- 1Laboratory of Regenerative Medicine in Sports Science, School of Physical Education and Sports Science, South China Normal University, Guangzhou, China
- 2Laboratory of Laser Sports Medicine, School of Physical Education and Sports Science, South China Normal University, Guangzhou, China
- 3Department of Physical Education, Guangdong Pharmaceutical University, Guangzhou, China
- 4China Institute of Sport Science, Beijing, China
Objective: The objective of this paper was to study the effects of long-term exercise on circulating microRNAs (miRNAs) in human plasma.
Methods: Whole blood was collected from 10 female elite athletes with at least 5 years of training experience in a Synchronized Swimming Group (S group) and 15 female college students without regular exercise training (C group). Plasma miRNAs were then isolated, sequenced, and semi-quantified by the second-generation sequencing technology, and the results were analyzed by bioinformatics methods.
Results: We found 380 differentially expressed miRNAs in the S group compared with the C group, among which 238 miRNAs were upregulated and 142 were downregulated. The top five abundant miRNAs in the 380 miRNAs of the S group are hsa-miR-451a, hsa-miR-486, hsa-miR-21-5p, hsa-miR-423-5p, and hsa-let-7b-5p. Muscle-specific/enriched miRNAs were not significantly different, except for miR-206 and miR-486. According to the Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis, a large proportion of the differentially expressed miRNAs are targeted in cancer-related pathways, including proteoglycans in cancer and miRNAs in cancer and basal cell carcinoma. As the levels of circulating miRNAs (ci-miRNAs) are commonly known to be significantly deregulated in cancer patients, we further compared the levels of some well-studied miRNAs in different types of cancer patients with those in the S group and found that long-term exercise regulates the level of ci-miRNAs in an opposite direction to those in cancer patients.
Conclusion: Long-term exercise significantly alters the profiles of plasma miRNAs in healthy young women. It may reduce the risk of certain types of cancers by regulating plasma miRNA levels.
What are the findings?
• Long-term exercise significantly alters the plasma miRNA profile of healthy young women.
• Long-term exercise may reduce the risk of lung, breast, pancreas, esophageal, thyroid neoplasms, melanoma and cholangiocarcinoma by regulating circulating miRNAs.
• Athletes may have a higher risk of heart failure and coronary artery disease.
Introduction
MicroRNAs (miRNAs) are small non-coding RNAs with approximately 22 nucleotides in length. Their function is to degrade the targeted messenger RNAs (mRNAs) or inhibit their translation by binding to the complementary region of the mRNA molecules, which in turn participate in various biological or pathological processes (Bartel, 2004). They are found to be abundant and stable in biofluids, including blood serum/plasma (Chim et al., 2008), cerebrospinal fluid (CSF) (Baraniskin et al., 2012; Sorensen et al., 2014; Akers et al., 2015), milk (Modepalli et al., 2014), saliva (Bahn et al., 2015), and urine (Cheng et al., 2014). These miRNAs in the biofluids are called circulating miRNAs (ci-miRNAs) and may come from dead cells or as by-products of routine microvesicle secretion or specifically secreted by cells (Turchinovich et al., 2012). The levels of ci-miRNAs will change under different physio- or pathological conditions, such as pregnancy (Sanders et al., 2015), myocardial infarction (Boon and Dimmeler, 2015), muscle injury (Desai et al., 2014), liver damage (Leelahavanichkul et al., 2015), diabetes (Ortega et al., 2014), and cancer (Volinia et al., 2010).
A few studies have shown that plasma ci-miRNAs can also be affected by a single bout of acute exercise (Baggish et al., 2011; Aoi et al., 2013; Nielsen et al., 2014; Cui et al., 2016; Shah et al., 2017; Barber et al., 2019) or sustained training from 28 to 140 days (Baggish et al., 2011; Aoi et al., 2013; Nielsen et al., 2014; Li et al., 2018; Barber et al., 2019). Most of them focused on some specific ci-miRNAs, such as ci-miRNAs involved in inflammation, skeletal and cardiac muscle contractility, and hypoxia/ischemia adaptation (Baggish et al., 2011), muscle-enriched miRNAs (Aoi et al., 2013), miRNAs related to cardiovascular disease (Barber et al., 2019), and those involved in angiogenesis and inflammation and are enriched in muscle and/or cardiac tissues (Li et al., 2018). Only one study (Nielsen et al., 2014) quantified the expression levels of 188 known ci-miRNAs in the plasma of 32 healthy, trained men before and after 12 weeks of endurance exercise and found nine miRNAs expressed differently. By far, the response of whole ci-miRNAs in plasma to long-term exercise (>5 years), especially among long-term trained professional athletes, remains unknown. In this study, we chose athletes of a Synchronized Swimming Group as our subjects since the movement difference between players is small and the team members usually have years of regular exercise experience. We compared the total plasma miRNAs of 10 players from a Synchronized Swimming Group and 15 female college students without regular exercise experience, using second-generation sequencing technology, to study the impact of long-term exercise on the plasma miRNA content.
Materials and Methods
Subjects
The participants were 10 Synchronized Swimming Group members and 15 female college students of Guangdong Pharmaceutical University. All participants were informed of the methods, procedures, and risks, and then they signed an informed consent document, which was approved by the Ethics Committee of South China Normal University School of Sports Science (approval number 2017102001).
MiRNA Isolation From Plasma
Blood samples were collected around 7:00 a.m., and participants did not have breakfast or vigorous exercise for 24 h. Whole blood (10 ml) from subjects was collected via a direct venous puncture into tubes with ethylenediaminetetraacetic acid (EDTA) as an anticoagulant. Blood was put on ice for half an hour and then separated by 2,000 rpm for 10 min at 4°C. Then, the plasma was transferred to an RNase-free tube and stored at −80°C. The RNA samples were extracted with a TRIzol reagent (Life Technologies, cat. no. 15596018). The purity and concentration of the RNA samples were assessed with Nanodrop and Qubit 2.0. RNA integrity was assessed using the RNA Nano 6000 Assay Kit of the Agilent Bioanalyzer 2100 system (Agilent Technologies, Santa Clara, CA, United States) to ensure the use of qualified samples for sequencing.
MiRNA Sequencing
RNA samples undergo a series of strict quality control. Qualified samples are used for library construction. Library construction is strictly in accordance with NEBNext Ultra Small RNA Sample Library Prep Kit for Illumina, qualified libraries for high-throughput sequencing. The sequencing platform was Illumina HiSeq X Ten, and read length was single-end (SE) 50 nt.
Data Analysis
Quality Control
Raw data (raw reads) of fastq format were firstly processed through in-house perl scripts. In this step, clean data (clean reads) were obtained by removing reads containing adapter or ploy-N and low-quality reads from raw data. The reads were trimmed and cleaned by removing the sequences smaller than 15 nt or longer than 35 nt. At the same time, Q20, Q30, GC content, and the sequence duplication level of the clean data were calculated. All the downstream analyses were based on clean data with high quality.
Comparative Analysis
Using Bowtie software, analysis of the clean reads was done, respectively, with the Silva database, GtRNAdb database, Rfam database, and Repbase database for sequence alignment, filter ribosomal RNA (rRNA), transfer RNA (tRNA), small nuclear RNA (snRNA), small nucleolar RNA (snoRNA), and other ncRNAs and repeats. The remaining reads were used to detect known miRNAs and novel miRNAs predicted by comparing with Genome and known miRNAs from miRBase. Randfold software was used for novel miRNA secondary structure prediction.
Differential Expression Analysis
The expression of miRNAs in each sample was counted and normalized by the Trusted Platform Module (TPM) algorithm (Fahlgren et al., 2007). The normalization formula for TPM is as follows: TPM = Actual miRNA read count/Total miRNA read count × 1,000,000. Differential expression analysis was performed using the DESeq R package (1.10.1). The resulting P values were adjusted using the Benjamini and Hochberg’s approach for controlling the false discovery rate (FDR). MiRNAs with |log2(FC, fold change)| ≥ 1 and FDR < 0.05 found by DESeq were assigned as differentially expressed miRNAs.
Target Gene Functional Annotation
Target gene prediction was done using miRanda (Doron Betel et al., 2008) and RNAhybrid (Rehmsmeier et al., 2004). Gene function was annotated based on the following databases: Nr (NCBI non-redundant protein sequences), Pfam (Protein family), KOG/COG (Clusters of Orthologous Groups of proteins), Swiss-Prot (a manually annotated and reviewed protein sequence database), KEGG (KEGG Ortholog database), and GO (Gene Ontology).
Enrichment Analysis
A GO term enrichment analysis1 was used to identify the biological processes, molecular functions, and cellular components associated with the target genes of differential expression miRNAs. KEGG pathway enrichment analysis2 was used to identify the miRNA targets associated with signaling pathways in the S vs. the C group.
MiRNAs as Biomarkers of Certain Diseases (Study Selection Criteria)
We screened ci-miRNA biomarkers related to certain types of cancers through the Human MicroRNA Disease Database (HMDD3). Studies were included if they (i) were original research and (ii) evaluated the ci-miRNA levels in a specific disease. Studies were excluded if they were published in a language other than English and are review articles, book chapters, conference abstracts, editorials/commentaries/expert opinion, theses, or dissertations.
Results
The Expression Patterns of Ci-MiRNAs Were Highly Similar Within the Originally Divided Participants
The participants included 10 Synchronized Swimming Group members (S group) and 15 female college students from Guangdong Pharmaceutical University in China (C group). The average age of these 10 players was 19.1 years compared with 19.4 years of the control, and there was no significant age difference between these two groups (19.1 ± 2.08 vs. 19.4 ± 0.83 years, P = 0.67). The subjects were all healthy and without known diseases. C group members had no exercise habit and did not enroll in any long-term sports or dancing in the last 5 years. For details, see Supplementary Table 1. Pearson’s correlation coefficient was used to test the similarity of these samples at a global level (Figure 1). The heat map of the correlations between each sample was generated and showed distinct expression pattern between the two groups. The clustering of samples showed that the same group of samples is very similar to the groups of samples originally divided.
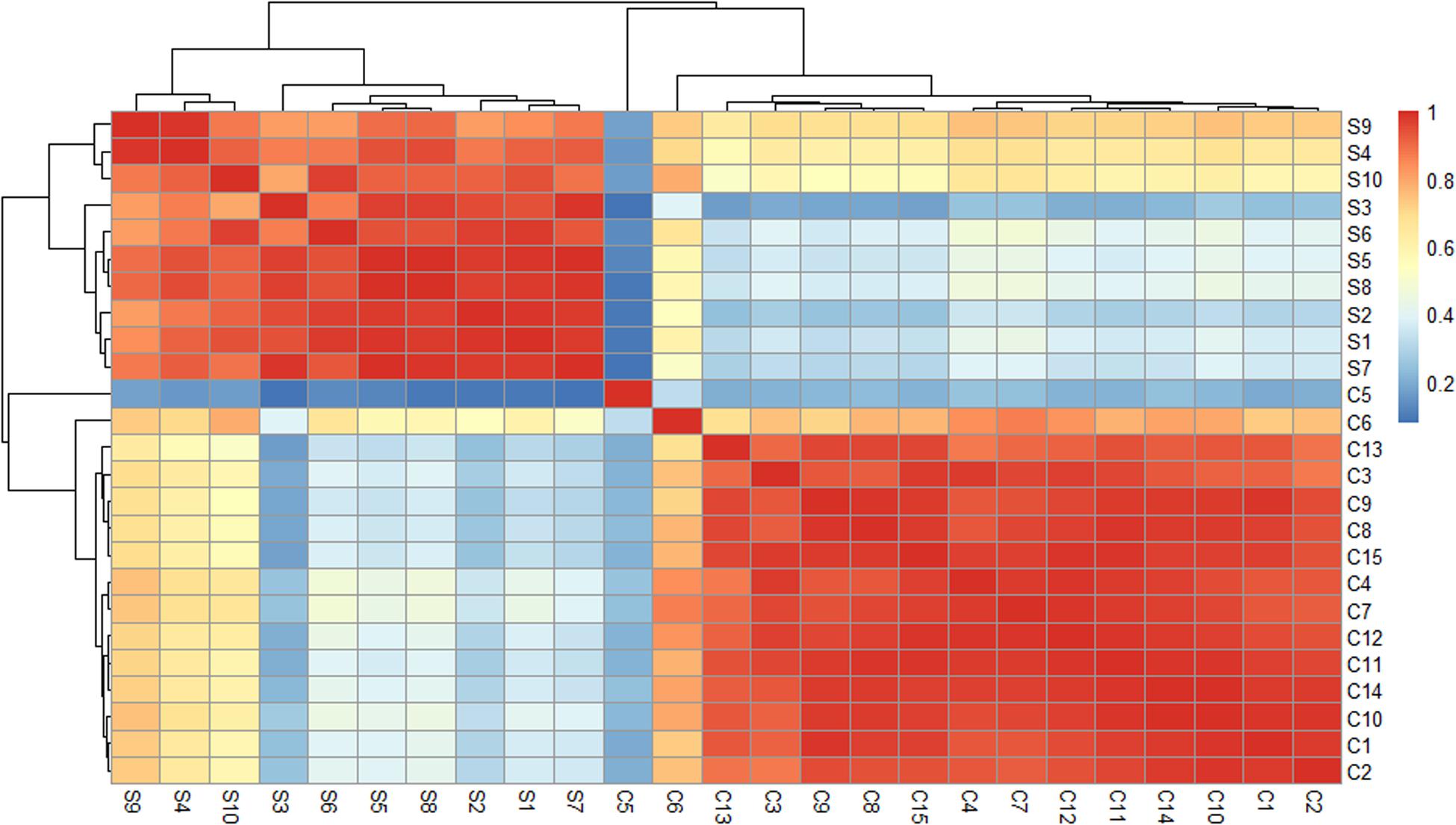
Figure 1. Heat maps of the Pearson’s correlation coefficients for different samples. When the linear relationship between the two variables is enhanced, the correlation coefficient tends to be 1 or –1. A positive correlation tends to be 1, while a negative correlation tends to be –1.
Differentially Expressed MiRNAs Between the S and C Groups
Twenty-five specimens were successfully sequenced by using Illumina Genome Analyzer II. The number of raw reads obtained per specimen ranged from 15,872,436 to 32,103,564 (average = 23,148,465). From the raw reads, an average of 22,508,866 clean reads (ranging from 15,569,728 to 31,145,335) were filtered and mapped to human genome. After analyzing and normalizing all the mapped reads in both groups, we identified the existence of 3,531 miRNAs with 1,733 known miRNAs and 1,798 novel miRNAs. Then, we applied a stringent filtering criterion to differentiate S and C (FDR < 0.05, | log2(FC)| ≥ 1) and identified 380 miRNAs that were differentially expressed (for details, see Supplementary Table 2). Specifically, 238 miRNAs were upregulated and 142 were downregulated (Figure 2A). In addition, 23 miRNAs have an extremely significant difference between the two groups (| log2(FC)| ≥ 5) (Figure 2B). Notably, all 23 miRNAs were upregulated in the S group, and the highest differentially expressed miRNA was novel-miR-1150 (| log2(FC)| = 8.09). Among the 380 differentially expressed miRNAs, the five most abundant in the S group were miR-451a, miR-486, miR-423-5p, let-7b-5p, and miR-21-5p, with | log2(FC)| = 4.77, 4.76, 2.13, 2.65, and 1.53 and TPM = 224,122, 119,250, 85,314, 43,867, and 26,667, respectively. On the other hand, the five most abundant miRNAs in the C group were miR-21-5p, let-7f-5p, miR-148a-3p, miR-423-5p, and miR-146a-5p. Taken together, there are consistent and significant differences in the plasma miRNA profiles between the athlete group and the normal college student group.
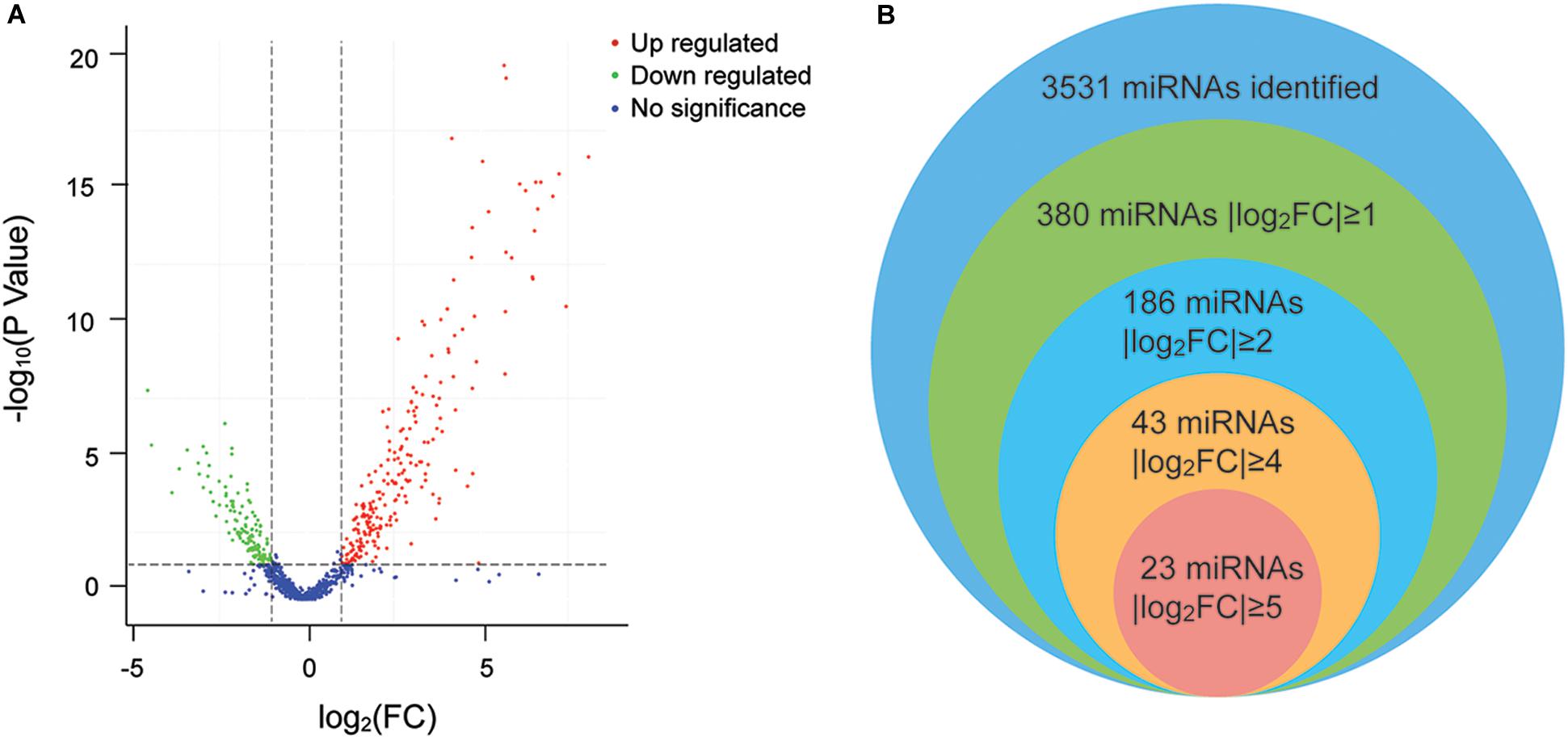
Figure 2. (A) Volcano plot of the differentially expressed miRNAs. The plot shows the log2 fold change on the X-axis vs. the adjusted P values (on the log10 scale) on the Y-axis. Red dots indicate upregulated miRNAs, green dots are downregulated miRNAs, and blue dots are miRNAs with no significant difference. (B) Venn diagram of the differentially expressed miRNAs. Each circle represents a comparison group. The overlapping part is the common miRNAs among the comparison groups.
Target Gene Prediction of Differentially Expressed MiRNAs
To explore the functions of chronic exercise induced differentially expressed miRNAs, the target genes of these miRNAs were predicted by combining miRanda and RNAhybrid to reduce the probability of false positives. From this analysis, 20,449 target genes corresponding to 3,531 miRNAs were identified. For details, see Supplementary Table 3.
Functional Annotation Analysis
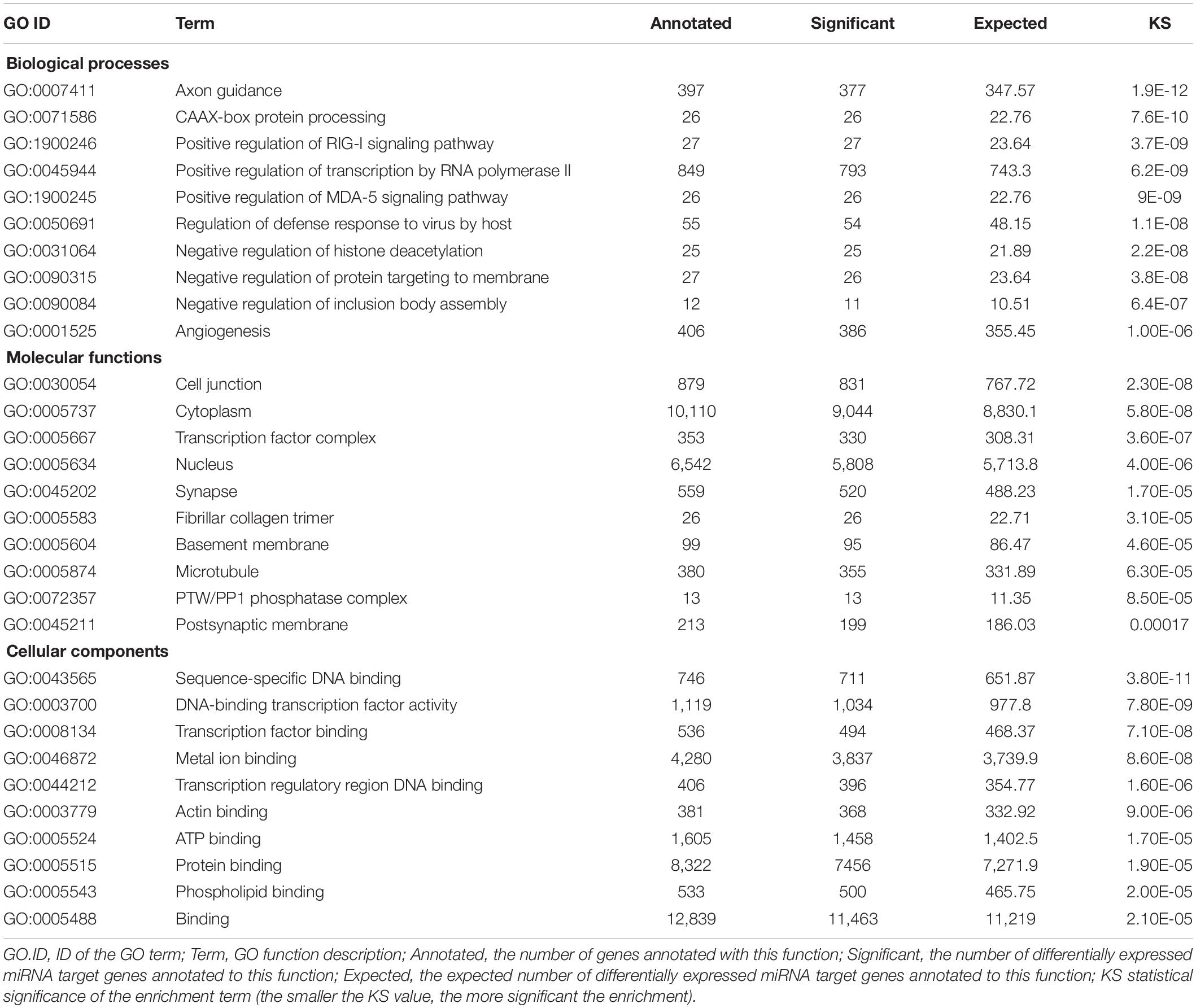
Gene ontology enrichment analysis of the 380 differentially expressed genes (DEGs) was implemented by the GOseq R packages based on Wallenius’ non-central hyper-geometric distribution. In the S group, 1,162 GO categories of biological processes, 142 of cellular component, and 203 of molecular function were found to be significantly affected (Kolmogorov–Smirnov, KS < 0.05). The 10 most significantly enriched GO categories of biological processes, cellular component, and molecular function are listed in Table 1.
Pathway Annotation Analysis
Pathway annotation analysis of the differentially expressed miRNA target genes was done using the KEGG database see text footnote 2). The results are shown in Figure 3.
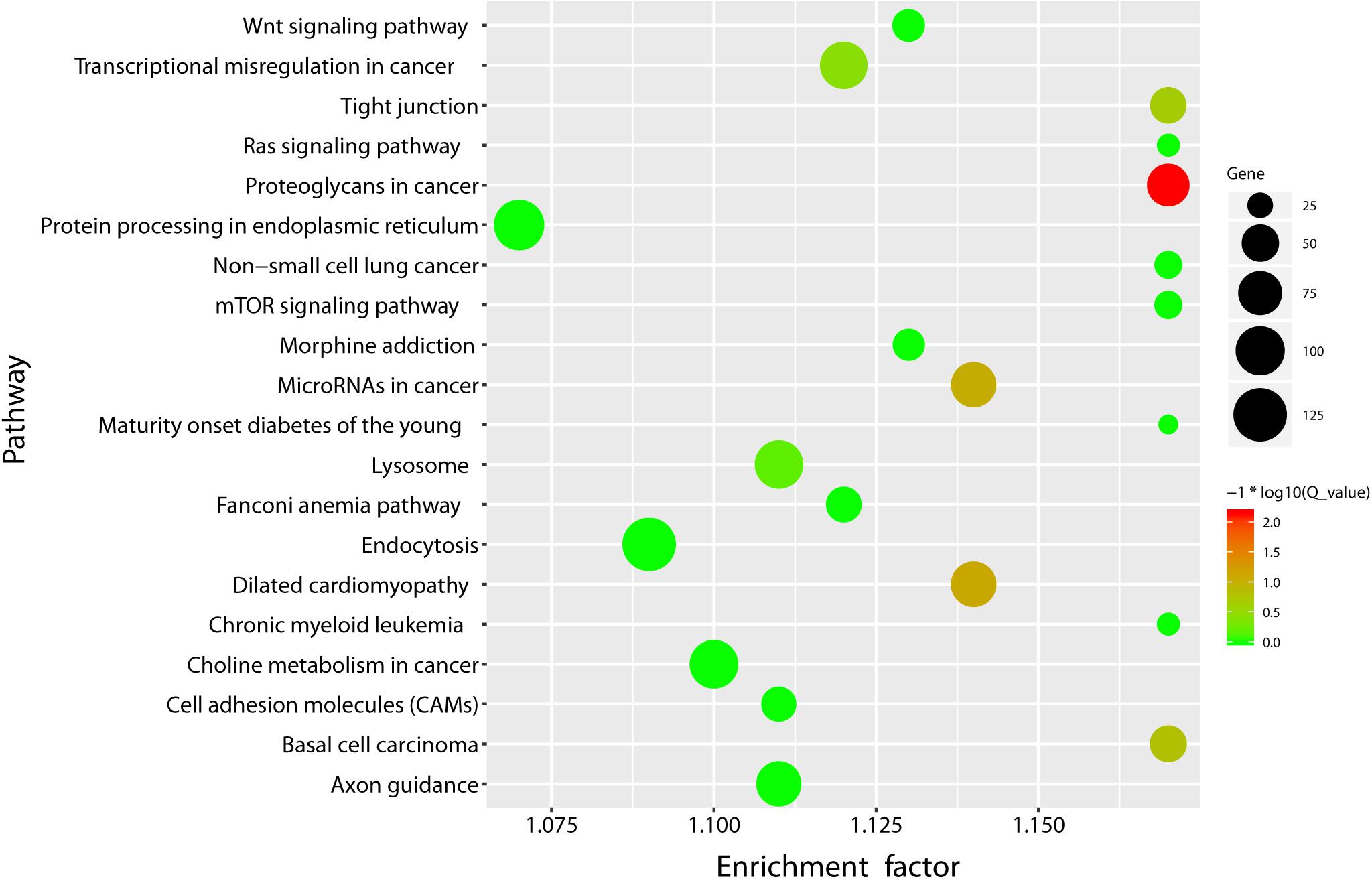
Figure 3. KEGG pathway enrichment bubble plot of the differentially expressed miRNAs. X-axis label represents rich factor (rich factor = amount of differentially expressed genes enriched in the pathway/amount of all genes in the background gene set) and Y-axis label represents pathway. The size and color of the bubble represent the amount of differentially expressed genes enriched in the pathway and the enrichment significance (Q_ value is the P value corrected after multiple hypothesis testing), respectively.
Circulating MiRNAs as Biomarkers of Different Diseases
We screened ci-miRNA biomarkers related to certain types of diseases through the HMDD. The results are shown in Table 2.
Discussion
Differentially Expressed MiRNAs in Athletes Are Likely Due to Long-Term Exercise
As shown in Figure 1, the expression profiles of the ci-miRNAs in athletes were significantly different from those of college students who had not received any regular sports training. Notably, all the participants from both groups in this study come from different parts of China, eating different food. In addition, there are no immediate training seasons prior to the blood withdrawal in the athlete group. In addition to synchronized swimming, the athletes also underwent strength training and aerobic and anaerobic training. Thus, the differentially expressed miRNAs in the athlete group may be a result of combined exercise training. Altogether, the ci-miRNAs’ profile changes in the athlete group are highly likely due to long-term exercise.
Ci-MiRNA Profile Changes in Athletes Are Correlated With Muscle-Enriched MiRNAs
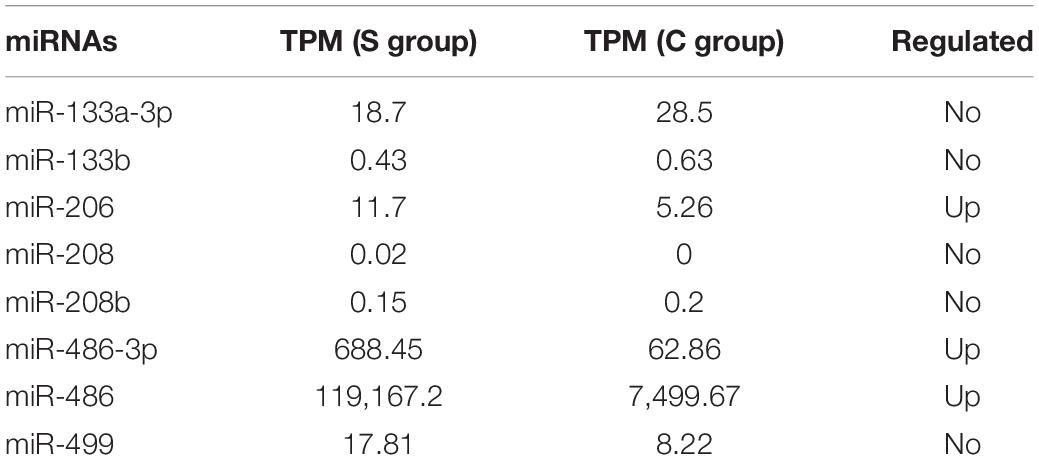
A set of miRNAs, called myo-miRs, have been identified in skeletal muscle and/or the myocardium, including miR-1, miR-133a, miR-133b, miR-206, miR-208a, miR-208b, and miR-499 (McCarthy and Esser, 2007; Callis et al., 2008; van Rooij et al., 2008; Small et al., 2010). Their expression is at least 20-fold higher than the mean expression value of the other tissues (Lee et al., 2008). Among them, miR-206 is skeletal muscle-specific (McCarthy, 2008) and miR-208a is cardiac muscle-specific (Sempere et al., 2004). MiR-486 is also a skeletal muscle and cardiac-enriched miRNA, which was included in myo-miRs in later studies (Horak et al., 2016). In our study, miR-1 was not detected in all samples. the TPMs of the remaining myo-miRs are shown in Table 3.
To test whether the level changes of the other differently expressed miRNAs were associated with muscle-specific/enriched miRNAs, we first calculated a Pearson’s correlation coefficient between miR-486 and the other differently expressed miRNAs. As a myo-miR, miR-486 mainly functions as a regulator of myoblast proliferation and migration (Alexander et al., 2011) and skeletal muscle size (Hitachi et al., 2014). The results showed that a large portion of the differently expressed miRNAs (>30%, R2 > 0.6) were highly correlated with miR-486-5p and miR-486-3p (for details, see Supplementary Table 4), which provide another evidence that the miRNA profile changes in the athlete group were likely to be induced by exercise.
Muscle injures are common among athletes. In the plasma of healthy young men, the expression level of miR-206 was significantly higher after high-intensity intermittent exercise (HIIE) or vigorous-intensity continuous exercise (VICE) (Cui et al., 2016). miR-206 is also increased in the serum of mdx and CXMDJ mice (Mizuno et al., 2011) or in response to cardiotoxin (CTX)-induced injury, but markedly decreased from day 3 to day 5 after CTX injury (Matsuzaka et al., 2014). This suggests that high levels of miR-206 in plasma may be associated with skeletal muscle injury. In addition, miR-133 has also been demonstrated to be associated with skeletal muscle injury (Laterza et al., 2009). In the present study, miR-206 was slightly increased in the S group (|log2FC| = 1.99, TPM = 0–42.44), and miR-133 did not show significant difference between the two groups. To examine whether the level changes of miRNAs in the athlete group were also associated with muscle injury, we also calculated Pearson’s correlation coefficient between miR-206 and the other differently expressed miRNAs and found that there were less than 3% of the differentially expressed miRNAs correlated with muscle injury (<3%, R2 > 0.6; for details, see Supplementary Table 4).
Long-Term Exercise May Reduce the Risk of Cancers by Regulating Ci-MiRNAs
Physical activity has been shown to be associated with lower cancer risks. From 30 to 60 min/day of moderate- to vigorous-intensity physical activity can decrease the risk of breast cancer, and physically active individuals have a lower risk of lung cancer (Lee, 2003). The overall cancer incidence was lower in athletes than in the general population (Robsahm et al., 2010; Sormunen et al., 2014). And former college athletes had a significantly lower risk of breast cancer than do the non-athletes (Wyshak and Frisch, 2000). However, the underlying mechanism regarding exercise reducing cancer risks remains to be discovered. Lately, a number of studies have indicated that some of the ci-miRNAs (see Table 2) can be used as biomarkers for cancer or other diseases (Komatsu et al., 2014; Zhang et al., 2016; Hara et al., 2017; Khalighfard et al., 2018). Strikingly, long-term exercise regulates the levels of these ci-miRNAs in an opposite direction to those in cancer patients, which suggests that long-term exercise may reduce the risk of cancers through regulating ci-miRNAs.
Interestingly, some of the elevated miRNAs, such as miR-126 and miR-423, in heart failure and coronary artery disease (CAD) were also upregulated in the athlete group. Coronary artery calcification (CAC) is a strong predictor of incident CAD (Detrano et al., 2008). In a prospective study, 27% higher odds of CAC have been seen among participants who exceeded physical activity guidelines vs. those below physical activity guidelines (Laddu et al., 2017). CAD is also a leading cause of sudden cardiac death in athletes over 35 years of age (Morrison et al., 2018). But the incidence rates of sports-related sudden cardiac deaths in noncompetitive and competitive athletes are not different (Risgaard et al., 2014). Does high-intensity or volume exercise increase the risk of CAD? Is miR-126 not a solid biomarker of CAD? Further research is needed to address these questions.
Conslusion
Long-term exercise significantly alters the plasma miRNA profiles in healthy young women, which may reduce the risk of lung, breast, pancreas, melanoma, cholangiocarcinoma, esophageal neoplasms, and colorectal carcinoma. Our current study described an overall effect of long-term exercise on circulating miRNA profiles in the plasma of young women. Further studies are needed to determine whether different types of exercises have different effects on ci-miRNAs and whether there are gender and age differences.
Data Availability Statement
The sequencing data has been deposited in the BioProject database (accession: PRJNA613853, https://www.ncbi.nlm.nih.gov/sra?linkname=bioproject_sra_all&from_uid=613853).
Ethics Statement
The studies involving human participants were reviewed and approved by the Ethics Committee of South China Normal University School of Sports Science (Approval Number 2017102001). Written informed consent to participate in this study was provided by the participants.
Author Contributions
RD and CL initiated and supported the project. FL and MB performed the experiments. FL and RD wrote the manuscript. All authors analyzed the data and commented on the manuscript.
Funding
This work was supported by the NSFC (31971096 and 31771256) and Guangdong Science and Technology Collaborative Innovation Center for Sports Science (2019B110210004).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors wish to thank all participants, staff of the China Institute of Sport Science, as well as coaches and medical staff for their participation in the study.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphys.2020.00372/full#supplementary-material
Footnotes
References
Abue, M., Yokoyama, M., Shibuya, R., Tamai, K., Yamaguchi, K., Sato, I., et al. (2015). Circulating miR-483-3p and miR-21 is highly expressed in plasma of pancreatic cancer. Int. J. Oncol. 46, 539–547. doi: 10.3892/ijo.2014.2743
Akers, J. C., Ramakrishnan, V., Kim, R., Phillips, S., Kaimal, V., Mao, Y., et al. (2015). miRNA contents of cerebrospinal fluid extracellular vesicles in glioblastoma patients. J. Neurooncol. 123, 205–216. doi: 10.1007/s11060-015-1784-3
Alexander, M. S., Casar, J. C., Motohashi, N., Myers, J. A., Eisenberg, I., Gonzalez, R. T., et al. (2011). Regulation of DMD pathology by an ankyrin-encoded miRNA. Skelet. Muscle 1:27. doi: 10.1186/2044-5040-1-27
Ali, S., Almhanna, K., Chen, W., Philip, P. A., and Sarkar, F. H. (2010). Differentially expressed miRNAs in the plasma may provide a molecular signature for aggressive pancreatic cancer. Am. J. Transl. Res. 3, 28–47.
Anfossi, S., Giordano, A., Gao, H., Cohen, E. N., Tin, S., Wu, Q., et al. (2014). High serum miR-19a levels are associated with inflammatory breast cancer and are predictive of favorable clinical outcome in patients with metastatic HER2+ inflammatory breast cancer. PLoS One 9:e83113. doi: 10.1371/journal.pone.0083113
Aoi, W., Ichikawa, H., Mune, K., Tanimura, Y., Mizushima, K., Naito, Y., et al. (2013). Muscle-enriched microRNA miR-486 decreases in circulation in response to exercise in young men. Front. Physiol. 4:80. doi: 10.3389/fphys.2013.00080
Baggish, A. L., Hale, A., Weiner, R. B., Lewis, G. D., Systrom, D., Wang, F., et al. (2011). Dynamic regulation of circulating microRNA during acute exhaustive exercise and sustained aerobic exercise training. J. Physiol. 589(Pt 16), 3983–3994. doi: 10.1113/jphysiol.2011.213363
Bahn, J. H., Zhang, Q., Li, F., Chan, T. M., Lin, X., Kim, Y., et al. (2015). The landscape of microRNA, Piwi-interacting RNA, and circular RNA in human saliva. Clin. Chem. 61, 221–230. doi: 10.1373/clinchem.2014.230433
Baraniskin, A., Kuhnhenn, J., Schlegel, U., Maghnouj, A., Zollner, H., Schmiegel, W., et al. (2012). Identification of microRNAs in the cerebrospinal fluid as biomarker for the diagnosis of glioma. Neurol Oncol. 14, 29–33. doi: 10.1093/neuonc/nor169
Barber, J. L., Zellars, K. N., Barringhaus, K. G., Bouchard, C., Spinale, F. G., and Sarzynski, M. A. (2019). The effects of regular exercise on circulating cardiovascular-related microRNAs. Sci. Rep. 9:7527. doi: 10.1038/s41598-019-43978-x
Basati, G., Emami Razavi, A., Abdi, S., and Mirzaei, A. (2014). Elevated level of microRNA-21 in the serum of patients with colorectal cancer. Med. Oncol. 31:205. doi: 10.1007/s12032-014-0205-3
Boon, R. A., and Dimmeler, S. (2015). MicroRNAs in myocardial infarction. Nat. Rev. Cardiol. 12, 135–142. doi: 10.1038/nrcardio.2014.207
Cai, E. H., Gao, Y. X., Wei, Z. Z., Chen, W. Y., Yu, P., and Li, K. (2012). Serum miR-21 expression in human esophageal squamous cell carcinomas. Asian Pac. J. Cancer Prev. 13, 1563–1567. doi: 10.7314/apjcp.2012.13.4.1563
Callis, T. E., Deng, Z., Chen, J. F., and Wang, D. Z. (2008). Muscling through the microRNA world. Exp. Biol. Med. (Maywood) 233, 131–138. doi: 10.3181/0709-MR-237
Cheng, L., Sun, X., Scicluna, B. J., Coleman, B. M., and Hill, A. F. (2014). Characterization and deep sequencing analysis of exosomal and non-exosomal miRNA in human urine. Kidney Int. 86, 433–444. doi: 10.1038/ki.2013.502
Chim, S. S., Shing, T. K., Hung, E. C., Leung, T. Y., Lau, T. K., Chiu, R. W., et al. (2008). Detection and characterization of placental microRNAs in maternal plasma. Clin. Chem. 54, 482–490. doi: 10.1373/clinchem.2007.097972
Cui, S. F., Wang, C., Yin, X., Tian, D., Lu, Q. J., Zhang, C. Y., et al. (2016). Similar responses of circulating microRNAs to acute high-intensity interval exercise and vigorous-intensity continuous exercise. Front. Physiol. 7:102. doi: 10.3389/fphys.2016.00102
Desai, V. G., Kwekel, J. C., Vijay, V., Moland, C. L., Herman, E. H., Lee, T., et al. (2014). Early biomarkers of doxorubicin-induced heart injury in a mouse model. Toxicol. Appl. Pharmacol. 281, 221–229. doi: 10.1016/j.taap.2014.10.006
Detrano, R., Guerci, A. D., Carr, J. J., Bild, D. E., Burke, G., Folsom, A. R., et al. (2008). Coronary calcium as a predictor of coronary events in four racial or ethnic groups. N. Engl. J. Med. 358, 1336–1345. doi: 10.1056/NEJMoa072100
Dickinson, B. A., Semus, H. M., Montgomery, R. L., Stack, C., Latimer, P. A., Lewton, S. M., et al. (2013). Plasma microRNAs serve as biomarkers of therapeutic efficacy and disease progression in hypertension-induced heart failure. Eur. J. Heart Fail. 15, 650–659. doi: 10.1093/eurjhf/hft018
Betel, D., Wilson, M., Gabow, A., Marks, D. S., and Sander, C. (2008). The microRNA.org resource: targets and expression. Nucleic Acids Res. 36(suppl. 1), D149–D153.
Fahlgren, N., Howell, M. D., Kasschau, K. D., Chapman, E. J., Sullivan, C. M., Cumbie, J. S., et al. (2007). High-throughput sequencing of Arabidopsis microRNAs: evidence for frequent birth and death of MIRNA genes. PLoS One 2:e219. doi: 10.1371/journal.pone.0000219
Fichtlscherer, S., De Rosa, S., Fox, H., Schwietz, T., Fischer, A., Liebetrau, C., et al. (2010). Circulating microRNAs in patients with coronary artery disease. Circ. Res. 107, 677–684. doi: 10.1161/CIRCRESAHA.109.215566
Goren, Y., Kushnir, M., Zafrir, B., Tabak, S., Lewis, B. S., and Amir, O. (2012). Serum levels of microRNAs in patients with heart failure. Eur. J. Heart Fail. 14, 147–154. doi: 10.1093/eurjhf/hfr155
Goto, T., Fujiya, M., Konishi, H., Sasajima, J., Fujibayashi, S., Hayashi, A., et al. (2018). An elevated expression of serum exosomal microRNA-191, – 21, -451a of pancreatic neoplasm is considered to be efficient diagnostic marker. BMC Cancer 18:116. doi: 10.1186/s12885-018-4006-5
Hara, N., Kikuchi, M., Miyashita, A., Hatsuta, H., Saito, Y., Kasuga, K., et al. (2017). Serum microRNA miR-501-3p as a potential biomarker related to the progression of Alzheimer’s disease. Acta Neuropathol. Commun. 5:10. doi: 10.1186/s40478-017-0414-z
Hitachi, K., Nakatani, M., and Tsuchida, K. (2014). Myostatin signaling regulates Akt activity via the regulation of miR-486 expression. Int. J. Biochem. Cell Biol. 47, 93–103. doi: 10.1016/j.biocel.2013.12.003
Horak, M., Novak, J., and Bienertova-Vasku, J. (2016). Muscle-specific microRNAs in skeletal muscle development. Dev. Biol. 410, 1–13. doi: 10.1016/j.ydbio.2015.12.013
Igglezou, M., Vareli, K., Georgiou, G. K., Sainis, I., and Briasoulis, E. (2014). Kinetics of circulating levels of miR-195, miR-155 and miR-21 in patients with breast cancer undergoing mastectomy. Anticancer Res. 34, 7443–7447.
Kanaan, Z., Rai, S. N., Eichenberger, M. R., Roberts, H., Keskey, B., Pan, J., et al. (2012). Plasma miR-21: a potential diagnostic marker of colorectal cancer. Ann. Surg. 256, 544–551. doi: 10.1097/SLA.0b013e318265bd6f
Khalighfard, S., Alizadeh, A. M., Irani, S., and Omranipour, R. (2018). Plasma miR-21, miR-155, miR-10b, and Let-7a as the potential biomarkers for the monitoring of breast cancer patients. Sci. Rep. 8:17981. doi: 10.1038/s41598-018-36321-3
Komatsu, S., Ichikawa, D., Hirajima, S., Kawaguchi, T., Miyamae, M., Okajima, W., et al. (2014). Plasma microRNA profiles: identification of miR-25 as a novel diagnostic and monitoring biomarker in oesophageal squamous cell carcinoma. Br. J. Cancer. 111, 1614–1624. doi: 10.1038/bjc.2014.451
Kumar, S., Keerthana, R., Pazhanimuthu, A., and Perumal, P. (2013). Overexpression of circulating miRNA-21 and miRNA-146a in plasma samples of breast cancer patients. Indian J. Biochem. Biophys. 50, 210–214.
Kurashige, J., Kamohara, H., Watanabe, M., Tanaka, Y., Kinoshita, K., Saito, S., et al. (2012). Serum microRNA-21 is a novel biomarker in patients with esophageal squamous cell carcinoma. J. Surg. Oncol. 106, 188–192. doi: 10.1002/jso.23064
Laddu, D. R., Rana, J. S., Murillo, R., Sorel, M. E., Quesenberry, C. P. Jr., Allen, N. B., et al. (2017). 25-year physical activity trajectories and development of subclinical coronary artery disease as measured by coronary artery calcium: the coronary artery risk development in young adults (CARDIA) study. Mayo Clin. Proc. 92, 1660–1670. doi: 10.1016/j.mayocp.2017.07.016
Laterza, O. F., Lim, L., Garrett-Engele, P. W., Vlasakova, K., Muniappa, N., Tanaka, W. K., et al. (2009). Plasma MicroRNAs as sensitive and specific biomarkers of tissue injury. Clin. Chem. 55, 1977–1983. doi: 10.1373/clinchem.2009.131797
Lee, E. J., Baek, M., Gusev, Y., Brackett, D. J., Nuovo, G. J., and Schmittgen, T. D. (2008). Systematic evaluation of microRNA processing patterns in tissues, cell lines, and tumors. RNA 14, 35–42. doi: 10.1261/rna.804508
Lee, I. M. (2003). Physical activity and cancer prevention–data from epidemiologic studies. Med. Sci. Sports Exerc. 35, 1823–1827. doi: 10.1249/01.MSS.0000093620.27893.23
Leelahavanichkul, A., Somparn, P., Panich, T., Chancharoenthana, W., Wongphom, J., Pisitkun, T., et al. (2015). Serum miRNA-122 in acute liver injury induced by kidney injury and sepsis in CD-1 mouse models. Hepatol. Res. 45, 1341–1352. doi: 10.1111/hepr.12501
Leidinger, P., Keller, A., Borries, A., Reichrath, J., Rass, K., Jager, S. U., et al. (2010). High-throughput miRNA profiling of human melanoma blood samples. BMC Cancer 10:262. doi: 10.1186/1471-2407-10-262
Li, Y., Yao, M., Zhou, Q., Cheng, Y., Che, L., Xu, J., et al. (2018). Dynamic regulation of circulating micrornas during acute exercise and long-term exercise training in basketball athletes. Front. Physiol. 9:282. doi: 10.3389/fphys.2018.00282
Liu, C. H., Huang, Q., Jin, Z. Y., Xie, F., Zhu, C. L., Liu, Z., et al. (2018). Circulating microRNA-21 as a prognostic, biological marker in cholangiocarcinoma. J. Cancer Res. Ther. 14, 220–225. doi: 10.4103/0973-1482.193125
Liu, X. G., Zhu, W. Y., Huang, Y. Y., Ma, L. N., Zhou, S. Q., Wang, Y. K., et al. (2012). High expression of serum miR-21 and tumor miR-200c associated with poor prognosis in patients with lung cancer. Med. Oncol. 29, 618–626. doi: 10.1007/s12032-011-9923-y
Marino, A. L., Evangelista, A. F., Vieira, R. A., Macedo, T., Kerr, L. M., Abrahao-Machado, L. F., et al. (2014). MicroRNA expression as risk biomarker of breast cancer metastasis: a pilot retrospective case-cohort study. BMC Cancer 14:739. doi: 10.1186/1471-2407-14-739
Markou, A., Zavridou, M., Sourvinou, I., Yousef, G., Kounelis, S., Malamos, N., et al. (2016). Direct comparison of metastasis-related mirnas expression levels in circulating tumor cells, corresponding plasma, and primary tumors of breast cancer patients. Clin. Chem. 62, 1002–1011. doi: 10.1373/clinchem.2015.253716
Matamala, N., Vargas, M. T., Gonzalez-Campora, R., Minambres, R., Arias, J. I., Menendez, P., et al. (2015). Tumor microRNA expression profiling identifies circulating microRNAs for early breast cancer detection. Clin. Chem. 61, 1098–1106. doi: 10.1373/clinchem.2015.238691
Matsuzaka, Y., Kishi, S., Aoki, Y., Komaki, H., Oya, Y., Takeda, S., et al. (2014). Three novel serum biomarkers, miR-1, miR-133a, and miR-206 for Limb-girdle muscular dystrophy, facioscapulohumeral muscular dystrophy, and Becker muscular dystrophy. Environ. Health Prev. Med. 19, 452–458. doi: 10.1007/s12199-014-0405-7
McCarthy, J. J. (2008). MicroRNA-206: the skeletal muscle-specific myomiR. Biochim. Biophys. Acta 1779, 682–691. doi: 10.1016/j.bbagrm.2008.03.001
McCarthy, J. J., and Esser, K. A. (2007). MicroRNA-1 and microRNA-133a expression are decreased during skeletal muscle hypertrophy. J. Appl. Physiol. (1985) 102, 306–313. doi: 10.1152/japplphysiol.00932.2006
Mizuno, H., Nakamura, A., Aoki, Y., Ito, N., Kishi, S., Yamamoto, K., et al. (2011). Identification of muscle-specific microRNAs in serum of muscular dystrophy animal models: promising novel blood-based markers for muscular dystrophy. PLoS One 6:e18388. doi: 10.1371/journal.pone.0018388
Modepalli, V., Kumar, A., Hinds, L. A., Sharp, J. A., Nicholas, K. R., and Lefevre, C. (2014). Differential temporal expression of milk miRNA during the lactation cycle of the marsupial tammar wallaby (Macropus eugenii). BMC Genomics 15:1012. doi: 10.1186/1471-2164-15-1012
Morrison, B. N., McKinney, J., Isserow, S., Lithwick, D., Taunton, J., Nazzari, H., et al. (2018). Assessment of cardiovascular risk and preparticipation screening protocols in masters athletes: the Masters Athlete Screening Study (MASS): a cross-sectional study. BMJ Open Sport Exerc. Med. 4:e000370. doi: 10.1136/bmjsem-2018-000370
Motawi, T. M., Sadik, N. A., Shaker, O. G., El Masry, M. R., and Mohareb, F. (2016). Study of microRNAs-21/221 as potential breast cancer biomarkers in Egyptian women. Gene 590, 210–219. doi: 10.1016/j.gene.2016.01.042
Munagala, R., Aqil, F., and Gupta, R. C. (2016). Exosomal miRNAs as biomarkers of recurrent lung cancer. Tumour Biol. 37, 10703–10714. doi: 10.1007/s13277-016-4939-8
Nielsen, S., Akerstrom, T., Rinnov, A., Yfanti, C., Scheele, C., Pedersen, B. K., et al. (2014). The miRNA plasma signature in response to acute aerobic exercise and endurance training. PLoS One 9:e87308. doi: 10.1371/journal.pone.0087308
Ortega, F. J., Mercader, J. M., Moreno-Navarrete, J. M., Rovira, O., Guerra, E., Esteve, E., et al. (2014). Profiling of circulating microRNAs reveals common microRNAs linked to type 2 diabetes that change with insulin sensitization. Diabetes Care 37, 1375–1383. doi: 10.2337/dc13-1847
Rehmsmeier, M., Steffen, P., Hochsmann, M., and Giegerich, R. (2004). Fast and effective prediction of microRNA/target duplexes. RNA 10, 1507–1517. doi: 10.1261/rna.5248604
Risgaard, B., Winkel, B. G., Jabbari, R., Glinge, C., Ingemann-Hansen, O., Thomsen, J. L., et al. (2014). Sports-related sudden cardiac death in a competitive and a noncompetitive athlete population aged 12 to 49 years: data from an unselected nationwide study in Denmark. Heart Rhythm 11, 1673–1681. doi: 10.1016/j.hrthm.2014.05.026
Robsahm, T. E., Hestvik, U. E., Veierod, M. B., Fagerlie, A., Nystad, W., Engebretsen, L., et al. (2010). Cancer risk in Norwegian world class athletes. Cancer Causes Control 21, 1711–1719. doi: 10.1007/s10552-010-9600-z
Russo, A., Caltabiano, R., Longo, A., Avitabile, T., Franco, L. M., Bonfiglio, V., et al. (2016). Increased levels of miRNA-146a in serum and histologic samples of patients with uveal melanoma. Front. Pharmacol. 7:424. doi: 10.3389/fphar.2016.00424
Sanders, A. P., Burris, H. H., Just, A. C., Motta, V., Amarasiriwardena, C., Svensson, K., et al. (2015). Altered miRNA expression in the cervix during pregnancy associated with lead and mercury exposure. Epigenomics 7, 885–896. doi: 10.2217/epi.15.54
Schwarzenbach, H., Milde-Langosch, K., Steinbach, B., Muller, V., and Pantel, K. (2012). Diagnostic potential of PTEN-targeting miR-214 in the blood of breast cancer patients. Breast Cancer Res Treat 134, 933–941. doi: 10.1007/s10549-012-1988-6
Sempere, L. F., Freemantle, S., Pitha-Rowe, I., Moss, E., Dmitrovsky, E., and Ambros, V. (2004). Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol. 5:R13. doi: 10.1186/gb-2004-5-3-r13
Shah, R., Yeri, A., Das, A., Courtright-Lim, A., Ziegler, O., Gervino, E., et al. (2017). Small RNA-seq during acute maximal exercise reveal RNAs involved in vascular inflammation and cardiometabolic health: brief report. Am. J. Physiol. Heart Circ. Physiol. 313, H1162–H1167. doi: 10.1152/ajpheart.00500.2017
Si, H., Sun, X., Chen, Y., Cao, Y., Chen, S., Wang, H., et al. (2013). Circulating microRNA-92a and microRNA-21 as novel minimally invasive biomarkers for primary breast cancer. J. Cancer Res. Clin. Oncol. 139, 223–229. doi: 10.1007/s00432-012-1315-y
Silakit, R., Loilome, W., Yongvanit, P., Thongchot, S., Sithithaworn, P., Boonmars, T., et al. (2017). Urinary microRNA-192 and microRNA-21 as potential indicators for liver fluke-associated cholangiocarcinoma risk group. Parasitol. Int. 66, 479–485. doi: 10.1016/j.parint.2015.10.001
Skrha, P., Horinek, A., Andel, M., and Skrha, J. (2015). miRNA-192, miRNA-21 and miRNA-200: new pancreatic cancer markers in diabetic patients?. Vnitr. Lek 61, 351–354.
Small, E. M., O’Rourke, J. R., Moresi, V., Sutherland, L. B., McAnally, J., Gerard, R. D., et al. (2010). Regulation of PI3-kinase/Akt signaling by muscle-enriched microRNA-486. Proc. Natl. Acad. Sci. U.S.A. 107, 4218–4223. doi: 10.1073/pnas.1000300107
Sorensen, S. S., Nygaard, A. B., Nielsen, M. Y., Jensen, K., and Christensen, T. (2014). miRNA expression profiles in cerebrospinal fluid and blood of patients with acute ischemic stroke. Transl. Stroke Res. 5, 711–718. doi: 10.1007/s12975-014-0364-8
Sormunen, J., Backmand, H. M., Sarna, S., Kujala, U. M., Kaprio, J., Dyba, T., et al. (2014). Lifetime physical activity and cancer incidence–a cohort study of male former elite athletes in Finland. J. Sci. Med. Sport 17, 479–484. doi: 10.1016/j.jsams.2013.10.239
Swellam, M., El Magdoub, H. M., Hassan, N. M., Hefny, M. M., and Sobeih, M. E. (2018). Potential diagnostic role of circulating MiRNAs in breast cancer: implications on clinicopathological characters. Clin. Biochem. 56, 47–54. doi: 10.1016/j.clinbiochem.2018.04.013
Tanaka, Y., Kamohara, H., Kinoshita, K., Kurashige, J., Ishimoto, T., Iwatsuki, M., et al. (2013). Clinical impact of serum exosomal microRNA-21 as a clinical biomarker in human esophageal squamous cell carcinoma. Cancer 119, 1159–1167. doi: 10.1002/cncr.27895
Tijsen, A. J., Creemers, E. E., Moerland, P. D., de Windt, L. J., van der Wal, A. C., Kok, W. E., et al. (2010). MiR423-5p as a circulating biomarker for heart failure. Circ. Res. 106, 1035–1039. doi: 10.1161/CIRCRESAHA.110.218297
Toraih, E. A., Mohammed, E. A., Farrag, S., Ramsis, N., and Hosny, S. (2015). Pilot study of serum microRNA-21 as a diagnostic and prognostic biomarker in egyptian breast cancer patients. Mol. Diagn. Ther. 19, 179–190. doi: 10.1007/s40291-015-0143-6
Turchinovich, A., Weiz, L., and Burwinkel, B. (2012). Extracellular miRNAs: the mystery of their origin and function. Trends Biochem. Sci. 37, 460–465. doi: 10.1016/j.tibs.2012.08.003
van Rooij, E., Liu, N., and Olson, E. N. (2008). MicroRNAs flex their muscles. Trends Genet. 24, 159–166. doi: 10.1016/j.tig.2008.01.007
Volinia, S., Galasso, M., Costinean, S., Tagliavini, L., Gamberoni, G., Drusco, A., et al. (2010). Reprogramming of miRNA networks in cancer and leukemia. Genome Res. 20, 589–599. doi: 10.1101/gr.098046.109
Wang, J., Huang, S. K., Zhao, M., Yang, M., Zhong, J. L., Gu, Y. Y., et al. (2014). Identification of a circulating microRNA signature for colorectal cancer detection. PLoS One 9:e87451. doi: 10.1371/journal.pone.0087451
Wang, X., Lian, Y., Wen, X., Guo, J., Wang, Z., Jiang, S., et al. (2017). Expression of miR-126 and its potential function in coronary artery disease. Afr. Health Sci. 17, 474–480. doi: 10.4314/ahs.v17i2.22
Wyshak, G., and Frisch, R. E. (2000). Breast cancer among former college athletes compared to non-athletes: a 15-year follow-up. Br. J. Cancer 82, 726–730. doi: 10.1054/bjoc.1999.0987
Yamada, A., Horimatsu, T., Okugawa, Y., Nishida, N., Honjo, H., Ida, H., et al. (2015). Serum miR-21, miR-29a, and miR-125b are promising biomarkers for the early detection of colorectal neoplasia. Clin. Cancer Res. 21, 4234–4242. doi: 10.1158/1078-0432.CCR-14-2793
Yan, L. X., Huang, X. F., Shao, Q., Huang, M. Y., Deng, L., Wu, Q. L., et al. (2008). MicroRNA miR-21 overexpression in human breast cancer is associated with advanced clinical stage, lymph node metastasis and patient poor prognosis. RNA 14, 2348–2360. doi: 10.1261/rna.1034808
Yang, J. S., Li, B. J., Lu, H. W., Chen, Y., Lu, C., Zhu, R. X., et al. (2015). Serum miR-152, miR-148a, miR-148b, and miR-21 as novel biomarkers in non-small cell lung cancer screening. Tumour Biol. 36, 3035–3042. doi: 10.1007/s13277-014-2938-1
Zhang, R., Pang, B., Xin, T., Guo, H., Xing, Y., Xu, S., et al. (2016). Plasma miR-221/222 family as novel descriptive and prognostic biomarkers for glioma. Mol. Neurobiol. 53, 1452–1460. doi: 10.1007/s12035-014-9079-9
Keywords: long-term exercise, circulating miRNAs, risk of cancer, human plasma, women
Citation: Li F, Bai M, Xu J, Zhu L, Liu C and Duan R (2020) Long-Term Exercise Alters the Profiles of Circulating Micro-RNAs in the Plasma of Young Women. Front. Physiol. 11:372. doi: 10.3389/fphys.2020.00372
Received: 09 November 2019; Accepted: 30 March 2020;
Published: 08 May 2020.
Edited by:
Sergej Ostojic, University of Novi Sad, SerbiaReviewed by:
Wuqiang Zhu, Mayo Clinic in Arizona, United StatesLi Fu, Tianjin Medical University, China
Jun Shi, The University of Texas Southwestern Medical Center, United States
Copyright © 2020 Li, Bai, Xu, Zhu, Liu and Duan. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chengyi Liu, bGl1dGN5QHNjbnUuZWR1LmNu; Rui Duan, ZHVhbnJ1aUBtLnNjbnUuZWR1LmNu
†These authors have contributed equally to this work
 Fan Li
Fan Li Muwei Bai2,3†
Muwei Bai2,3† Chengyi Liu
Chengyi Liu Rui Duan
Rui Duan