
95% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
CORRECTION article
Front. Pharmacol. , 05 April 2023
Sec. Renal Pharmacology
Volume 14 - 2023 | https://doi.org/10.3389/fphar.2023.1188615
This article is part of the Research Topic Education and Training in Renal Pharmacology: Basic and applied renal physiology and its contribution to drug development for kidney disease View all 7 articles
This article is a correction to:
Protective effects of mefunidone on ischemia-reperfusion injury/Folic acid-induced acute kidney injury
 Jiajia Li1,2,3†
Jiajia Li1,2,3† Yupeng Jiang1,2,3,4†
Yupeng Jiang1,2,3,4† Qin Dai1
Qin Dai1 Yue Yu1
Yue Yu1 Xin Lv1
Xin Lv1 Yan Zhang1
Yan Zhang1 Xiaohua Liao1
Xiaohua Liao1 Liyun Ao1
Liyun Ao1 Gaoyun Hu5
Gaoyun Hu5 Jie Meng2,6
Jie Meng2,6 Zhangzhe Peng1,2,3
Zhangzhe Peng1,2,3 Lijian Tao1,2,3
Lijian Tao1,2,3 Yanyun Xie1,2,3*
Yanyun Xie1,2,3*A Corrigendum on
Protective effects of mefunidone on ischemia-reperfusion injury/folic acid-induced acute kidney injury
by Li J, Jiang Y, Dai Q, Yu Y, Lv X, Zhang Y, Liao X, Ao L, Hu G, Meng J, Peng Z, Tao L and Xie Y (2022). Front. Pharmacol. 13:1043945. doi: 10.3389/fphar.2022.1043945
In the published article, there was an error in the legend for Figure 1F as published. Figure 1F was displayed as “Vimentin positive area (%).” The correct Figure 1F is “NGAL positive area (%).” The corrected legend appears below.
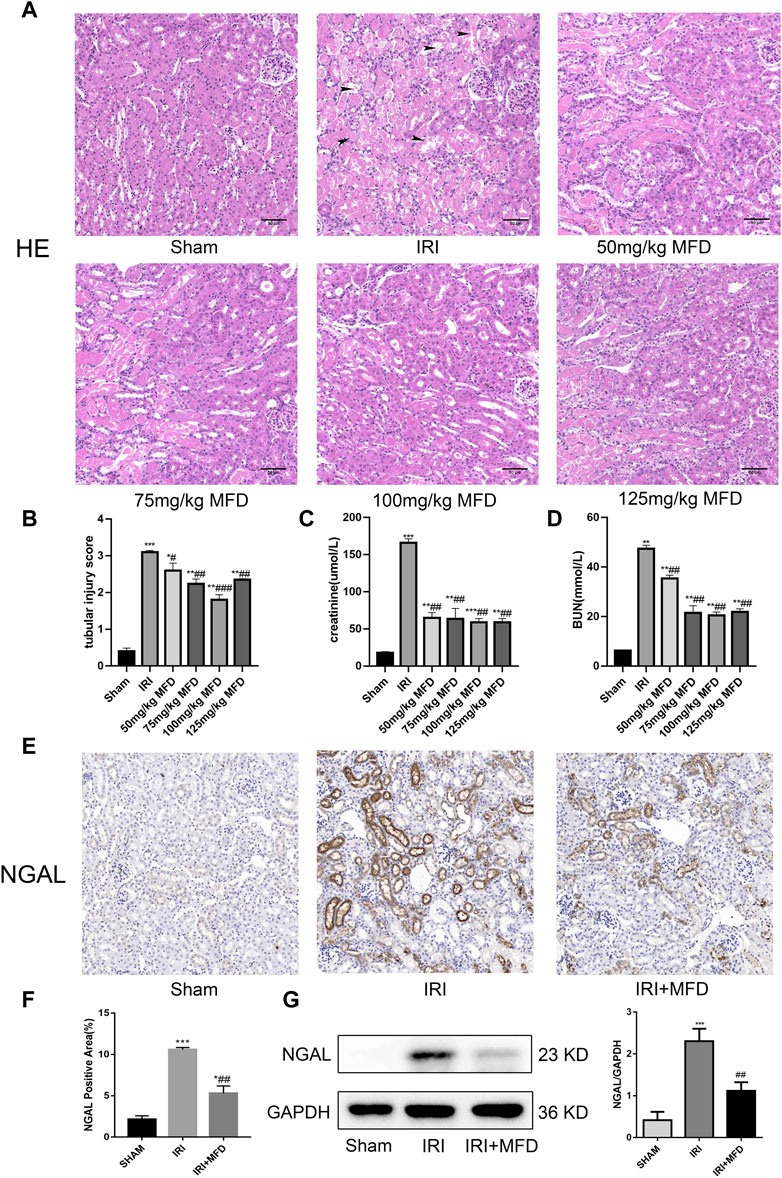
FIGURE 1. Mefunidone ameliorated IRI-induced AKI (A) HE staining showed protective effect of mefunidone at various doses of 50 mg/kg, 75 mg/kg, 100 mg/kg, 125 mg/kg on renal tubular injury on day 2 after IRI modeling (×200). arrows for renal tubular damage. (B) The tubular injury scores of HE staining for kidney damage. (C) Serum creatinine (SCr) levels of mefunidone at various doses of 50 mg/kg, 75 mg/kg, 100 mg/kg, 125 mg/kg on renal tubular injury on day 2 after IRI modeling. (D) Blood urea nitrogen (BUN) levels of mefunidone at various doses of 50 mg/kg, 75 mg/kg, 100 mg/kg, 125 mg/kg on renal tubular injury on day 2 after IRI modeling. (E,F) Histological images of immunohistochemical staining with NGAL and evaluation of NGAL positive area in each group on day 2 after IRI modeling (×200). Mefunidone: 100 mg/kg. (G) Western blot analysis and quantitative data of NGAL in each group on day 2 after IRI modeling. Mefunidone: 100 mg/kg. Data represent mean ± SEM (n = 3–5). *p < 0.05, vs. Sham group; **p < 0.01, vs. Sham group; ***p < 0.001, vs. Sham group; #p < 0.05, vs. IRI group; ##p < 0.01, vs. IRI group; ###p < 0.001, vs. IRI group.
Furthermore, there was an error in the Supplementary Material. Supplementary Figure S1 was displayed as “CCK-8 to determine the optimal drug concentration of mefunidone for 24 h for HK-2 cell viability”; Supplementary Figure S2 was displayed as “Mefunidone alleviated kidney fibrosis and inhibited EMT in IRI-induced CKD.” The correct Supplementary Figure S1 is “Mefunidone alleviated kidney fibrosis and inhibited EMT in IRI-induced CKD”; The correct Supplementary Figure S2 is “CCK-8 to determine the optimal drug concentration of mefunidone for 24 h for HK-2 cell viability.”
The authors apologize for this error and state that this does not change the scientific conclusions of the article in any way. The original article has been updated.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2023.1188615/full#supplementary-material
SUPPLEMENTARY FIGURE S1 | Mefunidone alleviated kidney fibrosis and inhibited EMT in IRI-induced CKD (A) HE staining (×200) and tubular injury scores showed protective effect of Mefunidone on renal tubular injury on day 14 after IRI modeling. arrows for renal tubular damage. (B) Masson's trichrome staining (×200) and kidney fibrotic scores showed protective effect of Mefunidone on renal tubular injury on day 14 after IRI modeling. arrows for renal tubular damage and renal fibrosis. (C) Histological images of immunohistochemical staining with collagen I and evaluation of collagen I positive area in each group on day 14 after IRI modeling (×200). (D) Histological images of immunohistochemical staining with vimentin and evaluation of vimentin positive area in each group on day 14 after IRI modeling (×200). (E) Western blot analysis and quantitative data of α-SMA and E-Cadherin in each group on day 14 after IRI modeling. Mefunidone: 100 mg/kg. Data represent mean ± SEM (n = 5-6). *p < 0.05, vs Sham group; **p < 0.01, vs Sham group; ***p < 0.001, vs Sham group; ****p < 0.0001, vs Sham group; #p < 0.05, vs IRI group; ##p < 0.01, vs IRI group; ###p < 0.001, vs IRI group.
SUPPLEMENTARY FIGURE S2 | CCK-8 to determine the optimal drug concentration of Mefunidone for 24 h for HK-2 cell viability. HK-2 cells were treated with Mefunidone at concentrations of 0, 20, 40, 80, 120 and 160 μg/ml. *p < 0.05, **p < 0.01.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Keywords: renal ischemia-reperfusion injury, folic acid, acute kidney injury, chronic kidney disease, mefunidone
Citation: Li J, Jiang Y, Dai Q, Yu Y, Lv X, Zhang Y, Liao X, Ao L, Hu G, Meng J, Peng Z, Tao L and Xie Y (2023) Corrigendum: Protective effects of Mefunidone on ischemia-reperfusion injury/folic acid-induced acute kidney injury. Front. Pharmacol. 14:1188615. doi: 10.3389/fphar.2023.1188615
Received: 17 March 2023; Accepted: 27 March 2023;
Published: 05 April 2023.
Edited and reviewed by:
Robert John Unwin, University College London, United KingdomCopyright © 2023 Li, Jiang, Dai, Yu, Lv, Zhang, Liao, Ao, Hu, Meng, Peng, Tao and Xie. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yanyun Xie, eGlleWFueXVuQGNzdS5lZHUuY24=
†These authors have contributed equally to this work
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.