
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Psychiatry , 06 August 2021
Sec. Child and Adolescent Psychiatry
Volume 12 - 2021 | https://doi.org/10.3389/fpsyt.2021.718485
This article is part of the Research Topic Fragile X Spectrum Disorders View all 24 articles
 Nell Maltman1,2
Nell Maltman1,2 Janna Guilfoyle1
Janna Guilfoyle1 Kritika Nayar1
Kritika Nayar1 Gary E. Martin3
Gary E. Martin3 Molly Winston1
Molly Winston1 Joseph C. Y. Lau1
Joseph C. Y. Lau1 Lauren Bush1
Lauren Bush1 Shivani Patel1
Shivani Patel1 Michelle Lee1
Michelle Lee1 John Sideris4
John Sideris4 Deborah A. Hall5
Deborah A. Hall5 Lili Zhou6
Lili Zhou6 Kevin Sharp6
Kevin Sharp6 Elizabeth Berry-Kravis6
Elizabeth Berry-Kravis6 Molly Losh1*
Molly Losh1*The FMR1 gene in its premutation (PM) state has been linked to a range of clinical and subclinical phenotypes among FMR1 PM carriers, including some subclinical traits associated with autism spectrum disorder (ASD). This study attempted to further characterize the phenotypic profile associated with the FMR1 PM by studying a battery of assessments examining clinical-behavioral traits, social-cognitive, and executive abilities in women carrying the FMR1 PM, and associations with FMR1-related variability. Participants included 152 female FMR1 PM carriers and 75 female controls who were similar in age and IQ, and screened for neuromotor impairments or signs of fragile X-associated tremor/ataxia syndrome. The phenotypic battery included assessments of ASD-related personality and language (i.e., pragmatic) traits, symptoms of anxiety and depression, four different social-cognitive tasks that tapped the ability to read internal states and emotions based on different cues (e.g., facial expressions, biological motion, and complex social scenes), and a measure of executive function. Results revealed a complex phenotypic profile among the PM carrier group, where subtle differences were observed in pragmatic language, executive function, and social-cognitive tasks that involved evaluating basic emotions and trustworthiness. The PM carrier group also showed elevated rates of ASD-related personality traits. In contrast, PM carriers performed similarly to controls on social-cognitive tasks that involved reliance on faces and biological motion. The PM group did not differ from controls on self-reported depression or anxiety symptoms. Using latent profile analysis, we observed three distinct subgroups of PM carriers who varied considerably in their performance across tasks. Among PM carriers, CGG repeat length was a significant predictor of pragmatic language violations. Results suggest a nuanced phenotypic profile characterized by subtle differences in select clinical-behavioral, social-cognitive, and executive abilities associated with the FMR1 PM in women.
The FMR1 gene plays a critical role in the expression of a range of clinical phenotypes, including both neurodevelopmental and neurodegenerative disorders. Located in the 5′ untranslated region (5′ UTR) on the long arm of the X chromosome, FMR1 encodes the Fragile X Mental Retardation Protein (FMRP), which is highly expressed in the brain and plays a role in synaptic plasticity (1–4). A full mutation of the FMR1 gene (>200 cytosine-guanine-guanine [CGG] trinucleotide repeats) causes methylation and subsequent silencing of the gene, inhibiting production of FMRP and causing fragile X syndrome (FXS), a rare condition (~1 in 4,000 males and ~1 in 8,000 females) that is the most common inherited cause of intellectual disability and monogenic cause of autism spectrum disorder (ASD) (5–7). The FMR1 premutation (PM; 55–200 CGG repeats), occurs in roughly 1 in 150–250 women, and is less prevalent in men (~1 in 430–460) (8–12). Though the PM was once believed to have limited phenotypic expression, a number of clinical and subclinical phenotypes have since been identified, including PM-specific disorders (i.e., fragile X-associated primary ovarian insufficiency [FXPOI], fragile X-associated tremor/ataxia syndrome [FXTAS], and fragile X-associated neuropsychiatric disorders [FXAND]) (4, 13, 14). FMR1-related molecular genetic variation has been associated with these phenotypes, including CGG repeat length, such as mid-range vulnerability (90–110 repeats), toxic gain-of-function production of mRNA, RAN translation, and FMRP variation (15–23). As such, detailed phenotypic characterization of the PM is important from a public health perspective, with potential to connect complex human traits to known genetic variation.
An important body of work has described a number of clinical-behavioral traits among carriers of the FMR1 PM, including ASD and subclinical ASD-related traits (24–27), anxiety and depression (28–33), and differences in social cognition and executive function (EF) (34–38). Because most prior work has examined these phenotypes in separate study samples, a key remaining question concerns whether such phenotypes co-occur, together constituting a phenotypic profile associated with the FMR1 PM. Examining the co-occurrence of key phenotypes within individuals can also help to address whether such features may interrelate causally. It could be, for instance, that personality traits previously reported in PM groups reflect underlying differences in social cognition or EF. This study attempted to build on prior work to address these gaps by studying a range of clinical-behavioral, social-cognitive, and executive phenotypes associated with the FMR1 PM within a relatively large sample of female PM carriers and controls.
Consistent with the strong phenotypic overlap observed between FXS and ASD (where most individuals with FXS exhibit at least some ASD symptoms, and many meet full diagnostic criteria for ASD) (24, 39–42), a number of studies have documented elevated rates of ASD among PM carriers, particularly males (~14%) (24, 25, 43). Subclinical ASD traits have also been noted in the PM more generally, including personality styles such as social reticence (28) and rigid and perfectionistic traits (27, 34). Collectively known as the broad autism phenotype (BAP), this constellation of subclinical personality and language traits mirror the central features of ASD and are thought to index genetic liability to the disorder as they are observed at higher rates among first degree relatives of individuals with ASD relative to the general population (44–46) and associated with increased polygenic burden for ASD (47).
Losh et al. (27) evaluated personality and pragmatic language features of the BAP in PM carriers. Using direct assessment measures, including the Modified Personality Assessment Scale (MPAS) (48) and Pragmatic Rating Scale (PRS) (49), they found similar profiles of BAP personality traits and increased pragmatic language violations in PM carriers and mothers of individuals with ASD compared to controls. Further, within-family associations were detected in the PM group, showing that children with FXS whose mothers exhibited BAP traits had more severe ASD symptoms. Such co-segregation of ASD-related phenotypes within a subgroup of families is intriguing, particularly when considering known interactions between a number of ASD risk genes and the FMR1 gene that might underlie these phenotypes (50, 51).
Beyond ASD-related risk, female PM carriers may display elevated symptoms of anxiety and depression, though rates vary considerably between studies (30, 32, 33, 52, 53). For instance, whereas Jiraanont et al. (53) reported higher diagnostic rates of depression (50%) and anxiety (33%) compared to controls (8.3 vs. 4.2%, respectively), Gossett et al. (52) reported no significant differences, but noted that the majority of the control group (~54%) had clinically elevated symptoms. Studies including younger PM carriers and adults without children have also reported elevated mood and anxiety disorders, suggesting that these symptoms may arise early on and are not merely related to the stress of parenting a child with FXS (25, 32). Interestingly, there is some evidence to suggest that mood and anxiety symptoms may co-occur with other phenotypes, such as FXPOI and EF deficits (e.g., working memory), among subgroups of PM carriers (54, 55), underscoring the importance of examining how these symptoms may co-occur with other phenotypes, such as social-cognitive deficits and ASD-related features.
Subtle differences in social cognition (i.e., understanding mental states and feelings essential for supporting fluent interpersonal interactions) have also been observed among PM carriers. Studies of male PM carriers reported lower social-cognitive performance, including decreased theory of mind (34, 56) and reduced social reward processing, which related to lower FMRP (37). Male PM carriers have been reported to exhibit reduced neural activation in the amygdala compared to controls when viewing faces with fearful expressions (35). Far less is known about the social-cognitive profiles of females and potential biological correlates. Klusek et al. (57) found that, compared to controls, female PM carriers displayed reduced visual attention to others' direct gaze, which can impact the ability to interpret others' intent. These results were hypothesized to reflect difficulty with interpreting ambiguous social information or with recognizing direct gaze as an important social cue.
Even subtly impaired social-cognitive abilities have been associated with increased pragmatic language violations in ASD and FXS and the FMR1 PM (58–62). For example, subgroups of parents of individuals with ASD who displayed reduced social-cognitive skills tended to show elevated pragmatic language violations during semi-structured conversation (60). In FXS, increased pragmatic language deficits have been reported to cosegregate with more severe impairments in social cognition (62), and some evidence suggests that differences in social cognition among PM carriers may also relate to pragmatic language. In a study of visual attention, Winston et al. (63) reported atypical visual scanning patterns of faces among female PM carriers, but found that these differences were associated with better social cognition and pragmatic language, perhaps suggesting that some female PM carriers may employ alternative strategies for deducing meaningful information in social exchanges. Such findings suggest that subgroups of female PM carriers may exhibit social-cognitive or attentional patterns that facilitate pragmatic language, whereas for others, these domains may not be related. Consistent with this possibility, Winston et al. (64) identified a subgroup of PM carriers who demonstrated social viewing patterns characteristic of those found among parents of individuals with ASD and greater co-occurring pragmatic difficulties, and also had children with more severe ASD symptoms.
Numerous studies have also identified EF differences among female PM carriers without FXTAS (36, 65–67), including differences in working memory across both visual and verbal modalities (68, 69), inhibition (23, 54, 70), and attention (71). PM carriers also exhibit differences in language domains that are thought to reflect underlying EF difficulties, such as verbal disfluencies (72–74). Additionally, a recent study reported inefficient language processing and eye-voice coordination on a task of rapid naming of familiar objects; difficulties were particularly evident during the latter portions of the task when executive demands have been shown to be the greatest (22). The overlap between these language tasks and EF could suggest that aspects of the PM phenotype, such as subtle differences in pragmatic language, could cluster together with EF difficulties. For instance, Kraan et al. (54) found that EF deficits co-occurred with neuropsychiatric symptoms (such as depression and anxiety) in female PM carriers, though far less is known about the co-occurrence of executive dysfunction and other clinical-behavioral or cognitive correlates.
The present study aimed to further characterize the phenotypic spectrum associated with the FMR1 PM by examining performance across a battery of clinical-behavioral, social-cognitive, and EF tasks in females with the FMR1 PM in comparison to controls. Further, we utilized latent profile analyses to examine whether homogenous phenotypic subgroups within the PM might be identified based on distinct constellations of these phenotypes. Finally, following prior evidence that FMR1-related molecular-genetic variation is often associated with clinical-behavioral and cognitive phenotypes in the PM (21, 23, 30, 33, 37, 56, 75–77), we examined associations between the phenotypic battery and CGG repeat length and FMRP.
Participants included 152 female FMR1 PM carriers and 75 adult female controls. Only females were included in this study to control for biological sex, and to ensure feasible ascertainment. All participants were native English speakers because of the language-based nature of many of the tasks. PM carriers were recruited from genetic clinics, physicians' offices, advocacy groups, and the Research Participant Registry Core of the Carolina Institute for Developmental Disabilities at the University of North Carolina at Chapel Hill. PM status was confirmed by genetic testing (direct or confirmation by prior medical records). Controls were recruited through community resources (e.g., schools and child care centers, local community events) and word of mouth, as well as through the Communication Research Registry at Northwestern University. Controls were recruited as part of larger family genetics studies of ASD/BAP and FMR1-related conditions, and were therefore screened for personal or family history of FXS, ASD, and genetically-based conditions associated with ASD. Table 1 summarizes participant characteristics. Groups did not differ significantly in age (p = 0.233), or Full-Scale IQ (FSIQ; p = 0.250). In the PM sample, 135 participants had at least one child with FXS. Nine participants were mothers of children without FXS, and 16 were not mothers.
Potential participants were asked to report any prior diagnosis of FXTAS or Parkinsonism, and were excluded if they endorsed such symptoms. Additionally, participants completed a reduced set of screening questions from the FXTAS Rating Scale (78, 79), assessing action or postural tremor, standing capacities, tandem gait, and handwriting-related items. Four individuals were excluded for rating positive on one or both of these indices.
All study procedures were approved by the Institutional Review Boards of participating universities. All participants provided informed consent to participate.
The Modified Personality Assessment Schedule (MPAS) (48) was used to assess three core personality traits associated with the BAP (i.e., social aloofness, rigidity, and untactfulness) among the PM carrier group. This instrument has been used extensively in family studies of ASD [e.g., (44, 45)], and consists of a direct-assessment clinical interview designed to elicit examples regarding the endorsement of each personality trait. Traits are rated based on concrete behavioral examples using a 3-point scale (0 = absent, 1 = partially present, and 2 = present). Given that the BAP occurs at relatively low rates in individuals without a family history of ASD (46, 80), and the overarching goal of assessing how BAP personality traits might relate to other phenotypes among PM carriers, controls were not administered the MPAS.
Each interview was coded by two independent raters who were trained to at least 80% reliability, and were blind to group status, with final scores determined through consensus. Individuals were rated BAP (+) for social features if they had a score of 2 on either social aloofness or untactfulness, and BAP (+) for rigid features if they had a score of 2 in the rigid domain. Finally, individuals were rated BAP (–) if they scored either a 0 or 1 on all domains. Average reliability prior to consensus coding was 76.28%.
Twenty-minute semi-structured conversations were conducted between examiners and participants concerning their “life history” [for detailed description, see (27)]. Participants were asked about a series of topics that pertained to early childhood and friendships, current employment, hobbies, and romantic relationships. To elicit specific pragmatic behaviors during the conversation, such as reciprocity and the ability to clarify a message, examiners were trained to periodically offer related personal information and feign confusion. Conversations were coded from video by two trained research assistants, who were blind to participant family diagnosis, using the Pragmatic Rating Scale (PRS) (49). The PRS captures pragmatic language features of the BAP, and assesses 26 different pragmatic skills (e.g., providing adequate detail and background information), which are rated on a three-point scale from 0 (not present), 1 (somewhat present), to 2 (definitely present). In addition to a total number of pragmatic language violations, scores on three factor scores [dominant, withdrawn, and suprasegmental factors; see (27)] were also examined. All files were consensus coded for a best estimate rating used in analyses. Reliability prior to consensus coding was 84.07%.
Depression symptoms were assessed using the Beck Depression Inventory-II (BDI-II) (81). The BDI-II is a 21-item measure of depressive symptoms on a 4-point scale, and has been normed in both typical and clinical populations. It follows the Diagnostic and Statistical Manual of Mental Disorders-Fourth Edition (DSM-IV) criteria for depression and assesses the presence and severity of depression with high reliability (Coefficient Alpha = 0.92). Scores on this measure range from 0 to 63; scores falling in the 0–13 range suggest the presence of minimal depressive symptoms, 14–19 indicate mild, 20–28 moderate, and 29–63 severe.
The State-Trait Anxiety Inventory (STAI) (82) assessed anxiety symptoms. Used extensively in psychiatric research and practice, the STAI consists of two 20-item questionnaires that evaluate current (i.e., state) and more persistent (i.e., trait) symptoms of anxiety. The STAI provides a continuous measure of anxiety, with a range for each subtest of 20–80, with higher scores indicating greater anxiety (83, 84). Clinically significant anxiety has been suggested for scores at or above 39 (83, 84). Standard scores were used in all analyses.
The Mini-International Neuropsychiatric Interview (MINI) (85) is a well-validated and widely used structured psychiatric diagnostic interview instrument to evaluate current and past depressive episodes along DSM-IV criteria. This measure is intended as a tool for dichotomous categorization (i.e., yes/no) of psychiatric symptoms of depression and anxiety. Two-sided Fisher's exact tests were used to compare rates of symptoms across groups.
The Eyes Task (86) was used to index the ability to infer psychological states from viewing the eye region of the face. Participants were shown 36 different images of eyes expressing different psychological states and were asked to select a corresponding term from an array of four words. Performance was measured as the proportion of correct responses.
The Trustworthiness of Faces Task (87, 88) assessed the ability to use facial expressions to infer the social attribute of trustworthiness. Participants were asked to rate the trustworthiness of a series of 42 faces, which varied in gender, expression, and gaze. Ratings were made on a seven-point scale (−3 to +3), with negative scores denoting less trustworthiness, a score of “0” denoting neutral trustworthiness, and positive scores indicating greater trustworthiness. Faces were categorized into “negatively valenced” and “positively valenced” based on valence ratings from the original control group (87).
The Movie Stills Task (89) measured the extent to which individuals use facial information to infer the emotional content of a scene. Participants were asked to determine the emotional state (happy, sad, afraid, angry, surprised, disgusted, or neutral) from a series of 16 movie stills. The first trial obscured the faces of the characters in the image, and the second trial presented the image with the faces intact. Following prior work (89), scores were derived from the control participants (n = 49). For each of the 16 images with faces, the proportion of controls who selected a given emotion was first calculated to determine the distribution of responses. Scores were then weighted based on the distribution of responses, and parametrically transformed to give partial credit to alternative responses. For instance, if on a given image, 50% of the controls chose angry, 40% chose afraid, and 10% chose neutral, angry was given a score of 1, afraid was weighted as 0.8, and a neutral response was weighted as 0.2, whereas all other responses received no credit. For each image, the emotion that was the most often chosen by the controls was determined as the target emotion. Scores were then averaged for each emotion condition.
The ability to recognize emotions through biological motion was assessed using a task where participants viewed light emitting diodes affixed to an actor's body, as the figure moved through a black space in different manners (90). Participants completed two versions of this task—one in which they judged basic emotions (e.g., happy, angry) and another where they judged the trustworthiness conveyed by the body movements. As with the Movie Stills task, each emotion was given a weighted score based on the proportion of controls who selected each emotion per trial. The weighted scores were used to calculate emotional accuracy.
During the second version of the task, participants were asked to rate the trustworthiness of each display based on its pattern of movement. Trustworthiness was rated on a scale of 1–5, with 1 representing the most trustworthy and 5 representing the least trustworthy. For analyses and interpretation of this version, images were divided into “lower” and “higher” trustworthiness based on original control ratings. Raw ratings of trustworthiness were used in analyses.
Participants completed the Behavior Rating Inventory of Executive Function-Adult Version (BRIEF-A) (91), a 75-item self-report questionnaire that assesses multiple domains of executive functioning. Participants had the option to complete this self-report questionnaire in the lab, or remotely via an online link to the questionnaire. Each item was rated on a three-point Likert scale (i.e., never, sometimes, often), indicating the extent to which a behavior occurred over the past 6 months. Raw scores were converted into standardized t-scores across nine domains: Inhibition, Shift, Emotional Control, Self-Monitor, Initiate, Working Memory, Plan/Organize, Task Monitor, and Organization of Materials. Higher scores indicate poorer executive functioning abilities. Scores from each domain yield an overall Global Executive Composite (GEC) score, and two composite scores, Behavioral Regulation Index (BRI) and Metacognitive Index (MI). T-scores 65 or greater indicate clinically significant deficits in executive functioning.
Polymerase chain reaction and Southern blot techniques were used to confirm PM status and determine CGG repeat length. FMRP was assayed in lymphocytes isolated from blood, using a Luminex Assay to reliably quantify levels of FMRP (92). Activation ratio (AR) measures the proportion of cells carrying the normal allele on the active X chromosome (78), and was determined by the ratio of the intensity of the normal FMR1 unmethylated band divided by the sum of the intensities of the normal unmethylated and methylated bands (93). Table 2 presents descriptive information regarding FMR1-related variation.
All variables were examined for normality of distribution. General linear models (i.e., analyses of variance, ANOVAs) were used to assess group differences across clinical-behavioral, social-cognitive, and EF measures. Effect sizes for correlational analyses were interpreted as small (0.20), medium (0.50), and large (0.80) (94). Effect sizes for ANOVAs are reflected as partial eta squared (), and were interpreted as small (0.01), medium (0.06), and large (0.14) (94). Bonferroni corrections were applied to adjust for multiple comparisons. Because groups did not differ significantly on age or IQ (p > 0.153), and age and IQ were generally not associated with variables of interest, we did not control for these variables in analyses.
To examine whether there may be subgroups who display specific patterns of performance across tasks, we conducted latent profile analyses (LPA) in the PM group. LPA serves to identify latent subpopulations having different configural profiles based on variables hypothesized to comprise meaningful phenotypes (95). A total of 14 variables reflecting performance across measures were included as numerical indicators in the LPA (see Table 3). Given numerical scale differences between measures, all variables were z-scored to improve interpretability.
Using an iterative process, we evaluated LPA solutions ranging from one to six potential PM profiles using the following fit measures to determine the best solution: the Akaike Information Criterion (AIC), the Bayesian Information Criterion (BIC), Entropy, a Bootstrap Likelihood Ratio Test (BLRT), and a Sattora–Bentler Scaled likelihood ratio chi-square difference test (TRd) (95–98). We also considered the theoretical interpretability of the profiles and profile size (for technical details on LPA, see Supplementary Materials). Profile subgroups were examined in follow-up analyses to evaluate any meaningful differences in age, IQ, FMR1-related genetic variation (i.e., CGG repeat length, AR, and quantitative FMRP), maternal status, or severity of ASD symptoms in their children (measured in oldest child in multiplex families using the ADOS-2) (99).
Associations between clinical-behavioral and cognitive phenotypes with molecular-genetic variables (i.e., FMR1 CGG repeat length, FMRP) were conducted in the PM group only using Pearson correlations. Subsequently, linear regression models were applied to examine the extent to which molecular-genetic variables might predict phenotypic outcomes. For regression models that included CGG repeat length, separate models were conducted using linear and curvilinear CGG terms (i.e., CGG squared) given evidence of varied associations between CGG repeat length and behavioral measures in the PM (20, 21, 33). Given the presence of two X chromosomes in females, in models that included CGG repeat length, AR and an interaction term (i.e., CGG × AR) were included as covariates.
Among those with MPAS data in the PM group (n = 87), 54% of the sample was characterized as BAP (+). Thirty-two percent of the sample exhibited either social or rigid BAP personality features (ns = 28), and 10% of the sample (n = 9) displayed both social and rigid BAP personality features. Prior studies using the MPAS indicate ~10–15% of controls display any BAP personality features (27, 46).
Females with the PM exhibited significantly higher total PRS scores (i.e., more violations) than the control group [F(1, 185) = 5.65, p =0.019, and = 0.03]. Examining factor scores on the PRS revealed that the PM group demonstrated a more dominant conversational style than controls [F(1,174) = 7.67, p =0.006, and = 0.04], but did not differ from controls in their withdrawn [F(1,174) = 0.60, p = 0.439, and = 0.00] or suprasegmental scores [F(1,174) = 2.08, p = 0.151, = 0.01, see Figure 1].
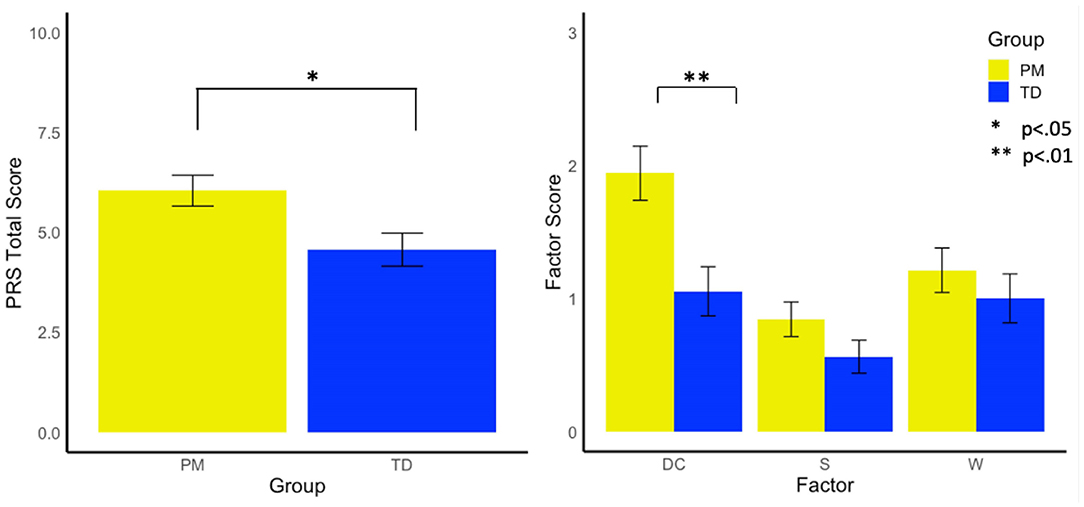
Figure 1. Group differences in pragmatic rating scale total scores and factors. DC, Dominates Conversation Factor; S, Suprasegmental Factor; W, Withdrawn Factor.
PM carriers did not significantly differ from controls on BDI-II total scores [F(1, 69) = 2.58, p = 0.113, = 0.04].
PM carriers endorsed marginally more Trait and State anxiety than controls [F(1 77) = 3.72, p = 0.058, = 0.05; F(1, 76) = 3.02, p = 0.086, = 0.04, respectively]. Clinically significant symptoms of anxiety (>39) were self-reported in 94.7% of PM carriers and 90% of controls.
PM carriers and controls did not differ on rates of current generalized anxiety disorder [22 vs. 14%, = 1.40, p = 0.285], past generalized anxiety disorder [19.6 vs. 11.9%, = 1.23, p = 0.338], current major depressive disorder [5 vs. 0%, = 2.75, p = 0.164], or past major depressive disorder [25.7 vs. 19.6%, = 0.75, p = 0.440].
PM carriers had marginally lower scores on the Eyes Task than controls [F(1, 203) = 1.49 p = 0.075, = 0.02].
PM carriers demonstrated significantly lower overall performance on movie stills with faces [F(1, 23) = 6.03, p = 0.015, = 0.03], but did not differ from controls in performance on stimuli without faces (p = 0.389). PM carriers had marginally lower scores on neutral and sad stimuli with faces (p = 0.074 and 0.069, respectively).
PM carriers scored significantly lower than controls on happy stimuli [F(1, 97) = 4.25, p = 0.042, = 0.04], but did not differ in overall performance (p = 0.256) or on other emotion types (p > 0.145).
No differences emerged between groups in overall point light trustworthiness scores [F(1, 24) = 0.94, p = 0.335, = 0.008], or for more trustworthy or less trustworthy stimuli [F(1, 24) = 0.74, p = 0.392, = 0.006; F(1, 24) = 0.65, p = 0.422, = 0.005, respectively].
PM carriers rated faces as significantly less trustworthy than controls overall [F(1, 147) = 9.31, p = 0.003, = 0.06]; differences were evident on both positive and negative valanced faces [F(1, 147) = 9.80, p = 0.002, = 0.06; F(1, 147) = 7.74, p = 0.006, = 0.05, respectively; see Figure 2].
Twenty-nine percent of PM carriers exceeded clinical cut-off on the GEC of the BRIEF-A, whereas only one female control exceeded clinical cut-off. PM carriers scored significantly higher (i.e., greater EF difficulty) than controls on the GEC, BRI, and MI scales [F(1, 58) = 6.55, p = 0.013, = 0.10; F(1, 58) = 4.47, p =0.039, = 0.07; F(1, 58) = 6.81, p = 0.012, = 0.12; see Figure 3].
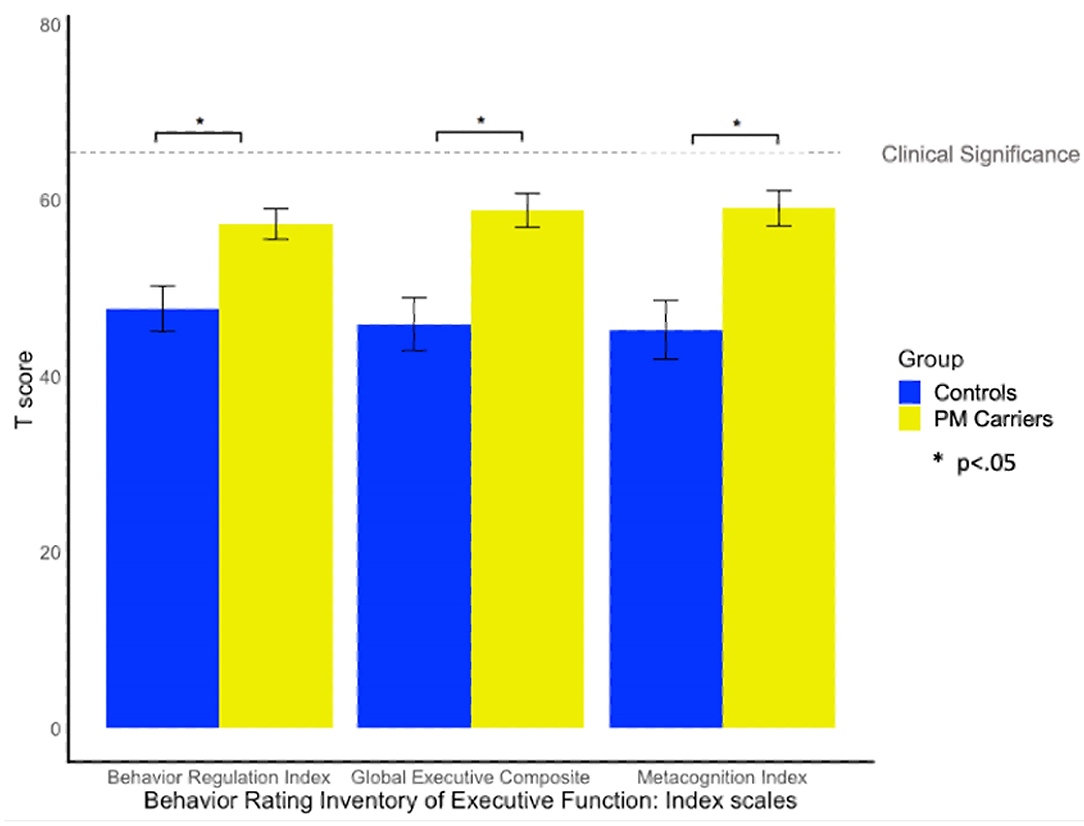
Figure 3. Group differences on the BRIEF-A. BRIEF-A T-scores > 65 indicate clinically elevated difficulties relative to the standardization sample.
LPA solutions for up to six models are presented in Table 4, and a three-profile solution was selected for interpretability.
The results of the three-profile solution are presented in Figure 4. The first profile (Profile 1) contained 77.6% of PM carriers (n = 118, average posterior probability = 0.983), and included individuals whose scores across domains consistently fell around the PM group mean, with limited variation across domains. The second profile (Profile 2) contained 17% of PM carriers (n = 26, average posterior probability = 0.871) and reflected individuals with increased mood and anxiety symptoms, slightly elevated social and rigid personality features of the BAP, and increased pragmatic language violations in the suprasegmental domain (e.g., intonation of voice, rate of speech, and volume modulation) as compared to other PM carriers. The final profile (Profile 3) contained 5.3% of PM carriers (n = 8, average posterior probability = 0.959) and included individuals who demonstrated elevated executive dysfunction, poorer social-cognitive abilities across tasks, elevated social and rigid personality features of the BAP, and increased pragmatic language violations in the listener expectation domain (e.g., unable to clarify, failure to reciprocate) relative to other PM carriers.
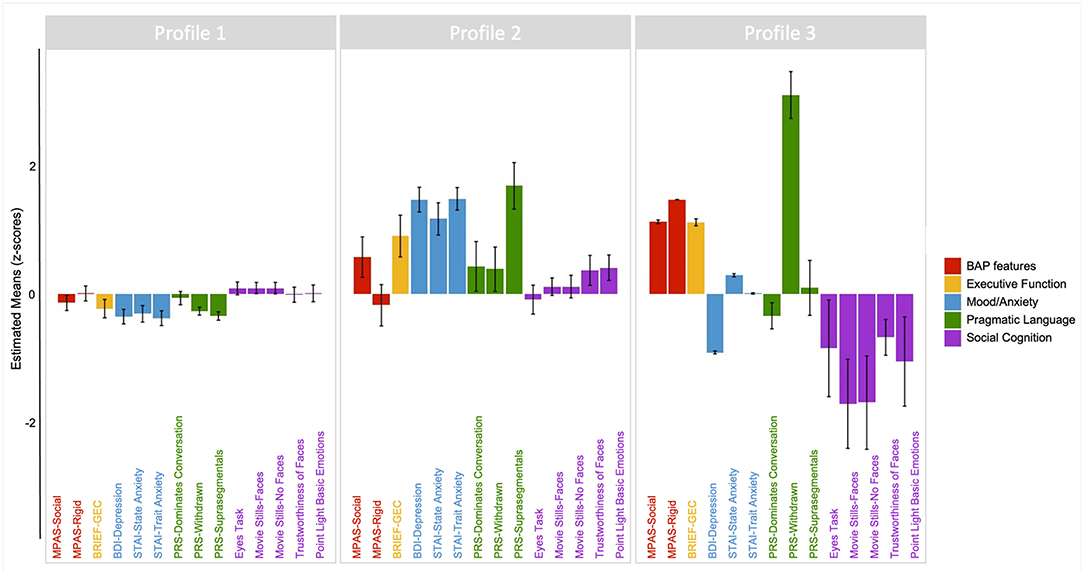
Figure 4. Latent profile groups emerging from performance profiles across clinical-behavioral, pragmatic language, social cognitive, and executive tasks. Positive scores reflect poorer or more atypical performance along the following: BAP personality features (red); Executive function (yellow); Mood/anxiety (blue); Pragmatic language features of the BAP (green). For social cognition (purple), negative scores reflect poorer performance relative to the mean.
Follow up comparisons showed no significant differences across the profile groups in age, IQ, FMR1-related genetic variation, maternal status, or presence of ASD diagnosis or severity of ASD symptoms in their children (all ps > 0.200).
A linear regression model including CGG repeat length predicted a significant amount of variance in PRS scores (R2 = 0.09, p = 0.046; see Table 5). No other associations were observed between CGG repeat length and other clinical-behavioral, social-cognitive, or EF measures (p > 0.169).
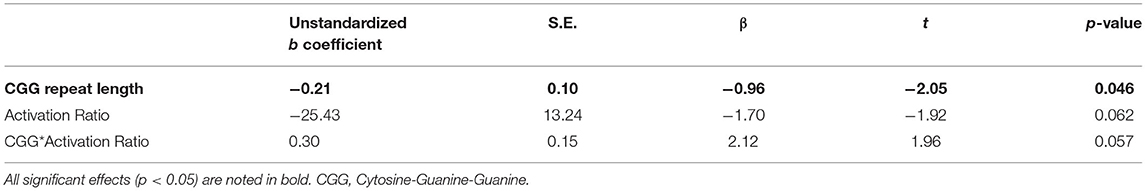
Table 5. CGG repeat length predicts elevated pragmatic language violations (on Pragmatic Rating Scale).
FMRP was a significant predictor of performance on one social-cognitive task: Movie Stills (Happy) with faces. Specifically, quantitative FMRP predicted 12% of the variance in Movie Stills (Happy) with faces (b = −0.06, p = 0.005). Quantitative FMRP levels were not significantly associated with other clinical-behavioral, social-cognitive, or EF measures (p > 0.131).
This study characterized clinical-behavioral, social-cognitive, and EF features in females with the FMR1 PM. Consistent with prior work, results revealed elevated rates of subclinical ASD-related personality and language features among women with the PM, as well as increased self-reported difficulties in executive functioning, but no differences in mood and anxiety symptoms compared to controls. Some differences in social-cognitive tasks were also observed, including differences in complex social-emotional judgements of trustworthiness of faces, and in accuracy identifying basic emotions when viewing complex scenes. Latent profile analysis revealed three subgroups within the PM group who exhibited distinct phenotypic profiles across clinical-behavioral, social-cognitive, and EF measures, which together with group differences, and some associations with FMR1-related variation, may provide insights into phenotypic profiles associated with the FMR1 PM.
Replicating prior work, personality and pragmatic language differences that define the BAP were observed at elevated rates in the PM carrier group. Direct measurement tools, scored by raters blind to group status, identified approximately half of the PM carrier group as displaying personality traits consistent with the BAP, compared with published rates among individuals without a family history of ASD or FXS ranging from ~5 to 10% [e.g., (46, 80)]. Pragmatic language differences have been repeatedly observed among PM carriers (27, 57, 100–102), and were evident in this study as well, with additional patterns noted across the types of pragmatic language violations occurring more frequently in the PM group, who tended to display a more dominant conversational style (e.g., overly detailed, tangential language) than controls. Differences in pragmatic language have consistently emerged as a phenotypic marker associated with the PM in females (27, 100–102), and among individuals with FXS, particularly those who meet criteria for ASD (62, 103–109) and may be of clinical importance. For instance, prior work has shown that pragmatic language violations among mothers of children with FXS were associated with reduced self-reported quality of life for mothers (102), as well as elevated ASD symptoms and weaker expressive and receptive language in their children (27, 100). Together, this suggests that pragmatic language may be relevant to the well-being of both mothers and their children.
In contrast to previous work demonstrating higher rates of mood and anxiety disorders in PM carriers (25, 28, 30), we found no significant differences in depression scores between PM carriers and controls and only marginal differences from controls for anxiety symptoms, with both groups self-reporting elevated anxiety symptoms (>90% of participants). Rates of anxiety and depression were far lower in both groups using the MINI, a standardized psychiatric interview (ranging from 0 to 25%), and also comparable to rates reported in prior work (31, 33, 110). In follow-up analyses, no differences were observed on depression and anxiety features according to maternal status, or the number of affected children in each family, suggesting these findings are not related to parenting factors.
Unique to the present study was the inclusion of multiple measures of social cognition within a single sample, permitting characterization of social cognitive strengths and weaknesses across different types of stimuli. Differences in social-cognitive profiles have been reported among PM carriers compared to controls, particularly among males (35, 37). This study identified key social-cognitive differences in female carriers of the PM in specific tasks, which echo findings observed among individuals with FXS (62, 111). PM carriers differed in rating trustworthiness in response to faces ranging in emotional valence, a task that draws on social-cognitive and social decision-making skills. Differences on this task have been reported in ASD and the BAP in clinically unaffected relatives (61), and also among patients with bilateral amygdala damage, implicating this brain region in atypical performance in these groups (87, 89, 112). In studies of the PM, differences in amygdala volume and activation have been linked to aspects of social cognition among male carriers (35, 37), and these findings may suggest similar relationships among women with the PM that will be important to investigate in future work.
PM carriers were also less accurate in inferring emotions from faces when viewing still movie scenes. These results may indicate that PM carriers use different strategies in making social judgments relative to controls when viewing faces. Prior work that used eye tracking to examine looking patterns in response to faces indicates that PM carriers use different visual strategies from controls to inform social judgments (63). In the present study, marginal differences were also observed on the task involving reading complex thoughts and emotions from the eye region of the face. Thus, sensitivity to gaze and emotion expression, as measured by these social-cognitive tasks, may be an objective behavioral marker of underlying neural processes associated with interpreting emotion within the PM (35).
Consistent with prior reports of EF impairment associated with the PM (113), the PM group exhibited significantly higher executive functioning difficulty than controls, with approximately one third of the PM group reporting clinically significant EF difficulties. Characterization of the cognitive phenotype associated with the PM is especially important for understanding the manifestation of the neurodegenerative disorder, FXTAS, which may reveal subclinical phenotypic markers that are evident in a subgroup of PM carriers who go on to develop the disorder, as dysexecutive symptoms are a hallmark of FXTAS, particularly in males (114). Nonetheless, females with the PM exhibit differences in EF symptoms even without a diagnosis of FXTAS (68, 69, 115, 116). This study highlights the value of using self-report measures of EF in studies of the PM, as prior work has typically used online behavioral measures [e.g., see (113) for review]. The use of the BRIEF-A among females with the PM has been limited to only a handful of prior studies (23, 117), but is useful in that clinical significance may be easily determined and a standardized self-report measure can enable cross-cohort comparisons. Of note, however, such self-report measures could lead to over-reporting of symptoms (118, 119), and so may best be interpreted within the context of results from studies employing direct assessment of executive skills.
Complementing results from group comparisons, latent profile analyses revealed three distinct groups of PM carriers who displayed different profiles of performance across the various domains. Profile 1 comprised the largest subgroup, and represented those scoring at the mean of the sample across measures, and who largely contributed to the group differences observed in pragmatic language and select social cognitive tasks and EF. Profile 2 included 17% of the sample, and was characterized by elevated and co-occurring mood and anxiety symptoms, mild expression of personality features of the BAP, and higher suprasegmental violations (e.g., atypical variation in intonation, volume, or rate of speech). This specific co-occurrence of features is not surprising in the context of social-emotional patterns commonly observed among individuals who are more anxious or depressed, in that their symptoms can interfere with their social relationships, and vice versa (120). Allen et al. (55) also reported distinct clusters of PM carriers who reported different mood and anxiety symptoms, and other work has reported the co-occurrence of mood/anxiety features with executive dysfunction among PM carriers (54). Consistent with prior work (121), this subgroup also exhibited slightly elevated BAP traits, which raises the possibility that the co-occurrence of such traits may also be common to a subgroup of family members of individuals with ASD (of note, individuals showing Profile 3, discussed below, demonstrated elevated BAP features in the absence of elevated mood/anxiety symptoms). Although suprasegmental speech violations committed by PM carriers in this subgroup does not constitute clinical impairment, among individuals with ASD, where suprasegmental atypicalities are more pronounced, such variation can pose a significant obstacle to social interactions (122). First-degree relatives of individuals with ASD also demonstrate subtle differences in suprasegmental aspects of language (e.g., prosody) (123, 124).
Profile 3 represented the smallest subgroup, comprised of ~5% of PM participants who exhibited starker differences across all clinical-behavioral, social-cognitive, and executive domains relative to other PM carriers. This subgroup displayed notable differences on social-cognitive tasks together with elevated BAP features, and high rates of pragmatic language violations in particular. This pattern of performance is markedly similar to features described in prior investigations of parents of individuals with ASD who display the BAP (60, 61). Given the large number of ASD risk genes known to interact with FMR1 (50, 51), it may be that this phenotypic profile reflects an increased genetic liability for ASD among this subgroup. However, the small size of Profile 3 warrants cautious interpretation. We found no group differences in age, IQ, or FMR1-related variation between the Profile subgroups, likely due in part to some missing data in Profile 3 and unequal sample sizes across groups, and further investigations in larger samples will be important for confirming these patterns. Importantly, membership within any of the profile groups was not associated with differences in age, IQ, or parenting stress-related factors (including number of affected children, or severity of child symptoms), suggesting the phenotypic profiles identified are likely reflective of inherent traits, rather than systematic environmental differences between subgroups.
Finally, some associations were detected between phenotypic profiles and FMR1-related molecular genetic variability in the PM carrier group. We found linear associations between CGG repeat length and pragmatic language, indicating greater violations at lower ends of the CGG continuum. The CGG range reflected in the participants studied extended from 59 to 126 CGGs; thus, it may be that inclusion of more PM carriers with higher repeats (i.e., over 120) might have altered the findings observed here. Prior work has observed curvilinear links between language and CGG length, with different patterns noted in the mid-range (90–110 repeats) as compared to those with CGG repeats beyond 120 (21). Interestingly, the linear CGG association became marginal after factoring in participants' activation ratios, suggesting the importance of considering the second, healthy X allele in phenotype-genotype associations of females with the FMR1 PM. Additionally, we found that higher levels of FMRP predicted poorer performance on one social-cognitive task. This finding is somewhat in contrast to those from Hessl et al. (37), who found associations between reduced FMRP and poorer performance on social processing tasks in male PM carriers. Interestingly, a prior study based on the PM sample of participants studied here reported increased FMRP related to poorer performance on a language fluency task that taps into executive functioning (22), which together may suggest that the findings observed here may be specific to our sample of PM carriers (and thus may not be replicable), or that FMRP from blood is not analogous to FMRP in the brain. The links between FMR1 and the phenotypes included in the present study are likely not straightforward, and it is possible that other FMR1-related factors (e.g., mosaicism, mRNA) could also help to elucidate FMR1-associated patterns not explored in this study (125, 126).
Together, findings contribute to an emerging profile of PM carriers that suggests substantial phenotypic variability, and the presence of distinct phenotypic subgroups that may reveal important differences in underlying mechanistic factors and etiology (e.g., involvement of ASD risk genes interacting with FMR1). Strengths of this study included the broad phenotypic characterization of a relatively large group of females with the PM. Examining an array of phenotypic measures in a single sample enabled us to investigate comparisons with controls as well as unique profiles among PM carriers. Nevertheless, a larger sample may have mitigated some of the concerns with the small subgroup sample sizes yielded in our latent profile analyses. We were limited in our attempts at a validation analysis of the three-profile model through comparison across such important factors as FMR1-related variation, though it is possible that other individual factors not available in the present study might have differentiated subgroups, such as direct measures of caregiving stress (127, 128), the presence of other co-occurring health conditions (55), or polygenic risk for ASD (47). There is emerging literature to suggest subgroups among PM carriers, and indeed the clinical disorders associated with the PM (i.e., FXTAS, FXPOI, and FXAND) only occur among a subset of PM carriers [e.g., see (55), and for review, (113, 129)]. Large scale studies of the PM are warranted to further investigate the interrelationships observed here, and to determine whether the phenotypes included in this study may co-occur with other meaningful clinical or health outcomes, as has been documented previously (55). Additionally, we were limited in FMR1-related information, as we did not have genetic data on the control group in this study. Further, there is emerging evidence to suggest that phenotypic associations may be observed across the range of CGG repeats (21, 130, 131). It may be that FMR1 relationships with phenotypes were not detected given that the range of repeats in the current study was limited to the PM range. It may also be that molecular parameters in blood do not correlate with cognitive assays in the brain in straightforward ways. We recognize that due to our exclusion criteria, our control group might be expected to have performed well on the measures employed here. Larger and more heterogeneous control groups should be included in future studies, or in comparison to mothers of children with ASD. Finally, it will be important for future work to examine whether the findings reported here may extend to males with the PM, given prior evidence to suggest females and males with the PM may exhibit somewhat different phenotypic profiles and associations with underlying biology (29, 132). Such studies may build on the present findings, and help to characterize the phenotypic profile associated with the FMR1 PM, and inform clinical efforts to promote the health and well-being of individuals with the PM and their families.
This study provided a comprehensive assessment of clinical and subclinical phenotypes associated with the FMR1 PM. We identified differences from controls on pragmatic language features of the BAP, executive functioning, and some aspects of social cognition, but did not observe differences in mood and anxiety. Using LPA, we found subgroups within the PM sample characterized by unique patterns of performance on these measures. This study adds to a growing literature suggestive of important phenotypic heterogeneity among PM carriers, and provides further insight into FMR1-associated phenotypes.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found below: https://nda.nih.gov/edit_collection.html?id=1958.
The studies involving human participants were reviewed and approved by Northwestern University Institutional Review Board. The patients/participants provided their written informed consent to participate in this study.
NM helped to conceptualize the project, drafted the initial manuscript, and analyzed the data. JG, KN, MW, and JL assisted in preparing the dataset and provided valuable feedback on manuscript drafts. GM contributed to manuscript preparation. LB, SP, and MLe contributed to data preparation and processing. JS provided further consultation and verification of statistical methods and results. DH advised on PM participant evaluation of neurocognitive functioning. LZ, KS, and EB-K provided resources for genetic analysis of the FMR1 gene, helped to draft the Materials and Methods section pertaining to genetic analyses, and helped with interpretation of results. MLo secured funding for the projects from which these data were drawn, conceptualized the project, helped to interpret data, and develop the manuscript. All authors reviewed and approved the manuscript.
The research reported in this publication was supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development of the National Institutes of Health under award numbers R01 HD038819, P30 HD03110, and T32 HD007489, the National Institute of Mental Health under award numbers R01 MH091131 and R03 MH079998, the National Institute on Deafness and Other Communication Disorders under award number R01 DC010191 and P30 DC012035, and the National Science Foundation under award number DGE-1324585. Additional funding was received from the Developmental Science Institute Doctoral Student Research Grant at Northwestern University.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
We are grateful for the many individuals who participated in this study.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpsyt.2021.718485/full#supplementary-material
1. Nolin SL, Lewis FA, Ye LL, Houck GE, Glicksman AE, Limprasert P, et al. Familial transmission of the FMR1 CGG repeat. Am J Hum Genet. (1996) 59:1252–61.
2. Hagerman RJ, Hagerman PJ. The fragile X premutation: into the phenotypic fold. Curr Opin Genet Dev. (2002) 12:278–83. doi: 10.1016/S0959-437X(02)00299-X
3. Huber KM, Gallagher SM, Warren ST, Bear MF. Altered synaptic plasticity in a mouse model of fragile X mental retardation. Proc Natl Acad Sci U S A. (2002) 99:7746–50. doi: 10.1073/pnas.122205699
4. Sullivan AK, Marcus M, Epstein MP, Allen EG, Anido AE, Paquin JJ, et al. Association of FMR1 repeat size with ovarian dysfunction. Hum Reprod. (2005) 20:402–12. doi: 10.1093/humrep/deh635
5. Coffee B, Keith K, Albizua I, Malone T, Mowrey J, Sherman SL, et al. Incidence of fragile X syndrome by newborn screening for methylated FMR1 DNA. Am J Hum Genet. (2009) 85:503–14. doi: 10.1016/j.ajhg.2009.09.007
6. Hunter J, Rivero-Arias O, Angelov A, Kim E, Fotheringham I, Leal J. Epidemiology of fragile X syndrome: a systematic review and meta-analysis. Am J Med Genet A. (2014) 164:1648–58. doi: 10.1002/ajmg.a.36511
7. Wang LW, Berry-Kravis E, Hagerman RJ. Fragile X: leading the way for targeted treatments in autism. Neurotherapeutics. (2010) 7:264–74. doi: 10.1016/j.nurt.2010.05.005
8. Seltzer MM, Baker MW, Hong J, Maenner M, Greenberg J, Mandel D. Prevalence of CGG expansions of the FMR1 gene in a US population-based sample. Am J Med Genet B Neuropsychiatr Genet. (2012) 159:589–97. doi: 10.1002/ajmg.b.32065
9. Tassone F, Iong KP, Tong TH, Lo J, Gane LW, Berry-Kravis E, et al. FMR1 CGG allele size and prevalence ascertained through newborn screening in the United States. Genome Med. (2012) 4:100. doi: 10.1186/gm401
10. Maenner MJ, Baker MW, Broman KW, Tian J, Barnes JK, Atkins A, et al. FMR1 CGG expansions: prevalence and sex ratios. Am J Med Genet B Neuropsychiatr Genet. (2013) 162B:466–73. doi: 10.1002/ajmg.b.32176
11. Macpherson JN, Murray A. Development of genetic testing for fragile X syndrome and associated disorders, and estimates of the prevalence of FMR1 expansion mutations. Genes (Basel). (2016) 7:110. doi: 10.3390/genes7120110
12. Owens KM, Dohany L, Holland C, Dare J, Mann T, Settler C, et al. FMR1 premutation frequency in a large, ethnically diverse population referred for carrier testing. Am J Med Genet A. (2018) 176:1304–8. doi: 10.1002/ajmg.a.38692
13. Hagerman RJ, Hagerman P. Fragile X-associated tremor/ataxia syndrome - features, mechanisms and management. Nat Rev Neurol. (2016) 12:403–12. doi: 10.1038/nrneurol.2016.82
14. Hagerman RJ, Protic D, Rajaratnam A, Salcedo-Arellano MJ, Aydin EY, Schneider A. Fragile X-associated neuropsychiatric disorders (FXAND). Front Psychiatry. (2018) 9:564. doi: 10.3389/fpsyt.2018.00564
15. Tassone F, Hagerman RJ, Taylor AK, Gane LW, Godfrey TE, Hagerman PJ. Elevated levels of FMR1 mRNA in carrier males: a new mechanism of involvement in the fragile-X syndrome. Am J Hum Genet. (2000) 66:6–15. doi: 10.1086/302720
16. Kenneson A, Zhang F, Hagedorn CH, Warren ST. Reduced FMRP and increased FMR1 transcription is proportionally associated with CGG repeat number in intermediate-length and premutation carriers. Hum Mol Genet. (2001) 10:1449–54. doi: 10.1093/hmg/10.14.1449
17. Ennis S, Ward D, Murray A. Nonlinear association between CGG repeat number and age of menopause in FMR1 premutation carriers. Eur J Hum Genet. (2006) 14:253–5. doi: 10.1038/sj.ejhg.5201510
18. Seltzer MM, Abbeduto L, Greenberg JS, Almeida D, Hong J, Witt W. Biomarkers in the study of families of individuals with developmental disabilities. Int Rev Res Ment Retard. (2009) 37:213–49. doi: 10.1016/S0074-7750(09)37007-X
19. Hagerman R, Hagerman P. Advances in clinical and molecular understanding of the FMR1 premutation and fragile X-associated tremor/ataxia syndrome. Lancet Neurol. (2013) 12:786–98. doi: 10.1016/S1474-4422(13)70125-X
20. Mailick MR, Hong J, Greenberg J, Smith L, Sherman S. Curvilinear association of CGG repeats and age at menopause in women with FMR1 premutation expansions. Am J Med Genet B Neuropsychiatr Genet. (2014) 165B:705–11. doi: 10.1002/ajmg.b.32277
21. Klusek J, Porter A, Abbeduto L, Adayev T, Tassone F, Mailick MR, et al. Curvilinear association between language disfluency and FMR1 CGG repeat size across the normal, intermediate, and premutation range. Front Genet. (2018) 9:344. doi: 10.3389/fgene.2018.00344
22. Nayar K, Mckinney W, Hogan AL, Martin GE, La Valle C, Sharp K, et al. Language processing skills linked to FMR1 variation: a study of gaze-language coordination during rapid automatized naming among women with the FMR1 premutation. PLoS One. (2019) 14:e0219924. doi: 10.1371/journal.pone.0219924
23. Klusek J, Hong J, Sterling A, Berry-Kravis E, Mailick MR. Inhibition deficits are modulated by age and CGG repeat length in carriers of the FMR1 premutation allele who are mothers of children with fragile X syndrome. Brain Cogn. (2020) 139:105511. doi: 10.1016/j.bandc.2019.105511
24. Clifford S, Dissanayake C, Bui QM, Huggins R, Taylor AK, Loesch DZ. Autism spectrum phenotype in males and females with fragile X full mutation and premutation. J Autism Dev Disord. (2007) 37:738–47. doi: 10.1007/s10803-006-0205-z
25. Bailey DB, Raspa M, Olmsted M, Holiday DB. Co-occurring conditions associated with FMR1 gene variations: findings from a national parent survey. Am J Med Genet A. (2008) 146A:2060–9. doi: 10.1002/ajmg.a.32439
26. Bourgeois JA, Coffey SM, Rivera SM, Hessl D, Gane LW, Tassone F, et al. A review of fragile X premutation disorders: expanding the psychiatric perspective. J Clin Psychiatry. (2009) 70:852–62. doi: 10.4088/JCP.08r04476
27. Losh M, Klusek J, Martin GE, Sideris J, Parlier M, Piven J. Defining genetically meaningful language and personality traits in relatives of individuals with fragile X syndrome and relatives of individuals with autism. Am J Med Genet B Neuropsychiatr Genet. (2012) 159B:660–8. doi: 10.1002/ajmg.b.32070
28. Franke P, Leboyer M, Gänsicke M, Weiffenbach O, Biancalana V, Cornillet-Lefebre P, et al. Genotype-phenotype relationship in female carriers of the premutation and full mutation of FMR-1. Psychiatry Res. (1998) 80:113–27. doi: 10.1016/S0165-1781(98)00055-9
29. Hunter JE, Allen EG, Abramowitz A, Rusin M, Leslie M, Novak G, et al. Investigation of phenotypes associated with mood and anxiety among male and female fragile X premutation carriers. Behav Genet. (2008) 38:493–502. doi: 10.1007/s10519-008-9214-3
30. Roberts JE, Bailey DB, Mankowski J, Ford A, Sideris J, Weisenfeld LA, et al. Mood and anxiety disorders in females with the FMR1 premutation. Am J Med Genet B Neuropsychiatr Genet. (2009) 150B:130–9. doi: 10.1002/ajmg.b.30786
31. Bourgeois JA, Seritan AL, Casillas EM, Hessl D, Schneider A, Yang Y, et al. Lifetime prevalence of mood and anxiety disorders in fragile X premutation carriers. J Clin Psychiatry. (2011) 72:175–82. doi: 10.4088/JCP.09m05407blu
32. Cordeiro L, Abucayan F, Hagerman R, Tassone F, Hessl D. Anxiety disorders in fragile X premutation carriers: preliminary characterization of probands and non-probands. Intractable Rare Dis Res. (2015) 4:123–30. doi: 10.5582/irdr.2015.01029
33. Roberts JE, Tonnsen BL, Mccary LM, Ford AL, Golden RN, Bailey DB. Trajectory and predictors of depression and anxiety disorders in mothers with the FMR1 premutation. Biol Psychiatry. (2016) 79:850–7. doi: 10.1016/j.biopsych.2015.07.015
34. Cornish K, Kogan C, Turk J, Manly T, James N, Mills A, et al. The emerging fragile X premutation phenotype: evidence from the domain of social cognition. Brain Cogn. (2005) 57:53–60. doi: 10.1016/j.bandc.2004.08.020
35. Hessl D, Rivera S, Koldewyn K, Cordeiro L, Adams J, Tassone F, et al. Amygdala dysfunction in men with the fragile X premutation. Brain. (2007) 130:404–16. doi: 10.1093/brain/awl338
36. Hunter JE, Abramowitz A, Rusin M, Sherman SL. Is there evidence for neuropsychological and neurobehavioral phenotypes among adults without FXTAS who carry the FMR1 premutation? A review of current literature. Genet Med. (2009) 11:79–89. doi: 10.1097/GIM.0b013e31818de6ee
37. Hessl D, Wang JM, Schneider A, Koldewyn K, Le L, Iwahashi C, et al. Decreased fragile X mental retardation protein expression underlies amygdala dysfunction in carriers of the fragile X premutation. Biol Psychiatry. (2011) 70:859–65. doi: 10.1016/j.biopsych.2011.05.033
38. Shelton AL, Cornish KM, Kolbe S, Clough M, Slater HR, Li X, et al. Brain structure and intragenic DNA methylation are correlated, and predict executive dysfunction in fragile X premutation females. Transl Psychiatry. (2016) 6:e984. doi: 10.1038/tp.2016.250
39. Abbeduto L, Mcduffie A, Thurman AJ. The fragile X syndrome-autism comorbidity: what do we really know? Front Genet. (2014) 5:355. doi: 10.3389/fgene.2014.00355
40. Yu TW, Berry-Kravis E. Autism and fragile X syndrome. Semin Neurol. (2014) 34:258–65. doi: 10.1055/s-0034-1386764
41. Thurman AJ, Mcduffie A, Kover ST, Hagerman RJ, Abbeduto L. Autism symptomatology in boys with fragile X syndrome: a cross sectional developmental trajectories comparison with nonsyndromic autism spectrum disorder. J Autism Dev Disord. (2015) 45:2816–32. doi: 10.1007/s10803-015-2443-4
42. Kaufmann WE, Kidd SA, Andrews HF, Budimirovic DB, Esler A, Haas-Givler B, et al. Autism spectrum disorder in fragile X syndrome: cooccurring conditions and current treatment. Pediatrics. (2017) 139:S194–S206. doi: 10.1542/peds.2016-1159F
43. Farzin F, Perry H, Hessl D, Loesch D, Cohen J, Bacalman S, et al. Autism spectrum disorders and attention-deficit/hyperactivity disorder in boys with the fragile X premutation. J Dev Behav Pediatr. (2006) 27:S137–S144. doi: 10.1097/00004703-200604002-00012
44. Piven J, Palmer P, Landa R, Santangelo SJD, Childress D. Personality and language characteristics in parents from multiple-incidence autism families. Am J Med Genet Pt B. (1997) 74:398–411. doi: 10.1002/(SICI)1096-8628(19970725)74:4<398::AID-AJMG11>3.0.CO;2-D
45. Murphy M, Bolton P, Pickles A, Fombonne E, Piven J, Rutter M. Personality traits of the relatives of autistic probands. Psychol Med. (2000) 30:1411–24. doi: 10.1017/S0033291799002949
46. Losh M, Childress D, Lam K, Piven J. Defining key features of the broad autism phenotype: a comparison across parents of multiple- and single-incidence autism families. Am J Med Genet B Neuropsychiatr Genet. (2008) 147B:424–33. doi: 10.1002/ajmg.b.30612
47. Nayar K, Sealock JM, Maltman N, Bush L, Cook EH, Davis LK, et al. Elevated polygenic burden for autism spectrum disorder is associated with the broad autism phenotype in mothers of individuals with autism spectrum disorder. Biol Psychiatry. (2021) 89:476–85. doi: 10.1016/j.biopsych.2020.08.029
48. Tyrer P (editor). Personality assessment schedule. In: Personality Disorders: Diagnosis, Management, and Course. London: Butterworth and Company (1988). p. 43–62.
49. Landa R, Piven J, Wzorek MM, Gayle JO, Chase GA, Folstein SE. Social language use in parents of autistic individuals. Psychol Med. (1992) 22:245–54. doi: 10.1017/S0033291700032918
50. Darnell JC, Van Driesche SJ, Zhang C, Hung KY, Mele A, Fraser CE, et al. FMRP stalls ribosomal translocation on mRNAs linked to synaptic function and autism. Cell. (2011) 146:247–61. doi: 10.1016/j.cell.2011.06.013
51. Iossifov I, Ronemus M, Levy D, Wang Z, Hakker I, Rosenbaum J, et al. De novo gene disruptions in children on the autistic spectrum. Neuron. (2012) 74:285–99. doi: 10.1016/j.neuron.2012.04.009
52. Gossett A, Sansone S, Schneider A, Johnston C, Hagerman R, Tassone F, et al. Psychiatric disorders among women with the fragile X premutation without children affected by fragile X syndrome. Am J Med Genet B Neuropsychiatr Genet. (2016) 171:1139–47. doi: 10.1002/ajmg.b.32496
53. Jiraanont P, Sweha SR, Alolaby RR, Silva M, Tang HT, Durbin-Johnson B, et al. Clinical and molecular correlates in fragile X premutation females. eNeurologicalSci. (2017) 7:49–56. doi: 10.1016/j.ensci.2017.04.003
54. Kraan CM, Hocking DR, Georgiou-Karistianis N, Metcalfe SA, Archibald AD, Fielding J, et al. Impaired response inhibition is associated with self-reported symptoms of depression, anxiety, and ADHD in female FMR1 premutation carriers. Am J Med Genet B Neuropsychiatr Genet. (2014) 165B:41–51. doi: 10.1002/ajmg.b.32203
55. Allen EG, Charen K, Hipp HS, Shubeck L, Amin A, He W, et al. Clustering of comorbid conditions among women who carry an FMR1 premutation. Genet Med. (2020) 22:758–66. doi: 10.1038/s41436-019-0733-5
56. Schneider A, Johnston C, Tassone F, Sansone S, Hagerman RJ, Ferrer E, et al. Broad autism spectrum and obsessive-compulsive symptoms in adults with the fragile X premutation. Clin Neuropsychol. (2016) 30:929–43. doi: 10.1080/13854046.2016.1189536
57. Klusek J, Schmidt J, Fairchild AJ, Porter A, Roberts JE. Altered sensitivity to social gaze in the FMR1 premutation and pragmatic language competence. J Neurodev Disord. (2017) 9:31. doi: 10.1186/s11689-017-9211-z
58. Capps L, Kehres J, Sigman M. Conversational abilities among children with autism and children with developmental delays. Autism. (1998) 2:325–44. doi: 10.1177/1362361398024002
59. Losh M, Capps L. Narrative ability in high-functioning children with autism or Asperger's syndrome. J Autism Dev Disord. (2003) 33:239–51. doi: 10.1023/A:1024446215446
60. Losh M, Piven J. Social-cognition and the broad autism phenotype: identifying genetically meaningful phenotypes. J Child Psychol Psychiatry. (2007) 48:105–12. doi: 10.1111/j.1469-7610.2006.01594.x
61. Losh M, Adolphs R, Poe MD, Couture S, Penn D, Baranek GT, et al. Neuropsychological profile of autism and the broad autism phenotype. Arch Gen Psychiatry. (2009) 66:518–26. doi: 10.1001/archgenpsychiatry.2009.34
62. Losh M, Martin GE, Klusek J, Hogan-Brown AL, Sideris J. Social communication and theory of mind in boys with autism and fragile x syndrome. Front Psychol. (2012) 3:266. doi: 10.3389/fpsyg.2012.00266
63. Winston M, Nayar K, Hogan AL, Barstein J, La Valle C, Sharp K, et al. Physiological regulation and social-emotional processing in female carriers of the FMR1 premutation. Physiol Behav. (2020) 214:112746. doi: 10.1016/j.physbeh.2019.112746
64. Winston M, Nayar K, Landau E, Maltman N, Sideris J, Zhou L, et al. A unique visual attention profile associated with the FMR1 premutation. Front Genet. (2021) 12:591211. doi: 10.3389/fgene.2021.591211
65. Grigsby J, Brega AG, Engle K, Leehey MA, Hagerman RJ, Tassone F, et al. Cognitive profile of fragile X premutation carriers with and without fragile X-associated tremor/ataxia syndrome. Neuropsychology. (2008) 22:48–60. doi: 10.1037/0894-4105.22.1.48
66. Grigsby J, Cornish K, Hocking D, Kraan C, Olichney JM, Rivera SM, et al. The cognitive neuropsychological phenotype of carriers of the FMR1 premutation. J Neurodev Disord. (2014) 6:28. doi: 10.1186/1866-1955-6-28
67. Baker EK, Arpone M, Aliaga SM, Bretherton L, Kraan CM, Bui M, et al. Incomplete silencing of full mutation alleles in males with fragile X syndrome is associated with autistic features. Mol Autism. (2019) 10:21. doi: 10.1186/s13229-019-0271-7
68. Shelton AL, Cornish KM, Godler DE, Clough M, Kraan C, Bui M, et al. Delineation of the working memory profile in female FMR1 premutation carriers: the effect of cognitive load on ocular motor responses. Behav Brain Res. (2015) 282:194–200. doi: 10.1016/j.bbr.2015.01.011
69. Shelton AL, Cornish K, Fielding J. Long term verbal memory recall deficits in fragile X premutation females. Neurobiol Learn Mem. (2017) 144:131–5. doi: 10.1016/j.nlm.2017.07.002
70. Shelton AL, Cornish K, Kraan C, Georgiou-Karistianis N, Metcalfe SA, Bradshaw JL, et al. Exploring inhibitory deficits in female premutation carriers of fragile X syndrome: through eye movements. Brain Cogn. (2014) 85:201–8. doi: 10.1016/j.bandc.2013.12.006
71. Goodrich-Hunsaker NJ, Wong LM, Mclennan Y, Tassone F, Harvey D, Rivera SM, et al. Adult female Fragile X Premutation carriers exhibit age- and CGG repeat length-related impairments on an attentionally based enumeration task. Front Hum Neurosci. (2011) 5:63. doi: 10.3389/fnhum.2011.00063
72. Jacquemont S, Hagerman RJ, Leehey M, Grigsby J, Zhang L, Brunberg JA, et al. Fragile X premutation tremor/ataxia syndrome: molecular, clinical, and neuroimaging correlates. Am J Hum Genet. (2003) 72:869–78. doi: 10.1086/374321
73. Moore CJ, Daly EM, Schmitz N, Tassone F, Tysoe C, Hagerman RJ, et al. A neuropsychological investigation of male premutation carriers of fragile X syndrome. Neuropsychologia. (2004) 42:1934–47. doi: 10.1016/j.neuropsychologia.2004.05.002
74. Sterling A, Mailick M, Greenberg J, Warren SF, Brady N. Language dysfluencies in females with the FMR1 premutation. Brain Cogn. (2013) 82:84–9. doi: 10.1016/j.bandc.2013.02.009
75. Johnston C, Eliez S, Dyer-Friedman J, Hessl D, Glaser B, Blasey C, et al. Neurobehavioral phenotype in carriers of the fragile X premutation. Am J Med Genet. (2001) 103:314–9. doi: 10.1002/ajmg.1561
76. Goodlin-Jones BL, Tassone F, Gane LW, Hagerman RJ. Autistic spectrum disorder and the fragile X premutation. J Dev Behav Pediatr. (2004) 25:392–8. doi: 10.1097/00004703-200412000-00002
77. Loesch DZ, Bui MQ, Hammersley E, Schneider A, Storey E, Stimpson P, et al. Psychological status in female carriers of premutation FMR1 allele showing a complex relationship with the size of CGG expansion. Clin Genet. (2015) 87:173–8. doi: 10.1111/cge.12347
78. Leehey MA, Berry-Kravis E, Goetz CG, Zhang L, Hall DA, Li L, et al. FMR1 CGG repeat length predicts motor dysfunction in premutation carriers. Neurology. (2008) 70:1397–402. doi: 10.1212/01.wnl.0000281692.98200.f5
79. Hall DA, Stebbins GT, Jacquemont S, Berry-Kravis E, Goetz CG, Hagerman R, et al. Clinimetric properties of the fragile X-associated Tremor Ataxia Syndrome Rating Scale. Mov Disord Clin Pract. (2019) 6:120–4. doi: 10.1002/mdc3.12708
80. Sasson NJ, Lam KS, Childress D, Parlier M, Daniels JL, Piven J. The broad autism phenotype questionnaire: prevalence and diagnostic classification. Autism Res. (2013) 6:134–43. doi: 10.1002/aur.1272
81. Beck AT, Steer RA, Brown GK. Manual for the Beck Depression Inventory-II. San Antonio, TX: Psychological Corporation (1996).
82. Spielberger CD, Gorsuch RL, Lushene R, Vagg PR, Jacobs GA. Manual for the State-Trait Anxiety Inventory. Palo Alto, CA: Consulting Psychologists Press (1983).
83. Knight RG, Waal-Manning HJ, Spears GF. Some norms and reliability data for the State–Trait Anxiety Inventory and the Zung Self-Rating Depression scale. Br J Clin Psychol. (1983) 22 (Pt 4):245–9. doi: 10.1111/j.2044-8260.1983.tb00610.x
84. Julian LJ. Measures of anxiety: State-Trait Anxiety Inventory (STAI), Beck Anxiety Inventory (BAI), and Hospital Anxiety and Depression Scale-Anxiety (HADS-A). Arthritis Care Res (Hoboken). (2011) 63(Suppl. 11):S467–S72. doi: 10.1002/acr.20561
85. Sheehan DV, Lecrubier Y, Sheehan KH, Amorim P, Janavs J, Weiller E, et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry. (1998) 59(Suppl. 20):22–33; quiz 34–57.
86. Baron-Cohen S, Wheelwright S, Hill J, Raste Y, Plumb I. The “reading the mind in the eyes” test revised version: a study with normal adults, and adults with Asperger syndrome or high-functioning autism. J Child Psychol Psychiatry Allied Discipl. (2001) 42:241–51. doi: 10.1111/1469-7610.00715
87. Adolphs R, Tranel D, Damasio AR. The human amygdala in social judgment. Nature. (1998) 393:470–4. doi: 10.1038/30982
88. Adolphs R, Sears L, Piven J. Abnormal processing of social information from faces in autism. J Cogn Neurosci. (2001) 13:232–40. doi: 10.1162/089892901564289
89. Adolphs R, Tranel D. Amygdala damage impairs emotion recognition from scenes only when they contain facial expressions Neuropsychologia. (2003) 41:1281–9. doi: 10.1016/S0028-3932(03)00064-2
90. Heberlein A, Ravahi S, Adolphs R, Tranel D. Deficits in attributing emotion to moving visual stimuli consequent to amygdala damage. Cogn Neurosci Soc Abstracts. (2000) 2000:56.
91. Roth RM, Isquith PK, Gioia GA. Behavior Rating Inventory of Executive Function-Adult Version. Lutz, FL: Psychological Assessment Resources (2005).
92. Lafauci G, Adayev T, Kascsak R, Nolin S, Mehta P, Brown WT, et al. Fragile X screening by quantification of FMRP in dried blood spots by a Luminex immunoassay. J Mol Diagn. (2013) 15:508–17. doi: 10.1016/j.jmoldx.2013.02.006
93. Berry-Kravis E, Lewin F, Wuu J, Leehey M, Hagerman R, Hagerman P, et al. Tremor and ataxia in fragile X premutation carriers: blinded videotape study. Ann Neurol. (2003) 53:616–23. doi: 10.1002/ana.10522
94. Cohen J. Statistical Power Analysis for the Behavioral Sciences. New York, NY: Routledge Academic (1988).
95. Spurk D, Hirschi A, Wang M, Valero D, Kauffeld S. Latent profile analysis: a review and “how to” guide of its application within vocational behavior research. J Vocat Behav. (2020) 120:103445. doi: 10.1016/j.jvb.2020.103445
96. Tein JY, Coxe S, Cham H. Statistical power to detect the correct number of classes in latent profile analysis. Struct Equ Model. (2013) 20:640–57. doi: 10.1080/10705511.2013.824781
97. Williams GA, Kibowski F. Latent class analysis and latent profile analysis. In: Jason LA, Glenwick DS, editors. Handbook of Methodological Approaches to Community-Based Research: Qualitative, Quantitative, and Mixed Methods. New York, NY: Oxford University Press (2016). p. 143–151.
98. Ferguson SL, Whitney E, Moore G, Hull D. Finding latent groups in observed data: a primer on latent profile analysis in Mplus for applied researchers. Int J Behav Dev. (2019) 44:458–68. doi: 10.1177/0165025419881721
99. Lord C, Rutter M, Dilavore PC, Risi S, Gotham K, Bishop S. Autism Diagnostic Observation Schedule, 2nd Edn. Torrance, CA: Western Psychological Services (2012).
100. Klusek J, Mcgrath SE, Abbeduto L, Roberts JE. Pragmatic language features of mothers with the FMR1 premutation are associated with the language outcomes of adolescents and young adults with fragile X syndrome. J Speech Lang Hear Res. (2016) 59:49–61. doi: 10.1044/2015_JSLHR-L-15-0102
101. Klusek J, Fairchild AJ, Roberts JE. Vagal tone as a putative mechanism for pragmatic competence: an investigation of carriers of the FMR1 premutation. J Autism Dev Disord. (2019) 49:197–208. doi: 10.1007/s10803-018-3714-7
102. Klusek J, Thurman AJ, Abbeduto L. Maternal pragmatic language difficulties in the FMR1 premutation and the broad autism phenotype: associations with individual and family outcomes. J Autism Dev Disord. (2021) 59:49–61. doi: 10.1007/s10803-021-04980-3
103. Abbeduto L, Brady N, Kover ST. Language development and fragile X syndrome: profiles, syndrome-specificity, and within-syndrome differences. Ment Retard Dev Disabil Res Rev. (2007) 13:36–46. doi: 10.1002/mrdd.20142
104. Kover ST, Abbeduto L. Expressive language in male adolescents with fragile X syndrome with and without comorbid autism. J Intellect Disabil Res. (2010) 54:246–65. doi: 10.1111/j.1365-2788.2010.01255.x
105. Hogan-Brown AL, Losh M, Martin GE, Mueffelmann DJ. An investigation of narrative ability in boys with autism and fragile X syndrome. Am J Intellect Dev Disabil. (2013) 118:77–94. doi: 10.1352/1944-7558-118.2.77
106. Martin GE, Losh M, Estigarribia B, Sideris J, Roberts J. Longitudinal profiles of expressive vocabulary, syntax and pragmatic language in boys with fragile X syndrome or Down syndrome. Int J Lang Commun Disord. (2013) 48:432–43. doi: 10.1111/1460-6984.12019
107. Klusek J, Martin GE, Losh M. A comparison of pragmatic language in boys with autism and fragile X syndrome. J Speech Lang Hear Res. (2014) 57:1692–707. doi: 10.1044/2014_JSLHR-L-13-0064
108. Martin GE, Barstein J, Hornickel J, Matherly S, Durante G, Losh M. Signaling of noncomprehension in communication breakdowns in fragile X syndrome, down syndrome, and autism spectrum disorder. J Commun Disord. (2017) 65:22–34. doi: 10.1016/j.jcomdis.2017.01.003
109. Martin GE, Bush L, Klusek J, Patel S, Losh M. A multimethod analysis of pragmatic skills in children and adolescents with fragile X syndrome, autism spectrum disorder, and down syndrome. J Speech Lang Hear Res. (2018) 61:3023–37. doi: 10.1044/2018_JSLHR-L-18-0008
110. Kessler RC, Berglund P, Demler O, Jin R, Koretz D, Merikangas KR, et al. The epidemiology of major depressive disorder: results from the National Comorbidity Survey Replication (NCS-R). JAMA. (2003) 289:3095–105. doi: 10.1001/jama.289.23.3095
111. Lewis P, Abbeduto L, Murphy M, Richmond E, Giles N, Bruno L, et al. Cognitive, language and social-cognitive skills of individuals with fragile X syndrome with and without autism. J Intellect Disabil Res. (2006) 50:532–45. doi: 10.1111/j.1365-2788.2006.00803.x
112. Adolphs R. Social cognition and the human brain. Trends Cogn Sci. (1999) 3:469–79. doi: 10.1016/s1364-6613(99)01399-6
113. Wheeler AC, Bailey DB, Berry-Kravis E, Greenberg J, Losh M, Mailick M, et al. Associated features in females with an FMR1 premutation. J Neurodev Disord. (2014) 6:30. doi: 10.1186/1866-1955-6-30
114. Brega AG, Goodrich G, Bennett RE, Hessl D, Engle K, Leehey MA, et al. The primary cognitive deficit among males with fragile X-associated tremor/ataxia syndrome (FXTAS) is a dysexecutive syndrome. J Clin Exp Neuropsychol. (2008) 30:853–69. doi: 10.1080/13803390701819044
115. Shelton AL, Cornish KM, Kraan CM, Lozano R, Bui M, Fielding J. Executive dysfunction in female FMR1 premutation carriers. Cerebellum. (2016) 15:565–9. doi: 10.1007/s12311-016-0782-0
116. Shelton AL, Cornish K, Clough M, Gajamange S, Kolbe S, Fielding J. Disassociation between brain activation and executive function in fragile X premutation females. Hum Brain Mapp. (2017) 38:1056–67. doi: 10.1002/hbm.23438
117. Movaghar A, Mailick M, Sterling A, Greenberg J, Saha K. Automated screening for Fragile X premutation carriers based on linguistic and cognitive computational phenotypes. Sci Rep. (2017) 7:2674. doi: 10.1038/s41598-017-02682-4
118. Birch RC, Hocking DR, Trollor JN. Prevalence and predictors of subjective memory complaints in adult male carriers of the FMR1 premutation. Clin Neuropsychol. (2016) 30:834–48. doi: 10.1080/13854046.2016.1145905
119. Hall D, Todorova-Koteva K, Pandya S, Bernard B, Ouyang B, Walsh M, et al. Neurological and endocrine phenotypes of fragile X carrier women. Clin Genet. (2016) 89:60–7. doi: 10.1111/cge.12646
120. Steger MF, Kashdan TB. Depression and everyday social activity, belonging, and well-being. J Couns Psychol. (2009) 56:289–300. doi: 10.1037/a0015416
121. Wallace GL, Budgett J, Charlton RA. Aging and autism spectrum disorder: evidence from the broad autism phenotype. Autism Res. (2016) 9:1294–303. doi: 10.1002/aur.1620
122. Mesibov GB. Treatment issues with high-functioning adolescents and adults with autism. In: Schopler E, Mesibov GB, editors. High Functioning Individuals With Autism. Current Issues in Autism. Boston, MA: Springer (1992). p. 143–55.
123. Patel SP, Kim JH, Larson CR, Losh M. Mechanisms of voice control related to prosody in autism spectrum disorder and first-degree relatives. Autism Res. (2019) 12:1192–210. doi: 10.1002/aur.2156
124. Patel SP, Nayar K, Martin GE, Franich K, Crawford S, Diehl JJ, et al. An acoustic characterization of prosodic differences in autism spectrum disorder and first-degree relatives. J Autism Dev Disord. (2020) 50:3032–45. doi: 10.1007/s10803-020-04392-9
125. Klusek J, Lafauci G, Adayev T, Brown WT, Tassone F, Roberts JE. Reduced vagal tone in women with the FMR1 premutation is associated with FMR1 mRNA but not depression or anxiety. J Neurodev Disord. (2017) 9:16. doi: 10.1186/s11689-017-9197-6
126. Mailick MR, Movaghar A, Hong J, Greenberg JS, Dawalt LS, Zhou L, et al. Health profiles of mosaic versus non-mosaic FMR1 premutation carrier mothers of children with fragile X syndrome. Front Genet. (2018) 9:173. doi: 10.3389/fgene.2018.00173
127. Bailey DB, Sideris J, Roberts J, Hatton D. Child and genetic variables associated with maternal adaptation to fragile X syndrome: a multidimensional analysis. Am J Med Genet A. (2008) 146A:720–9. doi: 10.1002/ajmg.a.32240
128. Seltzer MM, Barker ET, Greenberg JS, Hong J, Coe C, Almeida D. Differential sensitivity to life stress in FMR1 premutation carrier mothers of children with fragile X syndrome. Health Psychol. (2012) 31:612–22. doi: 10.1037/a0026528
129. Wheeler A, Raspa M, Hagerman R, Mailick M, Riley C. Implications of the FMR1 premutation for children, adolescents, adults, and their families. Pediatrics. (2017) 139:S172–S82. doi: 10.1542/peds.2016-1159D
130. Mailick MR, Hong J, Rathouz P, Baker MW, Greenberg JS, Smith L, et al. Low-normal FMR1 CGG repeat length: phenotypic associations. Front Genet. (2014) 5:309. doi: 10.3389/fgene.2014.00309
131. Adamsheck HC, Petty EM, Hong J, Baker MW, Brilliant MH, Mailick MR. Is low FMR1 CGG repeat length in males correlated with family history of BRCA-associated cancers? An exploratory analysis of medical records. J Genet Couns. (2017) 26:1401–10. doi: 10.1007/s10897-017-0116-5
Keywords: FMR1 premutation, broad autism phenotype, latent profile analysis, executive function, social cognition
Citation: Maltman N, Guilfoyle J, Nayar K, Martin GE, Winston M, Lau JCY, Bush L, Patel S, Lee M, Sideris J, Hall DA, Zhou L, Sharp K, Berry-Kravis E and Losh M (2021) The Phenotypic Profile Associated With the FMR1 Premutation in Women: An Investigation of Clinical-Behavioral, Social-Cognitive, and Executive Abilities. Front. Psychiatry 12:718485. doi: 10.3389/fpsyt.2021.718485
Received: 31 May 2021; Accepted: 12 July 2021;
Published: 06 August 2021.
Edited by:
Randi Jenssen Hagerman, UC Davis, United StatesReviewed by:
Tri Indah Winarni, Diponegoro University, IndonesiaCopyright © 2021 Maltman, Guilfoyle, Nayar, Martin, Winston, Lau, Bush, Patel, Lee, Sideris, Hall, Zhou, Sharp, Berry-Kravis and Losh. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Molly Losh, bS1sb3NoQG5vcnRod2VzdGVybi5lZHU=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.