
94% of researchers rate our articles as excellent or good
Learn more about the work of our research integrity team to safeguard the quality of each article we publish.
Find out more
ORIGINAL RESEARCH article
Front. Pharmacol., 12 January 2022
Sec. Translational Pharmacology
Volume 12 - 2021 | https://doi.org/10.3389/fphar.2021.812295
This article is part of the Research TopicVenoms, Animal and Microbial Toxins, Volume IIView all 21 articles
 Pamella G. Gutierres1
Pamella G. Gutierres1 Diego R. Pereira1
Diego R. Pereira1 Nataly L. Vieira1
Nataly L. Vieira1 Lilian F. Arantes2
Lilian F. Arantes2 Nelson J. Silva Jr.3
Nelson J. Silva Jr.3 Kristian A. Torres-Bonilla4
Kristian A. Torres-Bonilla4 Stephen Hyslop4
Stephen Hyslop4 Karen Morais-Zani5
Karen Morais-Zani5 Rosa M. B. Nogueira1
Rosa M. B. Nogueira1 Edward G. Rowan6
Edward G. Rowan6 Rafael S. Floriano1*
Rafael S. Floriano1*Varespladib (VPL) was primarily developed to treat inflammatory disturbances associated with high levels of serum phospholipase A2 (PLA2). VPL has also demonstrated to be a potential antivenom support agent to prevent PLA2-dependent effects produced by snake venoms. In this study, we examined the action of VPL on the coagulant, haemorrhagic and enzymatic activities of Lachesis muta rhombeata (South-American bushmaster) venom. Conventional colorimetric enzymatic assays were performed for PLA2, caseinolytic and esterasic activities; in vitro coagulant activities for prothrombin time (PT) and activated partial thromboplastin time (aPTT) were performed in rat citrated plasma through a quick timer coagulometer, whereas the dimensions of haemorrhagic haloes obtained after i.d. injections of venom in Wistar rats were determined using ImageJ software. Venom (1 mg/ml) exhibited accentuated enzymatic activities for proteases and PLA2 in vitro, with VPL abolishing the PLA2 activity from 0.01 mM; VPL did not affect caseinolytic and esterasic activities at any tested concentrations (0.001–1 mM). In rat citrated plasma in vitro, VPL (1 mM) alone efficiently prevented the venom (1 mg/ml)-induced procoagulant disorder associated to extrinsic (PT) pathway, whereas its association with a commercial antivenom successfully prevented changes in both intrinsic (aPTT) and extrinsic (PT) pathways; commercial antivenom by itself failed to avoid the procoagulant disorders by this venom. Venom (0.5 mg/kg)-induced hemorrhagic activity was slightly reduced by VPL (1 mM) alone or combined with antivenom (antivenom:venom ratio 1:3 ‘v/w’) in rats, with antivenom alone producing no protective action on this parameter. In conclusion, VPL does not inhibit other major enzymatic groups of L. m. rhombeata venom, with its high PLA2 antagonize activity efficaciously preventing the venom-induced coagulation disturbances.
Snakes of Lachesis genus are represented by three species found in Central America (L. stenophrys, L. melanocephala, and L. acrochorda) and one in South America (L. muta), with the latter being recognized as two subspecies distributed in the Amazon river basin (=L. muta muta) and Atlantic rainforest (=L. muta rhombeata) in Brazil (Costa and Bérnils, 2018; Nogueira et al., 2019; Diniz-Sousa et al., 2020). Together, these snakes are responsible by the second most frequent cases of snakebites in Americas, being exceeded only by Bothrops snakes (Magalhães et al., 2019; Diniz-Sousa et al., 2020).
Envenomation by Lachesis spp. is characterized by intense local pain, oedema and necrosis (Damico et al., 2006; Ferreira et al., 2009; Damico et al., 2012), systemic myotoxicity (Fuly et al., 2000; Fuly et al., 2003; Damico et al., 2006), renal failure (Damico et al., 2007), haemorrhage and coagulopathy (Sánchez et al., 1987; Sánchez et al., 1991; Sánchez et al., 1995; Fuly et al., 1997; Rucavado et al., 1999; Estêvão-Costa et al., 2000; Torres-Huaco et al., 2013), including severe cardiovascular disorders (Diniz and Oliveira, 1992; Giovanni-De-Simone et al., 1997; Dias et al., 2016a; Dias et al., 2016b). Such effects have been associated predominantly with the presence of phospholipases A2 (PLA2) (Cordeiro et al., 2015; Diniz-Sousa et al., 2018), metalloproteases (Cordeiro et al., 2018) and serine proteases (Wiezel et al., 2019), including biologically active peptides (Graham et al., 2005; Soares et al., 2005; Sanz et al., 2008; Pla et al., 2013; Pinheiro-Júnior et al., 2018), in these venoms.
Polyvalent antivenom (=anti-Bothrops/Lachesis serum) therapies comprise the main therapeutic options to treat the systemic envenomation by Lachesis snakes (Madrigal et al., 2017; Solano et al., 2018). Recently, several studies have demonstrated the value of varespladib, a PLA2 inhibitor drug (Lewin et al., 2016; Salvador et al., 2019), concerning its suppressive action on the biological effects of Elapidae and Viperidae venoms (Bittenbinder et al., 2018; Lewin et al., 2018; Wang et al., 2018; Zinenko et al., 2020; Gutiérrez et al., 2020a), including their toxins (Bryan-Quirós et al., 2019; Salvador et al., 2021). However, there are not reports about the action of varespladib, as a stand-alone therapy and/or combined with antivenoms, on the toxic effects caused by Lachesis venoms. In the present study, we have investigated the efficiency of this drug on some aspects of the envenomation by Lachesis muta rhombeata venom using in vitro and in vivo approaches for enzymatic, coagulant and haemorrhagic activities of this venom. We have also assessed an eventual synergic mechanism of action by varespladib when combined with a commercial antivenom used to treat envenomations by Lachesis in Brazil.
Varespladib (LY-315920) was obtained from Sigma-Aldrich Chemical Co. (St. Louis, MO, United States) and anti-Bothrops/Lachesis serum was from Butantan Institute (São Paulo, SP, Brazil); varespladib was dissolved in DMSO prior to use, whereas the antivenom was provided ready for injection and maintained under refrigeration. Azocasein (A2765), Nα-Benzoyl-dl-arginine 4-nitroanilide hydrochloride (B4875) and 4-nitro-3-octanoyloxy-benzoic acid (N1646) substrates were also from Sigma-Aldrich Chemical Co. (St. Louis, MO, United States). Lachesis m. rhombeata venom was provided by Center for Biological Studies and Research of the Pontifical Catholic University of Goiás (PUC Goiás, Goiânia, GO, Brazil) through Dr Nelson J. Silva Jr. A lyophilized pool of venom obtained from one female adult snake was stored at −20°C and dissolved in ultrapure water prior to use.
Wistar rats (300–350 g; 2–3 months old) obtained from Central Bioterium of the University of Western São Paulo (UNOESTE, Presidente Prudente, SP, Brazil) were housed in plastic cages (3 animals/cage) with a wood-shaving substrate, at 23 ± 1°C on a 12-h light/dark cycle with lights on at 6 a.m. The animals had free access to food and water. The experimental procedures were approved by an institutional Committee for Ethics in Animal Use (CEUA/UNOESTE, Protocol No. 6713/2021) and were done according to the general ethical guidelines for animal use established by the Brazilian Society of Laboratory Animal Science (SBCAL) and Brazilian Federal Law No. 11.794 of October 8, 2008, in conjunction with the guidelines for animal experiments established by the Brazilian National Council for Animal Experimentation (CONCEA).
PLA2 activity was assayed essentially as described elsewhere (Carregari et al., 2013). The standard assay mixture contained 200 μl of buffer (10 mM Tris-HCl, 10 mM CaCl2 and 100 mM NaCl, pH 8.0), 20 μl of substrate (3 mM 4-nitro-3-octanoyloxy-benzoic acid) and 20 μl of sample [venom alone (1 mg/ml) or venom (1 mg/ml) pre-incubated (for 30 min at 37°C) with varespladib (0.001–1 mM)] in a final volume of 240 μl. After adding sample, the mixture was incubated for 30 min at 37°C, with one unit of enzymatic activity being defined as an increase in absorbance of 0.001/min at 425 nm. All assays were done in triplicate with readings at 60-s intervals using a SpectraMax 340 multiwell plate reader (Molecular Devices, San Jose, CA, United States).
Caseinolytic activity was determined through colorimetric assay in a SpectraMax 340 multiwell plate reader (Molecular Devices, San Jose, CA, United States) using Azocasein as substrate, essentially as described elsewhere (Torres-Bonilla et al., 2020). The standard assay mixture contained 90 μl of substrate (212 mM Azocasein), 10 μl of reaction buffer (0.05 M Tris-HCl, 1 mM CaCl2, pH 8.0) and 10 μl of sample [venom alone (1 mg/ml) or venom (1 mg/ml) pre-incubated (for 30 min at 37°C) with varespladib (0.001–1 mM)] in a final volume of 110 μl. The mixture was incubated for 90 min at 37°C and then the reaction was terminated adding 200 μl of TCA 5% for 5 min at room temperature; the mixture was centrifuged (5 min at 8.000 g) and 150 μl of supernatant was transferred to the multiwell plate containing the same volume of NaOH (0.5 M). Finally, the absorbance was read at 440 nm via endpoint mode, with one activity unit being defined as an increase of absorbance of 0.001/min.
Esterase activity was assayed essentially as described by (Erlanger et al., 1961) and adapted by (Torres-Bonilla et al., 2020). The standard assay mixture contained 200 μl of substrate (100 mM Nα-Benzoyl-dl-arginine 4-nitroanilide hydrochloride), 50 μl of reaction buffer (10 mM Tris-HCl, 10 mM CaCl2, 100 mM NaCl, pH 8.0), 15 μl of ultrapure water and 5 μl of sample [venom alone (1 mg/ml) or venom (1 mg/ml) pre-incubated (for 30 min at 37°C) with varespladib (0.001–1 mM)] in a final volume of 270 μl. The mixture was incubated for 30 min at 37°C in a multiwell plate and then read under an absorbance at λ = 410 nm via endpoint mode, with one activity unit being defined as an increase of absorbance of 0.001/min.
Coagulant activity was performed using Labtest® commercial kits (Labtest Diagnóstica S.A., Lagoa Santa, MG, Brasil) performed in a quick timer Coagmaster 4.0 (Wama Diagnóstica Produtos para Laboratórios, São Carlos, SP, Brazil). Wistar rats were anesthetized by a non-lethal dose (1.8 mg/kg, i.p.) of thiopental (Cristália®, São Paulo, SP, Brazil) and, subsequently, subjected to intracardiac puncture in order to obtain blood samples using BD Vacutainer® Citrate Tubes with 3.2% buffered sodium citrate solution at an anticoagulant:blood ratio of 1:10 (v/v); after this procedure, the animals were euthanased in saturated atmosphere with CO2. Blood samples were centrifuged (2,500 g, 4°C, 15 min) in order to obtain citrated platelet-poor plasma used in the prothrombin time (PT) and activated partial thromboplastin clotting time (aPTT) assays at 37°C; for each assay, 190 μl of platelet-poor plasma was incubated at 37°C with 10 μl of sample [1 – saline solution, 2 – varespladib (1 mM) or 3 – antivenom (antivenom:venom ratio 1:3 ‘v/w’), 4 – L. m. rhombeata venom (1 mg/ml), 5 – L. m. rhombeata venom + VPL, 6 – L. m. rhombeata venom + antivenom, 7 – and L. m. rhombeata venom + VPL + antivenom]. Antivenom:venom ratio was based on the manufacturer’s stated neutralizing capacity for the antivenom, where 1 ml of antivenom neutralizes 3 mg of L. muta venom (Instituto Butantan, São Paulo, SP, Brazil). The minimum effective dose of varespladib (1 mM) was confirmed in pilot experiments. Protocols: 1 − L. m. rhombeata venom was pre-incubated with VPL and/or antivenom for 30 min at 37°C before PT- and aPTT-required clot formation recording; 2 − L. m. rhombeata venom was directly exposed to VPL and/or antivenom and followed by immediate PT- and aPTT-required clot formation recording.
Hemorrhagic activity was performed according to (Theakston and Reid, 1983). Male Wistar rats were anesthetized with sodium thiopental (1.8 mg/kg, i.p.) and their dorsal region was trichotomized in order to set the injection sites. Initially, some doses of L. m. rhombeata venom (0.01, 0.05, 0.1, 0.5 and 1 mg/kg) were tested in order to find the minimum haemorrhagic dose (0.5 mg/kg), which was used to investigate the neutralizing action of antivenom (antivenom:venom ratio of 1:3 ‘v/w’) and varespladib (1 mM). Protocol: after injection of venom (i.d.), the animals were subsequently treated with antivenom and/or varespladib via an intraperitoneal injection, followed by monitoring period of 24 h. Control sites were injected with 0.9% NaCl, DMSO (varespladib solvent) or antivenom. After 24 h, the rats were euthanized in saturated atmosphere with CO2, the dorsal skin was removed and the subcutaneous hemorrhagic halos in the inner surface were measured through ImageJ software (National Institute of Health, Bethesda, Maryland, United States).
All results (enzymatic, coagulant and haemorrhagic) were expressed as the mean ± SDM and statistical comparisons were done using Student’s t test or ANOVA followed by the Tukey–Kramer test, with p < 0.05 indicating significance. Data were analyzed using SAS University Edition software (SAS Institute Inc., Cary, NC, United States).
In colorimetric assays, L. m. rhombeata (1 mg/ml) exhibited moderate enzymatic activity for PLA2 which was promptly abolished from 0.01 mM of varespladib; the lowest concentration of varespladib (0.001 mM) did not produce inhibitory effect on the PLA2 activity of this venom (Figure 1A). Caseinolytic (Figure 1B) and esterasic (Figure 1C) activities were not affected by any of these concentrations of varespladib tested on artificial substrates (0.001–1 mM).
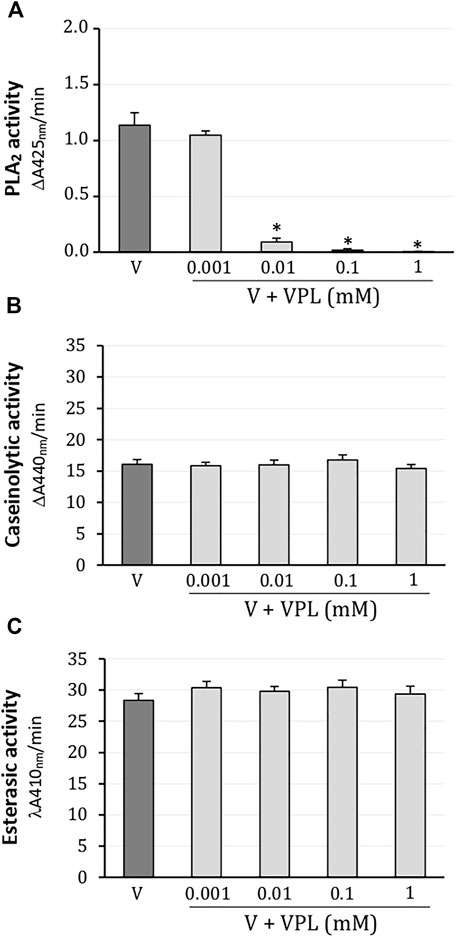
FIGURE 1. Action of varespladib on the major enzymatic activities of L. m. rhombeata venom. (A) Varespladib abolished the PLA2 activity at low concentrations. Unaffected (B) caseinolytic and (C) esterasic activities by varespladib at any tested concentration. Bars represent the mean ± SDM (n = 5). *p < 0.05 compared to venom alone.
In rat citrated plasma, L. m. rhombeata venom (1 mg/ml) exhibited procoagulant action on the aPPT (intrinsic pathway) and PT (extrinsic pathway), decreasing in approximately 56.5 and 55.7% these times, respectively (p < 0.05 compared to basal values for both, n = 6) (Figures 2A,B). L. m. rhombeata venom (1 mg/ml)-induced procoagulant action (aPTT and PT) was not prevented by pre-incubating venom with antivenom (antivenom:venom ratio of 1:3 ‘v/w’) alone for 30 min at 37°C before clotting assay; however, varespladib (1 mM) alone significantly prevented the venom-induced procoagulant action for PT, with approximately 16.4% of decreasing being verified (p < 0.05 compared to venom alone, n = 6), and it partially avoided the venom-induced procoagulant action for aPTT, being observed approximately 26.9% of decreasing; the combination of both agents successfully contributed to avoid both intrinsic and extrinsic disorders in rat citrated plasma, resulting in 8.4 and 18.6% of decreasing for PT and aPTT, respectively (p < 0.05 compared to venom alone, n = 6) (Figure 2A). In assays performed immediately after the exposure L. m. rhombeata venom (1 mg/ml) to antivenom (antivenom:venom ratio of 1:3 ‘v/w’) and/or varespladib (1 mM), both of agents slightly delayed the L. m. rhombeata venom (1 mg/ml)-induced procoagulant action for PT and aPTT; under this experimental condition, varespladib alone or combined with antivenom were more effective in avoiding only the venom-induced procoagulant action for PT, with approximately 23.3% of decreasing being verified using both agents (Figure 2B).
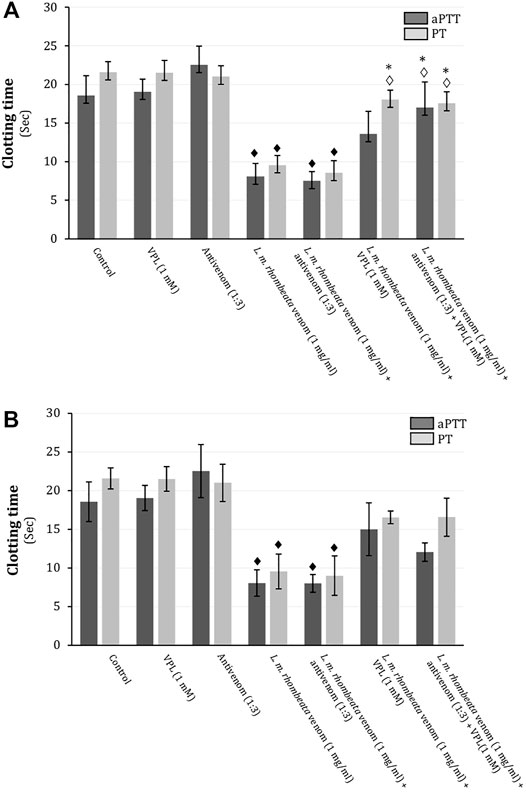
FIGURE 2. Action of varespladib on the procoagulant activity of L. m. rhombeata venom in rat citrated plasma in vitro. (A) Antivenom did not prevent the venom-induced procoagulant action applying the pre-incubation protocol, whereas varespladib alone avoided the procoagulant action for PT and its association with antivenom successfully prevented both intrinsic and extrinsic disorders. (B) Antivenom did not prevent the venom-induced procoagulant action applying the direct exposure protocol, whereas varespladib alone or combined with antivenom produced minor protective action on both coagulant pathways even combined with antivenom. Bars represent the mean ± SDM (n = 6). ♦p < 0.05 compared to control, ◊p < 0.05 compared to venom alone and *p < 0.05 compared to antivenom alone.
L. m. rhombeata venom at low doses (5, 50 and 100 μg/kg) did not produce subcutaneous haemorrhagic action in rats, however, with exceptionally one animal exhibiting ∼213 mm2 of haemorrhagic halo for 50 μg of venom/kg (opened arrow) and another one ∼301 mm2 for 100 μg of venom/kg (filled arrow); the higher dose of this venom (500 μg/kg) induced pronounced haemorrhagic halo formation (p < 0.05 compared to control saline, n = 6) (Figure 3A). Antivenom (antivenom:venom ratio of 1:3 ‘v/w’) administered (i.p) immediately after intradermic injections of L. m. rhombeata venom (500 μg/kg) was not able to prevent the venom-induced subcutaneous haemorrhage; varespladib (1 mM) alone or associated with antivenom (antivenom:venom ratio of 1:3 ‘v/w’) exhibited a mild protective effect on the venom-induced subcutaneous haemorrhage (Figure 3B).
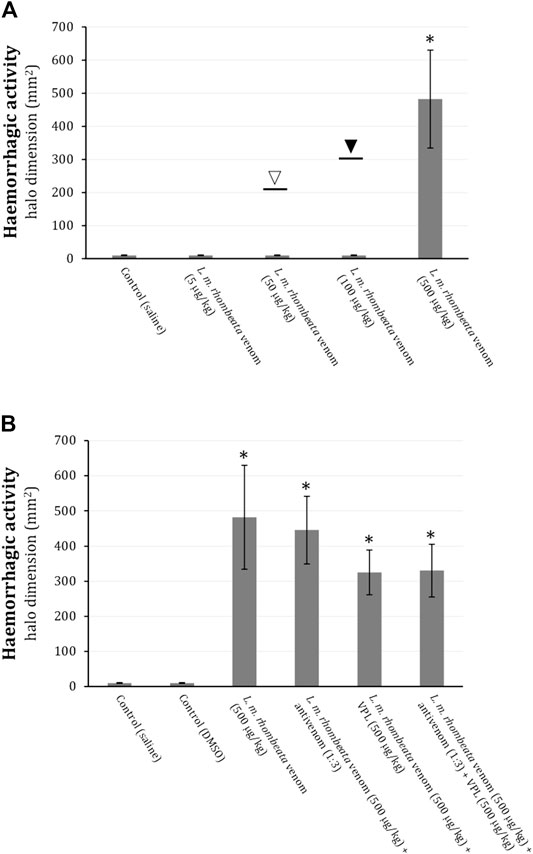
FIGURE 3. Action of varespladib on the haemorrhagic activity of L. m. rhombeata venom in rats. (A) L. m rhombeata venom produced intense subcutaneous haemorrhage at a dose of 500 μg of venom/kg, with the lowest doses (5, 50, and 100 μg/kg) being ineffective to produce haemorrhage; the opened and filled arrows indicate the haemorrhagic dimension caused in single animals envenomed with 50 and 100 μg of venom/kg, respectively. (B) Antivenom did not prevent the venom-induced subcutaneous haemorrhage, with varespladib alone or combined with antivenom producing discreet reduction of this alteration. Bars represent the mean ± SDM (n = 6). *p < 0.05 compared to control (saline).
Envenomations by Viperidae snakes comprise a serious public health problem in Latin America (Chippaux, 2017; Ochoa-Avilés et al., 2020; Gutiérrez et al., 2020b). In Brazil, these snakes are represented by three main genera, i.e., Bothrops, Crotalus and Lachesis (Viperidae–Crotalinae), being responsible for more than 20,000 cases of snakebites per year in this country, as reported by Notifiable Diseases Information System of the Brazilian Ministry of Health (SINAN, Brasília, DF, Brazil). Lachesis snakes found in South America (L. muta muta and L. m. rhombeata) occasionally cause severe human envenomations (Magalhães et al., 2019; Diniz-Sousa et al., 2020), which are characterized by pronounced local and systemic disorders, e.g., necrosis (Damico et al., 2006; Ferreira et al., 2009; Damico et al., 2012), haemorrhage, coagulopathy (Sánchez et al., 1987; Sánchez et al., 1991; Sánchez et al., 1995; Fuly et al., 1997; Rucavado et al., 1999; Estêvão-Costa et al., 2000; Torres-Huaco et al., 2013) and hypotension (Dias et al., 2016a; Dias et al., 2016b), strongly associated with a variety of enzymatically active proteins such as snake venom metalloproteases, serine proteases, PLA2, C-type lectins and l-amino acid oxidases (Weinberg et al., 2004; Junqueira-de-Azevedo et al., 2006; Bregge-Silva et al., 2012; Madrigal et al., 2012; Cordeiro et al., 2018; Diniz-Sousa et al., 2018; Wiezel et al., 2019) present in these venoms.
In recent years, the PLA2 antagonistic activity of varespladib (Lewin et al., 2016; Salvador et al., 2019; Salvador et al., 2021), a synthetic drug developed to treat disturbances of inflammatory cascades associated with high levels of secreted PLA2 (Varespladib, 2011), has been experimentally explored as an useful therapeutic alternative to complement antivenom therapies applied in envenomations by Elapidae and Viperidae snakes, with potential even to replace them in special situations in which these antivenoms are not available. Varespladib has high efficacy to suppress the systemic effects caused by several venoms from Elapidae (Lewin et al., 2016; Bittenbinder et al., 2018; Lewin et al., 2018; Wang et al., 2018; Oliveira et al., 2020; Gutiérrez et al., 2020a; Dashevsky et al., 2021; Kazandjian et al., 2021; Silva-Carvalho et al., 2021) and Viperidae (Lewin et al., 2016; Wang et al., 2018; Youngman et al., 2020; Zinenko et al., 2020; Gutiérrez et al., 2020a; Liu et al., 2021) snakes. However, there is no report associating the efficacy of varespladib with toxicological aspects of Lachesis venoms.
Based on this premise, we have unprecedentedly investigated the action of varespladib on the enzymatic, coagulant and haemorrhagic activities of Lachesis muta rhombeata venom to determine its efficiency as a single pharmacological tool or combined with a commercial antivenom used in Brazil. In summary, we have demonstrated that varespladib used as a single pharmacological tool abolishes the PLA2 activity of L. m. rhombeata venom at low concentrations, without affecting the catalytic activity for proteases (metalloprotease and serino protease) of the venom, indicating a very specific inhibitory activity; its high PLA2 antagonistic activity was reflected on the venom-induced procoagulant action, mostly interfering on the extrinsic pathway disorders produced by venom in rat citrated serum (pre-incubation protocol), whereas the drug did not prevent the haemorrhagic activity induced by L. m. rhombeata venom in rats. The association of varespladib with a commercial antivenom used in Brazil to treat envenomations by Lachesis spp. did not produce important synergic actions on the procoagulant (direct incubation protocol) and haemorrhagic effects induced by L. m. rhombeata venom; such interaction resulted in a major prevention of the venom-induced intrinsic and extrinsic coagulant disorders seen under pre-incubation protocol.
Although the procoagulant action of Viperidae venoms has been mostly associated with the presence of serine proteases in these venoms (Gutiérrez et al., 2021), varespladib can partially prevent the procoagulant action of L. m. rhombeata venom, indicating an eventual role of PLA2 in these processes. Accordingly, varespladib also contributes to prevent coagulating disorders induced by other groups of snake venoms, e.g., Bothrops (Viperidae-Crotalinae), Daboia, Echis, Oxyuranus, Naja, Pseudechis and Bitis spp., which exhibit high PLA2 activity (Bittenbinder et al., 2018; Xie et al., 2020; Youngman et al., 2020; Zdenek et al., 2020). On the other hand, varespladib does not affect the L. m. rhombeata venom-induced subcutaneous haemorrhage in rats, reflecting its disability in antagonizing the metalloproteases of this venom, since local and systemic haemorrhagic actions produced by Viperidae venoms are mainly mediated by this family of toxins (Escalante et al., 2011; Seo et al., 2017). In addition, although an Asp49 PLA2 (LmrTX) with anticoagulant activity has been already isolated from L. m. rhombeata venom (Damico et al., 2012), it does not appear to contribute expressively with the subcutaneous haemorrhage induced in rats, as reported in this study.
L. m. rhombeata venom has been an important object of study for structural characterization of toxins such as metalloproteases (Cordeiro et al., 2018), serine proteases (Aguiar et al., 1996; Wiezel et al., 2019), C-type lectins (Wiezel et al., 2019), basic and acid PLA2 (Damico et al., 2012; Cordeiro et al., 2015; Diniz-Sousa et al., 2018), phospholipase B and hialuronidase (Wiezel et al., 2015), including bradykinin-potentiating peptides (BPPs) (Pinheiro-Júnior et al., 2018). As previously commented, these toxins are responsible by developing the main toxicological aspects of the envenomation by Lachesis. However, the factual involvement of PLA2 toxins on the effects produced by Lachesis venoms have been poorly investigated, with a few reports describing their inhibitory action of platelet aggregation (Cordeiro et al., 2015), anticoagulant and antithrombotic activities (Damico et al., 2012), and cytotoxicity on C2C12 myotubes (Diniz-Sousa et al., 2018).
In Brazil, envenomations by L. m. muta and L. m. rhombeata are treated with anti-Bothrops/Lachesis serums, with their rescue action being dependent on early i.v. administration (Pla et al., 2013; Madrigal et al., 2017; Solano et al., 2018). There are some negative factors that resulting in deaths by accidents involving Lachesis snakes in Brazil: 1) limited availability of antivenoms, 2) difficulties in accessing health services in certain regions of the country, and 3) lack of a specific anti-Lachesis serum. Consequently, other types of antivenoms, e.g., anti-Bothrops serum and anti-Bothrops/Crotalus serum, have been inaccurately recommended to treat envenomations by L. muta in the absence of anti-Bothrops/Lachesis serum (Magalhães et al., 2019; Muniz et al., 2021). These challenges involving the treatment of envenomations by Lachesis snakes strengthen the search for therapeutically useful adjuncts, with varespladib rising as a plausible tool.
In conclusion, varespladib abolishes efficiently the PLA2 activity of L. m. rhombeata venom at low concentrations and does not affect other majority enzymatic groups of this venom, e.g., metalloproteases and serine proteases. Varespladib alone might partially prevent the procoagulant effect of L. m. rhombeata venom, with its combination with antivenom avoiding alterations in both intrinsic and extrinsic pathways. Varespladib does not reduce the subcutaneous haemorrhage formation induced by L. m. rhombeata venom in rats due to the lack of inhibitory action on the proteases of the venom. The association of varespladib with a recommended polyvalent antivenom does not produce synergic action on the venom-induced haemorrhagic action but it helps to prevent the venom-induced coagulation disorders.
The original contributions presented in the study are included in the Article/Supplementary Material, further inquiries can be directed to the corresponding author.
The animal study was reviewed and approved by the Committee for Ethics in Animal Use of University of Western São Paulo (CEUA/UNOESTE, Protocol No. 6713/2021).
RMBN and RSF contributed to conception and design of the study. PGG, DRP, and NLV organized the database. LFA performed the statistical analysis. PGG, DRP, and KAT-B wrote the first draft of the manuscript. NJSJ and SH wrote sections of the manuscript. KM-Z, EGR, and RSF contributed to manuscript revision, read, and approved the submitted version.
This work was funded by institutional resources from University of Western São Paulo (UNOESTE, Presidente Prudente, SP, Brazil) and Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, São Paulo, SP, Brazil, Grant No. 2020/04287-6 and 2021/14375-2). PGG was also supported by a studentship from FAPESP (Grant No. 2020/11268-8). NJSJ and SH are supported by a research fellowship from Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Brasília, DF, Brazil, Grant Nos. 309320/2016-0 and 310547/2014-8, respectively).
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Aguiar, A. S., Alves, C. R., Melgarejo, A., and Giovanni-De-Simone, S. (1996). Purification and Partial Characterization of a Thrombin-Like/gyroxin Enzyme from Bushmaster (Lachesis muta Rhombeata) Venom. Toxicon 34 (5), 555–565. doi:10.1016/0041-0101(95)00159-X
Bittenbinder, M. A., Zdenek, C. N., Op den Brouw, B., Youngman, N. J., Dobson, J. S., Naude, A., et al. (2018). Coagulotoxic Cobras: Clinical Implications of Strong Anticoagulant Actions of African Spitting Naja Venoms that Are Not Neutralised by Antivenom but Are by LY315920 (Varespladib). Toxins (Basel) 10 (12), E516. doi:10.3390/toxins10120516
Bregge-Silva, C., Nonato, M. C., de Albuquerque, S., Ho, P. L., Junqueira de Azevedo, I. L., Vasconcelos Diniz, M. R., et al. (2012). Isolation and Biochemical, Functional and Structural Characterization of a Novel L-Amino Acid Oxidase from Lachesis muta Snake Venom. Toxicon 60 (7), 1263–1276. doi:10.1016/j.toxicon.2012.08.008
Bryan-Quirós, W., Fernández, J., Gutiérrez, J. M., Lewin, M. R., and Lomonte, B. (2019). Neutralizing Properties of LY315920 toward Snake Venom Group I and II Myotoxic Phospholipases A2. Toxicon 157, 1–7. doi:10.1016/j.toxicon.2018.11.292
Chippaux, J. P. (2017). Snakebite Envenomation Turns Again into a Neglected Tropical Disease!. J. Venom. Anim. Toxins Incl. Trop. Dis. 23, 38–42. doi:10.1186/s40409-017-0127-6
Carregari, V. C., Stuani Floriano, R., Rodrigues-Simioni, L., Winck, F. V., Baldasso, P. A., Ponce-Soto, L. A., et al. (2013). Biochemical, Pharmacological, and Structural Characterization of New Basic PLA2 Bbil-TX from Bothriopsis Bilineata Snake Venom. Biomed. Res. Int. 2013, 612649. doi:10.1155/2013/612649
Cordeiro, F. A., Coutinho, B. M., Wiezel, G. A., de Castro Figueiredo Bordon, K., Bregge-Silva, C., Rosa-Garzon, N. G., et al. (2018). Purification and Enzymatic Characterization of a Novel Metalloprotease from Lachesis muta Rhombeata Snake Venom. J. Venom. Anim. Toxins Incl. Trop. Dis. 24, 32. doi:10.1186/s40409-018-0171-x
Cordeiro, F. A., Perini, T. G., Bregge-Silva, C., Cremonez, C. M., Rodrigues, R. S., Boldrini-França, J., et al. (2015). A New Phospholipase A₂ from Lachesis muta Rhombeata: Purification, Biochemical and Comparative Characterization with Crotoxin B. Protein Pept. Lett. 22 (9), 816–827. doi:10.2174/0929866522666150706112431
Costa, H. C., and Bérnils, R. S. (2018). Répteis Do Brasil e suas Unidades Federativas: Lista de espécies. Herpetologia Brasileira 8, 11–57.
Damico, D. C., Bueno, L. G., Rodrigues-Simioni, L., Marangoni, S., da Cruz-Höfling, M. A., and Novello, J. C. (2006). Functional Characterization of a Basic D49 Phospholipase A2 (LmTX-I) from the Venom of the Snake Lachesis muta Muta (Bushmaster). Toxicon 47 (7), 759–765. doi:10.1016/j.toxicon.2006.02.007
Damico, D. C., Nascimento, J. M., Lomonte, B., Ponce-Soto, L. A., Joazeiro, P. P., Novello, J. C., et al. (2007). Cytotoxicity of Lachesis muta Muta Snake (Bushmaster) Venom and its Purified Basic Phospholipase A2 (LmTX-I) in Cultured Cells. Toxicon 49 (5), 678–692. doi:10.1016/j.toxicon.2006.11.014
Damico, D. C., Vassequi-Silva, T., Torres-Huaco, F. D., Nery-Diez, A. C., de Souza, R. C., Da Silva, S. L., et al. (2012). LmrTX, a Basic PLA₂ (D49) Purified from Lachesis muta Rhombeata Snake Venom with Enzymatic-Related Antithrombotic and Anticoagulant Activity. Toxicon 60 (5), 773–781. doi:10.1016/j.toxicon.2012.06.010
Dashevsky, D., Bénard-Valle, M., Neri-Castro, E., Youngman, N. J., Zdenek, C. N., Alagón, A., et al. (2021). Anticoagulant Micrurus Venoms: Targets and Neutralization. Toxicol. Lett. 337, 91–97. doi:10.1016/j.toxlet.2020.11.010
Dias, L., Rodrigues, M. A., Inoue, B. R., Rodrigues, R. L., Rennó, A. L., de Souza, V. B., et al. (2016a). Pharmacological Analysis of Hemodynamic Responses to Lachesis muta (South American Bushmaster) Snake Venom in Anesthetized Rats. Toxicon 123, 25–44. doi:10.1016/j.toxicon.2016.10.002
Dias, L., Rodrigues, M. A., Rennó, A. L., Stroka, A., Inoue, B. R., Panunto, P. C., et al. (2016b). Hemodynamic Responses to Lachesis muta (South American Bushmaster) Snake Venom in Anesthetized Rats. Toxicon 123, 1–14. doi:10.1016/j.toxicon.2016.10.001
Diniz, M. R., and Oliveira, E. B. (1992). Purification and Properties of a Kininogenin from the Venom of Lachesis muta (Bushmaster). Toxicon 30 (3), 247–258. doi:10.1016/0041-0101(92)90867-5
Diniz-Sousa, R., Caldeira, C. A. S., Kayano, A. M., Paloschi, M. V., Pimenta, D. C., Simões-Silva, R., et al. (2018). Identification of the Molecular Determinants of the Antibacterial Activity of LmutTX, a Lys49 Phospholipase A2 Homologue Isolated from Lachesis muta muta Snake Venom (Linnaeus, 1766). Basic Clin. Pharmacol. Toxicol. 122 (4), 413–423. doi:10.1111/bcpt.12921
Diniz-Sousa, R., Moraes, J. D. N., Rodrigues-da-Silva, T. M., Oliveira, C. S., and Caldeira, C. A. D. S. (2020). A Brief Review on the Natural History, Venomics and the Medical Importance of Bushmaster (Lachesis) Pit viper Snakes. Toxicon X 7 (7), 100053. doi:10.1016/j.toxcx.2020.100053
Erlanger, B. F., Kokowsky, N., and Cohen, W. (1961). The Preparation and Properties of Two New Chromogenic Substrates of Trypsin. Arch. Biochem. Biophys. 95 (2), 271–278. doi:10.1016/0003-9861(61)90145-x
Escalante, T., Rucavado, A., Fox, J. W., and Gutiérrez, J. M. (2011). Key Events in Microvascular Damage Induced by Snake Venom Hemorrhagic Metalloproteinases. J. Proteomics 74 (9), 1781–1794. doi:10.1016/j.jprot.2011.03.026
Estêvão-Costa, M. I., Diniz, C. R., Magalhães, A., Markland, F. S., and Sanchez, E. F. (2000). Action of Metalloproteinases Mutalysin I and II on Several Components of the Hemostatic and Fibrinolytic Systems. Thromb. Res. 99 (4), 363–376. doi:10.1016/s0049-3848(00)00259-0
Ferreira, T., Camargo, E. A., Ribela, M. T., Damico, D. C., Marangoni, S., Antunes, E., et al. (2009). Inflammatory Oedema Induced by Lachesis muta muta (Surucucu) Venom and LmTX-I in the Rat Paw and Dorsal Skin. Toxicon 53 (1), 69–77. doi:10.1016/j.toxicon.2008.10.016
Fuly, A. L., Calil-Elias, S., Martinez, A. M., Melo, P. A., and Guimarães, J. A. (2003). Myotoxicity Induced by an Acidic Asp-49 Phospholipase A(2) Isolated from Lachesis muta Snake Venom. Comparison with Lysophosphatidylcholine. Int. J. Biochem. Cell Biol. 35 (10), 1470–1481. doi:10.1016/s1357-2725(03)00129-8
Fuly, A. L., Calil-Elias, S., Zingali, R. B., Guimarães, J. A., and Melo, P. A. (2000). Myotoxic Activity of an Acidic Phospholipase A2 Isolated from Lachesis muta (Bushmaster) Snake Venom. Toxicon 38 (7), 961–972. doi:10.1016/s0041-0101(99)00208-1
Fuly, A. L., Machado, O. L., Alves, E. W., and Carlini, C. R. (1997). Mechanism of Inhibitory Action on Platelet Activation of a Phospholipase A2 Isolated from Lachesis muta (Bushmaster) Snake Venom. Thromb. Haemost. 78 (5), 1372–1380. doi:10.1055/s-0038-1665414
Giovanni-De-Simone, S., Aguiar, A. S., Gimenez, A. R., Novellino, K., and de Moura, R. S. (1997). Purification, Properties, and N-Terminal Amino Acid Sequence of a Kallikrein-like Enzyme from the Venom of Lachesis muta Rhombeata (Bushmaster). J. Protein Chem. 16 (8), 809–818. doi:10.1023/a:1026372018547
Graham, R. L., Graham, C., McClean, S., Chen, T., O’Rourke, M., Hirst, D., et al. (2005). Identification and Functional Analysis of a Novel Bradykinin Inhibitory Peptide in the Venoms of New World Crotalinae Pit Vipers. Biochem. Biophys. Res. Commun. 338 (3), 1587–1592. doi:10.1016/j.bbrc.2005.10.130
Gutiérrez, J. M., Albulescu, L-O., Clare, R. H., Casewell, N. R., Abd El-Aziz, T. M., Escalante, T., et al. (2021). The Search for Natural and Synthetic Inhibitors that Would Complement Antivenoms as Therapeutics for Snakebite Envenoming. Toxins (Basel) 13 (7), 451. doi:10.3390/toxins13070451
Gutiérrez, J. M., Lewin, M. R., Williams, D. J., and Lomonte, B. (2020a). Varespladib (LY315920) and Methyl Varespladib (LY333013) Abrogate or Delay Lethality Induced by Presynaptically Acting Neurotoxic Snake Venoms. Toxins (Basel) 12 (2), 131. doi:10.3390/toxins12020131
Gutiérrez, J. M., Castillo, L., de Naves, K. M. D., Masís, J., and Alape-Girón, A. (2020b). Epidemiology of Snakebites in El Salvador (2014-2019). Toxicon 186, 26–28. doi:10.1016/j.toxicon.2020.07.027
Junqueira-de-Azevedo, I. L., Ching, A. T., Carvalho, E., Faria, F., Nishiyama, M. Y., Ho, P. L., et al. (2006). Lachesis muta (Viperidae) cDNAs Reveal Diverging Pit viper Molecules and Scaffolds Typical of Cobra (Elapidae) Venoms: Implications for Snake Toxin Repertoire Evolution. Genetics 173, 877–889. doi:10.1534/genetics.106.056515
Kazandjian, T. D., Arrahman, A., Still, K. B. M., Somsen, G. W., Vonk, F. J., Casewell, N. R., et al. (2021). Anticoagulant Activity of Naja nigricollis Venom Is Mediated by Phospholipase A2 Toxins and Inhibited by Varespladib. Toxins (Basel) 13, 302. doi:10.3390/toxins13050302
Lewin, M., Samuel, S., Merkel, J., and Bickler, P. (2016). Varespladib (LY315920) Appears to Be a Potent, Broad-Spectrum, Inhibitor of Snake Venom Phospholipase A2 and a Possible Pre-referral Treatment for Envenomation. Toxins (Basel) 8 (9), 248. doi:10.3390/toxins8090248
Lewin, M. R., Gilliam, L. L., Gilliam, J., Samuel, S. P., Bulfone, T. C., Bickler, P. E., et al. (2018). Delayed LY333013 (Oral) and LY315920 (Intravenous) Reverse Severe Neurotoxicity and rescue Juvenile Pigs from Lethal Doses of Micrurus fulvius (Eastern Coral Snake) Venom. Toxins (Basel) 10 (11), E479. doi:10.3390/toxins10110479
Liu, C. C., Wu, C. J., Hsiao, Y. C., Yang, Y. H., Liu, K. L., Huang, G. J., et al. (2021). Snake Venom Proteome of Protobothrops Mucrosquamatus in Taiwan: Delaying Venom-Induced Lethality in a Rodent Model by Inhibition of Phospholipase A2 Activity with Varespladib. J. Proteomics 234, 104084. doi:10.1016/j.jprot.2020.104084
Madrigal, M., Pla, D., Sanz, L., Barboza, E., Arroyo-Portilla, C., Corrêa-Netto, C., et al. (2017). Cross-reactivity, Antivenomics, and Neutralization of Toxic Activities of Lachesis Venoms by Polyspecific and Monospecific Antivenoms. Plos Negl. Trop. Dis. 11 (8), e0005793. doi:10.1371/journal.pntd.0005793
Madrigal, M., Sanz, L., Flores-Díaz, M., Sasa, M., Núñez, V., Alape-Girón, A., et al. (2012). Snake Venomics across Genus Lachesis. Ontogenetic Changes in the Venom Composition of Lachesis Stenophrys and Comparative Proteomics of the Venoms of Adult Lachesis Melanocephala and Lachesis Acrochorda. J. Proteomics 77, 280–297. doi:10.1016/j.jprot.2012.09.003
Magalhães, S. F. V., Peixoto, H. M., Moura, N., Monteiro, W. M., and de Oliveira, M. R. F. (2019). Snakebite Envenomation in the Brazilian Amazon: a Descriptive Study. Trans. R. Soc. Trop. Med. Hyg. 113, 143–151. doi:10.1093/trstmh/try121
Muniz, E. G., Noronha, M. D. D. N., Saraiva, M. D. G. G., Monteiro, W. M., and Oliveira, S. S. (2021). Neutralization of Hemostatic Disorders Induced by Lachesis muta Venom using Brazilian Antivenoms. Toxicon 191, 44–47. doi:10.1016/j.toxicon.2020.12.013
Nogueira, C. C., Argôlo, A. J. S., Arzamendia, V., Azevedo, J. A., Barbo, F. E., Bérnils, R. S., et al. (2019). Atlas of Brazilian Snakes: Verified point-locality Maps to Mitigate the Wallacean Shortfall in a Megadiverse Snake Fauna. South. Am. J. Herpetol. 14 (1), 1–274. doi:10.2994/sajh-d-19-00120.1
Ochoa-Avilés, A., Heredia-Andino, O. S., Escandón, S. A., Celorio-Carvajal, C. A., Arias-Peláez, M. C., Zaruma-Torres, F., et al. (2020). Viperidae Snakebites in Ecuador: a Review of Epidemiological and Ecological Aspects. Toxicon X 7, 100051. doi:10.1016/j.toxcx.2020.100051
Oliveira, I. C. F., Gutiérrez, J. M., Lewin, M. R., and Oshima-Franco, Y. (2020). Varespladib (LY315920) Inhibits Neuromuscular Blockade Induced by Oxyuranus Scutellatus Venom in a Nerve-Muscle Preparation. Toxicon 187, 101–104. doi:10.1016/j.toxicon.2020.08.023
Pinheiro-Júnior, E. L., Boldrini-França, J., de Campos Araújo, L. M. P., Santos-Filho, N. A., Bendhack, L. M., Cilli, E. M., et al. (2018). LmrBPP9: A Synthetic Bradykinin-Potentiating Peptide from Lachesis muta Rhombeata Venom that Inhibits the Angiotensin-Converting Enzyme Activity in vitro and Reduces the Blood Pressure of Hypertensive Rats. Peptides 102, 1–7. doi:10.1016/j.peptides.2018.01.015
Pla, D., Sanz, L., Molina-Sánchez, P., Zorita, V., Madrigal, M., Flores-Díaz, M., et al. (2013). Snake Venomics of Lachesis muta Rhombeata and Genus-wide Antivenomics Assessment of the Paraspecific Immunoreactivity of Two Antivenoms Evidence the High Compositional and Immunological Conservation across Lachesis. J. Proteomics 89, 112–123. doi:10.1016/j.jprot.2013.05.028
Rucavado, A., Flores-Sánchez, E., Franceschi, A., Magalhaes, A., and Gutiérrez, J. M. (1999). Characterization of the Local Tissue Damage Induced by LHF-II, a Metalloproteinase with Weak Hemorrhagic Activity Isolated from Lachesis muta muta Snake Venom. Toxicon 37 (9), 1297–1312. doi:10.1016/s0041-0101(98)00268-2
Salvador, G. H. M., Borges, R. J., Lomonte, B., Lewin, M. R., and Fontes, M. R. M. (2021). The Synthetic Varespladib Molecule Is a Multi-Functional Inhibitor for PLA2 and PLA2-like Ophidic Toxins. Biochim. Biophys. Acta Gen. Subj 1865 (7), 129913. doi:10.1016/j.bbagen.2021.129913
Salvador, G. H. M., Gomes, A. A. S., Bryan-Quirós, W., Fernández, J., Lewin, M. R., Gutiérrez, J. M., et al. (2019). Structural Basis for Phospholipase A2-like Toxin Inhibition by the Synthetic Compound Varespladib (LY315920). Sci. Rep. 9 (1), 17203. doi:10.1038/s41598-019-53755-5
Sánchez, E. F., Costa, M. I., Chavez-Olortegui, C., Assakura, M. T., Mandelbaum, F. R., and Diniz, C. R. (1995). Characterization of a Hemorrhagic Factor, LHF-I, Isolated from the Bushmaster Snake (Lachesis muta muta) Venom. Toxicon 33 (12), 1653–1667. doi:10.1016/0041-0101(95)00097-6
Sánchez, E. F., Magalhës, A., Mandelbaum, F. R., and Diniz, C. R. (1991). Purification and Characterization of the Hemorrhagic Factor II from the Venom of the Bushmaster Snake (Lachesis muta muta). Biochim. Biophys. Acta 1074 (3), 347–356. doi:10.1016/0304-4165(91)90084-t
Sánchez, E. F., Magalhães, A., and Diniz, C. R. (1987). Purification of a Hemorrhagic Factor (LHF-I) from the Venom of the Bushmaster Snake, Lachesis muta muta. Toxicon 25 (6), 611–619. doi:10.1016/0041-0101(87)90107-3
Sanz, L., Escolano, J., Ferretti, M., Biscoglio, M. J., Rivera, E., Crescenti, E. J., et al. (2008). Snake Venomics of the South and Central American Bushmasters. Comparison of the Toxin Composition of Lachesis muta Gathered from Proteomic versus Transcriptomic Analysis. J. Proteomics 71 (1), 46–60. doi:10.1016/j.jprot.2007.10.004
Seo, T., Sakon, T., Nakazawa, S., Nishioka, A., Watanabe, K., Matsumoto, K., et al. (2017). Haemorrhagic Snake Venom Metalloproteases and Human ADAMs Cleave LRP5/6, Which Disrupts Cell-Cell Adhesions in vitro and Induces Haemorrhage in vivo. FEBS J. 284 (11), 1657–1671. doi:10.1111/febs.14066
Silva-Carvalho, R., Gaspar, M. Z., Quadros, L. H. B., Lobo, L. G. G., Rogério, L. M., Santos, N. T. S., et al. (2021). In Vivo treatment with Varespladib, a Phospholipase A2 Inhibitor, Prevents the Peripheral Neurotoxicity and Systemic Disorders Induced by Micrurus corallinus (Coral Snake) Venom in Rats. Toxicol. Lett. 356, 54–63. doi:10.1016/j.toxlet.2021.11.003
Soares, M. R., Oliveira-Carvalho, A. L., Wermelinger, L. S., Zingali, R. B., Ho, P. L., Junqueira-de-Azevedo, I. L., et al. (2005). Identification of Novel Bradykinin-Potentiating Peptides and C-type Natriuretic Peptide from Lachesis muta Venom. Toxicon 46 (1), 31–38. doi:10.1016/j.toxicon.2005.03.006
Solano, G., Gómez, A., Corrales, G., Chacón, D., Estrada, R., and León, G. (2018). Contributions of the Snake Venoms of Bothrops asper, Crotalus Simus and Lachesis Stenophrys to the Paraspecificity of the Central American Polyspecific Antivenom (PoliVal-ICP). Toxicon 144, 1–6. doi:10.1016/j.toxicon.2018.01.016
Theakston, R. D., and Reid, H. A. (1983). Development of Simple Standard Assay Procedures for the Characterization of Snake Venom. Bull World Health Organ. 61 (6), 949–956.
Torres-Bonilla, K. A., Panunto, P. C., Pereira, B. B., Zambrano, D. F., Herrán-Medina, J., Bernal, M. H., et al. (2020). Toxinological Characterization of Venom from Leptodeira annulata (Banded Cat-Eyed Snake; Dipsadidae, Imantodini). Biochimie 174, 171–188. doi:10.1016/j.biochi.2020.04.006
Torres-Huaco, F. D., Werneck, C. C., Vicente, C. P., Vassequi-Silva, T., Nery-Diez, A. C., Mendes, C. B., et al. (2013). Rapid Purification and Procoagulant and Platelet Aggregating Activities of Rhombeobin: a Thrombin-like/gyroxin-like Enzyme from Lachesis muta Rhombeata Snake Venom. Biomed. Res. Int. 2013, 903292. doi:10.1155/2013/903292
Varespladib (2011). Varespladib. Am. J. Cardiovasc. Drugs 11:137–43. doi:10.2165/11533650-000000000-00000
Wang, Y., Zhang, J., Zhang, D., Xiao, H., Xiong, S., and Huang, C. (2018). Exploration of the Inhibitory Potential of Varespladib for Snakebite Envenomation. Molecules 23 (2), E391. doi:10.3390/molecules23020391
Weinberg, M. L., Felicori, L. F., Bello, C. A., Magalhães, H. P., Almeida, A. P., Magalhães, A., et al. (2004). Biochemical Properties of a Bushmaster Snake Venom Serine Proteinase (LV-Ka), and its Kinin Releasing Activity Evaluated in Rat Mesenteric Arterial Rings. J. Pharmacol. Sci. 96, 333–342. doi:10.1254/jphs.fpj04005x
Wiezel, G. A., Bordon, K. C., Silva, R. R., Gomes, M. S., Cabral, H., Rodrigues, V. M., et al. (2019). Subproteome of Lachesis muta Rhombeata Venom and Preliminary Studies on LmrSP-4, a Novel Snake Venom Serine Proteinase. J. Venom. Anim. Toxins Incl. Trop. Dis. 25, e147018. doi:10.1590/1678-9199-JVATITD-1470-18
Wiezel, G. A., dos Santos, P. K., Cordeiro, F. A., Bordon, K. C., Selistre-de-Araújo, H. S., Ueberheide, B., et al. (2015). Identification of Hyaluronidase and Phospholipase B in Lachesis muta Rhombeata Venom. Toxicon 107 (Pt B), 359–368. doi:10.1016/j.toxicon.2015.08.029
Xie, C., Albulescu, L. O., Still, K. B. M., Slagboom, J., Zhao, Y., Jiang, Z., et al. (2020). Varespladib Inhibits the Phospholipase A2 and Coagulopathic Activities of Venom Components from Hemotoxic Snakes. Biomedicines 8 (6), 165. doi:10.3390/biomedicines8060165
Youngman, N. J., Walker, A., Naude, A., Coster, K., Sundman, E., and Fry, B. G. (2020). Varespladib (LY315920) Neutralises Phospholipase A2 Mediated Prothrombinase-Inhibition Induced by Bitis Snake Venoms. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 236, 108818. doi:10.1016/j.cbpc.2020.108818
Zdenek, C. N., Youngman, N. J., Hay, C., Dobson, J., Dunstan, N., Allen, L., et al. (2020). Anticoagulant Activity of Black Snake (Elapidae: Pseudechis) Venoms: Mechanisms, Potency, and Antivenom Efficacy. Toxicol. Lett. 330, 176–184. doi:10.1016/j.toxlet.2020.05.014
Keywords: Viperidae snake, phospholipase A2 (PLA2), coagulating activity, haemorrhage, varespladib, antivenom, neutralization
Citation: Gutierres PG, Pereira DR, Vieira NL, Arantes LF, Silva Jr. NJ, Torres-Bonilla KA, Hyslop S, Morais-Zani K, Nogueira RMB, Rowan EG and Floriano RS (2022) Action of Varespladib (LY-315920), a Phospholipase A2 Inhibitor, on the Enzymatic, Coagulant and Haemorrhagic Activities of Lachesis muta rhombeata (South-American Bushmaster) Venom. Front. Pharmacol. 12:812295. doi: 10.3389/fphar.2021.812295
Received: 10 November 2021; Accepted: 09 December 2021;
Published: 12 January 2022.
Edited by:
Delavar Shahbazzadeh, Pasteur Institute of Iran (PII), IranReviewed by:
Manuela Berto Pucca, Federal University of Roraima, BrazilCopyright © 2022 Gutierres, Pereira, Vieira, Arantes, Silva, Torres-Bonilla, Hyslop, Morais-Zani, Nogueira, Rowan and Floriano. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Rafael S. Floriano, cmFmYWVsQHVub2VzdGUuYnI=
Disclaimer: All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Research integrity at Frontiers

Learn more about the work of our research integrity team to safeguard the quality of each article we publish.