
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Pharmacol., 25 April 2022
Sec. Pharmacology of Anti-Cancer Drugs
Volume 13 - 2022 | https://doi.org/10.3389/fphar.2022.873792
This article is part of the Research TopicNatural Products Modulate the Sensitivity of Cancer to Anti-PD-1 Based ImmunotherapyView all 13 articles
Myeloid-derived suppressor cells (MDSCs) are major immunosuppressive cells that accumulate in tumor-bearing hosts. Since MDSCs suppress anti-tumor immunity and promote tumor progression, they are promising targets for cancer immunotherapy. Granulocyte colony-stimulating factor (G-CSF) is an agent used for the treatment of chemotherapy-induced febrile neutropenia (FN) in patients with cancer. However, several reports have revealed that G-CSF plays crucial immune-related adverse roles in tumor progression through MDSCs. In this study, we showed that MDSCs differentiated in the presence of G-CSF in vitro exhibited enhanced proliferation and immunosuppressive activity compared to those differentiated without G-CSF. RNA sequencing analysis demonstrated that G-CSF enhanced the immunosuppressive function of MDSCs by upregulating gamma-glutamyltransferase (GGT) 1. Moreover, in the EL4 lymphoma-bearing neutropenic mouse model, administration of recombinant G-CSF increased the number of MDSCs and attenuated the anti-cancer effect of chemotherapy. We showed that the combination of GGsTop, a GGT inhibitor, could prevent G-CSF-induced tumor growth, without affecting the promotion of myelopoiesis by G-CSF. These results suggest that targeting GGT1 can mitigate G-CSF-induced enhanced immunosuppressive functions of MDSCs and can eliminate the tumor-promoting effect of G-CSF. Furthermore, GGsTop could be an attractive combination agent during G-CSF treatment for FN in patients with cancer.
Myeloid-derived suppressor cells (MDSCs) are immature myeloid cells that accumulate in cancer patients as well as in mouse tumor models. MDSCs play important roles in the suppression of anti-tumor immunity, resulting in the exacerbation of cancer (Fleming et al., 2018; Ostrand-Rosenberg and Fenselau, 2018; Veglia et al., 2021). Phenotypically and morphologically, MDSCs are classified into two subpopulations, namely monocytic (M)-MDSCs with CD11b+Ly-6G−Ly-6Chi phenotype and polymorphonuclear (PMN)-MDSCs with CD11b+Ly-6G+Ly-6Cint phenotype (Bronte et al., 2016). M-MDSCs reportedly suppress T cell proliferation via arginase 1 and inducible nitric oxide synthase (iNOS), and PMN-MDSCs show an inhibitory effect via arginase 1 and reactive oxygen species (ROS) (Bronte et al., 2016). However, the mechanisms underlying the immunosuppressive function of MDSCs remain largely unexplored.
Febrile neutropenia (FN) is a deadly complication associated with cancer chemotherapy, usually indicating infection and sepsis, and commonly occurs following the initial cycles of myelosuppressive therapy (Klastersky et al., 2016). Prevention of FN reduces lengthy hospitalization, morbidity, mortality, and risk of chemotherapy reductions and delays (Kuderer et al., 2006; Mehta et al., 2015; Lucas et al., 2018). Granulocyte colony-stimulating factor (G-CSF) is a principal cytokine promoting and mobilizing granulocytes. Recombinant human G-CSF as prophylaxis and therapy has been shown to significantly reduce FN incidence and mortality following chemotherapy (Klastersky et al., 2016; Lucas et al., 2018). However, several observations suggest that G-CSF promotes tumor growth, metastasis, and resistance to chemotherapy by increasing the number of circulating MDSCs (Kawano et al., 2015; Pilatova et al., 2018) and is associated with a poor clinical prognosis (Hollmén et al., 2016; Liu et al., 2020). Although G-CSF has become the main therapeutic agent in cancer therapy for the treatment of neutropenia and prevention of FN, it is necessary to evaluate its impact on tumor-bearing patients. Therefore, considering the possible side effects of G-CSF in promoting tumor progression through MDSCs, there is an urgent need to elucidate the direct mechanism underlying the effect of G-CSF on MDSCs.
Female C57BL/6J mice were purchased from Japan SLC (Shizuoka, Japan). All mice were bred and maintained under specific pathogen-free conditions and used for experiments at 6–8 weeks of age. All experimental procedures in this study were performed in accordance with the institutional guidelines for animal experiments at the Osaka University, Japan.
The EL4 cell lines were purchased from the American Type Culture Collection (ATCC) and maintained in RPMI-1640 medium (FUJIFILM Wako, Osaka, Japan) supplemented with 10% fetal bovine serum (FBS; Gibco, CA, United States) and 1% Antibiotic-Antimycotic Mixed Stock Solution (100 ×) (Nacalai Tesque, Kyoto, Japan). Cells were cultured according to the ATCC guidelines and used within 1 month of thawing from an early passage (<3 passages of original vial) lots.
The in vitro differentiation of bone marrow (BM) cells into MDSCs was performed as described previously (Xie et al., 2018). Briefly, BM cells from C57BL/6J mice were stimulated with 40 ng/ml recombinant Granulocyte-macrophage CSF (GM-CSF) (Peprotech, NJ, United States) for 4 days in the absence or presence of G-CSF (5 ng/ml, BioLegend, CA, United States) and/or GGsTop (50 mΜ, FUJIFILM Wako) to examine their effects on MDSCs.
Cells were pelleted and washed in phosphate-buffered saline (PBS) supplemented with 2% FBS (2% FBS/PBS). The cell suspension was first blocked with TruStain fcX (anti-mouse CD16/32) antibody (BioLegend) for 5 min, and then stained with the following antibodies (Abs) for 15 min at 4°C: APC anti-mouse CD11b, Pacific Blue anti-mouse Gr-1, PE anti-mouse F4/80, FITC anti-mouse CD11c, APC-Cy7 anti-mouse Ly-6C, FITC anti-mouse Ly-6G, Pacific Blue anti-mouse CD4, and FITC anti-mouse CD8α (BioLegend). Next, the cells were washed and resuspended in 2% FBS/PBS. Shortly before performing measurements, a 7-amino actinomycin D viability staining solution (BioLegend) was added to each sample to stain dead cells. Flow cytometry analysis was performed on a BD FACSCanto II flow cytometer (BD Biosciences), and results were analyzed using the FlowJo software (version10.7.0, BD Biosciences).
CD4+ T cells or CD8+ T cells were isolated from the spleens of C57BL/6J mice using the MojoSort magnetic cell separation system as previously described (Xie et al., 2018) and then labeled with the proliferation dye eFluor 670 (eBioscience, Thermo Fisher Scientific, CA, United States). The eFluor 670-labeled CD4+ T cells or CD8+ T cells were incubated with in vitro differentiated MDSCs at different ratios in a 96-well plate cultured with anti-mouse CD3ɛ Ab/anti-mouse CD28 Ab (BioLegend). After 3 days of incubation at 37°C in 5% CO2, the proliferation of CD4+ and CD8+ T cells determined by the eFluor 670 fluorescence intensity was analyzed using flow cytometry.
PMN-MDSCs (Ly-6G+Ly-6Cint) and M-MDSCs (Ly-6G−Ly-6Chi) were purified from in vitro-differentiated MDSCs cultured with or without the addition of G-CSF by fluorescence-activated cell sorting (purity >95%; JSAN, Bay bioscience Co., Ltd., Kobe, Japan). Total RNA was extracted from the in vitro differentiated MDSCs or sorted MDSC subsets and used to synthesize cDNA using the QuantiTect reverse transcription kit (Qiagen, Hilden, Germany) following the manufacturer’s instructions. qRT-PCR was performed using SYBR Premix Ex Taq (Tli RNaseH Plus; TaKaRa, Kusatsu, Japan) on a CFX96 touch real-time PCR detection system (Bio-Rad). The specific primer sequences are listed in Supplementary Table S1. Glyceraldehyde 3-phosphate dehydrogenase (Gapdh) was used as a reference gene, and relative expressions of other genes were calculated using the 2−ΔΔCt method.
Total RNA was extracted from cells using the miRNeasy Mini kit (Qiagen) according to the manufacturer’s protocol. RNA libraries were prepared for sequencing using a TruSeq stranded mRNA sample prep kit (Illumina, CA, United States) according to the manufacturer’s instructions. Whole transcriptome sequencing was performed on RNA samples using the Illumina HiSeq 2500 platform in a 75 bp single-end mode. Volcano plot representation and pathway enrichment analysis of the differentially expressed genes were performed using BioJupies tools with adjusted p < 0.05 and fold change >2 (Torre et al., 2018). Raw data from this study have been submitted to Gene Expression Omnibus (GEO) under the accession number GSE183066.
MDSCs (1 × 106 cells) were homogenized in 200 µL of GGT assay buffer and centrifuged at 13,000 × g for 10 min following which the supernatant was collected. After reacting with the GGT substrate at 37°C for 40 min, fluorescence (365/460 nm) was measured using a microplate reader SpectraMax iD5 (Molecular Devices). GGT activity was calculated according to the protocol of the GGT Activity Colorimetric Assay Kit (BioVision, CA, United States).
The in vitro MDSC culture medium was collected on day 4. Next, glutamate concentration was measured using Glutamate Assay Kit-WST (Dojindo, Kumamoto, Japan) according to the manufacturer’s protocol. Briefly, a glutamate standard or MDSC culture medium was added to a 96-well microplate; the working solution was then added to each well. After incubating the microplate at 37°C for 30 min, the absorbance was measured at 450 nm using SpectraMax iD5 (Molecular Devices). The glutamate concentration in each sample was calculated using a standard curve.
Intracellular ROS was assayed using a ROS Assay Kit-Highly Sensitive DCFH-DA (Dojindo) according to the manufacturer’s protocol. Briefly, 2 × 105 MDSCs were treated with 2′, 7′dichlorofluorescin diacetate (DCFH-DA) working solution and incubated for 30 min. Subsequently, the MDSCs were washed twice with Hanks’ Balanced Salt Solution, and ROS production was measured using a BD FACSCanto II flow cytometer (BD Biosciences).
EL4 lymphoma cells (4 × 105 cells/mouse) were injected subcutaneously into the lower right flank of C57BL/6J mice. Seven days after inoculation with EL4 cells, mice were intraperitoneally administered a single dose of CPA (100 mg/kg, FUJIFILM Wako) to create a neutropenic mouse model as described previously (Hattori et al., 1990). Starting from the same day, experimental mice received intraperitoneal injections of PBS, GGsTop (5 mg/kg, FUJIFILM Wako), and recombinant G-CSF (200 μg/kg, BioLegend) for six consecutive days. Before administration of the above reagent, mouse retro-orbital blood (approximately 75 μL) was collected for flow cytometry analysis and determining blood cell counts using a Sysmex XT-2000i automated hematology analyzer (Sysmex) daily. The tumor volume was calculated periodically up to day 15 using the following formula: Tumor Volume (cm3) = 0.5 × (Length × Width2).
Significant differences were assessed using Student’s t-test, or a one- or two-way analysis of variance (ANOVA) using GraphPad Prism 7.0 (GraphPad Software). Statistical significance was set at p < 0.05.
Previous studies have reported that GM-CSF stimulates BM cells to differentiate into MDSCs (hereafter referred to as in vitro MDSCs). Freshly isolated BM cells were cultured for 4 days in a medium supplemented with or without G-CSF and GM-CSF to examine whether G-CSF affects the differentiation of in vitro MDSCs. G-CSF did not affect the percentage of MDSCs (CD11b+Gr-1+), dendritic cells (DCs; Gr-1−CD11c+), or macrophages (Gr-1-F4/80+) differentiated from BM cells, as shown in Figure 1A. The proportion of M-MDSCs with the CD11b+Ly-6G−Ly-6Chi phenotype increased in the presence of G-CSF, whereas PMN-MDSCs with the CD11b+Ly-6G+Ly-6Cint phenotype decreased (Figure 1B). However, since whole BM cells largely increased with stimulation by G-CSF, there was no change in the number of PMN-MDSCs, and M-MDSCs substantially increased (Figure 1C). These results suggest that G-CSF promotes the proliferation of MDSCs, especially M-MDSCs.
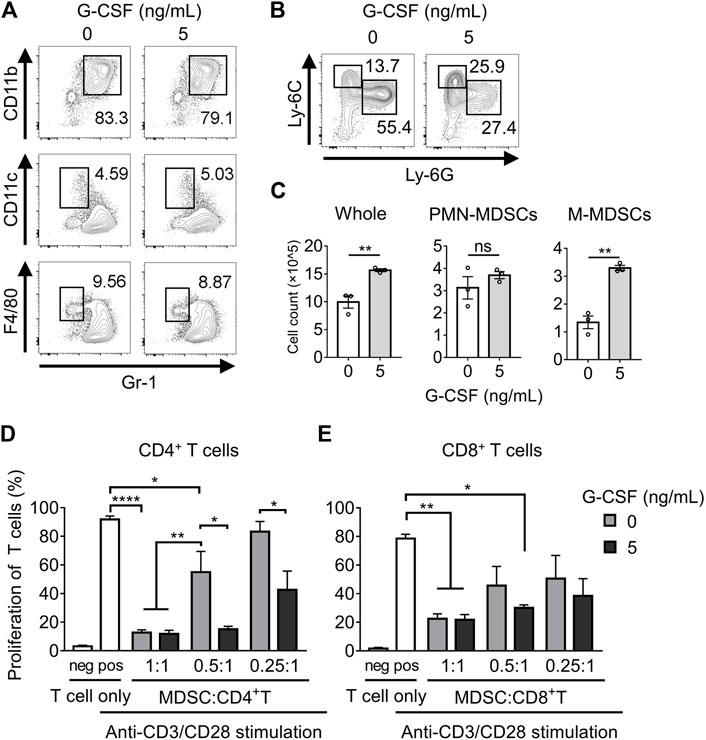
FIGURE 1. G-CSF regulates the differentiation and immunosuppressive activity of MDSCs. (A) Flow cytometry analysis of the percentage of MDSC (CD11b+Gr-1+), DC (Gr-1−CD11c+), and macrophage (Gr-1-F4/80+) populations after 4 days of culture in medium supplemented with GM-CSF (40 ng/ml) with or without G-CSF stimulation (5 ng/ml). (B) Flow cytometry analysis of the percentage of PMN-MDSCs (CD11b+Ly-6G+Ly-6Cint) and M-MDSCs (CD11b+Ly-6G−Ly-6Chi) among CD11b+ cells. (C) Total numbers of in vitro MDSCs and their subset dyed with trypan blue are represented as means ± SEM (n = 3, Student’s t-test: **p < 0.01). (D) CD4+ or CD8+ T cells derived from WT mice were co-cultured with in vitro MDSCs under the stimulation of anti-CD3ɛ/CD28 antibodies. After three-day-culture, T cell proliferation in the absence or presence of MDSCs was examined by flow cytometry. Data are represented as means ± SEM (n = 5 pooled with two independent experiments, two-way ANOVA: *p < 0.05, **p < 0.01, and ****p < 0.0001).
Next, we investigated whether G-CSF affected the immunosuppressive activity of in vitro MDSCs. Suppression of CD4+ T cell proliferation was observed in in vitro MDSCs that were not treated with G-CSF, and the immunosuppressive activity was impaired with decreasing numbers of MDSCs. Moreover, the immunosuppressive activity of in vitro MDSCs was significantly enhanced by G-CSF treatment (Figure 1D). Similar results were obtained in the CD8+ T cell suppression assay (Figure 1E). These observations revealed that T cells, especially CD4+ T cells, were less proliferative when co-cultured with G-CSF-conditioned in vitro MDSCs, suggesting that G-CSF strongly enhanced the immunosuppressive activity of in vitro MDSCs.
To explore the mechanism by which G-CSF enhances the immunosuppressive function of MDSCs, we first analyzed the expression of the main immunosuppressive molecules of MDSCs: arginase 1 and iNOS as well as ROS by qRT-PCR or flow cytometry analysis. We found that the mRNA levels of Arg1 and Nos2 were significantly upregulated by G-CSF and that ROS levels determined by DCFH-DA also increased (Figures 2A,B). Besides, G-CSF upregulated the expression of Arg1 and Nos2 in both PMN-MDSCs and M-MDSCs, indicating that the G-CSF-induced upregulation of those genes was because of the enhanced transcription of the genes but not because of the alteration of the population of MDSC subsets (Figures 2C,D).
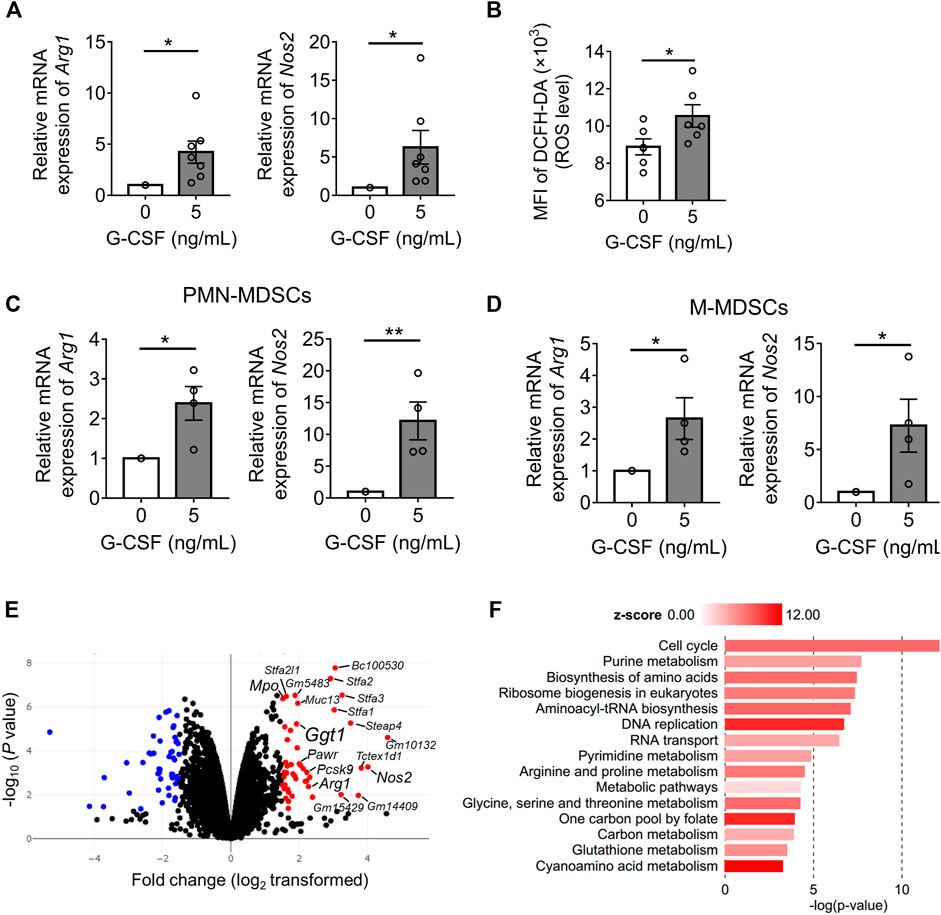
FIGURE 2. Identifying G-CSF-induced MDSC gene expression profile using by RNA sequencing. (A) The mRNA expression of Arg1 and Nos2 of in vitro MDSCs cultured with GM-CSF with or without the addition of G-CSF was measured by qRT-PCR. Data are represented as means ± SEM of three independent experiments and normalized to the expression of Gapdh housekeeping gene (Arg1: n = 7; Nos2: n = 7; Student’s t-test: *p < 0.05). (B) Intracellular ROS level was measured by flow cytometry analysis using DCFH-DA (means ± SEM, n = 4 pooled with two independent experiments, Student’s t-test: *p < 0.05). (C,D) The mRNA expressions of Arg1 and Nos2 in the sorted PMN-MDSCs and M-MDSCs were measured by qRT-PCR (means ± SEM, n = 4 pooled with two independent experiments, Student’s t-test: *p < 0.05, **p < 0.01). (E) Volcano plots showing differences in the mRNA expression profile of in vitro MDSCs (n = 2 biological replicates). (F) KEGG pathway enrichment analysis of significantly upregulated gene sets in MDSCs stimulated by G-CSF using BioJupies tools (n = 2 biological replicates).
Next, we performed an RNA-seq analysis, comparing the gene expression profiles of in vitro MDSCs differentiated with or without G-CSF. There were 228 genes differentially expressed with fold change >2 and adjusted p < 0.05 when comparing both groups. Among these genes, 89 genes were expressed at higher levels in MDSCs differentiated in the presence of G-CSF than in those differentiated without G-CSF, and 139 genes showed lower expression. Increased expression of Arg1, Nos2, and Mpo, which are the immunosuppressive molecules of MDSCs, was observed (Figure 2E), consistent with the above results. Additionally, pathway enrichment analysis revealed that the MDSCs differentiated with G-CSF displayed the activation of the cell cycle, metabolism, and DNA replication, which would account for the enhanced proliferation of MDSCs differentiated with G-CSF (Figure 2F).
Next, among the top 20 upregulated genes by G-CSF, genes associated with poor prognosis (Logrank p < 0.05) in more than four cancers, including Nos2, proprotein convertase subtilisin/kexin type 9 (Pcsk9), PRKC apoptosis WT1 regulator (Pawr), and gamma-glutamyltransferase 1 (Ggt1), were identified as candidate factors involved in the immunosuppressive ability of MDSCs using the GEPIA web server (Tang et al., 2017) (Supplementary Figure S1). GGT1 is a key enzyme involved in several metabolic processes, such as glutathione metabolism and cyanoamino acid metabolism (Verma et al., 2015), which were also enriched and active in MDSCs differentiated with G-CSF (Figure 2F). Furthermore, GGT is expressed on the cell surface, acts as a glutathione hydrolase that increases extracellular cysteinyl glycine and glutamate, and is reported to be associated with the progression of several tumors (Wang et al., 2017; Ince et al., 2020). Thus, to identify novel factors involved in the immunosuppressive ability of MDSCs, we focused on the Ggt1 gene.
The upregulation of the mRNA of Ggt1 by G-CSF in MDSCs was verified by qRT-PCR (Figure 3A). Besides, Ggt1 mRNA expression was more upregulated in M-MDSCs than in PMN-MDSCs upon G-CSF stimulation, suggesting that M-MDSCs can play an important role in increasing the G-CSF-promoted immunosuppressive effects (Figure 3B). To elucidate whether GGT1 is involved in the enhanced immunosuppressive function of MDSCs by G-CSF, we used GGsTop, a highly selective GGT inhibitor. The GGT activity of MDSCs was enhanced by G-CSF, which was inhibited below the detection limit in the GGsTop alone and G-CSF/GGsTop combination groups (Figure 3C). There was no change in the number of MDSCs in the presence of GGsTop (Figure 3D), suggesting that the inhibition of GGT did not affect the G-CSF-induced proliferation of BM cells. Meanwhile, the addition of GGsTop did not affect the proportion and number of M-MDSCs or PMN-MDSCs (Supplementary Figures S2A,B). We further investigated whether GGsTop cancels the enhancement of the immunosuppressive activity of in vitro MDSCs by G-CSF. Consistent with Ggt1 expression, the immunosuppressive activity of in vitro MDSCs was significantly increased by G-CSF, while the combination of GGsTop and G-CSF attenuated the immunosuppressive activity to the same level as that in the control group (Figures 3E–G). Additionally, GGsTop alone did not affect the immunosuppressive activity of in vitro MDSCs. Furthermore, we examined the effect of GGsTop alone or the G-CSF/GGsTop combination on Arg1, Nos2, and ROS expression. The G-CSF/GGsTop combination group downregulated the expression of Arg1 and Nos2. Unexpectedly, GGsTop alone upregulated their expression (Supplementary Figure S2C). As Arg1 and Nos2 upregulation by GGsTop alone did not increase the immunosuppressive ability of MDSCs (Figures 3E,F), suggesting that the upregulation of Arg1 and Nos2 did not play a decisive role in increasing the MDSC immunosuppressive ability caused by G-CSF. In addition, GGsTop did not abrogate the expression of ROS caused by G-CSF, suggesting that ROS is not a key factor either (Supplementary Figure S2D). These results suggest that inhibition of GGT1 activity eliminates G-CSF-induced enhanced immunosuppressive function of MDSCs, but has no effect on its ability to promote proliferation.
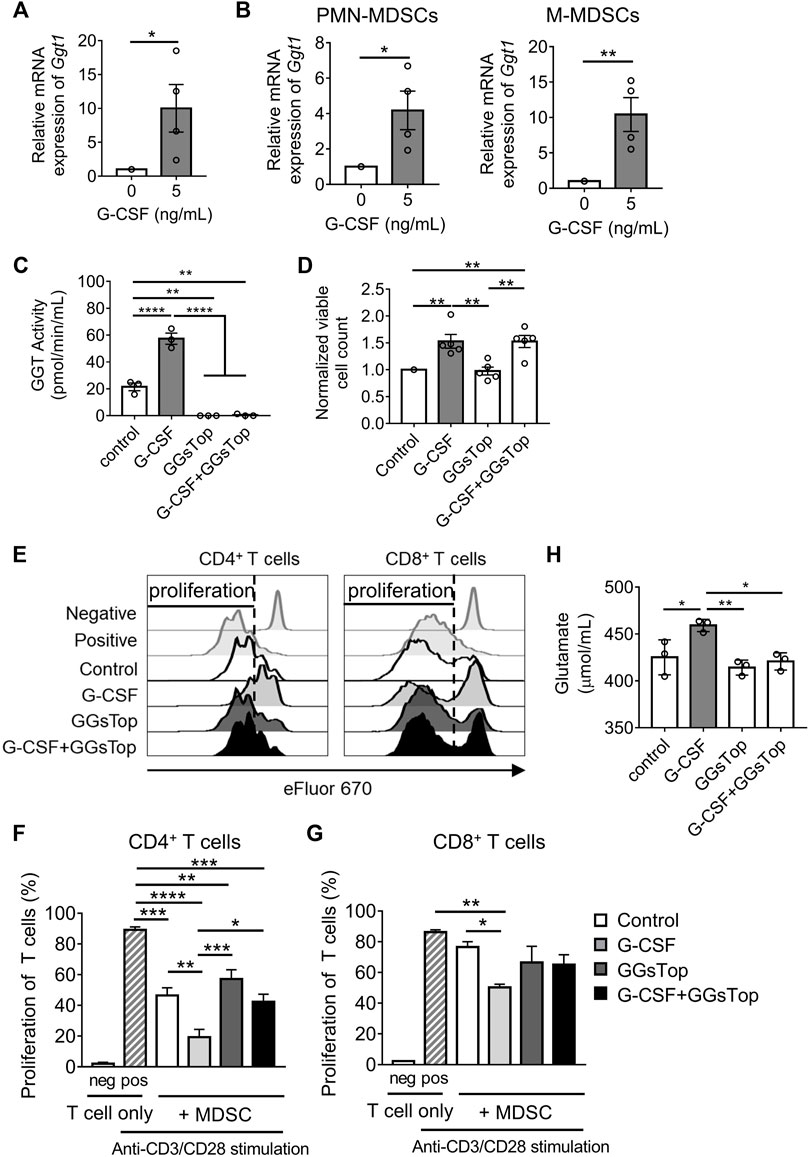
FIGURE 3. GGT1 is involved in the G-CSF-enhanced immunosuppressive function of MDSCs. (A) The mRNA expression of Ggt1 in in vitro MDSCs was measured by qRT-PCR. Data are represented as means ± SEM of three independent experiments (n = 4, Student’s t-test: *p < 0.05). (B) The mRNA expression of Ggt1 in the sorted PMN-MDSCs and M-MDSCs was measured by qRT-PCR (means ± S.E.M., n = 4 pooled with two independent experiments, Student’s t-test: *p < 0.05, **p < 0.01). (C) GGT activity of in vitro MDSCs cultured with or without the addition of G-CSF and/or GGsTop was measured using a GGT activity assay kit (means ± SEM, n = 4 pooled with two independent experiments, one-way ANOVA: *p < 0.05, **p < 0.01, and ****p < 0.0001). (D) The numbers of in vitro MDSCs were measured by trypan blue dye exclusion method (means ± SEM, n = 5 pooled with three independent experiments, one-way ANOVA: **p < 0.01). (E–G) MDSCs conditioned with or without G-CSF or GGsTop were combined in a 0.5:1 ratio with eFluor 670-labeled CD4+ or CD8+ T cells, followed by stimulation with anti-CD3ɛ/CD28 antibodies. (E) Representative histograms of eFluor 670 expressions in CD4+ or CD8+ T cells. (F,G) T cell proliferation was examined by flow cytometry. Data are represented as means ± SEM (two-way ANOVA: *p < 0.05, **p < 0.01, ***p < 0.001, and ****p < 0.0001). (H) The glutamate level was measured from the culture medium of in vitro MDSCs by glutamate assay-kit (means ± SEM, n = 4 pooled with two independent experiments, one-way ANOVA: *p < 0.05, **p < 0.01).
The key role of GGT1 is to hydrolyze glutathione. We speculated that metabolites of glutathione, such as glutamate and cysteinyl glycine, obtained by the action of GGT1, enhance the immunosuppressive function of MDSCs. Elevated glutamate concentrations are commonly observed in tumor patients, and studies indicate that glutamate could regulate the immunosuppressive activity of MDSCs on the proliferation of T cells (Morikawa et al., 2018; Wu et al., 2019). Therefore, we measured the concentration of glutamate to determine whether the level of glutamate is regulated by GGT1. Glutamate levels were increased on treating cells with G-CSF and were decreased on treatment with GGsTop both alone and in combination with G-CSF (Figure 3H). GGsTop alone or the G-CSF/GGsTop combination did not significantly reduce glutamate levels compared with the control, which explained why GGsTop alone did not inhibit MDSC-mediated T cell suppression. These observations revealed that elevated GGT activity induced by G-CSF increased glutamate levels, and that GGT1 is a novel factor involved in the enhanced immunosuppressive function of MDSCs induced by G-CSF.
Given that G-CSF enhances the immunosuppressive function and proliferation of MDSCs, it is possible that G-CSF causes side effects through MDSCs when used to prevent FN. We then tested whether G-CSF promoted MDSCs and tumor progression using the EL4 lymphoma-bearing neutropenic mouse model. In the EL4 tumor-bearing mouse model, administration of the anti-tumor drug CPA reduced white blood cells, including neutrophils, monocytes, and lymphocytes to 50% below the pretreatment value measured using an XT-2000i hematology analyzer, leading to neutropenia (Figures 4A–D). CPA strongly inhibited tumor growth (Figure 4F). In contrast, the accelerated recovery of white blood cells was observed in mice administered G-CSF after CPA. G-CSF dramatically increased neutrophil and monocyte counts from days 11–13 and facilitated the recovery of lymphocytes to control levels (Figures 4A–D), which would reduce the risk associated with CPA-induced neutropenia. Flow cytometry analysis revealed that the proportion of M-MDSCs and PMN-MDSCs in the G-CSF group increased significantly, suggesting that G-CSF promoted neutrophils and monocytes that were morphologically identified using an XT-2000i hematology analyzer to be mixed with most MDSCs (Figure 4E). Furthermore, tumor progression was observed after G-CSF administration, suggesting that G-CSF-induced MDSCs attenuated the anti-tumor effect of CPA (Figure 4F). Next, we explored whether GGsTop could inhibit the growth of tumors induced by G-CSF while maintaining myelopoiesis by G-CSF. As expected, GGsTop alone did not affect the dynamics of myelopoiesis (Figures 4A–E) and tumor progression (Figure 4F). The combination of GGsTop and G-CSF promoted the proliferation of white blood cells, neutrophils, and monocytes in the G-CSF group and inhibited tumor growth in the presence of CPA. Consistent with other studies (Jounaidi and Waxman, 2001; Kim et al., 2018), due to CPA drug toxicity and rapid tumor growth in the PBS and GGsTop groups, the biological significance of body weight was shown in the CPA-treated and control mice. Moreover, the results showed that GGsTop was well-tolerated, with no significant decrease observed in the body weight (Figure 4G). These results suggest that targeting GGT eliminates the effect of G-CSF in promoting tumor growth. GGsTop could be an attractive combination agent with G-CSF for the treatment of neutropenia in patients with cancer.
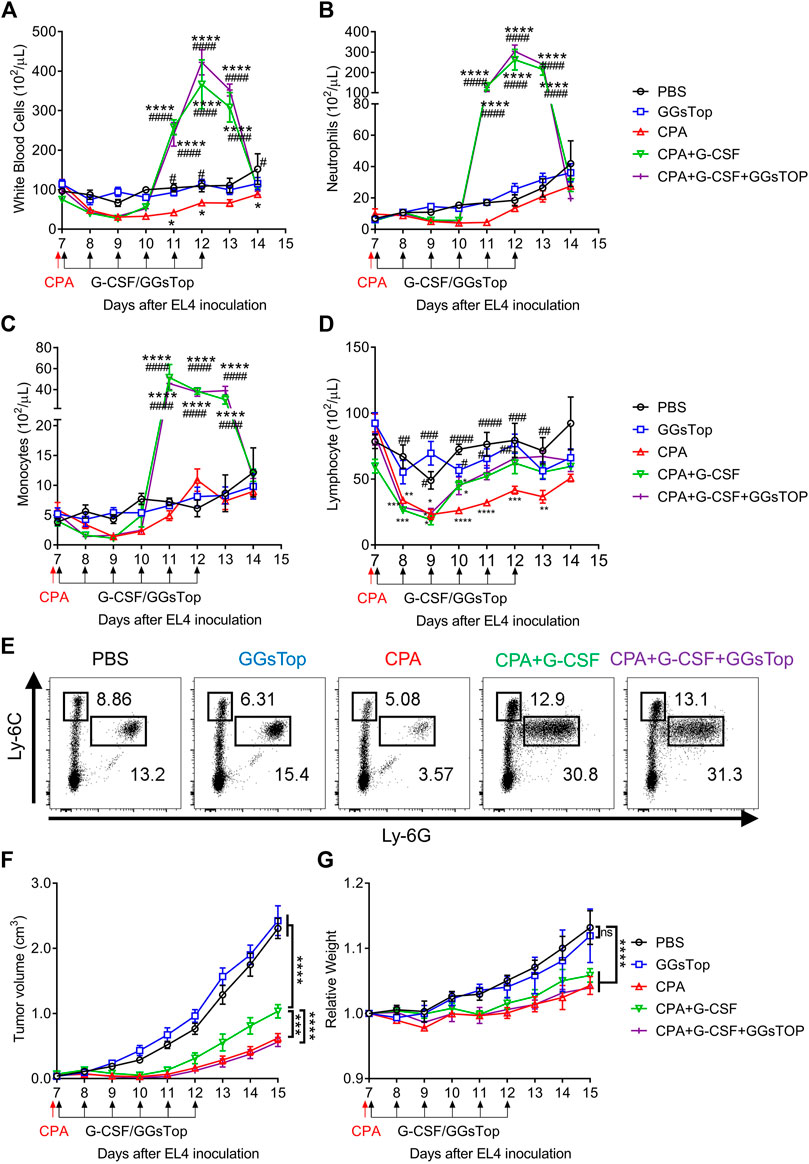
FIGURE 4. GGsTop eliminates the effect of G-CSF in promoting tumor growth. (A–D) Dynamics of circulating (A) white blood cells, (B) neutrophils, (C) monocytes, and (D) lymphocytes was examined using a Sysmex XT-2000i automated hematology analyzer (means ± SEM, pooled from two independent experiments with n = 6, *p < 0.05, **p < 0.01, ***p < 0.001 and ****p < 0.0001 compared with PBS group by two-way ANOVA; #p < 0.05, ##p < 0.01, ###p < 0.001 and ####p < 0.0001 compared with CPA group by two-way ANOVA). (E) Flow cytometric analysis of MDSC subsets in the blood at 4 days after CPA/G-CSF/GGsTop administration. (F,G) Tumor volumes and relative body weight were calculated periodically and are shown as means ± SEM, pooled from two independent experiments with n = 6 (***p < 0.001, and ****p < 0.0001 by two-way ANOVA).
To date, G-CSF administration is commonly used in the management of cancer patients and dramatically reduces the risks associated with chemotherapy-induced FN (Pilatova et al., 2018). However, an increasing number of studies have found that G-CSF enhances the pro-tumor effects of MDSCs, thereby leading to poor prognosis and chemoresistance in cancers (Hollmén et al., 2016; Liu et al., 2020; Karagiannidis et al., 2021). Therefore, the safety of G-CSF as an adjunct to cancer treatment should be addressed. Although further research is needed, our pre-clinical data showed that G-CSF promotes tumor growth in the CPA-induced neutropenia EL4 tumor-bearing model, which would result from the enhancement of immunosuppressive function and the increase in the number of MDSCs by G-CSF.
To eliminate the side effects of G-CSF, it is necessary to elucidate the direct mechanism underlying the action of G-CSF on MDSCs. We utilized in vitro MDSCs and RNA-seq analysis and identified that GGT1 played a pivotal role in enhancing the immunosuppressive function of MDSCs by G-CSF. GGT1 is a member of the GGT family, which also includes other proteins, such as GGT2, GGT3P, GGT4P, GGT5, GGT6, GGT7, and GGT8P. Among these, GGT1 and GGT5 are the only two enzymes with catalytic activity (Takemura et al., 2021), and the catalytic rate of GGT1 is approximately 46 times faster than that of GGT5 (Wickham et al., 2011). Therefore, GGT1 is the main catalytically active enzyme in the GGT family. It has been reported that the activity of GGT is increased in cancers resistant to chemotherapy, and the removal of glutathione, the substrate of GGT, in tumors inhibits tumor progression and metastasis (Mena et al., 2007; Hanigan, 2014). Our results also showed that G-CSF upregulated GGT1 expression on MDSCs, while GGsTop canceled the enhanced T cell proliferation inhibitory ability of MDSCs induced by G-CSF. Additionally, we showed that G-CSF increased the level of glutamate in the culture medium. GGT1 hydrolyzes extracellular glutathione to produce glutamate. Glutamate was originally discovered to be the main excitatory neurotransmitter in the central nervous system and can also act as a signaling molecule in other tissues (Affaticati et al., 2011). Recent studies have shown that glutamate could promote the activation of early MDSCs and the high concentrations of extracellular glutamate can suppress T cell activation (Poulopoulou et al., 2005; Wu et al., 2019). Previously, we found that glutamate signaling through metabotropic glutamate receptor 2/3 increases the potency of the immunosuppressive ability of MDSCs, especially the inhibition of CD4+ T cell proliferation, similar to G-CSF (Morikawa et al., 2018). The expression of glutamate receptors differs among different types of T lymphocytes (Pacheco et al., 2007); thus, CD8+ T cells may express lower glutamate receptor levels, resulting in the relative resistance of CD8+ T cells to the increased suppressive function of MDSCs cultured with G-CSF. These results indicate that the mechanism by which G-CSF enhances the immunosuppressive ability of MDSCs may involve GGT-induced increased glutamate levels.
GGsTop is now commercially available and its beneficial properties, such as being non-toxic, highly selective, and potent irreversible inhibitor of GGT activity, have been reported under a variety of experimental conditions (Kamiyama et al., 2016). For example, it has been reported that GGsTop could quickly and safely treat oral mucositis induced by cancer chemotherapy (Shimamura et al., 2019), reduce hepatic ischemia-reperfusion injury (Tamura et al., 2016), and may protect against chronic experimental autoimmune encephalomyelitis progression (Mendiola et al., 2020). Our results demonstrated its safety and effectiveness in an in vivo CPA-induced neutropenic tumor model, indicating that GGsTop can eliminate the effect of G-CSF in promoting tumor growth. Inhibition of GGT1 by GGsTop did not affect the viability and proliferation of MDSCs in vitro, as assessed by cell number quantification. The numbers of white blood cells, neutrophils, monocytes, and lymphocytes were similar after 6 days of treatment with GGsTop compared to PBS control in vivo. Accordingly, the inhibition of GGT1 by another inhibitor, acivicin, also showed no difference in microglia, macrophages, and whole blood cell numbers, but altered the glutathione metabolism of those cells (Mendiola et al., 2020). These results indicated that inhibiting GGT1 regulated the metabolism and activity of myeloid cells, including MDSCs, but had no effect on their proliferative activity.
In conclusion, our study showed that G-CSF directly enhanced the proliferation and immunosuppressive function of MDSCs and identified GGT1 as a novel factor involved in the enhanced immunosuppressive function of MDSCs induced by G-CSF. Furthermore, our results demonstrated that tumor progression in the chemotherapy-induced neutropenic mouse model was promoted by G-CSF, and the specific GGT inhibitor, GGsTop, prevented G-CSF-induced EL4 lymphoma progression. Although our findings suggest that GGsTop exerts its effects by targeting GGT1 on MDSCs, GGT1 is also expressed in a variety of cells other than MDSCs, and the possibility that GGsTop functions through other cellular mechanisms cannot be excluded, thus suggesting the requirement for further studies. Our study also still lacks clinical human experimental data to prove the safety and efficacy of GGsTop, the existing results suggest that GGsTop would be effective in future clinical applications. These findings should contribute to the development of safer and more effective treatment strategies for FN in patients with cancer.
The datasets presented in this study can be found in online repositories. The names of the repository/repositories and accession number(s) can be found in the article/Supplementary Material.
The animal study was reviewed and approved by Osaka University.
ZX and MT conceptualized the study, designed the experiments, and wrote the manuscript. ZX and TK performed the experiments. HZ assisted in the acquisition of data. DO helped with the RNA-seq experiment and wrote the associated part of the manuscript. NO helped with the design of the experiments and assisted with discussions. All authors helped with reviewing and revising the manuscript.
This work was supported, in part, by JSPS KAKENHI Grant Number JP19H04049 [Grants-in-aid for Scientific Research (B)] and Suzuken Memorial Foundation (MT). This research was also partially supported by the Platform Project for Supporting Drug Discovery and Life Science Research [Basis for Supporting Innovative Drug Discovery and Life Science Research (BINDS)] from AMED under Grant Numbers JP21am0101084 and JP21am0101123.
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors, and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2022.873792/full#supplementary-material
Affaticati, P., Mignen, O., Jambou, F., Potier, M. C., Klingel-Schmitt, I., Degrouard, J., et al. (2011). Sustained Calcium Signalling and Caspase-3 Activation Involve NMDA Receptors in Thymocytes in Contact with Dendritic Cells. Cell Death Differ 18, 99–108. doi:10.1038/cdd.2010.79
Bronte, V., Brandau, S., Chen, S. H., Colombo, M. P., Frey, A. B., Greten, T. F., et al. (2016). Recommendations for Myeloid-Derived Suppressor Cell Nomenclature and Characterization Standards. Nat. Commun. 7, 12150. doi:10.1038/ncomms12150
Fleming, V., Hu, X., Weber, R., Nagibin, V., Groth, C., Altevogt, P., et al. (2018). Targeting Myeloid-Derived Suppressor Cells to Bypass Tumor-Induced Immunosuppression. Front. Immunol. 9, 398. doi:10.3389/fimmu.2018.00398
Hanigan, M. H. (2014). Gamma-glutamyl Transpeptidase: Redox Regulation and Drug Resistance. Adv. Cancer Res. 122, 103–141. doi:10.1016/B978-0-12-420117-0.00003-7
Hattori, K., Shimizu, K., Takahashi, M., Tamura, M., Oheda, M., Ohsawa, N., et al. (1990). Quantitative In Vivo Assay of Human Granulocyte colony-stimulating Factor Using Cyclophosphamide-Induced Neutropenic Mice. Blood 75, 1228–1233. doi:10.1182/blood.v75.6.1228.bloodjournal7561228
Hollmén, M., Karaman, S., Schwager, S., Lisibach, A., Christiansen, A. J., Maksimow, M., et al. (2016). G-CSF Regulates Macrophage Phenotype and Associates with Poor Overall Survival in Human Triple-Negative Breast Cancer. Oncoimmunology 5, e1115177. doi:10.1080/2162402X.2015.1115177
Ince, V., Carr, B. I., Bag, H. G., Koc, C., Usta, S., Ersan, V., et al. (2020). Gamma Glutamyl Transpeptidase as a Prognostic Biomarker in Hepatocellular Cancer Patients Especially with >5 Cm Tumors, Treated by Liver Transplantation. Int. J. Biol. Markers 35, 91–95. doi:10.1177/1724600820921869
Jounaidi, Y., and Waxman, D. J. (2001). Frequent, Moderate-Dose Cyclophosphamide Administration Improves the Efficacy of Cytochrome P-450/cytochrome P-450 Reductase-Based Cancer Gene Therapy. Cancer Res. 61, 4437–4444.
Kamiyama, A., Nakajima, M., Han, L., Wada, K., Mizutani, M., Tabuchi, Y., et al. (2016). Phosphonate-based Irreversible Inhibitors of Human gamma-glutamyl Transpeptidase (GGT). GGsTop Is a Non-toxic and Highly Selective Inhibitor with Critical Electrostatic Interaction with an Active-Site Residue Lys562 for Enhanced Inhibitory Activity. Bioorg. Med. Chem. 24, 5340–5352. doi:10.1016/j.bmc.2016.08.050
Karagiannidis, I., Salataj, E., Said Abu Egal, E., and Beswick, E. J. (2021). G-CSF in Tumors: Aggressiveness, Tumor Microenvironment and Immune Cell Regulation. Cytokine 142, 155479. doi:10.1016/j.cyto.2021.155479
Kawano, M., Mabuchi, S., Matsumoto, Y., Sasano, T., Takahashi, R., Kuroda, H., et al. (2015). The Significance of G-CSF Expression and Myeloid-Derived Suppressor Cells in the Chemoresistance of Uterine Cervical Cancer. Sci. Rep. 5, 18217. doi:10.1038/srep18217
Kim, J. W., Choi, J.-S., Seol, D. J., Choung, J. J., and Ku, S. K. (2018). Immunomodulatory Effects of Kuseonwangdogo-Based Mixed Herbal Formula Extracts on a Cyclophosphamide-Induced Immunosuppression Mouse Model. Evidence-Based Complement. Altern. Med. 2018, 1–14. doi:10.1155/2018/6017412
Klastersky, J., de Naurois, J., Rolston, K., Rapoport, B., Maschmeyer, G., Aapro, M., et al. (2016). Management of Febrile Neutropaenia: ESMO Clinical Practice Guidelines. Ann. Oncol. 27, v111. doi:10.1093/annonc/mdw325
Kuderer, N. M., Dale, D. C., Crawford, J., Cosler, L. E., and Lyman, G. H. (2006). Mortality, Morbidity, and Cost Associated with Febrile Neutropenia in Adult Cancer Patients. Cancer 106, 2258–2266. doi:10.1002/cncr.21847
Liu, L., Liu, Y., Yan, X., Zhou, C., and Xiong, X. (2020). The Role of Granulocyte Colony-stimulating Factor in Breast Cancer Development: A Review. Mol. Med. Rep. 21, 2019–2029. doi:10.3892/mmr.2020.11017
Lucas, A. J., Olin, J. L., and Coleman, M. D. (2018). Management and Preventive Measures for Febrile Neutropenia. P T 43, 228–232.
Mehta, H. M., Malandra, M., and Corey, S. J. (2015). G-CSF and GM-CSF in Neutropenia. J. Immunol. 195, 1341–1349. doi:10.4049/jimmunol.1500861
Mena, S., Benlloch, M., Ortega, A., Carretero, J., Obrador, E., Asensi, M., et al. (2007). Bcl-2 and Glutathione Depletion Sensitizes B16 Melanoma to Combination Therapy and Eliminates Metastatic Disease. Clin. Cancer Res. 13, 2658–2666. doi:10.1158/1078-0432.CCR-06-2642
Mendiola, A. S., Ryu, J. K., Bardehle, S., Meyer-Franke, A., Ang, K. K., Wilson, C., et al. (2020). Transcriptional Profiling and Therapeutic Targeting of Oxidative Stress in Neuroinflammation. Nat. Immunol. 21, 513–524. doi:10.1038/s41590-020-0654-0
Morikawa, N., Tachibana, M., Ago, Y., Goda, H., Sakurai, F., and Mizuguchi, H. (2018). LY341495, an mGluR2/3 Antagonist, Regulates the Immunosuppressive Function of Myeloid-Derived Suppressor Cells and Inhibits Melanoma Tumor Growth. Biol. Pharm. Bull. 41, 1866–1869. doi:10.1248/bpb.b18-00055
Ostrand-Rosenberg, S., and Fenselau, C. (2018). Myeloid-derived Suppressor Cells: Immune-Suppressive Cells that Impair Antitumor Immunity and Are Sculpted by Their Environment. J.I. 200, 422–431. doi:10.4049/jimmunol.1701019
Pacheco, R., Gallart, T., Lluis, C., and Franco, R. (2007). Role of Glutamate on T-Cell Mediated Immunity. J. Neuroimmunol 185, 9–19. doi:10.1016/j.jneuroim.2007.01.003
Pilatova, K., Bencsikova, B., Demlova, R., Valik, D., and Zdrazilova-Dubska, L. (2018). Myeloid-derived Suppressor Cells (MDSCs) in Patients with Solid Tumors: Considerations for Granulocyte colony-stimulating Factor Treatment. Cancer Immunol. Immunother. 67, 1919–1929. doi:10.1007/s00262-018-2166-4
Poulopoulou, C., Markakis, I., Davaki, P., Nikolaou, C., Poulopoulos, A., Raptis, E., et al. (2005). Modulation of Voltage-Gated Potassium Channels in Human T Lymphocytes by Extracellular Glutamate. Mol. Pharmacol. 67, 856–867. doi:10.1124/mol.67.310.1124/mol.67.3
Shimamura, Y., Takeuchi, I., Terada, H., and Makino, K. (2019). Therapeutic Effect of GGsTop, Selective Gamma-Glutamyl Transpeptidase Inhibitor, on a Mouse Model of 5-Fluorouracil-Induced Oral Mucositis. Anticancer Res. 39, 201–206. doi:10.21873/anticanres.13098
Takemura, K., Board, P. G., and Koga, F. (2021). A Systematic Review of Serum γ-Glutamyltransferase as a Prognostic Biomarker in Patients with Genitourinary Cancer. Antioxidants (Basel) 10, 549. doi:10.3390/antiox10040549
Tamura, K., Hayashi, N., George, J., Toshikuni, N., Arisawa, T., Hiratake, J., et al. (2016). GGsTop, a Novel and Specific γ-glutamyl Transpeptidase Inhibitor, Protects Hepatic Ischemia-Reperfusion Injury in Rats. Am. J. Physiol. Gastrointest. Liver Physiol. 311, G305–G312. doi:10.1152/ajpgi.00439.2015
Tang, Z., Li, C., Kang, B., Gao, G., Li, C., and Zhang, Z. (2017). GEPIA: a Web Server for Cancer and normal Gene Expression Profiling and Interactive Analyses. Nucleic Acids Res. 45, W98–W102. doi:10.1093/nar/gkx247
Torre, D., Lachmann, A., and Ma'ayan, A. (2018). BioJupies: Automated Generation of Interactive Notebooks for RNA-Seq Data Analysis in the Cloud. Cell Syst 7, 556. doi:10.1016/j.cels.2018.10.007
Veglia, F., Sanseviero, E., and Gabrilovich, D. I. (2021). Myeloid-derived Suppressor Cells in the Era of Increasing Myeloid Cell Diversity. Nat. Rev. Immunol. 21, 485–498. doi:10.1038/s41577-020-00490-y
Verma, V. V., Gupta, R., and Goel, M. (2015). "Phylogenetic and Evolutionary Analysis of Functional Divergence Among Gamma Glutamyl Transpeptidase (GGT) Subfamilies". Biol. Direct 10, 49. doi:10.1186/s13062-015-0080-7
Wang, Q., Shu, X., Dong, Y., Zhou, J., Teng, R., Shen, J., et al. (2017). Tumor and Serum Gamma-Glutamyl Transpeptidase, New Prognostic and Molecular Interpretation of an Old Biomarker in Gastric Cancer. Oncotarget 8, 36171–36184. doi:10.18632/oncotarget.15609
Wickham, S., West, M. B., Cook, P. F., and Hanigan, M. H. (2011). Gamma-glutamyl Compounds: Substrate Specificity of Gamma-Glutamyl Transpeptidase Enzymes. Anal. Biochem. 414, 208–214. doi:10.1016/j.ab.2011.03.026
Wu, W. C., Sun, H. W., Chen, J., OuYang, H. Y., Yu, X. J., Chen, H. T., et al. (2019). Immunosuppressive Immature Myeloid Cell Generation Is Controlled by Glutamine Metabolism in Human Cancer. Cancer Immunol. Res. 7, 1605–1618. doi:10.1158/2326-6066.CIR-18-0902
Keywords: myeloid-derived suppressor cells, granulocyte colony-stimulating factor, GGT1, febrile neutropenia, GGsTop
Citation: Xie Z, Kawasaki T, Zhou H, Okuzaki D, Okada N and Tachibana M (2022) Targeting GGT1 Eliminates the Tumor-Promoting Effect and Enhanced Immunosuppressive Function of Myeloid-Derived Suppressor Cells Caused by G-CSF. Front. Pharmacol. 13:873792. doi: 10.3389/fphar.2022.873792
Received: 11 February 2022; Accepted: 07 April 2022;
Published: 25 April 2022.
Edited by:
Peixin Dong, Hokkaido University, JapanReviewed by:
Hiroaki Shime, Nagoya City University, JapanCopyright © 2022 Xie, Kawasaki, Zhou, Okuzaki, Okada and Tachibana. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Masashi Tachibana, dGFjY2lAcGhzLm9zYWthLXUuYWMuanA=, b3JjaWQub3JnLzAwMDAtMDAwMi0wMzc2LTUwOFg=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.