
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Psychiatry , 07 July 2022
Sec. Addictive Disorders
Volume 13 - 2022 | https://doi.org/10.3389/fpsyt.2022.947603
This article is part of the Research Topic Women in Psychiatry 2022: Addictive Disorders View all 5 articles
Background: Overdoses caused by synthetic mu-opioid receptor (MOR) agonists such as fentanyl are causing increasing mortality in the United States. The COVID-19 pandemic continues to have complex effects on public health, including opioid use disorders (OUD). It is unclear whether recent increases in mortality caused by synthetic opioids have reached a plateau (i.e., a stable period), after the onset of the COVID-19 pandemic.
Method: This study examined provisional overdose mortality data from the Centers for Disease Control and Prevention, for synthetic opioids excluding methadone (code T40.4; monthly data available from 39 States, plus New York City and Washington DC), for June 2019–November 2021. Data were first examined as crude mortality rates. The presence of a maximum plateau was analyzed for the last 4 months of available data. For authorities in which a plateau in mortality was detected, sigmoidal Boltzmann equations were used to model parameters of this phenomenon (e.g., level of the plateau).
Results: At the end of the study period, all but one authority (New Hampshire) reported increases in mortality rates for synthetic opioids, compared to the baseline month of June 2019 (range: 111–745% of baseline). A plateau was observed over the last 4 months of the study period (Aug 2021–Nov 2021) in 29 of the authorities. Ten other authorities had not reached a stable plateau at the end of the study period. For the authorities where a plateau was detected, a sigmoidal Boltzmann model revealed a fitted maximum of 262% rise in mortality over the study period, from the baseline month. The midpoint in the rise in mortality was fitted in September 2020. After separation of data into census regions, the highest plateau was observed in the West region, followed by South, Midwest, and Northeast (fitted plateau values were 409, 262, 204, and 149% of baseline, respectively).
Discussion: There were increases in overdose mortality due to synthetic opioids across most states, ranging considerably in magnitude. A plateau in overdose mortality was detected at the end of the study period in most of these authorities. The reasons for these plateaus should be explored, in order to develop optimized public health interventions.
Starting with its global spread from early 2020, the COVID-19 pandemic has caused complex public health challenges. Most public health measures (e.g., lockdowns) in industrialized countries such as the United States commenced in March 2020 (1). Among the reported comorbidities with COVID-19 is opioid use disorder (OUD) (2). The COVID-19 pandemic could potentially affect different stages in the trajectory of OUD and its medical care (3–5). OUD is caused by self-exposure to mu-opioid receptor (MOR) agonists, such as fentanyl derivatives, heroin, and prescription opioids (e.g., oxycodone). MOR-agonist overdose causes mortality primarily by centrally mediated respiratory depression (6–9). Fentanyl analogs have other deleterious effects (e.g., chest rigidity and laryngospasm), which may further increase their potential to cause morbidity and mortality (10–13).
Even prior to the COVID-19 pandemic, increases in opioid-induced overdose mortality were reported (14–16). For example, a recent wave in 2014–2017 was thought to be due to fentanyl derivatives, heroin and prescription opioids (17). The increasing presence of synthetic opioids in the illicit drug supply has resulted in a substantial increase in overdoses and mortality in the United States over the last several years (18–22). Consistent with this, data indicate that synthetic opioids (e.g., fentanyl derivatives), as opposed to other MOR-agonists such as heroin, are primarily responsible for the most recent increases in mortality in the United States, after the onset of COVID-19 (14, 23, 24).
The time-dependent increase in opioid-induced overdoses after the onset of COVID-19 was recently examined in 11 states (ending in August–December 2020) (25). This study reported heterogeneous profiles across these 11 states, for different kinds of opioids, including potential stabilizing trends in some states (i.e., tending toward a plateau). Another study reported on the increases in suspected opioid overdoses visits to ED in four states after onset of the COVID-19 lockdowns, ending in August 2020 (5). Other studies have also reported increases in all drug overdoses in individual states and counties in the onset of the COVID-19 pandemic (26–29).
The present study extends and complements the above reports by examining U.S. Centers for Disease Control (CDC) data from a larger national sample (39 states, plus New York City and Washington DC), on mortality due to synthetic opioids, over a more prolonged period after the onset of COVID-19 (starting June 2019 and ending November 2021). Furthermore, this study directly focused on whether plateaus in mortality have occurred across states (i.e., whether mortality levels have stabilized). Plateaus are important to examine in epidemiological studies, as they may provide information on continuing common sources of risk (30), as well as heterogeneity in behavioral and decision-making processes in the populations (31–34).
Provisional overdose monthly death counts from the U.S. Centers for Diseases Control and Prevention (CDC) were examined, for the 12-month periods ending June 2019 to November 2021 (therefore, covering a period before and after the onset of COVID-19 in the United States). The selected starting month (June 2019) provided 8 months of pre-COVID-19 data, to characterize a potential minimum plateau, important for non-linear sigmoidal regression analyses. The selected ending month was the latest available at the time of download (20 April 2022; Vital Statistics Rapid Release).1 We examined data for the authorities that report overdose deaths caused by “synthetic opioid excluding methadone” (category T40.4) (23). This category contains primarily fentanyl and its analogs, and can potentially include MOR-agonists from other synthetic scaffolds (e.g., analogs of etonitazene and U47700), although the latter are thought to be less common (16, 24). All data from an authority was excluded from non-linear regressions if it had more than two consecutive missing monthly values, or if values for the baseline month (June 2019) were not available.
Data for each 12-month ending period were first plotted per authority over the study period, expressed as crude mortality rate/million of population (to the nearest 0.1 million, based on U.S. Census numbers). Data were not cumulated across authorities, due to the possibility of differences in reporting standards (35, 36). For better visualization, separate figures were plotted for the four Census regions (i.e., West, Midwest, South, and Northeast).
Absolute baseline values (June 2019) differed widely across authorities, and the remaining calculations were made after normalizing each data set’s monthly data as a percent of its baseline month. For each data set, a maximum plateau was defined to occur if each of values for the last 4 months (i.e., August 2021–November 2021) was within ±5% of: (mean value for the last 4 months/baseline month). This therefore takes into account the differing magnitudes of increases relative to each authority’s baseline.
Sigmoidal equations (including the sigmodal Boltzmann model) have been used for epidemiological and opioid clinical pharmacology studies (32, 37, 38). The sigmoidal Boltzmann model was used to fit minimum and maximum (i.e., bottom and top) plateaus for mortality due to synthetic opioids, and the midpoint of the rise in mortality (23, 25). Data sets from the 29 authorities which had a detected maximum plateau in the last 4 months of the study period (as defined above; Table 1) were entered in an overall Boltzmann sigmoidal equation model, using GraphPad Prism (V.9) software. The equation was: mortality = [lower plateau + (top plateau - bottom plateau)]/1 + exp [(month of midpoint of top - bottom) - month]/slope].2 The regression was weighted by 1/Y2, to account for larger variation in the later months of the study period. The minimum plateau was constrained to be >0%. The main fitted values of interest were therefore the maximum plateau (±95%CI) and the month of the midpoint between the minimum and maximum plateau (i.e., to determine when the rise in mortality occurred). The minimum plateau is not reported here, as the values were normalized as percent of baseline. This overall sigmoidal model was followed by separate models for the states in the four Census regions, with identical methods.
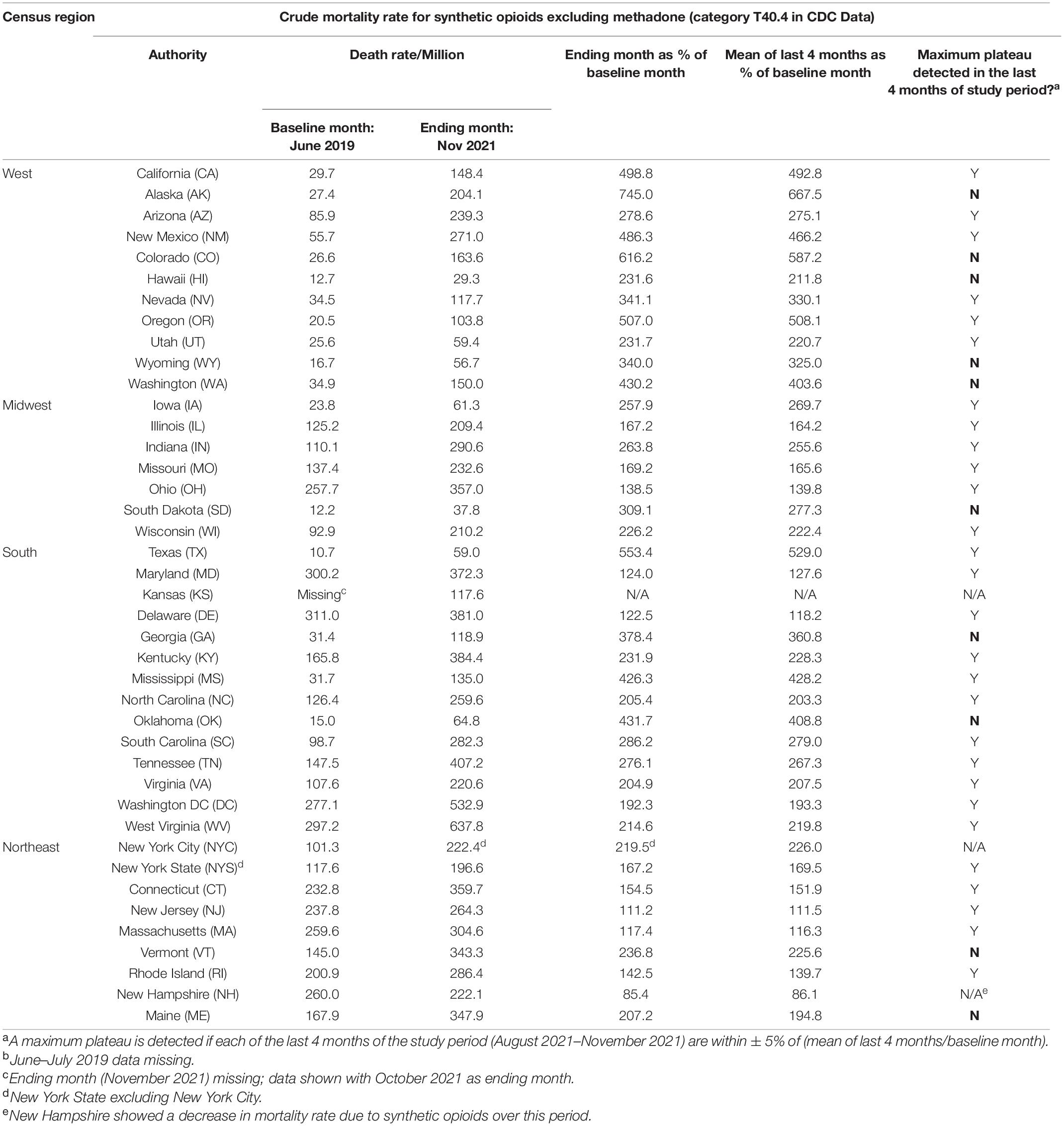
Table 1. Mortality rate for overdoses caused by synthetic opioids, before and after the onset of COVID-19.
Provisional overdose deaths due to synthetic opioids excluding methadone were analyzed, before and after the onset of COVID-19 in the United States (June 2019–November 2021). Monthly crude mortality rates/million are shown in Figure 1, separated by Census region. There was a considerable variation in baseline mortality rate (Table 1 and Figure 1). Of the 41 authorities studied, only New Hampshire showed a decrease in mortality rate over the study period (to 85% of baseline; Table 1). All other authorities reported an increase in mortality over the study period, and there was a considerable variation in the magnitude of time-dependent increases compared to each baseline (i.e., June 2019; Table 1 and Figure 1). Due to this variation, the potential sigmodal profile of some curves is less easily discernible than others, on a common y-axis axis. As an illustration, the four States in the South census region with lower absolute values are re-plotted on a compressed y-axis (Figure 2, compare to Figure 1, lower right panel).
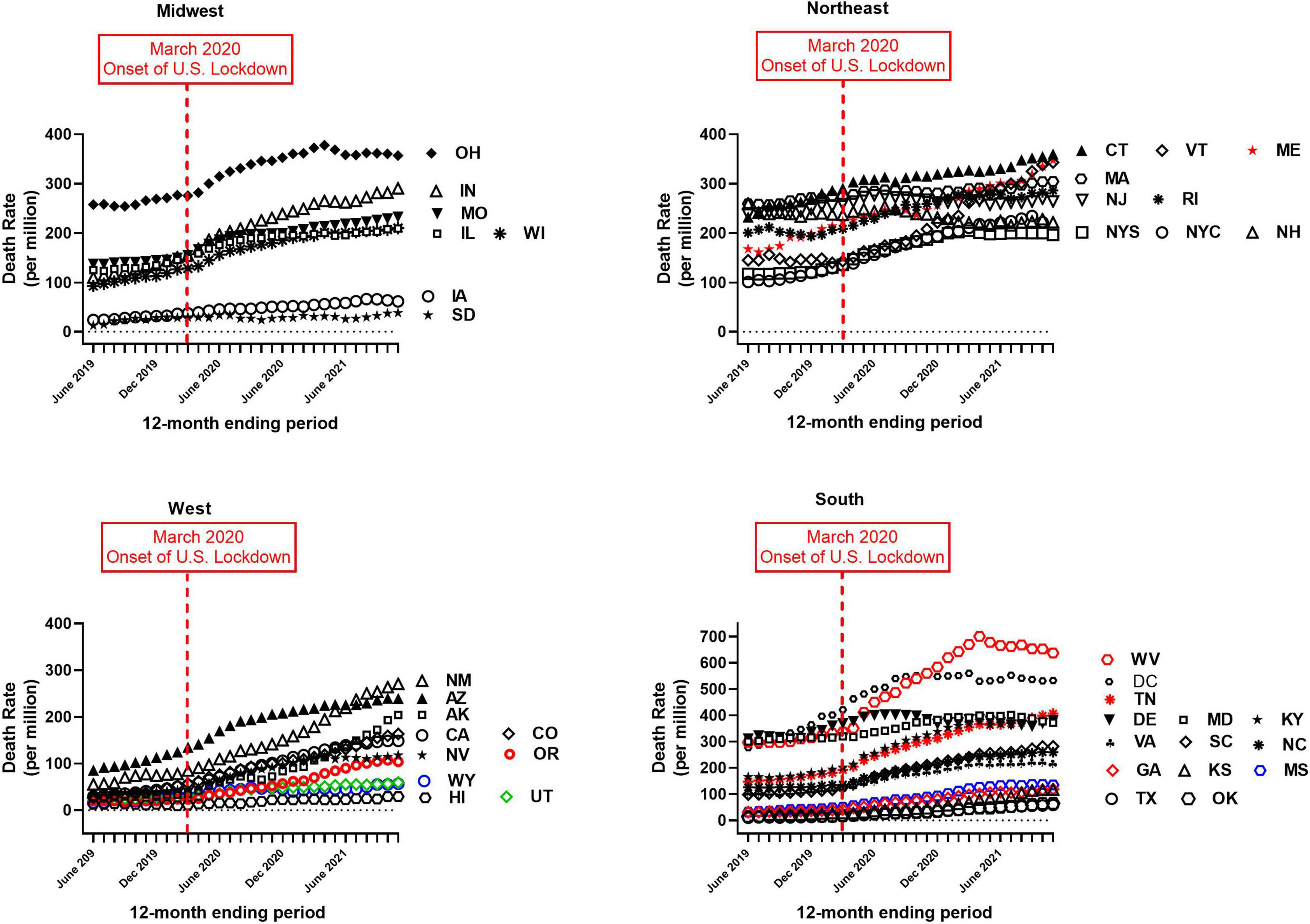
Figure 1. Crude monthly mortality rate (per million), due to “synthetic opioids excluding methadone,” for June 2019–November 2021. X-axes: month; Y-axes: mortality rate (per million population). Different panels show data separated by Census region (Midwest, Northeast, West and South). Note different Y-axis scale in the panel for the South region (lower right). Labels for each authority are placed near the ending month data for each curve, to aid visualization. Abbreviations for authorities are defined in Table 1.
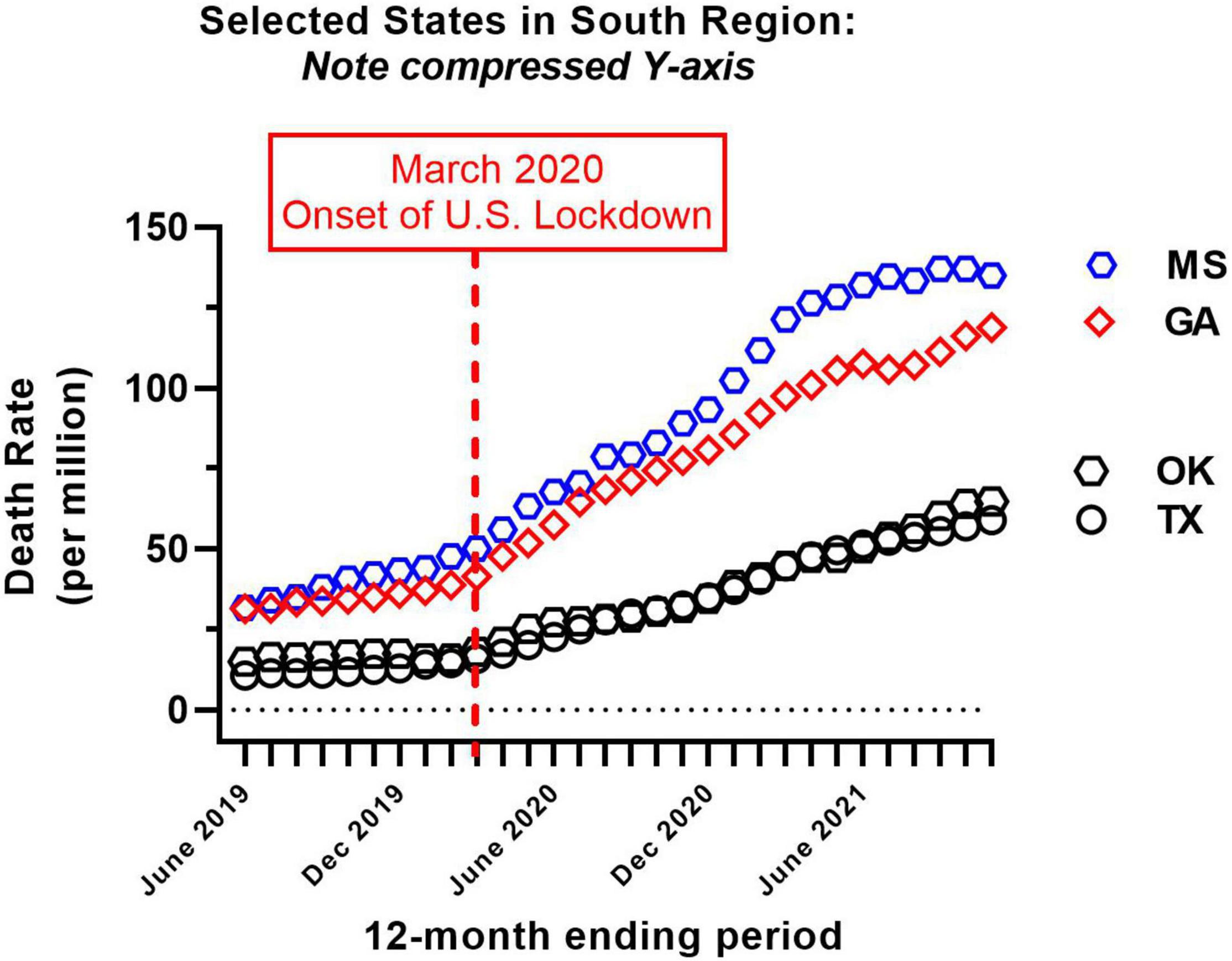
Figure 2. Re-plot of crude mortality rate of the four states from the South census region with the lowest absolute values (i.e., see Figure 1, lower right panel, for comparison). Note compressed Y-axis range; all other details as in Figure 1.
Crude mortality rates were then normalized as a percent of each data set’s baseline month (June 2019), and are presented in Table 1. As mentioned above, as of the final month of the study period (Nov 2021), only New Hampshire had a decrease (85% of baseline). All other data sets exhibited increases by the end of the study period, ranging in magnitude from 111 to 645% of baseline (New Jersey and Alaska, respectively).
The presence of a maximum plateau (i.e., a period of stability) in the last 4 months of the study period was examined for each data set. There were a total of 39 data sets which could be analyzed (New York City and Kansas were excluded due to missing data in some cells). Of these 39 data sets, 29 showed a maximum plateau as defined in the Methods (Table 1). By contrast, data sets from 10 states did not exhibit a maximum plateau in the last 4 months of the study; these were: Alaska, Colorado, Hawaii, Wyoming, Washington, South Dakota, Georgia, Oklahoma, Vermont, and Maine.
In order to examine the plateau phenomenon further, we entered data from the 29 authorities (28 states and Washington DC) with a detected maximum plateau (see Table 1) into an overall regression, as replicates. For this overall regression, the data fit a sigmoidal Boltzmann equation with a high weighted R2 (0.99) (Figure 3, left panel). The fitted maximum plateau for this overall regression was 262% of baseline (95%CI: 255–271%) (Figure 3, left panel). The fitted mid-point between the minimum and maximum plateau was detected in September 2020 (Table 2). In a follow up analysis, we divided these 29 authorities into their four respective Census regions, and carried out separate regressions (Figure 3, right panel; Table 2). These four regressions revealed that the regions had different maximum plateau levels, based on lack of overlap in their weighted 95%CI (Table 2). The region with the highest plateau was the West, followed in descending order by the South, Midwest and Northeast. These region-specific regressions also exhibited high weighted R2 (≈0.99).
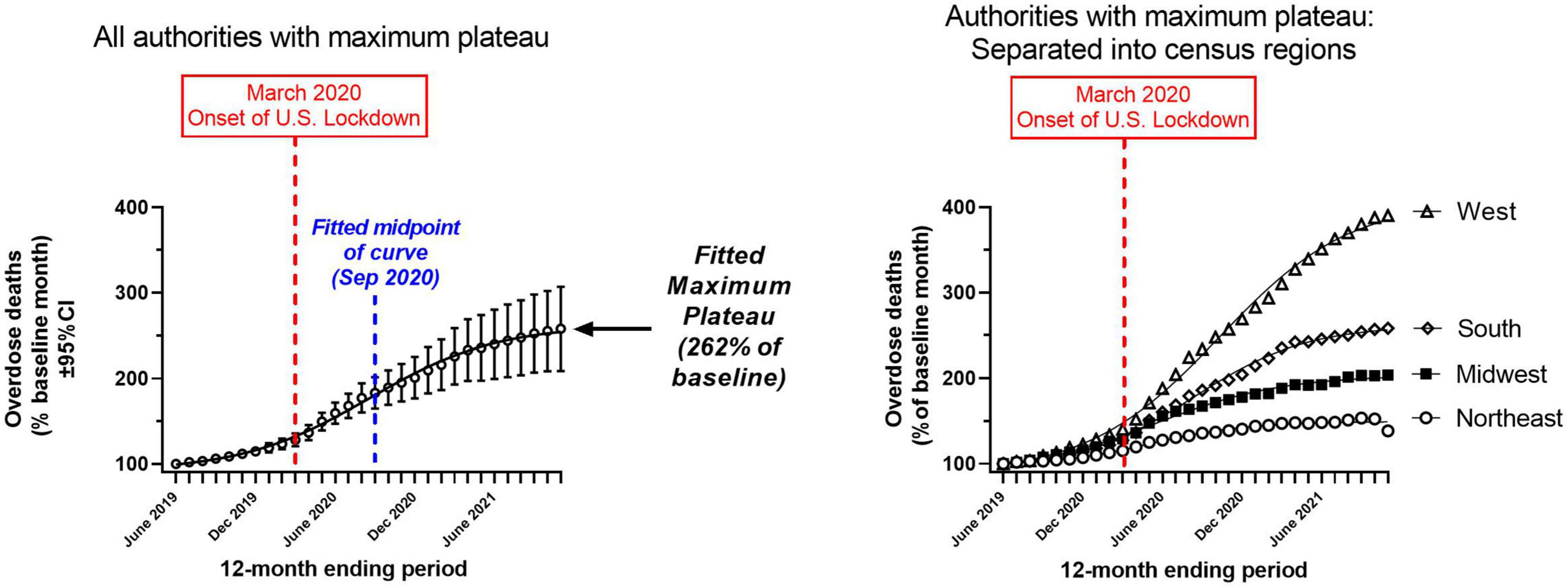
Figure 3. Sigmoidal regressions of mortality, normalized as percent of the baseline month (June 2019) for the 29 authorities which had a maximum plateau in the last 4 months of the study period (August 2021–November 2021; Table 1). Left panel: Overall regression for these 29 authorities. Right panel: Separate regressions for four census regions; confidence intervals not shown. X-axes: month; Y-axes: mortality rate (percent of baseline month). Table 2 shows the parameters of the sigmoidal fits.
Several studies have reported an increase in opioid-induced mortality after the onset of COVID-19 in the United States (commencing in March 2020), largely ascribed to synthetic MOR-agonists such as fentanyl analogs (10, 23, 24). Mortality had also been rising in prior years (e.g., a wave in 2014 onwards) due to synthetic MOR-agonists, heroin and prescription opioids (11, 14, 17, 23, 39).
As expected, nearly all of the 41 authorities under study exhibited an increase in mortality due to synthetic opioids after the onset of the COVID-19 lockdowns in March 2020. One authority only (New Hampshire) showed a decrease at the end of the study period. This study detected that 29 of these authorities exhibited a plateau in mortality caused by synthetic opioids, defined as stability in the last 4 months of this period (August 2021–November 2021). The data also show that 10 authorities had not exhibited such a plateau at the end of the study period.
A large variability in baseline mortality due to synthetic opioids and the magnitude of the post-COVID-19 increases was noted across authorities and regions. Overall, the highest plateau was observed in the West Census region, followed in descending order by South, Midwest and Northeast. Variability in these profiles is consistent with recent reports focusing on all opioid overdoses (i.e., not only those due to synthetic opioids), for selected states (25, 26, 40). Future studies could examine the relationship between different relative increases in mortality across regions and States, and diverse public health measures enacted after the onset of COVID-19. Pre-existing differences across regions and States could also be important factors, based on variations in health systems, socioeconomics, demographics and frequency of illicit synthetic opioids in the drug supply (41–46).
Using the pre-pandemic period from June 2019 as baseline, the sigmoidal regression models in this study document an increase in mortality due to synthetic opioids in the United States after the onset of COVID-19 (in March 2020 onwards). For the overall regression, the midpoint of this increase was detected in September 2020, approximately 6 months after the onset of the lockdowns in the United States. The increase of such overdoses during the early pandemic may have been due to changes in supply, distribution or demand, for illicit opioids and other drugs (41, 47). Increased adulteration with fentanyl analogs in different types of illicit drug products (including both opioid and non-opioid compounds) is another potential cause of this trend (39, 45, 48). Behavioral changes in the early pandemic period, such as injecting alone, or refusal of transport to the ED may have also contributed to this rise in mortality (5, 49).
The presence of plateaus in epidemiological studies has been ascribed to different phenomena, including the presence of a “continuing common source,” which in this case could be the extensive penetration of synthetic opioids in the illicit drug supply (30, 39). Other factors that have been recently examined in such plateaus include behaviorally based heterogeneity within populations, and behavioral change due to awareness of risk (31, 34).
It cannot be excluded that the plateaus observed at the end of the study period in 29 authorities are temporary, and that further increases may occur in the future (17, 31, 34, 50). Only one state examined here, New Hampshire, showed a decrease in mortality due to synthetic opioids, compared to baseline. A slight time-dependent decrease was also observed for New Hampshire for mortality due to all opioids (T40.0–T40.4, T40.6), not only synthetic opioids studied here (not shown).
The analysis for “synthetic opioids excluding methadone” (category T40.4) did not include all States, as some do not report this category separately. Nevertheless, this study included a large sample of reporting authorities, representing all census regions in the United States. These CDC mortality data are considered provisional, and have lag times for reporting (51); there are also potential differences in reporting standards across authorities (36), further justifying normalization by the local baseline. However, completeness is relatively high, and these data are among the most timely indicators of overdose mortality across the United States (see text footnote 1).
The available data do not allow differentiation of which specific synthetic opioid (e.g., fentanyl or an analog thereof) was detected (24). Also, these data sets do not imply that “synthetic opioids other than methadone” were the only drugs detected in the decedents (52). However synthetic opioids are thought to be the compounds primarily underlying the increases in mortality observed after the onset of COVID-19 (14, 41). For illustration, California (the most populous State) had a decrease in overdose mortality with heroin (T40.1) in the last month of the study period compared to baseline (to 80.9% of baseline; not shown). By contrast, there had been a robust increase in overdose mortality with “synthetic opioids other than methadone” (i.e., 498.8% of baseline; Table 1).
Because there was a broad variation across authorities in the crude mortality rate at baseline (i.e., June 2019), data were normalized data as percent of each baseline, for sigmoidal non-linear regressions. However a similar sigmoidal curve could also be fit on crude mortality rates (not shown), therefore the models shown here were not simply an artifact of normalization. Modeling of these monthly data with a sigmoidal Boltzmann equation was justified based on the observed profiles in most of the authorities in this study, and has been used to examine other epidemiological variables (32, 37). Nevertheless, equations other than the sigmoidal Boltzmann could be used in the future to explore this potential plateau phenomenon. In the sigmoidal models shown here, we included only authorities that had individually shown a stable maximum plateau in the last 4 months of the study period. As a follow-up, we also carried out a further Boltzmann model including all data sets without exclusion. Results of this equation were also fit to a sigmoidal model with high R2, and yielded similar maximum plateau to that reported above (not shown). Therefore, the fitted sigmoidal models here were not likely to be an artifact of the exclusion of the data sets that had not individually reached a plateau.
The COVID-19 pandemic has potentially affected different stages in OUD, including initiation, escalation, overdoses, and engagement with clinical care (3, 5, 21). We confirm here increases in mortality rates due to synthetic opioids after the onset of COVID-19 in the United States, which varied considerably in magnitude across census regions and the broad array of states examined here. This study shows that most, but not all, of the authorities had reached a stable plateau in August to November 2021. The magnitude of the observed plateau was the highest for the West census region, followed in descending order by South, Midwest and Northeast. Differing plateau levels in specific parts of the United States may be due to patterns of opioid initiation and use, but also use of other drugs, as illicit synthetic opioids can be mixed in the drug supply for ostensibly non-opioid drugs (14, 39, 53). The lack of observable plateaus at the end of the study period in 10 of the states studied here is also worthy of investigation, given the potential risk of further increases. Overall, future studies should explore the reasons underlying these divergent plateau profiles across states, as a potential guide for locally optimized prevention, mitigation and intervention approaches.
Publicly available datasets were analyzed in this study. This data can be found here: https://www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm.
All authors listed have made a substantial, direct, and intellectual contribution to the work, and approved it for publication.
This work was supported by NIH-NIDA (DA 018151), and NIH-National Institute on Allergy and Diseases NIH-NIAID (5P30 AI124414). This work was also supported by the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation. Funding sources had no role in the planning, writing, or submission of this publication.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
95%CI, 95% confidence interval; COVID-19, Coronavirus disease 2019; CDC, U.S. Centers for Disease Control and Preventions; ED, Emergency Department; MOR, mu-opioid receptor; OUD, opioid use disorder.
1. Kates J, Michaud J, Tolbert J. Stay-At-Home Orders to Fight COVID-19 in the United States: The Risks of a Scattershot Approach. (2020). Available online at: https://www.kff.org/policy-watch/stay-at-home-orders-to-fight-covid19/ (accessed June 7, 2021).
2. Wang QQ, Kaelber DC, Xu R, Volkow ND. COVID-19 risk and outcomes in patients with substance use disorders: analyses from electronic health records in the United States. Mol Psychiatry. (2020) 26:40. doi: 10.1038/s41380-020-00880-7
3. Bart G, Wastvedt S, Hodges JS, Rosenthal R. Did drug use increase following COVID-19 relaxation of methadone take-out regulations? 2020 was a complicated year. J Subst Abuse Treat. (2022) 133:108590. doi: 10.1016/j.jsat.2021.108590
4. Nunes EV, Levin FR, Reilly MP, El-Bassel N. Medication treatment for opioid use disorder in the age of COVID-19: can new regulations modify the opioid cascade? J Subst Abuse Treat. (2021) 122:108196. doi: 10.1016/j.jsat.2020.108196
5. Root ED, Slavova S, LaRochelle M, Feaster DJ, Villani J, Defiore-Hyrmer J, et al. The impact of the national stay-at-home order on emergency department visits for suspected opioid overdose during the first wave of the COVID-19 pandemic. Drug Alcohol Depend. (2021) 228:108977. doi: 10.1016/j.drugalcdep.2021.108977
6. Ding H, Trapella C, Kiguchi N, Hsu FC, Caló G, Ko MC. Functional profile of systemic and intrathecal cebranopadol in nonhuman primates. Anesthesiology. (2021) 135:482–93. doi: 10.1097/aln.0000000000003848
7. France CP, Ahern G, Averick S, Disney A, Enright HA, Esmaeli-Azad B, et al. Countermeasures for preventing and treating opioid overdose. Clin Pharmacol Ther. (2020) 109:578–90. doi: 10.1002/cpt.2098
8. Liu S, Kim DI, Oh TG, Pao GM, Kim JH, Palmiter RD, et al. Neural basis of opioid-induced respiratory depression and its rescue. Proc Natl Acad Sci USA. (2021) 118:e2022134118. doi: 10.1073/pnas.2022134118
9. Yang PK, Weinger MB, Negus SS. Elucidation of dose-effect relationships for different opiate effects using alfentanil in the spontaneously ventilating rat. Anesthesiology. (1992) 77:153–61. doi: 10.1097/00000542-199207000-00022
10. Britch SC, Walsh SL. Treatment of opioid overdose: current approaches and recent advances. Psychopharmacology (Berl). (2022) 239:2063–81. doi: 10.1007/s00213-022-06125-5
11. Comer SD, Cahill CM. Fentanyl: receptor pharmacology, abuse potential, and implications for treatment. Neurosci Biobehav Rev. (2019) 106:49–57. doi: 10.1016/j.neubiorev.2018.12.005
12. Lovrecic B, Lovrecic M, Gabrovec B, Carli M, Pacini M, Maremmani AGI, et al. Non-medical use of novel synthetic opioids: a new challenge to public health. Int J Environ Res Public Health. (2019) 16:177. doi: 10.3390/ijerph16020177
13. Schifano F, Chiappini S, Corkery JM, Guirguis A. Assessing the 2004-2018 fentanyl misusing issues reported to an international range of adverse reporting systems. Front Pharmacol. (2019) 10:46. doi: 10.3389/fphar.2019.00046
14. CDC. Increase in Fatal Drug Overdoses Across the United States Driven by Synthetic Opioids Before and During the COVID-19 Pandemic. Atlanta, GA: CDC (2020).
15. Rudd RA, Aleshire N, Zibbell JE, Gladden RA. Increases in drug and opioid overdose deaths — United States, 2000–2014. Morb Mortal Wkly Rep. (2015) 64:1378–82.
16. Seth P, Rudd RA, Noonan RK, Haegerich TM. Quantifying the epidemic of prescription opioid overdose deaths. Am J Public Health. (2018) 108:500–2. doi: 10.2105/ajph.2017.304265
17. Ciccarone D. The triple wave epidemic: supply and demand drivers of the US opioid overdose crisis. Int J Drug Policy. (2019) 71:183–8. doi: 10.1016/j.drugpo.2019.01.010
18. Cicero TJ, Ellis MS, Kasper ZA. Increases in self-reported fentanyl use among a population entering drug treatment: the need for systematic surveillance of illicitly manufactured opioids. Drug Alcohol Depend. (2017) 177:101–3. doi: 10.1016/j.drugalcdep.2017.04.004
19. Henry BF, Mandavia AD, Paschen-Wolff MM, Hunt T, Humensky JL, Wu E, et al. COVID-19, mental health, and opioid use disorder: old and new public health crises intertwine. Psychol Trauma. (2020) 12:S111–2. doi: 10.1037/tra0000660
20. Kosten TR, Petrakis IL. The hidden epidemic of opioid overdoses during the coronavirus disease 2019 pandemic. JAMA Psychiatry. (2021) 78:585–6. doi: 10.1001/jamapsychiatry.2020.4148
21. Volkow ND. Collision of the COVID-19 and addiction epidemics. Ann Intern Med. (2020) 173:61–2. doi: 10.7326/m20-1212
22. Wakeman SE, Green TC, Rich J. An overdose surge will compound the COVID-19 pandemic if urgent action is not taken. Nat Med. (2020) 26:819–20. doi: 10.1038/s41591-020-0898-0
23. Ahmad FB, Rossen LM, Sutton P. Provisional Drug Overdose Death Counts. Hyattsville, MD: National Center for Health Statistics (2021).
24. Skolnick P. Treatment of overdose in the synthetic opioid era. Pharmacol Ther. (2021) 233:108019. doi: 10.1016/j.pharmthera.2021.108019
25. Garcia GP, Stringfellow EJ, DiGennaro C, Poellinger N, Wood J, Wakeman S, et al. Opioid overdose decedent characteristics during COVID-19. Ann Med. (2022) 54:1081–8. doi: 10.1080/07853890.2022.2067350
26. Burgess-Hull AJ, Smith KE, Panlilio LV, Schriefer D, Preston KL, Alter A, et al. Nonfatal opioid overdoses before and after Covid-19: regional variation in rates of change. PLoS One. (2022) 17:e0263893. doi: 10.1371/journal.pone.0263893
27. Larson PS, Bergmans RS. Impact of the COVID-19 pandemic on temporal patterns of mental health and substance abuse related mortality in Michigan: an interrupted time series analysis. Lancet Reg Health Am. (2022) 10:100218. doi: 10.1016/j.lana.2022.100218
28. Macmadu A, Batthala S, Correia Gabel AM, Rosenberg M, Ganguly R, Yedinak JL, et al. Comparison of characteristics of deaths from drug overdose before vs during the COVID-19 pandemic in Rhode Island. JAMA Netw Open. (2021) 4:e2125538. doi: 10.1001/jamanetworkopen.2021.25538
29. Shreffler J, Shoff H, Thomas JJ, Huecker M. Brief report: the impact of COVID-19 on emergency department overdose diagnoses and county overdose deaths. Am J Addict. (2021) 30:330–3. doi: 10.1111/ajad.13148
31. Berestycki H, Desjardins B, Heintz B, Oury J-M. Plateaus, rebounds and the effects of individual behaviours in epidemics. Sci Rep. (2021) 11:18339. doi: 10.1038/s41598-021-97077-x
32. El Aferni A, Guettari M, Tajouri T. Mathematical model of Boltzmann’s sigmoidal equation applicable to the spreading of the coronavirus (Covid-19) waves. Environ Sci Pollut Res Int. (2021) 28:40400–8. doi: 10.1007/s11356-020-11188-y
33. Smaldino PE, Aplin LM, Farine DR. Sigmoidal acquisition curves are good indicators of conformist transmission. Sci Rep. (2018) 8:14015. doi: 10.1038/s41598-018-30248-5
34. Weitz JS, Park SW, Eksin C, Dushoff J. Awareness-driven behavior changes can shift the shape of epidemics away from peaks and toward plateaus, shoulders, and oscillations. Proc Natl Acad Sci USA. (2020) 117:32764–71. doi: 10.1073/pnas.2009911117
35. Slavova S, O’Brien DB, Creppage K, Dao D, Fondario A, Haile E, et al. Drug overdose deaths: let’s get specific. Public Health Rep. (2015) 130:339–42. doi: 10.1177/003335491513000411
36. Warner M, Paulozzi LJ, Nolte KB, Davis GG, Nelson LS. State variation in certifying manner of death and drugs involved in drug intoxication deaths. Acad Forensic Pathol. (2013) 3:231–7. doi: 10.23907/2013.029
37. Hartwig TS, Sørensen S, Jørgensen FS. The maternal age-related first trimester risks for trisomy 21, 18 and 13 based on Danish first trimester data from 2005 to 2014. Prenatal Diagnosis. (2016) 36:643–9. doi: 10.1002/pd.4833
38. Olofsen E, Boom M, Sarton E, van Velzen M, Baily P, Smith KJ, et al. Analgesic and respiratory depressant effects of R-dihydroetorphine: a pharmacokinetic-pharmacodynamic analysis in healthy male volunteers. Anesthesiology. (2019) 131:1327–39. doi: 10.1097/aln.0000000000002991
39. Fairbairn N, Coffin PO, Walley AY. Naloxone for heroin, prescription opioid, and illicitly made fentanyl overdoses: challenges and innovations responding to a dynamic epidemic. Int J Drug Policy. (2017) 46:172–9. doi: 10.1016/j.drugpo.2017.06.005
40. Holland KM, Jones C, Vivolo-Kantor AM, Idaikkadar N, Zwald M, Hoots B, et al. Trends in US emergency department visits for mental health, overdose, and violence outcomes before and during the COVID-19 pandemic. JAMA Psychiatry. (2021) 78:372–9. doi: 10.1001/jamapsychiatry.2020.4402
41. Ciccarone D. The rise of illicit fentanyls, stimulants and the fourth wave of the opioid overdose crisis. Curr Opin Psychiatry. (2021) 34:344–50. doi: 10.1097/yco.0000000000000717
42. Friedman J, Hansen H, Bluthenthal RN, Harawa N, Jordan A, Beletsky L. Growing racial/ethnic disparities in overdose mortality before and during the COVID-19 pandemic in California. Prev Med. (2021) 153:106845–106845. doi: 10.1016/j.ypmed.2021.106845
43. Friedman JR, Hansen H. Evaluation of Increases in drug overdose mortality rates in the us by race and ethnicity before and during the COVID-19 pandemic. JAMA Psychiatry. (2022) 79:379–81. doi: 10.1001/jamapsychiatry.2022.0004
44. Jalal H, Buchanich JM, Sinclair DR, Roberts MS, Burke DS. Age and generational patterns of overdose death risk from opioids and other drugs. Nat Med. (2020) 26:699–704. doi: 10.1038/s41591-020-0855-y
45. Kilmer B, Pardo B, Pujol TA, Caulkins JP. Rapid changes in illegally manufactured fentanyl products and prices in the United States. Addiction. (2022). doi: 10.1111/add.15942
46. Shover CL, Falasinnu TO, Dwyer CL, Santos NB, Cunningham NJ, Freedman RB, et al. Steep increases in fentanyl-related mortality west of the Mississippi River: recent evidence from county and state surveillance. Drug Alcohol Depend. (2020) 216:108314. doi: 10.1016/j.drugalcdep.2020.108314
47. Mars SG, Rosenblum D, Ciccarone D. Illicit fentanyls in the opioid street market: desired or imposed? Addiction. (2018) 114:774–80. doi: 10.1111/add.14474
48. Macmadu A, Carroll JJ, Hadland SE, Green TC, Marshall BD. Prevalence and correlates of fentanyl-contaminated heroin exposure among young adults who use prescription opioids non-medically. Addict Behav. (2017) 68:35–8. doi: 10.1016/j.addbeh.2017.01.014
49. Slavova S, Rock P, Bush HM, Quesinberry D, Walsh SL. Signal of increased opioid overdose during COVID-19 from emergency medical services data. Drug and Alcohol Depend. (2020) 214:108176. doi: 10.1016/j.drugalcdep.2020.108176
50. Jalal H, Burke DS. Carfentanil and the rise and fall of overdose deaths in the United States. Addiction. (2021) 116:1593–9. doi: 10.1111/add.15260
51. Spencer MR, Ahmad F. Timeliness of Death Certificate Data for Mortality Surveillance and Provisional Estimates. Washington, DC: National Center for Health Statistics (2016).
53. Bolinski RS, Walters S, Salisbury-Afshar E, Ouellet LJ, Jenkins WD, Almirol E, et al. The Impact of the COVID-19 pandemic on drug use behaviors, fentanyl exposure, and harm reduction service support among people who use drugs in rural settings. Int J Environ Res Public Health. (2022) 19:2230. doi: 10.3390/ijerph19042230
Keywords: COVID-19, overdose, opioid use disorder, fentanyl, plateau, opioid
Citation: Brown KG, Chen CY, Dong D, Lake KJ and Butelman ER (2022) Has the United States Reached a Plateau in Overdoses Caused by Synthetic Opioids After the Onset of the COVID-19 Pandemic? Examination of Centers for Disease Control and Prevention Data to November 2021. Front. Psychiatry 13:947603. doi: 10.3389/fpsyt.2022.947603
Received: 18 May 2022; Accepted: 17 June 2022;
Published: 07 July 2022.
Edited by:
Hossein Hassanian-Moghaddam, Shahid Beheshti University of Medical Sciences, IranReviewed by:
Scott Phillips, Washington Poison Center, United StatesCopyright © 2022 Brown, Chen, Dong, Lake and Butelman. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Eduardo R. Butelman, YnV0ZWxtZUByb2NrZWZlbGxlci5lZHU=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.