
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
MINI REVIEW article
Front. Pharmacol. , 26 February 2025
Sec. Neuropharmacology
Volume 16 - 2025 | https://doi.org/10.3389/fphar.2025.1500313
This article is part of the Research Topic Pain and Pain-Related Neuropsychiatric Disorders: From Mechanistic Insights to Innovative Therapeutic Strategies View all 16 articles
Chronic pain affects a significant portion of adults and is linked to psychosocial issues, cognitive dysfunction, and psychiatric disorders, complicating treatment. Attention deficit hyperactivity disorder (ADHD) is increasingly recognized as a contributing factor to chronic pain, particularly nociplastic pain, with a notable prevalence of comorbidity between ADHD and conditions like fibromyalgia and chronic low back pain. ADHD behaviors such as impulsivity and overactivity can exacerbate pain by leading patients to seek risky treatments or discontinue care prematurely. ADHD medications are expected to alleviate pain severity by improving associated cognitive dysfunction and addressing central sensitization, a fundamental mechanism in chronic pain. Brain abnormalities in ADHD contribute to increased spontaneous activity in the anterior cingulate cortex-posterior insular pathway due to neuroinflammation, alterations in action potential firing, and changes in transmission pathways in the spinal dorsal horn. Additionally, increased norepinephrine synthesis and reduced transmission efficiency amplify nociceptive information from the periphery and facilitate central sensitization in ADHD. Beyond typical ADHD medications like central stimulants, norepinephrine reuptake inhibitors, and alpha-2 receptor agonists, various antidepressants, mood stabilizers, antipsychotics, Parkinson’s disease medications, and antidementia medications have proven effective in alleviating ADHD symptoms. These medications, effective for ADHD, may offer innovative solutions for managing chronic pain by targeting both the cognitive/behavioral dysfunction and central sensitization observed in chronic pain comorbid with ADHD. Further research into these mechanisms could lead to new, more effective pharmacological treatments for chronic pain with comorbid ADHD, a condition that is often overlooked.
Chronic pain affects 13%–50% of adults (Mills et al., 2019) and is associated with psychosocial factors, cognitive dysfunction, and psychiatric disorders. Moreover, inadequate management of pain and the underlying physical conditions that cause it can also lead to cognitive impairment and psychiatric disorders. This complexity makes assessing and treating chronic pain particularly challenging and imposes an extremely high economic burden (Sarria-Santamera et al., 2022). Consequently, elucidating the pathophysiological mechanisms of chronic pain is vital for discovering new pharmacological targets.
Understanding the cognitive and behavioral characteristics of patients with chronic pain, both clinically and in daily life, alongside psychiatric disorders, is crucial for implementing effective pharmacological strategies targeting these mechanisms.
Patients with chronic pain resistant to multidisciplinary treatment often experience repeated accidents (Swanson et al., 1977), which has been linked to attention deficit hyperactivity disorder (ADHD), a neurodevelopmental disorder (Kaplan and Kaplan, 2006). Chronic pain patients frequently display three behavioral traits (Flor and Turk, 2015): (1) overconcentration on pain and susceptibility to distraction, (2) overactivity exceeding personal limits (traditionally called “ergomania”) (Vlaeyen and Morley, 2004), and (3) persistent, intense anger—features also characteristic of ADHD (American Psychiatric Association, 2013). Due to impulsivity and impatience, ADHD patients often pursue invasive, high-risk treatments (e.g., surgery, tooth extraction, opioids) for rapid relief or abandon treatments prematurely if results are not immediate, leading to “doctor shopping” (Kasahara et al., 2023d). Notably, 72.5% of chronic pain patients have comorbid ADHD, and ADHD medications can reduce the pain NRS score by 3.5 points (61.5%), indicating that ADHD comorbidity may drive cognitive and behavioral dysfunction in chronic pain (Kasahara et al., 2020).
Over 80% of adult ADHD cases go undiagnosed, especially in psychiatric settings (Ginsberg et al., 2014). Because most chronic pain patients receive care from orthopedic surgeons, rheumatologists, and pain specialists with limited ADHD expertise, comorbid ADHD often remains overlooked. Recognizing this gap could provide a breakthrough for new, behaviorally tailored pharmacotherapies.
Herein, we review clinical and basic research on the relationship between chronic pain and ADHD and explore the potential pathophysiological mechanisms involved. We also examine effective ADHD medications for chronic pain, offering insights into novel pharmacotherapeutic approaches.
An epidemiological study revealed an association between ADHD symptoms and work-related pain (Stickley et al., 2016). In 2017, chronic pain—previously categorized as psychogenic or somatoform—was redefined as nociplastic pain (NP), recognized alongside nociceptive and neuropathic pain (Kosek et al., 2016; Aydede and Shriver, 2018). NP is believed to result from central sensitization, involving plastic changes in central nervous system circuits that amplify nociceptive signals (Kosek et al., 2021; Treede et al., 2019; Nijs et al., 2021). NP rarely appears in isolation, instead presenting with hyperalgesia, fatigue, sensory hypersensitivity, sleep disturbances, mood disorders, or cognitive dysfunction in concentration and memory (Kosek et al., 2021), which—combined with psychosocial factors—makes treatment particularly challenging.
Recent findings indicate ADHD, a developmental disorder, frequently coexists with representative NP-related conditions such as fibromyalgia (Pallanti et al., 2021), chronic low back pain (Kasahara et al., 2021b), idiopathic orofacial pain (Kasahara et al., 2023b), chronic chest pain (Zain et al., 2023), and chronic abdominal pain (Asztély et al., 2019; Kasahara et al., 2024). ADHD may contribute to central sensitization and cognitive impairments in NP, including attention deficits and sensory hypersensitivity (Ibrahim and Hefny, 2022). Historical figures with NP, such as John F. Kennedy, who suffered from chronic low back pain (Kasahara et al., 2022a), and Margaret Mitchell, the author of Gone with the Wind, who struggled with fibromyalgia and experienced multiple car accidents (Kasahara et al., 2021a), were also speculated to have had comorbid ADHD.
The coexistence of ADHD and chronic pain is thought to stem from dual impairments in cognitive-emotional factors and motor control associated with ADHD. ADHD exacerbates pain, and pain, in turn, worsens ADHD, with central nervous system inflammation potentially perpetuating this relationship (Battison et al., 2023) (Figure 1). Individuals with ADHD have been shown to exhibit considerably more motor control issues and increased muscle tension than those without ADHD, which are also associated with widespread and severe pain (Stray et al., 2013).
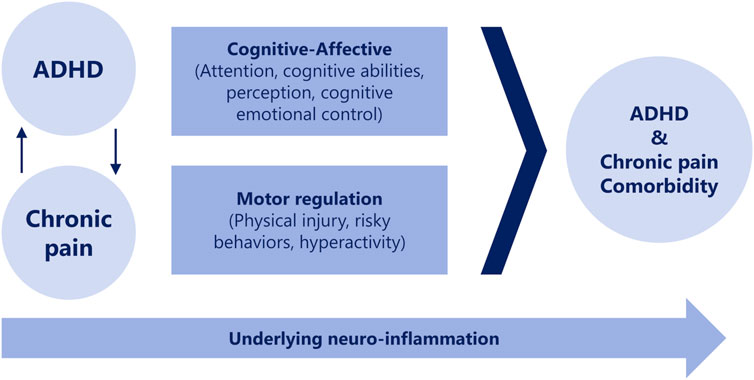
Figure 1. Potential associations between chronic pain and ADHD over time. (Battison et al., 2023). ADHD, attention-deficit hyperactivity disorder.
Furthermore, when ADHD accompanies NP, ADHD medications have been reported to improve pain and the associated cognitive dysfunctions. (Kasahara et al., 2017; Kasahara et al., 2022b; Kasahara et al., 2023a; Kasahara et al., 2023b; Kasahara et al., 2023c; Kasahara et al., 2023d; Kasahara et al., 2023e; Zain et al., 2023; Kasahara et al., 2024). ADHD medications administered to patients with NP have also been shown to regulate blood flow in the prefrontal cortex, precuneus, anterior cingulate cortex, and insular cortex—regions involved in the core pain matrix and related emotional processing areas (Garcia-Larrea and Peyron, 2013; Kasahara et al., 2023a; Kasahara et al., 2023c; Kasahara et al., 2023e; Kasahara et al., 2024). Notably, increased blood flow in the precuneus showed a positive correlation with NP severity scores. In the group that responded to methylphenidate, the precuneus was identified as a region of significantly elevated blood flow before treatment compared to that of after treatment. In typical cases, a decrease in precuneus hyperperfusion was accompanied by an increase in blood flow in the prefrontal cortex (Takahashi et al., 2024). Moreover, methylphenidate has been shown to alleviate motor control issues and high muscle tension in individuals with ADHD (Stray et al., 2009. See Additional Video Files 1–4.). Additionally, ADHD medications have been found to improve family relationships, which are significant psychosocial factors contributing to the maintenance and exacerbation of chronic pain (Kasahara et al., 2024). Therefore, ADHD medications are gaining attention as a new approach for treating NP, which is often difficult to manage.
However, as previously mentioned, many chronic pain patients are treated by non-psychiatric specialists such as orthopedic surgeons, rheumatologists, or pain specialists. For these physicians, diagnosing ADHD or prescribing ADHD medications is not straightforward. Therefore, when clinical behavioral characteristics of ADHD are observed, it is practical to use self-administered questionnaires such as the Adult ADHD Self-Report Scale (Kessler et al., 2005) or the Conners' Adult ADHD Rating Scale (Conners et al., 1999) for screening and to consult with a psychiatric specialist. Both scales have also been utilized in clinical research on chronic pain (Stickley et al., 2016; Kasahara et al., 2021b). Patients with coexisting ADHD often exhibit the following behavioral characteristics (Kooij and Francken, 2010): difficulty maintaining attention and concentration, which leads to omissions when responding to questionnaires; a tendency to become bored easily and frequently change jobs; procrastination on tasks requiring effort, such as returning to work or completing administrative procedures; reliance on family members for difficult tasks; disorganized speech, resulting in scattered conversations; tardiness or forgetting appointment dates; organizational difficulties, leading to overfilled bags; fidgeting with hands or feet even while seated; hyperactivity, resulting in involvement in numerous activities and subsequent exhaustion; excessive talking; impatience and impulsive pursuit of high-risk treatments, with demands such as ‘take the pain away immediately; ’ inability to control anger; over-involvement with others; and a dislike of waiting, resulting in a preference for the earliest possible appointment slots.”
NP is associated with central sensitization (Kosek et al., 2021). Recently, basic research suggests that ADHD is more likely to cause central sensitization. At least 21 types of rodent ADHD model animals have been reported in basic research, with eight frequently cited models being identified as the most notable (Kantak K. M., 2022; Table 1).
In spontaneously hypertensive rats (SHR), an animal model for ADHD, noxious stimulus-induced analgesia (NSIA) (Tambeli et al., 2009) was weaker compared to the control group. Additionally, in vivo studies have shown that the concentration of norepinephrine (NE) in the dorsal horn of the spinal cord, which increases in response to nociceptive stimuli, decreases in SHR (Suto et al., 2023). This decrease suggests a weakened endogenous analgesia, including the descending inhibitory system, in ADHD.
In spinal cord slices, SHR exhibited a larger immunostaining area for NE-synthesizing enzymes in the dorsal horn than that of the control group, alongside higher extracellular NE concentrations than those of the control group. This suggests more active NE synthesis under pain-free conditions (Suto et al., 2023). Conversely, SHR exhibited a larger staining area for the norepinephrine transporter (NET) but a smaller staining area for the α2A receptor, the target receptor for NE, compared to the control group. This pattern indicates that excessive NE production in a pain-free state leads to NET overexpression and α2A receptor downregulation, resulting in diminished NE activity during NSIA and weakened descending inhibition.
Similarly, 6-hydroxydopamine (6-OHDA) mice, another ADHD model, showed shorter latency and a lower threshold for licking hind limbs in response to thermal or mechanical stimuli than those shown by controls, indicating heightened baseline nociceptive sensitivity (Bouchatta et al., 2022). In these mice, inhibitory synaptic connections in the spinal dorsal horn lamina II were similar to controls, but excitatory connections were significantly increased (Bouchatta et al., 2022). This suggests that ADHD-related anatomical changes, including increased excitatory pathways in the dorsal horn, may promote pain sensitization.
Central sensitization mechanisms have also been studied by examining the pathway from the anterior cingulate cortex (ACC) to the posterior insula (PI), known as the ACC-PI pathway (Bouchatta et al., 2022). In 6-OHDA-induced ADHD, spontaneous activity in this pathway was higher than that in controls, and ACC activity evoked by mechanical stimulation of the contralateral hind limb was stronger than that evoked in controls. This intensified activity boosted firing in second-order nociceptive neurons in the spinal dorsal horn, further lowering the pain threshold. Conversely, inhibiting the ACC-PI pathway in 6-OHDA mice more effectively suppressed dorsal horn action potentials and raised the pain threshold. Hence, ADHD-driven ACC-PI activity can both increase and reduce pain sensitivity, potentially triggering overactivity (ergomania).
Regarding the cause of elevated spontaneous ACC-PI activity, a sex-specific neuroinflammatory response to dopamine (DA) neuronal depletion in 6-OHDA mice has been reported (Meseguer-Beltrán et al., 2023). In males, DA depletion triggered inflammation only in the ACC, resulting in hyperactivity but no hyperalgesia. In females, inflammation occurred in the ACC-PI pathway, leading to hyperalgesia but not hyperactivity; reducing this inflammation alleviated hyperalgesia. These findings may explain why females experience higher rates of conditions like fibromyalgia (Queiroz, 2013) or burning mouth syndrome (Khawaja et al., 2023), as DA dysfunction in females may manifest primarily as hyperalgesia rather than hyperactivity.
In summary, in SHR and 6-OHDA mice, which have been reported to demonstrate a relationship between pain and ADHD, brain abnormalities in ADHD are thought to increase the spontaneous activity of the ACC-PI pathway owing to neuroinflammation, changes in the firing of action potentials and transmission pathways in the spinal dorsal horn, increased NE synthesis, and reduced transmission efficiency. These factors may amplify peripheral nociceptive information, increasing susceptibility to central sensitization. Furthermore, it has been shown that in SHR and 6-OHDA mice, hyperalgesia caused by central sensitization and ADHD symptoms, such as hyperactivity and inattention, can be improved by atomoxetine—a selective norepinephrine reuptake inhibitor (NRI) used as an ADHD medication (Sifeddine et al., 2023; Suto et al., 2023).
However, among the ADHD models listed in Table 1, only the SHR and 6-OHDA mice have been reported to show a relationship between pain and ADHD, while such investigations have not been conducted for other ADHD model animals. Therefore, there is room to explore the relationship between pain and ADHD in these other models as well. Additionally, ADHD is believed to result from gene-environment interactions, highlighting the need to develop models that integrate both genetic and environmental factors. This integration remains a challenge for ADHD models as a whole (Regan et al., 2022). Furthermore, since all rodent ADHD models exhibit hyperactivity or impulsivity, developing a model that specifically represents inattentive-type ADHD without hyperactivity or impulsivity remains an important task.
Medications that are effective for treating ADHD may also be effective in treating chronic pain and its associated cognitive dysfunction mediated by central sensitization. Here, we briefly introduce the representative drug candidates (Table 2).
The main ADHD medications include the central stimulant methylphenidate, the selective NRI atomoxetine, the α2 receptor agonist guanfacine, and the central stimulant lisdexamfetamine (Stahl, 2021). There have been reports that the first three of these drugs have shown improvements in chronic pain and its associated cognitive dysfunction (Kasahara et al., 2023a; Kasahara et al., 2023e; Zain et al., 2023). However, even with these so-called ADHD medications approved by the Food and Drug Administration, more than 33% of children and over 50% of adults discontinue treatment within a year due to suboptimal efficacy or tolerance issues (Childress A., 2024). Therefore, alternative ADHD treatments that have proven effective for ADHD, as discussed below, are also considered pertinent (Buoli et al., 2016).
Other central stimulants such as amphetamines, modafinil, and pemoline are also considered effective for ADHD (Stahl, 2020). It has been suggested that the amphetamine, which John F. Kennedy used to treat back pain, may have also inadvertently improved ADHD symptoms (Kasahara et al., 2022a).
Additionally, antidepressants, such as duloxetine and venlafaxine, which are norepinephrine-serotonin reuptake inhibitors, have been reported to improve ADHD symptoms (Buoli et al., 2016) and NP (Kasahara et al., 2024). Traditional tricyclic antidepressants (TCAs), used to treat chronic pain (Feinmann and Harris, 1984), also improve ADHD (Banaschewski et al., 2004). TCAs inhibit norepinephrine reuptake. Dopamine reuptake occurs via norepinephrine transporters in the prefrontal cortex. Consequently, TCAs enhance both norepinephrine and dopamine levels in this region. Among TCAs, desipramine is considered the most effective for ADHD, whereas imipramine, nortriptyline, and amitriptyline are slightly less effective. On the other hand, clomipramine and protriptyline are deemed less effective and generally unsuitable due to intolerable side effects. The effects of TCAs on ADHD primarily affect behavioral symptoms rather than cognitive symptoms. Furthermore, it has been suggested that their effectiveness in treating chronic pain may have been due to the improvement of undiagnosed coexisting ADHD (Kasahara et al., 2023d). Other antidepressants, such as the selective serotonin reuptake inhibitor paroxetine, the NE-DA reuptake inhibitor bupropion, and the selective NRI reboxetine, are also considered effective for ADHD (Buoli et al., 2016). Regarding the role of serotonin in ADHD treatment, studies report that hyperactivity caused by high extracellular dopamine levels in 6-OHDA mice is regulated and improved by enhancing serotonergic tone (Regan et al., 2022). Furthermore, mood stabilizers such as lithium and lamotrigine are also considered effective for ADHD (Buoli et al., 2016; Sablaban and Sivananthan, 2022). These agents have been shown to improve ADHD symptoms associated with addictive behaviors, intense anger, and comorbid bipolar disorder (Buoli et al., 2016). Notably, the effectiveness of lithium in treating ADHD has been reported to be comparable to that of methylphenidate (Dorrego et al., 2002).
Haloperidol, chlorpromazine (Stahl, 2020), risperidone, and aripiprazole (Ghanizadeh, 2013; Lamberti et al., 2016) are effective treatments for ADHD. There have also been reports that risperidone (Kasahara et al., 2022b) and aripiprazole (Kasahara et al., 2011; Kasahara et al., 2023d) have improved chronic pain. Haloperidol and chlorpromazine are considered useful for managing hyperactivity and aggression in ADHD (Stahl, 2020). Risperidone and aripiprazole have been found beneficial for ADHD patients experiencing anxiety, irritability, depression, anger, or self-injurious behavior (Alsayouf, 2024). Antipsychotics are thought to ameliorate ADHD by addressing these cognitive-emotional factors. Notably, the effectiveness of risperidone in treating ADHD symptoms has been reported to be comparable to that of methylphenidate (Arabgol et al., 2015).
Additionally, although antipsychotics block dopamine D2 receptors and may initially seem counterproductive for ADHD, which is typically characterized by low dopamine levels, the “Complex DA Model” has been proposed to explain this phenomenon (Yanofski, 2010). According to this model, ADHD involves low tonic dopamine levels and high DA bursts. Stimulants act presynaptically to increase tonic dopamine, suppress DA bursts, and downregulate postsynaptic receptors. Conversely, antipsychotics like risperidone and aripiprazole function postsynaptically to increase tonic dopamine, suppress DA bursts, and upregulate postsynaptic receptors. Therefore, while stimulants and antipsychotics are each effective for ADHD when used alone, their combination proves particularly effective. This combined approach acts on both pre- and postsynaptic mechanisms, increasing tonic dopamine, suppressing DA bursts, and stabilizing postsynaptic receptor regulation. Consequently, this strategy is believed to prevent tolerance to treatment, minimize side effects due to receptor upregulation, and allow for low medication dosages.
DA agonists such as pramipexole (Kurlan et al., 2012) and the selective monoamine oxidase B inhibitor selegiline (Buoli et al., 2016; Akhondzadeh et al., 2003), commonly used to treat Parkinson’s disease, have also been found to be effective for ADHD by enhancing DA neurotransmission at the synapse. Pramipexole improves chronic low back pain (Kasahara et al., 2023c) and fibromyalgia (Holman and Myers, 2005). Clonidine, an α2 agonist like guanfacine and an antihypertensive, is considered effective for ADHD (Stahl, 2020) and has shown efficacy in treating chronic pain (Guay, 2001). Additionally, cholinesterase inhibitors such as donepezil (Doyle et al., 2006) and galantamine (Buoli et al., 2016), along with the N-methyl-D-aspartate receptor antagonist memantine (Surman et al., 2013), typically used as anti-dementia drugs, are known to activate dopamine neurotransmission and have been reported to be effective against ADHD. These medications may also potentially improve chronic pain.
The contents of this mini-review can be summarized as follows: The cognitive-emotional and motor control issues associated with ADHD mutually amplify and exacerbate both ADHD and pain. Neuroinflammation and alterations in neurotransmission within the ADHD brain contribute to central sensitization, amplifying nociceptive input and promoting pain chronicity. ADHD medications can correct the increased blood flow in the precuneus and decreased blood flow in the prefrontal cortex, thereby improving cognitive-emotional factors such as anxiety, depression, anger, and aggression, as well as motor control issues like increased muscle tension. These treatments have the potential to alleviate not only chronic pain itself but also its associated psychosocial issues. However, it is assumed that the ADHD phenotype encompasses multiple models, each responding differently to medication. Therefore, instead of limiting treatment to a single medication, it is pertinent to implement a treatment algorithm composed of multiple medications with different mechanisms of action, as discussed in this review (e.g., Kasahara et al., 2023d).
In the future, to develop innovative pharmacological treatments for chronic pain, it is considered important to discover approaches that utilize ADHD medications targeting the clinical features of ADHD coexisting with chronic pain and the pathophysiological mechanisms of central sensitization.
SK: Writing–original draft, Writing–review and editing. MT: Writing–original draft, Writing–review and editing. TS: Writing–original draft, Writing–review and editing. TM: Writing–original draft, Writing–review and editing. HO: Writing–original draft, Writing–review and editing. S-IN: Writing–original draft, Writing–review and editing.
The author(s) declare that financial support was received for the research, authorship, and/or publication of this article. This paper was supported by a Grant-in-Aid for Scientific Research (C) from the Japan Society for the Promotion of Science [Grant numbers: 20K07755, 24K13083].
We would like to thank Editage (www.editage.com) for English language editing.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The author(s) declare that no Generative AI was used in the creation of this manuscript.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
NP, nociplastic pain; 6-OHDA, 6-hydroxydopamine; SHR, spontaneously hypertensive rats; NSIA, noxious stimulus-induced analgesia; NE, norepinephrine; ACC, anterior cingulate cortex; PI, posterior insula; ACC-PI pathway, anterior cingulate cortex posterior insula; DA, dopamine; NRI, norepinephrine reuptake inhibitor.
Akhondzadeh, S., Tavakolian, R., Davari-Ashtiani, R., Arabgol, F., and Amini, H. (2003). Selegiline in the treatment of attention deficit hyperactivity disorder in children: a double blind and randomized trial. Prog. Neuropsychopharmacol. Biol. Psychiatry 27, 841–845. doi:10.1016/S0278-5846(03)00117-9
Alsayouf, H. A. (2024). Growing evidence of pharmacotherapy effectiveness in managing attention-deficit/hyperactivity disorder in young children with or without autism spectrum disorder: a minireview. Front. Psychiatry 15, 1408876. doi:10.3389/fpsyt.2024.1408876
American Psychiatric Association (2013). Diagnostic and Statistical Manual of Mental Disorders, DSM-5. Washington, DC: American Psychiatric Association.
Arabgol, F., Panaghi, L., and Nikzad, V. (2015). Risperidone versus methylphenidate in treatment of preschool children with attention-Deficit Hyperactivity Disorder. Iran. J. Pediatr. 25, e265. doi:10.5812/ijp.265
Asztély, K., Kopp, S., Gillberg, C., Waern, M., and Bergman, S. (2019). Chronic pain and health-related quality of life in women with autism and/or ADHD: A prospective longitudinal study. J. Pain Res. 12, 2925–2932. doi:10.2147/jpr.s212422
Aydede, M., and Shriver, A. (2018). Recently introduced definition of “nociplastic pain” by the International Association for the Study of Pain needs better formulation. Pain 159, 1176–1177. doi:10.1097/j.pain.0000000000001184
Banaschewski, T., Roessner, V., Dittmann, R. W., Janardhanan Santosh, P., and Rothenberger, A. (2004). Non?stimulant medications in the treatment of ADHD. Eur. Child Adolesc. Psychiatry 13, I102–I116. doi:10.1007/s00787-004-1010-x
Battison, E. A. J., Brown, P. C. M., Holley, A. L., and Wilson, A. C. (2023). Associations between chronic pain and Attention-Deficit Hyperactivity Disorder (ADHD) in youth: A scoping review. Child. (Basel) 10, 142. doi:10.3390/children10010142
Bouchatta, O., Aby, F., Sifeddine, W., Bouali-Benazzouz, R., Brochoire, L., Manouze, H., et al. (2022). Pain hypersensitivity in a pharmacological mouse model of attention-deficit/hyperactivity disorder. Proc. Natl. Acad. Sci. U. S. A. 119, e2114094119. doi:10.1073/pnas.2114094119
Buoli, M., Serati, M., and Cahn, W. (2016). Alternative pharmacological strategies for adult ADHD treatment: a systematic review. Expert Rev. Neurother. 16, 131–144. doi:10.1586/14737175.2016.1135735
Childress, A. (2024). Recent advances in pharmacological management of attention-deficit/hyperactivity disorder: moving beyond stimulants. Expert Opin. Pharmacother. 25, 853–866. doi:10.1080/14656566.2024.2358987
Conners, C. K., Erhardt, D., and Sparrow, E. P. (1999). Conners’ Adult ADHD Rating Scales (CAARS): Technical Manual. North Tonawanda, NY: Multi-Health Systems Inc.
Dorrego, M. F., Canevaro, L., Kuzis, G., Sabe, L., and Starkstein, S. E. (2002). A randomized, double-blind, crossover study of methylphenidate and lithium in adults with attention-deficit/hyperactivity disorder: preliminary findings. J. Neuropsychiatry Clin. Neurosci. 14, 289–295. doi:10.1176/jnp.14.3.289
Doyle, R. L., Frazier, J., Spencer, T. J., Geller, D., Biederman, J., and Wilens, T. (2006). Donepezil in the treatment of ADHD-like symptoms in youths with pervasive developmental disorder: a case series. J. Atten. Disord. 9, 543–549. doi:10.1177/1087054705284091
Feinmann, C., and Harris, M. (1984). Psychogenic facial pain. Part 2: Management and prognosis. Br. Dent. J. 156, 205–208. doi:10.1038/sj.bdj.4805304
Flor, H., and Turk, D. C. (2015). Chronic Pain: An Integrated Biobehavioral Approach. Philadelphia, PA: Lippincott Williams and Wilkins.
Garcia-Larrea, L., and Peyron, R. (2013). Pain matrices and neuropathic pain matrices: a review. Pain 154 (Suppl. 1), S29–S43. doi:10.1016/j.pain.2013.09.001
Ghanizadeh, A. (2013). Systematic review of clinical trials of aripiprazole for treating attention deficit hyperactivity disorder. Neurosci. (Riyadh) 18, 323–329.
Ginsberg, Y., Quintero, J., Anand, E., Casillas, M., and Upadhyaya, H. P. (2014). Underdiagnosis of attention-deficit/hyperactivity disorder in adult patients: A review of the literature. Prim. Care Companion CNS Disord 16. doi:10.4088/pcc.13r01600
Guay, D. R. P. (2001). Adjunctive agents in the management of chronic pain. Pharmacotherapy 21, 1070–1081. doi:10.1592/phco.21.13.1070.34622
Holman, A. J., and Myers, R. R. (2005). A randomized, double-blind, placebo-controlled trial of pramipexole, a dopamine agonist, in patients with fibromyalgia receiving concomitant medications. Arthritis Rheum. 52, 2495–2505. doi:10.1002/art.21191
Ibrahim, M. E., and Hefny, M. A. (2022). Central sensitization and adult attention deficit hyperactivity disorder in medical students with chronic back pain: a cross-sectional study. Egypt. Rheumatol. Rehabil 49, 24. doi:10.1186/s43166-022-00124-2
Kantak, K. M. (2022). Rodent models of attention-deficit hyperactivity disorder: An updated framework for model validation and therapeutic drug discovery. Pharmacol. Biochem. Behav. 216, 173378. doi:10.1016/j.pbb.2022.173378
Kaplan, M. S., and Kaplan, L. R. (2006). Why do chronic pain patients have multiple accidents? Pain Med 7, 466.1–466. doi:10.1111/j.1526-4637.2006.00208_1.x
Kasahara, S., Kanda, S., Takahashi, M., Fujioka, M., Morita, T., Matsudaira, K., et al. (2023a). Case Report: Guanfacine and methylphenidate improved chronic lower back pain in autosomal dominant polycystic kidney disease with comorbid attention deficit hyperactivity disorder and autism spectrum disorder. Front. Pediatr. 11, 1283823. doi:10.3389/fped.2023.1283823
Kasahara, S., Kato, Y., Takahashi, K., Matsudaira, K., Sato, N., Fukuda, K.-I., et al. (2023b). Improvement in persistent idiopathic facial pain with comorbid ADHD using the combination of a dopamine system stabilizer and psychostimulant: A case report. Clin. Case Rep. 11, e7552. doi:10.1002/ccr3.7552
Kasahara, S., Kato, Y., Takahashi, M., Matsudaira, K., Sato, N., Niwa, S.-I., et al. (2023c). Case report: Remission of chronic low back pain and oral dysesthesia comorbid with attention deficit/hyperactivity disorder by treatment with atomoxetine and pramipexole. Front. Pain Res. (Lausanne) 4, 1159134. doi:10.3389/fpain.2023.1159134
Kasahara, S., Kunii, Y., Mashiko, H., Otani, K., Konno, S.-I., and Niwa, S.-I. (2011). Four cases of chronic pain that improved dramatically following low-dose aripiprazole administration. Prim. Care Companion CNS Disord 13. doi:10.4088/pcc.10l01078
Kasahara, S., Matsudaira, K., Sato, N., and Niwa, S.-I. (2021a). Pain and attention-deficit/hyperactivity disorder: The case of Margaret Mitchell. Psychosom. Med. 83, 492–493. doi:10.1097/psy.0000000000000947
Kasahara, S., Matsudaira, K., Sato, N., and Niwa, S.-I. (2022a). Attention-Deficit/hyperactivity disorder and centralized pain: A review of the case of John F. Kennedy. Clin. Case Rep. 10, e6422. doi:10.1002/ccr3.6422
Kasahara, S., Niwa, S. I., Matsudaira, K., Sato, N., Oka, H., Fujii, T., et al. (2021b). High attention-deficit/hyperactivity disorder scale scores among patients with persistent chronic nonspecific low back pain. Pain Physician 24, E299–E307. doi:10.36076/ppj.2021/24/e299
Kasahara, S., Niwa, S.-I., Matsudaira, K., Sato, N., Oka, H., and Yamada, Y. (2020). Attention-deficit/hyperactivity disorder and chronic pain. Psychosom. Med. 82, 346–347. doi:10.1097/psy.0000000000000789
Kasahara, S., Okamura, Y., Matsudaira, K., Oka, H., Suzuki, Y., Murakami, Y., et al. (2017). Diagnosis and treatment of attention-deficit hyperactivity disorder in patients with chronic pain. Open J. Psychiatr. 07, 261–275. doi:10.4236/ojpsych.2017.74023
Kasahara, S., Takahashi, K., Matsudaira, K., Sato, N., Fukuda, K.-I., Toyofuku, A., et al. (2023d). Diagnosis and treatment of intractable idiopathic orofacial pain with attention-deficit/hyperactivity disorder. Sci. Rep. 13, 1678. doi:10.1038/s41598-023-28931-3
Kasahara, S., Takahashi, M., Morita, T., Matsudaira, K., Sato, N., Momose, T., et al. (2023e). Case report: Atomoxetine improves chronic pain with comorbid post-traumatic stress disorder and attention deficit hyperactivity disorder. Front. Psychiatry 14, 1221694. doi:10.3389/fpsyt.2023.1221694
Kasahara, S., Takahashi, M., Takahashi, K., Morita, T., Matsudaira, K., Sato, N., et al. (2024). Case Report: Methylphenidate and venlafaxine improved abdominal nociplastic pain in an adult patient with attention deficit hyperactivity disorder, autism spectrum disorder, and comorbid major depression. Front. Pain Res. (Lausanne) 5, 1394131. doi:10.3389/fpain.2024.1394131
Kasahara, S., Takao, C., Matsudaira, K., Sato, N., Tu, T. T. H., Niwa, S.-I., et al. (2022b). Case report: Treatment of persistent atypical odontalgia with attention deficit hyperactivity disorder and autism spectrum disorder with risperidone and atomoxetine. Front. Pain Res. (Lausanne) 3, 926946. doi:10.3389/fpain.2022.926946
Kessler, R. C., Adler, L., Ames, M., Demler, O., Faraone, S., Hiripi, E., et al. (2005). The World Health Organization Adult ADHD Self-Report Scale (ASRS): a short screening scale for use in the general population. Psychol. Med. 35, 245–256. doi:10.1017/s0033291704002892
Khawaja, S. N., Alaswaiti, O. F., and Scrivani, S. J. (2023). Burning mouth syndrome. Dent. Clin. North Am. 67, 49–60. doi:10.1016/j.cden.2022.07.004
Kooij, J. J., and Francken, M. H. (2010). Diagnostic Interview for ADHD in adults 2.0 (DIVA 2.0). The Hague, Netherlands: DIVA Foundation.
Kosek, E., Clauw, D., Nijs, J., Baron, R., Gilron, I., Harris, R. E., et al. (2021). Chronic nociplastic pain affecting the musculoskeletal system: clinical criteria and grading system. Pain 162, 2629–2634. doi:10.1097/j.pain.0000000000002324
Kosek, E., Cohen, M., Baron, R., Gebhart, G. F., Mico, J.-A., Rice, A. S. C., et al. (2016). Do we need a third mechanistic descriptor for chronic pain states? Pain 157, 1382–1386. doi:10.1097/j.pain.0000000000000507
Kurlan, R., Crespi, G., Coffey, B., Mueller-Vahl, K., Koval, S., Wunderlich, G., et al. (2012). A multicenter randomized placebo-controlled clinical trial of pramipexole for Tourette’s syndrome. Mov. Disord. 27, 775–778. doi:10.1002/mds.24919
Lamberti, M., Siracusano, R., Italiano, D., Alosi, N., Cucinotta, F., Di Rosa, G., et al. (2016). Head-to-head comparison of aripiprazole and risperidone in the treatment of ADHD symptoms in children with autistic spectrum disorder and ADHD: A pilot, open-label, randomized controlled study. Paediatr. Drugs 18, 319–329. doi:10.1007/s40272-016-0183-3
Meseguer-Beltrán, M., Sánchez-Sarasúa, S., Landry, M., Kerekes, N., and Sánchez-Pérez, A. M. (2023). Targeting neuroinflammation with abscisic acid reduces pain sensitivity in females and hyperactivity in males of an ADHD mice model. Cells 12, 465. doi:10.3390/cells12030465
Mills, S. E. E., Nicolson, K. P., and Smith, B. H. (2019). Chronic pain: a review of its epidemiology and associated factors in population-based studies. Br. J. Anaesth. 123, e273–e283. doi:10.1016/j.bja.2019.03.023
Nijs, J., George, S. Z., Clauw, D. J., Fernández-de-Las-Peñas, C., Kosek, E., Ickmans, K., et al. (2021). Central sensitisation in chronic pain conditions: latest discoveries and their potential for precision medicine. Lancet Rheumatol 3, e383–e392. doi:10.1016/S2665-9913(21)00032-1
Pallanti, S., Porta, F., and Salerno, L. (2021). Adult attention deficit hyperactivity disorder in patients with fibromyalgia syndrome: Assessment and disabilities. J. Psychiatr. Res. 136, 537–542. doi:10.1016/j.jpsychires.2020.10.027
Queiroz, L. P. (2013). Worldwide epidemiology of fibromyalgia. Curr. Pain Headache Rep 17, 356. doi:10.1007/s11916-013-0356-5
Regan, S. L., Williams, M. T., and Vorhees, C. V. (2022). Review of rodent models of attention deficit hyperactivity disorder. Neurosci. Biobehav. Rev. 132, 621–637. doi:10.1016/j.neubiorev.2021.11.041
Sablaban, I. M., and Sivananthan, M. (2022). Attention-deficit hyperactivity disorder–associated impulsive aggression treated with lamotrigine. Am. J. Ther. 29, e747–e748. doi:10.1097/mjt.0000000000001219
Sarria-Santamera, A., Kuntuganova, A., and Alonso, M. (2022). Economic costs of pain in the Spanish working population. J. Occup. Environ. Med. 64, e261–e266. doi:10.1097/jom.0000000000002497
Sifeddine, W., Ba-M’hamed, S., Landry, M., and Bennis, M. (2023). Effect of atomoxetine on ADHD-pain hypersensitization comorbidity in 6-OHDA lesioned mice. Pharmacol. Rep. 75, 342–357. doi:10.1007/s43440-023-00459-3
Stahl, S. M. (2020). Prescriber Guide: Stahl’s Essential Psychopharmacology. Cambridge University Press.
Stahl, S. M. (2021). Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications. Cambridge University Press.
Stickley, A., Koyanagi, A., Takahashi, H., and Kamio, Y. (2016). ADHD symptoms and pain among adults in England. Psychiatry Res 246, 326–331. doi:10.1016/j.psychres.2016.10.004
Stray, L. L., Kristensen, Ø., Lomeland, M., Skorstad, M., Stray, T., and Tønnessen, F. E. (2013). Motor regulation problems and pain in adults diagnosed with ADHD. Behav. Brain Funct. 9, 18. doi:10.1186/1744-9081-9-18
Stray, L. L., Stray, T., Iversen, S., Ruud, A., and Ellertsen, B. (2009). Methylphenidate improves motor functions in children diagnosed with Hyperkinetic Disorder. Behav. Brain Funct. 5, 21. doi:10.1186/1744-9081-5-21
Surman, C. B. H., Hammerness, P. G., Petty, C., Spencer, T., Doyle, R., Napolean, S., et al. (2013). A pilot open label prospective study of memantine monotherapy in adults with ADHD. World J. Biol. Psychiatry 14, 291–298. doi:10.3109/15622975.2011.623716
Suto, T., Kato, D., Koibuchi, I., Arai, Y., Ohta, J., Hiroki, T., et al. (2023). Rat model of attention-deficit hyperactivity disorder exhibits delayed recovery from acute incisional pain due to impaired descending noradrenergic inhibition. Sci. Rep. 13, 5526. doi:10.1038/s41598-023-32512-9
Swanson, D. W., Swenson, W. M., Maruta, T., and Floreen, A. C. (1977). The dissatisfied patient with chronic pain. Pain 4, 367–378. doi:10.1016/0304-3959(77)90147-6
Takahashi, M., Kasahara, S., Soma, T., Morita, T., Sato, N., Matsudaira, K., et al. (2024). Precuneal hyperperfusion in patients with attention-deficit/hyperactivity disorder-comorbid nociplastic pain. Front. Pharmacol. 15, 1480546. doi:10.3389/fphar.2024.1480546
Tambeli, C. H., Levine, J. D., and Gear, R. W. (2009). Centralization of noxious stimulus-induced analgesia (NSIA) is related to activity at inhibitory synapses in the spinal cord. Pain 143, 228–232. doi:10.1016/j.pain.2009.03.005
Treede, R.-D., Rief, W., Barke, A., Aziz, Q., Bennett, M. I., Benoliel, R., et al. (2019). Chronic pain as a symptom or a disease: the IASP classification of Chronic pain for the International Classification of diseases (ICD-11). Pain 160, 19–27. doi:10.1097/j.pain.0000000000001384
Vlaeyen, J. W. S., and Morley, S. (2004). Active despite pain: the putative role of stop-rules and current mood. Pain 110, 512–516. doi:10.1016/j.pain.2004.04.037
Yanofski, J. (2010). The dopamine dilemma: using stimulants and antipsychotics concurrently. Psychiatry (Edgmont) 7, 18–23.
Keywords: nociplastic pain, central sensitization, attention deficit hyperactivity disorder, ADHD medication, dopamine, norepinephrine, pain matrix, ergomania
Citation: Kasahara S, Takahashi M, Suto T, Morita T, Obata H and Niwa S-I (2025) Innovative therapeutic strategies using ADHD medications tailored to the behavioral characteristics of patients with chronic pain. Front. Pharmacol. 16:1500313. doi: 10.3389/fphar.2025.1500313
Received: 23 September 2024; Accepted: 07 February 2025;
Published: 26 February 2025.
Edited by:
Fabio Turco, Cannabiscientia SA, SwitzerlandReviewed by:
Rosmara Infantino, University of Galway, IrelandCopyright © 2025 Kasahara, Takahashi, Suto, Morita, Obata and Niwa. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Satoshi Kasahara, bmFtYWhhZ2VuYXRvckBnbWFpbC5jb20=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.