
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Pharmacol. , 19 February 2025
Sec. Drugs Outcomes Research and Policies
Volume 16 - 2025 | https://doi.org/10.3389/fphar.2025.1533709
Background: Statins are one of the most widely prescribed medicines in clinical practice. Their benefits have extended beyond cardiovascular applications to reduce serum uric acid levels. This study aims to investigate the relationship of hyperuricemia with the risk of all-cause mortality among individuals taking statins.
Method: A retrospective cohort study was performed using data from the National Health and Nutrition Examination Survey (NHANES) database between 2005 and 2018. The weighted Cox proportional hazards models were used to investigate the relationship between hyperuricemia and all cause-mortality.
Results: A total of 1,958 participants were enrolled for analysis. Of them 1,429 participants were with normal uric acid levels and 529 people were with hyperuricemia. After 12 years of follow-up, there were 267 participants who died from all-cause mortality in the non-hyperuricemia group and 136 died in the hyperuricemia group. Additionally, 32.49% of participants took more than five kinds of medicines in the non-hyperuricemia group compared to 42.05% participants in the hyperuricemia group. Even after adjusting for confounding factors, we found that the serum uric acid (SUA) level was significantly correlated with all-cause mortality among statin users (HR = 1.13, 95% CI:1.02–1.24, p = 0.0161). Additionally, hyperuricemia resulted in significant increases in all-cause mortality relative to non-hyperuricemia participants in three models (HR = 1.51, 95% CI:1.16–1.96, P = 0.0023).
Conclusion: Although statins have been shown to reduce uric acid levels, hyperuricemia is still significantly associated with the all-cause mortality in people taking statins. Those taking statins and having hyperuricemia should pay special attention to their SUA level.
Serum uric acid (SUA) is a crucial substance that exerts a substantial influence on metabolism and various physiological processes (Wang et al., 2024). Elevated SUA concentrations, as a metabolite of ingested or endogenous guanine and adenine, have been linked to an increased risk of all-cause mortality (Li et al., 2023; Yang et al., 2023). Epidemiological evidence has suggested that various metabolic conditions, including hypertension, diabetes, obesity, and metabolic syndrome, are associated with elevated SUA levels (Crawley et al., 2022).
Hyperuricemia is caused by an increase in the concentration of SUA, which is defined as SUA levels of ≥7 mg/dL in men and ≥6 mg/dL in women (Ren et al., 2024). It is estimated that approximately 38 million adults or 16.9% of the population in the United States are affected by hyperuricemia (Leung et al., 2022). As an important worldwide public health issue, hyperuricemia is reported to be associated with long-term mortality (Yin et al., 2024).
Statins, which are also known as 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitors, are the most broadly prescribed medical regimens to reduce low-density lipoprotein (Khatiwada and Hong, 2024). Due to their pleiotropic effects on physiological function, they are used for the primary prevention of cardiovascular disease in adults (Paparodis et al., 2024). High-quality trials showed that statin therapy was associated with a reduced risk of all-cause and cardiovascular mortality, as well as cardiovascular disease (CVD) events, even in people over the age of 75 years (Chou et al., 2022; Orkaby et al., 2020). Despite these profound effects, statins are renowned for their ability to reduce SUA levels, especially for atorvastatin and simvastatin (Akbari et al., 2024).
Although accumulating evidence reveals that hyperuricemia is associated with an increased risk of all-cause mortality, no studies have yet examined such a relationship among people taking statins. Considering the much-needed survival benefit by statins, it is quite necessary to make further exploration. In this cohort study, we sought to evaluate the relationship of hyperuricemia with the risk of all-cause mortality among individuals taking statins in a large cohort by using the National Health and Nutrition Examination Survey (NHANES) data.
All original data enrolled for analyses were collected from the NHANES database (https://www.cdc.gov/nchs/nhanes/index.htm), which is a publicly available database that includes the results of a nationally representative survey that assesses the health and nutrition status of the US civilian population at different time periods. A total of seven periods, namely, 2005–2006, 2007–2008, 2009–2010, 2011–2012, 2013–2014, 2015–2016, and 2017–2018, are used in the study.
Participants who completed the questionnaire “Prescription Medications” were enrolled in the analyses for further screening. Participants who have taken statins are enrolled for analysis, regardless of the type of statins. Those with missing data including the key laboratory index, smoking and drinking status, and survival status were excluded. All-cause mortality data were drawn from the National Center for Health Statistics (https://www.cdc.gov/nchs/data-linkage/mortality-public.htm). The NCHS Research Ethics Review Board approved all NHANES protocols of the survey (https://www.cdc.gov/nchs/nhanes/irba98.htm). All individual privacy is kept strictly confidential, and all anonymous data were exclusively used for academic research.
Baseline characteristics including laboratory parameters were extracted from the database year by year. The diagnostic criteria of hyperuricemia were defined as SUA ≥6.0 mg/dL in female individuals and ≥7.0 mg/dL in male individuals. Triglycerides (TG) were classified as follows: less than 150 mg/dL (normal), 150–199 mg/dL (elevated), 200–499 mg/dL (high), and 500 mg/dL or above (very high) (Miller et al., 2011). Total cholesterol levels were classified as normal (less than 200 mg/dL), borderline high (200–239 mg/dL), and high (over 240 mg/dL) (He et al., 2021). Glucose levels were determined according to WHO criteria, with a cutoff point of 126 mg/dL. Drinking was defined as either normal or heavy alcohol use, according to the definition of the National Institute on Alcohol Abuse and Alcoholism, with men drinking more than five standard cups and women drinking more than four standard cups on any given day categorized as heavy alcohol use (https://www.niaaa.nih.gov/alcohol-health/overview-alcohol-consumption/moderate-binge-drinking). Smoking statuses were defined using the following question: Do you now smoke cigarettes?
All analyses were performed using the statistical package R (version 4.2) and Empower (Empower XYS 6.0PC Version, X&Y Solutions, Inc., Boston, MA). According to the Centers for Disease Control and Prevention guidelines (https://wwwn.cdc.gov/nchs/nhanes/tutorials/default.aspx), the sample weight was assigned to each person participating in NHANES; therefore, the proposed weighting methodology was used in our analysis. Continuous variables were presented as weighted means and 95% CI, while categorical variables were described as weighted percentages (95% CI). Baseline characteristics were compared using Pearson’s chi-squared tests (categorical variables) or weighted linear regression (continuous variables). The Cox proportional hazard function model was used to investigate the association between hyperuricemia and all cause-mortality. Confounding factors including age, gender, BMI, smoking, drinking, ethnicity, the number of taken medicines, days of statin administration, direct HDL cholesterol, total cholesterol, glucose, creatinine, and triglycerides were adjusted in different models. The overall survival time was illustrated using a Kaplan–Meier curve and compared with a log-rank test with confounding factors adjusted. A two-tailed p-value < 0.05 was considered statistically significant.
The database contains 70,190 participants from 2005 to 2018. Of them, 7,465 had completed the “Prescription Medications” questionnaire and provided the names of statins. After excluding the participants with missing data, 1,958 people were left for further analysis. The selection process is illustrated in Figure 1. A total of 1,429 participants had normal uric acid levels, while 529 people had hyperuricemia. The average age in the non-hyperuricemia group is 62.32 years, while the hyperuricemia group averages 64.25 years.
There were 267 participants who died from of all-cause mortality in the non-hyperuricemia group and 136 people in the hyperuricemia group. Additionally, 907 participants in the non-hyperuricemia group took less than five kinds of medicines compared to 277 participants in the hyperuricemia group. There were 92 participants in the non-hyperuricemia group who had taken more than 10 kinds of medicines compared to 48 participants in the hyperuricemia group. The detailed basic demographic characteristics are shown in Table 1. Seven kinds of statins, namely, simvastatin, lovastatin, atorvastatin, fluvastatin, pitavastatin, pravastatin, and rosuvastatin, were found among these participants (Figure 2).
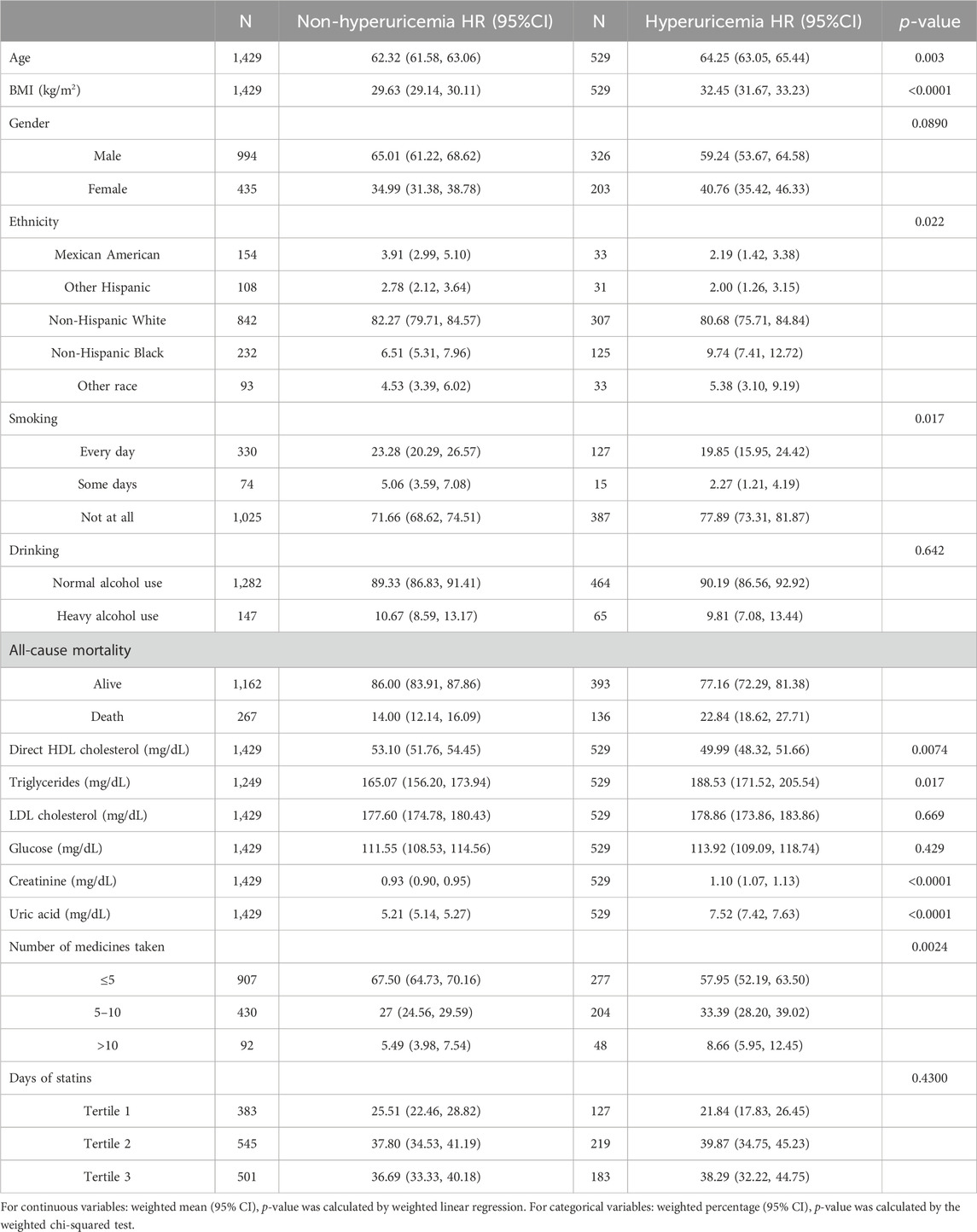
Table 1. Baseline characteristics of the enrolled participants. Data are presented as the weighted mean (95% CI) for continuous variables or weighted percentage (95% CI) for categorical variables.
To elucidate the relationship between hyperuricemia and all-cause mortality in people taking statins, we constructed three models using the Cox proportional hazard function model. The HRs and 95% CIs for these three models are listed in Table 2. Our results show that the SUA level was significantly correlated with the all-cause mortality in the crude model (HR = 1.18, 95% CI:1.06–1.31, P = 0.0028). Even after controlling for confounding factors, the difference was still significant (HR = 1.13, 95% CI:1.02–1.24, and p = 0.0161). Additionally, participants were further divided into hyperuricemia and non-hyperuricemia groups. Compared with the non-hyperuricemia group, hyperuricemia caused significant increases in all-cause mortality (HR = 1.71, 95% CI:1.29–2.27, P = 0.0002) in the crude model. This trend is still significant even after adjusting the confounding factors (HR = 1.51, 95% CI:1.16–1.96, P = 0.0023). The Kaplan–Meier curve is shown in Figure 3 (P = 0.0016).
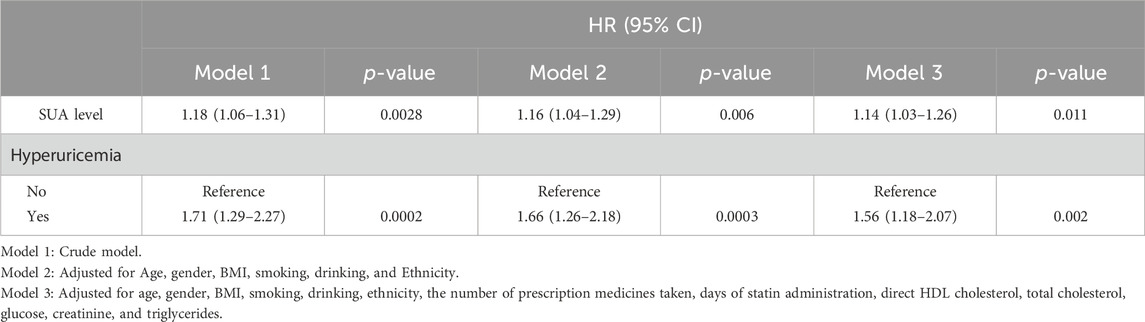
Table 2. Associations between hyperuricemia and the risk of all-cause mortality in participants who have taken statins.
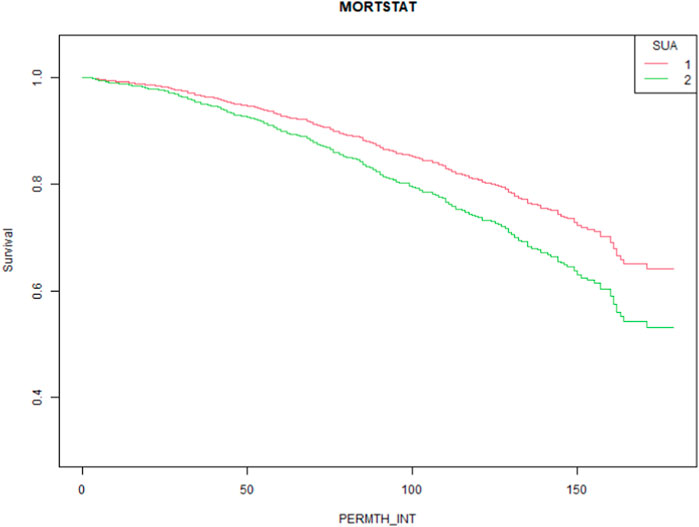
Figure 3. Kaplan–Meier survival curves for all-cause mortality in patients taking statins complicated with normal uric acid (1) and hyperuricemia (2).
We evaluated the interaction of various variables and the relationship between hyperuricemia and all-cause mortality (Table 3). The results revealed that there were no significant interactions found for age, BMI, drinking, smoking, gender, total medicine taken, days of statin administration, triglycerides, glucose, and total cholesterol. Stratified analysis of hyperuricemia and all-cause mortality by other variables in statin users can be found in the Supplementary Materials.
In a comprehensive analysis using the NHANES database, we evaluated the relationship between hyperuricemia and all-cause mortality in people taking statins. Our results revealed that hyperuricemia is significantly associated with the all-cause mortality in people taking statins. This trend still existed even after we adjusted the cofounding factors. To the best of our knowledge, our study is the first to assess the relationship between hyperuricemia and all-cause mortality in people taking statins.
Hyperuricemia, one of the serious health problems for individuals with gout and cardiovascular diseases, is a metabolic disorder resulting from prolonged impaired elimination of uric acid (Prabhakar and Lopez-Candales, 2024; Du L et al., 2024). Data from observational cohort studies and meta-analysis suggest that hyperuricemia is associated with all-cause mortality (Zuo et al., 2016; Han et al., 2023; Rao et al., 2024). In addition, Keller et al. found that statin use was associated with a lower risk of mortality in gout patients (Keller et al., 2018). However, no study has observed whether hyperuricemia is associated with a much higher risk of all-cause mortality in people taking statins. Thus, to address the knowledge gaps, we conducted this retrospective cohort study. We believe these findings warrant extra consideration, especially for people who have taken statins and have hyperuricemia.
Statins are one of the most widely prescribed medicines due to their ability to reduce the risk of cardiovascular and cerebrovascular diseases (Mangione et al., 2022; Lavie et al., 2021; Gan et al., 2024). Studies have revealed that it is not only anti-inflammatory but also aids in plaque stabilization (Cesaro et al., 2024). In addition, a recent meta-analysis found that statins can reduce serum uric acid levels, adding to the body of evidence supporting statins’ role in protecting people dying from CVD (Akbari et al., 2024; Derosa et al., 2016; Athyros et al., 2004). The mechanisms linking statins to lower SUA levels are still not fully elucidated. As SUA may play a role in the formation of free radicals and oxidative stress, we speculate that this effect may be associated with the anti-oxidative and anti-inflammatory effects (Sorensen et al., 2019; Athyros et al., 2004).
Although we observed a relationship between hyperuricemia and all-cause mortality, there are several limitations that should also be noted. First, we only observed all-cause mortality in people taking stains. As we know, statins mainly play a role in preventing cardiovascular and cerebrovascular events; nevertheless, the all-cause mortality can be influenced by a lot of factors. Thus, the results of this study should be carefully evaluated in a large cohort. Second, the limited sample size limits the importance of our research. Although the total number of participants in the NHANES was large, participants with missing variables that were incorporated in the analysis were removed from the study, resulting in a significantly decreased number of enrolled participants. Third, the retrospective design and reliance on observational data limit the ability to draw causal conclusions. In addition, some selection bias cannot be excluded from the analysis. Thus, the discovery of our findings warrants further validation in future clinical practice.
Statins are widely used in the prevention of cardiovascular and cerebrovascular diseases. Recent studies showed that statins can also help in reducing SUA levels. Our study revealed that hyperuricemia is significantly associated with the all-cause mortality in people taking statins. The findings hold significant clinical implications, emphasizing the importance of patients taking statins being aware of the potential negative impact of hyperuricemia. However, due to the bias caused by the small sample size, future studies in a large cohort are warranted to verify the conclusion.
The original contributions presented in the study are included in the article/Supplementary Material; further inquiries can be directed to the corresponding authors.
The studies involving humans were approved by the NCHS Research Ethics Review Board. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study.
BZ: investigation, methodology, and writing–review and editing. ZH: investigation and writing–review and editing. MW: data curation and writing–review and editing. JH: data curation and writing–review and editing. ZZ: supervision, validation, and writing–review and editing. WG: supervision, validation, and writing–review and editing. LY: data curation, supervision, validation, and writing–review and editing.
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
The authors would like to thank all investigators and participants of NHANES for their contribution to the data collection.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The authors declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2025.1533709/full#supplementary-material
Akbari, A., Razmi, M., Rafiee, M., Watts, G. F., and Sahebkar, A. (2024). The effect of statin therapy on serum uric acid levels: a systematic review and meta-analysis. Curr. Med. Chem. 31 (13), 1726–1739. doi:10.2174/0929867330666230207124516
Athyros, V. G., Elisaf, M., Papageorgiou, A. A., Symeonidis, A. N., Pehlivanidis, A. N., Bouloukos, V. I., et al. (2004). Effect of statins versus untreated dyslipidemia on serum uric acid levels in patients with coronary heart disease: a subgroup analysis of the Greek Atorvastatin and Coronary-heart-disease Evaluation (GREACE) study. Am. J. Kidney Dis. 43 (4), 589–599. doi:10.1053/j.ajkd.2003.12.023
Cesaro, A., Acerbo, V., Indolfi, C., Filardi, P. P., and Calabro, P. (2024). The clinical relevance of the reversal of coronary atherosclerotic plaque. Eur. J. Intern. Med. 129, 16–24. doi:10.1016/j.ejim.2024.08.010
Chou, R., Cantor, A., Dana, T., Wagner, J., Ahmed, A. Y., Fu, R., et al. (2022). Statin use for the primary prevention of cardiovascular disease in adults: updated evidence report and systematic review for the US preventive services task force. JAMA-J. Am. Med. Assoc. 328 (8), 754–771. doi:10.1001/jama.2022.12138
Crawley, W. T., Jungels, C. G., Stenmark, K. R., and Fini, M. A. (2022). U-shaped association of uric acid to overall-cause mortality and its impact on clinical management of hyperuricemia. Redox Biol. 51, 102271. doi:10.1016/j.redox.2022.102271
Derosa, G., Maffioli, P., Reiner, Z., Simental-Mendia, L. E., and Sahebkar, A. (2016). Impact of statin therapy on plasma uric acid concentrations: a systematic review and meta-analysis. Drugs 76 (9), 947–956. doi:10.1007/s40265-016-0591-2
Du, L., Zong, Y., Li, H., Wang, Q., Xie, L., Yang, B., et al. (2024). Hyperuricemia and its related diseases: mechanisms and advances in therapy. Signal Transduct. Target. Ther. 9 (1), 212. doi:10.1038/s41392-024-01916-y
Gan, T. M., Ye, Y. Y., Mo, G. L., and Li, J. Y. (2024). Progress of uric acid in cardiovascular disease. Cardiovasc. Endocrinol. Metab. 13 (2), e0300. doi:10.1097/XCE.0000000000000300
Han, Y., Cao, Y., Han, X., Di, H., Yin, Y., Wu, J., et al. (2023). Hyperuricemia and gout increased the risk of long-term mortality in patients with heart failure: insights from the National Health and Nutrition Examination Survey. J. Transl. Med. 21 (1), 463. doi:10.1186/s12967-023-04307-z
He, G. D., Liu, X. C., Liu, L., Yu, Y. L., Chen, C. L., Huang, J. Y., et al. (2021). A nonlinear association of total cholesterol with all-cause and cause-specific mortality. Nutr. Metab. 18 (1), 25. doi:10.1186/s12986-021-00548-1
Keller, S. F., Rai, S. K., Lu, N., Oza, A., Jorge, A. M., Zhang, Y., et al. (2018). Statin use and mortality in gout: a general population-based cohort study. Semin. Arthritis Rheum. 48 (3), 449–455. doi:10.1016/j.semarthrit.2018.03.007
Khatiwada, N., and Hong, Z. (2024). Potential benefits and risks associated with the use of statins. Pharmaceutics 16 (2), 214. doi:10.3390/pharmaceutics16020214
Lavie, G., Hoshen, M., Leibowitz, M., Benis, A., Akriv, A., Balicer, R., et al. (2021). Statin therapy for primary prevention in the elderly and its association with new-onset diabetes, cardiovascular events, and all-cause mortality. Am. J. Med. 134 (5), 643–652. doi:10.1016/j.amjmed.2020.09.058
Leung, N., Yip, K., Pillinger, M. H., and Toprover, M. (2022). Lowering and raising serum urate levels: off-label effects of commonly used Medications. Mayo Clin. Proc. 97 (7), 1345–1362. doi:10.1016/j.mayocp.2022.02.027
Li, B., Chen, L., Hu, X., Tan, T., Yang, J., Bao, W., et al. (2023). Association of serum uric acid with all-cause and cardiovascular mortality in diabetes. Diabetes Care 46 (2), 425–433. doi:10.2337/dc22-1339
Mangione, C. M., Barry, M. J., Nicholson, W. K., Cabana, M., Chelmow, D., Coker, T. R., et al. (2022). Statin use for the primary prevention of cardiovascular disease in adults: US preventive services task force recommendation statement. JAMA-J. Am. Med. Assoc. 328 (8), 746–753. doi:10.1001/jama.2022.13044
Miller, M., Stone, N. J., Ballantyne, C., Bittner, V., Criqui, M. H., Ginsberg, H. N., et al. (2011). Triglycerides and cardiovascular disease: a scientific statement from the American Heart Association. Circulation 123 (20), 2292–2333. doi:10.1161/CIR.0b013e3182160726
Orkaby, A. R., Driver, J. A., Ho, Y. L., Lu, B., Costa, L., Honerlaw, J., et al. (2020). Association of statin use with all-cause and cardiovascular mortality in US veterans 75 Years and older. JAMA-J. Am. Med. Assoc. 324 (1), 68–78. doi:10.1001/jama.2020.7848
Paparodis, R. D., Bantouna, D., Livadas, S., and Angelopoulos, N. (2024). Statin therapy in primary and secondary cardiovascular disease prevention. Curr. Atheroscler. Rep. 27 (1), 21. doi:10.1007/s11883-024-01265-9
Prabhakar, A. P., and Lopez-Candales, A. (2024). Uric acid and cardiovascular diseases: a reappraisal. Postgrad. Med. 136 (6), 615–623. doi:10.1080/00325481.2024.2377952
Rao, J., Lai, R., Jiang, L., Wen, W., and Chen, H. (2024). Hyperuricaemia elevates risk of short-term readmission and mortality in patients with heart failure. Open Heart 11 (2), e002830. doi:10.1136/openhrt-2024-002830
Ren, T., Zhou, E., Wu, J., Wang, C., and Yin, Y. (2024). Systemic inflammation markers independently associated with increased mortality in individuals with hyperuricemia: results from the NHANES prospective cohort study. Immun. Inflamm. Dis. 12 (10), e70032. doi:10.1002/iid3.70032
Sorensen, A. L., Hasselbalch, H. C., Nielsen, C. H., Poulsen, H. E., and Ellervik, C. (2019). Statin treatment, oxidative stress and inflammation in a Danish population. Redox Biol. 21, 101088. doi:10.1016/j.redox.2018.101088
Wang, W., Tu, M., Qiu, X. P., Tong, Y., and Guo, X. L. (2024). The interplay of systemic inflammation and oxidative stress in connecting perirenal adipose tissue to hyperuricemia in type 2 diabetes mellitus: a mediation analysis. J. Inflamm. Res. 17, 11319–11329. doi:10.2147/JIR.S488964
Yang, Z., Lv, T., Lv, X., Wan, F., Zhou, H., Wang, X., et al. (2023). Association of serum uric acid with all-cause and cardiovascular mortality in obstructive sleep apnea. Sci. Rep. 13 (1), 19606. doi:10.1038/s41598-023-45508-2
Yin, Y., Zhou, E., and Wu, J. (2024). Association between hyperuricemia and long-term mortality in patients with hypertension: results from the NHANES 2001-2018. Front. Cardiovasc. Med. 11, 1306026. doi:10.3389/fcvm.2024.1306026
Keywords: Statin (HMG-CoA reductase inhibitor), hyperuricemia, all-cause mortality, National Health and Nutrition Examination Survey, serum uric acid
Citation: Zhu B, He Z, Wu M, Huo J, Zhao Z, Guo W and Yang L (2025) Association between hyperuricemia and all-cause mortality in people taken Statins: a retrospective cohort study. Front. Pharmacol. 16:1533709. doi: 10.3389/fphar.2025.1533709
Received: 24 November 2024; Accepted: 20 January 2025;
Published: 19 February 2025.
Edited by:
Sheyu Li, Sichuan University, ChinaCopyright © 2025 Zhu, He, Wu, Huo, Zhao, Guo and Yang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Zhigang Zhao, MTAyMnp6Z0BzaW5hLmNvbQ==; Wei Guo, Z3Vvd2VpMTAxMEAxNjMuY29t; Li Yang, MDUzMHlhbmdsaUAxNjMuY29t
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.