- 1RIKEN Center for Sustainable Resource Science, Yokohama, Japan
- 2Graduate School of Bioagricultural Sciences, Nagoya University, Nagoya, Japan
- 3Department of Chemical and Biological Sciences, Faculty of Science, Japan Women’s University, Tokyo, Japan
- 4Department of International Environmental and Agricultural Science, Tokyo University of Agriculture and Technology, Tokyo, Japan
- 5NARO, Institute of Vegetable and Floriculture Science, Tsu, Japan
- 6Department of Biochemistry, Okayama University of Science, Okayama, Japan
Temperature is a critical environmental factor governing plant growth and development. The difference between day temperature (DT) and night temperature (NT), abbreviated as DIF, influences plant architecture. Subjecting plants to artificial DIF treatments is an effective strategy in ornamental horticulture. For example, negative DIF (when DT – NT < 0) generally inhibits stem elongation, resulting in dwarf plants. However, the mechanisms underlying stem growth regulation by DIF remains to be completely elucidated. In this study, we aimed to analyze the growth, transcriptome, and phytohormone profiles of tomato (Solanum lycopersicum) seedlings grown under different DIF treatments. Under positive DIF (when DT – NT > 0), in contrast to the control temperature (25°C/20°C, DT/NT), high temperature (30°C/25°C) increased stem length and thickness, as well as the number of xylem vessels. Conversely, compared with the positive high temperature DIF treatment (30°C/25°C), under negative DIF treatment (25°C/30°C) stem elongation was inhibited, but stem thickness and the number of xylem vessels were not affected. The negative DIF treatment decreased the expression of gibberellin (GA)-, auxin-, and cell wall-related genes in the epicotyl, as well as the concentrations of GAs and indole-3-acetic acid (IAA). The expression of these genes and concentrations of these hormones increased under high temperature compared to those under the control temperature positive DIF. Our results suggest that stem length in tomato seedlings is controlled by changes in GA and IAA biosynthesis in response to varying day and night temperatures.
Introduction
Plants modulate their growth according to their environmental conditions. In particular, temperature is a critical factor affecting plant growth. Each plant species has a suitable temperature range. Within this range, higher temperatures generally promote shoot growth, including leaf expansion and stem elongation and thickening. However, temperatures above the optimal range suppress growth. In addition to the absolute temperature, the difference in temperature between day and night can affect growth. Normally, night temperatures are lower than daytime temperatures, and plants modulate their growth pattern and metabolism in response to this temperature change. Altering the temperature difference between day and night is one method through which plant growth is controlled in ornamental horticulture. The difference between day temperature (DT) and night temperature (NT), abbreviated as DIF, is defined as DT– NT. Positive DIF occurs when DT is higher than NT, zero DIF occurs when DT is the same as NT, and negative DIF occurs when DT is lower than NT. Negative DIF can be used to control plant height as an alternative to agricultural chemicals (Shimizu, 2007). While the effects were plant species-dependent, it has been reported that negative DIF inhibits stem elongation compared to positive DIF in various plants, including Lilium longiflorum, Dendranthema grandiflora, Cucumis sativus (Shimizu, 2007), and Solanum lycopersicum (Went, 1944; de Koning, 1988).
Previous studies have suggested that phytohormones are involved in regulating plant growth in response to temperature. Gibberellin (GA) is known to play a key role in stem elongation (Davière and Achard, 2013; Binenbaum et al., 2018; Ferrero et al., 2019). Indole-3-acetic acid (IAA), an auxin, also plays an important role in cell elongation in the hypocotyl, the epicotyl, and other organs (Leyser, 2018; Zhao, 2018). In Arabidopsis thaliana, higher temperatures promote hypocotyl elongation mediated by phytochrome-interacting factor 4 (PIF4)-dependent auxin biosynthesis (Franklin et al., 2011; Nomoto et al., 2012; Sun et al., 2012). It has been demonstrated that PIF4 function is regulated by GA via DELLA proteins, which are key negative regulators of GA signaling (Koini et al., 2009; Stavang et al., 2009).
Studies have found that stem elongation under different DIF treatments is accompanied by a change in GA content in Campanula isophylla and Pisum sativum (Jensen et al., 1996; Grindal et al., 1998; Stavang et al., 2005). In P. sativum, inhibition of stem elongation under negative DIF was weaker in GA-related mutants than that in the wild type (Grindal et al., 1998). In A. thaliana, non-bioactive GA29 content was lower under a negative DIF treatment than that under a positive DIF treatment, while IAA concentration was higher under a positive DIF treatment than that under a negative DIF treatment (Thingnaes et al., 2003). In Raphaus sativus L., differences in stem elongation under DIF treatments followed a similar pattern to changes in IAA content (Hayata et al., 2001). These studies suggest the involvement of these hormones in the effect of DIF on stem elongation. However, these hormones and the expression of their genes have not been investigated in detail. Further, temperature not only affects stem elongation but also stem thickness; however, the effect of DIF on vascular development has not been properly characterized to date.
In this study, we aimed to elucidate the mechanisms underlying stem growth regulation by DIF. To this end, we examined the effects of different DIF treatments on tomato (S. lycopersicum), which is one of the most important vegetable crops. Our analyses of growth, transcriptomes, and hormones strongly suggest that negative DIF-dependent inhibition of stem elongation is mediated by the repression of GA and IAA synthesis accompanied by the regulation of cell wall-related genes. We also report that negative DIF treatment has a minimal effect on stem thickening in tomato seedling.
Results
Higher Temperatures Under Positive DIF Promote Stem Elongation and Thickening in Tomato Seedlings
To examine the effect of temperature under positive DIF on plant growth, tomato seedlings (Managua RZ) were grown under control (25°C/20°C, CT) and high temperatures (30°C/25°C, HT) (Supplementary Figures 1A,B). The stem, hypocotyl, and epicotyl lengths and diameters were significantly greater under HT than those under CT (Figures 1A–C). Histological analysis of hypocotyl cross sections showed that the number of xylem vessels (diameter > 50 μm) were significantly greater under HT than that under CT (Figures 1D,E), indicating that higher temperatures under positive DIF promote stem elongation, stem thickening, and vascular development in tomato seedlings.
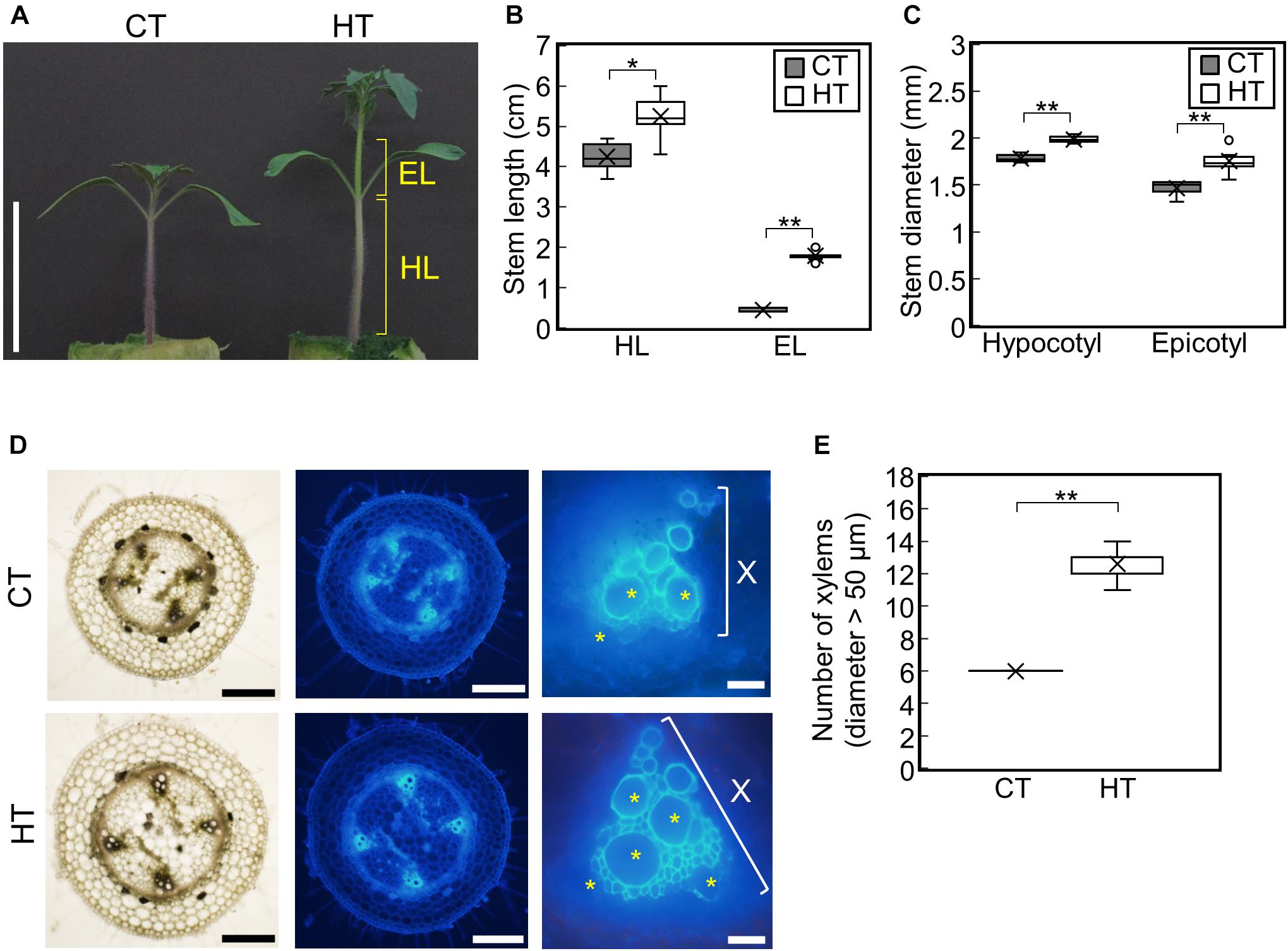
Figure 1. Effect of temperature on growth of tomato seedlings. Young tomato seedlings were grown for 7 days under 25°C/20°C (control temperature) and 30°C/25°C (high temperature). (A), Growth of seedlings. Tomato seedlings were photographed at 7 days. Scale bar = 5 cm. (B), Hypocotyl and epicotyl length. (C), Hypocotyl and epicotyl diameter. Data are shown as boxplots (n = 7). (D), Cross sections of hypocotyls. Asterisks indicate xylems (diameter > 50 μm). Scale bars = 500 μm (left and middle panels) and 50 μm (right panels). (E), Number of xylems (diameter > 50 μm) in the cross section. Data are shown as boxplots (n = 5). Cross marks in boxplot indicate the mean values. *P < 0.01; **P < 0.001 (Student’s t test). CT, control temperature; HT, high temperature; HL, hypocotyl length; EL, epicotyl length; X, xylem area.
Negative DIF Inhibits Stem Elongation, but Maintains Promotion of Stem Thickening
To examine the effect of negative DIF on tomato seedling growth, tomato seedlings were grown under HT (30°C/25°C, positive DIF) or with night and day temperatures reversed (25°C/30°C, negative DIF) (Supplementary Figures 1B,C). Under the negative DIF treatment, temperature-dependent stem elongation was inhibited both in the hypocotyl and epicotyl (Figures 2A,B). The difference in hypocotyl and epicotyl elongation between DIF treatments could be detected from 3 d after the onset of the treatments, and it became significant at 5 d (Supplementary Figures 2A–C). Conversely, hypocotyl and epicotyl thickness were similar under both DIF treatments (Figure 2C and Supplementary Figures 2D,E). The number of xylem vessels (diameter > 50 μm) was also similar (Figures 2D,E), indicating that the negative DIF treatment inhibited stem elongation without any negative effect on stem thickening. The size of cotyledons and true leaves was also similar under both treatments (Supplementary Figure 2F).
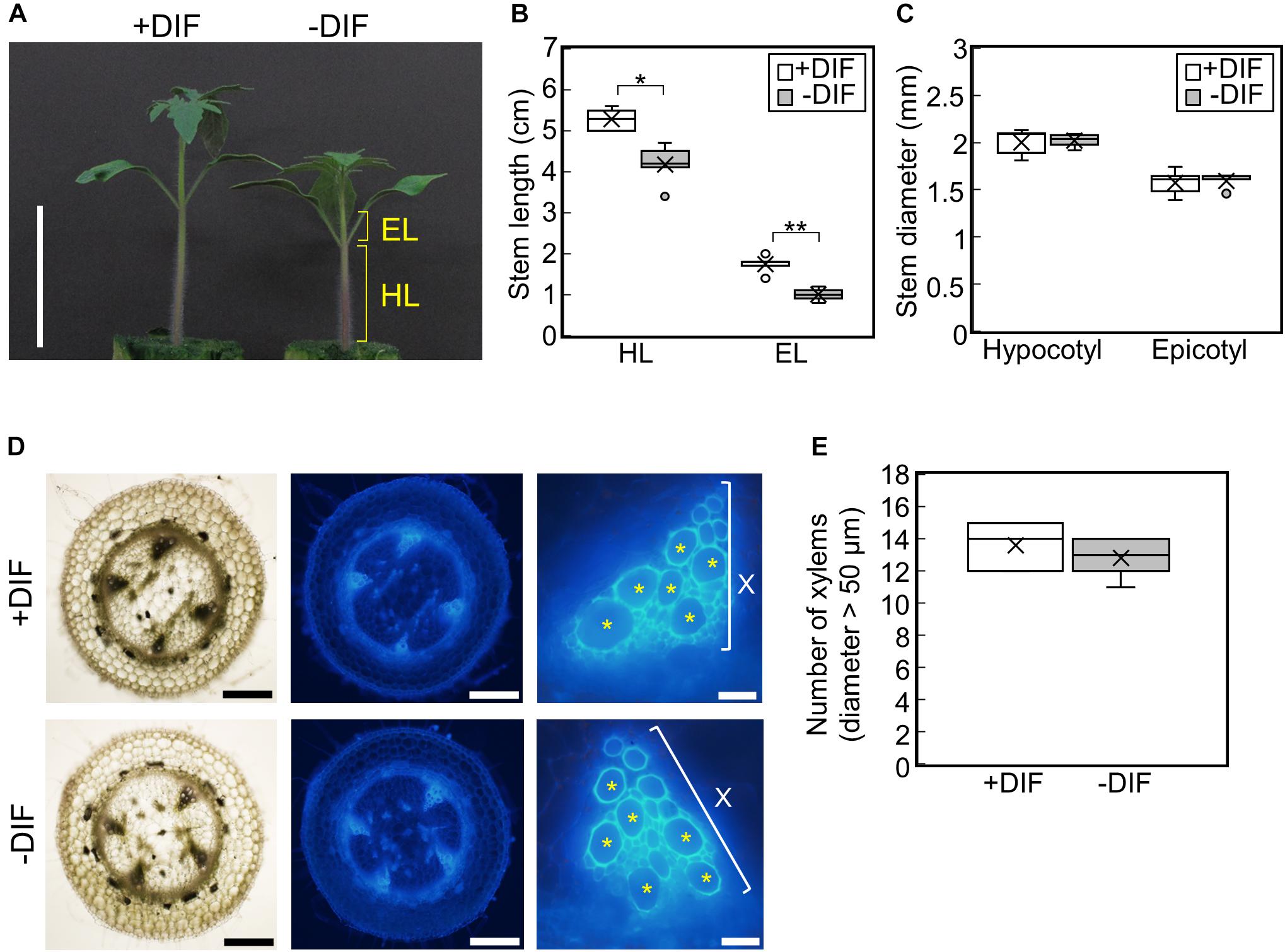
Figure 2. Effect of DIF treatment on growth of tomato seedlings. Young tomato seedlings were grown for 7 days under 30°C/25°C (positive DIF) and 25°C/30°C (negative DIF). (A), Growth of seedlings. Seedlings of tomato were photographed at 7 days. Scale bar = 5 cm. (B), Hypocotyl and epicotyl length. (C), Hypocotyl and epicotyl diameter. Data are shown as boxplots (n = 5). (D), Cross sections of hypocotyls. Asterisks indicate xylems (diameter > 50 μm). Scale bars = 500 μm (left and middle panels) and 50 μm (right panels). (E), Number of xylems (diameter > 50 μm) in the cross section. Data are shown as boxplots (n = 5). Cross marks in boxplot indicate the mean values. *P < 0.01; **P < 0.001 (Student’s t test). +DIF, positive DIF; -DIF, negative DIF; HL, hypocotyl length; EL, epicotyl length; X, xylem area.
To determine whether these responses were cultivar-specific, we tested the effect of the same DIF treatments on four other tomato cultivars. Similar to Managua RZ, the negative DIF treatment decreased stem length in all four tomato cultivars, but it did not affect stem thickness (Supplementary Figure 3). This suggests that the growth responses observed are a common feature among tomato seedlings.
Negative DIF Affects Gene Expression
To examine the mechanisms underlying stem growth regulation under the DIF treatments, we analyzed epicotyl transcriptomes in seedlings grown under positive or negative DIF for 7 d (Supplementary Figures 1B,C) and explored differentially expressed genes (DEGs). Microarray analysis revealed more than 5000 DEGs, with some upregulated and downregulated by negative DIF treatment (Figure 3A). We performed gene ontology (GO) analysis using the top 300 upregulated and downregulated genes (P < 0.05) (Supplementary Tables 1, 2). Enriched GO terms in biological process were found in cell wall macromolecule catabolic/metabolic process and (programmed) cell death (Figure 3B). Since stem elongation is related to cell wall modification, we focused on cell wall-related genes for further analyses. In addition, hormone-related genes could be involved in temperature-dependent regulation of epicotyl elongation; consequently, GA and IAA-related genes were also included in further analyses. In total, seven DEGs associated with the cell wall, GA, and IAA, as well as a PIF4 homolog possibly related to temperature-dependent hypocotyl elongation, were chosen for further analyses (Figure 3C).
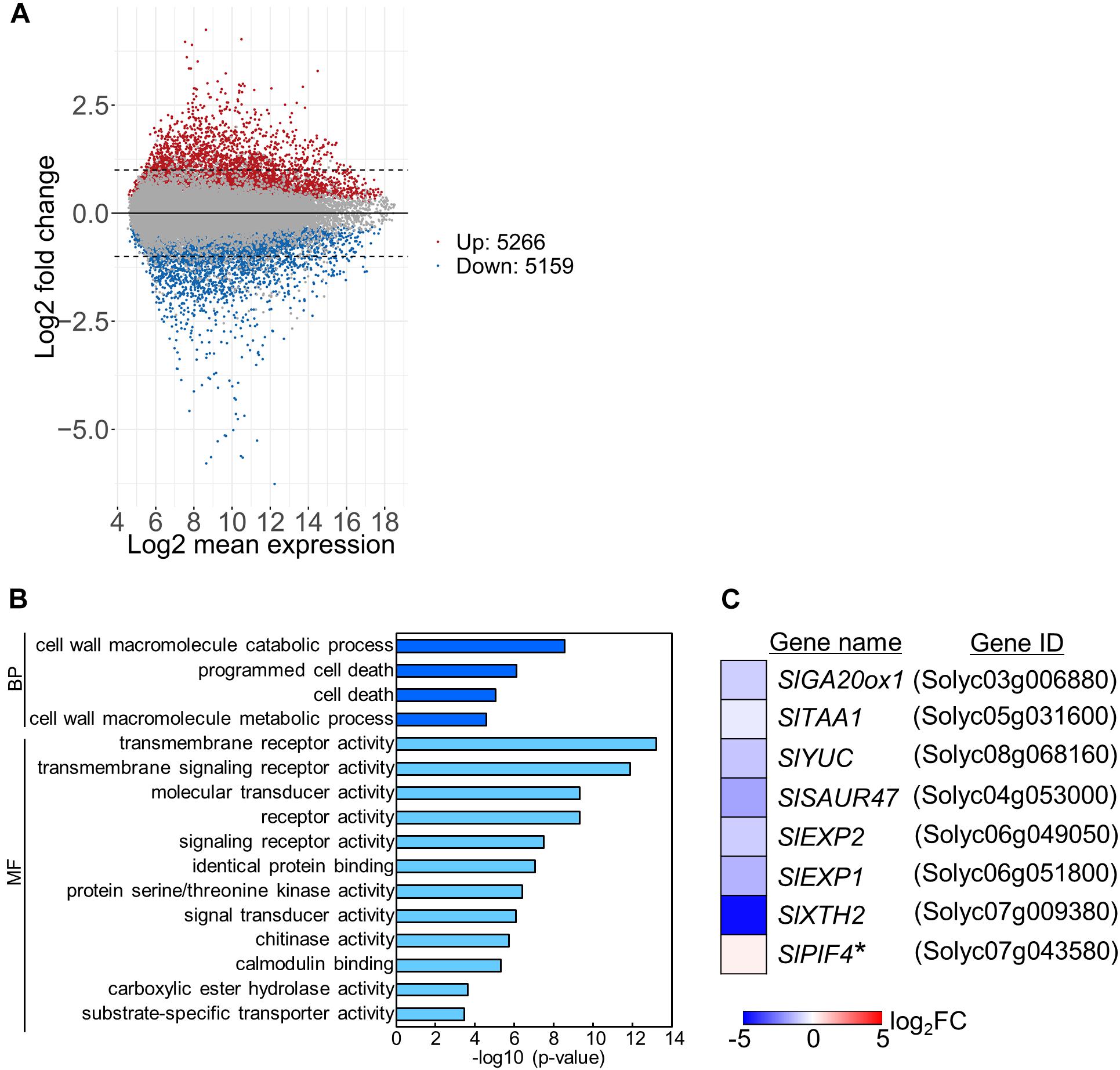
Figure 3. Transcriptome analysis of tomato under positive and negative DIF treatments. Total RNA was extracted from epicotyls of tomato seedlings grown for 7 days under positive DIF and negative DIF and subjected to microarray analysis with three biological replicates. (A), MA plots of microarray data in the positive DIF and negative DIF treatments. Red and blue dots represent genes upregulated (Up: 5266 genes) and downregulated (Down: 5159), respectively, under negative DIF in comparison with positive DIF. Gray dots represent genes that did not significantly differ between DIF treatments. (B), GO terms enriched among the DEGs (top 300 upregulated and top 300 downregulated genes; P < 0.05) identified. (C), List of 8 genes associated with stem elongation. The relative expression level of each gene under negative DIF compared with that under positive DIF is shown on the heatmap, which represents the log2 fold-change (FC). The color scale is shown at the bottom. +DIF, positive DIF; -DIF, negative DIF; BP, Biological Process; MF, Molecular Function.
GA and IAA Biosynthesis Genes Were Downregulated in Stems Under the Negative DIF Treatment
Among the identified DEGs, a key gene of de novo GA biosynthesis, namely GA20-oxidase (SlGA20ox1: Solyc03g006880), was downregulated (Figure 3C). Using reverse transcriptase real-time PCR, we confirmed that the expression of SlGA20ox1 was significantly downregulated in both hypocotyls and epicotyls under the negative DIF treatment (Figure 4A). The following genes involved in IAA biosynthesis were also among the downregulated DEGs: YUCCA (YUC), which encodes a flavin monooxygenase-like enzyme; TRYPTOPHAN AMINOTRANSFERASE OF ARABIDOPSIS (TAA); and IAA responsive factor SMALL AUXIN UP RNA (SAUR) (Figure 3C). Our reverse transcriptase real-time PCR analysis confirmed that SlTAA1 (Solyc05g031600) and SlYUC (Solyc08g068160) were downregulated in tomato epicotyl tissues under the negative DIF treatment (Figures 4B,C). SlSAUR47 (Solyc04g053000) was significantly downregulated in both hypocotyl and epicotyl tissues under the negative DIF treatment (Figure 4D).

Figure 4. Expression of GA-, IAA-, and cell wall-related genes in epicotyls and hypocotyls under negative and positive DIF treatments. The expression of SlGA20ox1 (A), IAA-related genes (SlTAA1, SlYUC, SlSAUR47) (B–D), cell wall-related genes (SlEXP2, SlEXP1, SlXTH2) (E–G), and SlPIF4 (H) in epicotyls and hypocotyls grown under positive and negative DIF treatments were analyzed using reverse transcriptase real-time PCR. Data are shown as boxplots (n = 3). Cross marks in boxplot indicate the mean values. *P < 0.1; **P < 0.05; ***P < 0.01 (Student’s t test). +DIF, positive DIF; -DIF, negative DIF.
Cell Wall Modification Genes Were Downregulated in Stems Under the Negative DIF Treatment
In our transcriptome analysis, expansins (SlEXP2; Solyc06g049050 and SlEXP1; Solyc06g051800) and Xyloglucan endotransglucosylase/hydrolase (SlXTH2; Solyc07g009380) were downregulated (Figure 3C). Our reverse transcriptase real-time PCR analysis revealed that SlEXP2 and SlXTH2 were significantly downregulated in both hypocotyls and epicotyls under the negative DIF treatment (Figures 4E,G). SlEXP1 was also downregulated in epicotyls under the negative DIF treatment (Figure 4F). These results are in line with the negative DIF treatment-driven repression of stem elongation.
We further examined the expression of PIF4, but the expression of SlPIF4 (Solyc07g043580), a homolog of A. thaliana PIF4, was not affected by DIF treatment (Figure 4H).
Higher Temperatures Under Positive DIF Upregulate Genes for GA and IAA Biosynthesis, as Well as Cell Wall Modification in Epicotyls
We next examined the expression of the DEGs under the CT (25°C/20°C) and HT (30°C/25°C) positive DIF treatments (Supplementary Figures 1A,B) using reverse transcriptase real-time PCR. SlGA20ox1, SlYUC, SlEXP2, and SlXTH2 were upregulated in epicotyls under the HT treatment (Figures 5A,C,E,G). SlSAUR47 and SlEXP1 were also upregulated in both hypocotyls and epicotyls under the HT treatment (Figures 5D,F). These results are consistent with the positive DIF treatment-driven promotion of stem elongation. In addition, SlPIF4 was upregulated in epicotyls under the HT treatment (Figure 5H).
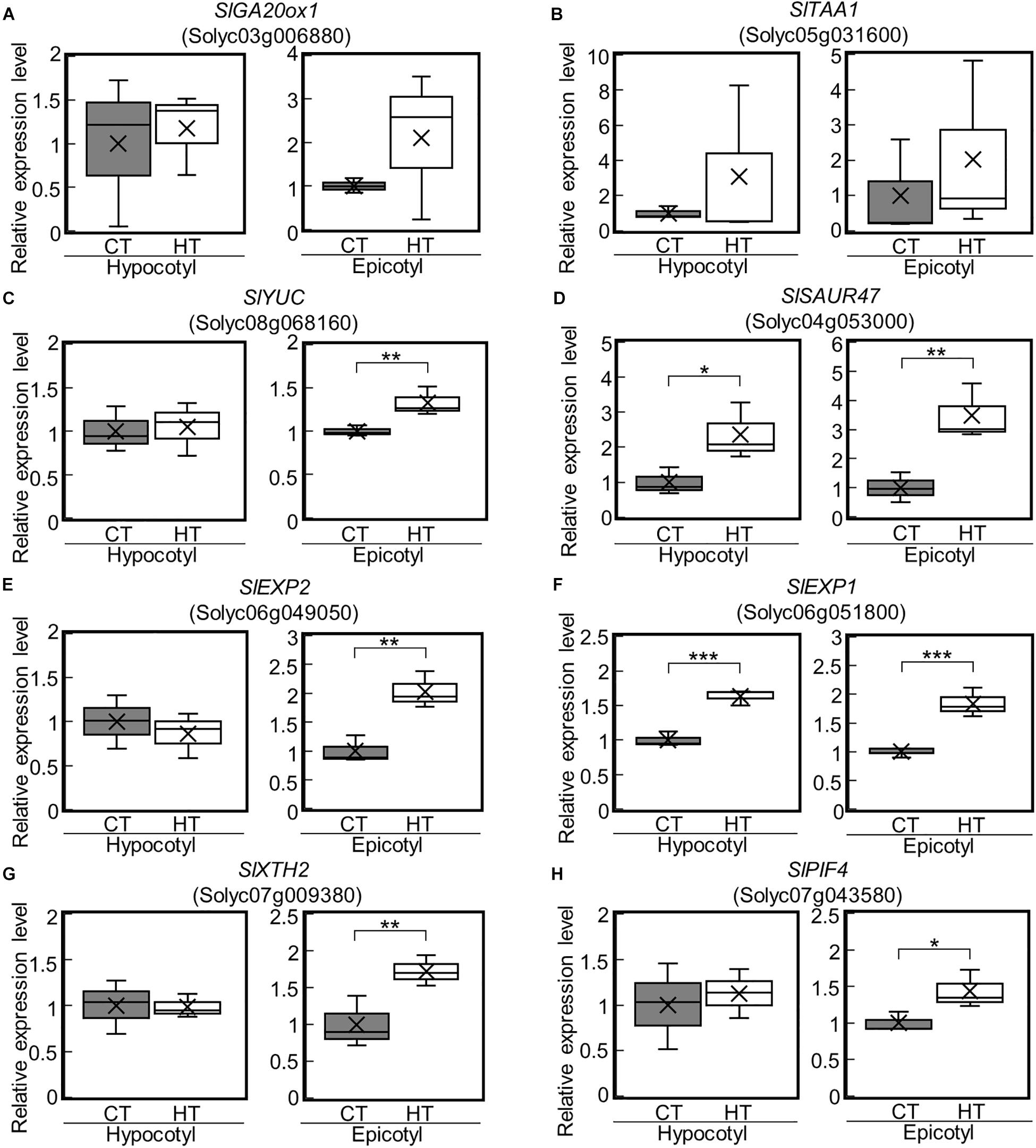
Figure 5. Expression of GA-, IAA-, and cell wall-related genes in epicotyls and hypocotyls under two different temperatures. The expression of SlGA20ox1 (A), IAA-related genes (SlTAA1, SlYUC, SlSAUR47) (B–D), cell wall-related genes (SlEXP2, SlEXP1, SlXTH2) (E–G), and SlPIF4 (H) in epicotyls and hypocotyls grown under control and high temperatures were analyzed by reverse transcriptase real-time PCR. Data are shown as boxplots (n = 3). Cross marks in boxplot indicate the mean values. *P < 0.1; **P < 0.05; ***P < 0.01 (Student’s t test). CT, control temperature; HT, high temperature.
Phytohormone Concentrations Are Consistent With Stem Elongation and Gene Expression Patterns in Response to DIF Treatments
We further investigated the effect of growth temperature on phytohormone concentrations in hypocotyls and epicotyls (Figure 6). Under positive and negative DIF treatments, GA1 and GA4, bioactive forms, could only be quantified in epicotyls under the positive DIF treatment, whereas they were below the quantification limit in all other tissues (Figures 6A,B). Another bioactive form, GA7, was also below the quantification limit (Figure 6C). The concentrations of the precursors, including GA9, GA19, GA20, GA24, and GA44, were lower in hypocotyls or epicotyls, while that of GA53 was higher, under the negative DIF treatment compared with those under the positive DIF treatment (Figures 6D,F–J).
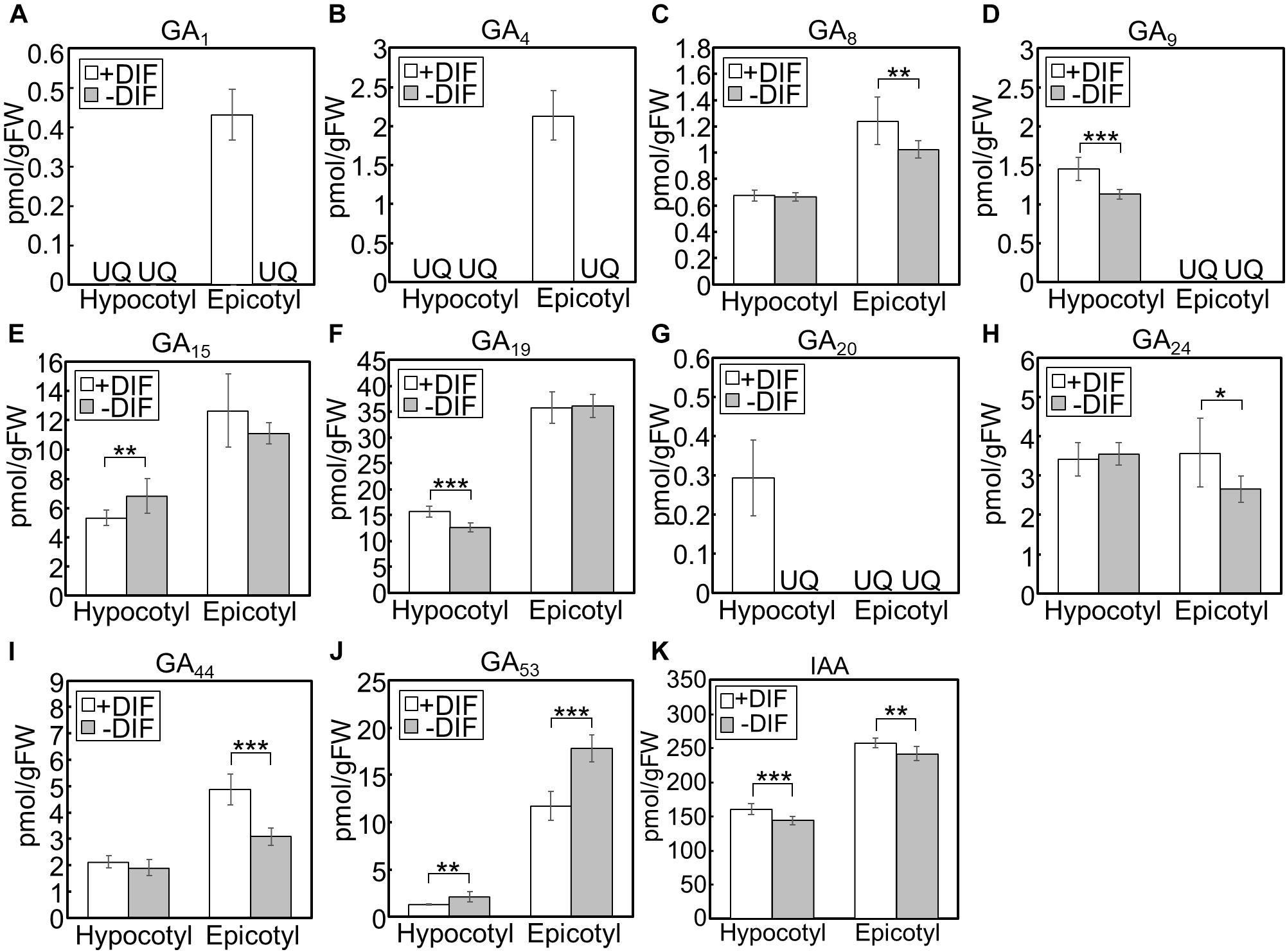
Figure 6. GA and IAA concentration in hypocotyls and epicotyls under negative and positive DIF treatments. Hypocotyls and epicotyls of tomato seedlings grown for 7 days under positive and negative DIF treatments were harvested and subjected to hormone analysis. The concentrations of GAs (A–J) and IAA (K) were quantified using liquid chromatography-tandem mass spectrometry. Data are represented as mean ± SD (n = 4 or 5). *P < 0.1; **P < 0.05; ***P < 0.01 (Student’s t test). GA7 and GA12 were below the quantification limit. +DIF, positive DIF; -DIF, negative DIF; FW, fresh weight; UQ, under quantification limit.
When we analyzed hormone species under the CT and HT positive DIF treatments (Figure 7), GA1 and GA4 were only detected in epicotyls under the HT treatment (Figures 7A,B). Concentrations of the inactive precursors GA15, GA24, and GA44 in epicotyls were higher under the HT treatment than those under the CT treatment (Figures 7E,H,I). Conversely, concentrations of GA53 were lower under the HT treatment those under the CT treatment (Figure 7J). These patterns were opposite to the changes that occurred following negative DIF treatment (Figure 6).
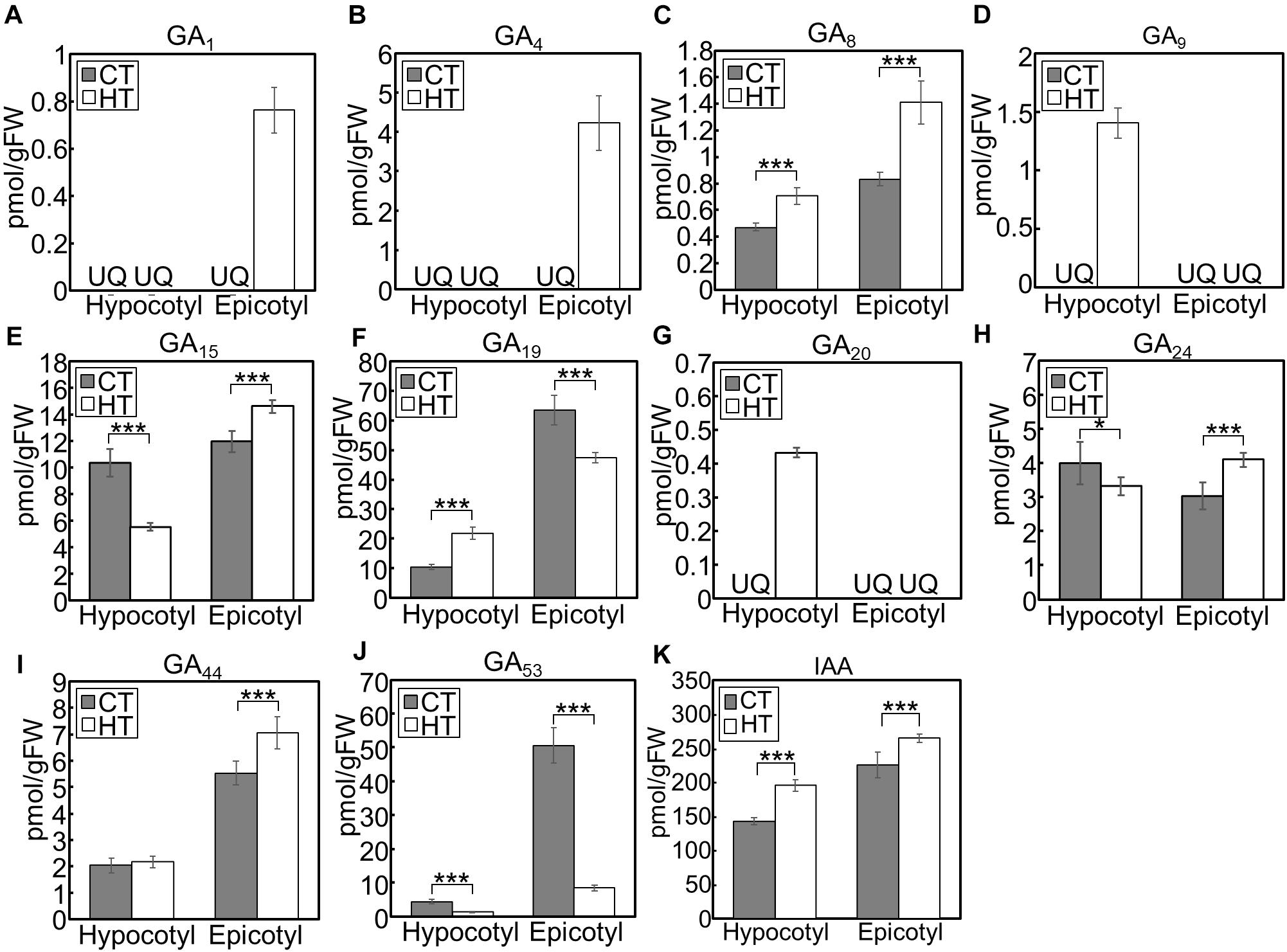
Figure 7. GA and IAA concentration in hypocotyls and epicotyls under two different temperatures. Hypocotyls and epicotyls of tomato seedlings grown for 7 days under control and high temperatures were harvested and subjected to hormone analysis. The concentrations of GAs (A–J) and IAA (K) were quantified using liquid chromatography-tandem mass spectrometry. Data are represented as mean ± SD (n = 4 or 5). *P < 0.1; ***P < 0.01 (Student’s t test). GA7 and GA12 were below the quantification limit. CT, control temperature; HT, high temperature; FW, fresh weight; UQ, under quantification limit.
IAA concentrations in negative DIF-treated hypocotyls and epicotyls were slightly but significantly lower than those in positive DIF-treated tissues (Figure 6K). In contrast, the concentrations of IAA in hypocotyls and epicotyls were higher under HT than those under CT (Figure 7K). These results were consistent with the expression pattern of IAA biosynthesis and signaling genes (Figures 4, 5).
We also quantified the concentration of cytokinins (CKs) because the involvement of this hormone in vascular development is well-documented (Kieber and Schaller, 2014). Several N6-(Δ2-isopentenyl) adenine (iP)-type and trans-zeatin-type (tZ-type) species had higher concentrations in negative DIF-treated epicotyls than positive DIF-treated epicotyls (Supplementary Figure 4). Conversely, an opposite effect was found under the CT and HT treatments (Supplementary Figure 5).
Discussion
In this study, we demonstrated that DT and NT affect stem growth in tomato seedlings, and that this is possibly mediated by the regulation of GA-, IAA-, and cell wall-related genes. Under the positive DIF treatment, higher temperatures promoted stem elongation, and this was accompanied by the upregulation of GA and IAA synthesis genes, resulting in higher concentrations of their active forms. Conversely, negative DIF inhibited stem elongation, which was accompanied by the downregulation of GA and IAA synthesis genes and decrease in GA and IAA concentrations. Although numerous studies have investigated plant growth regulation in response to temperature, our study provides information about possible mechanisms regulating stem growth under DIF treatments.
Under the negative DIF treatment, the concentrations of GA1, GA4, and some GA precursors were lower, whereas those of GA53 were higher, compared with those under positive DIF treatment. Furthermore, the expression of SlGA20ox1 was downregulated under the negative DIF treatment. In contrast, under the positive DIF treatment (i.e., higher temperatures), upregulation of SlGA20ox1 was observed and this treatment had the opposite effect on the profile of GA1, GA4 and the precursors, compared to that of the negative DIF treatment. Since GA20ox catalyzes the following multi-step reactions: GA12/GA53→GA15/GA44→GA24/GA19→GA9/GA20, these results were consistent with the downregulation or upregulation of SlGA20ox1 expression. In addition, as GA8 is the inactivated form of GA1, lower GA8 concentrations in negative DIF-treated epicotyls supported weakened GA activity. GA20ox plays a key role in the GA biosynthesis pathway and affects bioactive GA content (Yamaguchi, 2008). In A. thaliana, the over-expression of GA20ox enhances hypocotyl elongation (Huang et al., 1998; Coles et al., 1999; Ferrero et al., 2019), and the expression of GA20ox (GA20ox1) is upregulated by high temperatures in hypocotyls (Stavang et al., 2009). Thus, it is suggested that SlGA20ox1 plays a key role in regulating stem elongation in tomato seedlings in response to different DIF conditions.
IAA is involved in thermomorphogenesis, such as stem elongation, in response to higher temperatures (Quint et al., 2016). In A. thaliana hypocotyls, high temperatures induce the expression of YUC, TAA and SAUR, which facilitates stem elongation (Stavang et al., 2009; Franklin et al., 2011). In our analysis, these homologs were downregulated under the negative DIF treatment and upregulated under the high temperature positive DIF treatment, suggesting that these IAA-related genes also play a role in regulating stem elongation in tomato in response to temperature conditions.
Expression patterns of the cell wall-related genes SlEXP1, SlEXP2, and SlXTH2 were correlated with GA- and IAA-biosynthesis genes. EXPs are cell wall loosening proteins, which cause cell wall expansion (Marowa et al., 2016). Previous studies have shown that EXPs are involved in stem elongation of Oryza sativa (Cho and Kende, 1997a,b; Lee and Kende, 2001, 2002; Choi et al., 2003; Zou et al., 2015) and respond to GA (Cho and Kende, 1997b; Lee and Kende, 2001, 2002). EXP1 has been found to be regulated by temperature in Agrostis scabra and Agrostis stolonifera (Xu et al., 2007). XTH catalyzes Xyloglucan endohydrolysis and endotransglycosylation, which is involved in the modification of cell wall structures (Rose et al., 2002). It has also been reported that EXPs and XTHs are regulated by IAA (Goda et al., 2004; Majda and Robert, 2018; Lehman and Sanguinet, 2019). The response of cell wall-related genes to temperature might be mediated by GA and/or IAA action in tomato seedlings.
Previous studies investigating the molecular mechanisms behind temperature acclimation in A. thaliana identified PIF4 as a key regulator (Proveniers and van Zanten, 2013; de Wit et al., 2014; Quint et al., 2016). Expression of PIF4 was upregulated by high temperatures and was found to control GA and IAA biosynthesis and signaling (Koini et al., 2009; Stavang et al., 2009; Franklin et al., 2011; Sun et al., 2012). In our experiment, SlPIF4 was upregulated in epicotyls under the high temperature positive DIF treatment, suggesting that similar regulatory systems are employed in tomato seedlings. Conversely, SlPIF4 expression was not affected by the negative DIF treatment. The role of PIF4 in the regulation of stem elongation under negative DIF remains to be clarified. Future studies should focus on constructing a loss-of-function mutant to help understand its role.
Stem thickness and vessel development did not differ significantly between the negative and positive DIF treatments. Limited studies have reported the effect of temperature on stem thickness and vessel development. Among the phytohormones, CKs play an important role in vascular development (Kieber and Schaller, 2014). In a cytokinin biosynthesis mutant of A. thaliana, stem thickness and the number of xylems were significantly decreased (Matsumoto-Kitano et al., 2008). Another recent study suggests that CK is involved in the variation in xylem development in Dutch and Japanese tomato cultivars (Qi et al., 2020). However, in our experimental conditions, the concentration of endogenous cytokinin was increased in the negative DIF and decreased in the positive DIF treatments, suggesting that CK plays a minor role in the regulation of xylem development under these DIF treatments. Further analysis is needed to elucidate the mechanisms underlying temperature-dependent regulation of stem thickness and xylem development.
It is believed that the quality of seedlings greatly affects the final agricultural yield. Our results show that short-term negative DIF treatment can control plant height without affecting stem thickness. This might be an effective technique for growing tomatoes in nurseries. Since stem thickening is accompanied by vascular development, it may give a positive effect on mineral transport, the partition of assimilates, and fruit growth. Findings in this study would shed light on mechanisms of possible new agricultural practices.
Materials and Methods
Plant Materials and Growth Conditions
The tomato (S. lycopersicum) cultivar used in this study was Managua RZ (RIJK ZWAAN, Netherlands), except for the growth comparison of tomato cultivars under DIF treatments (Supplementary Figure 3). In the comparison experiment, we used CF Momotaro-York and Daiki B Baria (Takii Seed, Kyoto, Japan), and Rinka and Reiyo (Sakata Seed, Kanagawa, Japan).
Tomato seeds were imbibed in water in a petri dish at 28°C in the dark for 2 d. After imbibition, germinated seeds were transferred to a wet rockwool block (Nippon Rockwool Corporation, Japan) and were grown at 23°C/23°C (DT/NT, 16 h photoperiod) under 130 to 140 μmol photons m–2 s–1 of fluorescent illumination for 5–6 d. Next, the young seedlings, whose cotyledons were fully opened, were further grown on the rockwool block with liquid culture medium under the following two temperature-condition pairs: 25°C/20°C and 30°C/25°C, or 30°C/25°C and 25°C/30°C (DT/NT, 16 h photoperiod) under 300 μmol photons m–2 s–1 of light for 7 d (Supplementary Figure S1). The liquid culture medium contained nutrients at the following concentrations: 5 mM KNO3, 1 mM NH4H2PO4, 0.5 mM MgSO4, 5.5 mM Ca(NO3)2, 27 μM Fe-EDTA, 25 μM KCl, 10 μM H3BO3, 1 μM MnSO4, 1 μM ZnSO4, 0.25 μM CuSO4, and 0.04 μM Na2MoO4.
Fluorescent Observation of Xylem
The middle section of the tomato seedling hypocotyls was harvested and fixed in a 4% paraformaldehyde phosphate buffer solution (Nacalai Tesque, Kyoto, Japan). The fixed hypocotyls were thinly sliced (∼0.5 mm) with a razor blade, and cross sections were observed with fluorescence microscopy (Mirror unit with U-FUW, Olympus BX53, Olympus, Japan). The diameter of vessels was measured using the ImageJ software (Abramoff et al., 2004).
GA, IAA, and CK Quantification
Phytohormones were extracted and semi-purified as previously described (Kojima et al., 2009; Kojima and Sakakibara, 2012). CKs were quantified using an ultra-performance liquid chromatography (UPLC)-electrospray interface (ESI) tandem quadrupole mass spectrometer (qMS/MS) (AQUITY UPLCTM System/Xevo-TQS; Waters, Milford, MA, United States) as described previously (Kojima et al., 2009) with an ODS column (AQUITY UPLC HSS T3, 1.8mm, 2.1 x 100 mm; Waters). IAA and GAs were quantified using an ultra-high performance liquid chromatography (UHPLC)-ESI quadrupole-orbitrap mass spectrometer (UHPLC/Q-ExactiveTM; Thermo Fisher Scientific, United States) as described previously (Kojima and Sakakibara, 2012; Shinozaki et al., 2015) with an ODS column (AQUITY UPLC HSS T3, 1.8mm, 2.1 x 100 mm; Waters).
RNA Extraction
Hypocotyls and epicotyls were frozen in liquid nitrogen and ground to a fine powder with a mortar and pestle. Total RNA was extracted using an RNeasy Mini kit with an RNase-Free DNase Set (Cat. No. 74104/79254, Qiagen, Hilden, Germany). The RNA in samples was quantified using a NanoDrop-1000 spectrophotometer and the quality was monitored using an Agilent 2100 Bioanalyzer (Agilent Technologies, Santa Clara, CA, United States).
Microarray and Data Analysis
Total RNA was extracted from epicotyls of young seedlings grown for 7 d under 30°C/25°C (positive DIF) and 25°C/30°C (negative DIF) (DT/NT, 16 h photoperiod) as described in the preceding subsection. Three biological replicates were used for microarray analysis. Target labeling was performed according to the manual of the Low Input Quick Amp Labeling Kit, One-Color (Agilent Technologies). We used a tomato custom-designed microarray (platform ID “GPL21511”). Hybridization was performed according to the manufacturer’s instructions. We scanned the microarray images using an Agilent DNA Microarray Scanner G2565CA (Agilent Technologies). Scanned images were converted to signal data using Feature Extraction software (Agilent Technologies). Value definition in the data matrix was Log2. GO enrichment analysis among DEGs (top 300 upregulated and top 300 downregulated genes; P < 0.05) was performed using the GO Analysis Toolkit and Database for Agricultural Community (AgriGO, http://systemsbiology.cau.edu.cn/agriGOv2/) (Tian et al., 2017).
Reverse Transcriptase Real-time PCR
First-strand cDNA was synthesized using SuperScriptTM III First-Strand Synthesis SuperMix (Invitrogen, Waltham, MA, United States). Real-time PCR was performed using a StepOnePlus Real Time PCR system (Applied Biosystems, Waltham, MA, United States) and a KAPA SYBR FAST qPCR Master Mix (2×) Kit (Kapa Biosystems, London, United Kingdom) under the following conditions: 95°C for 3 min; followed by 40 cycles of 95°C for 3 s and 60°C for 20 s. Gene expression was calculated using the ΔΔCT method and normalized to that of the ubiquitin homolog as the housekeeping gene (Solyc01g056940). The following primers were used: for the housekeeping gene (Solyc01g056940), forward primer 5′-CG TGGTGGTGCTAAGAAGAG-3′, reverse primer 5′-ACGAAG CCTCTGAACCTTTC-3′; for SlGA20ox1 (Solyc03g006880), forward primer 5′-TGGCGTTCCATCAGTCCAAA-3′, reverse primer 5′-TTCGAGGGTTGTTGGAGTCC-3′; for SlTAA1 (Solyc05g031600), forward primer 5′-TGAAGCACACCCTGC ATTTG-3′, reverse primer 5′-ACTTCCAAATCTTCTTCCACT CCTT-3′; for SlYUC (Solyc08g068160), forward primer 5′-GC CCTCGTGGCTAAAGGAA-3′, reverse primer 5′-CCACTGCA TAAAGTCCACACTCTC-3′; for SlSAUR47 (Solyc04g053000), forward primer 5′-GAAGAACAGTTTGGCTTCGATTAC-3′, reverse primer 5′-CGGTATGTGAGATCAACAAACA-3′; for SlEXP2 (Solyc06g049050), forward primer 5′-TTCGAAGGGTG CCCTGTAT-3′, reverse primer 5′-TGAATATCACCAGCAC CTCCA-3′; for SlEXP1 (Solyc06g051800), forward primer 5′-CGCTGGCATTGTTCCTGT-3′, reverse primer 5′-CTGC ACCTGCTACATTCGTG-3′; for SlXTH2 (Solyc07g009380), forward primer 5′-TATGCACAAGGCAAGGGAGA-3′, reverse primer 5′-TGTATTGTCTTATTGGTGTCCCATC-3′; for SlPIF4 (Solyc07g043580), forward primer 5′-ATCAAGCAGCTGCAAT GTGC-3′, reverse primer 5′-CTGCTGAGTTTGCTGTGCTG-3′.
Data Availability Statement
Microarray data have been deposited in the National Center for Biotechnology Information Gene Expression Omnibus (NCBI GEO) database under accession number GSE131496.
Author Contributions
KO, AY, SI, and HS designed research. KO, AY, YK, KF, MK, YT, and KY performed research. KO and YK analyzed the data. KO and HS wrote the manuscript. All authors contributed to the article and approved the submitted version.
Funding
This research was supported by the Cross-ministerial Strategic Innovation Promotion Program (SIP) and partially by JSPS KAKENHI Grant Number JP19H05462.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Momo Okumura, Mayumi Tanigawa, and Mizuki Yamada for their advice on tomato cultivation. Microarray analysis was assisted by the National Agriculture and Food Research Organization (NARO).
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2020.577235/full#supplementary-material
Supplementary Figure 1 | Schematic representation of growth conditions in the study. (A) Control temperature treatment, 25°C/20°C (DT/NT). (B) High temperature/positive DIF treatment, 30°C/25°C (DT/NT). (C) Negative DIF treatment, 25°C/30°C (DT/NT). Young tomato seedlings were grown for 7 days under each condition. White bars show the light period, black bars show the dark period, and red arrowheads show the sampling time for the experiments. Further details are explained in the Materials and Methods.
Supplementary Figure 2 | Growth of tomato plants under negative and positive DIF treatments. Young tomato seedlings were grown for 7 days under positive and negative DIF treatments. (A) Growth of seedlings. Seedlings of tomato were photographed at 0, 1, 3, 5, and 7 days. Scale bars = 5 cm. (B,C) Hypocotyl and epicotyl length, respectively. (D,E) Hypocotyl and epicotyl diameters, respectively. (F) Cotyledons and true leaves photographed at 7 days after onset of DIF treatments. Scale bars = 5 cm. Data are represented as mean ± SD (n = 3 or 4). ∗P < 0.05; ∗∗P < 0.01 (Student’s t test). +DIF, positive DIF; -DIF, negative DIF.
Supplementary Figure 3 | Growth comparison of five tomato cultivars under DIF treatments. Seedlings of five tomato cultivars, Daiki B baria (DaikiB), Rinka, CF momotaro-york (CFMY), Managua RZ (MA), and Reiyo (RYO) were grown for 7 days under positive and negative DIF. (A) Growth of seedlings. Seedlings of tomato were photographed at 7 days. Scale bars = 5 cm. (B,C) Hypocotyl and epicotyl length, respectively. (D,E) Hypocotyl and epicotyl diameter, respectively. Data are shown as boxplots (n = 3). Cross marks in boxplot indicate the mean values. ∗P < 0.05; ∗∗P < 0.01 (Student’s t test). +DIF, positive DIF; -DIF, negative DIF.
Supplementary Figure 4 | Concentration of CKs in hypocotyls and epicotyls under positive and negative DIF treatments. Hypocotyls and epicotyls of tomato seedlings grown for 7 days under positive and negative DIF treatments were harvested and subjected to hormone analysis. The concentration of CKs were quantified using liquid chromatography-tandem mass spectrometry. Data are represented as mean ± SD (n = 3 to 5). tZ, trans-zeatin; tZR, tZ riboside; tZRPs, tZR 5′-phosphates; tZ7G, tZ-7N-glucoside; tZ9G, tZ-9N-glucoside; tZOG, tZ-O-glucoside; tZROG, tZR-O-glucoside; iP, N6-(Δ2-isopentenyl)adenine; iPR, iP riboside; iPRPs, iPR 5′-phosphates; iP7G, iP-7N-glucoside; iP9G, iP-9N-glucoside. ∗P < 0.1; ∗∗P < 0.05; ∗∗∗P < 0.01 (Student’s t test). tZRPOG, tZRP-O-glucoside was below the quantification limit. +DIF, positive DIF; -DIF, negative DIF; FW, fresh weight; UQ, under quantification limit.
Supplementary Figure 5 | Concentration of CKs in hypocotyls and epicotyls under two different temperatures. Hypocotyls and epicotyls of tomato seedlings grown for 7 days under control and high temperatures were harvested and subjected to hormone analysis. The concentration of CKs were quantified using liquid chromatography-tandem mass spectrometry. Data are represented as mean ± SD (n = 3 to 5). tZ, trans-zeatin; tZR, tZ riboside; tZRPs, tZR 5′-phosphates; tZ7G, tZ-7N-glucoside; tZ9G, tZ-9N-glucoside; tZOG, tZ-O-glucoside; tZROG, tZR-O-glucoside; iP, N6-(Δ2-isopentenyl)adenine; iPR, iP riboside; iPRPs, iPR 5′-phosphates; iP7G, iP-7N-glucoside; iP9G, iP-9N-glucoside. ∗P < 0.1; ∗∗P < 0.05; ∗∗∗P < 0.01 (Student’s t test). tZRPOG, tZRP-O-glucoside was below the quantification limit. CT, control temperature; HT, high temperature; FW, fresh weight; UQ, under quantification limit.
Abbreviations
CT, control temperature; DT, day temperature; +DIF, positive DIF; -DIF, negative DIF; HT, high temperature; NT, night temperature.
References
Abramoff, M. D., Magalhaes, P. J., and Ram, S. J. (2004). Image processing with ImageJ. Biophoton. Int. 11, 36–42.
Binenbaum, J., Weinstain, R., and Shani, E. (2018). Gibberellin localization and transport in plants. Trends Plant Sci. 23, 410–421. doi: 10.1016/j.tplants.2018.02.005
Cho, H. T., and Kende, H. (1997a). Expansins and internodal growth of deepwater rice. Plant Physiol. 113, 1145–1151. doi: 10.1104/pp.113.4.1145
Cho, H. T., and Kende, H. (1997b). Expression of expansin genes is correlated with growth in deepwater rice. Plant Cell 9, 1661–1671. doi: 10.1105/tpc.9.9.1661
Choi, D., Lee, Y., Cho, H. T., and Kende, H. (2003). Regulation of expansin gene expression affects growth and development in transgenic rice plants. Plant Cell 15, 1386–1398. doi: 10.1105/tpc.011965
Coles, J. P., Phillips, A. L., Croker, S. J., Garcia-Lepe, R., Lewis, M. J., and Hedden, P. (1999). Modification of gibberellin production and plant development in Arabidopsis by sense and antisense expression of gibberellin 20-oxidase genes. Plant J. 17, 547–556. doi: 10.1046/j.1365-313X.1999.00410.x
Davière, J. M., and Achard, P. (2013). Gibberellin signaling in plants. Development 140, 1147–1151. doi: 10.1242/dev.087650
de Koning, A. N. M. (1988). The effect of different day/night temperature regimes on growth, development and yield of glasshouse tomatoes. J. Hortic. Sci. 63, 465–471. doi: 10.1080/14620316.1988.11515880
de Wit, M., Lorrain, S., and Fankhauser, C. (2014). Auxin-mediated plant architectural changes in response to shade and high temperature. Physiol. Plant. 151, 13–24. doi: 10.1111/ppl.12099
Ferrero, L. V., Viola, I. L., Ariel, F. D., and Gonzalez, D. H. (2019). Class I TCP transcription factors target the gibberellin biosynthesis gene GA20ox1 and the growth-promoting genes HBI1 and PRE6 during thermomorphogenic growth in Arabidopsis. Plant Cell Physiol. 60, 1633–1645. doi: 10.1093/pcp/pcz137
Franklin, K. A., Lee, S. H., Patel, D., Kumar, S. V., Spartz, A. K., Gu, C., et al. (2011). Phytochrome-interacting factor 4 (PIF4) regulates auxin biosynthesis at high temperature. Proc. Natl. Acad. Sci. U.S.A. 108, 20231–20235. doi: 10.1073/pnas.1110682108
Goda, H., Sawa, S., Asami, T., Fujioka, S., Shimada, Y., and Yoshida, S. (2004). Comprehensive comparison of auxin-regulated and brassinosteroid-regulated genes in Arabidopsis. Plant Physiol. 134, 1555–1573. doi: 10.1104/pp.103.034736
Grindal, G., Ernstsen, A., Reid, J. B., Junttila, O., Lindgård, B., and Moe, R. (1998). Endogenous gibberellin A1 levels control thermoperiodic stem elongation in Pisum sativum. Physiol. Plant. 102, 523–531. doi: 10.1034/j.1399-3054.1998.1020406.x
Hayata, Y., Kotani, K., and Li, X. X. (2001). Effects of day and night temperatures on hypocotyl elongation and endogenous levels of indole-3-acetic and abscisic acids in radish (Raphaus sativus L.). Engei Gakkai Zasshi 70, 443–447. doi: 10.2503/jjshs.70.443
Huang, S., Raman, A. S., Ream, J. E., Fujiwara, H., Cerny, R. E., and Brown, S. M. (1998). Overexpression of 20-oxidase confers a gibberellin-overproduction phenotype in Arabidopsis. Plant Physiol. 118, 773–781. doi: 10.1104/pp.118.3.773
Jensen, E., Eilertsen, S., Ernsten, A., Juntilla, O., and Moe, R. (1996). Thermoperiodic control of stem elongation and endogenous gibberellins in Campanula isophylla. J. Plant Growth Regul. 15, 167–171. doi: 10.1007/BF00190580
Kieber, J. J., and Schaller, G. E. (2014). Cytokinins. Arabidopsis Book 12:e0168. doi: 10.1199/tab.0168
Koini, M. A., Alvey, L., Allen, T., Tilley, C. A., Harberd, N. P., Whitelam, G. C., et al. (2009). High temperature-mediated adaptations in plant architecture require the bHLH transcription factor PIF4. Curr. Biol. 19, 408–413. doi: 10.1016/j.cub.2009.01.046
Kojima, M., Kamada-Nobusada, T., Komatsu, H., Takei, K., Kuroha, T., Mizutani, M., et al. (2009). Highly sensitive and high-throughput analysis of plant hormones using MS-probe modification and liquid chromatography-tandem mass spectrometry: an application for hormone profiling in Oryza sativa. Plant Cell Physiol. 50, 1201–1214. doi: 10.1093/pcp/pcp057
Kojima, M., and Sakakibara, H. (2012). Highly sensitive high-throughput profiling of six phytohormones using MS-probe modification and liquid chromatography-tandem mass spectrometry. Methods Mol. Biol. 918, 151–164. doi: 10.1007/978-1-61779-995-2_11
Lee, Y., and Kende, H. (2001). Expression of β-expansins is correlated with internodal elongation in deepwater rice. Plant Physiol. 127, 645–654. doi: 10.1104/pp.010345
Lee, Y., and Kende, H. (2002). Expression of α-expansin and expansin-like genes in deepwater rice. Plant Physiol. 130, 1396–1405. doi: 10.1104/pp.008888
Lehman, T. A., and Sanguinet, K. A. (2019). Auxin and cell wall crosstalk as revealed by the Arabidopsis thaliana cellulose synthase mutant radially swollen 1. Plant Cell Physiol. 60, 1487–1503. doi: 10.1093/pcp/pcz055
Majda, M., and Robert, S. (2018). The role of auxin in cell wall expansion. Int. J. Mol. Sci. 19:951. doi: 10.3390/ijms19040951
Marowa, P., Ding, A., and Kong, Y. (2016). Expansins: roles in plant growth and potential applications in crop improvement. Plant Cell Rep. 35, 949–965. doi: 10.1007/s00299-016-1948-4
Matsumoto-Kitano, M., Kusumoto, T., Tarkowski, P., Kinoshita-Tsujimura, K., Václavíková, K., Miyawaki, K., et al. (2008). Cytokinins are central regulators of cambial activity. Proc. Natl. Acad. Sci. U.S.A. 105, 20027–20031. doi: 10.1073/pnas.0805619105
Nomoto, Y., Kubozono, S., Miyachi, M., Yamashino, T., Nakamichi, N., and Mizuno, T. (2012). A circadian clock- and PIF4-mediated double coincidence mechanism is implicated in the thermosensitive photoperiodic control of plant architectures in Arabidopsis thaliana. Plant Cell Physiol. 53, 1965–1973. doi: 10.1093/pcp/pcs141
Proveniers, M. C., and van Zanten, M. (2013). High temperature acclimation through PIF4 signaling. Trends Plant Sci. 18, 59–64. doi: 10.1016/j.tplants.2012.09.002
Qi, X., Takahashi, H., Kawasaki, Y., Ohta, Y., Isozaki, M., Kojima, M., et al. (2020). Differences in xylem development between Dutch and Japanese tomato (Solanum lycopersicum) correlate with cytokinin levels in hypocotyls. Ann. Bot. 126, 315–322. doi: 10.1093/aob/mcaa094
Quint, M., Delker, C., Franklin, K. A., Wigge, P. A., Halliday, K. J., and Van Zanten, M. (2016). Molecular and genetic control of plant thermomorphogenesis. Nat. Plants 2:15190. doi: 10.1038/NPLANTS.2015.19
Rose, J. K. C., Braam, J., Fry, S. C., and Nishitani, K. (2002). The XTH family of enzymes involved in xyloglucan endotransglucosylation and endohydrolysis: current perspectives and a new unifying nomenclature. Plant Cell Physiol. 43, 1421–1435. doi: 10.1093/pcp/pcf171
Shimizu, H. (2007). Effect of day and night temperature alternations on plant morphogenesis. Environ. Control Biol. 45, 259–265. doi: 10.2525/ecb.45.259
Shinozaki, Y., Hao, S., Kojima, M., Sakakibara, H., Ozeki-Iida, Y., Zheng, Y., et al. (2015). Ethylene suppresses tomato (Solanum lycopersicum) fruit set through modification of gibberellin metabolism. Plant J. 83, 237–251. doi: 10.1111/tpj.12882
Stavang, J. A., Gallego-Bartolomé, J., Gómez, M. D., Yoshida, S., Asami, T., Olsen, J. E., et al. (2009). Hormonal regulation of temperature-induced growth in Arabidopsis. Plant J. 60, 589–601. doi: 10.1111/j.1365-313X.2009.03983.x
Stavang, J. A., Lindgård, B., Erntsen, A., Lid, S. E., Moe, R., and Olsen, J. E. (2005). Thermoperiodic stem elongation involves transcriptional regulation of gibberellin deactivation in pea. Plant Physiol. 138, 2344–2353. doi: 10.1104/pp.105.063149
Sun, J., Qi, L., Li, Y., Chu, J., and Li, C. (2012). PIF4-mediated activation of YUCCA8 expression integrates temperature into the auxin pathway in regulating Arabidopsis hypocotyl growth. PLoS Genet. 8:e1002594. doi: 10.1371/journal.pgen.1002594
Thingnaes, E., Torre, S., Ernstsen, A., and Moe, R. (2003). Day and night temperature responses in Arabidopsis: effects on gibberellin and auxin content, cell size, morphology and flowering time. Ann. Bot. 92, 601–612. doi: 10.1093/aob/mcg176
Tian, T., Liu, Y., Yan, H., You, Q., Yi, X., Du, Z., et al. (2017). agriGO v2.0: a GO analysis toolkit for the agricultural community, 2017 update. Nucleic Acids Res. 45, W122–W129. doi: 10.1093/nar/gkx382
Went, F. W. (1944). Plant growth under controlled conditions. II. Thermoperiodicity in growth and fruiting of tomato. Am. J. Bot. 31, 135–150. doi: 10.2307/2437636
Xu, J., Tian, J., Belanger, F. C., and Huang, B. (2007). Identification and characterization of an expansin gene AsEXP1 associated with heat tolerance in C3 Agrostis grass species. J. Exp. Bot. 58, 3789–3796. doi: 10.1093/jxb/erm229
Yamaguchi, S. (2008). Gibberellin metabolism and its regulation. Annu. Rev. Plant Biol. 59, 225–251. doi: 10.1146/annurev.arplant.59.032607.092804
Zhao, Y. (2018). Essential roles of local auxin biosynthesis in plant development and in adaptation to environmental changes. Annu. Rev. Plant Biol. 29, 417–435. doi: 10.1146/annurev-arplant-042817-040226
Keywords: auxin, DIF, gibberellin, Solanum lycopersicum, stem elongation, tomato
Citation: Ohtaka K, Yoshida A, Kakei Y, Fukui K, Kojima M, Takebayashi Y, Yano K, Imanishi S and Sakakibara H (2020) Difference Between Day and Night Temperatures Affects Stem Elongation in Tomato (Solanum lycopersicum) Seedlings via Regulation of Gibberellin and Auxin Synthesis. Front. Plant Sci. 11:577235. doi: 10.3389/fpls.2020.577235
Received: 29 June 2020; Accepted: 16 November 2020;
Published: 08 December 2020.
Edited by:
M. Iqbal R. Khan, Jamia Hamdard University, IndiaReviewed by:
Dr. Aditi Gupta, Centre for Research in Agricultural Genomics (CRAG), SpainCris Argueso, Colorado State University, United States
Copyright © 2020 Ohtaka, Yoshida, Kakei, Fukui, Kojima, Takebayashi, Yano, Imanishi and Sakakibara. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Hitoshi Sakakibara, c2FrYWtpQGFnci5uYWdveWEtdS5hYy5qcA==
 Kinuka Ohtaka
Kinuka Ohtaka Akiko Yoshida1,4
Akiko Yoshida1,4 Yusuke Kakei
Yusuke Kakei Kosuke Fukui
Kosuke Fukui Kanako Yano
Kanako Yano Hitoshi Sakakibara
Hitoshi Sakakibara