
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Plant Sci., 21 November 2018
Sec. Plant Breeding
Volume 9 - 2018 | https://doi.org/10.3389/fpls.2018.01690
This article is part of the Research TopicThe Applications of New Multi-Locus GWAS Methodologies in the Genetic Dissection of Complex TraitsView all 18 articles
 Kaixin Zhang1,2†
Kaixin Zhang1,2† Shulin Liu3†
Shulin Liu3† Wenbin Li1,2
Wenbin Li1,2 Shiping Liu1,2
Shiping Liu1,2 Xiyu Li1,2
Xiyu Li1,2 Yanlong Fang1,2
Yanlong Fang1,2 Jun Zhang4
Jun Zhang4 Yue Wang1,2
Yue Wang1,2 Shichao Xu1,2
Shichao Xu1,2 Jianan Zhang1,2
Jianan Zhang1,2 Jie Song1,2
Jie Song1,2 Zhongying Qi1,2
Zhongying Qi1,2 Xiaocui Tian1,2
Xiaocui Tian1,2 Zhixi Tian3
Zhixi Tian3 Wen-Xia Li1,2*
Wen-Xia Li1,2* Hailong Ning1,2*
Hailong Ning1,2*Protein content (PC), an important trait in soybean (Glycine max) breeding, is controlled by multiple genes with relatively small effects. To identify the quantitative trait nucleotides (QTNs) controlling PC, we conducted a multi-locus genome-wide association study (GWAS) for PC in 144 four-way recombinant inbred lines (FW-RILs). All the FW-RILs were phenotyped for PC in 20 environments, including four locations over 4 years with different experimental treatments. Meanwhile, all the FW-RILs were genotyped using SoySNP660k BeadChip, producing genotype data for 109,676 non-redundant single-nucleotide polymorphisms. A total of 129 significant QTNs were identified by five multi-locus GWAS methods. Based on the 22 common QTNs detected by multiple GWAS methods or in multiple environments, pathway analysis identified 8 potential candidate genes that are likely to be involved in protein synthesis and metabolism in soybean seeds. Using superior allele information for 22 common QTNs in 22 elite and 7 inferior lines, we found higher superior allele percentages in the elite lines and lower percentages in the inferior lines. These findings will contribute to the discovery of the polygenic networks controlling PC in soybean, increase our understanding of the genetic foundation and regulation of PC, and be useful for molecular breeding of high-protein soybean varieties.
Soybean [Glycine max (L.) Merr.] is a globally important high-protein crop, with protein accounting for about 40% of the seed’s dry weight. Soybean is one of humans’ main sources of dietary protein; therefore, breeding high-protein varieties of soybean is an ongoing, important objective of plant breeders. The efficiency of plant breeding has been greatly accelerated by the emergence of molecular markers and molecular technology, such as random amplified polymorphic DNA, restriction fragment length polymorphism, amplified fragment length polymorphism, simple sequence repeats, specific-locus amplified fragment, and single-nucleotide polymorphism (SNP). For genome-wide association studies (GWAS) in soybean, the acquisition of a large number of molecular markers is extremely important (Song et al., 2013), and SNPs are well suited for such analyses because of their high densities throughout the genome (Gaur et al., 2012).
Breeders and molecular geneticists have routinely used populations derived from biparental crosses for development of new varieties and mapping quantitative trait loci (QTLs) for traits of interest. However, the richness of allelic and phenotypic variation in biparental inter-mated populations is somewhat limited. To overcome this limitation, animal breeders have developed the multi-parental inter-mated population design, for example by using a population descended from eight mouse strains (Yalcin et al., 2005). Recently, multi-parent advanced generation inter-cross (MAGIC) lines have also been developed in plants, including wheat (Triticum aestivum; Huang et al., 2012; Mackay et al., 2014), maize (Zea mays; Dell’Acqua et al., 2015), Arabidopsis thaliana (Kover et al., 2009), barley (Hordeum vulgare; Sannemann et al., 2015), tomato (Solanum lycopersicum; Pascual et al., 2015), and rice (Oryza sativa; Bandillo et al., 2013; Meng et al., 2016a,b). MAGIC populations have more diverse alleles than bi-parental populations, which increases genetic variation. However, obtaining a MAGIC population is labor intensive because of the repeated crossing and requires large population sizes to include recombinants for all the desirable traits. An intermediate design, the four-way cross population, is easier to obtain while providing some of the same benefits as an eight-way cross population.
In recent years, GWAS with high-density SNPs have emerged as very powerful tools for dissecting the genetic basis of complex traits. This approach has been applied to MAGIC populations of many crop plants (Bandillo et al., 2013; Pascual et al., 2015; Meng et al., 2016a,b) but not, as of yet, to soybean. As in soybean, either biparental populations or natural populations have been used in all previous QTL mapping studies (Li et al., 2018; Wang et al., 2018). This previous research indicates that PC in soybean is a typical quantitative trait controlled by multiple genes with relatively small genetic effects, whose identification will require a more efficient method to detected QTLs. Multi-locus GWAS is a suitable method for identifying significant QTNs, especially for relatively small effects; it also has a low false positive rate, and has been used in many studies (Liu et al., 2016; Wang et al., 2016; Tamba et al., 2017; Zhang Y. et al., 2017; Wen et al., 2018).
In this study, we used 144 recombinant inbred lines (FW-RILs) from a four-way cross, which were genotyped by SNPs and phenotyped seed protein content (PC) in different environments. We then combined these data to identify significant QTNs for PC in soybean using multi-locus GWAS methods. The objective was to find common QTNs that were identified by multiple methods or in multiple environments and then deduce potential candidate genes and identify elite lines in the FW-RIL population, as a means to accelerate molecular breeding to increase PC in soybean.
Four soybean varieties, Kenfeng14 (PC 41.08%), Kenfeng15 (PC 41.42%), Kenfeng19 (PC 43.06%), and Heinong48 (PC 43.55%), were used to construct a four-way recombinant inbred line (FW-RIL) population. Among these, Kenfeng14, Kenfeng15, and Kenfeng19 were bred by the Heilongjiang Academy of Agricultural Reclamation and derived from the crosses Suinong 10 × Changnong 5, Suinong 14 × Kenjiao 9307, and Hefeng 25 × (Kenfeng 4 × Gong 8861-0), respectively; and Heinong48 was bred by Heilongjiang Academy of Agricultural Science and derived from the cross Ha 90-6719 × Sui 90-5888.
First, two single crosses, Kengfeng14 × Kenfeng15 and Kenfeng19 × Heinong48, were carried out in Harbin (45.75°N, 126.63°E), Heilongjiang Province, China, and the F1 seeds were harvested in 2008. Second, a cross was conducted between two sets of single-cross F1 seeds, and F1 seeds of the resulting four-way cross (Kengfeng14 × Kenfeng15) × (Kenfeng19 × Heinong48) were harvested in 2009. Third, the four-way cross F1 seeds were self-crossed for six generations continuously by alternate sowing in Yacheng (17.5°N, 109.00°E), Hainan Province, China, in the winter and in Harbin in the summer from 2010 to 2014, using the single-seed descent method to select single seeds from individual plants in each generation. Finally, a total of 144 FW-RILs were obtained for this study.
The four parental lines and 144 FW-RILs were planted in 20 environments with different locations, years, seedling densities, fertilizers, and sowing dates. The detailed planting schedule is summarized in Supplementary Table S1. All plant materials in each environment were grown in three-row 5 m × 0.7 m plots in a completely randomized block design with three replications. The experimental plots were managed identically to local soybean crops. Ten plants from the middle of the plots for each line (four parents and 144 FW-RILs) were harvested and the seeds threshed separately for each of the 20 environments. The total PC of seeds (dry seeds, with water content of about 10%) was determined in three random samples from mixed seed of each line by the near-infrared analyzer (Infratec 1241, Foss, Denmark) at the Key Laboratory of Soybean Biology of the Chinese Education Ministry at the Northeast Agricultural University in China. The calibration regression technique was Partial Least Square (PLS), which involved combining spectral data with laboratory data (Kjeldahl method) to calculate seed PC, described by the percentage of seed weight. The phenotypic values given for each parental and FW-RIL used in this study were all the mean values of three repetitions.
Mean, standard deviation, minimum, maximum, range, skewness, kurtosis, and coefficient of variation (CV) for FW-RILs in each environment were calculated. A correlation analysis between each pair of environments was performed. Analysis of variance (ANOVA) for single environment and jointly multiple environments was conducted with “varietal effect model of genotype” and “environment + genotype + environment × genotype interaction,” respectively, and the broad-sense heritability (h2) was estimated by the following equation:
where is the genotypic variance, is the variance due to the genotype × environment interaction, σ2 is the error variance, e is the number of environments, and r is the number of replications within an environment. The statistical analysis was implemented by SAS 9.2 (SAS Institute, Cary, NC, United States).
Juvenile leaves from parents and FW-RIL plants were collected, frozen in liquid nitrogen, and immediately ground into powder. Total genomic DNA was extracted using the CTAB method (Doyle et al., 1990) and eluted in 50 μl deionized water. The DNA concentration was determined using a UV752N spectrophotometer (Shanghai Jingke Science Instrument Co. Ltd.) and was diluted to 100 ng ± 1 ng in deionized water. SNP genotyping was performed at Beijing Boao Biotechnology Co. Ltd, using the SoySNP660K BeadChip. A total of 109,676 SNPs across 20 chromosomes remained after quality filtering; the SNP markers identified were filtered for minor allele frequency (MAF > 0.05), and the maximum missing sites per SNP was < 10% (Belamkar et al., 2016). Heterozygous loci were then marked as missing to obtain better estimates of marker effects, and the SNP markers were re-filtered using the same filtering criteria and used for the next analysis of population structure, kinship, and GWAS.
The analysis of population structure was performed with the software STRUCTURE 2.3.4 (Pritchard et al., 2000). For each run, the number of burn-in iterations was 10,000, followed by 2000 Markov chain Monte Carlo (MCMC) replications after burn-in. The admixture and allele frequencies correlated models were considered in the analysis. Ten impended iterations were used in the STRUCTURE analysis. The hypothetical number of subpopulations (K) ranged from 1 to 10. The best K was identified according to Evanno et al. (2005) using STRUCTURE HARVESTER (Earl and Vonholdt, 2012).
TASSEL 5.0 was utilized to analyze linkage disequilibrium (LD) (Bradbury et al., 2007) by analyzing r2 values of all pairs of SNPs located within 10 Mb physical distance, the LD decay trend was found following regression of the negative natural logarithm, and the physical distance of LD decay was estimated as the position where r2 dropped to half of its maximum value.
The software mrMLM.GUI (version 3.0) was used to perform the GWAS. Five multi-locus GWAS methods within mrMLM.GUI were used to identify significant QTNs, including mrMLM (Wang et al., 2016), FASTmrMLM (Tamba et al., 2017), FASTmrEMMA (Wen et al., 2018), pLARmEB (Zhang J. et al., 2017), and ISIS EM-BLASSO (Tamba et al., 2017). The critical P-value parameters for these methods at the first stage were set to 0.01 except for FASTmrEMMA, where the critical P-value was set to 0.005, and the critical LOD score was set to 3 for significant QTN at the last stage. All these five methods involved the population structure and kinship matrices in this study, and the kinship matrix was calculated with the software mrMLM.GUI 3.0.
We considered the QTNs we detected from multiple environments or by multiple methods as common QTNs. Based on the effect values of each common QTN and the genotype for code 1, we could determine the superior alleles of each QTN. If the effect value of the QTN is positive, the genotype for code 1 is the superior allele; if the effect value is negative, another genotype is the superior allele. For each QTN, the superior allele percentage in the 144 FW-RILs was equal to the number of lines containing the superior allele divided by the total number of lines. For each line, the proportion of superior alleles in these QTNs was calculated as the number of superior alleles divided by the total number of QTNs. A heat map visualizing the percentage of superior alleles was obtained in the R (heatmap package) program (Mellbye and Schuster, 2014).
The search for potential candidate genes based on the common QTNs detected by multiple methods or in multiple environments was performed using four steps. First, the intervals that include each common QTN were selected on the Phytozome website1. These intervals were determined by the rate of LD decay. Second, genes highly expressed in the form process of seed protein through the Bio-Analytic Resource for Plant Biology (BAR) website2 were identified. Third, based on the experimental data in the Plant Expression Database (PLEXdb) website3, the differentially expressed genes among high and low protein lines were identified from the above high expression genes. Finally, all the differentially expressed genes were put together for analysis of pathways on the Kyoto Encyclopedia of Genes and Genomes (KEGG) website4, and potential candidate genes were identified by the result of pathway analysis.
We measured the PC phenotypes of the parents and the 144 FW-RILs in 20 environments, which are presented in Supplementary Table S2 and Supplementary Figure S1. Graphing the average value for PC of each line in 20 environments revealed that the 144 lines show extensive variation in PC (Supplementary Figure S1). Examination of the values (Supplementary Table S2) showed that the parental lines Kenfeng14 and Kenfeng15 had lower PC than Kenfeng19 and Heinong48 in all 20 environments. And the “Range” (Range = PCMax – PCMin) of the four parents in 20 environments was from 1.70 to 4.04%; the “Range” of FW-RILs was 5.20–11.56%, representing a large difference in PC, especially in FW-RILs. Kurtosis and skewness (absolute value) were less than 1, indicating the continuous normal distribution of the PC values (Supplementary Table S2 and Figure 1). We found a high coefficient of variation in PC across all the environments. The ANOVA results of parents and FW-RILs both indicated that extremely significant variation exists in genotype, environment, and genotype-by-environment (Tables 1, 2). The mean square values for the genotype-by-environment interaction were all less than the mean square values of genotype, and the estimated broad-based heritability was high, being 85.46%. The correlation coefficients between each pair of environments were almost all positive, and many were significant or extremely significant (Supplementary Table S3), indicating high consistency across various environments.
To define the subpopulations within the panel of 144 lines, as described by Pritchard et al. (2009), we selected 5375 of the 109,676 SNPs that had better polymorphisms and were randomly distributed across the 20 soybean chromosomes. Delta K (ΔK) was calculated using STRUCTURE 2.3.4 (Figure 2A; K = 1–10), revealing the presence of two subpopulations (selected K = 2) based on ΔK values (Figure 2B). These two subgroups contained 53 (36.81%) and 91 (63.19%) lines.
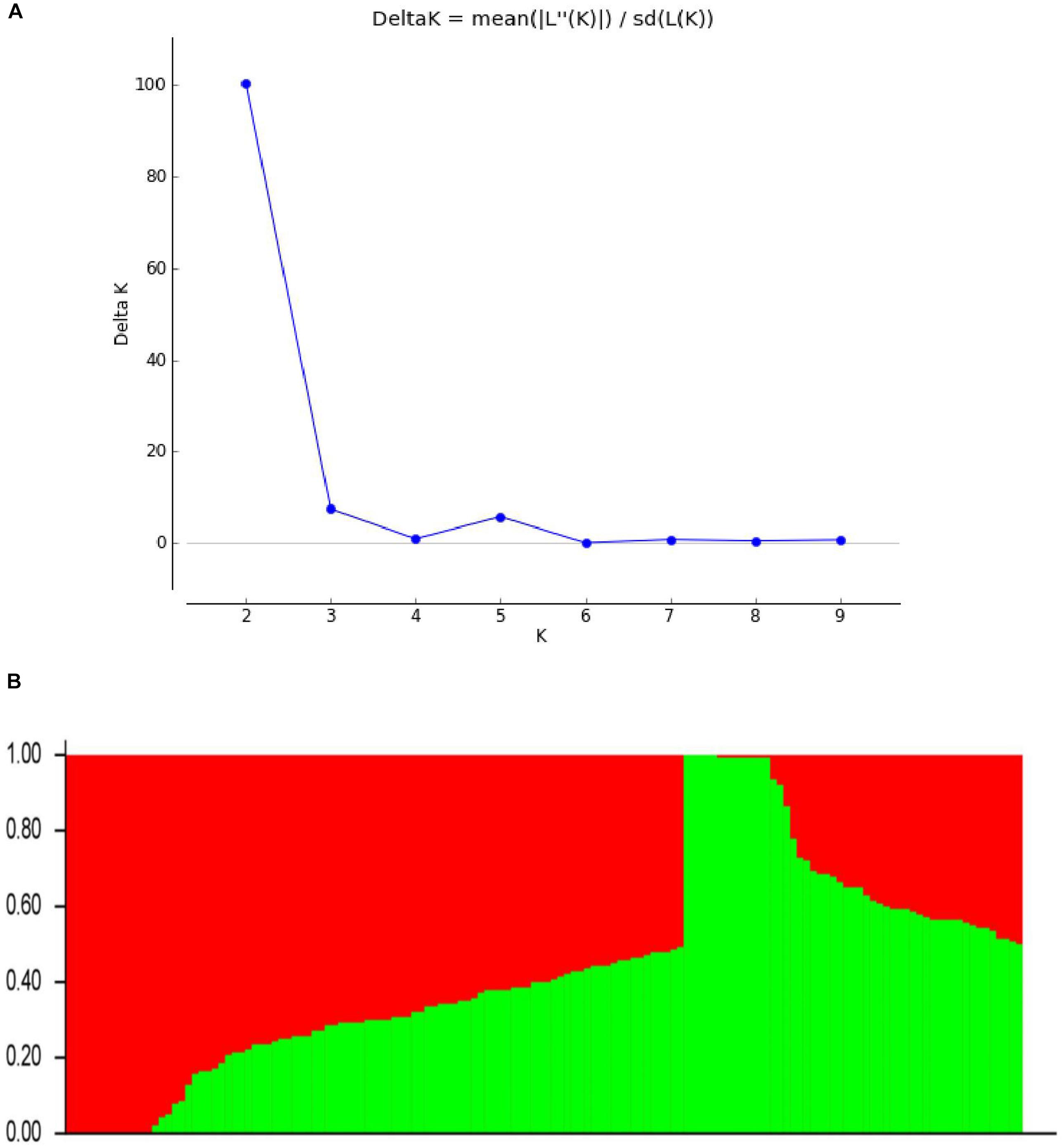
FIGURE 2. Population structure based on 5375 SNPs distributed across 20 chromosomes. (A) Plot of ΔK calculated for K = 1–10. (B) Population structure (K = 2); the areas of the two colors (green and red) illustrate the proportion of each subgroup.
We analyzed the r2 values of all pairs of SNPs located within 10 Mb of each other and determined the LD decay trend based on regression to the negative natural logarithm. As shown in Supplementary Figure S2, r2 decreased gradually with increased distance, and the LD decay distance was estimated at 1.2 Mb, where r2 dropped to half of its maximum value (0.45). Because the population used in this study is derived from parents, the speed of LD decay is slower and the LD decay distance is much longer than that of a natural population.
We identified 19, 18, 12, 37, and 43 significant QTNs for PC using mrMLM, FASTmrMLM, FASTmrEMMA, pLARmEB, and ISIS EM-BLASSO, respectively, and 10, 11, 10, 2, 1, 2, 3, 6, 11, 10, 0, 8, 4, 9, 9, 7, 1, 5, 12, and 8 significant QTNs, respectively, in the 20 environments (Figure 3 and Supplementary Table S4). No significant QTN was detected in the eleventh environment (E11).
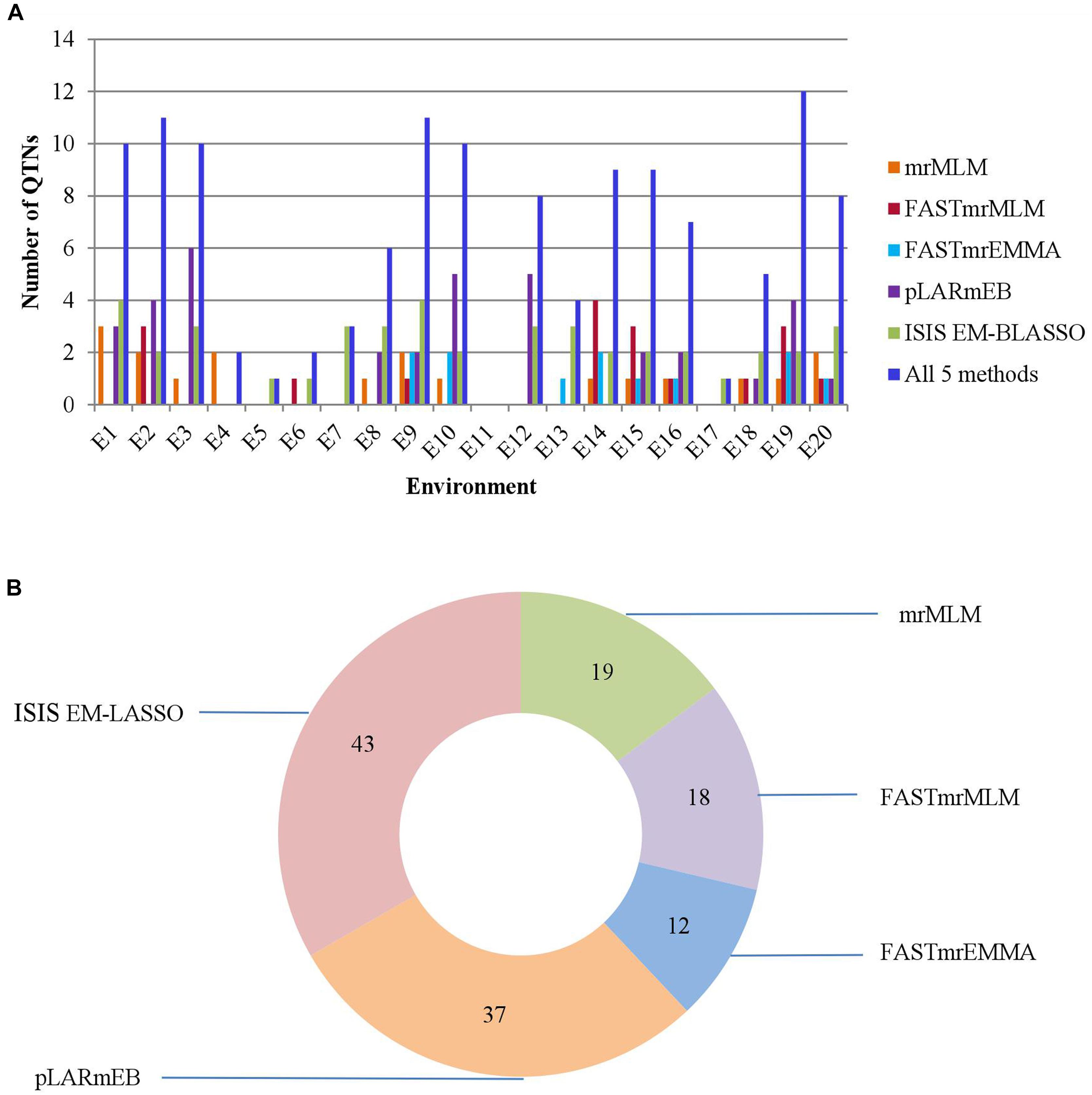
FIGURE 3. (A) The total numbers of significant QTNs detected in 20 environments across 5 methods. (B) The total numbers of significant QTNs detected using each of 5 multi-locus GWAS methods in 20 environments.
We further checked the common QTNs across multiple environments. As discussed above, only one such common QTN was identified in three environments (Table 3). The single QTN (AX-157298785) was located on chromosome 18, with the LOD values ranging from 4.02 to 5.33 (Table 3). The proportion of phenotypic variance explained (PVE) by the QTN ranged from 8.40 to 11.02%, and the QTN direction of effect (positive or negative) was consistent across different environments and different methods (Table 3).
Comparing the results across the different approaches, we found that 22 common QTNs (including AX-157298785) were identified simultaneously by at least two approaches (Table 4); these were located on chromosomes 1, 2, 3, 4, 6, 7, 9, 10, 12, 14, 16, and 18. Their LOD values ranged from 3.06 to 6.90, the proportion of PVE by each QTN ranged from 3.84 to 19.21%, and the direction of effect (positive or negative) of each QTN was also consistent across the different methods (Table 4). Of the 22 common QTNs, 12, 5, and 5 were identified simultaneously by 2, 3, and 4 approaches, respectively. Among the five methods, ISIS EM-BLASSO detected the highest number of common QTNs (Figure 4A), and among the combinations of two methods, FASTmrMLM combined with pLARmEB detected the highest number of common QTNs (Figure 4B).
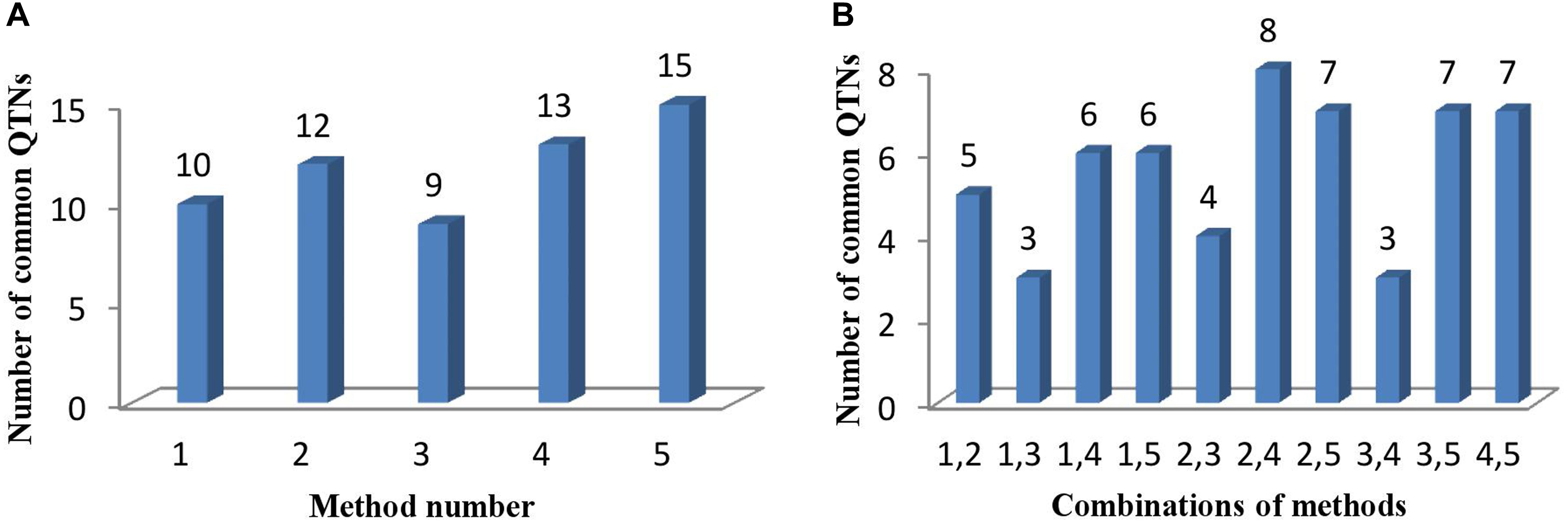
FIGURE 4. (A) The number of common QTNs detected by different methods and (B) different combinations of methods. Method numbers correspond to (1) mrMLM, (2) FASTmrMLM, (3) FASTmrEMMA, (4) pLARmEB, and (5) ISIS EM-BLASSO.
We found only one stable QTN that was identified not only in multiple environments but also by multiple methods (Table 3): AX-157298785, located on chromosome 18, which was detected by mrMLM, FASTmrMLM, FASTmrEMMA, and ISIS EM-BLASSO in environments E14, E16, and E20, with LOD values ranging from 4.02 to 5.33 and PVE values ranging from 5.86 to 11.02% (Table 3).
Based on the PC averages in 20 environments for each FW-RIL, we found that 22 lines had higher phenotypic values (43.07–44.21%) and 7 lines had lower phenotypic values (40.60–40.98%) (Table 5). For each of the 22 elite lines, the percentages of superior alleles (PSA) across 22 common QTNs ranged from 36 to 82% (Table 5), 91% (20 of the 22 lines) showed PSAs of ≥50%, and only 9% showed PSAs of <50%. For each of the 7 lines with lower phenotypic values, the PSAs ranged from 32 to 50% (Table 5), only 2 lines (28%) had PSAs of ≥50%, and the remaining 5 (71%) had PSAs of <50%. Thus, the elite lines with higher PC have more superior alleles than the lines with lower PC (Figure 5).
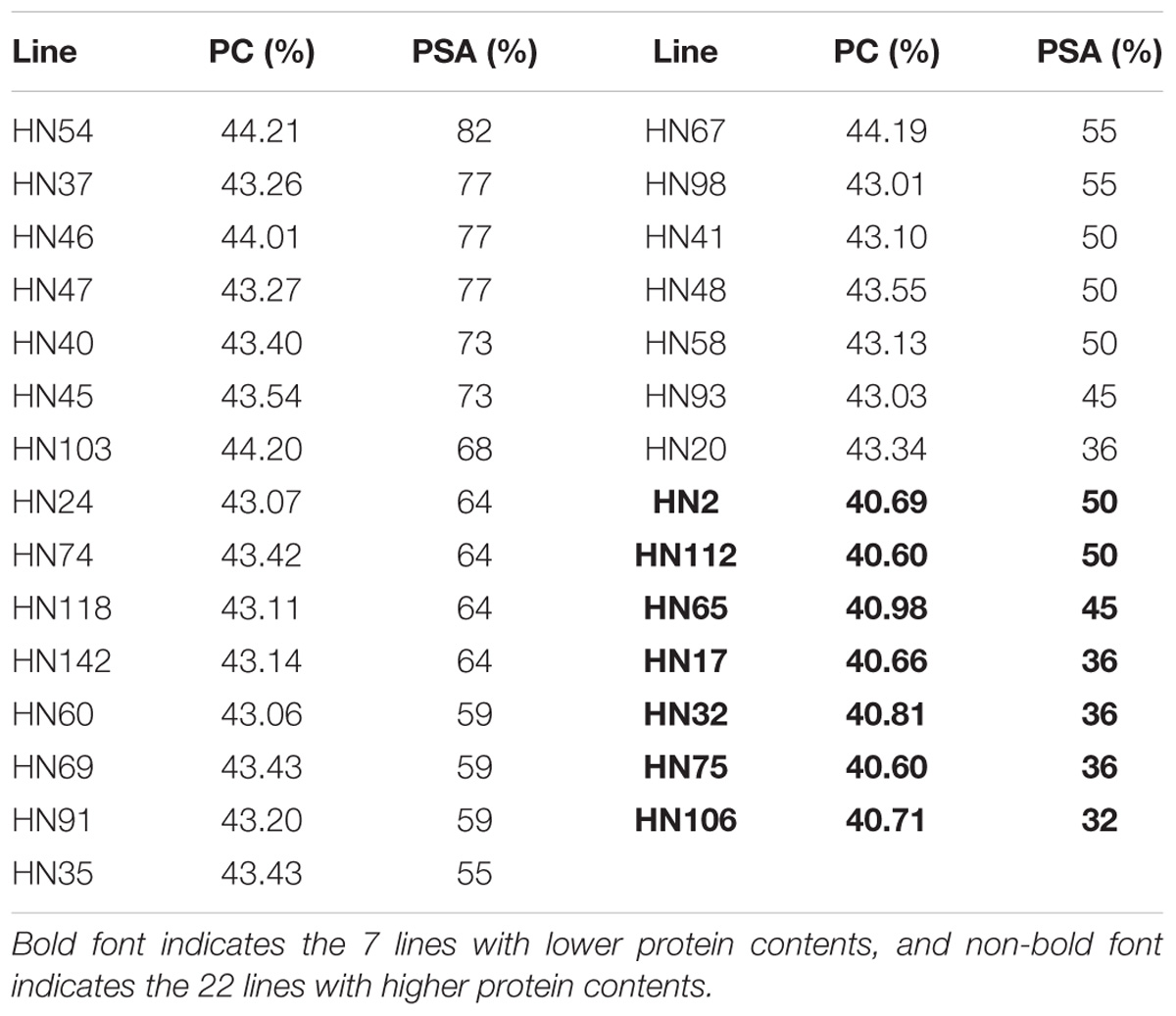
TABLE 5. Phenotypic averages of seed protein content and proportion of superior alleles in 29 lines across 22 common QTNs.
Based on the superior allele information for the 22 common QTNs in 29 lines, the PSAs for each QTN ranged from 36 to 95% in the 22 elite lines, with 16 QTNs showing ≥ 50% superior alleles while the remaining 6 QTNs showed < 50%. The range of PSAs for each QTN was 0–71% in the 7 lines with lower phenotypic values; 8 of the QTNs had PSAs ≥ 50% and the remaining 14 QTNs had PSAs < 50% (Table 6 and Figure 6). The number of QTNs with ≥50% superior alleles was higher in the 22 elite lines than in the 7 inferior lines. Based on these results, we can easily find elite lines by identifying superior alleles for application in breeding higher PC soybean.
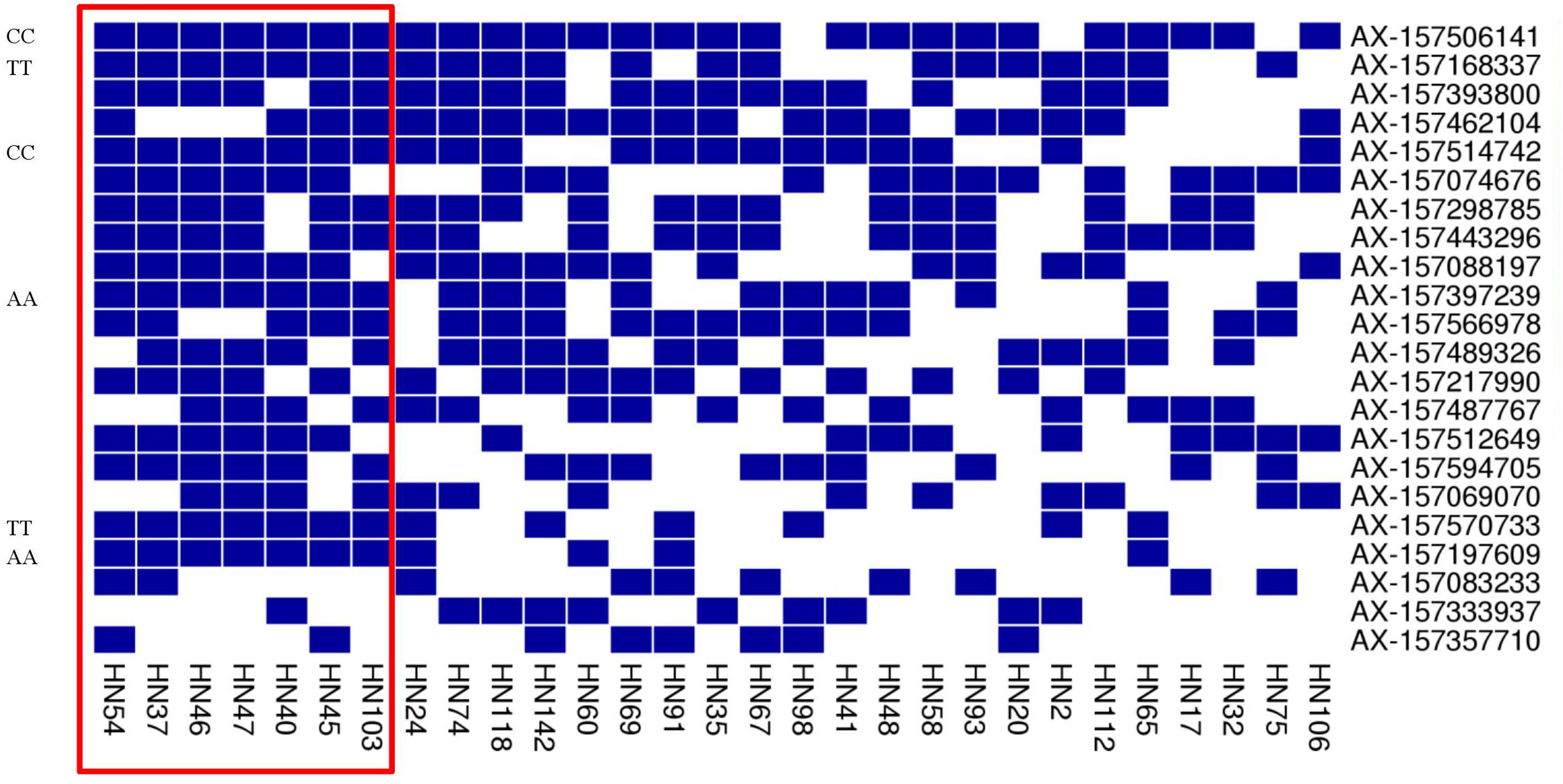
FIGURE 6. Heat map of the superior allele distribution for the 22 common QTNs in the 29 high- and low-PC lines. Blue and white colors represent superior and inferior alleles, respectively.
In addition, we found some common superior alleles in multiple elite lines: for example, the seven lines HN54, HN37, HN46, HN47, HN40, HN45, and HN103 all contained the superior alleles AX-157506141, AX-157168337, AX-157514742, AX-157397239, AX-157570733, and AX-15719760 (Figure 6), and the superior allele AX-157506141 occurred in 21 of the 22 elite lines. We suspect that common superior alleles may have a particularly strong influence on PC. In further research, we hope to make use of this information to breed better soybean using marker-assisted selection.
We used the LD decay distance to select potential candidate genes within a specific distance of each common QTN. Because of the nature of the population (i.e., derived from parents), the LD decay distance is large, so we determined the range of potential candidate genes according to the position of the fastest decay rate. In Supplementary Figure S2B, we can see that LD decays fastest before 200 kb, and then tends to flatten, so we searched for potential candidate genes in the interval of 100 kb on either side of each QTN. Following the four steps described in the Materials and Methods, a total of 288 genes were found in these intervals and 96 genes were expressed highly in seed at the form process of seed protein. Among the 96 genes, 34 genes were differentially expressed among high and low protein lines, and these 34 genes were used to do pathway analysis.
From the annotation data, we found that 17 of 34 genes (51.4% of the genes we submitted) were previously annotated in 14 pathways and 3 protein families in the KEGG database (Supplementary Tables S5, S6 and Figure 7). Of these, 8 were considered potential candidate genes based on the information of their annotation and functions in metabolic pathways (Table 7 bold text).
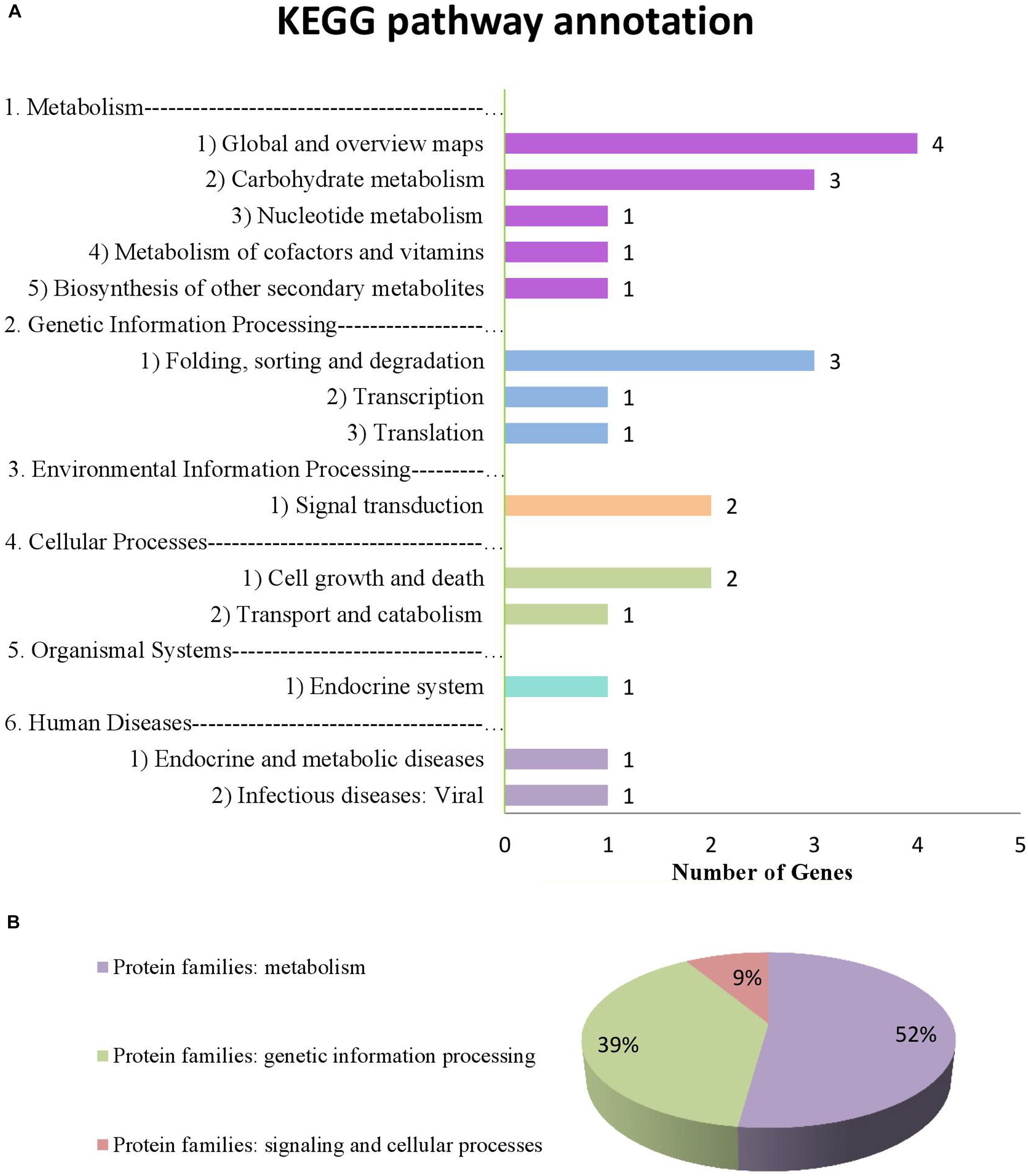
FIGURE 7. Information on pathways and orthologous protein families of 17 genes. (A) shows the information on pathway. (B) shows the information on orthologous protein families.
In this study, we employed multi-locus GWAS with an FW-RIL population of the MAGIC population type to identify QTNs related to PC of soybean. Twenty-two common QTNs were detected by multiple methods or in multiple environments (Tables 3, 4). Based on the SoyBase database and the results of recent studies, 12 of the 22 common QTNs appeared to be near QTLs associated with PC in soybean that had been mapped in earlier studies (Lee et al., 1996; Brummer et al., 1997; Csanádi et al., 2001; Jun et al., 2008; Lu et al., 2013; Mao et al., 2013; Pathan et al., 2013; Hwang et al., 2014; Qi et al., 2014; Vaughn et al., 2014; Warrington et al., 2015): AX-157074676, AX-157594705, AX-157397239, AX-157489326, AX-157462104, AX-157506141, AX-157069070, AX-157357710, AX-157217990, AX-157570733, AX-157566978, and AX-157168337 (Table 4, bold text). This indirectly confirmed the accuracy of our QTN detection. The remaining ten QTNs in this study were new (Table 4, non-bold text). For this population, the detection of significant QTNs is not only a way to identify the genes related to PC, but can also identify good lines based on the superior allele information to support breeding high PC soybean.
Based on the 22 common QTNs detected here and their pathway annotation, we have identified 8 genes that may be related to protein anabolism (Table 7, bold text). Glyma.03G100800 is intimately involved in the biosynthesis of amino acids, and the pentose phosphate pathway which it is involved in also indirectly affects the biosynthesis of proteins (Xu et al., 2018). Glyma.10G207300, Glyma.14G081600, and Glyma.12G019300 are mainly involved in the proteasome pathway and work as protease to degrade proteins to small peptides and amino acids, so we think that these three genes are closely related to protein degradation. Glyma.18G071100 may play an important role in protein anabolism; it participates in the process of glycosylation in endoplasmic reticulum, and the function of glycosylation is to enable proteins to resist the digestive enzymes, so as to protect protein from degradation (Jayaprakash and Surolia, 2017). Glyma.12G112900 is a kind of riboflavin synthase and it participates in the biosynthesis of riboflavin, which plays an important role in energy metabolism including carbohydrate, protein and fat metabolism (Tuan et al., 2014; Zhao et al., 2014). Glyma.18G071300 participates in RNA transport, and this process is essential in protein synthesis; as we know, it mainly carries amino acids into ribosomes and synthesizes proteins under the guidance of mRNA. Glyma.18G028600 is a kind of translocation protein, and it mainly works in post-translational transport and post-translational modification in the process of protein synthesis, so it plays an important role in protein synthesis (Kwon et al., 1999). Based on these 8 genes, further work will be needed to determine which of these actually significantly affect PC in soybeans, and then to identify the target genes. This information can be used as the basis for further exploration of the gene network for the trait.
In recent years, GWAS has been widely applied to crop plants such as rice (Huang et al., 2010; Ma et al., 2016), maize (Tian et al., 2011), and soybean (Li et al., 2018), and the model mainly used for GWAS is mixed linear model (MLM). It belongs to single-locus GWAS, for which the screening criterion for significance is P = 0.05/m (where m is the number of markers) (Perneger, 1998). For a large number of SNPs, some important loci may be undetectable under this screening criterion.
In this study, we also used MLM to carry out the GWAS of soybean PC, with the calculation of population structure based on STRUCTURE 2.3.4 and the calculation of kinship and GWAS based on TASSEL 5.0. However, we did not detect any significant SNPs using the model. We believe that this was related to the screening criteria of single-locus GWAS and to the type of target trait as well as the population type. In light of this negative result, we tried changing the screening criteria, and replaced the strict Bonferroni correction with a less stringent false discovery rate (FDR) correction. The q-values were equal to the P-values adjusted with the Benjamini and Hochberg (2000) procedure; we used the cut-off of q < 0.05 and P < 0.0001as the threshold value to identify significant QTNs. Based on these screening criteria, we still did not find significantly associated SNP markers. To further reduce the stringency of the screening criteria, we next tried using a P-value of < 0.0001 directly as a cut-off without any corrections. With this criterion, a total of 15 SNP markers were identified with 1 cluster (bold text in Table 8). The range for the one cluster was 83.40 kb.
Because of the nature of single-locus methods, even if we identified these significant SNP markers under less stringent screening methods, we lack confidence about our ability to control for the false positive rate of results obtained without the correction for multiple tests. A previous study yielded a similar outcome to ours (Fang et al., 2017), so it seems evident that the single-locus method is not always suitable for detecting the genetic basis of complex traits.
To make up for the shortcomings of the above methods, multi-locus GWAS methods have recently been explored, including the five methods we used in this study: mrMLM (Wang et al., 2016), FASTmrMLM (Tamba et al., 2017), FASTmrEMMA (Wen et al., 2018), pLARmEB (Zhang J. et al., 2017), and ISIS EM-BLASSO (Tamba et al., 2017). Using the five methods, we detected a total of 19, 18, 12, 37, and 43 significant QTNs, respectively (Figure 3B and Supplementary Table S4). The differences in the numbers of QTNs detected by the five methods are presumed to be due to the different principles underlying the different methods: even though all five are two-stage combined approaches, they differ in the models and methods for screening and estimation. Because the main purpose of this study required the most accurate QTNs possible, we felt it desirable to take into consideration the results of all five methods, and took QTNs detected by multiple methods as the credible QTNs to use in further experiments. This practice adds an extra screening to the multi-locus GWAS approach and thus makes us more confident about the results.
Based on the data from the QTNs we detected, we found that the absolute values of QTN effects were all relatively low, in the range of 0.17–1.35, which indirectly explained why we could not detect significant QTNs by MLM with the standard Bonferroni correction. Thus, the greatest advantage of the multi-locus GWAS approach was its ability to find loci with relatively small effects. In addition, the multi-locus GWAS method was more suitable for the FW-RIL population used in this study than single-locus GWAS. This is because the single-locus GWAS method is generally based on SNPs for which there are only two alleles at one locus, meaning that the multi-allelic variation that exists at some genetic loci in the FW-RIL population cannot be detected. However, the multi-locus GWAS method overcomes this limitation: because it is based on a multi-locus and multi-allele model, it can identify genome-wide QTNs in a more comprehensive fashion along with the multiple alleles. This is the other reason that we were able to detect significant QTNs with the multi-locus GWAS method.
Combining five multi-locus GWAS methods, we identified 22 common QTNs, including one stable expression QTN AX-157298785. Around these QTNs, 8 potential candidate genes were identified. Moreover, we selected elite lines for breeding higher seed protein content.
W-XL and HN conceived and designed the experiments. WL, ShiL, SX, JiZ, KZ, XL, YF, YW, JS, ZQ, and XT made major contributions to the field experiments and determination of quality traits. ShuL and ZT performed the genome sequencing. KZ, HN, and JuZ analyzed and interpreted the results. KZ and HN drafted the manuscript. All the authors contributed to the manuscript revision.
The authors gratefully acknowledge the financial support for this study provided by grants from the National Key Research and Development Program of China (2017YFD0101303-6) to W-XL and Project of Research and Development on Applied Technology of Harbin in Heilongjiang Province (2017RAXXJ019) to HN.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpls.2018.01690/full#supplementary-material
FIGURE S1 | The mean values of seed protein content for 144 lines and 4 parents across 20 environments.
FIGURE S2 | Linkage disequilibrium (LD) decays in the four-way recombinant line (FW-RIL) population. (A) The LD decay rate was estimated as the squared correlation coefficient (r2) using all pairs of SNPs located within 10 Mb of physical distance. The dashed line in red indicates the position where r2 dropped to half of its maximum value, and the dashed line in green indicates the position where r2 dropped fast and then tended to flatten. (B) Enlarged display of the area in the purple frame in (A).
TABLE S1 | Summary of details of planting conditions in field experiments.
TABLE S2 | Statistical characteristics of protein content in the parents and the FW-RIL populations grown in twenty environments.
TABLE S3 | The correlation analysis between each pair of environment.
TABLE S4 | Significant QTNs for seed protein content across 20 environments using the five multi-locus GWAS methods.
TABLE S5 | The information of pathways of 17 genes annotated in the KEGG database.
TABLE S6 | The information of orthologous protein families of 17 genes annotated in the KEGG database.
Bandillo, N., Raghavan, C., Muyco, P. A., Sevilla, M. A. L., Lobina, I. T., Dilla-Ermita, C. J., et al. (2013). Multi-parent advanced generation inter-cross (MAGIC) populations in rice: progress and potential for genetics research and breeding. Rice 6:11. doi: 10.1186/1939-8433-6-11
Belamkar, V., Farmer, A. D., Weeks, N. T., Kalberer, S. R., Blackmon, W. J., and Cannon, S. B. (2016). Genomics-assisted characterization of a breeding collection of Apios americana, an edible tuberous legume. Sci. Rep. 6:34908. doi: 10.1038/srep34908
Benjamini, Y., and Hochberg, Y. (2000). On the adaptive control of the false discovery rate in multiple testing with independent statistics. J. Educ. Behav. Stat. 25, 60–83. doi: 10.2307/1165312
Bradbury, P. J., Zhang, Z., Kroon, D. E., Casstevens, T. M., Ramdoss, Y., and Buckler, E. S. (2007). TASSEL: software for association mapping of complex traits in diverse samples. Bioinformatics 23, 2633–2635. doi: 10.1093/bioinformatics/btm308
Brummer, E. C., Graef, G. L., Orf, J., Wilcox, J. R., and Shoemaker, R. C. (1997). Mapping QTL for seed protein and oil content in eight soybean populations. Crop Sci. 37, 370–378. doi: 10.1007/BF00224058
Csanádi, G., Vollmann, J., Stift, G., and Lelley, T. (2001). Seed quality QTLs identified in a molecular map of early maturing soybean. Theor. Appl. Genet. 103, 912–919. doi: 10.1007/s001220100621
Dell’Acqua, M., Gatti, D. M., Pea, G., Cattonaro, F., Coppens, F., Magris, G., et al. (2015). Genetic properties of the MAGIC maize population: a new platform for high definition QTL mapping in Zea mays. Genome Biol. 16:167. doi: 10.1186/s13059-015-0716-z
Doyle, J. J., Doyle, J. L., and Brown, A. H. D. (1990). Analysis of a polyploid complex in Glycine with chloroplast and nuclear DNA. Aust. Syst. Bot. 3, 125–136. doi: 10.1071/SB9900125
Earl, D. A., and vonholdt, B. M. (2012) Structure harvester: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv. Genet. Resour. 4, 359–361. doi: 10.1007/s12686-011-9548-7
Evanno, G., Regnaut, S., and Goudet, J. (2005). Detecting the number of clusters of individuals using the software structure: a simulation study. Mol. Ecol. 14, 2611–2620. doi: 10.1111/j.1365-294X.2005.02553.x
Fang, C., Ma, Y., Wu, S., Liu, Z., Wang, Z., Yang, R., et al. (2017). Genome-wide association studies dissect the genetic networks underlying agronomical traits in soybean. Genome Biol. 18:161. doi: 10.1186/s13059-017-1289-9
Gaur, R., Azam, S., Jeena, G., Khan, A. W., Choudhary, S., Jain, M., et al. (2012). High-throughput SNP discovery and genotyping for constructing a saturated linkage map of chickpea (Cicer arietinum L.). DNA Res. 19, 357–373. doi: 10.1093/dnares/dss018
Huang, B. E., George, A. W., Forrest, K. L., Kilian, A., Hayden, M. J., Morell, M. K., et al. (2012). A multiparent advanced generation inter-cross population for genetic analysis in wheat. Plant Biotechnol. J. 10, 826–839. doi: 10.1111/j.1467-7652.2012.00702.x
Huang, X., Wei, X., Sang, T., Zhao, Q. A., Feng, Q., Zhao, Y., et al. (2010). Genomewide association studies of 14 agronomic traits in rice landraces. Nat. Genet. 42, 961–976. doi: 10.1038/ng.695
Hwang, E., Song, Q., Jia, G., Specht, J. E., Hyten, D. L., Costa, J., et al. (2014). A genome-wide association study of seed protein and oil content in soybean. BMC Genomics 15:1. doi: 10.1186/1471-2164-15-1
Jayaprakash, N. G., and Surolia, A. (2017). Role of glycosylation in nucleating protein folding and stability. Biochem. J. 474, 2333–2347. doi: 10.1042/BCJ20170111
Jun, T., Van, K., Kim, M. Y., Lee, S., and Walker, D. R. (2008). Association analysis using SSR markers to find QTL for seed protein content in soybean. Euphytica 162, 179–191. doi: 10.1007/s10681-007-9491-6
Kover, P. X., Valdar, W., Trakalo, J., Scarcelli, N., Ehrenreich, I. M., Purugganan, M. D., et al. (2009). A multiparent advanced generation inter-cross to fine-map quantitative traits in Arabidopsis thaliana. PLoS Genet. 5:e1000551. doi: 10.1371/journal.pgen.1000551
Kwon, Y. T., Kashina, A. S., and Varshavsky, A. (1999). Alternative splicing results in differential expression, activity, and localization of the two forms of arginyl-tRNA-protein transferase, a component of the N-end rule pathway. Mol. Cell. Biol. 19, 182–193. doi: 10.1128/MCB.19.1.182
Lee, S. H., Bailey, M. A., Mian, M. A., Carter, T. E. Jr., Shipe, E. R., Ashley, D. A., et al. (1996). RFLP loci associated with soybean seed protein and oil content across populations and locations. Theor. Appl. Genet. 93, 649–657. doi: 10.1007/BF00224058
Li, J., Li, H., Li, Y., Li, Y., Li, W., Reif, J. C., et al. (2018). Genome-wide association mapping of QTL underlying seed oil and protein contents of a diverse panel of soybean accessions. Plant Sci. 266, 95–101. doi: 10.1016/j.plantsci.2017.04.013
Liu, X., Huang, M., Fan, B., Buckler, E. S., and Zhang, Z. (2016). Iterative usage of fixed and random effect models for powerful and efficient genome-wide association studies. PLoS Genet. 12:e1005767. doi: 10.1371/journal.pgen.1005767
Lu, W., Wen, Z., Li, H., Yuan, D., Li, J., Zhang, H., et al. (2013). Identification of the quantitative trait loci (QTL) underlying water soluble protein content in soybean. Theor. Appl. Genet. 126, 425–433. doi: 10.1007/s00122-012-1990-8
Ma, X., Feng, F., Wei, H., Mei, H., Xu, K., Chen, S., et al. (2016). Genome-wide association study for plant height and grain yield in rice under contrasting moisture regimes. Front. Plant Sci. 7:1801. doi: 10.3389/fpls.2016.01801
Mackay, I. J., Bansept-Basler, P., Barber, T., Bentley, A. R., Cockram, J., Gosman, N., et al. (2014). An eight-parent multiparent advanced generation inter-cross population for winter-sown wheat: creation, properties, and validation. G3 4, 1603–1610. doi: 10.1534/g3.114.012963
Mao, T., Jiang, Z., Han, Y., Teng, W., Zhao, X., Li, W., et al. (2013). Identification of quantitative trait loci underlying seed protein and oil contents of soybean across multi-genetic backgrounds and environments. Plant Breed. 132, 630–641. doi: 10.1111/pbr.12091
Mellbye, B., and Schuster, M. (2014). Physiological framework for the regulation of quorum sensing-dependent public goods in Pseudomonas aeruginosa. J. Bacteriol. 196, 1155–1164. doi: 10.1128/JB.01223-13
Meng, L., Guo, L., Ponce, K., Zhao, X., and Ye, G. (2016a). Characterization of three rice multiparent advanced generation intercross (MAGIC) populations for quantitative trait loci identification. Plant Genome 9, 2–15. doi: 10.3835/plantgenome2015.10.0109
Meng, L., Zhao, X., Ponce, K., Ye, G., and Leung, H. (2016b). QTL mapping for agronomic traits using multi-parent advanced generation inter-cross (MAGIC) populations derived from diverse elite indica rice lines. Field Crops Res. 189, 19–42. doi: 10.1016/j.fcr.2016.02.004
Pascual, L., Desplat, N., Huang, B. E., Desgroux, A., Bruguier, L., Bouchet, J., et al. (2015). Potential of a tomato MAGIC population to decipher the genetic control of quantitative traits and detect causal variants in the resequencing era. Plant Biotechnol. J. 13, 565–577. doi: 10.1111/pbi.12282
Pathan, S. M., Vuong, T., Clark, K., Lee, J., Shannon, J. G., Roberts, C. A., et al. (2013). Genetic mapping and confirmation of quantitative trait loci for seed protein and oil contents and seed weight in soybean. Crop Sci. 53, 765–774. doi: 10.2135/cropsci2012.03.0153
Perneger, T. V. (1998). What’s wrong with bonferroni adjustments. BMJ 316, 1236–1238. doi: 10.1136/bmj.316.7139.1236
Pritchard, J. K., Stephens, M., Rosenberg, N. A., and Donnelly, P. (2000). Association mapping in structured populations. Am. J. Hum. Genet. 67, 170–181. doi: 10.1086/302959
Pritchard, J. K., Wen, X., and Falush, D. (2009). Documentation for Structure Software: Version 2.3. Chicago, IL: The University of Chicago Press.
Qi, Z., Hou, M., Han, X., Liu, C., Jiang, H., Xin, D., et al. (2014). Identification of quantitative trait loci (QTLs) for seed protein concentration in soybean and analysis for additive effects and epistatic effects of QTLs under multiple environments. Plant Breed. 133, 499–507. doi: 10.1111/pbr.12179
Sannemann, W., Huang, B. E., Mathew, B., and Léon, J. (2015). Multi-parent advanced generation inter-cross in barley: high-resolution quantitative trait locus mapping for flowering time as a proof of concept. Mol. Breed. 35, 1–16. doi: 10.1007/s11032-015-0284-7
Song, Q., Hyten, D. L., Jia, G., Quigley, C. V., Fickus, E. W., Nelson, R. L., et al. (2013). Development and evaluation of SoySNP50K, a high-density genotyping array for soybean. PLoS One 8:e54985. doi: 10.1371/journal.pone.0054985
Tamba, C. L., Ni, Y., and Zhang, Y. (2017). Iterative sure independence screening EM-bayesian LASSO algorithm for multi-locus genome-wide association studies. PLoS Comput. Biol. 13:e1005357. doi: 10.1371/journal.pcbi.1005357
Tian, F., Bradbury, P. J., Brown, P. J., Hung, H., Sun, Q., Flint-Garcia, S., et al. (2011). Genome-wide association study of leaf architecture in the maize nested association mapping population. Nat. Genet 159–162. doi: 10.1038/ng.746
Tuan, P. A., Zhao, S., Kim, J. K., Kim, Y. B., Yang, J., Li, C. H., et al. (2014). Riboflavin accumulation and molecular characterization of cDNAs encoding bifunctional GTP cyclohydrolase II/3,4-dihydroxy-2-butanone 4-phosphate synthase, lumazine synthase, and riboflavin synthase in different organs of Lycium chinense plant. Molecules 19, 17141–17153. doi: 10.3390/molecules191117141
Vaughn, J. N., Nelson, R. L., Song, Q., Cregan, P. B., and Li, Z. (2014). The genetic architecture of seed composition in soybean is refined by genome-wide association scans across multiple populations. G3 4, 2283–2294. doi: 10.1534/g3.114.013433
Wang, S. B., Feng, J. Y., Ren, W. L., Huang, B., Zhou, L., Wen, Y., et al. (2016). Improving power and accuracy of genome-wide association studies via a multi-locus mixed linear model methodology. Sci. Rep. 6:19444. doi: 10.1038/srep19444
Wang, Y., Li, Y., Wu, H., Hu, B., Zheng, J., Zhai, H., et al. (2018). Genotyping of soybean cultivars with medium-density array reveals the population structure and QTNs underlying maturity and seed traits. Front. Plant Sci. 9:610. doi: 10.3389/fpls.2018.00610
Warrington, C. V., Abdel-Haleem, H., Hyten, D. L., Cregan, P. B., Orf, J. H., Killam, A. S., et al. (2015). QTL for seed protein and amino acids in the benning × danbaekkong soybean population. Theor. Appl. Genet 128, 839–850. doi: 10.1007/s00122-015-2474-4
Wen, Y. J., Zhang, H., Ni, Y. L., Huang, B., Zhang, J., Feng, J. Y., et al. (2018). Methodological implementation of mixed linear models in multi-locus genome-wide association studies. Brief. Bioinform. 19, 700–712. doi: 10.1093/bib/bbw145
Xu, J., Yang, H., and Zhang, W. (2018). NADPH metabolism: a survey of its theoretical characteristics and manipulation strategies in amino acid biosynthesis. Crit. Rev. Biotechnol. 38, 1061–1076. doi: 10.1080/07388551.2018.1437387
Yalcin, B., Flint, J., and Mott, R. (2005). Using progenitor strain information to identify quantitative trait nucleotides in outbred mice. Genetics 171, 673–681. doi: 10.1534/genetics.104.028902
Zhang, J., Feng, J. Y., Ni, Y. L., Wen, Y. J., Niu, Y., Tamba, C. L., et al. (2017). pLARmEB: integration of least angle regression with empirical Bayes for multilocus genome-wide association studies. Heredity 118, 517–524. doi: 10.1038/hdy.2017.8
Zhang, Y., Ge, F., Hou, F., Sun, W., Zheng, Q., Zhang, X., et al. (2017). Transcription factors responding to Pb stress in maize. Genes 8:E231. doi: 10.3390/genes8090231
Keywords: protein content, soybean, multi-locus GWAS, QTNs, four-way recombinant inbred lines
Citation: Zhang K, Liu S, Li W, Liu S, Li X, Fang Y, Zhang J, Wang Y, Xu S, Zhang J, Song J, Qi Z, Tian X, Tian Z, Li W-X and Ning H (2018) Identification of QTNs Controlling Seed Protein Content in Soybean Using Multi-Locus Genome-Wide Association Studies. Front. Plant Sci. 9:1690. doi: 10.3389/fpls.2018.01690
Received: 15 June 2018; Accepted: 31 October 2018;
Published: 21 November 2018.
Edited by:
Yuan-Ming Zhang, Huazhong Agricultural University, ChinaReviewed by:
Dongying Gao, University of Georgia, United StatesCopyright © 2018 Zhang, Liu, Li, Liu, Li, Fang, Zhang, Wang, Xu, Zhang, Song, Qi, Tian, Tian, Li and Ning. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Wen-Xia Li, bGl3ZW54aWFuZWF1QDEyNi5jb20= Hailong Ning, bmluZ2hhaWxvbmduZWF1QDEyNi5jb20=
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.