
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Psychiatry , 27 April 2023
Sec. Mood Disorders
Volume 14 - 2023 | https://doi.org/10.3389/fpsyt.2023.1083244
This article is part of the Research Topic The Neurobiology of Suicide: The 'Suicidal Brain' View all 11 articles
Suicide is a leading cause of death in the United States. Historically, scientific inquiry has focused on psychological theory. However, more recent studies have started to shed light on complex biosignatures using MRI techniques, including task-based and resting-state functional MRI, brain morphometry, and diffusion tensor imaging. Here, we review recent research across these modalities, with a focus on participants with depression and Suicidal Thoughts and Behavior (STB). A PubMed search identified 149 articles specific to our population of study, and this was further refined to rule out more diffuse pathologies such as psychotic disorders and organic brain injury and illness. This left 69 articles which are reviewed in the current study. The collated articles reviewed point to a complex impairment showing atypical functional activation in areas associated with perception of reward, social/affective stimuli, top-down control, and reward-based learning. This is broadly supported by the atypical morphometric and diffusion-weighted alterations and, most significantly, in the network-based resting-state functional connectivity data that extrapolates network functions from well validated psychological paradigms using functional MRI analysis. We see an emerging picture of cognitive dysfunction evident in task-based and resting state fMRI and network neuroscience studies, likely preceded by structural changes best demonstrated in morphometric and diffusion-weighted studies. We propose a clinically-oriented chronology of the diathesis-stress model of suicide and link other areas of research that may be useful to the practicing clinician, while helping to advance the translational study of the neurobiology of suicide.
The World Health Organization (WHO) reports that suicide is the second leading cause of death among individuals aged 15–29 years (1) with an estimate of approximately 800,000 people dying from suicide each year - a global mortality rate of one person every 40 s. In the United States, the Center for Disease Control and Prevention (CDC) reports Suicide is the 12th leading cause of death for both Hispanic and non-Hispanic people of all races (2). In 2020, suicide was the second leading cause of death for children (age 10–14 years, accounting for 581 deaths) and third leading cause of death for young individuals (age 15–24 years, accounting for 6,062 deaths) (3). Suicide research has been very important in developing clinical suicide risk assessments (4). However, recent neuroimaging work with suicidal patients holds significant promise for the clinician to directly access information without it being filtered through the situation, suspicion or in fact impaired processes a suicidal individual may use to disclose the risk assessment information. This has been accelerated by parallel theories in neurobiology and cognitive neuroscience (5–7) and a clinical focus from the likes of McGirr (8) and Turecki (9). Recent reviews (10, 11) have also contributed to bridging this translational gap. In spite of this, there still is not a clinically-accessible link between witnessed patient histories, symptoms, and deficits and what is rapidly being discovered in the fields of functional, network, and morphometric neurobiology.
Multiple general theories have been developed in an attempt to understand the risk factors, thoughts, distortions, cognitive/behavioral differences, atypical brain structures, systems and functions that distinguish suicidal individuals from those without past attempts, or a high risk. As clearly laid out in Van Heeringen’s book “the Neuroscience of Suicidal Behavior,” (6) five main neurobiological theories precede the recent work of Schmaal, Auerbach, and others. These include the “Cry of Pain” model (12), the Interpersonal model (7), the Integrated Motivational model (13), the Clinical Stress-Diathesis model (8, 14), and Jollant’s Neurocognitive model (5). The most relevant to this paper is Van Heeringen’s Neurobiological model (6). This model synthesizes Molecular, Morphological, Cognitive, and Functional evidence into a theory based on predictive coding, also known as computational psychology. This model suggests that the main difference between suicidal individuals and those that are not suicidal, have an impaired system of evaluating old beliefs, evaluating the importance and certainty (precision) of new information, and appropriately changing current beliefs and strategies accordingly. Author states that learning is directly affected, proposing that humans are generally biased toward positive valenced stimuli and predictions. However, those with abnormal serotonin systems, which play a role in learning and extinction of behaviors that lead to aversive events, may be biased toward learning more negatively valenced behaviors (called Pavlovian instrumental transfer). Author further proposes that belief updating (modifying old beliefs based on new sensory information) is dependent on the right inferior frontal gyrus (IFG) and bilateral superior frontal gyrus (SFG) for positive valenced information, and on the left IFG and right inferior parietal lobule (IPL) for negative information. Lastly, as with disrupted serotonin and NMDA systems, blunted cortisol reactivity to stress creates a founding diathesis and may account for the higher rates of suicidality amongst individuals with trauma and adverse childhood events.
Our goal is to consider the current body of neuroimaging research on STBs (defined here as suicidal ideation and attempts) among those with Major Depressive Disorder (MDD) (15). Our goal is to clarify relevant concepts for the clinicians working with these patients and to propose the framework of a testable timeline of the suicidal brain in this population that may be developed into a clinical tool.
A search on functional, structural, and diffusion-weighted MRI studies of the suicidal brain was performed in the search engine ‘PubMed’ for both original and review research articles that were published before December 2021. The literature search was conducted using the following terms in the title:
((BOLD[Title]) OR (fMRI[Title]) OR (functional MRI[Title]) OR (functional magnetic resonance imaging[Title]) OR (morphometry[Title]) OR (thickness[Title]) OR (surface area[Title]) OR (volume[Title]) OR (gyrification[Title]) OR (folding[Title]) OR (MRI[Title]) OR (DTI[Title]) OR (magnetic resonance imaging[Title]) OR (diffusion tensor imaging[Title]) OR (brain connectivity[Title]) OR (connectivity[Title]) OR (brain activation[Title]) OR (neural activation[Title]) OR (neuroimaging[Title]) OR (spectroscopy[Title]) OR (white-matter[Title]) OR (gray-matter[Title]) OR (neural correlates[Title]) OR (neural representations[Title])) AND ((suicide[Title]) OR (suicidal[Title]) OR (suicidality[Title]) OR (suicide risk[Title]) OR (self-harm[Title]) OR (suicidal ideation[Title])) NOT ((Inflammation[Title]) OR (Inflammatory[Title]) OR (Plasma[Title]) OR (Tumor[Title]) OR (Immunity[Title]) OR (habenula[Title]) OR (mRNA[Title]) OR (RNA[Title]) OR (DNA[Title]) OR (Gene[Title]) OR (Genetics[Title]) OR (Glucose[Title]) OR (Brain expression[Title]) OR (Pain[Title])). The search was performed without a time limit. This resulted in a total of 149 articles. The articles were further filtered through an inspection of the abstracts. A total of 80 articles that included studies focusing on patients with clinical conditions other than MDD, studies focusing on non-suicidal self-injury, and studies relating to neuroimaging modalities other than MRI (i.e., MEG, PET) were excluded.
A total of 69 research articles, focusing on STBs associated with depression, were identified, and reviewed in the current study (Tables 1–4).
The task-based fMRI research in STBs depends on the ability of a participant to complete a task in real time, and thus, may be a more proximally clinically important difference amongst patients. Task-based fMRI studies of the suicidal brain have focused on six primary tasks according to Dr. Van Heeringen: (1) Decision Making/Reward-Based Learning; (2) Emotional Pain and Affect Regulation; (3) Sensitivity to Social Stressors; (4) Cognitive Control/Response Inhibition; (5) Hopelessness; and (6) Impulsivity and Aggression. Unfortunately, within these 6 domains, only the first 4 have functional neuroimaging data consistent with our focused limitations.
Examining the fMRI activation differences during a learning task which requires participants to maximize rewards earned through a sequence of lever pulls (the Three-Armed Bandit Task), Brown et al. found that while non-suicidal participants showed decreased impulsivity with increasing connectivity between the ventromedial prefrontal cortex (vmPFC) and parietal cortex, suicidal participants had increasing impulsivity with increased connectivity between these regions (16).
Suicide attempts have been found to be associated with greater activation in the right lateral orbitofrontal cortex (OFC) and decreased activation in the right superior frontal gyrus (SFG) while performing decision-making tasks in response to prototypical angry versus neutral faces (17).
In a recent meta-analyses that included work on decision making/reward-based learning, Li et al. concluded that brain activation in suicide attempters increased in the left insula but decreased in the bilateral fusiform gyrus compared to non-attempters across multiple learning-based fMRI tasks (18).
As a proximal and distal risk factor for suicide, social exclusion is of immediate importance to the suicidal patient. This is consistent with the concepts of “thwarted belongingness” in Joiner/Van Orden’s interpersonal theory of suicide, shining light on the vital importance of social support amongst persons at high risk for suicide.
Two fMRI studies (19, 20) have used the Cyberball task (where the participant is progressively excluded from a game) to examine social exclusion in suicidal populations. Both found a decreased activation in the anterior cingulate cortex (ACC) in their higher suicide risk groups. Specifically, Olie et al. found that suicide attempters displayed decreased activation in the left insula (as well as supramarginal gyrus) when compared to patients without any history of suicide attempt and healthy controls (19). Caceda et al. (20) found that the activation of the anterior insula during inclusion trials in suicide attempters was significantly decreased compared to depressed patients with and without suicidal ideation.
Emotion regulation is clinically relevant to multiple conditions, from self-injurious behavior to PTSD. Supportive of the clinical impression that emotion regulation is important in STBs, Miller et al. found that adolescents with suicidal ideation (SUI) showed increased activation in the dorsolateral prefrontal cortex (dlPFC) compared to healthy controls (21).
Many studies have tried to examine the connection between the structures involved in affective processing and regions of interest in the suicidal brain. Clinically, this may be consistent with the concept of a cognitive distortion and related to such possible risks as low self-esteem, isolation, and unwillingness to seek help, though a comparison of the neurobiological changes associated with Cognitive Therapy is beyond the scope of this review.
In a study by Pan et al. participants made a gender selection for images of faces with or without affective valences (22). Consistent with the idea of a sensitivity to social stressors, adolescents with past suicide attempts showed an increased activation in ACC, dlPFC, sensory cortex, and temporal cortex during angry trials but a decreased activation in the same areas during neutral or happy face trials.
Ai et al. similarly utilized a gender discrimination task with individuals with past suicide attempts. Authors found that they had lower activation within the fusiform gyrus during emotional face processing across all stimulus types: happy, angry, sad, and scared (23).
Two major studies mixed resting state data with task-based functional tasks. Alarcon et al. found that depressed participants who had attempted suicide or had high suicidal ideation showed greater functional connectivity between the amygdala, dlPFC, dorsomedial prefrontal cortex (dmPFC), and precuneus compared to controls completing an emotional self-face recognition task where they considered if the faces looked like them, with valence varying between happy, sad, and neutral (24). Malhi et al. examined resting state functional connectivity (RSFC) in suicidal adults using an emotional face-word stroop paradigm in which congruent and incongruent images were flanked by the word “happy” or “sad.” During negative valenced incongruent trials, depressed participants (with and without suicidal thoughts and behaviors) had increased activity relative to healthy controls in the prefrontal cortex, frontopolar cortex, ACC, and posterior parietal cortex. At the same time, participants with STB’s showed increased activity among basal ganglia structures but decreased activity among the Medial Frontoparietal Network/Default Mode Network (M-FPN/DMN) structures with increasing suicide risk on measures, including the Columbia-Suicide Severity Rating Scale (C-SSRS) (25).
A recent machine learning study by Just et al. supported the involvement of structures important to all these processes among participants with suicidal ideation. When presented with affectively valenced verbal stimuli, specifically the words death, cruelty, trouble, carefree, good, and praise, group differences in activation in the medial superior frontal cortex, inferior parietal, medial temporal, ACC, and inferior frontal gyrus (IFG) were found (26).
Impairments in top-down cognitive control and response inhibition have clear clinical relevance to suicide but have been studied very little in task-based fMRI research in this population. However, Richard-Devantoy et al. used a Go-No-Go task and found that deficits in cognitive inhibition (in relation to the IFG, thalamus, OFC, and parietal cortex) were related to the depressive, but not specifically, STB vulnerability risk (27). In contrast, the meta-analysis involving neuroimaging studies using the monetary incentive delay task and the stop signal task with over 5,000 participants aged 9–11 could not delineate between those with suicidal ideation from those with suicidal behaviors (28). Although our belief is that there is a difference in this domain between those with suicidal ideation and those who attempt, evidence is scant and will depend on future work of our own and others.
While performance on fMRI tasks and task-based connectivity studies can show abnormal/atypical recruitment of structures theorized to be essential in relevant cognitive tasks, resting-state fMRI (rsfMRI) studies allow a view of default self-referential thought processes while the participant is not engaged in a specific task. It, therefore, is used to analyze which relevant systems have robust or weakened ‘default’ connections or communications (29–31) within and between networks, defined as the level of increased, decreased, or mixed functional connectivity between them, or RSFC. Each defined network is organized based on theoretical common functions. In the interest of clarity, we will use Uddin et al.’s (32) definitions of major networks as a reference for the resting-state studies in our review, as they clarify the involved neuroanatomy and are analogous to established networks familiar in the research domain.
Uddin proposes the following six main networks and their primary functions: (1) the Lateral Fronto-Parietal Network/Cognitive Control Network (L-FPN/CCN) = functions include executive functions, such as goal-oriented cognition, working-memory, inhibition, and task switching; (2) the Pericentral/Somatomotor Network (PN/Somatomotor) = functions include involvement in motor processes and somatosensory processing; (3) Occipital Network/Visual Network (ON/VN) = functions include visual processing; (4) Dorsal Frontoparietal Network/Attention Network (D-FPN/AN) = plays a broad role in visuospatial attention. The functions of this system are to prepare and apply top-down selection for stimuli and responses; (5) The Midcingulo-insular Network/Salience Network (M-CIN/SN) = has a broad role in identifying important, or salient, information. Salience processing involves the detection of behaviorally-relevant environmental stimuli and may include internally-generated (i.e., remembered) information; and the (6) M-FPN/DMN = functions likely involve formation, temporal binding, and dynamic reconfiguration of associative representations based on current goal-states, detecting the associative relevance of internal and external stimuli, and providing value coding and elaboration to perceived events. Other accounts suggest M-FPN function accommodates predictive coding, semantic associations, and plays a role continuously monitoring the environment.
Though abnormal RSFC can be complicated, ranging from nodal (i.e., region to region) to whole brain (as it sounds) analysis and involving established networks or networks defined within each individual study, it may become increasingly relevant to clinical practice. One limitation may be the inherent assumption of the specific functions of each interconnected area. We present RSFC studies grouped according to main findings. The most consistent findings among suicidal participants seem to occur between the M-FPN (default network/DMN) and the M-CIN (salience network/SN). Many studies showed decreased RSFC between M-FPN/DMN and M-CIN/SN. When we talk about connections between networks, we clearly mean connections between the regions or nodes that comprise networks. In other words, resting state or intrinsic brain networks in fMRI are best thought of as a collection of regions that show correlations in terms of their fluctuating activity.
Qiu et al. (33) looked at depressed participants with a history of suicide attempts compared to those without and found that in both groups, as suicidal ideation increased, functional connectivity decreased between the pregenual anterior cingulate cortex (pgACC part of M-CIN/SN) and the superior frontal gyrus (M-FPN/DMN). Among depressed suicidal adults, Du et al. (34) found that the SUI group exhibited decreased functional connectivity between the right ACC (M-CIN/SN), the orbito-medial prefrontal cortex (M-FPN/DMN), and the right middle temporal pole (within the D-FPN/AN) compared to non-suicidal depressed and control groups. Yang et al. (35) found that between depressed participants with past attempts and those without attempts, those with past attempts showed decreased RSFC (decreased positive correlation) in the right inferior front orbital gyrus (within the M-FPN/DMN) to the left inferior parietal lobule (within the M-CIN/SN). They also found that compared with non-attempters, those with past attempts had decreased gray matter volume (GMV) in the right inferior frontal orbital gyrus (IFOG) and left caudate (CAU) but increased GMV in the left calcarine fissure.
In a study by Stange et al. (36), suicidal participants showed decreased connectivity between the right middle frontal gyrus/SFG (L-FPN/CCN) and the M-FPN/DMN and decreased connectivity between the precuneus (L-FPN/CCN or M-FPN/DMN) and the Salience Network (SN). The difference in RSFC within areas of M-FPN/DMN was greater than that between M-FPN/DMN to M-CIN or to L-FPN/CCN.
Cao et al. (37) analyzed low frequency RSFC and found participants with STB’s had increased connectivity in the right superior temporal gyrus (STG) (M-FPN/DMN), left MTG (M-FPN/DMN), and left middle occipital gyrus (ON/VN) but decreased connectivity in the left SFG (M-FPN/DMN) and left MFG (M-FPN/DMN), at least compared to non-suicidal clinical participants. Another study by this group (38) found that participants with past attempts had decreased RSFC between the left MFG (L-FPN/CCN) and the left SPG (D-FPN/AN) compared to the non-attempt group and decreased RSFC between the left superior frontal gyrus (M-FPN/DMN) and the right ACC (M-CIN/SN).
Several studies showed an increase in connectivity between the M-FPN/DMN and M-CIN/SN. Zhang et al. (39) specifically focused on RSFC between the bilateral amygdala and whole-brain activation and found increased connectivity between the right amygdala (M-CIN/SN) and bilateral paracentral lobule/precuneus (part of M-FPN/DMN) in a suicidal behavior (suicidal ideation and/or past suicide attempt) group relative to non-suicidal and healthy-control groups.
Kang et al. (40) found mixed results, with suicide attempters displaying significantly increased functional connectivity of the left amygdala (within the M-CIN/SN) with the right insula (within the M-CIN/SN) and the left superior orbitofrontal area (within the DMN) and increased functional connectivity of the right amygdala (within the M-CIN/SN) with the left middle temporal area (within the D-FPN/Attn). Wei et al.’s (41) study similarly found that suicidal patients with depression showed greater amygdala (within the M-CIN/SN) to precuneus/cuneus (within the M-FPN/DMN) RSFC compared with non-suicidal patients and healthy controls.
Cao et al. (42) showed that a suicidal (history of an attempt) depressed group demonstrated decreased RSFC connectivity between the anterior M-FPN/DMN and left L-FPN/CCN but increased connectivity between the L-FPN/CCN and M-CIN/SN.
The next most common findings were abnormal RSFCs between the M-FPN/DMN and L-FPN (CCN), adding to the findings of Stange, Cao and others.
An RSFC analysis of treatment changes by Shu et al. (43) showed that prior to treatment, participants showed increased baseline activity in the left posterior cerebellar lobe, right ACC (within the M-FPN/DMN), left caudate (within the L-FPN/CCN) nucleus, and left superior frontal cortex (within the M-CIN/SN). After combined treatment, patients showed increased activity in the left middle occipital cortex and left precuneus (within the L-FPN/CCN).
After this, intra-network abnormalities show up in multiple studies, focusing on the M-FPN/DMN.
Zhang et al. (44) used an independent component analysis to show that RSFC within the M-FPN/DMN was increased in the left cerebellum but decreased in the posterior cingulate cortex (PCC) and right precuneus among suicidal (SUI & STBs) versus healthy controls. Network analysis by Chen et al. (45) found increased connectivity between the frontal (M-FPN/DMN) and parietal lobes in comparison to the healthy controls.
Yang’s work (35), mentioned above, also found participants with past attempts had increased RSFC between the right IFOG and left rectus gyrus (both in M-FPN/DMN).
In a study examining trauma and suicide in adults with PTSD, increased functional connectivity between reward and control regions (primarily under the M-FPN/DMN of Uddin’s definition) was found to be positively correlated with suicidality (46).
Lee et al. (47) found that suicidal patients (past attempts) with depression had significantly increased RSFC in tracts from an anteriorly defined division of the right parahippocampal gyrus (within the M-FPN/DMN) to a posteriorly defined division of the left parahippocampus.
Schreiner et al. (48) examined suicidal adolescents and found more evidence for involvement of the precuneus/cuneus (within the M-FPN/DMN) in the suicidal mind. They showed that in suicidal participants, as suicidality increased, RSFC increased between the right precuneus (M-FPN/DMN), right IFG (M-FPN/DMN), and cerebellum; and between the left PCC (M-FPN/DMN), left cerebellum, and cingulate gyrus.
Using whole brain analysis, Kim et al. (49) proposed a subnetwork of decreased RSFC among participants with suicidal ideation consisting of the “orbitofrontal cortex (within the M-FPN/DMN), especially the left SFT (M-FPN/DMN), pars orbitalis, left MFG, and right olfactory cortex.”
Most promising may be the recent attempts to apply machine learning to one or more MRI modalities. Gosnell et al. (50) distilled five prominent RSFC patterns amongst participants with suicidal ideation or past attempts, including: (1) decreased RSFC between the rSFG (M-FPN/DMN) and insula (M-CIN/SN); (2) increased RSFC between the left habenula (possibly M-CIN/SN) and right parahippocampus (M-FPN/DMN); (3) increased connectivity between the left frontal middle orbital gyrus (M-FPN/DMN) and left rolandic operculum (L-FPN/CCN); (4) increased connectivity between the left putamen (within the FPN) and the cerebellar vermis; and (5) decreased connectivity between the amygdala (within the SN) and middle temporal pole (within the D-FPN/AN).
Dai et al. (51) used ICA (machine learning) to conclude that the relevant structures were the right inferior temporal gyrus (within the L-FPN/CCN), left IFG (within the DMN), right angular gyrus (within the L-FPN/CCN), left inferior parietal cortex (IPC) (within the L-FPN/CCN), left rolandic operculum (within the L-FPN/CCN), and right dorsolateral superior frontal gyrus (within the DMN-M-FPN).
Suicide attempt-related altered RSFC was also observed in a Graph Analytics analysis (machine learning) by Stumps et al. (52) within the M-CIN(SN), cognitive-control (L-FPN), M-FPN, and visual networks.
There were a variety of studies showing abnormal connections between other networks, including the D-FPN/Attention Network, PN/Somatomotor Network, and ON/Visual Network.
In line with the research of Du and Kang above, Chase et al. (53) found that patients with SUI (but not necessarily historical attempts) had increased connectivity between the dorsal PCC(M-FPN/DMN) and MTG (D-FPN/Attn).
Serafini et al. (54) in a review of RSFC work showed a mixed increased connectivity/decreased connectivity pattern among networks but could not distinguish findings between pathologies.
Along with abnormalities in the M-FPN/DMN above, Lee also found increased RSFC in tracks from the temporooccipital part of the right inferior temporal gyrus (within the L-FPN/CCN) to the frontal eye fields of the Dorsal Attention Network (i.e., within the D-FPN). They also found decreased RSFC between the medial frontal cortex (within the M-FPN/DMN) and the right supplementary motor cortex (within the PN/Somatomotor network).
Another way to examine the suicidal brain is through morphometric analysis. Here, we try to make it more directly accessible to the clinician. This modality measures the physical makeup of brain structures by examining measures such as cortical thickness, cortical surface area, and/or cortical volume of the relevant brain areas. The implication is that the neural systems which may be hypo/hyperactive during functional processing may have altered physical attributes. Many morphometric studies have been done to find alterations in those with STBs.
The current review identified such studies which reported associations between these morphometric measures and suicidal behavior.
One of the earlier studies addressing the association between history of suicidal attempts in elderly, depressed individuals and both cortical and sub-cortical abnormalities was attempted by Hwang et al. (55). In that study, authors used voxel-based analysis and were able to show widespread gray matter volume (GMV) reduction in the frontal (i.e., left medial, bilateral superior, right middle, right inferior, and left posterior frontal cortices), parietal (i.e., left superior, right inferior, and left lateral parietal cortices), occipital gyrus (i.e., left cuneus), left STG, and sub-cortical (i.e., bilateral insula, left lentiform nucleus, right claustrum, bilateral midbrain, bilateral culmen, and right inferior and bilateral superior semilunar lobules) brain regions in late-onset geriatric depressed individuals with a history of suicide attempts compared to those without previous attempts. Here, volume reduction was most prominent within the dmPFC, consistent with impairment in reward-based learning and top-down executive control.
Wagner et al. reported cortical thinning in the left dorsolateral, ventrolateral prefrontal, and ACC in depressed adults with documented suicidal behavior, i.e., high-risk group of suicide as compared to depressed adults with a non-high risk for suicide (56).
A study by Huber et al. specifically found white-matter volume differences in the left ACC between veterans with a history of attempting suicide relative to veterans with a history of SUI (57). Along with RSFC data previously mentioned, the work by Barredo and colleagues found that cortical thickness of the ACC/PCC was shown to predict the functional connectivity between the lateral pars orbitalis and anterior cingulate/paracingulate control regions (46).
Wang et al. specifically reported significant differences in GMV in the bilateral MFG across patients with mood disorders and suicidal behavior, patients with mood disorders without suicidal behavior, and healthy controls (58). However, they did not find significant differences between participants with SUI and those with a history of actual suicide attempts.
Ding et al. used a region-specific approach to study differences between suicide attempters with a past history of mood disorders and suicidal behavior, participants with a mood disorder but not suicidal behavior, and healthy controls with neither (59). Reduced cortical volume was observed within the left ventrolateral prefrontal cortex in suicide attempters compared to both control groups. In addition, the orbitofrontal and dorsal prefrontal cortices (but not medial prefrontal cortex) also showed reduced cortical measures in suicide attempters compared to healthy controls. This is consistent with task-based and resting-state fMRI studies showing generally both reduced top-down executive control and impairment in reward-based learning regions and networks. Structurally, there were significant GMV decreases among suicide attempters across clinical conditions compared to non-attempters. Diffusion Tensor Imaging (DTI) findings also showed significantly reduced fractional anisotropy among those with past attempts versus those without (60). In a study of future suicide attempters (i.e., individuals attempting suicide between baseline and follow-up assessment) with mood disorders, participants showed lower baseline ventral and rostral prefrontal GMV compared to those who did not attempt (61). Besides the studies examining participants with depression and/or previous suicide attempts, distinct markers that included an involvement of frontal regions, particularly reduced cortical volume within the left MFG and cortical thinning within the posterior frontal lobe including the bilateral precentral gyrus, were also found in individuals with current SUI as compared to healthy controls without even a family history of psychiatric disorders or suicide attempts (62). Interestingly, greater cortical surface area and cortical volume within the left dorsolateral prefrontal gyrus were reported to be associated with reduced SUI in a non-clinical population with mild levels of stress and perceived lack of social support (63). This would be consistent with increasing behavioral control and top-down influence on impulsivity with decreasing pathology.
In a recent study by Yang et al. authors found that suicidal depressed patients had reduced GMV in the right IFG and left caudate but increased GMV in the left calcarine fissure (35), areas associated with visual processing not irrelevant to potential affective or social stimuli.
A recent study by Kang et al. involving individuals with depression with and without a history of suicide attempts showed altered morphometry in the lateral parietal and occipital brain regions along with frontal areas. In that study, depressed patients with past suicide attempts were found to have larger surface area within the left postcentral and left lateral occipital areas and large cortical volume within the left postcentral and left lateral orbitofrontal areas, whereas smaller surface area within the left SFG was found (64). In another recent study by Harenski et al. criminal offenders with a history of suicide attempts had widespread decreased gray matter within both cortical and sub-cortical regions, including the PCC/precuneus, IPC, dorsal prefrontal cortex, amygdala, insula, superior occipital gyrus, cuneus, and cerebellum (65). Using local shape volume analysis, researchers specifically found significant volumetric differences between suicidal and non-suicidal depressed individuals in the left amygdala, left hippocampus, left putamen, bilateral pallidum, and bilateral thalamus (66). In another study however, it was only the reduced volume of the right hippocampus that was most prominent in participants with a recent history of suicide attempts within the past 2 months as compared to healthy individuals (67). This may represent limited or impaired processing of new information relative to old beliefs and memories, consistent with Van Heeringen’s framework. Interestingly, in a postmortem study, compared to healthy controls, the suicidal depressed participants had an increased neuron number in CA2/3 subregions of the hippocampus gyrus (68).
In a study of suicide attempters with a family history of suicide and personal history of violent suicide attempts, Jollant et al. found an association between family history of suicide and reduced volume within the bilateral temporal regions, right dlPFC, and left putamen, as well as between violent methods of attempt and increased bilateral caudate and left putamen volumes (69). Reduced GMV in the dorsal striatal structures, particularly bilateral putamen and caudate, were associated with greater implicit SUI observed from suicide-related outcomes from the death version of the Implicit Association Test (IAT) (70, 71).
Several studies also reported cortical alterations within the temporal and parietal lobes but not the frontal lobe.
Reduced GMV within the right STG was observed in adolescents with MDD and a history of suicide attempts compared to adolescents with MDD but without any history of suicide attempts (72), whereas care-giver reported STBs were also associated with decreased volume at the left bank of the superior temporal sulcus in children (73). In another study on adolescents, McLellan et al. reported reduced volume of the right STG in adolescents with treatment-resistant depression and a history of suicide attempts as compared to healthy adolescents (74). Compared to healthy controls, patients with MDD and a history of suicide attempts in Peng et al.’s work showed decreased GMV within the right MTG and increased GMV within the right parietal lobe (75). Authors reported decreased GMV in the left limbic cingulate gyrus for the depressed suicidal group compared to the depressed non-suicidal group. Somewhat contrary to Peng’s work, patients with an attempt history have also been shown to have decreased GMV in the left anterolateral region of the parietal lobe as well as in the right cerebellum (76).
A recent study by the ENIGMA-MDD working group of over 18,925 participants examined morphometric differences between healthy controls, depressed participants, and participants with a history of attempted suicide and found multiple differences between the groups (77). Regarding volumetric differences, the thalamus and right pallidum were significantly smaller in depressed attempters compared to depressed and healthy controls. Regarding cortical surface area, depressed attempters had smaller cortical surface area of the left cuneus, left inferior parietal, left rostral middle frontal, and right pericalcarine cortex compared with healthy controls, but only the inferior parietal cortex was clinically distinct from depressed clinical controls. Lastly, in terms of cortical thickness, although there was not a significant difference between depressed attempters and clinical controls, attempters did display significantly lower cortical thickness in the left rostral middle frontal region. The authors concluded that these findings suggested impairment in decision making, impulsivity, and planning as well as attention and the concept of self (77). However, please note that a significant difference in such large studies does not mean the effect size is clinically meaningful. In other words, a clinically insignificant volumetric difference could be statistically significant due to the large number of subjects studied. For example, in this study, for the left pallidum and right nucleus accumbens subcortical volumes, the difference between clinical and healthy controls did not reach statistical significance after correction for multiple comparisons. Therefore, statistically significant differences may not necessarily translate to clinical significance, but it can inform next steps and build a future-focused plan for translational researchers.
Most recently, Sarkinaite et al. (78) published findings that showed volumetric differences between participants with past suicide attempts and healthy controls in the frontal and temporal cortex thickness and volume of the hippocampus. Notably, with number of attempts as a covariate, participants with increasing number of past suicide attempts showed decreasing thickness of temporal cortex in the inferior middle and temporal cortex.
In Figures 1, 2, we provide an overview of brain regions (Figure 1) and networks (Figure 2) that are most commonly found to be involved in functional MRI and brain morphometry research of suicidal thoughts and behavior. Both the figures are generated through FreeSurfer 7.3.2 (82, 83) and Yeo’s 17-network atlas (84).
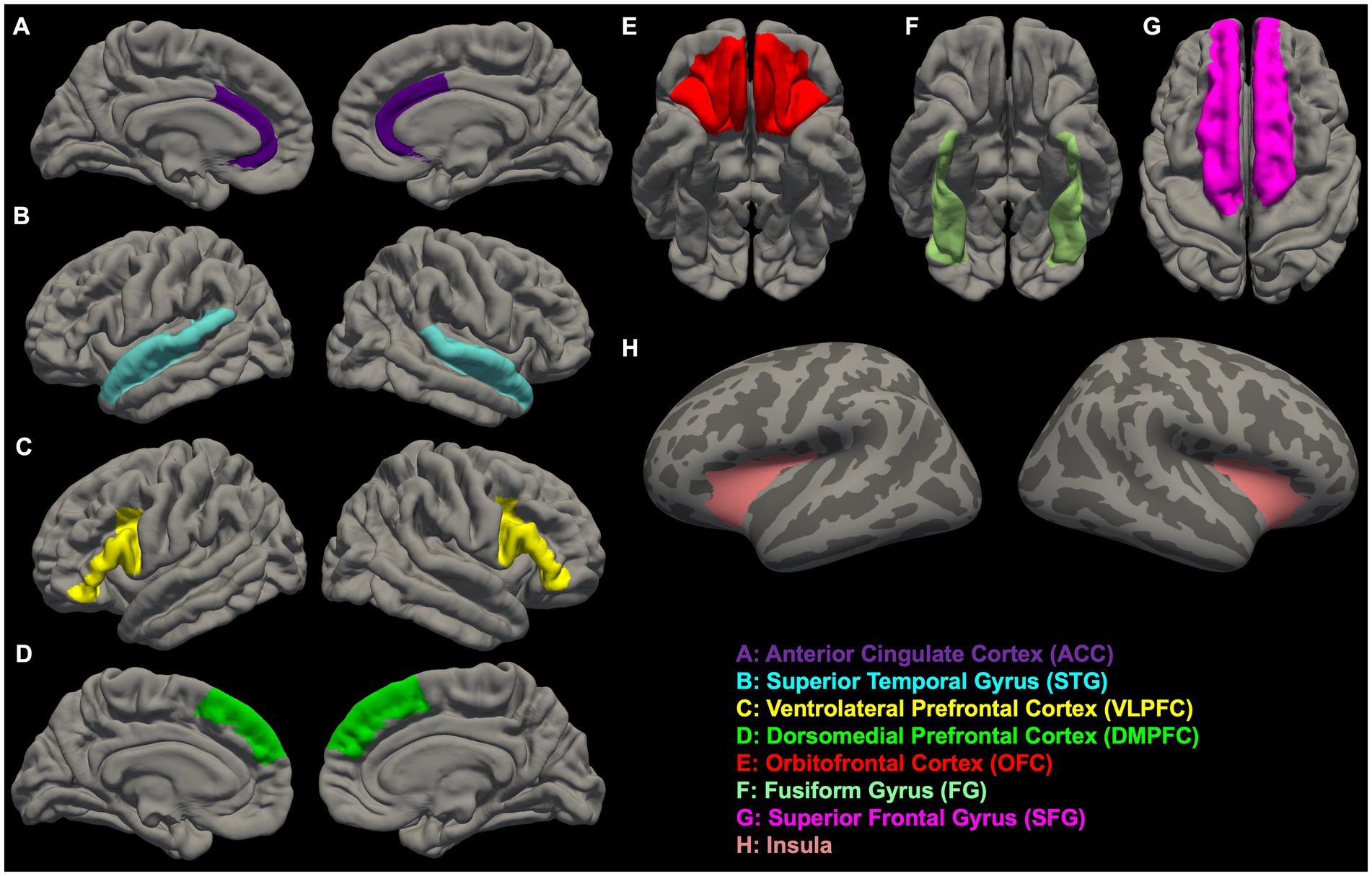
Figure 1. Overview of brain regions (A–H) that are most commonly found to be involved in functional MRI and brain morphometry research of suicidal thoughts and behavior.
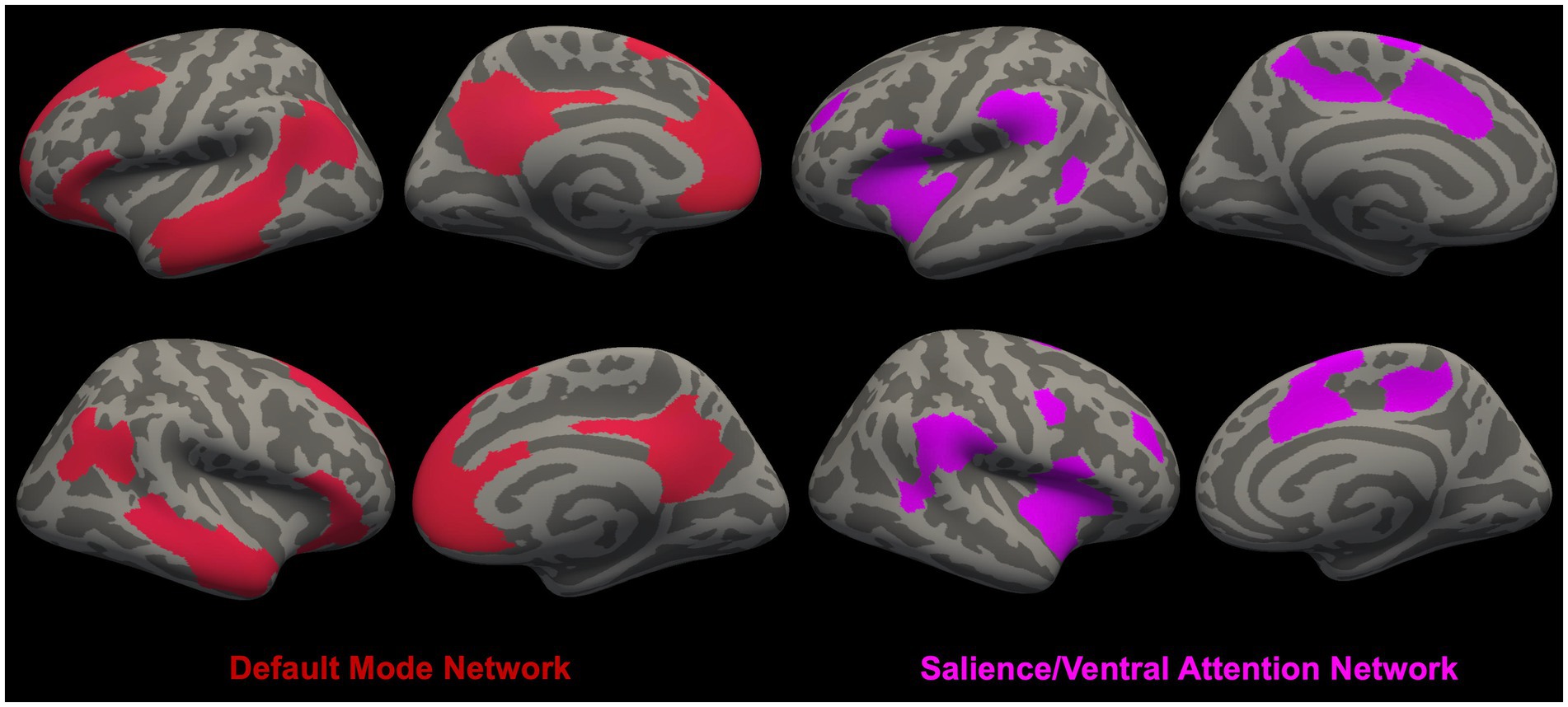
Figure 2. Overview of brain networks [i.e., Default Mode Network (DMN) (79) and Salience/Ventral Attention Network (SN/VAN) (80)] (81) that are most commonly found to be involved in functional MRI and brain morphometry research of suicidal thoughts and behavior.
The fourth neurobiological modality that we included in this study was Diffusion Tensor Imaging (DTI). This method analyzes the robustness of water diffusion within white-matter structures of the brain to find associated differences between patient populations and healthy controls or other cohorts. DTI also examines the physical “highways” within and between significant structures. While fMRI gives information on “in the moment” electrical communication that could conceivably indicate functional robustness of structures and pathways, DTI is a direct measure of the physical robustness of those structures and pathways. To that end, it may be considered as a more concrete/persistent measure to estimate the difference between healthy controls and the suicidal patients. Again, most of the modalities can be accomplished in a relatively short scanning session and are more potentially accessible to the working clinician.
Recent advancements in imaging have allowed the scientists to study differences in white-matter integrity, compactness, or structural connectivity in clinical populations – the “quality of the highways” so to speak. These studies use parameters such as apparent diffusion coefficient (ADC-speed of water flow regardless of direction), fractional anisotropy (FA-diffusion of water molecules in a particular direction), and edge-weight (white-matter structural connectivity) to measure connections between regions of interest (85, 86). Edge weight has been considered a potentially more appropriate parameter to measure the strength of structural connectivity because it takes into account both the number of white-matter fibers and the size of the regions of interest (87). Our focused review identified studies which reported associations between these measures and suicidal behavior.
In a study of young adult healthy controls and young adults with MDD with and without a history of suicide attempts conducted by Jia et al. reduced FA was found in the (a) left anterior limb of the internal capsule (ALIC) for suicide attempters compared to non-attempters and healthy controls, (b) right frontal lobe (subgyral) for suicide attempters compared to healthy controls, and (c) right lentiform nucleus (putamen) for suicide attempters compared to non-attempters (88). In another similar study by Jia et al. it was also found that compared to healthy controls, depressed suicide attempters had significantly lower mean percentage of fibers projecting from the ALIC to the left medial frontal cortex, left OFC, and left thalamus. Compared to depressed non-suicide attempters, depressed suicide attempters had significantly lower mean percentage of fibers projecting from the ALIC to the left OFC and left thalamus (89). However, in a study involving panic disorder and suicide attempt, several regions, including the retrolenticular part of the internal capsule, splenium of the corpus callosum, superior and posterior corona radiata, posterior thalamic radiations, sagittal stratum (including the inferior longitudinal fasciculus and inferior fronto-occipital fasciculus), and superior longitudinal fasciculus, showed increased FA in individuals with panic disorder and history of suicidal attempt (PD + SA) compared to individuals with panic disorder but without any history of suicidal attempt (90). For the PD + SA group, Kim et al. also found that for two regions (i.e., right retrolenticular part of the internal capsule and bilateral posterior thalamic radiations), there was a significant positive association between FA and SUI.
In another study, lower baseline FA was found in the left ALIC, bilateral dmPFC, and right dorsal cingulum for future suicide attempters (i.e., who attempted suicide between the baseline and follow-up assessment) compared to non-attempters (61). In that study, compared to the non-future suicide attempt group, both future suicide attempters with or without a history of suicide attempts had lower FA for the left dmPFC, right dlPFC, and left ALIC. Future suicide attempters with a history of suicide attempts also showed lower FA in the right dmPFC and right dorsal cingulum. Authors found that after an exclusion of four participants with alcohol/substance use disorder, the left ventral prefrontal cortex also had lower FA for the future suicide attempters relative to non-attempters.
Fan et al. also found lower FA in the dorsal and ventral frontal regions that included the uncinate fasciculus for individuals with MDD and a history of suicide attempts as compared to non-suicide attempters (60).
In terms of structural connectivity parameters, it was determined that compared to individuals with MDD without SUI, individuals with MDD and SUI had reduced structural connectivity/edge weights across cortical (i.e., rostral middle frontal cortex, superior parietal cortex), subdivisions of the inferior frontal cortex (i.e., pars triangularis and pars orbitalis, frontal pole, and lateral occipital cortex), and sub-cortical (i.e., pallidum, thalamus, putamen, and caudate) regions in the left hemisphere (91). In terms of FA also, the frontal areas, especially right dmPFC and white-matter bundles in several regions, including the bilateral inferior fronto-occipital fasciculus, bilateral uncinate fasciculus, body of corpus callosum, right anterior limb of internal capsule, right external capsule, left posterior thalamic radiation, and bilateral posterior corona radiata, showed lower FA amongst suicide attempters with MDD compared to non-attempters with MDD and healthy controls (92, 93).
Another structural connectivity study showed that compared to euthymic non-attempters and healthy controls, there was significantly decreased structural connectivity in euthymic suicide attempters in the connections between the left olfactory cortex and left anterior cingulate gyrus, as well as a clear trend of decreased connectivity between the right medial orbital, SFG, and the right rectal gyrus and between the right calcarine fissure and both the left superior and middle occipital gyrus (94). Hwang et al. reported greater edge weight in the left PCC and greater structural connectivity strength of local connections amongst participants who were military veterans with prior suicide attempts in comparison to those with SUI only and with no suicidal behavior (95).
A 2021 study by Chen et al. evaluated white-matter integrity (generalized fractional anisotropy) and white-matter compactness (normalized quantitative anisotropy) among depressive patients with and without past suicide attempts (96). On a voxel-based (region of interest) analysis, participants with past suicide attempts had significantly lower white matter compactness and integrity in the corpus callosum, cingulate gyrus, and caudate than both the depressed non-attempt and the healthy control groups, with differences between the attempt group and healthy control group reaching statistical significance (96).
The same researchers evaluated white matter density and integrity among depressed patients with suicidal ideation but no history of attempts (97). In the voxel-based analysis, white matter integrity was found to be decreased in the suicidal ideation group, specifically in the corpus callosum and the ACC compared to depressed, non-SI, and healthy control participants. On RSFC, the suicidal ideation group had weaker connections within subnetworks of the frontal lobe compared to healthy controls but did not find differences between suicidal ideation and depression in suicidal participants.
The concept of maladaptive thinking in depression is well understood by most clinicians (98). The notion that patients may develop maladaptive thinking in/or about social interactions is likely not surprising. However, to grasp the comprehensive picture of what is happening neurobiologically, providers must look to the evolving literature in neuroscience and neurobiology. Among our sample we have found many indications that functional, structural, RSFC, and diffusion-weighted MRI studies are beginning to bridge this translational gap well.
Task based fMRI studies show abnormal activation in prefrontal, subcortical, and limbic regions. Specifically, areas important for emotional processing, reward-based learning (value estimation), emotional regulation/social exclusion, relative representation/sensitivity to affective stimuli, and cognitive control/response inhibition show abnormal activation, though in some cases (18), they contradict other studies. It may be that the same regions (e.g., left insula) are increasingly active in some cognitive challenges but less activated than controls in others, but this would not be contradictory to Van Heeringen’s model. Complicating the current growing body of research is the comparison of suicidal individuals with past attempts, versus those without, versus those with ideation and those without. At this stage it may simply be important to keep the focus on what regions and processes are relevant as data grows and models continue to develop.
Among our sample, rsfMRI enriches the functional data by showing the major networks with abnormal connectivity among patients with MDD and STBs. From machine learning to strictly RSFC studies, the most relevant networks are clearly the M-FPN/DMN and M-CIN/SN and their communication within and between each other and with top-down control areas of the L-FPN/CCN. With the vast and complex roles of the M-FPN/DMN and M-CIN/SN, it is clear that even in the resting state, areas important for learning, affective and social processing, and cognitive control are affected, but this research also lends depth to the increased weight of an abnormally functioning “salience judge” at the cost of new information coming in from all sides. The mix of participants (adults, adults and adolescents, older adults, combat veterans, and convicted criminals) and type of STB being studied (standard scale score, suicide attempt, or suicidal ideation) of course complicates these already complicated findings, but data continues to grow.
Helping to enlighten a picture of abnormal communication amongst regions and networks, DTI data among our sample similarly supported differences in major tracts such as the ALIC and uncinate fasciculus (UF), important in communicating between structures of the reward-based learning network, along with differences in edge weight and FA among and between structures important for emotional processing, reward-based learning (value estimation), emotional regulation/social exclusion, relative representation/sensitivity to affective stimuli, and cognitive control/response inhibition. The clear difference among our DTI sample and the other methodologies, however, is that among participants with MDD and STBs, all of these measures were found to be decreased in comparison to control groups.
This is further supported in our sample by the morphometric studies generally showing that structures involved in both top-down and bottom-up emotional processing, visual and language processing, impulse control, and affective processing are atypical across the board. Decreased GMV/thickness/area were found in frontal systems such as the IFG and OFC, ventral-lateral prefrontal cortex, dlPFC and temporal regions, and in suicidal individuals, there are clear reductions in GMV in subcortical (putamen and caudate) and limbic (hippocampus-MFPN/DMN) areas as well. Intuitively, these measured differences represent more long-term changes among the relevant structures and networks that a patient would depend on as stressors and, hence, the risk of STBs accumulate.
Previous literature reviews are generally consistent with our results, though many have incorporated different samples with different conditions that inhibit direct comparison. Especially relevant to our review are the work of Jollant (5), Desmyter (99), Zhang (100), Martin (101), Schmaal (10), and Auerbach (11), among others (102–106). Studies over the last 20 years have increasingly showed a relation between emotional pain and physical pain (107–109). Work by Olie et al. has specifically examined the increasing relevance of social exclusion to affective pain and suicide and further discussed the association between neuroimaging findings of social exclusion and suicide risk (110). They found that while the normal response to the affective pain of social exclusion increases activity in the anterior insula, ACC, and inferior OFC in normal controls, suicidal individuals show a decreased activation in these same regions, even compared with non-suicidal patients with a mood disorder.
In short, we are not proposing a grand new theory that is all encompassing, for that would take many more variables into account, which have their own emerging literature, such as genetic, socioeconomic, and cultural factors. Also biochemical, MEG, SPECT, and PET scanning and incorporating the rich and ever evolving psychometric data would also need to be considered. We are simply proposing a framework to begin applying emerging neurobiological data in a clinical and chronological way in conjunction with already used measures, such as psychometrics and clinical assessments, so that as a patient encounters various diatheses and stressors, their clinician will be able to look to this framework to address a complex problem in comprehensive but clinically feasible way. With genuine and earnest collaboration in translational medicine, imaging, and neuroscience, combined with machine learning and worldwide research consortiums focusing on suicidal thoughts and behaviors, and replication of findings, especially in these diverse and complicated modalities, collaboration can shrink the time from new discoveries to clinical intervention. There are few things more urgent than attempting it.
Our simple framework is this: First, morphometric changes may be more observable early on from genetic and environmental stressors but also long standing atypical cognitive processing. Second, abnormalities in diffusion-weighted projections will become apparent, implying increasingly longstanding atypical networks and relevant ROI communication, and demonstrable on scanning. Third, abnormalities in processing new information, especially negative social and affective valenced-relevant stimuli tethered to language and facial processing, will be demonstrated on rsfMRI/RSFC analysis, as evident by increased communication between networks (M-CIN/SN to L-FPN/CCN), implying maladaptive rumination of faulty negative information and decreased communication between new, contradicting affective/relevant processing areas and value estimation/strategy adjustment networks (M-CIN/SN TO M-FPN/DMN). Fourth, the gap between value and risk estimation will widen on fMRI tasks immediately prior to the STBs.
Ultimately, we hope to start to construct a chronological framework of early diatheses, developing stressors, whether distal or proximal, and correlating their neurobiological fingerprint across MRI modalities and behavioral task performance. We will do this through continued task/theory-based fMRI and network studies, structural/morphometric, RSFC, and DTI research. We will continue to develop machine learning evaluations through each modality and across them first using classification and machine learning techniques to quickly determine biosignatures that directly affect suicide risk and improve our model. We will then use a regression analysis to analyze level of risk and sequence mining to predict proximal neurobiological changes. Through this comprehensive and accelerated approach, we hope to begin to capture a clinically relevant and useful point-of-care tool that can accurately and thoroughly assess risk of suicidal ideation and attempt. Then, with extensive collaboration, those in the field of neurobiological suicide research can shift into evaluating the most effective interventions at each specific time that will prevent it. Lastly, given that functional connectivity studies and diffusion tensor imaging have a particular drawback: neither are in a position to assess directed functional or effective connectivity (111). In other words, one gets a single number for the connectivity between two regions – as opposed to separate estimates of the directed influence of one region on another, and the reciprocal influence. This is important when talking about the distinction between bottom-up and top-down processes in functional brain hierarchies. In consequence, given our expertise in cutting-edge directed functional (e.g., Granger causality) and effective (e.g., dynamic causal modeling) brain connectivity techniques (29–31, 112–117), we will aim to see how analyses of directional connectivity nuance the emerging picture of suicidal thoughts and behavior described above. Emerging research in directed functional and effective connectivity will surely prove invaluable.
MD conceived the presented idea, performed the literature search and wrote the initial draft. KB substantially contributed to interpreting the relevant literature and writing the manuscript. EC performed the literature search and contributed to writing of the manuscript. JB substantially contributed to the conception, interpretation, and writing of the manuscript. AD contributed to the writing of the manuscript and edited various versions of the draft. SB conceived the presented idea, performed literature search, wrote the initial draft, and supervised all aspects of the study. All authors contributed to the article and approved the submitted version.
This work was funded by GRT-00092 at Boys Town National Research Hospital.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. World Health Organization . Preventing suicide: a global imperative. Geneva, Switzerland:WHO Press (2014). Available at: https://apps.who.int/iris/bitstream/handle/10665/131056/9789241564878_eng.pdf
2. Curtin, SC, and Heron, M. Death rates due to suicide and homicide among persons aged 10-24: United States, 2000-2017. NCHS Data Brief. Washington, DC, United States: U.S. Department of Health and Human Services (2019) 352:1–8. Available at: https://pubmed.ncbi.nlm.nih.gov/31751202/
4. Posner, K, Brown, GK, Stanley, B, Brent, DA, Yershova, KV, Oquendo, MA, et al. The Columbia-suicide severity rating scale: initial validity and internal consistency findings from three multisite studies with adolescents and adults. Am J Psychiatr. (2011) 168:1266–77. doi: 10.1176/appi.ajp.2011.10111704
5. Jollant, F, Lawrence, NL, Olié, E, Guillaume, S, and Courtet, P. The suicidal mind and brain: a review of neuropsychological and neuroimaging studies. World J Biol Psychiatry. (2011) 12:319–39. doi: 10.3109/15622975.2011.556200
6. van Heeringen, K . The neuroscience of suicidal behavior. Cambridge, England: Cambridge University Press (2018).
7. Van Orden, KA, Witte, TK, Cukrowicz, KC, Braithwaite, SR, Selby, EA, and Joiner, TEJ. The interpersonal theory of suicide. Psychol Rev. (2010) 117:575–600. doi: 10.1037/a0018697
8. McGirr, A, and Turecki, G. The relationship of impulsive aggressiveness to suicidality and other depression-linked behaviors. Curr Psychiatry Rep. (2007) 9:460–6. doi: 10.1007/s11920-007-0062-2
9. Turecki, G, and Brent, DA. Suicide and suicidal behaviour. Lancet. (2016) 387:1227–39. doi: 10.1016/S0140-6736(15)00234-2
10. Schmaal, L, van Harmelen, AL, Chatzi, V, Lippard, ETC, Toenders, YJ, Averill, LA, et al. Imaging suicidal thoughts and behaviors: a comprehensive review of 2 decades of neuroimaging studies. Mol Psychiatry. (2020) 25:408–27. doi: 10.1038/s41380-019-0587-x
11. Auerbach, RP, Pagliaccio, D, Allison, GO, Alqueza, KL, and Alonso, MF. Neural correlates associated with suicide and nonsuicidal self-injury in youth. Biol Psychiatry. (2021) 89:119–33. doi: 10.1016/j.biopsych.2020.06.002
12. Williams, JMG, and Williams, M. Cry of pain: understanding suicide and the suicidal mind. London, England:Piatkus (2014)
13. O’Connor, RC, and Kirtley, OJ. The integrated motivational-volitional model of suicidal behaviour. Philos Trans R Soc Lond Ser B Biol Sci. (2018) 373:20170268. doi: 10.1098/rstb.2017.0268
14. Mann, JJ, Waternaux, C, Haas, GL, and Malone, KM. Toward a clinical model of suicidal behavior in psychiatric patients. Am J Psychiatry. (1999) 156:181–9. doi: 10.1176/ajp.156.2.181
15. Brådvik, L . Suicide risk and mental disorders. Int J Environ Res Public Health. (2018) 15:2028. doi: 10.3390/ijerph15092028
16. Brown, VM, Wilson, J, Hallquist, MN, Szanto, K, and Dombrovski, AY. Ventromedial prefrontal value signals and functional connectivity during decision-making in suicidal behavior and impulsivity. Neuropsychopharmacology. (2020) 45:1034–41. doi: 10.1038/s41386-020-0632-0
17. Jollant, F, Lawrence, NS, Giampietro, V, Brammer, MJ, Fullana, MA, Drapier, D, et al. Orbitofrontal cortex response to angry faces in men with histories of suicide attempts. Am J Psychiatr. (2008) 165:740–8. doi: 10.1176/appi.ajp.2008.07081239
18. Li, H, Chen, Z, Gong, Q, and Jia, Z. Voxel-wise meta-analysis of task-related brain activation abnormalities in major depressive disorder with suicide behavior. Brain Imaging Behav. (2020) 14:1298–308. doi: 10.1007/s11682-019-00045-3
19. Olié, E, Jollant, F, Deverdun, J, De Champfleur, NM, Cyprien, F, Le Bars, E, et al. The experience of social exclusion in women with a history of suicidal acts: a neuroimaging study. Sci Rep. (2017) 7:89. doi: 10.1038/s41598-017-00211-x
20. Cáceda, R, James, GA, Stowe, ZN, Delgado, PL, Kordsmeier, N, and Kilts, CD. The neural correlates of low social integration as a risk factor for suicide. Eur Arch Psychiatry Clin Neurosci. (2020) 270:619–31. doi: 10.1007/s00406-019-00990-6
21. Miller, AB, McLaughlin, KA, Busso, DS, Brueck, S, Peverill, M, and Sheridan, MA. Neural correlates of emotion regulation and adolescent suicidal ideation. Biol Psychiatry Cogn Neurosci Neuroimaging. (2018) 3:125–32. doi: 10.1016/j.bpsc.2017.08.008
22. Pan, LA, Hassel, S, Segreti, AM, Nau, SA, Brent, DA, and Phillips, ML. Differential patterns of activity and functional connectivity in emotion processing neural circuitry to angry and happy faces in adolescents with and without suicide attempt. Psychol Med. (2013) 43:2129–42. doi: 10.1017/S0033291712002966
23. Ai, H, van Tol, MJ, Marsman, JBC, Veltman, DJ, Ruhé, HG, van der Wee, NJA, et al. Differential relations of suicidality in depression to brain activation during emotional and executive processing. J Psychiatr Res. (2018) 105:78–85. doi: 10.1016/j.jpsychires.2018.08.018
24. Alarcón, G, Sauder, M, Teoh, JY, Forbes, EE, and Quevedo, K. Amygdala functional connectivity during self-face processing in depressed adolescents with recent suicide attempt. J Am Acad Child Adolesc Psychiatry. (2019) 58:221–31. doi: 10.1016/j.jaac.2018.06.036
25. Malhi, GS, Das, P, Outhred, T, Gessler, D, John Mann, J, and Bryant, R. Cognitive and emotional impairments underpinning suicidal activity in patients with mood disorders: an fMRI study. Acta Psychiatr Scand. (2019) 139:454–63. doi: 10.1111/acps.13022
26. Just, MA, Pan, L, Cherkassky, VL, McMakin, DL, Cha, C, Nock, MK, et al. Machine learning of neural representations of suicide and emotion concepts identifies suicidal youth. Nat Hum Behav. (2017) 1:911–9. doi: 10.1038/s41562-017-0234-y
27. Richard-Devantoy, S, Ding, Y, Lepage, M, Turecki, G, and Jollant, F. Cognitive inhibition in depression and suicidal behavior: a neuroimaging study. Psychol Med. (2016) 46:933–44. doi: 10.1017/S0033291715002421
28. van Velzen, LS, Dauvermann, MR, Colic, L, Villa, LM, Savage, HS, Toenders, YJ, et al. Structural brain alterations associated with suicidal thoughts and behaviors in young people: results across 21 international studies from the ENIGMA suicidal thoughts and behaviours consortium. Med Rxiv. (2021). doi: 10.1038/s41380-022-01734-0
29. Bajaj, S, Adhikari, BM, Friston, KJ, and Dhamala, M. Bridging the gap: dynamic causal modeling and granger causality analysis of resting state functional magnetic resonance imaging. Brain Connect. (2016) 6:652–61. doi: 10.1089/brain.2016.0422
30. Bajaj, S, Adhikari, BM, and Dhamala, M. Higher frequency network activity flow predicts lower frequency node activity in intrinsic low-frequency BOLD fluctuations. PLoS One. (2013) 8:e64466. doi: 10.1371/journal.pone.0064466
31. Bajaj, S, Raikes, AC, Razi, A, Miller, MA, and Killgore, WDS. Blue-light therapy strengthens resting-state effective connectivity within default-mode network after mild TBI. J Cent Nerv Syst Dis. (2021) 13:11795735211015076. doi: 10.1177/11795735211015076
32. Uddin, LQ, Yeo, BTT, and Spreng, RN. Towards a universal taxonomy of macro-scale functional human brain networks. Brain Topogr. (2019) 32:926–42. doi: 10.1007/s10548-019-00744-6
33. Qiu, H, Cao, B, Cao, J, Li, X, Chen, J, Wang, W, et al. Resting-state functional connectivity of the anterior cingulate cortex in young adults depressed patients with and without suicidal behavior. Behav Brain Res. (2020) 384:112544. doi: 10.1016/j.bbr.2020.112544
34. Du, L, Zeng, J, Liu, H, Tang, D, Meng, H, Li, Y, et al. Fronto-limbic disconnection in depressed patients with suicidal ideation: a resting-state functional connectivity study. J Affect Disord. (2017) 215:213–7. doi: 10.1016/j.jad.2017.02.027
35. Yang, Y, Chattun, MR, Yan, R, Zhao, K, Chen, Y, Zhu, R, et al. Atrophy of right inferior frontal orbital gyrus and frontoparietal functional connectivity abnormality in depressed suicide attempters. Brain Imaging Behav. (2020) 14:2542–52. doi: 10.1007/s11682-019-00206-4
36. Stange, JP, Jenkins, LM, Pocius, S, Kreutzer, K, Bessette, KL, Deldonno, SR, et al. Using resting-state intrinsic network connectivity to identify suicide risk in mood disorders. Psychol Med. (2019) 50:2324–34. doi: 10.1017/S0033291719002356
37. Cao, J, Chen, X, Chen, J, Ai, M, Gan, Y, Wang, W, et al. Resting-state functional MRI of abnormal baseline brain activity in young depressed patients with and without suicidal behavior. J Affect Disord. (2016) 205:252–63. doi: 10.1016/j.jad.2016.07.002
38. Cao, J, Chen, X, Chen, J, Ai, M, Gan, Y, He, J, et al. The association between resting state functional connectivity and the trait of impulsivity and suicidal ideation in young depressed patients with suicide attempts. Front Psych. (2021) 12:567976. doi: 10.3389/fpsyt.2021.567976
39. Zhang, R, Zhang, L, Wei, S, Wang, P, Jiang, X, Tang, Y, et al. Increased amygdala-paracentral lobule/precuneus functional connectivity associated with patients with mood disorder and suicidal behavior. Front Hum Neurosci. (2020) 14:585664. doi: 10.3389/fnhum.2020.611008
40. Kang, SG, Na, KS, Choi, JW, Kim, JH, Son, YD, and Lee, YJ. Resting-state functional connectivity of the amygdala in suicide attempters with major depressive disorder. Prog Neuro-Psychopharmacol Biol Psychiatry. (2017) 77:222–7. doi: 10.1016/j.pnpbp.2017.04.029
41. Wei, S, Chang, M, Zhang, R, Jiang, X, Wang, F, and Tang, Y. Amygdala functional connectivity in female patients with major depressive disorder with and without suicidal ideation. Ann General Psychiatry. (2018) 17:37. doi: 10.1186/s12991-018-0208-0
42. Cao, J, Ai, M, Chen, X, Chen, J, Wang, W, and Kuang, L. Altered resting-state functional network connectivity is associated with suicide attempt in young depressed patients. Psychiatry Res. (2020) 285:112713. doi: 10.1016/j.psychres.2019.112713
43. Shu, Y, Kuang, L, Huang, Q, and He, L. Fractional amplitude of low-frequency fluctuation (fALFF) alterations in young depressed patients with suicide attempts after cognitive behavioral therapy and antidepressant medication cotherapy: a resting-state fMRI study. J Affect Disord. (2020) 276:822–8. doi: 10.1016/j.jad.2020.07.038
44. Zhang, S, Chen, JM, Kuang, L, Cao, J, Zhang, H, Ai, M, et al. Association between abnormal default mode network activity and suicidality in depressed adolescents. BMC Psychiatry. (2016) 16:337. doi: 10.1186/s12888-016-1047-7
45. Chen, V, Chou, Y-S, Tsai, Y-H, Huang, Y-C, McIntyre, RS, and Weng, J-C. Resting-state functional connectivity and brain network abnormalities in depressive patients with suicidal ideation. Brain Topogr. (2021) 34:234–44. doi: 10.1007/s10548-020-00817-x
46. Barredo, J, Aiken, E, Vant Wout-Frank, M, Greenberg, BD, Carpenter, LL, and Philip, NS. Neuroimaging correlates of suicidality in decision-making circuits in posttraumatic stress disorder. Front Psychiatry. (2019) 10:44. doi: 10.3389/fpsyt.2019.00044
47. Lee, S, Lee, SM, Kang, WS, Jahng, GH, Ryu, CW, and Park, JK. Altered resting-state functional connectivity in depressive disorder patients with suicidal attempts. Neurosci Lett. (2019) 696:174–8. doi: 10.1016/j.neulet.2018.12.037
48. Schreiner, MW, Klimes-Dougan, B, and Cullen, KR. Neural correlates of suicidality in adolescents with major depression: resting-state functional connectivity of the precuneus and posterior cingulate cortex. Suicide Life Threat Behav. (2019) 49:899–913. doi: 10.1111/sltb.12471
49. Kim, K, Kim, SW, Myung, W, Han, CE, Fava, M, Mischoulon, D, et al. Reduced orbitofrontal-thalamic functional connectivity related to suicidal ideation in patients with major depressive disorder. Sci Rep. (2017) 7:15772. doi: 10.1038/s41598-017-15926-0
50. Gosnell, SN, Fowler, JC, and Salas, R. Classifying suicidal behavior with resting-state functional connectivity and structural neuroimaging. Acta Psychiatr Scand. (2019) 140:20–9. doi: 10.1111/acps.13029
51. Dai, Z, Shen, X, Tian, S, Yan, R, Wang, H, Wang, X, et al. Gradually evaluating of suicidal risk in depression by semi-supervised cluster analysis on resting-state fMRI. Brain Imaging Behav. (2020) 15:2149–58. doi: 10.1007/s11682-020-00410-7
52. Stumps, A, Jagger-Rickels, A, Rothlein, D, Amick, M, Park, H, Evans, T, et al. Connectome-based functional connectivity markers of suicide attempt. J Affect Disord. (2020). 430–40. doi: 10.1016/j.jad.2020.11.061
53. Chase, HW, Segreti, AM, Keller, TA, Cherkassky, VL, Just, MA, Pan, LA, et al. Alterations of functional connectivity and intrinsic activity within the cingulate cortex of suicidal ideators. J Affect Disord. (2017) 212:78–85. doi: 10.1016/j.jad.2017.01.013
54. Serafini, G, Pardini, M, Pompili, M, Girardi, P, and Amore, M. Understanding suicidal behavior: The contribution of recent resting-state fMRI techniques. Front Psychiatry. (2016) 7:69. doi: 10.3389/fpsyt.2016.00069
55. Hwang, JP, Lee, TW, Tsai, SJ, Chen, TJ, Yang, CH, Lirng, JF, et al. Cortical and subcortical abnormalities in late-onset depression with history of suicide attempts investigated with MRI and voxel-based morphometry. J Geriatr Psychiatry Neurol. (2010) 23:171–84. doi: 10.1177/0891988710363713
56. Wagner, G, Schultz, CC, Koch, K, Schachtzabel, C, Sauer, H, and Schlösser, RG. Prefrontal cortical thickness in depressed patients with high-risk for suicidal behavior. J Psychiatr Res. (2012) 46:1449–55. doi: 10.1016/j.jpsychires.2012.07.013
57. Huber, RS, McGlade, EC, Legarreta, M, Subramaniam, P, Renshaw, PF, and Yurgelun-Todd, DA. Cingulate white matter volume and associated cognitive and behavioral impulsivity in veterans with a history of suicide behavior. J Affect Disord. (2021) 281:117–24. doi: 10.1016/j.jad.2020.11.126
58. Wang, P, Zhang, R, Jiang, X, Wei, S, Wang, F, and Tang, Y. Gray matter volume alterations associated with suicidal ideation and suicide attempts in patients with mood disorders. Ann General Psychiatry. (2020) 19:69. doi: 10.1186/s12991-020-00318-y
59. Ding, Y, Lawrence, N, Olie, E, Cyprien, F, le Bars, E, Bonafe, A, et al. Prefrontal cortex markers of suicidal vulnerability in mood disorders: a model-based structural neuroimaging study with a translational perspective. Transl Psychiatry. (2015) 5:e516. doi: 10.1038/tp.2015.1
60. Fan, S, Lippard, ETC, Sankar, A, Wallace, A, Johnston, JAY, Wang, F, et al. Gray and white matter differences in adolescents and young adults with prior suicide attempts across bipolar and major depressive disorders. J Affect Disord. (2019) 245:1089–97. doi: 10.1016/j.jad.2018.11.095
61. Lippard, ETC, Johnston, JAY, Spencer, L, Quatrano, S, Fan, S, Sankar, A, et al. Preliminary examination of gray and white matter structure and longitudinal structural changes in frontal systems associated with future suicide attempts in adolescents and young adults with mood disorders. J Affect Disord. (2019) 245:1139–48. doi: 10.1016/j.jad.2018.11.097
62. Segreti, AM, Chase, HW, Just, M, Brent, D, and Pan, L. Cortical thickness and volume reductions in young adults with current suicidal ideation. J Affect Disord. (2019) 245:126–9. doi: 10.1016/j.jad.2018.10.081
63. Bajaj, S, Raikes, AC, Smith, R, Vanuk, JR, and Killgore, WDS. The role of prefrontal cortical surface area and volume in preclinical suicidal ideation in a non-clinical sample. Front Psychiatry. (2019) 10:445. doi: 10.3389/fpsyt.2019.00445
64. Kang, SG, Cho, SE, Na, KS, Lee, JS, Joo, SW, Cho, SJ, et al. Differences in brain surface area and cortical volume between suicide attempters and non-attempters with major depressive disorder. Psychiatry Res Neuroimaging. (2020) 297:111032. doi: 10.1016/j.pscychresns.2020.111032
65. Harenski, CL, Harenski, KA, Calhoun, VD, and Kiehl, KA. Source-based morphometry reveals gray matter differences related to suicidal behavior in criminal offenders. Brain Imaging Behav. (2020) 14:1–9. doi: 10.1007/s11682-018-9957-2
66. Kang, W, Shin, JH, Han, KM, Kim, A, Kang, Y, Kang, J, et al. Local shape volume alterations in subcortical structures of suicide attempters with major depressive disorder. Hum Brain Mapp. (2020) 41:4925–34. doi: 10.1002/hbm.25168
67. Gosnell, SN, Velasquez, KM, Molfese, DL, Molfese, PJ, Madan, A, Fowler, JC, et al. Prefrontal cortex, temporal cortex, and hippocampus volume are affected in suicidal psychiatric patients. Psychiatry Res Neuroimaging. (2016) 256:50–6. doi: 10.1016/j.pscychresns.2016.09.005
68. Chen, F, Bertelsen, AB, Holm, IE, Nyengaard, JR, Rosenberg, R, and Dorph-Petersen, KA. Hippocampal volume and cell number in depression, schizophrenia, and suicide subjects. Brain Res. (2020) 1727:146546. doi: 10.1016/j.brainres.2019.146546
69. Jollant, F, Wagner, G, Richard-Devantoy, S, Köhler, S, Bär, KJ, Turecki, G, et al. Neuroimaging-informed phenotypes of suicidal behavior: a family history of suicide and the use of a violent suicidal means. Transl Psychiatry. (2018) 8:120. doi: 10.1038/s41398-018-0170-2
70. Ho, TC, Cichocki, AC, Gifuni, AJ, Camacho, MC, Ordaz, SJ, Singh, MK, et al. Reduced dorsal striatal gray matter volume predicts implicit suicidal ideation in adolescents. Soc Cogn Affect Neurosci. (2018) 13:1215–24. doi: 10.1093/scan/nsy089
71. Ho, TC, Teresi, GI, Ojha, A, Walker, JC, Kirshenbaum, JS, Singh, MK, et al. Smaller caudate gray matter volume is associated with greater implicit suicidal ideation in depressed adolescents. J Affect Disord. (2021) 278:650–7. doi: 10.1016/j.jad.2020.09.046
72. Pan, LA, Ramos, L, Segreti, A, Brent, DA, and Phillips, ML. Right superior temporal gyrus volume in adolescents with a history of suicide attempt. Br J Psychiatry. (2015) 206:339–40. doi: 10.1192/bjp.bp.114.151316
73. Vidal-Ribas, P, Janiri, D, Doucet, GE, Pornpattananangkul, N, Nielson, DM, Frangou, S, et al. Multimodal neuroimaging of suicidal thoughts and behaviors in a U.S. population-based sample of school-age children. Am J Psychiatr. (2021) 178:321–32. doi: 10.1176/appi.ajp.2020.20020120
74. McLellan, Q, Wilkes, TC, Swansburg, R, Jaworska, N, Langevin, LM, and Mac Master, FP. History of suicide attempt and right superior temporal gyrus volume in youth with treatment-resistant major depressive disorder. J Affect Disord. (2018) 239:291–4. doi: 10.1016/j.jad.2018.07.030
75. Peng, H, Wu, K, Li, J, Qi, H, Guo, S, Chi, M, et al. Increased suicide attempts in young depressed patients with abnormal temporal-parietal-limbic gray matter volume. J Affect Disord. (2014) 165:69–73. doi: 10.1016/j.jad.2014.04.046
76. Lee, YJ, Kim, S, Gwak, AR, Kim, SJ, Kang, SG, Na, KS, et al. Decreased regional gray matter volume in suicide attempters compared to suicide non-attempters with major depressive disorders. Compr Psychiatry. (2016) 67:59–65. doi: 10.1016/j.comppsych.2016.02.013
77. Campos, AI, Thompson, PM, Veltman, DJ, Pozzi, E, van Veltzen, LS, Jahanshad, N, et al. Brain correlates of suicide attempt in 18, 925 participants across 18 international cohorts. Biol Psychiatry. (2021) 90:243–52. doi: 10.1016/j.biopsych.2021.03.015
78. Sarkinaite, M, Gleizniene, R, Adomaitiene, V, Dambrauskiene, K, Raskauskiene, N, and Steibliene, V. Volumetric MRI analysis of brain structures in patients with history of first and repeated suicide attempts: a cross sectional study. Diagnostics. (2021) 11:488. doi: 10.3390/diagnostics11030488.
79. Raichle, ME . The brain’s default mode network. Annu Rev Neurosci. (2015) 38:433–47. doi: 10.1146/annurev-neuro-071013-014030
80. Chand, GB, and Dhamala, M. The salience network dynamics in perceptual decision-making. Neuroimage. (2016) 134:85–93. doi: 10.1016/j.neuroimage.2016.04.018
81. Chand, GB, and Dhamala, M. Interactions among the brain default-mode, salience, and central-executive networks during perceptual decision-making of moving dots. Brain Connect. (2015) 6:249–54. doi: 10.1089/brain.2015.0379
82. Fischl, B, Sereno, MI, and Dale, AM. Cortical surface-based analysis. II: inflation, flattening, and a surface-based coordinate system. NeuroImage. (1999) 9:195–207. doi: 10.1006/nimg.1998.0396
83. Dale, AM, Fischl, B, and Sereno, MI. Cortical surface-based analysis. I. Segmentation and surface reconstruction. Neuroimage. (1999) 9:179–94. doi: 10.1006/nimg.1998.0395
84. Yeo, BT, Krienen, FM, Sepulcre, J, Sabuncu, MR, Lashkari, D, Hollinshead, M, et al. The organization of the human cerebral cortex estimated by intrinsic functional connectivity. J Neurophysiol. (2011) 106:1125–65. doi: 10.1152/jn.00338.2011
85. Provenzale, JM, Isaacson, J, Chen, S, Stinnett, S, and Liu, C. Correlation of apparent diffusion coefficient and fractional anisotropy values in the developing infant brain. AJR Am J Roentgenol. (2010) 195:W456–62. doi: 10.2214/AJR.10.4886
86. Cheng, H, Wang, Y, Sheng, J, Kronenberger, WG, Mathews, VP, Hummer, TA, et al. Characteristics and variability of structural networks derived from diffusion tensor imaging. NeuroImage. (2012) 61:1153–64. doi: 10.1016/j.neuroimage.2012.03.036
87. Huang, HQ, and Ding, MZ. Linking functional connectivity and structural connectivity quantitatively: a comparison of methods. Brain Connect. (2016) 6:99–108. doi: 10.1089/brain.2015.0382
88. Jia, Z, Huang, X, Wu, Q, Zhang, T, Lui, S, Zhang, J, et al. High-field magnetic resonance imaging of suicidality in patients with major depressive disorder. Am J Psychiatr. (2010) 167:1381–90. doi: 10.1176/appi.ajp.2010.09101513
89. Jia, Z, Wang, Y, Huang, X, Kuang, W, Wu, Q, Lui, S, et al. Impaired frontothalamic circuitry in suicidal patients with depression revealed by diffusion tensor imaging at 3.0 T. J Psychiatry Neurosci. (2014) 39:170–7. doi: 10.1503/jpn.130023
90. Kim, B, Oh, J, Kim, MK, Lee, S, Tae, WS, Kim, CM, et al. White matter alterations are associated with suicide attempt in patients with panic disorder. J Affect Disord. (2015) 175:139–46. doi: 10.1016/j.jad.2015.01.001
91. Myung, W, Han, CE, Fava, M, Mischoulon, D, Papakostas, GI, Heo, JY, et al. Reduced frontal-subcortical white matter connectivity in association with suicidal ideation in major depressive disorder. Transl Psychiatry. (2016) 6:e835. doi: 10.1038/tp.2016.110
92. Olvet, DM, Peruzzo, D, Thapa-Chhetry, B, Sublette, ME, Sullivan, GM, Oquendo, MA, et al. A diffusion tensor imaging study of suicide attempters. J Psychiatr Res. (2014) 51:60–7. doi: 10.1016/j.jpsychires.2014.01.002
93. Wei, S, Womer, FY, Edmiston, EK, Zhang, R, Jiang, X, Wu, F, et al. Structural alterations associated with suicide attempts in major depressive disorder and bipolar disorder: a diffusion tensor imaging study. Prog Neuro-Psychopharmacol Biol Psychiatry. (2020) 98:109827. doi: 10.1016/j.pnpbp.2019.109827
94. Bijttebier, S, Caeyenberghs, K, van den Ameele, H, Achten, E, Rujescu, D, Titeca, K, et al. The vulnerability to suicidal behavior is associated with reduced connectivity strength. Front Hum Neurosci. (2015) 9:632. doi: 10.3389/fnhum.2015.00632
95. Hwang, J, Legarreta, M, Bueler, CE, DiMuzio, J, McGlade, E, Lyoo, IK, et al. Increased efficiency of brain connectivity networks in veterans with suicide attempts. Neuroimage Clin. (2018) 20:318–26. doi: 10.1016/j.nicl.2018.04.021
96. Chen, V, Kao, C-J, Tsai, Y-H, McIntyre, RS, and Weng, J-C. Mapping brain microstructure and network alterations in depressive patients with suicide attempts using generalized Q-sampling MRI. J Pers Med. (2021) 11:174. doi: 10.3390/jpm11030174
97. Chen, VC-H, Kao, C-J, Tsai, Y-H, Cheok, MT, McIntyre, RS, and Weng, J-C. Assessment of disrupted brain structural connectome in depressive patients with suicidal ideation using generalized Q-sampling MRI. Front Hum Neurosci. (2021) 15:711731. doi: 10.3389/fnhum.2021.711731
99. Desmyter, S, Bijttebier, S, and Heeringen, K. The role of neuroimaging in our understanding of the suicidal brain. CNS Neurol Disord Drug Targets. (2013) 12:921–9. doi: 10.2174/18715273113129990093
100. Zhang, H, Chen, Z, Jia, Z, and Gong, Q. Dysfunction of neural circuitry in depressive patients with suicidal behaviors: a review of structural and functional neuroimaging studies. Prog Neuro-Psychopharmacol Biol Psychiatry. (2014) 53:61–6. doi: 10.1016/j.pnpbp.2014.03.002
101. Martin, PC, Zimmer, TJ, and Pan, LA. Magnetic resonance imaging markers of suicide attempt and suicide risk in adolescents. CNS Spectr. (2014) 20:355–8. doi: 10.1017/S1092852915000048
102. Sher, L . Functional magnetic resonance imaging in studies of the neurobiology of suicidal behavior in adolescents with alcohol use disorders. Int J Adolesc Med Health. (2007) 19:11–8. doi: 10.1515/IJAMH.2007.19.1.11
103. van Heeringen, K, Bijttebier, S, Desmyter, S, Vervaet, M, and Baeken, C. Is there a neuroanatomical basis of the vulnerability to suicidal behavior? A coordinate-based meta-analysis of structural and functional MRI studies. Front Hum Neurosci. (2014) 8:824. doi: 10.3389/fnhum.2014.00824
104. Desmyter, S, van Heeringen, C, and Audenaert, K. Structural and functional neuroimaging studies of the suicidal brain. Prog Neuro-Psychopharmacol Biol Psychiatry. (2011) 35:796–808. doi: 10.1016/j.pnpbp.2010.12.026
105. Bani-Fatemi, A, Tasmim, S, Graff-Guerrero, A, Gerretsen, P, Strauss, J, Kolla, N, et al. Structural and functional alterations of the suicidal brain: an updated review of neuroimaging studies. Psychiatry Res Neuroimaging. (2018) 278:77–91. doi: 10.1016/j.pscychresns.2018.05.008
106. Olié, E, Ding, Y, Le Bars, E, de Champfleur, NM, Mura, T, Bonafé, A, et al. Processing of decision-making and social threat in patients with history of suicidal attempt: a neuroimaging replication study. Psychiatry Res Neuroimaging. (2015) 234:369–77. doi: 10.1016/j.pscychresns.2015.09.020
107. Simons, LE, Elman, I, and Borsook, D. Psychological processing in chronic pain: a neural systems approach. Neurosci Biobehav Rev. (2014) 39:61–78. doi: 10.1016/j.neubiorev.2013.12.006
108. Kross, E, Berman, MG, Mischel, W, Smith, EE, and Wager, TD. Social rejection shares somatosensory representations with physical pain. Proc Natl Acad Sci U S A. (2011) 108:6270–5. doi: 10.1073/pnas.1102693108
109. Eisenberger, NI . The pain of social disconnection: examining the shared neural underpinnings of physical and social pain. Nat Rev Neurosci. (2012) 13:421–34. doi: 10.1038/nrn3231
110. Olié, E, and Courtet, P. Interest of neuroimaging of social exclusion in suicide. J Neurosci Res. (2020) 98:581–7. doi: 10.1002/jnr.24314
111. Friston, KJ . Functional and effective connectivity: a review. Brain Connect. (2011) 1:13–36. doi: 10.1089/brain.2011.0008
112. Bajaj, S, Lamichhane, B, Adhikari, BM, and Dhamala, M. Amygdala mediated connectivity in perceptual decision-making of emotional facial expressions. Brain Connect. (2013) 3:386–97. doi: 10.1089/brain.2013.0145
113. Bajaj, S, and Killgore, WDS. Association between emotional intelligence and effective brain connectome: a large-scale spectral DCM study. NeuroImage. (2021) 229:117750. doi: 10.1016/j.neuroimage.2021.117750
114. Bajaj, S, Housley, SN, Wu, D, Dhamala, M, James, GA, and Butler, AJ. Dominance of the unaffected hemisphere motor network and its role in the behavior of chronic stroke survivors. Front Hum Neurosci. (2016) 10:650. doi: 10.3389/fnhum.2016.00650
115. Killgore, WDS, Vanuk, JR, Shane, BR, Weber, M, and Bajaj, S. A randomized, double-blind, placebo-controlled trial of blue wavelength light exposure on sleep and recovery of brain structure, function, and cognition following mild traumatic brain injury. Neurobiol Dis. (2020) 134:104679. doi: 10.1016/j.nbd.2019.104679
116. Bajaj, S, Butler, AJ, Drake, D, and Dhamala, M. Brain effective connectivity during motor-imagery and execution following stroke and rehabilitation. Neuroimage Clin. (2015) 8:572–82. doi: 10.1016/j.nicl.2015.06.006
Keywords: suicidal thoughts and behavior, depression, review, clinical relevance, task-based fMRI, resting state fMRI, brain morphometry, diffusion tensor imaging
Citation: Dobbertin M, Blair KS, Carollo E, Blair JR, Dominguez A and Bajaj S (2023) Neuroimaging alterations of the suicidal brain and its relevance to practice: an updated review of MRI studies. Front. Psychiatry. 14:1083244. doi: 10.3389/fpsyt.2023.1083244
Received: 28 October 2022; Accepted: 04 April 2023;
Published: 27 April 2023.
Edited by:
Çiçek Hocaoğlu, Recep Tayyip Erdoğan University, TürkiyeReviewed by:
Karl Friston, University College London, United KingdomCopyright © 2023 Dobbertin, Blair, Carollo, Blair, Dominguez and Bajaj. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Matthew Dobbertin, bWF0dGhldy5kb2JiZXJ0aW5AYm95c3Rvd24ub3Jn
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.