
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
REVIEW article
Front. Psychiatry, 24 November 2022
Sec. Social Neuroscience
Volume 13 - 2022 | https://doi.org/10.3389/fpsyt.2022.1054065
This article is part of the Research TopicReviews in Psychiatry 2022: Social NeuroscienceView all 4 articles
Despite its negative reputation, egoism – the excessive concern for one’s own welfare – can incite prosocial behavior. So far, however, egoism-based prosociality has received little attention. Here, we first provide an overview of the conditions under which egoism turns into a prosocial motive, review the benefits and limitations of egoism-based prosociality, and compare them with empathy-driven prosocial behavior. Second, we summarize studies investigating the neural processing of egoism-based prosocial decisions, studies investigating the neural processing of empathy-based prosocial decisions, and the small number of studies that compared the neural processing of prosocial decisions elicited by the different motives. We conclude that there is evidence for differential neural networks involved in egoism and empathy-based prosocial decisions. However, this evidence is not yet conclusive, because it is mainly based on the comparison of different experimental paradigms which may exaggerate or overshadow the effect of the different motivational states. Finally, we propose paradigms and research questions that should be tackled in future research that could help to specify how egoism can be used to enhance other prosocial behavior and motivation, and the how it could be tamed.
“The most disinterested love is, after all, but a kind of bargain, in which the dear love of our own selves always proposes to be the gainer some way or other” (1).
“We have only one relation to each other, that of usableness, of utility, of use. We owe each other nothing, for what I seem to owe you I owe at most to myself. If I show you a cheery air in order to cheer you likewise, then your cheeriness is of consequence to me, and my air serves my wish; to a thousand others, whom I do not aim to cheer, I do not show it” (2).
Egoism is commonly related to the “dark side” of human nature, that is, anti-social behavior such as hostility and aggression (3). It is defined as “the excessive concern with one’s own pleasure or advantage at the expense of community well-being” (4). Similarly defined terms that are often synonymous with egoism are selfishness, defined as “an inordinate focus on one’s own welfare, regardless of the well-being of others” (5), and egocentrism, defined as an “excessive interest in oneself and concern for one’s own welfare or advantage at the expense of or in disregard of others” (6). On the motivational level, an egoistic motive drives behavior with the ultimate goal of increasing one’s own welfare (7).
Interestingly, egoism has also been related to behaviors on the “bright side” of human nature such as prosocial behaviors that benefit others than oneself, including a variety of actions like helping, sharing, comforting and cooperating (8). There is evidence that an egoistic motive can drive prosocial behaviors, related to the expectation of a reward. These rewards can be financial incentives, or social rewards such as gain of a positive (i.e., prosocial) reputation, but also by the avoidance of punishment (9). It has been suggested that egoistic motives are promising for promoting the common good because they are easily aroused and are strongly motivating (10).
Another motive for prosocial behavior that is traditionally associated with the “bright side” of human nature is empathy. Empathy is a multidimensional construct (11) with many different definitions (12). Reflecting this multidimensionality, researchers commonly distinguish between affective empathy, i.e., the sharing of others emotions, and cognitive empathy, i.e., the sharing of others cognitive representations (ideas, thoughts, intentions (13–16). It is assumed that both, affective and cognitive empathy can incite prosocial behaviors (13, 17, 18), i.e., behaviors that maximize the outcome of another person at costs to oneself (19, 20). This claim is bolstered by many studies showing a consistent relationship between individual differences in empathy ratings and individual difference in prosocial decisions (19–23). Based on this evidence, an influential social psychology model has proposed that empathy motivates prosocial behavior, because it induces an “altruistic motivation” (24, 25). Building on this model, in this review we refer to empathy as a motive, i.e., a process that can motivate prosocial behavior, in agreement with other researchers of the field (19, 20, 26–28).
Empathy-induced prosocial behavior has been addressed in a number of previous review papers (29–32). In contrast, egoism-based prosociality has received considerably less attention. In the following, we review behavioral and neuroscientific evidence for prosocial behavior that is driven by egoism and discuss its advantages and disadvantages compared to empathy-driven prosocial behavior.
In general, behavior is rarely driven by a single motive and multiple motives hardly operate in isolation. There is broad evidence that different motives are often activated simultaneously and most complex behaviors are driven by the interaction between different motives (33). To understand and predict human behavior, it is necessary to investigate the interactions between different motives. However, based on behavioral observations this is challenging, because different motives can result in the identical behavioral outcome. For instance, the same prosocial decision can be driven by an egoistic motive, i.e., the expectation of a reward, or an empathy motive, i.e., the sharing of other’s emotions eliciting concern for the other’s welfare. Thus, motives and their interactions are not directly observable in overt behavior. Moreover, subjectively reported motives tend to be biased by social desirability (34).
Recent studies have started to use neuroscientific methods to distinguish different motives that result in the same behavior and to investigate multi-motive interactions (21, 22, 35–37). On a conceptual level, models of multi-motive interactions and their neural computation help to understand their impact on prosocial decisions. Practically, disentangling different motives and their interactions can shed light on the motivational basis of an observed behavior and inform interventions that foster or suppress certain behavioral outcomes. For example, differentiating between an egoistic and an empathy motive and/or investigating the interactions between both motivational forces can help improving incentive structures that shape individual prosocial behavior. The current review first provides a summary of the benefits and limitations of egoism- and empathy-based prosocial behaviors. Second, we review relevant neuroscience studies investigating the neural processing of egoism-based and empathy-based prosocial decisions, and the small number of studies that compared the neural processing of prosocial decisions elicited by the different motives. Third, we highlight future directions and open questions for future research.
Before reflecting on potential differences between egoistically and empathically motivated prosocial behavior, a definition of both alternatives is necessary. Egoism-based prosocial behavior depends on the probability that prosocial behavior is associated with a reward (38–42), or the avoidance of costs (43). At the personal level, the pursuit of material goods is associated with lower psychological well-being both cross-sectionally and over time (44; for meta-analysis, see 45).
Although easily elicited, egoism-based prosocial behavior is fragile. There is evidence that incentives can undermine the behaviors that they were supposed to foster (46–50), known as crowding-out effects (51). One aspect of crowding-out relates to initial incentives vs. later prosocial behavior. In this context, it is proposed that crowding-out effects occur when a person receives a reward for a prosocial action and in consequence, is less likely to do the prosocial act for free later (52). Documenting the importance of crowding-out effects for real-life incentive structures, a large number of studies from behavioral economics (e.g., 53) have tested conditions that may alter the impact of incentives on prosocial behaviors. As another separate key outcome, they propose that incentives only undermine prosocial behaviors if they are offered in public (47, 53–55). If social interaction partners are aware that someone receives a payment that enhances prosocial behavior, this person loses their prosocial reputation (i.e., reputation loss, see Table 1). Consequently, this person is no longer perceived as a prosocial and nice person. For instance, Ariely et al. (47) asked participants to make donations to charitable organizations. These donations could be made publicly or privately and were incentivized or not. Incentives offered in private resulted in an increase in prosocial decision. In contrast, receiving payment for prosocial behavior in public reduced the prosocial decision frequency. Crowding-out effects have been observed in a variety of prosocial behaviors, including the collection of door to door money for charity (56), voluntary cooperation (57), and blood donations (58). According to other findings, the negative (i.e., decreasing) effect of reward on behavior is particularly strong when participants’ decisions are not anonymous and thus, their reputation is at stake. For instance, Newman and Cain (59) showed that individuals’ charitable behaviors were rated as more negative when they fulfilled self-interests compared to similar behaviors that did not result in a charitable benefit – a phenomenon they called “tainted altruism”. Other research confirmed reputational costs for egoistically motivated prosocial behavior (59–61).

Table 1. Summary of positive and negative aspects of egoism and empathy as motives of prosocial behavior.
In summary, there may be a narrow and situation-specific region in public charitable behavior within which individuals can unleash their egoistic motive without fearing the loss of reputational benefits. This process may be dimensional rather than categorical, i.e., there is no general rule that indicates a tipping point at which egoism-based charity leads to reputational loss rather than gain.
The increase of own welfare is the most obvious aspect of an egoistic motive (i.e., resource gain, see Table 1), as incentivized prosocial behavior is rewarded by the value of the incentive and, depending on the context, can lead additionally to an increase in reputation. As an everyday example, in Germany a financial donation does not only support the work of a charity and increase the donor’s reputation. The donors also reduce their own tax burdens and thereby receive a financial advantage in return. Importantly, while in the case of egoism-based prosociality, a behavior that increases one’s own welfare also has a positive effect on others (e.g., monetary donations, paid blood/plasma donations), these benefits for others occur merely as a side effect but increasing one’s own welfare is the primary goal. The positive relationship between self-care and care for others has also been documented in recent research on egoistic motives (62) and the measurement of individual differences in trait egoism (63).
Traditionally, egoism has been linked to aggression (e.g., 3). However, under certain circumstances, even egoism-based aggression can be perceived as a prosocial act. For example, in a variety of models investigating social behavior, punishing norm deviating behavior is considered as a mechanism that facilitates the development of altruism, which makes the punishers to be perceived as altruists (64–66). A hitherto rarely addressed aspect is that egoistically motivated individuals may also punish other egoistic individuals in social interactions. By doing so, these individuals contribute to more prosocial behavior in general, although they ultimately aim to increase their chances of getting the maximum benefit by switching to an egoistic strategy at a later point. However, this kind of egoistic behavior could also occur in response to the presumably egoistic behavior of others, which is labeled “reactive egoism” (67). Egoists might fear exploitation by the egoistic behavior of others if they behave charitably, and are therefore motivated to behave in accordance with their own self-interest (e.g., 68, 69).
In contrast to egoistically-driven prosocial behavior, empathy-based prosocial behavior has the goal to increase the welfare of the other person (70). In more detail, although affective empathy results in two distinct reactions – compassion or personal distress (12, 71), only compassion is associated with empathy-based prosocial behavior. Therefore, while egoistically motivated behavior will stop in the absence of reward or distress, empathy-driven prosocial behavior based on compassion will continue until the welfare of the other is secured. There is a large body of evidence showing that empathic responses motivate cooperation and helping behavior (e.g., 25, 72). Practically, this means donating to charity, sharing with others, and volunteering time to help others (73).
However, acting on the basis of empathic feelings can also lead to less/a lack of prosocial behavior in general due to biases, as individual’s emotions can cloud their judgment regarding the best course of action (74). In the case of so called empathic vampirism (75), individuals experience the world vicariously through others and thus share their emotions, but without considering their interests. While this does not mean that empathic vampirism necessarily leads to disadvantageous effects, it is a possibility. An example for less or a lack of prosocial behavior is the identified victim effect. Experiments showed that participants donated more money for a the treatment of an ill child that was introduced with a picture and a name than to a non-identified child suffering from the same illness (76). Other empathy-induced biases include an increased empathy toward members of the empathizer’s group compared to members of different groups (77). Other research highlighted that the empathy toward the suffering of one particular person results in neglecting the fate of many other individuals, compared to the collective suffering of many individuals (e.g., 78). In this sense, empathy acts like a spotlight focusing on certain people or situations, thereby excluding others (79).
Besides biases leading to a reduction of prosocial behavior, sharing the emotions of others can also be disadvantageous for the empathizer, because it can elicit personal distress (80–83). Personal distress is a self-centered negative affect when being exposed to the suffering of others that may result in withdrawal instead of prosocial behavior toward the other. If withdrawal is not possible, personal distress may be the source of egoistically motivated prosociality, i.e., prosocial behavior with the goal to escape the distressing situation (i.e., to increase own welfare), instead of focusing on the welfare of the other person (24, 84–86). As an example, Piferi and colleagues (87) showed that personal distress was the most frequently reported motive for donations immediately following the terrorist attacks of September 11, whereas donations to alleviate the suffering of others was the most frequently cited motive one year after the attacks.
Taken together, both egoism and empathy have their bright and dark sides. Table 1 summarizes the positive and negative aspects of egoism and empathy as motives of prosocial behavior.
Based on the mere observation of overt behavior, empathic and egoistic motivation are hard to distinguish, because they can result in the identical behavior, i.e., the decision to help another person. There is evidence that neuroscience methods can help to specify the type of motive that drives a given decision. For example, Tusche and colleagues (37) identified empathy and perspective taking as motives for generous behavior and used functional magnetic resonance imaging (fMRI) to disentangle differential neural responses associated with each motive. Hein et al. (35) and Saulin et al. (22) showed that two different motives, i.e., empathy and reciprocity, incite a comparable frequency of prosocial decisions. However, based on the patterns of brain connectivity (35) and neural decision parameters (22), the authors were able to distinguish between the two motives and their interrelation. Moreover, in a recent meta-analysis using a data-driven graph-based approach, Rhoads and colleagues classified the neural circuitries of prosocial decisions based on task features. The results revealed three different clusters that were labeled as altruistic, cooperative or equity-based prosocial decisions. Compared to prosocial decisions driven by cooperation and equity, altruism-based prosocial decision uniquely recruited the dorsal anterior cingulate cortex (ACC) and the anterior insula (AI), i.e., regions that have been linked to the processing of empathy (20, 88, 89).
Inspired by these findings, the following section provides a concise review of the neural networks that have been related to the processing of egoism-related and empathy-related prosocial behavior and summarize the results of first studies that aimed to disentangle these motives based on these networks.
The egoistic motivation of increasing one’s own outcomes and welfare (e.g., 25) has been linked to neural responses in reward circuitries (e.g., 90, 91). Human reward circuitries have been addressed in several seminal review articles (e.g., 92–95), showing that the regulation of behavioral and psychological responses to reward stimuli is coordinated by a series of cortical and subcortical structures (96). At the center of this circuit is the striatum, a subcortical structure involved in reward-related learning and resulting approach behaviors. Therefore, a variety of fMRI experiments have examined how the striatum with its potent connections to cortical regions and dopaminergic sites in the midbrain contributes to reward processing (e.g., 97–99).
In the context of prosocial decision behavior, the neural circuitries related to the processing of basic rewards have been linked to the intrinsic positive value of prosocial acts for the individual, known as “warm-glow.” For instance, in an early study Harbaugh et al. (100) asked participants to either observe money being transferred to a charity in a tax-like manner (mandatory condition) or voluntarily donated to the charity (voluntary condition). As a result, both mandatory and voluntary donations triggered similar neural activity in the ventral striatum (VS) as when participants received money themselves. Nevertheless, voluntary donation resulted in stronger striatal activation than mandatory donation, consistent with the concept of warm-glow giving. It was proposed that the act of giving to others itself is rewarding and results in a “warm-glow”, because it contributes to a positive self-image and a gain in reputation (55). Consequently, prosocial decisions that are driven by the expectation of a resulting “warm-glow” would be classified as egoistic, because they focus on a personal, intrinsic reward and an increase in reputation, instead of the welfare of the other (101–103).
Specifying these results, other studies using monetary donation task showed that the VS is part of a larger network, including the ventromedial prefrontal cortex (vmPFC; 104–106), as well as the junction between the temporal and the parietal cortex (TPJ; 107, 108). VS, vmPFC, and medial orbitofrontal cortex (mOFC) are associated with the computation of a common neural value currency for different types of goods, including social goods (95, 98, 109). Increases of activity within the TPJ during social decision-making has been linked to altruistic and generous decision-making (37, 107, 110), thereby promoting generosity. Moreover, in concert with the vmPFC, the TPJ has been related to overcoming egoistic impulses in prosocial decisions (e.g., 107, 108). For example, in an fMRI study by Strombach and colleagues, participants choose to keep a reward for themselves (egoistic decision) or divide this reward between themselves and a (close or socially distant) other person (prosocial decision; 91). An analysis of egoistic decisions alone revealed that vmPFC activity correlated with the value of the egoistic reward. However, when considering both egoistic and prosocial decisions, the results showed that an increase in prosocial decisions was associated with increased activation in the TPJ and the vmPFC. The individual strength of the connectivity between TPJ and vmPCF was larger for prosocial compared to egoistic decisions, an effect that was proposed to reflect the overcoming of egoistic impulses (111, 112). Recently, Sellitto et al. (113) tested framing effects on prosocial behavior in a social discounting task. Participants chose between egoistic and prosocial decisions either in a gain frame, i.e., prosocial decisions led to a reward for the other person, or in a loss frame, i.e., prosocial decisions prevented the other person from losing a previously received endowment. Supporting the results of other studies (e.g., 108), the authors demonstrated that activation in the TPJ and the vmPFC was linked to prosocial decisions in the gain frame, whereas insula activation was associated with prosocial behavior in the loss frame.
Another group of studies addressed how self-related reward-processing affects other motives. For example, Murayama et al. (50) studied whether a financial incentive undermines participants’ performance in a stopwatch task. In this task, participants were presented with an automatically starting stopwatch. The goal was to press a button to make the button press fall within 50 ms of the 5-s time point. In the first part of the study, participants received a performance-based reward, in the second part of the study this reward was no longer given. A control group completed the same task without performance-based reward in both periods. The results showed stronger activation in the striatum and midbrain in the reward group compared to the control group during the first part when the rewards were provided. In the second part of the study, this contrast was reversed, reflecting a decrease of striatal activation in the reward group compared to the control group in absence of an incentive. Based on this results, the authors concluded that the highly rewarding nature of money undermined an intrinsic motivation to successfully complete a task (see also 114). Although this research does not have a prosocial focus per se, it is important for us to understand how performance-based incentive systems practically guide human behavior.
Targeting neural activation and a potentially undermining effect of social reward (i.e., reputation effects) on monetary rewards in a social decision-making paradigm, Izuma et al. (115) asked participants to keep money for themselves or donate money to a charity while either being observed by others or not (control condition). At the behavioral level, participants donated more when they were observed by others. The neural results showed that activation in the striatum increased when participants made donations while being observed (compared to the absence of others), but also when they kept money for themselves without being observed (compared to the presence of others). As such, reputation effects increase the willingness to donate to a charity, which is encoded in striatal brain areas in a similar manner to egoistic decisions, such as keeping money (i.e., not donating).
The association of brain regions with empathy-based prosocial behavior were mainly deduced from studies focusing on empathy-for-pain inductions as a manipulation for decision making. In these studies, participants are confronted with images or videos from persons or confederates suffering from pain compared to control conditions [for meta-analysis, see (11, 77, 116)]. The separation between cognitive empathy [or theory of mind (ToM)] and affective empathy is equally reflected at the neuronal level (9, 14, 15, 17, 117, 118). Cognitive empathy is commonly related to neural activation of the mPFC, the superior temporal sulcus (STS), the temporal poles (TP), and the TPJ (9, 13, 15, 16, 117). In contrast, affective empathy is usually linked to activity in the AI, ACC, and inferior frontal gyrus (IFG) (9, 13, 15, 16, 117, 119, 120). The relationship between different facets of empathy and prosocial decision-making has been reviewed in a large number of review articles and meta-analyses (121–124). Across these different previous reviews there is a broad agreement that neural activation in regions associated with affective empathy (e.g., AI, ACC) and cognitive empathy (e.g., TPJ, mPFC) are linked to empathy-based social decision-making in contrast to social decision-making without explicit activation of empathy for pain.
In an early study, Weiland et al. (125) aimed at disentangling the egoistic from the empathic motive in economic decision making, by comparing two different economic games, i.e., the ultimatum game (UG; where a proposer provides a monetary offer to a receiver who can decide to accept or reject the offer, but rejecting results in a payoff of zero for both), and the dictator game (where the allocator determines how to split an endowment and the receiver has no influence on the outcome). Weiland and colleagues assumed that fair offers in the ultimatum game are driven by strategic considerations with an underlying egoistic motive, and that fair offers in the dictator game are driven by an empathy motive. Within the UG, fair compared to unfair offers were associated with increased activation in the striatum, the superior temporal area and the temporal pole. In the dictator game, fair offers were associated with increased activity in the dorsal part of the ACC and the posterior cingulate cortex. Given that an increase in ACC activation has been associated with affective empathy (126, 127), the authors concluded that prosocial dictator giving might be driven by an empathy motive. However, as these results are based on the comparison of mechanistically different paradigms (i.e., UG and DG), they might exaggerate or overshadow the effects of the motivational states underlying the behavior and their neural correlates.
More recently, Cutler and Campbell-Meiklejohn (9) conducted an fMRI-based meta-analysis in which they synthesized findings from 36 studies focusing on prosocial giving (1,150 participants in total). The authors investigated the neural networks underlying “strategic” prosocial decisions (i.e., decisions that might increase the likelihood of extrinsic rewards) and “altruistic” decisions (i.e., decisions without the chance to receive extrinsic rewards). Strategic decisions included decisions based on the avoidance of punishment, reciprocity, rewards through cooperation or increased reputation, whereas altruistic decisions included decision driven by empathic concern or self-enhancement through compliance with social norms. According to the results, both altruistically motivated and strategically motivated prosocial decisions commonly recruit reward-related brain areas. Contrasting altruistically motivated and strategically motivated prosocial behaviors across these different paradigms revealed stronger activation in the ACC for decisions motivated by the predefined “altruistic” motives (including affective empathy) as compared to decisions motivated by “strategic,” egoistic motives. Overall, these results suggest that neural correlates can differentiate between giving to others based on egoistic motives and prosocial behavior without a chance to receive extrinsic rewards. However, the “altruistic” and “strategic” motive categories aggregated a number of very different motives. Thus, the neural differentiation between empathy-based and egoism-based remains rather unspecific. In addition, distinct types of decisions might also pertain to both the “altruistic” and the “strategic” category. For instance, self-enhancement through compliance with social norms could also categorized as “strategic” because decisions in domain may later entail positive consequences for the agent.
Disentangling egoism-based and empathy-based prosocial behavior requires paradigms that allow for investigating the effect of both motives on the same behavior. In a recent study, Iotzov and colleagues induced empathy for pain and egoism and investigated the effect of both motives on an identical social decision task (21). In one condition (empathy-alone condition) participants observed their partner experiencing pain. In the other condition (empathy-bonus condition), participants also observed the other in pain, but were offered a financial bonus if they made prosocial decisions in the majority of trials in a subsequent task, in which they could allocate points in favor of themselves or in favor of the other person. Thus, in the empathy-bonus condition, the egoistic motive of outcome maximization (winning the bonus) was activated simultaneously with the empathy motive. Later, participants performed the same social decision task in which they could allocate points in favor of themselves or in favor of the other person. The results showed more prosocial decisions in the empathy-bonus compared to the empathy-alone condition. Clarifying the mechanism through which the bonus facilitated empathy-based decisions, drift-diffusion modeling (DDM) revealed an increase in the efficiency of information processing (drift rate). On the neural level, the bonus-related increase in drift rate was captured by changes of neural responses in the AI, i.e., the same region that also correlated with empathy ratings. Interestingly, the effect of the bonus on empathy-related neural responses in AI was stronger the lower an individual scored on empathy. In contrast, in highly empathic individuals the bonus had little effect.
Comparing the neural network involved in prosocial decisions in the empathy-alone compared to the empathy-bonus condition revealed activations in the ACC and midcingulate cortex (MCC), as well as the AI (Figure 1A), the neural network that has been associated with the processing of affective empathy for pain (11, for meta-analyses, see 120, 128). The reverse contrast (empathy-bonus vs empathy-alone) revealed activation in the TPJ (Figure 1B), a brain region that has been associated with cognitive empathy or theory of mind (129, 130) and overcoming egoistic impulses (107, 108). Together, these findings imply that an egoistic motive (incited by a monetary incentive) alters the cognitive and neural processing of empathy-based prosocial decisions. Moreover, they indicate different neural networks for the processing of egoism-based and empathy-based prosocial behavior, in line with other recent evidence (e.g., 9).
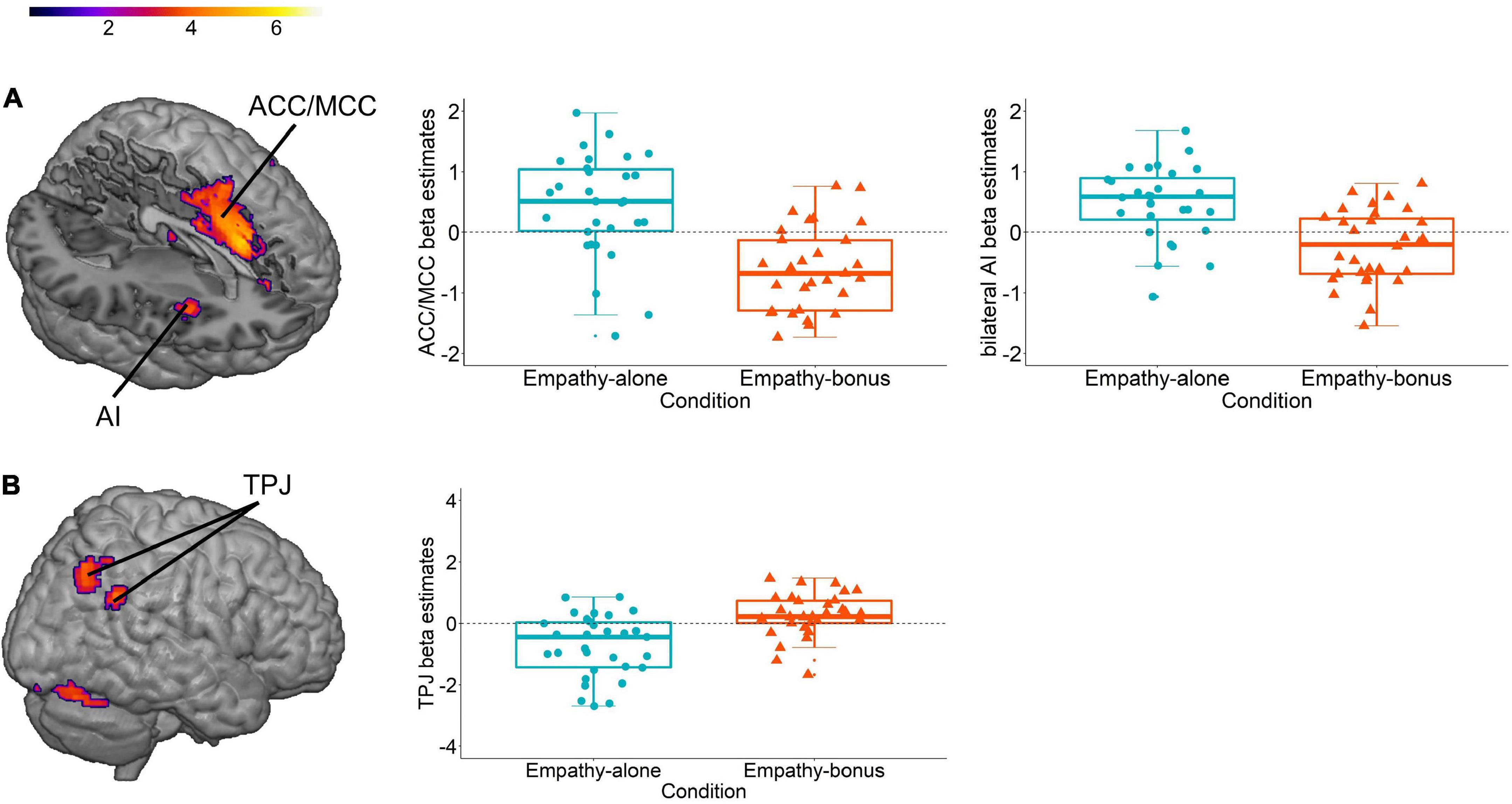
Figure 1. Neural response during prosocial decisions (1% uncorrected cluster-based inference with p < 0.001 cluster-forming threshold; k = 0). We used data (N = 31) from Iotzov et al. (21) and reanalyzed them for the purpose of this review. (A) Neural contrast between prosocial decisions in the empathy-alone vs the empathy-bonus condition and visualization of the average beta-values extracted from the clusters of activation in the bilateral anterior insula (AI; center co-ordinates left x = –36, y = 14, z = 8 and right X = 30, y = 11, z = –16) and the anterior and mid cingulate cortex (ACC/MCC; center co-ordinates x = –9, y = 14, z = 35). (B) Neural contrast between prosocial decisions in the empathy-bonus vs the empathy-alone condition and visualization of the average beta-values extracted from two clusters of activation in the temporal parietal junction (TPJ; center co-ordinates x = –45, y = –55, z = 32 and x = –36, y = –70, z = 47).
In the first part of our review, we reviewed behavioral evidence for egoism-based prosocial behavior, compared to prosocial behaviors driven by empathy. We conclude that egoism can be a strong driver of prosocial behaviors, that is however limited in several aspects. First, compared to empathy-based prosocial behavior, egoism-based prosocial behavior focuses on increasing one’s own well-being, whereas increasing the well-being of the other person plays only a subordinate role and can be seen more as a means to an end. Second, the two motives differ in their effects on reputation. While empathy-based prosocial behavior has a positive effect on reputation, adding an egoistic motive can lead to a reduction in reputation (47, 53–55).
That being said, egoism and empathy can incite the same behavioral outcomes, e.g., prosocial decisions. Thus, it can be difficult to infer an egoistic motivation from overt behavior. If one only focuses on the behavioral outcome per se, it may not seem necessary to specify the motivational source of the behavior, i.e., to draw a distinction between an egoistic and another motive. However, differentiating between different motives is crucial for behavioral predictions. Given that it is conditional on reward, egoistically motivated prosocial behavior will stop if this reward is not provided. In contrast, prosocial behavior driven by empathy should continue until the wellbeing of the other person is secured.
In the second part of the review, we asked if neuroscientific evidence can help to specify the motive that drives a given behavior, for example, by specifying if a prosocial decision is driven by an egoistic or an empathic motive. The reviewed neural evidence indeed suggests differential neural circuitries for the processing of egoism-based and empathy-based prosocial decisions.
However, neuroscientific studies that directly compare egoism-based prosocial behavior with prosocial behavior that is driven by a specific other motive are rare. Moreover, they are mostly based on the comparison between different paradigms (e.g., DG vs. UG), one of which is assumed to promote egoistic decisions and the other is assumed to promote “altruistic” decisions (9, 125). Based on these approaches it remains unclear whether potential neural differences reflect differences in motivational states or in task requirements. One possible solution to this challenge is to design carefully controlled experiments in which individuals perform the same prosocial task driven by egoism and driven by another motive see (e.g., 21). These paradigms should be combined with state-of-the-art neuroscientific methods to assess changes in neural activation and neural connectivity in complex motivational states (i.e., states characterized by different interacting motives) and simple motivational states (in which one particular motive prevails). Moreover, computational methods should be used to specify the effect of interacting motives on the decision process. For example, first studies have started to use drift-diffusion modeling (131, 132) to determine how different motivational states alter the different components of the decision process (i.e., influence the initial decision bias or the efficiency of information accumulation (21, 133). As another line of future research, it would be interesting to investigate how “state”-egoism, i.e., egoism that is induced by incentives, interacts with trait egoism, and how both forms of egoism shape other specific motives.
MW and GH: conceptualization and writing – original draft. GH: supervision. MW and VI: visualization. MW, VI, YZ, and GH: writing – review and editing. All authors contributed to the article and approved the submitted version.
GH was supported by grants from the German Research Foundation (HE 4566/5-1 and HE 4566/3-2). This publication was supported by the Open Access Publication Fund of the University of Würzburg.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
1. de La Rochefoucauld D. Moral Maxims and Reflections, in Four Parts. London: Gillyflower, Sare, &Everingham (1691).
2. Stirner M. The Owner. In: D Leopold editor. The Ego and Its Own. Cambridge: Cambridge University Press (1995). p. 155–319.
3. Park LE, Troisi JD, Maner JK. Egoistic versus altruistic concerns in communal relationships. J Soc Pers Relat. (2011) 28:315–35.
4. Weigel RH, Hessing DJ, Elffers H. Egoism: concept, measurement and implications for deviance. Psychol Crime Law. (1999) 5:349–78.
5. Raine A, Uh S. The Selfishness questionnaire: egocentric, adaptive, and pathological forms of selfishness. J Pers Assess. (2018) 101:503–14. doi: 10.1080/00223891.2018.1455692
7. Batson CD, Shaw LL. Evidence for altruism: toward a pluralism of prosocial motives. Psychol Inq. (1991) 2:107–22.
8. Batson CD, Powell AA. Altruism and prosocial behavior. In: Gilbert DT, Fiske ST, Lindzey G editors. The Handbook of Social Psychology. Hoboken, NJ: John Wiley & Sons, Inc (2003). p. 463–84.
9. Cutler J, Campbell-Meiklejohn D. A comparative fMRI meta-analysis of altruistic and strategic decisions to give. Neuroimage. (2019) 184:227–41. doi: 10.1016/j.neuroimage.2018.09.009
10. Batson CD, Ahmad N, Tsang JA. Four motives for community involvement. J Soc Issues. (2002) 58:429–45.
11. Timmers I, Park AL, Fischer MD, Kronman CA, Heathcote LC, Hernandez JM, et al. Is empathy for pain unique in its neural correlates? A meta-analysis of neuroimaging studies of empathy. Front Behav Neurosci. (2018) 12:289. doi: 10.3389/fnbeh.2018.00289
12. Hein G, Qi Y, Han S. The biological foundations and modulation of empathy. 3rd ed. In: Van Lange PAM, Higgins ET, Kruglanski AW editors. Social Psychology: Handbook of Basic Principles. New York, NY: The Guilford Press (2021). p. 127–39.
13. Stietz J, Jauk E, Krach S, Kanske P. Dissociating empathy from perspective-taking: evidence from intra–and inter-individual differences research. Front Psychiatry. (2019) 10:126. doi: 10.3389/fpsyt.2019.00126
14. Cox CL, Uddin LQ, Di Martino A, Castellanos FX, Milham MP, Kelly C. The balance between feeling and knowing: affective and cognitive empathy are reflected in the brain’s intrinsic functional dynamics. Soc Cogn Affect Neurosci. (2012) 7:727–37. doi: 10.1093/scan/nsr051
15. Preckel K, Kanske P, Singer T. On the interaction of social affect and cognition: empathy, compassion and theory of mind. Curr Opin Behav Sci. (2018) 19:1–6.
16. Schurz M, Radua J, Tholen MG, Maliske L, Margulies DS, Mars RB, et al. Toward a hierarchical model of social cognition: a neuroimaging meta-analysis and integrative review of empathy and theory of mind. Psychol Bull. (2021) 147:293–327. doi: 10.1037/bul0000303
17. Kanske P, Böckler A, Trautwein FM, Singer T. Dissecting the social brain: introducing the EmpaToM to reveal distinct neural networks and brain-behavior relations for empathy and Theory of Mind. Neuroimage. (2015) 122:6–19. doi: 10.1016/j.neuroimage.2015.07.082
18. Zaki J, Ochsner K. The neuroscience of empathy: progress, pitfalls and promise. Nat Neurosci. (2012) 15:675–80. doi: 10.1038/nn.3085
19. Batson CD, Turk CL, Shaw LL, Klein TR. Information function of empathic emotion: learning that we value the other’s welfare. J Pers Soc Psychol. (1995) 68:300–13.
20. Decety J, Bartal IBA, Uzefovsky F, Knafo-Noam A. Empathy as a driver of prosocial behaviour: highly conserved neurobehavioural mechanisms across species. Philos Trans R Soc B Biol Sci. (2016) 371:20150077. doi: 10.1098/rstb.2015.0077
21. Iotzov V, Saulin A, Kaiser J, Han S, Hein G. Financial incentives facilitate stronger neural computation of prosocial decisions in lower empathic adult females. Soc Neurosci. (2022) 0:1–21. doi: 10.1080/17470919.2022.2115550
22. Saulin A, Horn U, Lotze M, Kaiser J, Hein G. The neural computation of human prosocial choices in complex motivational states. Neuroimage. (2022) 247:118827. doi: 10.1016/j.neuroimage.2021.118827
23. Hartmann H, Forbes PAG, Rütgen M, Lamm C. Placebo analgesia reduces costly prosocial helping to lower another person’s pain. Psychol Sci. (2022):09567976221119727. doi: 10.1177/09567976221119727
24. Batson CD. The Altruism Question: Toward a Social Psychological Answer. Hillsdale, NJ: Erlbaum (1991).
25. Batson CD. Empathy-induced altruistic motivation. In: Mikulincer M, Shaver PR editors. Prosocial Motives, Emotions, and Behavior: The Better Angels of Our Nature. Washington, DC: American Psychological Association (2010). p. 15–34.
26. Lockwood PL, Apps MAJ, Valton V, Viding E, Roiser JP. Neurocomputational mechanisms of prosocial learning and links to empathy. Proc Natl Acad Sci USA. (2016) 113:9763–8. doi: 10.1073/pnas.1603198113
27. FeldmanHall O, Dalgleish T, Evans D, Mobbs D. Empathic concern drives costly altruism. Neuroimage. (2015) 105:347–56. doi: 10.1016/j.neuroimage.2014.10.043
28. Morelli SA, Rameson LT, Lieberman MD. The neural components of empathy: predicting daily prosocial behavior. Soc Cogn Affect Neurosci. (2014) 9:39–47. doi: 10.1093/scan/nss088
29. Toumbourou JW. Beneficial Action within altruistic and prosocial behavior. Rev Gen Psychol. (2016) 20:245–58.
30. Penner LA, Dovidio JF, Piliavin JA, Schroeder DA. Prosocial behavior: multilevel perspectives. Annu Rev Psychol. (2005) 56:365–92.
31. Kurzban R, Burton-Chellew MN, West SA. The evolution of altruism in humans. Annu Rev Psychol. (2015) 66:575–99.
32. de Waal FBM. Putting the altruism back into altruism: the evolution of empathy. Annu Rev Psychol. (2008) 59:279–300.
33. Kwang T, Swann WB. Do people embrace praise even when they feel unworthy? a review of critical tests of self-enhancement versus self-verification. Pers Soc Psychol Rev. (2010) 14:263–80. doi: 10.1177/1088868310365876
34. Windmann S, Hein G. Altruism from the perspective of the social neurosciences. e Neuroforum. (2018) 24:A11–8.
35. Hein G, Morishima Y, Leiberg S, Sul S, Fehr E. The brain’s functional network architecture reveals human motives. Science. (2016) 351:1074–8.
36. Rhoads SA, Cutler JO, Marsh AA. A feature-based network analysis and fMRI meta-analysis reveal three distinct types of prosocial decisions. Soc Cogn Affect Neurosci. (2021) 2021:1–20. doi: 10.1093/scan/nsab079
37. Tusche A, Böckler A, Kanske P, Trautwein FM, Singer T. Decoding the charitable brain: empathy, perspective taking, and attention shifts differentially predict altruistic giving. J Neurosci. (2016) 36:4719–32.
38. Berridge KC. Reward learning: reinforcement, incentives, and expectations. In: DL Medin editor. The Psychology of Learning and Motivation: Advances in Research and Theory. (Vol. 40), San Diego, CA: Academic Press (2001). p. 223–78.
39. Kane RL, Johnson PE, Town RJ, Butler M. A structured review of the effect of economic incentives on consumers’ preventive behavior. Am J Prev Med. (2004) 27:327–52.
40. Strohacker K, Galarraga O, Williams DM. The impact of incentives on exercise behavior: a systematic review of randomized controlled trials. Ann Behav Med. (2014) 48:92–9.
41. Garbers Y, Konradt U. The effect of financial incentives on performance: a quantitative review of individual and team-based financial incentives. J Occup Organ Psychol. (2014) 87:102–37.
43. Wilson DS, Wilson EO. Rethinking the theoretical foundation of sociobiology. Q Rev Biol. (2007) 82:327–48. doi: 10.1086/522809
44. Kasser T, Rosenblum KL, Sameroff AJ, Deci EL, Niemiec CP, Ryan RM, et al. Changes in materialism, changes in psychological well-being: evidence from three longitudinal studies and an intervention experiment. Motiv Emot. (2014) 38:1–22.
45. Dittmar H, Bond R, Hurst M, Kasser T. The relationship between materialism and personal well-being: a meta-analysis. J Pers Soc Psychol. (2014) 107:879–924.
46. Andreoni J, Payne AA. Is crowding out due entirely to fundraising? Evidence from a panel of charities. J Public Econ. (2011) 95:334–43.
47. Ariely D, Bracha A, Meier S. Doing good or doing well? Image motivation and monetary incentives in behaving prosocially. Am Econ Rev. (2009) 99:544–55.
48. Deci EL, Koestner R, Ryan RM. A meta-analytic review of experiments examining the effects of extrinsic rewards on intrinsic motivation. Psychol Bull. (1999) 125:627–68; discussion 692–700. doi: 10.1037/0033-2909.125.6.627
49. Gneezy U, Meier S, Rey-Biel P. When and why incentives (Don’t) work to modify behavior. J Econ Perspect. (2011) 25:191–210.
50. Murayama K, Matsumoto M, Izuma K, Matsumoto K. Neural basis of the undermining effect of monetary reward on intrinsic motivation. Proc Natl Acad Sci USA. (2010) 7:20911–6.
51. Janssen MCW, Mendys-Kamphorst E. The price of a price: on the crowding out and in of social norms. J Econ Behav Organ. (2004) 55:377–95.
52. Wollbrant CE, Knutsson M, Martinsson P. Extrinsic rewards and crowding-out of prosocial behaviour. Nat Hum Behav. (2022) 6:774–81.
55. Exley C. Incentives for prosocial behavior: the role of reputations. Manag Sci. (2017) 64:2460–71.
57. Fehr E, Gächter S. Do Incentive Contracts Undermine Voluntary Cooperation? Rochester, NY: Social Science Research Network (2002).
58. Mellström C, Johannesson M. Crowding out in blood donation: was titmuss right? J Eur Econ Assoc. (2008) 6:845–63.
59. Newman GE, Cain DM. Tainted altruism: when doing some good is evaluated as worse than doing no good at all. Psychol Sci. (2014) 25:648–55. doi: 10.1177/0956797613504785
60. Carlson RW, Zaki J. Good deeds gone bad: Lay theories of altruism and selfishness. J Exp Soc Psychol. (2018) 75:36–40.
61. Lin-Healy F, Small DA. Cheapened altruism: discounting personally affected prosocial actors. Organ Behav Hum Decis Process. (2012) 117:269–74.
62. Crocker J, Canevello A. From egosystem to ecosystem: motivations of the self in a social world. Adv Motiv Sci. (2018) 5:41–86.
63. Kaufman SB, Jauk E. Healthy selfishness and pathological altruism: measuring two paradoxical forms of selfishness. Front Psychol. (2020) 11:1006. doi: 10.3389/fpsyg.2020.01006
66. Boyd R, Gintis H, Bowles S, Richerson PJ. The evolution of altruistic punishment. Proc Natl Acad Sci USA. (2003) 100:3531–5.
67. Epley N, Caruso EM, Bazerman MH. When perspective taking increases taking: reactive egoism in social interaction. J Pers Soc Psychol. (2006) 91:872–89. doi: 10.1037/0022-3514.91.5.872
68. Kelley HH, Stahelski AJ. Social interaction basis of cooperators’ and competitors’ beliefs about others. J Pers Soc Psychol. (1970) 16:66–91.
69. Messé LA, Sivacek JM. Predictions of others’ responses in a mixed-motive game: self-justification or false consensus? J Pers Soc Psychol. (1979) 37:602–7.
70. Batson CD, Ahmad N, Stocks EL. Four forms of prosocial motivation. In: D Dunning editor. Social Motivation. Milton Park: Taylor & Francis Group (2010). p. 103–27.
72. Hoffman ML. Empathy and prosocial behavior. In: Lewis M, Haviland-Jones J, Barrett L editors. Handbook of Emotions. New York, NY: Guilford Press (2008). p. 440–55.
73. Eisenberg N, Miller PA. The relation of empathy to prosocial and related behaviors. Psychol Bull. (1987) 101:91–119.
75. Schiffrin HH, Liss M, Miles-McLean H, Geary KA, Erchull MJ, Tashner T. Helping or hovering? The effects of helicopter parenting on college students’ well-being. J Child Fam Stud. (2014) 23:548–57.
76. Kogut T, Ritov I. The “identified victim” effect: an identified group, or just a single individual? J Behav Decis Mak. (2005) 18:157–67.
77. Hein G, Silani G, Preuschoff K, Batson CD, Singer T. Neural responses to ingroup and outgroup members’ suffering predict individual differences in costly helping. Neuron. (2010) 68:149–60. doi: 10.1016/j.neuron.2010.09.003
79. Ryan DL. Paul bloom: against empathy: the case for rational compassion. Society. (2018) 55:216–7. doi: 10.1097/TA.0000000000002071
81. Trommsdorff G, Friedlmeier W, Mayer B. Sympathy, distress, and prosocial behavior of preschool children in four cultures. Int J Behav Dev. (2007) 31:284–93.
82. Eisenberg N, Fabes RA, Spinrad TL. Handbook of Child Psychology. (Vol. 3). New York, NY: John Wiley & Sons (2006).
83. Decety J, Lamm C. Empathy versus personal distress: recent evidence from social neuroscience. In: Decety J, Ickes W editors. The Social Neuroscience of Empathy. Cambridge, MA: The MIT Press (2009). p. 199–213.
84. Carlo G, Eisenberg N, Troyer D, Switzer G, Speer AL. The altruistic personality: in what contexts is it apparent? J Pers Soc Psychol. (1991) 61:450–8.
85. Eisenberg N, Fabes RA, Miller PA, Fultz J, Shell R, Mathy RM, et al. Relation of sympathy and personal distress to prosocial behavior: a multimethod study. J Pers Soc Psychol. (1989) 57:55–66. doi: 10.1037//0022-3514.57.1.55
86. Tice DM, Bratslavsky E, Baumeister RF. Emotional distress regulation takes precedence over impulse control: if you feel bad, do it! J Pers Soc Psychol. (2001) 80:53–67.
87. Piferi RL, Jobe RL, Jones WH. Giving to others during national tragedy: the effects of altruistic and egoistic motivations on long-term giving. J Soc Pers Relat. (2006) 23:171–84.
88. Brethel-Haurwitz KM, Cardinale EM, Vekaria KM, Robertson EL, Walitt B, VanMeter JW, et al. Extraordinary altruists exhibit enhanced self–other overlap in neural responses to distress. Psychol Sci. (2018) 29:1631–41. doi: 10.1177/0956797618779590
89. O’Connell K, Brethel-Haurwitz KM, Rhoads SA, Cardinale EM, Vekaria KM, Robertson EL, et al. Increased similarity of neural responses to experienced and empathic distress in costly altruism. Sci Rep. (2019) 9:10774. doi: 10.1038/s41598-019-47196-3
90. Gêsiarz F, Crockett MJ. Goal-directed, habitual and pavlovian prosocial behavior. Front Behav Neurosci. (2015) 9:135. doi: 10.3389/fnbeh.2015.00135
91. Sonne JWH, Gash DM. Psychopathy to altruism: neurobiology of the selfish–selfless spectrum. Front Psychol. (2018) 9:575. doi: 10.3389/fpsyg.2018.00575
93. Kable JW, Glimcher PW. The neurobiology of decision: consensus and controversy. Neuron. (2009) 63:733–45. doi: 10.1016/j.neuron.2009.09.003
94. Rangel A, Camerer C, Montague PR. A framework for studying the neurobiology of value-based decision making. Nat Rev Neurosci. (2008) 9:545–56.
95. Ruff CC, Fehr E. The neurobiology of rewards and values in social decision making. Nat Rev Neurosci. (2014) 15:549–62.
96. Haber SN, Knutson B. The reward circuit: linking primate anatomy and human imaging. Neuropsychopharmacology. (2010) 35:4–26.
97. Bartra O, McGuire JT, Kable JW. The valuation system: a coordinate-based meta-analysis of BOLD fMRI experiments examining neural correlates of subjective value. Neuroimage. (2013) 76:412–27. doi: 10.1016/j.neuroimage.2013.02.063
98. Clithero JA, Rangel A. Informatic parcellation of the network involved in the computation of subjective value. Soc Cogn Affect Neurosci. (2014) 9:1289–302. doi: 10.1093/scan/nst106
99. Delgado MR. Reward-related responses in the human striatum. Ann N Y Acad Sci. (2007) 1104:70–88.
100. Harbaugh WT, Mayr U, Burghart DR. Neural responses to taxation and voluntary giving reveal motives for charitable donations. Science. (2007) 316:1622–5. doi: 10.1126/science.1140738
101. Altman M. Real-World Decision Making: An Encyclopedia of Behavioral Economics: An Encyclopedia of Behavioral Economics. Santa Barbara, CA: ABC-CLIO (2015). 532 p.
102. Andreoni J. Giving with impure altruism: applications to charity and ricardian equivalence. J Polit Econ. (1989) 97:1447–58.
103. Barasch A, Levine EE, Berman JZ, Small DA. Selfish or selfless? On the signal value of emotion in altruistic behavior. J Pers Soc Psychol. (2014) 107:393–413.
104. Hackel LM, Wills JA, Van Bavel JJ. Shifting prosocial intuitions: neurocognitive evidence for a value-based account of group-based cooperation. Soc Cogn Affect Neurosci. (2020) 15:371–81. doi: 10.1093/scan/nsaa055
105. Pärnamets P, Shuster A, Reinero DA, Van Bavel JJA. Value-based framework for understanding cooperation. Curr Dir Psychol Sci. (2020) 29:227–34.
106. Fareri DS, Chang LJ, Delgado MR. Computational substrates of social value in interpersonal collaboration. J Neurosci. (2015) 35:8170–80. doi: 10.1523/JNEUROSCI.4775-14.2015
107. Morishima Y, Schunk D, Bruhin A, Ruff CC, Fehr E. Linking brain structure and activation in temporoparietal junction to explain the neurobiology of human altruism. Neuron. (2012) 75:73–9. doi: 10.1016/j.neuron.2012.05.021
108. Strombach T, Weber B, Hangebrauk Z, Kenning P, Karipidis II, Tobler PN, et al. Social discounting involves modulation of neural value signals by temporoparietal junction. Proc Natl Acad Sci USA. (2015) 112:1619–24. doi: 10.1073/pnas.1414715112
109. Levy DJ, Glimcher PW. Comparing apples and oranges: using reward-specific and reward-general subjective value representation in the brain. J Neurosci. (2011) 31:14693–707. doi: 10.1523/JNEUROSCI.2218-11.2011
110. Park SQ, Kahnt T, Dogan A, Strang S, Fehr E, Tobler PN. A neural link between generosity and happiness. Nat Commun. (2017) 8:1–10. doi: 10.1038/ncomms15964
111. Strang S, Park SQ. Human cooperation and its underlying mechanisms. In: Wöhr M, Krach S editors. Social Behavior from Rodents to Humans: Neural Foundations and Clinical Implications. Cham: Springer International Publishing (2017). p. 223–39. doi: 10.1007/7854_2016_445
112. Soutschek A, Tobler PN. Motivation for the greater good: neural mechanisms of overcoming costs. Curr Opin Behav Sci. (2018) 22:96–105.
113. Sellitto M, Neufang S, Schweda A, Weber B, Kalenscher T. Arbitration between insula and temporoparietal junction subserves framing-induced boosts in generosity during social discounting. Neuroimage. (2021) 238:118211. doi: 10.1016/j.neuroimage.2021.118211
114. Murayama K, Sakaki M, Meliss S, Yomogida Y, Matsumori K, Sugiura A, et al. Motivated for near impossibility: how task type and reward modulates intrinsic motivation and the striatal activation for an extremely difficult task. bioRxiv. (2019) [Preprint]. 828756. doi: 10.1101/828756v1
115. Izuma K, Saito DN, Sadato N. Processing of the incentive for social approval in the ventral striatum during charitable donation. J Cogn Neurosci. (2010) 22:621–31. doi: 10.1162/jocn.2009.21228
116. Preis MA, Schmidt-Samoa C, Dechent P, Kroener-Herwig B. The effects of prior pain experience on neural correlates of empathy for pain: an fMRI study. Pain. (2013) 154:411–8.
117. Dvash J, Shamay-Tsoory SG. Theory of mind and empathy as multidimensional constructs: neurological foundations. Topics Lang Disord. (2014) 34:282–95.
118. Shamay-Tsoory SG, Aharon-Peretz J, Perry D. Two systems for empathy: a double dissociation between emotional and cognitive empathy in inferior frontal gyrus versus ventromedial prefrontal lesions. Brain. (2009) 132:617–27. doi: 10.1093/brain/awn279
119. Fan Y, Duncan NW, de Greck M, Northoff G. Is there a core neural network in empathy? An fMRI based quantitative meta-analysis. Neurosci Biobehav Rev. (2011) 35:903–11. doi: 10.1016/j.neubiorev.2010.10.009
120. Walter H. Social cognitive neuroscience of empathy: concepts, circuits, and genes. Emot Rev. (2012) 4:9–17.
121. Bellucci G, Camilleri JA, Eickhoff SB, Krueger F. Neural signatures of prosocial behaviors. Neurosci Biobehav Rev. (2020) 118:186–95.
122. Davis MH. Empathy and prosocial behavior. In: Schroeder DA, Graziano WG editors. The Oxford Handbook of Prosocial Behavior. New York, NY: Oxford University Press (2015). p. 282–306.
123. Lamm C, Rütgen M, Wagner IC. Imaging empathy and prosocial emotions. Neurosci Lett. (2019) 693:49–53.
124. Stevens F, Taber K. The neuroscience of empathy and compassion in pro-social behavior. Neuropsychologia. (2021) 159:107925.
125. Lockwood PL, O’Nell KC, Apps MAJ. Anterior cingulate cortex: a brain system necessary for learning to reward others? PLoS Biol. (2020) 18:e3000735. doi: 10.1371/journal.pbio.3000735
126. Lockwood PL, Wittmann M, Nili H, Matsumoto-Ryan M, Abdurahman A, Cutler J, et al. Distinct neural representations for prosocial and self-benefitting effort. bioRxiv. (2021) [Preprint]. bioRxiv 2021.09.27.461936. doi: 10.1101/2021.09.27.461936v1
127. Lamm C, Decety J, Singer T. Meta-analytic evidence for common and distinct neural networks associated with directly experienced pain and empathy for pain. Neuroimage. (2011) 54:2492–502. doi: 10.1016/j.neuroimage.2010.10.014
128. Saxe R, Kanwisher N. People thinking about thinking people: the role of the temporo-parietal junction in “theory of mind”. Neuroimage. (2003) 19:1835–42.
129. Schurz M, Radua J, Aichhorn M, Richlan F, Perner J. Fractionating theory of mind: a meta-analysis of functional brain imaging studies. Neurosci Biobehav Rev. (2014) 42:9–34.
130. Weiland S, Hewig J, Hecht H, Mussel P, Miltner WHR. Neural correlates of fair behavior in interpersonal bargaining. Soc Neurosci. (2012) 7:537–51. doi: 10.1080/17470919.2012.674056
131. Ratcliff R, McKoon G. The diffusion decision model: theory and data for two-choice decision tasks. Neural Comput. (2008) 20:873–922.
132. Smith PL, Ratcliff R. Psychology and neurobiology of simple decisions. Trends Neurosci. (2004) 27:161–8.
Keywords: egoism, incentives, prosociality, social motives, fMRI
Citation: Weiß M, Iotzov V, Zhou Y and Hein G (2022) The bright and dark sides of egoism. Front. Psychiatry 13:1054065. doi: 10.3389/fpsyt.2022.1054065
Received: 26 September 2022; Accepted: 01 November 2022;
Published: 24 November 2022.
Edited by:
Sören Krach, University of Lübeck, GermanyReviewed by:
Emanuel Jauk, Medical University of Graz, AustriaCopyright © 2022 Weiß, Iotzov, Zhou and Hein. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Martin Weiß, d2Vpc3NfbTExQHVrdy5kZQ==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.