- Behavioral Science, Ariel University, Science Park, Ariel, Israel
There is a growing use of novel psychoactive substances containing synthetic cannabinoids. Synthetic cannabinoid products have effects similar to those of natural cannabis, yet, these drugs are more potent and dangerous, and have been associated with dangerous adverse effects. Here, we review current literature on the epidemiology, acute, and chronic effects of synthetic and natural cannabinoid-based drugs. Synthetic drugs contain a mixture of psychoactive compounds that mostly bind cannabinoid receptors with high potency. These synthetic drugs replicate the effects of natural cannabis and Δ9-tetrahydrocannabinol but they induce more severe adverse effects including respiratory difficulties, hypertension, tachycardia, chest pain, muscle twitches, acute renal failure, anxiety, agitation, psychosis, suicidal ideation, and cognitive impairment. Chronic use of synthetic cannabinoids has been associated with serious psychiatric and medical conditions and even death. Given the growing popularity in the use of cannabinoid-based drugs and their harmful potential, there is a need for further research in this field.
Introduction
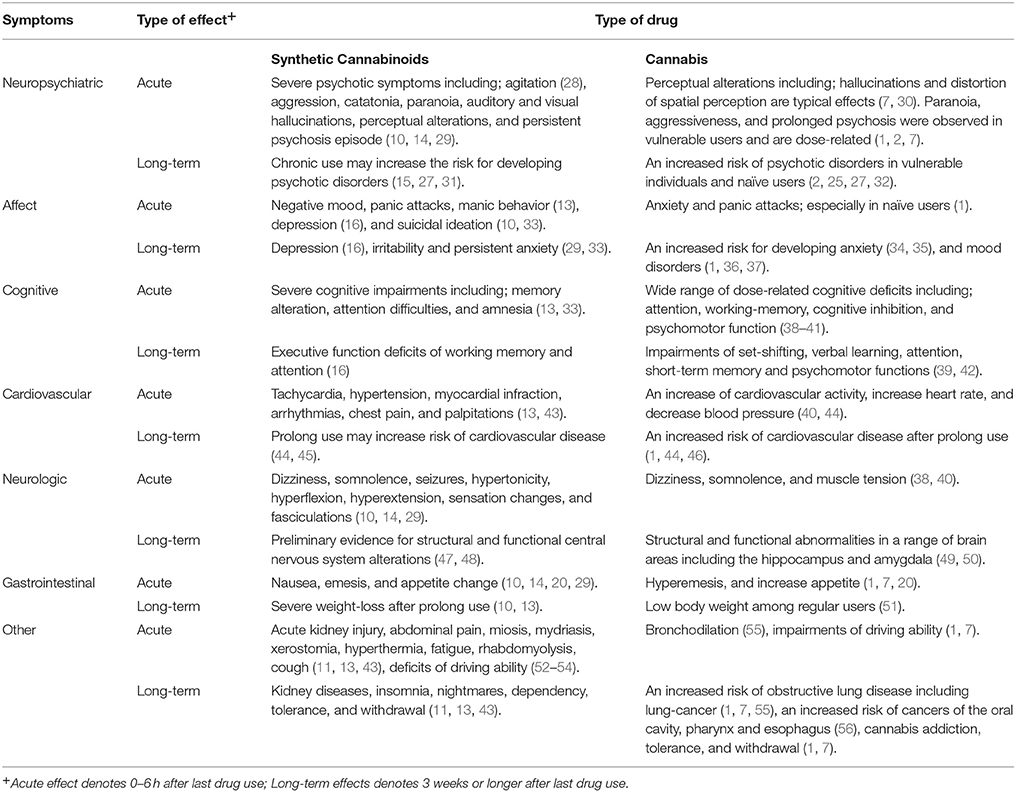
Cannabis is the most widely available and used drug across the world (1, 2). According to the United Nations Office on Drugs and Crime (UNODC) ~4% of the global adult population have used cannabis in their life. In the United States of America (USA) alone, 11% (36 million people) of adults used cannabis at least once in their past (3). In addition, the therapeutic use of cannabis and its derivatives is increasing and has been evaluated for a various health conditions including; pain, anorexia, side-effects of chemotherapy, multiple sclerosis, and muscle spasms (4–6). The primary psychoactive constituent within cannabis is Δ-9 tetra-hydro-cannabinol (THC), which interacts with CB1 and CB2 receptors and it consequently elicits its main effects (7–9). Novel Psychoactive Substances (NPS) which contain Synthetic Cannabinoids (SCs) have recently started to be used recreationally, especially by young adults (10, 11). In contrast to the decline in use of many NPSs such as the cathinones and piperazines, it appears that the number of SC users is increasing (12). Although SC drugs mimic the psychotropic effects of cannabis, their undesired effects are unpredictable and more severe than those associated with cannabis (10, 13–16). Although, there is an increasing interesting on the therapeutic potential of cannabinoid-based medications (6, 17) repeated exposure to cannabinoid-agonists in either organic or synthetic forms is associated with both physically and psychological adverse effects (2, 10, 15, 18). The most notorious psychological side effects are mental disorders including psychotic-states, schizophrenia, and affective disorders (1, 2, 10). The aim of the current review is to describe the available knowledge regarding acute and repeated consumption of both organic and synthetic cannabinoid drugs and their side effects from a public health prospective.
Epidemiology and Pattern of Use of Cannabinoids
According to national and regional representative surveys, lifetime prevalence of SC use in the general population is between 0.2 and 4% (19). By comparison, lifetime use of cannabis tends to be greater; and to range between one-quarter to one-third of the population (1). However, Winstock and Barratt reported that SC products are widely popular among recreational cannabis users (20). Among high school seniors in USA, the annual prevalence of SC usages was higher than any other drugs, with the exception of cannabis (21). Evidence accumulated from several surveys shows that between 6 and 17% of college students in USA have used SC drugs at least once during their study period (22, 23). Other than that, SC use was relatively frequent among adolescents and young-teenagers. Approximately 1% of European people between the ages of 14–18 used SC drugs at least once in their lifetime (24). This is especially important since both clinical and preclinical studies indicate that exposure to SC as well as THC during adolescence is associated with an increased risk of developing schizophrenia later in life (25–27) (see Table 1 for comparison of cannabis and SC adverse effects). Prompted by the alarming growth of the SC drugs phenomenon, legal measures to control the distribution of these drugs have been taken in many countries (57, 58). For example, in the United Kingdom (UK), “first generation” SCs were controlled in 2009 and further legislation to control so-called “second generation” of SCs drugs was enacted in 2013. Yet, subsequent manipulation of the chemical structure of these compounds has resulted in a novel generation of SC's that are not currently legally controlled in the UK (57). Unfortunately, a similar pattern was observed in other countries as well, manufacturers of SCs are aware of the chemical analog loopholes in the law and continue to manipulate SCs as necessary to keep them legal for distribution (26, 57–59). In line with this, a recent epidemiological study by Waugh et al. (57) reported substantial increase in SC drug use in 2011 despite prior legislation efforts. Suggesting, that legislation efforts alone have an insufficient effect on the distribution and use of SC drugs, and further prevention efforts are required to control this phenomenon (57).
Naive consumers typically report using SCs for various reasons, such as curiosity, high availability, easy access, and lower costs compared with cannabis. Since SCs are mostly undetectable via a simple urine test, a major motivation for consuming SC drugs is the desire to experience “cannabis-like” effects without the danger of being detected (11). Other motivations to use SCs are their relatively high availability and low prices (11, 19). In in contrast to cannabis, these synthetic drugs are typically not designed to be mixed with tobacco, probably to achieve the most intense effects (13).
Psychoactive Ingredients of Cannabinoids
The main psychoactive ingredient in cannabis is THC, which is a CB1 and CB2 receptors partial-agonist and the most potent cannabinoid that is present in the organic forms of cannabis (8, 27). Besides THC, organic cannabis products contain additional cannabinoids which do not induce psychoactive effects, such as Cannabinol, Δ8-Tetrahydrocannabinol, and Cannabidiol (CBD) (9, 60, 61). CBD is considered a non-psychoactive cannabinoid that also moderates the psycho tropic effects of THC (32, 62). Moreover, evidence is increasing that CBD has anxiolytic and antipsychotic properties (17, 62). In a broader context, CBD appears to have the ability to counteract psychotic symptoms and cognitive impairment associated with cannabis use as well as with acute THC administration (17, 63–65).
In contrast to cannabis, which contains mostly a mixture of agonist and antagonist cannabinoids (1, 7). SC's compounds show differences in their selectivity, their potency and their function (10, 26, 27, 66), in general they are more potent and efficacious cannabinoid receptor agonists than THC (11, 67). In addition, SC drugs have additional ingredients such as preservatives, additives, fatty acids, amides, esters, benzodiazepines, and O-desmethyltramadol- an active metabolite of the opioid medication tramadol (26, 31, 68). It is suggested that these additional compounds are probably added to these drugs in purposely to induce greater psychoactive effect, act as masking agents to confuse the identification of the main psychoactive substances within these drugs (31, 68).
Since 2008, at least 200 different types of SCs have been isolated from herbal mixtures in several countries (27, 69, 70). This wide variability of psychoactive compounds is probably a result of (a) the impermanent production processes of these drugs, and (b) again their ever-changing compositions in an attempt to dodge prevention and legal actions (31).
One of the earliest compounds that was identified as a psychoactive component in SC drugs is JWH-018. Contrary to THC, JWH-018 has four times the affinity for CB1 receptors and 10 times the affinity for the CB2 receptors (67). Later-on, additional cannabinomimetic compounds such as CP 47,497, cannabicyclohexanol, HU-210, and the fatty acid, oleamide have been detected in samples of SC drugs in different areas around the globe (10, 11). Prior studies examined and indicated the therapeutic potential of SCs; HU-210 proposed to have anti-depressant effects (71), HU-211 proposed to have anti-inflammatory effect on brain trauma (72), and nabilone has antiemetic and analgesic effects (73, 74). However, while these studies report an effect that is induced by exclusive compound, within SC based-drugs, a mixture these cannabinoids as well as additional psychoactive ingredients generates a condition that incudes synergistic interactions which may underline their extrema and unpredicted adverse effects (10, 31).
Adverse Health Effects of Cannabinoids
Acute Effects of Cannabinoids
The intoxication effects of cannabis are characterized by cannabis users as; mild euphoria, relaxation, and a general pleasant feeling (1, 7). These desired psychotropic effects are considered to be related to the presence of THC (8, 9). In laboratory settings, cannabis and THC induce dose-related impairment in several functions including; motor coordination and executive function (38, 75–78).
Further undesired symptoms including anxiety, panic attacks, and psychotic episodes were associated with cannabis intoxication, all of which are most often reported by naïve users and vulnerable individuals (56, 79). Similar to cannabis, the intoxication of SCs may induce reactions such as relaxation, euphoria, perceptual alteration, altered sense of time, and mild cognitive impairments (80).
These cognitive alterations increase the risk of road accidents if cannabis or SC users drive while intoxicated (1, 15). Epidemiological evidence demonstrates that cannabis is the most common illicit drug to be detected in drivers involved in fatal road accidents or stopped for dangerous driving (1). Accordingly, cannabis use by drivers is associated with an increased risk of being involved in motor vehicle crashes (81). There is less epidemiological data on the association between SC drugs and road accidents, yet, several case studies have documented driving under influence of SCs. Musshoff et al. reported 8 drivers in Germany that were stopped by police for reckless driving. Several types of SCs were identified in all suspected serum samples, drivers showed slow responses, retarded movements and an impairment of fine motor skills (52). Yeakel and Logan described 12 cases of driving under the influence of SCs, in all cases drivers presented a generally poor motor coordination (53). In a Norwegian study conducted during a 7-week period, about 2.2% of 726 blood samples that were collected from drivers that were suspected to drive under the influence of drugs, confirmed positive for SCs and majority of the drivers were involved in vehicle accidents (54). Taken together it seems that as with driving under the influence of cannabis (1, 81), driving after SCs may also increase the risk of being involved in road accidents (10, 15).
Despite the resemblances, there are major differences between the effect of cannabis and SC drugs, both in terms of spectrum and intensity of these effects (13). Case report studies indicate a wide range of undesired somatic effects ranging in intensity from nausea to more severe symptoms such as psychomotor agitation, diaphoresis, and palpitations (13, 29). Some symptoms such as seizures; agitation, hypertension, emesis, and hypokalemia are features of SC intoxication and are not present even after consuming high doses of organic cannabis (29, 31, 33).
Psychotropic effects of SC intoxication vary significantly. Some users report a feeling of sedation while others experience agitation, fatigue, and flushes (29). This variation may occur due to different concentrations of SCs within different brands (27). Compared with the intoxication of organic cannabis products which have a slow effect and gradually fade (7, 82), SCs have a shorter duration and peak earlier (83). For example, the effects of JWH-018 last approximately for 1–2 h, while CP-47,497 induce effects for ~5–6 h (20). Moreover, some adverse effects such as anxiety, hallucinations, insomnia, and psychotic episodes may be experienced for days and weeks after consuming SC products (84).
Chronic Effects of Cannabinoids
Several studies demonstrate an association between repeated cannabis use and long-lasting cognitive impairments (2, 39, 85, 86), and an increase in risk for developing a variety of mental disorders. These include anxiety (34, 35), bipolar disorder (36), depression (37), and schizophrenia (2, 25, 32). There is growing evidence that SC drugs are associated with severe negative psychiatric and medical conditions (10, 15, 29, 31). This evidence demonstrates that repeated exposures to SCs induce overall negative side-effects which are more severe and long-lasting than those related with THC (10, 15, 29, 31).
A recent study by Cohen et al. shows deficiencies in a variety of high-order cognitive functions observed among SC users compared with recreational cannabis users including working memory, attention and recall (18). Another study indicates that SC users who had acute psychotic disorder induced by SC drugs, show cognitive impairments similar to those of schizophrenic patients (87). These results are compatible with evidence from rodent studies that show that repeated exposures to SCs induces long-lasting behavioral and cognitive impairments that resemble rodent models of schizophrenia (88, 89).
There is evidence indicating adverse effects of cannabis on cardiovascular function (1, 44, 46). Studies demonstrate that cannabis use can increase the risk of serious cardiovascular condition, including artery thrombosis, vasospasm, and myocardial infarction (90). Cannabinoid agonists, as well as THC, increase heart rate in a dose-dependent manner (1). Therefore, it is likely that these effects are mediated through cannabinoid-agonist's increase in catecholamines and increased cardiac workload together with a decreased supply of oxygen (91). Since SC drugs contain psychoactive compounds which have much higher affinity at CB1 receptors (67), it is not surprising that there are several reports that indicate an association between SC use and serious cardiovascular problems such as myocardial infarction and tachycardia in both adults and adolescents (92, 93).
Cannabinoids and Psychosis
The association between cannabinoids and psychosis is reported and it is well-recognized (2, 77, 79, 94), yet, casual relations between these two factors were not established (2, 79). However, converging evidence suggests that cannabis use has the potential for inducing psychosis (31, 77, 79, 95–98). SC products contain compounds which act as highly potent CB1 and CB2 full agonists, and in contrast to natural cannabis, contain no CBD (27, 31, 79). Due to the psychoactive features of SC drug ingredients it is not surprising that there are numerous reports on healthy and vulnerable individuals who suffer from recurrent psychosis after an acute or repeated consumption of SC drugs (15, 45, 99). Recent reports in Europe suggest that 15% of SC users who report to emergency departments present psychotic symptoms (100). These figures are far greater compared to those using other types of psychoactive substance (100). In addition, compared with cannabis, psychotic symptoms that are associated with SC are more severe and gross, in some cases they can even last for weeks following last use (10, 99).
Effects of Cannabinoids on the Central Nervous System
A large number of recent studies present a range of functional and structural neuronal abnormalities associated with regular use of cannabis (32, 49, 50). Generally, cannabis users show volumetric, gray matter, and white matter structural changes in the brain, particular within limbic and prefrontal areas (49, 50, 101). In addition, pharmacological studies draw a link between cannabis use and alterations of dopamine synthesis (102). Greater dose of THC, and an earlier age of onset area associated with these neuronal alterations (2, 32).
Compared with cannabis, there are fewer brain imaging studies exploring the neural correlates of SC use. Nurmedov et al. compared 20 males who used SC products with 20 healthy control participants and reported that SC users showed smaller gray-matter volume in the thalamus and the cerebellum (47). Recently, Zorlu et al. found a reduction of white matter volumes in several brain regions including the left temporal lobe and subcortical structures among SC users (48). In another single case study, a young SC user reported severe symptoms induced following a voluntary abstinence from SC drugs. In this patient, dopamine D2 and D3 receptors availability was lower in the striatum and in extra-striatal regions in comparison with healthy control participants during abstinence but it recovered after treatment (103). These initial studies highlight some the neurotoxic potential of SC products but since they are still preliminary. Further research is warranted.
Health Hazards and Withdrawal
In contrast to cannabis, the use of SC has been associated with severe hazardous health effects on multiple systems and with death (28, 104, 105). During the last 8 years in the United Kingdom, there were 510 reports associated with SC use that required urgent medical intervention (57). In USA there were 37,500 reported cases of seizures and 3,682 reported cases of poisonings related to SC use during 2014 (106). In addition, while there is no available documentation on an overdose death as a result of cannabis use (1), there are numbers of reports indicating a fatal outcome following consumption of SC drugs. Tait et al. identified at least 26 cases of individuals who used SC products and exhibited side-effect complications that have led to their death. The major complications were cardiovascular events, respiratory depression, pulmonary complications and acute kidney injury (107). Prolonged consumption of SC is associated with serious withdrawal symptoms including; agitation, irritability, anxiety, and mood swings (43). Some of these symptoms are similar to cannabis, yet, the differences in presentation may reflect the presence of extraneous psychoactive compounds, including amphetamine-like stimulants, and the high affinity of these SCs (45, 43).
Conclusions
Cannabinoid based drugs became increasingly popular despite the risks associated with their use (31, 45, 57). While the psychotropic effects associated with natural cannabis are related to the presence of THC (1), SC products contain a wide range of high-potent full agonists of the cannabinoid receptors that induce “THC-like” effects, but they are more severe and enduring (10, 15, 29, 31). While cannabis use usually induces psychotropic effects such as euphoria, relaxation, and a general pleasant feeling, it is associated with severe side-effects (1). The use of SC drugs is associated with more undesired effects including; agitation, irritability, confusion, hallucinations, delusions, psychosis, and death (28, 104, 105).
Chronic use of SC is associated with a greater risk for developing serious mental health disorder than cannabis or other psychoactive substances (100). Chronic use of cannabinoids is associated with structural and functional neuronal alterations (15, 32, 49), these alterations are moderated by the age of onset and the type of cannabinoids (32). This is crucial since the use of SC drugs is widely popular among young adults and teenager (10, 11). In addition, legal measures to control the use of SCs are reported as not effective enough to diminished the use of these drugs (26, 31, 106, 108), thus, further prevention programs should be conducted (31). Prevention programs to control SC drugs use and distribution may be based on information communication technology (ICT) tools and target young adults and students (109–111). Initial research regarding the efficiency of ICT-based prevention programs indicated on promising results in monitoring and control NPS use and distribution (109, 111). In conclusion, SC drugs show greater toxicity than organic cannabis, and therefore further investigation of their long-lasting and acute adverse effects is required as well as better detection and controlling measures against the spread of the use of SC products.
Author Contributions
All authors listed have made a substantial, direct and intellectual contribution to the work, and approved it for publication.
Funding
The study was funded by the National Institute for Psychobiology in Israel.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We would like to thank Shlomo Tekuzener for linguistic editing of the article.
References
1. Hall W, Degenhardt L. Adverse health effects of non-medical cannabis use. Lancet (2009) 374:1383–91. doi: 10.1016/S0140-6736(09)61037-0
2. Volkow ND, Swanson JM, Evins AE, DeLisi LE, Meier MH, Gonzalez R, et al. Effects of cannabis use on human behavior, including cognition, motivation, and psychosis: a review. JAMA Psychiatry (2016) 73:292–7. doi: 10.1001/jamapsychiatry.2015.3278
4. Borgelt LM, Franson KL, Nussbaum AM, Wang GS. The pharmacologic and clinical effects of medical cannabis. Pharmacotherapy (2013) 33:195–209. doi: 10.1002/phar.1187
5. Amato L, Minozzi S, Mitrova Z, Parmelli E, Saulle R, Cruciani F, et al. Systematic review of safeness and therapeutic efficacy of cannabis in patients with multiple sclerosis, neuropathic pain, and in oncological patients treated with chemotherapy. Epidemiol Preven. (2017) 41:279–93. doi: 10.19191/EP17.5-6.AD01.069
6. Whiting PF, Wolff RF, Deshpande S, Di Nisio M, Duffy S, Hernandez AV, et al. Cannabinoids for medical use: a systematic review and meta-analysis. JAMA (2015) 313:2456–73. doi: 10.1001/jama.2015.6358
7. Ashton CH. Pharmacology and effects of cannabis: a brief review. Br J Psychiatry (2001) 178:101–6. doi: 10.1192/bjp.178.2.101
8. Gaoni Y, Mechoulam R. Isolation, structure, and partial synthesis of an active constituent of hashish. J Am Chem Soc. (1964) 86:1646–7. doi: 10.1021/ja01062a046
9. Pertwee RG. Pharmacological actions of cannabinoids. In R. G. Pertwee editor. Handbook of Experimental Pharmacology, Vol. 168. New York, NY: Springer (2005). p. 1–51.
10. Castaneto MS, Gorelick DA, Desrosiers NA, Hartman RL, Pirard S, Huestis MA. Synthetic cannabinoids: epidemiology, pharmacodynamics, and clinical implications. Drug Alcohol Depend. (2014) 144:12–41. doi: 10.1016/j.drugalcdep.2014.08.005
11. Gunderson EW, Haughey HM, Ait-Daoud N, Joshi AS, Hart CL. A survey of synthetic cannabinoid consumption by current cannabis users. Subst Abuse (2014) 35:184–9. doi: 10.1080/08897077.2013.846288
12. Winstock A, Lynskey M, Borschmann R, Waldron J. Risk of emergency medical treatment following consumption of cannabis or synthetic cannabinoids in a large global sample. J Psychopharmacol. (2015) 29:698–703. doi: 10.1177/0269881115574493
13. Spaderna M, Addy PH, D'Souza DC. Spicing things up: synthetic cannabinoids. Psychopharmacology (2013) 228:525–40. doi: 10.1007/s00213-013-3188-4
14. Tournebize J, Gibaja V, Kahn JP. Acute effects of synthetic cannabinoids: update 2015. Subst Abuse (2017) 38:344–66. doi: 10.1080/08897077.2016.1219438
15. Weinstein AM, Rosca P, Fattore L, London ED. Synthetic cathinone and cannabinoid designer drugs pose a major risk for public health. Front Psychiatry (2017) 8:156. doi: 10.3389/fpsyt.2017.00156
16. Cohen K, Weinstein A. The effects of cannabinoids on executive functions: evidence from cannabis and synthetic cannabinoids—a systematic review. Brain Sci. (2018) 8:40. doi: 10.3390/brainsci8030040
17. Iseger TA, Bossong MG. A systematic review of the antipsychotic properties of cannabidiol in humans. Schizophr Res. (2015) 162:153–61. doi: 10.1016/j.schres.2015.01.033
18. Cohen K, Kapitány-Fövény M, Mama Y, Arieli M, Rosca P, Demetrovics Z, et al. The effects of synthetic cannabinoids on executive function. Psychopharmacology (2017) 234:1121–34. doi: 10.1007/s00213-017-4546-4
19. Loeffler G, Delaney E, Hann M. International trends in spice use: prevalence, motivation for use, relationship to other substances, and perception of use and safety for synthetic cannabinoids. Brain Res Bull. (2016) 126:8–28. doi: 10.1016/j.brainresbull.2016.04.013
20. Winstock AR, Barratt MJ. Synthetic cannabis: a comparison of patterns of use and effect profile with natural cannabis in a large global sample. Drug Alcohol Depend. (2013) 131:106–11. doi: 10.1016/j.drugalcdep.2012.12.011
21. Weaver MF, Hopper JA, Gunderson EW. Designer drugs 2015: assessment and management. Addict Sci Clin Pract. (2015) 10:10–19. doi: 10.1186/s13722-015-0024-7
22. Egan KL, Suerken CK, Reboussin BA, Spangler J, Wagoner KG, Sutfin EL, et al. K2 and Spice use among a cohort of college students in southeast region of the USA. Am J Drug Alcohol Abuse (2015) 41:317–22. doi: 10.3109/00952990.2015.1043438
23. Palamar JJ, Acosta P. Synthetic cannabinoid use in a nationally representative sample of US high school seniors. Drug Alcohol Depend. (2015) 149:194–202. doi: 10.1016/j.drugalcdep.2015.01.044
24. Cottencin O, Rolland B, Karila L. New designer drugs (synthetic cannabinoids and synthetic cathinones): review of literature. Curr Pharm Design (2013) 20:4106–11. doi: 10.2174/13816128113199990622
25. Di Forti M, Marconi A, Carra E, Fraietta S, Trotta A, Bonomo M, et al. Proportion of patients in south London with first-episode psychosis attributable to use of high potency cannabis: a case-control study. Lancet Psychiatry (2015) 2:233–8. doi: 10.1016/S2215-0366(14)00117-5
26. Fattore L, Fratta W. Beyond THC: the new generation of cannabinoid designer drugs. Front Behav Neurosci. (2011) 5:60. doi: 10.3389/fnbeh.2011.00060
27. van Amsterdam J, Brunt T, van den Brink W. The adverse health effects of synthetic cannabinoids with emphasis on psychosis-like effects. J Psychopharmacol. (2015) 29:254–63. doi: 10.1177/0269881114565142
28. Hermanns-Clausen M, Kneisel S, Szabo B, Auwärter V. Acute toxicity due to the confirmed consumption of synthetic cannabinoids: clinical and laboratory findings. Addiction (2013) 108:534–44. doi: 10.1111/j.1360-0443.2012.04078.x
29. Seely KA, Lapoint J, Moran JH, Fattore L. Spice drugs are more than harmless herbal blends: a review of the pharmacology and toxicology of synthetic cannabinoids. Progr NeuroPsychopharmacol Biol Psychiatry (2012) 39:234–43. doi: 10.1016/j.pnpbp.2012.04.017
30. Morrison PD, Zois V, McKeown DA, Lee TD, Holt DW, Powell JF, et al. The acute effects of synthetic intravenous Δ9-tetrahydrocannabinol on psychosis, mood and cognitive functioning. Psychol Med. (2009) 39:1607–16. doi: 10.1017/S0033291709005522
31. Fattore L. Synthetic cannabinoids—further evidence supporting the relationship between cannabinoids and psychosis. Biol Psychiatry (2016) 79:539–48. doi: 10.1016/j.biopsych.2016.02.001
32. Lorenzetti V, Solowij N, Yücel M. The role of cannabinoids on neuroanatomical alterations in cannabis users. Biol Psychiatry (2015) 79:17–31. doi: 10.1016/j.biopsych.2015.11.013
33. Castellanos D, Singh S, Thornton G, Avila M, Moreno A. Synthetic cannabinoid use: a case series of adolescents. J Adolesc Health (2011) 49:347–9. doi: 10.1016/j.jadohealth.2011.08.002
34. Crippa JA, Zuardi AW, Martín-Santos R, Bhattacharyya S, Atakan Z, McGuire P, et al. Cannabis and anxiety: a critical review of the evidence. Hum Psychopharmacol. (2009) 24:515–23. doi: 10.1002/hup.1048
35. Hill KP. Cannabis use and risk for substance use disorders and mood or anxiety disorders. JAMA (2017) 317:1070–1. doi: 10.1001/jama.2016.19706
36. Weinstock LM, Gaudiano BA, Wenze SJ, Epstein-Lubow G, Miller IW. Demographic and clinical characteristics associated with comorbid cannabis use disorders (CUDs) in hospitalized patients with bipolar I disorder. Comprehens Psychiatry (2016) 65:57–62. doi: 10.1016/j.comppsych.2015.10.003
37. Degenhardt L, Hall W, Lynskey M. Exploring the association between cannabis use and depression. Addiction (2003) 98:1493–504. doi: 10.1046/j.1360-0443.2003.00437.x
38. Curran VH, Brignell C, Fletcher S, Middleton P, Henry J. Cognitive and subjective dose-response effects of acute oral Δ9-tetrahydrocannabinol (THC) in infrequent cannabis users. Psychopharmacology (2002) 164:61–70. doi: 10.1007/s00213-002-1169-0
39. Broyd SJ, van Hell HH, Beale C, Yücel M, Solowij N. Acute and chronic effects of cannabinoids on human cognition—a systematic review. Biol Psychiatry (2016) 79:557–67. doi: 10.1016/j.biopsych.2015.12.002
40. Weinstein A, Brickner O, Lerman H, Greemland M, Bloch M, Lester H, et al. Brain imaging study of the acute effects of Δ9-tetrahydrocannabinol (THC) on attention and motor coordination in regular users of marijuana. Psychopharmacology (2008) 196:119–31. doi: 10.1007/s00213-007-0940-7
41. Crean RD, Crane NA, Mason BJ. An evidence based review of acute and long-term effects of cannabis use on executive cognitive functions. J Addict Med. (2011) 5:1–8. doi: 10.1097/ADM.0b013e31820c23fa
42. Pope HG, Gruber AJ, Hudson JI, Huestis MA, Yurgelun-Todd D. Neuropsychological performance in long-term cannabis users. Arch Gen Psychiatry (2001) 58:909–15. doi: 10.1001/archpsyc.58.10.909
43. Nacca N, Vatti D, Sullivan R, Sud P, Su M, Marraffa J. The synthetic cannabinoid withdrawal syndrome. J Addict Med. (2013) 7:296–8. doi: 10.1097/ADM.0b013e31828e1881
44. Pacher P, Steffens S, Haskó G, Schindler TH, Kunos G. Cardiovascular effects of marijuana and synthetic cannabinoids: the good, the bad, and the ugly. Nat Rev Cardiol. (2018) 15:151. doi: 10.1038/nrcardio.2017.130
45. Davidson C, Opacka-Juffry J, Arevalo-Martin A, Garcia-Ovejero D, Molina-Holgado E, Molina-Holgado F. Spicing up pharmacology: a review of synthetic cannabinoids from structure to adverse events. In: Kendall D, Alexander SPH, editors. Advances in Pharmacology, Vol. 80, San Diego, CA: Elsevier (2017). p. 135–68.
46. Reece AS, Norman A, Hulse GK. Cannabis exposure as an interactive cardiovascular risk factor and accelerant of organismal ageing: a longitudinal study. BMJ Open (2016) 6:91–118. doi: 10.1136/bmjopen-2016-011891
47. Nurmedov S, Metin B, Ekmen S, Noyan O, Yilmaz O, Darcin A, et al. Thalamic and cerebellar gray matter volume reduction in synthetic cannabinoids users. Eur Addict Res. (2015) 21:315–20. doi: 10.1159/000430437
48. Zorlu N, Di Biase MA, Kalayci ÇÇ, Zalesky A, Bagci B, Oguz N, et al. Abnormal white matter integrity in synthetic cannabinoid users. Eur Neuropsychopharmacol. (2016) 26:1818–25. doi: 10.1016/j.euroneuro.2016.08.015
49. Batalla A, Bhattacharyya S, Yücel M, Fusar-Poli P, Crippa JA, Nogué S, et al. Structural and functional imaging studies in chronic cannabis users: a systematic review of adolescent and adult findings. PLoS ONE (2013) 8:e55821. doi: 10.1371/journal.pone.0055821
50. Weinstein A, Livny A, Weizman A. Brain imaging studies on the cognitive, pharmacological and neurobiological effects of cannabis in humans: evidence from studies of adult users. Curr Pharm Des. (2016) 22:6366–79. doi: 10.2174/1381612822666160822151323
51. Hayatbakhsh MR, O'Callaghan MJ, Mamun AA, Williams GM, Clavarino A, Najman JM. Cannabis use and obesity and young adults. Am J Drug Alcohol Abuse (2010) 36:350–6. doi: 10.3109/00952990.2010.500438
52. Musshoff F, Madea B, Kernbach-Wighton G, Bicker W, Kneisel S, Hutter M, et al. Driving under the influence of synthetic cannabinoids (“Spice”): a case series. Int J Legal Med. (2014) 128:59–64. doi: 10.1007/s00414-013-0864-1
53. Yeakel JK, Logan BK. Blood synthetic cannabinoid concentrations in cases of suspected impaired driving. J Analyt Toxicol. (2013) 37:547–51. doi: 10.1093/jat/bkt065
54. Tuv SS, Krabseth H, Karinen R, Olsen KM, Øiestad EL, Vindenes V. Prevalence of synthetic cannabinoids in blood samples from Norwegian drivers suspected of impaired driving during a seven weeks period. Accid Anal Prev. (2014) 62:26–31. doi: 10.1016/j.aap.2013.09.009
55. Tetrault JM, Crothers K, Moore BA, Mehra R, Concato J, Fiellin DA. Effects of marijuana smoking on pulmonary function and respiratory complications: a systematic review. Arch Intern Med. (2007) 167:221–8. doi: 10.1001/archinte.167.3.221
56. Hall W, Solowij N. Adverse effects of cannabis. Lancet (1998) 352:1611–6. doi: 10.1016/S0140-6736(98)05021-1
57. Waugh J, Najafi J, Hawkins L, Hill SL, Eddleston M, Vale JA, et al. Epidemiology and clinical features of toxicity following recreational use of synthetic cannabinoid receptor agonists: a report from the United Kingdom National Poisons Information Service. Clin Toxicol. (2016) 54:512–8. doi: 10.3109/15563650.2016.1171329
58. Loeffler G, Hurst D, Penn A, Yung K. Spice, bath salts, and the US military: the emergence of synthetic cannabinoid receptor agonists and cathinones in the US Armed Forces. Mil Med. (2012) 177:1041–8. doi: 10.7205/MILMED-D-12-00180
59. Brewer TL, Collins M. A review of clinical manifestations in adolescent and young adults after use of synthetic cannabinoids. J Spec Pediatr Nurs. (2014) 19:119–26. doi: 10.1111/jspn.12057
60. Huffman JW, Bushell SM, Miller JRA, Wiley JL, Martin BR. 1-Methoxy-, 1-deoxy-11-hydroxy-and 11-hydroxy-1-methoxy-Δ 8-tetrahydrocannabinols: new selective ligands for the CB 2 receptor. Bioorg Med Chem. (2002) 10:4119–29. doi: 10.1016/S0968-0896(02)00331-0
61. Rhee MH, Vogel Z, Barg J, Bayewitch M, Levy R, Hanuš L, et al. Cannabinol derivatives: binding to cannabinoid receptors and inhibition of adenylylcyclase. J Med Chem. (1997) 40:3228–33. doi: 10.1021/jm970126f
62. Mechoulam R, Peters M, Murillo-Rodriguez E, Hanus LO. Cannabidiol–recent advances. Chem Biodivers. (2007) 4:1678–92. doi: 10.1002/cbdv.200790147
63. Leweke FM, Schneider U, Radwan M, Schmidt E, Emrich HM. Different effects of nabilone and cannabidiol on binocular depth inversion in man. Pharmacol Biochem Behav. (2000) 66:175–81. doi: 10.1016/S0091-3057(00)00201-X
64. Englund A, Morrison PD, Nottage J, Hague D, Kane F, Bonaccorso S, et al. Cannabidiol inhibits THC-elicited paranoid symptoms and hippocampal-dependent memory impairment. J Psychopharmacol. (2013) 27:19–27. doi: 10.1177/0269881112460109
65. Bhattacharyya S, Crippa JA, Allen P, Martin-Santos R, Borgwardt S, Fusar-Poli P, et al. Induction of psychosis byδ9-tetrahydrocannabinol reflects modulation of prefrontal and striatal function during attentional salience processing. Arch Gen Psychiatry (2012) 69:27–36. doi: 10.1001/archgenpsychiatry.2011.161
66. Wiley JL, Marusich JA, Huffman JW. Moving around the molecule: relationship between chemical structure and in vivo activity of synthetic cannabinoids. Life Sci. (2014) 97:55–63. doi: 10.1016/j.lfs.2013.09.011
67. Pertwee RG. Ligands that target cannabinoid receptors in the brain: from THC to anandamide and beyond. Addict Biol. (2008) 13:147–59. doi: 10.1111/j.1369-1600.2008.00108.x
68. Zuba D, Byrska B. Prevalence and co-existence of active components of “legal highs.” Drug Test Anal. (2013) 5:420–9. doi: 10.1002/dta.1365
69. Hudson S, Ramsey J. The emergence and analysis of synthetic cannabinoids. Drug Test Anal. (2011) 3:466–78. doi: 10.1002/dta.268
70. Uchiyama N, Kikura-Hanajiri R, Ogata J, Goda Y. Chemical analysis of synthetic cannabinoids as designer drugs in herbal products. Forens Sci Int. (2010) 198:31–8. doi: 10.1016/j.forsciint.2010.01.004
71. Hill MN, Gorzalka BB. Pharmacological enhancement of cannabinoid CB1 receptor activity elicits an antidepressant-like response in the rat forced swim test. Eur Neuropsychopharmacol. (2005) 15:593–99. doi: 10.1016/j.euroneuro.2005.03.003
72. Shohami E, Novikov M, Bass R. Long-term effect of HU-211, a novel non-competitive NMDA antagonist, on motor and memory functions after closed head injury in the rat. Brain Res. (1995) 674:55–62. doi: 10.1016/0006-8993(94)01433-I
73. Lewis IH, Campbell DN, Barrowcliffe MP. Effect of nabilone on nausea and vomiting after total abdominal hysterectomy. Br J Anaesth. (1994) 73:244–6. doi: 10.1093/bja/73.2.244
74. John Redmond W, Goffaux P, Potvin S, Marchand S. Analgesic and antihyperalgesic effects of nabilone on experimental heat pain. Curr Med Res Opin. (2008) 24:1017–24. doi: 10.1185/030079908X280635
75. Asmaro D, Carolan PL, Liotti M. Electrophysiological evidence of early attentional bias to drug-related pictures in chronic cannabis users. Addict Behav. (2014) 39:114–21. doi: 10.1016/j.addbeh.2013.09.012
76. Bolla KI, Eldreth DA, Matochik JA, Cadet JL. Neural substrates of faulty decision-making in abstinent marijuana users. Neuroimage (2005) 26:480–92. doi: 10.1016/j.neuroimage.2005.02.012
77. D'Souza DC, Perry E, MacDougall L, Ammerman Y, Cooper T, Wu Y, et al. The psychotomimetic effects of intravenous delta-9-tetrahydrocannabinol in healthy individuals: implications for psychosis. Neuropsychopharmacology (2004) 29:1558–72. doi: 10.1038/sj.npp.1300496
78. Gorelick DA, Goodwin RS, Schwilke E, Schwope DM, Darwin WD, Kelly DL, et al. Tolerance to effects of high-dose oral Δ9-tetrahydrocannabinol and plasma cannabinoid concentrations in male daily cannabis smokers. J Analyt Toxicol. (2013) 37:11–6. doi: 10.1093/jat/bks081
79. Burns JK. Pathways from cannabis to psychosis: a review of the evidence. Front Psychiatry (2012) 4:128. doi: 10.3389/fpsyt.2013.00128
80. Wilson B, Tavakoli H, DeCecchis D, Mahadev V. Synthetic cannabinoids, synthetic cathinones, and other emerging drugs of abuse. Psychiatr Ann. (2013) 43:558–64. doi: 10.3928/00485713-20131206-08
81. Li MC, Brady JE, DiMaggio CJ, Lusardi AR, Tzong KY, Li G. Marijuana use and motor vehicle crashes. Epidemiol Rev. (2011) 34:65–72. doi: 10.1093/epirev/mxr017
82. Ramaekers JG, Kauert G, Theunissen EL, Toennes SW, Moeller MR. Neurocognitive performance during acute THC intoxication in heavy and occasional cannabis users. J Psychopharmacol. (2009) 23:266–77. doi: 10.1177/0269881108092393
83. Fantegrossi WE, Moran JH, Radominska-Pandya A, Prather PL. Distinct pharmacology and metabolism of K2 synthetic cannabinoids compared to Δ 9-THC: mechanism underlying greater toxicity? Life Sci. (2014) 97:45–54. doi: 10.1016/j.lfs.2013.09.017
84. Papanti D, Schifano F, Botteon G, Bertossi F, Mannix J, Vidoni D, et al. “Spiceophrenia”: a systematic overview of “Spice”-related psychopathological issues and a case report. Hum Psychopharmacol. (2013) 28:379–89. doi: 10.1002/hup.2312
85. Bossong M, Jager G, Bhattacharyya S, Allen P. Acute and non-acute effects of cannabis on human memory function: a critical review of neuroimaging studies. Curr Pharm Des. (2014) 20:2114–25 doi: 10.2174/13816128113199990436.
86. Solowij N, Battisti R. The chronic effects of cannabis on memory in humans: a review. Curr Drug Abuse Rev. (2008) 1:81–98. doi: 10.2174/1874473710801010081
87. Altintas M, Inanc L, Oruc GA, Arpacioglu S, Gulec H. Clinical characteristics of synthetic cannabinoid-induced psychosis in relation to schizophrenia: a single-center cross-sectional analysis of concurrently hospitalized patients. Neuropsychiatr Dis Treat. (2016) 12:1893. doi: 10.2147/NDT.S107622
88. Bambico FR, Nguyen NT, Katz N, Gobbi G. Chronic exposure to cannabinoids during adolescence but not during adulthood impairs emotional behaviour and monoaminergic neurotransmission. Neurobiol Dis. (2010) 37:641–55. doi: 10.1016/j.nbd.2009.11.020
89. Renard J, Krebs MO, Jay TM, Le Pen G. Long-term cognitive impairments induced by chronic cannabinoid exposure during adolescence in rats: a strain comparison. Psychopharmacology (2013) 225:781–90. doi: 10.1007/s00213-012-2865-z
90. Jouanjus E, Raymond V, Lapeyre-Mestre M, Wolff V. What is the current knowledge about the cardiovascular risk for users of cannabis-based products? A systematic review. Curr Atheroscl Rep. (2017) 19:1–15. doi: 10.1007/s11883-017-0663-0
91. Aryana A, Williams MA. Marijuana as a trigger of cardiovascular events: speculation or scientific certainty? Int J Cardiol. (2007) 118:141–4. doi: 10.1016/j.ijcard.2006.08.001
92. Barceló B, Pichini S, López-Corominas V, Gomila I, Yates C, Busardò FP, et al. Acute intoxication caused by synthetic cannabinoids 5F-ADB and MMB-2201: a case series. Forensic Sci Int. (2017) 273:10–14. doi: 10.1016/j.forsciint.2017.01.020
93. McIlroy G, Ford L, Khan JM. Acute myocardial infarction, associated with the use of a synthetic adamantyl-cannabinoid: a case report. BMC Pharmacol Toxicol. (2016) 17:1–4. doi: 10.1186/s40360-016-0045-1
94. Radhakrishnan R, Wilkinson ST, D'Souza DC. Gone to pot-a review of the association between cannabis and psychosis. Front Psychiatry (2014) 5:50–4. doi: 10.3389/fpsyt.2014.00054
95. Bioque M, Cabrera B, García-Bueno B, Mac-Dowell KS, Torrent C, Saiz PA, et al. Dysregulated peripheral endocannabinoid system signaling is associated with cognitive deficits in first-episode psychosis. J Psychiatr Res. (2016) 75:14–21. doi: 10.1016/j.jpsychires.2016.01.002
96. Bloomfield MAP, Morgan CJA, Egerton A, Kapur S, Curran HV, Howes OD. Dopaminergic function in cannabis users and its relationship to cannabis-induced psychotic symptoms. Biol Psychiatry (2014) 75:470–8. doi: 10.1016/j.biopsych.2013.05.027
97. D'Souza DC, Sewell RA, Ranganathan M. Cannabis and psychosis/schizophrenia: human studies. Eur Arch Psychiatry Clin Neurosci. (2009) 259:413–31. doi: 10.1007/s00406-009-0024-2
98. Fergusson DM, Poulton R, Smith PF, Boden JM. Cannabis and psychosis. BMJ (2006) 332:172–5. doi: 10.1136/bmj.332.7534.172
99. Khan M, Pace L, Truong A, Gordon M, Moukaddam N. Catatonia secondary to synthetic cannabinoid use in two patients with no previous psychosis. Am J Addict. (2016) 25:25–7. doi: 10.1111/ajad.12318
100. Vallersnes OM, Dines AM, Wood DM, Yates C, Heyerdahl F, Hovda KE, et al. Psychosis associated with acute recreational drug toxicity: a European case series. BMC Psychiatry (2016) 16:293. doi: 10.1186/s12888-016-1002-7
101. Yücel M, Solowij N, Respondek C, Whittle S, Fornito A, Pantelis C, et al. Regional brain abnormalities associated with long-term heavy cannabis use. Arch Gen Psychiatry (2008) 65:694–701. doi: 10.1001/archpsyc.65.6.694
102. Bloomfield MAP, Ashok AH, Volkow ND, Howes OD. The effects of Δ9-tetrahydrocannabinol on the dopamine system. Nature (2016) 539:369–77. doi: 10.1038/nature20153
103. Rominger A, Cumming P, Xiong G, Koller G, Förster S, Zwergal A, et al. Effects of acute detoxification of the herbal blend “Spice Gold”on dopamine D 2/3 receptor availability: a [18 F] fallypride PET study. Eur Neuropsychopharmacol. (2013) 23:1606–10. doi: 10.1016/j.euroneuro.2013.01.009
104. Mir A, Obafemi A, Young A, Kane C. Myocardial infarction associated with use of the synthetic cannabinoid K2. Pediatrics (2011) 128:e1622–7. doi: 10.1542/peds.2010-3823
105. Schaefer N, Peters B, Bregel D, Kneisel S, Auwärter V, Schmidt PH, et al. A fatal case involving several synthetic cannabinoids. Toxichem Krimtech (2013) 80:248–51.
106. Keyes KM, Rutherford C, Hamilton A, Palamar JJ. Age, period, and cohort effects in synthetic cannabinoid use among US adolescents, 2011–2015. Drug Alcohol Depend. (2016) 166:159–67. doi: 10.1016/j.drugalcdep.2016.07.018
107. Tait RJ, Caldicott D, Mountain D, Hill SL, Lenton S. A systematic review of adverse events arising from the use of synthetic cannabinoids and their associated treatment. Clin Toxicol. (2016) 54:1–13. doi: 10.3109/15563650.2015.1110590
108. Bilgrei OR. From “herbal highs” to the “heroin of cannabis”: exploring the evolving discourse on synthetic cannabinoid use in a Norwegian Internet drug forum. Int J Drug Policy (2016) 29:1–8. doi: 10.1016/j.drugpo.2016.01.011
109. Corazza O, Schifano F, Simonato P, Fergus S, Assi S, Stair J, et al. Phenomenon of new drugs on the Internet: the case of ketamine derivative methoxetamine. Hum Psychopharmacol. (2012) 27:145–9. doi: 10.1002/hup.1242
110. Corazza O, Assi S, Trincas G, Simonato P, Corkery J, Deluca P, et al. Novel Drugs, Novel Solutions: exploring the potentials of web-assistance and multimedia approaches for the prevention of drug abuse. Italian J Addict. (2011) 1:25–30.
Keywords: cannabis, synthetic cannabinoids, novel psychoactive drugs, drug abuse, addiction
Citation: Cohen K and Weinstein AM (2018) Synthetic and Non-synthetic Cannabinoid Drugs and Their Adverse Effects-A Review From Public Health Prospective. Front. Public Health 6:162. doi: 10.3389/fpubh.2018.00162
Received: 09 January 2018; Accepted: 14 May 2018;
Published: 07 June 2018.
Edited by:
Blair Henry, Sunnybrook Health Science Centre, CanadaReviewed by:
Mandakini Sadhir, University of Kentucky, United StatesDaniel Rossignol, Rossignol Medical Center, United States
Copyright © 2018 Cohen and Weinstein. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Aviv M. Weinstein, YXZpdndlaW5zdGVpbkB5YWhvby5jb20=
 Koby Cohen
Koby Cohen Aviv M. Weinstein
Aviv M. Weinstein