
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Pharmacol. , 31 March 2025
Sec. Pharmacology of Infectious Diseases
Volume 16 - 2025 | https://doi.org/10.3389/fphar.2025.1533952
 Wen-Ming Long1,2†
Wen-Ming Long1,2† Wei-Xin Xu1†
Wei-Xin Xu1† Qin Hu3†
Qin Hu3† Qiang Qu3
Qiang Qu3 Xiao-Li Wu4
Xiao-Li Wu4 Ying Chen5
Ying Chen5 Qing Wan6
Qing Wan6 Tian-Tian Xu6
Tian-Tian Xu6 Yue Luo7
Yue Luo7 Jian Qu1,8*
Jian Qu1,8*Introduction: Carbapenem-resistant Pseudomonas aeruginosa (CRPA) infections pose a critical clinical challenge. Although ceftazidime/avibactam (CAZ/AVI) and polymyxin B (PMB) are frontline therapies, their comparative effectiveness in terms of 30-day survival, renal safety profiles, and clinical success rates remains poorly characterized. To address this knowledge gap, a multicenter real-world study was conducted.
Methods: CRPA-infected patients treated with PMB or CAZ/AVI-based regimens were enrolled from five hospitals between January 1, 2021, to July 31, 2023. Propensity score matching (PSM) and binary logistic regression analysis were performed to evaluate efficacy and acute renal injury (AKI) occurrence, and a multivariable COX proportional hazards regression of the 30-day all-cause mortality was performed.
Results: 170 CRPA-infected patients were enrolled, among whom 124 (72.9%) had difficult-to-treat resistant P. aeruginosa (DTR-PA) infections and 77 (45.3%) received CAZ/AVI-based regimens. After 1:1 PSM, the results demonstrated that the CRPA clearance rate was significantly higher in the CAZ/AVI group compared to the PMB group (61.0% vs. 24.4%, p = 0.001); however, no significant differences were observed in clinical success rates (55.6% vs. 44.4%), incidence of AKI (26.8% vs. 39.0%), or 30-day all-cause mortality (7.3% vs. 12.2%) between the two groups (all p > 0.05). Compared with the PMB-based regimens, CAZ/AVI-based regimens were significantly associated with CRPA clearance success (OR 0.185, 95%CI 0.061–0.564, p < 0.001); additionally, multi-site infection (OR 0.295, 95%CI 0.097–0.899, p = 0.032) and the number of combined anti-PA antibiotics (OR 0.435, 95%CI 0.213–0.888, p = 0.022) were associated with enhanced CRPA clearance. The occurrence of AKI in patients with CRPA infection was associated with underlying diseases, including sepsis/septic shock (OR 3.405, 95%CI 1.007–11.520, p = 0.049), and diabetes mellitus (OR 3.600, 95%CI 1.018–12.733, p = 0.047). In addition, other CREs infection (HR 40.849, 95%CI 3.323–502.170, p = 0.004), APACHE II score (HR 1.072, 95%CI 1.032–1.114, p < 0.001) were found to be independent predictors of 30-day all-cause mortality.
Conclusion: In conclusion, CAZ/AVI-based regimens demonstrated superior efficacy in clearing CRPA compared to PMB-based regimens. Furthermore, several factors associated with AKI and mortality in CRPA-infected patients were identified, highlighting the need for further research to optimize treatment strategies.
The escalating prevalence of multidrug-resistant organisms (MDROs) poses a significant threat to global public health. In critical infections, particularly within intensive care units (ICUs), the identification of causative pathogens is exceptionally challenging due to infection complexity and potential polymicrobial involvement. Among these, Pseudomonas aeruginosa (PA) represents one of the paramount concerns (WHO, 2024; Borgatta et al., 2017). As a common pathogen with multifaceted resistance mechanisms, PA is recognized as one of the six most lethal multidrug-resistant pathogens under the ESKAPE classification (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, P. aeruginosa, and Enterobacter spp.) (Antimicrobial, 2022).
Since in clinical practice it is difficult to ascertain the multiple resistance mechanisms present in every PA strain, and particularly carbapenem-resistance, the concept of difficult to treat resistant PA (DTR-PA) has emerged (Cosentino et al., 2023). Data from the CHINET surveillance system (http://www.chinets.com) revealed that in 2023, PA accounted for 7.8% of 445,199 bacterial isolates collected through active surveillance across 74 tertiary hospitals in China. Notably, 17.4% and 21.9% of these PA isolates exhibited resistance to meropenem and imipenem, respectively. Alarmingly, the prevalence of carbapenemase production in carbapenem-resistant PA (CRPA) in China has reached 41% (Zhang et al., 2023), with approximately 34%–38% of CRPA strains classified as DTR-PA (Dong et al., 2025; Yuan et al., 2023). The attributable mortality of CRPA infections is estimated between 20.0% and 30.8% (Lodise et al., 2022), while mortality associated with DTR-PA may escalate to 43% (Yuan et al., 2023).
Current therapeutic options for CRPA infections remain severely limited (Reig et al., 2022). Ceftazidime/avibactam (CAZ/AVI) is considered as one of the first-line treatment for CRPA infections, whereas polymyxin B (PMB) is reserved as a last-resort therapy (Pulmonary, 2022; Tamma et al., 2024). However, emerging reports of CRPA resistance to both PMB and CAZ/AVI, coupled with the nephrotoxicity associated with these agents, have constrained their clinical utility (Howard-Anderson et al., 2022; Wang et al., 2025; Shi et al., 2024; Chang et al., 2022). Specifically, PMB and CAZ/AVI are linked to drug-related renal insufficiency, which complicates dosing regimens and exacerbates treatment failure risks. This highlights the critical need to elucidate the real-world clinical efficacy and renal safety profiles of CAZ/AVI-based and PMB-based regimens in the treatment of CRPA infections, with a focus on comprehensive clinical considerations.
While some small-sample studies have been conducted, they have provided limited insights. For instance, Xu et al. reported a clinical cure rate of 63.1% for CAZ/AVI in treating CRPA, but their study did not conclusively determine whether monotherapy or combination therapy was more effective, as no significant difference in clinical efficacy was found between the two approaches (Xu et al., 2024). Additionally, a single-center retrospective cohort study (Chen et al., 2022) compared PMB and CAZ/AVI in CRPA treatment and found that CAZ/AVI seemed to offer better survival benefits than PMB, with age, CAZ/AVI use, and central venous catheter placement identified as independent predictors of 30-day survival. However, this study had notable limitations, as it did not evaluate the safety of PMB and CAZ/AVI, and the assessment of their microbiological efficacy was not comprehensive. The existing literature thus falls short in providing a complete picture of the comparative effectiveness and safety of these treatments, as well as the factors influencing their outcomes. This multicenter retrospective cohort study aims to compare the clinical effectiveness of PMB and CAZ/AVI in treating CRPA infections, with specific emphasis on evaluating the impact of antibiotic treatment regimens, DTR-PA infections, microbiological clearance rates, and AKI incidence on 30-day all-cause mortality, while systematically identifying associated clinical influencing factors.
This multicenter retrospective cohort study, conducted from January 2021 to July 2023 at five tertiary hospitals in China (Second Xiangya Hospital (3,500 beds), Xiangya Hospital (3,500 beds), Second Affiliated Hospital of Guangzhou Medical University (2,500 beds), First Affiliated Hospital of Nanchang University (6,000 beds), and Renmin Hospital of Wuhan University (3,500 beds)). Hospital selection criteria included: (1) Provincial-level tertiary centers with >2000 beds; (2) Established antimicrobial stewardship programs; (3) Complete electronic medical record systems covering ICU and general wards.
The study was conducted in accordance with the ethical standards outlined in the Helsinki Declaration (1964). Approval was obtained from the Ethics Committees of the Second Xiangya Hospital of Central South University (LYF-2020021) and other ethicscommittees at each study site. Given the retrospective and observational design of the study, the requirement for written informed consent was waived.
CRPA infection in a patient was defined as the detection of CRPA accompanied by a body temperature >38.3°C or <36°C, along with a white blood cell count >12 × 109/L or <4 × 109/L, C-reactive protein (CRP) > 50 mg/L (measured by immunoturbidimetry) or procalcitonin (PCT) ≥0.5 ng/mL (measured by electrochemiluminescence), new onset of purulent sputum or changes in sputum characteristics, and progression of infiltrates on chest imaging within 72 h. Inclusion criteria of patients were: (1) patients confirmed to have CRPA infection by bacterial culture and sensitivity testing; (2) patients treated with PMB or CAZ/AVI-based therapy for ≥72 h; (3) patients with infection-related indicators (Body temperature, white blood cell count, neutrophil count, CRP, and PCT) to assess treatment efficacy. Exclusion criteria were: (1) age <18 years; (2) pregnant patients; (3) patients unable to assess efficacy; (4) PMB maintenance dose <50 mg q12h; (5) cases of resistance of CRPA to CAZ/AVI or PMB; (6) Patients for whom the microbiological efficacy could not be determined at the end of treatment due to the irregular re-examination of pathogens.
Demographic characteristics, clinical features, microbiological data, etc., including age, weight, comorbidities, Acute Physiology and Chronic Health Evaluation II (APACHE II) score, site of infection, pathogens causing infection, details of antibiotic use, and inflammatory markers, were extracted from the hospital electronic medical record system. The primary outcome of interest was 30-day all-cause mortality, with secondary outcomes including microbiological clearance, clinical efficacy, and AKI.
All isolates were identified by MALDI-TOF MS (bioMérieux) with ≥98.7% confidence. Antimicrobial susceptibility testing utilized VITEK®2 platforms, supplemented by CLSI M07-compliant broth microdilution for CAZ/AVI. Minimum inhibitory concentration (MIC) interpretations uniformly applied CLSI M100-Ed33 (2023) criteria, except where unavailable: EUCAST ECOFFs (v13.0) guided PMB interpretation. Historical MIC data were reanalyzed using 2023 standards to eliminate temporal guideline discrepancies, adhering to China’s WS/T 639–2018 mandate prioritizing CLSI. CRPA required meropenem MIC ≥8 mg/L (CLSI 2023). Given the real-world retrospective design focusing on CRPA, systematic β-lactamase/carbapenemase phenotypic testing was not performed. DTR-PA was defined as PA exhibiting non-susceptibility to all of the following: piperacillin-tazobactam, ceftazidime, cefepime, aztreonam, meropenem, imipenem-cilastatin, ciprofloxacin, and levofloxacin (Tamma et al., 2024).
Patients with CRPA infection were treated with PMB or CAZ/AVI-based regimens, focusing on single CRPA strains. Microbiological clearance group: all infection sites sampled for microbial culture after treatment with PMB or CAZ/AVI were negative of CRPA.
Patients with CRPA infection treated with PMB or CAZ/AVI-based regimens. Clinical efficacy group: hemodynamically stable without the need for vasopressors, body temperature <37.5°C for 72 h, white blood cell count <10 × 109/L; improvement in clinical symptoms, infection indicators (CRP, PCT), and microbiological indicators. Clinical inefficacy group: did not meet any criteria of the clinical efficacy group, worsened condition leading to treatment discontinuation, or cases resulting in hospital mortality.
After completing treatment with PMB or CAZ/AVI-based regimens, creatinine levels should be monitored. Changes in renal function were categorized according to the KDIGO classification. The AKI group was defined as an increase in creatinine levels by either 26.5 μmol/L (observed within 48 h of CAZ/AVI or PMB administration) or 1.5 times the baseline level by the end of treatment. Due to the retrospective nature of this study, we were unable to observe changes in urine output.
Patients with CRPA infection treated with PMB or CAZ/AVI-based regimens. Mortality group: all-cause mortality or treatment discontinuation due to worsened condition within 30 days after PMB or CAZ/AVI treatment.
Statistical analyses were conducted using SPSS version 25.0. Continuous variables were summarized as mean ± standard deviation for normally distributed data or median with interquartile range (IQR) for non-normally distributed data. Comparisons between groups for continuous variables were performed using independent samples t-tests for normally distributed data and non-parametric tests (e.g., Mann-Whitney U test) for non-normally distributed data. Categorical variables were presented as numbers and percentages and analyzed using chi-square tests or Fisher’s exact tests, as appropriate. Propensity score matching (PSM) was performed in a 1:1 ratio, incorporating variables with p < 0.1 in the univariate analysis of CAZ/AVI and PMB, as well as covariates influencing the matched cohort. The matching tolerance was set at 0.2, and the order of cases was randomly permuted during the matching process to minimize selection bias. Treatment outcomes, including therapeutic efficacy, microbiological clearance rate, mortality, and AKI, were analyzed using chi-square tests or Fisher’s exact tests before and after PSM. Survival time and time to microbiological clearance were analyzed using non-parametric tests (e.g., Kaplan-Meier analysis with log-rank test). In the analysis of the impact of different administration methods on outcomes, the clinical efficacy, microbial clearance rate, and incidence of AKI were assessed using either the chi-square test or Fisher’s exact test, as appropriate. The 30-day all-cause mortality was evaluated using the log-rank test. In the subgroup analysis of microbiological clearance rates for different treatment regimens, we compared the microbiological clearance rates for monotherapy and combination therapy with CAZ/AVI and PMB before and after PSM. These comparisons were analyzed using chi-square tests or Fisher’s exact tests. For analysis of factors influencing therapeutic efficacy and all-cause mortality, variables with p < 0.05 in the univariate analysis and clinically relevant covariates were included in binary logistic regression or Cox proportional hazards regression models using enter selection. A P value of less than 0.05 was considered statistically significant. During multiple factor analysis, the predicted probabilities from the models were saved and used to construct receiver operating characteristic (ROC) curves using GraphPad software to evaluate the predictive performance on the treatment outcomes.
A total of 170 patients infected with CRPA and treated with CAZ/AVI or PMB for ≥3 days were included in the study based on the inclusion and exclusion criteria. Among them, 93 (54.7%) cases were treated with PMB-based therapy for CRPA infection (Figure 1). The characteristics of the included cases are shown in Table 1. There were 137 (80.6%) male patients, with an average age of 61.2 ± 17.6 years. A total of 44 (25.9%) patients presented with multi-site infections of CRPA, while 106 (62.4%) patients were concurrently found to harbor multiple species of carbapenem-resistant Gram-negative bacteria (CRGNB). A total of 124 (72.9%) strains of CRPA met the DTR-PA criteria. Among the CRPA strains, 15 (8.8%) remained sensitive to ceftazidime (Table 2). A combination therapy regimen was employed in 103 (60.6%) patients. The details of PMB and CAZ/AVI use were shown in Supplementary Material S1. Among the patients treated with CAZ/AVI, ten required dose adjustments following the initial administration due to changes in renal function. Specifically, three patients had an increase in the single-dose amount, while seven patients had a decrease in the single-dose amount.
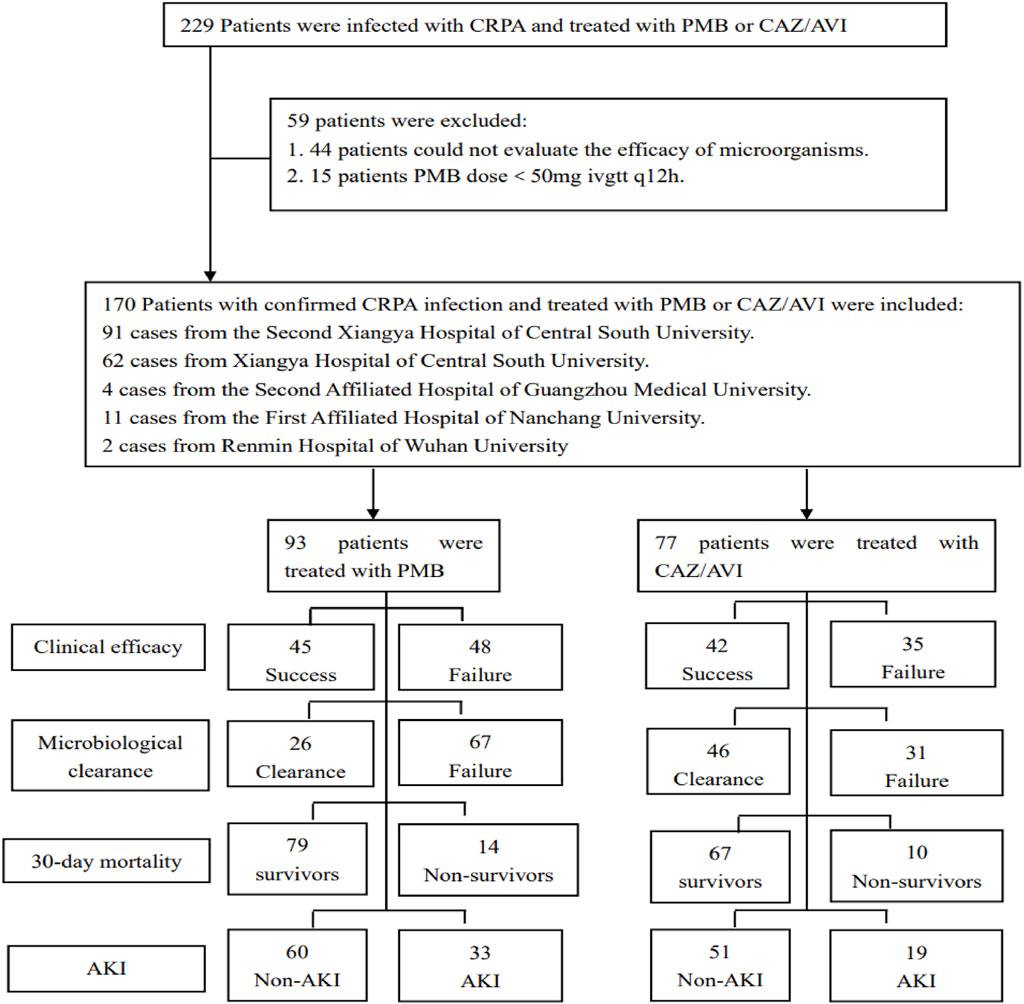
Figure 1. Flow diagram illustrating the process of determining the inclusion and exclusion criteria for patients.
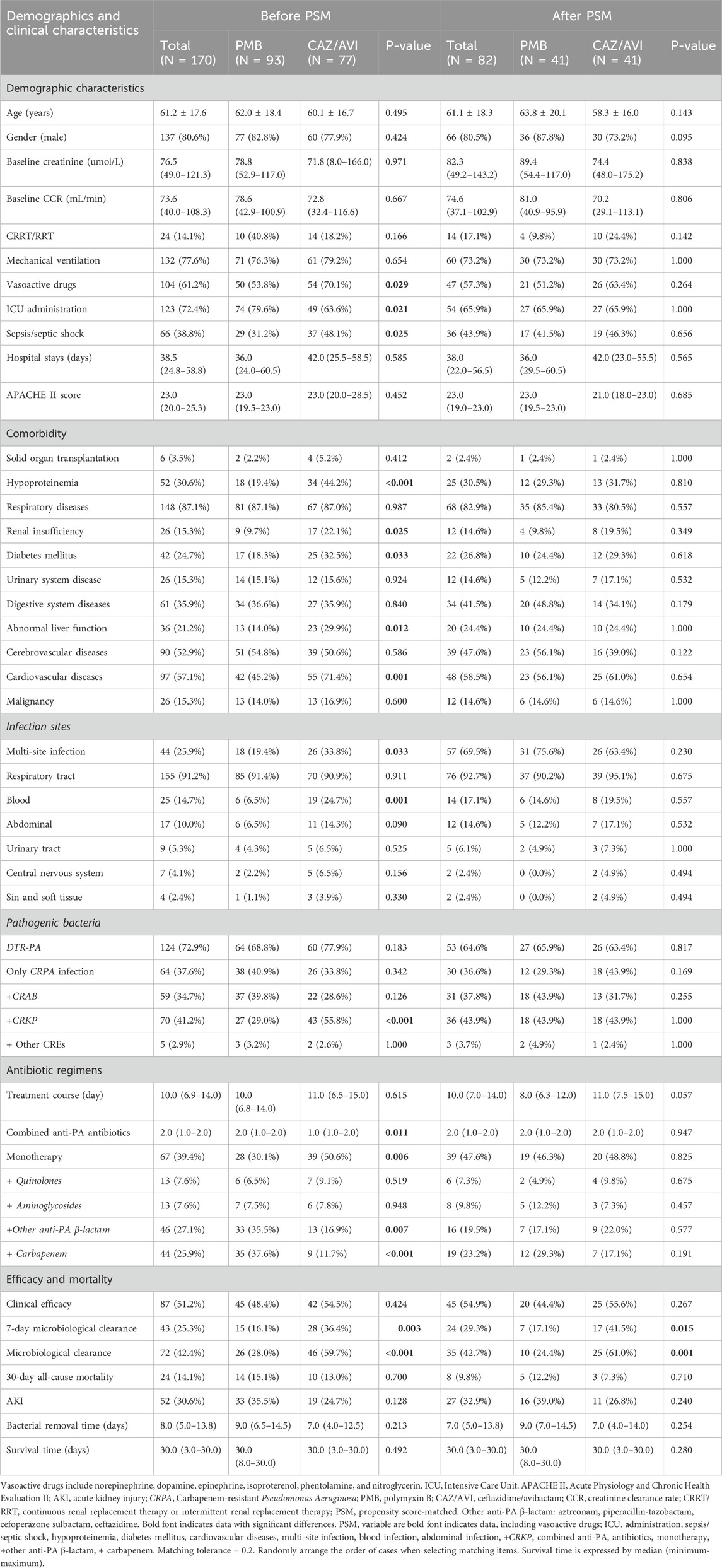
Table 1. Demographics and clinical characteristics of CRPA-infected patients treated with different regimens.
The group that received CAZ/AVI presented with more complex clinical profiles, characterized by a higher burden of comorbidities, diverse infection types, and greater disease severity compared to the PMB-treated cohort. Specifically, CAZ/AVI recipients demonstrated significantly higher rates of vasopressor use (CAZ/AVI:70.1% vs. PMB:53.8%, p = 0.029), sepsis/septic shock (CAZ/AVI:48.1% vs. PMB:31.2%, p = 0.025), and bloodstream infections (CAZ/AVI:24.7% vs. PMB:6.5%, p = 0.001). Additionally, CAZ/AVI monotherapy was more frequently employed than PMB monotherapy (50.6% vs. 30.1%, p = 0.006). To address these differences, we performed a 1:1 PSM, resulting in 82 matched cases. Univariate analysis revealed no significant differences in comorbidities, types of infections, or disease severity (details are provided in Table 1).
The clinical course analysis included 170 patients undergoing PMB-based regimens or CAZ/AVI-based regimens, with a median treatment duration of 10.0 days (IQR 6.9–14.0). Key therapeutic outcomes demonstrated clinical improvement in 51.2% of cases (87/170), while 14.1% (24/170) either succumbed during hospitalization or required treatment cessation secondary to clinical deterioration. Early microbiological response was observed in 25.3% (43/170) showing CRPA clearance within 7 days of antimicrobial initiation. Cumulative CRPA clearance rates reached 42.4% (73/170), with a median clearance time of 8.0 days (IQR 5.0–13.8). Treatment-emergent nephrotoxicity manifested as AKI in 30.6% (52/170) at therapy completion.
When comparing antimicrobial regimens, CAZ/AVI-based regimens exhibited superior microbiological efficacy with both 7-day clearance (36.4% vs. 16.1%) and cumulative clearance rates (59.7% vs. 28.0%) demonstrating statistical significance (p < 0.05), findings maintained after propensity score adjustment. Nevertheless, intergroup analyses revealed comparable clinical response rates, equivalent 30-day all-cause mortality trajectories, and analogous renal safety profiles (Table 1, “Efficacy and Mortality”). Supplementary Material S2 delineates the dose-response relationships between antimicrobial regimens and treatment outcomes, stratified by dosing intensity.
To clarify antibiotic treatment regimens for polymicrobial infections, we conducted detailed medication treatment regimens analyses across three clinical scenarios in Supplementary Material S3. Regarding CRAB co-infections (n = 59), these were managed with appropriate adjunct agents (e.g., high-dose sulbactam in 13.5% cases, tigecycline in 13.6%), while CAZ/AVI was specifically used for its anti-CRPA activity. Notably, least 23% (n = 15/65) of CRAB-positive cases demonstrated persistent colonization patterns (≥3 consecutive positive cultures) without associated inflammatory markers elevation (median CRP 8.2 mg/L vs. 42.7 mg/L in invasive infections, p < 0.001) or organ dysfunction, supporting non-pathogenic carriage status per ESCMID 2023 guidelines. In contrast, CRPA detection in these cases was associated with progressive clinical worsening, including rising inflammatory markers and aggravated symptoms.
Although there was no significant difference in the clearance rate of CRPA between monotherapy and combination therapy (p > 0.05), Subgroup analysis revealed that, after PSM, the CAZ/AVI-based combination regimens achieved significantly higher CRPA clearance rates compared to the PMB-based combination regimens (70.8% vs. 29.2%, p = 0.004) (Figure 2b). However, no statistically significant differences in CRPA clearance were observed in the monotherapy subgroup or other combination therapy subgroups (p > 0.05) (Figures 2a, c, d). Detailed results are presented in Figure 2; Supplementary Materials S4, S5.
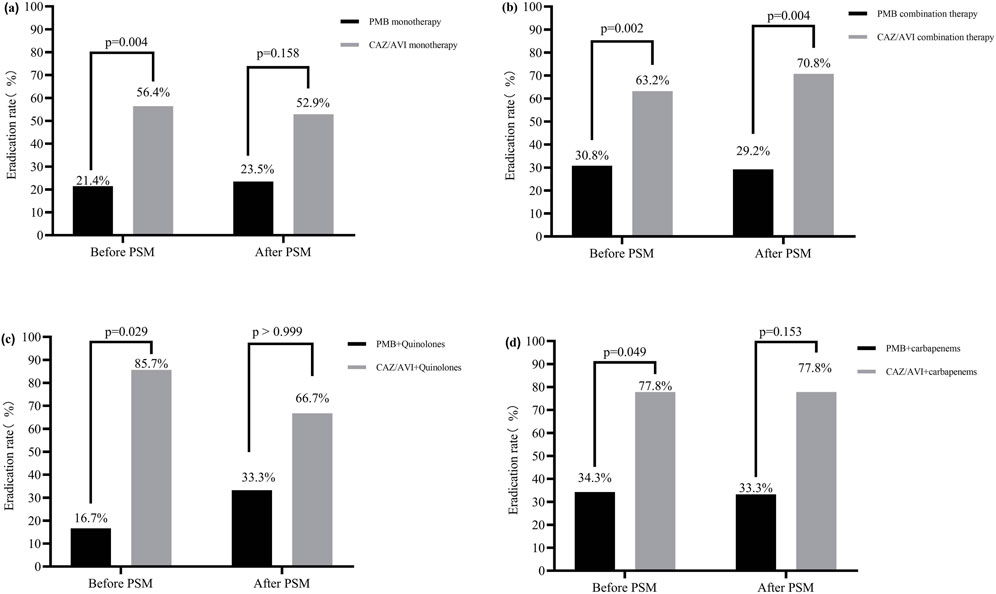
Figure 2. Microbiological efficacy subgroup analysis between PMB and CAZ/AVI-based regimens. (a) CRPA clearance rate with CAZ/AVI monotherapy vs PMB monotherapy; (b) CRPA clearance rate with CAZ/AVI combination therapy vs PMB combination therapy; (c) CRPA clearance rate with CAZ/AVI combined with quinolones vs. PMB combined with quinolones; (d) CRPA clearance rate with CAZ/AVI combined with carbapenems vs PMB combined with carbapenems.
To better understand the clinical outcomes of CRPA infections, we conducted a further analysis of factors influencing the clinical efficacy of CAZ/AVI and PMB in treating CRPA infections. In the cohort study comparing the success and failure groups, data both before and after PSM revealed that patients in the failure group had a higher severity of illness, as evidenced by a greater need for Mechanical ventilation (88.0% vs. 67.8%, p = 0.002) and vasoactive agent use (74.7% vs. 48.3%, p < 0.001), and the infection situation were more complex, as DTR-PA infections (81.9% vs. 64.4%, p = 0.010), compared to the success group (Table 3). Multivariate analysis (Table 4) identified the following influencing factors before PSM: CAZ/AVI-based regimens (OR 0.596,95%CI 0.305–1.166, p = 0.131), DTR-PA infections (OR 2.272, 95%CI 1.060–4.869, p = 0.035), and the use of vasoactive agents (OR 2.399, 95%CI 1.137–5.161, p = 0.022). However, PSM-adjusted multivariable analysis revealed no clinically significant differences in baseline characteristics (p > 0.05).
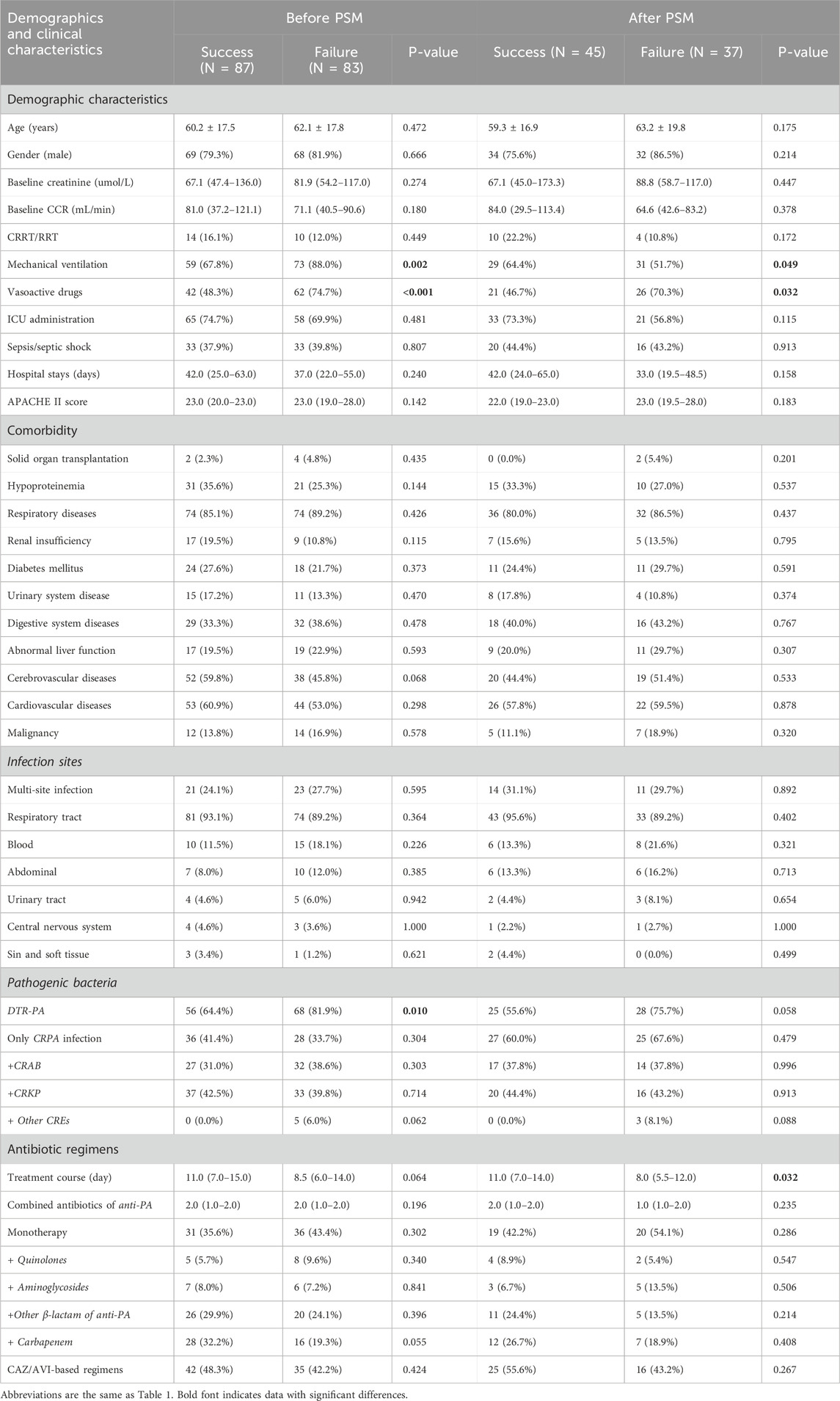
Table 3. Univariate analysis of factors associated with clinical efficacy in CRPA-infected patients.
In the analysis of factors influencing the clearance rate of CRPA infections treated with PMB and CAZ/AVI, a comparison between the clearance success group and the clearance failure group (Table 5) revealed the following findings based on data before and after PSM. Regarding the median differences, the duration of treatment in the clearance failure group was significantly shorter than that in the clearance success group [9.0 (5.9–14.0) vs. 11.0 (8.0–15.0), p = 0.038]. In terms of treatment regimens, the utilization rate of CAZ/AVI in the clearance failure group was lower than that in the clearance success group (31.6% vs. 63.9%, p < 0.001). Multivariate analysis identified the following influencing factors before PSM: treatment with CAZ/AVI (OR 0.218, 95%CI 0.108–0.440, p < 0.001) and DTR-PA infections (OR 2.139, 95%CI 1.011–4.529, p = 0.047). After PSM, treatment with CAZ/AVI remained a significant protective factor (OR 0.185, 95%CI 0.061–0.564, p = 0.003). Furthermore, multi-site infection (OR 0.295, 95%CI 0.097–0.899, p = 0.032) and the number of combined anti-PA antibiotics (OR 0.435, 95%CI 0.213–0.888, p = 0.022) were identified as protective factors associated with improved CRPA clearance rates (Table 6). The ROC curves demonstrated robust discriminatory performance of the multivariable regression model, with AUC values maintaining >0.70 across sensitivity analyses (Figures 3a, b).
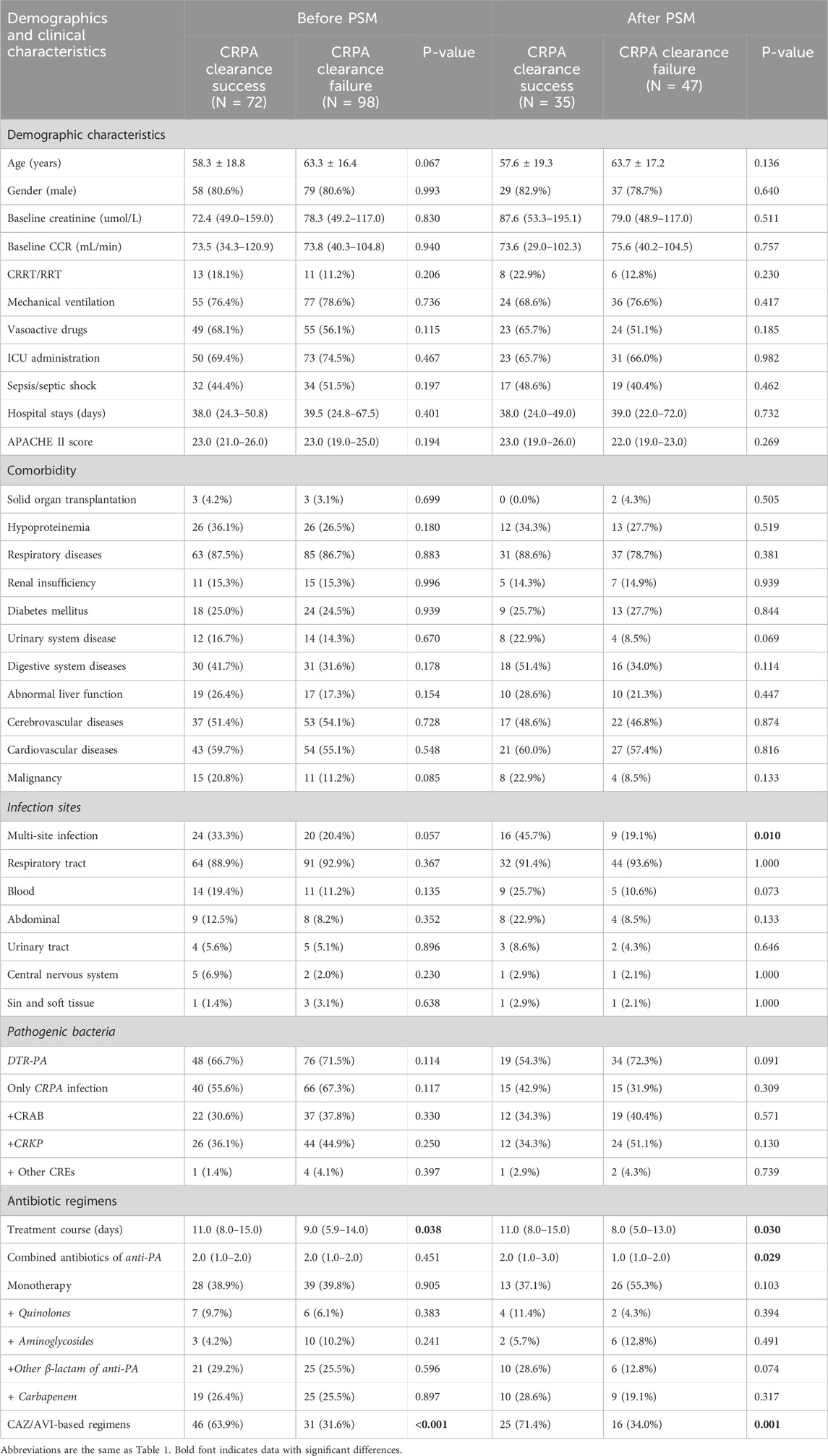
Table 5. Univariate analysis of factors associated with microbiological efficacy in CRPA-infected patients.
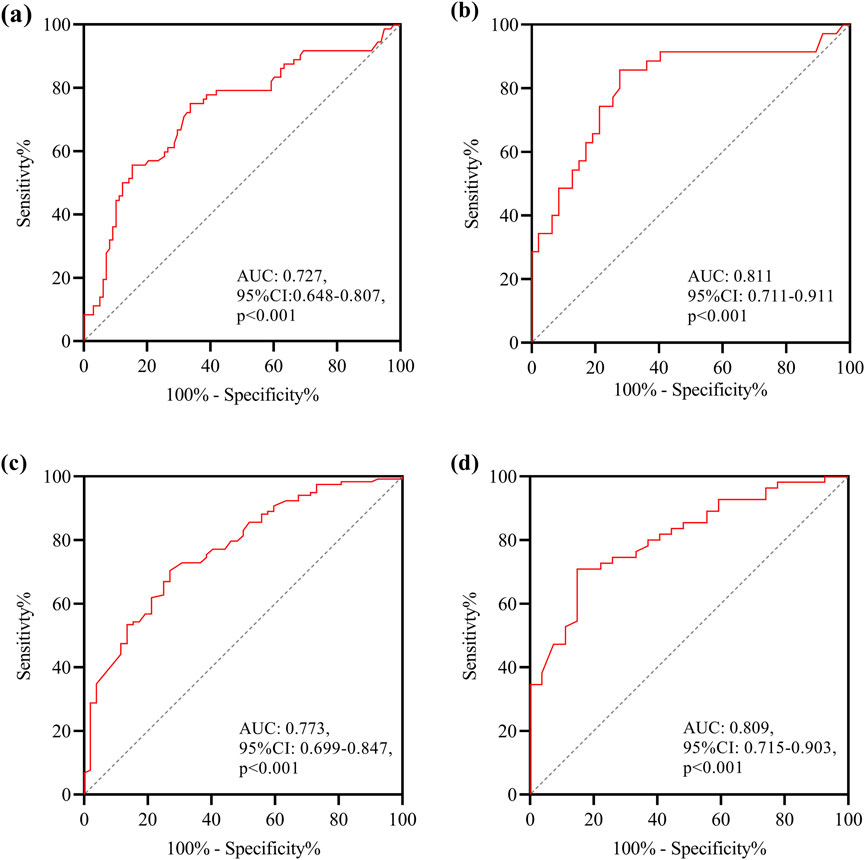
Figure 3. The multifactor analysis model ROC curve for predicting the occurrence of microbiological clearance and AKI. (a) Data before PSM for predicting the occurrence of microbiological clearance failure; (b) data after PSM for predicting the occurrence of microbiological clearance failure; (c) data before PSM for predicting the occurrence of AKI; (d) data after PSM for predicting the occurrence of AKI.
To evaluate the safety of PMB and CAZ/AVI in the treatment of CRPA infections, we conducted an analysis of factors influencing the development of AKI. In the comparison between the AKI group and the non-AKI group (Table 7), the AKI group exhibited higher rates of comorbidities, disease severity, and specific infection types compared to the non-AKI group. However, there were no significant differences between the AKI group and the non-AKI group in terms of baseline creatinine values [101.6 (54.7–160.7) vs. 71.4 (49.0–117.0), p = 0.098], creatinine clearance rates [66.9 (36.7–89.4) vs. 79.0 (40.3–118.6), p = 0.085], or the use of combined antibiotics of anti-PA [2.0 (1.0–2.0) vs. 2.0 (1.0–2.0), p = 0.880]. Multivariate analysis (Table 8) identified the following influencing factors before PSM: hypoproteinemia (OR 0.375, 95%CI 0.146–0.9620, p = 0.041), renal insufficiency (OR 5.360, 95% CI 1.929–14.898, p = 0.001), diabetes mellitus (OR 2.778, 95% CI 1.166–6.623, p = 0.027), digestive system diseases (OR 2.503, 95% CI 1.094–5.726, p = 0.030), and PMB-based regimens (OR 2.510, 95%CI 1.053–5.984, p = 0.038). After PSM, diabetes mellitus remained a significant influencing factor (OR 3.600, 95%CI 1.018–12.733, p = 0.047), and the sepsis/septic shock (OR 3.405, 95%CI 1.007–11.520, p = 0.049) increased the risk of AKI. The ROC curves demonstrated robust discriminatory performance of the multivariable regression model, with AUC values maintaining >0.70 across sensitivity analyses (Figures 3c, d).
The all-cause mortality rates at 30 days after treatment with PMB-based regimens and CAZ/AVI-based regimens were 15.1% (14/93) and 13.0% (10/77), respectively. Incorporating microbiological efficacy outcomes and the incidence of AKI into the analysis of 30-day all-cause mortality (Table 9), the univariate analysis showed that the proportion of patients with sepsis-induced shock in the non-survival group was significantly higher than that in the survival group before and after PSM (before PSM: 66.6% vs. 34.2%, p = 0.003; after PSM: 87.5% vs. 39.1%, p = 0.025). The median APACHE II score was also higher in the non-survival group compared to the survival group [before PSM: 29.00 (23.00–36.25) vs. 23.00 (19.00–23.00), p < 0.001], but the difference in APACHE II scores between the two groups was not statistically significant after PSM. The multivariate COX regression analysis revealed that sepsis/septic shock (HR 2.702, 95%CI 1.115–6.548, p = 0.028), APACHE II (HR 1.072, 95%CI 1.032–1.114, p < 0.001) were independent risk factors for 30-day all-cause mortality in patients with CRPA infection, with APACHE II (HR 1.103, 95%CI 1.105–1.198, p = 0.021) remained statistically significant in the multivariate analysis after PSM, and the other CREs infections (HR 40.849, 95%CI 3.323–502.170, p = 0.004) increased the risk of 30-day all-cause mortality (Table 10). Notably, neither DTR-PA infection status (HR 0.916, 95%CI 0.340–2.471) nor the treatment selection between CAZ/AVI and PMB regimens (HR 2.426, 95%CI 0.886–6.646) showed significant impact on 30-day all-cause mortality outcomes in the multivariate COX regression analysis (p > 0.05).
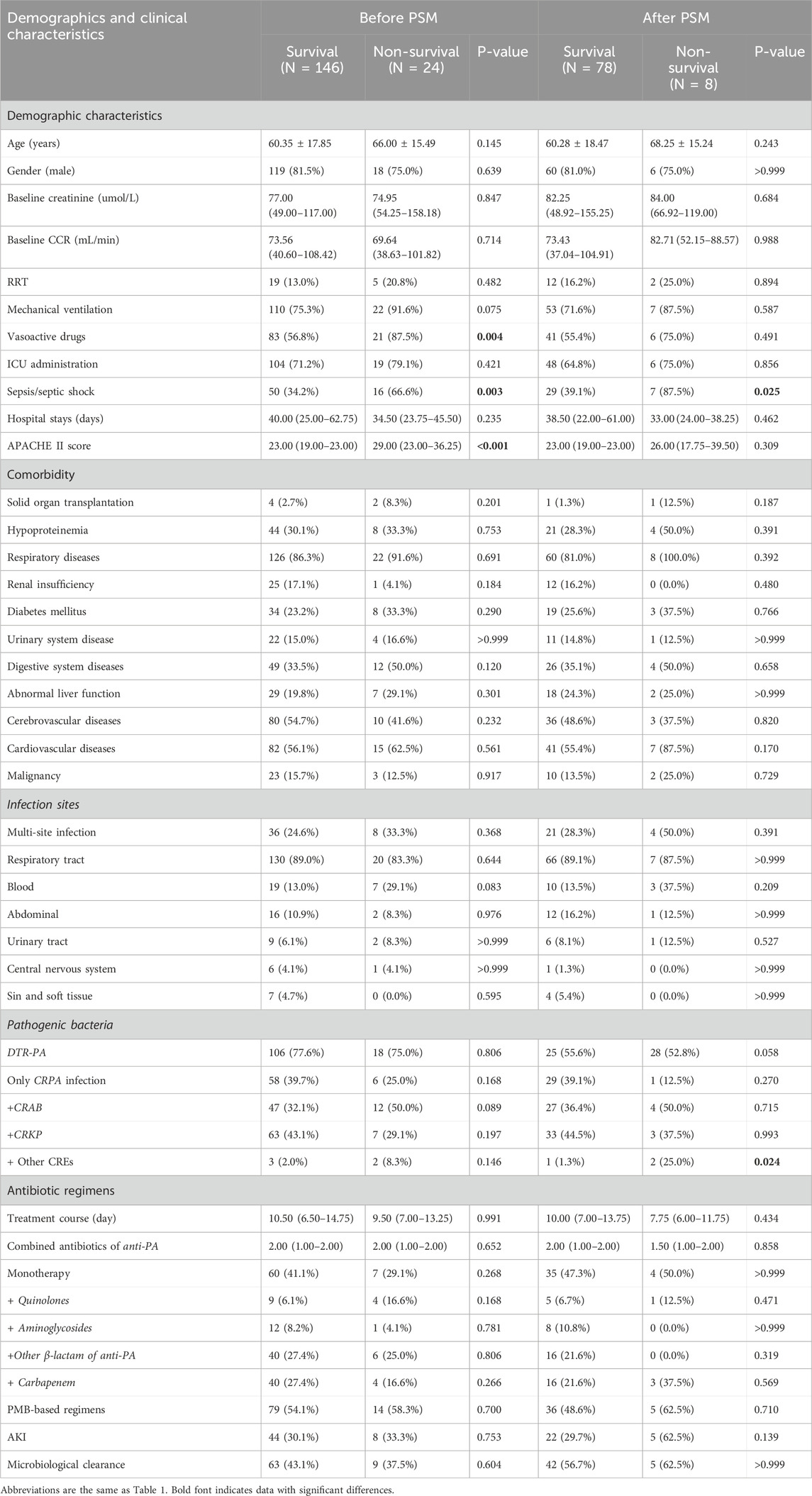
Table 9. Univariate analysis of factors associated with 30-day all-cause mortality in CRPA-infected patients.
This study, based on real-world multicentre data, aims to investigate the efficacy and safety of PMB-based regimens and CAZ/AVI-based regimens in the treatment of CRPA infections. It was the first study to examine the clearance rate of CRPA, the incidence of AKI, and the influencing factors associated with these two treatment regimens. Both 1:1 PSM and multivariable analyses independently demonstrated significantly superior microbiological clearance of CRPA with CAZ/AVI-based regimens versus PMB-based regimens. However, neither analytical approach revealed statistically significant differences in clinical efficacy, AKI incidence, or 30-day all-cause mortality between treatment groups.
In terms of clinical efficacy, there was no statistically significant difference in clinical success rates between CAZ/AVI-based and PMB-based regimens. The overall clinical success rate for CRPA infections was consistent with previously reported data (51.2% vs. 63.1%) (Xu et al., 2024). Before PSM, multivariable regression analysis identified DTR-PA infections and vasopressor requirements as independent risk factors for treatment failure in CRPA infections. PA multifaceted resistance mechanisms (efflux pump overexpression/porin loss/β-lactamase production) pose therapeutic challenges, and DTR-PA had also been proposed (Cosentino et al., 2023). Notably, PA virulence determinants (exotoxin A, type III secretion system) have been demonstrated to be critical drivers of ventilator-associated pneumonia (VAP) progression, with severe cases predisposing to multiorgan dysfunction and consequent escalation of vasopressor dependency (Alonso et al., 2020). This finding underscores the imperative for comprehensive analysis of DTR-PA virulence determinants, particularly given their demonstrated role in treatment failure and clinical deterioration.
The CRPA clearance rates observed in this study were higher compared to previous studies (CAZ/AVI: 59.7% vs. 45.1%; PMB: 28.0% vs. 14.3%), possibly due to the exclusion of patients receiving low-dose PMB (Chen et al., 2022). In regimens of medication choices, both the CRPA clearance rates for CAZ/AVI monotherapy and combination therapy were superior to the PMB-based regimens. Multifactorial analysis of CRPA microbiological efficacy also shows that the CAZ/AVI regimen was an independent predictor for CRPA clearance, consistent with guidelines recommending CAZ/AVI as the preferred treatment for CRPA infections (Tamma et al., 2024, Pulmonary Infection Assembly of Chinese Thoracic, 2022; Tamma et al., 2022). Furthermore, multivariable analysis after PSM indicated that a higher number of combined anti-PA antibiotics was associated with improved CRPA clearance rates, indirectly suggesting the potential benefits of appropriate combination therapy in the management of CRPA infections.
However, when in vitro susceptibility results indicated sensitivity to first-line drugs like CAZ/AVI, combination antibiotic therapy was not recommended (Tamma et al., 2024; Pulmonary Infection Assembly of Chinese Thoracic, 2022). Nevertheless, the total microbiological clearance rate for PMB treatment of CRPA infections with a median duration of 10.0 days was only 28.0% (monotherapy 21.4% vs. combination 30.8%), suggesting that combination therapy was a viable option for increasing the CRPA clearance rate with PMB-based regimens (Wang et al., 2022). Intriguingly, 25% of the patients in our cohort received carbapenems as part of their treatment regimen. Although in vitro studies had demonstrated synergistic effects of meropenem and colistin against CRPA (Gunalan et al., 2021), the in vivo and in vitro efficacy of CAZ/AVI combined with carbapenem antibiotics for CRPA infections had not yet been reported. In our study, nine patients received CAZ/AVI in combination with carbapenems.
In hospitalized patients, the occurrence of AKI was mainly related to sepsis, hypotension, and medications. The management of nephrotoxic drugs was one of the main strategies for AKI management (Kellum et al., 2021). Real-world data suggested that the incidence of PMB-induced AKI in the Chinese population is around 33.5%, mainly related to loading dose, concomitant nephrotoxic drugs, and baseline creatinine levels (Chang et al., 2022). Our study results shown that the incidence of AKI in the PMB group was similar to previous studies on PMB-related AKI (35.5% vs. 33.5%) (Chang et al., 2022). CAZ/AVI was generally well-tolerated, with most adverse events being mild to moderate. The incidence of AKI in real-world data for CAZ/AVI ranges from 10% to 38% (Shi et al., 2024; Feldman et al., 2022). Our study results suggested that the incidence of AKI in patients treated with CAZ/AVI is 24.7%. The incidence of AKI was higher following PMB treatment compared to CAZ/AVI, although the difference was not statistically significant (35.5% vs. 24.7%). This finding was consistent with previous studies (Chen et al., 2024).
However, multivariate analysis demonstrated that, prior to PSM, the risk of AKI was significantly higher with the PMB regimen compared to the CAZ/AVI regimen. After PSM, this difference was no longer statistically significant. This attenuation of significance might be attributable to the relatively preserved baseline renal function among the CRPA-infected patients included in our study, and given that baseline renal function constitutes the primary independent predictor of PMB-associated AKI(Wu et al., 2022). Specifically, the median baseline serum creatinine levels were 82.3 μmol/L after PSM, and the median creatinine clearance was 74.6 mL/min after PSM. Moreover, there were no significant differences in baseline renal function between the PMB and CAZ/AVI cohorts.
In our PMB cohort study, 91.4% of patients had CRPA lung infection. Adherence to the recommended PMB dosage was suboptimal, with only 73.1% of patients received the prescribed dose of 50 mg q12h, and merely 49.5% received a loading dose. Current PMB dosing guidelines suggested a loading dose ranging from 2.0–2.5 mg/kg and a maintenance dose of 1.25–1.5 mg/kg infused every 12 h (Tsuji et al., 2019). Nebulized PMB was proposed as a potential alternative to intravenous administration in ventilator-associated pneumonia patients, considering nephrotoxicity concerns (Shi et al., 2023). However, loading doses of PMB were independently associated with nephrotoxicity risks, and monitoring PMB blood concentrations was crucial in critically ill patients (Chang et al., 2022; Nation et al., 2017). Multi-centre studies suggested that combined nebulized PMB did not significantly impact the cure rate of ventilator-associated pneumonia in CRGNB-infected patients (Liu et al., 2022).
In the case of CAZ/AVI, the recommended dosage for adult patients with a CCR >50 mL/min was 2.5 g q8h administered as a continuous intravenous infusion over 2 h (Das et al., 2019). Adjustments to the CAZ/AVI dosage were warranted for patients with a CCR≤50 mL/min (Das et al., 2019). Blood concentrations of CAZ/AVI differ based on renal function status, with dosing adjustments required for patients with renal impairment (Kang et al., 2021; Teng et al., 2022). The 2.5 g q8h regimen was deemed feasible for critically ill patients with MDR-PA lung infections undergoing CRRT (Soukup et al., 2019). Patients with augmented renal clearance (CCR>130 mL/min) might necessitate higher CAZ/AVI dosages to achieve PK/PD targets (Dai et al., 2021). In our study of CAZ/AVI cohort patients, 22.1% had pre-existing renal insufficiency. Most patients (85.7%) adhered to the recommended dosing regimen, while 9.1% required dose adjustments during treatment.
In the primary endpoints of this study, there was no difference in the 30-day all-cause mortality before and after PSM. We compared the characteristics of survivors and non-survivors at 30 days and explored potential independent influencing factors through multivariate analysis. In the univariate analysis, AKI and bacterial clearance rate did not show significant differences between the survival and non-survival groups. Notably, our study revealed a higher prevalence of DTR-PA infections compared to previous reports (72.9% vs. 34%–38%) (Dong et al., 2025; Yuan et al., 2023); interestingly, in our study, DTR-PA cases demonstrated a 30-day all-cause mortality rate of 14.5%, numerically lower than the 43% rate historically reported (Yuan et al., 2023). This discrepancy may be attributable to the inclusion of cases exclusively from high-volume tertiary care centres in China, which typically have advanced antimicrobial stewardship programs and critical care capabilities.
The multivariate COX regression analysis revealed that APACHE II score were independent risk factors for 30-day all-cause mortality. In previous studies treating CRPA or DTR-PA infections with CAZ/AVI (Xu et al., 2024), the APACHE II score and sepsis/sepsis shock at the onset of infection shown significant differences between the survival and non-survival groups, which seems like our findings. In another study comparing PMB with CAZ/AVI for CRPA treatment (Chen et al., 2022), sepsis shock was also confirmed as an independent predictor for 30-day all-cause mortality, and the CAZ/AVI regimen was an independent predictor for 30-day survival compared to the PMB regimen. However, in our study, the CAZ/AVI regimens and DTR-PA infections did not significantly impact 30-day survival rates compared to the PMB regimen.
This study has several limitations. Firstly, the observational design introduced immortal-time bias, as patients had to survive long enough to receive CAZ/AVI or PMB therapy. This bias was compounded by the fact that newer antibiotics were often reserved for resistant cases, potentially excluding patients who died prematurely. Additionally, the requirement for 72 h of effective therapy further exacerbated this bias. Secondly, the inclusion of polymicrobial infections, particularly those involving CRKP (41.2%), represented a limitation. Although CRKP presence was matched, data on the susceptibility of co-pathogens to CAZ/AVI and PMB were not included. Thirdly, as a retrospective study, not all patients underwent rechecking for CRPA colonization at the end of drug therapy in real-world clinical settings, which may have impacted our results. Fourthly, we were unable to ascertain the proportion of carbapenemase-producing strains among the CRPA isolates, as resistance mechanisms were not characterized for any included strains. Lastly, our sample size was limited, and the varying medical standards across different centres could have influenced the results. Furthermore, we did not categorize CRPA based on genotype or biofilm formation status, which necessitates further evaluation of the efficacy of different resistance mechanisms.
In conclusion, for the treatment of CRPA infection, CAZ/AVI demonstrates superior efficacy in microbiological clearance of CRPA compared to PMB. However, the clinical efficacy was comparable between the two treatment regimens. These findings warrant validation through large-scale prospective studies to further elucidate the comparative effectiveness of these antimicrobial agents.
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
The studies involving humans were approved by the Ethics Committees of the Second Xiangya Hospital of Central South University. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation was not required from the participants or the participants’ legal guardians/next of kin in accordance with the national legislation and institutional requirements.
W-ML: Data curation, Funding acquisition, Writing–original draft. W-XX: Writing–original draft. QH: Investigation, Writing–review and editing. QQ: Writing–original draft. X-LW: Investigation, Writing–review and editing. YC: Funding acquisition, Investigation, Writing–review and editing. QW: Data curation, Investigation, Writing–review and editing. T-TX: Investigation, Writing–review and editing. YL: Writing–review and editing. JQ: Conceptualization, Data curation, Funding acquisition, Writing–review and editing.
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the Scientific Research Project of Hunan Health Commission (No. 202213014496) and Project of Hunan Natural Science Foundation (2023JJ50446), Hubei Province Health and Family Planning Scientific Research Project (WJ2017Q006), BEIJING Medical and health foundation (YWJKJJHKYJJ-TYU139N), and Degree and Postgraduate Education Reform Project of Central South University (2023JGB123).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2025.1533952/full#supplementary-material
Alonso, B., Fernandez-Barat, L., Di Domenico, E. G., Marin, M., Cercenado, E., Merino, I., et al. (2020). Characterization of the virulence of Pseudomonas aeruginosa strains causing ventilator-associated pneumonia. BMC Infect. Dis. 20, 909. doi:10.1186/s12879-020-05534-1
Antimicrobial, R. C., Ikuta, K. S., Sharara, F., Swetschinski, L., Robles Aguilar, G., Gray, A., et al. (2022). Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis. Lancet 399, 629–655. doi:10.1016/s0140-6736(21)02724-0
Borgatta, B., Gattarello, S., Mazo, C. A., Imbiscuso, A. T., Larrosa, M. N., Lujan, M., et al. (2017). The clinical significance of pneumonia in patients with respiratory specimens harbouring multidrug-resistant Pseudomonas aeruginosa: a 5-year retrospective study following 5667 patients in four general ICUs. Eur. J. Clin. Microbiol. Infect. Dis. 36, 2155–2163. doi:10.1007/s10096-017-3039-z
Chang, K., Wang, H., Zhao, J., Yang, X., Wu, B., Sun, W., et al. (2022). Risk factors for polymyxin B-associated acute kidney injury. Int. J. Infect. Dis. 117, 37–44. doi:10.1016/j.ijid.2022.01.055
Chen, J., Hu, Q., Zhou, P., and Deng, S. (2024). Ceftazidime-avibactam versus polymyxins in treating patients with carbapenem-resistant Enterobacteriaceae infections: a systematic review and meta-analysis. Infection 52, 19–28. doi:10.1007/s15010-023-02108-6
Chen, J., Liang, Q., Chen, X., Wu, J., Wu, Y., Teng, G., et al. (2022). Ceftazidime/avibactam versus polymyxin B in the challenge of carbapenem-resistant Pseudomonas aeruginosa infection. Infect. Drug Resist 15, 655–667. doi:10.2147/IDR.S350976
Cosentino, F., Viale, P., and Giannella, M. (2023). MDR/XDR/PDR or DTR? Which definition best fits the resistance profile of Pseudomonas aeruginosa? Curr. Opin. Infect. Dis. 36, 564–571. doi:10.1097/QCO.0000000000000966
Dai, Y., Chang, W., Zhou, X., Yu, W., Huang, C., Chen, Y., et al. (2021). Evaluation of ceftazidime/avibactam administration in enterobacteriaceae and Pseudomonas aeruginosa bloodstream infections by Monte Carlo simulation. Drug Des. Devel Ther. 15, 2899–2905. doi:10.2147/DDDT.S309825
Das, S., Li, J., Riccobene, T., Carrothers, T. J., Newell, P., Melnick, D., et al. (2019). Dose selection and validation for ceftazidime-avibactam in adults with complicated intra-abdominal infections, complicated urinary tract infections, and nosocomial pneumonia. Antimicrob. Agents Chemother. 63, e02187. doi:10.1128/AAC.02187-18
Dong, L., Huang, Y., Zhang, S., Xu, B., Li, B., and Cao, Y. (2025). Risk factors for development and mortality of carbapenem-resistant Pseudomonas aeruginosa bloodstream infection in a Chinese teaching hospital: a seven-year retrospective study. Infect. Drug Resist 18, 979–991. doi:10.2147/IDR.S495240
Feldman, S., Russo, A., Ceccarelli, G., Borrazzo, C., Madge, C., Venditti, M., et al. (2022). Ceftazidime-avibactam for the treatment of carbapenem-resistant Klebsiella pneumoniae infections in patients with liver cirrhosis. J. Clin. Exp. Hepatol. 12, 1293–1300. doi:10.1016/j.jceh.2022.04.016
Gunalan, A., Sarumathi, D., Sastry, A. S., Ramanathan, V., Rajaa, S., and Sistla, S. (2021). Effect of combined colistin and meropenem against meropenem resistant Acinetobacter baumannii and Pseudomonas aeruginosa by checkerboard method: a cross sectional analytical study. Indian J. Pharmacol. 53, 207–212. doi:10.4103/ijp.ijp_1013_20
Howard-Anderson, J., Davis, M., Page, A. M., Bower, C. W., Smith, G., Jacob, J. T., et al. (2022). Prevalence of colistin heteroresistance in carbapenem-resistant Pseudomonas aeruginosa and association with clinical outcomes in patients: an observational study. J. Antimicrob. Chemother. 77, 793–798. doi:10.1093/jac/dkab461
Kang, Y., Zhou, Q., and Cui, J. (2021). Pharmacokinetic/pharmacodynamic modelling to evaluate the efficacy of various dosing regimens of ceftazidime/avibactam in patients with pneumonia caused by Klebsiella pneumoniae carbapenemase (KPC)-producing K. pneumoniae: a multicentre study in northern China. J. Glob. Antimicrob. Resist 27, 67–71. doi:10.1016/j.jgar.2021.07.020
Kellum, J. A., Romagnani, P., Ashuntantang, G., Ronco, C., Zarbock, A., and Anders, H. J. (2021). Acute kidney injury. Nat. Rev. Dis. Prim. 7, 52. doi:10.1038/s41572-021-00284-z
Liu, J., Shao, M., Xu, Q., Liu, F., Pan, X., Wu, J., et al. (2022). Low-dose intravenous plus inhaled versus intravenous polymyxin B for the treatment of extensive drug-resistant Gram-negative ventilator-associated pneumonia in the critical illnesses: a multi-center matched case-control study. Ann. Intensive Care 12, 72. doi:10.1186/s13613-022-01033-5
Lodise, T. P., Bassetti, M., Ferrer, R., Naas, T., Niki, Y., Paterson, D. L., et al. (2022). All-cause mortality rates in adults with carbapenem-resistant Gram-negative bacterial infections: a comprehensive review of pathogen-focused, prospective, randomized, interventional clinical studies. Expert Rev. Anti Infect. Ther. 20, 707–719. doi:10.1080/14787210.2022.2020099
Nation, R. L., Garonzik, S. M., Thamlikitkul, V., Giamarellos-Bourboulis, E. J., Forrest, A., Paterson, D. L., et al. (2017). Dosing guidance for intravenous colistin in critically-ill patients. Clin. Infect. Dis. 64, 565–571. doi:10.1093/cid/ciw839
Pulmonary, I. A. O. C. T. S. (2022). Chinese expert consensus on the management of lower respiratory tract infections of Pseudomonas aeruginosa in adults. Zhonghua Jie He He Hu Xi Za Zhi 45, 739–752. doi:10.3760/cma.j.cn112147-20220407-00290
PULMONARY INFECTION ASSEMBLY OF CHINESE THORACIC, S (2022). Chinese expert consensus on the management of lower respiratory tract infections of Pseudomonas aeruginosa in adults. Zhonghua Jie He He Hu Xi Za Zhi 45, 739–752. doi:10.3760/cma.j.cn112147-20220407-00290
Reig, S., Le Gouellec, A., and Bleves, S. (2022). What is new in the anti-Pseudomonas aeruginosa clinical development pipeline since the 2017 WHO alert? Front. Cell Infect. Microbiol. 12, 909731. doi:10.3389/fcimb.2022.909731
Shi, R., Fu, Y., Gan, Y., Wu, D., Zhou, S., and Huang, M. (2023). Use of polymyxin B with different administration methods in the critically ill patients with ventilation associated pneumonia: a single-center experience. Front. Pharmacol. 14, 1222044. doi:10.3389/fphar.2023.1222044
Shi, Y., Wu, J., Mi, W., Zhang, X., Ren, X., Shen, C., et al. (2024). Ceftazidime-avibactam induced renal disorders: past and present. Front. Pharmacol. 15, 1329307. doi:10.3389/fphar.2024.1329307
Soukup, P., Faust, A. C., Edpuganti, V., Putnam, W. C., and Mckinnell, J. A. (2019). Steady-state ceftazidime-avibactam serum concentrations and dosing recommendations in a critically ill patient being treated for Pseudomonas aeruginosa pneumonia and undergoing continuous venovenous hemodiafiltration. Pharmacotherapy 39, 1216–1222. doi:10.1002/phar.2338
Tamma, P. D., Aitken, S. L., Bonomo, R. A., Mathers, A. J., Van Duin, D., and Clancy, C. J. (2022). Infectious diseases society of America 2022 guidance on the treatment of extended-spectrum β-lactamase producing enterobacterales (ESBL-E), carbapenem-resistant enterobacterales (CRE), and Pseudomonas aeruginosa with difficult-to-treat resistance (DTR-P. aeruginosa). Clin. Infect. Dis. 75, 187–212. doi:10.1093/cid/ciac268
Tamma, P. D., Heil, E. L., Justo, J. A., Mathers, A. J., Satlin, M. J., and Bonomo, R. A. (2024). Infectious diseases society of America 2024 guidance on the treatment of antimicrobial-resistant gram-negative infections. Clin. Infect. Dis., ciae403. doi:10.1093/cid/ciae403
Teng, X. Q., Qu, Q., Luo, Y., Long, W. M., Zhuang, H. H., Xu, J. H., et al. (2022). Therapeutic drug monitoring of ceftazidime-avibactam concentrations in carbapenem-resistant K. Pneumoniae-infected patients with different kidney statuses. Front. Pharmacol. 13, 780991. doi:10.3389/fphar.2022.780991
Tsuji, B. T., Pogue, J. M., Zavascki, A. P., Paul, M., Daikos, G. L., Forrest, A., et al. (2019). International consensus guidelines for the optimal use of the polymyxins: endorsed by the American college of clinical pharmacy (ACCP), European society of clinical microbiology and infectious diseases (ESCMID), infectious diseases society of America (IDSA), international society for anti-infective pharmacology (ISAP), society of critical care medicine (SCCM), and society of infectious diseases pharmacists (SIDP). Pharmacotherapy 39, 10–39. doi:10.1002/phar.2209
Wang, Y., Li, C., Wang, J., Bai, N., Zhang, H., Chi, Y., et al. (2022). The efficacy of colistin combined with amikacin or levofloxacin against Pseudomonas aeruginosa biofilm infection. Microbiol. Spectr. 10, e0146822. doi:10.1128/spectrum.01468-22
Wang, Y., Sholeh, M., Yang, L., Shakourzadeh, M. Z., Beig, M., and Azizian, K. (2025). Global trends of ceftazidime-avibactam resistance in gram-negative bacteria: systematic review and meta-analysis. Antimicrob. Resist Infect. Control 14, 10. doi:10.1186/s13756-025-01518-5
WHO (2024). WHO bacterial priority pathogens list, 2024. Available online at: https://www.who.int/publications/i/item/9789240093461 (Accessed March 7, 2025).
Wu, X. L., Long, W. M., Lu, Q., Teng, X. Q., Qi, T. T., Qu, Q., et al. (2022). Polymyxin B-associated nephrotoxicity and its predictors: a retrospective study in carbapenem-resistant gram-negative bacterial infections. Front. Pharmacol. 13, 672543. doi:10.3389/fphar.2022.672543
Xu, C., Zeng, F., Huang, Y., Xu, Q., Yang, Y., Gong, W., et al. (2024). Clinical efficacy of ceftazidime/avibactam combination therapy for severe hospital-acquired pulmonary infections caused by carbapenem-resistant and difficult-to-treat Pseudomonas aeruginosa. Int. J. Antimicrob. Agents 63, 107021. doi:10.1016/j.ijantimicag.2023.107021
Yuan, Q., Guo, L., Li, B., Zhang, S., Feng, H., Zhang, Y., et al. (2023). Risk factors and outcomes of inpatients with carbapenem-resistant Pseudomonas aeruginosa bloodstream infections in China: a 9-year trend and multicenter cohort study. Front. Microbiol. 14, 1137811. doi:10.3389/fmicb.2023.1137811
Keywords: ceftazidime/avibactam, polymyxin B, carbapenem-resistant Pseudomonas aeruginosa, real-world study, propensity score-matched, microbiological efficacy
Citation: Long W-M, Xu W-X, Hu Q, Qu Q, Wu X-L, Chen Y, Wan Q, Xu T-T, Luo Y and Qu J (2025) The efficacy and safety of ceftazidime/avibactam or polymyxin B based regimens for carbapenem-resistant Pseudomonas aeruginosa infection: a multicenter real-world and propensity score-matched study. Front. Pharmacol. 16:1533952. doi: 10.3389/fphar.2025.1533952
Received: 09 December 2024; Accepted: 20 March 2025;
Published: 31 March 2025.
Edited by:
Sonia Alejandra Gomez, National Scientific and Technical Research Council (CONICET), ArgentinaReviewed by:
Maximiliano Gabriel Castro, Jose Bernando Iturraspe Hospital, ArgentinaCopyright © 2025 Long, Xu, Hu, Qu, Wu, Chen, Wan, Xu, Luo and Qu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jian Qu, cXVqaWFuc3RhbmxleUBjc3UuZWR1LmNu
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.