
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Plant Sci. , 25 August 2020
Sec. Plant Abiotic Stress
Volume 11 - 2020 | https://doi.org/10.3389/fpls.2020.01311
Potassium transporters play an essential role in maintaining cellular ion homeostasis, turgor pressure, and pH, which are critical for adaptation under salt stress. We identified a salt responsive Avicennia officinalis KUP/HAK/KT transporter family gene, AoKUP2, which has high sequence similarity to its Arabidopsis ortholog AtKUP2. These genes were functionally characterized in mutant yeast cells and Arabidopsis plants. Both AoKUP2 and AtKUP2 were induced by salt stress, and AtKUP2 was primarily induced in roots. Subcellular localization revealed that AoKUP2 and AtKUP2 are localized to the plasma membrane and mitochondria. Expression of AtKUP2 and AoKUP2 in Saccharomyces cerevisiae mutant strain (BY4741 trk1Δ::loxP trk2Δ::loxP) helped to rescue the growth defect of the mutant under different NaCl and K+ concentrations. Furthermore, constitutive expression of AoKUP2 and AtKUP2 conferred enhanced salt tolerance in Arabidopsis indicated by higher germination rate, better survival, and increased root and shoot length compared to the untreated controls. Analysis of Na+ and K+ contents in the shoots and roots showed that ectopic expression lines accumulated less Na+ and more K+ than the WT. Two stress-responsive transcription factors, bHLH122 and WRKY33, were identified as direct regulators of AtKUP2 expression. Our results suggest that AtKUP2 plays a key role in enhancing salt stress tolerance by maintaining cellular ion homeostasis.
Salinity is a major environmental threat for crop production because high concentration of salt in the soil severely affects plant performance by disturbing cellular metabolism. Such adverse effects of increased salinity occur mainly due to the osmotic stress and continuous accumulation of toxic ions within the plant cells. Additionally, this can lead to oxidative stress and nutritional imbalance (Munns and Tester, 2008). Ion transport is a critical step controlling ion homeostasis in plants. This includes the transport of ions across the root cell plasma membrane or through vacuolar membrane and also occurs via xylem and phloem in order to facilitate the accumulation and excretion of salt by specialized cells (Volkov and Beilby, 2017). The physiological and morphological disturbances under saline conditions are often caused by the accumulation of toxic ions such as Na+ and Cl-. This ion toxicity can be minimized by three possible ways, (i) compartmentalization of Na+ and Cl- ions into the vacuole (ii) through exclusion of Na+ and Cl- outside the cell and (iii) through limiting net Na+ and Cl− uptake (Munns and Tester, 2008; Roy et al., 2014).
The disproportionate accumulation of Na+ in the intracellular and extracellular compartments adversely affects the uptake and homeostasis of essential nutrients such as, K+ and Ca2+. The activation of Na+ and K+ transport systems helps in retaining the cellular ion homeostasis by maintaining low Na+ content in leaves and high K+/Na+ ratio in cytosol. The optimum maintenance of K+/Na+ ratio in cytosol can be achieved by either limiting the accumulation of Na+ in plant tissues or by preventing the loss of K+ across the plasma membrane (Almeida et al., 2017; Assaha et al., 2017). Thus far, most of the plant breeders and physiologists have focused mainly on the former mechanism. There is increasing evidence to show that the ability of a plant to maintain high K+/Na+ ratio in cytosol seems to be crucial for plant survival under salt stress. Rather than vacuolar content, the cytosolic K+ homeostasis is crucial for plant metabolic processes (Wu et al., 2018).
There are mainly two kinds of transport systems in plants, (a) through channels or (b) via transporters (Adams and Shin, 2014; Ruiz-Lau et al., 2016). Four multi-gene K+ transporter families are found in plants, (i) KUP/HAK/KT, (ii) HKT/Trk, (iii) CHX, and (iv) KEA transporters (Véry and Sentenac, 2003; Sharma et al., 2013; Yang et al., 2014; Gupta et al., 2018). The large family of high affinity potassium transporters (HAK/KUP/KT) in plants mainly contribute to root K+ acquisition under a wide range of external K+ levels. They also mediate K+ movement within the plant and K+ efflux into the environment, thereby maintaining ion homeostasis (Liang et al., 2020). Among these transporters, HAKs have been extensively studied. In rice, induction of OsHAK1 expression was observed upon NaCl treatment or by K+ deficiency (Chen et al., 2015). Constitutive expression of OsHAK5 in BY2 tobacco cells improved the K+ accumulation but not the Na+ accumulation under salt treatment and conferred increased salinity tolerance (Horie et al., 2011). Most of the AtKT/KUPs are known to be localized to the root hairs and root tip (Ahn et al., 2004). The physiological functions in relation to K+ transportation, acquisition, and improving salinity tolerance of other distinct members of plant KUP/KT/HAK transporter family have not been well studied. Maintenance of K+ uptake under high external Na+ is crucial for K+/Na+ homeostasis leading to salt tolerance (Munns and Tester, 2008). On the other hand, increased Na+ level suppresses the expression of several HAK/KUP/KT K+ transporters at low K+ conditions (Nieves-Cordones et al., 2010). Therefore, in order to devise strategies for crop improvement, it is important to identify and characterize KUP transporters that can function under elevated NaCl conditions.
The KUP/HAK/KT transporter family was first identified in bacteria through mutagenesis of K+-dependent Escherichia coli strains harboring the kdpABC5 mutation (Epstein and Kim, 1971). The KUP family has 13 genes in Arabidopsis and 27 genes in rice. Their ubiquitous presence in plants suggests that they play a significant role in acquisition of nutrients and survival in potassium-poor environments. In Arabidopsis, it was shown that AtKUPs (KUP1, 2, 4, 5-7, 10, and 11) complement a mutant E. coli strain that lacks K+ uptake gene, which proves that these KUP proteins play an important function in transport of K+ (Fu and Luan, 1998; Kim et al., 1998; Ahn et al., 2004; Gierth and Mäser, 2007; Horie et al., 2011; Chen et al., 2015; Han et al., 2016). In Arabidopsis, the semi-dominant mutant of AtKUP2 also named as shy3-1causes growth-related defects such as short hypocotyl, short flowering stem and small leaves (Elumalai et al., 2002). Since potassium is a main solute, defect in cellular homeostasis of K+ might affect the turgor pressure and perturb the expansion rate of cells. Increased expression of KUP members has shown contrasting results. While overexpression of GhKT1 and VvKUPs (VvKUP1 and VvKUP2) was shown to increase the turgor pressure to drive rapid elongation of cotton fibres and Vitis vinifera, respectively (Ruan et al., 2001; Davies et al., 2006), enhanced cell expansion was observed in the triple mutant kup268 where the expression of KUP2, KUP6 and KUP8 are disrupted along with guard cell outward rectifying K+ channel gene, GORK (Osakabe et al., 2013). KUP2, KUP6 and KUP8 together are shown to have a role in lateral root (LR) formation by positively affecting the ABA and osmotic stress responses. Also, mutation of KUP leads to enhanced expression of auxin responsive LBD genes, which are involved in control of lateral root formation (Okushima et al., 2007; Lee et al., 2009). Altogether, KUP2, KUP6, and KUP8 are involved in initiation of lateral root formation and in the development of antagonistic signal crosstalk between auxin and ABA. Similarly, AtKUP4 and AtKUP9 contribute to auxin homeostasis in Arabidopsis root. The disruption of AtKUP4/TRH1 (Tiny Root Hair 1) affects the root hair elongation due to auxin deficiency and AtKUP9 mediates K+ and auxin efflux to maintain meristem activity under low K+ stress (Rigas et al., 2001; Rigas et al., 2013; Daras et al., 2015; Zhang et al., 2020). This further highlights the functional importance of this gene family and the need to better understand them in plants.
Regulation of genes invariably involves specific transcription factors (TFs). Hence, in order to understand the molecular regulatory mechanisms behind the expression of KUPs and how they function in salinity stress tolerance, the associated TFs should be studied. Plants have developed complicated stress response strategies that include differential expression of genes encoding TFs. Various TFs such as bHLH, WRKY, MYC, NAC, MYB, and ERF/AP2 have been identified to be associated with salt tolerance mechanism (Okushima et al., 2007; Golldack et al., 2011; Hoang et al., 2017; Meraj et al., 2020). Nevertheless, information on how these TFs modulate their respective downstream target genes is quite limited in this area.
One of the approaches used to address salinity stress is through the investigation of salt responsive genes in halophytic species. Halophytes employ three mechanisms to confer salt tolerance; compartmentalization, reduction of the Na+ influx, and excretion of Na+ ions (Mishra and Tanna, 2017). In attempts to better understand the molecular mechanisms behind salinity tolerance, salt-responsive genes have been isolated from specific halophytes and expressed in glycophytes (Himabindu et al., 2016). Avicennia officinalis is a halophyte with unique characteristics such as efficient salt filtration at the roots and salt secreting glands on leaves (Krishnamurthy et al., 2017). However, not much work is done to understand the functioning of their ion transporters which might serve as potential candidates for generating salt tolerant crops.
In this report, the physiological function and expression patterns of AoKUP2 from A. officinalis and its Arabidopsis ortholog, AtKUP2 were characterized. AoKUP2 and AtKUP2 expression were studied in Arabidopsis under varying salt stress conditions. We show that expression of AtKUP2 and AoKUP2 in transgenic Arabidopsis and yeast improved growth, K+ uptake and salinity tolerance. Our findings show that AtKUP2 expression is directly regulated by bHLH122 and WRKY33 while playing an essential function in mediating K+ transport and maintaining plant growth under salt stress.
T-DNA insertional mutants, atkup2 (SAIL_504_A07), atbhlh122 (SALK_002286), atwrky33 (SALK_064436) and wild-type (WT) Arabidopsis thaliana, ecotype Columbia-0 were obtained from the Arabidopsis Biological Resource Center (ABRC) seed stock. Ectopic expression lines, 35S::AoKUP2 and 35S::AtKUP2 were generated by us for this study. A. officinalis L. (A. officinalis) propagules were collected and salt treated as described in (Krishnamurthy et al., 2014; Krishnamurthy et al., 2017).
Mutant line atkup2 (SAIL_504_A07) with T-DNA insertion was obtained from the SALK collection (Alonso et al., 2003). Positions of T-DNA insertion sites are shown in Supplementary Figure S1A. Plants homozygous for the T-DNA insertion were selected by genotyping with primers designed using the T-DNA primer design tool (http://signal.salk.edu/tdnaprimers.2.html) (Supplementary Figure S1B). To check the suppression of AtKUP2 in mutant, qRT-PCR was carried out. Seeds were collected from only those lines that showed more than 80% suppression of AtKUP2 (Supplementary Figure S1C). For generation of ectopic expression lines in Arabidopsis, coding DNA sequences (CDS) of AoKUP2 and AtKUP2 were cloned into pGreen binary vector. The constructs 35S::AoKUP2, 35S::AtKUP2 and pAtKUP2::GUS were electroporated into Agrobacterium tumefaciens strain GV3101:pMP90 and introduced into WT by the floral dip method (Clough and Bent, 1998). Basta-resistant T1 transgenic plants were selected and gene expression was confirmed by genotyping PCR and qRT-PCR (Supplementary Figures S1D, E) analyses. T3 generation plants were used for all the experiments. For chromatin immunoprecipitation (ChIP) assay, CDS of AtbHLH122 and AtWRKY33 were cloned into pGreen binary vector with hemagglutinin (HA) fusion tag. All the plasmids were sequence verified before use and the primers used in the study are listed in Supplementary Table S1.
Total RNA was extracted from leaf and root tissues of control and treated (500 mM NaCl for varying time periods; 0, 1, 3, 6, 12, and 24 h) greenhouse-grown A. officinalis and control and treated (50 mM NaCl for varying time periods; 0, 1, 3, 6, 12, and 24 h) tissues of 1-week-old WT Arabidopsis seedlings using TRIzol™ reagent (Life Technologies) following the manufacturer’s instructions. An aliquot of this RNA (1 µg) was used to synthesize cDNA using Maxima first strand cDNA synthesis kit for qRT-PCR (Thermo Scientific) following the manufacturer’s instructions. For genotyping and expression analysis of mutants and the heterologous expression lines, DNA and RNA were extracted from leaves of four-week-old seedlings. The qRT-PCR for selected genes was performed as described earlier (Krishnamurthy et al., 2019). The primers used in the study are listed in Supplementary Table S1. Constitutively expressed AtUbiquitin 10 was used as internal control.
For histochemical study, transcriptional reporter line pAtKUP2::GUS was generated. To carry out GUS staining assays, cold stratified T3 seeds were sown on MS plates. One-week-old, untreated and salt treated (50 mM NaCl for 12 h) seedlings and 4-week-old seedling parts were immersed into the GUS staining solution [0.1 M sodium phosphate buffer (pH 7.0), 10 mM EDTA, 0.1% Triton-X, and 2 mM 5-bromo-4-chloro-3-indolyl glucuronide (X-Gluc)]. The tissues were vacuum infiltrated for 2 min and then processed as described (Ravindran et al., 2017). GUS expression in different parts of seedlings was quantified based on the relative intensities of blue coloration using ImageJ software. Data presented are mean ± SE of three biological replicates, each biological replicate consisting of at least six plants.
The CDS of AoKUP2 and AtKUP2 were fused in-frame to both C- and N-terminal of GFP in the 35S::pGREEN vector and sequence confirmed. Empty vector, 35S::GFP-pGREEN was used as control. These plasmids as well as the plasmids containing subcellular markers (PM and mitochondria) were introduced into Agrobacterium. The subcellular marker plasmids (MT-rk CD3‐991 and PM-rk CD3-1007) were obtained from TAIR. About 3- to 4-week-old leaves of N. benthamiana were co-infiltrated with Agrobacterium harboring 35S::GFP : At/AoKUP2 or 35S::At/AoKUP2:GFP and the related subcellular marker constructs. N. benthamiana leaf epidermal cells were examined for GFP-Ao/AtKUP2 expression along with mCherry-tagged markers using a confocal laser scanning microscope (Olympus FV3000) at 488 and 561 nm wavelengths, respectively.
Saccharomyces cerevisiae strain BY4741 (MATa his3Δ1 leu2Δ met15Δ ura3Δ; EUROSCARF) and its derivative: BYT12 (BY4741 trk1Δ::loxP trk2Δ::loxP) (Petrezsélyová et al., 2011) were used to carry out the complementation experiments. Yeast strains were cultivated at 30°C either in standard YPD (1% yeast extract, 2% peptone, and 2% glucose) or the synthetic minimal medium YNB (Difco; 0.67% yeast nitrogen base without amino acids and 2% glucose). Synthetic minimal medium was supplemented with the appropriate auxotrophic requirements. Semi-solid medium was prepared by adding 2% agar. The Escherichia coli strain DH5α was used to amplify the plasmid DNA and was grown in Luria–Bertani (LB) broth with 100 μg/mL ampicillin at 37°C. The coding sequence of AtKUP2 and AoKUP2 were cloned downstream of the respective promoter into the yeast multicopy vector YEp352 and the primers used for amplification are listed in Supplementary Table S1. WT transformed with empty YEp352 (Hill et al., 1986) plasmid was used as control. The yeast complementation assays with cells expressing AtKUP2 and AoKUP2 were performed on semi-solid YNB medium containing appropriate supplements. For drop tests, cell suspensions were adjusted to OD600 = 0.2, and 10-fold serial dilutions were made. A 10 μL aliquots of each sample were inoculated onto YNB semi-solid medium containing various concentrations of KCl (500, 1,000, and 1,500 mM) and NaCl (100, 200, and 500 mM). The growth of cells on plates was recorded for 5 days. The growth rate of the transformed WT and Δtrk1trk2 strains was monitored. These strains were also grown in liquid YPD to log phase (OD600nm = 0.6–0.8). The cells were collected by centrifugation (5,000×g, 10 min) and diluted to an OD600nm of 0.1 prior to culture in liquid SD-Ura with and without salts at 30°C, 160 rpm. The OD600nm was measured every 24 h.
After surface sterilization, seeds were cold stratified for 3 days at 4°C, before sowing on Murashige-Skoog (MS) agar medium. They were allowed to germinate under 16 h of light/8 h of dark at 22°C. After 1 week of germination, Arabidopsis seedlings were carefully removed from MS plate and exposed to salt (50 mM NaCl) treatment. The salt-treated plant tissues were collected at various time intervals (0, 1, 3, 6, 12, and 24 h) and frozen in liquid nitrogen for total RNA isolation. For seed germination assay, the surface sterilized and cold stratified seeds were sown on MS agar plate with and without NaCl treatment (50 and 75 mM) and allowed to germinate as described above. The number of germinated seeds was counted from day one to day four. For root length studies, the sterilized and cold stratified seeds were sown on MS agar plate with and without NaCl, and the root lengths were measured and photographed after 1 week of germination. Similarly, photographs were taken after 14 days for lateral root development assay. For studies in older seedlings, seeds were sown on compost soil and transferred to cold room for 4 days. Trays with cold-stratified seeds were incubated in growth chambers at 23°C and 75% RH under 16 h of light/8 h of dark. Four-week-old seedlings were treated with 150 mM NaCl for 1 week. The soil was rinsed with water twice to remove the soil-bound NaCl followed by a recovery growth in NaCl-free water for 1 week. For cell viability analyses, Arabidopsis seeds were germinated on MS plates. Three-day-old seedlings were transferred to MS supplemented with 50 mM NaCl. After the treatment, seedlings were incubated with propidium iodide (pI) for 1 min.
Control and salt-treated 4-week-old Arabidopsis plants were harvested and rinsed briefly with distilled water to remove surface contaminating Na+. Pools of four plants were taken as one replicate, and at least three independent replicates were used to generate the mean values reported. Leaves and roots from plants were separated at collection and left to dry at 50°C for 2 days. The dried tissue was ground into a powder in liquid nitrogen, and acid digestion and ion analysis were carried out as described earlier (Krishnamurthy et al., 2014).
Mesophyll protoplasts were isolated from leaves of 3- to 4-week-old WT Arabidopsis (Col-0) plants and transfected as described earlier (Yoo et al., 2007) with minor modifications. For each transfection, 8–15 μg of purified plasmid DNA (35S::AtWRKY33 and 35S::AtbHLH122) was used. Polyethylene glycol (PEG)–CaCl2 transfection solution used was as follows: 25% PEG, 0.4 M mannitol, and 150 mM CaCl2. The transfected protoplasts were incubated for 20 h at room temperature and fixed with formaldehyde. Protoplasts without any plasmids were used as the negative control. ChIP was performed as described previously (Kaufmann et al., 2010), with minor changes. Anti-HA monoclonal antibody (Santa Cruz Biotechnology) bound to Protein-A agarose beads (Sigma) was used to immunoprecipitate the genomic DNA fragments. ChIP-qPCR analysis was carried out as described (Krishnamurthy et al., 2019).
AtKUP2 promoter fragment of 1.5 kb was cloned into pGreen II-0800-LUC vector. Mesophyll protoplasts isolated from atwrky33 and atbhlh22 mutants were transfected with 35S::AtWRKY33 and 35S::AtbHLH122, respectively. The protoplasts transfected with pAtKUP2::LUC construct was used as control. 16–24 h after transfection, protoplasts were pelleted by centrifugation (14,000×g for 30 s). Protoplasts were lysed in 1x passive lysis buffer (the Dual-Luciferase® Reporter Assay System, Promega) and incubated at room temperature for 15 min. Following the incubation, 20 μL (approximately 6.6 × 104 cells) of lysed protoplasts were added to 100 μL LARII (the Dual-Luciferase® Reporter Assay System, Promega), vortexed briefly, and measured immediately using GloMax discover (Promega). The luminescence of Luciferase was quenched and Renilla luminescence measured by the addition of 100 μL of Stop & Glo® Buffer (the Dual-Luciferase® Reporter Assay System, Promega) (Iwata et al., 2011). Firefly luciferase activity was normalized to Renilla luciferase activity. Data presented are from five independent biological replicates, each with three technical replicates.
Data presented are the mean values ± SE/SD. Significance difference between multiple samples was estimated by one-way ANOVA followed by Tukey’s test. Means with same letters are not significantly different, P > 0.05. Binary comparisons of data were statistically analyzed by Student’s t-test (P < 0.05 and P < 0.01).
Full length coding sequence of A. officinalis potassium transporter, AoKUP2 was obtained from our earlier transcriptomic study (Krishnamurthy et al., 2017). AoKUP2 cDNA of 2,391 nucleotides in length encodes a polypeptide of 796 amino acids with theoretical pI of 6.74 and molecular weight of 88.32 kDa and its ortholog, the 2,385 bp long AtKUP2 cDNA encodes a polypeptide of 794 amino acids with theoretical pI of 7.14 and molecular weight of 88.63 kDa. Phylogenetic analysis of KUP family derived amino acid sequences revealed that KUP2 from Arabidopsis, Avicennia, and rice are grouped into same clade, which suggests that they are homologous to each other (Figure 1A). Multiple sequence alignment of deduced amino acid sequences of AoKUP2 revealed that it has 78% sequence identity and 87% sequence similarity with AtKUP2. Similarly, with Oryza sativa, both AtKUP2 and AoKUP2 share about 72% sequence identity and 82% sequence similarity (Figure 1B). The number of transmembrane (TM) domains were predicted using TMHMM server, AoKUP2 contains 13 TM helices (Figure 1C) and AtKUP2 consists of 12 TM helices (Figure 1D). Sequences highlighted with black box (GVVYGDLSISPLY) are characteristic of the first transmembrane fragment, which is conserved in all three classes of K+ transporters (AtKUP2, AoKUP2, and OsKUP2).
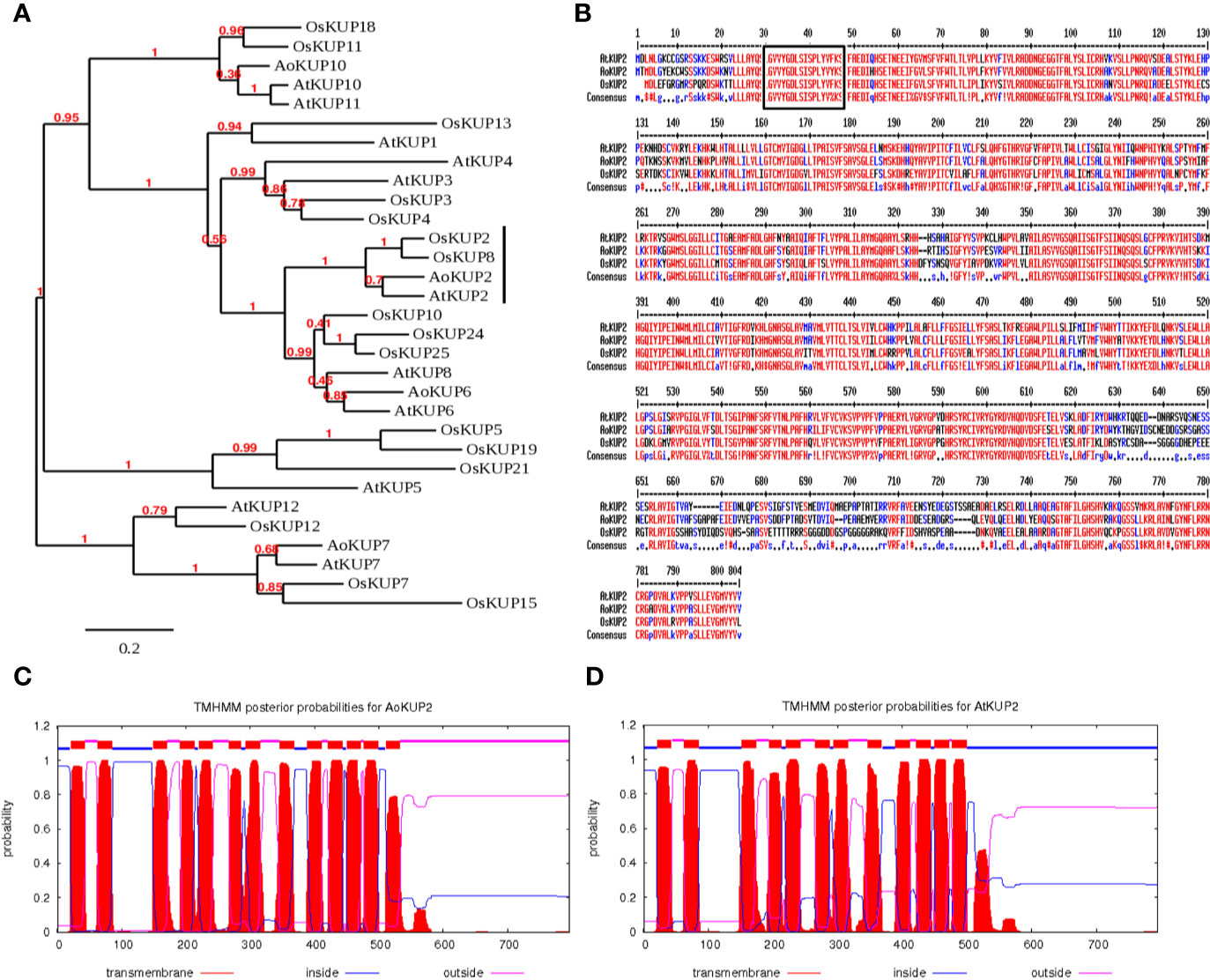
Figure 1 In-silico analysis of AoKUP2 and AtKUP2. (A) A phylogenetic tree of KUP family proteins among A. officinalis, A. thaliana, and O. sativa. The phylogenetic tree was constructed using Phylogeny.fr by bootstrap method. The scale bar indicates the branch length. (B) Sequence alignment of the deduced amino acid sequences of AoKUP2 with Arabidopsis and O. sativa KUPs. The black box depicts the characteristic of the first transmembrane fragment, which is conserved in all three transporters. (C, D) The number of transmembrane domains of AoKUP2 and AtKUP2 were predicted using TMHMM sever 2.0.
To gain insight into the transcriptional responses of AoKUP2 and AtKUP2 genes to NaCl stress, expression levels of AoKUP2 and AtKUP2 were assessed using qRT-PCR after exposure of Avicennia and Arabidopsis seedlings to salinity for varying durations. The transcript level of AoKUP2 was induced (~6-fold) after 3 h in roots, and around 5-fold induction was observed in leaves after 6 h of salt treatment (Figure 2A). There was a decrease in the expression level of AoKUP2 after 12 h of salt treatment in both leaves and roots. In Arabidopsis, we found that AtKUP2 is constitutively expressed in all tissue types with the highest level of expression in flowers and roots (Figure 2B). Similar to AoKUP2, the expression of AtKUP2 was induced (~4-fold) in roots after 6 h of NaCl treatment, and gradual decrease in expression level was observed after 12 h of treatment (Figure 2C). These results suggest that AoKUP2 and AtKUP2 might play an important role in salinity.
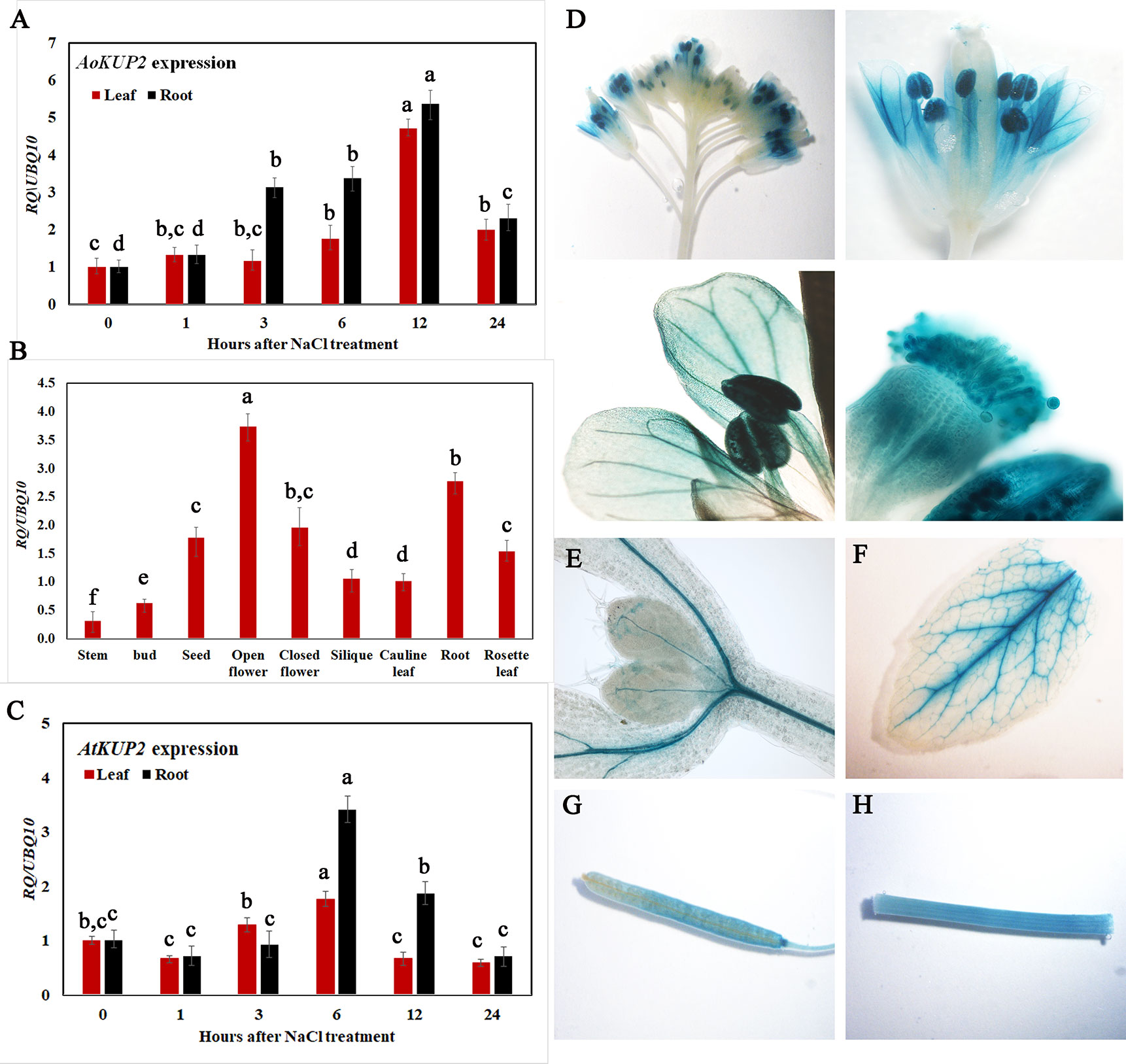
Figure 2 qRT-PCR analysis of Arabidopsis and A. officinalis KUP2 and GUS expression pattern of AtKUP2. (A) Time course analysis of AoKUP2 in 2-month-old A. officinalis leaves and root after salt treatment (B) tissue-specific expression of AtKUP2, (C) Time course analysis of AtKUP2 in 1-week-old Arabidopsis leaves and roots after salt treatment. Relative expression levels of transcripts with reference to Ubiquitin 10 transcript levels are plotted, and qRT-PCR data are mean ± SD from three biological replicates, each with three technical replicates. (D–H) Tissue specific expression of pAtKUP2::GUS in (D) open flower, (E) cotyledon, (F) leaf, (G) silique, and (H) stem. Different letters in A and C indicate statistically significant differences between various time points among leaves and roots, as determined by the ANOVA employing the Tukey-Kramer posthoc test (P < 0.05). Same letters indicate no statistical difference between them. In B, different letters indicate significant differences among various tissues.
Consistent with qRT-PCR results, GUS expression driven by the native promoter of AtKUP2 was also observed in all the plant parts. AtKUP2 promoter-driven GUS expression was mainly seen in leaf and shoot vasculature, flower, stem, silique, and root stele (Figures 2D–H). After salt treatment, no significant difference was observed in the leaves (Figure 3A_I), but in the shoots and roots, an increase was seen in GUS expression surrounding the vasculature (Figure 3A_II and III). Also, the GUS expression in the root tip and lateral root was significantly increased upon salt treatment (Figures 3A_IV and V and C).
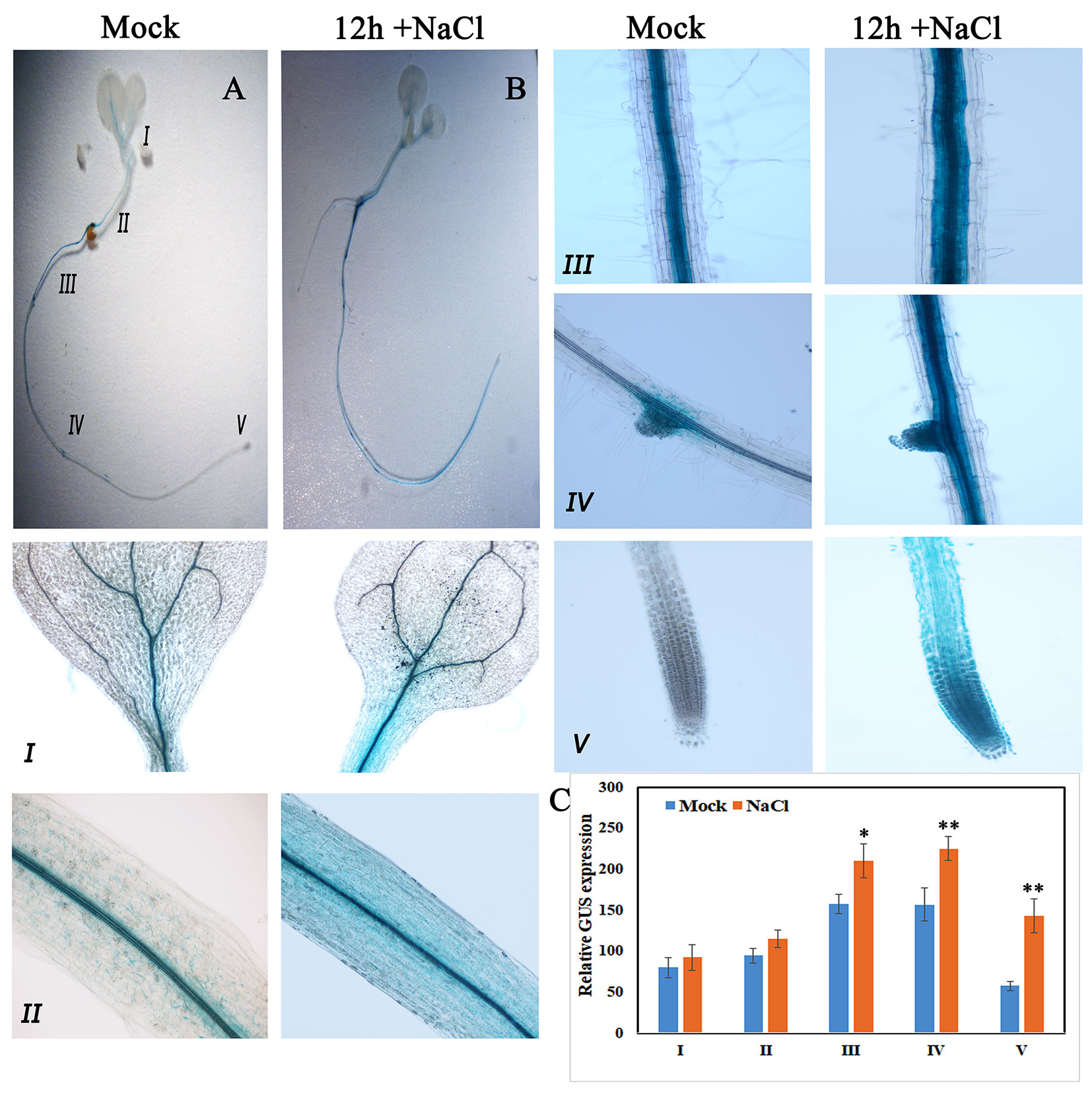
Figure 3 GUS expression pattern of AtKUP2 before and after salt treatment. (A, B) Tissue-specific expression of pAtKUP2::GUS in 1-week-old Arabidopsis seedlings. (I-V) Close-up images of (A, B) [relative positions labeled in the image (A)] before and after 50 mM NaCl treatment for 12 h. (C) Relative quantification of GUS intensity in I-V before (mock) and after NaCl treatment. Data are mean ± SE of three biological replicates, each biological replicate consisting of at least six plants. Asterisks indicate statistically significant differences (*P< 0.05 and **P< 0.01) as measured by Student’s t-test between mock and the treated.
Although KUP2 transporter is known to be involved in transmembrane transport of K+ ions, there is no experimental evidence for its localization in the plant plasma membrane. In order to confirm the localization, AoKUP2 and AtKUP2 cDNAs were cloned into both C-terminal and N-terminal GFP tagged vectors and transiently expressed in Nicotiana benthamiana leaf epidermal cells along with PM marker. Both C-terminal and N-terminal tagged fusion proteins showed subcellular distribution pattern of AoKUP2 and AtKUP2 to the plasma membrane (Figures 4A, D). In addition, localization of AoKUP2 and AtKUP2 was also observed in the mitochondria (Figures 4B, C, E). The arrows (Figures 4B, C, E) indicating yellow merged signals show the colocalization of AoKUP2 and AtKUP2 with the mitochondrial marker.
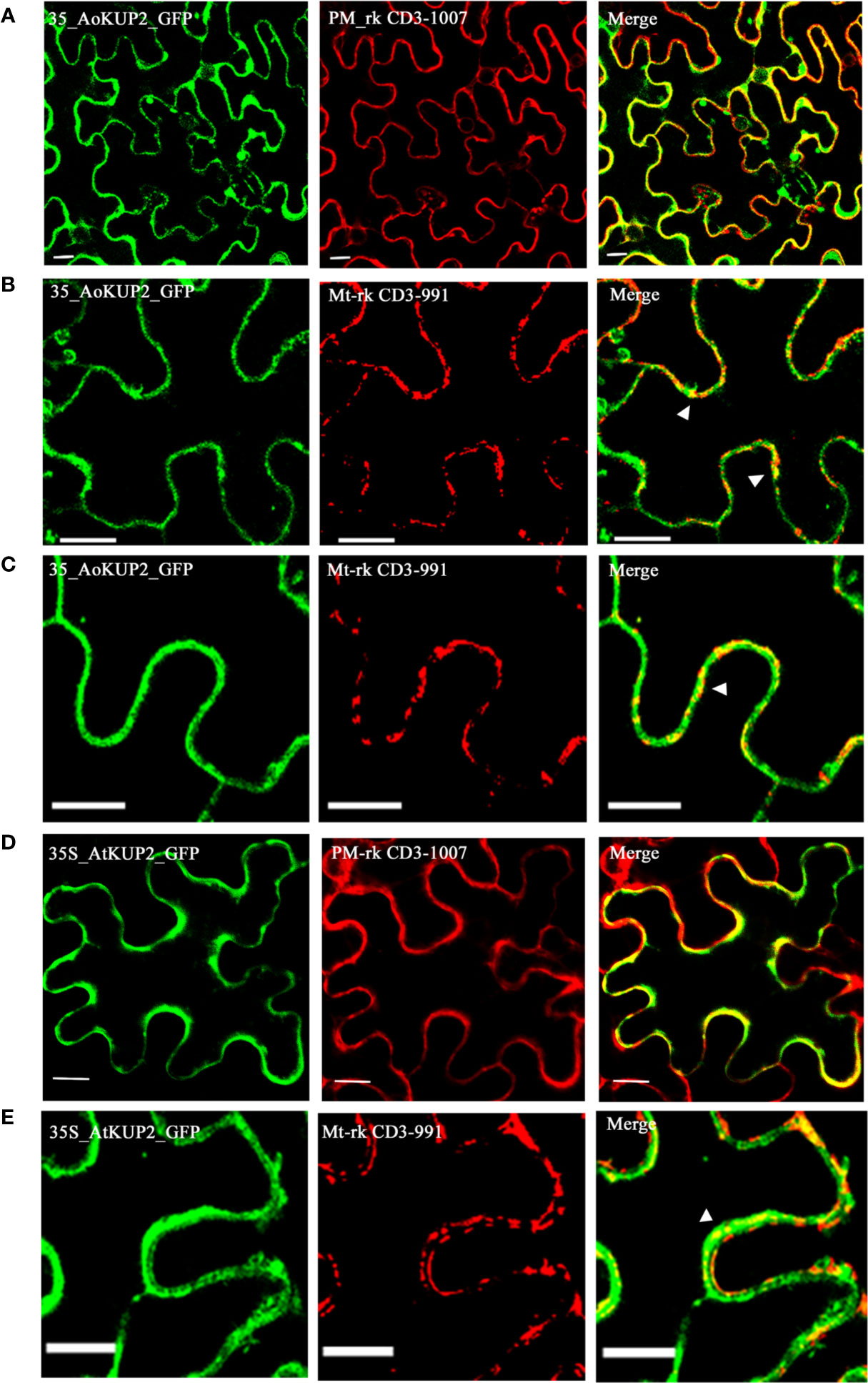
Figure 4 Subcellular localization of AoKUP2 and AtKUP2. Confocal microscopy images of Nicotiana benthamiana leaf epidermal cells (A) Expression of 35S::AoKUP2:GFP along with PM marker (PM-rk CD3-1007) and (B, C) mitochondria marker (mt-rk CD3-991). (D) Expression of 35S::AtKUP2:GFP along with PM marker (E) and mitochondria marker. The expression of 35S::AoKUP2:GFP and 35S::AtKUP2:GFP were visualized with λ = 488 nm (green) while the expression of PM and mitochondria markers were visualized with λ = 561 nm (Red). Co-localization of 35S::AoKUP2:GFP and 35S::AtKUP2:GFP with PM and mitochondria are shown in the merged images. Scale bar = 15 µm.
In order to carry out functional characterization of AtKUP2 and AoKUP2 transporters, we introduced AtKUP2 and AoKUP2 genes into S. cerevisiae deletion mutants lacking plasma membrane TRK potassium transport system, BYT12 (BY4741 trk1Δ::loxP trk2Δ::loxP). The deletion mutant lacking TRK genes and the transgenic yeasts harboring AtKUP2 and AoKUP2 did not grow on SD-Ura, while growth of WT (BY4741) harboring YEp352 vector was not affected. Therefore, they were grown on medium with different concentrations of K+ (10 mM to 1 M). All the yeast strains grew well on 100 mM KCl but the mutants could not grow well under low K+ (20 and 50 mM) concentrations (Figures 5A, C–E). In addition, when KCl concentration was increased to 1 M, growth of the mutant was inhibited but transgenic yeasts harboring AtKUP2 and AoKUP2 were not affected (Figures 5B, F). Similarly, the mutant displayed sensitivity to NaCl (200 and 500 mM) treatment. But this NaCl sensitivity was rescued in the mutant yeast expressing AtKUP2 and AoKUP2 (Figures 5B, G, H). These results suggest that KUP2 is involved in K+ uptake and might help in maintaining intracellular K+/Na+ homeostasis which is essential for survival under salt stress.
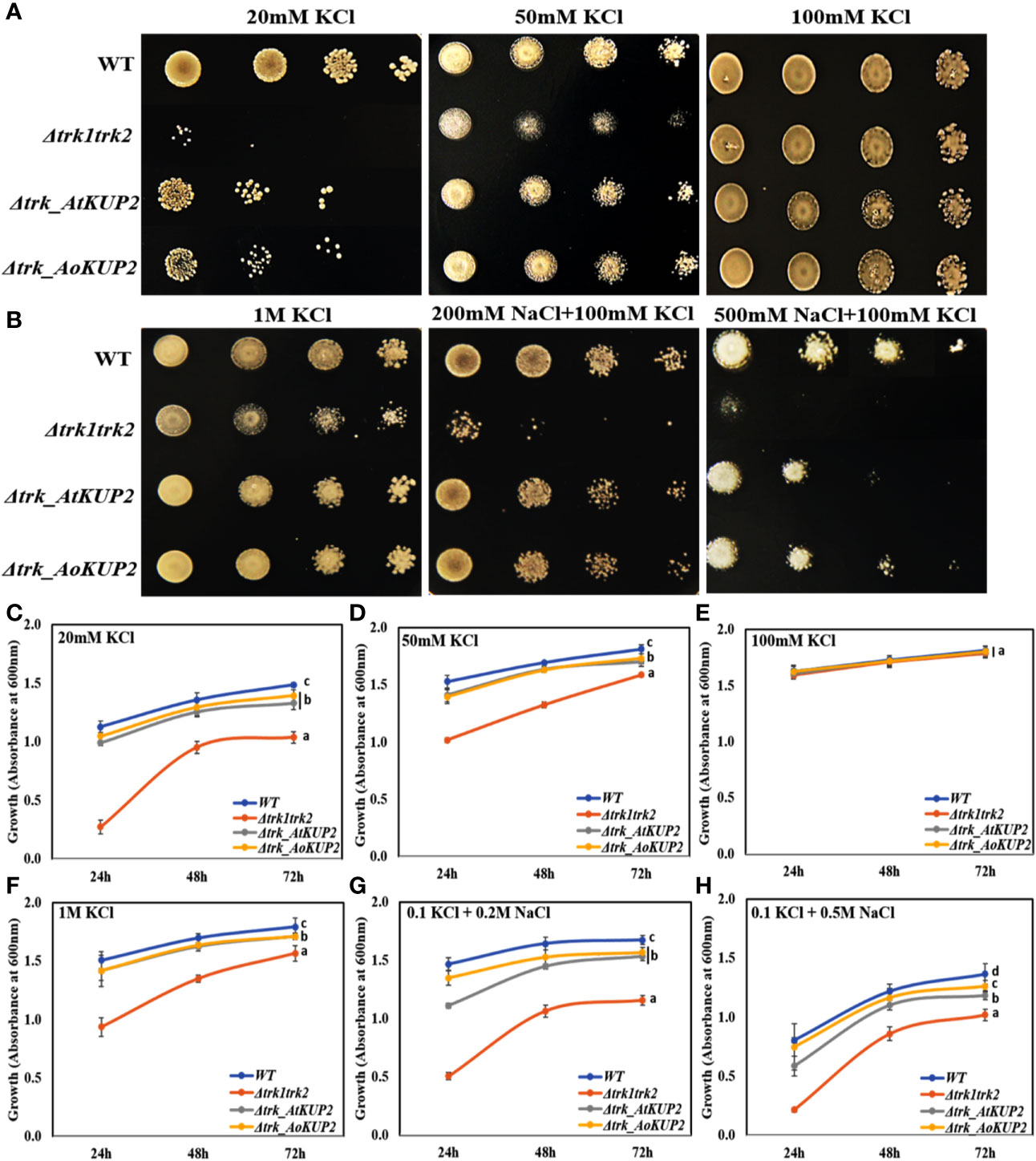
Figure 5 AtKUP2 and AoKUP2 functionally complement the K+ and Na+ sensitivity of yeast mutant strains lacking the TRK1 and TRK2 genes. Growth of BY4741 strain, its derivative BYT12 (trk1Δ::loxP trk2Δ::loxP) harboring AtKUP2 and AoKUP2 in YEp352 plasmid was tested on SD-Ura medium with (A) (10–100 mM KCl) and (B) with various salts (1 M KCl, 200 mM NaCl with 100 mM KCl, and 500 mM NaCl with 100 mM KCl). (C–H) The density of cells containing Δtrk1trk2 mutant transformed with AtKUP2 and AoKUP2 and WT cells in liquid SD-Ura with various salts (OD600) was monitored. Data are mean ± SE of three independent experiments. Means with same letters (shown only for the 72h time point) are not significantly different, P > 0.05 (one-way ANOVA followed by Tukey’s test).
In order to investigate whether ectopic expression of AoKUP2 and AtKUP2 in Arabidopsis enhances salinity tolerance, WT, mutant, 35S::AoKUP2 and 35S::AtKUP2 Arabidopsis seedlings were treated with NaCl during both seed germination and vegetative growth stages. Under untreated condition, there was no significant difference in the germination rate of atkup2 and ectopic expression lines compared to WT. But, cotyledon expansion in atkup2 was severely inhibited by NaCl treatment, while 35S::AtKUP2 and 35S::AoKUP2 lines showed reduced sensitivity to NaCl stress (Figures 6A–C). Around 20–30% decrease in germination rate was observed in the mutant compared to the ectopic expression lines. Similarly, we also examined the root growth in 35S::AtKUP2 and 35S::AoKUP2 lines under salt treatment. Under untreated condition, there was no growth difference between WT, mutant and ectopic expression seedlings. Significant reduction in root length was noted in the mutant and WT seedlings upon 50 mM (Supplementary Figure S2) and 75 mM (Figures 6D, E) NaCl treatment compared to the ectopic expression lines. 35S::AtKUP2 and 35S::AoKUP2 lines showed about 2.5-fold increase in root length compared to the mutant and ˜1.5-fold increase in root length compared to the WT under 75 mM NaCl treatment (Figure 6F).
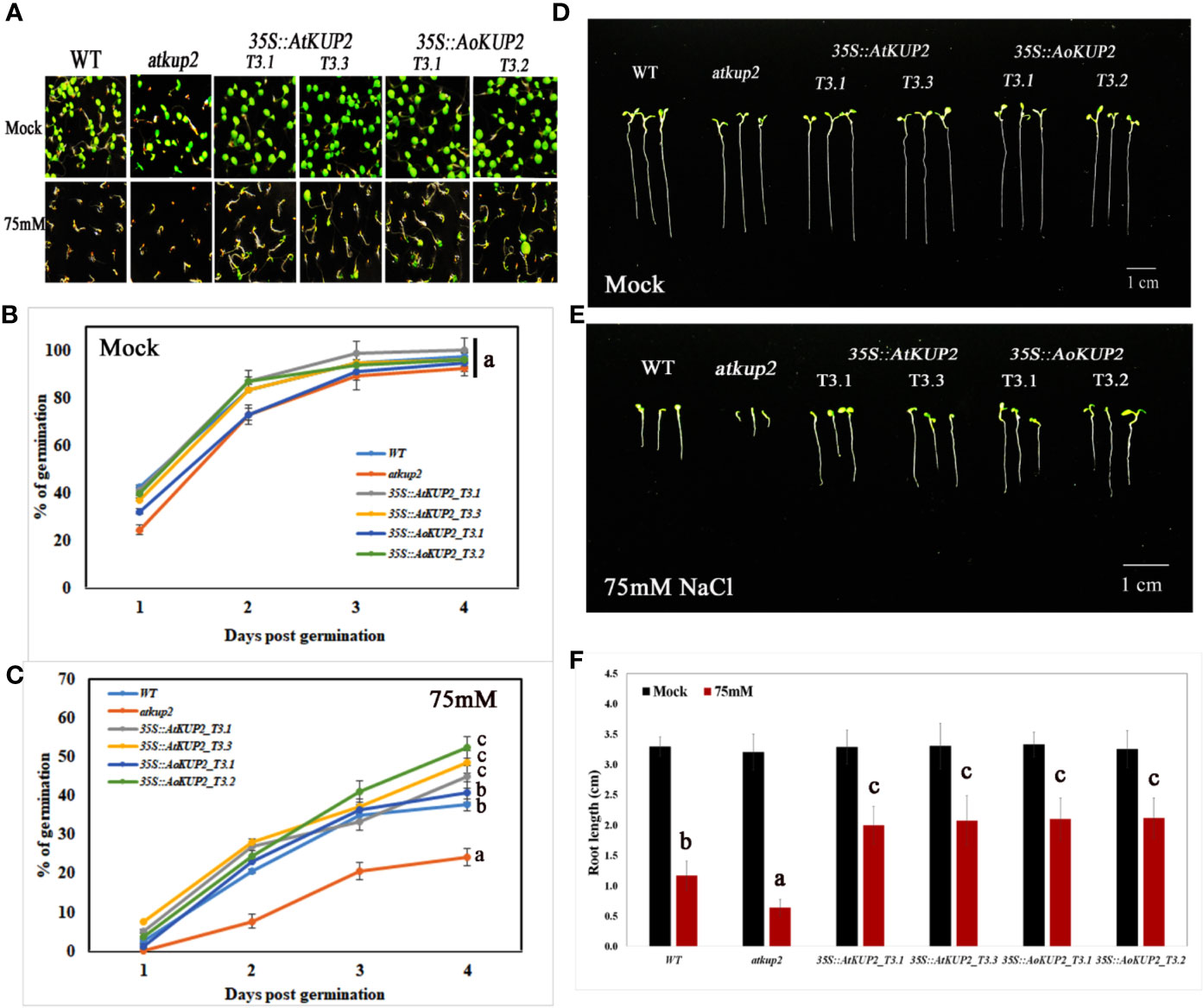
Figure 6 Ectopic expression lines showed reduced sensitivity to NaCl compared to WT and atkup2. Germination rate was compared between WT, atkup2 mutant, and ectopically expressed in WT background (two independent lines each for 35S::AtKUP2 and 35S::AoKUP2). (A–C) Germination assay of WT, atkup2, 35S::AtKUP2, and 35S::AoKUP2 lines. The number of germinated seeds were counted from day 1 to 4 and the photographs were taken after 1 week of germination. (D–F) Comparison of root length among WT, atkup2, 35S::AtKUP2, and 35S::AoKUP2 lines with and without NaCl (75 mM) treatment. Root length measurements and photographs were taken after 1 week of germination. Data are mean ± SE of three independent experiments, each with at least 15 replicates per experiment. Means with same letters are not significantly different, P > 0.05 (one-way ANOVA followed by Tukey’s test). Scale bar = 10 mm.
Salinity tolerance in mature plants grown in the soil was also investigated. One-month-old WT, mutant and ectopic expression Arabidopsis plants were treated with 150 mM NaCl for 1 week. WT and mutant plants showed stunted phenotypes with severe chlorosis and eventually died, but the 35S::AtKUP2 and 35S::AoKUP2 plants appeared greener and healthier than WT and mutant (Figures 7A–C). Similarly, shoot length assay was also performed under salt treatment and analyzed after recovery. Under salt treatment, WT and mutant displayed reduced growth compared to ectopic expression lines. After 1 week of recovering with normal watering, the WT and mutant could not recover completely, and exhibited short siliques with poor seed set (Figures 7D–F). But 35S::AtKUP2 and 35S::AoKUP2 lines showed better performance of about 1.5-fold increase in shoot length compared to that of the mutant (Figure 7F). These results clearly indicate that 35S::AtKUP2 and 35S::AoKUP2 ectopic expression lines increased the salt tolerance of Arabidopsis.
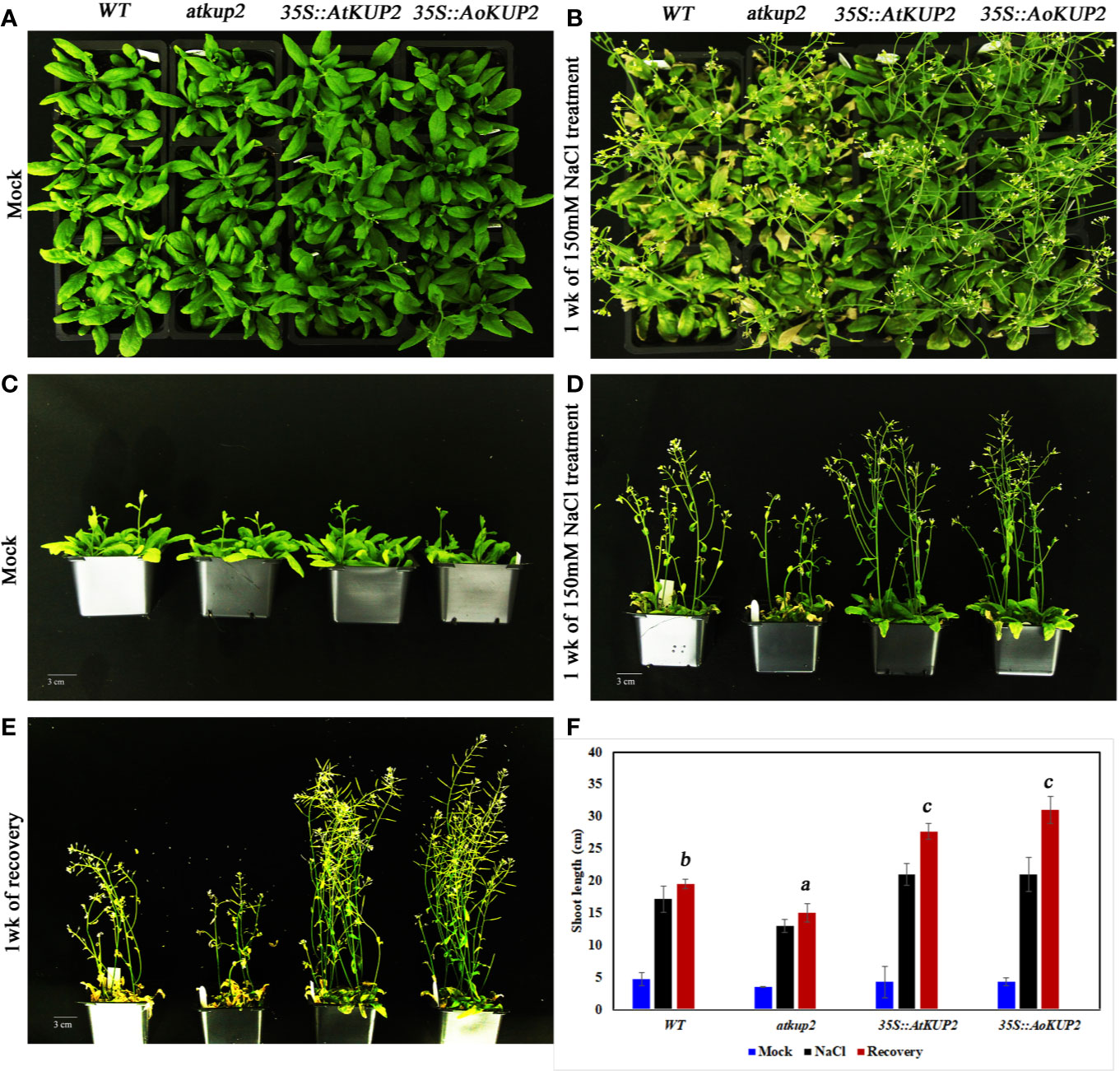
Figure 7 Ectopic expression of AtKUP2 and AoKUP2 increases salt tolerance of Arabidopsis plants. (A–E) Growth response to salt (150 mM NaCl) was monitored in 1-month-old WT, atkup2, and ectopic expression lines (35S::AtKUP2 and 35S::AoKUP2) grown on soil. Growth of WT, atkup2, 35S::AtKUP2, and 35S::AoKUP2 lines under (A, C) untreated and (B, D) salt treated conditions, (E) growth of WT, atkup2, and 35S::AtKUP2 and 35S::AoKUP2 lines after recovery growth in normal water for 1 week, and (F) shoot length analysis of WT, atkup2 and 35S::AtKUP2 and 35S::AoKUP2 under treated and untreated conditions. Scale bar = 30 mm. Data are mean ± SE of three biological replicates, each with at least three plants. Means with the same letter within a data set are not significantly different, P > 0.05 (one-way ANOVA followed by Tukey’s test). Scale bar = 30 mm.
In order to examine the K+/Na+ ratio in plants, we analysed the ion content of WT, mutant, 35S::AoKUP2 and 35S::AtKUP2 plants before and after exposure to NaCl stress. Without NaCl stress, Na+ contents were similar in WT, atkup2 and ectopic expression lines. Upon NaCl treatment, Na+ levels increased in all the plants, but the shoot Na+ content in the ectopic expression lines was significantly lower (~28 mg/g DW) compared to the WT (~46 mg/g DW) and mutant (~53 mg/g DW) lines (Figure 8A). Without NaCl treatment, the K+ content did not vary in the shoots of WT, mutant and ectopic expression lines but varied in the roots. Although K+ levels decreased upon NaCl treatment in all the plants, the extent of decrease was significantly lower in the shoots and roots of ectopic expression lines compared to that of the WT and mutant (Figure 8B). The K+/Na+ ratio decreased with NaCl treatment in both roots and shoots (Figure 8C). However, ectopic expression lines maintained a significantly higher K+/Na+ ratio than in the WT (~2.5-fold)) and mutant (~3.5-fold) under salt-stressed conditions.
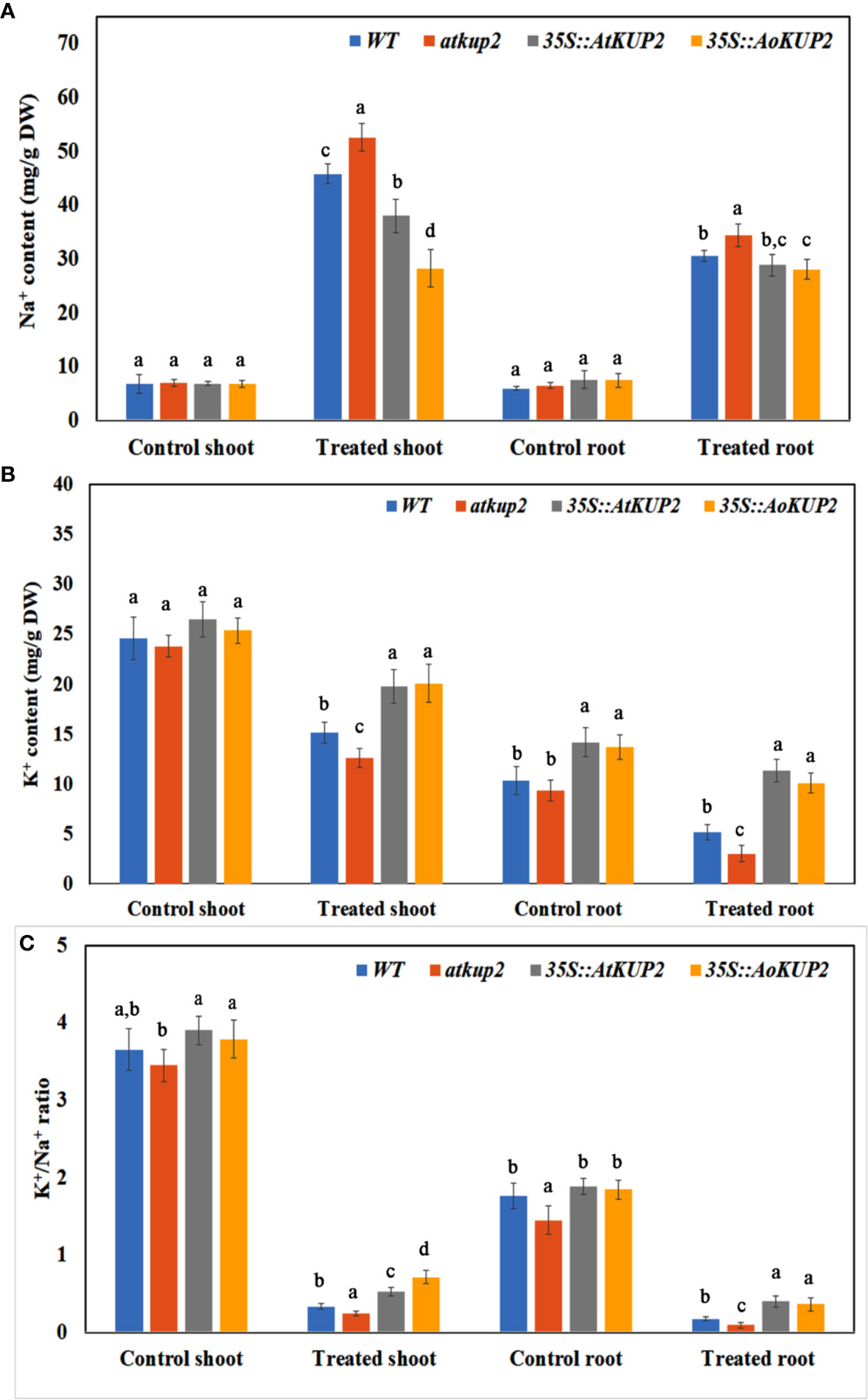
Figure 8 Ectopic expression lines accumulated more K+ than WT under NaCl stress. The Na+ and K+ content in the shoots and leaves of WT, atkup2, 35S::AtKUP2, and 35S::AoKUP2 lines before and after NaCl treatment. (A) Na+ content of the shoot and root (B) K+ content of the shoot and root (C) K+ to Na+ ratio of the shoot and root. Data are mean ± SE of three biological replicates, each with at least three plants. Means with same letter within a data set are not significantly different, P > 0.05 (one-way ANOVA followed by Tukey’s test).
We also investigated the effect of NaCl on lateral root growth and membrane integrity by treating WT, atkup2 and 35S::AtKUP2 with 50 mM NaCl. Better lateral root growth was observed in 35S::AtKUP2 than WT and atkup2 under both untreated and treated conditions (Supplementary Figure S3A). In untreated condition, no membrane damage was found in WT and 35S::AtKUP2. But after treatment, the proportion of cell death in the roots gradually increased and comparatively more cell death was observed in atkup2 and WT than 35S::AtKUP2 (Supplementary Figure S3B). This suggests that AtKUP2 might help in preventing general cell damage caused by salinity stress.
Promoter analysis of AtKUP2 revealed various cis-regulatory elements in the 5’ upstream region. The putative binding motifs in the upstream region included cis-elements for bHLH and WRKY TFs (Figure 9A). By coincidence, in a previous RNA-Seq analysis, bHLH and WRKY were among the major groups of TFs co-induced upon salt treatment in A. officinalis roots (Krishnamurthy et al., 2019). Since it is already reported that bHLH and WRKY are regulated by salt stress, we checked the expression of AtKUP2 in atbhlh122 and atwrky33 T-DNA insertional mutants. In atwrky33, the expression of AtKUP2 was suppressed by about 80% and in atbhlh122, the suppression level was even higher (~90%) compared to WT (Figure 9B). We then carried out ChIP assay to investigate the interaction between AtKUP2 and bHLH and WRKY transcription factors using Arabidopsis protoplasts. AtWRKY33-HA pulldown samples displayed ~3-fold enrichment and AtbHLH122-HA pull down samples displayed ~4-fold enrichment of AtKUP2 promoter fragment (Figure 9C) compared to the vector controls. In addition, we carried out luciferase assay using atwrky33 and atbhlh122 protoplasts transfected with 35S::AtWRKY33 and 35S::AtbHLH122, respectively, together with pAtKUP2::LUC in order to check whether AtWRKY33 and AtbHLH122 can activate the expression of AtKUP2. atwrky33 protoplast transfected with AtWRKY33 showed ~3.6-fold higher luciferase activity compared to the control (Figure 9D). Similarly, atbhlh122 protoplasts transfected with AtbHLH122 showed ~3.4-fold higher luciferase activity compared to the control (Figure 9E). We also checked whether both TFs have additive effect on AtKUP2 expression by co-transfecting both 35S::AtWRKY33 and 35S::AtbHLH122 with pAtKUP2::LUC in atwrky33 mutant (Figure 9D). It showed ~5.4-fold higher activity compared to pAtKUP2::LUC. These results clearly indicate that AtbHLH122 and AtWRKY33 TFs act together as upstream regulators of AtKUP2.
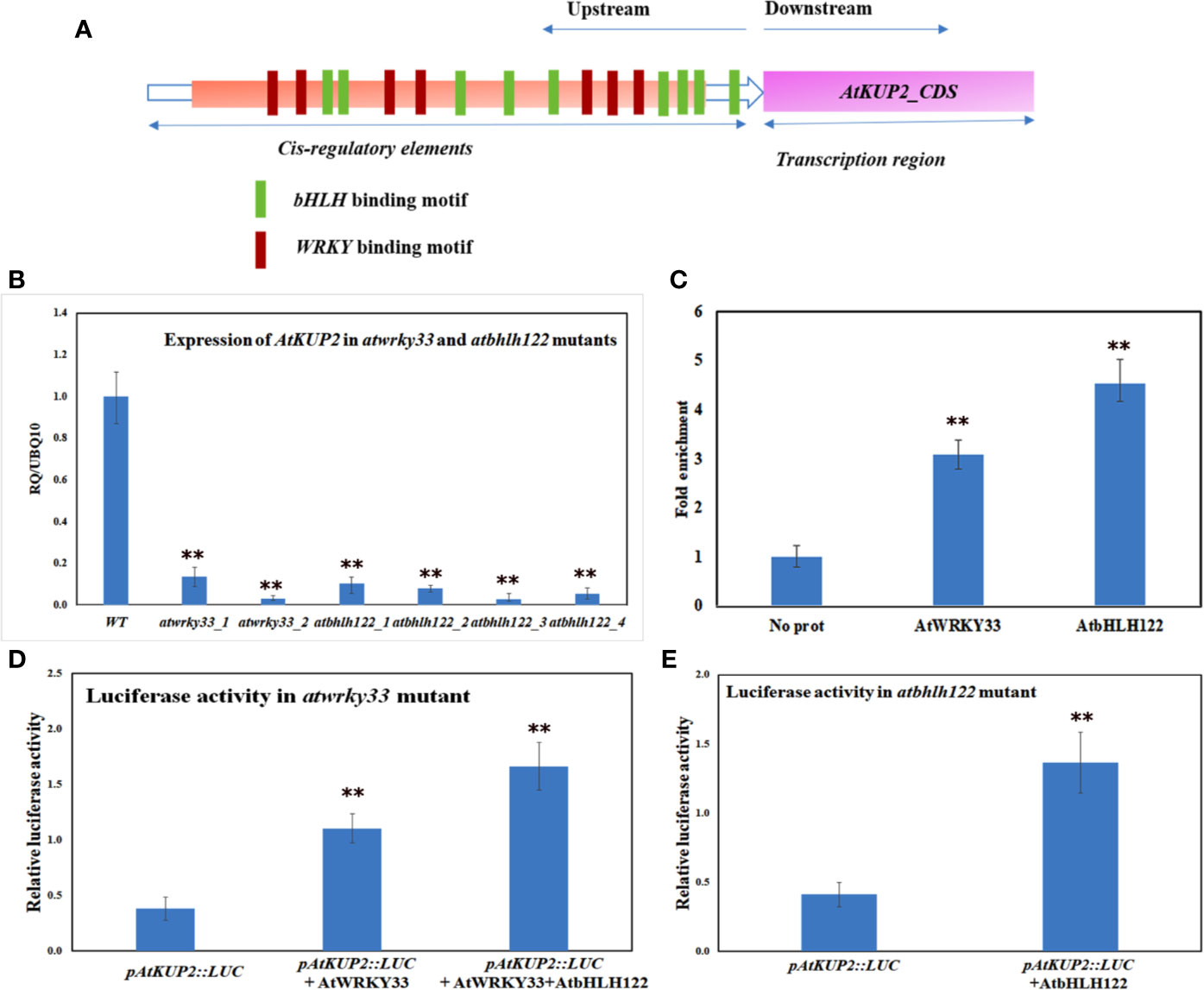
Figure 9 AtKUP2 is regulated by transcription factors, AtbHLH122 and AtWRKY33 (A) Schematic representation of the presence of bHLH and WRKY transcription factors binding motifs in the upstream region of AtKUP2. (B) Suppression in the transcript levels of AtKUP2 in the atbhlh122 and atwrky33 T-DNA insertional mutants compared to WT. (C) ChIP-qPCR analysis shows enrichment of AtKUP2 promoter fragment in AtWRKY33-HA and AtbHLH122-HA ChIP samples. (D) Luciferase assay using the mesophyll protoplasts obtained from the leaves of atwrky33 mutants. (E) Luciferase assay using atbhlh122 mutants. pAtKUP2::LUC was used as the reporter and 35S::AtWRKY33 and 35S::AtbHLH122 was used as the effector. Firefly luciferase activity was normalized to Renilla luciferase activity and plotted. Data are mean ± SD of five independent biological replicates, each with three technical replicates. Asterisks indicate statistically significant differences (**P < 0.01) as measured by Student’s t-test between WT and the mutants in B and between control and test in (C, E).
In plants, ionic and osmotic stresses are the two major stresses imposed by soil salinity. Ion transport is a critical step, which controls the ion homeostasis under such stress conditions (Volkov and Beilby, 2017). High affinity KUP/HAK/KT transporters belong to a large K+ transporter gene family and have been identified in several crop species including Arabidopsis, rice and maize (Gupta et al., 2008). Based on our knowledge, KUPs have not been characterized in any of the mangrove species and their heterologous expression has not yet been studied. Mangroves possess efficient salt tolerance mechanisms in order to grow under extreme saline conditions (Kodikara et al., 2018). Therefore, it is possible that their KUPs could function more effectively under salt stress than the corresponding proteins from the non-halophytes. In the present study, we identified a specific KUP2 from A. officinalis that was differentially expressed upon salt treatment (Figure 2A). High sequence similarity of AoKUP2 with Arabidopsis, O. sativa and other plant species show that it may have a similar function in A. officinalis. The expression of AtKUP2 in different tissues of Arabidopsis indicated by qRT-PCR (Figure 2B) and transcriptional GUS-marker (Figures 2D–J) reveals its ubiquitous presence in plants, with higher level of expression in roots and flowers, which is similar to the tissue-specific expression data of HAKs in O. sativa (Su et al., 2002; Yang et al., 2014; Chen et al., 2015). The increased expression of AtKUP2 in the root tissues rather than leaves under salt stress suggests that it may have an important role under such conditions and is similar to the expression pattern of OsHAK1 under potassium stress conditions (Chen et al., 2015). Increase in AtKUP2 expression specifically in the younger parts of the roots, and the tissues surrounding root vasculature (Figures 3A, B) further suggests its role in regulating influx/efflux of K+ ions in order to maintain the intracellular ion homeostasis as reported in earlier studies (Ketehouli et al., 2019; Ragel et al., 2019). There was a significant upregulation of AoKUP2 in both leaves and roots under salt stress suggesting that KUP2 might be involved in transport of K+ in both leaves and roots under salt stress. Lateral root growth and root elongation are highly affected by limitation of K+ (Jung et al., 2009; Kellermeier et al., 2013). The importance of Arabidopsis KUP6 in lateral root initiation and development in ABA and auxin signalling pathways is reported (Osakabe et al., 2013). Our observation of enhanced GUS expression in LRP and better lateral root growth in 35S::AtKUP2 in both treated and untreated conditions (Supplementary Figure S3A) imply that AtKUP2 has some role in lateral root growth. For better understanding of the underlying mechanism, detailed studies need to be carried out. In addition, localization of AoKUP2 and AtKUP2 in plasma membrane (Figure 4) could be required for increased transport of K+ across the root plasma membrane in order to maintain intracellular K+/Na+ ratio under salt stressed conditions. The reduced cell damage in the roots of 35S::AtKUP2 compared to WT and atkup2 under salt treatment (Supplementary Figure S3B) further confirms that AtKUP2 might help in preserving the membrane integrity, which is important for survival under salt stress by maintaining a high K+/Na+ ratio. Also, mitochondrial localization of AoKUP2 and AtKUP2 suggests that they might be involved in ROS cell signalling process during oxidative stress as reported previously for durum wheat mitochondrial K+ channel (PmitoKATP) (Pastore et al., 1999; Pastore et al., 2013). Hence, further experiments may help to reveal if these KUPs play any role in the ROS pathway besides salinity tolerance mechanism.
Most of the plant KUP/HAK/KT transporters function in high‐affinity K+ transport, although some members from this family were shown to function as low-affinity K+ transporters. For instance, KUP1 from Arabidopsis mediates K+ uptake at both low and high external supply of K+ in Arabidopsis‐suspension cells and yeast (Ahn et al., 2004; Ketehouli et al., 2019). In the current study, we show that the expression of AtKUP2 and AoKUP2 in the mutant yeast strain, trk1Δtrk2Δ could rescue the growth defect at both low (50 mM) and high external (1 M) K+ conditions. Similarly, expression of OsHAK2, OsHAK7 and HvHAK2 rescued the sensitive phenotype of mutant E. coli strain with the defect of K+ uptake under low K+ supply (Mangano et al., 2008; Alemán et al., 2009; Nieves-Cordones et al., 2010; Horie et al., 2011). Our results show that the growth suppression of the yeast mutants in the 500 mM NaCl with 100 mM KCl medium could be rescued by the introduction of AtKUP2 and AoKUP2 (Figure 5) suggests that KUP2 mediates K+ transport (and could regulate K+ and Na+ homeostasis) under NaCl stress in yeast cells. We can speculate that it may function in a similar manner in the plants. Several important Na+ transporters such as NHXs, SOS1 and HKTs found in glycophytes as well as in some halophytes are known to mediate salt resistance in plants (Horie et al., 2009; Yang et al., 2009; Mishra et al., 2014; Hamamoto et al., 2015; Ma et al., 2019; Al-Harrasi et al., 2020). Ectopic expression of some of these Na+ transporters in specific targeted tissues or in whole plants have been reported to increase the salinity tolerance of plants (Yang et al., 2009; Li et al., 2011; Mian et al., 2011; Gao et al., 2012; Huang et al., 2018; Zhang et al., 2019; Al-Harrasi et al., 2020), implying that improvement in detoxification mechanisms of Na+ possibly is a typical approach to generate salt tolerant crops. In addition, maintenance of high K+/Na+ ratio in shoots as wells as roots is essential for salinity tolerance. High-affinity K+ uptake was proved to be essential for salinity tolerance in plants (Shabala and Cuin, 2008; Almeida et al., 2017; Wu et al., 2018). The significant increase in salt tolerance of 35S::AtKUP2 and 35S::AoKUP2 lines and increase in shoot and root K+/Na+ ratio of 35S::AtKUP2 and 35S::AoKUP2 compared to WT and mutant upon salt stress suggests that both AtKUP2 and AoKUP2 mediate K+ transport and accumulation during salt stress, which might help in maintaining turgor pressure or membrane potential leading to better survival of plants.
Our computational analysis of AtKUP2 sequence indicated the presence of various putative TF binding sites recognized as abiotic stress-responsive elements within the promoter region. Amongst these TF binding sites, bHLH and WRKY are abundantly distributed within the promoter region of AtKUP2 (Figure 9A). Our previous study showed that upon salt treatment, the expression of AtNHX1 and AtNHX6 are regulated by bHLH TFs, AtbHLH122 and AtMYC2 (Krishnamurthy et al., 2019). Another study reported that overexpressing Gossypium hirsutum WRKY34 in Arabidopsis increased the salt tolerance by developing the plant’s ability for the selective uptake of K+ and Na+ and maintain high K+/Na+ ratio in leaves and roots of transgenic plants (Zhou et al., 2015). The suppression of AtKUP2 expression in atbhlh122 and atwrky33 mutants coupled with the enrichment of AtKUP2 promoter fragments in ChIP and enhancement of AtKUP2 promoter-driven luciferase expression (Figures 9B, C) collectively show that AtWRKY33 and AtbHLH122 act as the upstream regulators of AtKUP2 under salt stress. The regulatory link between AtKUP2 and the two TFs identified in this study helps to explain a part of the molecular mechanism of action for these different players. Thus, bHLH122 was reported to improve stress tolerance in Arabidopsis by reducing the activity of ROS and enhancing the levels of proline (Liu et al., 2014; Liu et al., 2015; Wang et al., 2018; Krishnamurthy et al., 2019). Overexpression of WRKY33 was also shown to improve salt stress tolerance in Arabidopsis (Bao et al., 2018). WRKY33 is not only salt-responsive but also is regulated by oxidative stress (Jiang and Deyholos, 2009; Birkenbihl et al., 2012). Furthermore, WRKY33 regulated genes are found to be associated with ROS detoxification mechanisms (Jiang and Deyholos, 2009) suggesting role of WRKY TFs as important regulators in various stress adaptation. Our observations helped to identify the TFs as necessary for the salt-mediated upregulation of AtKUP2. So far, there have not been any reports identifying TFs that regulate the expression of AtKUP2. Another question that remains to be clarified with further experiments is the nature of interaction between bHLH122 and WRKY33 transcription factors in order to act as a molecular regulator of gene expression.
In conclusion, our data suggest that KUP2 from both Avicennia and Arabidopsis are induced by salt stress, and ectopic expression of both AoKUP2 and AtKUP2 plays an important role in salt remediation of Arabidopsis. Our results with heterologous expression of AtKUP2 in selected yeast strains show that it complements the function of plasma membrane K+ transport and in Arabidopsis helps to maintain higher K+/Na+ ratio. In addition, using ChIP and luciferase, we show that transcriptional regulation of AtKUP2 occurs by AtWRKY33 and AtbHLH122. In future, studies on KUP2 need to be carried out in crop plants such as rice and wheat in order to produce salt tolerant crop plants that will help to address food security despite the increasing soil salinization occurring globally.
All datasets presented in this study are included in the article/Supplementary Material.
SR, PK, and PPK conceived the research plans. SR and PK designed the experiments. SR carried out all the experiments, analyzed the data, and wrote the article with contributions from all the authors.
This research grant was supported by a PhD research scholarship to SR from the National University of Singapore.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors thank Dr. Hana Sychrová for providing the yeast strains used in this study.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2020.01311/full#supplementary-material
Adams, E., Shin, R. (2014). Transport, signaling, and homeostasis of potassium and sodium in plants. J. Integr. Plant Biol. 56, 231–249. doi: 10.1111/jipb.12159
Ahn, S. J., Shin, R., Schachtman, D. P. (2004). Expression of KT/KUP genes in Arabidopsis and the role of root hairs in K+ uptake. Plant Physiol. 134, 1135–1145. doi: 10.1104/pp.103.034660
Alemán, F., Nieves-Cordones, M., Martínez, V., Rubio, F. (2009). Differential regulation of the HAK5 genes encoding the high-affinity K+ transporters of Thellungiella halophila and Arabidopsis thaliana. Environ. Exp. Bot. 65, 263–269. doi: 10.1016/j.envexpbot.2008.09.011
Al-Harrasi, I., Jana, G. A., Patankar, H. V., Al-Yahyai, R., Rajappa, S., Kumar, P. P., et al. (2020). A novel tonoplast Na+/H+ antiporter gene from date palm (PdNHX6) confers enhanced salt tolerance response in Arabidopsis. Plant Cell Rep. 39, 1079–1093. doi: 10.1007/s00299-020-02549-5
Almeida, D. M., Margarida Oliveira, M., Saibo, N. J. M. (2017). Regulation of Na+ and K+ homeostasis in plants: Towards improved salt stress tolerance in crop plants. Genet. Mol. Biol. 40, 326–345. doi: 10.1590/1678-4685-gmb-2016-0106
Alonso, J. M., Stepanova, A. N., Leisse, T. J., Kim, C. J., Chen, H., Shinn, P., et al. (2003). Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301, 653–657. doi: 10.1126/science.1086391
Assaha, D. V. M., Ueda, A., Saneoka, H., Al-Yahyai, R., Yaish, M. W. (2017). The role of Na+ and K+ transporters in salt stress adaptation in glycophytes. Front. Physiol. 8:509. doi: 10.3389/fphys.2017.00509
Bao, W., Wang, X., Chen, M., Chai, T., Wang, H. (2018). A WRKY transcription factor, PcWRKY33, from Polygonum cuspidatum reduces salt tolerance in transgenic Arabidopsis thaliana. Plant Cell Rep. 37, 1033–1048. doi: 10.1007/s00299-018-2289-2
Birkenbihl, R. P., Diezel, C., Somssich, I. E. (2012). Arabidopsis WRKY33 is a key transcriptional regulator of hormonal and metabolic responses toward Botrytis cinerea infection. Plant Physiol. 159, 266–285. doi: 10.1104/pp.111.192641
Chen, G., Hu, Q., Luo, L., Yang, T., Zhang, S., Hu, Y., et al. (2015). Rice potassium transporter OsHAK1 is essential for maintaining potassium-mediated growth and functions in salt tolerance over low and high potassium concentration ranges. Plant Cell Environ. 38, 2747–2765. doi: 10.1111/pce.12585
Clough, S. J., Bent, A. F. (1998). Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743. doi: 10.1046/j.1365-313X.1998.00343.x
Daras, G., Rigas, S., Tsitsekian, D., Iacovides, T. A., Hatzopoulos, P. (2015). Potassium transporter TRH1 subunits assemble regulating root-hair elongation autonomously from the cell fate determination pathway. Plant Sci. 231, 131–137. doi: 10.1016/j.plantsci.2014.11.017
Davies, C., Shin, R., Liu, W., Thomas, M. R., Schachtman, D. P. (2006). Transporters expressed during grape berry (Vitis vinifera L.) development are associated with an increase in berry size and berry potassium accumulation. J. Exp. Bot. 57, 3209–3216. doi: 10.1093/jxb/erl091
Elumalai, R. P., Nagpal, P., Reed, J. W. (2002). A mutation in the Arabidopsis KT2/KUP2 potassium transporter gene affects shoot cell expansion. Plant Cell 14, 119–131. doi: 10.1105/TPC.010322
Epstein, W., Kim, B. S. (1971). Potassium transport loci in Escherichia coli K-12. J. Bacteriol. 108, 639–644. doi: 10.1128/JB.108.2.639-644.1971
Fu, H. H., Luan, S. (1998). AtKUP1: A dual-affinity K+ transporter from Arabidopsis. Plant Cell 10, 63–73. doi: 10.1105/tpc.10.1.63
Gao, S., Yuan, L., Zhai, H., Liu, C. L., He, S. Z., Liu, Q. C. (2012). Overexpression of SOS genes enhanced salt tolerance in sweetpotato. J. Integr. Agric. 11, 378–386. doi: 10.1016/S2095-3119(12)60022-7
Gierth, M., Mäser, P. (2007). Potassium transporters in plants - Involvement in K+ acquisition, redistribution and homeostasis. FEBS Lett. 581, 2348–2356. doi: 10.1016/j.febslet.2007.03.035
Golldack, D., Lüking, I., Yang, O. (2011). Plant tolerance to drought and salinity: Stress regulating transcription factors and their functional significance in the cellular transcriptional network. Plant Cell Rep. 30, 1383–1391. doi: 10.1007/s00299-011-1068-0
Gupta, M., Qiu, X., Wang, L., Xie, W., Zhang, C., Xiong, L., et al. (2008). KT/HAK/KUP potassium transporters gene family and their whole-life cycle expression profile in rice (Oryza sativa). Mol. Genet. Genomics 280, 437–452. doi: 10.1007/s00438-008-0377-7
Gupta, D. K., Tiwari, S., Chatterjee, S., Walther, C. (2018). Potassium and its role in cesium transport in plants. Biol. (Bratisl) 73, 885–896. doi: 10.2478/s11756-018-0110-x
Hamamoto, S., Horie, T., Hauser, F., Deinlein, U., Schroeder, J., II, Uozumi, N. (2015). HKT transporters mediate salt stress resistance in plants: From structure and function to the field. Curr. Opin. Biotechnol. 32, 113–120. doi: 10.1016/j.copbio.2014.11.025
Han, M., Wu, W., Wu, W. H., Wang, Y. (2016). Potassium transporter KUP7 is involved in K+ acquisition and translocation in Arabidopsis root under K+-limited conditions. Mol. Plant 9, 437–446. doi: 10.1016/j.molp.2016.01.012
Hill, J. E., Myers, M. A., Koerner, T. J., Tzagoloff, A. (1986). Yeast/E. coli shuttle vectors with multiple unique restriction sites. Yeast 2, 163–167. doi: 10.1002/yea.320020304
Himabindu, Y., Chakradhar, T., Reddy, M. C., Kanygin, A., Redding, K. E., Chandrasekhar, T. (2016). Salt-tolerant genes from halophytes are potential key players of salt tolerance in glycophytes. Environ. Exp. Bot. 124, 39–63. doi: 10.1016/J.ENVEXPBOT.2015.11.010
Hoang, X. L. T., Nhi, D. N. H., Thu, N. B. A., Thao, N. P., Tran, L.-S. P. (2017). Transcription factors and their roles in signal transduction in plants under abiotic stresses. Curr. Genomics 18, 483. doi: 10.2174/1389202918666170227150057
Horie, T., Hauser, F., Schroeder, J., II (2009). HKT transporter-mediated salinity resistance mechanisms in Arabidopsis and monocot crop plants. Trends Plant Sci. 14, 660–668. doi: 10.1016/j.tplants.2009.08.009
Horie, T., Sugawara, M., Okada, T., Taira, K., Kaothien-Nakayama, P., Katsuhara, M., et al. (2011). Rice sodium-insensitive potassium transporter, OsHAK5, confers increased salt tolerance in tobacco BY2 cells. J. Biosci. Bioeng. 111, 346–356. doi: 10.1016/j.jbiosc.2010.10.014
Huang, Y., Zhang, X., Li, Y., Ding, J., Du, H., Zhao, Z., et al. (2018). Overexpression of the Suaeda salsa SsNHX1 gene confers enhanced salt and drought tolerance to transgenic Zea mays. J. Integr. Agric. 17, 2612–2623. doi: 10.1016/S2095-3119(18)61998-7
Iwata, Y., Lee, M.-H., Koizumi, N. (2011). Analysis of a transcription factor using transient assay in Arabidopsis protoplasts. Methods Mol. Biol. 754, 107–117. doi: 10.1007/978-1-61779-154-3_6
Jiang, Y., Deyholos, M. K. (2009). Functional characterization of Arabidopsis NaCl-inducible WRKY25 and WRKY33 transcription factors in abiotic stresses. Plant Mol. Biol. 69, 91–105. doi: 10.1007/s11103-008-9408-3
Jung, J. Y., Shin, R., Schachtman, D. P. (2009). Ethylene mediates response and tolerance to potassium deprivation in Arabidopsis. Plant Cell 21, 607–621. doi: 10.1105/tpc.108.063099
Kaufmann, K., Muiño, J. M., Østerås, M., Farinelli, L., Krajewski, P., Angenent, G. C. (2010). Chromatin immunoprecipitation (ChIP) of plant transcription factors followed by sequencing (ChIP-SEQ) or hybridization to whole genome arrays (ChIP-CHIP). Nat. Protoc. 5, 457–472. doi: 10.1038/nprot.2009.244
Kellermeier, F., Chardon, F., Amtmann, A. (2013). Natural variation of Arabidopsis root architecture reveals complementing adaptive strategies to potassium starvation. Plant Physiol. 161, 1421–1432. doi: 10.1104/pp.112.211144
Ketehouli, T., Carther, K. F., II, Noman, M., Wang, F. W., Li, X. W., Li, H. Y. (2019). Adaptation of plants to salt stress: Characterization of Na+ and K+ transporters and role of Cbl gene family in regulating salt stress response. Agronomy 9, 687. doi: 10.3390/agronomy9110687
Kim, E. J., Kwak, J. M., Uozumi, N., Schroeder, J., II (1998). AtKUP1: An Arabidopsis gene encoding high-affinity potassium transport activity. Plant Cell 10, 51–62. doi: 10.1105/tpc.10.1.51
Kodikara, K. A. S., Jayatissa, L. P., Huxham, M., Dahdouh-Guebas, F., Koedam, N. (2018). The effects of salinity on growth and survival of mangrove seedlings changes with age. Acta Bot. Brasilica 32, 37–46. doi: 10.1590/0102-33062017abb0100
Krishnamurthy, P., Jyothi-Prakash, P. A., Qin, L., He, J., Lin, Q., Loh, C. S., et al. (2014). Role of root hydrophobic barriers in salt exclusion of a mangrove plant Avicennia officinalis. Plant Cell Environ. 37, 1656–1671. doi: 10.1111/pce.12272
Krishnamurthy, P., Mohanty, B., Wijaya, E., Lee, D. Y., Lim, T. M., Lin, Q., et al. (2017). Transcriptomics analysis of salt stress tolerance in the roots of the mangrove Avicennia officinalis. Sci. Rep. 7, 1–19. doi: 10.1038/s41598-017-10730-2
Krishnamurthy, P., Vishal, B., Khoo, K., Rajappa, S., Loh, C. S., Kumar, P. P. (2019). Expression of AoNHX1 increases salt tolerance of rice and Arabidopsis, and bHLH transcription factors regulate AtNHX1 and AtNHX6 in Arabidopsis. Plant Cell Rep. 38, 1299–1315. doi: 10.1007/s00299-019-02450-w
Lee, H. W., Kim, N. Y., Lee, D. J., Kim, J. (2009). LBD18/ASL20 regulates lateral root formation in combination with LBD16/ASL18 downstream of ARF7 and ARF19 in Arabidopsis. Plant Physiol. 151, 1377–1389. doi: 10.1104/pp.109.143685
Li, M., Li, Y., Li, H., Wu, G., Näsholm, T. (2011). Overexpression of AtNHX5 improves tolerance to both salt and drought stress in Broussonetia papyrifera (L.) Vent. Tree Physiol. 31, 349–357. doi: 10.1093/TREEPHYS/TPR003
Liang, M., Gao, Y., Mao, T., Zhang, X., Zhang, S., Zhang, H., et al. (2020). Characterization and expression of KT/HAK/KUP transporter family genes in willow under potassium deficiency, drought, and salt stresses. Bio Med Res. Int. 2020 (6), 1–12. doi: 10.1155/2020/2690760
Liu, W., Tai, H., Li, S., Gao, W., Zhao, M., Xie, C., et al. (2014). bHLH122 is important for drought and osmotic stress resistance in Arabidopsis and in the repression of ABA catabolism. New Phytol. 201, 1192–1204. doi: 10.1111/nph.12607
Liu, Y., Ji, X., Nie, X., Qu, M., Zheng, L., Tan, Z., et al. (2015). Arabidopsis AtbHLH112 regulates the expression of genes involved in abiotic stress tolerance by binding to their E-box and GCG-box motifs. New Phytol. 207, 692–709. doi: 10.1111/nph.13387
Ma, Y., Wang, L., Wang, J., Zhong, Y., Cheng, Z.-M. (2019). Isolation and expression analysis of Salt Overly Sensitive gene family in grapevine (Vitis vinifera) in response to salt and PEG stress. PloS One 14, e0212666. doi: 10.1371/journal.pone.0212666 (Max).
Mangano, S., Silberstein, S., Santa-María, G. E. (2008). Point mutations in the barley HvHAK1 potassium transporter lead to improved K+-nutrition and enhanced resistance to salt stress. FEBS Lett. 582, 3922–3928. doi: 10.1016/j.febslet.2008.10.036
Meraj, T. A., Fu, J., Raza, M. A., Zhu, C., Shen, Q., Xu, D., et al. (2020). Transcriptional factors regulate plant stress responses through mediating secondary metabolism. Genes (Basel) 11, 346. doi: 10.3390/genes11040346
Mian, A., Oomen, R. J. F. J., Isayenkov, S., Sentenac, H., Maathuis, F. J. M., Véry, A. A. (2011). Over-expression of an Na+- and K+-permeable HKT transporter in barley improves salt tolerance. Plant J. 68, 468–479. doi: 10.1111/j.1365-313X.2011.04701.x
Mishra, A., Tanna, B. (2017). Halophytes: Potential resources for salt stress tolerance genes and promoters. Front. Plant Sci. 8, 829. doi: 10.3389/fpls.2017.00829
Mishra, S., Alavilli, H., Lee, B., Panda, S. K., Sahoo, L. (2014). Cloning and Functional Characterization of a vacuolar Na+/H+ antiporter gene from mungbean (VrNHX1) and its ectopic expression enhanced salt tolerance in Arabidopsis thaliana. PloS One 9, e106678. doi: 10.1371/journal.pone.0106678
Munns, R., Tester, M. (2008). Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 59, 651–681. doi: 10.1146/annurev.arplant.59.032607.092911
Nieves-Cordones, M., Alemán, F., Martínez, V., Rubio, F. (2010). The Arabidopsis thaliana HAK5 K+ transporter is required for plant growth and K+ acquisition from low K+ solutions under saline conditions. Mol. Plant 3, 326–333. doi: 10.1093/mp/ssp102
Okushima, Y., Fukaki, H., Onoda, M., Theologis, A., Tasaka, M. (2007). ARF7 and ARF19 regulate lateral root formation via direct activation of LBD/ASL genes in Arabidopsis. Plant Cell 19, 118–130. doi: 10.1105/tpc.106.047761
Osakabe, Y., Arinaga, N., Umezawa, T., Katsura, S., Nagamachi, K., Tanaka, H., et al. (2013). Osmotic stress responses and plant growth controlled by potassium transporters in Arabidopsis. Plant Cell 25, 609–624. doi: 10.1105/tpc.112.105700
Pastore, D., Stoppelli, M. C., Di Fonzo, N., Passarella, S. (1999). The existence of the K+ channel in plant mitochondria. J. Biol. Chem. 274, 26683–26690. doi: 10.1074/jbc.274.38.26683
Pastore, D., Soccio, M., Laus, M. N., Trono, D. (2013). The uniqueness of the plant mitochondrial potassium channel. BMB Rep. 46, 391–397. doi: 10.5483/BMBRep.2013.46.8.075
Petrezsélyová, S., Ramos, J., Sychrová, H. (2011). Trk2 transporter is a relevant player in K+ supply and plasma-membrane potential control in Saccharomyces cerevisiae. Folia Microbiol. (Praha) 56, 23–28. doi: 10.1007/s12223-011-0009-1
Ragel, P., Raddatz, N., Leidi, E. O., Quintero, F. J., Pardo, J. M. (2019). Regulation of K+ nutrition in plants. Front. Plant Sci. 10, 281. doi: 10.3389/fpls.2019.00281
Ravindran, P., Verma, V., Stamm, P., Kumar, P. P. (2017). A novel RGL2–DOF6 complex contributes to primary seed dormancy in Arabidopsis thaliana by regulating a GATA transcription factor. Mol. Plant 10, 1307–1320. doi: 10.1016/j.molp.2017.09.004
Rigas, S., Debrosses, G., Haralampidis, K., Vicente-Agullo, F., Feldmann, K. A., Grabov, A., et al. (2001). TRH1 encodes a potassium transporter required for tip growth in arabidopsis root hairs. Plant Cell 13, 139–151. doi: 10.1105/tpc.13.1.139
Rigas, S., Ditengou, F. A., Ljung, K., Daras, G., Tietz, O., Palme, K., et al. (2013). Root gravitropism and root hair development constitute coupled developmental responses regulated by auxin homeostasis in the Arabidopsis root apex. New Phytol. 197, 1130–1141. doi: 10.1111/nph.12092
Roy, S. J., Negrão, S., Tester, M. (2014). Salt resistant crop plants. Curr. Opin. Biotechnol. 26, 115–124. doi: 10.1016/j.copbio.2013.12.004
Ruan, Y.-L., Llewellyn, D. J., Furbank, R. T. (2001). The control of single-celled cotton fiber elongation by developmentally reversible gating of plasmodesmata and coordinated expression of sucrose and K transporters and expansin. Plant Cell. 1, 47–60. doi: 10.1105/tpc.13.1.47
Ruiz-Lau, N., Bojórquez-Quintal, E., Benito, B., Echevarría-Machado, I., Sánchez-Cach, L. A., De Fátima Medina-Lara, M., et al. (2016). Molecular cloning and functional analysis of a Na+-insensitive K+ transporter of Capsicum chinense jacq. Front. Plant Sci. 7, 1980. doi: 10.3389/fpls.2016.01980
Shabala, S., Cuin, T. A. (2008). Potassium transport and plant salt tolerance. Physiol. Plant 133, 651–669. doi: 10.1111/j.1399-3054.2007.01008
Sharma, T., Dreyer, I., Riedelsberger, J. (2013). The role of K+ channels in uptake and redistribution of potassium in the model plant Arabidopsis thaliana. Front. Plant Sci. 4, 224. doi: 10.3389/fpls.2013.00224
Su, H., Golldack, D., Zhao, C., Bohnert, H. J. (2002). The expression of HAK-Type K+ transporters is regulated in response to salinity stress in common ice plant. Plant Physiol. 129, 1482–1493. doi: 10.1104/pp.001149
Véry, A.-A., Sentenac, H. (2003). Molecular mechanisms and regulation of K+ transport in higher plants. Annu. Rev. Plant Biol. 54, 575–603. doi: 10.1146/annurev.plant.54.031902.134831
Volkov, V., Beilby, M. J. (2017). Editorial: Salinity tolerance in plants: Mechanisms and regulation of ion transport. Front. Plant Sci. 8, 1795. doi: 10.3389/fpls.2017.01795
Wang, P., Su, L., Gao, H., Jiang, X., Wu, X., Li, Y., et al. (2018). Genome-wide characterization of bHLH genes in grape and analysis of their potential relevance to abiotic stress tolerance and secondary metabolite biosynthesis. Front. Plant Sci. 9, 64. doi: 10.3389/fpls.2018.00064
Wu, H., Zhang, X., Giraldo, J. P., Shabala, S. (2018). It is not all about sodium: revealing tissue specificity and signalling roles of potassium in plant responses to salt stress. Plant Soil 431, 1–17. doi: 10.1007/s11104-018-3770-y
Yang, Q., Chen, Z. Z., Zhou, X. F., Yin, H. B., Li, X., Xin, X. F., et al. (2009). Overexpression of SOS (salt overly sensitive) genes increases salt tolerance in transgenic Arabidopsis. Mol. Plant 2, 22–31. doi: 10.1093/mp/ssn058
Yang, T., Zhang, S., Hu, Y., Wu, F., Hu, Q., Chen, G., et al. (2014). The role of a potassium transporter OsHAK5 in potassium acquisition and transport from roots to shoots in rice at low potassium supply levels. Plant Physiol. 166, 945–959. doi: 10.1104/pp.114.246520
Yoo, S. D., Cho, Y. H., Sheen, J. (2007). Arabidopsis mesophyll protoplasts: A versatile cell system for transient gene expression analysis. Nat. Protoc. 2, 1565–1572. doi: 10.1038/nprot.2007.199
Zhang, J., Wang, L., Liu, Y., Li, D., Feng, S., Yang, J., et al. (2019). Improving salt tolerance in potato through overexpression of AtHKT1 gene. BMC Plant Biol. 19, 357. doi: 10.1186/s12870-019-1963-z
Zhang, M., Huang, P., Ji, Y., Wang, S., Wang, S., Li, Z., et al. (2020). KUP9 maintains root meristem activity by regulating K+ and auxin homeostasis in response to low K. EMBO Rep. 21, e50164. doi: 10.15252/embr.202050164
Keywords: salt tolerance, mangrove, KUP2, bHLH, WRKY, ChIP
Citation: Rajappa S, Krishnamurthy P and Kumar PP (2020) Regulation of AtKUP2 Expression by bHLH and WRKY Transcription Factors Helps to Confer Increased Salt Tolerance to Arabidopsis thaliana Plants. Front. Plant Sci. 11:1311. doi: 10.3389/fpls.2020.01311
Received: 27 May 2020; Accepted: 11 August 2020;
Published: 25 August 2020.
Edited by:
Rosa M. Rivero, Spanish National Research Council, SpainReviewed by:
Yanchun Cui, Chinese Academy of Sciences, ChinaCopyright © 2020 Rajappa, Krishnamurthy and Kumar. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Prakash P. Kumar, ZGJza3VtYXJAbnVzLmVkdS5zZw==
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.