- 1Guangxi Scientific Experimental Center of Traditional Chinese Medicine, Guangxi University of Chinese Medicine, Nanning, China
- 2Guangxi Collaborative Innovation Center of Study on Functional Ingredients of Agricultural Residues, Nanning, China
- 3Guangxi Key Laboratory of Efficacy Study on Chinese Materia Medica, Nanning, China
- 4Guangxi International Zhuang Medicine Hospital Affiliated to Guangxi University of Chinese Medicine, Nanning, China
- 5Office of the President, Guangxi University of Chinese Medicine, Nanning, China
- 6Guangxi Dahai Sunshine Pharmaceutical, Nanning, China
- 7Guangxi Heli Pharmaceutical, Nanning, China
Tetrandrine (Tet), derived from the traditional Chinese herb Fangji, is a class of natural alkaloids with the structure of bisbenzylisoquinoline, which has a wide range of physiological activities and significant pharmacfological effects. However, studies and clinical applications have revealed a series of drawbacks such as its poor water solubility, low bioavailability, and the fact that it can be toxic to humans. The results of many researchers have confirmed that chemical structural modifications and nanocarrier delivery can address the limited application of Tet and improve its efficacy. In this paper, we summarize the anti-tumor efficacy and mechanism of action, anti-inflammatory efficacy and mechanism of action, and clinical applications of Tet, and describe the progress of Tet based on chemical structure modification and nanocarrier delivery, aiming to explore more diverse structures to improve the pharmacological activity of Tet and provide ideas to meet clinical needs.
1 Introduction
Natural products play a key role in drug discovery, and many of the widely used contemporary drugs are derived from natural products. For example, paclitaxel, found in the bark of the gymnosperm redbud, and pergolide, found in the periwinkle plant of the oleaceae family, are widely used as clinical chemotherapy drugs for the treatment of pancreatic cancer, non-small cell lung cancer, breast cancer, acute leukemia, and many other cancers (Khan et al., 2022), Penicillin, found in Penicillium, is a commonly used antibiotic in clinical practice. Chinese medicine is the accumulation of thousands of years of practical experience of the Chinese people in the application of natural products for the treatment of diseases, and is a treasure trove of natural product discovery. For example, artemisinin, which has significant efficacy against malaria, and berberine, which has significant efficacy against tumors and diabetes, etc., have been discovered from Chinese medicine. The continuous development of new natural products, especially those from traditional Chinese medicine, is of great significance to modern medicine. However, natural products often suffer from poor solubility and low bioavailability, so the discovery of natural products with therapeutic effects and modification to make them more suitable for clinical use are important aspects of drug development to enable their application.
The root of Stephania Tetrandra S. Moore, known as Fangji in China, is a traditional Chinese medicine, has long been used in the treatment of rheumatism and paralysis, edema and foot pain, urinary discomfort, eczema, and sores, and other conditions. Research on its chemical composition shows that it contains alkaloids, flavonoids, volatile oils, sterols, organic acids and other components. It is rich in alkaloids, mainly containing dozens of alkaloids such as Tetrandrine, Fangchinoline, Stephenanthrine, etc. Among them, Tetrandrine is one of the most concerned active ingredients, and is also considered to be one of the main ingredients for its medicinal effects. Tetrandrine (Tet), also known as powdered alkaloids, is a bisbenzylisoquinoline alkaloid. Tet has a variety of pharmacological activities, including anti-tumor, anti-inflammatory, anti-cellular fibrosis, cardiovascular protection as well as antioxidant, antiviral, and immune enhancement. It protects the cardiovascular system through hypotension, antiarrhythmia, and anti-myocardial ischemia and reperfusion injury (Yu et al., 2001; Pinelli et al., 2010; Xu et al., 2010; Huang et al., 2011; Zhang et al., 2017a; Li et al., 2020), in addition to its antibacterial (Lee et al., 2012; Li et al., 2021) and activation of mesenchymal stem cells to enhance immune regulation (Yang et al., 2016).
Currently, clinical administration of Tet is mainly by oral administration and injection, but there are several problems in its application, such as poor water solubility, low oral bioavailability, short half-life (Que et al., 2019), and poor pharmacokinetic profile (Tian et al., 2016a), the need for high doses leading to poor patient compliance (6–15 tablets per day) (Zhao et al., 2013), gastrointestinal and renal damage (Liang et al., 2010), and toxic effects on liver and lung effects (Tainlin et al., 1982; Yan et al., 2006; Jin et al., 2011; Shi et al., 2016a; Tian et al., 2016b; Chu et al., 2022), among others. In addition, Tet has poor solubility in physiological environments due to the presence of quaternary ammonium salts (Tet saturation = 0.015 mg/ml in pH 7.4 phosphate buffered saline) (Liu et al., 2018). Structural modification of Tet to improve its aqueous solubility and bioavailability, reduce side effects and enhance its toxic effect on cancer cells is an urgent issue to be addressed. In this paper, a comprehensive and systematic discussion of the structural modification of Tet is presented to provide ideas for exploring more diverse structures to improve the pharmacological activity of Tet as well as to meet clinical needs.
2 Pharmacological research and clinical application of Tetrandrine
2.1 Anti-tumor efficacy of Tetrandrine
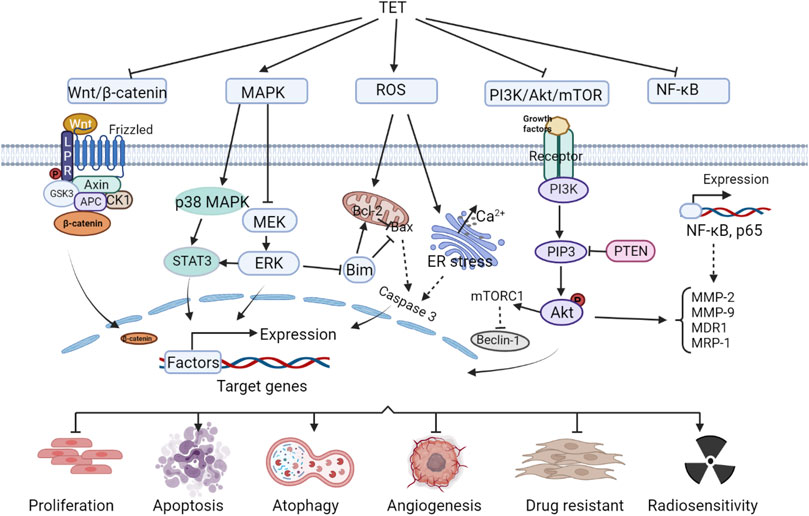
Tet has good antitumor effects against a variety of solid tumors and leukemias, including: nasopharyngeal carcinoma (Wang et al., 2020a),bladder cancer (Li et al., 2011a),triple-negative breast cancer (Yuan et al., 2018),lung cancer (Chen et al., 2018),liver cancer (Chen et al., 2018; Zhang et al., 2018),gastric cancer (Wei et al., 2007)and leukemia (Hu et al., 2020), etc. It inhibits tumor cell migration and invasion, enhances radiosensitivity, induces apoptosis, inhibits tumor cell proliferation and tumor growth, induces autophagy, inhibits vascular regeneration, and reverses tumor multidrug resistance. The anti-tumor efficacy and anti-tumor molecular mechanisms of Tet are comprehensively summarized in Figure 1 and Table 1.
2.2 Anti-tumor mechanism of action of Tetrandrine
2.2.1 Wnt/β-catenin signaling pathway
Wnt is a protein that activates the Wnt/β-catenin signaling pathway, and the name originates from wingless (a Drosophila somatic node polarity gene) and int (a mouse proto-oncogene), a hybrid of these two homologous genes. The human genome encodes at least 19 Wnt proteins, which activate the Wnt signaling pathway, causing the transcription factor complex to disassemble and release the transcription factor β-catenin, which enters the nucleus and activates gene expression. Accumulation of β-catenin in the nucleus is associated with cancer development (Vallée et al., 2021). Tet inhibits Wnt/β-catenin pathway activity and reduces metastatic tumor antigen 1 (MTA1) expression, preventing metastasis in human hepatocellular carcinoma (HCC) (Zhang et al., 2018). Tet can also inactivate Wnt/β-catenin signaling by downregulating IGFBP-5 expression, inhibit LoVo cell proliferation and induce apoptosis, and suppress tumor growth (Wu et al., 2015a). Tet significantly enhances cisplatin-induced growth inhibition and apoptosis in ovarian cancer cells, causing a redistribution of the cell cycle, which is associated with its regulation of the Wnt/cadherin signaling pathway (Zhang et al., 2011). And it was shown that Tet enhanced the sensitivity of ovarian cancer paclitaxel-resistant SKOV3/PTX cells to paclitaxel (PTX) (Jiang and Hou, 2020), induced apoptosis and inhibited the growth of xenograft tumors in colon cancer cells (He et al., 2011a), all associated with Wnt/β-catenin signaling blockade.
2.2.2 Mitogen-activated protein kinase signaling pathway
MAPK, mitogen-activated protein kinase, or MAP kinase, is a class of serine/threonine kinases. MAPK signaling pathway is a tyrosine kinase receptor-mediated signaling pathway that is widely present in cells from yeast to mammals. The classical MAPK (ERK) involves a cascade reaction of four proteins, Ras-Raf-MEK-ERK, and many tumor cells show abnormal cascade protein transduction, which is an important cause of excessive cell proliferation and tumorigenesis (Dillon et al., 2021). On the one hand, Tet inhibits MAPK signaling pathway, which can block the migration and invasion of nasopharyngeal carcinoma cells NPC-TW-039 (Wu et al., 2020). In addition, it significantly enhances the growth inhibition of nasopharyngeal carcinoma cell lines CNE1, CNE2, and C666-1 by radiation, enhances the radiosensitivity of transplanted tumors, and increases the apoptosis rate of transplanted tumor cells (Wang et al., 2020a). On the other hand, Tet activates the MAPK signaling pathway, inhibits cell viability and induces apoptosis in glucocorticoid-resistant human leukemia Jurkat T-cell lines (Xu et al., 2019). Tet induction of apoptosis and inhibition of colon cancer tumor growth have also been shown to be at least partially associated with activation of the p38MAPK signaling pathway.
2.2.3 Reactive oxygen species signaling pathway
Reactive oxygen species (Ros) in mammalian cells are mainly derived from hydrogen peroxide produced during the folding of mitochondrial electron transport chain, nitrogen oxidase and endoplasmic reticulum proteins (Sarmiento-Salinas et al., 2021). Pathological conditions such as cancer, inflammatory diseases and neurodegenerative diseases have been shown to be associated with excessive production of Ros (Liu et al., 2020a). Lin et al. (2016) reported that Tet caused a g0/g1 phase block in human nasopharyngeal carcinoma npc-tw-076 cells, which increased Ros and ca2+ production and eventually led to apoptosis. Tet in human hepatocellular carcinoma induced apoptosis by activating Ros and inhibiting Akt activity to induce apoptosis (Liu et al., 2011), and in human leukemia cells inhibits proliferation and induces autophagy and promotes cell differentiation also due to the accumulation of Ros (Liu et al., 2015; Wu et al., 2018). Tet induces apoptosis in cancer cells by activating the cystatinase pathway, a downstream event of its induction of Ros production, and has therapeutic effects in breast and pancreatic cancers (Wu et al., 2019).
2.2.4 PI3K/Akt/mTOR signaling pathway and NF-κB signaling pathway
PI3K protein consists of a catalytic subunit and a regulatory subunit, which, when bound to tyrosine kinase receptors or cytokine receptors, deregulate the PH structural domain, transactivate Akt proteins, and activate or inhibit the activity of a series of downstream substrates such as apoptosis-associated proteins Bad and Caspase9 by phosphorylation. PI3K/Akt downstream targets are mammalian target of rapamycin (mTOR) proteins. The PI3K/Akt/mTOR signaling pathway is one of the most important intracellular signaling pathways that regulate cell growth, motility, survival, metabolism, and angiogenesis (Yang et al., 2019). PI3K/Akt/mTOR signaling in tumor cells mediates chemoresistance in the tumor microenvironment by shielding immune responses and activating multiple survival signaling pathways in human cancers (Kaboli et al., 2021). Tet downregulates this signaling pathway and inhibits proliferation and invasion of glioma U87 cells (Wu et al., 2014), and also promotes PI3K/Akt/mTOR signaling pathway-mediated cell apoptosis and exert anti-gemcitabine-resistant pancreatic cancer effects (Song et al., 2021a). Bai et al. (2018a) found that Tet induced autophagy and apoptosis in human gastric cancer cells by inducing autophagy and apoptosis involving the Akt/mTOR pathway. Tet inhibited the proliferation of MDA-MB-231 cells and induced autophagy by inhibiting the PI3K/AKT/mTOR pathway (Guo and Pei, 2019).
In addition, Akt in the PI3K/Akt/mTOR signaling pathway can also activate IKK, which has a cross-talk with NF-κB signaling pathway. Core components of NF-κB signaling pathway include nuclear factor κB, NF-κB inhibitory protein, IκB kinase, etc. The signals that activate the NF-κB signaling pathway include the action of signaling molecules such as cytokines (e.g., tumor necrosis factor α, interleukins), growth factors (e.g., EGF, PDGF, and NGF), free radicals (reactive oxygen species), and physical signal stimuli such as radiation, as well as pathogenic infections such as bacteria and viruses. During the immune process of the body, NF-κB coordinates many signals that promote cell activation and proliferation, but also those that may lead to inflammation and autophagy in tumorigenesis (Silke and O’REILLY, 2021). Tet ultimately prevents multidrug resistance in U-2OS osteosarcoma cell lines by inhibiting the NF-κB signaling pathway and thereby suppressing P-gp overexpression (Lu et al., 2017). Tet reduces the cell number of SW620 cells and inhibited cell adhesion and migration due to inhibition of nuclear factor-κB, matrix metalloproteinase-2 and matrix metalloproteinase-9 signaling pathways (Juan et al., 2018a). Studies have shown that Tet inhibition of breast cancer angiogenesis and metastasis, as well as reversal of multidrug resistance in the leukemia-resistant cell line K562/A02, are associated with reduced NF-κB activity (Chen et al., 2008; Gao et al., 2013a).
2.3 Anti-inflammatory efficacy and mechanism of Tetrandrine
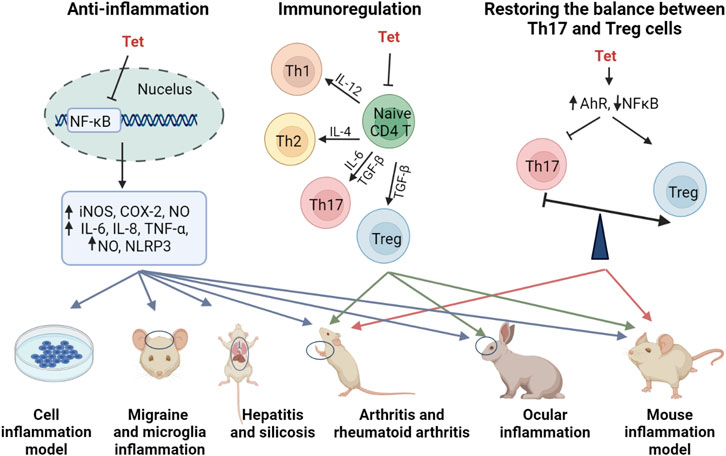
Tet has some anti-inflammatory effects and exerts good anti-inflammatory effects in a variety of inflammatory models in vitro and in vivo (Choi et al., 2000a; Feng et al., 2008; Wu et al., 2011; Wu et al., 2015b; Xu et al., 2016a; Yuan et al., 2016a; Feifei and Ming, 2020). In a glial cell inflammation model, Tet inhibited amyloid-β-induced inflammatory cytokines by inhibiting the NF-κB pathway in mouse BV2 microglia (He et al., 2011b). Qin et al. (2018) established a rat model of migraine and found that Tet pretreatment inhibited S100B and p-ERK activation in satellite glial cells of the trigeminal ganglion and attenuated injury perception in rats. In a model of joint inflammation, Tet reduced foot swelling, bone erosion, immunosuppression, and reduced inflammation in a rat model of arthritis and rheumatoid arthritis (Gao et al., 2016a; Jia et al., 2018; Li et al., 2018). The anti-inflammatory effects and anti-inflammatory molecular mechanisms of Tet are comprehensively summarized in Figure 2 and Table 2.
Although inflammation involves the regulation of multiple signaling pathways, MAPK signaling pathway and NF-κB signaling pathway are considered to be the main inflammatory signaling pathways (COGGINS and ROSENZWEIG, 2012). In contrast, Tet mainly regulates inflammation-related pathways such as NF-κB signaling pathway and NLRP3 inflammatory vesicles. Feng et al. (2008) induced hepatitis in mice by injecting them with concomitant cutaneous globulin A (ConA), and treatment with Tet revealed that Tet inhibited the production of various inflammatory mediators in the liver, and the molecular mechanism of action was inhibition of NF-κB activation. Tet reduced hepatitis in a severe acute pancreatitis rat models of systemic inflammatory response syndrome (SIRS) and multiple organ dysfunction syndrome (MODS), again mediated through the NF-κB pathway, improving the pro/anti-inflammatory imbalance (Wu et al., 2015b). As the only drug approved for the treatment of silicosis in China, Tet was found to alleviate silicosis by inhibiting the typical and atypical NLRP3 inflammatory vesicle pathway in lung macrophages (Song et al., 2021).
2.4 Clinical application of Tetrandrine
Tet has obvious antitumor, anti-inflammatory and anti-fibrosis effects in cell and animal experiments. Meanwhile, it showed the above functions in clinical trial. In a clinical study, Tet has been proven as an effective medicine for silicosis without other side effects (Miao et al., 2012). It was also demonstrated in another clinical trial of acetylcysteine combined with Tet tablets for silicosis. In addition, the incidence of adverse reactions was significantly lower in patients who took the acetylcysteine Tet tablets, and their lung function was significantly higher after treatment, compared with those in the conventional treatment group (Guo et al., 2020a). In December 2019, COVID-19 virus outbreak made the world trying different methods to fight this pandemic. In China, it was found that traditional Chinese medicine had an obvious effect on COVID-19 virus. Tet as traditional Chinese medicines, could improve the prognosis of COVID-19 patients and reduce the incidence of pulmonary fibrosis in patients during recovery.
The treatment of tumor mainly includes surgical excision and drug chemotherapy. However, most drug chemotherapy has high toxicity and multidrug resistance (MDR), which leads to low survival rate and poor prognosis. One of the complex mechanisms involved in the development of multidrug resistance is the MDR gene and p-glycoprotein, which is also one of the main reasons for the failure of Acute Myelogenous Leukemia (AML) (Cao et al., 2018). Tet is an effective inhibitor of MDR-1 efflux pump. It can reverse MDR in cancer cells by increasing the intracellular concentration of chemotherapeutics drugs (Sun and WINK, 2014). In a clinical trial, Tet combined with daunorubicin, etoposide and cytarabine treated 38 patients with AML. Among them, 36 patients had symptom of hypoplastic bone marrow after chemotherapy, 16 patients (42%) was completely remission or slow recovery, 9 patients achieved partial response (PR), and 13 patients treatment failure (Xu et al., 2006).
In addition, Tet has antihepatic fibrosis (Teng et al., 2015; Zhang, 2016) and cardiomyocyte fibrosis (Gan et al., 2018) and has been used to treat silicosis with fibrosis. In addition, Tet (Yu et al., 2001; Meng et al., 2008; Pinelli et al., 2010; Xu et al., 2010; Huang et al., 2011; Zhang et al., 2017a; Li et al., 2020) protects the cardiovascular system through mechanisms such as antihypertensive, antiarrhythmic and anti-myocardial ischemia and reperfusion injury. Also, Tet has antibacterial (Lee et al., 2012; Li et al., 2020) and activates mesenchymal stem cells to enhance immunomodulation (Yang et al., 2016).
3 Chemical-based structural modification of Tet
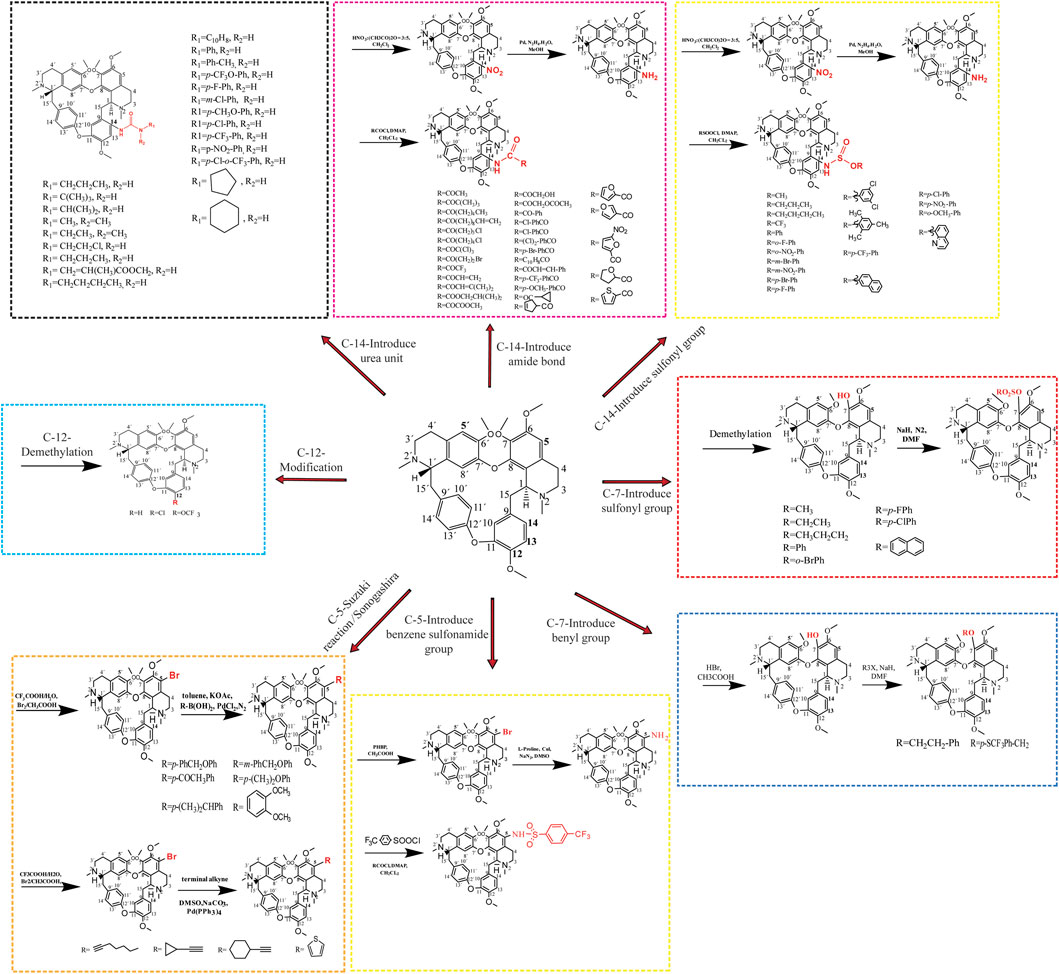
The introduction of key chemical groups to the original structure of a compound can modulate its key pharmacological properties, such as metabolism or efficacy, among others, without the need to develop or redesign the synthetic method as well as restart the synthesis (Moir et al., 2019). Tet (6,6′,7,12-tetramethoxy-2,2′-dimethyl-(1β)-berbaman), whose structure belongs to the structure of Tet (6,6′,7,12-tetramethoxy-2,2′-dimethyl-(1β)-berbaman) is a bis(benzylisoquinoline) alkaloid with molecular formula C38H42N2O6, molecular weight 622.76 g/mol and CAS No. 518-34-3. The structural formula of Tet in the figure below shows that the multiple methoxyl groups on the benzene ring make the benzene ring electron-rich and its electron cloud density is in the order of C-5 position > C-14 position > C-5′ position > C -12 position. Secondly, the tertiary nitrogen atoms on the isoquinoline ring are more basic and can easily form salts in the presence of acid, and can also form quaternary ammonium salts in the form of haloalkanes, so the derivatization reactions of Tet, i.e., structural modifications, mostly occur at the above-mentioned sites (Xiangyu et al., 2022). The chemical structure modification of Tet is summarized in Figure 3.
3.1 Structural modification of the C-5 position
The C-5 position of hanbanzide is mostly reacted with halogen substitution as the first step, and then other key pharmacophore groups are introduced, usually aryl groups, terminal alkynyl groups, heterocycles containing nitrogen and sulfur atoms, etc. Similar to the Sonogashira cross-coupling reaction, the Suzuki reaction is a cross-coupling reaction of aryl halides with aryl boronic acids and is a widely known and versatile method used to construct C-C bonds. Niu et al. (2019) synthesized 12 5-alkynyl-Tet derivatives by the Sonogashira cross-coupling reaction and found that most of the derivatives had better anti-tumor activity than Tet. The mechanism of action was related to the triggering of an intrinsically activated apoptotic pathway, upregulating pro-apoptotic genes (Bax, caspase-3), downregulating anti-apoptotic Bcl-2 and releasing Cyt-C. Wei et al. (2016) synthesized 15 new Tet by the Suzuki reaction using 5-bromohampanoid alkaloids as key intermediates. The in vitro evaluation of anti-HL60 and A549 cellular activity showed that the anticancer activity was enhanced by the introduction of heteroatomic thiophene derivatives and acetylated acetyl phenyl derivatives. (Wu et al., 2013) also synthesized 20 new powdered hamphenine derivatives by combining the Sonogashira and Suzuki reactions using 5-bromohamphenine as the key intermediate. The introduction of an aromatic ring group at the C-5 position significantly increased the proliferative activity of Tet against human breast cancer cell line (MCF-7) and hepatocellular carcinoma cell line (HepG2), compared with sunitinib up to 29.2-fold, with a mechanism of action related to the induction of apoptosis in cancer cells (Li et al., 2017). In addition, the introduction of sulfonamide group at the C-5 position was effective in inducing multiple myeloma cell (RPMI-8226) death, and in comparison to hanpacryl methacin itself, the 5-(4-trifluoromethylbenzenesulfonamido)-hanpacryl methacin derivative had significant anti cell line activity, with nearly 2-fold increased activity against myeloma RPMI-8226 cell line (Wang et al., 2022).
3.2 Structural modification of C-14 position
The nitration reaction of C-14 position of Hanbanzin, catalyzed by Pd/C, is reduced to amino group with hydrazine hydrate (N2H4-H2O), and acylation or sulfonylation with a series of acyl halides and carboxylic acid compounds or sulfonyl chlorides to obtain Tet derivatives of amide or sulfonamide. The introduction of the urea unit on C14 of Tet enhances the antitumor activity of Tet, with derivatives having up to 12-fold the anticancer activity of Tet, 31-fold that of 5-fluorouracil, and 26-fold that of cisplatin, with a mechanism of action related to the induction of cell death by the endogenous apoptotic pathway (Lan et al., 2018). The introduction of an amide bond on C14 of Tet was shown to enhance the cytotoxic effect of Tet on hepatocellular carcinoma cell lines MHCC97L and PLC/PRF/5, the most potent derivative of antitumor activity, 15.8-fold that of Tet and 30.3-fold that of sorafenib, with a mechanism of action endoplasmic reticulum stress-related apoptotic pathway and activation of JNK and caspase pathways associated (Lan et al., 2017), where the introduction of electron-absorbing groups (-F, -CI, -Br) at the C-14 position has higher antitumor activity than electron-donating groups (-OH, -OMe), and amide side chains with aromatic ring substitutions have superior antitumor activity than mono- or multi-substituted benzene ring derivatives. The introduction of sulfonamide groups at the C-14 position significantly enhances the anticancer activity of sulfonamide-antihexine derivatives compared to the parent compounds. Among them, the derivatives in which the benzene ring was substituted with an electron-absorbing group at the 14-amino position showed a stronger increase in antiproliferative activity, and the mechanism of action may be related to the increased expression of the apoptotic protein Bax and a corresponding decrease in the expression of the anti-apoptosis-related proteins (Bcl-xl and Bcl-2) (Song et al., 2018).
3.3 Structural modification of the C-7 position
The introduction of fluorinated benzyl bromide into Tet increased the inhibitory activity against A549 lung cancer cells while reducing the cytotoxicity against the human normal hepatocyte line HL-7702 Gao et al. (2021) while Tet was first demethylated to synthesize the antihexenolines, after a series of reactions, and then different benzyl bromide groups were added to the antihexenoline structure, three of the ten derivatives obtained were comparable to the antihexenoline The other 7 sulfonyl analogs showed stronger antitumor activity than the parent compound, and the most active compound was up to 12.15 times that of Tet.
3.4 Structural modification of the C-12 position
The methoxy group at the para position of C-12 is the key group that induces the hepatotoxicity of Tet. Reducing CYP3A4-mediated toxicity by replacing or eliminating the metabolically unstable C12-methoxy group is one of the important means to improve the clinical application of Tet and explore Tet derivatives with more efficient antitumor activity. Demethylation of the methoxy group at the 12-position of TetC or by a metabolically stable trifluoromethoxy or chloro substituent directly inhibits the proliferation of vincristine-resistant leukemia cells (VCR-RCEM), and its mechanism of action may disrupt mitochondrial membranes potential, affecting early apoptosis-related (Schütz et al., 2020).
3.5 Forming salts
The solubility of a drug will affect its pharmacokinetic properties, chemical stability and the choice of dosage form, which is an important content in the evaluation of druggability. Salt formation can change the solubility of the drug, improve the water solubility of the drug, improve the compliance of the drug, and improve its stability through salt formation. After the tetrandrine is formed into a salt, on the one hand, the influence of the lipid partition coefficient of the derivative on the cytotoxicity is improved; on the other hand, the defect of its own low solubility is changed to meet the selection of different dosage forms. The Tet structure contains two tertiary nitrogen atoms with strong basicity, which are easy to form salts with acids or quaternary salts with halogenated alkanes. Using 1M HCl (inmethanol) and CH2Cl2 as reaction conditions, tetrandrine hydrochloride was synthesized. Compared with tetrandrine itself, it could significantly inhibit the proliferation activity of HEL, K562, MDA-MB-231, PC3, and WM9 cells (Lan et al., 2018). Tetrandrine citrate is synthesized by tetrandrine and citric acid in a ratio of 4:1. The solubility in water reaches 500 mg/ml. It is a new type of oral active tetrandrine salt, which can inhibit ima. The proliferation of tinib (IM)-resistant chronic myeloid leukemia (CML) cells without obvious toxicity in a nude mouse xenograft model may be related to the loss of p210Bcr-Abl and β-catenin proteins (Xu et al., 2012).
4 Structural modification of tetrandrine based on nano-carrier
In recent years, with the vigorous development of new technologies and new materials, the development of nanomaterials has attracted wide attention. Nanomaterials are defined as materials with inherent quantum limitations in at least one dimension, which is also reflected in their electronic structure and other physical properties (Alfieri et al., 2022). Compared with ordinary pharmaceutical preparations, nanomaterials have the following advantages as drug carriers: 1) they can improve their pharmacokinetics by changing the physical and chemical properties of drug molecules (water solubility, fat solubility, etc.). Help drug molecules cross physiological and pathological barriers, thus improving bioavailability (Shi et al., 2017); 2) it can achieve active and passive targeting, increase the local concentration of nano-drugs in the focus, improve the efficacy and reduce the occurrence of side effects, so as to achieve a more safe and effective diagnosis and treatment of diseases (Petros and Desimone, 2010); 3) reduce immune recognition and clearance of reticuloendothelial system by ingenious “camouflage”, protect active molecules from enzymolysis, increase drug retention time, prolong drug half-life, and enhance drug efficacy (Lu et al., 2016); 4) prolong drug circulation time, control drug release, and improve patient compliance; 5) simultaneous visualization of tumor therapeutic effects based on new imaging techniques; 6) to achieve diversity and intelligence. In addition to bioactive chemical molecules, nano-carriers can also contain bioactive substances such as peptides and nucleic acids, as well as contrast agents outside clinical treatment; 7) the drug was delivered to cancer cells by endocytosis, so that the cytotoxicity was not decreased due to the decrease of intracellular pH, and the physiological drug resistance induced by pH was reversed (Li et al., 2011b).
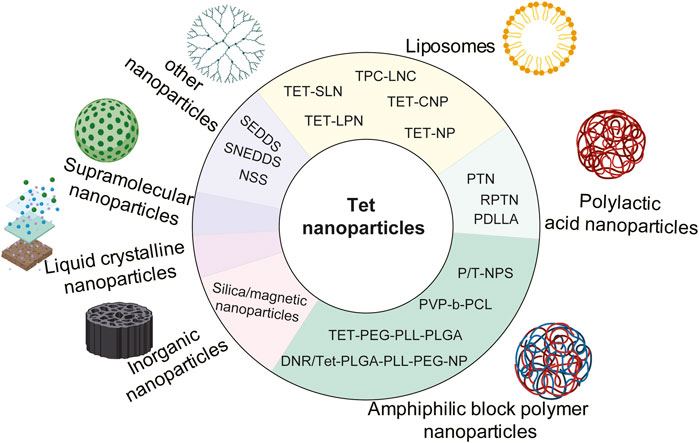
Nanoparticle-drug delivery system is an indispensable new drug delivery system, including organic nanoparticles (for example, polymer nanoparticles, dendrimer nanoparticles, liposomes, micelles, solid lipid nanoparticles, and hydrogels, etc.), inorganic nanoparticles (metal, magnetic, and semiconductor nanoparticles, and carbon nanotubes, etc.). And some nanocrystals designed to improve the rate of dissolution and absorption (Zhang et al., 2020). At present, in order to improve the water solubility, targeting, bioavailability and stability of Tet, many nanomaterials have been developed and utilized, including liposomes, polymer nanoparticles, supramolecular nanoparticles, inorganic nanoparticles, and liquid crystal nanoparticles. The structural modifications of Tet based on nanosystem delivery are summarized in Figure 4.
4.1 Liposomes
Liposomes are nanomaterials formed by hydrophilic cores and hydrophobic lipid bilayers (Van Der Koog et al., 2021). Liposome nanoparticles are multicomponent lipid systems, which usually contain phospholipids, ionizable lipids, cholesterol and polyethylene glycol lipids. The traditional type of liposome nanoparticles refers to liposomes, which was first proposed by British hematologist AlecDBangham in 1961. Importantly, liposomes may overcome the limitations of a single Tet due to the following characteristics: 1) liposomes are amphiphilic and can disperse Tet to avoid aggregation; 2) liposomes are multi-functional nano-carriers that can be targeted and triggered by multiple stimuli because of their surface modification flexibility, size control and adjustable encapsulation. 3) Liposomes avoid drug exposure to normal tissues, organs, and blood circulation, so as to prevent drug degradation, immune reaction and toxicity before reaching the desired lesions; 4) liposomes are composed of natural phospholipids. It is a biocompatible and biodegradable material, minimizing the risk caused by formula. 5) Compared with free Tet, liposome preparation can improve the therapeutic efficiency by prolonging tissue penetration and retention. (Cheng et al., 2021).
Fan et al. (2013) prepared spherical liposomes by pH gradient loading method, which enhanced the local delivery of Tet, thus enhancing the anti-arthritis effect of Tet. Li et al. (2011c) prepared Tet solid lipid nanoparticles (SLNs) by melt emulsification and ultrasound. It was found that TET-SLN had higher concentration in plasma, lower clearance rate, higher release rate and was absorbed by reticuloendothelial system organs. Li et al. (2014b) prepared cationic solid lipid nanoparticles (TET-CNP) and solid lipid nanoparticles (TET-NP) by emulsion evaporation-curing method at low temperature, which were used to load Tet. Compared with Tet ophthalmic solution (TET-SOL), they delayed the release of Tet and significantly improved their bioavailability. Li et al. (2006) used ultrasonic treatment to prepare solid lipid nanoparticles (SLN), which were used to deliver Tet. Stability evaluation found that SLN after ultrasonic treatment was more stable, simple and efficient. Zhao et al. (2013) prepared lipid nanocapsules (TPC-LNCs) loaded with Tet-phospholipid complex by phase inversion method. Compared with traditional tablets, oral bioavailability was greatly improved. Tet was encapsulated with nanoliposomes and g DSPE-MPEG 2000 (DP) was added as a stabilizer, and the results showed that the nanoliposomes could significantly improve the physicochemical properties of Tet, making it safer and more efficient (Song et al., 2022).
As an excellent multi-functional nano-drug carrier, liposomes are widely used to enhance tumor targeting (Perche and Torchilin, 2013). The iRGD peptide modified lipid-polymer hybrid nanosystem (LPN) prepared by Zhang et al. (2017c) is a targeted liposome nano-carrier for the co-delivery of PTX and Tet. This system is more toxic to cells than free drug combinations and non-targeted LPN, significantly induce apoptosis of drug-resistant cells, and has the advantages of high drug loading rate and good stability. Transferrin is a kind of plasma glycoprotein bound to iron. Vincristine plus Tet liposome modified by transferrin is also a kind of targeted liposome, which can significantly prolong the circulation time, make the drug accumulate obviously in the part of brain tumor, and overcome the multidrug resistance of malignant glioma cells (Song et al., 2017).
4.2 Polymer nanoparticles
Nano-polymer refers to the polymer with at least one-dimensional size within 100 nm, including spherical, linear, tubular and other structures. Polymer nanoparticles are an important part of nano-polymers. They have stable structure and can be designed and prepared at the molecular level, such as the selection of polymerization methods or monomers. Because their size and particle uniformity can be controlled, they not only have small size effect, surface effect and quantum tunneling effect, but also have other functions such as temperature, pH, electric field and magnetic field (Xiaobei, 2014). Polymer nanoparticles mainly include poly (DL-lactide-glycolide), polyethylene glycol, poly (lactic acid), poly (vinyl alcohol), Polyaniline, polypyrrole, polyethyleneimine and so on.
4.2.1 Polylactic acid nanoparticles
Poly (lacticacid) (PLA) is a polymer structure formed by dehydration and condensation between individual lactic acid molecules. Its molecular formula can be expressed as (C3H4O2) n, and its molecular weight is determined by different degrees of polymerization (Guoqiang and Kai, 2002). FDA (FoodandDrugAdministration) approved polylactic acid and its derivatives as pharmaceutical excipients in 1995. Polylactic acid is widely used in the field of drug release, surgical suture materials, biomedical instruments and so on (Gang and FENG, 2003; Siripitayananon, 2005; Musumeci et al., 2006; Gao et al., 2014). At present, poly (lactic acid) or poly (lactide) (PLA) is the most widely studied and used biodegradable and renewable thermoplastic based polyester, which has great potential to replace traditional petrochemical-based polymers (Bhuvanesh and Revagade, 2007; Rasal et al., 2010; Saeidlou, 2012).
Meng et al. (2016) found that drug release and cell uptake can be accurately controlled simultaneously by adjusting the surface charge of polylactic acid-glycolic acid (PLGA) nanoparticles (TPNs) multilayer coatings containing Tetrandrine. Shi et al. (2015) prepared polylactic acid-glycolic acid copolymer (PLGA) nanoparticles by emulsion solvent diffusion method. At the same drug concentration, the nanoparticles showed better performance than pure drugs. Among them, the inhibitory effect of DMAB stable particles on A549 cells was the strongest, while that of PVA stable particles was the weakest. (Que et al. (2019) found that compared with the above TET-PLGA nanoparticles (PTN), the erythrocyte membrane camouflaged TET polylactic acid-glycolic acid copolymer (RPTN) nanoparticles (RPTN) significantly reduced the swallowing of RAW264.7 macrophages due to the retention of natural membrane proteins. The combination of TET RPTN and adriamycin (ADR) significantly enhanced the sensitivity of drug-resistant cells MCF-7/ADR to ADR. Racemic polylactic acid (PDLLA) membrane is a new type of drug sustained release and anti-adhesion material in vivo and in vitro, which can significantly inhibit the proliferation and collagen synthesis of fibroblasts, improve the local bioavailability of Tet, and induce apoptosis and death of fibroblasts (Yao et al., 2020).
4.2.2 Amphiphilic block polymer nanoparticles
In drug delivery systems, polymer nanoparticles with amphiphilic diblock or triblock polymers as drug carriers are the mainstream in the field of drug delivery systems. These nanoparticles can escape the clearance of the reticuloendothelial system (RES) and prolong circulation in blood vessels, so they accumulate in the tumor tissue through the EPR effect (Van Vlerken et al., 2007; Zhang et al., 2011c). Compared with free Tet, Tet loaded nanoparticles Tet-NP could more effectively inhibit the proliferation and induce apoptosis of osteosarcoma cells (Tian et al., 2016a). Poly (lactide-co-glycolide) nanoparticles without surfactant were prepared from PLGA by nano-precipitation method, and Tet was encapsulated in it, which can significantly improve the anticancer activity of Tet (Shi et al., 2016b). The nano-drug delivery system based on PEG-PLL-PLGA polymer, carriers daunorubicin (DNR) and Tet, were injected into nude mice carrying MDR leukemia cell K562/A02 xenografts, targeting reversing multidrug resistance MDR (Guo et al., 2015). DNR/Tet-PLGA-PLL-PEG-NP copolymer nano-carriers were constructed by improved double emulsion solvent evaporation/separation technique, and further modified with transferrin (Tf), which improved the solubility of DNR and Tet and increased the targeting of drugs to tumors (Liu et al., 2015d). Xu et al. (2016b) reported that spherical core-shell Tet loaded nanoparticles were prepared by nano-precipitation method using amphoteric poly (N-vinylpyrrolidone)-block-poly (ε-caprolactone) (PVP-b-PCL) copolymers. Tet was incorporated into NP with high encapsulation efficiency and released continuously. Compared with free Tet, the toxic effect of Tet-NP on A549 cells was stronger and positively correlated with dose and time. The ability to induce apoptosis is also stronger.
Li et al. (2010) reported for the first time a Trojan strategy, that is, a simple method to produce nanoparticles containing Tet based on amphiphilic block copolymers, and the results in vitro showed that compared with the same dose of free Tet (1–8 μg/ml), Tet-np at lower concentration (1–8 μg/ml) significantly inhibited the proliferation of cancer cells, and the stability, solubility and anti-tumor activity of Tet were improved. It has been found that the co-delivery of Tet and Ptx in mPEG-PCL nanoparticles has a significant anti-hepatoma effect (Li et al., 2012b), however, it is difficult for drug-loaded nanoparticles to penetrate into the tumor mass and inhibit the growth of cancer cells far from the injection site (Emerich et al., 2002; Ding et al., 2011). The mPEG-PCL nanoparticles (P/T-NPs) co-loaded with Tet and PTX were encapsulated into physically cross-linked gelatin hydrogel and then implanted into the tumor site, the drug could be continuously released. The results showed that P/T-NPs inhibited the growth and invasion of BGC-823 gastric cancer cells more effectively than free drugs or non-Rigner combination (Zhang et al., 2016b).
4.3 Inorganic nanoparticles
Inorganic nanoparticles mainly include metal nanoparticles (Ag, Au, Zn, Co., MoS, Ni, CuS, Gd, TiO2, Fe, etc), magnetic nanoparticles (FeO, Fe3O4, FeCo, FeSe3, etc), semiconductor nanoparticles (Mn, C3N4-BiOCl, TiO2, CuS, etc.) and carbon nanotubes (Amidated carbon nanotube, Carboxylated multi-walled carbon nanotube, Hydroxylated single-walled carbon nanotube, etc) (Zhang et al., 2020). Multifunctional mesoporous silica nanoparticles were prepared by self-assembly in situ loading method, and a co-delivery system of antineoplastic drug PTX and multidrug resistance reversal agent Tet was established. This system inhibited the growth of tumor cells more effectively than only delivering PTX or free PTX, and the drug-loaded nanoparticles completely reversed the resistance of MCF-7/ADR cells to PTX at the molar ratio of 4.4 PTX/TET to PTX. The resistance reversal index was 72.3, and the mechanism was related to cell cycle arrest (Jia et al., 2015).
Wang et al. (2019b) proposed a new MACS ®technique for purification of magnetic nanoparticles for the first time. PLGA-based multifunctional nanoparticles were successfully synthesized and purified by co-encapsulation of Tet and magnetic materials (Fe3O4), which improved the encapsulation efficiency and anti-proliferation effect on A549 lung cancer cells. The mechanism is related to activating the mitochondrial pathway and inducing A549 cell apoptosis by loading lysosomes. Shi et al. (2016c) developed poly (lactic acid-glycolic acid) particles co-loaded with Tet-magnetite, which is a novel polymer magnetic delivery system that releases Tet to inhibit cancer-related TASK3 channels in a dose-dependent manner.
4.4 Liquid crystalline nanoparticles
Liquid crystalline nanoparticles (LCNPs) are formed by amphiphilic lipids in the presence of excess water, which provide higher encapsulation than other drug delivery systems, and they have a higher proportion of lipids in the particles, so they have a larger surface area, so they are proposed as potential drug delivery carriers (Avachat and PARPANI, 2015). Liu et al. (2016) found that Tet-LCNPs, as a new ophthalmic drug delivery system for Tet, compared with Tet ophthalmic solution, has many advantages, such as increasing drug solubility, better sustained release effect, significantly promoting corneal penetration of Tet and improving bioavailability.
4.5 Supramolecular nanoparticles
PTX-SA-RGD is a new type of supramolecular nanomaterial formed by coupling paclitaxel (PTX) with tumor-specific peptide RGD (arginine-glycine-aspartic acid) and succinic acid (SA). Li et al. (2020e) constructed carrier-free nanofibers using PTX-SA-RGD as drug carriers, and obtained nanofiber drug delivery systems co-loaded with PTX and Tet, which improved the poor solubility of PTX and Tet. The toxic effect of their combination on tumor cells was significantly enhanced, and the mechanism was related to the induction of mitochondrial apoptosis.
4.6 Other nano-carrier systems
SNEDDS is an isotropic mixture of oil, surfactant, hydrophilic cosurfactant and drug substances. When introduced into the aqueous medium, fine oil-in-water microemulsion is formed under the gentle agitation of digestive movements of the stomach and intestines. SNEDDS is a relatively new term used to describe preparations whose pellet size is smaller than 100 nm. Self-emulsifying drug delivery system (SEDDS) is a relatively new, lipid-based technological innovation, which has a great prospect in improving the oral bioavailability of drugs (Singh et al., 2009). Liu et al. (2018) improved the dissolution and oral bioavailability of Tet through self-nano-emulsified drug delivery system (SNEDDS), and the oral bioavailability was about 2.33 times higher than that of commercial tablets. In the rapidly developing nano-drug delivery system, nano-suspension (NSS) is a favorable strategy in drug design for cancer treatment, which can overcome the shortcomings of other nano-carriers, such as poor physical stability, low drug loading and low encapsulation ability. Guo et al. (2020b) prepared Tet-NS nano-suspension by wet grinding. The experimental results showed that the cumulative dissolution of Tet-NS increased by 4–5 times within 2 h, which increased the solubility of Tet. High concentration of Tet-NS significantly induced apoptosis and cell uptake of A549 cells.
5 Conclusion
Tet, as a natural product from the Chinese herbal medicine Fangji, is an alkaloid with multiple pharmacological activities, and we focus on the anti-tumor and anti-inflammatory activities of Tet and its current clinical applications and the problems encountered at present. Tet is currently used in clinical practice mainly for the treatment of silicosis, hypertension, and cardiac arrhythmias, but modern research has shown that it also has good anti-tumor, anti-inflammatory, antioxidant, anti-bacterial, anti-viral, and immunity-boosting effects. In up to 24 tumor types, Tet has been shown to have a significant effect through Wnt/β-catenin signaling pathway, MAPK signaling pathway, Reactive oxygen species signaling pathway, PI3K/Akt/mTOR signaling pathway and NF-κB signaling pathway mediating its role in tumors. In addition, Tet also has anti-inflammatory effects, and the mechanism by which it exerts its anti-inflammatory effects is through mediating the NF-κB signaling pathway and CD4 T-cells, upregulating the levels of INOD, COX-2, NO, IL-6, IL-8, TNF-a, NLRP3, Th1, Th2, Th17 and Treg factors, and by acting on AhR and NF-κB signaling pathway to restore the balance between Th17 and Treg cells. Thus Tet is a natural product with great potential for clinical application development.
Despite its multiple pharmacological activities, the disadvantages of Tet, such as its low bioavailability and side effects in humans, have limited its clinical application. Therefore, many researchers have designed and synthesized new Tet derivatives by structurally modifying the C-5, C-14, C-7, and C-12 positions to improve their pharmacokinetic and therapeutic properties. In addition to chemical structural modifications, delivery via nanosystems is also an important way to improve Tet. In this review, liposomal nanoparticles, polymeric nanoparticles, inorganic nanoparticles, liquid crystalline nanoparticles, novel supramolecular material nanoparticles and other nanoparticles are shown to have potential in improving Tet’s clinical application limitations.
In general, we comprehensively summarized the latest research progress on the pharmacological mechanism of Tet in anti-tumor and anti-inflammation, as well as the current clinical application and problems of Tet. The latest progress in the improvement of its physical and chemical properties (water solubility, fat solubility, etc.) by chemical structural modification and structural modification based on nanosystem delivery was also summarized. We hope that this review will help to discover the new pharmacological activities of Tet, expand its clinical application in tumors and inflammatory diseases, and help researchers modify the structure of other active natural products, so that they can be better used in the treatment of clinical diseases.
Author contributions
LM: Writing—original draft, including substantive translation; FZ: Writing—review and editing; FC: Visualization; LX: Literature curation; YH: Literature curation; YM: Literature search; LZ: Literature search; DH: Funding acquisition; SH: Funding acquisition; JD: Project administration; EH: Conceptualization; ZD: Conceptualization.
Funding
This review was supported by Doctoral Research Start-up Fund (Grant No. 2019BS028) and Guangxi Zhuang Autonomous Region Postgraduate Innovation Project (Grant No. YCSW2021238).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Alfieri, A., Anantharaman, S., Zhang, H., and Jariwala, D. (2022). Nanomaterials for quantum information science and engineering. Adv. Mater, e2109621. doi:10.1002/adma.202109621
Avachat, A., and Parpani, S. (2015). Formulation and development of bicontinuous nanostructured liquid crystalline particles of efavirenz. Colloids Surf. B Biointerfaces 126, 87–97. doi:10.1016/j.colsurfb.2014.12.014
Bai, X., Liu, Y., Song, W., Li, Y. Y., Hou, D. S., Luo, H. M., et al. (2018). Anticancer activity of tetrandrine by inducing pro-death apoptosis and autophagy in human gastric cancer cells. J. Pharm. Pharmacol. 70 (8), 1048–1058. doi:10.1111/jphp.12935
Bai, X. Y., Liu, Y. G., and Song, W. (2018). Anticancer activity of tetrandrine by inducing pro-death apoptosis and autophagy in human gastric cancer cells [J].
Bao, G., Li, C., Qi, L., Wang, N., and He, B. (2016). Tetrandrine protects against oxygen-glucose-serum deprivation/reoxygenation-induced injury via PI3K/AKT/NF-κB signaling pathway in rat spinal cord astrocytes. Biomed. Pharmacother. = Biomedecine Pharmacother. 84, 925–930. doi:10.1016/j.biopha.2016.10.007
Bhuvanesh, G., and Revagade, N. (2007). Poly(lactic acid) fiber: An overview [J]. Prog. Polym. Sci. 32 (4), 455–482.
Cao, Z., Li, D., Liu, L., and Yang, P. (2018). Effect of five novel 5-substituted tetrandrine derivatives on P-glycoprotein-mediated inhibition and transport in Caco-2 cells. Oncol. Lett. 16 (5), 6808–6814. doi:10.3892/ol.2018.9492
Chang, K., Liao, H., Chang, H., Chen, Y. Y., Yu, M. C., Chou, C. J., et al. (2004). Inhibitory effect of tetrandrine on pulmonary metastases in CT26 colorectal adenocarcinoma-bearing BALB/c mice. Am. J. Chin. Med. 32 (6), 863–872. doi:10.1142/S0192415X04002478
Chaudhary, P., and Vishwanatha, J. K. (2014). c-Jun NH2-terminal kinase-induced proteasomal degradation of c-FLIPL/S and Bcl2 sensitize prostate cancer cells to Fas- and mitochondria-mediated apoptosis by tetrandrine. Biochem. Pharmacol. 91 (4), 457–473. doi:10.1016/j.bcp.2014.08.014
Chen, B., Su, A., Cheng, J., Zhao, H., Li, G., and Wang, X. (2008). Effects of tetrandrine on nuclear factor-kappaB expression in leukemia multidrug-resistant cell line K562/A02. J. Chin. Integr. Med. 6(9), 956–959. doi:10.3736/jcim20080916
Chen, C. (2011). Toxicology in vitro.Tetrandrine down-regulates ERK/NF-κB signaling and inhibits activation of mesangial cells [J]
Chen, J., Hwang, J., Chiu, W., and Chan, Y. C. (2014). Tetrandrine and caffeine modulated cell cycle and increased glioma cell death via caspase-dependent and caspase-independent apoptosis pathways. Nutr. Cancer 66 (4), 700–706. doi:10.1080/01635581.2014.902974
Chen, Q. Z., Yang, L., Shao, Y., Zeng, Y. H., Ren, W. Y., Liu, R. X., et al. (2017). TGF-β1/PTEN/PI3K signaling plays a critical role in the anti-proliferation effect of tetrandrine in human colon cancer cells. Int. J. Oncol. 50 (3), 1011–1021. doi:10.3892/ijo.2017.3875
Chen, S., Liu, W., Wang, K., Fan, Y., Chen, J., Ma, J., et al. (2017). Tetrandrine inhibits migration and invasion of human renal cell carcinoma by regulating Akt/NF-κB/MMP-9 signaling. PloS one 12 (3), e0173725. doi:10.1371/journal.pone.0173725
Chen, T., Ji, B., and Chen, Y. (2014). Tetrandrine triggers apoptosis and cell cycle arrest in human renal cell carcinoma cells. J. Nat. Med. 68 (1), 46–52. doi:10.1007/s11418-013-0765-0
Chen, Y., Chen, J., and Tseng, S. (2009). Effects of tetrandrine plus radiation on neuroblastoma cells. Anticancer Res. 29 (8), 3163–3171.
Chen, Y., Chen, J., and Tseng, S. (2009). Tetrandrine suppresses tumor growth and angiogenesis of gliomas in rats. Int. J. Cancer 124 (10), 2260–2269. doi:10.1002/ijc.24208
Chen, Z., Zhao, L., Zhao, F., Yang, G., and Wang, J. J. (2018). Tetrandrine suppresses lung cancer growth and induces apoptosis, potentially via the VEGF/HIF-1α/ICAM-1 signaling pathway. Oncol. Lett. 15 (5), 7433–7437. doi:10.3892/ol.2018.8190
Cheng, X., Gao, J., Ding, Y., Lu, Y., Wei, Q., Cui, D., et al. (2021). Multi-functional liposome: A powerful theranostic nano-platform enhancing photodynamic therapy. Adv. Sci. 8 (16), e2100876. doi:10.1002/advs.202100876
Cho, H. S., Chang, S. H., Chung, Y. S., Shin, J. Y., Park, S. J., Lee, E. S., et al. (2009). Synergistic effect of ERK inhibition on tetrandrine-induced apoptosis in A549 human lung carcinoma cells. J. Vet. Sci. 10 (1), 23–28. doi:10.4142/jvs.2009.10.1.23
Choi, H., Kim, H., Min, K., Kim, Y., Lim, H. K., Chang, Y. K., et al. (2000). Anti-inflammatory effects of fangchinoline and tetrandrine. J. Ethnopharmacol. 69 (2), 173–179. doi:10.1016/s0378-8741(99)00141-5
Choi, H. S., Kim, H. S., Min, K. R., Kim, Y., Lim, H. K., Chang, Y. K., et al. (2000). Anti-inflammatory effects of fangchinoline and tetrandrine. J. Ethnopharmacol. 69 (2), 173–179. doi:10.1016/s0378-8741(99)00141-5
Chow, L. W. C., Cheng, K. S., Leong, F., Cheung, C. W., Shiao, L. R., Leung, Y. M., et al. (2019). Enhancing tetrandrine cytotoxicity in human lung carcinoma A549 cells by suppressing mitochondrial ATP production. Naunyn. Schmiedeb. Arch. Pharmacol. 392 (4), 427–436. doi:10.1007/s00210-018-01601-2
Chu, S., Lu, Y., Liu, W., Ma, X., Peng, J., Wang, X., et al. (2022). Ursolic acid alleviates tetrandrine-induced hepatotoxicity by competitively binding to the substrate-binding site of glutathione S-transferases. Phytomedicine. 104, 154325. doi:10.1016/j.phymed.2022.154325
Coggins, M., and Rosenzweig, A. (2012). The fire within: Cardiac inflammatory signaling in health and disease. Circ. Res. 110 (1), 116–125. doi:10.1161/CIRCRESAHA.111.243196
Cui, X., Xiao, D., and Wang, X. (2019). Inhibition of laryngeal cancer stem cells by tetrandrine. Anticancer. Drugs 30 (9), 886–891. doi:10.1097/CAD.0000000000000803
Cui, X., Zhu, W., Wang, P., and Wang, X. (2015). Tetrandrine inhibits the intracellular calcium ion level and upregulates the expression of Brg1 and AHNAK in hep-2 cells. Clin. Lab. 61 (10), 1569–1576. doi:10.7754/clin.lab.2015.141242
Deng, W. Y., Luo, S. X., Zhou, M. Q., Li, N., Chen, X. B., and Han, L. L. (2008). [The study of anti-tumor effect of Tetrandrine combined with Nedaplatin on human liver cancer cell line 7402]. Zhong Yao Cai 31 (10), 1522–1525.
Dillon, M., Lopez, A., Lin, E., Sales, D., Perets, R., and Jain, P. (2021). Progress on Ras/MAPK signaling research and targeting in blood and solid cancers. Cancers 13 (20), 5059. doi:10.3390/cancers13205059
Ding, D., Zhu, Z., Liu, Q., Wang, J., Hu, Y., Jiang, X., et al. (2011). Cisplatin-loaded gelatin-poly(acrylic acid) nanoparticles: Synthesis, antitumor efficiency in vivo and penetration in tumors. Eur. J. Pharm. Biopharm. 79 (1), 142–149. doi:10.1016/j.ejpb.2011.01.008
Duan, B., Yu, Z., Liu, R., Li, J., Song, Z., Zhou, Q., et al. (2022). Tetrandrine-induced downregulation of lncRNA NEAT1 inhibits rheumatoid arthritis progression through the STAT3/miR-17-5p pathway. Immunopharmacol. Immunotoxicol., 1–8. doi:10.1080/08923973.2022.2092748
Emerich, D., Snodgrass, P., Lafreniere, D., Dean, R. L., Salzberg, H., Marsh, J., et al. (2002). Sustained release chemotherapeutic microspheres provide superior efficacy over systemic therapy and local bolus infusions. Pharm. Res. 19 (7), 1052–1060. doi:10.1023/a:1016434926649
Fan, C., Li, X., Zhou, Y., Zhao, Y., Ma, S., Li, W., et al. (2013). Enhanced topical delivery of tetrandrine by ethosomes for treatment of arthritis. Biomed. Res. Int. 2013, 161943. doi:10.1155/2013/161943
Feifei, W., and Ming, G. (2020). Effects of tetrandrine on the inflammatory response of BV2 cells injured by hypoxia and glucose [J]. Chin. J. Clin. Pharmacol. 36 (10), 1350–1352.
Feng, D., Mei, Y., Wang, Y., Zhang, B., Wang, C., and Xu, L. (2008). Tetrandrine protects mice from concanavalin A-induced hepatitis through inhibiting NF-kappaB activation. Immunol. Lett. 121 (2), 127–133. doi:10.1016/j.imlet.2008.10.001
Gan, L., Liang, C., and Dongmei, S. (2018). Effects of tetrandrine on the proliferation and activation of myocardial fibroblasts [J]. J. Peking Univ. Med. Ed. 50 (02), 331–334.
Gang, R., and Feng, S. S. (2003). Preparation and characterization of poly(lactic acid)-poly(ethylene glycol)-poly(lactic acid) (PLA-PEG-PLA) microspheres for controlled release of paclitaxel [J]. Biomaterials 24 (27), 5037–5044.
Gao, A., Liu, F., and Xue, L. (2014). Preparation and evaluation of heparin-immobilized poly (lactic acid) (PLA) membrane for hemodialysis. J. Membr. Sci. 452, 390–399. doi:10.1016/j.memsci.2013.10.016
Gao, J., Ji, X., He, T., Zhang, Q., He, K., Zhao, Y., et al. (2013). Tetrandrine suppresses cancer angiogenesis and metastasis in 4T1 tumor bearing mice. Evid. Based. Complement. Altern. Med. 2013, 265061. doi:10.1155/2013/265061
Gao, J. L., Ji, X., He, T. C., Zhang, Q., He, K., Zhao, Y., et al. (2013). Tetrandrine suppresses cancer angiogenesis and metastasis in 4T1 tumor bearing mice. Evid. Based. Complement. Altern. Med. 2013 (-5-25), 265061. doi:10.1155/2013/265061
Gao, L., Feng, Q., Zhang, X., Wang, Q. S., and Cui, Y. L. (2016). Tetrandrine suppresses articular inflammatory response by inhibiting pro-inflammatory factors via NF-κB inactivation. J. Orthop. Res. 34 (9), 1557–1568. doi:10.1002/jor.23155
Gao, L. N., Feng, Q. S., Zhang, X. F., Wang, Q. S., and Cui, Y. L. (2016). Tetrandrine suppresses articular inflammatory response by inhibiting pro-inflammatory factors via NF-κB inactivation. J. Orthop. Res. 34 (9), 1557–1568. doi:10.1002/jor.23155
Gao, N. B., Chandrashekar, K. R., and Prabhu, A. (2019). “Tetrandrine isolated from Cyclea peltata induces cytotoxicity and apoptosis through ROS and caspase pathways in breast and pancreatic cancer cells [J],” in Vitrocellular & developmental biology - animal.
Gao, X., Lv, X., Zhang, R., Luo, Y., Wang, M. X., Chen, J. S., et al. (2021). Design, synthesis and in vitro anticancer research of novel tetrandrine and fangchinoline derivatives. Bioorg. Chem. 109, 104694. doi:10.1016/j.bioorg.2021.104694
Gong, K., Chen, C., Zhan, Y., Chen, Y., Huang, Z., and Li, W. (2012). Autophagy-related gene 7 (ATG7) and reactive oxygen species/extracellular signal-regulated kinase regulate tetrandrine-induced autophagy in human hepatocellular carcinoma. J. Biol. Chem. 287 (42), 35576–35588. doi:10.1074/jbc.M112.370585
Guangcheng, Q., and Gui, B. (2018). Tetrandrine alleviates nociception in a rat model of migraine via suppressing S100B and p-ERK activation in satellite glial cells of the trigeminal ganglia [J]. J. Mol. Neurosci. MN.
Guo, J., Gu, X., Mai, Y., Zhao, Y., Gou, G., and Yang, J. (2020). Preparation and characterisation of tetrandrine nanosuspensions and in vitro estimate antitumour activity on A549 lung cancer cell line. J. Microencapsul. 37 (5), 384–393. doi:10.1080/02652048.2020.1761905
Guo, L., Zhang, H., Wang, F., Liu, P., Wang, Y., Xia, G., et al. (2015). Targeted multidrug-resistance reversal in tumor based on PEG-PLL-PLGA polymer nano drug delivery system. Int. J. Nanomedicine 10, 4535–4547. doi:10.2147/IJN.S85587
Guo, X., Qi, J., Li, H., and Xing, Z. (2020). Clinical efficacy of acetylcysteine combined with tetrandrine tablets on patients with silicosis and its effect on exercise tolerance and pulmonary function. Exp. Ther. Med. 20 (2), 1285–1290. doi:10.3892/etm.2020.8858
Guo, Y., and Pei, X. (2019). Tetrandrine-induced autophagy in MDA-MB-231 triple-negative breast cancer cell through the inhibition of PI3K/AKT/mTOR signaling. Evid. Based. Complement. Altern. Med. 2019, 7517431. doi:10.1155/2019/7517431
He, B., Gao, J., Zhang, B., Luo, Q., Shi, Q., Kim, S. H., et al. (2011). Tetrandrine inhibits Wnt/β-catenin signaling and suppresses tumor growth of human colorectal cancer. Mol. Pharmacol. 79 (2), 211–219. doi:10.1124/mol.110.068668
He, F. C., Tang, R. Y., and Yao, D. F. (1989). [Effects of tetrandrine on vascular permeability and neutrophil function in acute inflammation]. Zhongguo yao li xue bao = Acta Pharmacol. Sin. 10 (3), 249–251.
He, F., Qiu, B., Li, T., Xie, Q., Cui, D. J., Huang, X. L., et al. (2011). Tetrandrine suppresses amyloid-β-induced inflammatory cytokines by inhibiting NF-κB pathway in murine BV2 microglial cells. Int. Immunopharmacol. 11 (9), 1220–1225. doi:10.1016/j.intimp.2011.03.023
Ho, W. I. N. G. (2013). Tetrandrine inhibits hepatocellular carcinoma cell growth through the caspase pathway and G2/M phase [J]. Oncol. Rep. 29 (6), 2205–2210.
Horng, C., Yang, J., Chiang, J., Lu, C. C., Lee, C. F., Chiang, N. N., et al. (2016). Inhibitory effects of tetrandrine on epidermal growth factor-induced invasion and migration in HT29 human colorectal adenocarcinoma cells. Mol. Med. Rep. 13 (1), 1003–1009. doi:10.3892/mmr.2015.4635
Hu, S., Yang, J., Chen, C., Song, J. R., and Pan, W. D. (2020). Design, synthesis of novel tetrandrine-14-l-amino acid and tetrandrine-14-l-amino acid-urea derivatives as potential anti-cancer agents. Molecules 25 (7), E1738. doi:10.3390/molecules25071738
Huang, A., Lien, J., Lin, M., Yang, J. S., Wu, P. P., Chang, S. J., et al. (2013). Tetrandrine induces cell death in SAS human oral cancer cells through caspase activation-dependent apoptosis and LC3-I and LC3-II activation-dependent autophagy. Int. J. Oncol. 43 (2), 485–494. doi:10.3892/ijo.2013.1952
Huang, P., Xu, Y., Wei, R., Li, H., Tang, Y., Liu, J., et al. (2011). Efficacy of tetrandrine on lowering intraocular pressure in animal model with ocular hypertension. J. Glaucoma 20 (3), 183–188. doi:10.1097/IJG.0b013e3181d7882a
Jang, B., Lim, K., Paik, J., Cho, J. W., Baek, W. K., Suh, M. H., et al. (2004). Tetrandrine-induced apoptosis is mediated by activation of caspases and PKC-delta in U937 cells. Biochem. Pharmacol. 67 (10), 1819–1829. doi:10.1016/j.bcp.2004.01.018
Jia, L., Li, Z., Shen, J., Zheng, D., Tian, X., Guo, H., et al. (2015). Multifunctional mesoporous silica nanoparticles mediated co-delivery of paclitaxel and tetrandrine for overcoming multidrug resistance. Int. J. Pharm. 489, 318–330. doi:10.1016/j.ijpharm.2015.05.010
Jia, Y., Miao, Y., Yue, M., Shu, M., Wei, Z., and Dai, Y. (2018). Tetrandrine attenuates the bone erosion in collagen-induced arthritis rats by inhibiting osteoclastogenesis via spleen tyrosine kinase. FASEB J. official Publ. Fed. Am. Soc. Exp. Biol. 32 (6), 3398–3410. doi:10.1096/fj.201701148RR
Jiang, L., and Hou, R. (2020). Tetrandrine reverses paclitaxel resistance in human ovarian cancer via inducing apoptosis, cell cycle arrest through β-catenin pathway. Onco. Targets. Ther. 13, 3631–3639. doi:10.2147/OTT.S235533
Jiang, Y., Cheng, H., Kuo, C., Way, T., Lien, J., Chueh, F., et al. (2018). Tetrandrine inhibits human brain glioblastoma multiforme GBM 8401 cancer cell migration and invasion in vitro. Environ. Toxicol. 34 (4), 364–374. doi:10.1002/tox.22691
Jin, H., Li, L., Zhong, D., Liu, J., Chen, X., and Zheng, J. (2011). Pulmonary toxicity and metabolic activation of tetrandrine in CD-1 mice. Chem. Res. Toxicol. 24 (12), 2142–2152. doi:10.1021/tx200290s
Jin, Q., Kang, C., Soh, Y., Sohn, N. W., Lee, J., Cho, Y. H., et al. (2002). Tetrandrine cytotoxicity and its dual effect on oxidative stress-induced apoptosis through modulating cellular redox states in Neuro 2a mouse neuroblastoma cells. Life Sci. 71 (17), 2053–2066. doi:10.1016/s0024-3205(02)01989-6
Juan, T., Liu, K., Kuo, C., Yang, M., Chu, Y., Yang, J., et al. (2018). Tetrandrine suppresses adhesion, migration and invasion of human colon cancer SW620 cells via inhibition of nuclear factor-κB, matrix metalloproteinase-2 and matrix metalloproteinase-9 signaling pathways. Oncol. Lett. 15 (5), 7716–7724. doi:10.3892/ol.2018.8286
Kaboli, P., Imani, S., Jomhori, M., and Ling, K. H. (2021). Chemoresistance in breast cancer: PI3K/Akt pathway inhibitors vs the current chemotherapy. Am. J. Cancer Res. 11 (10), 5155–5183.
Khan, A., Farooq, M., Haseeb, M., Valdivielso, J. M., Bozic, M., Lopez-Mejia, I. C., et al. (2022). Glucose starvation or pyruvate dehydrogenase activation induce a broad, ERK5-mediated, metabolic remodeling leading to fatty acid oxidation. Cells 11 (8), 1392. doi:10.3390/cells11091392
Kobayashi, S., Inaba, K., Kimura, I., and KiMuraM., (1998). Inhibitory effects of tetrandrine on angiogenesis in adjuvant-induced chronic inflammation and tube formation of vascular endothelial cells. Biol. Pharm. Bull. 21 (4), 346–349. doi:10.1248/bpb.21.346
Kou, B., Liu, W., Xu, X., Yang, Y., Yi, Q., Guo, F., et al. (2017). Autophagy induction enhances tetrandrine-induced apoptosis via the AMPK/mTOR pathway in human bladder cancer cells. Oncol. Rep. 38 (5), 3137–3143. doi:10.3892/or.2017.5988
Lai, Y., Chen, Y., Wu, T., Wang, S. Y., Chang, K. H., Chung, C. H., et al. (1998). Induction of apoptosis in human leukemic U937 cells by tetrandrine. Anticancer. Drugs 9 (1), 77–81. doi:10.1097/00001813-199801000-00009
Lan, J., Huang, L., Lou, H., Chen, C., Liu, T., Hu, S., et al. (2018). Design and synthesis of novel C14-urea-tetrandrine derivatives with potent anti-cancer activity. Eur. J. Med. Chem. 143, 1968–1980. doi:10.1016/j.ejmech.2017.11.007
Lan, J., Wang, N., Huang, L., Liu, Y., Ma, X., Lou, H., et al. (2017). Design and synthesis of novel tetrandrine derivatives as potential anti-tumor agents against human hepatocellular carcinoma. Eur. J. Med. Chem. 127, 554–566. doi:10.1016/j.ejmech.2017.01.008
Lee, J., Kang, G., Kim, K., Kim, K., Park, D., Choi, B., et al. (2002). Tetrandrine-induced cell cycle arrest and apoptosis in A549 human lung carcinoma cells. Int. J. Oncol. 21 (6), 1239–1244. doi:10.3892/ijo.21.6.1239
Lee, Y., Han, S., Lee, S., Kim, Y. G., Park, C. B., Kang, O. H., et al. (2012). The mechanism of antibacterial activity of tetrandrine against Staphylococcus aureus. Foodborne Pathog. Dis. 9 (8), 686–691. doi:10.1089/fpd.2011.1119
Li, D., Liu, H., Liu, Y., Zhang, Q., Liu, C., Zhao, S., et al. (2017). Design, synthesis and biological activities of tetrandrine and fangchinoline derivatives as antitumer agents. Bioorg. Med. Chem. Lett. 27 (3), 533–536. doi:10.1016/j.bmcl.2016.12.029
Li, G., Li, W., Jin, Y., Jie, Q., Wang, C., and Wu, L. (2020). Tetrandrine attenuated doxorubicin-induced acute cardiac injury in mice. Biomed. Res. Int. 2020, 2616024. doi:10.1155/2020/2616024
Li, J., Guo, X., Liu, Z., Okeke, C. I., Li, N., Zhao, H., et al. (2014). Preparation and evaluation of charged solid lipid nanoparticles of tetrandrine for ocular drug delivery system: Pharmacokinetics, cytotoxicity and cellular uptake studies. Drug Dev. Ind. Pharm. 40 (7), 980–987. doi:10.3109/03639045.2013.795582
Li, J. N., Wang, Q. H., Wang, Z. B., Cui, N., Yang, B., Niu, W., et al. (2019). Tetrandrine inhibits colon carcinoma HT-29 cells growth via the Bcl-2/Caspase 3/PARP pathway and G1/S phase. Biosci. Rep. 39 (5), BSR20182109. doi:10.1042/BSR20182109
Li, P., Zou, J., Dong, Y., Jiang, J., Liang, W., and Li, D. (2021). Tetrandrine, a potent antifungal agent, inhibits mycelial growth and virulence of botrytis cinerea. Phytopathology 111 (7), 1152–1157. doi:10.1094/PHYTO-10-20-0446-R
Li, R., Xie, L., Zhu, Z., Liu, Q., Hu, Y., Jiang, X., et al. (2011). Reversion of pH-induced physiological drug resistance: A novel function of copolymeric nanoparticles. PloS one 6 (9), e24172. doi:10.1371/journal.pone.0024172
Li, S., Ji, Z., Zou, M., Nie, X., Shi, Y., and Cheng, G. (2011). Preparation, characterization, pharmacokinetics and tissue distribution of solid lipid nanoparticles loaded with tetrandrine. AAPS PharmSciTech 12 (3), 1011–1018. doi:10.1208/s12249-011-9665-3
Li, X., Lu, X., Xu, H., Zhu, Z., Yin, H., Qian, X., et al. (2012). Paclitaxel/tetrandrine coloaded nanoparticles effectively promote the apoptosis of gastric cancer cells based on "oxidation therapy. Mol. Pharm. 9 (2), 222–229. doi:10.1021/mp2002736
Li, X., Su, B., Liu, R., Wu, D., and He, D. (2011). Tetrandrine induces apoptosis and triggers caspase cascade in human bladder cancer cells. J. Surg. Res. 166 (1), e45–51. doi:10.1016/j.jss.2010.10.034
Li, X., Wu, Z., He, B., and Zhong, W. (2018). Tetrandrine alleviates symptoms of rheumatoid arthritis in rats by regulating the expression of cyclooxygenase-2 and inflammatory factors. Exp. Ther. Med. 16 (3), 2670–2676. doi:10.3892/etm.2018.6498
Li, X., Xu, H., Dai, X., Zhu, Z., Liu, B., and Lu, X. (2012). Enhanced in vitro and in vivo therapeutic efficacy of codrug-loaded nanoparticles against liver cancer. Int. J. Nanomedicine 7, 5183–5190. doi:10.2147/IJN.S34886
Li, X., Yu, N., Li, J., Bai, J., Ding, D., Tang, Q., et al. (2020). Novel "Carrier-Free" nanofiber codelivery systems with the synergistic antitumor effect of paclitaxel and tetrandrine through the enhancement of mitochondrial apoptosis. ACS Appl. Mat. Interfaces 12 (9), 10096–10106. doi:10.1021/acsami.9b17363
Li, X., Zhen, D., Lu, X., Xu, H., Shao, Y., Xue, Q., et al. (2010). Enhanced cytotoxicity and activation of ROS-dependent c-Jun NH2-terminal kinase and caspase-3 by low doses of tetrandrine-loaded nanoparticles in Lovo cells--a possible Trojan strategy against cancer. Eur. J. Pharm. Biopharm. 75 (3), 334–340. doi:10.1016/j.ejpb.2010.04.016
Li, Y., Dong, L., Jia, A., Chang, X. m., and Xue, H. (2006). [Preparation of tetrandrine solid lipid nanoparticles]. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 28 (5), 686–689.
Li, Y., Li, D., Wang, P., Zhu, W., and Yin, W. (2020). Tetrandrine partially reverses multidrug resistance of human laryngeal cancer cells. J. Int. Med. Res. 48 (8), 300060520944706. doi:10.1177/0300060520944706
Li, Y., Zhao, Q., Tan, B., Fan, L. Q., Liu, Q. W., Jiao, Z. K., et al. (2014). [Effect and mechanisms of TET on human gastric carcinoma cell line SGC7901 and SGC7901/ADR]. Zhongguo Zhong xi yi jie he za zhi Zhongguo Zhongxiyi jiehe zazhi = Chin. J. Integr. traditional West. Med. 34 (1), 66–70.
Liang, Q., Ni, C., Yan, X., Xie, M., Zhang, Y., Zhang, Q., et al. (2010). [Comparative study on metabonomics and on liver and kidney toxicity of Aristolochia fangchi and Stephania tetrandra]. Zhongguo Zhong yao za zhi = Zhongguo zhongyao zazhi = China J. Chin. materia medica 35 (21), 2882–2888.
Liao, C., Ma, Y., Hsia, T., Chou, Y. C., Lien, J. C., Peng, S. F., et al. (2021). Tetrandrine suppresses human brain glioblastoma GBM 8401/luc2 cell-xenografted subcutaneous tumors in nude mice in vivo. Mol. (Basel, Switz. 26 (23), 7105. doi:10.3390/molecules26237105
Lien, J., Lin, M., Chang, S., Lai, K. C., Huang, A. C., Yu, F. S., et al. (2017). Tetrandrine induces programmed cell death in human oral cancer CAL 27 cells through the reactive oxygen species production and caspase-dependent pathways and associated with beclin-1-induced cell autophagy. Environ. Toxicol. 32 (1), 329–343. doi:10.1002/tox.22238
Lin, W., Wang, W., Lin, Y., Leu, J. D., Cheng, S. Y., Chen, Y. J., et al. (2018). Synergistic effects of tetrandrine combined with ionizing radiation on a murine colorectal carcinoma‑bearing mouse model. Oncol. Rep. 40 (3), 1390–1400. doi:10.3892/or.2018.6568
Lin, Y., Peng, S., Lin, M., Kuo, C. L., Lu, K. W., Liao, C. L., et al. (2016). Tetrandrine induces apoptosis of human nasopharyngeal carcinoma NPC-tw 076 cells through reactive oxygen species accompanied by an endoplasmic reticulum stress signaling pathway. Molecules 21 (10), E1353. doi:10.3390/molecules21101353
Liu, C., Gong, K., Mao, X., and Li, W. (2011). Tetrandrine induces apoptosis by activating reactive oxygen species and repressing Akt activity in human hepatocellular carcinoma. Int. J. Cancer 129 (6), 1519–1531. doi:10.1002/ijc.25817
Liu, C., Lv, L., Guo, W., Mo, L., Huang, Y., Li, G., et al. (2018). Self-nanoemulsifying drug delivery system of tetrandrine for improved bioavailability: Physicochemical characterization and pharmacokinetic study. Biomed. Res. Int. 2018, 6763057. doi:10.1155/2018/6763057
Liu, J. F., Feng, W., Huo, J. W., Wei, D. F., and Pan, D. F. (2020). [Tetrandrine induces HL-60 cell differentiation]. Zhongguo Shi Yan Xue Ye Xue Za Zhi 28 (2), 400–404. doi:10.19746/j.cnki.issn.1009-2137.2020.02.007
Liu, J., Li, Y., Chen, S., Lin, Y., Lai, H., Chen, B., et al. (2020). Biomedical application of reactive oxygen species-responsive nanocarriers in cancer, inflammation, and neurodegenerative diseases. Front. Chem. 8, 838. doi:10.3389/fchem.2020.00838
Liu, K., Lin, Y., Hsiao, Y., Lin, M. L., Yang, J. L., Huang, Y. P., et al. (2017). Tetrandrine induces apoptosis in human nasopharyngeal carcinoma NPC-tw 039 cells by endoplasmic reticulum stress and Ca2+/calpain pathways. Anticancer Res. 37 (11), 6107–6118. doi:10.21873/anticanres.12059
Liu, R., Wang, S., Fang, S., Wang, J., Chen, J., Huang, X., et al. (2016). Liquid crystalline nanoparticles as an ophthalmic delivery system for tetrandrine: Development, characterization, and in vitro and in vivo evaluation. Nanoscale Res. Lett. 11 (1), 254. doi:10.1186/s11671-016-1471-0
Liu, R., Wang, Y., Li, X., Bao, W., Xia, G., Chen, W., et al. (2015). Synthesis and characterization of tumor-targeted copolymer nanocarrier modified by transferrin. Drug Des. devel. Ther. 9, 2705–2719. doi:10.2147/DDDT.S80948
Liu, T., Men, Q., Wu, G., Yu, C., Huang, Z., Liu, X., et al. (2015). Tetrandrine induces autophagy and differentiation by activating ROS and Notch1 signaling in leukemia cells. Oncotarget 6 (10), 7992–8006. doi:10.18632/oncotarget.3505
Liu, T., Zhang, Z., Yu, C., Zeng, C., Xu, X., Wu, G., et al. (2017). Tetrandrine antagonizes acute megakaryoblastic leukaemia growth by forcing autophagy-mediated differentiation. Br. J. Pharmacol. 174 (23), 4308–4328. doi:10.1111/bph.14031
Liu, W., Kou, B., Ma, Z., Tang, X. S., Lv, C., Ye, M., et al. (2015). Tetrandrine suppresses proliferation, induces apoptosis, and inhibits migration and invasion in human prostate cancer cells. Asian J. Androl. 17 (5), 850–853. doi:10.4103/1008-682X.142134
Lu, Y., Aimetti, A. A., Langer, R., and Gu, Z. (2016). Bioresponsive materials. Nat. Rev. Mat. 2 (1), 16075. doi:10.1038/natrevmats.2016.75
Lu, Y., Li, F., Xu, T., and Sun, J. (2017). Tetrandrine prevents multidrug resistance in the osteosarcoma cell line, U-2OS, by preventing Pgp overexpression through the inhibition of NF-κB signaling. Int. J. Mol. Med. 39 (4), 993–1000. doi:10.3892/ijmm.2017.2895
Lyu, L., Hu, Y., Yin, S., Wang, L., Ye, F., Wang, M., et al. (2021). Autophagy inhibition enhances anti-pituitary adenoma effect of tetrandrine. Phytother. Res. 35 (7), 4007–4021. doi:10.1002/ptr.7117
Ma, H., Yao, L., Pang, L., Li, X., and Yao, Q. (2016). Tetrandrine ameliorates sevofluraneinduced cognitive impairment via the suppression of inflammation and apoptosis in aged rats. Mol. Med. Rep. 13 (6), 4814–4820. doi:10.3892/mmr.2016.5132
Ma, J., Zhang, Y., Li, R., Ye, J., Li, H., Zhang, Y., et al. (2015). Tetrandrine suppresses human glioma growth by inhibiting cell survival, proliferation and tumour angiogenesis through attenuating STAT3 phosphorylation. Eur. J. Pharmacol. 764, 228–239. doi:10.1016/j.ejphar.2015.06.017
Ma, J., Zhang, Y., Ye, J., Li, R., Wen, Y. L., Huang, J. X., et al. (2017). Tetrandrine exerts a radiosensitization effect on human glioma through inhibiting proliferation by attenuating ERK phosphorylation. Biomol. Ther. 25 (2), 186–193. doi:10.4062/biomolther.2016.044
Meng, L.-H., Zhang, H., Hayward, L., Takemura, H., Shao, R. G., and Pommier, Y. (2004). Tetrandrine induces early G1 arrest in human colon carcinoma cells by down-regulating the activity and inducing the degradation of G1-S-specific cyclin-dependent kinases and by inducing p53 and p21Cip1. Cancer Res. 64 (24), 9086–9092. doi:10.1158/0008-5472.CAN-04-0313
Meng, R., Li, K., Chen, Z., and Shi, C. (2016). Multilayer Coating of Tetrandrine-loaded PLGA nanoparticles: Effect of surface charges on cellular uptake rate and drug release profile. J. Huazhong Univ. Sci. Technol. Med. Sci. = Hua zhong ke ji da xue xue bao Yi xue Ying De wen ban = Huazhong keji daxue xuebao Yixue Yingdewen ban 36 (1), 14–20. doi:10.1007/s11596-016-1535-5
Meng, Z., Xinwei, C., and Shilin, C. (2008). Study on the correlation between the antioxidant capacity of tetrandrine and myocardial protection [J]. World Sci. Technol. - Mod. Traditional Chin. Med. (03), 21–26.
Miao, R., Fang, Z., and Yao, Y. (2012). [Therapeutic efficacy of tetrandrine tablets combined with matrine injection in treatment of silicosis]. Zhonghua lao dong wei sheng zhi ye bing za zhi = Zhonghua laodong weisheng zhiyebing zazhi = Chin. J. industrial Hyg. Occup. Dis. 30 (10), 778–780.
Moir, M., Danon, J., Reekie, T., and Kassiou, M. (2019). An overview of late-stage functionalization in today's drug discovery. Expert Opin. Drug Discov. 14 (11), 1137–1149. doi:10.1080/17460441.2019.1653850
Musumeci, T., Ventura, C. A., Giannone, I., Ruozi, B., Montenegro, L., Pignatello, R., et al. (2006). PLA/PLGA nanoparticles for sustained release of docetaxel. Int. J. Pharm. 325 (1-2), 172–179. doi:10.1016/j.ijpharm.2006.06.023
Niu, B., Wei, S., Sun, J., Zhao, H., Wang, B., and Chen, G. (2022). Deciphering the molecular mechanism of tetrandrine in inhibiting hepatocellular carcinoma and increasing sorafenib sensitivity by combining network pharmacology and experimental evaluation. Pharm. Biol. 60 (1), 75–86. doi:10.1080/13880209.2021.2017468
Niu, N., Qu, T., Xu, J., Lu, X., Bodwell, G. J., and Zhao, Z. (2019). Synthesis of 5-alkynyltetrandrine derivatives and evaluation of their anticancer activity on A549 cell lines. Anticancer. Agents Med. Chem. 19 (12), 1454–1462. doi:10.2174/1871520619666190408132249
Perche, F., and Torchilin, V. (2013). Recent trends in multifunctional liposomal nanocarriers for enhanced tumor targeting. J. Drug Deliv. 2013, 705265. doi:10.1155/2013/705265
Petros, R., and Desimone, J. (2010). Strategies in the design of nanoparticles for therapeutic applications. Nat. Rev. Drug Discov. 9 (8), 615–627. doi:10.1038/nrd2591
Pinelli, A., Trivulzio, S., Brenna, S., Galmozzi, G., and Rossoni, G. (2010). Pretreatment with tetrandrine has protective effects against isoproterenol-induced myocardial infarction in rabbits. Vivo 24 (3), 265–270.
Qian, X., Liu, B., Hu, J., Li, M., Hu, W., Sun, J., et al. (2008). Inhibitory effect of tetrandrine on angiogenesis. Chin. J. Cancer 27 (10), 1050.
Qin, G., Gui, B., Xie, J., Chen, L., Chen, L., Cui, Z., et al. (2018). Tetrandrine alleviates nociception in a rat model of migraine via suppressing S100B and p-ERK activation in satellite glial cells of the trigeminal ganglia. J. Mol. Neurosci. 64 (1), 29–38. doi:10.1007/s12031-017-0999-5
Qin, R., Shen, H., Cao, Y., Fang, Y., Li, H., Chen, Q., et al. (2013). Tetrandrine induces mitochondria-mediated apoptosis in human gastric cancer BGC-823 cells. PloS one 8 (10), e76486. doi:10.1371/journal.pone.0076486
Que, X., Su, J., Guo, P., Kamal, Z., Xu, E., Liu, S., et al. (2019). Study on preparation, characterization and multidrug resistance reversal of red blood cell membrane-camouflaged tetrandrine-loaded PLGA nanoparticles. Drug Deliv. 26 (1), 199–207. doi:10.1080/10717544.2019.1573861
Rasal, R. M., Janorkar, A. V., and Hirt, D. (2010). Poly(lactic acid) modifications. Prog. Polym. Sci. 35 (3), 338–356. doi:10.1016/j.progpolymsci.2009.12.003
Ren, D., Fu, Y., Wang, L., Liu, J., Zhong, X., Yuan, J., et al. (2021). Tetrandrine ameliorated alzheimer's disease through suppressing microglial inflammatory activation and neurotoxicity in the 5XFAD mouse. Phytomedicine 90, 153627. doi:10.1016/j.phymed.2021.153627
Samsuzzaman, M., and Jang, B. (2022). Growth-suppressive and apoptosis-inducing effects of tetrandrine in SW872 human malignant liposarcoma cells via activation of caspase-9, down-regulation of XIAP and STAT-3, and ER stress. Biomolecules 12 (6), 843. doi:10.3390/biom12060843
Sarmiento-Salinas, F., Perez-Gonzalez, A., Acosta-Casique, A., Ix-Ballote, A., Diaz, A., Treviño, S., et al. (2021). Reactive oxygen species: Role in carcinogenesis, cancer cell signaling and tumor progression. Life Sci. 284, 119942. doi:10.1016/j.lfs.2021.119942
Schütz, R., MüLLER, M., Geisslinger, F., Vollmar, A., Bartel, K., and Bracher, F. (2020). Synthesis, biological evaluation and toxicity of novel tetrandrine analogues. Eur. J. Med. Chem. 207, 112810. doi:10.1016/j.ejmech.2020.112810
Shang, W., Zhang, J., Song, H., Zhu, S., Zhang, A., Hua, Y., et al. (2021). Mechanism of tetrandrine against endometrial cancer based on network pharmacology. Drug Des. devel. Ther. 15, 2907–2919. doi:10.2147/DDDT.S307670
Shen, H., Xu, W., Chen, Q., Wu, Z., Tang, H., and Wang, F. (2010). Tetrandrine prevents acquired drug resistance of K562 cells through inhibition of mdr1 gene transcription. J. Cancer Res. Clin. Oncol. 136 (5), 659–665. doi:10.1007/s00432-009-0704-3
Shi, C., Ahmad Khan, S., Wang, K., and Schneider, M. (2015). Improved delivery of the natural anticancer drug tetrandrine. Int. J. Pharm. 479 (1), 41–51. doi:10.1016/j.ijpharm.2014.12.022
Shi, C., Thum, C., Zhang, Q., Tu, W., Pelaz, B., Parak, W. J., et al. (2016). Inhibition of the cancer-associated TASK 3 channels by magnetically induced thermal release of Tetrandrine from a polymeric drug carrier. J. Control. Release 237, 50–60. doi:10.1016/j.jconrel.2016.06.044
Shi, C., Zeng, F., and Fu, D. (2016). Surfactant-free poly(lactide-co-glycolide) nanoparticles for improving in vitro anticancer efficacy of tetrandrine. J. Microencapsul. 33 (3), 249–256. doi:10.3109/02652048.2016.1156175
Shi, J., Kantoff, P., Wooster, R., and Farokhzad, O. C. (2017). Cancer nanomedicine: Progress, challenges and opportunities. Nat. Rev. Cancer 17 (1), 20–37. doi:10.1038/nrc.2016.108
Shi, J., Li, S., Ma, Z., Gao, A. L., Song, Y. J., and Zhang, H. (2016). Acute and sub-chronic toxicity of tetrandrine in intravenously exposed female BALB/c mice. Chin. J. Integr. Med. 22 (12), 925–931. doi:10.1007/s11655-015-2303-2
Shishodia, G., Koul, S., Dong, Q., and Koul, H. K. (2018). Tetrandrine (TET) induces death receptors apo trail R1 (DR4) and apo trail R2 (DR5) and sensitizes prostate cancer cells to TRAIL-induced apoptosis. Mol. Cancer Ther. 17 (6), 1217–1228. doi:10.1158/1535-7163.MCT-17-1157
Silke, J., and O'Reilly, L. (2021). NF-κB and pancreatic cancer; chapter and verse. Cancers 13 (18), 4510. doi:10.3390/cancers13184510
Singh, B., Bandopadhyay, S., Kapil, R., Singh, R., and Katare, O. (2009). Self-emulsifying drug delivery systems (SEDDS): Formulation development, characterization, and applications. Crit. Rev. Ther. Drug Carr. Syst. 26 (5), 427–521. doi:10.1615/critrevtherdrugcarriersyst.v26.i5.10
Singh, K., Dong, Q., Timirishanmugam, P., Koul, S., and Koul, H. K. (2018). Tetrandrine inhibits deregulated cell cycle in pancreatic cancer cells: Differential regulation of p21Cip1/Waf1, p27Kip1 and cyclin D1. Cancer Lett. 425, 164–173. doi:10.1016/j.canlet.2018.03.042
Sinya, E. S. Tetrandrine increases the sensitivity of human lung adenocarcinoma PC14 cells to gefitinib by lysosomal inhibition [J]. Anticancer Res. 39 (12), 6585–6593.
Siripitayananon, B. M. (2005). Synthesis, characterization and melt spinning of a block copolymer of L-lactide and epsilon-caprolactone for potential use as an absorbable monofilament surgical suture [J]. J. Mater. Sci. Mater. Med.
Song, J., Lan, J., Chen, C., Hu, S., Song, J., Liu, W., et al. (2018). Design, synthesis and bioactivity investigation of tetrandrine derivatives as potential anti-cancer agents. MedChemComm 9 (7), 1131–1141. doi:10.1039/c8md00125a
Song, J., Liu, Y., Guo, Y., Zhong, W. X., and Guo, L. (2022). Nano–liposomes double loaded with curcumin and tetrandrine: Preparation, characterization, hepatotoxicity and anti–tumor effects. Int. J. Mol. Sci. 23 (12), 6858. doi:10.3390/ijms23126858
Song, J., Xu, J., Guo, J., Shang, Y., Wang, J., and Wang, T. (2021). The enhancement of Tetrandrine to gemcitabine-resistant PANC-1 cytochemical sensitivity involves the promotion of PI3K/Akt/mTOR-mediated apoptosis and AMPK-regulated autophagy. Acta Histochem. 123 (6), 151769. doi:10.1016/j.acthis.2021.151769
Song, M., Wang, J., Sun, Y., Han, Z., Zhou, Y., Liu, , et al. (2021). Tetrandrine alleviates silicosis by inhibiting canonical and non-canonical NLRP3 inflammasome activation in lung macrophages. Acta Pharmacol. Sin. 43 (5), 1274–1284. doi:10.1038/s41401-021-00693-6
Song, X., Liu, S., Jiang, Y., Gu, L. Y., Xiao, Y., Wang, X., et al. (2017). Targeting vincristine plus tetrandrine liposomes modified with DSPE-PEG2000-transferrin in treatment of brain glioma. Eur. J. Pharm. Sci. 96, 129–140. doi:10.1016/j.ejps.2016.09.024
Sun, X. C., Cheng, H. Y., Deng, Y. X., Shao, R. G., and Ma, J. (2008). Tetrandrine: A potent abrogator of G2 checkpoint function in tumor cells and its mechanism [J]. Biomed. Environ. Sci. 20 (006), 495–501.
Sun, Y., and Wink, M. (2014). Tetrandrine and fangchinoline, bisbenzylisoquinoline alkaloids from Stephania tetrandra can reverse multidrug resistance by inhibiting P-glycoprotein activity in multidrug resistant human cancer cells. Phytomedicine 21, 1110–1119. doi:10.1016/j.phymed.2014.04.029
Tainlin, L., Tingyi, H., Changqi, Z., Peipei, Y., and Qiong, Z. (1982). Studies of the chronic toxicity of tetrandrine in dogs: An inhibitor of silicosis. Ecotoxicol. Environ. Saf. 6 (6), 528–534. doi:10.1016/0147-6513(82)90034-3
Tao, L., Zhou, X., Shen, C., Liang, C. Z., Liu, B., Tao, Y., et al. (2014). Tetrandrine induces apoptosis and triggers a caspase cascade in U2-OS and MG-63 cells through the intrinsic and extrinsic pathways. Mol. Med. Rep. 9 (1), 345–349. doi:10.3892/mmr.2013.1761
Teng, G., Svystonyuk, D., Mewhort, H., Turnbull, J. D., Belke, D. D., Duff, H. J., et al. (2015). Tetrandrine reverses human cardiac myofibroblast activation and myocardial fibrosis. Am. J. Physiol. Heart Circ. Physiol. 308 (12), H1564–H1574. doi:10.1152/ajpheart.00126.2015
Tha, B., Sx, A., and Rd, A. (2019). Targeting the Ca 2+/Calmodulin-dependent protein kinase II by Tetrandrine in human liver cancer cells [J]. Biochem. Biophysical Res. Commun. 508 (4), 1227–1232.
Tian, D., Zhang, R., Wu, N., Yuan, W., Luo, S. H., Chen, H. Q., et al. (2017). Tetrandrine inhibits the proliferation of human osteosarcoma cells by upregulating the PTEN pathway. Oncol. Rep. 37 (5), 2795–2802. doi:10.3892/or.2017.5560
Tian, Y., Shen, S., Jiang, Y., Shen, Q., Zeng, S., and Zheng, J. (2016). CYP3A5 mediates bioactivation and cytotoxicity of tetrandrine. Arch. Toxicol. 90 (7), 1737–1748. doi:10.1007/s00204-015-1584-8
Tian, Y., Yin, H., and Xu, H. (2016). Enhanced pro-apoptotic effect of tetrandrine loaded nanoparticles against osteosarcoma cells. Curr. Drug Deliv. 13 (6), 946–952. doi:10.2174/1567201813666160303104439
Tsai, S., Wu, W., and Yang, J. (2021). Tetrandrine inhibits epithelial-mesenchymal transition in IL-6-induced HCT116 human colorectal cancer cells. Onco. Targets. Ther. 14, 4523–4536. doi:10.2147/OTT.S324552
Vallée, A., Lecarpentier, Y., and VALLéE, J. (2021). The key role of the WNT/β-Catenin pathway in metabolic reprogramming in cancers under normoxic conditions. Cancers 13 (21), 5557. doi:10.3390/cancers13215557
Van Der Koog, L., Gandek, T., and Nagelkerke, A. (2021). Liposomes and extracellular vesicles as drug delivery systems: A comparison of composition, pharmacokinetics, and functionalization. Adv. Healthc. Mat. 11, e2100639. doi:10.1002/adhm.202100639
Van Vlerken, L., Vyas, T., and Amiji, M. (2007). Poly(ethylene glycol)-modified nanocarriers for tumor-targeted and intracellular delivery. Pharm. Res. 24 (8), 1405–1414. doi:10.1007/s11095-007-9284-6
Wan, J., Liu, T., Mei, L., Li, J., Gong, K., Yu, C., et al. (2013). Synergistic antitumour activity of sorafenib in combination with tetrandrine is mediated by reactive oxygen species (ROS)/Akt signaling. Br. J. Cancer 109 (2), 342–350. doi:10.1038/bjc.2013.334
Wang, C., Yang, J., Guo, Y., Shen, J., and Pei, X. (2020). Anticancer activity of tetrandrine by inducing apoptosis in human breast cancer cell line MDA-MB-231 in vivo. Evid. Based. Complement Alternat. Med., 6823520. doi:10.1155/2020/6823520
Wang, J., Chang, L., Lai, X., Li, X., Wang, Z., Huang, Z., et al. (2018). Tetrandrine enhances radiosensitivity through the CDC25C/CDK1/cyclin B1 pathway in nasopharyngeal carcinoma cells. Cell. cycleGeorget. Tex) 17 (6), 671–680. doi:10.1080/15384101.2017.1415679
Wang, J., Yao, Z., Lai, X., Bao, H., Li, Y., Li, S., et al. (2020). Tetrandrine sensitizes nasopharyngeal carcinoma cells to irradiation by inducing autophagy and inhibiting MEK/ERK pathway. Cancer Med. 9 (19), 7268–7278. doi:10.1002/cam4.3356
Wang, K., Hu, H., Zhang, Q., Zhang, Y., and Shi, C. (2019). Synthesis, purification, and anticancer effect of magnetic Fe3O4-loaded poly (lactic-co-glycolic) nanoparticles of the natural drug tetrandrine. J. Microencapsul. 36 (4), 356–370. doi:10.1080/02652048.2019.1631403
Wang, N., Yang, S., Tan, T., Huang, Y., Chen, Y., Dong, C., et al. (2021). Tetrandrine suppresses the growth of human osteosarcoma cells by regulating multiple signaling pathways. Bioengineered 12 (1), 5870–5882. doi:10.1080/21655979.2021.1967034
Wang, T., Wan, J., Gong, X., Li, H. Z., and Cheng, Y. (2012). Tetrandrine enhances cytotoxicity of cisplatin in human drug-resistant esophageal squamous carcinoma cells by inhibition of multidrug resistance-associated protein 1. Oncol. Rep. 28 (5), 1681–1686. doi:10.3892/or.2012.1999
Wang, X., Chen, Y., Li, J., Guo, S., Lin, X., Zhang, H., et al. (2019). Tetrandrine, a novel inhibitor of ether-à-go-go-1 (Eag1), targeted to cervical cancer development. J. Cell. Physiol. 234 (5), 7161–7173. doi:10.1002/jcp.27470
Wang, X., Muzaffar, J., Kirtane, K., Song, F., Johnson, M., Schell, M. J., et al. (2022). T cell repertoire in peripheral blood as a potential biomarker for predicting response to concurrent cetuximab and nivolumab in head and neck squamous cell carcinoma. J. Immunother. Cancer 10 (6), e004512. doi:10.1136/jitc-2022-004512
Wang, Y., Yue, W., Lang, H., Ding, X., Chen, X., and Chen, H. (2021). Resuming sensitivity of tamoxifen-resistant breast cancer cells to tamoxifen by tetrandrine. Integr. Cancer. Ther. 20, 1534735421996822. doi:10.1177/1534735421996822
Wei, J., Liu, B., Wang, L., Qian, X., Ding, Y., and Yu, L. (2007). Synergistic interaction between tetrandrine and chemotherapeutic agents and influence of tetrandrine on chemotherapeutic agent-associated genes in human gastric cancer cell lines. Cancer Chemother. Pharmacol. 60 (5), 703–711. doi:10.1007/s00280-007-0416-9
Wei, X., Qu, T., Yang, Y., Xu, J. F., Li, X. W., Zhao, Z. B., et al. (2016). Design and synthesis of new tetrandrine derivatives and their antitumor activities. J. Asian Nat. Prod. Res. 18 (10), 966–975. doi:10.1080/10286020.2016.1188085
Wong, C., Seow, W., O'Callaghan, J., and Thong, Y. H. (1992). Comparative effects of tetrandrine and berbamine on subcutaneous air pouch inflammation induced by interleukin-1, tumour necrosis factor and platelet-activating factor [J]. Agents & Actions 36 (1-2), 112–118.
Wu, C., Lai, L., Hu, X., Lei, R. R., and Yang, Y. F. (2013). Synthesis and antitumor activity of tetrandrine derivatives. J. Asian Nat. Prod. Res. 15 (9), 993–1002. doi:10.1080/10286020.2013.823950
Wu, C., Wang, Y., Lin, C., Chen, H. H., and Chen, Y. J. (2011). Tetrandrine down-regulates ERK/NF-κB signaling and inhibits activation of mesangial cells. Toxicol. Vitro 25 (8), 1834–1840. doi:10.1016/j.tiv.2011.09.024
Wu, G., Liu, T., Li, H., Li, Y., Li, D., and Li, W. (2018). c-MYC and reactive oxygen species play roles in tetrandrine-induced leukemia differentiation. Cell. Death Dis. 9 (5), 473. doi:10.1038/s41419-018-0498-9
Wu, J. M., Chen, Y., Chen, J. C., Lin, T. Y., and Tseng, S. H. (2010). Tetrandrine induces apoptosis and growth suppression of colon cancer cells in mice. Cancer Lett. 287 (2), 187–195. doi:10.1016/j.canlet.2009.06.009
Wu, K., Zhou, M., Wu, Q., Yuan, S., Wang, D., Jin, J., et al. (2015). The role of IGFBP-5 in mediating the anti-proliferation effect of tetrandrine in human colon cancer cells. Int. J. Oncol. 46 (3), 1205–1213. doi:10.3892/ijo.2014.2800
Wu, N. B., Chandrashekar, K., Prabhu, A., and Rekha, P. (2019). Tetrandrine isolated from Cyclea peltata induces cytotoxicity and apoptosis through ROS and caspase pathways in breast and pancreatic cancer cells [J]. Dev. Biol. Animal 55 (5), 331–340. In vitro cellular.
Wu, S., Chueh, F., Chou, Y., Ma, Y. S., Peng, S. F., Lin, C. C., et al. (2020). Tetrandrine inhibits cell migration and invasion in human nasopharyngeal carcinoma NPC-TW 039 cells through inhibiting MAPK and RhoA signaling pathways. J. Food Biochem. 44, e13387. doi:10.1111/jfbc.13387
Wu, S. J., and Ng, L. T. (2007). Tetrandrine inhibits proinflammatory cytokines, iNOS and COX-2 expression in human monocytic cells. Biol. Pharm. Bull. 30 (1), 59–62. doi:10.1248/bpb.30.59
Wu, X. L., Li, J. X., Li, Z. D., Liu, D. S., Lu, S. H., Liu, K. L., et al. (2015). Protective effect of tetrandrine on sodium taurocholate-induced severe acute pancreatitis. Evid. Based. Complement. Altern. Med. 2015 (-10-8), 129103–129108. doi:10.1155/2015/129103
Wu, X., Li, J., Li, Z., Liu, D., Lu, S., Liu, K., et al. (2015). Protective Effect of Tetrandrine on Sodium Taurocholate-Induced Severe Acute Pancreatitis. Evid Based Complement. Alternat. Med. 2015, 129103. doi:10.1155/2015/129103
Wu, Z., Wang, G., Xu, S., Li, Y., Tian, Y., Niu, H., et al. (2014). Effects of tetrandrine on glioma cell malignant phenotype via inhibition of ADAM17. Tumour Biol. 35 (3), 2205–2210. doi:10.1007/s13277-013-1293-y
Wxb, C. . Tetrandrine and cepharanthine induce apoptosis through caspase cascade regulation, cell cycle arrest, MAPK activation and PI3K/Akt/mTOR signal modification in glucocorticoid resistant human leukemia Jurkat T cells [J]. Chemico-biological Interact. 310, 108726.
Xiangyu, Z., Shijie, C., and Xiaoying, W. (2022). Research progress of tetrandrine and its derivatives [J]. Chin. J. Med. Chem. 32 (01), 43–52.
Xiao, J., and Chiou, G. (1996). Tetrandrine inhibits breakdown of blood-aqueous barrier induced by endotoxin and interleukin-1 alpha in rats. J. Ocul. Pharmacol. Ther. 12 (3), 323–329. doi:10.1089/jop.1996.12.323
Xiao, J., Wang, Y., Wu, S., Li, J., and Zhang, S. (1992). Inhibitory effect of tetrandrine on lens proteins-induced ocular inflammation in rabbits. J. Ocul. Pharmacol. 8 (4), 309–315. doi:10.1089/jop.1992.8.309
Xiao, J., Wu, S., Wang, Y., Zhang, M., and Li, J. (1994). Therapeutic actions and mechanism of tetrandrine on experimental uveitis [J]. CHINESE JOURNAL OF OCULAR FUNDUS DISEASES 3, 149–152.
Xiaobei, W. (2014). Construction and properties of multifunctional polymer nanoparticles [D]. Available at: https://kns.cnki.net/KCMS/detail/detail.aspx?dbname=CDFDLAST2015&filename=1015531344.nh
Xu, H., Li, X., Kong, H., Zeng, X., Jin, Y., Qi, Y., et al. (2016). Characterization of the uptake efficiency and cytotoxicity of tetrandrine-loaded poly(N-vinylpyrrolidone)-Block-Poly(ε-caprolactone) (PVP-b-PCL) nanoparticles in the A549 lung adenocarcinoma cell line. J. Biomed. Nanotechnol. 12 (8), 1699–1707. doi:10.1166/jbn.2016.2293
Xu, J., Liu, D., Yin, Q., and Guo, L. (2016). Tetrandrine suppresses β‑glucan‑induced macrophage activation via inhibiting NF‑κB, ERK and STAT3 signaling pathways. Mol. Med. Rep. 13 (6), 5177–5184. doi:10.3892/mmr.2016.5187
Xu, M., Sheng, L., Zhu, X., Zeng, S., Chi, D., and Zhang, G. J. (2010). Protective effect of tetrandrine on doxorubicin-induced cardiotoxicity in rats. Tumori 96 (3), 460–464. doi:10.1177/030089161009600314
Xu, W., Debeb, B., Lacerda, L., Li, J., Woodward, W., et al. (1925). Tetrandrine, a compound common in Chinese traditional medicine, preferentially kills breast cancer tumor initiating cells (TICs) in vitro [J]. Cancers 3 (2).
Xu, W., Shen, H., Ao, Z., Chen, B. A., Xia, W., Gao, F., et al. (2006). Combination of tetrandrine as a potential-reversing agent with daunorubicin, etoposide and cytarabine for the treatment of refractory and relapsed acute myelogenous leukemia. Leuk. Res. 30 (4), 407–413. doi:10.1016/j.leukres.2005.08.005
Xu, W., Wang, X., Chen, S., Wu, H., Tanaka, S., Onda, K., et al. (2020). Tetrandrine enhances glucocorticoid receptor translocation possibly via inhibition of P-glycoprotein in daunorubicin-resistant human T lymphoblastoid leukemia cells. Eur. J. Pharmacol. 881, 173232. doi:10.1016/j.ejphar.2020.173232
Xu, W., Wang, X., Tu, Y., Masaki, H., Tanaka, S., Onda, K., et al. (2019). Tetrandrine and cepharanthine induce apoptosis through caspase cascade regulation, cell cycle arrest, MAPK activation and PI3K/Akt/mTOR signal modification in glucocorticoid resistant human leukemia Jurkat T cells. Chem. Biol. Interact. 310, 108726. doi:10.1016/j.cbi.2019.108726
Xu, X., Gan, Y., Xu, G., Chen, T., Zhou, H., Tang, J. f., et al. (2012). Tetrandrine citrate eliminates imatinib-resistant chronic myeloid leukemia cells in vitro and in vivo by inhibiting Bcr-Abl/β-catenin axis. J. Zhejiang Univ. Sci. B 13 (11), 867–874. doi:10.1631/jzus.B1200021
Yan, C., Xin-Ming, Q., Li-Kun, G., Lin-Lin, L., Fang-Ping, C., Ying, X., et al. (2006). Tetrandrine-induced apoptosis in rat primary hepatocytes is initiated from mitochondria: Caspases and endonuclease G (endo G) pathway. Toxicology 218 (1), 1–12. doi:10.1016/j.tox.2005.08.024
Yang, J., Nie, J., Ma, X., Wei, Y., Peng, Y., and Wei, X. (2019). Targeting PI3K in cancer: Mechanisms and advances in clinical trials. Mol. Cancer 18 (1), 26. doi:10.1186/s12943-019-0954-x
Yang, Y., and Zheng, J. (2016). Tetrandrine suppresses metastatic phenotype of prostate cancer cells by regulating Akt/mTOR/MEMP-9 signaling pathway [J]. Oncol. Rep. 35 (5), 2880–2886.
Yang, Z., Concannon, J., Ng, K. S., Seyb, K., Mortensen, L. J., Ranganath, S., et al. (2016). Tetrandrine identified in a small molecule screen to activate mesenchymal stem cells for enhanced immunomodulation. Sci. Rep. 6, 30263. doi:10.1038/srep30263
Yao, H., Cao, Z., Peng, L., Liu, J., Zhang, X., and Deng, Z. (2020). A novel controlled release tetrandrine-loaded PDLLA film: Evaluation of drug release and anti-adhesion effects in vitro and in vivo. Drug Deliv. Transl. Res. 10 (1), 13–22. doi:10.1007/s13346-019-00654-x
Yu, B., Yuan, B., Li, J., Kiyomi, A., Kikuchi, H., Hayashi, H., et al. (2020). JNK and autophagy independently contributed to cytotoxicity of arsenite combined with tetrandrine via modulating cell cycle progression in human breast cancer cells [J]. Front. Pharmacol., 11.
Yu, F., Yu, C., Chen, J., Yang, J. L., Lu, H. F., Chang, S. J., et al. (2016). Tetrandrine induces apoptosis via caspase-8, -9, and -3 and poly (ADP ribose) polymerase dependent pathways and autophagy through beclin-1/LC3-I, II signaling pathways in human oral cancer HSC-3 cells. Environ. Toxicol. 31 (4), 395–406. doi:10.1002/tox.22053
Yuan, B., Yao, M., Wang, X., Sato, A., Okazaki, A., Komuro, H., et al. (2018). Antitumor activity of arsenite in combination with tetrandrine against human breast cancer cell line MDA-MB-231 in vitro and in vivo [J]. Cancer Cell. Int. 18 (1), 113.
Yu, X., Wu, S., Wang, G., Shan, J., Wong, T. M., Chen, C. F., et al. (2001). Cardiac effects of the extract and active components of radix stephaniae tetrandrae. II. Myocardial infarct, arrhythmias, coronary arterial flow and heart rate in the isolated perfused rat heart. Life Sci. 68 (25), 2863–2872. doi:10.1016/s0024-3205(01)01067-0
Yuan, B., Yao, M., Wang, X., Sato, A., Okazaki, A., Komuro, H., et al. (2018). Antitumor activity of arsenite in combination with tetrandrine against human breast cancer cell line MDA-MB-231 in vitro and in vivo. Cancer Cell. Int. 18, 113. doi:10.1186/s12935-018-0613-0
Yuan, X., Tong, B., Dou, Y., Wu, X., Wei, Z., and Dai, Y. (2016). Tetrandrine ameliorates collagen-induced arthritis in mice by restoring the balance between Th17 and Treg cells via the aryl hydrocarbon receptor. Biochem. Pharmacol. 101, 87–99. doi:10.1016/j.bcp.2015.11.025
Zhang, A., Meng, K., Liu, Y., Pan, Y., Qu, W., Chen, D., et al. (2020). Absorption, distribution, metabolism, and excretion of nanocarriers in vivo and their influences. Adv. Colloid Interface Sci. 284, 102261. doi:10.1016/j.cis.2020.102261
Zhang, H., Tian, Y., Zhu, Z., Xu, H., Li, X., Zheng, D., et al. (2016). Efficient antitumor effect of co-drug-loaded nanoparticles with gelatin hydrogel by local implantation. Sci. Rep. 6, 26546. doi:10.1038/srep26546
Zhang, H., Xie, B., Zhang, Z., Sheng, X., and Zhang, S. (2019). Tetrandrine suppresses cervical cancer growth by inducing apoptosis in vitro and in vivo. Drug Des. devel. Ther. 13, 119–127. doi:10.2147/DDDT.S187776
Zhang, J., Wang, L., Fai Chan, H., Xie, W., Chen, S., He, C., et al. (2017). Co-delivery of paclitaxel and tetrandrine via iRGD peptide conjugated lipid-polymer hybrid nanoparticles overcome multidrug resistance in cancer cells. Sci. Rep. 7, 46057. doi:10.1038/srep46057
Zhang, L., He, Y., Yu, M., and Song, C. (2011). Paclitaxel-loaded polymeric nanoparticles based on PCL-PEG-PCL: Preparation, in vitro and in vivo evaluation. J. Control. Release 152 Suppl 1, e114–6. doi:10.1016/j.jconrel.2011.08.160
Zhang, T., Guo, R., Li, X., Wang, Y. W., and Li, Y. J. (2017). Tetrandrine cardioprotection in ischemia-reperfusion (I/R) injury via JAK3/STAT3/Hexokinase II. Eur. J. Pharmacol. 813, 153–160. doi:10.1016/j.ejphar.2017.08.019
Zhang, X. (2016). Therapeutic effect of tetrandrine on bovine serum albumin-induced liver fibrosis in rats [J]. Chin. J. Hepatology 8 (02), 29–33.
Zhang, Y., Liu, W., He, W., Deng, X., Ma, Y., et al. (2016). Tetrandrine reverses epithelial-mesenchymal transition in bladder cancer by downregulating Gli-1. Int. J. Oncol. 48 (5), 2035–2042. doi:10.3892/ijo.2016.3415
Zhang, Y., Wang, C., Wang, H., Wang, K., Du, Y., and Zhang, J. (2011). Combination of Tetrandrine with cisplatin enhances cytotoxicity through growth suppression and apoptosis in ovarian cancer in vitro and in vivo. Cancer Lett. 304 (1), 21–32. doi:10.1016/j.canlet.2011.01.022
Zhang, Y., Wen, Y., Ma, J., Ye, J. C., Wang, X., Huang, J. X., et al. (2017). Tetrandrine inhibits glioma stem-like cells by repressing β-catenin expression. Int. J. Oncol. 50 (1), 101–110. doi:10.3892/ijo.2016.3780
Zhang, Z., Liu, T., Yu, M., Li, K., and Li, W. (2018). The plant alkaloid tetrandrine inhibits metastasis via autophagy-dependent Wnt/β-catenin and metastatic tumor antigen 1 signaling in human liver cancer cells. J. Exp. Clin. Cancer Res. 37 (1), 7. doi:10.1186/s13046-018-0678-6
Zhao, Q., Jia, X., Zhang, Y., Dong, Y., Lei, Y., Tan, X., et al. (2019). Tetrandrine induces apoptosis in human neuroblastoma through regulating the Hippo/YAP signaling pathway. Biochem. Biophys. Res. Commun. 513 (4), 846–851. doi:10.1016/j.bbrc.2019.04.075
Zhao, X. M., Hu, W. X., Wu, Z. F., Chen, Y. X., and Zeng, Z. C. (2018). Tetrandrine enhances radiosensitization in human hepatocellular carcinoma cell lines. Radiat. Res. 190 (4), 385–395. doi:10.1667/RR14981.1
Zhao, Y., Wang, L., Ma, C., Zhao, K., Liu, Y., and Feng, N. p. (2013). Preparation and characterization of tetrandrine-phospholipid complex loaded lipid nanocapsules as potential oral carriers. Int. J. Nanomedicine 8, 4169–4181. doi:10.2147/IJN.S50557
Zhou, X., Jin, N., and Chen, B. (2022). Tetrandrine overcomes drug resistance mediated by bone marrow microenvironment by regulating the expression of P-glycoprotein in acute leukemia. Hematol. Amst. Neth. 27 (1), 274–279. doi:10.1080/16078454.2022.2034256
Zhou, Y., Mu, L., Liu, X. L., Li, Q., Ding, L. X., Chen, H. C., et al. (2021). Tetrandrine inhibits proliferation of colon cancer cells by BMP9/PTEN/PI3K/AKT signaling. Genes. & Dis. 8 (3), 373–383. doi:10.1016/j.gendis.2019.10.017
Zhu, R., Liu, T., Tan, Z., Wu, X., Li, M., Jiang, L., et al. (2014). Tetrandrine induces apoptosis in gallbladder carcinoma in vitro. Int. J. Clin. Pharmacol. Ther. 52 (10), 900–905. doi:10.5414/CP202123
Zhuo, C., Liang, Z., and Feng, Z. (2018). Tetrandrine suppresses lung cancer growth and induces apoptosis, potentially via the VEGF/HIF-1α/ICAM-1 signaling pathway [J]. Oncol. Lett. 15.
Keywords: tetrandrine (TET), poor water solubility, low bioavailability, structure modification, nanocarrier delivery systems
Citation: Mo L, Zhang F, Chen F, Xia L, Huang Y, Mo Y, Zhang L, Huang D, He S, Deng J, Hao E and Du Z (2022) Progress on structural modification of Tetrandrine with wide range of pharmacological activities. Front. Pharmacol. 13:978600. doi: 10.3389/fphar.2022.978600
Received: 26 June 2022; Accepted: 18 July 2022;
Published: 16 August 2022.
Edited by:
Milad Ashrafizadeh, Sabancı University, TurkeyReviewed by:
Esmaeel Sharifi, Hamadan University of Medical Sciences, IranGautam Sethi, National University of Singapore, Singapore
Copyright © 2022 Mo, Zhang, Chen, Xia, Huang, Mo, Zhang, Huang, He, Deng, Hao and Du. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jiagang Deng, NzU5MzE5NTA3QHFxLmNvbQ==; Erwei Hao, NTE2MTEwNDkzQHFxLmNvbQ==; Zhengcai Du, NzcxNTI4OTI4QHFxLmNvbQ==
†These authors have contributed equally to this work
 Liuying Mo
Liuying Mo Fan Zhang1,2,3,4†
Fan Zhang1,2,3,4†