doi: 10.1007/s00417-021-05477-6
He, L., Tan, P., Huang, Y., and Zhou, Y. (2021). Design of smart antibody mimetics with photosensitive switches. Adv. Biol. 5:e2000541. doi: 10.1002/adbi.202000541
Hellemans, K. G., Everitt, B. J., and Lee, J. L. (2006). Disrupting reconsolidation of conditioned withdrawal memories in the basolateral amygdala reduces suppression of heroin seeking in rats. J. Neurosci. 26, 12694–12699. doi: 10.1523/JNEUROSCI.3101-06.2006
Hemmati, S., Behzadipour, Y., and Haddad, M. (2020). Decoding the proteome of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) for cell-penetrating peptides involved in pathogenesis or applicable as drug delivery vectors. Infect Genet. Evol. 85:104474. doi: 10.1016/j.meegid.2020.104474
Hori, Y., and Kikuchi, K. (2019). Chemical tools with fluorescence switches for verifying epigenetic modifications. Acc. Chem. Res. 52, 2849–2857. doi: 10.1021/acs.accounts.9b00349
Hosseinpanah, F., Serahati, S., Barzin, M., Aryannezhad, S., Rezaie, M., Valizadeh, M., et al. (2019). Trends of obesity in 10-years of follow-up among tehranian children and adolescents: tehran lipid and glucose study (TLGS). Iran J. Public Health 48, 1714–1722.
Hsiao, P. Y., Wu, M. C., Lin, Y. Y., Fu, C. C., and Chiang, A. S. (1408). Optogenetic manipulation of selective neural activity in free-moving drosophila adults. Methods Mol. Biol. 37:2016. doi: 10.1007/978-1-4939-3512-3_26
Hua, T., Chen, B., Lu, D., Sakurai, K., Zhao, S., Han, B. X., et al. (2020). General anesthetics activate a potent central pain-suppression circuit in the amygdala. Nat. Neurosci. 23, 854–868. doi: 10.1038/s41593-020-0632-8
Huang, X., Huang, P., Huang, L., Hu, Z., Liu, X., Shen, J., et al. (2021). A visual circuit related to the nucleus reuniens for the spatial-memory-promoting effects of light treatment. Neuron 109, 347–362e7. doi: 10.1016/j.neuron.2020.10.023
Ingram, T. L., Shephard, F., Sarmad, S., Ortori, C. A., Barrett, D. A., and Chakrabarti, L. (2020). Sex specific inflammatory profiles of cerebellar mitochondria are attenuated in Parkinson’s disease. Aging 12, 17713–17737. doi: 10.18632/aging.103937
Ishikawa, T., and Sakaguchi, Y. (2013). Both movement-end and task-end are critical for error feedback in visuomotor adaptation: a behavioral experiment. PLoS One 8:e55801. doi: 10.1371/journal.pone.0055801
Jacob, J., and Szerb, J. (1951). [Effect of various alarm stimuli on reaction to pain in mice]. Arch. Int. Pharmacodyn Ther. 87, 251–253.
Jamaluddin, N. D., Mazlan, N. F., Tan, L. L., Yusof, N. Y. M., and Khalid, B. (2021). G-quadruplex microspheres-based optical RNA biosensor for arthropod-borne virus pathogen detection: a proof-of-concept with dengue serotype 2. Int. J. Biol. Macromol. 199, 1–9. doi: 10.1016/j.ijbiomac.2021.12.047
Jennings, J. H., Ung, R. L., Resendez, S. L., Stamatakis, A. M., Taylor, J. G., Huang, J., et al. (2015). Visualizing hypothalamic network dynamics for appetitive and consummatory behaviors. Cell 160, 516–527. doi: 10.1016/j.cell.2014.12.026
Ji, J., He, Q., Luo, X., Bang, S., Matsuoka, Y., McGinnis, A., et al. (2021). IL-23 enhances C-fiber-mediated and blue light-induced spontaneous pain in female mice. Front. Immunol. 12:787565. doi: 10.3389/fimmu.2021.787565
Josselyn, S. A., Kohler, S., and Frankland, P. W. (2015). Finding the engram. Nat. Rev. Neurosci. 16, 521–534. doi: 10.1038/nrn4000
Karim, F., Hu, H. J., Adwanikar, H., and Kaplan, D. (2006). Gereau RWt: impaired inflammatory pain and thermal hyperalgesia in mice expressing neuron-specific dominant negative mitogen activated protein kinase kinase (MEK). Mol. Pain 2:2. doi: 10.1186/1744-8069-2-2
Kessler, T. M., Birder, L. A., and Gomery, P. (2019). Neuromodulation of urinary tract function. New Engl. J. Med. 380, 2067–2069.
Khakpoor, M., Nasehi, M., Vahdati, A., Hoseyni, S. E., and Zarrindast, M. R. (2016). Additive effect of BLA GABAA receptor mechanism and (+)-MK-801 on memory retention deficit, an isobologram analysis. Pharmacol. Biochem. Behav. 143, 57–64. doi: 10.1016/j.pbb.2016.02.001
Kim, K. D., Bae, S., Capece, T., Nedelkovska, H., de Rubio, R. G., Smrcka, A. V., et al. (2017). Targeted calcium influx boosts cytotoxic T lymphocyte function in the tumour microenvironment. Nat. Commun. 8:15365. doi: 10.1038/ncomms15365
Kim, W. S., Hong, S., Gamero, M., Jeevakumar, V., Smithhart, C. M., Price, T. J., et al. (2021). Organ-specific, multimodal, wireless optoelectronics for high-throughput phenotyping of peripheral neural pathways. Nat. Commun. 12:157. doi: 10.1038/s41467-020-20421-8
Kinouchi, K., Mikami, Y., Kanai, T., and Itoh, H. (2021). Circadian rhythms in the tissue-specificity from metabolism to immunity: insights from omics studies. Mol. Aspects Med. 80:100984. doi: 10.1016/j.mam.2021.100984
Kirmayer, L. J., Young, A., and Hayton, B. C. (1995). The cultural context of anxiety disorders. Psychiatr. Clin. North Am. 18, 503–521. doi: 10.1016/s0193-953x(18)30037-6
Klavir, O., Prigge, M., Sarel, A., Paz, R., and Yizhar, O. (2017). Manipulating fear associations via optogenetic modulation of amygdala inputs to prefrontal cortex. Nat. Neurosci. 20, 836–844. doi: 10.1038/nn.4523
Kramer, M. M., Muhlhauser, W. W. D., Weber, W., and Radziwill, G. (2021). Multichromatic control of signaling pathways in mammalian cells. Adv. Biol. 5:e2000196. doi: 10.1002/adbi.202000196
Kravitz, A. V., Freeze, B. S., Parker, P. R., Kay, K., Thwin, M. T., Deisseroth, K., et al. (2010). Regulation of parkinsonian motor behaviours by optogenetic control of basal ganglia circuitry. Nature 466, 622–626. doi: 10.1038/nature09159
Lacagnina, A. F., Brockway, E. T., Crovetti, C. R., Shue, F., McCarty, M. J., Sattler, K. P., et al. (2019). Distinct hippocampal engrams control extinction and relapse of fear memory. Nat. Neurosci. 22, 753–761. doi: 10.1038/s41593-019-0361-z
Lee, H., Kim, D. W., Remedios, R., Anthony, T. E., Chang, A., Madisen, L., et al. (2014). Scalable control of mounting and attack by Esr1+ neurons in the ventromedial hypothalamus. Nature 509, 627–632. doi: 10.1038/nature13169
Lee, S., Augustine, V., Zhao, Y., Ebisu, H., Ho, B., Kong, D., et al. (2019). Chemosensory modulation of neural circuits for sodium appetite. Nature 568, 93–97. doi: 10.1038/s41586-019-1053-2
Li, T., Chen, X., Qian, Y., Shao, J., Li, X., Liu, S., et al. (2021). A synthetic BRET-based optogenetic device for pulsatile transgene expression enabling glucose homeostasis in mice. Nat. Commun. 12:615. doi: 10.1038/s41467-021-20913-1
Liu, D. Y., and Zhang, L. (2019). MicroRNA-132 promotes neurons cell apoptosis and activates tau phosphorylation by targeting GTDC-1 in Alzheimer’s disease. Eur. Rev. Med. Pharmacol. Sci. 23, 8523–8532. doi: 10.26355/eurrev_201910_19166
Liu, H., Cheng, Y., Chu, J., Wu, M., Yan, M., Wang, D., et al. (2021). Baicalin attenuates angiotensin II-induced blood pressure elevation and modulates MLCK/p-MLC signaling pathway. Biomed. Pharmacother 143:112124. doi: 10.1016/j.biopha.2021.112124
Liu, Y., Li, N., Zhu, X., and Qi, Y. (2021). How wide is the application of genetic big data in biomedicine. Biomed. Pharmacother. 133:111074. doi: 10.1016/j.biopha.2020.111074
Llewellyn, M. E., Thompson, K. R., Deisseroth, K., and Delp, S. L. (2010). Orderly recruitment of motor units under optical control in vivo. Nat. Med. 16, 1161–1165. doi: 10.1038/nm.2228
Lu, D., Huang, Y., Kong, Y., Tao, T., and Zhu, X. (2020). Gut microecology: why our microbes could be key to our health. Biomed Pharmacother 131:110784. doi: 10.1016/j.biopha.2020.110784
Lu, W., Yao, J., Zhu, X., and Qi, Y. (2020). Nanomedicines: redefining traditional medicine. Biomed Pharmacother 134:111103. doi: 10.1016/j.biopha.2020.111103
Lu, Y., Zhong, C., Wang, L., Wei, P., He, W., Huang, K., et al. (2016). Optogenetic dissection of ictal propagation in the hippocampal-entorhinal cortex structures. Nat. Commun. 7:10962.
Luo, P. W., Han, H. W., Yang, C. S., Shrestha, L. K., Ariga, K., and Hsu, S. H. (2019). Optogenetic modulation and reprogramming of bacteriorhodopsin-transfected human fibroblasts on self-assembled fullerene C60 nanosheets. Adv. Biosyst. 3:e1800254. doi: 10.1002/adbi.201800254
Mai, L., Zhu, X., Huang, F., He, H., and Fan, W. (2020). p38 mitogen-activated protein kinase and pain. Life Sci. 256:117885. doi: 10.1016/j.lfs.2020.117885
Malaquias, A. D. M., Marques, L. E. C., Pereira, S. S., de Freitas Fernandes, C., Maranhao, A. Q., Stabeli, R. G., et al. (2021). A review of plant-based expression systems as a platform for single-domain recombinant antibody production. Int. J. Biol. Macromol. 193, 1130–1137. doi: 10.1016/j.ijbiomac.2021.10.126
Martinez-Gonzalez, J. M., Vilar Lopez, R., Becona Iglesias, E., and Verdejo-Garcia, A. (2016). Self-deception as a mechanism for the maintenance of drug addiction. Psicothema 28, 13–19. doi: 10.7334/psicothema2015.139
Martinez-Juarez, I. E., Lopez-Zapata, R., Gomez-Arias, B., Bravo-Armenta, E., Romero-Ocampo, L., Estevez-Cruz, Z., et al. (2012). [Refractory epilepsy: use of the new definition and related risk factors. a study in the mexican population of a third-level centre]. Rev. Neurol. 54, 159–166.
Meda, S. A., Koran, M. E., Pryweller, J. R., Vega, J. N., and Thornton-Wells, T. A. (2013). Alzheimer’s disease neuroimaging i: genetic interactions associated with 12-month atrophy in hippocampus and entorhinal cortex in Alzheimer’s disease neuroimaging initiative. Neurobiol. Aging 34:1518.e9. doi: 10.1016/j.neurobiolaging.2012.09.020
Melero-Fernandez de Mera, R. M., Li, L. L., Popinigis, A., Cisek, K., Tuittila, M., Yadav, L., et al. (2017). A simple optogenetic MAPK inhibitor design reveals resonance between transcription-regulating circuitry and temporally-encoded inputs. Nat. Commun. 8:15017. doi: 10.1038/ncomms15017
Michoud, F., Seehus, C., Schonle, P., Brun, N., Taub, D., Zhang, Z., et al. (2021). Epineural optogenetic activation of nociceptors initiates and amplifies inflammation. Nat. Biotechnol. 39, 179–185. doi: 10.1038/s41587-020-0673-2
Mickle, A. D., Won, S. M., Noh, K. N., Yoon, J., Meacham, K. W., Xue, Y., et al. (2019). A wireless closed-loop system for optogenetic peripheral neuromodulation. Nature 565, 361–365. doi: 10.1038/s41586-018-0823-6
Mickoleit, F., Rosenfeldt, S., Toro-Nahuelpan, M., Schaffer, M., Schenk, A. S., Plitzko, J. M., et al. (2021). High-yield production, characterization, and functionalization of recombinant magnetosomes in the synthetic bacterium rhodospirillum rubrum “magneticum”. Adv. Biol. 5:e2101017. doi: 10.1002/adbi.202101017
Molnar, K., and Labouesse, M. (2021). The plastic cell: mechanical deformation of cells and tissues. Open Biol. 11:210006. doi: 10.1098/rsob.210006
Moreira, J. M., Itskov, P. M., Goldschmidt, D., Baltazar, C., Steck, K., Tastekin, I., et al. (2019). optoPAD, a closed-loop optogenetics system to study the circuit basis of feeding behaviors. Elife 8:e43924.
Murawiec, S., Chudek, J., Nieves, W., Almgren-Rachtan, A., and Jedrzejczak, J. (2020). Increasing the dosage of pregabalin in patients with focal epilepsy decreases the frequency of seizures and ameliorates symptoms of anxiety, depression and insomnia. Eur. Rev. Med. Pharmacol. Sci. 24, 13015–13024. doi: 10.26355/eurrev_202012_24207
Nabavi, S., Fox, R., Proulx, C. D., Lin, J. Y., Tsien, R. Y., and Malinow, R. (2014). Engineering a memory with LTD and LTP. Nature 511, 348–352. doi: 10.1038/nature13294
News, S. (2010). Insights of the decade. stepping away from the trees for a look at the forest. introduction. Science 330, 1612–1613. doi: 10.1126/science.330.6011.1612
Ng, Y. S., Bindoff, L. A., Gorman, G. S., Horvath, R., Klopstock, T., Mancuso, M., et al. (2019). Consensus-based statements for the management of mitochondrial stroke-like episodes. Wellcome Open Res. 4:201. doi: 10.12688/wellcomeopenres.15599.1
Nieh, E. H., Matthews, G. A., Allsop, S. A., Presbrey, K. N., Leppla, C. A., Wichmann, R., et al. (2015). Decoding neural circuits that control compulsive sucrose seeking. Cell 160, 528–541. doi: 10.1016/j.cell.2015.01.003
Nowak, V. A., Pereira, E. A., Green, A. L., and Aziz, T. Z. (2010). Optogenetics–shining light on neurosurgical conditions. Br. J. Neurosurg. 24, 618–624. doi: 10.3109/02688697.2010.520764
Oh, T. J., Fan, H., Skeeters, S. S., and Zhang, K. (2021). Steering molecular activity with optogenetics: recent advances and perspectives. Adv. Biol. 5:e2000180. doi: 10.1002/adbi.202000180
Ostolaza, A., Blanco-Luquin, I., Urdanoz-Casado, A., Rubio, I., Labarga, A., Zandio, B., et al. (2020). Circular RNA expression profile in blood according to ischemic stroke etiology. Cell Biosci. 10:34. doi: 10.1186/s13578-020-00394-3
Ostrovsky, M. A., and Kirpichnikov, M. P. (2019). Prospects of optogenetic prosthesis of the degenerative retina of the eye. Biochemistry 84, 479–490. doi: 10.1134/S0006297919050031
Padmanabhan, S., Perez-Castano, R., and Elias-Arnanz, M. (2019). B12-based photoreceptors: from structure and function to applications in optogenetics and synthetic biology. Curr. Opin Struct. Biol. 57, 47–55. doi: 10.1016/j.sbi.2019.01.020
Peters, A. E., Caban, S. J., McLaughlin, E. A., Roman, S. D., Bromfield, E. G., Nixon, B., et al. (2021). The Impact of Aging on Macroautophagy in the Pre-ovulatory Mouse Oocyte. Front Cell Dev Biol 9:691826. doi: 10.3389/fcell.2021.691826
Pozarickij, A., Williams, C., and Guggenheim, J. A. (2020). and the UKBE, vision C: non-additive (dominance) effects of genetic variants associated with refractive error and myopia. Mol. Genet. Genom. 295, 843–853. doi: 10.1007/s00438-020-01666-w
Rajalingham, R., Sorenson, M., Azadi, R., Bohn, S., DiCarlo, J. J., and Afraz, A. (2021). Chronically implantable LED arrays for behavioral optogenetics in primates. Nat. Methods 18, 1112–1116. doi: 10.1038/s41592-021-01238-9
Riksen, N. P., and Netea, M. G. (2020). Immunometabolic control of trained immunity. Mol. Aspects Med. 77:100897. doi: 10.1016/j.mam.2020.100897
Roet, M., Hescham, S. A., Jahanshahi, A., Rutten, B. P. F., Anikeeva, P. O., and Temel, Y. (2019). Progress in neuromodulation of the brain: a role for magnetic nanoparticles? Prog. Neurobiol. 177, 1–14. doi: 10.1016/j.pneurobio.2019.03.002
Roy, D. S., Arons, A., Mitchell, T. I., Pignatelli, M., Ryan, T. J., and Tonegawa, S. (2016). Memory retrieval by activating engram cells in mouse models of early Alzheimer’s disease. Nature 531, 508–512. doi: 10.1038/nature17172
Roy-O’Reilly, M. A., Ahnstedt, H., Spychala, M. S., Munshi, Y., Aronowski, J., Sansing, L. H., et al. (2020). Aging exacerbates neutrophil pathogenicity in ischemic stroke. Aging 12, 436–461. doi: 10.18632/aging.102632
Saltmarche, A. E., Naeser, M. A., Ho, K. F., Hamblin, M. R., and Lim, L. (2017). Significant improvement in cognition in mild to moderately severe dementia cases treated with transcranial plus intranasal photobiomodulation: case series report. Photomed. Laser Surg. 35, 432–441. doi: 10.1089/pho.2016.4227
Samineni, V. K., Mickle, A. D., Yoon, J., Grajales-Reyes, J. G., Pullen, M. Y., Crawford, K. E., et al. (2017). Optogenetic silencing of nociceptive primary afferents reduces evoked and ongoing bladder pain. Sci. Rep. 7:15865. doi: 10.1038/s41598-017-16129-3
Santos, D. S., Morais, J. A. V., Vanderlei, I. A. C., Santos, A. S., Azevedo, R. B., Muehlmann, L. A., et al. (2021). Oral delivery of fish oil in oil-in-water nanoemulsion: development, colloidal stability and modulatory effect on in vivo inflammatory induction in mice. Biomed Pharmacother 133:110980. doi: 10.1016/j.biopha.2020.110980
Senok, A., Monecke, S., Nassar, R., Celiloglu, H., Thyagarajan, S., Muller, E., et al. (2021). Lateral flow immunoassay for the detection of panton-valentine leukocidin in staphylococcus aureus from skin and soft tissue infections in the united arab emirates. Front. Cell Infect Microbiol. 11:754523. doi: 10.3389/fcimb.2021.754523
Shao, J., Wang, M., Yu, G., Zhu, S., Yu, Y., Heng, B. C., et al. (2018). Synthetic far-red light-mediated CRISPR-dCas9 device for inducing functional neuronal differentiation. Proc. Natl. Acad. Sci. U.S.A. 115, E6722–E6730. doi: 10.1073/pnas.1802448115
Shu, S., Zhu, H., Tang, N., Chen, W., Li, X., Li, H., et al. (2016). Selective degeneration of entorhinal-CA1 synapses in Alzheimer’s disease via activation of DAPK1. J. Neurosci. 36, 10843–10852. doi: 10.1523/JNEUROSCI.2258-16.2016
Song, C., Kong, Y., Huang, L., Luo, H., and Zhu, X. (2020). Big data-driven precision medicine: starting the custom-made era of iatrology. Biomed. Pharmacother 129:110445. doi: 10.1016/j.biopha.2020.110445
Spierings, M. J., and ten Cate, C. (2014). Zebra finches are sensitive to prosodic features of human speech. Proc. Biol. Sci. 281:20140480. doi: 10.1098/rspb.2014.0480
Spreafico, R., Hayes, N. L., and Rustioni, A. (1981). Thalamic projections to the primary and secondary somatosensory cortices in cat: single and double retrograde tracer studies. J. Comput. Neurol. 203, 67–90. doi: 10.1002/cne.902030107
Srinivasan, S. S., Maimon, B. E., Diaz, M., Song, H., and Herr, H. M. (2018). Closed-loop functional optogenetic stimulation. Nat. Commun. 9:5303. doi: 10.1038/s41467-018-07721-w
Steinbeck, J. A., Choi, S. J., Mrejeru, A., Ganat, Y., Deisseroth, K., Sulzer, D., et al. (2015). Optogenetics enables functional analysis of human embryonic stem cell-derived grafts in a Parkinson’s disease model. Nat. Biotechnol. 33, 204–209. doi: 10.1038/nbt.3124
Sternson, S. M., and Atasoy, D. (2014). Agouti-related protein neuron circuits that regulate appetite. Neuroendocrinology 100, 95–102. doi: 10.1159/000369072
Thornton, S. M., and Fitzsimons, J. T. (1995). The effects of centrally administered porcine relaxin on drinking behaviour in male and female rats. J. Neuroendocrinol. 7, 165–169. doi: 10.1111/j.1365-2826.1995.tb00743.x
Tremblay, S., Acker, L., Afraz, A., Albaugh, D. L., Amita, H., Andrei, A. R., et al. (2020). An open resource for non-human primate optogenetics. Neuron 108:e6. doi: 10.1016/j.neuron.2020.09.027
Tretyn, A. (1999). Calcium-dependent signal transduction pathways in plants–phytochrome mechanism of action as an example. Pol. J. Pharmacol. 51, 145–151.
Tsai, Z. R., Zhang, H. W., Tseng, C. H., Peng, H. C., Kok, V. C., Li, G. P., et al. (2021). Late-onset epilepsy and subsequent increased risk of dementia. Aging 13, 3573–3587. doi: 10.18632/aging.202299
Vahaba, D. M., Macedo-Lima, M., and Remage-Healey, L. (2017). Sensory coding and sensitivity to local estrogens shift during critical period milestones in the auditory cortex of male songbirds. eNeuro 4:ENEURO.0317-17.2017. doi: 10.1523/ENEURO.0317-17.2017
Vajtay, T. J., Bandi, A., Upadhyay, A., Swerdel, M. R., Hart, R. P., Lee, C. R., et al. (2019). Optogenetic and transcriptomic interrogation of enhanced muscle function in the paralyzed mouse whisker pad. J. Neurophysiol. 121, 1491–1500. doi: 10.1152/jn.00837.2018
Vallee, A., Vallee, J. N., Guillevin, R., and Lecarpentier, Y. (2020). Riluzole: a therapeutic strategy in Alzheimer’s disease by targeting the WNT/beta-catenin pathway. Aging 12, 3095–3113. doi: 10.18632/aging.102830
Vassalli, Q. A., Colantuono, C., Nittoli, V., Ferraioli, A., Fasano, G., Berruto, F., et al. (2021). Onecut regulates core components of the molecular machinery for neurotransmission in photoreceptor differentiation. Front. Cell Dev. Biol. 9:602450. doi: 10.3389/fcell.2021.602450
Vetere, G., Tran, L. M., Moberg, S., Steadman, P. E., Restivo, L., Morrison, F. G., et al. (2019). Memory formation in the absence of experience. Nat. Neurosci. 22, 933–940. doi: 10.1038/s41593-019-0389-0
Wang, J. J., Yao, W. Q., Chen, Y. J., Ma, L., and Tao, Y. Z. (2014). [Neurons in NAc core and BLA are activated during cocaine context-associated reward memory retrieval in mice]. Sheng Li Xue Bao 66, 545–558.
Wolf, G., Schulkin, J., and Simson, P. E. (1984). Multiple factors in the satiation of salt appetite. Behav. Neurosci. 98, 661–673. doi: 10.1037//0735-7044.98.4.661
Wu, M. C., Chu, L. A., Hsiao, P. Y., Lin, Y. Y., Chi, C. C., Liu, T. H., et al. (2014). Optogenetic control of selective neural activity in multiple freely moving drosophila adults. Proc. Natl. Acad. Sci. U.S.A. 111, 5367–5372. doi: 10.1073/pnas.1400997111
Xu, Q., Zhang, J., Qin, T., Bao, J., Dong, H., Zhou, X., et al. (2021). The role of the inflammasomes in the pathogenesis of uveitis. Exp. Eye Res. 208:108618. doi: 10.1016/j.exer.2021.108618
Yang, X., Sun, X., Liu, J., Huang, Y., Peng, Y., Xu, Y., et al. (2021). Photo-crosslinked GelMA/collagen membrane loaded with lysozyme as an antibacterial corneal implant. Int. J. Biol. Macromol. 191, 1006–1016. doi: 10.1016/j.ijbiomac.2021.09.144
Yang, X., Yao, C., Tian, T., Li, X., Yan, H., Wu, J., et al. (2018). A novel mechanism of memory loss in Alzheimer’s disease mice via the degeneration of entorhinal-CA1 synapses. Mol. Psychiatry 23, 199–210. doi: 10.1038/mp.2016.151
Yang, Y., Wu, M., Vazquez-Guardado, A., Wegener, A. J., Grajales-Reyes, J. G., Deng, Y., et al. (2021). Wireless multilateral devices for optogenetic studies of individual and social behaviors. Nat. Neurosci. 24, 1035–1045. doi: 10.1038/s41593-021-00849-x
Yao, J. P., Hou, W. S., and Yin, Z. Q. (2012). Optogenetics: a novel optical manipulation tool for medical investigation. Int. J. Ophthalmol. 5, 517–522. doi: 10.3980/j.issn.2222-3959.2012.04.22
Yi, M., Ma, Y., Zhu, S., Luo, C., Chen, Y., Wang, Q., et al. (2020). Comparative proteomic analysis identifies biomarkers for renal aging. Aging 12, 21890–21903. doi: 10.18632/aging.104007
Yi, Y., Li, Y., Meng, Q., Li, Q., Li, F., Lu, B., et al. (2021). A PRC2-independent function for EZH2 in regulating rRNA 2’-O methylation and IRES-dependent translation. Nat. Cell Biol. 23, 341–354. doi: 10.1038/s41556-021-00653-6
Yilmaz, I., Karaarslan, N., Yasar Sirin, D., and Ozbek, H. (2020). Pharmaco-molecular assessment of the effects of anandamide and its antagonists on hippocampal tissue in Wistar albino rats. Eur. Rev. Med. Pharmacol. Sci. 24, 11871–11882. doi: 10.26355/eurrev_202011_23845
Yu, C., Cassar, I. R., Sambangi, J., and Grill, W. M. (2020). Frequency-specific optogenetic deep brain stimulation of subthalamic nucleus improves parkinsonian motor behaviors. J. Neurosci. 40, 4323–4334. doi: 10.1523/JNEUROSCI.3071-19.2020
Yu, L., Wang, J., Zou, Y., Zeng, H., Cheng, W., and Jing, X. (2021). Qingfei oral liquid inhibited autophagy to alleviate inflammation via mTOR signaling pathway in RSV-infected asthmatic mice. Biomed. Pharmacother 138:111449. doi: 10.1016/j.biopha.2021.111449
Yu, S. P., Tung, J. K., Wei, Z. Z., Chen, D., Berglund, K., Zhong, W., et al. (2019). Optochemogenetic stimulation of transplanted iPS-NPCs enhances neuronal repair and functional recovery after ischemic stroke. J. Neurosci. 39, 6571–6594. doi: 10.1523/JNEUROSCI.2010-18.2019
Zhang, D., Chen, H. D., Zulfiqar, H., Yuan, S. S., Huang, Q. L., Zhang, Z. Y., et al. (2021). iBLP: an xgboost-based predictor for identifying bioluminescent proteins. Comput. Math Methods Med. 2021:6664362. doi: 10.1155/2021/6664362
Zhang, M., Eshraghian, E. A., Jammal, O. A., Zhang, Z., and Zhu, X. (2020). CRISPR technology: the engine that drives cancer therapy. Biomed Pharmacother 133:111007. doi: 10.1016/j.biopha.2020.111007
Zhang, Z., Liu, Q., Wen, P., Zhang, J., Rao, X., Zhou, Z., et al. (2017). Activation of the dopaminergic pathway from VTA to the medial olfactory tubercle generates odor-preference and reward. Elife 6:e25423. doi: 10.7554/eLife.25423
Zhao, Z. D., Yang, W. Z., Gao, C., Fu, X., Zhang, W., Zhou, Q., et al. (2017). A hypothalamic circuit that controls body temperature. Proc. Natl. Acad. Sci. U.S.A. 114, 2042–2047. doi: 10.1073/pnas.1616255114
Zhou, M., Zhu, J. Q., and Kang, L. Y. (2013). [Progress on chemical components and anti-cerebral injury effects of storax]. Zhongguo Zhong Yao Za Zhi 38, 3825–3828.
Zhu, R., Guo, W., Xu, X. J., and Zhu, L. (2020). An integrating immune-related signature to improve prognosis of hepatocellular carcinoma. Comput. Math Methods Med. 2020:8872329. doi: 10.1155/2020/8872329
Keywords: nanoparticles, nervous system, neural circuits, neurobiology, neuron, optogenetics
Citation: Chen W, Li C, Liang W, Li Y, Zou Z, Xie Y, Liao Y, Yu L, Lin Q, Huang M, Li Z and Zhu X (2022) The Roles of Optogenetics and Technology in Neurobiology: A Review. Front. Aging Neurosci. 14:867863. doi: 10.3389/fnagi.2022.867863
Received: 01 February 2022; Accepted: 21 March 2022;
Published: 19 April 2022.
Edited by:
Min Tang, Jiangsu University, ChinaReviewed by:
Ling Li, University of North Dakota, United StatesKaihua Guo, Sun Yat-sen University, China
Copyright © 2022 Chen, Li, Liang, Li, Zou, Xie, Liao, Yu, Lin, Huang, Li and Zhu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zesong Li, bHpzc2NAZW1haWwuc3p1LmVkdS5jbg==; Xiao Zhu, YmlveHpodUB5YWhvby5jb20=
†These authors have contributed equally to this work
 Wenqing Chen1,2†
Wenqing Chen1,2† Chen Li
Chen Li Zhuoheng Zou
Zhuoheng Zou Lin Yu
Lin Yu Qianyi Lin
Qianyi Lin Zesong Li
Zesong Li Xiao Zhu
Xiao Zhu