- 1Department of Clinical Sciences, University of Pennsylvania School of Veterinary Medicine, Philadelphia, PA, United States
- 2Department of Biostatistics, Epidemiology and Informatics, University of Pennsylvania Perelman School of Medicine, Philadelphia, PA, United States
Background: Robust measurement and tracking of antimicrobial use (AMU) is a fundamental component of stewardship interventions. Feeding back AMU metrics to individual clinicians is a common approach to changing prescribing behavior. Metrics must be meaningful and comprehensible to clinicians. Little is known about how veterinary clinicians working in the United States (US) hospital setting think about AMU metrics for antimicrobial stewardship.
Objective: To identify hospital-based veterinary clinicians' attitudes toward audit and feedback of AMU metrics, their perceptions of different AMU metrics, and their response to receiving an individualized prescribing report.
Methods: Semi-structured interviews were conducted with veterinarians working at two hospitals in the Eastern US. Interviews elicited perceptions of antimicrobial stewardship in veterinary medicine. Respondents were shown a personalized AMU Report characterizing their prescribing patterns relative to their peers and were asked to respond. Interviews were recorded, transcribed, and analyzed using the framework method with matrices.
Results: Semi-structured interviews were conducted with 34 veterinary clinicians (22 small animal and 12 large animal). Respondents generally felt positive about the reports and were interested in seeing how their prescribing compared to that of their peers. Many respondents expressed doubt that the reports accurately captured the complexities of their prescribing decisions and found metrics associated with animal daily doses (ADDs) confusing. Only 13 (38.2%) respondents felt the reports would change how they used antimicrobials. When asked how the impact of the reports could be optimized, respondents recommended providing a more detailed explanation of how the AMU metrics were derived, education prior to report roll-out, guidance on how to interpret the metrics, and development of meaningful benchmarks for goal-setting.
Conclusions: These findings provide important insight that can be used to design veterinary-specific AMU metrics as part of a stewardship intervention that are meaningful to clinicians and more likely to promote judicious prescribing.
Introduction
Antimicrobial stewardship has been defined as “coordinated interventions designed to improve and measure the appropriate use of [antimicrobial] agents by promoting the selection of the optimal [antimicrobial] drug regimen including dosing, duration of therapy, and route of administration” (1). In people, antimicrobial stewardship programs (ASPs) have been shown to improve patient outcomes, shorten the length of stay, reduce antimicrobial resistance, and save money in the inpatient setting (2).
A frequently used stewardship initiative is the provision of periodic feedback on a prescriber's AMU, oftentimes situating their use relative to their peers'. In human medicine, this type of intervention has been shown to decrease AMU, improve clinical outcomes, and decrease costs in the outpatient clinical setting (3–6). In animal agriculture, AMU is tracked at the farm level in many (mostly European) countries, and individual AMU data are regularly provided to producers and veterinarians for purposes of benchmarking in the context of regulatory programs or quality assurance schemes. These types of initiatives are thought to be major contributors to the decline in AMU observed in many livestock sectors in these countries (7–9).
In veterinary hospitals, AMU data are rarely tracked and much less frequently fed back to clinicians. Veterinary hospitals represent fundamentally different prescribing ecosystems than farms, and very little is known about the attitudes of veterinary clinicians working in these settings toward antimicrobial stewardship initiatives involving tracking and reporting of antimicrobial use, especially in the United States (US). With increasing interest in antimicrobial stewardship within US veterinary hospitals (10, 11), more information is needed on best practices for implementing systems that involve the feedback of AMU prescribing data to veterinarians. In particular, there is no consensus on which metric(s) to use. In farm-level benchmarking schemes, a variety of metrics are used, including count-based, mass-based, daily dose-based, and course-based indicators (12). Each metric has advantages and disadvantages and may or may not be applicable to the hospital setting, where individual animals rather than herds are treated.
Because the goal of providing AMU prescribing data to clinicians is to effect behavior change related to antimicrobial prescribing, the best metrics to use will ultimately be those that are understood, accepted by individual clinicians, and make sense for the context in which they work (13). Feedback of AMU data to human medicine clinicians has mostly been successful in outpatient primary care settings (3–6). It has been less successful in improving the behavior of hospital-based clinicians (14). This may partially be explained by the fact that the social context of work in hospitals is different than in outpatient offices, where multiple clinicians may care for the same patient and responsibility for a prescription is not clearly linked to an individual clinician (15, 16). Sociobehavioral research on the way human medicine clinicians respond to AMU data targeted at them individually demonstrates that considerable skepticism and lack of trust can surround feedback reports, contributing to gaming and workarounds (17). Before implementing an ASP intervention, clinician confidence in the measurement system must be secured to boost clinicians' acceptance of feedback data and increase motivation to change (18, 19). The goal of this study was therefore to identify the perceptions that veterinary clinicians working in the US hospital setting hold toward different AMU metrics used for the systematic tracking and reporting of AMU for purposes of stewardship.
Methods
Study Design, Setting, and Participants
We conducted in-depth, semi-structured interviews with a purposive sample of veterinary clinicians from two hospitals (one large animal and one small animal) within a health system in the Eastern US. The small animal hospital sees ~35,000 patients per year and can house 250 inpatients at any time. The large animal hospital sees 4,900 patients per year and can house 200 inpatients at any time. The health system in which we gathered data did not have a formal stewardship program in place that utilized audit and feedback of AMU metrics at the time of data collection. However, both hospitals had implemented individual antimicrobial stewardship initiatives, had held educational sessions on AMU in veterinary medicine, and emphasized that improving the use of antimicrobials was an institutional priority.
This qualitative study was led by a medical sociologist (JES) with expertise in mixed-methods research on antimicrobial prescribing and stewardship interventions, in collaboration with a veterinary epidemiologist (LER) with expertise in developing novel stewardship measures and metrics in veterinary medicine. At each hospital, we sought to interview veterinary clinicians from different specialties who commonly prescribed antimicrobials including internal medicine, surgery, dermatology, and emergency medicine. To identify respondents, we worked with key contacts at each hospital to identify the names and email address of eligible clinicians. The study team recruited respondents by email. Respondents were offered a $50 Visa gift card as an incentive. Potential respondents were assured that their specific comments would not be shared with key contacts beyond a report of de-identified aggregated themes. Our protocol was approved by the University of Pennsylvania Institutional Review Board (IRB Protocol # 832630).
Data Collection
Data were gathered from April to July 2019. Interviews were conducted in person by the medical sociologist and a senior research associate (BMM) with graduate training in anthropology and advanced interview technique. A semi-structured interview guide was created based on a review of the literature and the authors' previous research (see Supplementary Material for the guide). Questions were designed to be open-ended in order to elicit in-depth responses from veterinarians with minimal prompting by the interviewer (20). Key thematic domains in the interview guide included respondent perceptions of antimicrobial resistance and overuse in veterinary medicine, the application of principles of antimicrobial stewardship to the veterinary hospital context, and perceptions of a personalized Antimicrobial Use Report (described in more detail below). All interviews were, with permission, recorded. Respondents were made aware that the purpose of the study was to better understand their opinions and perceptions of antimicrobial stewardship interventions and AMU metrics in veterinary medicine in order to inform the development of future health system interventions. Interviewers kept ongoing data collection memos to monitor for identification of novel insights and saturation of key themes in order to determine sample size adequacy (21).
Personalized Antimicrobial Use Report
The last part of the interview involved presenting the respondent with a hard copy of a personalized Antimicrobial Use Report (see Supplementary Material for sample report). These reports were generated from our veterinary hospital administrative and electronic medical record databases as previously described (22). Briefly, individuals' antimicrobial prescribing patterns from 2013 to 2018 were characterized relative to their peers using metrics that are frequently used to characterize AMU, including count- and dose-based metrics involving the animal-defined daily dose [ADD—also known as the DDDvet (12)], a metric that represents the average maintenance dose of a drug for its main indication in a specified species (12, 23). Standard doses used for the calculation of the ADDs (i.e., the defined daily dose) were obtained either from the drug labels or based on convention in our hospitals. Specifically, the report provided data on (1) the percent of visits in which an antimicrobial or highest priority critically important antimicrobial (HP-CIA—an antimicrobial class that is the sole, or one of limited available therapies, to treat serious bacterial infections in people) (24) was prescribed; (2) the prescription rate, or number of antimicrobial ADDs per 1,000 patient-days; (3) the average number of antimicrobial ADDs per patient; (4) the average number of antimicrobial classes prescribed per visit; (5) and rankings of the most frequently prescribed classes and combinations of antimicrobials. Data were presented via both prose and graphics, and detailed definitions of the ADD metrics along with examples of its use were provided (see example report).
For the large animal hospital clinicians, prescribing patterns were situated relative to peers within their service. Because some of the small animal hospital clinicians were involved in multiple services, their prescribing patterns were situated relative to peers within the entire hospital rather than within their service. For clinicians for whom there were insufficient prescribing data to create a personalized report (e.g., clinicians who were not employed by the hospitals from 2013 to 2018), a mock report was generated using data from a randomly selected de-identified clinician who was present during the time period of interest. These clinicians were made aware when presented the report that it did not contain their actual data, and they were asked to imagine how it might feel to receive such a report. The interviewer gave the respondent as much time as they needed to review the report and then asked them a series of questions to elicit their perceptions about the report and each of the metrics.
Data Analysis
All audio files were transcribed and uploaded into NVivo 12 software for coding (25). Data were independently analyzed using a flexible coding approach by two coders (26). Themes were systematically identified in a two-stage process. First, a codebook based on the interview guides and a review of the data collection memos was developed. Codes were defined clearly and discussed among the team. Second, the coders applied the codebook to the transcripts. Intercoder reliability was monitored throughout, and modifications were made to the coding procedure to ensure agreement exceeded 95%. Once line-by-line coding was complete, we utilized a framework matrix to identify variation in patterns across codes, respondent classifications, and hospitals (27).
Results
Characteristics of Study Subjects
Interviews were conducted with 34 veterinarians. The majority of respondents worked in a small animal hospital setting and had been in practice 10 years or less (Table 1). Interviews ranged in length from 18 to 68 min, with a median of 32 min. Twenty-one (61.8%) respondents were shown their actual prescribing data while 13 (38.2%) were shown mock prescribing data.
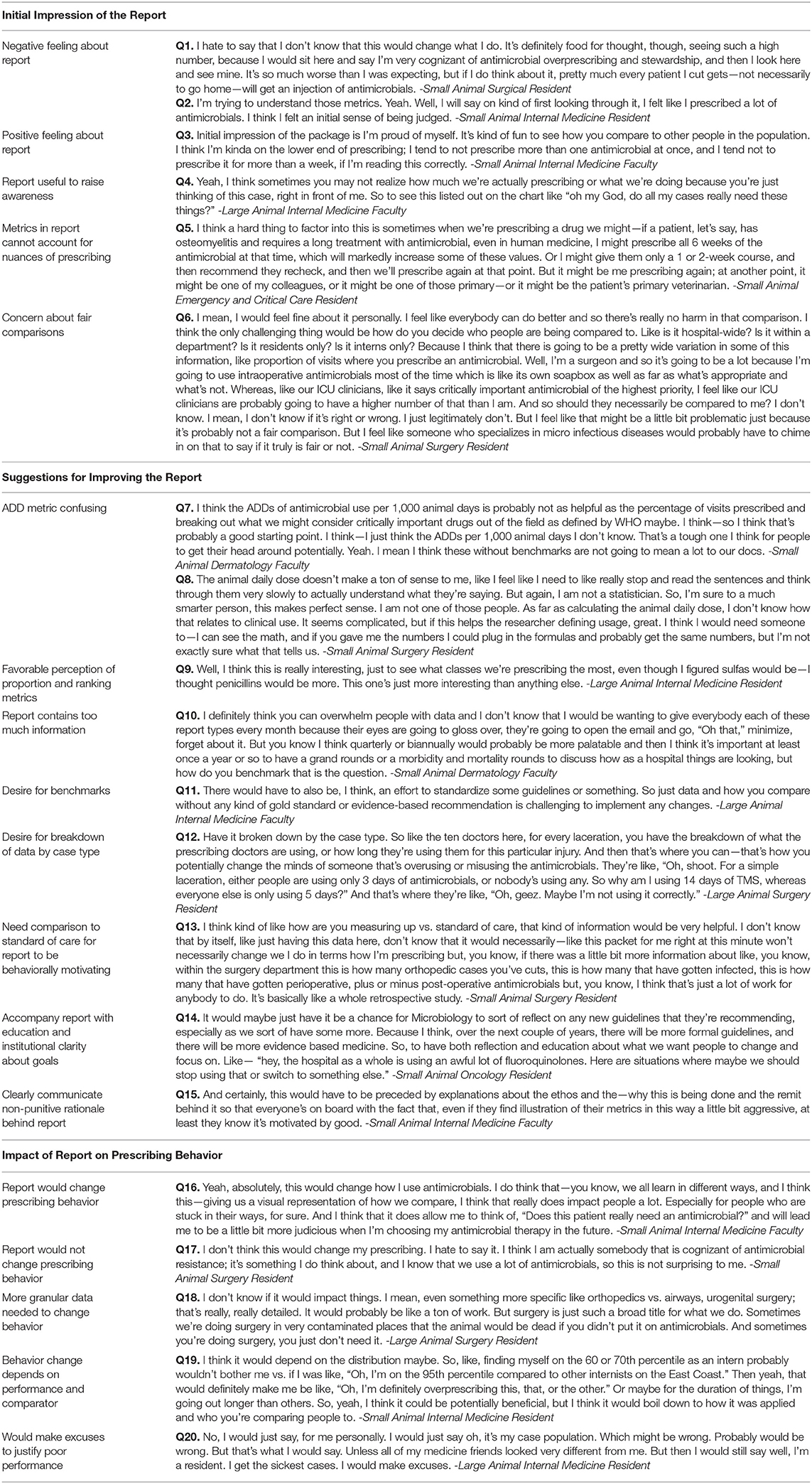
Initial Impression of Report
Upon initial presentation, 8 (23.5%) respondents expressed negative feelings about the report, 13 (38.2%) expressed positive feelings, while 13 (38.2%) were neutral in their response. Of those respondents who were shown their actual prescribing data, 6 (28.5%) expressed negative feelings about the report while 11 (52.3%) expressed positive feelings about the report. The primary reason respondents gave for feeling negatively about the report was believing that their actual antimicrobial use performance was better than the report indicated (Table 2, Quote 1 [Q1]). The surprise at seeing one's poorer than expected performance coupled with comparison to colleagues led some respondents to explain that the report made them feel “judged” (Q2). Respondents who were pleased explained that their data was on par with or better than how they perceived their actual use of antimicrobials (Q3).
After considering the data in the reports carefully, all respondents expressed appreciation for the metrics. Many said they had not ever seen data like this before and felt, in general, that communicating any data about antimicrobial use could be an important technique to encourage veterinarians to think about their prescribing decisions in the aggregate, which could improve antimicrobial use (Q4). Respondents' critical feedback of the reports primarily focused on doubt that the data could account for the nuances of prescribing in diverse clinical scenarios that might cause some veterinarians to justifiably use more antimicrobials than their peers (Q5). In reflecting on how it felt to be compared to colleagues, respondents generally found this approach to be informative and motivating for behavior change. However, some expressed concern that the comparisons might not be fair based on each individual veterinarian's case mix (Q6).
Specific Suggestions for Improving Report
While some respondents (n = 14, 41.2%) felt that the report was satisfactory and did not need changes, most had several suggestions as to how to improve the clarity and impact of the data. The majority of respondents (n = 22, 64.7%) found the animal defined daily doses metric to be the least meaningful and most confusing of all the metrics despite an explanatory page at the back of the report (Q7). Respondents suggested that this metric was not intuitive or clinically relevant and would take too much time for a non-statistically savvy, busy veterinarian to understand and find it meaningful (Q8). In comparison, respondents felt more favorably toward the proportion metrics and the ranking of antimicrobial classes (Q9). Some suggested moving the explanatory page that described how the metrics were calculated to the front of the report. Other respondents felt that the amount of information provided in the reports was overwhelming and suggested delivering less information all at once or using a phased approach so people could get used to receiving a report of this length (Q10).
One of the most common reactions to the report was a desire for benchmarks that could help put the individual's performance in context (Q11). We found, as respondents looked at the reports and thought aloud, that they felt the data would be more meaningful if it could be broken down further. Specific suggestions included organizing data by case or procedure type (Q12) and comparing the following: systemic vs. regional use of antimicrobials, colleagues only within the same specialty, prescribing data by species, farm vs. inpatient large animal use of antimicrobials, and prescribing data from other universities or hospitals.
The majority (n = 26, 76.5%) of respondents felt that supplementary information should be provided with reports to make them more actionable. For example, some respondents suggested that the metrics would only be meaningful and likely to produce behavioral change if it was clear that there was a “gold standard” or guideline that suggested what performance on each metric was “good” (Q13). Without a goal to strive for, many respondents expressed, the numbers alone would not motivate them to change. Others felt that the reports would be more useful if they were accompanied by institution-wide discussion about the trends and education about specific classes of antimicrobials that are overused or clinical scenarios in which prescribing could be improved (Q14). Most importantly, the “ethos” behind the report would need to be clearly communicated so prescribers would know that the intent of the report was to improve the quality of care delivered and not to punish individuals (Q15).
Influence of Report on Prescribing Behavior
Respondents varied in whether they felt their prescribing behavior would change in response to receiving the reports. Some (n = 13, 38.2%) felt that the report would change how they prescribed antimicrobials (Q16). A smaller number (n = 8, 23.5%) felt that they are already making the most judicious prescribing choices possible and did not believe the report would change the way they prescribe (Q17). Others suggested that without a goal to strive for, the data were not motivating (Q13).
Other respondents (n = 13, 38.2%) were undecided as to whether the data would change their prescribing and offered a variety of reasons for this equivocation. They explained that prescribing choices are influenced by an individual's accumulated clinical experience and the circumstances of each case; therefore, it was difficult for them to imagine a report with such high-level, aggregate metrics prompting change in practice as a whole (Q18). These respondents found the data to be informative but felt that more detail would be needed before the reports would change how they use antimicrobials. Multiple individuals said that the impact of the reports would depend on where they found themselves on the distribution compared to their colleagues and whether they felt the comparison was fair (Q19). Other respondents were unsure if the reports would motivate them to change and admitted that they would likely think of “excuses” to justify their high-prescribing rates in comparison to their colleagues (Q20).
Discussion
With increasing interest in antimicrobial stewardship in veterinary medicine (11, 28), methods to achieve stewardship goals are needed (29). In this study, we demonstrated the feasibility of providing individualized antimicrobial use reports to clinicians in veterinary hospitals, and we obtained feedback on the perceived utility of the reports in general and of the individual metrics used in the reports. The reports were generally well-received by clinicians, and all clinicians were appreciative of being able to visualize their antimicrobial prescribing patterns using different metrics.
Antimicrobial stewardship initiatives involving the provision of periodic feedback to clinicians on their antimicrobial prescribing have been used successfully in human medicine (6, 30–34) and in animal agriculture (7, 35, 36). In veterinary hospitals, such initiatives are being proposed (37, 38). However, as made clear by the participants of our study, who expressed a desire for information with which to gauge the appropriateness of their AMU, prescribing behavior should be benchmarked against universally accepted guidelines to be useful. In veterinary medicine, a general consensus about broad tenets of judicious AMU exist (39), but defined AMU guidelines exist for only a few select clinical conditions in small animal medicine (40–43). Adherence to these guidelines have been investigated within veterinary institutions (44, 45), but, to our knowledge, no use has been made of them to provide feedback to clinicians in a veterinary hospital setting. In small animals, antimicrobial use reports targeted to the clinical conditions for which guidelines exist represent the most logical option for a feedback-related antimicrobial stewardship intervention. Such interventions in the context of acute respiratory tract infections have been performed in human medicine (6, 33). However, targeted reports evaluate only a small proportion of an individual's prescriptions and may limit who can be evaluated, as veterinarians in certain specialties may not see patients with the conditions of interest. This is particularly problematic for large animal medicine, where AMU guidelines for specific conditions are lacking. The dearth of defined AMU guidelines represents an impediment to promoting appropriate antimicrobial use in the veterinary hospital, and more research is needed to develop such guidelines.
An important theme that emerged from our interviews was that of comparability. When benchmarking AMU across different units (e.g., clinicians, services, hospitals), comparability of patient populations seen by different clinicians is critical. Many of the veterinarians interviewed in this study expressed skepticism that their results could be meaningfully compared to those of their peers, as patient populations attended to differed greatly across and even within services. Situating an individual's prescribing patterns relative to peers within the same service is necessary but does not appear sufficient. Additional methods of ensuring comparability of patient populations seen by a clinician are needed. In human medicine, scoring systems such as the Medicare Severity Diagnosis Related Group or Charlson comorbidity score have been used when benchmarking AMU (46, 47). While scoring systems exist for specific conditions [e.g., equine colic (48, 49)], to our knowledge, there are no validated methods of more generally characterizing the disease severity or comorbidities of veterinary patients. Until such scoring systems are developed, clinicians may view attempts to compare their prescribing to peers' as invalid or unfair.
The question of which metrics to use when benchmarking or evaluating the appropriateness of AMU is a critical one that remains unresolved in both human medicine (50–52) and veterinary medicine (12, 53). In human medicine, a wide variety of AMU metrics exist for purposes of antimicrobial stewardship, many of which are similar to or equivalent to those used in our study (54). In animal agriculture, different metrics are used in different countries and systems (35, 55, 56), and an expansive body of literature has described and evaluated the advantages and disadvantages of these metrics (12, 23, 57–59). However, because herds of animals are treated on farms more often than individual animals, the metrics used on the farm setting may not translate well to the hospital setting. While there is some consensus among experts about which metrics should be used in the human hospital setting (60), others have argued that certain metrics, most notably the dose-based metrics which are equivalent to the ADD-based metrics used in this study, should not be used for purposes of antimicrobial stewardship (1, 52). This was corroborated by our study, as most of the clinicians found the ADD-based metrics to be confusing, even when provided with detailed explanations. While ADD-based metrics are appealing because they can be calculated from drug volumes that are often accessible in pharmacy or billing records, they are not intuitive. Moreover, we have shown that in veterinary medicine they are not, as they should theoretically be, equivalent to more intuitive duration-based metrics such as days of therapy (61) which have been used in human medicine for stewardship purposes (34). Dose-based metrics are useful for benchmarking AMU at the farm, regional, national, and international levels (62–67) and are generally recommended in these settings to monitor trends in use over time (12). Moreover, because the ADD represents a scaling factor more than an indicator of absolute AMU (23), it is well-suited for comparing a prescriber or user to his/her peers. However, at the level of the individual clinician working in a hospital setting, the ADD-based metrics are not immediately intuitive. It is unknown if prolonged exposure to these metrics will enhance a clinician's ease with these metrics, or if, as one clinician noted, they are just “tough […] for people to get their head around.” It has been suggested that duration-based metrics (e.g., individual days treated or number of individuals daily treated) may be more appropriate for hospital settings (12, 68). Unfortunately, these metrics were not easily accessible from our database and could not be extracted to present to clinicians.
In contrast, the proportion metrics (e.g., percent of visits where an antimicrobial or HP-CIA was prescribed) and the ranking of antimicrobial classes were the most well-received metrics. Both of these metrics or variants thereof [e.g., antimicrobial spectrum score (34, 69)] have been used successfully to gauge appropriateness of antimicrobial prescribing by physicians (6, 70) and veterinarians (44, 45, 71). These metrics capture information about the frequency and choice of antimicrobial prescriptions and can therefore be useful in encouraging clinicians to limit unnecessary prescribing of antimicrobials and optimize empiric antimicrobial regimens (e.g., targeted therapy over broad spectrum, lower vs. higher priority antimicrobials) (71, 72). Because they are also inherently intuitive, they were likely to be more readily accepted by clinicians.
Audit and feedback is a commonly used quality improvement intervention in human medicine. It has been demonstrated, however, that the effects of these interventions vary greatly and are not improving over time (13, 73). Simply providing professionals with data is not enough to stimulate change in the way they perform their work (74). Numbers must have salience and meaning to the individuals whose behavior is targeted for change. Antimicrobial stewardship feedback interventions that are consistent with the priorities, beliefs, and concerns of prescribers and that make meaningful social comparisons may be more successful than those that do not (5). Our study generates knowledge that can be used to inform the design and implementation of stewardship interventions in veterinary medicine. To advance the science of stewardship, interventions need to account for the social and behavioral mechanisms that are particular to the professional culture of veterinary medicine (75, 76).
Our study has several limitations. First, because we adopted a qualitative approach our findings may not be generalizable beyond the settings we studied. Second, despite explicit efforts to minimize their effects, our sample may be biased. It is possible that the respondents who agreed to participate to an interview possessed systematically different characteristics that influenced their willingness to participate and shaped their perceptions compared to those not interviewed. We were unable to assess the characteristics of respondents vs. non-respondents. It is also possible that interview respondents did not honestly share their perceptions about the Personalized Antimicrobial Use Reports in order to please the interviewer. Given that respondents did express criticism of the AMU metrics and their feelings about antimicrobial stewardship interventions, we believe the impact of social desirability bias on our data to be minimal.
Conclusion
Providing feedback on antimicrobial prescribing to individual veterinary clinicians may be an effective antimicrobial stewardship intervention, as long as the metrics used to describe prescribing patterns are understood by clinicians. Prescribing frequency, durations of therapy, and ranking of antimicrobial classes appear to be the metrics most well-received by veterinary clinicians, while dose-based metrics associated with the ADD are less intuitive. However, more research is needed to establish antimicrobial use guidelines against which prescribing can be measured.
Data Availability Statement
The raw data generated and analyzed during this study is not publicly available due to the sensitive nature of the data and ethics restrictions on data sharing. Respondents did not consent to have their data publicly shared. A de-identified dataset may be made available by reasonable request.
Ethics Statement
The studies involving human participants were reviewed and approved by Institutional Review Board of the University of Pennsylvania. The patients/participants provided their written informed consent to participate in this study.
Author Contributions
LR produced individual antimicrobial use reports for all clinicians, contributed to data analysis, and manuscript writing. BM interviewed participants and contributed to data coding and analysis. JS interviewed participants and contributed to data coding, data analysis, and manuscript writing. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by funding from the University Research Foundation of the University of Pennsylvania. JS received funding from the US Centers for Disease Control and Prevention's Cooperative Agreement [FOA#CK16-004] Epicenters for the Prevention of Healthcare Associated Infections.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fvets.2020.00582/full#supplementary-material
References
1. Society for Healthcare Epidemiology of A Infectious Diseases Society of A Pediatric Infectious Diseases S. Policy statement on antimicrobial stewardship by the Society for Healthcare Epidemiology of America (SHEA), the Infectious Diseases Society of America (IDSA), and the Pediatric Infectious Diseases Society (PIDS). Infect Control Hosp Epidemiol. (2012) 33:322–7. doi: 10.1086/665010
2. Dellit TH, Owens RC, McGowan JE Jr, Gerding DN, Weinstein RA, Burke JP, et al. Infectious diseases society of America and the society for healthcare epidemiology of America guidelines for developing an institutional program to enhance antimicrobial stewardship. Clin Infect Dis. (2007) 44:159–77. doi: 10.1086/510393
3. Meeker D, Linder JA, Fox CR, Friedberg MW, Persell SD, Goldstein NJ, et al. Effect of behavioral interventions on inappropriate antibiotic prescribing among primary care practices: a randomized clinical trial. JAMA. (2016) 315:562–70. doi: 10.1001/jama.2016.0275
4. Shively NR, Buehrle DJ, Wagener MM, Clancy CJ, Decker BK. Improved antibiotic prescribing within a veterans affairs primary care system through a multifaceted intervention centered on peer comparison of overall antibiotic prescribing rates. Antimicrob Agents Chemother. (2019) 64:e00928–19. doi: 10.1128/AAC.00928-19
5. Milani RV, Wilt JK, Entwisle J, Hand J, Cazabon P, Bohan JG. Reducing inappropriate outpatient antibiotic prescribing: normative comparison using unblinded provider reports. BMJ Open Qual. (2019) 8:e000351. doi: 10.1136/bmjoq-2018-000351
6. Gerber JS, Prasad PA, Fiks AG, Localio AR, Grundmeier RW, Bell LM, et al. Effect of an outpatient antimicrobial stewardship intervention on broad-spectrum antibiotic prescribing by primary care pediatricians: a randomized trial. JAMA. (2013) 309:2345–52. doi: 10.1001/jama.2013.6287
7. Speksnijder DC, Mevius DJ, Bruschke CJ, Wagenaar JA. Reduction of veterinary antimicrobial use in the Netherlands. The Dutch success model. Zoonoses Public Health. (2015) 62(Suppl. 1):79–87. doi: 10.1111/zph.12167
8. Agunos A, Leger DF, Carson CA, Gow SP, Bosman A, Irwin RJ, et al. Antimicrobial use surveillance in broiler chicken flocks in Canada, 2013-2015. PLoS ONE. (2017) 12:e0179384. doi: 10.1371/journal.pone.0179384
9. BMEL. Report of the Federal Ministry of Food and Agriculture on the Evaluation of the Antimicrobials Minimisation Concept Introduced With the 16th Act to Amend the Medicinal Products Act (16th AMG Amendment). Bonn, Germany (2019).
10. Jacob ME, Hoppin JA, Steers N, Davis JL, Davidson G, Hansen B, et al. Opinions of clinical veterinarians at a US veterinary teaching hospital regarding antimicrobial use and antimicrobial-resistant infections. J Am Vet Med Assoc. (2015) 247:938–44. doi: 10.2460/javma.247.8.938
11. AVMA Task Force for Antimicrobial Stewardship in Companion Animal Practice. Understanding companion animal practitioners' attitudes toward antimicrobial stewardship. J Am Vet Med Assoc. (2015) 247:883–4. doi: 10.2460/javma.247.8.883
12. Collineau L, Belloc C, Stärk KD, Hémonic A, Postma M, Dewulf J, et al. Guidance on the selection of appropriate indicators for quantification of antimicrobial usage in humans and animals. Zoonoses Public Health. (2017) 64:165–84. doi: 10.1111/zph.12298
13. Brehaut JC, Colquhoun HL, Eva KW, Carroll K, Sales A, Michie S, et al. Practice feedback interventions: 15 suggestions for optimizing effectiveness. Ann Intern Med. (2016) 164:435–41. doi: 10.7326/M15-2248
14. Rosa RM, Bushman AM. Impact of peer comparison on carbapenem use among inpatient prescribers at a community hospital. Infect Control Hosp Epidemiol. (2020) 41:376–8. doi: 10.1017/ice.2019.377
15. Charani E, Castro-Sanchez E, Sevdalis N, Kyratsis Y, Drumright L, Shah N, et al. Understanding the determinants of antimicrobial prescribing within hospitals: the role of “prescribing etiquette”. Clin Infect Dis. (2013) 57:188–96. doi: 10.1093/cid/cit212
16. Charani E, Ahmad R, Rawson TM, Castro-Sanchèz E, Tarrant C, Holmes AH. The differences in antibiotic decision-making between acute surgical and acute medical teams: an ethnographic study of culture and team dynamics. Clin Infect Dis. (2018) 69:12–20. doi: 10.1093/cid/ciy844
17. Szymczak JE, Feemster KA, Zaoutis TE, Gerber JS. Pediatrician perceptions of an outpatient antimicrobial stewardship intervention. Infect Control Hosp Epidemiol. (2014) 35(Suppl. 3):S69–78. doi: 10.1086/677826
18. Bork JT, Morgan DJ, Heil EL, Pineles L, Kleinberg M. Peer comparison of anti-MRSA agent prescription in the inpatient setting. Infect Control Hosp Epidemiol. (2017) 38:1506–8. doi: 10.1017/ice.2017.219
19. Landis-Lewis Z, Kononowech J, Scott WJ, Hogikyan RV, Carpenter JG, Periyakoil VS, et al. Designing clinical practice feedback reports: three steps illustrated in Veterans Health Affairs long-term care facilities and programs. Implement Sci. (2020) 15:7. doi: 10.1186/s13012-019-0950-y
20. Weiss RS. Learning From Strangers: The Art and Method of Qualitative Interview Studies. New York, NY: Free Press (1994).
21. Malterud K, Siersma VD, Guassora AD. Sample size in qualitative interview studies: guided by information power. Qual Health Res. (2016) 26:1753–60. doi: 10.1177/1049732315617444
22. Redding LE, Lavigne S, Aceto HW, Nolen-Walston RD. Antimicrobial prescribing patterns of clinicians and clinical services at a large animal veterinary teaching hospital. Am J Vet Res. (2020) 81:103–15. doi: 10.2460/ajvr.81.2.103
23. Jensen VF, Jacobsen E, Bager F. Veterinary antimicrobial-usage statistics based on standardized measures of dosage. Prev Vet Med. (2004) 64:201–15. doi: 10.1016/j.prevetmed.2004.04.001
24. World Health Organization. Critically Important Antimicrobials for Human Medicine. Geneva: WHO (2017).
25. QSR International Pty Ltd. NVivo (Version 12) (2019). Available online at: https://www.qsrinternational.com/nvivo-qualitative-data-analysissoftware/home
26. Deterding NM, Waters MC. Flexible coding of in-depth interviews: a twenty-first-century approach. Sociol Methods Res. (2018) 0049124118799377. doi: 10.1177/0049124118799377
27. Gale NK, Heath G, Cameron E, Rashid S, Redwood S. Using the framework method for the analysis of qualitative data in multi-disciplinary health research. BMC Med Res Methodol. (2013) 13:117. doi: 10.1186/1471-2288-13-117
28. Prescott JF, Boerlin P. Antimicrobial use in companion animals and good stewardship practice. Vet Record. (2016) 179:486–8. doi: 10.1136/vr.i5908
29. Guardabassi L, Prescott JF. Antimicrobial stewardship in small animal veterinary practice: from theory to practice. Vet Clin North Am Small Anim Pract. (2015) 45:361–76. doi: 10.1016/j.cvsm.2014.11.005
30. Arnold FW, McDonald LC, Smith RS, Newman D, Ramirez JA. Improving antimicrobial use in the hospital setting by providing usage feedback to prescribing physicians. Infect Control Hosp Epidemiol. (2016) 27:378–82. doi: 10.1086/503336
31. Hux JE, Melady MP, DeBoer D. Confidential prescriber feedback and education to improve antibiotic use in primary care: a controlled trial. CMAJ. (1999) 161:388–92.
32. Zwar N, Wolk J, Gordon J, Sanson-Fisher R, Kehoe L. Influencing antibiotic prescribing in general practice: a trial of prescriber feedback and management guidelines. Fam Pract. (1999) 16:495–500. doi: 10.1093/fampra/16.5.495
33. Tong I, Dean K, Park D, Thompson J, Yan L, Liu C, et al. 195. The effect of two antibiotic stewardship interventions in a telemedicine practice. Open Forum Infect Dis. (2018) 5(Suppl. 1):S85–S6. doi: 10.1093/ofid/ofy210.208
34. McIntyre MT, Naik L, Bell CM, Morris AM. Development and assessment of a physician-specific antimicrobial usage and spectrum feedback tool. Open Forum Infect Dis. (2017) 4:ofx124. doi: 10.1093/ofid/ofx124
35. Craig AL, Buijs S, Morrison S. Evaluation of veterinary antimicrobial benchmarking systems at farm- level in Europe: implications for the UK ruminant sector. Vet Record. (2020). doi: 10.1136/vr.105727. [Epub ahead of print].
36. Canadian Integrated Program for Antimicrobial Resistance. Reductions in Antimicrobial Use and Resistance: Preliminary Evidence of the Effect of the Canadian Chicken Industry's Elimination of Use of Antimicrobials of Very High Importance to Human Medicine. (2016). Available online at: https://www.canada.ca/en/public-health/services/publications/drugs-health-products/canadian-integrated-programantimicrobial-resistances-surveillance-bulletin.html (accessed August 21, 2020).
37. CAVSNET. Companion Animal Veterinary Surveillance Network: University of Minnesota. Available online at: https://cavsnet.umn.edu/about-cavsnet (accessed August 21, 2020).
38. SAVSNET. Small Animal Veterinary Surveillance Network University of Liverpool. Available online at: https://www.liverpool.ac.uk/savsnet/ (accessed August 21, 2020).
39. Weese JS, Giguere S, Guardabassi L, Morley PS, Papich M, Ricciuto DR, et al. ACVIM consensus statement on therapeutic antimicrobial use in animals and antimicrobial resistance. J Vet Intern Med. (2015) 29:487–98. doi: 10.1111/jvim.12562
40. Lappin MR, Blondeau J, Boothe D, Breitschwerdt EB, Guardabassi L, Lloyd DH, et al. Antimicrobial use guidelines for treatment of respiratory tract disease in dogs and cats: antimicrobial guidelines working group of the international society for companion animal infectious diseases. J Vet Intern Med. (2017) 31:279–94. doi: 10.1111/jvim.14627
41. Weese JS, Blondeau JM, Boothe D, Breitschwerdt EB, Guardabassi L, Hillier A, et al. Antimicrobial use guidelines for treatment of urinary tract disease in dogs and cats: antimicrobial guidelines working group of the international society for companion animal infectious diseases. Vet Med Int. (2011) 2011:263768. doi: 10.4061/2011/263768
42. Marks SL, Rankin SC, Byrne BA, Weese JS. Enteropathogenic bacteria in dogs and cats: diagnosis, epidemiology, treatment, and control. J Vet Intern Med. (2011) 25:1195–208. doi: 10.1111/j.1939-1676.2011.00821.x
43. Hillier A, Lloyd DH, Weese JS, Blondeau JM, Boothe D, Breitschwerdt E, et al. Guidelines for the diagnosis and antimicrobial therapy of canine superficial bacterial folliculitis (Antimicrobial Guidelines Working Group of the International Society for Companion Animal Infectious Diseases). Vet Dermatol. (2014) 25:163–e43. doi: 10.1111/vde.12118
44. Banfield Pet Hospital. Are We Doing Our Part to Prevent Superbugs? Antimicrobial Usage Patterns Among Companion Animal Veterinarians (2017).
46. Polk RE, Hohmann SF, Medvedev S, Ibrahim O. Benchmarking risk-adjusted adult antibacterial drug use in 70 US academic medical center hospitals. Clin Infect Dis. (2011) 53:1100–10. doi: 10.1093/cid/cir672
47. Ibrahim OM, Polk RE. Benchmarking antimicrobial drug use in hospitals. Expert Rev Anti Infect Ther. (2012) 10:445–57. doi: 10.1586/eri.12.18
48. Furr MO, Lessard P, White NA II. Development of a colic severity score for predicting the outcome of equine colic. Vet Surg. (1995) 24:97–101. doi: 10.1111/j.1532-950X.1995.tb01302.x
49. Grulke S, Olle E, Detilleux J, Gangl M, Caudron I, Serteyn D. Determination of a gravity and shock score for prognosis in equine surgical colic. J Vet Med Ser A. (2001) 48:465–73. doi: 10.1046/j.1439-0442.2001.00374.x
50. van den Bosch CMA, Geerlings SE, Natsch S, Prins JM, Hulscher MEJL, Perl TM. Quality indicators to measure appropriate antibiotic use in hospitalized adults. Clin Infect Dis. (2014) 60:281–91. doi: 10.1093/cid/ciu747
51. Spivak ES, Cosgrove SE, Srinivasan A. Measuring appropriate antimicrobial use: attempts at opening the black box. Clin Infect Dis. (2016) 63:1639–44. doi: 10.1093/cid/ciw658
52. Morris AM. Antimicrobial stewardship programs: appropriate measures and metrics to study their impact. Curr Treat Options Infect Dis. (2014) 6:101–12. doi: 10.1007/s40506-014-0015-3
53. Mills HL, Turner A, Morgans L, Massey J, Schubert H, Rees G, et al. Evaluation of metrics for benchmarking antimicrobial use in the UK dairy industry. Vet Record. (2018) 182:379. doi: 10.1136/vr.104701
54. Ibrahim OM, Polk RE. Antimicrobial use metrics and benchmarking to improve stewardship outcomes: methodology, opportunities, and challenges. Infect Dis Clin North Am. (2014) 28:195–214. doi: 10.1016/j.idc.2014.01.006
55. Carmo LP, Schupbach-Regula G, Muntener C, Chevance A, Moulin G, Magouras I. Approaches for quantifying antimicrobial consumption per animal species based on national sales data: a Swiss example 2006 to 2013. Euro Surveill. (2017) 22:30458. doi: 10.2807/1560-7917.ES.2017.22.6.30458
56. Deckert A, Gow S, Rosengren L, Leger D, Avery B, Daignault D, et al. Canadian integrated program for antimicrobial resistance surveillance (CIPARS) farm program: results from finisher pig surveillance. Zoonoses Public Health. (2010) 57(Suppl. 1):71–84. doi: 10.1111/j.1863-2378.2010.01356.x
57. Bosman AL, Loest D, Carson CA, Agunos A, Collineau L, Leger DF. Developing Canadian defined daily doses for animals: a metric to quantify antimicrobial use. Front Vet Sci. (2019) 6:220. doi: 10.3389/fvets.2019.00220
58. Postma M, Sjolund M, Collineau L, Losken S, Stark KD, Dewulf J, et al. Assigning defined daily doses animal: a European multi-country experience for antimicrobial products authorized for usage in pigs. J Antimicrob Chemother. (2015) 70:294–302. doi: 10.1093/jac/dku347
59. Timmerman T, Dewulf J, Catry B, Feyen B, Opsomer G, de Kruif A, et al. Quantification and evaluation of antimicrobial drug use in group treatments for fattening pigs in Belgium. Prev Vet Med. (2006) 74:251–63. doi: 10.1016/j.prevetmed.2005.10.003
60. Morris AM, Brener S, Dresser L, Daneman N, Dellit TH, Avdic E, et al. Use of a structured panel process to define quality metrics for antimicrobial stewardship programs. Infect Control Hosp Epidemiol. (2015) 33:500–6. doi: 10.1086/665324
61. Redding LE, Grunwald H, Melofchik C, Meily P, Henry A, Stefanovski D. Comparison of animal daily doses and days of therapy for antimicrobials in species of veterinary importance. Prev Vet Med. (2020) 176:104942. doi: 10.1016/j.prevetmed.2020.104942
62. Echtermann T, Muentener C, Sidler X, Kummerlen D. Antimicrobial drug consumption on swiss pig farms: a comparison of Swiss and European defined daily and course doses in the field. Front Vet Sci. (2019) 6:240. doi: 10.3389/fvets.2019.00240
63. Sjolund M, Postma M, Collineau L, Losken S, Backhans A, Belloc C, et al. Quantitative and qualitative antimicrobial usage patterns in farrow-to-finish pig herds in Belgium, France, Germany and Sweden. Prev Vet Med. (2016) 130:41–50. doi: 10.1016/j.prevetmed.2016.06.003
64. Grave K, Torren-Edo J, Mackay D. Comparison of the sales of veterinary antibacterial agents between 10 European countries. J Antimicrob Chemother. (2010) 65:2037–40. doi: 10.1093/jac/dkq247
65. Sarrazin S, Joosten P, Van Gompel L, Luiken REC, Mevius DJ, Wagenaar JA, et al. Quantitative and qualitative analysis of antimicrobial usage patterns in 180 selected farrow-to-finish pig farms from nine European countries based on single batch and purchase data. J Antimicrob Chemother. (2018) 74:807–16. doi: 10.1093/jac/dky503
66. DANMAP. DANMAP (2015) - Use of Antimicrobial Agents and Occurrence of Antimicrobial Resistance in Bacteria From Food Animals, Food and Humans in Denmark. Copenhagen (2015).
67. European Medicines Agency. Sales of Veterinary Antimicrobial Agents in 29 European Countries in 2014. Amsterdam, Netherlands (2016).
68. Hamilton KW, Fishman NO. Antimicrobial stewardship interventions: thinking inside and outside the box. Infect Dis Clin North Am. (2014) 28:301–13. doi: 10.1016/j.idc.2014.01.003
69. Madaras-Kelly K, Jones M, Remington R, Hill N, Huttner B, Samore M. Development of an antibiotic spectrum score based on veterans affairs culture and susceptibility data for the purpose of measuring antibiotic de-escalation: a modified Delphi approach. Infect Control Hosp Epidemiol. (2014) 35:1103–13. doi: 10.1086/677633
70. Grijalva CG, Nuorti JP, Griffin MR. Antibiotic prescription rates for acute respiratory tract infections in US ambulatory settings. JAMA. (2009) 302:758–66. doi: 10.1001/jama.2009.1163
71. Hopman NEM, Portengen L, Hulscher MEJL, Heederik DJJ, Verheij TJM, Wagenaar JA, et al. Implementation and evaluation of an antimicrobial stewardship programme in companion animal clinics: a stepped-wedge design intervention study. PLoS ONE. (2019) 14:e0225124. doi: 10.1371/journal.pone.0225124
72. McNeil V, Cruickshank M, Duguid M. Safer use of antimicrobials in hospitals: the value of antimicrobial usage data. Med J Australia. (2010) 193:S114–S7. doi: 10.5694/j.1326-5377.2010.tb04026.x
73. Grimshaw JM, Ivers N, Linklater S, Foy R, Francis JJ, Gude WT, et al. Reinvigorating stagnant science: implementation laboratories and a meta-laboratory to efficiently advance the science of audit and feedback. BMJ Qual Saf . (2019) 28:416. doi: 10.1136/bmjqs-2018-008355
74. van Braak M, Visser M, Holtrop M, Statius Muller I, Bont J, van Dijk N. What motivates general practitioners to change practice behaviour? A qualitative study of audit and feedback group sessions in Dutch general practice. BMJ Open. (2019) 9:e025286. doi: 10.1136/bmjopen-2018-025286
75. Gude WT, Brown B, van der Veer SN, Colquhoun HL, Ivers NM, Brehaut JC, et al. Clinical performance comparators in audit and feedback: a review of theory and evidence. Implement Sci. (2019) 14:39. doi: 10.1186/s13012-019-0887-1
Keywords: veterinarian, antimicrobial use, metric, antimicrobial stewardship, feedback
Citation: Redding LE, Muller BM and Szymczak JE (2020) Small and Large Animal Veterinarian Perceptions of Antimicrobial Use Metrics for Hospital-Based Stewardship in the United States. Front. Vet. Sci. 7:582. doi: 10.3389/fvets.2020.00582
Received: 03 May 2020; Accepted: 20 July 2020;
Published: 08 September 2020.
Edited by:
Lucie Collineau, Agence Nationale de Sécurité Sanitaire de l'Alimentation, de l'Environnement et du Travail (ANSES), FranceReviewed by:
Simon More, University College Dublin, IrelandAngelina Louise Bosman, University of Guelph, Canada
Copyright © 2020 Redding, Muller and Szymczak. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Laurel E. Redding, bHJlZGRpbmdAdmV0LnVwZW5uLmVkdQ==
 Laurel E. Redding
Laurel E. Redding Brandi M. Muller
Brandi M. Muller Julia E. Szymczak2
Julia E. Szymczak2