
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Physiol. , 18 November 2021
Sec. Invertebrate Physiology
Volume 12 - 2021 | https://doi.org/10.3389/fphys.2021.742871
 Govindaraju Ramkumar1
Govindaraju Ramkumar1 Ramasamy Asokan1†
Ramasamy Asokan1† N. R. Prasannakumar2
N. R. Prasannakumar2 B. Kariyanna1
B. Kariyanna1 Sengodan Karthi3
Sengodan Karthi3 Mona S. Alwahibi4
Mona S. Alwahibi4 Mohamed Soliman Elshikh4
Mohamed Soliman Elshikh4 Ahmed Abdel-Megeed5
Ahmed Abdel-Megeed5 Aml Ghaith6
Aml Ghaith6 Sengottayan Senthil-Nathan3*
Sengottayan Senthil-Nathan3* Kandaswamy Kalaivani7
Kandaswamy Kalaivani7 Wayne Brian Hunter8
Wayne Brian Hunter8 Patcharin Krutmuang9,10
Patcharin Krutmuang9,10The South American pinworm Tuta absoluta (Meyrick) (Family: Gelechiidae) is one of the most devastating lepidopteran pests in the developing countries of South America, Africa, and Asia. This pest is classified as the most serious threat for tomato production worldwide. In the present study, we analyzed RNAi-mediated control through exogenously applied dsRNA delivery on tomato. The dsRNA treatments were made to target the juvenile hormone binding protein and the v-ATPase B. Both mRNA targets were cloned, validated by sequencing, and used to produce each dsRNA. After treatments the relative transcript expression was analyzed using qRTPCR to assess to efficacy of RNAi. A leaf-dip assay was used to provide late 2nd instar larvae three feeding access periods: 24, 48, and 72 h, to evaluate the effect of gene silencing of each target. Larvae were fed tomato leaves coated with five different RNAi concentrations (10, 20, 30, 40, and 50 micrograms/centimeter-squared), that suppressed two genes (juvenile hormone protein, JHBP, and vacuolar-type adenosine triphosphatase enzyme, v-ATPase). Treatments with dsRNA showed a significant increase in mortality at 24, 48, and 72 h after ingestion (P < 0.01, α = 0.05), along with reduced leaf damage, and increased feeding deterrence. The results suggest that these two RNAi products may provide a suitable treatment for control of this and other lepidopteran pests.
Tuta absoluta Meyrick (Lepidoptera: Gelechiidae) is an oligophagous pest infesting many Solanaceous crops (Global Distribution Map: CABI, Campos et al., 2017; Biondi et al., 2018; Rwomushana et al., 2019). Since the 1960s, this moth has become one of the key pests of tomato in South America (Garcia and Espul, 1982). In Europe, T. absoluta was first reported in Spain in late 2006. Thereafter, it was reported in many countries including India (Shashank et al., 2014). Cost-benefit analysis showed that T. absoluta significantly increased costs of pest management. Its primary host is tomato, although potato, brinjal, common bean, and various wild Solanaceous plants are also suitable hosts. Synthetic pesticides are commonly used for pest control worldwide (Guedes et al., 2019). Application of these chemicals against T. absoluta has been reported with little success, mainly because the pest has developed resistance (Siqueira et al., 2000; Senthil-Nathan, 2020). Efforts to develop botanical pesticides, like citrus peel extract have met with limited success (Senthil-Nathan, 2013; Miresmailli and Isman, 2014; Campolo et al., 2017). Development of alternative methods for pest control like RNAi biopesticides provide highly specific pesticides that do not harm parasitoids, pollinators, or predators (Chen et al., 2018; Niu et al., 2018; Christiaens et al., 2020; Fletcher et al., 2020; Kunte et al., 2020; Taning et al., 2020; Yan et al., 2020a,b; Sarmah et al., 2021). Research also shows that RNAi biopesticides can be exogenously applied in liquid sprays (Dalakouras et al., 2016; Koch et al., 2016; McLoughlin et al., 2018; Dubrovina and Kiselev, 2019; Dubrovina et al., 2019, 2020; Jalaluddin et al., 2019; Mezzetti et al., 2020) or bound with a carrier-like clay or nanotubes in sprays (Worrall et al., 2019; Fletcher et al., 2020) or applied in water or clay pellets as a soil treatment (Ghosh et al., 2018) or as plant-expressed silencing, which has met with significant success in field crops (Younis et al., 2014; Bramlett et al., 2020; Das and Sherif, 2020; Veillet et al., 2020).
Juvenile Hormone (JH) is essential for regulating the maturation, reproduction, and development of insects. JH inhibits the metamorphosis, leading to growth arrest of insects in their pre-metamorphosis stage, induces insect diapauses, and affects the migratory behavior of insects. It is transported into the target cells via JH binding protein (JHBP) (Gilbert et al., 2000). It is present throughout late embryonic and larval development. Changing ratios of JH/20E regulate molting to the next developmental stage and thereby allow for continued growth of insect larvae (Riddiford, 1994).
Insect vacuolar ATPase synthase genes have been compared across many insect orders (Pan et al., 2017). RNA interference, which prompts specific gene silencing through the delivery of homologous double-stranded RNA (dsRNA) fragments, is referred to as the “trigger” (Fire et al., 1998; Mello and Conte, 2004). Application of RNAi to manage insect pests or viral pathogens is widely supportive of integrated pest management strategies and shows great potential (Bramlett et al., 2020; Christiaens et al., 2020; Das and Sherif, 2020). Sarmah et al. (2021) report that while Tuta absoluta is sensitive to RNAi treatments demonstrating significantly increased mortality when ingesting the dsRNA made to the alphaCOP (αCOP) (Coatomer subunit alpha protein) mRNA transcript, there was no significant increase in mortality when treated individuals were fed on by the mirid predator Nesidiocoris tenuis (Hemiptera: Miridae). Thus, they concluded that RNAi-mediated control of T. absoluta would be a safe addition to biological control programs as it would not negatively affect the pest's natural enemies (Sarmah et al., 2021).
Functional gene studies have provided significant advances in understanding insect physiology across many orders of arthropods including Coleoptera, Diptera, Hemiptera, Hymenoptera, and Lepidoptera (Chaitanya et al., 2017). In the present study we have chosen to silence the genes of v-ATPase B and JHBP, which are involved in the growth and development of insects, as effective targets for the management of pests.
Tuta absoluta was obtained from the Division of Biotechnology, ICAR-Indian Institute of Horticultural Research, Bangalore, Karnataka, India. The cultures were maintained on tomato leaves at a 28 ± 1°C temperature, 60–70% relative humidity, and 14:10 h of light: dark photoperiod in the laboratory.
Genes encoding v-ATPase B and JHBP genes were chosen based on previous successful reports of RNAi used for insect control (Chaitanya et al., 2017). Since no sequence information was available for T. absoluta genes, degenerative primers were designed based on conserved amino acid sequence regions from aligned homologs of Plutella xylostella (JN410829), Manduca sexta (S56567), and Bombyx mori (NM_001043483). Based on these orthologous genes, the mRNA for the complete v-ATPase B coding sequence was estimated to be around 1,500 bp (Supplementary Table 1).
Target transcripts were amplified from cDNA using a nested PCR-based method with degenerate primer pairs in a 20 μL reaction containing 2 μg of cDNA, 3 mM of MgCl2, 100 μM of dNTP, 1 μM of each primer, and 2 U of Taq DNA polymerase (Bioline reagents, Germany). Amplifications were done in a PepseqTM thermo cycler, programmed to cycle at 95°C for 5 min, and followed by 35 cycles of 95°C for 30 s, 60°C for 30 s, 72°C for 15 s, and a final cycle at 72°C for 10 min. Amplification products were analyzed by gel electrophoresis (1.5% agarose gels, voltage 100 mV, for 30 min); fragments were excised, purified using a NucleoSpin® Gel and PCR Clean-up kitTM (Fermentas, GmbH, Germany), ligated into a TA cloning vector (PTZ57R/TB) (GenJET™ Plasmid MiniPrep kit), and used to transform the DH5α E. coli strain as per the manufacturer's protocol. After blue-white colonies were screened, plasmids were isolated from “white” colonies using a GenJET™ Plasmid MiniPrep kit (Fermentas, GmbH, Germany) and were sequenced (XCelris Labs, Ahmadabad, India).
The phylogenetic tree analysis was performed using MEGAX 11.0 software in the NCBI public database. Significant Lepidoptera species included, but were not limited to: Spodoptera littoralis; Plutella xylostella, Helicoverpa armigera (Noctuidae); Bombyx mori (Bombycidae); Galleria mellonella (Pyralidae); and Amyelois transitella (Pyralidae).
Unmodified, canonical syntaxin-1A dsRNA was synthesized using the Ambion® MEGAscript® RNAi Kit (Ref. No. AM1626) per manual instructions. dsRNA was uniquely designed with specific primers along with T7-polymerase promoter sequences. The reaction volume was made up to 50 μl, v-ATPase B and JHBP plasmid clones were used for the DNA template with primer annealing at 63°C /40 s. The amplified products were run on 1.3% agarose gel, expected bands were eluted, and then were used as templates (1 μg) for synthesis of dsRNA following the manufacturer's protocol (Thermo Scientific, Germany). Finally, dsRNAs were quantified using Thermo NanoDropTM (Thermo Scientific) and verified by agarose gel electrophoresis (Rebijith et al., 2015; Chaitanya et al., 2017). The dsRNA for v-ATPase was 192 nt without T7, and the JHBP was 136 nt (Supplementary Table 1).
Tomato leaflet feeding bioassays were carried out as described by Rebijith et al. (2015) and Chaitanya et al. (2017) with slight modifications. Briefly, a fresh and young tomato leaf (Lycopersicon esculentum Mill) was rinsed in 1% Triton-X, rinsed with double-ionized distilled water, and then dried and placed on moist cotton in a Petri dish. The leaflets were treated with 200 μl of the solution topically applied with the following dsRNA concentrations (10, 20, 30, 40, and 50 μg/cm2) and spread across the leaf surface using a fine hairbrush. The treated leaves were allowed to sit until dry, ~15–20 min. Then, five T. absoluta larvae (late 2nd instars) were transferred to cages with the dsRNA-coated leaves, with three leaves per treatment. Experiments were replicated three times for a total of 45 insects per treatment. Control received nuclease free water applied in the same manner. Mortality was recorded after 24, 48, and 72 h of feeding access. Extra cohort cages provided a source of live insects for qPCR analyses that were sampled over time from each treatment concentration.
RNA was extracted from 100 mg of fourth instar T. absoluta larvae using an Isolate II RNA mini kit (Bioline reagents, Germany). RNA were quantified using a NanoDropTM Lite Spectrophotometer (Thermo Scientific, Germany) and further analyzed by electrophoresis in 1.5% denaturing agarose gels. The cDNA was synthesized using a Bioline kit manufacture protocol, taking 2.0 μg of RNA and adding oligo- (dT)18 primers. The mixtures were incubated at 65°C for 5 min and immediately cooled in ice. A total of 5 × reverse transcriptase buffer, 2.5 mM of dNTPs, and 10 U/μl of RNase inhibitor were added into the tube. The mixtures were incubated at 42°C for 90 min. Finally, reverse transcriptase was added and incubated at 72°C for 15 min as per the manufacturer's instructions (Bioline, Germany).
The v-ATPase subunit B and JHBP genes were assessed by RT-qPCR (Supplementary Table 1). All expression studies were carried out following MIQE. Live larvae were used for RNA isolation using the MyTaq™ One-Step RT-PCR Kit (Catalog No. BIO-65049, Meridian Bioscience™) as per the manufacturer's protocol. In brief, samples were diluted 1:5 before RT-qPCR. The final reaction volume was adjusted to 20 μl with RNAse free water and SYBR Green (TaKaRa, Japan). The cDNA from all samples was prepared as normalized concentrations of 5 ng/μl in 10 μl, then diluted 1:5 before RT-qPCR reactions as per the instructions. All selected primers used a 10-μM scale with β-actin (KU872540) as the reference gene (Pfaffl, 2001; Pfaffl et al., 2002). The expressed β actin constitutively was used for loading normalization. RT-qPCR was carried out with the following conditions, viz., 95°C for 5 min, followed by 40 amplification cycles at 95°C for 30 s, and 60°C for 1 min in a Light Cycler 480 II (Roche Applied Science, Switzerland). Relative expression was calculated by the 2−ΔΔct method (Pfaffl et al., 2002).
Probit studies were performed using SPSS v 16.0. The qRT-PCR gene expression data were analyzed using GraphPad Prism 5.0 software (www.graphpad.com). Significant differences analyzed by one-way ANOVA, followed by post-hoc Tukey (P ≤ 0.05). The correlation between mortality and downregulation was analyzed. T-tests for independent samples or Mann–Whitney U-tests, depending on data distribution, were used to test for significant differences in expression levels (ΔΔCt values) of the target genes between the experimental and control.
v-ATPase subunit B and JHBP gene cDNA sequencing resulted in 851 and 584 bp products, respectively. NCBI-BLASTX analysis indicated a 98% amino acid sequence match with Plutella xylostella sequences (Rebijith et al., 2015). Our target gene sequences and alignments were deposited at NCBI (v-ATPase B, Accession number; MN414200; JHBP Accession number; OK066277). The sequences of v-ATPase B and JHBP were clustered with publicly available sequences from Lepidoptera, BLASTn, NCBI, and the nr database. Significant identified Lepidoptera species in the NCBI public database included Spodoptera littoralis (Noctuidae); Plutella xylostella (Plutellidae), Helicoverpa armigera (Noctuidae) (Ni et al., 2017); Bombyx mori (Bombycidae); Galleria mellonella (Pyralidae); and Amyelois transitella (Pyralidae).
Three T. absoluta larvae were given feeding access periods of 24 and 48 h on single leaflets (n = 3), with three leaflets per treatment (n = 9), per concentration of dsRNA triggers (10, 20, 30, 40, and 50 μg/cm2), of v-ATPase B or JHBP sequences, topically applied to leaflets. Significant larval mortality was observed in larvae feeding at the increasing concentrations of each dsRNA (Figure 1, JHBP; Figure 2, v-ATPase B and LC50 values of 3.426 and 4.121 μg/μl; 5.126). The increased mortality rates were observed at all three feeding access periods, 24, 48, and 72 h. The treatment leaf damage decreased as the dsRNA concentration increased (Figure 3). The delivery of dsRNA triggers at different concentrations (10, 20, 30, 40, and 50μg/cm2) per treatment were sampled at three time-points during the feeding access period (24, 48, and 72 h). Results showed that the expression of both v-ATPase B and JHBP transcripts decreased progressively with increasing concentrations of dsRNA represented in Relative Fold Change per treatment (Figures 4, 5). The qRT-PCR analysis showed that relative expression of each target gene was significantly downregulated (~3.1 fold for JHBP; ~2.6 fold for vATPase B).
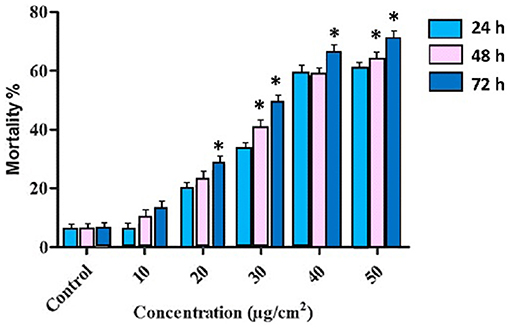
Figure 1. Percentage of mortality rates of T. absoluta larvae (late 2nd instars) post ingestion of JHBP dsRNA through treated leaves. Mortality was positively correlated with dsRNA concentration and feeding access time period. The mortality is averaged across three trials each with three biological replicates. The three time-points, 24, 48, and 72 h, were sampled across each dsRNA concentration treatment. One-way ANOVA followed by post-hoc Tukey's multiple comparison test were completed. Error bars indicate standard errors of each mean values. Asterisk (*) shows statistically significant at various time intervals at different concentrations (P < 0.05).
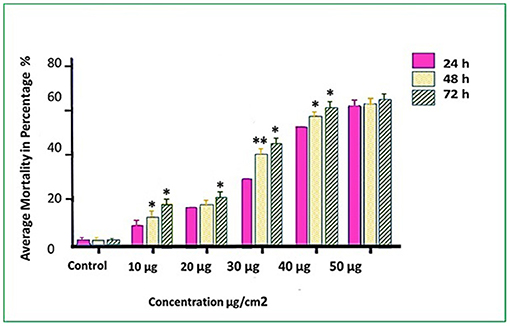
Figure 2. Mortality rates (%) of T. absoluta larvae (late 2nd instars) post feeding access period on v-ATPase B dsRNA-treated leaves. Observed mortality was recorded every 24, 48, and 72 h. The average values were obtained over three biological replications. One-way ANOVA followed with Tukey's multiple comparison (P < 0.05) were completed. Error bars are the SE ± means-averaged mortality across three trials each with three biological replicates. Asterisk shows statistically significant in various time intervals at different concentrations (*P < 0.05 and **P < 0.01).

Figure 3. Single leaf feeding assay. There was an observed correlation between decreased feeding damage to leaves with increasing dsRNA concentrations (A) 10 μg dsRNA /cm2, up to (E) 50 μg dsRNA /cm2. Individual leaves were coated with dsRNA solution at different concentrations of: (A) 10; (B) 20; (C) 30; (D) 40; (E) 50 μg/cm2 leaf. The dsRNA concentration of treated leaves at 10 μg/cm2 had >50% of the leaf surface eaten, and larger larvae than the 50 μg/cm2 treatment, while the dsRNA concentrations of 20–50 (μg/cm2) had <50% leaf damage.
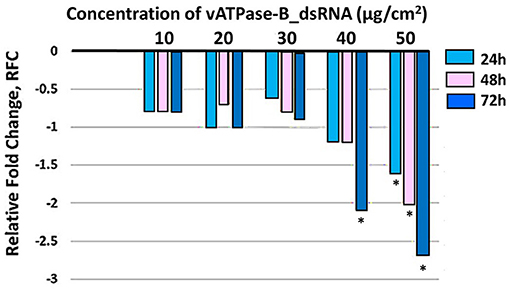
Figure 4. Relative fold change in expression of v-ATPase B transcript in T. absoluta larvae post feeding post treatment. Comparing relative expression of controls (set to “0,” to all other concentration treatments, at each of three time-points during feeding access period). RT-qPCR was used to quantify expression levels with internal control (β actin). Treatment concentrations were 10, 20, 30, 40, and 50μg/ cm2 leaf). Relative expression levels were determined with respect to control larvae fed on untreated tomato leaves. Analysis within each time-point across treatments compared to control was carried out by a one-way ANOVA (*P< 0.0001) multiple comparison test (n = 9) biological replicates, with three technical replicates.
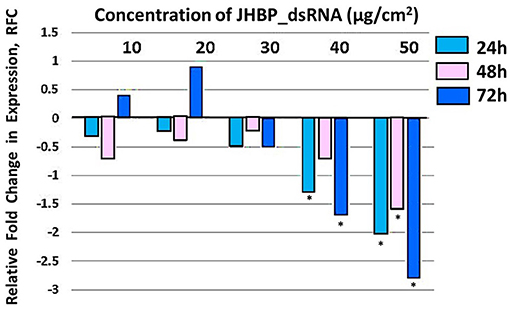
Figure 5. Relative fold change in expression of JHBP transcript in T. absoluta larvae, post feeding access on JHBP-dsRNA-treated leaf. RT-qPCR was used to access the expression levels at 24, 48, and 72 h post feeding access periods on dsRNA treatments at different concentrations (10, 20, 30, 40, and 50 μg/cm2 leaf). Relative expression levels were determined with respect to expression in the control at each time-point. Analysis was carried out by one-way ANOVA (*P < 0.0001); multiple comparison (n = 9), biological replicates with three technical replicates.
There is an urgent need to develop alternative strategies for Tuta absoluta pest control. In the present study, we demonstrate that oral delivery and RNAi-based silencing of JHBP and v-ATPase B transcripts cause significantly increased mortality in T. absoluta. Juvenile Hormone (JH) regulation is essential for development and reproductive maturation in insects (REF). In hemolymphs, JH appears complexed with a glycoprotein, the juvenile hormone-binding protein (JHBP), which serves as a carrier to release the hormone to target cells at appropriate developmental points. RNAi silencing of JHBP is reported to significantly increase mortality in several lepidopteran pests including Helicoverpa armigera (Lepidoptera: Noctuidae) fed on transgenic cotton. However, the JH gene family has not been extensively studied with only a few genes thus far demonstrated to be efficient targets for pest control (Yu et al., 2013).
The function of v-ATPase plays an essential role in the Lepidoptera midgut by keeping the midgut lumen alkaline and energizing secondary amino acid absorption. It is present at high density across the plasma membrane (Vitavska et al., 2003, 2005). The major challenge to implementing an effective RNAi strategy for controlling agricultural pests involves reliable delivery of dsRNA into the insects and choice of effective target genes that can confer pest protection. The use of RNAi in crop production requires delivery systems to provide dsRNA continuously as a diet component that is ingested by insects, either through transgenic plants expressing hairpin triggers, or by application of exogenous dsRNA by topical sprays, soil applications, baits, etc. (see reviews: Bramlett et al., 2020; Dubrovina et al., 2020; Fletcher et al., 2020; Kunte et al., 2020; Samada and Tambunan, 2020). Our results support previous RNAi studies on T. absoluta which demonstrated a high sensitivity to dsRNA triggers including Camargo et al. (2016) who first showed that RNAi of v-ATPase A could reduce T. absoluta in treated tomato. RNAi caused suppression and significant mortality of T. absoluta after feeding. And the report by Bento et al. (2020) demonstrated increased mortality of larvae T. absoluta fed on diets containing bacterial-expressed dsRNA to six different target genes (juvenile hormone inducible protein (JHP); juvenile hormone epoxide hydrolase protein (JHEH); ecdysteroid 25-hydroxylase (PHM); chitin synthase A (CHI); carboxylesterase (COE); and arginine kinase (AK). Finally, Rahmani and Bandani (2021) reported on the RNAi efficiency of silencing v-ATPase A in T. absoluta as an effective biopesticide. In the lepidopteran Helicoverpa armigera similar results using RNAi silencing of JHBP were reported (Ni et al., 2017).
Success of RNAi for pest control is dependent upon the choice of suitable gene target(s) (Terenius et al., 2011). Inhibition of RNAi activity in some pests is caused by excessive enzymes produced in the mouth and/ or gut of the target insects (Allen and Walker, 2012). Additionally the strength of the RNAi activity is dependent upon the concentration of dsRNA absorbed into the cells which are expressing the mRNA targeted for silencing (Terenius et al., 2011; Burand and Hunter, 2013; Scott et al., 2013). For chewing insects, exogenously applied or plant-absorbed dsRNA can be effectively introduced into the insect system (Koch et al., 2016; Faustinelli et al., 2018; Ghosh et al., 2018; Dubrovina and Kiselev, 2019; Dubrovina et al., 2019, 2020). While 130 lepidopteran genes have been screened for RNAi silencing, only 48% of these genes were silenced at a significant level, while 14% of the attempts resulted in failure (Terenius et al., 2011). Rebijith et al. (2015) reported that oral delivery of dsRNA, when effective, offers the best prospects for pest control under field conditions.
Modern agricultural biotechnologies are the most likely solution to growing demands for food, feed, and fibers, providing safer, more specific management of pathogens and pests (Adeyinka et al., 2020; Bramlett et al., 2020; Kunte et al., 2020; Raybould and Burns, 2020; Yan et al., 2020a,b; Sarmah et al., 2021). Furthermore, the increasing public acceptance and safety record (Kleter, 2020; Papadopoulou et al., 2020) continue to provide evidence for their adoption in the management of insect pests (Khalid et al., 2017; Sinisterra-Hunter and Hunter, 2018; ISAAA, 2019; Jalaluddin et al., 2019; Samada and Tambunan, 2020). Our results further support the concept of using RNAi to improve the management of T. absoluta (Camargo et al., 2016; Rahmani and Bandani, 2021; Sarmah et al., 2021) and other lepidopteran pests (Ni et al., 2017; Bramlett et al., 2020; Yan et al., 2020a). The capacity to use RNAi to reduce T. absoluta through exogenously treated leaves, plants, or diets with bacterial-expressed dsRNA provides ample evidence for moving forward toward commercialization of RNAi biopesticides for the management of this economically devastating lepidopteran pest. Furthermore, development of an exogenously applied treatment could be readily applied to many crop plants making pest suppression more effective than treating just a single crop or plant species (Dubrovina and Kiselev, 2019; Jalaluddin et al., 2019). Based on these results, RNAi strategies could be effective in targeting larvae of T. absoluta, with either exogenously applied, or plant-expressed double-stranded RNAs (Burand and Hunter, 2013; Scott et al., 2013; Younis et al., 2014; Zhang et al., 2015; Camargo et al., 2016; Chen et al., 2018; Jalaluddin et al., 2019; Bramlett et al., 2020; Samada and Tambunan, 2020).
RNAi for specific-target gene silencing through administration of double-stranded RNA (dsRNA) has been a useful tool for developing management of insect pests (Huvenne and Smagghe, 2010; Niu et al., 2018; Adeyinka et al., 2020; Christiaens et al., 2020; Romeis and Widmer, 2020; Yan et al., 2020a,b). In this study, we produced dsRNA for v-ATPase and JHBP from T. absoluta. Our results show that exogenous application on tomato leaves and oral ingestion of these dsRNA triggers to v-ATPase and JHBP mRNA successfully induced RNAi silencing resulting in a significant increase in larval mortality (50 μg dsRNA /cm2 leaf). An additional benefit from RNAi biopesticides are their demonstrated specificity to the pest target, while not harming beneficial non-target insects that may feed on treated pests, thus protecting predators (Sarmah et al., 2021), parasitoids, and pollinators like bees (Hunter et al., 2010; Tan et al., 2016; Vogel et al., 2019). With all these breakthroughs in RNAi for pest management, our study identifies two dsRNA triggers, v-ATPase B and JHBP, that may provide suitable targets for development of RNAi-based management of Tuta absoluta, a devastating global lepidopteran pest.
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding authors.
GR, RA, and SS-N designed the research plan, drafted, revised, and formatted the manuscript. GR, RA, and BK performed the experimental works and data compilation. GR, RA, SK, BK, NP, WH, MA, ME, AA-M, AG, KK, and PK coordinated the work and discussed the results. All authors have read and agreed to the published version of the manuscript.
The authors are grateful to DST-SERB, Government of India for providing financial support of this research (Grant No. PDF-LS/2017/001639; dated: 06/10/2017).
Mention of trade names or commercial products herein is solely for the purpose of providing specific information and does not imply recommendation or endorsement, to the exclusion of other similar products or services by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The authors thank the Division of Biotechnology, ICAR-Indian Institute of Horticultural Research (IIHR), Bangalore for providing their infrastructural facility. The authors extend their appreciation to the Researchers Supporting Project number (RSP-2021/173), King Saud University, Riyadh, Saudi Arabia.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphys.2021.742871/full#supplementary-material
Adeyinka, O. S., Riaz, S., Toufiq, N., Yousaf, I., Bhatti, M. U., Batcho, A., et al. (2020). Advances in exogenous RNA delivery techniques for RNAi mediated pest control.Mol. Biol. Rep. 47, 6309–6319. doi: 10.1007/s11033-020-05666-2
Allen, M. L., and Walker, W. B. (2012). Saliva of Lygus lineolaris digests double stranded ribonucleic acids. J. Insect Phys. 58, 391–396. doi: 10.1016/j.jinsphys.2011.12.014
Bento, F. M., Marques, R. N., Campana, F. B., Demétrio, C. G., Leandro, R. A., Parra, J. R. P., et al. (2020). Gene silencing by RNAi via oral delivery of dsRNA by bacteria in the South American tomato pinworm, Tuta absoluta. Pest Manag. Sci. 76, 287–295. doi: 10.1002/ps.5513
Biondi, A., Guedes, R. N. C., Wan, F. H., and Desneux, N. (2018). Ecology, worldwide spread and management of the invasive South American tomato pinworm, Tuta absoluta: past, present, and future. Annu. Rev. Entomol. 63, 239–258. doi: 10.1146/annurev-ento-031616-034933
Bramlett, M., Plaetinck, G., and Maienfisch, P. (2020). RNA-based biocontrols-a new paradigm in crop protection. Engineering 6, 522–527. doi: 10.1016/j.eng.2019.09.008
Burand, J. P., and Hunter, W. B. (2013). RNAi: future in insect management. J. Invertebr. Pathol. 112, 68–74. doi: 10.1016/j.jip.2012.07.012
Camargo, R. A., Barbosa, G. O., Possignolo, I. P., Peres, L. E. P., Lam, E., Lima, J. E., et al. (2016). RNA interference as a gene silencing tool to control Tuta absoluta in tomato (Solanum lycopersicum) PeerJ. 4:e2673. doi: 10.7717/peerj.2673
Campolo, O., Cherif, A., Ricupero, M., Siscaro, G., Grissa-Lebdi, K., Russo, A., et al. (2017). Citrus peel essential oil nano-formulations to control the tomato borer, Tuta absoluta: chemical properties and biological activity. Sci. Rep. 7:13036. doi: 10.1038/s41598-017-13413-0
Campos, M. R., Biondi, A., Adiga, A., Guedes, R. N. C., and Desneux, N. (2017). From the Western Palaearctic region to beyond: Tuta absoluta 10 years after invading Europe. J. Pest Sci. 90, 787–796. doi: 10.1007/s10340-017-0867-7
Chaitanya, B. N., Asokan, R., Sita, T., Rebijith, K. B., Ram Kumar, P., and Krishna Kumar, N. K. (2017). Silencing of JHEH and EcR genes of Plutella xylostella (Lepidoptera: Plutellidae) through double stranded RNA oral delivery. J. Asia-Pacific Entomol. 20, 637–643. doi: 10.1016/j.aspen.2017.03.020
Chen, J. X., Lyu, Z. H., Wang, C. Y., Cheng, J., and Lin, T. (2018). RNA interference of a trehalose-6-phosphate synthase gene reveals its roles in the biosynthesis of chitin and lipids in Heortia vitessoides (Lepidoptera: Crambidae). Insect Sci. 27, 1–12. doi: 10.1111/1744-7917.12650
Christiaens, O., Niu, J., and Taning, C. N. T.. (2020). RNAi in insects: a revolution in fundamental research and pest control. Insects 11:415. doi: 10.3390/insects11070415
Dalakouras, A., Wassenegger, M., McMillan, J. N., Cardoza, V., Maegele, I., Dadami, E., et al. (2016). Induction of silencing in plants by high-pressure spraying of in vitro-synthesized small RNAs. Front. Plant Sci. 7:1327. doi: 10.3389/fpls.2016.01327
Das, P. R., and Sherif, S. M. (2020). Application of exogenous dsRNAs induced RNAi in agriculture: challenges and triumphs. Front. Plant Sci. 11:946. doi: 10.3389/fpls.2020.00946
Dubrovina, A. S., Aleynova, O. A., Kalachev, A. V., Suprun, A. R., Ogneva, Z. V., and Kiselev, K. V. (2019). Induction of transgene suppression in plants via external application of synthetic dsRNA. Int. J. Mol. Sci. 20:1585. doi: 10.3390/ijms20071585
Dubrovina, A. S., Aleynova, O. A., Suprun, A. R., Ogneva, Z. V., and Kiselev, K. V. (2020). Transgene suppression in plants by foliar application of in vitro-synthesized small interfering RNAs. Appl. MicroBiol. Biotechnol. 104, 2125–2135. doi: 10.1007/s00253-020-10355-y
Dubrovina, A. S., and Kiselev, K. V. (2019). Exogenous RNAs for gene regulation and plant resistance. Int. J. Mol. Sci. 20:2282; doi: 10.3390/ijms20092282
Faustinelli, P. C., Power, I. L., and Arias, R. S. (2018). Detection of exogenous double-stranded RNA movement in in vitro peanut plants. Plant Biol. 20, 444–449. doi: 10.1111/plb.12703
Fire, A., Xu, S., Montgomery, M. K., Kostas, S. A., Driver, S. E., and Mello, C. C. (1998). Potent and specific genetic interference by double stranded RNA in Caenorhabditis elegans. Nature 391, 806–811. doi: 10.1038/35888
Fletcher, S. J., Reeves, P. T., Tram Hoang, B., and Mitter, N. (2020). A perspective on RNAi-based biopesticides. Front. Plant Sci. 11:51. doi: 10.3389/fpls.2020.00051
Garcia, M. F., and Espul, J. C. (1982). Bioecology of the tomato moth (Scrobipalpula absoluta) in Mendoza, Argentine Republic. Revista de Investigaciones Agropecuarias 17, 135–146.
Ghosh, S. K. B., Hunter, W. B., Park, A. L., and Gundersen-Rindal, D. E. (2018). Double-stranded RNA oral delivery methods to induce RNA interference in phloem and plant-sap-feeding hemipteran insects. J. Vis. Exp. 135:e57390. doi: 10.3791/57390
Gilbert, L. I., Granger, N. A., and Roe, R. M. (2000). The juvenile hormones: historical facts and speculations on future research directions. Insect Biochem. Mol. Biol. 30, 617–644. doi: 10.1016/S0965-1748(00)00034-5
Guedes, R. N. C., Roditakis, E., Campos, M. R., Haddi, K., Bielza, P., Siqueira, H. A. A., et al. (2019). Insecticide resistance in the tomato pinworm Tuta absoluta: patterns, spread, mechanisms, management and outlook. J. Pest Sci. 92, 1329–1342. doi: 10.1007/s10340-019-01086-9
Hunter, W., Ellis, J., VanEngelsdorp, D., Hayes, J., Westervelt, D., and Glick, E. (2010). Large-scale field application of RNAi technology reducing Israeli acute paralysis virus disease in honey bees (Apis mellifera, Hymenoptera: Apidae). PLoS Path 6:e1001160. doi: 10.1371/journal.ppat.1001160
Huvenne, H., and Smagghe, G. (2010). Mechanisms of dsRNA uptake in insects and potential of RNAi for pest control: a review. J. Insect Phys. 56, 227–235. doi: 10.1016/j.jinsphys.2009.10.004
ISAAA (2019). “International service for the acquisition of agri-biotech applications, global status of commercialized biotech/gm crops in 2018: biotech crops continue to help meet the challenges of Increased Population and Climate Change (ISAAABrief No. 54),” in International Service for the Acquisition of Agri-Biotech Applications, Ithaca, NY.
Jalaluddin, N. S. M., Othman, R. Y., and Harikrishna, J. A. (2019). Global trends in research and commercialization of exogenous and endogenous RNAi technologies for crops. Crit. Rev. Biotechnol. 39, 67–78. doi: 10.1080/07388551.2018.1496064
Khalid, A., Zhang, Q., Mand, Y., and Li, F. (2017). Small RNA based genetic engineering for plant viral resistance: application in crop protection. Front Microbiol 8:43. doi: 10.3389/fmicb.2017.00043
Kleter, G. A. (2020). Food safety assessment of crops engineered with RNA interference and other methods to modulate expression of endogenous and plant pest genes. Pest Manag. Sci. 76, 3333–3339. doi: 10.1002/ps.5957
Koch, A., Biedenkopf, D., Furch, A., Weber, L., Rossbach, O., and Abdellatef, E. (2016). An RNAi-based control of Fusarium graminearum infections through spraying of long dsRNAs involves a plant passage and is controlled by the fungal silencing machinery. PLoS Pathog. 12:e1005901. doi: 10.1371/journal.ppat.1005901
Kunte, N., McGraw, E., and Bell, S. (2020). Prospects, challenges and current status of RNAi through insect feeding. Pest Manag. Sci. 76, 26–41. doi: 10.1002/ps.5588
McLoughlin, A. G., Wytinck, N., Walker, P. L., Girard, I. J., Rashid, K. Y., de Kievit, T., et al. (2018). Identification and application of exogenous dsRNA confers plant protection against Sclerotinia sclerotiorum and Botrytis cinerea. Sci. Rep. 8:7320. doi: 10.1038/s41598-018-25434-4
Mello, C. C., and Conte, D. (2004). Revealing the world of RNA interference. Nature 43, 338–342. doi: 10.1038/nature02872
Mezzetti, B., Smagghe, G., Arpaia, S., Christiaens, O., Dietz-Pfeilstetter, A., Jones, H., et al. (2020). RNAi: What is its position in agriculture? J. Pest Sci. 93, 1125–1130. doi: 10.1007/s10340-020-01238-2
Miresmailli, S., and Isman, M. B. (2014). Botanical insecticides inspired by plant–herbivore chemical interactions. Trends Plant Sci. 19, 29–35. doi: 10.1016/j.tplants.2013.10.002
Ni, M., Ma, W., Wang, X., Gao, M., and Dai, Y. (2017). Next-generation transgenic cotton: Pyramiding RNAi and Bt counters insect resistance. Plant Biotechnol. J. 15, 1204–1213. doi: 10.1111/pbi.12709
Niu, J., Taning, C. N. T., Christiaens, O., Smagghe, G., and Wang, J. J. (2018). Chapter one rethink RNAi in insect pest control: challenges and perspectives. Adv. Insect Physiol. 55, 1–17. doi: 10.1016/bs.aiip.2018.07.003
Pan, H., Yang, X., Bidne, B., Hellmich, R. L., Siegfried, B. D., and Zhou, X. (2017). Dietary risk assessment of V-ATPase A dsRNAs on monarch butterfly larvae. Front. Plant Sci. 8:242. doi: 10.3389/fpls.2017.00242
Papadopoulou, N., Devos, Y., Álvarez-Alfageme, F., Lanzoni, A., and Waigmann, E. (2020). Risk assessment considerations for genetically modified RNAi plants: EFSA's activities and perspective. Front. Plant Sci. 11:445. doi: 10.3389/fpls.2020.00445
Pfaffl, M. W. (2001). A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res. 29:E45. doi: 10.1093/nar/29.9.e45
Pfaffl, M. W., Horgan, G. W., and Dempfle, L. (2002). Relative Expression Software Tool (REST©) for group wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Res. 30:E36. doi: 10.1093/nar/30.9.e36
Rahmani, S., and Bandani, A. R. (2021). A gene silencing of V ATPase subunit A interferes with survival and development of the tomato leafminer, Tuta absoluta. Arch. Insect Biochem. Physiol. 106:e21753. doi: 10.1002/arch.21753
Raybould, A., and Burns, A. (2020). Problem formulation for off-target effects of externally applied double-stranded RNA-based products for pest control. Front. Plant Sci. 11:424. doi: 10.3389/fpls.2020.00424
Rebijith, K. B., Asokan, R., Ranjitha Hande, H., Krishna Kumar, N. K., and Krishna, V. (2015). RNA interference of odorant-binding protein 2 (OBP2) of the cotton aphid, Aphis gossypii (glover), resulted in altered electrophysiological responses. Appl. Biochem. Biotechnol. 15, 1869–1877. doi: 10.1007/s12010-015-1869-7
Riddiford, L. M. (1994). Cellular and molecular actions of juvenile hormones. I: general considerations and premetamorphic actions. Adv. Insect Physiol. 24, 213–273. doi: 10.1016/S0065-2806(08)60084-3
Romeis, J., and Widmer, F. (2020). Assessing the risks of topically applied dsRNA-based products to non-target arthropods. Front. Plant Sci. 11:679. doi: 10.3389/fpls.2020.00679
Rwomushana, I., Tambo, J., Pratt, C., Gonzalez-Moreno, P., Beale, T., Lamontagne-Godwin, J., et al. (2019). Tomato leafminer (Tuta absoluta): impacts and coping strategies for Africa. Evidence Note. Available online at: https://www.invasive-species.org/wp-content/uploads/sites/2/2019/04/Tuta-Evidence-Note_FINAL.pdf
Samada, L. H., and Tambunan, U. S. F. (2020). Biopesticides as promising alternatives to chemical pesticides: a review of their current and future status. Online J. Biol. Sci. 20, 66–76. doi: 10.3844/ojbsci.2020.66.76
Sarmah, N., Kaldis, A., Taning, C. N. T., Perdikis, D., Smagghe, G., and Voloudakis, A. (2021). dsRNA-mediated pest management of Tuta absoluta is compatible with its biological control agent Nesidiocoris tenuis. Insects 12:274. doi: 10.3390/insects12040274
Scott, J. G., Michel, K., Bartholomay, L. C., Siegfried, B. D., Hunter, W. B., and Smagghe, G. (2013). Towards the elements of successful insect RNAi. J. Insect Physiol. 59, 1212–1221. doi: 10.1016/j.jinsphys.2013.08.014
Senthil-Nathan, S. (2013). Physiological and biochemical effect of neem and other Meliaceae plants secondary metabolites against Lepidopteran insects. Front. Physiol. 4:359. doi: 10.3389/fphys.2013.00359
Senthil-Nathan, S. (2020). A review of resistance mechanisms of synthetic insecticides and botanicals, phytochemicals and essential oils as alternative larvicidal agents against mosquitoes. Front. Physiol. 15:91. doi: 10.3389/fphys.2019.01591
Shashank, P. R., Chandrashekar, K., Meshram, N. M., and Sreedevi, K. (2014). Occurrence of Tuta absoluta (Lepidoptera: Gelechiidae) an invasive pest from India. Ind. J. Ento. 77, 323–329. doi: 10.5958/0974-8172.2015.00070.X
Sinisterra-Hunter, X., and Hunter, W. B. (2018). Towards a holistic integrated pest management: lessons learned from plant-insect mechanisms in the field. J. Chpt. 10, 204–226. doi: 10.1201/9781315119571-10
Siqueira, H. A. A., Guedes, R. N. C., and Picanco, M. C. (2000). Cartap resistance and synergism in populations of Tuta absoluta (Lep., Gelechiidae). J. Appl. Entomol. 124, 233–238. doi: 10.1046/j.1439-0418.2000.00470.x
Tan, J. G., Levine, S. L., Bachman, P. M., Jensen, P. D., Mueller, G. M., Uffman, J. P., et al. (2016). No impact of DvSnf7 RNA on honey bee (Apis mellifera L.) adults and larvae in dietary feeding tests. Environ. Toxicol. Chem. 35, 287–294. doi: 10.1002/etc.3075
Taning, C. N. T., Arpaia, S., Christiaens, O., Dietz-Pfeilstetter, A., Jones, H., and Mezzetti, B. (2020). RNA-based biocontrol compounds: current status and perspectives to reach the market. Pest Man. Sci. 76, 841–845. doi: 10.1002/ps.5686
Terenius, O., Papanicolao, A., Garbutt, J. S., Eleftherianos, I., Huvenne, H., and Kanginakudru, S. (2011). RNA interference in Lepidoptera: an overview of successful and unsuccessful studies and implications for experimental design. J. Insect Physiol. 57, 231–245. doi: 10.1016/j.jinsphys.2010.11.006
Veillet, F., Durand, M., Kroj, T., Cesari, S., and Gallois, J. L. (2020). Precision breeding made real with CRISPR: illustration through genetic resistance. Pathogens. 20:102. doi: 10.1016/j.xplc.2020.100102
Vitavska, O., Wieczorek, H., and Merzendorfer, H. (2003). A novel role for subunit C in mediating binding of the H+V-ATPase to the actin cytoskeleton. J. Biol. Chem. 278, 18499–18505. doi: 10.1074/jbc.M212844200
Vitavska, O., Wieczorek, H., and Merzendorfer, H. (2005). The V-ATPase subunit C binds to polymeric F-actin as well as to monomeric G-actin and induces cross-linking of actin filaments. J. Biol. Chem. 280, 1070–1076. doi: 10.1074/jbc.M406797200
Vogel, E., Snatos, D., Mingels, L., Verdonckt, T.-W., and Broeck, J. V. (2019). RNA interference in insects: protecting beneficials and controlling pests. Front. Physiol. 9:1912. doi: 10.3389/fphys.2018.01912
Worrall, E. A., Bravo-Cazar, A., Nilon, A. T., Fletcher, S. J., Robinson, K. E., Carr, J. P., et al. (2019). Exogenous application of RNAi-inducing double-stranded RNA inhibits aphid-mediated transmission of a plant virus. Front. Plant Sci. 10:265. doi: 10.3389/fpls.2019.00265
Yan, S., Qian, J., and Cai, C. (2020b). Spray method application of transdermal dsRNA delivery system for efficient gene silencing and pest control on soybean aphid Aphis glycines. J. Pest Sci. 93, 449–459. doi: 10.1007/s10340-019-01157-x
Yan, S., Ren, B., Zeng, B., and Shen, J. (2020a). Improving RNAi efficiency for pest control in crop species. BioTechniques 68, 283–290. doi: 10.2144/btn-2019-0171
Younis, A., Siddique, M. I., Kim, C. K., and Lim, K. B. (2014). RNA interference (RNAi) induced gene silencing: a promising approach of hi-tech plant breeding. Int. J. Biol. Sci. 10, 1150–1158. doi: 10.7150/ijbs.10452
Yu, N., Christiaens, O., Liu, J. S., Niu, J., Kaat, C., and Silvia, C. (2013). Delivery of dsRNA for RNAi in insects: an overview and future directions. Insect Sci. 20, 4–14. doi: 10.1111/j.1744-7917.2012.01534.x
Keywords: tomato leaf miner, v-ATPase, juvenile hormone binding protein, JHBP, dsRNA, RNAi, lepidoptera, pest management
Citation: Ramkumar G, Asokan R, Prasannakumar NR, Kariyanna B, Karthi S, Alwahibi MS, Elshikh MS, Abdel-Megeed A, Ghaith A, Senthil-Nathan S, Kalaivani K, Hunter WB and Krutmuang P (2021) RNA Interference Suppression of v-ATPase B and Juvenile Hormone Binding Protein Genes Through Topically Applied dsRNA on Tomato Leaves: Developing Biopesticides to Control the South American Pinworm, Tuta absoluta (Lepidoptera: Gelechiidae). Front. Physiol. 12:742871. doi: 10.3389/fphys.2021.742871
Received: 16 July 2021; Accepted: 20 September 2021;
Published: 18 November 2021.
Edited by:
Jose Eduardo Serrão, Universidade Federal de Viçosa, BrazilReviewed by:
Frank Chidawanyika, International Centre of Insect Physiology and Ecology (ICIPE), KenyaCopyright © 2021 Ramkumar, Asokan, Prasannakumar, Kariyanna, Karthi, Alwahibi, Elshikh, Abdel-Megeed, Ghaith, Senthil-Nathan, Kalaivani, Hunter and Krutmuang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sengottayan Senthil-Nathan, c2VudGhpbEBtc3VuaXYuYWMuaW4=
†Senior author
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.